Rippite, K2(Nb,Ti)2(Si4O12)O(O,F), a New K-Nb-Cyclosilicate from Chuktukon Carbonatite Massif, Chadobets Upland, Krasnoyarsk Territory, Russia
Abstract
1. Introduction
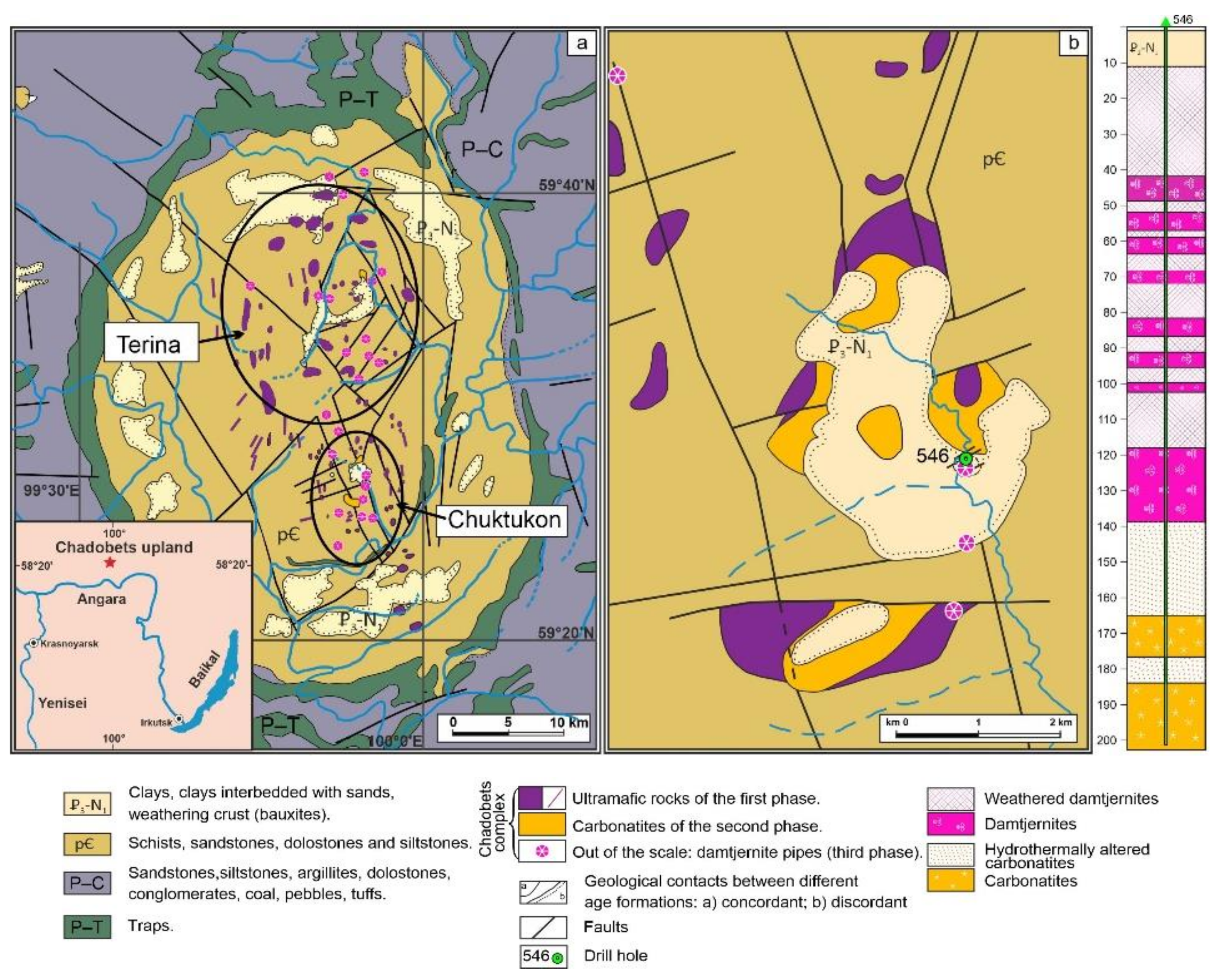
2. Brief Geological Background for the Chuktukon Massif
