Arsenic Forms in Soils of Various Settings in the Historical Ore Mining and Processing Site of Radzimowice, Western Sudetes
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil Samples and the Scope of Examination
2.2. Basic Properties and Chemical Analyses
2.3. Mineralogical Analyses
2.4. Statistics
3. Result
3.1. Screening
3.2. Detailed Chemical Analysis of Representative Samples
3.2.1. Basic Properties
3.2.2. Total and Extractable As
3.2.3. Sequential Extraction of As
3.3. Mineralogical Analysis of Representative Samples
3.3.1. Microscopic Observations and X-ray Diffraction (XRD) Analyses
3.3.2. Scanning Electron Microscopy–Energy-Dispersive Spectroscopy (SEM-EDS) Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wojciechowski, A.; Wierchowiec, J. Auriferous wastes from the abandoned arsenic and gold mine in Złoty Stok (Sudetes Mts., SW Poland). Geol. Quart. 2009, 53, 233–240. [Google Scholar]
- Mikulski, S.Z.; Speczik, S. The auriferous ore mineralisation and its zonal distribution around the Variscan Kłodzko–Złoty Stok granitoid pluton in the Sudetes (SW Poland)—An overview. Geol. Quart. 2016, 60, 650–674. [Google Scholar] [CrossRef][Green Version]
- Kaźmierczak, U.; Strzałkowski, P.; Lorenc, M.W.; Szumska, E.; Sánchez, A.A.P.; Baker, K.A. Post-mining Remnants and Revitalization. Geoheritage 2019. [Google Scholar] [CrossRef]
- Karczewska, A.; Krysiak, A.; Mokrzycka, D.; Jezierski, P.; Szopka, K. Arsenic distribution in soils of former As mining area and processing. Pol. J. Environ. Stud. 2013, 22, 175–181. [Google Scholar]
- Madziarz, M. Recognition formerly exploited polimetallic ore deposits as results of prospecting and mining works after II world war in Sudety Mts. Min. Sci. 2009, 128, 141. [Google Scholar]
- Mikulski, S.Z. Geological, mineralogical and geochemical characteristics of the Radzimowice Au-As-Cu deposit from the Kaczawa Mountains (Western Sudetes, Poland): An example of the transition of porphyry and epithermal style. Miner. Dep. 2005, 39, 904–920. [Google Scholar] [CrossRef]
- Karczewska, A.; Bogda, A.; Krysiak, A. Arsenic in soils in the areas of former arsenic mining and processing in Lower Silesia, SW Poland. In Arsenic in Soil and Groundwater Environments; Bhattacharya, P., Mukherjee, A.B., Loeppert, R.H., Eds.; Biogeochemical Interactions; Elsevier: Amsterdam, The Netherlands, 2007; Volume 9, pp. 411–440. [Google Scholar]
- Kałaska, M.; Siuda, R.; Sierpień, P. Application of Light Detection and Ranging (LiDAR) and geochemical survey to investigations of old mining center in Radzimowice (Lower Silesia, SW Poland). IX Symposium ProGEO: 25-28 June, 2018, Chęciny, Poland. In Programme and Abstract Book; Głowniak, E., Wasiłowska, A., Leonowicz, P., Eds.; Faculty of Geology, University of Warsaw: Warsaw, Poland, 2018; pp. 74–75. [Google Scholar]
- Marszalek, H.; Rysiukiewicz, M.; Wasik, M.; Costa, M.R. Hydrogeochemistry of groundwater from abandoned Radzimowice mine (Sudetes, SW Poland). In Proceedings of the SGEM2012 12th International Multidisciplinary Scientific GeoConference and EXPO, Albena, Bulgaria, 17–23 June 2012; Volume 2, pp. 135–142. [Google Scholar]
- Parafiniuk, J.; Siuda, R.; Borkowski, A. Sulphate and arsenate minerals as environmental indicators in the weathering zones of selected ore deposits, Western Sudetes, Poland. Acta Geol. Pol. 2016, 66, 493–508. [Google Scholar] [CrossRef]
- Wenzel, W.W. Arsenic. In Heavy Metals in Soils. Trace Metals and Metalloids in Soils and Their Bioavailability, 3rd ed.; Alloway, B.J., Ed.; Springer: Berlin, Germany, 2013; pp. 241–282. [Google Scholar]
- Kabata-Pendias, A.; Mukherjee, A.B. Trace Elements from Soil to Human; Springer Science & Business Media: New York, NY, USA, 2007. [Google Scholar]
- Bowell, R.J.; Alpers, C.N.; Jamieson, H.E.; Nordstrom, D.K.; Majzlan, J. The environmental geochemistry of arsenic—An overview—. Rev. Mineral. Geochem. 2014, 79, 1–16. [Google Scholar] [CrossRef]
