Use of Temperature Controlled Stage Confocal Raman Microscopy to Study Phase Transition of Lead Dioxide (Plattnerite)
Abstract
1. Introduction
2. Experiment
2.1. Materials
2.2. Laboratory Instrumentation
2.2.1. Raman Spectroscopy
Horiba Jobin Yvon LabRam micro-spectrometer
Renishaw InVia Raman spectrometer
2.2.2. PANalytical Xpert PRO Diffractometer
3. Results and Discussion
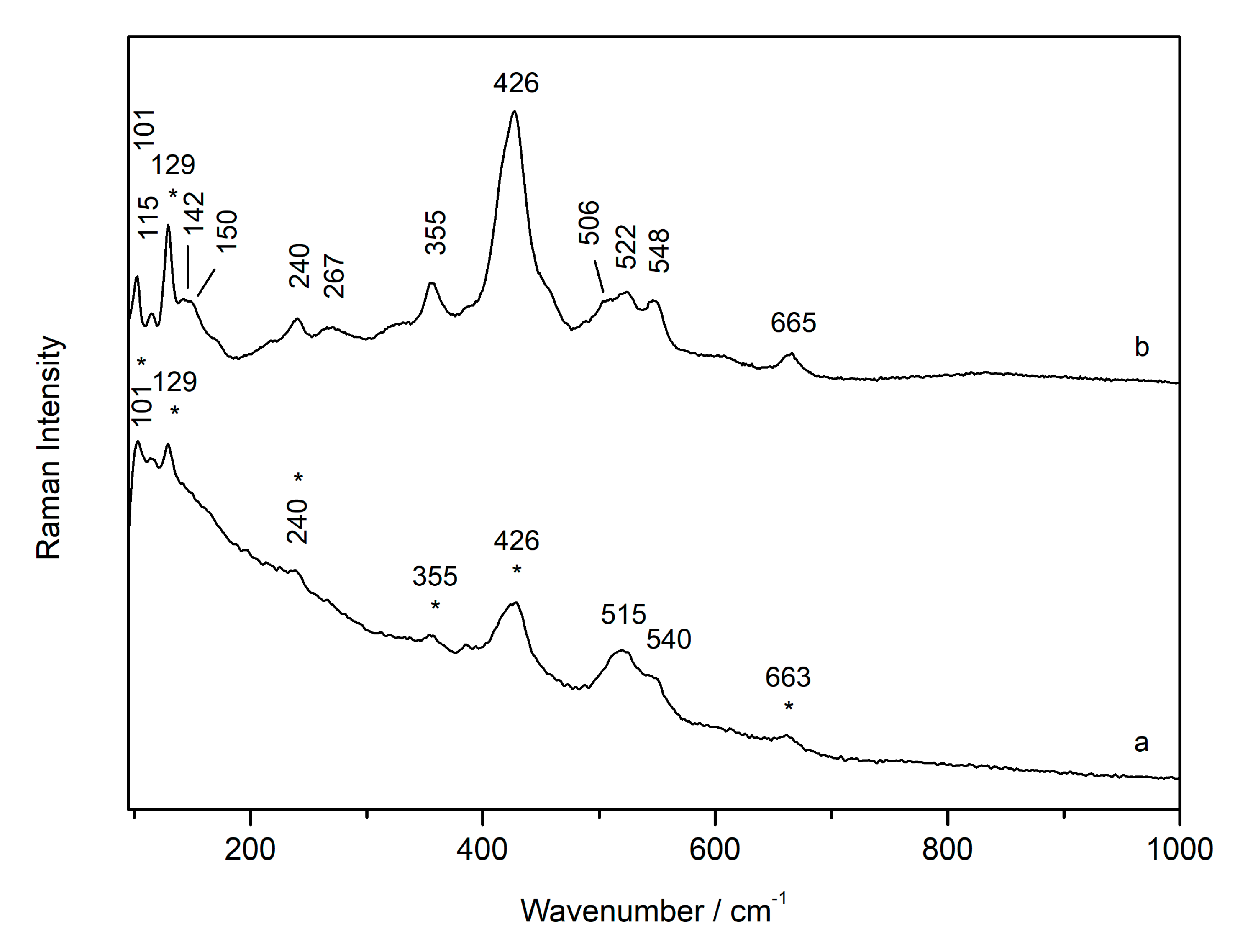
3.1. Laser-Induced Degradation of Plattnerite with 633 nm Laser Excitation
3.1.1. Laser-Induced Degradation of Plattnerite taken from the Mural Paintings
3.1.2. Laser-Induced Degradation of Commercial Plattnerite
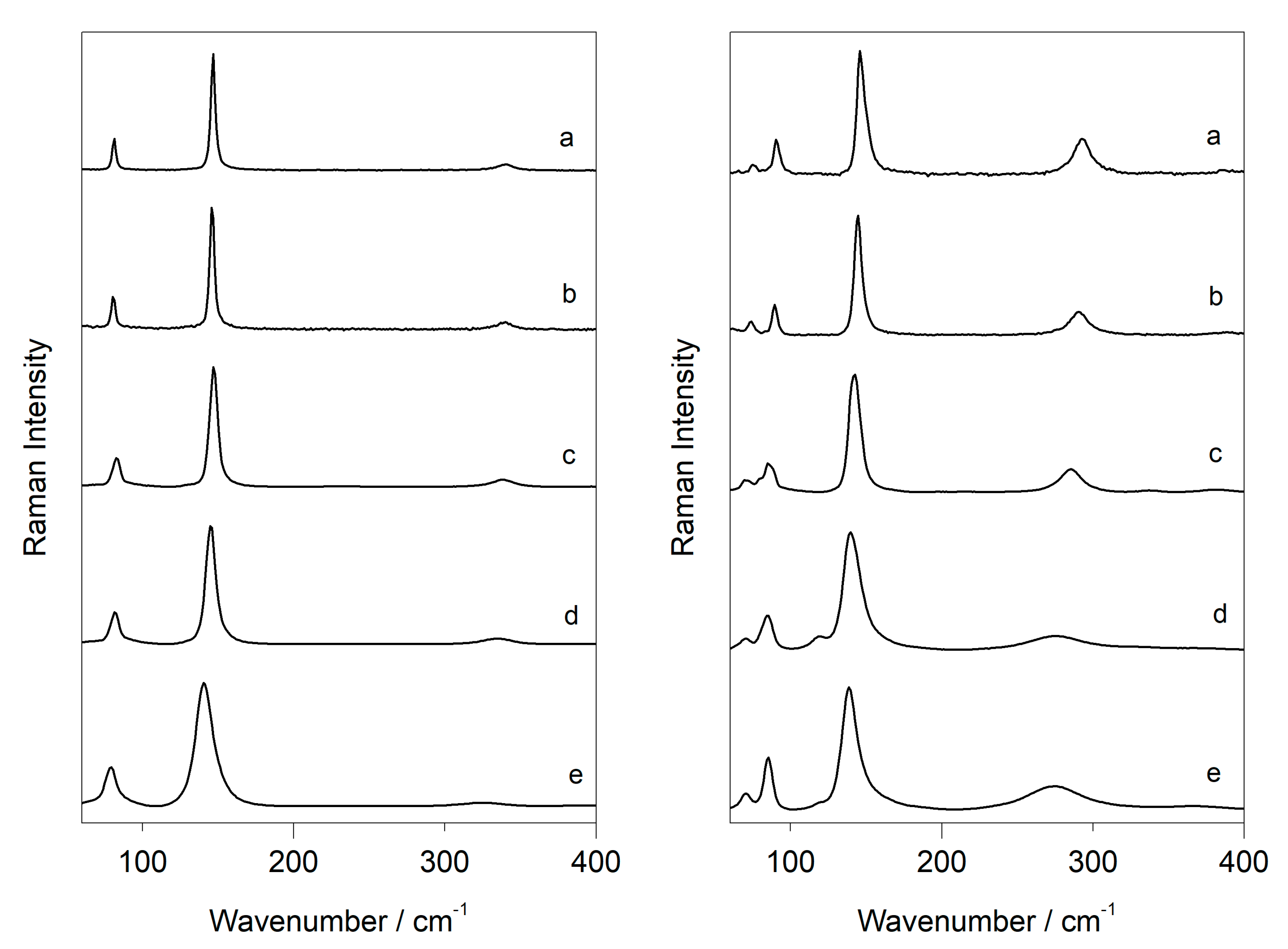
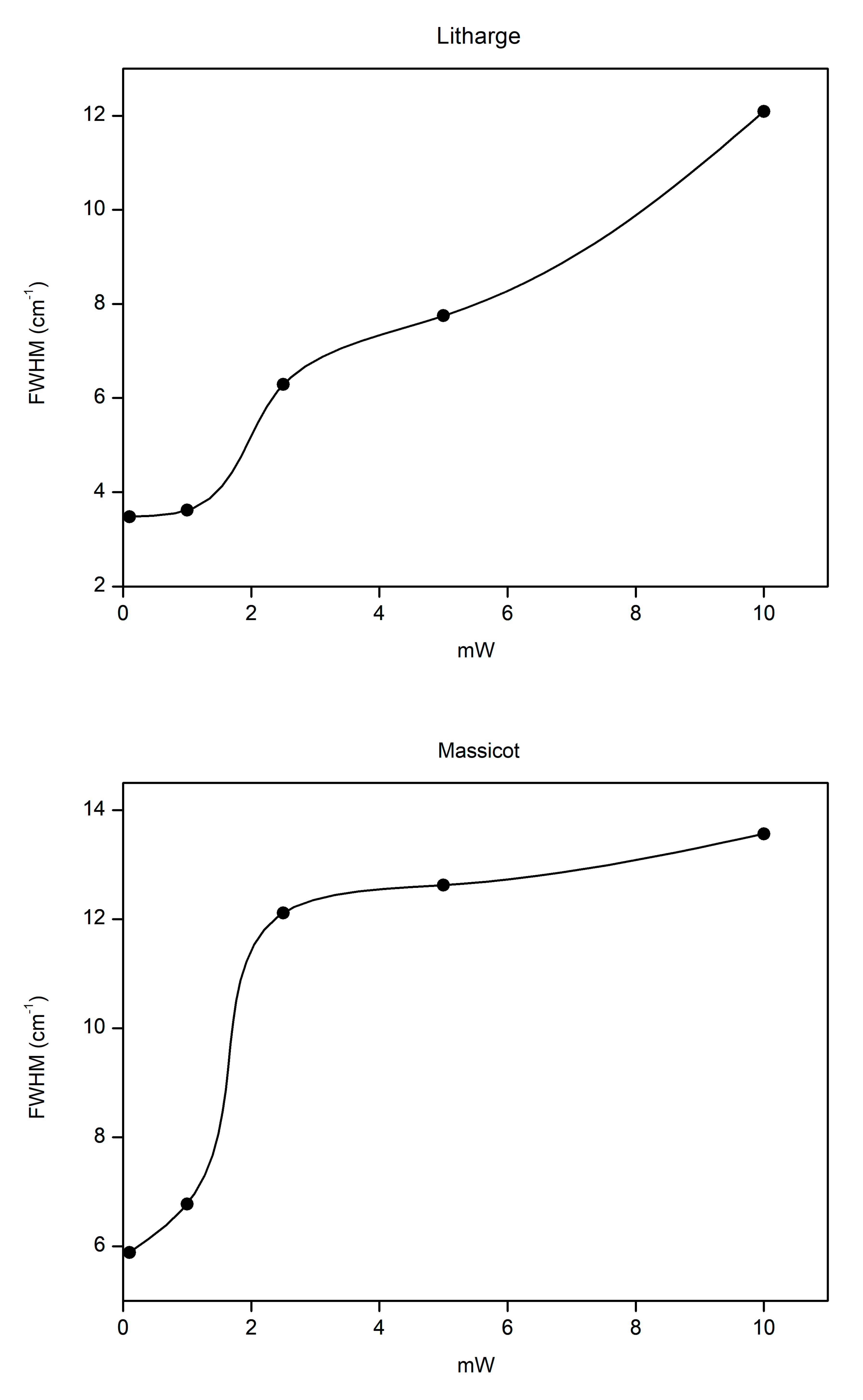
3.2. Laser-Induced Degradation of Plattnerite with 785 nm Laser
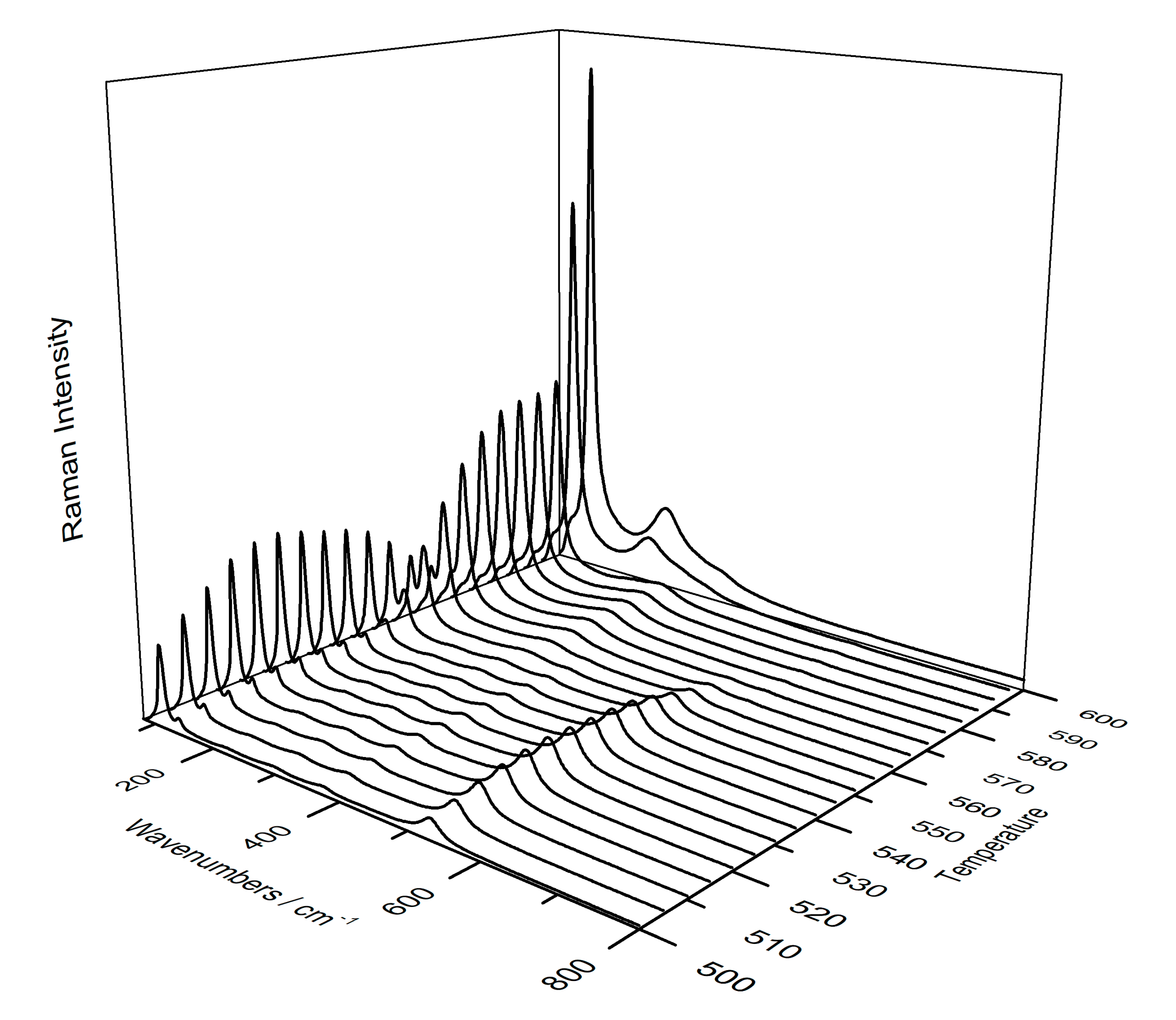
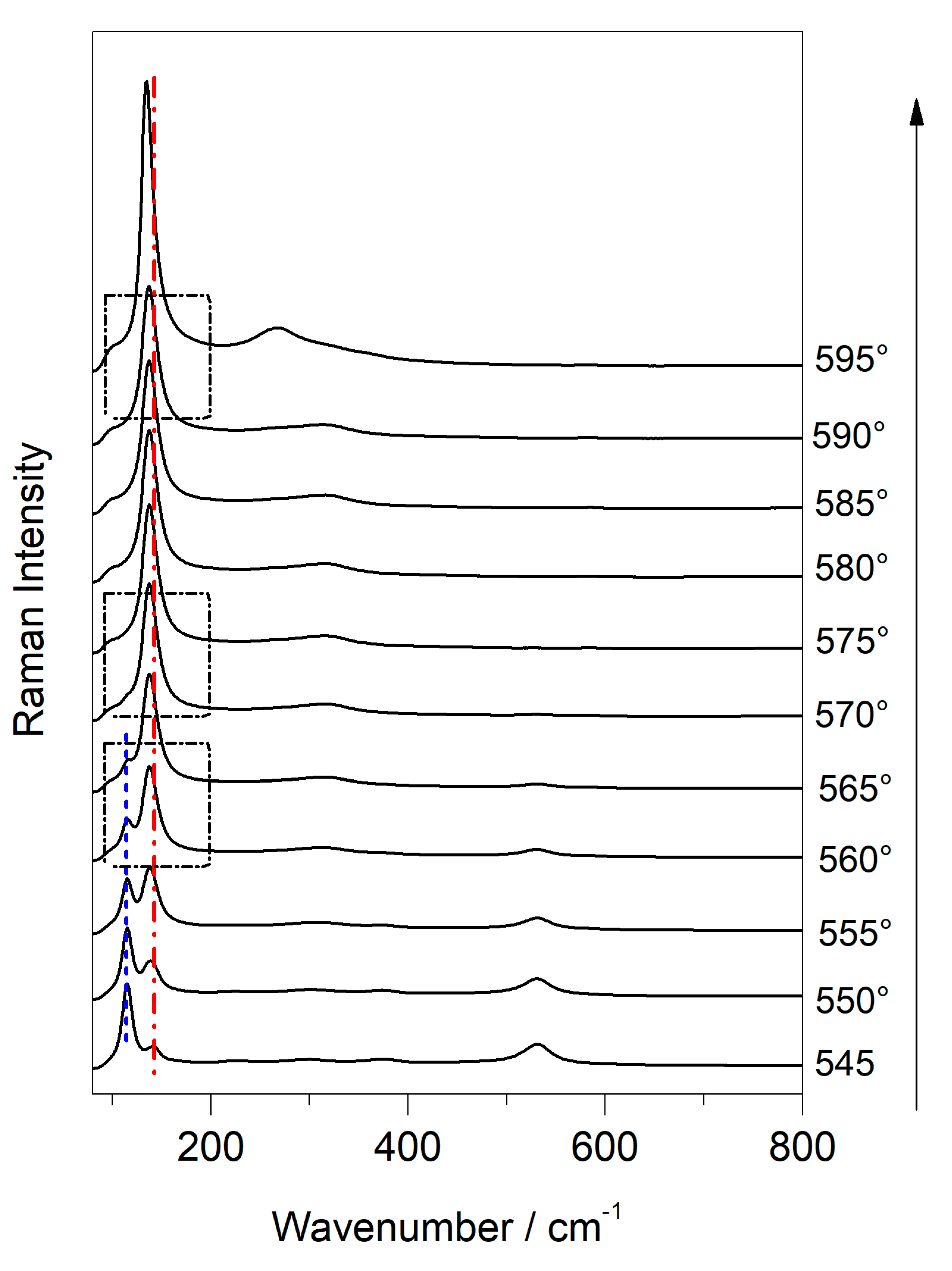
3.3. Use of Temperature-Controlled Stage to Study Phase Transition
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aze, S.; Vallet, J.-M.; Pomey, M.; Baronnet, A.; Grauby, O. Red Lead Darkening in Wall Paintings: Natural Ageing of Experimental Wall Paintings versus Artificial Ageing Tests. Eur. J. Miner. 2007, 19, 883–890. [Google Scholar] [CrossRef]
- Vagnini, M.; Vivani, R.; Viscuso, E.; Favazza, M.; Brunetti, B.G.; Sgamellotti, A.; Miliani, C. Investigation on the Process of Lead White Blackening by Raman Spectroscopy, XRD and Other Methods: Study of Cimabue’s Paintings in Assisi. Vib. Spectrosc. 2018, 98, 41–49. [Google Scholar] [CrossRef]
