Worth from Waste: Utilizing a Graphite-Rich Fraction from Spent Lithium-Ion Batteries as Alternative Reductant in Nickel Slag Cleaning
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Experimental Procedure of High-Temperature Process
2.3. Characterization of Sample Composition
| Cu | Co | Li | Ni | Mn | Pb | Sn | Zn | |
|---|---|---|---|---|---|---|---|---|
| External standard for slag | NIST610 [36] | |||||||
| Internal standard for slag | 29Si | |||||||
| Detection limit for slag (ppmw) for the isotope used | 63Cu: 0.010 | 59Co: 0.005 | 7Li: 0.025 | 60Ni: 0.093 | 55Mn: 0.023 | 208Pb: 0.001 | 118Sn: 0.008 | 68Zn: 0.159 |
3. Results and Discussion
3.1. Production of Graphite-Rich Fraction with Froth Flotation
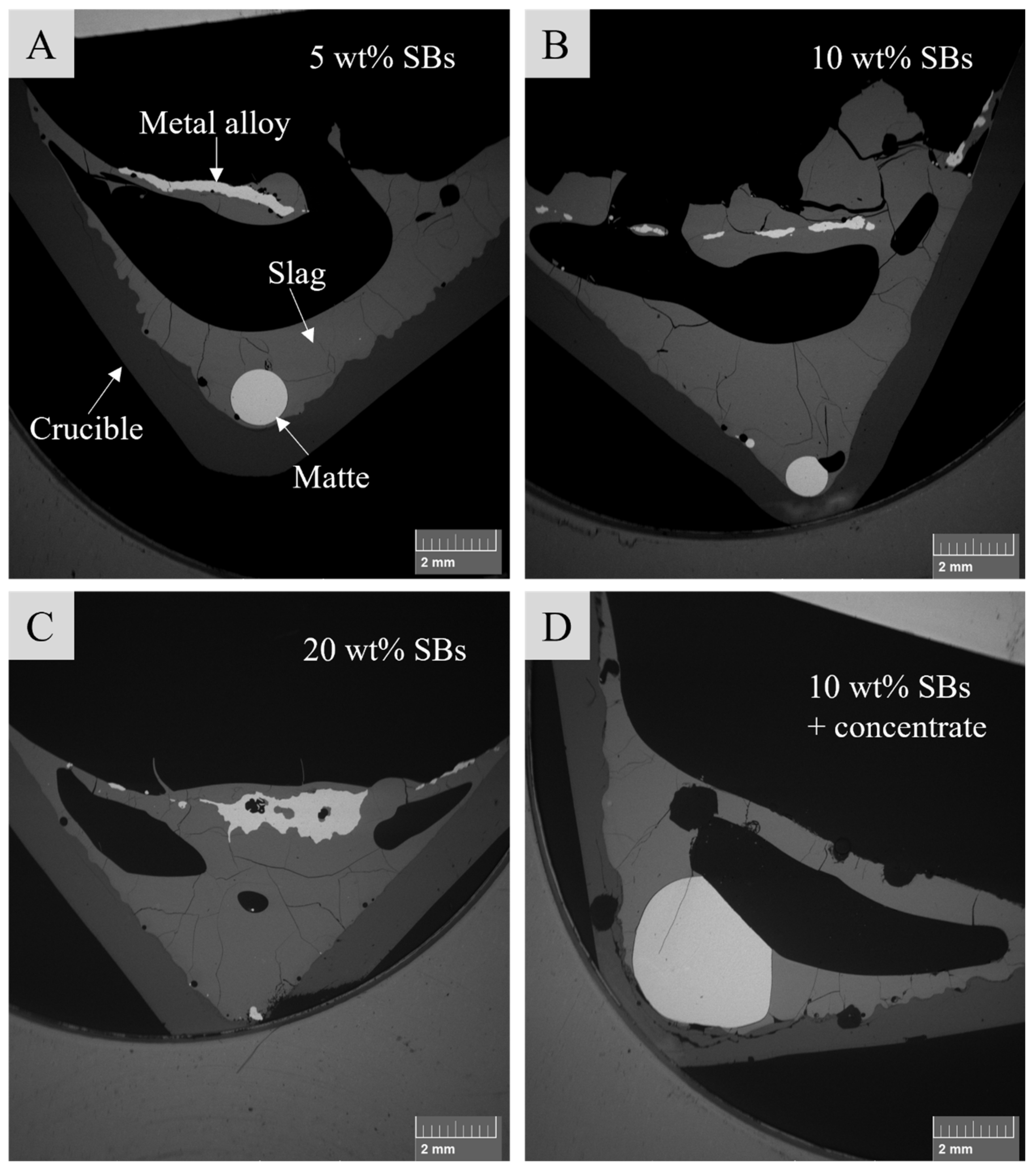
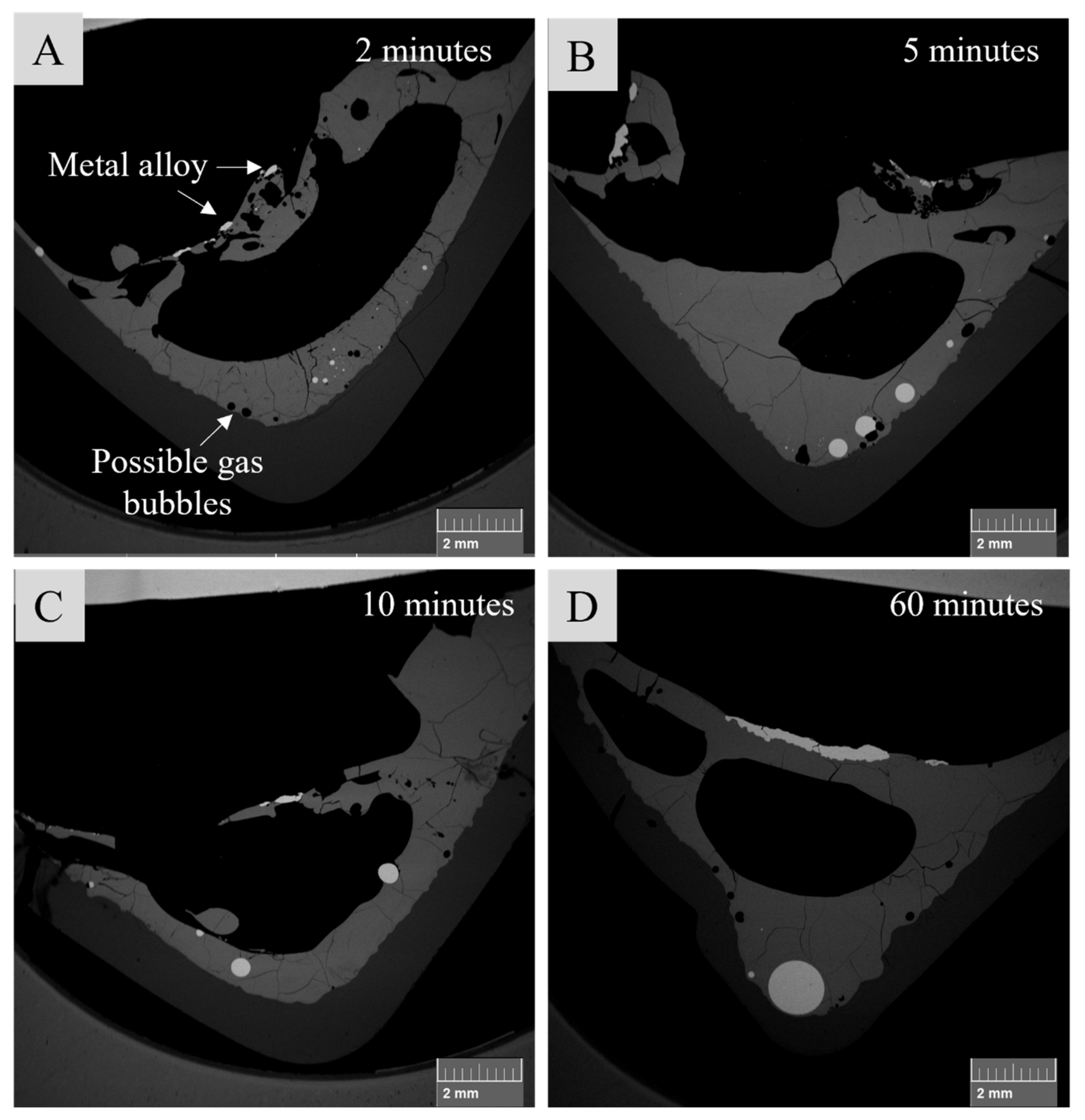
3.2. Sample Microstructure
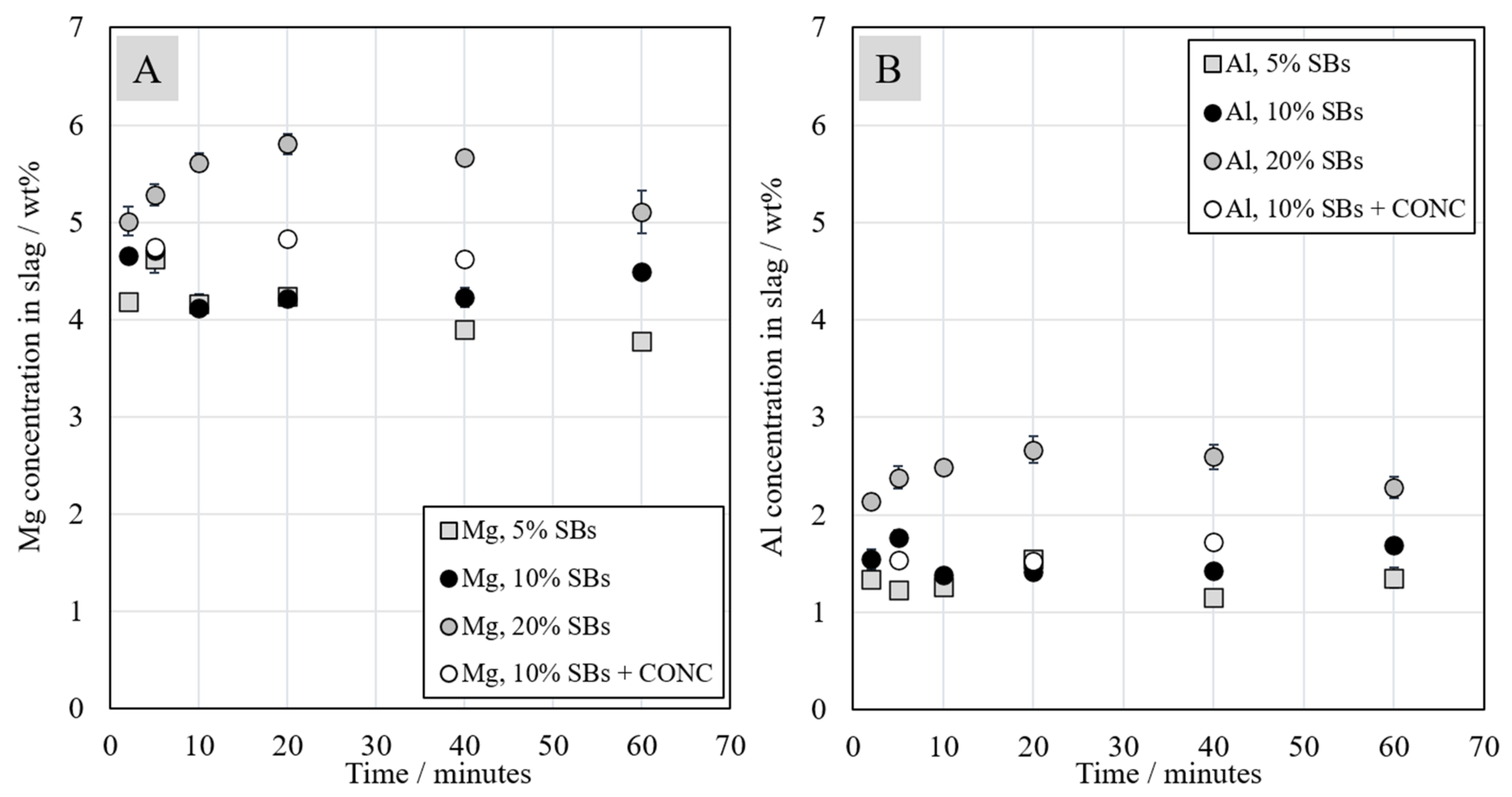
3.3. The Concentrations of Major Elements in the Slag
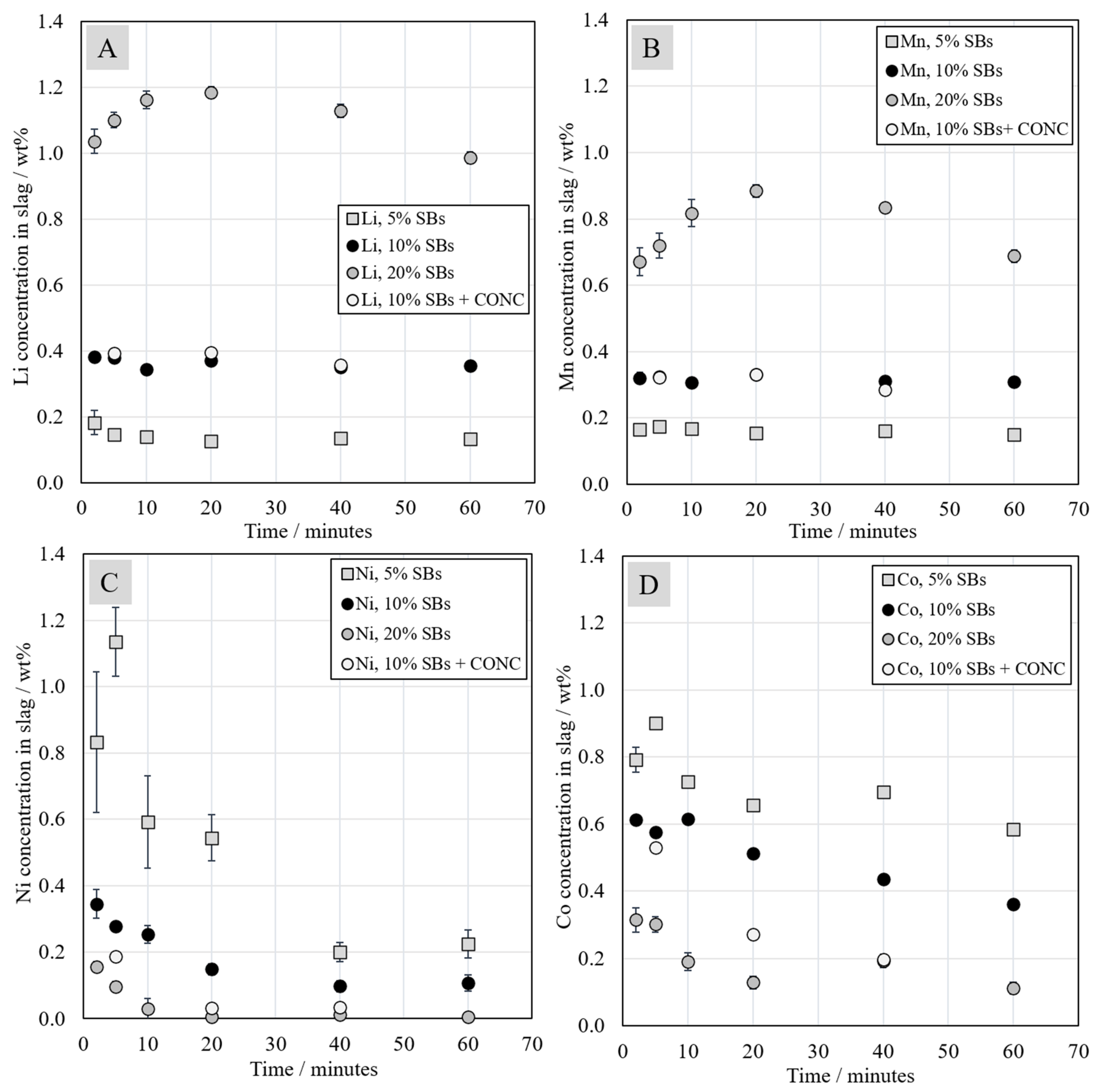