3. Analytical Methods
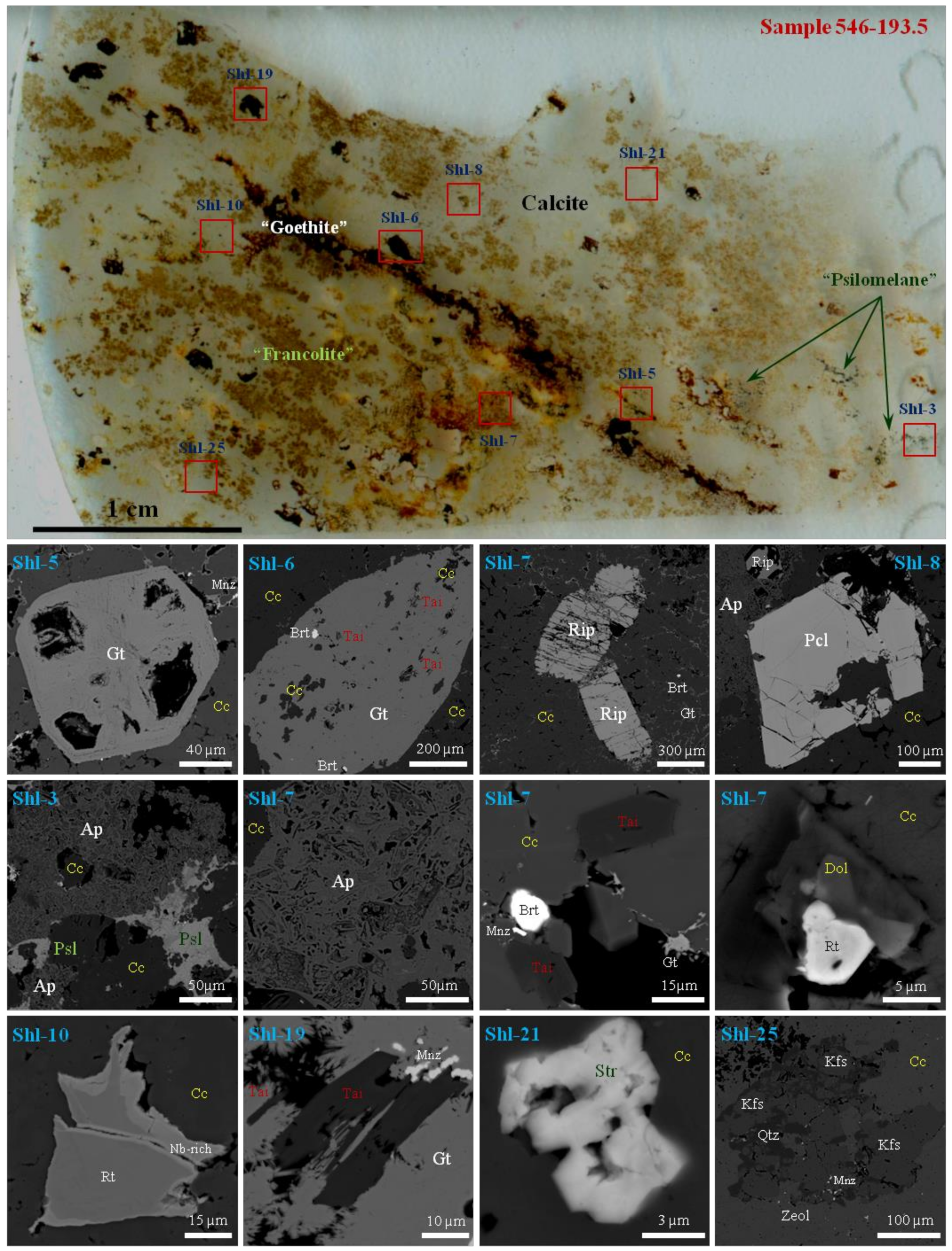
4. Mineralogy and Petrography of Calciocarbonatites from