Assessing Options for Remediation of Contaminated Mine Site Drainage Entering the River Teign, Southwest England
Abstract
1. Introduction
2. Methodology
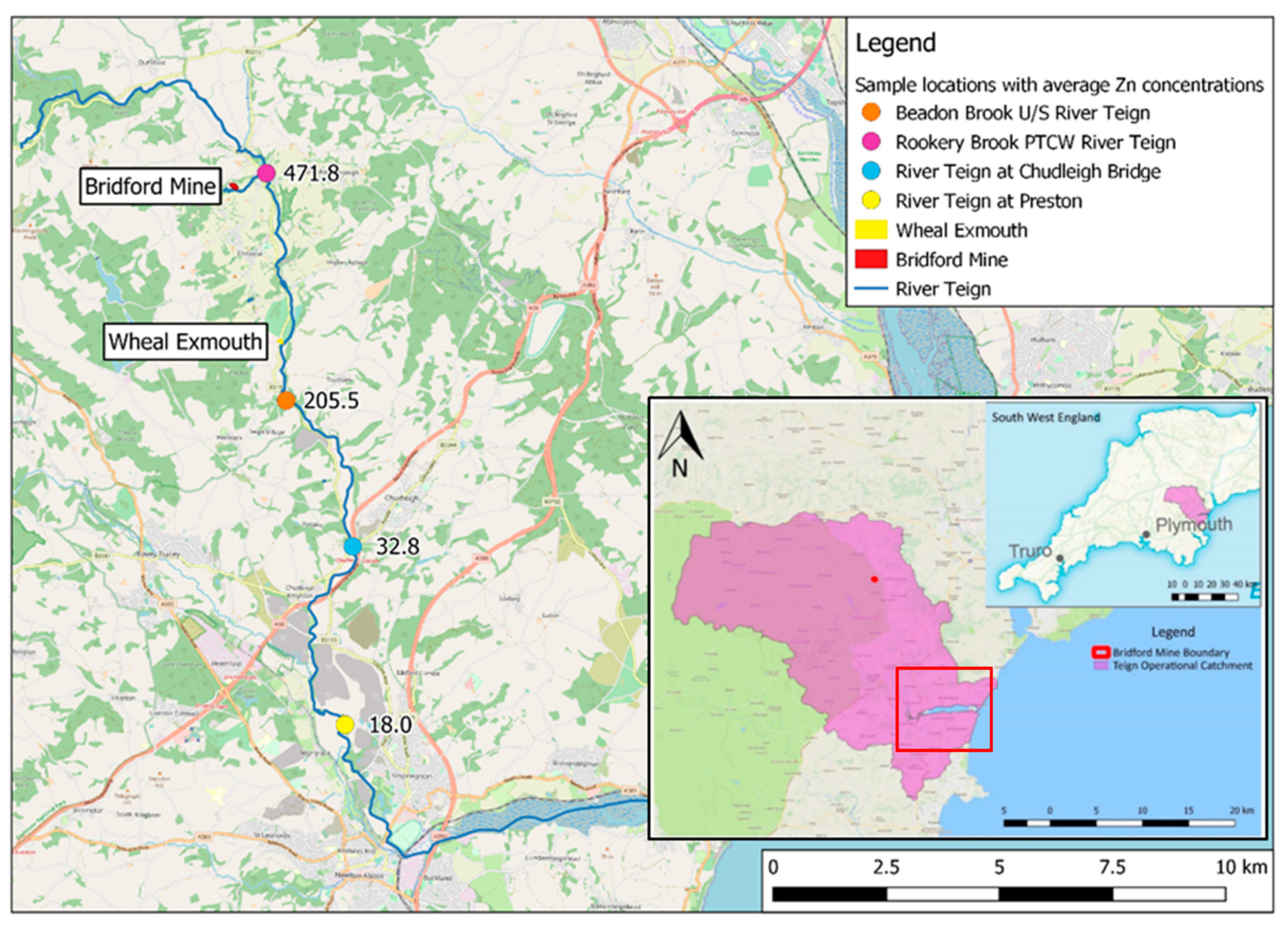
2.1. Study Area