- Naidu, R.; Smith, E.; Owens, G.; Bhattacharya, P. Managing Arsenic in the Environment: From Soil to Human Health; CSIRO Publishing: Collingwood, Australia, 2006. [Google Scholar]
- Yamamura, S.; Kurasawa, H.; Kashiwabara, Y.; Hori, T.; Aoyagi, T.; Nakajima, N.; Amachi, S. Soil microbial communities involved in reductive dissolution of arsenic from arsenate-laden minerals with different carbon sources. Environ. Sci. Technol. 2019, 53, 12398–12406. [Google Scholar] [CrossRef]
- Chen, X.; Zeng, X.C.; Kawa, Y.K.; Wu, W.; Zhu, X.; Ullah, Z.; Wang, Y. Microbial reactions and environmental factors affecting the dissolution and release of arsenic in the severely contaminated soils under anaerobic or aerobic conditions. Ecotoxicol. Environ. Saf. 2020, 189. [Google Scholar] [CrossRef] [PubMed]
- Lewińska, K.; Karczewska, A.; Siepak, M.; Szopka, K.; Gałka, B.; Iqbal, M. Effects of waterlogging on the solubility of Sb and As in variously treated shooting range soils. Appl. Geochem. 2019, 105, 7–16. [Google Scholar] [CrossRef]
- Szopka, K.; Gruss, I.; Gruszka, D.; Karczewska, A.; Gediga, K.; Gałka, B.; Dradrach, A. The Effects of Forest Litter and Waterlogging on the Ecotoxicity of Soils Strongly Enriched in Arsenic in a Historical Mining Site. Forests 2021, 12. [Google Scholar] [CrossRef]
- Lewińska, K.; Karczewska, A.; Siepak, M.; Gałka, B. Potential of Fe-Mn wastes produced by a water treatment plant for arsenic immobilization in contaminated soils. J. Geochem. Explor. 2016, 184, 226–231. [Google Scholar] [CrossRef]
- Karczewska, A.; Lewińska, K.; Siepak, M.; Gałka, B.; Dradrach, A.; Szopka, K. Transformation of beech forest litter as a factor that triggers arsenic solubility in soils developed on historical mine dumps. J. Soils Sediments 2018, 18, 2749–2758. [Google Scholar] [CrossRef]
- Drahota, P.; Knappová, M.; Kindlová, H.; Culka, A.; Majzlan, J.; Mihaljevič, M.; Jehlička, J. Mobility and attenuation of arsenic in sulfide-rich mining wastes from the Czech Republic. Sci. Total Environ. 2016, 557, 192–203. [Google Scholar] [CrossRef]
- Igarashi, T.; Herrera, P.S.; Uchiyama, H.; Miyamae, H.; Iyatomi, N.; Hashimoto, K.; Tabelin, C.B. The two-step neutralization ferrite-formation process for sustainable acid mine drainage treatment: Removal of copper, zinc and arsenic, and the influence of coexisting ions on ferritization. Sci. Total Environ. 2020, 715. [Google Scholar] [CrossRef]
- Cheng, H.; Hu, Y.; Luo, J.; Xu, B.; Zhao, J. Geochemical processes controlling fate and transport of arsenic in acid mine drainage (AMD) and natural systems. J. Hazard Mater. 2009, 165, 13–26. [Google Scholar] [CrossRef]
- Ramos, O.E.R.; Rötting, T.S.; French, M.; Sracek, O.; Bundschuh, J.; Quintanilla, J.; Bhattacharya, P. Geochemical processes controlling mobilization of arsenic and trace elements in shallow aquifers and surface waters in the Antequera and Poopó mining regions, Bolivian Altiplano. J. Hydrol. 2014, 518, 421–433. [Google Scholar] [CrossRef]
- Rieuwerts, J.; Mighanetara, K.; Braungardt, C.; Rollinson, G.; Pirrie, D.; Azizi, F. Geochemistry and mineralogy of arsenic in mine wastes and stream sediments in a historic metal mining area in the UK. Sci. Total Environ. 2014, 472, 226–234. [Google Scholar] [CrossRef]
- Beane, S.J.; Comber, S.D.; Rieuwerts, J.; Long, P. Abandoned metal mines and their impact on receiving waters: A case study from Southwest England. Chemosphere 2016, 153, 294–306. [Google Scholar] [CrossRef]
- González-Fernández, B.; Rodríguez-Valdés, E.; Boente, C.; Menéndez-Casares, E.; Fernández-Braña, A.; Gallego, J.R. Long-term ongoing impact of arsenic contamination on the environmental compartments of a former mining-metallurgy area. Sci. Total Environ. 2018, 610, 820–830. [Google Scholar] [CrossRef]