the Chuktukon Massif
5. Morphology, Optical and Physical Properties of Rippite
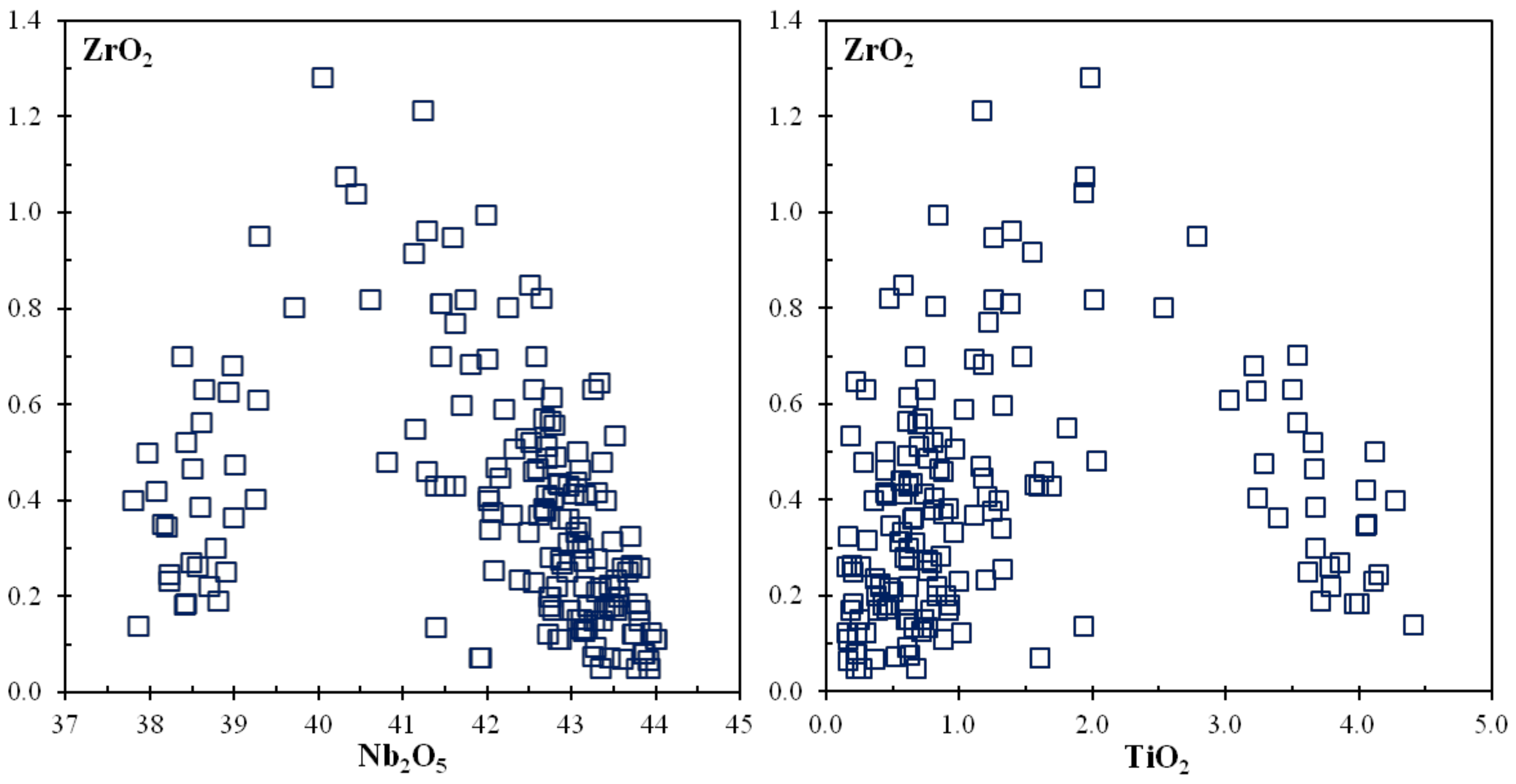