2.2. Current Studies Using Red Media Technology Products
2.3. Alternative Treatment Method Using Biochar
2.4. River Teign Metal Concentrations
2.5. Real-World Application Model
3. Results
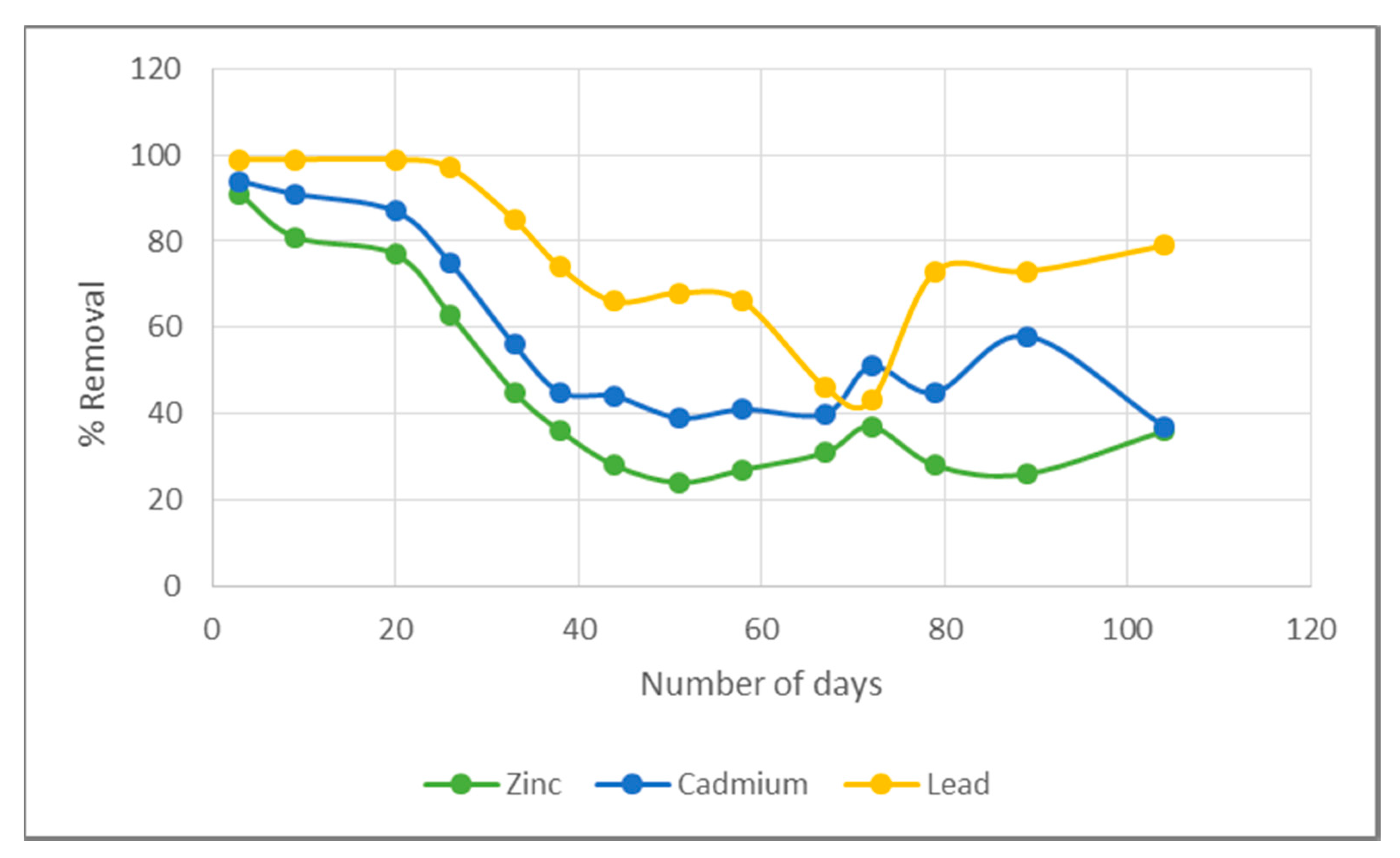
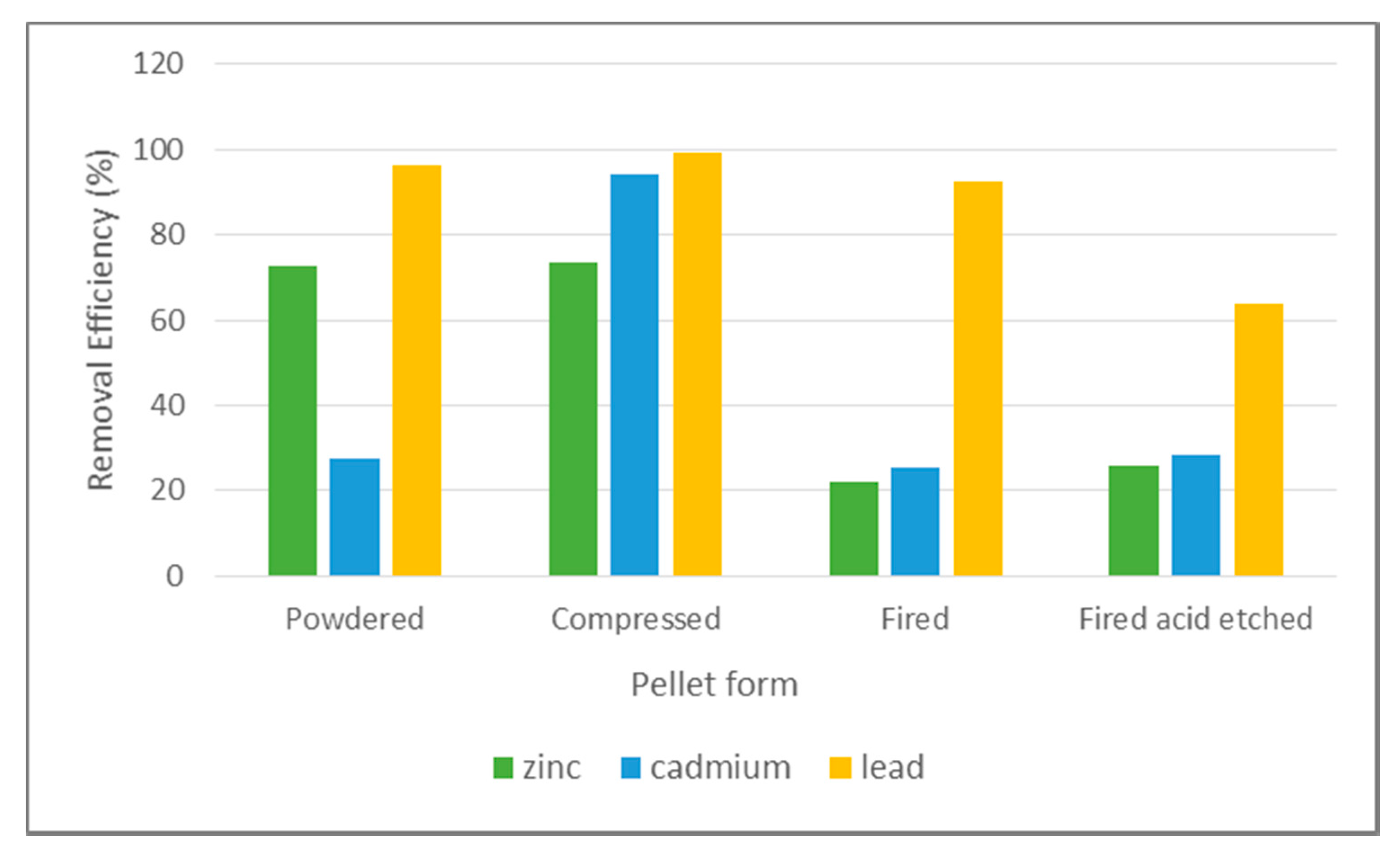
