Spontaneous Symmetry-Breaking in the Corrosion Transformation of Ancient Bronzes
Abstract
1. Introduction
2. Experimental
3. Results
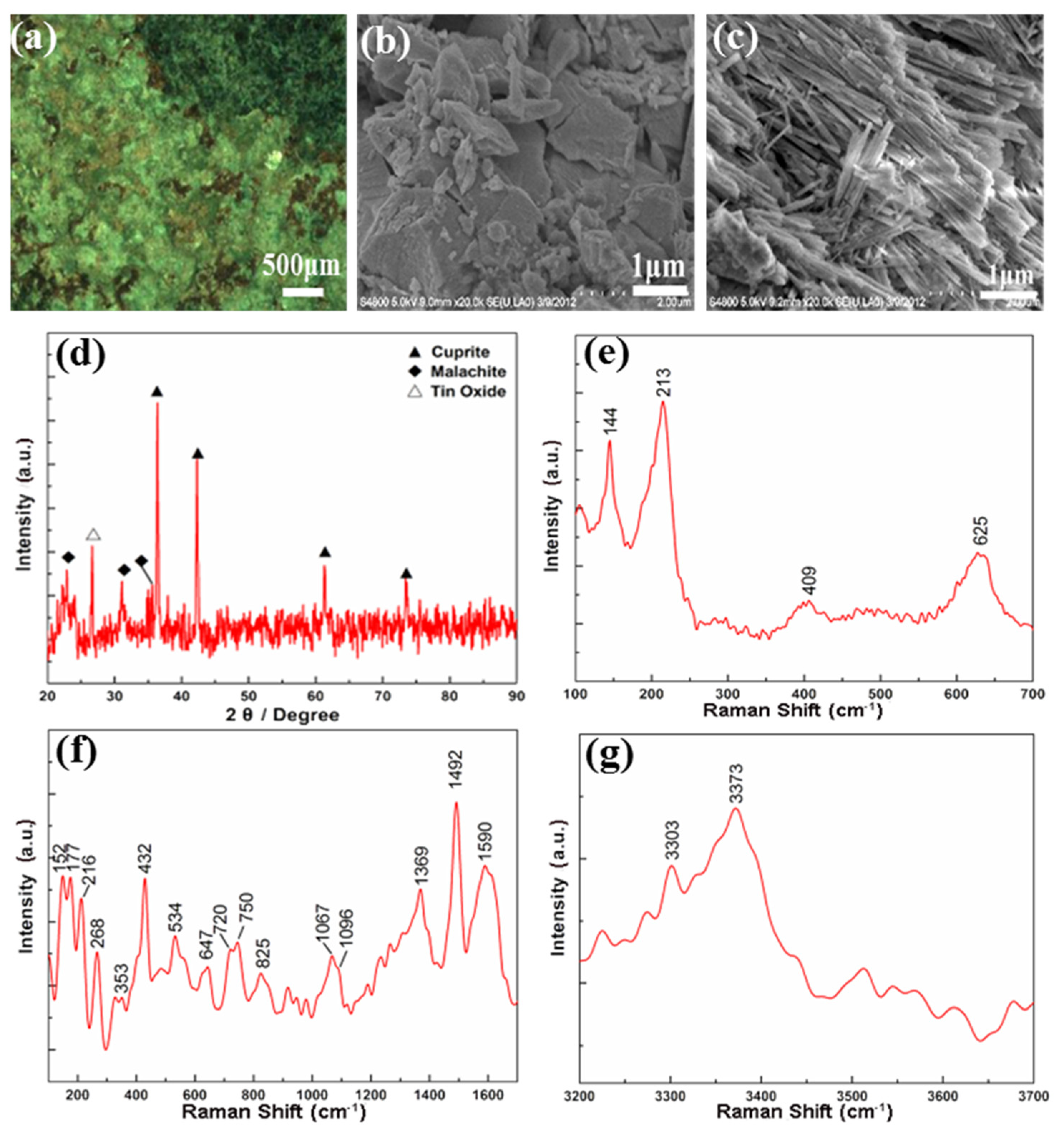
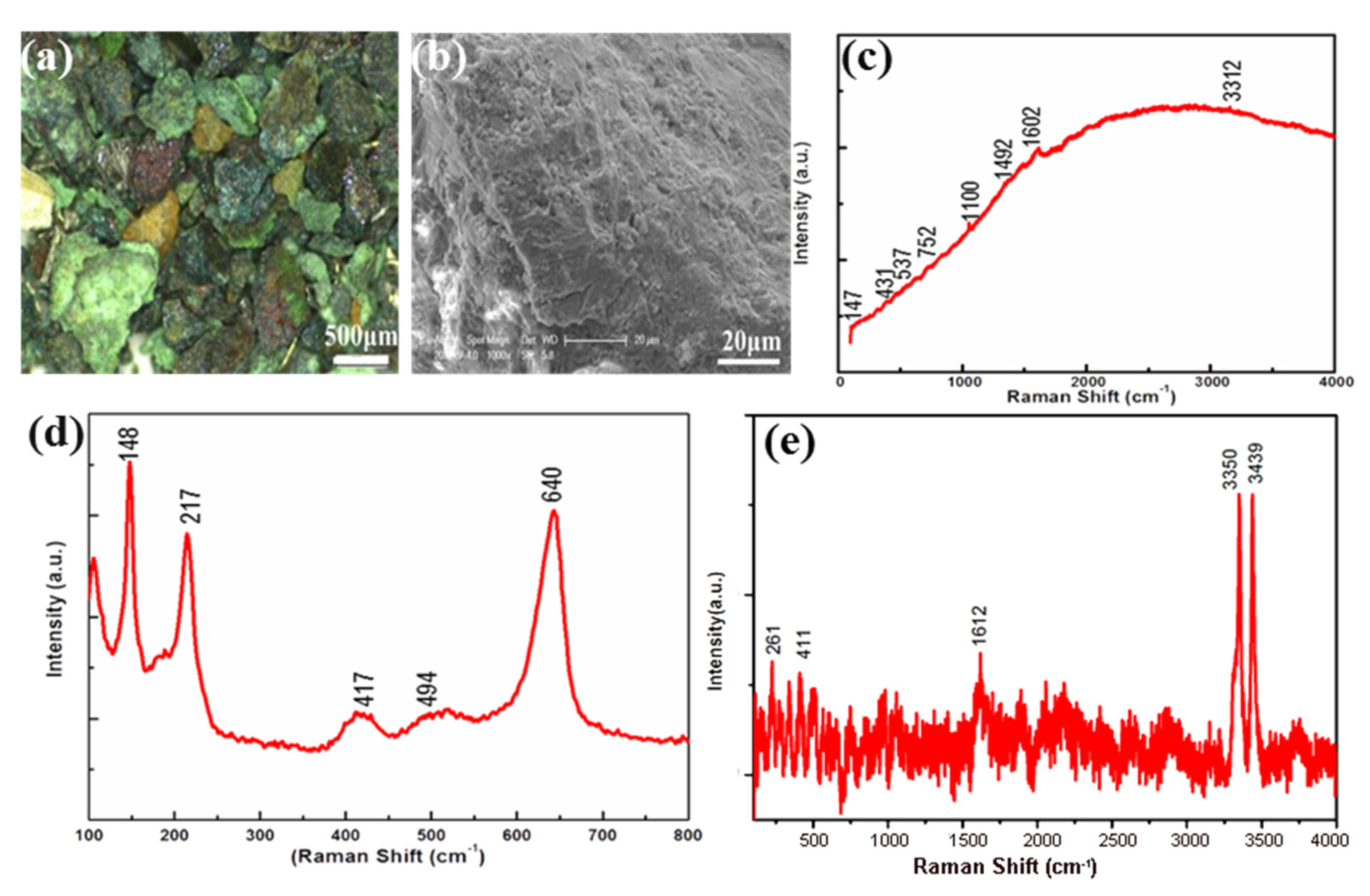
3.1. Corrosions on the Bronze Vase (Gu)
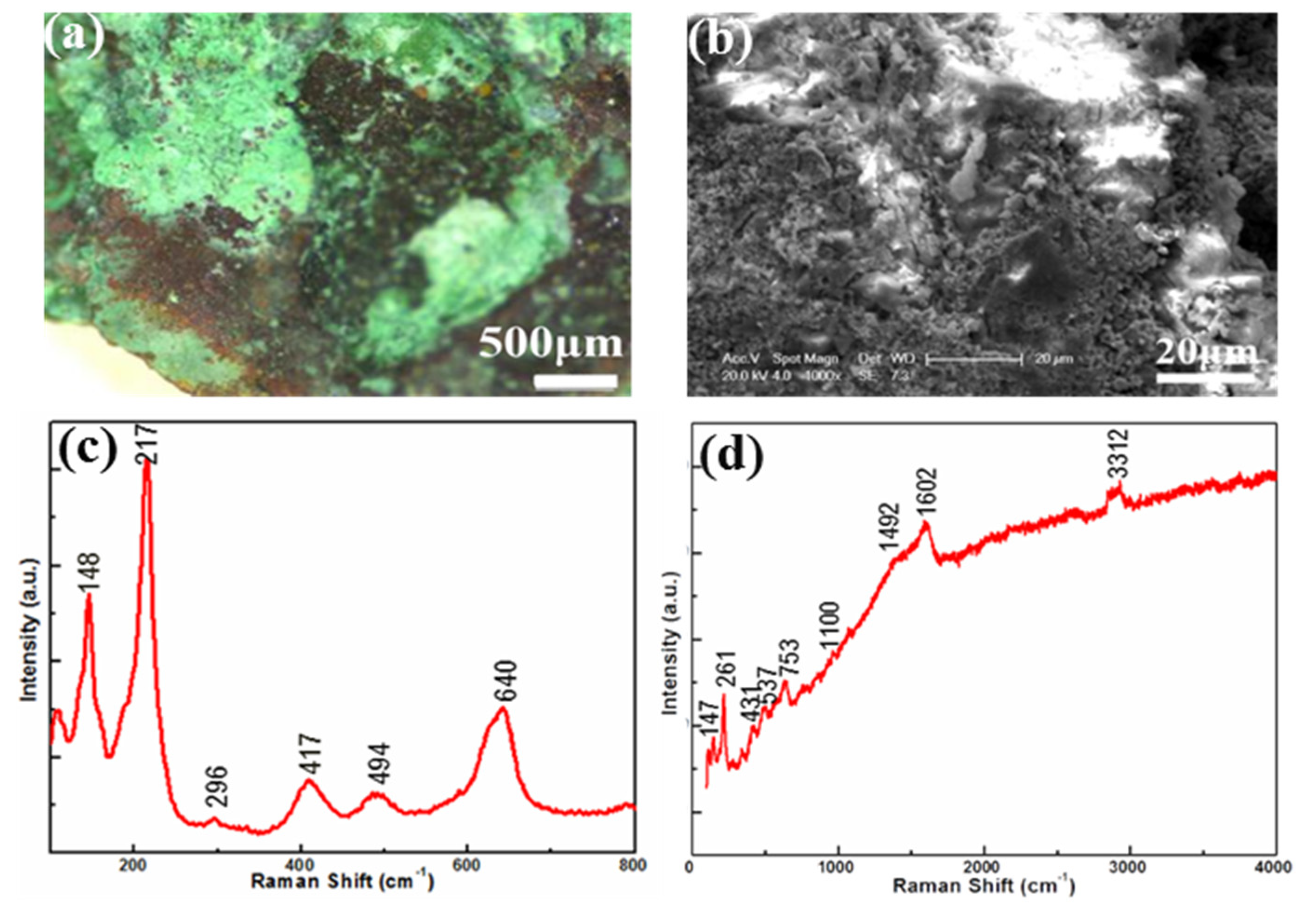
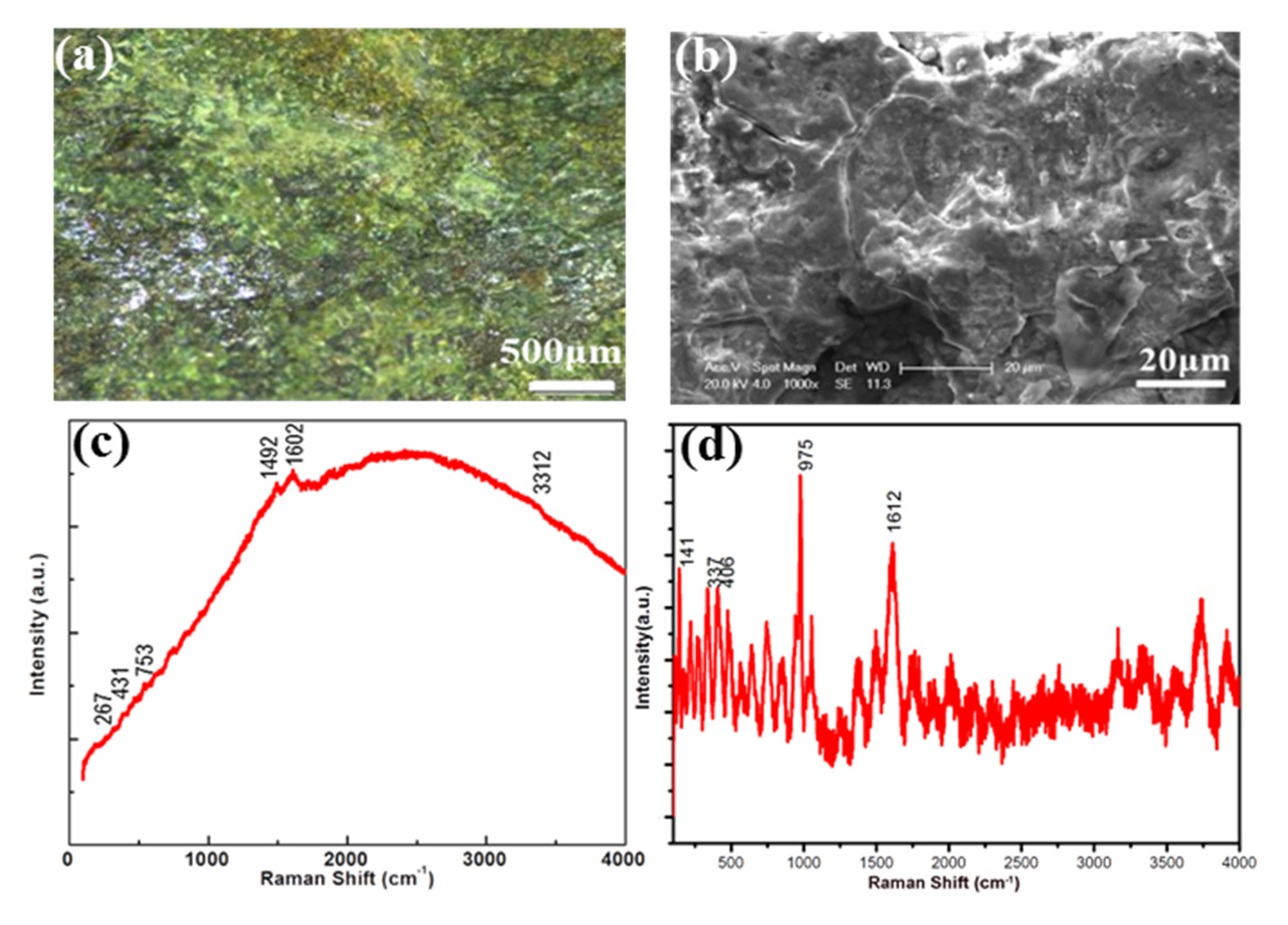
3.2. Corrosions on the Bronze Goblet (Jue Bei)
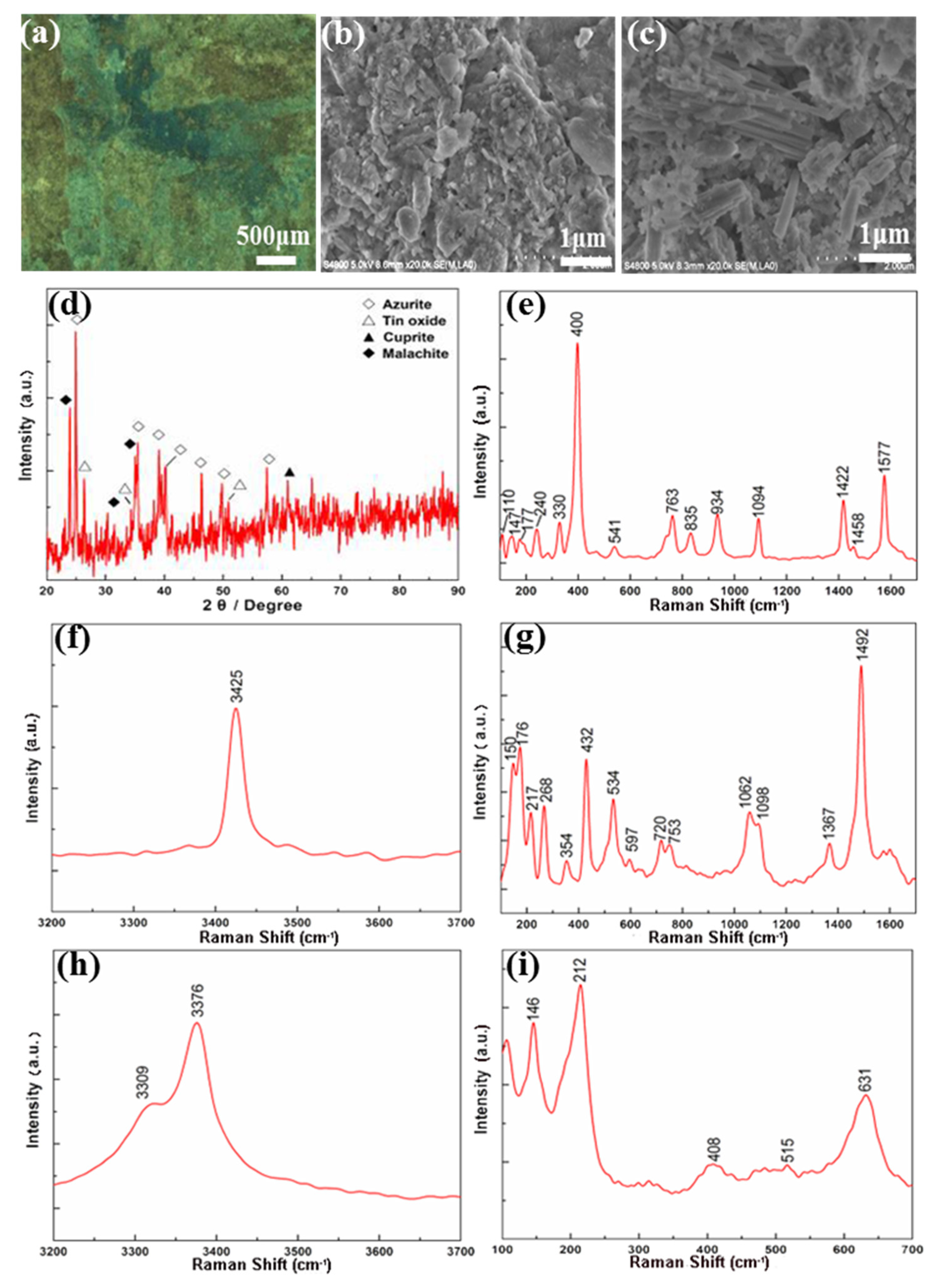
3.3. Corrosions on the Bronze Basin (Fu)
3.4. Corrosions on the Bronze Vessel (Ding)
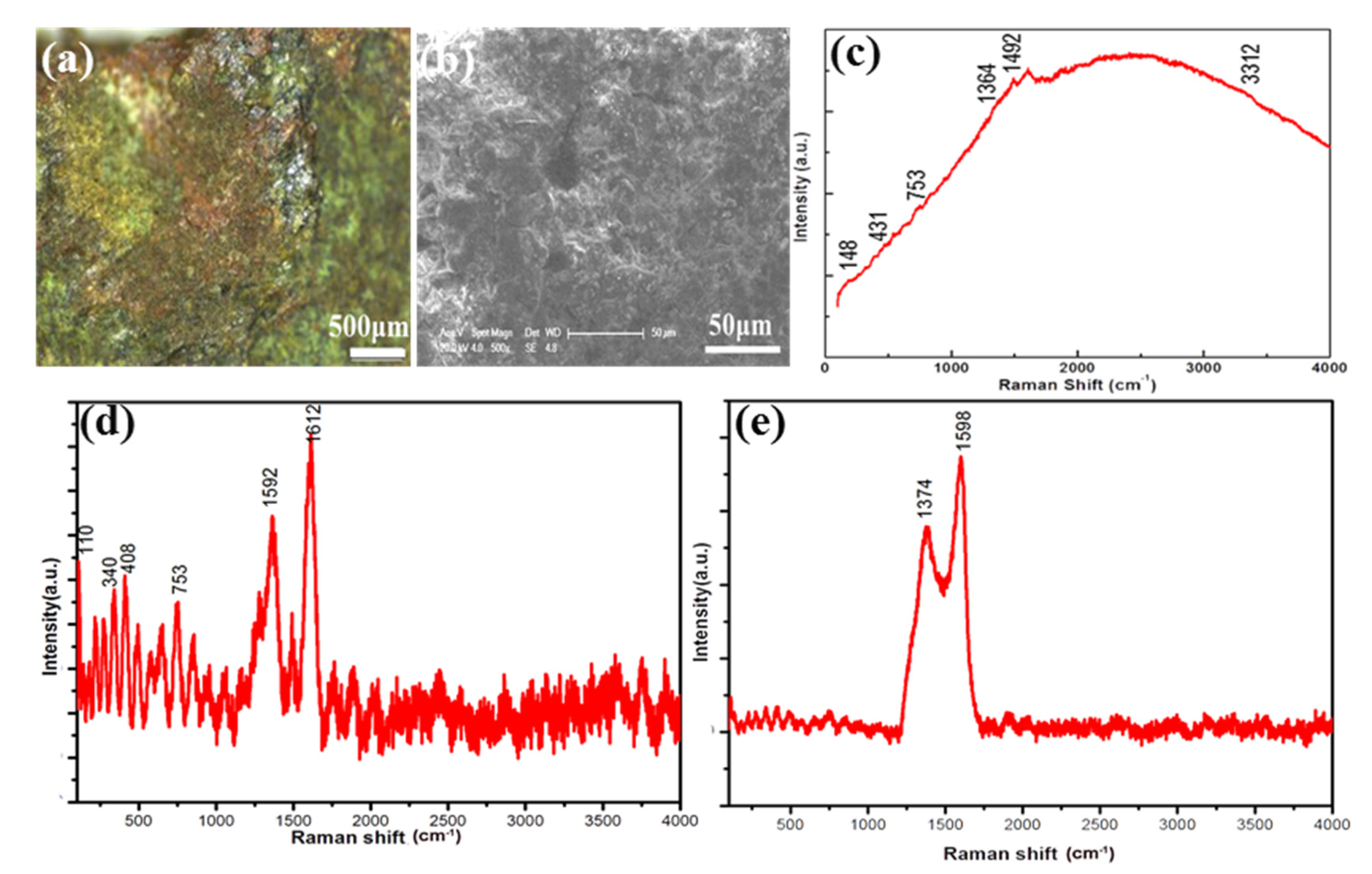
3.5. Corrosions on the Bronze Sword
3.6. Corrosions on the Bronze Bowl (Bo)
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liao, Y. Research on erosion mechanism of bronze cultural relics and transform agent on harmful rust. Sci. Conserv. Archaeol. 2003, 15, 20–23. [Google Scholar]
- Scott, D.A. Bronze disease: A review of some chemical problems and the role of relative humidity. J. Am. Inst. Conserv. 1990, 29, 193–206. [Google Scholar] [CrossRef]
