1. Introduction
Froth flotation has underpinned the continued economic production of coal and minerals since its discovery 100 years ago and, arguably, is the most important method of fine particle beneficiation today [
1]. The importance of the process has stemmed from its ability to process low-grade feeds more economically than any other separation technology [
2]. However, today, new challenges are arising due to the ongoing decline in feed quality and the increasing need to recover finer particles.
Attempts have been made to adapt the flotation process to meet the challenge of declining grades. For example, the volume of the conventional mechanical cells has been increased progressively over the years to achieve sufficient economy of scale, reaching over 600 m
3 in recent years. These large cells reflect the fact that flotation superficial velocities remain very low at only ~1 cm/s. Advances have nevertheless been made to improve the bubbles–particles adhesion kinetics [
3,
4]. However, flotation kinetics remains relatively poor for particles finer than 10 µm. Moreover, as feed quality continues to decline there will be a need to consider using ever larger flotation cells, though in reality this may not be feasible. This means that new, innovative methods will be needed to address these challenges.
We have been developing a new approach for recovering and concentrating hydrophobic particles, inspired by conventional oil agglomeration [
5]. Conventional oil agglomeration disperses oil droplets that selectively wet and agglomerate hydrophobic particles. Although generally applied to coal, the process has also been applied to mineral systems [
6,
7,
8]. Although the conventional approach was often highly effective, the process was not economically viable due to the significant oil consumption and hence prohibitive operating cost.
Here, a selective agglomeration process utilizing a high internal phase emulsion binder [
9] replaced the pure oil used in conventional oil agglomeration. The viscous emulsion binder consists of 95% aqueous solution dispersed within 5% organic liquid (including the emulsifier). The tightly packed droplets of water are separated by a thin continuous layer of oil. Thus, the binder is hydrophobic, while consisting primarily of water. Therefore, the emulsion functions like an oil, collecting and agglomerating only the hydrophobic particles in a suspension. However, as the emulsion consists primarily of water, the level of organic liquid required is reduced by an order of magnitude [
10] compared with the level required for conventional oil agglomeration. For example, it has been demonstrated that ultrafine coal tailings can be agglomerated using 1–2 wt% organic liquid [
11]. Of course, this value is still relatively high compared with the dose required in flotation; however, the application of the binder has other advantages.
In previous work by van Netten and co-workers, it was demonstrated that the agglomeration process is very fast, requiring only a few seconds in a batch system and less than a second in a continuous plug flow system [
9,
12]. Furthermore, the kinetics appears to be independent of the particle size, with particles much finer than 10 microns recovered in a few seconds [
10]. Product moisture is also lower [
13].
Thus far, the agglomeration process using the emulsion binder has been applied successfully to coal, silica, magnetite and graphite, with a suitable collector applied when it is necessary to make the particles hydrophobic [
9,
10,
14]. Moreover, fundamental studies of the behavior of the emulsion have demonstrated that the oil films within the emulsion are permeable and that water is transported into the emulsion during the agglomeration process [
10] via osmosis. Indeed, it is this permeability that is thought to account for the ability of the emulsion to recover ultrafine particles (even below 1 μm [
10]), eliminating the viscous resistance to thin film drainage of water between the particle and binder. Indeed, Davis and Zinchenko [
15] conducted a fundamental theoretical study, showing that permeable drops offer negligible hydrodynamic resistance to particle collection.
Originally, a batch system consisting of a high-speed blender was used. In order to establish a simpler industrial system, an experiment was undertaken using a continuous plug flow system, delivering promising results [
11,
12].