- Giovannoni, S.; Matteini, M.; Moles, A. Studies and Developments Concerning the Problem of Altered Lead Pigments in Wall Painting. Stud. Conserv. 1990, 35, 21–25. [Google Scholar]
- Coccato, A.; Moens, L.; Vandenabeele, P. On the Stability of Mediaeval Inorganic Pigments: A Literature Review of the Effect of Climate, Material Selection, Biological Activity, Analysis and Conservation Treatments. Herit. Sci. 2017, 5, 1–12. [Google Scholar]
- Daniilia, S.; Minopoulou, E. A Study of Smalt and Red Lead Discolouration in Antiphonitis Wall Paintings in Cyprus. Appl. Phys. A 2009, 96, 701–711. [Google Scholar]
- Kotulanová, E.; Bezdička, P.; Hradil, D.; Hradilová, J.; Švarcová, S.; Grygar, T. Degradation of Lead-Based Pigments by Salt Solutions. J. Cult. Herit. 2009, 10, 367–378. [Google Scholar] [CrossRef]
- Hradil, D.; Hradilová, J.; Kočí, E.; Švarcová, S.; Bezdička, P.; Maříková-Kubková, J. Unique Pre-Romanesque Murals in Kostoľany Pod Tríbečom, Slovakia: The Painting Technique and Causes of Damage. Archaeometry 2013, 55, 691–706. [Google Scholar] [CrossRef]
- Aze, S.; Vallet, J.-M.; Detalle, V.; Grauby, O.; Baronnet, A. Chromatic Alterations of Red Lead Pigments in Artworks: A Review. Phase Transit. 2008, 81, 145–154. [Google Scholar] [CrossRef]