- Ingo, G.M.; Caro, T.D.; Riccucci, C.; Angelini, E.; Grassini, S.; Balbi, S.; Bernardini, P.; Salvi, D.; Bousselmi, L.; Cilingiroglu, A.; et al. Large scale investigation of chemical composition, structure and corrosion mechanism of bronze archeological artefacts from Mediterranean basin. Appl. Phys. A 2006, 83, 513–520. [Google Scholar] [CrossRef]
- Bosi, C.; Garagnani, G.L.; Imbeni, V.; Martini, C.; Mazzeo, R.; Poli, G. Unalloyed copper inclusion in ancient bronze artifacts. J. Mater. Sci. 2002, 37, 4285–4298. [Google Scholar] [CrossRef]
- Robbiola, L.; Blengino, J.M.; Fiaud, C. Morphology and mechanisms of formation of natural patinas on archaeological Cu-Sn alloys. Corros. Sci. 1998, 40, 2083–2111. [Google Scholar] [CrossRef]
- Feng, S.B.; Hu, F.H.; Feng, L.T. Research status of corrosion of bronze wares. Corros. Prot. 2009, 30, 7–10. [Google Scholar]
- Frankel, G.S. Pitting corrosion of metals: A review of the critical factors. J. Electrochem. Soc. 1998, 145, 2186–2198. [Google Scholar] [CrossRef]
- Edwards, M.; Ferguson, J.F.; Reiber, S.H. The pitting corrosion of copper. J. Am. Water Work. Assoc. 1994, 86, 74–90. [Google Scholar] [CrossRef]
- Zhang, X.M.; Yuan, S.X.; Liu, Y.; Zhou, B.Z. Research on the corrosion of bronzes from Zhouyuan Site and Yu State cemeteries. Sci. Conserv. Archaeol. 1999, 11, 7–18. [Google Scholar]