- Tan, K.H. Soil Sampling, Preparation and Analysis, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- ISO 11466: 1995. Soil Quality—Extraction of Trace Elements Soluble in Aqua Regia; International Organization for Standardization: Geneva, Switzerland, 1995. [Google Scholar]
- ISO 17586: 2016. Soil Quality—Extraction of Trace Elements Using Dilute Nitric Acid; International Organization for Standardization: Geneva, Switzerland, 2016. [Google Scholar]
- ISO 19730: 2008. Soil Quality—Extraction of Trace Elements from Soil Using Ammonium Nitrate Solution; International Organization for Standardization: Geneva, Switzerland, 2008. [Google Scholar]
- Wenzel, W.W.; Kirchbaumer, N.; Prochaska, T.; Stingeder, G.; Lombi, E.; Adriano, D.C. Arsenic fractionation in soils using improved sequential extraction procedure. Anal. Chim. Acta 2001, 436, 309–323. [Google Scholar] [CrossRef]
- Meunier, L.; Walker, S.R.; Wragg, J.; Parsons, M.B.; Koch, I.; Jamieson, H.E.; Reimer, K.J. Effects of soil composition and mineralogy on the bioaccessibility of arsenic from tailings and soil in gold mine districts of Nova Scotia. Environ. Sci. Technol. 2010, 44, 2667–2674. [Google Scholar] [CrossRef] [PubMed]
- Drahota, P.; Filippi, M. Secondary arsenic minerals in the environment: A review. Environ. Int. 2009, 35, 1243–1255. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Schreiber, M.E. Arsenic release from arsenopyrite weathering: Insights from sequential extraction and microscopic studies. J. Hazard. Mat. 2013, 262, 896–904. [Google Scholar] [CrossRef]
- Drahota, P.; Kulakowski, O.; Culka, A.; Knappová, M.; Rohovec, J.; Veselovský, F.; Racek, M. Arsenic mineralogy of near-neutral soils and mining waste at the Smolotely-Líšnice historical gold district, Czech Republic. App. Geochem. 2018, 89, 243–254. [Google Scholar] [CrossRef]
- Murciego, A.; Álvarez-Ayuso, E.; Sanz-Arranz, A.; Medina-García, J.; Rull-Pérez, F.; Villar-Alonso, P. Characterization of secondary products in arsenopyrite-bearing mine wastes: Influence of cementation on arsenic attenuation. J. Hazard. Mat. 2009, 373, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Filippi, M.; Drahota, P.; Machovič, V.; Böhmová, V.; Mihaljevič, M. Arsenic mineralogy and mobility in the arsenic-rich historical mine waste dump. Sci. Total Environ. 2015, 536, 713–728. [Google Scholar] [CrossRef] [PubMed]
- Anawar, H.M. Sustainable rehabilitation of mining waste and acid mine drainage using geochemistry, mine type, mineralogy, texture, ore extraction and climate knowledge. J. Environ. Manag. 2015, 158, 111–121. [Google Scholar] [CrossRef]
- Drahota, P.; Filippi, M.; Ettler, V.; Rohovec, J.; Mihaljevič, M.; Šebek, O. Natural attenuation of arsenic in soils near a highly contaminated historical mine waste dump. Sci. Total Environ. 2012, 414, 546–555. [Google Scholar] [CrossRef]
- Kralova, L.; Száková, J.; Kubík, Š.; Tlustoš, P.; Balík, J. The variability of arsenic and other risk element uptake by individual plant species growing on contaminated soil. Soil Sediment Contam. 2010, 19, 617–634. [Google Scholar] [CrossRef]
- Stachnik, Ł.; Korabiewski, B.; Raczyk, J.; Łopuch, M.; Wieczorek, I. Arsenic pollution in Quaternary sediments and water near a former gold mine. Sci. Rep. 2020, 10, 18458. [Google Scholar] [CrossRef] [PubMed]
- Krysiak, A.; Karczewska, A. Arsenic extractability in soils in the areas of former arsenic mining and smelting, SW Poland. Sci. Total Environ. 2007, 379, 190–200. [Google Scholar] [CrossRef]
- Dradrach, A.; Karczewska, A.; Szopka, K.; Lewińska, K. Accumulation of arsenic by plants growing in the sites strongly contaminated by historical mining in the Sudetes region of Poland. IJERPH 2020, 17, 3342. [Google Scholar] [CrossRef]
- Regulation of the Minister for the Environment of 1 September 2016. On the Method How to Carry Out the Assessment of Soil Contamination. (Journal of Laws of 2016, Item 1395). In Polish. Available online: http://isap.sejm.gov.pl/isap.nsf/download.xsp/WDU20160001395/O/D20161395.pdf (accessed on 20 March 2021).
