Gold in the Farallones Block of the Shale-Hosted, Clastic-Dominated Castellanos Zinc-Lead Deposit (Northwest Cuba)
Abstract
1. Introduction
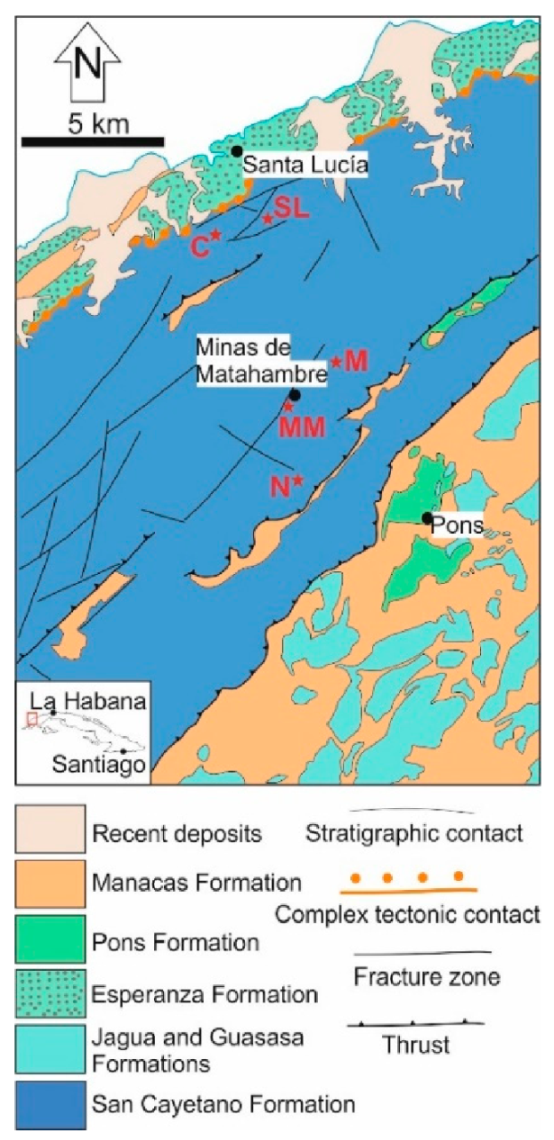
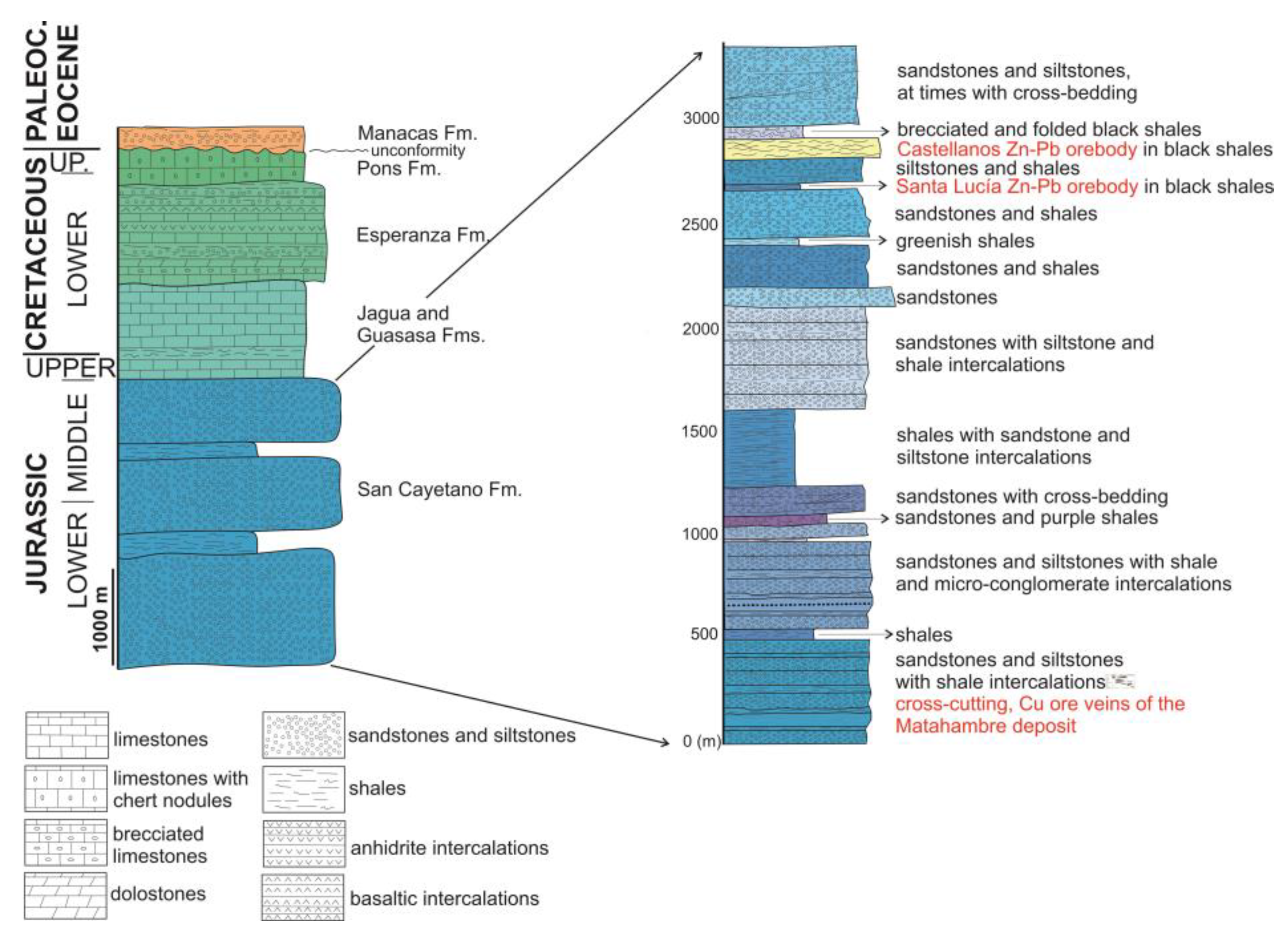
2. Geological Setting
3. Methods
4. The Castellanos Zn-Pb Orebody
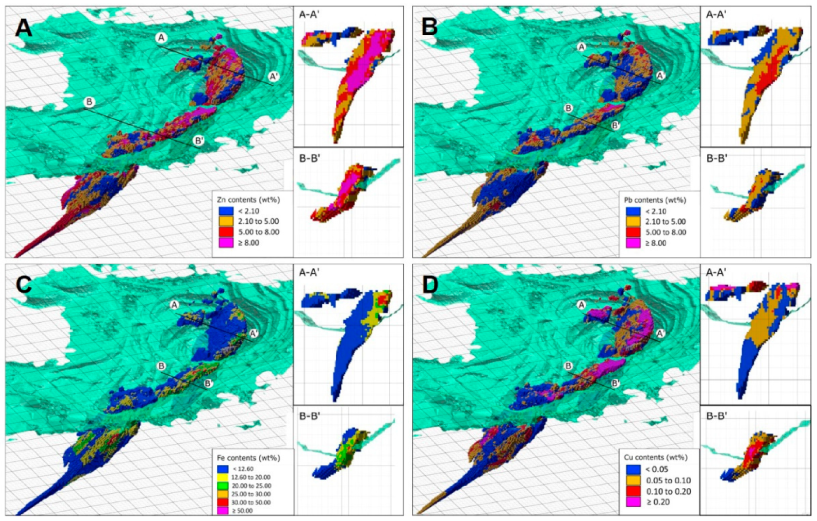
4.1. Morphology and Internal Structure
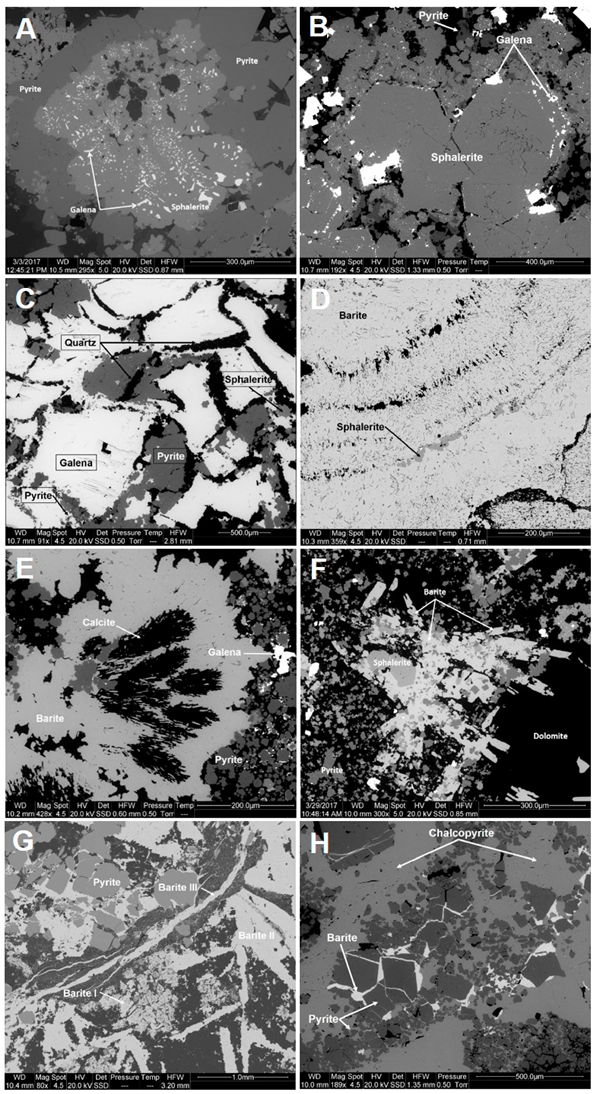
4.2. Mineral Assemblages and Textures
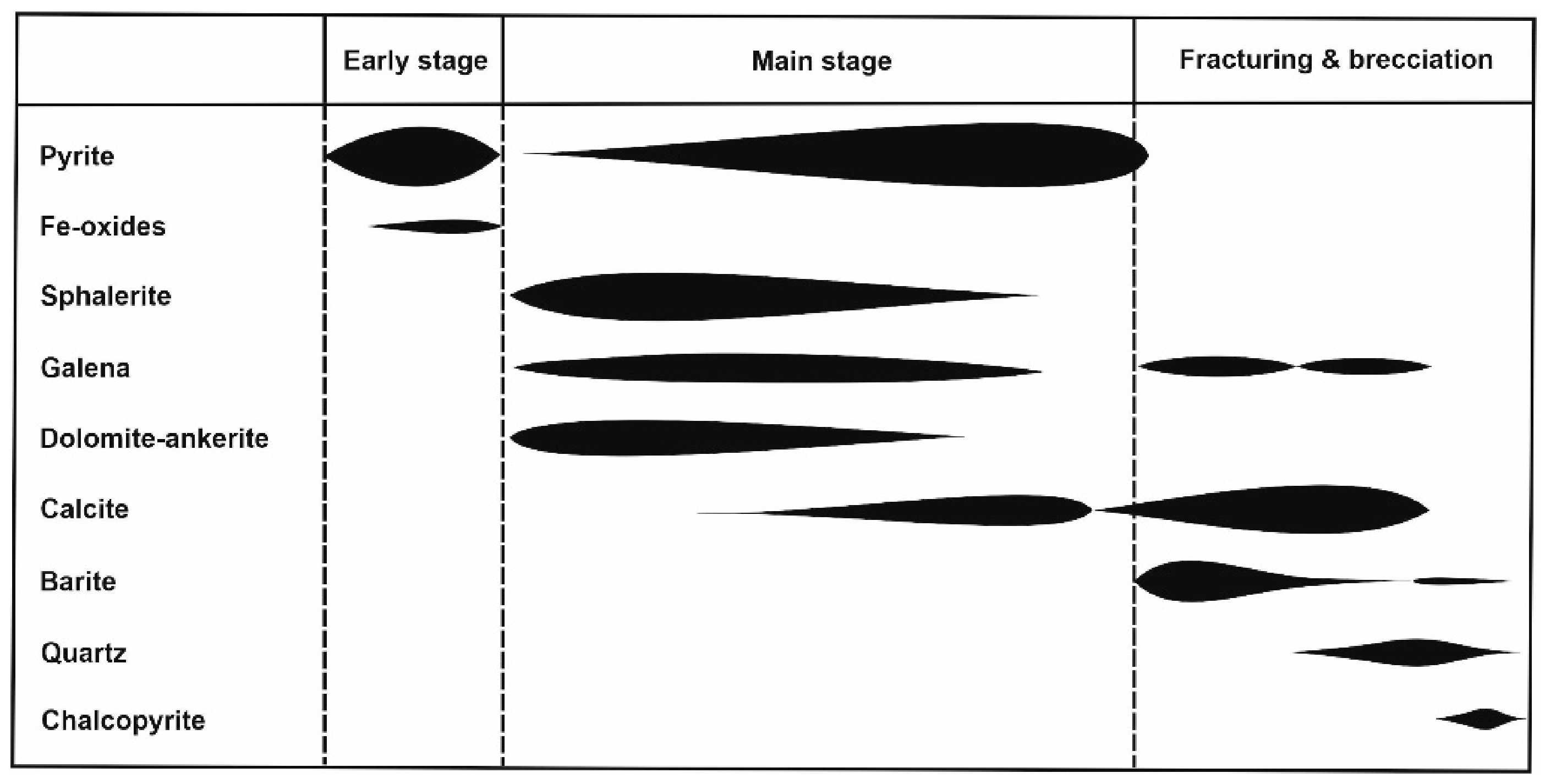
4.3. Sequence of Ore Deposition
- (i.)
- Early stage: formation of the fine-grained, framboidal and euhedral pyrite ore disseminated in the host black shale, coeval to or preceding the development of Fe-oxides (presently magnetite)-rich domains. Textural relationships between these Fe sulfides and oxides are not well known yet due to the scattered distribution of the later and the absence of samples where both fine-grained pyrite and Fe oxides coexist.
- (ii.)
- Main stage: formation of the main ore mineral assemblages as a consequence of the overlap of several events of sulfide crystallization. Early events started with the formation of colloform sphalerite including galena (often skeletal) (Figure 5A) and surrounded by collophorm pyrite (with a tendency to develop subhedral crystals at the outer rims), with intergranular fine-grained dolomite-ankerite. During late events, sulfides and carbonates tended to form euhedral, coarse-grained and zoned crystals (Figure 5B), and carbonates became progressively richer in calcite (Figure 4F). Each individual crystallization event involved partial dissolution of the previous assemblage frequently obliterating early textural relations (Figure 4G,H). The existence of a basal pyrite-rich zone where carbonates, phyllosilicates, minor sphalerite and rarely galena mainly occur as partly dissolved remains in pyrite, points to replacement processes of early Zn–Pb sulfide ores associated with the precipitation and growth of pyrite. These textural relationships are similar to those observed in pyrite filling the cross-cutting veins of the underlying stockwork. Pyrite from the basal pyrite-rich zone also shows pseudomorphic textures after replacement of laminar magnetite (Figure 4D) and becomes recrystallized giving rise to massive aggregates.
- (iii.)
- Late stage: it is characterized by multiple events (at least three) of fracturing and brecciation. The onset of this stage is marked by the formation of late banded colloform pyrite that cements previous sulfide aggregates shortly followed by the crystallization of early barite, frequently intergrown with calcite (Figure 5E). Later on, remobilization of galena, precipitation of at least three younger generations of barite filling fractures, and a silicification event took place. Finally, crystallization of chalcopyrite occurred along brecciated zones, both in the stratiform orebody (Figure 5H) and within pyrite veins of the stockwork (Figure 4A).