- Galvele, J.R.; Dedemicheli, S.M. Mechanism of intergranular corrosion of Al-Cu alloys. Corros. Sci. 1970, 10, 795–807. [Google Scholar] [CrossRef]
- Culpan, E.A.; Foley, A.G. The detection of selective phase corrosion in cast nickel aluminium bronze by acoustic emission techniques. J. Mater. Sci. 1982, 17, 953–964. [Google Scholar] [CrossRef]
- Ingo, G.M.; Caro, T.D.; Riccucci, C.; Khosroff, S. Uncommon corrosion phenomena of archaeological bronze alloys. Appl. Phys. A 2006, 83, 581–588. [Google Scholar] [CrossRef]
- Kosec, T.; Ćurković, H.O.; Legat, A. Investigation of the corrosion protection of chemically and electrochemically formed patinas on recent bronze. Electrochim. Acta 2010, 56, 722–731. [Google Scholar] [CrossRef]
- Couture-Rigert, D.E.; Sirois, P.J.; Moffatt, E.A. An investigation into the cause of corrosion on indoor bronze sculpture. Stud. Conserv. 2012, 57, 142–163. [Google Scholar] [CrossRef]
- Popkov, V.; Evans, M.R.; Mukamel, D. Spontaneous symmetry breaking in a bridge model fed by junctions. J. Phys. A-Math Theor. 2008, 41, 1751–8113. [Google Scholar] [CrossRef]
