Do New-Generation Recycled Phosphorus Fertilizers Increase the Content of Potentially Toxic Elements in Soil and Plants?
Abstract
1. Introduction
2. Materials and Methods
2.1. Fertilizers
2.2. Experimental Design and Agronomic Management
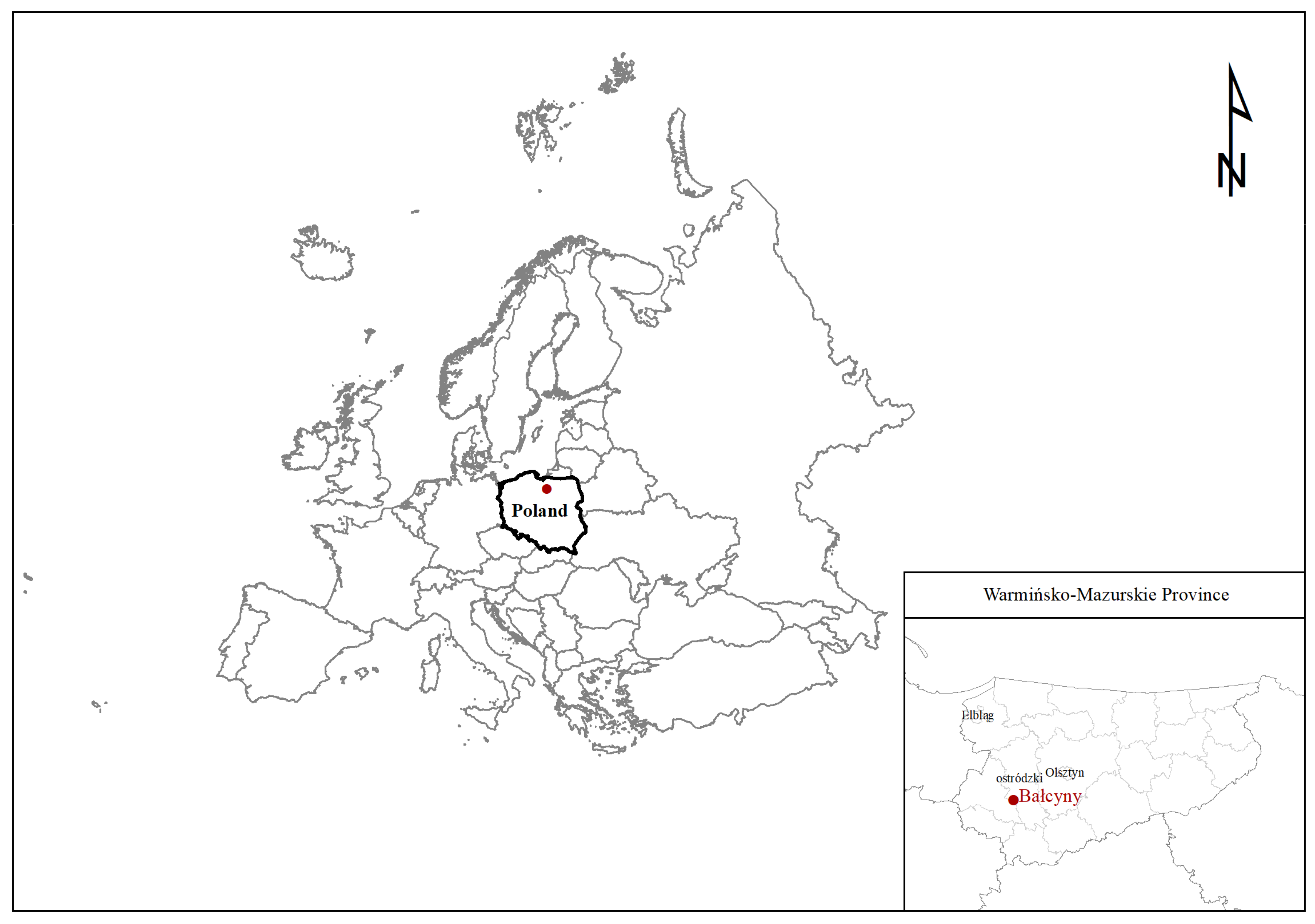
2.3. Study Site, Soil and Meteorological Conditions
2.4. Sampling
2.4.1. Soil
2.4.2. Wheat Grain
2.4.3. Wheat Straw
2.4.4. Weeds
2.4.5. Post-Harvest Residues
2.5. Chemical Analyses
2.6. Calculations and Statistical Analysis
3. Results and Discussion
3.1. PTE Input from Fertilizers to Soil
3.2. PTE in Soil
3.3. PTE in Plant Biomass
3.3.1. Wheat Grain and Straw
3.3.2. Weeds
3.3.3. Post-Harvest Residues
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kopittke, P.M.; Menzies, N.W.; Wang, P.; McKenna, B.A.; Lombi, E. Soil and the intensification of agriculture for global food security. Environ. Int. 2019, 132, 105078. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, R.K.; Mukherjee, A.; Chakraborty, S.K.; Verma, J.P. Impact of agrochemical application in sustainable agriculture. In New and Future Developments in Microbial Biotechnology and Bioengineering; Verma, J.P., Macdonald, C.A., Gupta, V.K., Podile, A.R., Eds.; Elsevier: Amsterdam, The Netherlands; Oxford, UK; Cambridge, MA, USA, 2021; pp. 15–24. [Google Scholar]
- Alengebawy, A.; Abdelkhalek, S.T.; Qureshi, S.R.; Wang, M.-Q. Heavy Metals and pesticides toxicity in agricultural soil and plants: Ecological risks and human health implications. Toxics 2021, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Kabata-Pendias, A. Trace Elements in Soils and Plants, 4th ed.; CRC Press: Boca Raton, FL, USA, 2010; pp. 1–520. [Google Scholar]
- Radziemska, M.; Bęś, A.; Gusiatin, Z.M.; Majewski, G.; Mazur, Z.; Bilgin, A.; Jaskulska, I.; Brtnický, M. Immobilization of potentially toxic elements (PTE) by mineral-based amendments: Remediation of contaminated soils in post-industrial sites. Minerals 2020, 10, 87. [Google Scholar] [CrossRef]
- Marschner, P. Marschner’s Mineral. Nutrition of Higher Plants, 3rd ed.; Academic Press: San Diego, CA, USA, 2012; pp. 1–651. [Google Scholar]
- Gruszecka-Kosowska, A. Potentially harmful element concentrations in the vegetables cultivated on arable soils, with human health-risk implications. Int. J. Environ. Res. Public Health 2019, 16, 4053. [Google Scholar] [CrossRef]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy metal toxicity and the environment. Mol. Clin. Exp. Toxicol. 2012, 101, 133–164. [Google Scholar] [CrossRef]
- Alloway, B.J. Heavy metals and metalloids as micronutrients for plants and animals. In Heavy Metals in Soils: Trace Metals and Metalloids in Soils and Their Bioavailability; Alloway, B.J., Ed.; Springer: Dordrecht, The Netherlands, 2013; pp. 195–209. [Google Scholar]
- Okereafor, U.; Makhatha, M.; Mekuto, L.; Uche-Okereafor, N.; Sebola, T.; Mavumengwana, V. Toxic metal implications on agricultural soils, plants, animals, aquatic life and human health. Int. J. Environ. Res. Public Health 2020, 17, 2204. [Google Scholar] [CrossRef]
- Ali, H.; Khan, E. Bioaccumulation of non-essential hazardous heavy metals and metalloids in freshwater fish. Risk to human health. Environ. Chem. Lett. 2018, 16, 903–917. [Google Scholar] [CrossRef]
- Rai, P.K.; Lee, S.S.; Zhang, M.; Tsang, Y.F.; Kim, K.-H. Heavy metals in food crops: Health risks, fate, mechanisms, and management. Environ. Int. 2019, 125, 365–385. [Google Scholar] [CrossRef] [PubMed]
- Gall, J.E.; Boyd, R.S.; Rajakaruna, N. Transfer of heavy metals through terrestrial food webs: A review. Environ. Monit. Assess. 2015, 187, 1–21. [Google Scholar] [CrossRef]
- Pu, W.; Sun, J.; Zhang, F.; Wen, X.; Liu, W.; Huang, C. Effects of copper mining on heavy metal contamination in a rice agrosystem in the Xiaojiang River Basin, southwest China. Acta Geochim. 2019, 38, 753–773. [Google Scholar] [CrossRef]
- Chen, X.-X.; Liu, Y.-M.; Zhao, Q.-Y.; Cao, W.-Q.; Chen, X.-P.; Zou, C.-Q. Health risk assessment associated with heavy metal accumulation in wheat after long-term phosphorus fertilizer application. Environ. Pollut. 2020, 262, 114348. [Google Scholar] [CrossRef]
- Kpomblekou-A, K.; Tabatabai, M.A. Metal contents of phosphate rocks. Commun. Soil Sci. Plant. Anal. 1994, 25, 2871–2882. [Google Scholar] [CrossRef]
- Sabiha-Javied, M.T.; Chaudhry, M.M.; Tufail, M.; Irfan, N. Heavy metal pollution from phosphate rock used for the production of fertilizer in Pakistan. Microchem. J. 2009, 91, 94–99. [Google Scholar] [CrossRef]
- Faridullah, F.; Umar, M.; Alam, A.; Sabir, M.A.; Khan, D. Assessment of heavy metals concentration in phosphate rock deposits, Hazara basin, Lesser Himalaya Pakistan. Geosci. J. 2017, 21, 743–752. [Google Scholar] [CrossRef]
- Mar, S.S.; Okazaki, M. Investigation of Cd contents in several phosphate rocks used for the production of fertilizer. Microchem. J. 2012, 104, 17–21. [Google Scholar] [CrossRef]
- Siddique, A.; Hassan, A.; Khan, S.R.; Inayat, A.; Nazir, A.; Iqbal, M. Appraisal of heavy metals and nutrients from phosphate rocks, Khyber Pakhtunkhwa, Pakistan. Chem. Int. 2018, 4, 1–6. [Google Scholar]
- Wang, X.; Xiong, J.; He, Z. Activated dolomite phosphate rock fertilizers to reduce leaching of phosphorus and trace metals as compared to superphosphate. J. Environ. Manag. 2020, 255, 109872. [Google Scholar] [CrossRef] [PubMed]
- Nziguheba, G.; Smolders, E. Inputs of trace elements in agricultural soils via phosphate fertilizers in European countries. Sci. Total Environ. 2008, 390, 53–57. [Google Scholar] [CrossRef] [PubMed]
- da Silva, F.B.V.; do Nascimento, C.W.A.; Araújo, P.R.M. Environmental risk of trace elements in P-containing fertilizers marketed in Brazil. J. Soil Sci. Plant Nutr. 2017, 17, 635–647. [Google Scholar] [CrossRef]
- EU. Regulation (EU) 2019/1009 of the European Parliament and of the Council of 5 June 2019 laying down rules on the making available on the market of EU fertilising products and amending Regulations (EC) No 1069/2009 and (EC) No 1107/2009 and repealing Regulation (EC) No 2003/2003 (Text with EEA relevance). Off. J. Eur. Union 2019, L170, 1–114. [Google Scholar]
- Mortvedt, J.J. Heavy metal contaminants in inorganic and organic fertilizers. Fert. Environ. 1995, 43, 55–61. [Google Scholar] [CrossRef]
- Ulrich, A.E. Cadmium governance in Europe’s phosphate fertilizers: Not so fast? Sci. Total Environ. 2019, 650, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Barquet, K.; Järnberg, L.; Rosemarin, A.; Macura, B. Identifying barriers and opportunities for a circular phosphorus economy in the Baltic Sea region. Water Res. 2020, 171, 115433. [Google Scholar] [CrossRef]
- van Dijk, K.C.; Lesschen, J.P.; Oenema, O. Phosphorus flows and balances of the European Union Member States. Sci. Total Environ. 2016, 542, 1078–1093. [Google Scholar] [CrossRef]
- Schröder, J.J.; Smit, A.L.; Cordell, D.; Rosemarin, A. Improved phosphorus use efficiency in agriculture: A key requirement for its sustainable use. Chemosphere 2011, 84, 822–831. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Xu, C.-c.; Ridoutt, B.G.; Wang, X.-c.; Ren, P.-a. Nitrogen and phosphorus losses and eutrophication potential associated with fertilizer application to cropland in China. J. Clean. Prod. 2017, 159, 171–179. [Google Scholar] [CrossRef]
- Zhu, J.; Li, M.; Whelan, M. Phosphorus activators contribute to legacy phosphorus availability in agricultural soils: A review. Sci. Total Environ. 2018, 612, 522–537. [Google Scholar] [CrossRef] [PubMed]
- El Wali, M.; Golroudbary, S.R.; Kraslawski, A. Circular economy for phosphorus supply chain and its impact on social sustainable development goals. Sci. Total Environ. 2021, 777, 146060. [Google Scholar] [CrossRef] [PubMed]
- Chojnacka, K.; Moustakas, K.; Witek-Krowiak, A. Bio-based fertilizers: A practical approach towards circular economy. Bioresour. Technol. 2020, 295, 122223. [Google Scholar] [CrossRef] [PubMed]