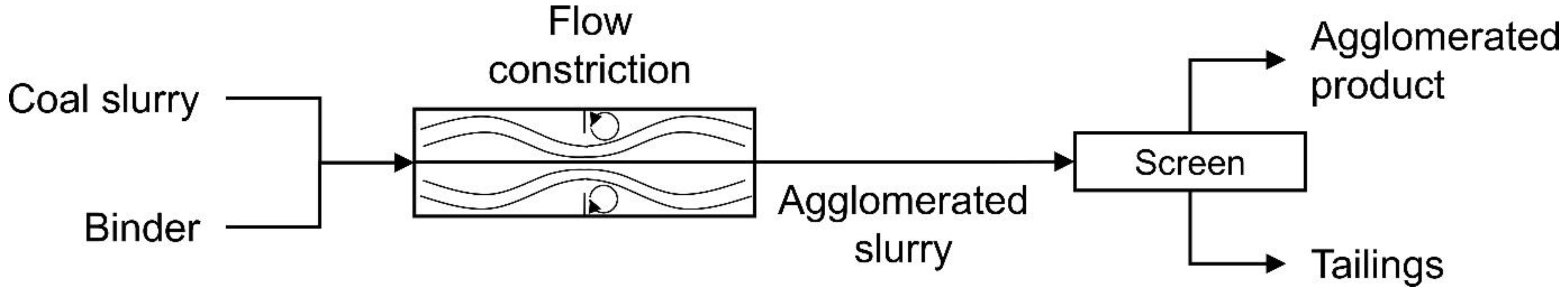
Figure 1 presents a simplified representation of the process. As shown in the figure, the high shear zone was created using a flow constriction and a mechanical screen was used to recover the agglomerates from the hydrophilic tailings. The plug flow approach appeared to perform significantly better than the previous high-speed blender.
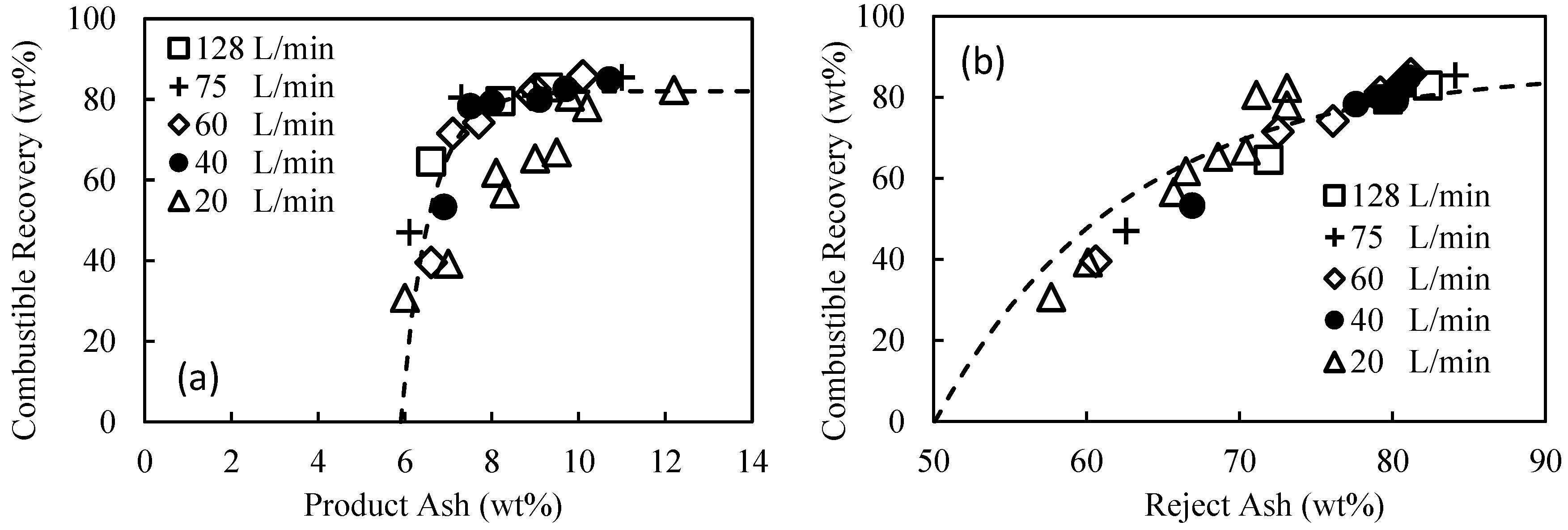
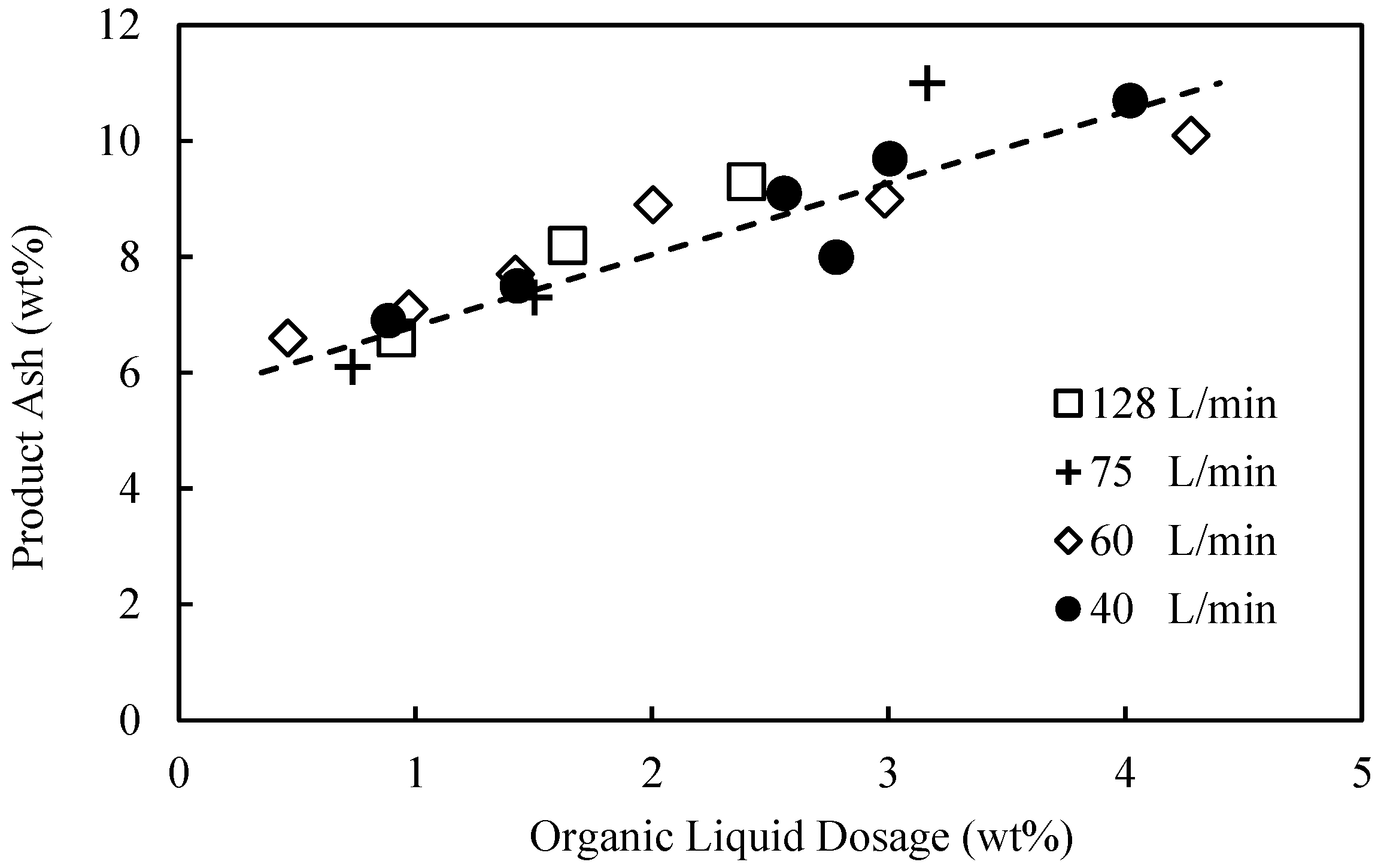
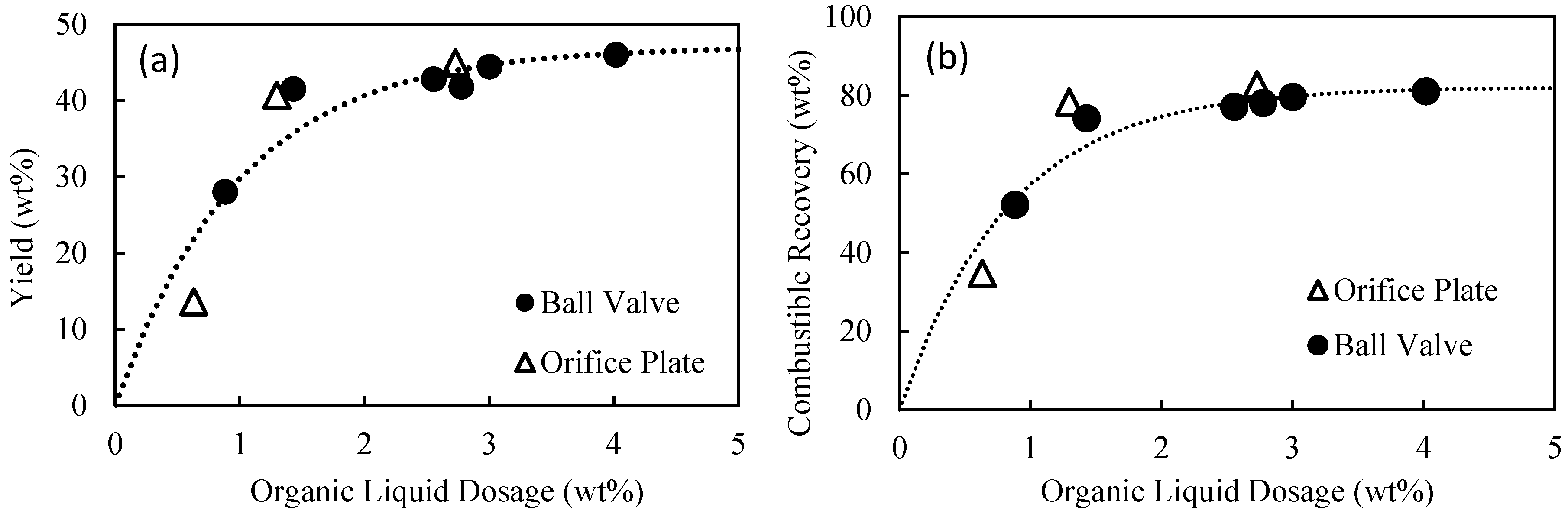
In the current study, for the first time, a detailed and systematic investigation into the influence of flowrate and pressure drop on the plug flow agglomeration was undertaken, with a strong focus on hydrophobic particle recovery, selectivity, and binder performance. The performance of the binder was measured by determining the minimum dosages required to achieve the maximum combustible recovery, with lower dosages equating to better performance. The feed used in