- Karczewska, A.; Kabała, C. Environmental risk assessment as a new basis for evaluation of soil contamination in Polish law. Soil Sci. Ann. 2017, 68, 67–80. [Google Scholar] [CrossRef]
- Knappová, M.; Drahota, P.; Falteisek, L.; Culka, A.; Penížek, V.; Trubač, J.; Matoušek, T. Microbial sulfidogenesis of arsenic in naturally contaminated wetland soil. Geoch. Cosmochim. Acta 2019, 267, 33–50. [Google Scholar] [CrossRef]
- Craw, D.; Falconer, D.; Youngson, J.H. Environmental arsenopyrite stability and dissolution: Theory, experiment, and field observations. Chem. Geol. 2013, 199, 71–82. [Google Scholar] [CrossRef]
- Murciego, A.; Álvarez-Ayuso, E.; Pellitero, E.; Rodríguez, M.; García-Sánchez, A.; Tamayo, A.; Rubin, J. Study of arsenopyrite weathering products in mine wastes from abandoned tungsten and tin exploitations. J. Hazard. Mat. 2011, 186, 590–601. [Google Scholar] [CrossRef] [PubMed]
- Corkhill, C.L.; Vaughan, D.J. Arsenopyrite oxidation—A review. Appl. Geochem. 2009, 24, 2342–2361. [Google Scholar] [CrossRef]
- Bari, A.F.; Lamb, D.; Choppala, G.; Bolan, N.; Seshadri, B.; Rahman, M.A.; Rahman, M.M. Geochemical fractionation and mineralogy of metal (loid) s in abandoned mine soils: Insights into arsenic behaviour and implications to remediation. J. Hazard. Mat. 2020, 399. [Google Scholar] [CrossRef]