- Saeid, A.; Wyciszkiewicz, M.; Jastrzebska, M.; Chojnacka, K.; Gorecki, H. A concept of production of new generation of phosphorus-containing biofertilizers. BioFertP project. Przem. Chem. 2015, 94, 361–365. [Google Scholar] [CrossRef]
- Wyciszkiewicz, M.; Sojka, M.; Saeid, A. Production of phosphorus biofertilizer based on the renewable materials in large laboratory scale. Open Chem. 2019, 17, 893–901. [Google Scholar] [CrossRef]
- Herzel, H.; Stemann, J.; Simon, S.; Adam, C. Comparison of thermochemical treatment of sewage sludge ash with sodium sulphate in laboratory-scale and pilot-scale experiments. Int. J. Environ. Sci. Technol. 2021, 1–10. [Google Scholar] [CrossRef]
- Ottosen, L.M.; Kirkelund, G.M.; Jensen, P.E. Extracting phosphorous from incinerated sewage sludge ash rich in iron or aluminum. Chemosphere 2013, 91, 963–969. [Google Scholar] [CrossRef]
- Nanzer, S.; Oberson, A.; Huthwelker, T.; Eggenberger, U.; Frossard, E. The molecular environment of phosphorus in sewage sludge ash: Implications for bioavailability. J. Environ. Qual. 2014, 43, 1050–1060. [Google Scholar] [CrossRef]
- Lekfeldt, J.D.S.; Rex, M.; Mercl, F.; Kulhánek, M.; Tlustoš, P.; Magid, J.; de Neergaard, A. Effect of bioeffectors and recycled P-fertiliser products on the growth of spring wheat. Chem. Biol. Technol. Agric. 2016, 3, 22. [Google Scholar] [CrossRef]
- Römer, W.; Steingrobe, B. Fertilizer effect of phosphorus recycling products. Sustainability 2018, 10, 1166. [Google Scholar] [CrossRef]
- Weigand, H.; Bertau, M.; Hübner, W.; Bohndick, F.; Bruckert, A. RecoPhos: Full-scale fertilizer production from sewage sludge ash. Waste Manag. 2013, 33, 540–544. [Google Scholar] [CrossRef]
- Krüger, O.; Adam, C. Recovery potential of German sewage sludge ash. Waste Manag. 2015, 45, 400–406. [Google Scholar] [CrossRef]
- Smol, M.; Adam, C.; Kugler, S.A. Thermochemical treatment of sewage sludge ash (SSA)-potential and perspective in Poland. Energies 2020, 13, 5461. [Google Scholar] [CrossRef]
- Smol, M. Inventory of wastes generated in Polish sewage sludge incineration plants and their possible circular management directions. Resources 2020, 9, 91. [Google Scholar] [CrossRef]
- Staroń, A.; Kowalski, Z.; Banach, M.; Wzorek, Z. Sposoby termicznej utylizacji odpadów z przemysłu mięsnego. Czas. Techn. Chemia 2010, 107, 323–332. [Google Scholar]
- Nelson, N.; Mikkelsen, R. Meeting the phosphorus requirement on organic farms. Better Crops 2008, 92, 12–14. [Google Scholar]
- Wyciszkiewicz, M.; Saeid, A.; Malinowski, P.; Chojnacka, K. Valorization of phosphorus secondary raw materials by Acidithiobacillus ferrooxidans. Molecules 2017, 22, 473. [Google Scholar] [CrossRef]
- Wisniak, J. Phosphorus-from discovery to commodity. Indian J. Chem. Technol. 2005, 12, 108–122. [Google Scholar]
- EU. Commission Regulation (EU) No 142/2011 of 25 February 2011 implementing Regulation (EC) No 1069/2009 of the European Parliament and of the Council laying down health rules as regards animal by-products and derived products not intended for human consumption and implementing Council Directive 97/78/EC as regards certain samples and items exempt from veterinary checks at the border under that Directive Text with EEA relevance. Off. J. Eur. Union 2011, L54, 1–254. [Google Scholar]
- Załuszniewska, A.; Nogalska, A. The effect of meat and bone meal (MBM) on the seed yield and quality of winter oilseed rape. Agronomy 2020, 10, 1952. [Google Scholar] [CrossRef]
- Kivelä, J.; Chen, L.; Muurinen, S.; Kivijärvi, P.; Hintikainen, V.; Helenius, J. Effects of meat bone meal as fertilizer on yield and quality of sugar beet and carrot. Agric. Food Sci. 2015, 24, 68–83. [Google Scholar] [CrossRef]
- Delin, S.; Engström, L.; Lundkvist, A. Optimal Placement of Meat Bone Meal Pellets to Spring Oats. Front. Sustain. Food Syst. 2018, 2, 27. [Google Scholar] [CrossRef]
- Jatana, B.S.; Kitchens, C.; Ray, C.; Tharayil, N. Regulating the nutrient release rates from proteinaceous agricultural byproducts using organic amendments and its effect on soil chemical and microbiological properties. Biol. Fertil. Soils 2020, 56, 747–758. [Google Scholar] [CrossRef]
- Jeng, A.S.; Haraldsen, T.K.; Grønlund, A.; Pedersen, P.A. Meat and bone meal as nitrogen and phosphorus fertilizer to cereals and rye grass. Nutr. Cycl. Agroecosystems 2006, 76, 183–191. [Google Scholar] [CrossRef]
- Balawejder, M.; Matłok, N.; Gorzelany, J.; Pieniążek, M.; Antos, P.; Witek, G.; Szostek, M. Foliar fertilizer based on calcined bones, boron and molybdenum—a study on the development and potential effects on maize grain production. Sustainability 2019, 11, 5287. [Google Scholar] [CrossRef]
- Balawejder, M.; Szostek, M.; Gorzelany, J.; Antos, P.; Witek, G.; Matłok, N. A study on the potential fertilization effects of microgranule fertilizer based on the protein and calcined bones in maize cultivation. Sustainability 2020, 12, 1343. [Google Scholar] [CrossRef]
- Piccolla, C.D.; Hesterberg, D.; Muraoka, T.; Novotny, E.H. Optimizing pyrolysis conditions for recycling pig bones into phosphate fertilizer. Waste Manag. 2021, 131, 249–257. [Google Scholar] [CrossRef]
- Saeid, A.; Labuda, M.; Chojnacka, K.; Górecki, H. Use of microorganism in production of phosphorus fertilizers. Przem. Chem. 2012, 91, 956–958. [Google Scholar]
- Jastrzębska, M.; Kostrzewska, M.K.; Saeid, A. Can phosphorus from recycled fertilisers replace conventional sources? An agronomic evaluation in field-scale experiments on temperate Luvisols. Appl. Sci. 2019, 9, 2086. [Google Scholar] [CrossRef]
- Jastrzȩbska, M.; Saeid, A.; Kostrzewska, M.K.; Baśladyńska, S. New phosphorus biofertilizers from renewable raw materials in the aspect of cadmium and lead contents in soil and plants. Open Chem. 2018, 16, 35–49. [Google Scholar] [CrossRef]
- Rolewicz, M.; Rusek, P.; Mikos-Szymańska, M.; Cichy, B.; Dawidowicz, M. Obtaining of suspension fertilizers from incinerated sewage sludge ashes (ISSA) by a method of solubilization of phosphorus compounds by Bacillus megaterium bacteria. Waste Biomass Valorization 2016, 7, 871–877. [Google Scholar] [CrossRef]
- Rolewicz, M.; Rusek, P.; Borowik, K. Obtaining of granular fertilizers based on ashes from combustion of waste residues and ground bones using phosphorous solubilization by bacteria Bacillus megaterium. J. Environ. Manag. 2018, 216, 128–132. [Google Scholar] [CrossRef]
- Polish Standard. General Requirements for the Competence of Testing and Calibration Laboratories; PN-EN ISO/IEC 17025:2005; Committee for Standardization: Warsaw, Poland, 2005. [Google Scholar]
- MARD-PL. Regulation of the Minister of Agriculture and Rural Development of 18 June 2008 regarding the implementation of certain provisions of the Act on fertilizers and fertilization. J. Laws 2008, 119, 765. [Google Scholar]
- Woś, A. Klimat Polski; Wydawnictwo Naukowe PWN: Warsaw, Poland, 1999. [Google Scholar]
- Grabowski, J. Charakterystyka stosunków termicznych występujących w Rolniczym Zakładzie Doświadczalnym w Bałcynach w latach 1972–1990. Acta Acad. Agricult. Techn. Olst. Agricultura 1995, 60, 3–13. [Google Scholar]
- Food and Agriculture Organization of the United Nations. World reference base for soil resources 2014. In International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; Food and Agriculture Organization of the United Nations: Rome, Italy, 2014. [Google Scholar]
- Polish Standard. Soil Quality and Determination of pH; PN-ISO 10390:1997; Polish Committee for Standardization: Warsaw, Poland, 1997. [Google Scholar]
- Czarnowska, K. Total content of heavy metals in parent rocks as reference background levels of soils. Soil Sci. Annu. 1996, 47, 43–50. [Google Scholar]
- Kucharczak-Moryl, E.; Moryl, A.; Żmuda, R. Influence of the environment on the content of arsenic in cultivated soils in Zgorzelec-Bogatynia region. Ecol. Eng. Environ. Technol. 2014, 37, 107–116. [Google Scholar]
- Kabata-Pendias, A.; Pendias, H. Biogeochemistry of Trace Elements; Polish Scientific Publishing Company: Warsaw, Poland, 1999; pp. 1–400. [Google Scholar]