the investigation was a hydrocyclone overflow, typically sent to the tailings thickener. This paper presents an extensive set of results covering flowrates from 20 to 128 L/min, detailing the pressure drop across the flow constriction used to quantify the degree of shear. The effect of constriction geometry was also investigated, with an orifice plate used in place of the ball valve in a series of experiments.
This work is significant because it clearly demonstrates the remarkable speed of hydrophobic interactions in an applied sense, made possible through the application of a permeable hydrophobic binder and a plug flow process. There is an industrial need for improved kinetics in fine particle recovery, given the known limitations observed in froth flotation. The plug flow agglomeration demonstrates the potential for vastly faster processing rates than have ever been realized, and is a possible solution to this problem. This novel and highly applied investigation should therefore inspire further fundamental work to build a clearer framework for further advancement.
2. Materials and Methods
2.1. Emulsion Binder
The composition of the emulsion binder was 2.5% kerosene, 2.5% emulsifier and 95% aqueous salt solution. This composition was shown to produce the best performance in previous work [
9]. The kerosene and emulsifier, which formed the continuous phase, were sourced from Recochem Inc., Australia Division (Brisbane, QLD, Australia) and VicChem Pty Ltd. (Coolaroo, VIC, Australia), respectively. The emulsifier was an industrial-grade mixture consisting of 60% sorbitan monooleate and 40% glycerol monooleate, referred to as SMO. The aqueous salt solution that formed the dispersed phase of the emulsion was prepared by dissolving 3 wt% NaCl, sourced from Cerebos Ltd. (Preston, VIC, Australia), in tap water. It must be noted that the salt level was not optimized; hence, much lower levels would ultimately be used.