- Langner, P.; Mikutta, C.; Kretzschmar, R. Arsenic sequestration by organic sulphur in peat. Nat. Geosci. 2012, 5, 66–73. [Google Scholar] [CrossRef]
- Ding, Z.; Zheng, B.; Long, J.; Belkin, H.E.; Finkelman, R.B.; Chen, C.; Zhou, Y. Geological and geochemical characteristics of high arsenic coals from endemic arsenosis areas in southwestern Guizhou Province, China. Appl. Geochem. 2001, 16, 1353–1360. [Google Scholar] [CrossRef]
- Drahota, P.; Mikutta, C.; Falteisek, L.; Duchoslav, V.; Klementová, M. Biologically induced formation of realgar deposits in soil. Geochim. Cosmochim. Acta 2017, 218, 237–256. [Google Scholar] [CrossRef]
- Falteisek, L.; Drahota, P.; Culka, A.; Laufek, F.; Trubač, J. Bioprecipitation of As4S4 polymorphs in an abandoned mine adit. Appl. Geochem. 2020, 113. [Google Scholar] [CrossRef]
- Paktunc, D.; Bruggeman, K. Solubility of nanocrystalline scorodite and amorphous ferric arsenate: Implications for stabilization of arsenic in mine wastes. Appl. Geochem. 2010, 25, 674–683. [Google Scholar] [CrossRef]
- Ciardelli, M.C.; Xu, H.; Sahai, N. Role of Fe (II), phosphate, silicate, sulfate, and carbonate in arsenic uptake by coprecipitation in synthetic and natural groundwater. Water Res. 2008, 42, 615–624. [Google Scholar] [CrossRef] [PubMed]

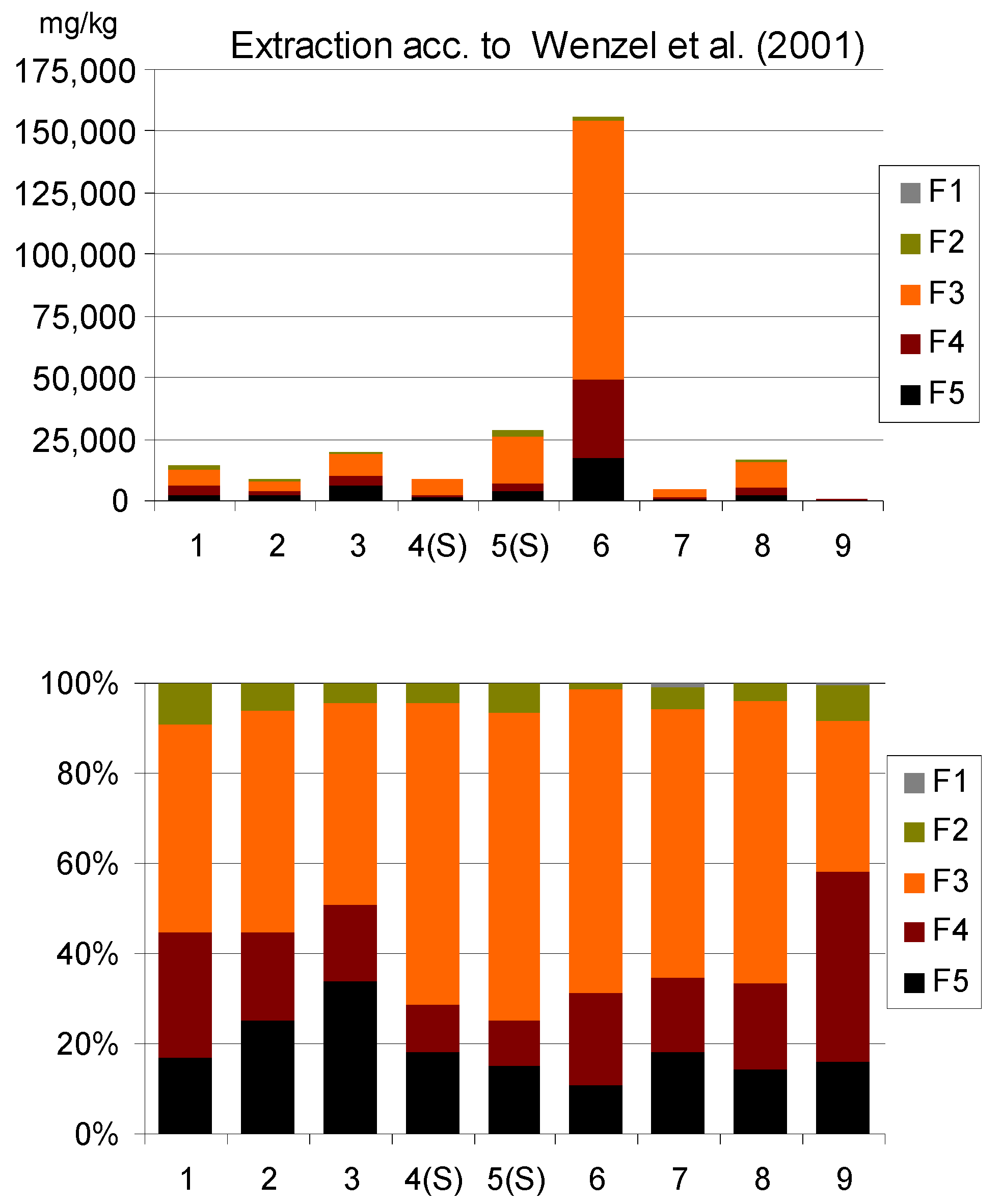
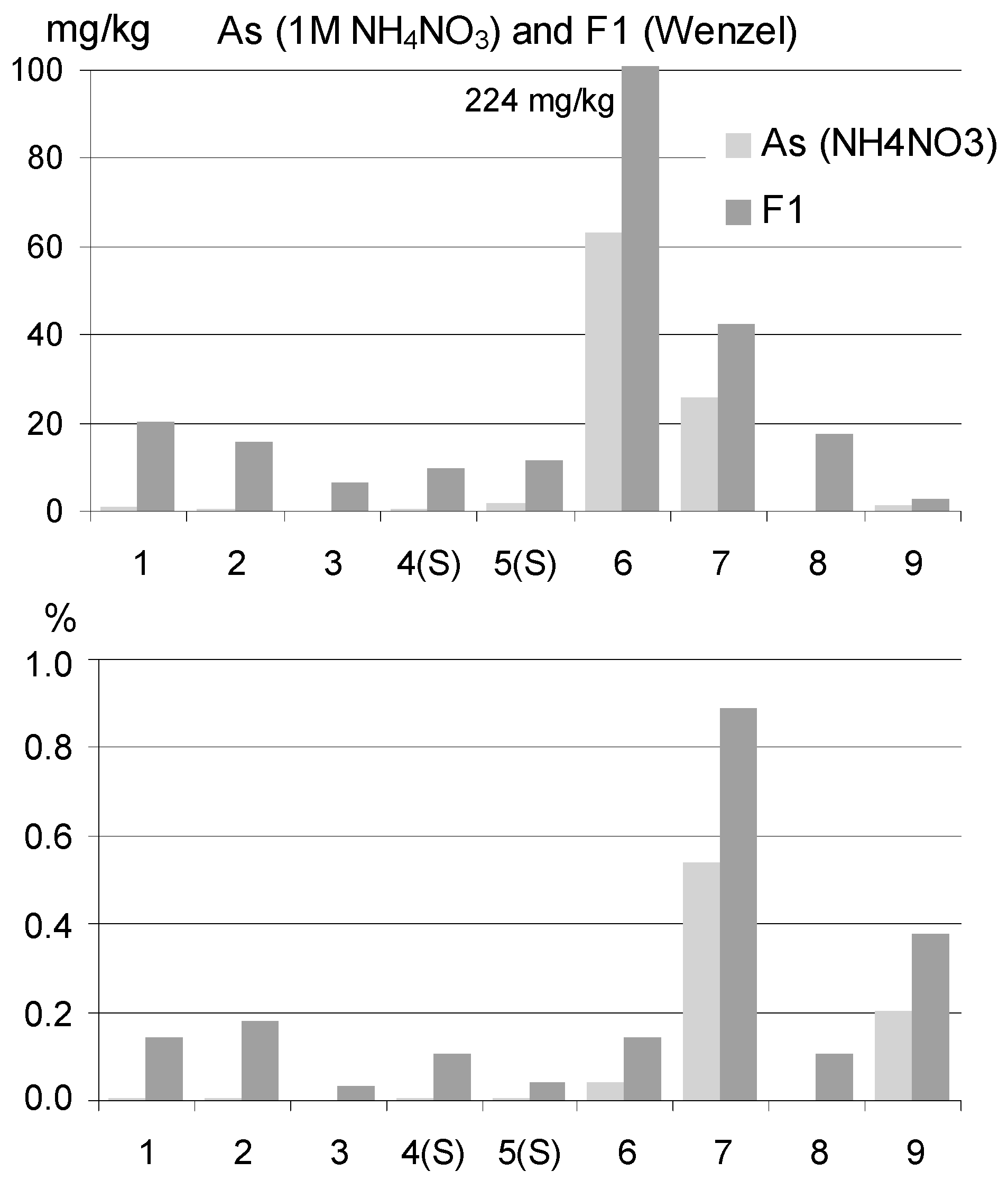


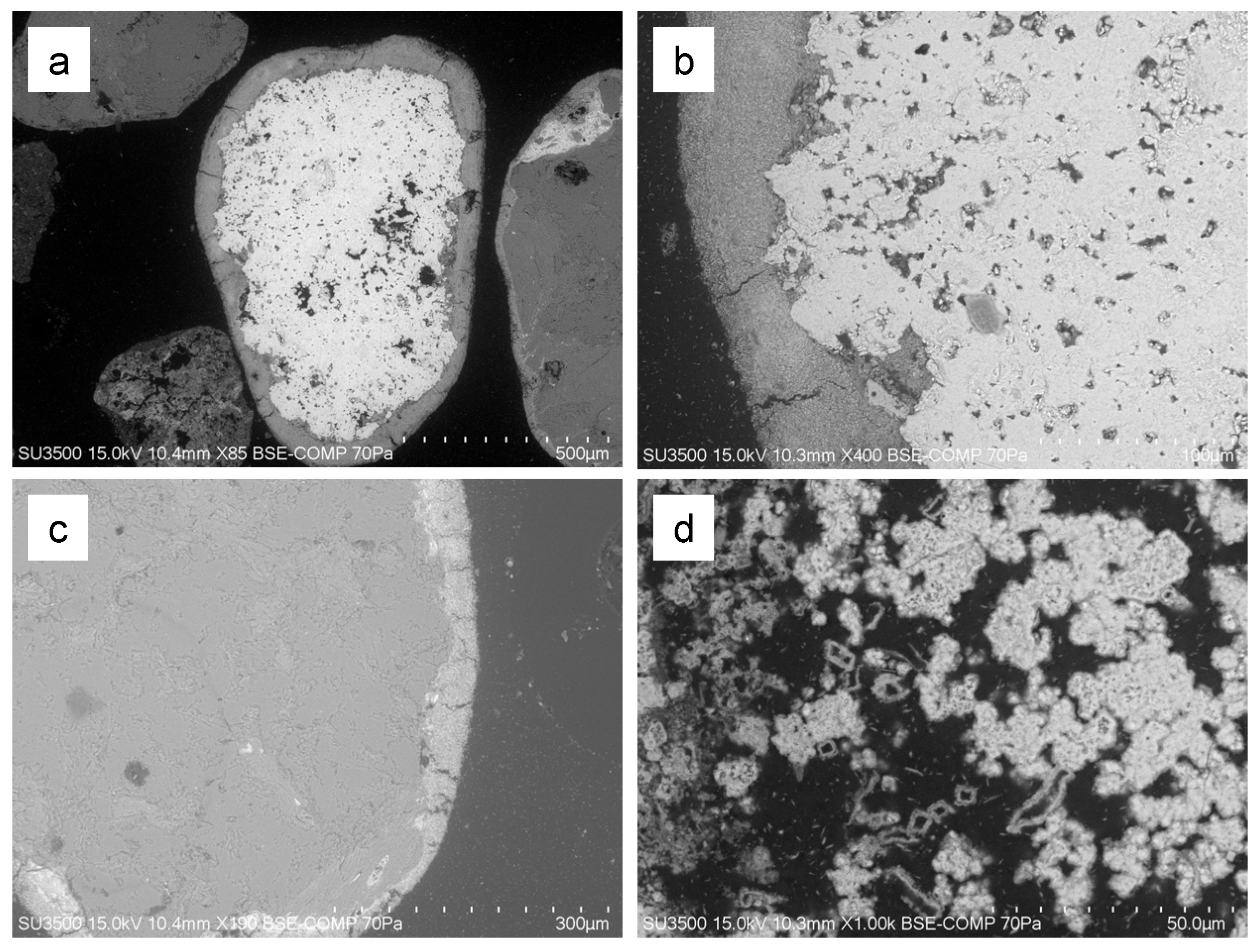
| Group | Description | Source of As in Soils | Numberof Samples | Total As in Soils, mg/kg | |