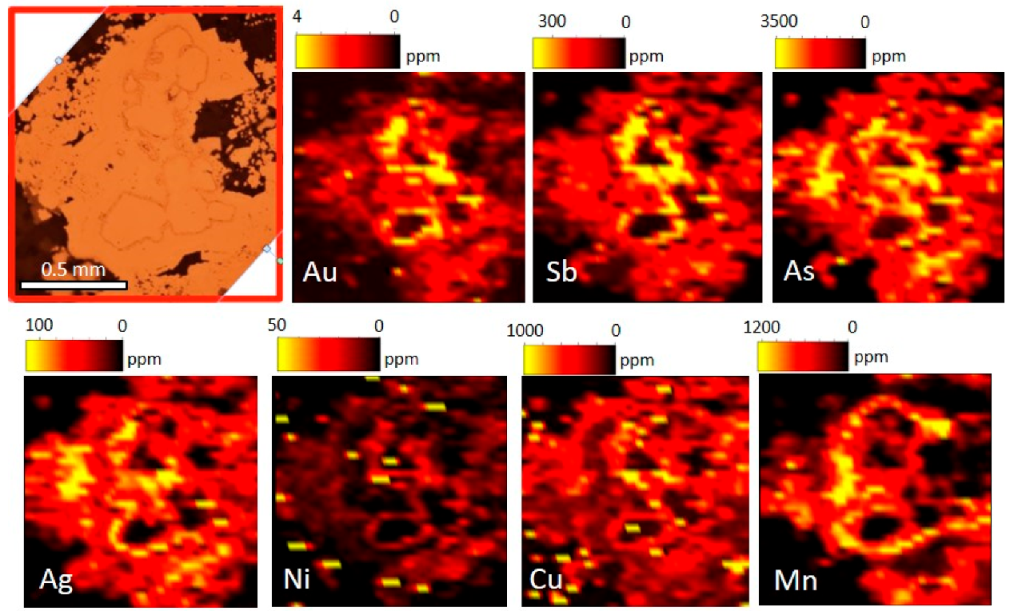
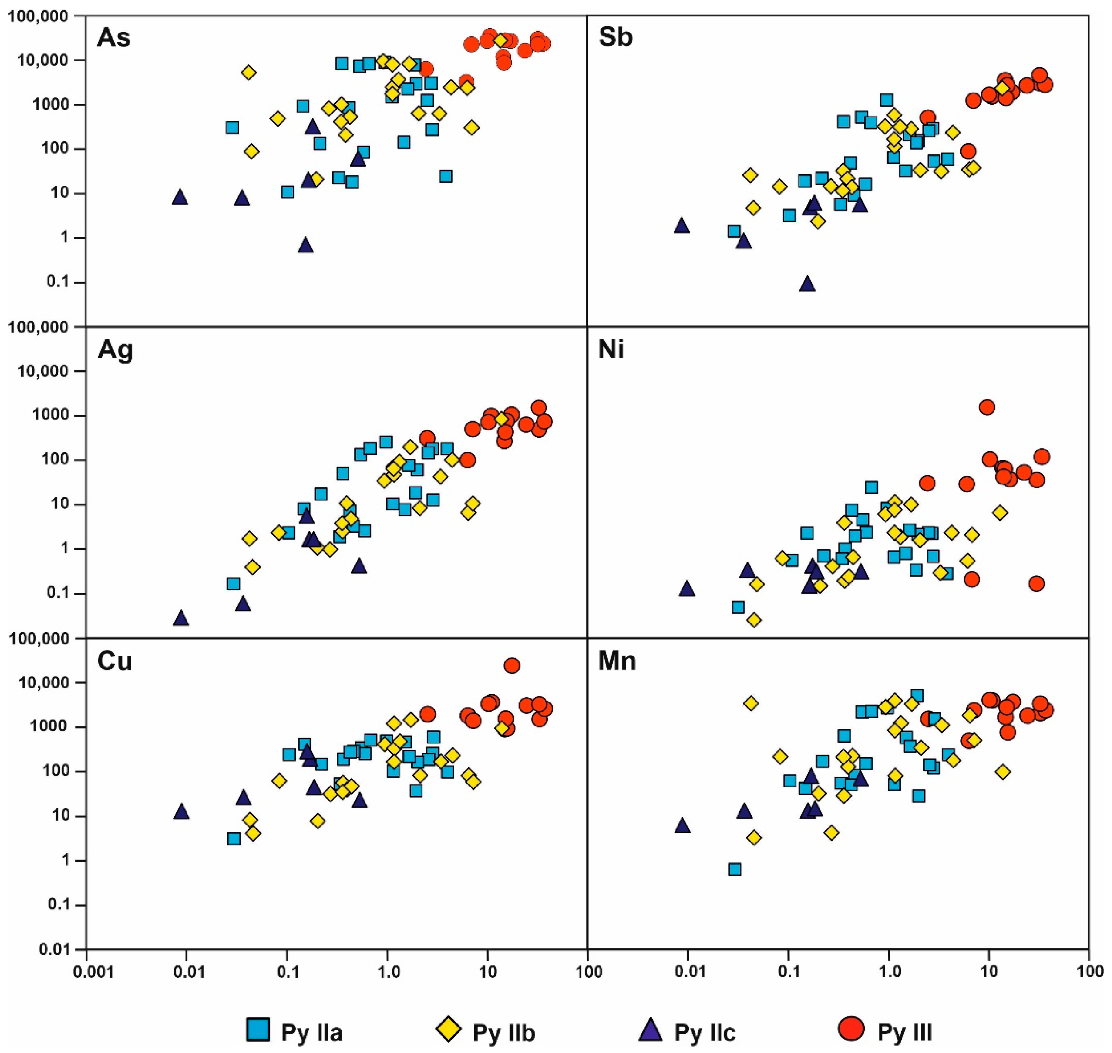
5. Au Distribution
6. Discussion
6.1. Ore-Forming Stages
6.2. Timing of Au Precipitation
6.3. Au Distribution in Zn-Pb Ores
7. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Isotope | 34S | 55Mn | 59Co | 60Ni | 65Cu | 66Zn | 75As | 77Se | 95Mo | 107Ag | 111Cd | 121Sb | 125Te | 197Au | 206Pb | 209Bi | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ||
| Reference material MASS-1 used for calibration of pyrite analyses | |||||||||||||||||
| MASS-1 | Working values | 276,000 | 280 | 60 | 97 | 134,000 | 210,000 | 65 | 51 | 59 | 50 | 60 | 60 | 15 | 47 | 68 | 60 |
| S.D | 1000 | 80 | 10 | 15 | 500 | 5000 | 3 | 4 | 9 | 5 | 7 | 9 | 7 | ||||
| Values obtained for UQAC-FeS1 and GSE-1g | |||||||||||||||||
| UQAC-FeS1 | Working values | 389,566 | 55.0 | 636.2 | 25,730.00 | 22,664.00 | 257.5 | 1055.00 | 330.00 | 66.0 | 165.6 | 8.2 | 149.5 | 65.0 | 90.00 | 120.00 | |
| S.D. | 41,000 | 5.8 | 47.0 | 1500.00 | 1300.00 | 27.5 | 105.00 | 42.00 | 3.5 | 15.5 | 8.8 | 17.0 | 6.5 | 9.00 | 20.00 | ||
| This study | average | 394,750 | 76.7 | 576.5 | 20,715.00 | 20,915.00 | 322.5 | 1169.50 | 295.55 | 62.45 | 171.25 | 2.45 | 73.75 | 192.85 | 62.25 | 100.4 | 136.1 |
| n = 2 | S.D. | 9263 | 2.8 | 3.5 | 4235.57 | 205.06 | 72.8 | 71.42 | 12.09 | 1.1 | 13.79 | 0.36 | 3.5 | 6.2 | 1.1 | 32.0 | 42.28 |
| Rel. Diff. | 1.01 | 1.39 | 0.91 | 0.81 | 0.92 | 1.25 | 1.11 | 0.90 | 0.95 | 1.03 | 0.85 | 1.29 | 0.96 | 1.12 | 1.13 | ||
| GSE-1g | Working values | 590.00 | 380.00 | 440.00 | 380.00 | 460.00 | 260.00 | 20.00 | 390.00 | 200.00 | 160.00 | 450.00 | 7.00 | 378.00 | 320.00 | ||
| S.D. | 20.00 | 20.00 | 30.00 | 40.00 | 10.00 | 90.00 | 16.00 | 30.00 | 20.00 | 50.00 | 110.00 | 12.00 | 30.00 | ||||
| This study | average | 3550 | 790.65 | 361.45 | 470.50 | 640.00 | 490.00 | 349.55 | 64.05 | 389.00 | 195.20 | 244.00 | 305.75 | 294.85 | 8.51 | 351.45 | 310.15 |
| n = 2 | S.D. | 636 | 63.14 | 1.2 | 58.69 | 14.14 | 70.71 | 19.02 | 6.72 | 12.73 | 6.93 | 3.11 | 3.46 | 9.69 | 0.3 | 33.59 | 5.87 |
| Rel. Diff. | 1.34 | 0.95 | 1.07 | 1.68 | 1.07 | 1.34 | 3.20 | 1.0 | 0.98 | 1.53 | 0.68 | 1.22 | 0.93 | 0.97 | |||
| 197Au | 75As | 121Sb | 107Ag | 59Co | 60Ni | 65Cu | 66Zn | 206Pb | 111Cd | 55Mn | 77Se | 95Mo | 125Te | 209Bi | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample/Spot_Line (Py) | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm |
| CAST-001_c1.d (Py IIa) | 0.37 | 8450.00 | 394.00 | 50.00 | 0.04 | 0.98 | 189.00 | 990.00 | 506.00 | 1.87 | 612.00 | 0.17 | 1.51 | 0.01 | bdl |
| CAST-001_c2.d (Py IIa) | 0.55 | 7290.00 | 497.00 | 135.00 | 0.10 | 4.50 | 346.00 | 830.00 | 709.00 | 2.31 | 2139.00 | 0.20 | 4.50 | 0.01 | bdl |
| CAST-001_c3.d (Py IIa) | 0.68 | 8440.00 | 371.00 | 186.30 | 0.37 | 24.49 | 512.00 | 11,600.00 | 1610.00 | 29.30 | 2218.00 | 0.43 | 10.36 | 0.15 | 0.01 |
| CAST-001_c6.d (Py IIa) | 0.98 | 9070.00 | 1205.00 | 258.00 | 0.15 | 8.14 | 491.00 | 1200.00 | 719.00 | 1.73 | 2638.00 | 0.21 | 3.59 | 0.02 | bdl |
| Cast-004_C1-2.d (Py III) | 14.90 | 11,800.00 | 3370.00 | 272.00 | 2.35 | 67.00 | 910.00 | 55.00 | 340.00 | 0.94 | 1620.00 | 0.80 | 6.20 | 1.00 | 0.02 |
| Cast-004_C1-3.d (Py III) | 33.00 | 30,100.00 | 2900.00 | 500.00 | 0.10 | 35.70 | 1540.00 | 140.00 | 510.00 | 0.50 | 2000.00 | 0.01 | 6.80 | 2.00 | 0.01 |
| Cast-004_C2-1.d (Py III) | 37.30 | 23,440.00 | 2680.00 | 757.00 | 1.30 | 120.00 | 2630.00 | 31.10 | 250.00 | 0.66 | 2330.00 | 1.02 | 5.40 | 1.10 | 0.01 |
| Cast-004_C2-2.d (Py III) | 24.60 | 16,300.00 | 2560.00 | 647.00 | 0.56 | 53.00 | 3130.00 | 360.00 | 210.00 | 0.63 | 1740.00 | 8.00 | 4.69 | 1.26 | 0.01 |
| Cast-004_C2-3.d (Py III) | 6.40 | 3200.00 | 82.00 | 101.00 | 0.96 | 29.00 | 1830.00 | 68.00 | 240.00 | 0.13 | 470.00 | 1.00 | 4.10 | 0.08 | 0.02 |
| Cast-004_C3-1.d (Py III) | 17.60 | 26,900.00 | 1820.00 | 1080.00 | 1.08 | 37.00 | 25,000.00 | 540.00 | 1600.00 | 0.51 | 3620.00 | 0.80 | 9.80 | 0.55 | 0.04 |
| Cast-004_C3-2.d (Py III) | 11.00 | 35,000.00 | 1440.00 | 1010.00 | 4.80 | 104.00 | 3700.00 | 1600.00 | 7600.00 | 0.77 | 3870.00 | 5.70 | 9.50 | 3.20 | 29.00 |
| Cast-004_C3-3.d (Py III) | 32.90 | 22,600.00 | 4420.00 | 1560.00 | 0.32 | 0.16 | 3320.00 | 72.00 | 300.00 | 2.90 | 3270.00 | 2.80 | 8.30 | 1.06 | 0.04 |
| Cast-004_C3-4.d (Py III) | 7.20 | 22,400.00 | 1170.00 | 509.00 | 5.50 | 0.20 | 1410.00 | 730.00 | 3100.00 | 13.00 | 2320.00 | 0.30 | 26.80 | 0.13 | 0.04 |
| Cast-004_C3-5.d (Py III) | 10.30 | 27,000.00 | 1590.00 | 730.00 | 6.20 | 1600.00 | 3400.00 | 1900.00 | 1700.00 | 0.99 | 3960.00 | 3.70 | 19.00 | 0.38 | 0.05 |