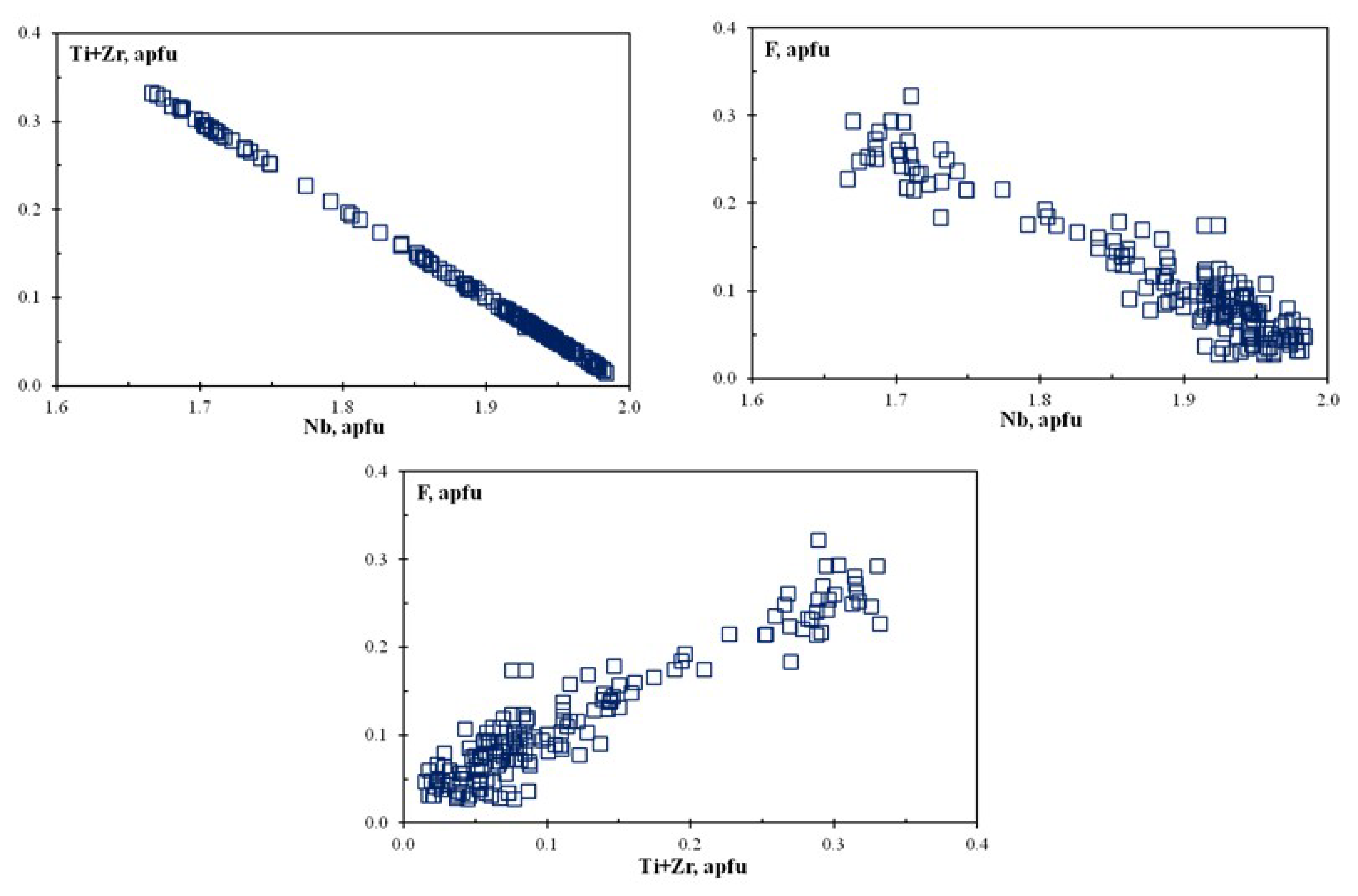
6. Chemical Composition of Rippite
6.1. SEM and EMPA data
6.2. LA-ICP-MS and SIMS Data