The high internal phase emulsion binder was prepared immediately prior to an experiment in 2 kg batches. To prepare the emulsion binder, the organic constituents were placed in a stainless-steel mixing bowl and mixed using a hand-held Russell Hobbs mixer (RHMX1, Spectrum Brands Austalia Pty Ltd., Braeside, VIC, Australia) set at maximum speed. The aqueous salt solution was then added to the organic mixture in an incremental manner and subjected to continued mixing. The aqueous solution was added incrementally, at approximately 5 g/s, to ensure that it formed the dispersed phase; adding the solution all at once would have resulted in the oil phase dispersing within the water to form a dilute emulsion. Thus, it was critical to add the aqueous phase gradually. The total time required for the addition of the aqueous solution was 6 min; however, the mixing continued for a total of 10 min. If prepared correctly, the emulsion appeared as a white and very viscous gel.
Prior to an experiment, the prepared emulsion binder was fragmented into a larger volume of water using a 20 L mixing vessel, 250 mm in diameter and 400 mm high, with three equally spaced baffles located at the wall, each with a 25 mm radial protrusion. An overhead stirrer set at 1500 rpm was used to achieve the fragmentation and suspension of the binder portions. The concentration of the binder suspension was 20 wt% emulsion binder and 80 wt% tap water. This procedure resulted in the formation of a water-in-oil-in-water double emulsion—in effect, fragments of the binder suspended in water.
2.2. Continous Plug Flow Agglomeration
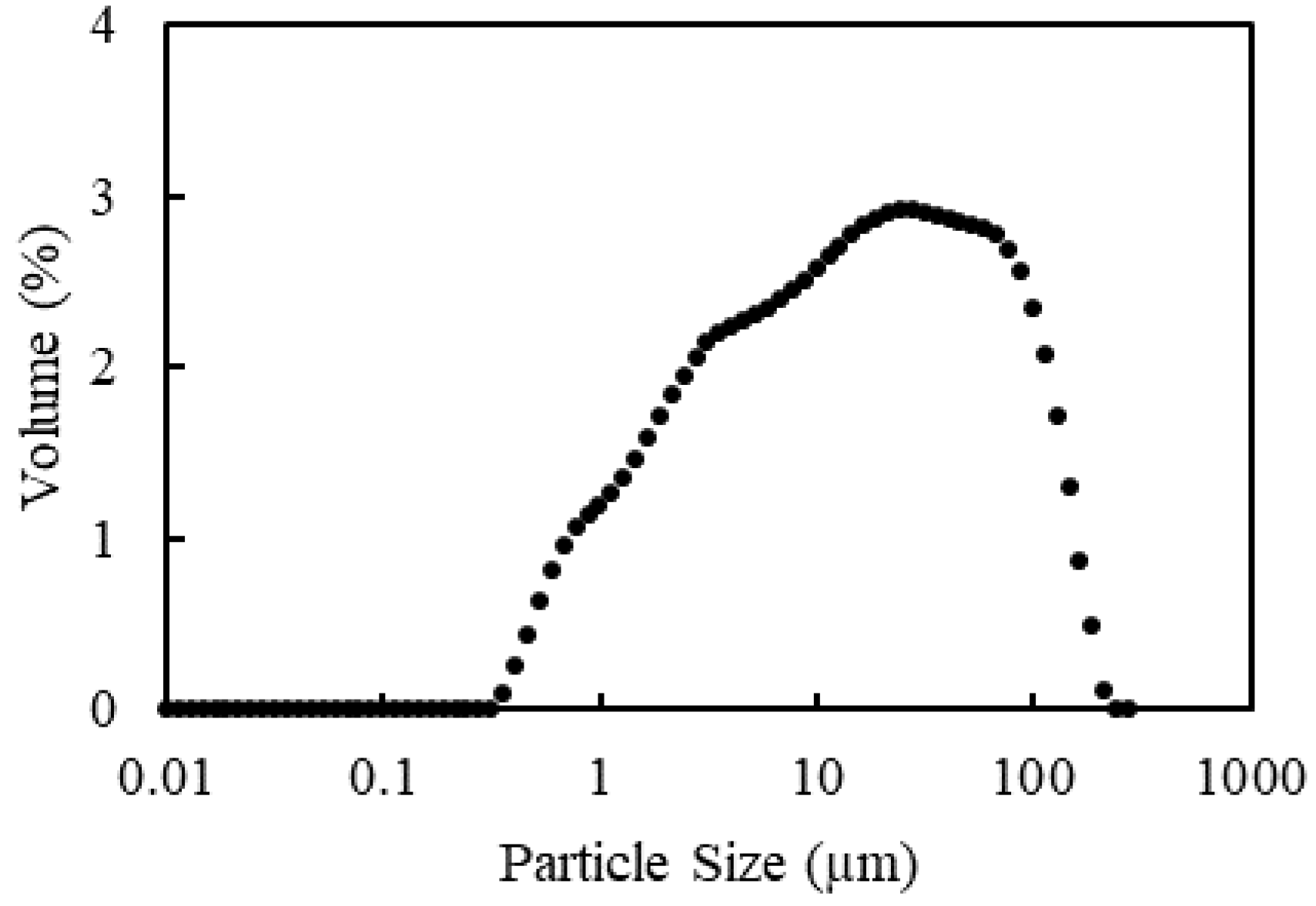
The coal feed, a hydrocyclone overflow with a mineral matter content equivalent to 50.1% ash, was sourced from a coal preparation plant in the upper Hunter Valley, NSW, Australia. The material had a size distribution with the 50th percentile,
d50, at 15.4 µm, and Sauter mean diameter,
d32, of 4.5 µm. The coal suspension, used as received, had a pulp density of 3.1 to 3.3 wt% solids.
Figure 2 shows the size distribution of the feed, as measured through laser diffraction using a Malvern Mastersizer 3000 (Malvern Panalytical, Malvern, UK).
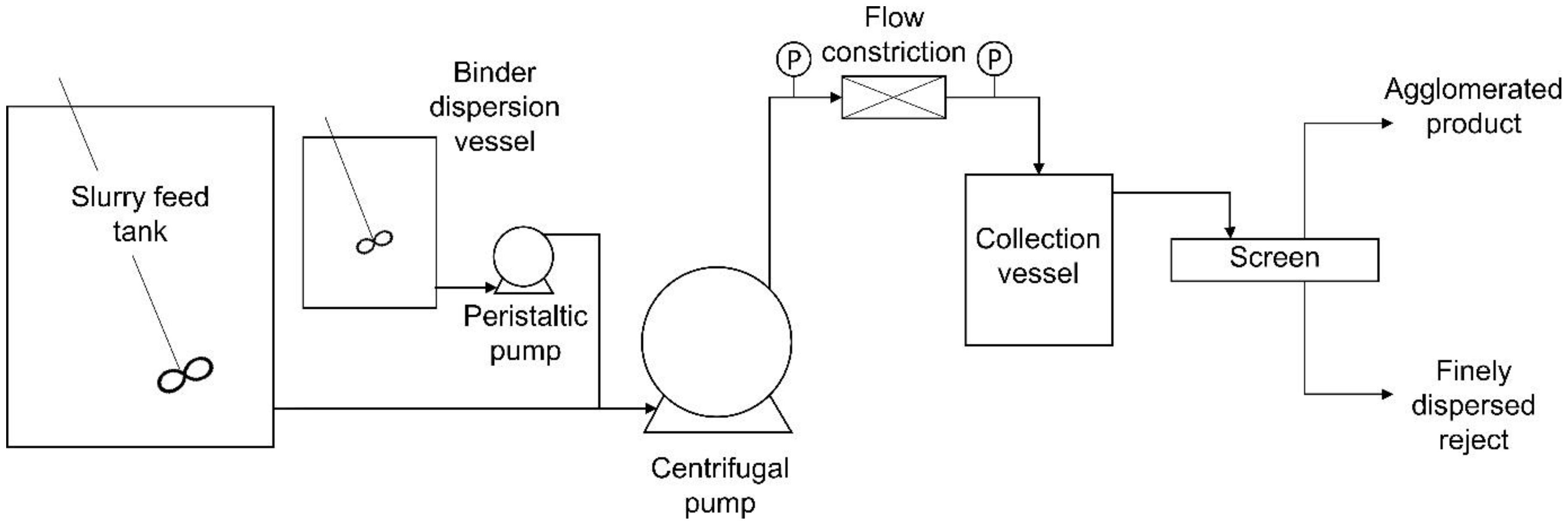
Figure 3 shows a schematic representation of the experimental configuration of the continuous plug flow agglomeration system used in this investigation.