|---|---|---|---|---|---|
| Range | Median | ||||
| I | Dry meadow | Weathering of primary minerals, air-borne particles | 10 | 161–14,200 | 265 |
| II | Soils on mine dumps | Mainly: weathering of primary minerals, | 12 | 110–19,900 | 8160 |
| III | Alluvial soils along the Olszanka stream | Mainly: transported primary minerals, secondary minerals, possibly: leaching from processing (calciner) wastes | 14 | 89–156,000 | 5170 |
| IV | Mixed-stand forests on the slopes | Air-borne particles, (and weathering of primary minerals) | 9 | 167–9990 | 358 |
| V | Bottom sediments of the Olszanka stream | AMD, ARD, leaching from processing wastes, transported primary minerals and secondary minerals | 7 | 1270–28,400 | 9630 |
| Group | 0.43 M HNO3—Extractable As | 1 M NH4NO3—Extractable As | ||||||
|---|---|---|---|---|---|---|---|---|
| mg/kg | Percent of Total As | mg/kg | Percent of Total As | |||||
| Range | Median | Range | Median | Range | Median | Range | Median | |
| I | 14–3320 | 32 | 4.1–23.4 | 13.0 | 0.05–0.78 | 0.07 | 0.01–0.25 | 0.03 |
| II | 5.2–2890 | 1360 | 4.8–44.7 | 20.3 | 0.06–1.56 | 0.23 | <0.01–0.05 | 0.01 |
| III | 6.2–18,900 | 504 | 3.0–39.6 | 13.9 | 0.02–63.2 | 0.65 | <0.01–0.75 | 0.02 |
| IV | 9.5–179 | 20 | 1.7–22.6 | 6.5 | 0.10–4.63 | 0.30 | <0.01–1.28 | 0.02 |
| V | 206–2740 | 1330 | 4.7–23.4 | 16.2 | 0.20–1.78 | 0.34 | <0.01–0.02 | <0.01 |