7. Optical Studies for Rippite
7.1. Cathodoluminescence
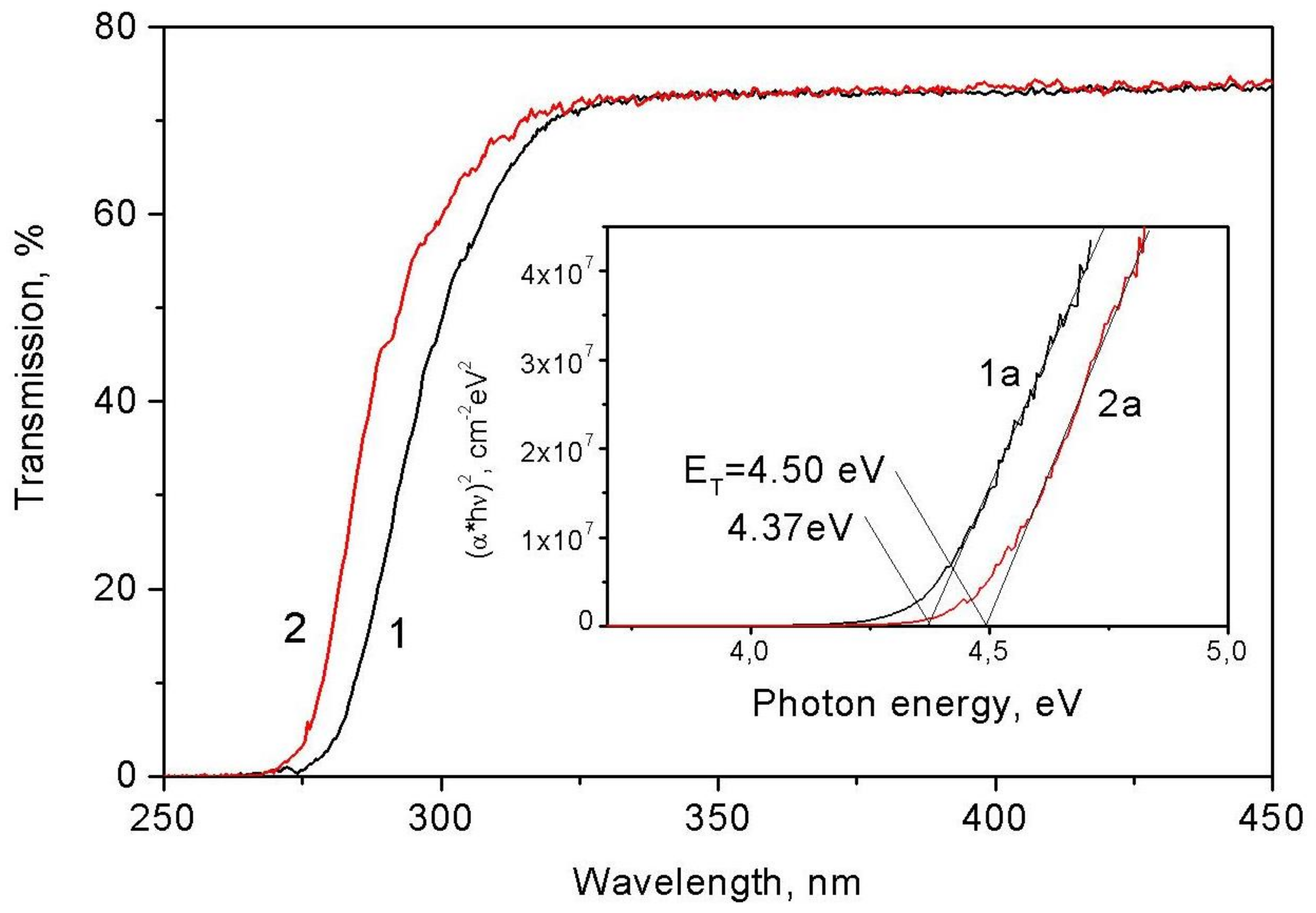
7.2. Transmission Spectra