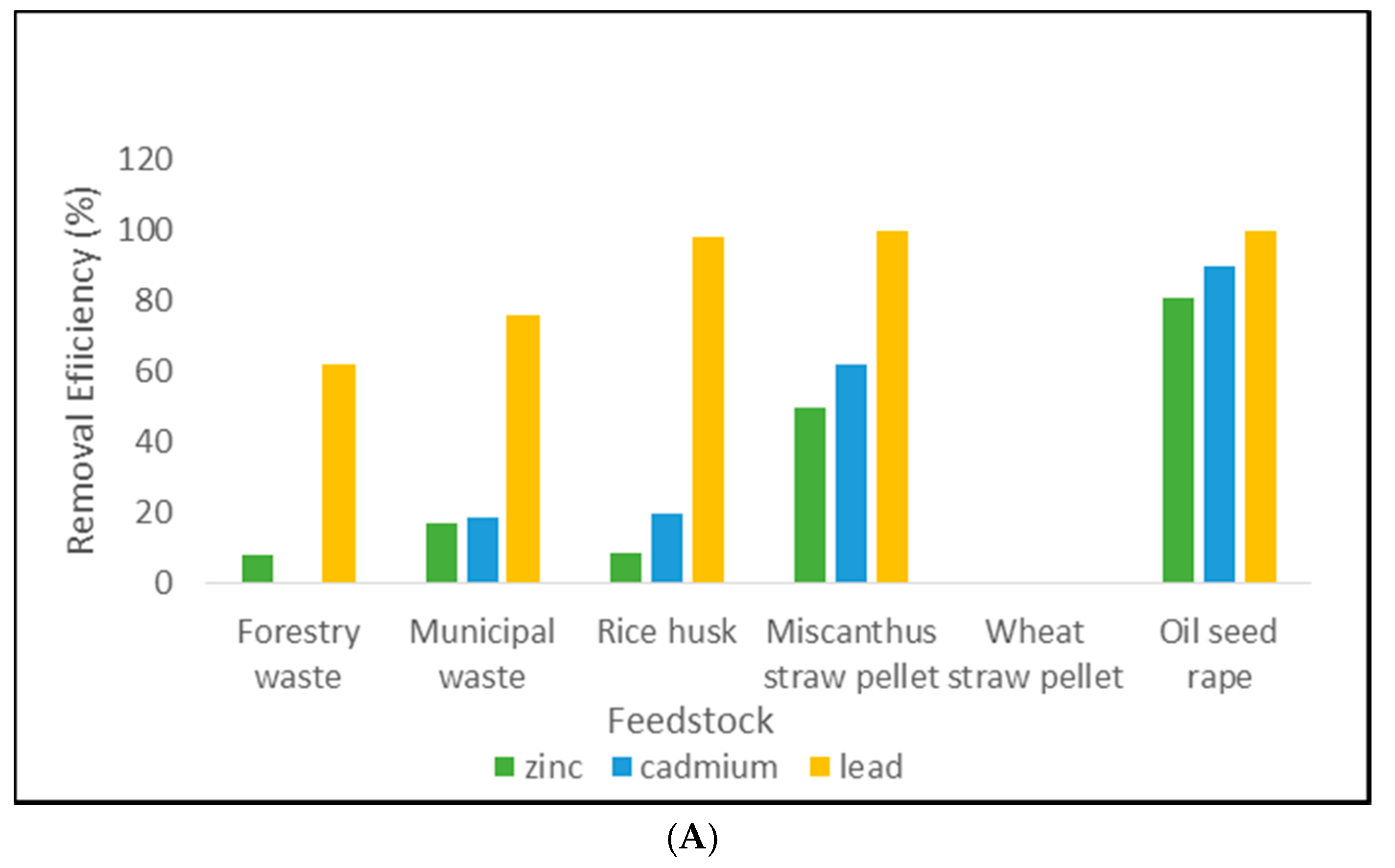
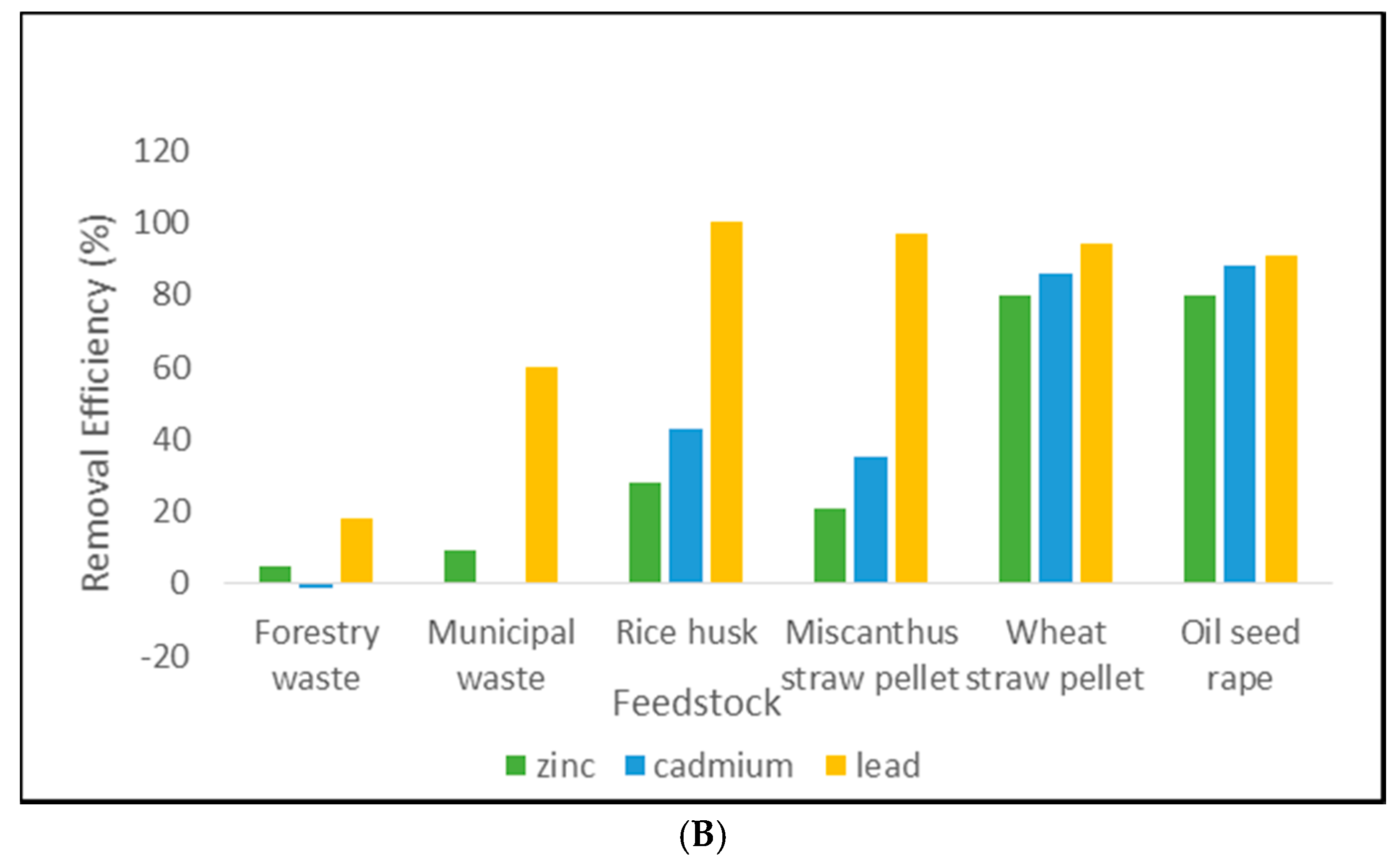
3.1. Removal Efficiency Results
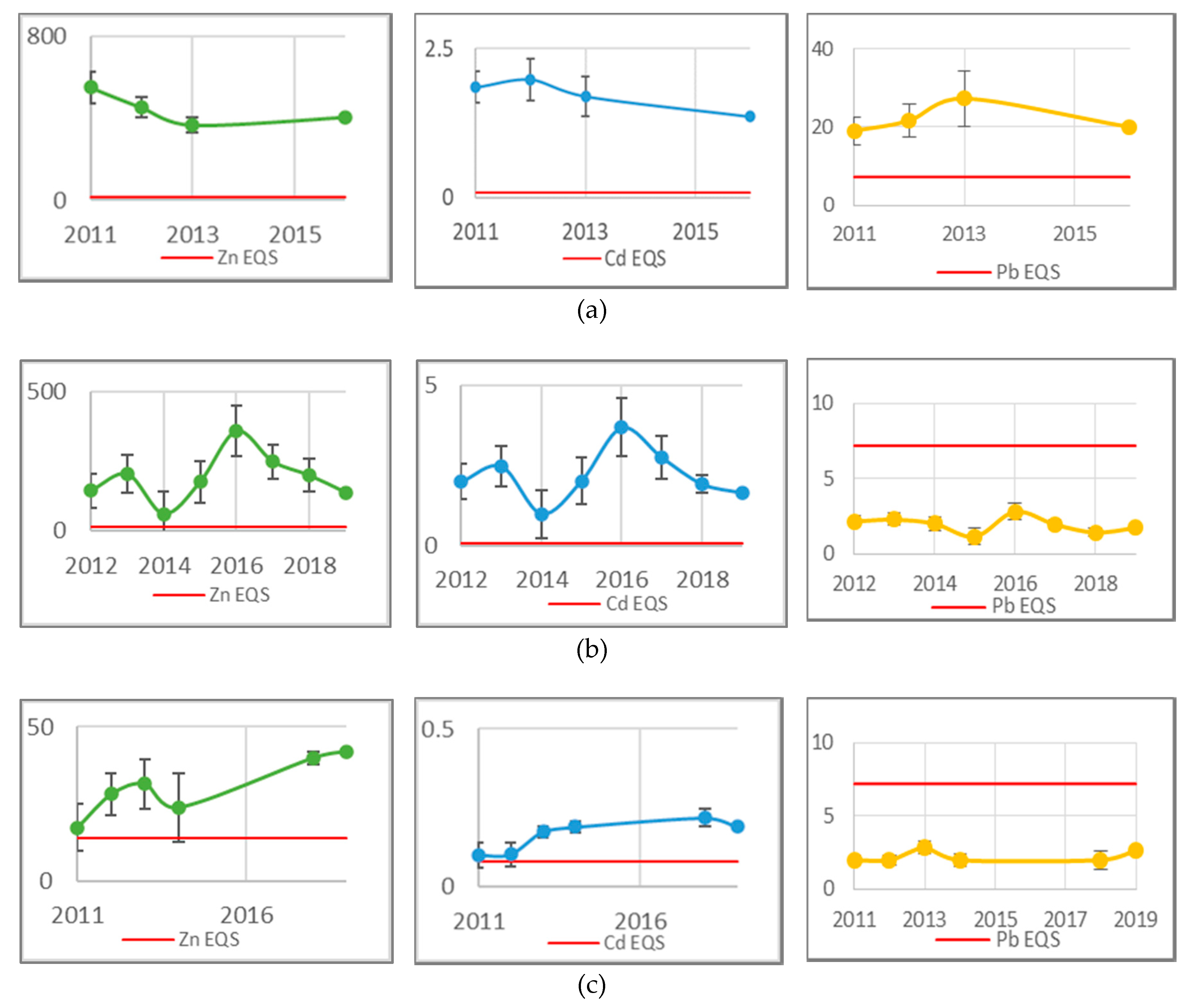
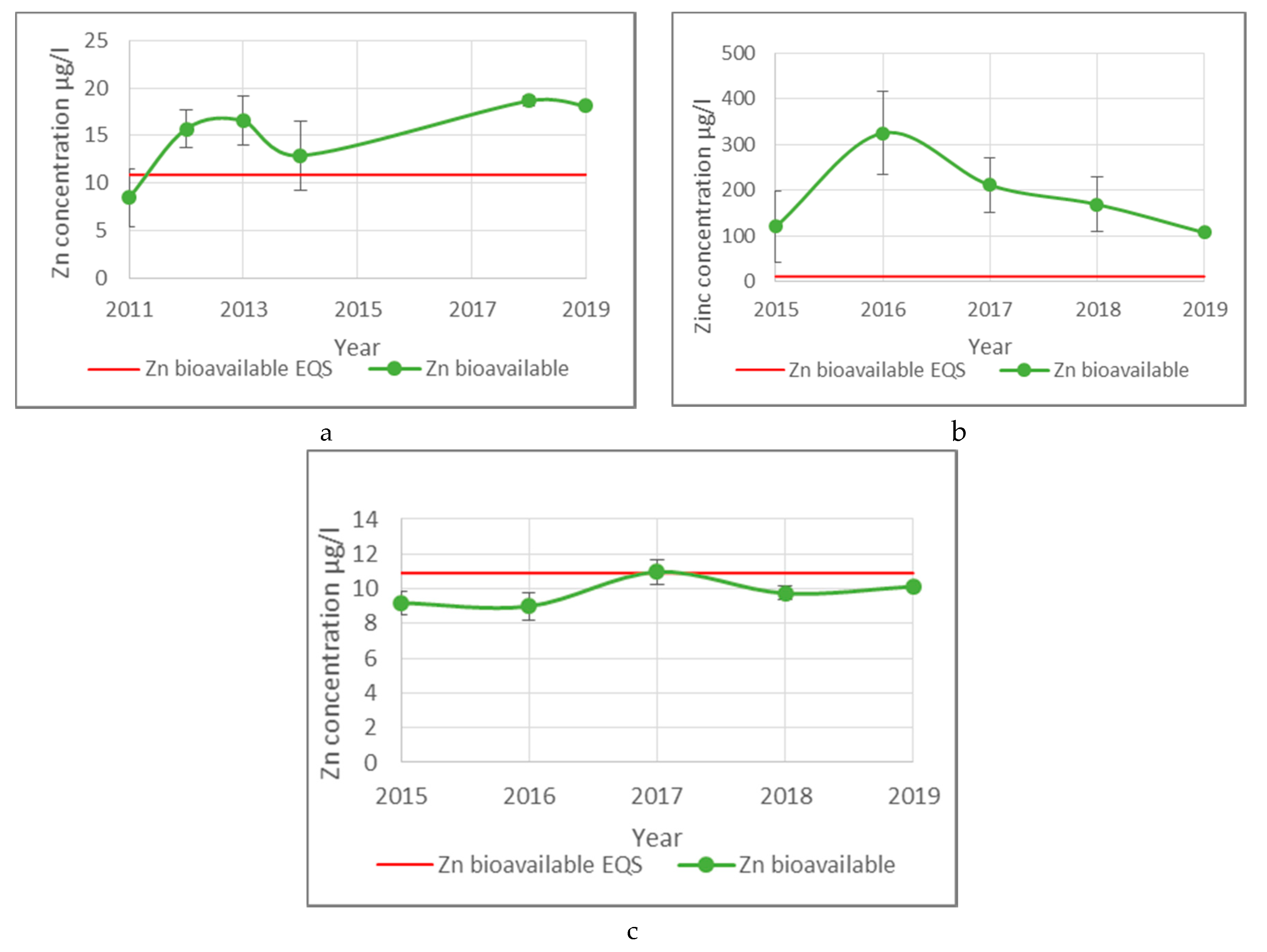
3.2. River Teign Metal Concentration Results
3.3. Real-World Application Model
4. Discussion
4.1. Pellet Removal Efficiency
4.2. Biochar Removal Efficiency
4.3. River Teign Compliance
4.4. Application to Bridford Mine
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Defra and Environment Agency (2015) South West River Basin Management Plan. Available online: https://www.gov.uk/government/publications/south-west-river-basin-district-river-basin-management-plan (accessed on 8 April 2020).