- Daniilia, S.; Sotiropoulou, S.; Bikiaris, D.; Salpistis, C.; Karagiannis, G.; Chryssoulakis, Y.; Price, B.A.; Carlson, J.H. Panselinos’ Byzantine Wall Paintings in the Protaton Church, Mount Athos, Greece: A Technical Examination. J. Cult. Herit. 2000, 1, 91–110. [Google Scholar] [CrossRef]
- Petushkova, J.P.; Lyalikova, N.N. Microbiological Degradation of Lead-Containing Pigments in Mural Paintings. Stud. Conserv. 1986, 31, 65–69. [Google Scholar]
- Feng, Q.; Zhang, X.; Ma, X. Effects of Microbes on Color Changes of Red Lead in Murals. J. Gen. Appl. Microbiol. 1999, 45, 85–88. [Google Scholar]
- Rosado, T.; Gil, M.; Mirão, J.; Candeias, A.; Caldeira, A.T. Darkening on Lead-Based Pigments: Microbiological Contribution. Color Res. Appl. 2016, 41, 294–298. [Google Scholar] [CrossRef]
- Aze, S.; Vallet, J.-M.; Baronnet, A.; Grauby, O. The Fading of Red Lead Pigment in Wall Paintings: Tracking the Physico-Chemical Transformations by Means of Complementary Micro-Analysis Techniques. Eur. J. Mineral. 2006, 18, 835–843. [Google Scholar] [CrossRef]
- Holakooei, P.; Karimy, A.-H. Early Islamic Pigments Used at the Masjid-i Jame of Fahraj, Iran: A Possible Use of Black Plattnerite. J. Archaeol. Sci. 2015, 54, 217–227. [Google Scholar] [CrossRef]
- De Ferri, L.; Mazzini, F.; Vallotto, D.; Pojana, G. In Situ Non-Invasive Characterization of Pigments and Alteration Products on the Masonry Altar of S. Maria Ad Undas (Idro, Italy). Archaeol. Anthropol. Sci. 2019, 11, 609–625. [Google Scholar] [CrossRef]