| CAST-004-Py.d (Py III) | 15.62 | 27,890.00 | 2587.00 | 771.00 | 0.38 | 63.70 | 944.00 | 50,000.00 | 84.00 | 155.00 | 731.00 | 2.12 | 57.20 | 2.03 | 0.03 |
| CAST-004-Py2.d (Py III) | 15.20 | 8720.00 | 1350.00 | 436.00 | 0.51 | 42.20 | 1570.00 | 102,000.00 | 190.00 | 300.00 | 2740.00 | 1.25 | 21.40 | 1.08 | 0.02 |
| CAST-004-Py3.d (Py III) | 2.53 | 6310.00 | 481.00 | 315.00 | 0.33 | 30.10 | 1980.00 | 190.00 | 220.00 | 0.79 | 1470.00 | 0.98 | 13.30 | 0.07 | 0.03 |
| CE-61c3a.d (Py IIa) | 2.83 | 3070.00 | 275.00 | 184.00 | 0.06 | 2.21 | 256.20 | 3600.00 | 2020.00 | 10.40 | 115.00 | 0.25 | 1.02 | 0.05 | 0.01 |
| CE-61c2a.d (Py IIa) | 2.59 | 1255.00 | 240.00 | 149.10 | 0.21 | 2.28 | 187.00 | 29.00 | 1310.00 | 0.26 | 134.30 | 0.29 | 1.55 | 0.05 | 0.01 |
| CE-61c2b.d (Py IIa) | 3.98 | 24.60 | 54.90 | 183.80 | 0.01 | 0.27 | 98.50 | 13.20 | 298.00 | 0.06 | 229.90 | 0.22 | 0.02 | 0.03 | bdl |
| CE-61c3a.d (Py IIa) | 2.02 | 2950.00 | 145.00 | 61.20 | 0.12 | 2.10 | 163.00 | 680.00 | 1332.00 | 1.93 | 26.50 | 0.10 | 0.29 | 0.03 | bdl |
| CE-61c5.d (Py IIb) | 13.93 | 27,810.00 | 2242.00 | 873.00 | 0.11 | 6.49 | 947.00 | 2590.00 | 1648.00 | 7.90 | 93.10 | 0.74 | 7.24 | 0.07 | bdl |
| CE-61c6.d (Py IIb) | 4.47 | 2470.00 | 221.00 | 103.00 | 0.02 | 2.30 | 232.00 | 6330.00 | 1556.00 | 16.10 | 171.00 | 0.19 | 0.31 | 0.05 | bdl |
| SR-208-5_50c1a.d (Py IIa) | 1.66 | 2260.00 | 196.00 | 77.20 | 1.92 | 2.63 | 218.00 | 32.00 | 467.00 | 0.12 | 353.00 | 0.21 | 3.87 | bdl | 0.03 |
| SR-208-5_50c2a.d (Py IIa) | 1.15 | 1490.00 | 60.50 | 10.31 | 0.22 | 0.64 | 102.20 | 2.90 | 1080.00 | 0.06 | 47.90 | 0.14 | 0.03 | bdl | 0.01 |
| SR-208-5_50c2b.d (Py IIa) | 0.22 | 135.00 | 20.40 | 17.10 | 0.81 | 0.68 | 147.00 | 37.80 | 228.00 | 0.05 | 162.00 | 0.06 | 0.06 | 0.01 | 0.03 |
| SR-208-5_50c3a.d (Py IIa) | 0.03 | 307.00 | 1.27 | 0.16 | 0.01 | 0.05 | 2.99 | 0.49 | 38.60 | bdl | 0.57 | 0.09 | 0.01 | bdl | bdl |
| SR-208-5_50c3b.d (Py IIa) | 1.94 | 7860.00 | 130.40 | 18.40 | 0.56 | 0.33 | 36.70 | 20.40 | 940.00 | 0.08 | 5030.00 | 0.02 | 0.50 | 0.03 | 0.03 |
| SR-208-5_50c1b.d (Py IIb) | 1.34 | 3660.00 | 296.00 | 93.40 | 0.13 | 1.84 | 480.00 | 13.14 | 263.00 | 0.07 | 1187.00 | bdl | 0.70 | bdl | 0.06 |
| SR-208-5_50c4a.d (Py IIb) | 0.05 | 89.00 | 4.29 | 0.38 | 0.04 | 0.16 | 3.96 | 2.39 | 142.00 | 0.10 | 3.00 | 0.01 | 0.06 | bdl | bdl |
| SR-208-5_50c4b.d (Py IIb) | 0.04 | 5280.00 | 23.87 | 1.67 | 0.01 | 0.02 | 8.00 | 20.60 | 15.90 | 0.07 | 3334.00 | 0.15 | 0.10 | bdl | bdl |
| SR-208-5_50c6c.d (Py IIb) | 0.20 | 20.90 | 2.13 | 1.05 | 0.15 | 0.14 | 7.60 | 0.92 | 17.60 | bdl | 29.90 | 0.02 | 0.02 | bdl | bdl |
| SR-208-5_50c8a.d (Py IIb) | 0.40 | 210.00 | 19.60 | 10.63 | 0.04 | 0.23 | 39.20 | 8.26 | 289.00 | 0.04 | 121.00 | 0.01 | 1.10 | 0.02 | 0.01 |
| SR-208-5_50c8b.d (Py IIb) | 2.13 | 640.00 | 30.90 | 8.28 | 7.80 | 1.55 | 82.00 | 12.50 | 780.00 | 0.14 | 328.00 | 1.87 | 1.17 | bdl | 0.21 |
| SR-208-5_50c9a.d (Py IIb) | 3.43 | 631.00 | 29.20 | 42.80 | 0.20 | 0.28 | 168.20 | 29.70 | 234.00 | 0.04 | 1070.00 | 0.04 | 0.26 | bdl | bdl |
| SR-208-5_50c9b.d (Py IIb) | 0.44 | 531.00 | 12.77 | 4.75 | 0.19 | 0.64 | 45.90 | 2.93 | 996.00 | 0.08 | 214.00 | 2.08 | 0.01 | 0.12 | 0.02 |
| SR-208-5_50c10a.d (Py IIb) | 6.50 | 2400.00 | 32.00 | 6.50 | 0.31 | 0.53 | 81.00 | 20.10 | 22.00 | 0.07 | 1790.00 | 0.08 | 0.11 | bdl | 0.02 |
| SR-208-5_50c10b.d (Py IIb) | 7.20 | 306.00 | 35.20 | 10.57 | 1.61 | 2.05 | 58.30 | 3.89 | 626.00 | 0.07 | 486.00 | 0.34 | 0.07 | 0.05 | 0.03 |
| SR-208-5_50c11a.d (Py IIb) | 1.17 | 1750.00 | 156.00 | 64.60 | 1.35 | 2.29 | 167.00 | 29.90 | 634.00 | 0.02 | 815.00 | 0.13 | 2.76 | 0.07 | 0.13 |
| SR-208-5_50c11b.d (Py IIb) | 0.36 | 415.00 | 10.63 | 3.74 | 5.61 | 3.85 | 34.10 | 3.15 | 492.00 | 0.03 | 203.00 | 3.68 | bdl | 0.05 | 1.17 |
| CAST-018Ac3b.d (Py IIa) | 0.15 | 920.00 | 17.50 | 8.02 | 1.05 | 2.24 | 417.00 | 56.00 | 1337.00 | 0.21 | 39.40 | 0.20 | 0.19 | 0.02 | 0.15 |
| CAST-018A_c8a.d (Py IIa) | 0.43 | 860.00 | 44.80 | 7.09 | 1.11 | 7.40 | 276.00 | 25.10 | 1620.00 | 0.12 | 47.70 | 0.15 | 0.42 | 0.01 | 0.03 |
| CAST-018Ac4.d (Py IIb) | 0.08 | 484.00 | 13.16 | 2.29 | 10.10 | 0.60 | 61.60 | 26.20 | 392.00 | 0.05 | 205.00 | 0.38 | 0.05 | 0.02 | 0.58 |
| CAST-018Ac5a.d (Py IIb) | 0.95 | 9560.00 | 303.00 | 34.60 | 0.01 | 6.09 | 417.00 | 61.10 | 0.38 | bdl | 2770.00 | 0.22 | 1.04 | 0.02 | bdl |
| CAST-018Ac5b.d (Py IIb) | 1.18 | 2510.00 | 108.00 | 48.00 | 0.15 | 11.40 | 332.00 | 2.71 | 549.00 | 0.05 | 76.00 | 0.19 | 0.38 | 0.16 | 0.12 |
| CAST-018Ac6a.d (Py IIb) | 1.72 | 8320.00 | 267.30 | 201.00 | 0.03 | 9.97 | 1490.00 | 28.70 | 9.22 | 0.03 | 3230.00 | 0.31 | 0.87 | 0.14 | bdl |