- Großkinsky, S.; Schütz, G.M.; Willmann, R.D. Rigorous Results on Spontaneous Symmetry Breaking in a One-Dimensional Driven Particle System. J. Stat. Phys. 2007, 128, 587–606. [Google Scholar] [CrossRef][Green Version]
- Yao, N.Y.; Potter, A.C.; Potirniche, I.D.; Vishwanath, A. Discrete Time Crystals: Rigidity, Criticality, and Realizations. Phys. Rev. Lett. 2017, 118, 030401. [Google Scholar] [CrossRef]
- Chen, S.P.; Wang, C.F.; Zhou, H.T.; Tan, Y.H.; Wen, H.R.; Tang, Y.Z. Symmetry breaking and switchable thermal dielectric behaviors triggered by order-disorder phase transition in a neutral co-crystallized organic adduct. Chem. Phys. Lett. 2018, 715, 45–50. [Google Scholar] [CrossRef]
- Frost, R.L.; Martens, W.N.; Rintoul, L.; Mahmutagic, E.; Kloprogge, J.T. Raman spectroscopic study of azurite and malachite at 298 and 77 K. J. Raman Spectrosc. 2002, 33, 252–259. [Google Scholar] [CrossRef]
- Bersani, D.; Lottici, P.P. Raman spectroscopy of minerals and mineral pigments in archaeometry. J. Raman Spectrosc. 2016, 47, 499–530. [Google Scholar] [CrossRef]
- Wu, T.T.; Meng, W.W.; Bao, Z.R.; Li, Y.; Pan, C.X. Study of synthesizing malachite corrosion product on the bronze surface in excessive CO2 atmosphere environment. J. Chin. Soc. Corr. Prot. 2014, 34, 82–88. (In Chinese) [Google Scholar]
