Ultrafast Elemental Mapping of Platinum Group Elements and Mineral Identification in Platinum-Palladium Ore Using Laser Induced Breakdown Spectroscopy
Abstract
1. Introduction
2. Materials and Methods
2.1. LIBS CORIOSITY Analyser
2.2. Ore Samples
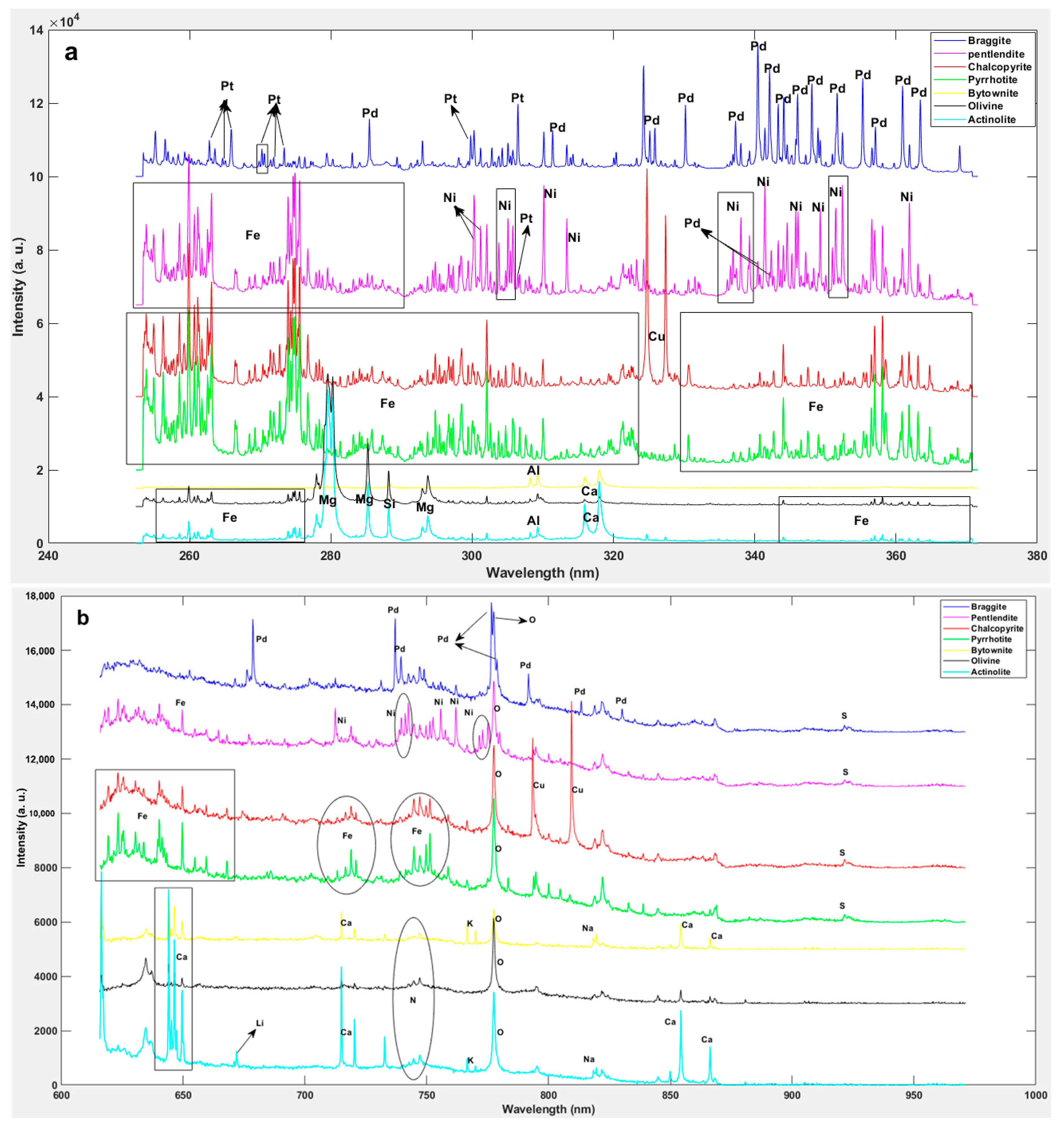
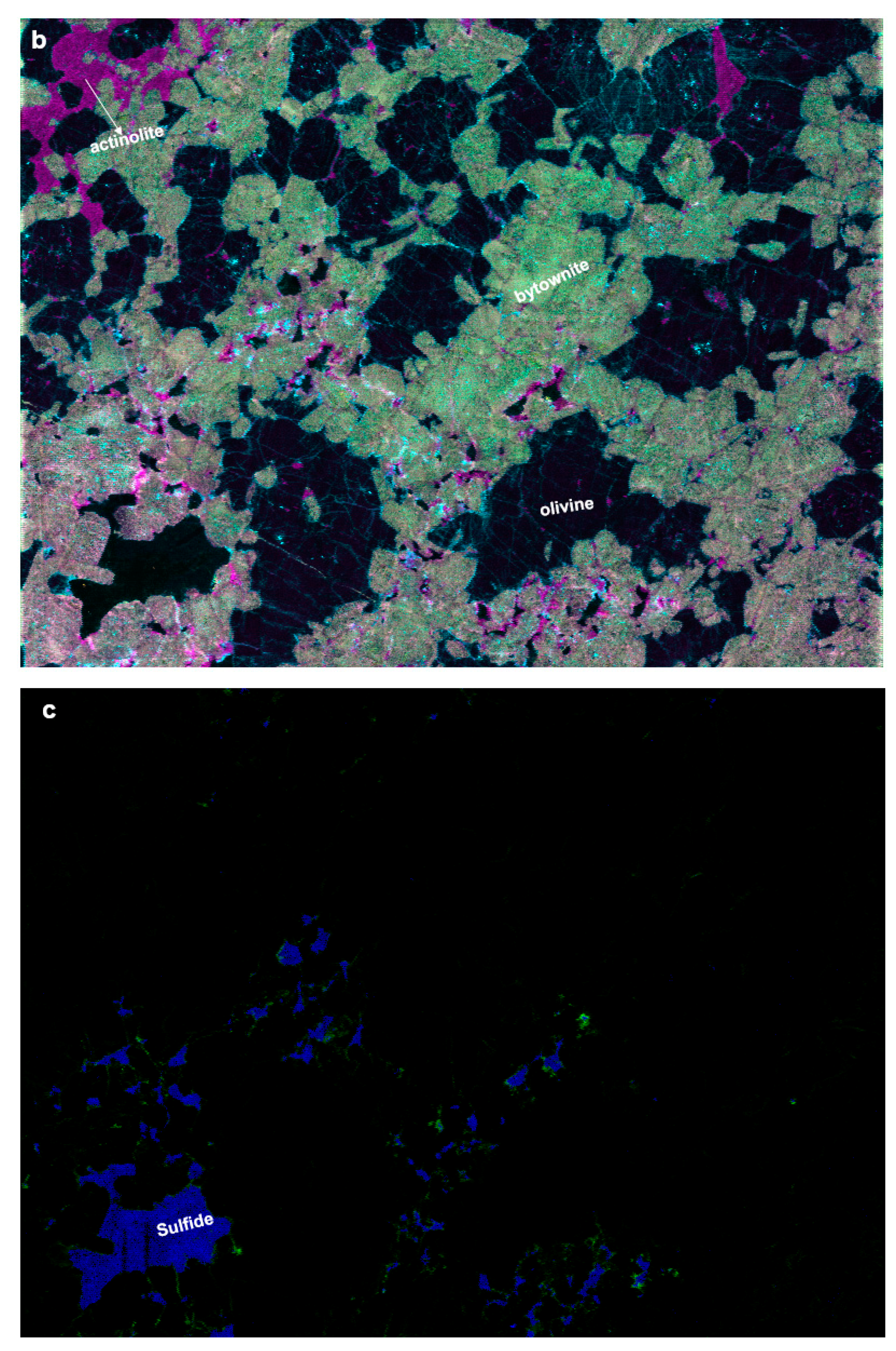
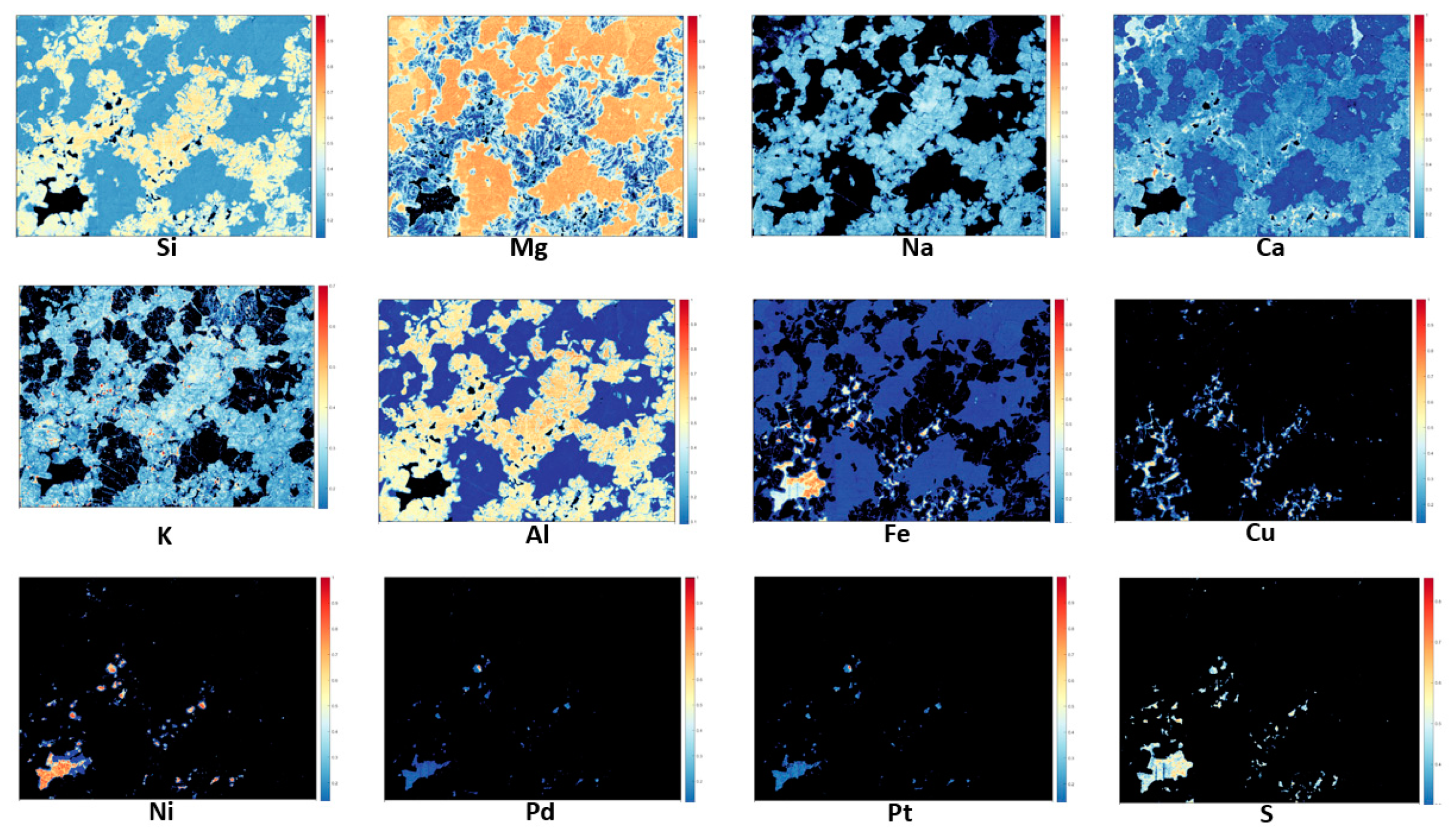
2.3. Identification of the Mineralogical Classes
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zientek, M.L.; Loferski, P.J.; Parks, H.L.; Schulte, R.F.; Seal, R.R., II. Platinum-Group Elements; Schulz, K.J., DeYoung, J.H., Eds.; Critical Mineral Resources of the United States—Economic and Environmental Geology and