| CAST-018Ac6b.d (Py IIb) | 0.36 | 1010.00 | 30.10 | 2.45 | 0.18 | 0.19 | 55.60 | 1.20 | 871.00 | 0.03 | 26.80 | 0.09 | 0.05 | bdl | 0.04 |
| CAST-018Ac7a.d (Py IIb) | 1.17 | 8030.00 | 552.00 | 70.80 | 0.02 | 7.55 | 1230.00 | 39.60 | 8.40 | 0.04 | 3890.00 | 0.28 | 1.15 | 0.31 | bdl |
| CAST-018A_c7b.d (Py IIb) | 0.27 | 821.00 | 13.49 | 0.96 | 1.58 | 0.40 | 31.00 | 0.46 | 405.00 | bdl | 3.87 | 0.11 | 0.01 | 0.04 | 0.06 |
| SR-185-0_6c1.d (Py IIa) | 0.34 | 22.90 | 5.19 | 1.84 | 0.58 | 0.59 | 53.20 | 30.60 | 49.30 | 0.07 | 51.80 | 0.10 | 0.31 | 0.01 | 0.02 |
| SR-185-0_6c4a.d (Py IIa) | 0.11 | 10.90 | 2.92 | 2.27 | 0.50 | 0.54 | 242.00 | 20.00 | 35.90 | 0.13 | 58.00 | 0.13 | 0.31 | 0.02 | 0.03 |
| SR-185-0_6c5.d (Py IIa) | 2.90 | 274.50 | 49.70 | 12.64 | 1.34 | 0.67 | 603.00 | 319.00 | 649.00 | 1.60 | 1486.00 | 0.20 | 0.84 | 0.03 | 0.15 |
| SR-185-0_6c6.d (Py IIa) | 0.60 | 85.50 | 14.97 | 2.51 | 4.58 | 2.34 | 255.10 | 880.00 | 165.40 | 2.54 | 143.00 | 0.14 | 2.67 | 0.03 | 0.02 |
| SR-185-0_6c8.d (Py IIa) | 1.52 | 142.80 | 29.90 | 7.64 | 1.73 | 0.78 | 469.00 | 344.00 | 429.00 | 1.84 | 566.00 | 0.17 | 0.90 | 0.05 | 0.02 |
| SR-185-0_6c9.d (Py IIa) | 0.46 | 18.20 | 8.29 | 3.20 | 1.34 | 1.94 | 286.60 | 271.00 | 125.20 | 1.63 | 76.40 | 0.14 | 0.18 | 0.03 | 0.13 |
| SR-185-0_6c2.d (Py IIc) | 0.01 | 8.60 | 1.76 | 0.03 | 0.07 | 0.13 | 12.74 | 13.60 | 1.32 | 0.08 | 5.80 | 0.17 | 1.41 | 0.03 | bdl |
| SR-185-0_6c3.d (Py IIc) | 0.17 | 20.50 | 4.47 | 1.64 | 0.50 | 0.41 | 193.00 | 90.00 | 66.10 | 0.27 | 76.00 | 0.09 | 0.47 | 0.03 | 0.01 |
| SR-185-0_6c4b.d (Py IIc) | 0.16 | 0.71 | 0.08 | 5.62 | 0.02 | 0.15 | 288.90 | 36.80 | 3.07 | 0.10 | 12.29 | 0.10 | bdl | 0.01 | bdl |
| SR-185-0_6c7a.d (Py IIc) | 0.19 | 324.00 | 5.67 | 1.63 | 0.14 | 0.31 | 44.40 | 5.90 | 249.00 | 0.03 | 13.90 | 0.45 | 0.31 | 0.05 | 0.24 |
| SR-185-0_6c7b.d (Py IIc) | 0.53 | 62.20 | 5.22 | 0.42 | 0.07 | 0.30 | 22.90 | 33.00 | 32.80 | 0.04 | 66.00 | 0.04 | 0.96 | bdl | 0.03 |
| SR-185-0_6c10.d (Py IIc) | 0.04 | 8.30 | 0.78 | 0.06 | 0.81 | 0.33 | 26.60 | 45.00 | 4.40 | 0.05 | 12.30 | 0.40 | 1.10 | 0.03 | bdl |
| 197Au | 75As | 121Sb | 107Ag | 59Co | 60Ni | 65Cu | 66Zn | 206Pb | 111Cd | 55Mn | 77Se | 95Mo | 125Te | 209Bi | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample/Spot_Line | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm |
| Cast-004_C1-1.d | bdl | 0.12 | 2.90 | 6.40 | 4.60 | 0.43 | 360.0 | 682,000.0 | 11.9 | 590.0 | 846.0 | 1.59 | 0.10 | 0.31 | 0.03 |
| Cast-004_C2-4.d | bdl | 0.80 | 7.50 | 8.30 | 3.27 | 0.70 | 2500.0 | 664,000.0 | 220.0 | 2750.0 | 808.0 | 1.70 | 0.12 | 0.21 | 0.04 |
| SR-208-5_50c5.d | 0.12 | 0.45 | 8.41 | 50.90 | 0.16 | 0.04 | 4550.0 | 577,900.0 | 550.0 | 1552.0 | 608.0 | 0.32 | 0.01 | 0.03 | bdl |
| SR-208-5_50c7a.d | 0.01 | 0.76 | 0.46 | 6.67 | 0.36 | 0.02 | 120.0 | 616,300.0 | 14.9 | 1600.0 | 600.0 | 0.52 | 0.01 | 0.03 | 0.01 |
| CAST-001_c4.d | 0.07 | 4.50 | 10.90 | 52.70 | 6.18 | 2.34 | 6010.0 | 568,800.0 | 118.0 | 2830.0 | 717.0 | 0.30 | 0.02 | 0.03 | 0.01 |
| CAST-001_c5.d | 0.04 | 6.00 | 6.67 | 34.70 | 5.68 | 0.76 | 2310.0 | 549,000.0 | 207.0 | 2763.0 | 319.0 | 0.26 | 0.01 | 0.26 | 0.01 |
| CE-61c4a.d | 0.05 | 1.60 | 40.10 | 125.80 | 0.01 | 0.13 | 4230.0 | 607,800.0 | 7500.0 | 3152.0 | 203.0 | 0.12 | 0.01 | 0.01 | bdl |
| CE-61c7.d | 0.05 | 5.30 | 18.50 | 88.10 | bdl | 0.09 | 449.0 | 597,100.0 | 651.0 | 2757.0 | 603.0 | 0.09 | 0.01 | 0.02 | bdl |
| CE-61c8.d | 0.33 | 12.00 | 87.20 | 163.00 | 0.01 | 0.07 | 767.0 | 540,600.0 | 122,000.0 | 3283.0 | 890.0 | 0.11 | 0.01 | 0.03 | 0.01 |
| SR-208-5_50c6b.d | 0.76 | 57.00 | 141.00 | 374.00 | 7.82 | 2.10 | 12,000.0 | 7740,000.0 | 5100.0 | 26,780.0 | 6140.0 | 6.10 | 0.67 | 0.20 | 0.04 |
| 197Au | 75As | 121Sb | 107Ag | 59Co | 60Ni | 65Cu | 66Zn | 206Pb | 111Cd | 55Mn | 77Se | 95Mo | 125Te | 209Bi | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample/Spot_Line | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm |
| SR-208-5_50c6a.d | bdl | 0.23 | 402.00 | 427.00 | bdl | 0.01 | 0.56 | 0.11 | 768,300.0 | 8.27 | 0.02 | 1.30 | 0.01 | 0.02 | 0.21 |
| SR-208-5_50c7b.d | 0.03 | 0.23 | 161.40 | 195.50 | bdl | 0.03 | 1.37 | 0.01 | 787,200.0 | 10.92 | 0.02 | 32.50 | 0.01 | 0.07 | 0.18 |
| CE-61c1.d | 0.02 | 10.40 | 349.10 | 383.70 | 0.03 | 2.30 | 4.62 | 142.00 | 724,000.0 | 10.29 | 5.00 | 2.15 | 0.06 | 0.75 | 0.03 |
| CE-61c4b.d | 0.01 | 2.30 | 423.40 | 466.20 | 0.01 | 0.67 | 3.10 | 24.40 | 761,000.0 | 16.40 | 0.50 | 0.86 | 0.06 | 0.29 | 0.03 |
| CAST-018Ac2.d | 0.01 | 0.08 | 92.80 | 114.60 | 0.01 | 0.01 | 1.16 | 0.62 | 798,700.0 | 17.47 | 0.18 | 6.56 | bdl | 0.12 | 1.40 |