- Anthony, J.W.; Bideaux, R.A.; Bladh, K.W.; Nichols, M.C. Handbook of Mineralogy; Mineralogical Society of America: Chantilly, VA, USA, 1990; Available online: http://www.handbookofmineralogy.org/ (accessed on 5 October 2019).
- Wagman, D.D.; Evans, W.H.; Parker, V.B.; Schumm, R.H.; Halow, I.; Bailey, S.M.; Churney, K.L.; Nuttal, R.L. The NBS tables of chemical thermodynamic properties. J. Phys. Chem. Ref. Data 1982, 11, 1807–1812. [Google Scholar]
- MacLeod, I.D. Bronze disease: An electrochemical explanation. ICCM Bull. 1981, 7, 16–26. [Google Scholar] [CrossRef]
- He, L.; Liang, J.Y.; Zhao, X.; Jiang, B.L. Corrosion behavior and morphological features of archeological bronze coins from ancient China. Microchem. J. 2011, 99, 203–212. [Google Scholar] [CrossRef]

| Name | Collection Number in the Museums | Period | Burial Conditions | Sampling Position |
|---|---|---|---|---|
| Bronze vase (Gu) | ABG-2209-1 | Shang Dynasty (1600 BC–1046 BC) | Soil | Mouth edge, as shown in Figure 1a |