- Simons, B.; Pirrie, D.; Rollinson, G.K.; Shail, R.K. Geochemical and Mineralogical Record of the Impact of Mining on the Teign Estuary; The Ussher Society: Devon, UK, 2011. [Google Scholar]
- Akcil, A.; Koldas, S. Acid Mine Drainage (AMD): Causes, treatment and case studies. J. Clean. Prod. 2006, 14, 1139–1145. [Google Scholar] [CrossRef]
- Johnson, D.B.; Hallberg, K.B. Acid mine drainage remediation options: A review. Sci. Total Environ. 2005, 338, 3–14. [Google Scholar] [CrossRef]
- Younger, P.L.; Wolkersdorfer, C. Mining impacts on the fresh-water environment: Technical and managerial guidelines for catchment scale management. Mine Water Environ. 2004, 23, s2–s80. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, L.; Li, Y.; Li, H.; Wang, W.; Ye, B. Impacts of lead/zinc mining and smelting on the environment and human health in China. Environ. Monit. Assess. 2012, 184, 2261–2273. [Google Scholar] [CrossRef] [PubMed]
- Desaulty, A.; Petelet-Giraud, E. Zinc isotope composition as a tool for tracing sources and fate of metal contaminants in rivers. Sci. Total Environ. 2020, 138599. [Google Scholar] [CrossRef] [PubMed]
- Environment Agency Data Catchment Explorer. 2019. Available online: https://environment.data.gov.uk/catchment-planning/OperationalCatchment/3451 (accessed on 6 July 2020).
- Department for Environment Food & Rural Affairs. Water Framework Directive Implementation in England and Wales: New and Updated Standards to Protect the Water Environment. 2014. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/307788/river-basin-planning-standards.pdf (accessed on 6 July 2020).
- Comber, S.D.W.; Merrington, G.; Sturdy, L.; Delbeke, K.; Van Assche, F. Copper and zinc water quality standards under the EU Water Framework Directive: The use of a tiered approach to estimate the levels of failure. Sci. Total Environ. 2008, 403, 12–22. [Google Scholar] [CrossRef]
- Valencia-Avellan, M.; Slack, R.; Stockdale, A.; Mortimer, R.J.G. Understanding the mobilisation of metal pollution associated with historical mining in a carboniferous upland catchment. Environ. Sci. Process. Impacts 2017, 19, 1061–1074. [Google Scholar] [CrossRef]
- Kalin, M. Passive mine water treatment: The correct approach? Ecol. Eng. 2004, 22, 299–304. [Google Scholar] [CrossRef]
- Nichols, C. Efficiency of Biochar to Reduce Heavy Metal Bioavailability and Increase Ryegrass Growth: Effect of Pyrolysis Temperature, Feedstock Material, Application Rate and Earthworms. Master’s Thesis, University of Plymouth, Plymouth, UK, 2015. [Google Scholar]
- Kołodyńska, D.; Wnętrzak, R.; Leahy, J.J.; Hayes, M.H.B.; Kwapiński, W.; Hubicki, Z.J.C.E.J. Kinetic and adsorptive characterization of biochar in metal ions removal. Chem. Eng. J. 2012, 197, 295–305. [Google Scholar] [CrossRef]
- Chen, X.; Chen, G.; Chen, L.; Chen, Y.; Lehmann, J.; McBride, M.B.; Hay, A.G. Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour. Technol. 2011, 102, 8877–8884. [Google Scholar] [CrossRef]
- Enunwa, L. Effect of Biochar Feedstock Material on the Adsorption of Zinc from Contaminated Mine Water. Master’s Thesis, University of Plymouth, Plymouth, UK, 2015. [Google Scholar]
- Bhatnagar, A.; Vilar, V.J.; Botelho, C.M.; Boaventura, R.A. A review of the use of red mud as adsorbent for the removal of toxic pollutants from water and wastewater. Environ. Technol. 2011, 32, 231–249. [Google Scholar] [CrossRef] [PubMed]
- Banks, V.J.; Palumbo-Roe, B. Synoptic monitoring as an approach to discriminating between point and diffuse source contributions to zinc loads in mining impacted catchments. J. Environ. Monit. 2010, 12, 1684–1698. [Google Scholar] [CrossRef] [PubMed]
- Hill, R. An Investigation Into The Ability Of Different Red Mud Media To Adsorb Metals From Mine Adit Water To Reach Environmental Quality Standards. Master’s Thesis, University of Plymouth, Plymouth, UK, 2016. [Google Scholar]
- Comber, S. Report of Pilot Scale Feasibility Field Trial for Metal Removal at Wheal Augusta. Bridford: Devon, UK, 2015. [Google Scholar]
- Turner, A. An Assessment of the Capability of Red Mud Pellets at Adsorbing Heavy Metals, with Evaluation of its Suitability at Bridford Mine to Reduce Heavy Metal Concentrations to the River Teign. Master’s Thesis, University of Plymouth, Plymouth, UK, 2017. [Google Scholar]
- Merefield, J.R. Heavy metals in Teign Valley sediments: Ten years after. Proc. Ussher Soc. 1987, 6, 529–535. [Google Scholar]
- Wright, C. Revegetation: An Investigation of Soil Contamination and Plant Community Composition at Bridford Mine, Teign Valley, UK. Master’s Thesis, University of Plymouth, Plymouth, UK, 2017. [Google Scholar]
- RMT. Red Media Technologies, Colchester, Essex. 2020. Available online: https://www.redmediatech.com/ (accessed on 7 July 2020).