3.4. Trace Elements in the Slag
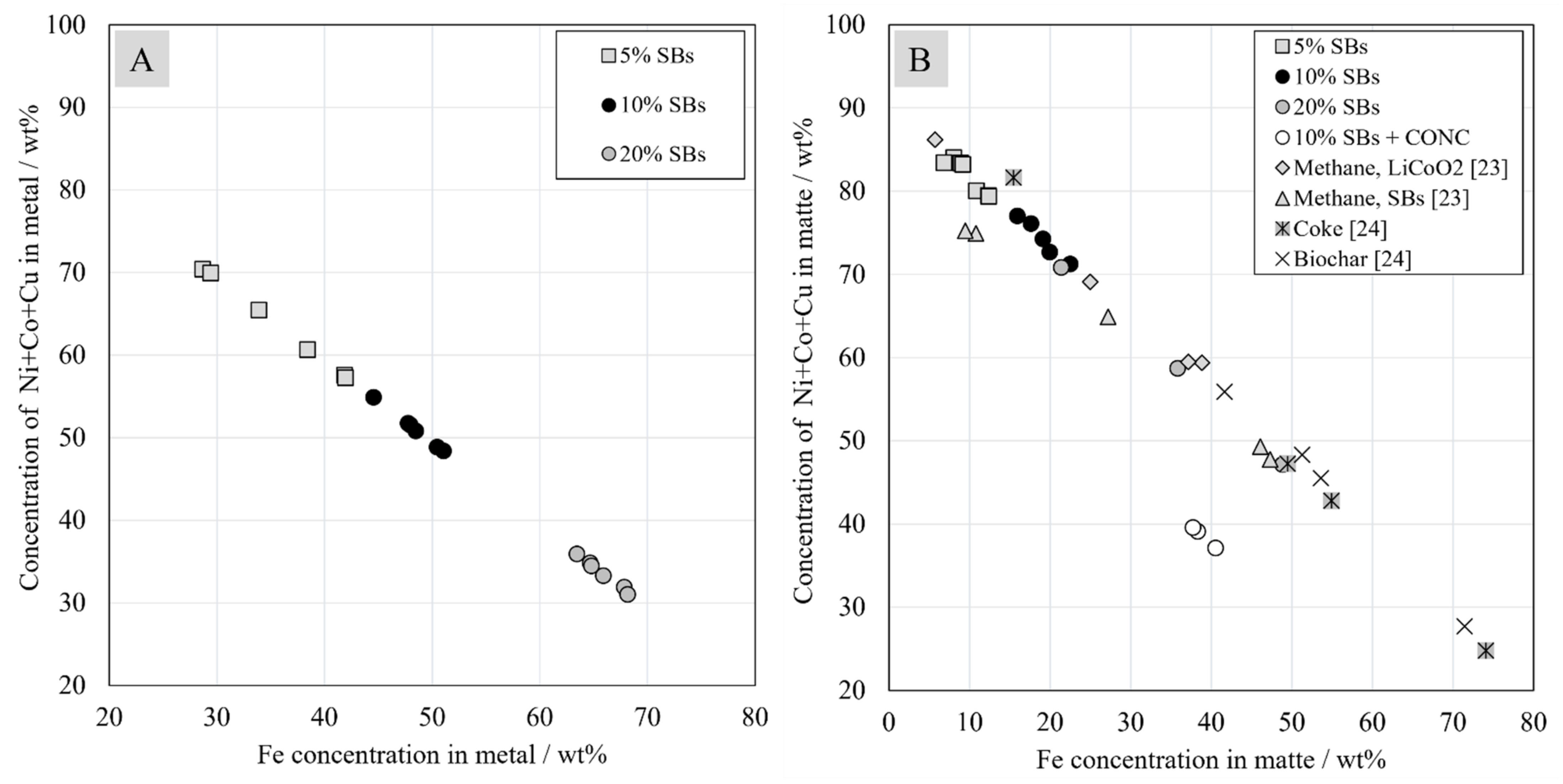
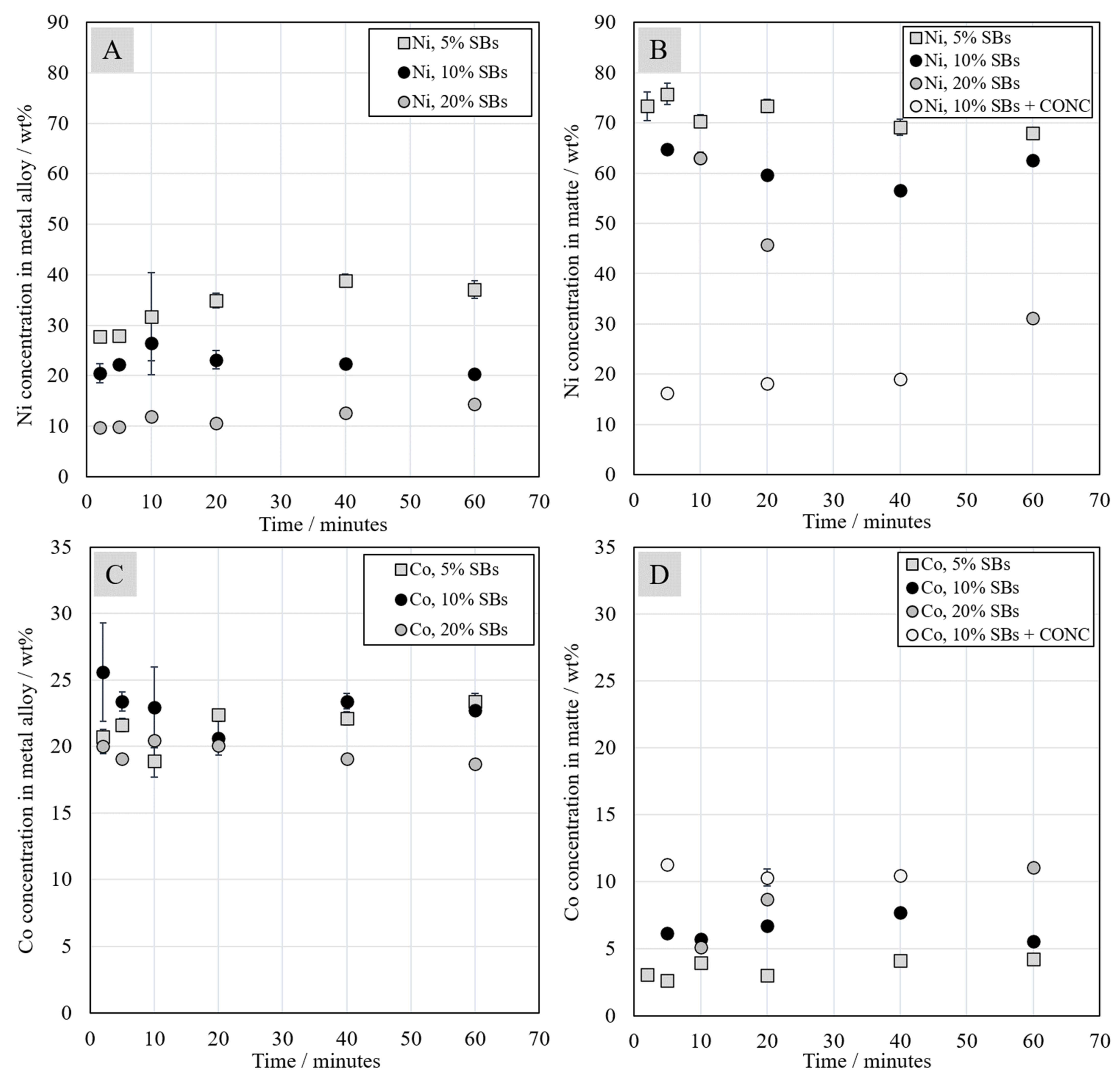
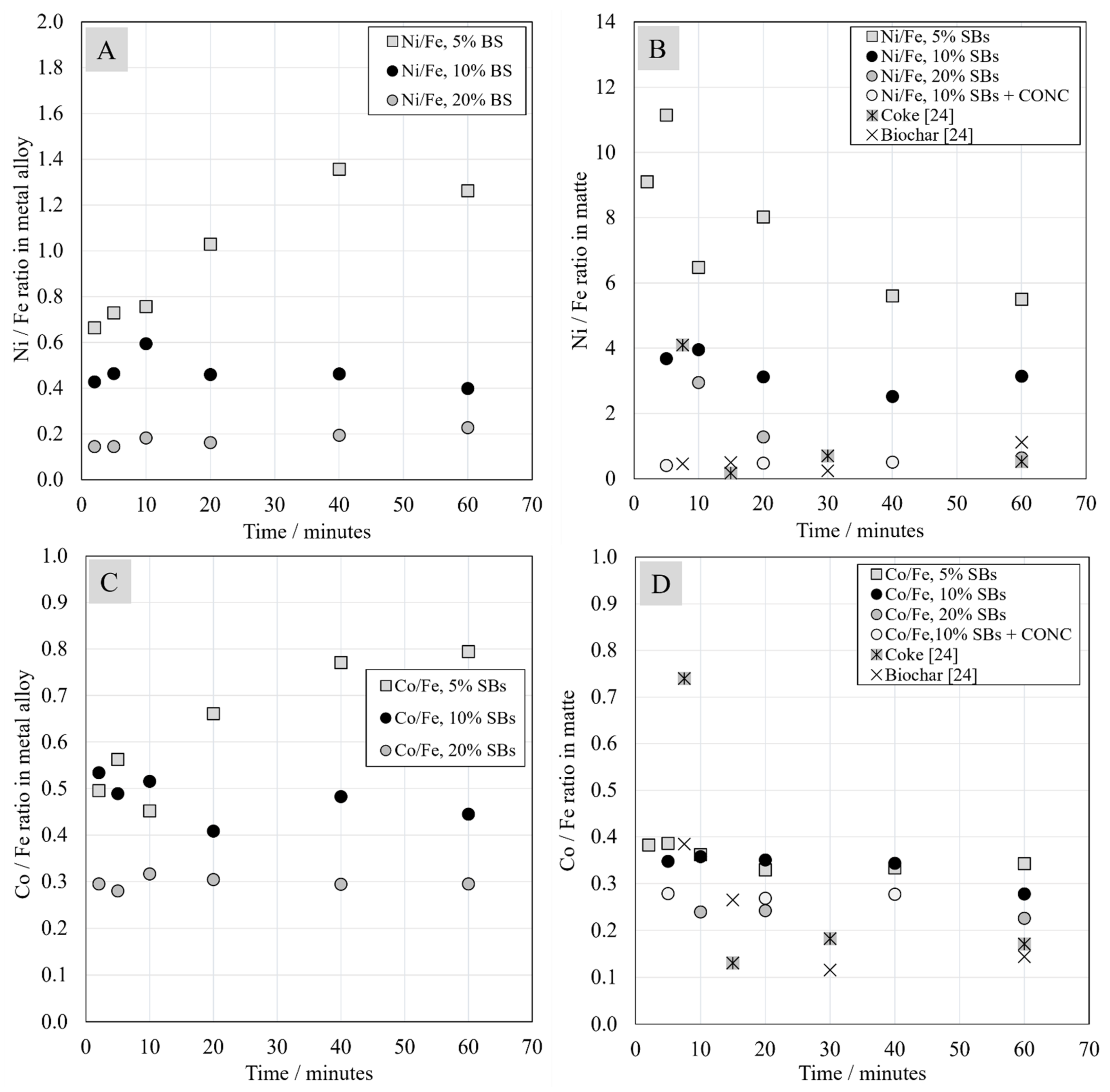
3.5. Chemical Composition of Metal and Matte
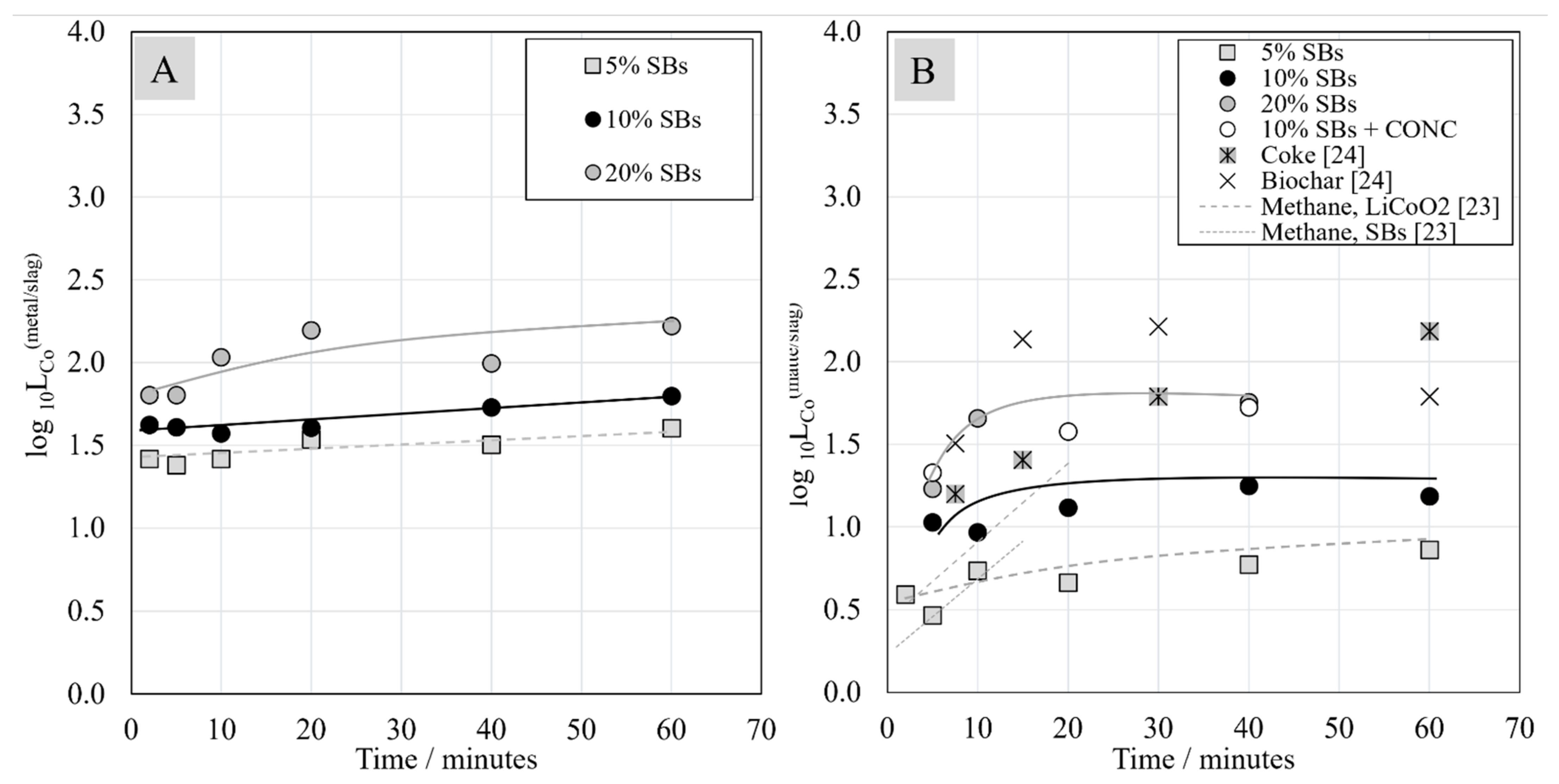
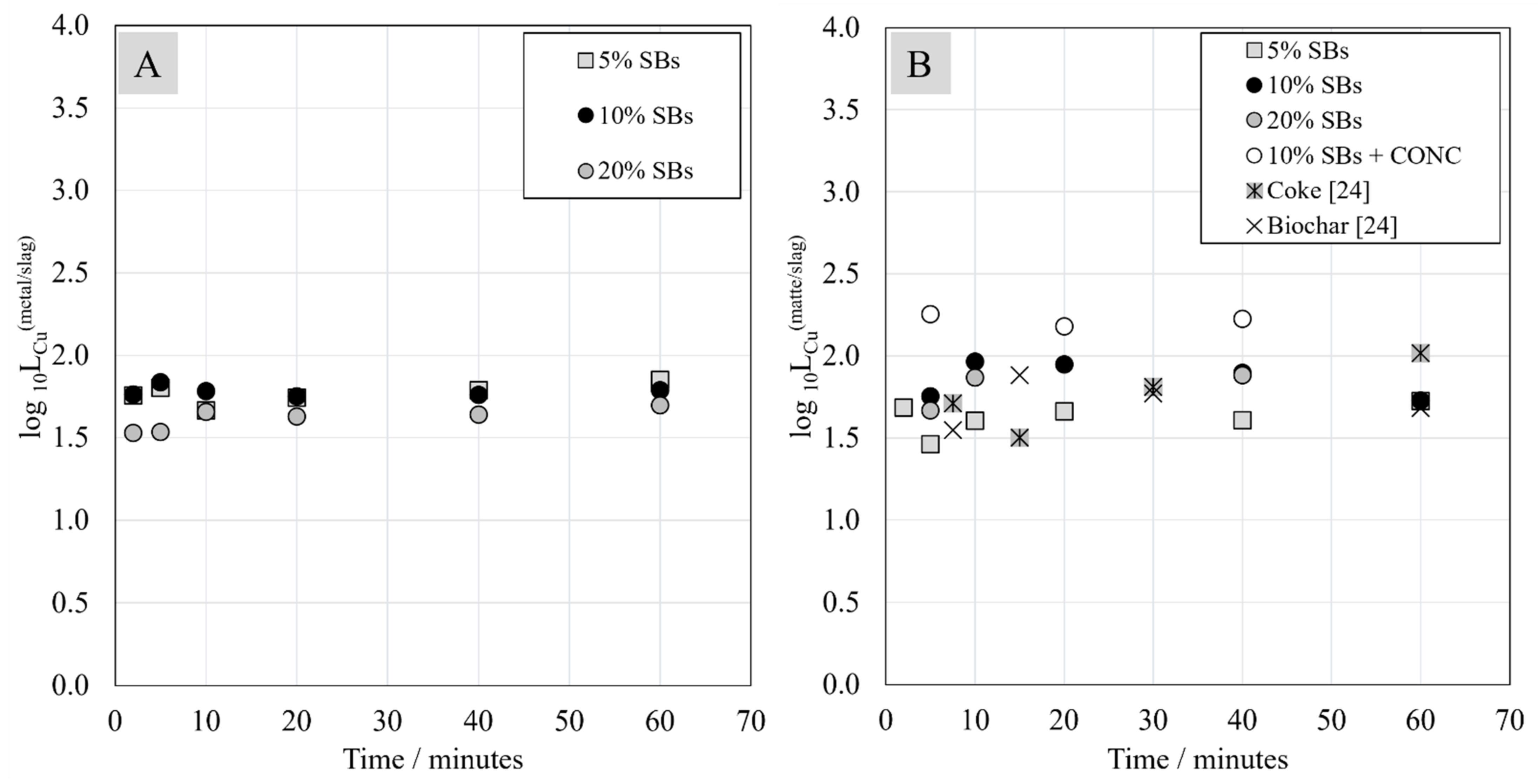
3.6. Distribution Coefficients of Ni, Co, and Cu between Metal, Matte, and Slag
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- European Commission. Questions and Answers on Sustainable Batteries Regulation. Available online: https://ec.europa.eu/commission/presscorner/detail/en/QANDA_20_2311 (accessed on 22 February 2021).