| CAST-018Ac3a.d | bdl | 0.13 | 73.90 | 150.40 | 0.01 | 0.03 | 1.06 | 0.69 | 790,800.0 | 26.13 | 0.52 | 31.80 | bdl | 1.08 | 3.03 |
| CAST-018Ac8b.d | bdl | 0.06 | 93.60 | 108.00 | 0.05 | 0.09 | 1.87 | 3.75 | 798,000.0 | 22.27 | 0.85 | 2.61 | 0.52 | 0.04 | 0.45 |
Appendix B

References
- Emsbo, P. Gold in sedex deposits. Rev. Econ. Geol. 2000, 13, 427–437. [Google Scholar]
- Emsbo, P.; Hutchinson, R.W.; Hofstra, A.H.; Volk, J.A.; Bettles, K.H.; Baschuk, G.J.; Johnson, C.A. Syngenetic Au on the Carlin trend: Implications for Carlin-Type deposits. Geology 1999, 27, 59–62. [Google Scholar] [CrossRef]
- Oudin, E. Geochemistry of submarine sulfides. In Marine Minerals: Advances in Research and Resource Assessment; Teleki, P.G., Dobson, M.R., Moore, J.R., von Stackelberg, U., Eds.; Springer: Dordrecht, The Netherlands, 1987; pp. 349–362. [Google Scholar]
- Large, D.E.; Walcher, E. The Rammelsberg massive sulphide Cu-Zn-Pb-Ba-deposit, Germany: An example of sediment-hosted, massive sulphide mineralization. Miner. Depos. 1999, 34, 522–538. [Google Scholar] [CrossRef]
- Cooke, D.R.; Bull, S.W.; Large, R.R.; McGoldrick, P.J. The Importance of Oxidized Brines for the Formation of Australian Proterozoic Stratiform Sediment-Hosted Pb-Zn (Sedex) Deposits. Econ. Geol. 2000, 95, 1–18. [Google Scholar] [CrossRef]
- Whitehead, R.E.; Davies, J.F.; Valdes-Nodarse, E.L.; Diaz-Carmona, A. Mineralogical and chemical variations, Castellanos shale-hosted Zn-Pb-Ba deposit, northwestern Cuba. Econ. Geol. 1996, 91, 713–722. [Google Scholar] [CrossRef]
- Cobiella-Reguera, J.L. Jurassic and Cretaceous Geological History of Cuba. Int. Geol. Rev. 2000, 42, 594–616. [Google Scholar] [CrossRef]
- DeGolyer, E. The Geology of Cuban Petroleum Deposits. AAPG Bull. 1918, 2, 133–167. [Google Scholar] [CrossRef]
- Haczewski, G. Sedimentological reconnaissance of the San Cayetano Formation: An accumulative continental margin in the Jurassic of western Cuba. Acta Geol. Pol. 1976, 26, 331–353. [Google Scholar]
- Meyerhoff, A.A.; Hatten, C.W. Bahamas Salient of North America. Geol. Cont. Margins 1974, 58, 429–446. [Google Scholar] [CrossRef]
- García-Casco, A.; Iturralde-Vinent, M.A.; Pindell, J. Latest Cretaceous Collision/Accretion between the Caribbean Plate and Caribeana: Origin of Metamorphic Terranes in the Greater Antilles. Int. Geol. Rev. 2008, 50, 781–809. [Google Scholar] [CrossRef]
- Maynard, J.B.; Elswick, E.R.; Hower, J.C. Reflectance of dispersed vitrinite in shales hosting Pb–Zn–Cu ore deposits in western Cuba: Comparison with clay crystallinity. Int. J. Coal Geol. 2001, 47, 161–170. [Google Scholar] [CrossRef]
- Valdés-Nodarse, E.L. Pb-Zn “SEDEX” deposits and their copper stockwork roots, western Cuba. Miner. Depos. 1998, 33, 560–567. [Google Scholar] [CrossRef]
- Pérez-Valdez, R.G.; Melgarejo, J.C. El yacimiento Matahambre (Pinar del Rio, Cuba): Estructura y mineralogía. Acta Geol. Hisp. 1998, 33, 133–152. [Google Scholar]
- Cobiella-Reguera, J.L. Reconstrucción palinspástica del paleomargen mesozoico de América del Norte en Cuba occidental y el sudeste del Golfo de México. Implicaciones para la evolución del SE del Golfo de México. Rev. Mex. Cienc. Geol. 2008, 25, 382–401. [Google Scholar]
- Astajov, K.; Solianik, V.; Vasilev, V.; Martínez, D.; Fernández de Lara, R.; Oubiña, J.; Demidov, S.; Santamaría, Z. Informe sobre los trabajos de levantamiento geológico a escala 1: 50,000 en parte noroeste de Pinar del Río (hojas 3484-III—3483-III—3483-IIIa) (unpublished charts). Oficina Nac. de Recur. Miner. Minist. de Energía y Minas La Habana 1980, 497. [Google Scholar]
- Gordon, M.; Mann, P.; Cáceres, D.; Flores, R. Cenozoic tectonic history of the North Ameria-Caribbean plate boundary in westetern Cuba. J. Gophys. Res. 1997, 102, 10005–10082. [Google Scholar] [CrossRef]
- Bralower, T.; Iturralde-Vinent, M. Micropaleontological dating of the collision between the North America and Caribean plates in western Cuba. Palaios 1997, 12, 133–150. [Google Scholar] [CrossRef]
- Paton, C.; Hellstrom, J.; Paul, B.; Woodhead, J.; Hergt, J. Iolite: Freeware for the visualization and processing of mass spectrometric data. J. Anal. At. Spectrom. 2011, 26, 2508–2518. [Google Scholar] [CrossRef]
- Sawlowicz, Z. Pyrite framboids and their development: A new conceptual mechanism. Acta Diabetol. 1993, 82, 148–156. [Google Scholar] [CrossRef]
- Liu, Z.; Chen, D.; Zhang, J.; Lü, X.; Wang, Z.; Liao, W.; Shi, X.; Tang, J.; Xie, G. Pyrite Morphology as an Indicator of Paleoredox Conditions and Shale Gas Content of the Longmaxi and Wufeng Shales in the Middle Yangtze Area, South China. Minerals 2019, 9, 428. [Google Scholar] [CrossRef]