| Bronze goblet (Jue Bei) | 3253 | Shang Dynasty (1600 BC–1046 BC) | Tomb | Mouth edge, as shown in Figure 1b |
| Bronze basin (Fu) | ABF-5155 | Spring and Autumn Period (770 BC–476 BC) | Soil | Mouth edge, as shown in Figure 1c |
| Bronze vessel (Ding) | 1233 | Western Han Dynasty (202 BC–25 AD) | Soil | Bottom, as shown in Figure 1d |
| Bronze sword | 1938 | Western Han Dynasty (202 BC–25 AD) | Tomb | Blade tip, as shown in Figure 1e |
| Bronze bowl (Bo) | 3668 | Tang Dynasty (618 AD–907 AD) | Tomb | Mouth edge, as shown in Figure 1f |
| Sample Number | Primary Corrosion | Secondary Corrosion | |
|---|---|---|---|
| 1 | Bronze vase (Gu) (No.: ABG-2209-1) | cuprite (cubic), cassiterite (tetrahedral) and amorphous mixtures | malachite (monoclinic) |
| 2 | Bronze goblet (Jue Bei) (No.: 3253) | cuprite (cubic) | malachite (monoclinic) |
| 3 | Bronze basin (Fu) (No.: ABF-5155) | cuprite (cubic), cassiterite (tetrahedral) and malachite (monoclinic) | azurite (monoclinic) |
| 4 | Bronze vessel (Ding) (No.: 1233) | malachite (monoclinic) and carbon | azurite (monoclinic) |
| 5 | Bronze sword (No.: 1938) | cuprite (cubic) and malachite (monoclinic) | atacamite (orthohombic) |