- Makuza, B.; Tian, Q.; Guo, X.; Chattopadhyay, K.; Yu, D. Pyrometallurgical options for recycling spent lithium-ion batteries: A comprehensive review. J. Power Sources 2021, 491, 229622. [Google Scholar] [CrossRef]
- Holzer, A.; Windisch-Kern, S.; Ponak, C.; Raupenstrauch, H. A Novel Pyrometallurgical Recycling Process for Lithium-Ion Batteries and Its Application to the Recycling of LCO and LFP. Metals 2021, 11, 149. [Google Scholar] [CrossRef]
- Velázquez-Martínez, O.; Valio, J.; Santasalo-Aarnio, A.; Reuter, M.; Serna-Guerrero, R. A Critical Review of Lithium-Ion Battery Recycling Processes from a Circular Economy Perspective. Batteries 2019, 5, 68. [Google Scholar] [CrossRef] [Green Version]
- Velázquez-Martinez, O.; Porvali, A.; Van Den Boogaart, K.G.; Santasalo-Aarnio, A.; Lundström, M.; Reuter, M.; Serna-Guerrero, R. On the use of statistical entropy analysis as assessment parameter for the comparison of lithium-ion battery recycling processes. Batteries 2019, 5, 41. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Gaustad, G.; Babbitt, C.W. Targeting high value metals in lithium-ion battery recycling via shredding and size-based separation. Waste Manag. 2016, 51, 204–213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ball, S.; Clark, J.; Cookson, J. Battery Materials Technology Trends and Market Drivers for Automotive Applications. Johns. Matthey Technol. Rev. 2020, 64, 287–297. [Google Scholar] [CrossRef]
- Ruismäki, R.; Rinne, T.; Dańczak, A.; Taskinen, P.; Serna Guerrero, R.; Jokilaakso, A. Integrating Flotation and Pyrometallurgy for Recovering Graphite and Valuable Metals from Battery Scrap. Metals 2020, 10, 680. [Google Scholar] [CrossRef]
- Ren, G.; Xiao, S.; Xie, M.; Pan, B.; Chen, J.; Wang, F.; Xia, X. Recovery of valuable metals from spent lithium ion batteries by smelting reduction process based on FeO–SiO2–Al2O3 slag system. Trans. Nonferrous Met. Soc. China 2017, 27, 450–456. [Google Scholar] [CrossRef]
- Wentker, M.; Greenwood, M.; Leker, J. A Bottom-Up Approach to Lithium-Ion Battery Cost Modeling with a Focus on Cathode Active Materials. Energies 2019, 12, 504. [Google Scholar] [CrossRef] [Green Version]
- European Commission. Critical Raw Materials Resilience: Charting a Path towards Greater Security and Sustainability. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:52020DC0474 (accessed on 22 February 2021).
- Mudd, G.M. Global trends and environmental issues in nickel mining: Sulfides versus laterites. Ore Geol. Rev. 2010, 38, 9–26. [Google Scholar] [CrossRef]
- Widijatmoko, S.D.; Fu, G.; Wang, Z.; Hall, P. Recovering lithium cobalt oxide, aluminium, and copper from spent lithium-ion battery via attrition scrubbing. J. Clean. Prod. 2020, 260, 120869. [Google Scholar] [CrossRef]
- Sommerville, R.; Shaw-Stewart, J.; Goodship, V.; Rowson, N.; Kendrick, E. A review of physical processes used in the safe recycling of lithium ion batteries. Sustain. Mater. Technol. 2020, 25, e00197. [Google Scholar] [CrossRef]
- Porvali, A.; Aaltonen, M.; Ojanen, S.; Velazquez-Martinez, O.; Eronen, E.; Liu, F.; Wilson, B.P.; Serna-Guerrero, R.; Lundström, M. Mechanical and hydrometallurgical processes in HCl media for the recycling of valuable metals from Li-ion battery waste. Resour. Conserv. Recycl. 2019, 142, 257–266. [Google Scholar] [CrossRef]
- Porvali, A.; Chernyaev, A.; Shukla, S.; Lundström, M. Lithium ion battery active material dissolution kinetics in Fe (II)/Fe (III) catalyzed Cu-H2SO4 leaching system. Sep. Purif. Technol. 2020, 236, 116305. [Google Scholar] [CrossRef]
- Peng, C.; Liu, F.; Aji, A.T.; Wilson, B.P.; Lundström, M. Extraction of Li and Co from industrially produced Li-ion battery waste—Using the reductive power of waste itself. Waste Manag. 2019, 95, 604–611. [Google Scholar] [CrossRef]
- Guoxing, R.; Songwen, X.; Meiqiu, X.; Bing, P.; Youqi, F.; Fenggang, W.; Xing, X. Recovery of Valuable Metals from Spent Lithium-Ion Batteries by Smelting Reduction Process Based on MnO-SiO2-Al2O3 Slag System. In Advances in Molten Slags, Fluxes, and Salts, Proceedings of the 10th International Conference on Molten Slags, Fluxes and Salts, Grand Hyatt Seattle, WA, USA, 22 May 2016; Reddy, R.G., Chaubal, P., Pistorius, P.C., Pal, U., Eds.; Springer: Cham, Switzerland, 2016. [Google Scholar] [CrossRef]
- Sommerfeld, M.; Vonderstein, C.; Dertmann, C.; Klimko, J.; Oráč, D.; Miškufová, A.; Havlík, T.; Friedrich, B. A Combined Pyro- and Hydrometallurgical Approach to Recycle Pyrolyzed Lithium-Ion Battery Black Mass Part 1: Production of Lithium Concentrates in an Electric Arc Furnace. Metals 2020, 10, 1069. [Google Scholar] [CrossRef]
- Kucharski, M.; Sak, T.; Madej, P.; Wedrychowicz, M.; Mroz, W. A Study on the Copper Recovery from the Slag of the Outokumpu Direct-to-Copper Process. Metall. Mater. Trans. B 2014, 45, 590–602. [Google Scholar] [CrossRef]
- Klimko, J.; Oráč, D.; Miškufová, A.; Vonderstein, C.; Dertmann, C.; Sommerfeld, M.; Friedrich, B.; Havlík, T. A Combined Pyro- and Hydrometallurgical Approach to Recycle Pyrolyzed Lithium-Ion Battery Black Mass Part 2: Lithium Recovery from Li Enriched Slag—Thermodynamic Study, Kinetic Study, and Dry Digestion. Metals 2020, 10, 1558. [Google Scholar] [CrossRef]
- International Energy Agency. Coal 2020. Available online: https://www.iea.org/reports/coal-2020 (accessed on 24 February 2021).