- Burgio, L.; Clark, R.J.H.; Firth, S. Raman Spectroscopy as a Means for the Identification of Plattnerite (PbO2), of Lead Pigments and of Their Degradation Products. Analyst 2001, 126, 222–227. [Google Scholar] [CrossRef]
- Andalò, C.; Bicchieri, M.; Bocchini, P.; Casu, G.; Galletti, G.C.; Mandò, P.A.; Nardone, M.; Sodo, A.; Plossi Zappalà, M. The Beautiful “Trionfo d’Amore” Attributed to Botticelli: A Chemical Characterisation by Proton-Induced X-Ray Emission and Micro-Raman Spectroscopy. Anal. Chim. Acta 2001, 429, 279–286. [Google Scholar] [CrossRef]
- Smith, G.D.; Burgio, L.; Firth, S.; Clark, R.J.H. Laser-Induced Degradation of Lead Pigments with Reference to Botticelli’s Trionfo d’Amore. Anal. Chim. Acta 2001, 440, 185–188. [Google Scholar] [CrossRef]
- Santis, A.D.; Mattei, E.; Pelosi, C. Micro-Raman and Stratigraphic Studies of the Paintings on the ‘Cembalo’ Model Musical Instrument (A.D. 1650) and Laser-Induced Degradation of the Detected Pigments. J. Raman Spectrosc. 2007, 38, 1368–1378. [Google Scholar] [CrossRef]
- Vila, M.; Díaz-Guerra, C.; Piqueras, J. Laser Irradiation-Induced α to δ Phase Transformation in Bi2O3 Ceramics and Nanowires. Appl. Phys. Lett. 2012, 101, 71905. [Google Scholar] [CrossRef]