- Roberts, T. How does Biochar Influence the Mobility of Heavy Metals Lead, Zinc and Cadmium in Mine Wastes? Master’s Thesis, University of Plymouth, Plymouth, UK, 2018. [Google Scholar]
- Robinson, A. Removal Mechanisms of Trace Metals from Acid Minewaters Using Sorptive Media. Master’s Thesis, University of Plymouth, Plymouth, UK, 2019. [Google Scholar]
- Abdus-Salam, N.; Adekola, F.A. The influence of pH and adsorbent concentration on adsorption of lead and zinc on a natural goethite. Afr. J. Sci. Technol. 2005, 6. [Google Scholar] [CrossRef]
- Gupta, V.K.; Sharma, S. Removal of cadmium and zinc from aqueous solutions using red mud. Environ. Sci. Technol. 2002, 36, 3612–3617. [Google Scholar] [CrossRef]
- Ayala, J.; Fernández, B. Treatment from abandoned mine landfill leachates. Adsorption technology. J. Mater. Res. Technol. 2019, 8, 2732–2740. [Google Scholar] [CrossRef]
- De Schamphelaere, K.A.; Lofts, S.; Janssen, C.R. Bioavailability models for predicting acute and chronic toxicity of zinc to algae, daphnids, and fish in natural surface waters. Environ. Toxicol. Chem. Int. J. 2005, 24, 1190–1197. [Google Scholar] [CrossRef]
- Balafrej, H.; Bogusz, D.; Triqui, Z.E.A.; Guedira, A.; Bendaou, N.; Smouni, A.; Fahr, M. Zinc Hyperaccumulation in plants: A review. Plants 2020, 9, 562. [Google Scholar] [CrossRef]
- Bowell, R.J.; Bruce, I. Geochemistry of iron ochres and mine waters from Levant Mine, Cornwall. Appl. Geochem. 1995, 10, 237–250. [Google Scholar] [CrossRef]
- O’Connor, D.; Peng, T.; Zhang, J.; Tsang, D.C.; Alessi, D.S.; Shen, Z.; Bolan, N.S.; Hou, D. Biochar application for the remediation of heavy metal polluted land: A review of in situ field trials. Sci. Total Environ. 2018, 619, 815–826. [Google Scholar] [CrossRef]
- Puga, A.P.; Abreu, C.A.; Melo, L.C.A.; Paz-Ferreiro, J.; Beesley, L. Cadmium, lead, and zinc mobility and plant uptake in a mine soil amended with sugarcane straw biochar. Environ. Sci. Pollut. Res. 2015, 22, 17606–17614. [Google Scholar] [CrossRef] [PubMed]
- Fellet, G.; Marchiol, L.; Delle Vedove, G.; Peressotti, A. Application of biochar on mine tailings: Effects and perspectives for land reclamation. Chemosphere 2011, 83, 1262–1267. [Google Scholar] [CrossRef] [PubMed]
- Kurniawan, T.A.; Chan, G.Y.; Lo, W.H.; Babel, S. Comparisons of low-cost adsorbents for treating wastewaters laden with heavy metals. Sci. Total Environ. 2006, 366, 409–426. [Google Scholar] [CrossRef] [PubMed]