As shown in the figure, the experimental system consisted of two tanks, a centrifugal pump, a peristaltic pump, the flow constriction and a collection vessel. The flow constriction was installed in a one-inch diameter stainless steel pipe and a one-inch diameter flexible hose was used to connect the tanks, via the pumps, to this pipe. Two pressure gauges were installed on the steel line, one on either side of the flow constriction, to provide a measure of the resulting pressure drop.
The coal feed was suspended in a 350 L tank, and the emulsion binder was dispersed separately in a 20 L tank. The centrifugal pump, which controlled the total flowrate through the system, was switched on first, followed by the peristaltic pump used to control the flowrate of the binder slurry into the system. As shown in
Figure 2, the binder slurry was introduced into the main line via a T-piece.
After the particle/binder slurry passed through the flow constriction, it was directed into a series of 20 L collection vessels to dissipate the energy of the high-velocity stream. Due to the short residence time within the flow constriction, the system reached a steady state almost immediately. However, the system was run for 30 s before samples were taken. After sampling, the agglomerated slurry was poured over a mechanical screen (aperture 180 µm) to separate the agglomerates from the hydrophilic slurry and washed. The resulting samples were then dried in an oven for 24 h. The dry samples were weighed to determine the mass yield, and the ash% analysis was conducted by an external laboratory to determine the mineral matter content of each sample.
Table 1 presents the flowrates and the corresponding pressure drops in each of the experiments. The feed flowrate was measured after the centrifugal pump and therefore included the binder suspension and the feed slurry. Also, as noted in
Table 1, the partially closed ball valve was used as the flow constriction in all but one of the experiments. The pressure drops reflect changes in the pumping rate only, apart from the run with the highest flowrate of 128 L/min which involved a more open ball valve constriction [