- Ruismäki, R.; Dańczak, A.; Klemettinen, L.; Taskinen, P.; Lindberg, D.; Jokilaakso, A. Integrated Battery Scrap Recycling and Nickel Slag Cleaning with Methane Reduction. Minerals 2020, 10, 435. [Google Scholar] [CrossRef]
- Avarmaa, K.; Järvenpää, M.; Klemettinen, L.; Marjakoski, M.; Taskinen, P.; Lindberg, D.; Jokilaakso, A. Battery Scrap and Biochar Utilization for Improved Metal Recoveries in Nickel Slag Cleaning Conditions. Batteries 2020, 6, 58. [Google Scholar] [CrossRef]
- Yu, D.; Chattopadhyay, K. Enhancement of the nickel converter slag-cleaning operation with the addition of spent potlining. Int. J. Miner. Metall. Mater. 2018, 25, 881–891. [Google Scholar] [CrossRef]
- Zhou, S.; Wei, Y.; Bo, L.; Wang, H. Cleaner recycling of iron from waste copper slag by using walnut shell char as green reductant. J. Clean. Prod. 2019, 217, 423–431. [Google Scholar] [CrossRef]
- Pei, M.; Petäjäniemi, M.; Regnell, A.; Wijk, O. Toward a Fossil Free Future with HYBRIT: Development of Iron and Steelmaking Technology in Sweden and Finland. Metals 2020, 10, 972. [Google Scholar] [CrossRef]
- Bellemans, I.; De Wilde, E.; Moelans, N.; Verbeken, K. Metal losses in pyrometallurgical operations—A review. Adv. Colloid Interface Sci. 2018, 255, 47–63. [Google Scholar] [CrossRef]
- Hittner, H.J.; Nguyen, Q.C. Stabilization of Fluorides of Spent Potlining by Chemical Dispersion. U.S. Patent No. 5,024,822, 18 June 1991. [Google Scholar]
- Cho, Y.S.; Laskowski, J.S. Bubble coalescence and its effect on dynamic foam stability. Can. J. Chem. Eng. 2002, 80, 299–305. [Google Scholar] [CrossRef]
- Klemettinen, L.; Aromaa, R.; Dańczak, A.; O’Brien, H.; Taskinen, P.; Jokilaakso, A. Distribution Kinetics of Rare Earth Elements in Copper Smelting. Sustainability 2020, 12, 208. [Google Scholar] [CrossRef] [Green Version]
- Wan, X.; Shen, L.; Jokilaakso, A.; Eriç, H.; Taskinen, P. Experimental Approach to Matte-Slag Reactions in the Flash Smelting Process. Miner. Process. Extr. Metall. Rev. 2021, 42, 231–241. [Google Scholar] [CrossRef]
- Dańczak, A.; Klemettinen, L.; O’Brien, H.; Taskinen, P.; Lindberg, D.; Jokilaakso, A. Slag chemistry and behavior of nickel and tin in black copper smelting with alumina and magnesia-containing slags. J. Sustain. Metall. 2021, 7, 1–14. [Google Scholar] [CrossRef]
- Bastin, G.F.; Heijligers, H.J.M. Quantitative Electron Probe Microanalysis of Ultralight Elements (Boron-Oxygen). Scanning 1990, 12, 225–236. [Google Scholar] [CrossRef] [Green Version]
- Van Achterberg, E.; Ryan, C.G.; Jackson, S.E.; Griffin, W.L. Data reduction software for LA-ICP-MS: Appendix. In Laser Ablation-ICP-Mass Spectrometry in the Earth Sciences: Principles and Applications; Short Course Series; Sylvester, P.J., Ed.; Mineralogical Association of Canada: Ottawa, ON, Canada, 2001; pp. 239–243. [Google Scholar]
- Jochum, K.P.; Weiss, U.; Stoll, B.; Kuzmin, D.; Yang, Q.; Raczek, I.; Jacob, D.E.; Stracke, A.; Birbaum, K.; Frick, D.A.; et al. Determination of Reference Values for NIST SRM 610–617 Glasses Following ISO Guidelines. Geostand. Geoanalytical Res. 2011, 35, 397–429. [Google Scholar] [CrossRef]
- Warner, A.; Díaz, C.; Dalvi, A.; Mackey, P. JOM World Nonferrous Smelter Survey Part IV: Nickel: Sulfide. JOM 2007, 59, 58–72. [Google Scholar] [CrossRef]
- Waanders, F.B.; Nell, J. Phase Chemical Composition of Slag from a Direct Nickel Flash Furnace and Associated Slag Cleaning Furnace. Hyperfine Interact. 2013, 218, 101–105. [Google Scholar] [CrossRef]
- Heo, J.H.; Chung, Y.; Park, J.H. Recovery of iron and removal of hazardous elements from waste copper slag via a novel aluminothermic smelting reduction (ASR) process. J. Clean. Prod. 2016, 137, 777–787. [Google Scholar] [CrossRef]
- Tirronen, T.; Sukhomlinov, D.; O’Brien, H.; Taskinen, P.; Lundström, M. Distribution of lithium-ion and nickel-metal hydride battery elements in copper converting. J. Clean. Prod. 2017, 168, 399–409. [Google Scholar] [CrossRef]
- Dańczak, A.; Klemettinen, L.; Kurhila, M.; Taskinen, P.; Lindberg, D.; Jokilaakso, A. Behavior of Battery Metals Lithium, Cobalt, Manganese and Lanthanum in Black Copper Smelting. Batteries 2020, 6, 16. [Google Scholar] [CrossRef] [Green Version]
- Riekkola-Vanhanen, M. Finnish Expert Report on Best Available Techniques in Nickel Production; Finnish Environment Institute: Helsinki, Finland, 1999; pp. 1–66. ISBN 952-11-0507-0. Available online: https://helda.helsinki.fi/bitstream/handle/10138/40684/FE_317.pdf?sequence=1&isAllowed=y (accessed on 8 April 2021).