- Taylor, K.G.; Macquaker, J.H.S. Iron Minerals in Marine Sediments Record Chemical Environments. Elements 2011, 7, 113–118. [Google Scholar] [CrossRef]
- Ohfuji, H.; Rickard, D. Experimental synthesis of framboids—A review. Earth Sci. Rev. 2005, 71, 147–170. [Google Scholar] [CrossRef]
- Ohmoto, H. Formation of volcanogenic massive sulfide deposits: The Kuroko perspective. Ore Geol. Rev. 1996, 10, 135–177. [Google Scholar] [CrossRef]
- Almodóvar, G.R.; Yesares, L.; Sáez, R.; Toscano, M.; González, F.; Pons, J.M. Massive Sulfide Ores in the Iberian Pyrite Belt: Mineralogical and Textural Evolution. Minerals 2019, 9, 653. [Google Scholar] [CrossRef]
- Lyons, T.W.; Gellatly, A.M.; McAurick, P.J.; Kah, L.C. Proterozoic sedimentary exhalative (SEDEX) deposits andlinks to evolving global ocean chemistry. In Evolution of Early Earth’s Atmosphere, Hydrosphere, and Biosphere—Constraints from ore Deposits; Kesler, S.E., Ohmoto, H., Eds.; Geological Society of America Memoirs: McLean, VA, USA, 2006. [Google Scholar]
- Johnson, C.A.; Emsbo, P.; Poole, F.G.; Rye, R.O. Sulfur- and oxygen-isotopes in sediment-hosted stratiform barite deposits. Geochim. et Cosmochim. Acta 2009, 73, 133–147. [Google Scholar] [CrossRef]
- Seewald, J.S.; Seyfried, W.E. The effect of temperature on metal mobility in subseafloor hydrothermal systems: Constraints from basalt alteration experiments. Earth Planet. Sci. Lett. 1990, 101, 388–403. [Google Scholar] [CrossRef]
- Seward, T.M.; Williams-Jones, A.E.; Migdisov, A.A. The chemistry of metal transport and deposition by ore-forming hydrothermal fluids. In Treatise on Geochemistry, 2nd ed.; Holland, H.D., Turekian, K.K., Eds.; Elsevier-Pergamon: Amsterdam, The Netherland, 2014; Volume 13. [Google Scholar]
- Emsbo, P.; Seal, R.R.; Breit, G.N.; Diehl, S.F.; Shah, A.K. Sedimentary Exhalative (Sedex) Zinc-Lead-Silver Deposit Model; U.S. Geological Survey Investigations Report; U.S. Geological Survey: Reston, VA, USA, 2016. [Google Scholar]
- Lydon, J.W. Chemical parameters controlling the origin and deposition of sediment-hosted stratiform lead-zinc deposits. In Short Course in Sediment-Hosted Stratiform Lead Zinc Deposits; Sangster, D.F., Ed.; Mineralogical Association of Canada, Short Course Handbook: Toronto, ON, Canada, 1983; Volume 8, pp. 175–250. [Google Scholar]
- Hunt, J.M. Petroleum Geochemistry and Geology; Freeman: New York, NY, USA, 1996. [Google Scholar]
- Huston, D.L.; Large, R.R. A chemical model for the concentration of gold in volcanogenic massive sulphide deposits. Ore Geol. Rev. 1989, 4, 171–200. [Google Scholar] [CrossRef]
- Jennings, D.S.; Jilson, G.A. Geology and sulphide deposits of the Anvil Range, Yukon. In Mineral Deposits of Northern Cordillera; Morin, J.A., Ed.; Canadian Institute of Mining and Metallurgy: Montreal, QC, Canada, 1986; Volume 39, pp. 39–361. [Google Scholar]
- Turner, R.J.W.; Otto, B.B. Structural and stratigraphic setting of the Triumph stratiform zinc-lead-silver deposit, Devonian Milligen Formation, Central Idaho. US Geol. Surv. Bull. 1995, 2064, E1–E27. [Google Scholar]
- Mercier-Langevin, P.; Hannington, M.D.; Dubé, B.; Bécu, V. The gold content of volcanogenic massive sulfide deposits. Miner. Depos. 2010, 46, 509–539. [Google Scholar] [CrossRef]
- Mercier-Langevin, P.; Hannington, M.D.; Dubé, B.; Piercey, S.J.; Peter, J.M.; Pehrsson, S.J. Precious metal enrichment processes in volcanogenic massive sulphide deposits—A summary of key features, with emphasis on TIGI-4 research contributions. In Targeted Geoscience Initiative 4: Contributions to the Understanding of Volcanogenic Massive Sulphide Deposits Genesis and Exploration Methods Development; Peter, J.M., Mercier-Langevin, P., Eds.; Geological Survey of Canada Open File: Ottawa, ON, Canada, 2015; Volume 7853, pp. 117–130. [Google Scholar]
- Sack, P.J.; Large, R.R.; Gregory, D.D. Geochemistry of shale and sedimentary pyrite as a proxy for gold fertility in the Selwyn basin area, Yukon. Miner. Depos. 2018, 53, 997–1018. [Google Scholar] [CrossRef]
- Palenick, C.S.; Ustunomiya, S.; Reich, M.; Kesler, S.E.; Wang, L.; Ewing, R.C. “Invisible” Au revealed: Direct imagining of Au nanoparticles in a Carlin-type deposit. Am. Miner. 2004, 89, 1359–1366. [Google Scholar] [CrossRef]
- Deditius, A.P.; Reich, M.; Kesler, S.E.; Utsunomiya, S.; Chryssoulis, S.L.; Walshe, J.; Rodney, C.; Ewing, R.C. The coupled geochemistry of Au and as in pyrite from hydrothermal ore deposits. Geochim. Cosmichim. Acta 2014, 140, 644–670. [Google Scholar] [CrossRef]
- Pokrovski, G.S.; Kara, S.; Roux, J. Stability and solubility of arsenopyrite, FeAsS, in crustal fluids. Geochim. Cosmochim. Acta 2002, 66, 2361–2378. [Google Scholar] [CrossRef]