- ME-PL. Ordinance by the Minister of the Environment of 1 September 2016 on assessment procedures for the land surface pollution. J. Laws 2016, 2016, 1395. [Google Scholar]
- IUNG. The Monitoring of the Chemistry of the Polish Arable Soils; Institute of Soil Science and Plant Cultivation: Puławy, Poland, 2017. [Google Scholar]
- MH-PL. Ordinance by the Minister of Health of 13 January 2003 on maximum concentrations of chemical and biological impurities which may be present in food, food ingredients, permitted supplementary substances and substances helpful in food processing. J. Laws 2003, 37, 326. [Google Scholar]
- MARD-PL. Ordinance by the Minister of Agriculture and Rural Development of 25 August 2014 amending the ordinance on the content of undesirable substances in animal feed. J. Laws 2014, 37, 1213. [Google Scholar]
- MARD-PL. Ordinance by the Minister of Agriculture and Rural Development (Poland) of 29 June 2019 amending the ordinance on the content of undesirable substances in animal feed. J. Laws 2018, 2018, 1213. [Google Scholar]
- FAO-WHO. Codex Alimentarius. General Standard for Contaminants and Toxins in Food and Feed. CXS 193-1995; Food and Agricultural Organization ff the United Nation, World Health Organization: Rome, Italy, 2019. [Google Scholar]
- EU. Commission Regulation (EU) 2015/1006 of 25 June 2015 amending Regulation (EC) No 1881/2006 as regards maximum levels of inorganic arsenic in foodstuffs (Text with EEA relevance). Off. J. Eur. Union 2015, L161, 14–16. [Google Scholar]
- USDA-FAS. China Releases the Standard for Maximum Levels of Contaminants in Foods; Office of Agricultural Affairs: Beijing, China, 2018; pp. 1–17. [Google Scholar]
- Kabata-Pendias, A.; Motowicka-Terelak, T.; Piotrowska, M.; Terelak, H.; Witek, T. Ocena Stopnia Zanieczyszczenia Gleb i Roślin Metalami Ciężkimi i Siarką. Ramowe Wytyczne dla Rolnictwa; IUNG: Puławy, Poland, 1993; pp. 1–20. [Google Scholar]
- Pres, J.; Kinal, S. Aktualne spojrzenie na sprawe zaopatrzenia zwierzat w mikroelementy. Zesz. Probl. Post. Nauk Rol. 1996, 434, 1042–1061. [Google Scholar]
- Wiśniowska-Kielian, B. Effect of soil moisture content on the absorption of heavy metals by plants. Zesz. Probl. Post. Nauk Rol. 2000, 472, 679–688. [Google Scholar]
- Gancarczyk-Gola, M.; Palowski, B. Heavy metals and acidity of surface soil horizons in surroundings of industrial centers and in non-contaminated regions. Rocz. Glebozn. 2005, 56, 59–66. [Google Scholar]
- Meteorological Data. Ten Days and Monthly Precipitation (Sum) and Air Temperature (Average) in Bałcyny in 2014–2016; 2014–2016 (Data in Exel Worksheet form Made Available Upon Request); Meteorological Station in Bałcyny of the University of Warmia and Mazury in Olsztyn: Olsztyn, Poland, 2021.
- Górecka, H.; Chojnacka, K.; Górecki, H. The application of ICP-MS and ICP-OES in determination of micronutrients in wood ashes used as soil conditioners. Talanta 2006, 70, 950–956. [Google Scholar] [CrossRef]
- Statsoft Inc. Statistica (Data Analysis Software System), Version 13.3; Statsoft Inc.: Tulsa, OK, USA, 2016. [Google Scholar]
- EC. Final Implementation Report for Directive 86/278/EEC on Sewage Sludge: 2013–2015; Eunomia Research & Consulting Ltd.: Bristol, UK, 2018. [Google Scholar]
- Kantek, K.; Korzeniowska, J. Potrzeby nawożenia zbóż mikroelementami w Województwie Dolnośląskim. Stud. Rap. IUNG-PIB 2013, 34, 145–157. [Google Scholar]
- Stępień, A.; Wojtkowiak, K. Effect of foliar application of Cu, Zn, and Mn on yield and quality indicators of winter wheat grain. Chil. J. Agric. Res. 2016, 76, 220–227. [Google Scholar] [CrossRef]
- Tani, F.H.; Barrington, S. Zinc and copper uptake by plants under two transpiration rates. Part I. Wheat (Triticum aestivum L.). Environ. Pollut. 2005, 138, 538–547. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kapoor, D.; Wang, J.; Shahzad, B.; Kumar, V.; Bali, A.S.; Jasrotia, S.; Zheng, B.; Yuan, H.; Yan, D. Chromium bioaccumulation and its impacts on plants: An overview. Plants 2020, 9, 100. [Google Scholar] [CrossRef]
- Singh, H.P.; Mahajan, P.; Kaur, S.; Batish, D.R.; Kohli, R.K. Chromium toxicity and tolerance in plants. Environ. Chem. Lett. 2013, 11, 229–254. [Google Scholar] [CrossRef]
- Wei, B.; Yu, J.; Cao, Z.; Meng, M.; Yang, L.; Chen, Q. The availability and accumulation of heavy metals in greenhouse soils associated with intensive fertilizer application. Int. J. Environ. Res. Public Health 2020, 17, 5359. [Google Scholar] [CrossRef]
- Martinez, J.; Peu, P. Nutrient fluxes from a soil treatment process for pig slurry. Soil Use Manag. 2000, 16, 100–107. [Google Scholar] [CrossRef]
- Xu, Y.; Yu, W.; Ma, Q.; Zhou, H. Accumulation of copper and zinc in soil and plant within ten-year application of different pig manure rates. Plant Soil Environ. 2013, 59, 492–499. [Google Scholar] [CrossRef]
- Jiao, W.; Chen, W.; Chang, A.C.; Page, A.L. Environmental risks of trace elements associated with long-term phosphate fertilizers applications: A review. Environ. Pollut. 2012, 168, 44–53. [Google Scholar] [CrossRef]
- Chen, W.; Chang, A.C.; Wu, L. Assessing long-term environmental risks of trace elements in phosphate fertilizers. Ecotoxicol. Environ. Saf. 2007, 67, 48–58. [Google Scholar] [CrossRef]
- Atafar, Z.; Mesdaghinia, A.; Nouri, J.; Homaee, M.; Yunesian, M.; Ahmadimoghaddam, M.; Mahvi, A.H. Effect of fertilizer application on soil heavy metal concentration. Environ. Monit. Assess. 2010, 160, 83–89. [Google Scholar] [CrossRef]
- Belhaj, D.; Elloumi, N.; Jerbi, B.; Zouari, M.; Abdallah, F.B.; Ayadi, H.; Kallel, M. Effects of sewage sludge fertilizer on heavy metal accumulation and consequent responses of sunflower (Helianthus annuus). Environ. Sci. Pollut. Res. 2016, 23, 20168–20177. [Google Scholar] [CrossRef] [PubMed]
- Uprety, D.; Hejcman, M.; Száková, J.; Kunzová, E.; Tlustoš, P. Concentration of trace elements in arable soil after long-term application of organic and inorganic fertilizers. Nutr. Cycl. Agroecosystems 2009, 85, 241–252. [Google Scholar] [CrossRef]
- Thielecke, F.; Nugent, A.P. Contaminants in grain—a major risk for whole grain safety? Nutrients 2018, 10, 1213. [Google Scholar] [CrossRef]
- Hashemi, M. Heavy metals concentrations in dairy cow feedstuffs from the south of Iran. Food Addit. Contam. Part B 2020, 13, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Kulczycki, G.; Sacała, E. Sulfur application alleviates chromium stress in maize and wheat. Open Chem. 2020, 18, 1093–1104. [Google Scholar] [CrossRef]
- Korzeniowska, J.; Kantek, K. Biofortifcation of wheat grain with copper through soil fertilization. Environ. Prot. Nat. Resour. 2014, 25, 23–27. [Google Scholar] [CrossRef]
- Wang, M.; Kong, F.; Liu, R.; Fan, Q.; Zhang, X. Zinc in wheat grain, processing, and food. Front. Nutr. 2020, 7, 124. [Google Scholar] [CrossRef]
- Liu, W.-X.; Liu, J.-W.; Wu, M.-Z.; Li, Y.; Zhao, Y.; Li, S.-R. Accumulation and translocation of toxic heavy metals in winter wheat (Triticum aestivum L.) growing in agricultural soil of Zhengzhou, China. Bull. Environ. Contam. Toxicol. 2009, 82, 343–347. [Google Scholar] [CrossRef]
- Al-Othman, Z.A.; Ali, R.; Al-Othman, A.M.; Ali, J.; Habila, M.A. Assessment of toxic metals in wheat crops grown on selected soils, irrigated by different water sources. Arab. J. Chem. 2016, 9, S1555–S1562. [Google Scholar] [CrossRef]
- Kundu, R.; Bhattacharyya, K.; Majumder, A.; Pal, S. Response of wheat cultivars to arsenic contamination in polluted soils of West Bengal, India. Cereal Res. Commun. 2013, 41, 66–77. [Google Scholar] [CrossRef]
- Kumar, R.; Mehrotra, N.K.; Nautiyal, B.D.; Kumar, P.; Singh, P.K. Effect of copper on growth, yield and concentration of Fe, Mn, Zn and Cu in wheat plants (Triticum aestivum L.). J. Environ. Biol. 2009, 30, 485–488. [Google Scholar]
- Korzeniowska, J.; Stanislawska-Glubiak, E. Copper concentration in the top plant tissue as an indicator of Cu toxicity. Electron. J. Pol. Agric. Univ. 2003, 6. Available online: http://www.ejpau.media.pl/volume6/issue1/environment/art-02.html (accessed on 12 July 2021).