Prospects for Future Supply: U.S. Geological Survey Professional Paper; USGS Publications: Reston, VA, USA, 2017; pp. N1–N91. [CrossRef]
- Harmon, R.S.; Russo, R.E.; Hark, R.R. Applications of laser-induced breakdown spectroscopy for geochemical and environmental analysis: A comprehensive review. Spectrochim. Acta B 2013, 87, 11–26. [Google Scholar] [CrossRef]
- Ma, Q.L.; Motto-Ros, V.; Lei, W.Q.; Boueri, M.; Zheng, L.J.; Zeng, H.P.; Bar-Matthews, M.; Ayalon, A. Multi-elemental mapping of a speleothem using laser-induced breakdown spectroscopy. Spectrochim. Acta B 2010, 65, 707–714. [Google Scholar] [CrossRef]
- Cáceres, J.O.; Pelascini, F.; Motto-Ros, V.; Moncayo, S.; Trichard, F.; Panczer, G.; Marín Roldán, A.; Cruz, J.A.; Coronado, I.; Martín-Chivelet, J. Megapixel multi-elemental imaging by laser induced breakdown spectroscopy, a technology with considerable potential for paleoclimate studies. Sci. Rep. 2017, 7, 5080. [Google Scholar] [CrossRef] [PubMed]
- Haavisto, O.; Kauppinen, T.; Häkkänen, H. Laser-induced breakdown spectroscopy for rapid elemental analysis of drillcore. IFAC Proc. Vol. 2013, 46, 25–28. [Google Scholar] [CrossRef]
- Rifai, K.; Doucet, F.; Özcan, L.; Vidal, F. LIBS core imaging at kHz speed: Paving the way for real-time geochemical applications. Spectrochim. Acta B 2018, 150, 43–48. [Google Scholar] [CrossRef]
- Klus, J.; Pořízka, P.; Prochazka, D.; Mikysek, P.; Novotný, J.; Novotný, K.; Slobodník, M.; Kaiser, J. Application of self-organizing maps to the study of U-Zr-Ti-Nb distribution in sandstone-hosted uranium ores. Spectrochim. Acta B 2017, 131, 66–73. [Google Scholar] [CrossRef]
- Pagnotta, S.; Lezzerini, M.; Campanella, B.; Gallello, G.; Grifoni, E.; Legnaioli, S.; Lorenzetti, G.; Poggialini, F.; Raneri, S.; Safi, A.; et al. Fast quantitative elemental mapping of highly inhomogeneous materials by micro-Laser-Induced Breakdown Spectroscopy. Spectrochim. Acta 2018, 146, 9–15. [Google Scholar] [CrossRef]
- A Breakthrough Design for a Breakthrough Technology. Available online: http://www.elemission.ca/en/ (accessed on 25 February 2020).