| 6 | Bronze bowl (Bo) (No.: 3668) | malachite (monoclinic) | brochantite (monoclinic) |
| Name | Molecular Formula | Chemical Composition | Crystal System (Lattice Type) | Space Group | Cell Data | |
|---|---|---|---|---|---|---|
| Copper | Cu | Cu | Cubic (face-centered cubic lattice) | Fm 3m (#225) | a = 3.615 Å Z = 4 | 0 |
| Cuprite | Cu2O | Cu: 88.82% | Cubic (Simple cubic lattice) | Pn 3m (#224) | a = 4.2685 Å Z = 2 | −146.0 |
| Cassiterite | SnO2 | SnO2: 99.60%, Cu: 0.0014% | Tetrahedral (Simple tetragonal lattice) | P42/mnm (#136) | a = b = 4.7382 Å c = 3.1871 Å Z = 2 | −515.8 |
| Atacamite (harmful corrosion) | CuCl2·3Cu(OH)2 | Cu: 59.51%, H: 1.42%, Cl: 22.47%, O: 16.60% | Orthohombic (Simple orthorhombic lattice) | Pnam (#62) | a = 6.030 Å b = 9.120 Å c = 6.865 Å Z = 4 | −1339.5 |
| Malachite | CuCO3·Cu(OH)2 | CuO: 71.95%, CO2: 19.90%, H2O: 8.15% | Monoclinic (Simple monoclinic lattice) | P21/c (#14) | a = 9.502 Å b = 11.974 Å c = 3.240 Å β = 98°45′ Z = 4 | −893.6 |
| Azurite | 2(CuCO3)·Cu(OH)2 | CuO: 69.24%, CO2: 25.53%, H2O: 5.23% | Monoclinic (Simple monoclinic lattice) | P21/c (#14) | a = 5.0109 Å b = 5.8485 Å c = 10.345 Å β = 92.43° Z = 2 | −1315.5 |