- Garnett, T.P.; Graham, R.D. Distribution and remobilization of iron and copper in wheat. Ann. Bot. 2005, 95, 817–826. [Google Scholar] [CrossRef]
- Hill, J.; Robson, A.D.; Loneragan, J.F. The effects of copper and nitrogen supply on the retranslocation of copper in four cultivars of wheat. Aust. J. Agric. Res. 1978, 29, 925–939. [Google Scholar] [CrossRef]
- Jastrzębska, M.; Kostrzewska, M.K.; Treder, K.; Makowski, P.; Jastrzębski, W.P. Functional properties of granulated ash and bone-based phosphorus biofertilizers in the field assessment. Part 2. Impact on selected morphological and physiological atributes of winter wheat. Przem. Chem. 2016, 95, 1586–1590. [Google Scholar] [CrossRef]
- Liu, D.-Y.; Liu, Y.-M.; Zhang, W.; Chen, X.-P.; Zou, C.-Q. Zinc uptake, translocation, and remobilization in winter wheat as affected by soil application of Zn fertilizer. Front. Plant Sci. 2019, 10, 426. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.-F.; Zhang, W.; Liu, D.-Y.; Yue, S.-C.; Cui, Z.-L.; Chen, X.-P.; Zou, C.-Q. Effects of nitrogen management on root morphology and zinc translocation from root to shoot of winter wheat in the field. Field Crop. Res. 2014, 161, 38–45. [Google Scholar] [CrossRef]
- Pascoalino, J.A.L.; Thompson, J.A.; Wright, G.; Franco, F.A.; Scheeren, P.L.; Pauletti, V.; Moraes, M.F.; White, P.J. Grain zinc concentrations differ among brazilian wheat genotypes and respond to zinc and nitrogen supply. PLoS ONE 2018, 13, e0199464. [Google Scholar] [CrossRef]
- Gondek, K. Wpływ nawożenia mineralnego, obornikiem i osadem garbarskim na zawartość wybranych metali ciężkich w roślinach i glebie. Acta Agrophys. 2004, 3, 465–478. [Google Scholar]
- Iżewska, A.; Wołoszyk, C. Contents of heavy metals in plants and soil fertilization of ash from sewage sludge combustion. Ecol. Chem. Eng. A 2013, 20, 1019–1027. [Google Scholar] [CrossRef]
- ME-PL. Regulation of the Minister of the Environment of January 20, 2015 regarding the R10 recovery process. J. Laws 2015, 2015, 132. [Google Scholar]
- ME-PL. Regulation of the Minister of the Environment, Republic of Poland, Catalogue of waste. J. Laws 2020, 2020, 10. [Google Scholar]
- Kirchmann, H.; Börjesson, G.; Kätterer, T.; Cohen, Y. From agricultural use of sewage sludge to nutrient extraction: A soil science outlook. Ambio 2017, 46, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Wierzbowska, J.; Sienkiewicz, S.; Sternik, P.; Busse, M.K. Using ash from incineration of municipal sewage sludge to fertilize Virginia fanpetals. Ecol. Chem. Eng. A 2015, 22, 497–507. [Google Scholar] [CrossRef]
- Zalewska, M.; Stępień, A.; Wierzbowska, J. Agronomic evaluation of dried sewage sludge and sewage sludge ash as sources of nutrients for maize. J. Elem. 2020, 25, 771–785. [Google Scholar] [CrossRef]
- Arroyo, M.d.M.D.; Hornedo, R.M.D.I.; Peralta, F.A.; Almestre, C.R.; Sánchez, J.V.M. Heavy metals concentration in soil, plant, earthworm and leachate from poultry manure applied to agricultural land. Rev. Int. Contam. Ambient. 2014, 30, 43–50. [Google Scholar]
- Wang, F.; Wang, Z.; Kou, C.; Ma, Z.; Zhao, D. Responses of wheat yield, macro- and micro-nutrients, and heavy metals in soil and wheat following the application of manure compost on the North China Plain. PLoS ONE 2016, 11, e0146453. [Google Scholar] [CrossRef]
- Staugaitis, G.; Narutytė, I.; Arbačauskas, J.; Vaišvila, Z.; Rainys, K.; Mažeika, R.; Masevičienė, A.; Žičkienė, L.; Šumskis, D. The influence of composts on yield and chemical elements of winter wheat and spring barley. Zemdirb. Agric. 2016, 103, 355–362. [Google Scholar] [CrossRef][Green Version]
- Godlewska, A. Changes in the contents of selected heavy metals in test plants fertilised with sewage sludge and hard coal ash. J. Ecol. Eng. 2017, 18, 97–102. [Google Scholar] [CrossRef]
- Chetyrbotskiy, V.A.; Chetyrbotskiy, A.N.; Levin, B.V. Mathematical modeling of the dynamics of plant mineral nutrition in the fertilizer–soil–plant system. Biophysics 2020, 65, 1036–1045. [Google Scholar] [CrossRef]
- Harre, N.T.; Young, B.G. Early-season nutrient competition between weeds and soybean. J. Plant. Nutr. 2020, 43, 1887–1906. [Google Scholar] [CrossRef]
- Jarrell, W.M.; Beverly, R.B. The dilution effect in plant nutrition studies. In Advances in Agronomy; Brady, N.C., Ed.; Academic Press: London, UK, 1981; Volume 34, pp. 197–224. [Google Scholar]
- Wołejko, E.; Łozowicka, B.; Kaczyński, P.; Konecki, R.; Grobela, M. The influence of chemical protection on the content of heavy metals in wheat (Triticum aestivum L.) growing on the soil enriched with granular sludge. Environ. Monit. Assess. 2017, 189, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Babar, M.A.; Khan, A.; Azam, S.; Ikramullah; Arif, M.; Hussain, S. Weeds in wheat crop: Weed whole plants nutrient concentration and uptake under fertilizer application and irrigation frequencies. Pure Appl. Biol. 2019, 8, 1724–1735. [Google Scholar] [CrossRef]
- Ashrafi, A.; Zahedi, M.; Fahmi, K.; Nadi, R. Neighbour effects of purslane (Portulaca oleracea L.) on Cd bioaccumulation by soybean in saline soil. Plant Soil Environ. 2014, 60, 439–445. [Google Scholar] [CrossRef]
- Gove, B.; Hutchinson, J.J.; Young, S.D.; Craigon, J.; McGrath, S.P. Uptake of metals by plants sharing a rhizosphere with the hyperaccumulator Thlaspi caerulescens. Int. J. Phytoremediation 2002, 4, 267–281. [Google Scholar] [CrossRef]
- Stupnicka-Rodzynkiewicz, E.; Labza, T.; Hochol, T. Content of some mineral components in weeds with respect to environmental conditions. Acta Agrar. Silvestria Ser. Agrar. 1996, 34, 125–130. [Google Scholar]
- Łabza, T.; Stupnicka-Rodzynkiewicz, E.; Hochół, T. Zawartość niklu w wybranych chwastach segetalnych. Zesz. Probl. Post. Nauk Rol. 1997, 448, 183–188. [Google Scholar]
- Roy, S.; Mondal, S. Brassicaceae plants response and tolerance to metal/metalloid toxicity. In The Plant Family Brassicaceae; Springer: Singapore, 2020; pp. 363–377. [Google Scholar]
- Sharma, P.; Tripathi, S.; Chandra, R. Phytoremediation potential of heavy metal accumulator plants for waste management in the pulp and paper industry. Heliyon 2020, 6, e04559. [Google Scholar] [CrossRef]
- Zubkova, V.; Belozubova, N.; Arslanbekova, F.; Gaponenko, A.; Gorbunova, V. Species Peculiarities of Weeds in Terms of Heavy Metal Accumulation. KnE Life Sci. 2021, 774–779. [Google Scholar] [CrossRef]
- Słomka, A.; Sutkowska, A.; Szczepaniak, M.; Malec, P.; Mitka, J.; Kuta, E. Increased genetic diversity of Viola tricolor L. (Violaceae) in metal-polluted environments. Chemosphere 2011, 83, 435–442. [Google Scholar] [CrossRef]
- Łabza, T.; Stupnicka-Rodzynkiewicz, E.; Hochol, T. Zawartość cynku i miedzi w wybranych chwastach segetalnych. Zesz. Probl. Post. Nauk Rol. 1996, 434, 307–312. [Google Scholar]