| Group | Soil No. | Texture | C org g/kg | pH | As, mg/kg | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| >2mm% | Fine Soil (<2 mm) | Total | 0.43 M HNO3—Extractable | 1 M NH4NO3—Extractable | |||||||
| Percent of Fractions, mm | Textural Group | ||||||||||
| 0.05–2.0 | 0.002–0.05 | <0.002 | |||||||||
| I | 1 | 45 | 63 | 35 | 2 | SL | 33.4 | 3.6 | 14,200 | 3320 | 0.78 |
| II | 2 | 55 | 57 | 36 | 7 | SL | 12.2 | 6.9 | 8670 | 953 | 0.48 |
| 3 | 47 | 53 | 39 | 8 | SL | 8.8 | 3.0 | 19,900 | 2890 | 0.23 | |
| III | 6 | 19 | 82 | 16 | 3 | LS | 31.6 | 4.6 | 156,200 | 18,900 | 63.2 |
| 7 | 28 | 46 | 50 | 4 | SL | 88.0 | 6.0 | 4770 | 2160 | 25.6 | |
| 8 | 37 | 51 | 47 | 2 | SL | 254.0 | 4.3 | 16,400 | 649 | 0.04 | |
| IV | 9 | 52 | 56 | 37 | 7 | SL | 42.0 | 3.1 | 737 | 145 | 1.48 |
| V | 4(S) | 5 | 56 | 35 | 9 | SL | 21.5 | 6.9 | 8950 | 814 | 0.34 |
| 5(S) | 3 | 61 | 26 | 13 | SL | 20.8 | 6.7 | 28,400 | 1330 | 1.78 | |
| Sample | Point | Element | Fe:S:As | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| C | O | Al | Si | S | K | Ca | Fe | As | |||
| 2/1 | 1 | 21.0 | 6.7 | 0.7 | 1.6 | 12.9 | 26.7 | 30.5 | 0.88:0.42:1 | ||
| 4 | 22.1 | 7.5 | 0.8 | 1.7 | 12.2 | 25.4 | 30.2 | 0.84:0.40:1 | |||
| 2/2 | 5 | 20.5 | 6.6 | 0.6 | 1.3 | 13.4 | 25.6 | 31.9 | 0.80:0.42:1 | ||
| 6 | 21.3 | 8.5 | 0.6 | 1.3 | 12.0 | 24.6 | 30.5 | 0.81:0.39:1 | |||