- Camacho-López, M.A.; Escobar-Alarcón, L.; Picquart, M.; Arroyo, R.; Córdoba, G.; Haro-Poniatowski, E. Micro-Raman Study of the m-MoO2 to α-MoO3 Transformation Induced by Cw-Laser Irradiation. Opt. Mater. 2011, 33, 480–484. [Google Scholar] [CrossRef]
- Vásquez, G.C.; Peche-Herrero, M.A.; Maestre, D.; Gianoncelli, A.; Ramírez-Castellanos, J.; Cremades, A.; González-Calbet, J.M.; Piqueras, J. Laser-Induced Anatase-to-Rutile Transition in TiO2 Nanoparticles: Promotion and Inhibition Effects by Fe and Al Doping and Achievement of Micropatterning. J. Phys. Chem. C 2015, 119, 11965–11974. [Google Scholar] [CrossRef]
- Costantini, I.; Lottici, P.P.; Bersani, D.; Pontiroli, D.; Casoli, A.; Castro, K.; Madariaga, J.M. Darkening of Lead- and Iron-Based Pigments on Late Gothic Italian Wall Paintings: Energy Dispersive X-Ray Fluorescence, μ-Raman, and Powder X-Ray Diffraction Analyses for Diagnosis: Presence of β-PbO2 (Plattnerite) and α-PbO2 (Scrutinyite). J. Raman Spectrosc. 2020, 51, 680–692. [Google Scholar] [CrossRef]
- Clark, G.L.; Schieltz, N.C.; Quirke, T.T. A New Study of the Preparation and Properties of the Higher Oxides of Lead. J. Am. Chem. Soc. 1937, 59, 2305–2308. [Google Scholar] [CrossRef]
- Greenwood, N.N.; Earnshaw, A. Chemistry of the Elements, 2nd ed.; Elsevier Butterworth-Heinemann: Oxford, UK, 1997. [Google Scholar]
- Bordalo, R.; Morais, P.J.; Gouveia, H.; Young, C. Laser Cleaning of Easel Paintings: An Overview. Laser Chem. 2006, 2006, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bordalo, R. Characterisation of Laser-Induced Physical Alterations of Pigmented Oil Layers. e-PS 2012, 9, 47–59. [Google Scholar]