- Głowacka, A. Content and uptake of microelements (Cu, Zn, Mn, Fe) by maize (Zea mays L.) and accompanying weeds. Acta Agrobot. 2012, 65, 179–188. [Google Scholar] [CrossRef]
- Kumar, N.; Bauddh, K.; Kumar, S.; Dwivedi, N.; Singh, D.P.; Barman, S.C. Accumulation of metals in weed species grown on the soil contaminated with industrial waste and their phytoremediation potential. Ecol. Eng. 2013, 61, 491–495. [Google Scholar] [CrossRef]
- Zhao, S.; Duo, L. Bioaccumulation of cadium, copper, zinc, and nickel by weed species from municipal solid waste compost. Pol. J. Environ. Stud. 2015, 24, 413–417. [Google Scholar] [CrossRef]
- Wang, J.; Xiong, Y.; Zhang, J.; Lu, X.; Wei, G. Naturally selected dominant weeds as heavy metal accumulators and excluders assisted by rhizosphere bacteria in a mining area. Chemosphere 2020, 243, 125365. [Google Scholar] [CrossRef] [PubMed]
- Galal, T.M.; Shehata, H.S. Impact of nutrients and heavy metals capture by weeds on the growth and production of rice (Oryza sativa L.) irrigated with different water sources. Ecol. Indic. 2015, 54, 108–115. [Google Scholar] [CrossRef]
- Kaur, S.; Kaur, R.; Chauhan, B.S. Understanding crop-weed-fertilizer-water interactions and their implications for weed management in agricultural systems. Crop Prot. 2018, 103, 65–72. [Google Scholar] [CrossRef]
- Palansooriya, K.N.; Shaheen, S.M.; Chen, S.S.; Tsang, D.C.W.; Hashimoto, Y.; Hou, D.; Bolan, N.S.; Rinklebe, J.; Ok, Y.S. Soil amendments for immobilization of potentially toxic elements in contaminated soils: A critical review. Environ. Int. 2020, 134, 105046. [Google Scholar] [CrossRef] [PubMed]
- Fu, B.; Chen, L.; Huang, H.; Qu, P.; Wei, Z. Impacts of crop residues on soil health: A review. Environ. Pollut. Bioavailab. 2021, 33, 164–173. [Google Scholar] [CrossRef]
- Barman, S.C.; Sahu, R.K.; Bhargava, S.K.; Chaterjee, C. Distribution of heavy metals in wheat, mustard, and weed grown in field irrigated with industrial effluents. Bull. Environ. Contam. Toxicol. 2000, 64, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.D.; Liu, D.S.; Tian, J.C.; He, F.L. Toxicity and accumulation of arsenic in wheat (Triticum aestivum L.) varieties of China. Phyton 2009, 78, 147–154. [Google Scholar]
- Asdeo, A. Toxic metal contamination of staple crops (wheat and millet) in periurban area of Western Rajasthan. Int. Ref. J. Eng. Sci. 2014, 3, 8–18. [Google Scholar]
- Impa, S.M.; Morete, M.J.; Ismail, A.M.; Schulin, R.; Johnson-Beebout, S.E. Zn uptake, translocation and grain Zn loading in rice (Oryza sativa L.) genotypes selected for Zn deficiency tolerance and high grain Zn. J. Exp. Bot. 2013, 64, 2739–2751. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Canqui, H.; Ruis, S.J.; Proctor, C.A.; Creech, C.F.; Drewnoski, M.E.; Redfearn, D.D. Harvesting cover crops for biofuel and livestock production: Another ecosystem service? Agron. J. 2020, 112, 2373–2400. [Google Scholar] [CrossRef]
| Fertilizer Symbol | Raw Material | Bacteria | Form |
|---|---|---|---|
| AsBm | sewage sludge ash (SSA) | Bacillus megaterium | suspension |
| BsBm | bones | Bacillus megaterium | suspension |
| AgAf | SSA | Acidithiobacillus ferrooxidans | granules |
| ABgAf | SSA + bones | Acidithiobacillus ferrooxidans | granules |
| ABg | SSA + bones | no bacteria | granules |
| ABgBm | SSA + bones | Bacillus megaterium | granules |
| Element | Unit | AsBm | BsBm | AgAf | ABgAf | ABg | ABgBm |
|---|---|---|---|---|---|---|---|
| P | % mass | 0.176 | 0.259 | 9.24 | 7.50 | 6.10 | 5.87 |
| N | 0.255 | 0.350 | 0.530 | 3.190 | 3.82 | 3.14 | |
| K | 0.487 | 0.217 | 0.963 | 0.727 | 0.846 | 0.772 | |
| Ca | 0.694 | 0.373 | 12.513 | 12.012 | 11.369 | 10.725 | |
| Mg | 0.119 | 0.009 | 2.442 | 1.176 | 1.296 | 1.188 | |
| S | 0.055 | 0.046 | 1.38 | 1.50 | 2.28 | 1.91 | |
| Na | 0.049 | 0.037 | 0.338 | 0.569 | 0.382 | 0.366 | |
| C | 0.590 | 1.650 | 6.71 | 23.6 | 17.6 | 16.1 | |
| Fe | g kg−1 | 1.679 | 0.022 | 33.0 | 15.1 | 15.4 | 14.4 |
| Al | 1.774 | 0.008 | 24.4 | 13.9 | 12.8 | 11.3 | |
| Zn | 0.117 | 0.007 | 2.00 | 1.18 | 1.75 | 1.56 | |
| As | mg·kg−1 | <0.5 | <0.5 | 4.35 | 2.55 | 26.5 | <0.5 |
| Cd | 0.274 | 0.01 | 0.43 | 0.24 | 0.38 | 0.84 | |
| Cr | 5.94 | 0.218 | 135 | 60.0 | 114 | 94.7 | |
| Cu | 55.0 | 0.433 | 880 | 398 | 470 | 444 | |
| Ni | 2.45 | 0.212 | 57.7 | 25.1 | 52.3 | 44.3 | |
| Pb | 10.4 | 1.04 | 21.6 | 11.2 | 34.0 | 14.5 |
| Regulation | As | Cd | Cr | Cu | Hg | Ni | Pb | Zn |
|---|---|---|---|---|---|---|---|---|
| Poland [64] | 50 | 50 | n.s. | n.s. | 2.0 | n.s. | 140 | n.s. |
| EU [24] | 40 | 60 1 | n.s. | n.s. | 1.0 | 100 | 120 | n.s. |
| Experiment | Year | Test Plant | Recycled Fertilizers | Reference Treatments | P Doses, kg·ha−1 | Plant Protection (PP) |
|---|---|---|---|---|---|---|
| I | 2014 | spring wheat | AsBm, BsBm | No P, SP | 21 | PP− |
| II | 2015 | spring wheat | AsBm | No P, SP | 17.6, 26.4, 35.2 | PP−, PP+ |
| III | 2015 | winter wheat | AgAf, ABgAf | No P, SP | 17.6, 26.4, 35.2 | PP+ |
| IV | 2016 | winter wheat | ABg, ABgBm | No P, SP | 17.6, 26.4, 35.2 | PP+ |
| Item | Experiment | |||
|---|---|---|---|---|
| I | II | III | IV | |
| Wheat cultivar | Trappe | Monsun | Julius | Julius |
| Previous crop | spring barley | cereal-legume mixture | winter rape | winter rape |
| Soil tillage system | plough tillage | plough tillage | plough tillage | plough tillage |
| Fertilization | ||||
| – K, kg·ha−1 | 99.6 | 83 | 83 | 83 |
| potassium chloride | potassium chloride | potassium chloride | potassium chloride | |
| – N, kg·ha−1 | 100 | 110 | 130 | 120 |
| ammonium sulphate | ammonium sulphate | ammonium sulphate | ammonium sulphate | |
| Plant protection | ||||
| – herbicides | MCPA 1 | 2.4-D + florasulam | 2.4-D + florasulam | |
| – fungicides | azoxystrobin 1 + propiconazole | fenpropimorph + epoxiconazole + metrafenon | fenpropimorph + epoxiconazole + metrafenon | |
| cyproconazole 1 | fluksapyroksad + piraklostrobina + epoxiconazole | fluksapyroksad + piraklostrobina + epoxiconazole | ||
| – insecticides | lambda-cyhalothrin 1 | deltamethrin | deltamethrin | |
| – growth regulators | trinexapac ethyl | trinexapac ethyl | ||
| Sowing date | 25.04.2014 | 9.04.2015 | 2.10.2014 | 15.10.2015 |
| Harvest date | 11.08.2014 | 11.08.2015 | 5.08.2015 | 9.08.2016 |
| Properties | Experiment | ||||
|---|---|---|---|---|---|
| I (n = 16) | II (n = 56) | III (n = 40) | IV (n = 40) | ||
| Soil type 1 | Luvisols | Luvisols | Luvisols | Luvisols | |
| Soil texture 2 | sandy clay loam | sandy loam | sandy loam | sandy loam | |
| pH in KCl 3 | 6.23 | 5.32 | 5.51 | 5.23 | |
| Total, 4 | C, g·kg−1 | 8.31 | 8.90 | 8.87 | 7.15 |
| N, g·kg−1 | 1.30 | 1.35 | 1.36 | 1.09 | |
| P, mg·kg−1 | 574 | 566 | 433 | 548 | |
| K, mg·kg−1 | 2979 | 2895 | 3210 | 3304 | |
| Mg, mg·kg−1 | 2070 | 2007 | 2253 | 2195 | |