- McClenaghan, S.H.; Lentz, D.R.; Martin, J.; Diegor, W.G. Gold in the Brunswick No. 12 volcanogenic massive sulfide deposit, Bathurst Mining Camp, Canada: Evidence from bulk ore analysis and laser ablation ICP─MS data on sulfide phases. Miner. Depos. 2009, 44, 523–557. [Google Scholar] [CrossRef]
- Dehnavi, A.S.; McFarlane, C.R.M.; Lentz, D.R.; Walker, J.A. Assessment of pyrite composition by LA-ICP-MS techniques from massive sulfide deposits of the Bathust Mining Camp, Canada: From textural and chemical evolution to its application as a vectorial tool for the exploration of VMS deposits. Ore. Geol. Rev. 2018, 92, 656–671. [Google Scholar] [CrossRef]
- García-Rodríguez, M. Control Textural en la Distribución del oro en Piritas de la Faja Pirítica Ibérica. Master’s Thesis, University of Huelva, Huelva, Spain, 2020. [Google Scholar]
- Gaspar, O.C. mineralogy and sulfide mineral chemistry of the neves corvo ores, portugal: Insight into their genesis. Can. Miner. 2002, 40, 611–636. [Google Scholar] [CrossRef]
- Leistel, J.M.; Marcoux, E.; Deschamps, Y.; Joubert, M. Antithetic behaviour of gold in the volcanogenic massive sulphide deposits of the Iberian Pyrite Belt. Miner. Depos. 1997, 33, 82–97. [Google Scholar] [CrossRef]
- Velasco, F.; Sánchez-España, J.; Yanguas, A.; Tornos, F. The occurrence of gold in the sulfide deposits of the Iberian Pyrite Belt: Evidence of precious metal remobilization. In Volcanic Environments and Massive Sulfide Deposits; Gemmel, J.B., Pontgrats, J., Eds.; Codes and SEG; University of Huelva: Huelva, Spain, 2000; pp. 221–223. [Google Scholar]



| Sample | Texture | N | Minimum | Maximum | Average |
|---|---|---|---|---|---|
| Cast-001 | Colloform (Py IIa) | 4 | 0.37 | 0.98 | 0.65 |
| Top | |||||
| Cast-004 | Colloform, banded (Py III) | 13 | 2.53 | 37.30 | 17.58 |
| CE-61 | Colloform (Py IIa) | 4 | 2.02 | 3.98 | 2.86 |
| Subhedral (Py IIb) | 2 | 4.47 | 13.93 | 9.02 | |
| SR-208/5.5–5.8 | Colloform (Py IIa) | 6 | 0.03 | 1.94 | 1.06 |
| Euhedral (Py IIb) | 11 | 0.04 | 7.20 | 1.99 | |
| Interior | |||||
| Cast-018A | Colloform (Py IIa) | 2 | 0.15 | 0.43 | 0.29 |
| Subhedral (Py IIb) | 7 | 0.08 | 1.72 | 0.82 | |
| Bottom | |||||
| SR-185/0.6–0.7 | Pseudomorphic (Py IIa) | 6 | 0.11 | 2.90 | 0.99 |
| Massive (Py IIc) | 6 | 0.01 | 0.53 | 0.18 | |
| Ag | As | Au | Bi | Cd | Co | Cu | Mn | Mo | Ni | Pb | Sb | Se | Te | Zn | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ag | 1 | ||||||||||||||
| As | 0.88 | 1 | |||||||||||||
| Au | 0.81 | 0.76 | 1 | ||||||||||||
| Bi | 0.32 | 0.39 | 0.09 | 1 | |||||||||||
| Cd | 0.21 | 0.18 | 0.22 | −0.03 | 1 | ||||||||||
| Co | 0.09 | 0.14 | 0.00 | 0.25 | −0.06 | 1 | |||||||||
| Cu | 0.56 | 0.48 | 0.39 | 0.10 | 0.02 | 0.04 | 1 | ||||||||
| Mn | 0.52 | 0.62 | 0.41 | 0.26 | 0.13 | 0.06 | 0.40 | 1 | |||||||
| Mo | 0.55 | 0.62 | 0.39 | 0.08 | 0.61 | 0.13 | 0.20 | 0.28 | 1 | ||||||
| Ni | 0.28 | 0.36 | 0.16 | 0.04 | 0.01 | 0.33 | 0.13 | 0.32 | 0.27 | 1 | |||||
| Pb | 0.40 | 0.49 | 0.10 | 0.82 | −0.05 | 0.31 | 0.22 | 0.27 | 0.18 | 0.16 | 1 | ||||
| Sb | 0.87 | 0.82 | 0.90 | 0.11 | 0.22 | 0.02 | 0.36 | 0.46 | 0.52 | 0.21 | 0.15 | 1 | |||
| Se | 0.55 | 0.49 | 0.45 | 0.47 | 0.10 | 0.32 | 0.21 | 0.30 | 0.29 | 0.33 | 0.38 | 0.47 | 1 | ||
| Te | 0.69 | 0.76 | 0.70 | 0.64 | 0.34 | 0.11 | 0.26 | 0.42 | 0.53 | 0.12 | 0.49 | 0.70 | 0.60 | 1 | |
| Zn | 0.20 | 0.18 | 0.21 | −0.02 | 1.00 | −0.07 | 0.02 | 0.14 | 0.59 | 0.02 | -0.05 | 0.21 | 0.10 | 0.34 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Vivo, D.; Gervilla, F.; Piña, R.; Hernández-Díaz, R.; Azor, A. Gold in the Farallones Block of the Shale-Hosted, Clastic-Dominated Castellanos Zinc-Lead Deposit (Northwest Cuba). Minerals 2021, 11, 414. https://doi.org/10.3390/min11040414
Gómez-Vivo D, Gervilla F, Piña R, Hernández-Díaz R, Azor A. Gold in the Farallones Block of the Shale-Hosted, Clastic-Dominated Castellanos Zinc-Lead Deposit (Northwest Cuba). Minerals. 2021; 11(4):414. https://doi.org/10.3390/min11040414
Chicago/Turabian StyleGómez-Vivo, David, Fernando Gervilla, Rubén Piña, Rebeca Hernández-Díaz, and Antonio Azor. 2021. "Gold in the Farallones Block of the Shale-Hosted, Clastic-Dominated Castellanos Zinc-Lead Deposit (Northwest Cuba)" Minerals 11, no. 4: 414. https://doi.org/10.3390/min11040414
APA StyleGómez-Vivo, D., Gervilla, F., Piña, R., Hernández-Díaz, R., & Azor, A. (2021). Gold in the Farallones Block of the Shale-Hosted, Clastic-Dominated Castellanos Zinc-Lead Deposit (Northwest Cuba). Minerals, 11(4), 414. https://doi.org/10.3390/min11040414