| Brochantite | CuSO4·3Cu(OH)2 | Cu: 56.20%, H: 1.34%, S: 7.09%, O: 35.37% | Monoclinic (Simple monoclinic lattice) | P21/c (#14) | a = 6.02 Å b = 9.85 Å c = 13.08 Å β = 103°22′ Z = 4 | −1817.7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.; Cao, X.; Li, Y.; Wang, Z.; Li, B.; Jiang, X.; Jia, J.; Pan, C. Spontaneous Symmetry-Breaking in the Corrosion Transformation of Ancient Bronzes. Minerals 2020, 10, 656. https://doi.org/10.3390/min10080656
Yang Y, Cao X, Li Y, Wang Z, Li B, Jiang X, Jia J, Pan C. Spontaneous Symmetry-Breaking in the Corrosion Transformation of Ancient Bronzes. Minerals. 2020; 10(8):656. https://doi.org/10.3390/min10080656
Chicago/Turabian StyleYang, Yanpeng, Xiaojuan Cao, Yang Li, Zhongchi Wang, Bingjie Li, Xudong Jiang, Junji Jia, and Chunxu Pan. 2020. "Spontaneous Symmetry-Breaking in the Corrosion Transformation of Ancient Bronzes" Minerals 10, no. 8: 656. https://doi.org/10.3390/min10080656
APA StyleYang, Y., Cao, X., Li, Y., Wang, Z., Li, B., Jiang, X., Jia, J., & Pan, C. (2020). Spontaneous Symmetry-Breaking in the Corrosion Transformation of Ancient Bronzes. Minerals, 10(8), 656. https://doi.org/10.3390/min10080656