| Cd, mg·kg−1 | 0.025 (0.365) | 0.292 (0.827) | 0.309 (1.083) | 0.133 (0.553) | |
| Pb, mg·kg−1 | 4.855 (13.198) | 7.959 (25.099) | 7.059 (22.895) | 2.297 (18.180) | |
| As, mg·kg−1 | 2.012 (8.388) | 0.574 (3.013) | <0.5 (<0.5) | 2.067 (8.390) | |
| Cu, mg·kg−1 | 10.66 (14.97) | 10.28 (14.92) | 11.90 (19.44) | 9.113 (10.860) | |
| Cr, mg·kg−1 | 19.96 (23.72) | 19.60 (23.95) | 20.85 (32.07) | 23.34 (28.37) | |
| Ni, mg·kg−1 | 7.16 (11.25) | 8.53 (14.78) | 8.05 (18.27) | 10.69 (15.71) | |
| Zn, mg·kg−1 | 33.60 (43.52) | 60.30 (78.80) | 51.08 (64.98) | 41.69 (56.22) | |
| Content | As | Cr | Cu | Ni | Zn |
|---|---|---|---|---|---|
| In Soil | |||||
| Geochemical background for Poland [69,70] | 2−13 | 2–64 | 0.4–23.5 | 0.5–28.5 | 5.0–59 |
| In surface level of mineral soils in Poland [71] | 0.6−10 | 5−100 | 0.5–25 | 0.5–60 | 10–225 |
| Permissible in Poland in arable land [72] | |||||
| – I 1 | 10 | 150 | 100 | 100 | 300 |
| – II | 20 | 300 | 150 | 150 | 500 |
| – III | 50 | 500 | 300 | 300 | 1000 |
| Monitoring of arable soils [73] | |||||
| – Poland | 0.73–20.7 | 2.4–49.1 | 1.2–320.1 | 1.0–71.1 | 5.9–6668.3 |
| – – average | 3.63 | 11.0 | 10.3 | 9.8 | 78.8 |
| – – median | 2.77 | 8.7 | 6.2 | 6.1 | 32.0 |
| – region 2 | 1.52–3.87 | 4.5–30.6 | 3.3−17.1 | 2.6–29.8 | 19.4–61.9 |
| In Plant | |||||
| Sufficient or normal [4] | 1−1.7 | 0.1–0.5 | 5–30 | 0.1–5 | 27−150 |
| Excessive or toxic [4] | 5–20 | 5–30 | 20−100 | 10−100 | 100–400 |
| Average in cereal grain [71] | 0.03–0.6 | 0.003–0.4 | 2.6–6 | 0.1–0.5 | 15–60 |
| Permissible in Poland | |||||
| – in cereal grain [74] 3 | 0.20 | n.s. | n.s. | n.s. | n.s. |
| – in feed materials of plant origin [75,76] | 2 | n.s. | n.s. | n.s. | n.s. |
| Other standards for food grain | |||||
| – FAO/WHO [77] | 0.35 (0.2) 4 | n.s. | n.s. | n.s. | n.s. |
| – EU [78] | 0.1–0.25 5 | n.s. | n.s. | n.s. | n.s. |
| – PR of China [79] | 0.5 (0.2) 6 | 0.1 | n.s. | n.s. | n.s. |
| Suggested maximum values in plant material [80,81] | |||||
| – for food purposes | n.i. | n.i. | 20 | 10 | 50 |
| – for feed purposes | n.i. | 20 | 25–50 | 50 | 100 |
| Year | Days | Month | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| I | II | III | IV | V | VI | VII | VIII | IX | X | XI | XII | ||
| Precipitation, mm | |||||||||||||
| 2014 | 1−10 | 16.7 | 15.0 | 15.7 | 11.8 | 37.3 | 12.6 | 0.0 | 9.5 | 0.0 | |||
| 11–20 | 5.6 | 2.3 | 21.5 | 8.6 | 6.8 | 0.0 | 20.8 | 6.2 | 12.8 | ||||
| 21–31 | 3.8 | 17.6 | 35.0 | 0.0 | 15.1 | 18.2 | 0.5 | 5.5 | 43.8 | ||||
| Total | 26.1 | 34.9 | 72.2 | 20.4 | 59.2 | 30.8 | 21.3 | 21.2 | 56.6 | ||||
| 2015 | 1−10 | 20.7 | 5.7 | 19.2 | 8.1 | 7.2 | 0.1 | 13.6 | 2.0 | 44.0 | 0.0 | 30.7 | 9.3 |
| 11–20 | 0.4 | 2.0 | 0.0 | 10.9 | 17.7 | 11.5 | 22.0 | 0.0 | 6.8 | 18.6 | 39.3 | 26.4 | |
| 21–31 | 7.4 | 1.1 | 26.8 | 4.4 | 0.5 | 31.4 | 35.4 | 11.0 | 0.4 | 2.2 | 10.8 | 44.7 | |
| Total | 28.5 | 8.8 | 46.0 | 23.4 | 25.4 | 43.0 | 71.0 | 13.0 | 51.2 | 20.8 | 80.8 | 80.4 | |
| 2016 | 1−10 | 0.3 | 11.8 | 0.6 | 4.8 | 7.5 | 12.1 | 39.6 | 54.5 | 7.5 | 65.1 | 39.9 | 41.4 |
| 11–20 | 9.0 | 23.7 | 7.0 | 19.2 | 53.7 | 28.1 | 34.0 | 10.4 | 1.2 | 9.2 | 27.7 | 19.2 | |
| 21–31 | 19.4 | 15.0 | 12.9 | 9.1 | 9.6 | 26.1 | 65.0 | 7.0 | 8.4 | 22.0 | 10.6 | 17.2 | |
| Total | 28.7 | 50.5 | 20.5 | 33.1 | 70.8 | 66.3 | 138.6 | 71.9 | 17.1 | 96.3 | 78.2 | 77.8 | |
| 1981–2010 | Total | 30.1 | 23.1 | 30.7 | 29.8 | 62.3 | 72.9 | 81.2 | 70.6 | 56.2 | 51.2 | 46.1 | 42.6 |
| Air temperature, °C | |||||||||||||
| 2014 | 1−10 | 7.0 | 8.9 | 16.5 | 20.5 | 22.2 | 15.0 | 11.3 | 8.5 | −1.7 | |||
| 11–20 | 8.5 | 13.3 | 14.2 | 19.6 | 17.2 | 17.0 | 12.2 | 4.7 | 3.5 | ||||
| 21–31 | 12.9 | 17.1 | 13.8 | 22.8 | 14.6 | 11.6 | 5.4 | 0.0 | −3.2 | ||||
| Average | 9.5 | 13.3 | 14.8 | 21.0 | 17.9 | 14.5 | 9.5 | 4.4 | −0.6 | ||||
| 2015 | 1−10 | 0.0 | −1.8 | 4.2 | 4.1 | 11.7 | 15.7 | 19.8 | 23.3 | 15.2 | 7.9 | 6.3 | 4.7 |
| 11–20 | 2.7 | −0.1 | 4.7 | 7.1 | 11.6 | 15.9 | 16.9 | 21.1 | 15.9 | 6.0 | 7.9 | 3.1 | |
| 21–31 | −0.9 | 3.3 | 4.9 | 10.4 | 13.0 | 15.5 | 17.4 | 19.5 | 11.6 | 6.0 | 1.2 | 3.5 | |
| Average | 0.6 | 0.3 | 4.6 | 7.2 | 12.1 | 15.7 | 18.0 | 21.3 | 14.2 | 6.6 | 5.1 | 3.8 | |
| 2016 | 1−10 | −8.4 | 4.1 | 2.9 | 10.3 | 14.1 | 16.4 | 17.5 | 17.7 | 17.5 | 9.0 | 2.7 | 1.9 |
| 11–20 | −4.3 | 2.0 | 2.2 | 9.1 | 11.8 | 16.3 | 18.1 | 15.6 | 15.1 | 5.7 | 2.7 | −0.3 | |
| 21–31 | 0.7 | 2.1 | 5.5 | 6.9 | 18.5 | 21.3 | 19.9 | 19.3 | 11.7 | 6.1 | 2.1 | 1.4 | |
| Average | −3.8 | 2.7 | 3.6 | 8.8 | 14.9 | 18.0 | 18.5 | 17.6 | 14.7 | 6.9 | 2.5 | 1.0 | |
| 1981–2010 | Average | −2.4 | −1.6 | 1.8 | 7.7 | 13.2 | 15.8 | 18.3 | 17.7 | 13.0 | 8.1 | 2.8 | −1.0 |
| Fertilizers | As | Cr | Cu | Ni | Zn |
|---|---|---|---|---|---|
| Input per 1 ha | |||||
| g | kg | kg | kg | kg | |
| SP 1 | 2.633–5.265 | 0.019–0.038 | 0.002–0.005 | 0.005–0.009 | 0.025–0.049 |
| AsBm | 5.00−10.00 2 | 0.059–0.119 | 0.550−1.100 | 0.025–0.049 | 1.170–2.340 |
| BsBm | 3.398–6.795 2 | 0.001–0.003 | 0.003–0.006 | 0.001–0.003 | 0.048–0.095 |
| AgAf | 0.829−1.657 | 0.026–0.051 | 0.168–0.335 | 0.011–0.022 | 0.381–0.762 |
| ABgAf | 0.598−1.197 | 0.014–0.028 | 0.093–0.187 | 0.006–0.012 | 0.277–0.554 |
| ABg | 7.646−15.292 | 0.033–0.066 | 0.136–0.271 | 0.015–0.030 | 0.505−1.010 |
| ABgBm | 0.150–0.300 | 0.028–0.057 | 0.133–0.266 | 0.013–0.027 | 0.468–0.935 |
| Limit values per year 3 | n.s | n.s. (0.04−12.0) 4 | 12 | 3 | 30 |
| Potential Increase in Soil Content, µg kg−1 of Soil DM | |||||
| SP | 0.59−1.17 | 4.26–8.52 | 0.51−1.02 | 1.01–2.03 | 5.47−10.94 |
| AsBm | 1.11–2.22 2 | 13.20–26.40 | 122.2–244.4 | 5.44−10.89 | 260.0–520 |
| BsBm | 0.76−1.51 2 | 0.33–0.66 | 0.65−1.31 | 0.32–0.64 | 10.6–21.14 |
| AgAf | 0.18–0.37 | 5.71−11.43 | 37.2–74.5 | 2.44–4.88 | 84.7−169 |
| ABgAf | 0.13–0.27 | 3.13–6.26 | 20.8–41.5 | 1.31–2.62 | 61.5−123 |
| ABg | 1.70–3.40 | 7.31−14.62 | 30.1–60.3 | 3.35–6.71 | 112–224 |
| ABgBm | 0.03–0.07 | 6.31−12.62 | 29.6–59.2 | 2.95–5.90 | 104–208 |
| Experiment | P-Fertilizer | P-Dose, kg·ha−1 | Plant Protection | As | Cr | Cu | Ni | Zn |
|---|---|---|---|---|---|---|---|---|
| I | No P | 0 | PP− | 1.854 | 18.1 | 7.07 ↓ | 5.31 ↓ | 26.1 ↓ |
| SP 1 | 21 | 2.096 | 19.6 | 8.20 ↓ | 5.06 ↓ | 28.2 ↓ | ||
| AsBm | 21 | 1.512 | 19.3 | 8.12 ↓ | 6.03 ↓ | 27.6 ↓ | ||
| BsBm | 21 | 0.996 | 19.0 | 9.32 ↓ | 6.38 ↓ | 27.2 ↓ | ||
| Me | <DL | 18.6 | 8.51 | 5.43 | 27.2 | |||
| Max | 6.885 | 22.8 | 10.93 | 9.04 | 30.8 | |||
| II | No P | 0 | PP− | 0.776 | 15.8 ↓2 | 6.17 ↓ | 7.67 ↓ | 43.3 ↓ |
| SP | 17.6 | <DL | 16.5 ↓ | 6.87 ↓ | 7.10 ↓ | 43.2 ↓ | ||
| 26.4 | <DL | 15.3 ↓ | 6.54 ↓ | 7.71 ↓ | 42.3 ↓ | |||
| 35.2 | 2.598 | 15.3 ↓ | 5.37 ↓ | 7.94 ↓ | 42.7 ↓ | |||
| AsBm | 17.6 | 3.047 | 15.8 ↓ | 6.47 ↓ | 7.98 ↓ | 42.9 ↓ | ||
| 26.4 | 0.814 | 15.4 ↓ | 5.80 ↓ | 7.59 ↓ | 42.3 ↓ | |||
| 35.2 | <DL | 15.8 ↓ | 5.48 ↓ | 7.94 ↓ | 42.9 ↓ | |||
| average | 1.248 | 15.7 ↓ | 6.10 ↓ | 7.71 ↓ | 42.8 ↓ | |||