- Xiao, S.; Ren, G.; Xie, M. Recovery of Valuable Metals from Spent Lithium-Ion Batteries by Smelting Reduction Process Based on MnO–SiO2–Al2O3 Slag System. J. Sustain. Metall. 2017, 3, 703–710. [Google Scholar] [CrossRef]
- Sukhomlinov, D.; Klemettinen, L.; Avarmaa, K.; O’Brien, H.; Taskinen, P.; Jokilaakso, A. Distribution of Ni, Co, precious and platinum group metals in copper making process. Metall. Mater. Trans. B 2019, 50, 1752–1765. [Google Scholar] [CrossRef] [Green Version]





| Part of the Setup | Details, Producer |
|---|---|
| Heating elements | Four silicon carbide elements, positioned around the work tube, Kanthal |
| Working tube | Impervious pure alumina, Ø 38 mm ID, Frialit AL23, Friatec AG, Mannheim, Germany |
| Inner tube | Installed inside the top part of the work tube through a top screw, impervious pure alumina, Ø 22 mm ID, Frialit AL23, Friatec AG, Mannheim, Germany |
| Thermocouple | S-type Pt/Pt–10%Rh, Johnson-Matthey Noble Metals, UK, accuracy of ±3 °C |
| Pt-resistance thermometer | Used for measuring room temperature, Pt100, SKS-Group, Vantaa, Finland, tolerance class B 1/10 |
| Multimeters | The thermocouple was connected to a Keithley 2010 digital multimeter (DMM) and the resistance thermometer was connected to a Keithley 2000 DMM |
| Temperature logging | LabView software (version 8.6.1) |
| Gas atmosphere | Inert, 400–500 mL/min, Ar 5.0 (99.999% purity, AGA Linde, Espoo, Finland) |
| Element | Standard Material and X-ray Line Analyzed |
|---|---|
| Al | aluminum, Kα |
| Ca | anhydrite, Kα |
| Co | cobalt, Kα |
| Cu | copper, Lα |
| K | sanidine, Kα |
| Mg | magnesium, Kα |
| Mn | manganese, Kα |
| Ni | nickel, Kα |
| O | quartz, Kα |
| S | marcasite, Kα |
| Si | quartz, Kα |
| Zn | zinc, Lα |
| (a) | Co | Ni | Mn | Cu | ||||
| Head Grade (%) | Head Grade (%) | Head Grade (%) | Head Grade (%) | |||||
| 1 | 17.8 | 2.06 | 1.92 | 23.74 | ||||
| 2 | 18.1 | 2.14 | 1.94 | 21.49 | ||||
| 3 | 18.0 | 2.14 | 1.86 | 21.97 | ||||
| 4 | 21.6 | 2.53 | 2.20 | 18.10 | ||||
| (b) | Co | Ni | Mn | Cu | ||||
| Recovery (%) | Grade (%) | Recovery (%) | Grade (%) | Recovery (%) | Grade (%) | Recovery (%) | Grade (%) | |
| 1 | 31.4 | 32.85 | 29.7 | 3.60 | 34.3 | 3.86 | 1.9 | 2.71 |
| 2 | 32.1 | 33.86 | 29.6 | 3.70 | 34.4 | 3.89 | 1.7 | 2.16 |
| 3 | 36.4 | 36.37 | 32.6 | 3.86 | 38.7 | 3.99 | 2.3 | 2.76 |
| 4 | 37.8 | 37.51 | 33.6 | 3.90 | 38.2 | 3.85 | 3.5 | 2.91 |
| Element | Al | Co | Cu | Fe | Li | Mn | Ni | P | F | Cg | C * |
|---|---|---|---|---|---|---|---|---|---|---|---|
| wt.% | 2.88 | 23.35 | 1.28 | 0.36 | 3.01 | 2.84 | 2.25 | 0.21 | 2.2 | 19.85 | 39.65 |
| Concentrations (wt.%) in Starting Mixture | |||
|---|---|---|---|
| Substance | Ni-Slag/5% SBs | Ni-Slag/10% SBs | Ni-Slag/20% SBs |
| Al * | 0.14 | 0.29 | 0.58 |
| Al2O3 | 2.96 | 2.81 | 2.50 |
| MgO | 6.78 | 6.43 | 5.71 |
| CaO | 1.57 | 1.49 | 1.32 |
| SiO2 | 32.17 | 30.47 | 27.09 |
| Co | 1.60 | 2.75 | 5.04 |
| Cu | 0.56 | 0.60 | 0.67 |
| Fe | 34.08 | 32.31 | 28.76 |
| Ni | 3.40 | 3.34 | 3.22 |
| Li | 0.15 | 0.30 | 0.60 |
| Zn | 0.06 | 0.05 | 0.05 |
| P | 0.01 | 0.02 | 0.04 |
| S | 0.14 | 0.14 | 0.12 |
| F | 0.11 | 0.22 | 0.44 |
| Cg | 0.99 | 1.98 | 3.97 |
| C ** | 1.98 | 3.97 | 7.93 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dańczak, A.; Ruismäki, R.; Rinne, T.; Klemettinen, L.; O’Brien, H.; Taskinen, P.; Jokilaakso, A.; Serna-Guerrero, R. Worth from Waste: Utilizing a Graphite-Rich Fraction from Spent Lithium-Ion Batteries as Alternative Reductant in Nickel Slag Cleaning. Minerals 2021, 11, 784. https://doi.org/10.3390/min11070784
Dańczak A, Ruismäki R, Rinne T, Klemettinen L, O’Brien H, Taskinen P, Jokilaakso A, Serna-Guerrero R. Worth from Waste: Utilizing a Graphite-Rich Fraction from Spent Lithium-Ion Batteries as Alternative Reductant in Nickel Slag Cleaning. Minerals. 2021; 11(7):784. https://doi.org/10.3390/min11070784
Chicago/Turabian StyleDańczak, Anna, Ronja Ruismäki, Tommi Rinne, Lassi Klemettinen, Hugh O’Brien, Pekka Taskinen, Ari Jokilaakso, and Rodrigo Serna-Guerrero. 2021. "Worth from Waste: Utilizing a Graphite-Rich Fraction from Spent Lithium-Ion Batteries as Alternative Reductant in Nickel Slag Cleaning" Minerals 11, no. 7: 784. https://doi.org/10.3390/min11070784
APA StyleDańczak, A., Ruismäki, R., Rinne, T., Klemettinen, L., O’Brien, H., Taskinen, P., Jokilaakso, A., & Serna-Guerrero, R. (2021). Worth from Waste: Utilizing a Graphite-Rich Fraction from Spent Lithium-Ion Batteries as Alternative Reductant in Nickel Slag Cleaning. Minerals, 11(7), 784. https://doi.org/10.3390/min11070784