- Pouli, P.; Emmony, D.C.; Madden, C.E.; Sutherland, I. Studies towards a Thorough Understanding of the Laser-Induced Discoloration Mechanisms of Medieval Pigments. J. Cult. Herit. 2003, 4, 271–275. [Google Scholar] [CrossRef]
- Cooper, M.I.; Fowles, P.S.; Tang, C.C. Analysis of the Laser-Induced Discoloration of Lead White Pigment. Appl. Surf. Sci. 2002, 201, 75–84. [Google Scholar] [CrossRef]
- Aze, S.; Delaporte, P.; Vallet, J.M.; Detalle, V.; Baronnet, A. Towards the restoration of darkened red lead containing mural paintings. In Lasers in the Conservation of Artworks, Proceedings of the International Conference Lacona VII, Madrid, Spain, 17–21 September 2007; Castillejo, P.M., Moreno, M., Oujja, R., Radvan, J.R., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 11–13. [Google Scholar]
- Bayne, J.M.; Butler, I.S. Variable-Temperature and High-Pressure Micro-Raman Spectra of Inorganic Artists’ Pigments: Crystalline Wulfenite, Lead(II) Molybdate(VI), PbMoO4. Spectrosc. Lett. 2014, 47, 616–620. [Google Scholar] [CrossRef]
- Bayne, J.M.; Butler, I.S. Effect of Temperature and Pressure on Selected Artists’ Pigments. New J. Chem. 2013, 37, 3833–3839. [Google Scholar] [CrossRef]