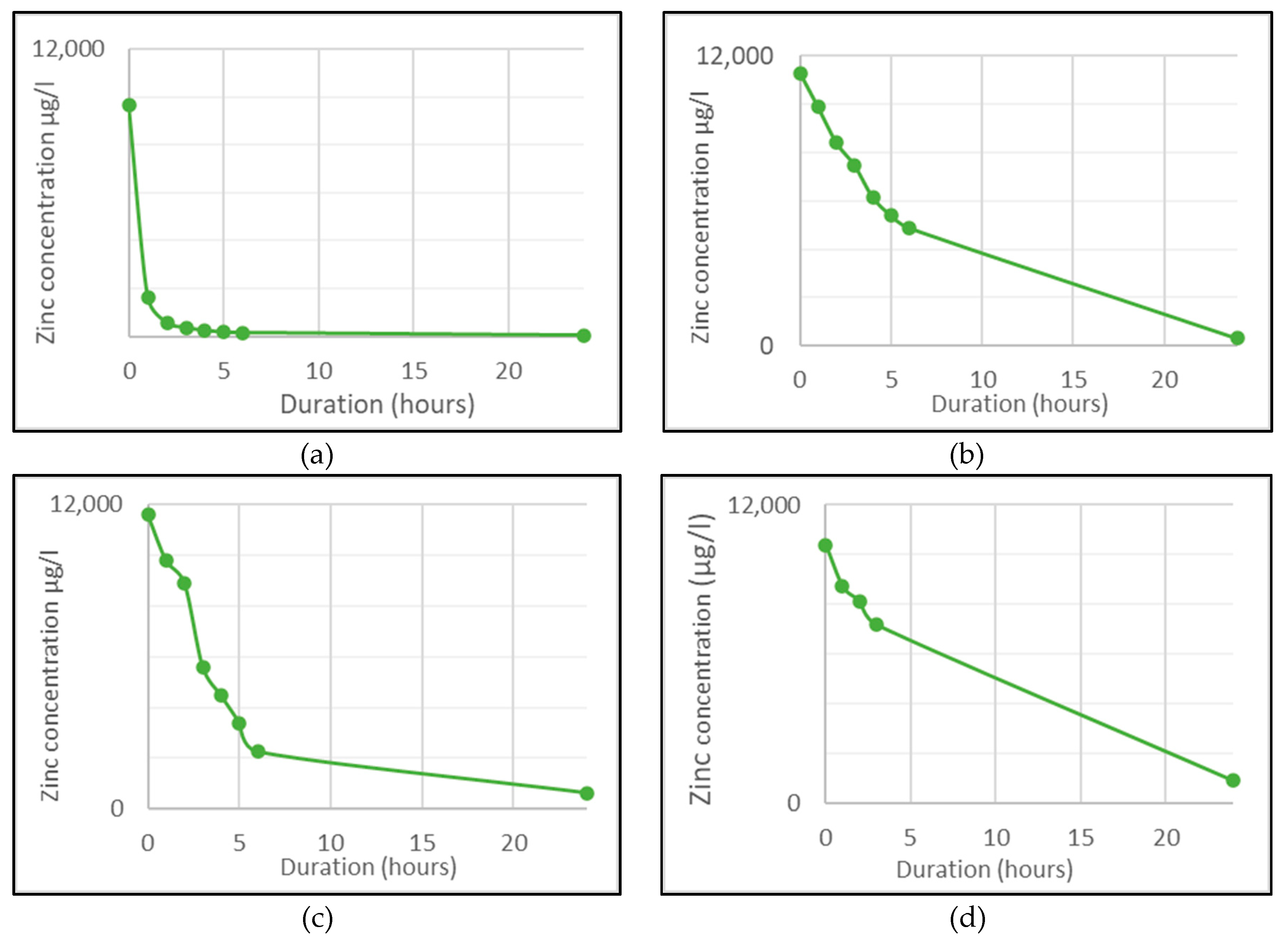
| Duration | pH of Mine Adit Water | |||
|---|---|---|---|---|
| Compressed Pellets (CP) | Fired Pellets (FP) | Fired Acid Etched Pellets (FAE) | Powdered Pellets (PP) | |
| Start of Experiment (0 h) | 4.65 | 4.65 | 3.78 | 4.59 |
| End of Experiment | 7.80 (53 h) | 9.33 (53 h) | 5.5 (6 h) | 8.84 (24 h) |
| Hours | Removal Efficiency for Zinc (%) | |||
|---|---|---|---|---|
| Compressed | Fired | FAE | Powdered | |
| 2 | 73.7 | 22.0 | 25.6 | 23.3 |
| 24 | 99.5 | 99.3 | 97.2 | 94.7 |
| 53 | 99.8 | 99.9 | - | - |
| Highest Adsorption Capacity Reached | Adsorption Capacity of Pellets (mg/kg) | ||
|---|---|---|---|
| Zn | Cd | Pb | |
| Compressed Pellet | >105.6 | >1.1 | >5.36 |
| Fired Pellet | >150 | >1.56 | >3.89 |
| Field-Scale Trial (Compressed Pellet) | 8743 | 35.40 | 2089 |
| Treatment Method | Tonnes/yr Required (Assuming 100% Efficiency) | Tonnes/yr Required Based on Removal Efficiencies from This Study | Cost |
|---|---|---|---|
| Compressed Pellet (CP) | 138 | 383 (36% efficiency after 3 months) | £34,067 a year |
| Fired Pellet (FP) | 8064 | 8064 (99.9% efficiency after 53 h) | £717,292 a year |
| Agricultural Biochar | 110 | 137.5 (80% efficiency after 2 h) | £12,230 |
| Wood Biochar | 3056 | 15,280 (20% efficiency after 2 h) | £1,359,156 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jordan, A.; Hill, R.; Turner, A.; Roberts, T.; Comber, S. Assessing Options for Remediation of Contaminated Mine Site Drainage Entering the River Teign, Southwest England. Minerals 2020, 10, 721. https://doi.org/10.3390/min10080721
Jordan A, Hill R, Turner A, Roberts T, Comber S. Assessing Options for Remediation of Contaminated Mine Site Drainage Entering the River Teign, Southwest England. Minerals. 2020; 10(8):721. https://doi.org/10.3390/min10080721
Chicago/Turabian StyleJordan, Abigail, Rachel Hill, Adrienne Turner, Tyrone Roberts, and Sean Comber. 2020. "Assessing Options for Remediation of Contaminated Mine Site Drainage Entering the River Teign, Southwest England" Minerals 10, no. 8: 721. https://doi.org/10.3390/min10080721
APA StyleJordan, A., Hill, R., Turner, A., Roberts, T., & Comber, S. (2020). Assessing Options for Remediation of Contaminated Mine Site Drainage Entering the River Teign, Southwest England. Minerals, 10(8), 721. https://doi.org/10.3390/min10080721