| No P | 0 | PP+ | <DL | 15.8 ↓ | 6.51 ↓ | 7.34 ↓ | 43.3 ↓ | |
| SP | 17.6 | 1.925 | 16.0 ↓ | 5.62 ↓ | 7.57 ↓ | 43.1 ↓ | ||
| 26.4 | <DL | 17.4 ↓ | 5.79 ↓ | 7.56 ↓ | 43.5 ↓ | |||
| 35.2 | 3.280 | 16.3 ↓ | 7.37 ↓ | 7.38 ↓ | 42.0 ↓ | |||
| AsBm | 17.6 | <DL | 14.7 ↓ | 6.11 ↓ | 7.59 ↓ | 42.9 ↓ | ||
| 26.4 | <DL | 15.3 ↓ | 5.37 ↓ | 7.65 ↓ | 41.8 ↓ | |||
| 35.2 | <DL | 16.3 ↓ | 7.82 ↓ | 7.16 ↓ | 42.3 ↓ | |||
| average | 1.101 | 16.0 ↓ | 6.37 ↓ | 7.46 ↓ | 42.7 ↓ | |||
| Me | <DL | 14.7 | 5.54 | 7.34 | 42.4 | |||
| Max | 8.841 | 21.5 | 11.0 | 14.8 | 51.5 | |||
| III | No P | 0 | PP+ | 1.257 | 19.8 ↓ | 7.29 ↓ | 8.77 | 38.0 ↓ |
| SP | 17.6 | 1.428 | 19.7 ↓ | 8.67 ↓ | 5.53 | 40.1 ↓ | ||
| 26.4 | <DL | 19.3 ↓ | 8.62 ↓ | 8.43 | 41.9 ↓ | |||
| 35.2 | <DL | 19.0 ↓ | 8.34 ↓ | 5.87 | 40.4 ↓ | |||
| AgAf | 17.6 | <DL | 19.2 ↓ | 9.32 ↓ | 7.15 | 40.5 ↓ | ||
| 26.4 | 1.113 | 19.8 ↓ | 9.26 ↓ | 8.76 | 37.3 ↓ | |||
| 35.2 | <DL | 19.3 ↓ | 8.16 ↓ | 6.04 | 39.1 ↓ | |||
| ABgAf | 17.6 | 0.711 | 19.3 ↓ | 8.23 ↓ | 5.67 | 39.2 ↓ | ||
| 26.4 | 0.806 | 19.6 ↓ | 7.20 ↓ | 6.24 | 38.7 ↓ | |||
| 35.2 | 0.705 | 20.0 ↓ | 8.26 ↓ | 8.35 | 38.2 ↓ | |||
| Me | <DL | 19.9 | 7.84 | 7.37 | 38.6 | |||
| Max | 2.950 | 23.2 | 13.56 | 12.2 | 47.4 | |||
| IV | No P | 0 | PP+ | 1.319 | 18.7 ↓ | 7.57 ↓ | 9.91 | 37.6 ↓ |
| SP | 17.6 | 1.242 | 20.9 ↓ | 8.13 ↓ | 9.95 | 38.9 ↓ | ||
| 26.4 | 1.356 | 19.6 ↓ | 7.65 ↓ | 9.40 | 38.1 ↓ | |||
| 35.2 | 1.286 | 18.1 ↓ | 7.33 ↓ | 9.99 | 38.2 ↓ | |||
| ABg | 17.6 | 0.931 | 18.8 ↓ | 7.96 ↓ | 9.67 | 36.5 ↓ | ||
| 26.4 | 1.154 | 19.7 ↓ | 7.76 ↓ | 10.30 | 37.1 ↓ | |||
| 35.2 | 1.573 | 20.4 ↓ | 7.99 ↓ | 10.20 | 43.1 ↓ | |||
| ABgBm | 17.6 | 0.819 | 15.5 ↓ | 7.99 ↓ | 10.39 | 40.5 ↓ | ||
| 26.4 | 1.513 | 20.5 ↓ | 8.62 ↓ | 10.20 | 37.9 ↓ | |||
| 35.2 | 1.423 | 19.6 ↓ | 7.75 ↓ | 10.12 | 36.2 ↓ | |||
| Me | 0.758 | 19.2 | 7.97 | 10.0 | 37.8 | |||
| Max | 4.082 | 23.8 | 10.6 | 15.1 | 55.0 |
| Experiment | P-Fertilizer | P-Dose, kg·ha−1 | Plant Protection | As | Cr | Cu | Ni | Zn |
|---|---|---|---|---|---|---|---|---|
| I | No P | 0 | PP− | <DL | 0.307 | 2.85 | 0.182 | 22.1 |
| SP 1 | 21 | <DL | 0.118 | 2.77 | 0.132 | 21.9 | ||
| AsBm | 21 | <DL | 0.125 | 3.00 | 0.079 | 23.2 | ||
| BsBm | 21 | <DL | 0.223 | 2.74 | 0.034 | 21.5 | ||
| Me | <DL | 0.141 | 2.79 | 0.096 | 22.1 | |||
| Max | 0.050 | 0.748 | 3.45 | 0.362 | 25.9 | |||
| II | No P | 0 | PP− | 0.054 | 0.233 | 3.70 | 0.028 | 40.9 |
| SP | 17.6 | 0.059 | <DL | 3.70 | 0.065 | 42.3 | ||
| 26.4 | <DL | 0.193 | 3.32 | 0.027 | 39.1 | |||
| 35.2 | <DL | 0.196 | 3.55 | 0.086 | 41.2 | |||
| AsBm | 17.6 | <DL | 0.008 | 3.76 | 0.031 | 47.6 | ||
| 26.4 | <DL | 0.058 | 3.64 | 0.058 | 39.7 | |||
| 35.2 | <DL | 0.084 | 3.87 | 0.033 | 42.8 | |||
| average | 0.047 | 0.111 | 3.65 b 2 | 0.047 | 41.9 | |||
| No P | 0 | PP+ | 0.074 | 0.072 | 4.32 | 0.008 | 39.9 | |
| SP | 17.6 | <DL | 0.083 | 3.91 | 0.008 | 37.2 | ||
| 26.4 | <DL | 0.063 | 4.07 | 0.032 | 40.1 | |||
| 35.2 | 0.052 | <DL | 4.01 | 0.068 | 38.7 | |||
| AsBm | 17.6 | <DL | 0.131 | 3.92 | 0.034 | 41.2 | ||
| 26.4 | <DL | 0.157 | 3.63 | 0.050 | 37.6 | |||
| 35.2 | <DL | 0.018 | 3.58 | 0.016 | 41.3 | |||
| average | 0.051 | 0.076 | 3.92 a | 0.031 | 39.4 | |||
| Me | <DL | 0.011 | 3.79 | 0.022 | 41.0 | |||
| Max | 0.125 | 0.521 | 4.67 | 0.235 | 56.4 | |||
| III | No P | 0 | PP+ | <DL | 0.033 | 2.31 | 0.179 | 26.4 |
| SP | 17.6 | 0.062 | 0.015 | 2.09 | 0.215 | 25.5 | ||
| 26.4 | <DL | <DL | 2.17 | 0.180 | 24.0 | |||
| 35.2 | <DL | 0.159 | 2.09 | 0.245 | 24.2 | |||
| AgAf | 17.6 | 0.057 | 0.132 | 2.50 | 0.149 | 24.7 | ||
| 26.4 | 0.081 | <DL | 2.39 | 0.187 | 24.2 | |||
| 35.2 | <DL | 0.062 | 2.17 | 0.122 | 27.1 | |||
| ABgAf | 17.6 | 0.063 | 0.112 | 2.39 | 0.287 | 27.5 | ||
| 26.4 | 0.058 | 0.133 | 2.43 | 0.182 | 24.6 | |||
| 35.2 | <DL | 0.165 | 2.36 | 0.230 | 27.2 | |||
| Me | <DL | <DL | 2.30 | 0.164 | 25.4 | |||
| Max | 0.173 | 0.630 | 2.93 | 0.691 | 33.2 | |||
| IV | No P | 0 | PP+ | 0.056 | 0.228 | 3.85 | 0.189 | 26.0 |
| SP | 17.6 | <DL | 0.068 | 3.74 | 0.113 | 24.1 | ||
| 26.4 | <DL | 0.176 | 3.57 | 0.125 | 24.1 | |||
| 35.2 | <DL | 0.405 | 3.36 | 0.087 | 22.7 | |||
| ABg | 17.6 | <DL | 0.591 | 4.04 | 0.167 | 24.9 | ||
| 26.4 | 0.065 | 0.314 | 3.74 | 0.144 | 24.3 | |||
| 35.2 | <DL | 0.180 | 3.92 | 0.138 | 25.7 | |||
| ABgBm | 17.6 | 0.052 | 0.143 | 4.05 | 0.149 | 24.2 | ||
| 26.4 | <DL | 0.179 | 3.95 | 0.185 | 25.6 | |||
| 35.2 | <DL | 0.247 | 3.90 | 0.250 | 27.0 | |||
| Me | <DL | 0.168 | 3.81 | 0.126 | 24.8 | |||
| Max | 0.087 | 0.992 | 5.11 | 0.390 | 33.4 |
| Experiment | P-Fertilizer | P-Dose, kg·ha−1 | Plant Protection | As | Cr | Cu | Ni | Zn |
|---|---|---|---|---|---|---|---|---|
| I | No P | 0 | PP− | 0.277 | 0.327 | 2.55 | 0.321 | 5.18 |
| SP 1 | 21 | 0.127 | 0.288 | 2.54 | 0.292 | 2.83 | ||
| AsBm | 21 | 0.250 | 0.311 | 2.61 | 0.245 | 3.27 | ||
| BsBm | 21 | 0.381 | 0.486 | 2.71 | 0.344 | 3.75 | ||
| Me | 0.249 | 0.358 | 2.63 | 0.299 | 1.41 | |||
| Max | 0.578 | 0.988 | 3.07 | 0.496 | 10.83 | |||
| II | No P | 0 | PP− | 0.171 | 2.99 | 3.36 | 0.421 | 18.52 |
| SP | 17.6 | 0.050 | 1.95 | 3.51 | 0.458 | 14.01 | ||
| 26.4 | 0.156 | 3.18 | 3.78 | 0.535 | 13.69 | |||
| 35.2 | 0.073 | 3.46 | 3.53 | 0.576 | 14.51 | |||
| AsBm | 17.6 | 0.081 | 2.49 | 3.43 | 0.454 | 13.30 | ||
| 26.4 | 0.106 | 1.98 | 3.49 | 0.337 | 14.22 | |||
| 35.2 | 0.076 | 2.06 | 3.66 | 0.352 | 13.73 | |||
| average | 0.102 | 2.59 | 3.54 | 0.447 | 14.57 a 2 | |||
| No P | 0 | PP+ | 0.084 | 3.08 | 3.63 | 0.339 | 10.44 | |
| SP | 17.6 | 0.085 | 3.66 | 4.68 | 0.606 | 9.20 | ||
| 26.4 | 0.121 | 2.05 | 4.01 | 0.307 | 9.41 | |||
| 35.2 | 0.069 | 2.16 | 3.76 | 0.263 | 8.30 | |||
| AsBm | 17.6 | 0.112 | 3.58 | 3.69 | 0.692 | 9.35 | ||
| 26.4 | 0.067 | 1.57 | 3.10 | 0.133 | 8.78 | |||
| 35.2 | 0.050 | 2.24 | 4.47 | 0.396 | 9.84 | |||
| average | 0.084 | 2.62 | 3.90 | 0.391 | 9.33 b | |||
| Me | 0.050 | 1.98 | 3.67 | 0.409 | 11.12 | |||
| Max | 0.474 | 5.97 | 5.76 | 1.420 | 24.95 | |||
| III | No P | 0 | PP+ | 0.516 | 0.415 | 6.43 | 0.530 | 6.16 |
| SP | 17.6 | 0.393 | 0.493 | 6.13 | 0.710 | 6.78 | ||
| 26.4 | 0.373 | 0.484 | 6.11 | 0.284 | 6.41 | |||
| 35.2 | 0.556 | 0.385 | 6.06 | 0.440 | 5.98 | |||
| AgAf | 17.6 | 0.343 | 0.315 | 5.83 | 0.277 | 5.98 | ||
| 26.4 | 0.582 | 0.531 | 6.37 | 0.221 | 6.28 | |||
| 35.2 | 0.458 | 0.492 | 4.08 | 0.236 | 6.20 | |||
| ABgAf | 17.6 | 0.515 | 0.430 | 7.02 | 0.248 | 5.84 | ||
| 26.4 | 0.465 | 0.690 | 6.20 | 0.384 | 5.75 | |||
| 35.2 | 0.415 | 0.599 | 6.08 | 0.558 | 6.12 | |||
| Me | 0.426 | 0.450 | 6.17 | 0.284 | 6.29 | |||
| Max | 0.961 | 1.546 | 9.26 | 1.492 | 8.23 | |||
| IV | No P | 0 | PP+ | 0.453 | 0.99 | 4.12 | 0.234 | 7.36 |
| SP | 17.6 | 0.643 | 0.71 | 6.34 | 0.310 | 7.54 | ||
| 26.4 | 0.738 | 1.11 | 5.00 | 0.235 | 6.79 | |||
| 35.2 | 0.448 | 1.03 | 5.39 | 0.256 | 6.82 | |||