- Smith, G.D.; Firth, S.; Clark, R.J.H.; Cardona, M. First- and Second-Order Raman Spectra of Galena (PbS). J. Appl. Phys. 2002, 92, 4375–4380. [Google Scholar] [CrossRef]
- Zoellner, M.T.; Ziemann, M.A. Plattnerite-Scrutinyite in a Central Asian Wall Painting of a Buddhist Cave Temple of the Northern Silk Road. In Abstracts Conference Programme, Proceedings of the 10th International Congress on the Application of Raman Spectroscopy in Art and Archaeology, Potsdam, Germany, 3–7 September 2019; Institute of Geosciences at the University of Potsdam: Potsdam, Germany, 2019. [Google Scholar]
- Anderson, J.S.; Sterns, M. The Intermediate Oxides of Lead. J. Inorg. Nucl. Chem. 1959, 11, 272–285. [Google Scholar] [CrossRef]
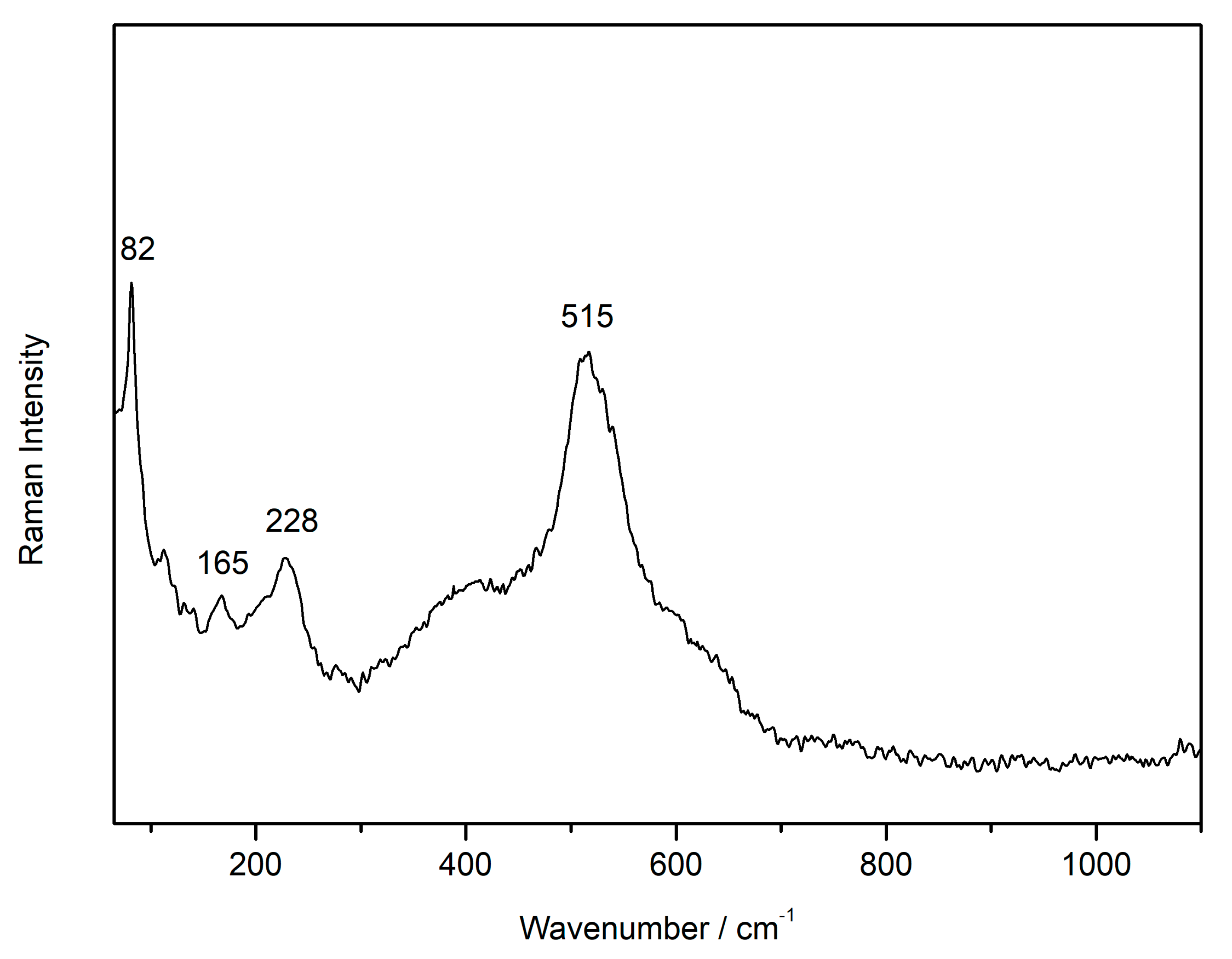
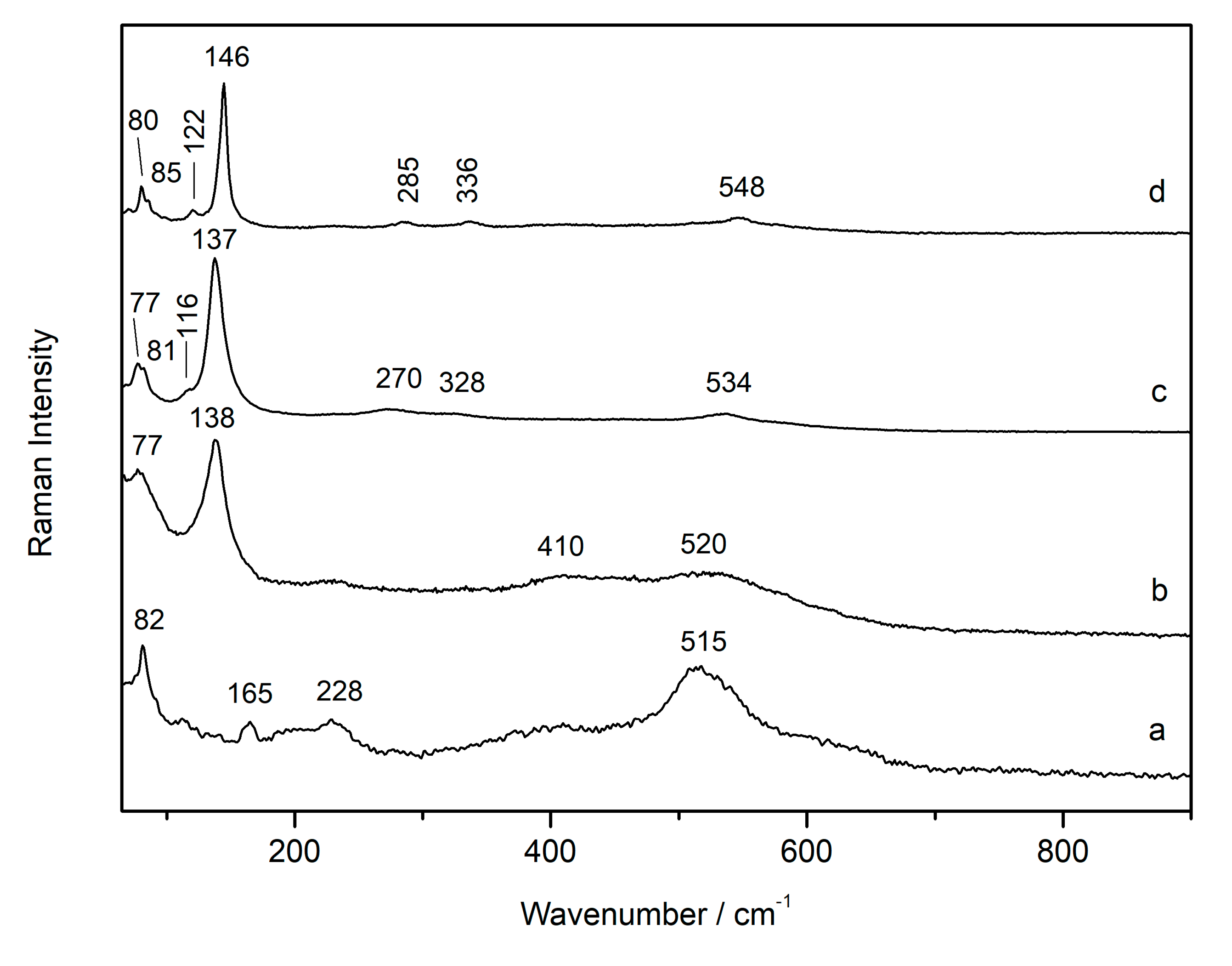
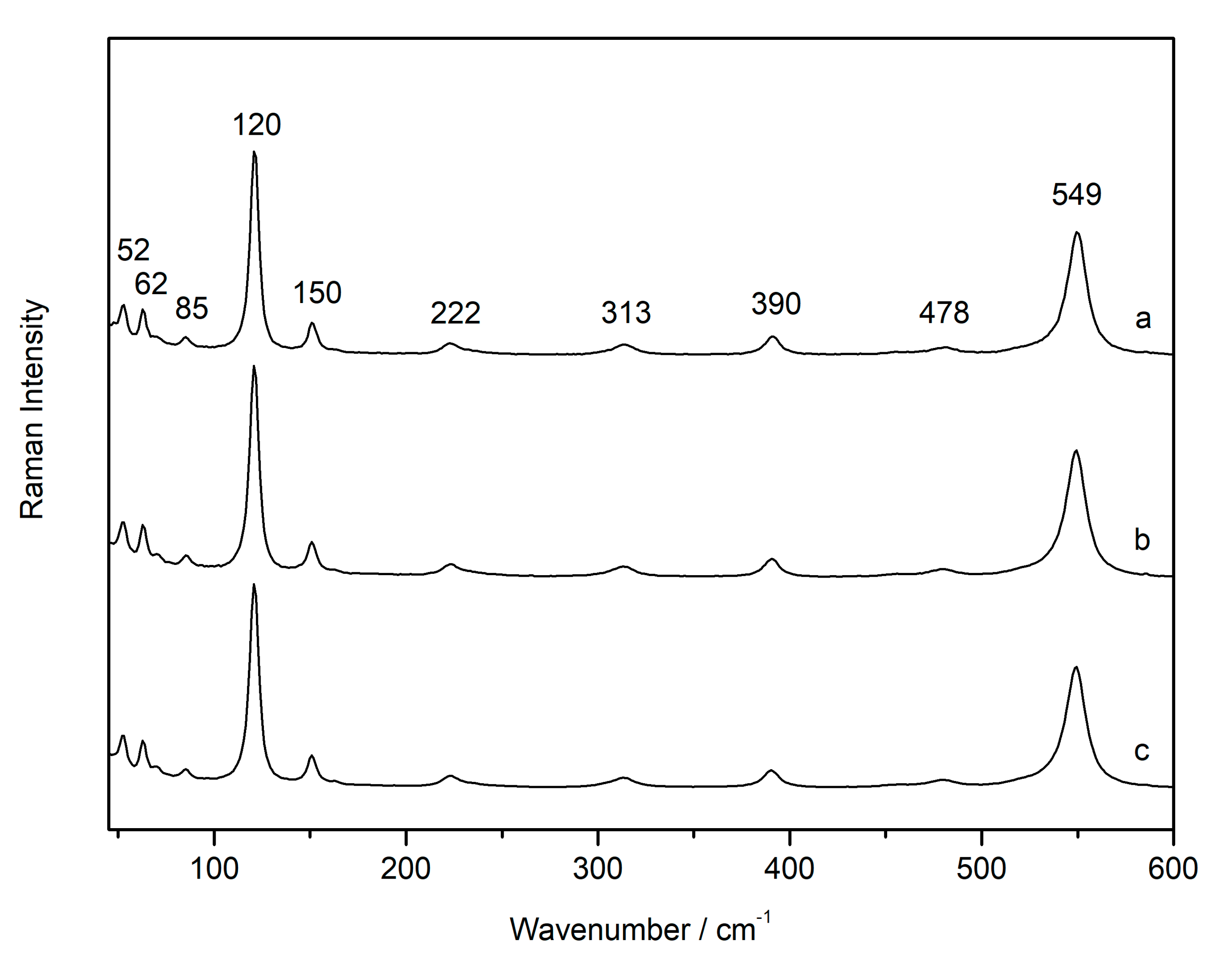
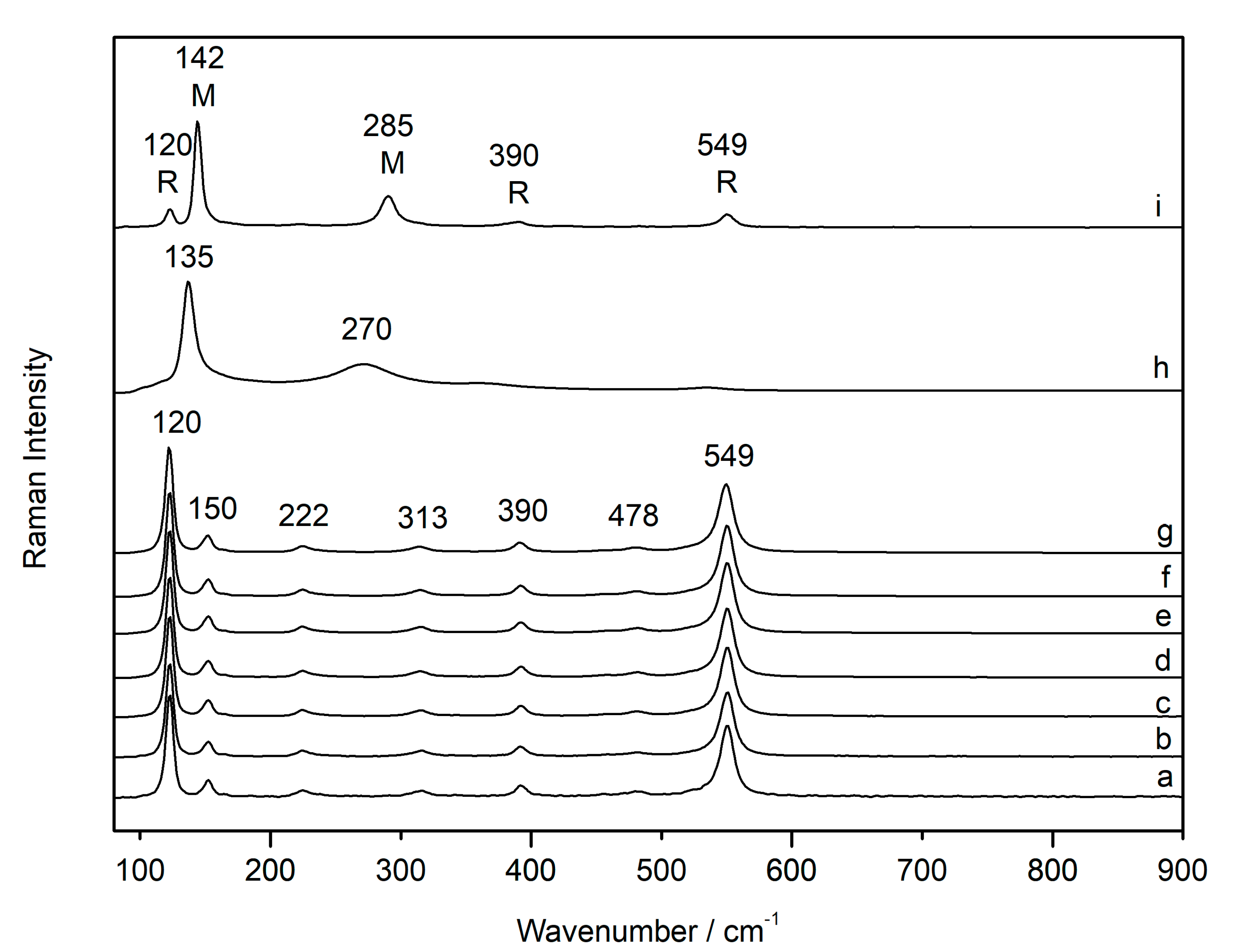
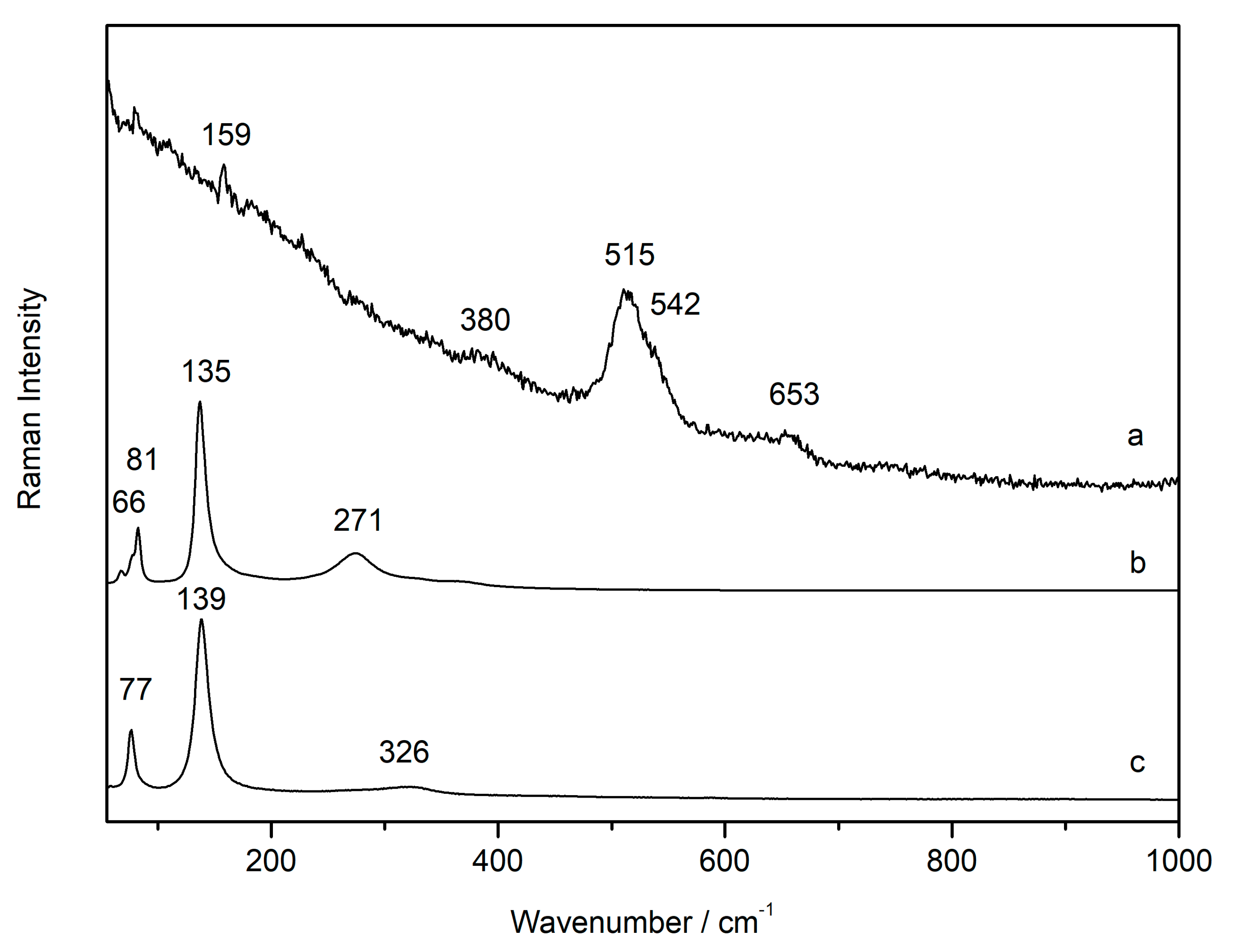
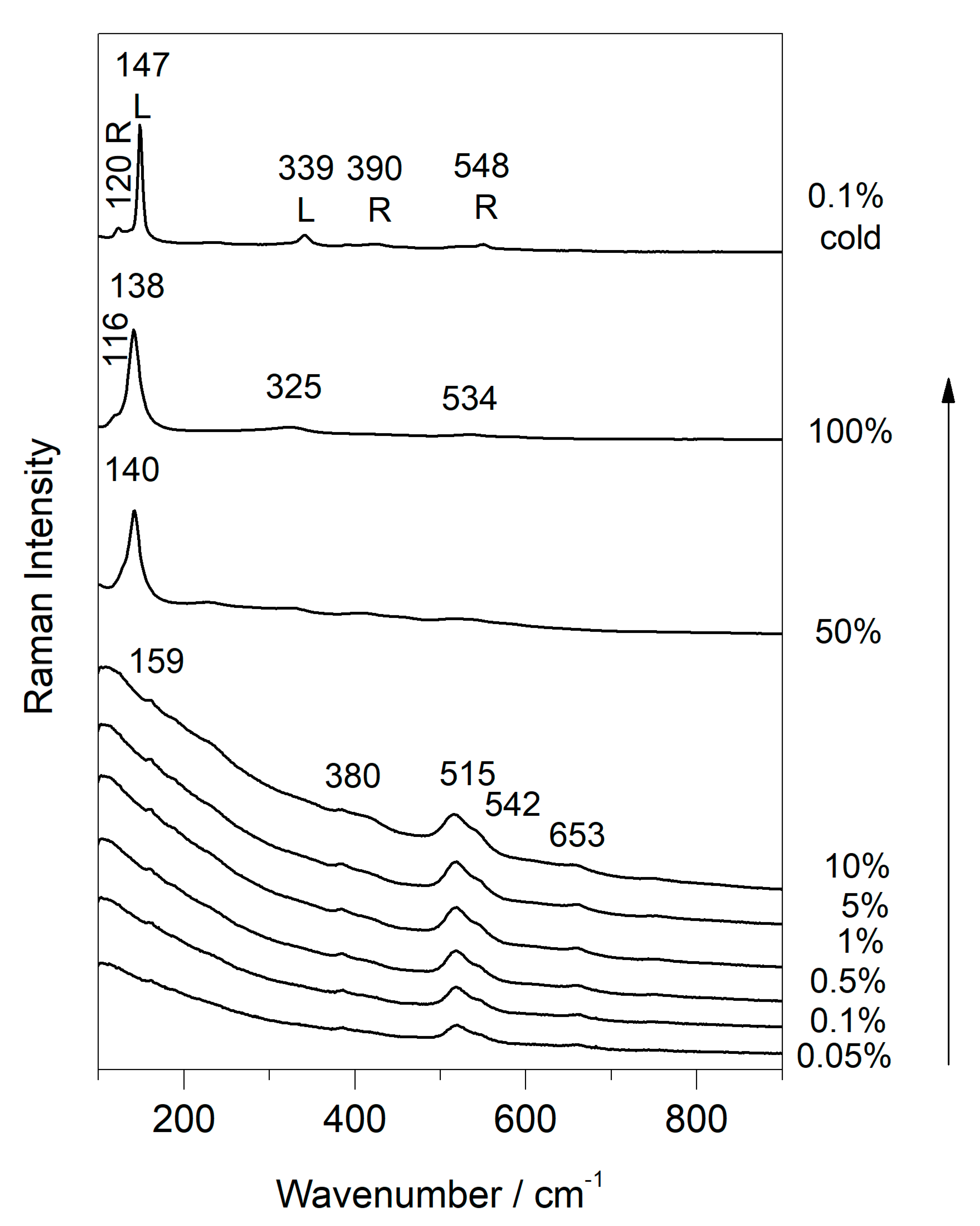

| Compounds | D2 | D0.6 | D0.0 | D2 |
|---|---|---|---|---|
| Plattnerite (β- PbO2) | 515 | |||
| Scrutiniyte (α-PbO2) | 82, 165, 228 | |||
| Red lead Pb3O4 | 116 (shi), 534 (shi) | 122, 548 | ||
| Litharge (α-PbO) | 77 (shi) | 77 (shi), 328 (shi) | 80, 146, 336 | |
| Massicot (β-PbO) | 138 (br/shi) | 137 (br/shi), 81 (shi), 270 (shi) | 85, 142 (sh), 285 |
| Litharge α-PbO-Tetragonal | |||
|---|---|---|---|
| D2 | 81 m | 146 vs | 340 w |
| D1 | 81 m | 145 vs | 338 w |
| D0.6 | 82 m | 145 vs | 337 w |