10]. In a separate experiment, an orifice plate with an internal diameter of 7 mm was used. The flowrates through the main pipe correspond to superficial flow velocities of up to 4.2 m/s, significantly higher than the flow velocity through conventional flotation cells of order 1 cm/s. Of course, it is still necessary to separate the agglomerates from the tailings, but it is clear the fundamental hydrophobic interaction is extraordinarily fast and efficient.
4. Conclusions
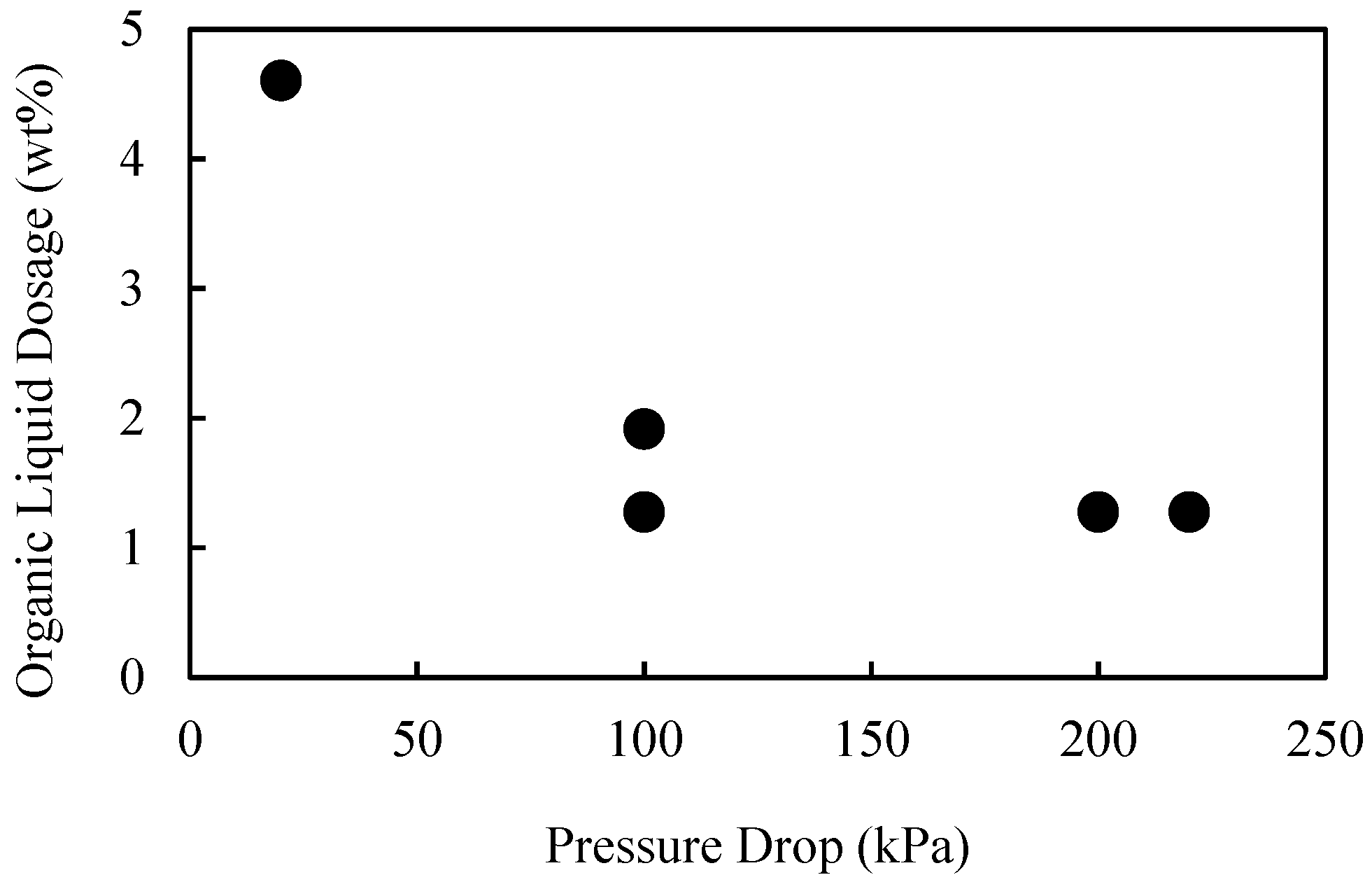
In this work, the influence of flow conditions on the selective agglomeration of high-ash fine coal using a high internal phase emulsion binder was investigated. The emulsion comprised 5% organic liquid (including the emulsifier) and 95% aqueous solution. To examine the performance of the binder under different flow conditions, flowrates in the range of 20 to 128 L/min were used. These flowrates equated to pressure drops in the range of 20–220 kPa, and extraordinary superficial velocity of up to 4.2 m/s. It was found that the flowrate and thus pressure drop across the flow constriction had a significant influence on the performance of the binder, with the dosage required for agglomeration decreasing from 4.6 wt% at 20 kPa to 2 wt% at 100 kPa. However, these performance improvements then appear to become marginal when operating above 150 kPa. It was therefore concluded that the performance of the binder is improved if the binder/particle suspension is subjected to a higher level of shear for a shorter period.
It was also found that the selectivity of the emulsion binder in the continuous plug flow system was remarkably good, given the high superficial flow velocity, provided there was sufficient shear within the flow constriction. When operating above 100 kPa, the system routinely delivered a product ash of 8% at a combustible recovery of 78% at the optimum binder dosage.
This work illustrates that the high internal phase emulsion binder can achieve rapid and selective agglomeration of fine particles under continuous plug flow conditions and that the performance of the binder improves under a shorter and more intense application of shear. Further work is now required to establish a method for the continuous separation of the agglomerates from the hydrophilic tailings material.