| ABg | 17.6 | 0.505 | 0.79 | 5.96 | 0.503 | 7.25 | ||
| 26.4 | 0.475 | 1.29 | 5.04 | 0.296 | 6.17 | |||
| 35.2 | 0.611 | 0.91 | 3.85 | 0.230 | 7.81 | |||
| ABgBm | 17.6 | 0.609 | 0.40 | 5.76 | 0.304 | 7.50 | ||
| 26.4 | 0.786 | 1.25 | 5.72 | 0.288 | 8.34 | |||
| 35.2 | 0.786 | 0.94 | 4.54 | 0.402 | 8.64 | |||
| Me | 0.530 | 0.861 | 4.66 | 0.221 | 7.01 | |||
| Max | 1.561 | 2.651 | 8.97 | 0.896 | 12.48 |
| Experiment | P-Fertilizer | P-Dose, kg·ha−1 | Plant Protection | As | Cr | Cu | Ni | Zn |
|---|---|---|---|---|---|---|---|---|
| I | No P | 0 | PP− | 0.180 | 0.537 | 5.13 | 0.331 | 16.4 |
| SP 1 | 21 | 0.329 | 0.404 | 5.28 | 0.339 | 16.2 | ||
| AsBm | 21 | 0.318 | 0.670 | 5.13 | 0.377 | 17.7 | ||
| BsBm | 21 | 0.254 | 0.524 | 4.91 | 0.384 | 14.7 | ||
| Me | 0.242 | 0.539 | 5.19 | 0.335 | 16.2 | |||
| Max | 0.571 | 0.988 | 5.95 | 0.628 | 20.3 | |||
| II | No P | 0 | PP− | 0.935 | 2.217 | 7.62 | 1.254 | 76.8 |
| SP | 17.6 | 0.942 | 1.983 | 8.01 | 1.460 | 77.7 | ||
| 26.4 | 0.704 | 2.247 | 8.72 | 1.211 | 68.8 | |||
| 35.2 | 0.677 | 1.937 | 8.15 | 1.355 | 80.5 | |||
| AsBm | 17.6 | 0.822 | 1.717 | 7.94 | 1.395 | 79.5 | ||
| 26.4 | 1.095 | 2.178 | 8.73 | 1.429 | 76.1 | |||
| 35.2 | 0.916 | 2.652 | 8.16 | 1.776 | 68.7 | |||
| average | 0.870 a 2 | 2.133 a | 8.19 | 1.411 a | 75.4 a | |||
| No P | 0 | PP+ | 0.140 | 1.545 | 8.80 | 0.908 | 64.1 | |
| SP | 17.6 | 0.322 | 1.718 | 8.18 | 0.850 | 44.2 | ||
| 26.4 | 0.443 | 1.550 | 7.85 | 1.094 | 48.3 | |||
| 35.2 | 0.703 | 1.830 | 7.57 | 0.993 | 49.8 | |||
| AsBm | 17.6 | 0.216 | 1.323 | 7.92 | 1.059 | 52.1 | ||
| 26.4 | 0.146 | 2.129 | 7.11 | 1.294 | 52.0 | |||
| 35.2 | 0.113 | 1.861 | 9.99 | 1.189 | 59.8 | |||
| average | 0.298 b | 1.708 b | 8.20 | 1.055 b | 52.9 b | |||
| Me | 0.476 | 1.832 | 8.04 | 1.178 | 65.5 | |||
| Max | 1.748 | 4.446 | 11.02 | 2.300 | 105.7 | |||
| III | No P | 0 | PP+ | 1.838 | 0.853 | 10.7 | 3.49 | 75.6 |
| SP | 17.6 | 1.586 | 0.298 | 9.7 | 4.86 | 83.1 | ||
| 26.4 | 2.174 | 0.822 | 14.4 | 2.34 | 82.9 | |||
| 35.2 | 1.311 | 0.723 | 10.3 | 4.08 | 82.2 | |||
| AgAf | 17.6 | 1.532 | 0.628 | 11.1 | 4.25 | 82.2 | ||
| 26.4 | 1.354 | 1.254 | 13.8 | 3.27 | 89.8 | |||
| 35.2 | 2.204 | 0.493 | 12.6 | 4.10 | 115.1 | |||
| ABgAf | 17.6 | 1.656 | 0.215 | 15.9 | 3.62 | 74.3 | ||
| 26.4 | 1.648 | 0.351 | 10.3 | 4.76 | 105.6 | |||
| 35.2 | 1.649 | 0.612 | 12.7 | 5.91 | 112.6 | |||
| Me | 1.634 | 1.093 | 10.6 | 3.92 | 102.5 | |||
| Max | 3.073 | 3.073 | 26.4 | 9.39 | 140.5 | |||
| IV | No P | 0 | PP+ | 0.873 | 0.547 | 7.60 | 1.379 | 48.4 |
| SP | 17.6 | 0.985 | 1.878 | 7.89 | 2.161 | 50.1 | ||
| 26.4 | 0.672 | 0.240 | 9.05 | 1.265 | 51.3 | |||
| 35.2 | 0.836 | 1.251 | 8.06 | 1.488 | 40.7 | |||
| ABg | 17.6 | 0.981 | 1.586 | 8.06 | 1.832 | 49.6 | ||
| 26.4 | 1.059 | 1.032 | 8.36 | 2.060 | 55.9 | |||
| 35.2 | 0.856 | 0.578 | 7.60 | 1.765 | 43.6 | |||
| ABgBm | 17.6 | 0.836 | 0.245 | 9.41 | 2.234 | 62.2 | ||
| 26.4 | 0.539 | 0.458 | 8.81 | 1.948 | 59.5 | |||
| 35.2 | 0.774 | 0.736 | 8.92 | 1.838 | 56.7 | |||
| Me | 0.894 | 0.687 | 8.13 | 1.735 | 51.7 | |||
| Max | 1.799 | 3.792 | 12.45 | 4.442 | 76.2 |
| Experiment | P-Fertilizer | P-Dose, kg·ha−1 | Plant Protection | As | Cr | Cu | Ni | Zn |
|---|---|---|---|---|---|---|---|---|
| I | No P | 0 | PP− | 0.577 | 5.64 | 3.52 | 2.08 | 7.79 |
| SP 1 | 21 | 0.627 | 5.75 | 3.90 | 2.26 | 9.61 | ||
| AsBm | 21 | 0.598 | 6.28 | 4.04 | 2.49 | 9.82 | ||
| BsBm | 21 | 0.408 | 5.96 | 4.27 | 2.53 | 8.91 | ||
| Me | 0.541 | 5.92 | 3.87 | 2.31 | 9.17 | |||
| Max | 0.778 | 7.06 | 5.10 | 2.92 | 11.71 | |||
| II | No P | 0 | PP− | 0.438 | 7.97 | 4.17 | 2.24 | 26.0 |
| SP | 17.6 | 0.369 | 6.46 | 4.79 | 1.94 | 19.5 | ||
| 26.4 | 0.603 | 8.64 | 4.59 | 2.26 | 19.7 | |||
| 35.2 | 0.385 | 8.50 | 4.95 | 2.29 | 20.6 | |||
| AsBm | 17.6 | 0.392 | 8.58 | 4.57 | 2.21 | 21.2 | ||
| 26.4 | 0.487 | 8.88 | 4.11 | 2.49 | 25.2 | |||
| 35.2 | 0.548 | 7.23 | 4.41 | 2.31 | 19.9 | |||
| average | 0.460 a 2 | 8.04 | 4.51 b | 2.25 | 21.7 | |||
| No P | 0 | PP+ | 0.372 | 10.83 | 4.67 | 1.88 | 24.0 | |
| SP | 17.6 | 0.318 | 11.35 | 5.64 | 1.95 | 21.9 | ||
| 26.4 | 0.245 | 7.92 | 6.01 | 1.87 | 20.9 | |||
| 35.2 | 0.369 | 9.10 | 5.03 | 2.12 | 25.0 | |||
| AsBm | 17.6 | 0.320 | 7.16 | 4.82 | 2.84 | 22.0 | ||
| 26.4 | 0.309 | 9.05 | 5.90 | 1.74 | 19.0 | |||
| 35.2 | 0.395 | 8.13 | 4.81 | 2.32 | 23.3 | |||
| average | 0.332 b | 9.08 | 5.27 a | 2.10 | 22.3 | |||
| Me | 0.375 | 8.32 | 4.84 | 2.04 | 21.9 | |||
| Max | 0.873 | 16.78 | 8.04 | 4.07 | 34.2 | |||
| III | No P | 0 | PP+ | 0.634 | 8.92 | 5.33 | 2.73 | 23.5 |
| SP | 17.6 | 0.355 | 9.13 | 5.41 | 2.31 | 22.8 | ||
| 26.4 | 0.333 | 9.81 | 5.63 | 2.74 | 20.8 | |||
| 35.2 | 0.232 | 9.78 | 5.59 | 2.60 | 24.3 | |||
| AgAf | 17.6 | 0.452 | 10.35 | 6.10 | 3.12 | 29.5 | ||
| 26.4 | 0.247 | 10.93 | 5.38 | 3.10 | 24.1 | |||
| 35.2 | 0.407 | 9.80 | 5.27 | 2.68 | 23.0 | |||
| ABgAf | 17.6 | 0.361 | 8.80 | 5.34 | 2.19 | 22.8 | ||
| 26.4 | 0.080 | 10.05 | 5.63 | 2.53 | 24.5 | |||
| 35.2 | 0.209 | 8.51 | 5.74 | 2.77 | 25.7 | |||
| Me | 0.227 | 9.75 | 5.32 | 2.51 | 24.3 | |||
| Max | 1.074 | 13.40 | 8.76 | 4.24 | 34.3 | |||
| IV | No P | 0 | PP+ | 0.425 | 6.98 | 4.86 | 4.48 | 20.5 |
| SP | 17.6 | 0.734 | 8.78 | 4.85 | 7.26 | 21.9 | ||
| 26.4 | 0.674 | 8.80 | 5.19 | 6.72 | 22.0 | |||
| 35.2 | 0.379 | 7.81 | 4.48 | 5.28 | 20.0 | |||
| ABg | 17.6 | 0.685 | 9.25 | 4.56 | 6.36 | 22.5 | ||
| 26.4 | 0.695 | 7.43 | 4.13 | 6.46 | 21.3 | |||
| 35.2 | 0.398 | 6.84 | 4.19 | 5.02 | 20.6 | |||
| ABgBm | 17.6 | 0.607 | 7.34 | 4.58 | 5.94 | 19.8 | ||
| 26.4 | 0.975 | 9.82 | 5.12 | 7.86 | 22.0 | |||
| 35.2 | 0.779 | 8.02 | 5.56 | 6.56 | 24.2 | |||
| Me | 0.652 | 7.757 | 4.61 | 6.32 | 21.4 | |||
| Max | 1.801 | 15.297 | 7.19 | 11.54 | 29.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jastrzębska, M.; Kostrzewska, M.K.; Saeid, A.; Jastrzębski, W.P. Do New-Generation Recycled Phosphorus Fertilizers Increase the Content of Potentially Toxic Elements in Soil and Plants? Minerals 2021, 11, 999. https://doi.org/10.3390/min11090999
Jastrzębska M, Kostrzewska MK, Saeid A, Jastrzębski WP. Do New-Generation Recycled Phosphorus Fertilizers Increase the Content of Potentially Toxic Elements in Soil and Plants? Minerals. 2021; 11(9):999. https://doi.org/10.3390/min11090999
Chicago/Turabian StyleJastrzębska, Magdalena, Marta K. Kostrzewska, Agnieszka Saeid, and Wiesław P. Jastrzębski. 2021. "Do New-Generation Recycled Phosphorus Fertilizers Increase the Content of Potentially Toxic Elements in Soil and Plants?" Minerals 11, no. 9: 999. https://doi.org/10.3390/min11090999
APA StyleJastrzębska, M., Kostrzewska, M. K., Saeid, A., & Jastrzębski, W. P. (2021). Do New-Generation Recycled Phosphorus Fertilizers Increase the Content of Potentially Toxic Elements in Soil and Plants? Minerals, 11(9), 999. https://doi.org/10.3390/min11090999