- Özcan, L.C.; Doucet, F.R. Method and System for Analysis of Samples Using Laser Induced Breakdown Spectroscopy. U.S. Patent 62/213,431, 2 September 2015. [Google Scholar]
- Brereton, R.G. Chemometrics Data Analysis for the Laboratory and Chemical Plant; John Wiley & Sons: Chichester, UK, 2003; pp. 191–210. [Google Scholar]
- Moncayo, S.; Duponchel, L.; Mousavipak, N.; Panczer, G.; Trichard, F.; Bousquet, B.; Pelascini, F.; Motto-Ros, V. Exploration of megapixel hyperspectral LIBS images using principal component analysis. J. Anal. At. Spectrom. 2018, 33, 210–220. [Google Scholar] [CrossRef]
- Atomic Spectra Database. Available online: https://www.nist.gov/pml/atomic-spectra-database (accessed on 25 February 2020).
- Page, N.J.; Zientek, M.L.; Czamanske, G.K.; Foose, M.P. Sulfide mineralization in the Stillwater complex and underlaying rocks. Stillwater Complex Mont. Geol. Guide Mont. Bur. Mines Geol. Spec. Publ. 1985, 92, 93–96. [Google Scholar]
- Zientek, M.L.; Czamanske, G.K.; Irvine, T.N. Stratigraphy and nomenclature for the Stillwater complex. Stillwater Complex Mont. Geol. Guide Mont. Bur. Mines Geol. Spec. Publ. 1985, 92, 21–32. [Google Scholar]
- Gervais, F.; Rifai, K.; Plamondon, P.; Özcan, L.; Doucet, F.; Vidal, F. Compositional tomography of a gold-bearing sample by Laser-induced breakdown spectroscopy. Terra Nova 2019, 31, 479–484. [Google Scholar] [CrossRef]
- Singh, V.; Rao, M. Application of image processing and radial basis neural network techniques for ore sorting and ore classification. Miner. Eng. 2005, 18, 1412–1420. [Google Scholar] [CrossRef]
- Duca, D.; Mancini, M.; Rossini, G.; Mengarelli, C.; Pedretti, E.F.; Toscano, G.; Pizzi, A. Soft independent modelling of class analogy applied to infrared spectroscopy for rapid discrimination between hardwood and softwood. Energy 2016, 117, 251–258. [Google Scholar] [CrossRef]
- Makvandi, S.; Ghasemzadeh-Barvarz, M.; Beaudoin, G.; Grunsky, E.C.; McClenaghan, M.B.; Duchesne, C.; Boutroy, E. Partial least squares-discriminant analysis of trace element compositions of magnetite from various VMS deposit subtypes: Application to mineral exploration. Ore Geol. Rev. 2016, 78, 388–408. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rifai, K.; Özcan, L.-Ç.; Doucet, F.R.; Rhoderick, K.; Vidal, F. Ultrafast Elemental Mapping of Platinum Group Elements and Mineral Identification in Platinum-Palladium Ore Using Laser Induced Breakdown Spectroscopy. Minerals 2020, 10, 207. https://doi.org/10.3390/min10030207
Rifai K, Özcan L-Ç, Doucet FR, Rhoderick K, Vidal F. Ultrafast Elemental Mapping of Platinum Group Elements and Mineral Identification in Platinum-Palladium Ore Using Laser Induced Breakdown Spectroscopy. Minerals. 2020; 10(3):207. https://doi.org/10.3390/min10030207
Chicago/Turabian StyleRifai, Kheireddine, Lütfü-Çelebi Özcan, François R. Doucet, Kyle Rhoderick, and François Vidal. 2020. "Ultrafast Elemental Mapping of Platinum Group Elements and Mineral Identification in Platinum-Palladium Ore Using Laser Induced Breakdown Spectroscopy" Minerals 10, no. 3: 207. https://doi.org/10.3390/min10030207
APA StyleRifai, K., Özcan, L.-Ç., Doucet, F. R., Rhoderick, K., & Vidal, F. (2020). Ultrafast Elemental Mapping of Platinum Group Elements and Mineral Identification in Platinum-Palladium Ore Using Laser Induced Breakdown Spectroscopy. Minerals, 10(3), 207. https://doi.org/10.3390/min10030207