| D0.3 | 82 m | 144 vs | 335 w |
| D0.0 | 77 m | 140 vs | 325 w |
| Massicot β-PbO-Orthorhombic | ||||
|---|---|---|---|---|
| D2 | 74 w | 91 m | 145 vs | 292 m |
| D1 | 73 w | 89 m | 143 vs | 290 m |
| D0.6 | 70 w | 84 m | 142 vs | 285 m |
| D0.3 | 67 w | 83 m | 138 vs | 280 m |
| D0.0 | 66 w | 81 m | 135 vs | 271 m |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costantini, I.; Lottici, P.P.; Castro, K.; Madariaga, J.M. Use of Temperature Controlled Stage Confocal Raman Microscopy to Study Phase Transition of Lead Dioxide (Plattnerite). Minerals 2020, 10, 468. https://doi.org/10.3390/min10050468
Costantini I, Lottici PP, Castro K, Madariaga JM. Use of Temperature Controlled Stage Confocal Raman Microscopy to Study Phase Transition of Lead Dioxide (Plattnerite). Minerals. 2020; 10(5):468. https://doi.org/10.3390/min10050468
Chicago/Turabian StyleCostantini, Ilaria, Pier Paolo Lottici, Kepa Castro, and Juan Manuel Madariaga. 2020. "Use of Temperature Controlled Stage Confocal Raman Microscopy to Study Phase Transition of Lead Dioxide (Plattnerite)" Minerals 10, no. 5: 468. https://doi.org/10.3390/min10050468
APA StyleCostantini, I., Lottici, P. P., Castro, K., & Madariaga, J. M. (2020). Use of Temperature Controlled Stage Confocal Raman Microscopy to Study Phase Transition of Lead Dioxide (Plattnerite). Minerals, 10(5), 468. https://doi.org/10.3390/min10050468