| 2/6 | 2 | 25.8 | 11.9 | 0.9 | 3.3 | 11.0 | 1.8 | 21.7 | 23.6 | 0.92:0.47:1 | |
| 2/12 | 2 | 26.7 | 10.4 | 1.7 | 3.2 | 11.9 | 0.8 | 20.8 | 24.4 | 0.85:0.49:1 | |
| 2/17 | 1 | 25.7 | 9.5 | 1.2 | 2.9 | 11.9 | 23.8 | 25.1 | 0.95:0.47:1 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lewińska, K.; Duczmal-Czernikiewicz, A.; Karczewska, A.; Dradrach, A.; Iqbal, M. Arsenic Forms in Soils of Various Settings in the Historical Ore Mining and Processing Site of Radzimowice, Western Sudetes. Minerals 2021, 11, 491. https://doi.org/10.3390/min11050491
Lewińska K, Duczmal-Czernikiewicz A, Karczewska A, Dradrach A, Iqbal M. Arsenic Forms in Soils of Various Settings in the Historical Ore Mining and Processing Site of Radzimowice, Western Sudetes. Minerals. 2021; 11(5):491. https://doi.org/10.3390/min11050491
Chicago/Turabian StyleLewińska, Karolina, Agata Duczmal-Czernikiewicz, Anna Karczewska, Agnieszka Dradrach, and Muhammad Iqbal. 2021. "Arsenic Forms in Soils of Various Settings in the Historical Ore Mining and Processing Site of Radzimowice, Western Sudetes" Minerals 11, no. 5: 491. https://doi.org/10.3390/min11050491
APA StyleLewińska, K., Duczmal-Czernikiewicz, A., Karczewska, A., Dradrach, A., & Iqbal, M. (2021). Arsenic Forms in Soils of Various Settings in the Historical Ore Mining and Processing Site of Radzimowice, Western Sudetes. Minerals, 11(5), 491. https://doi.org/10.3390/min11050491