7.3. Infrared and Raman Spectroscopy
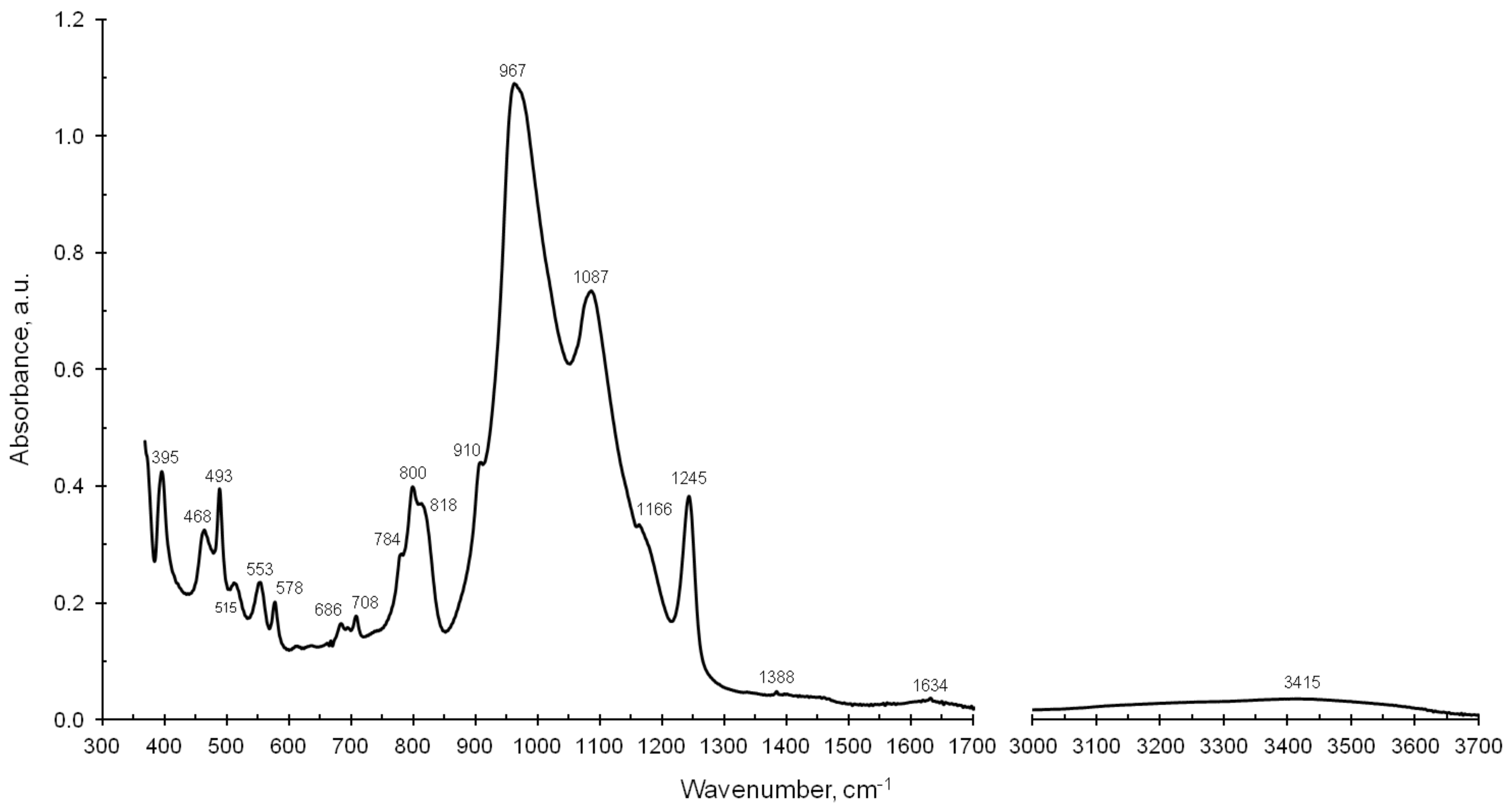
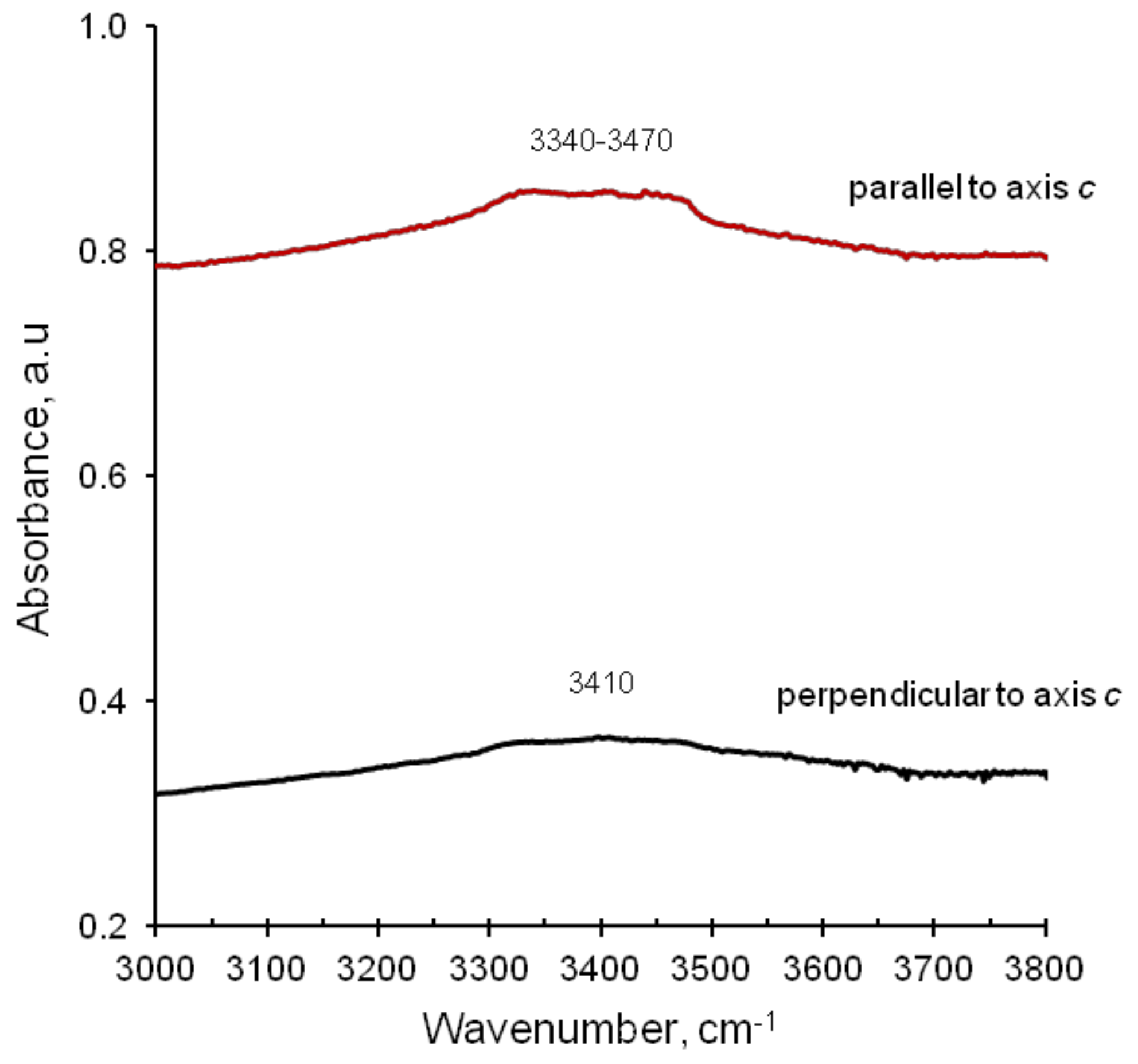
7.3.1. Infrared Spectra
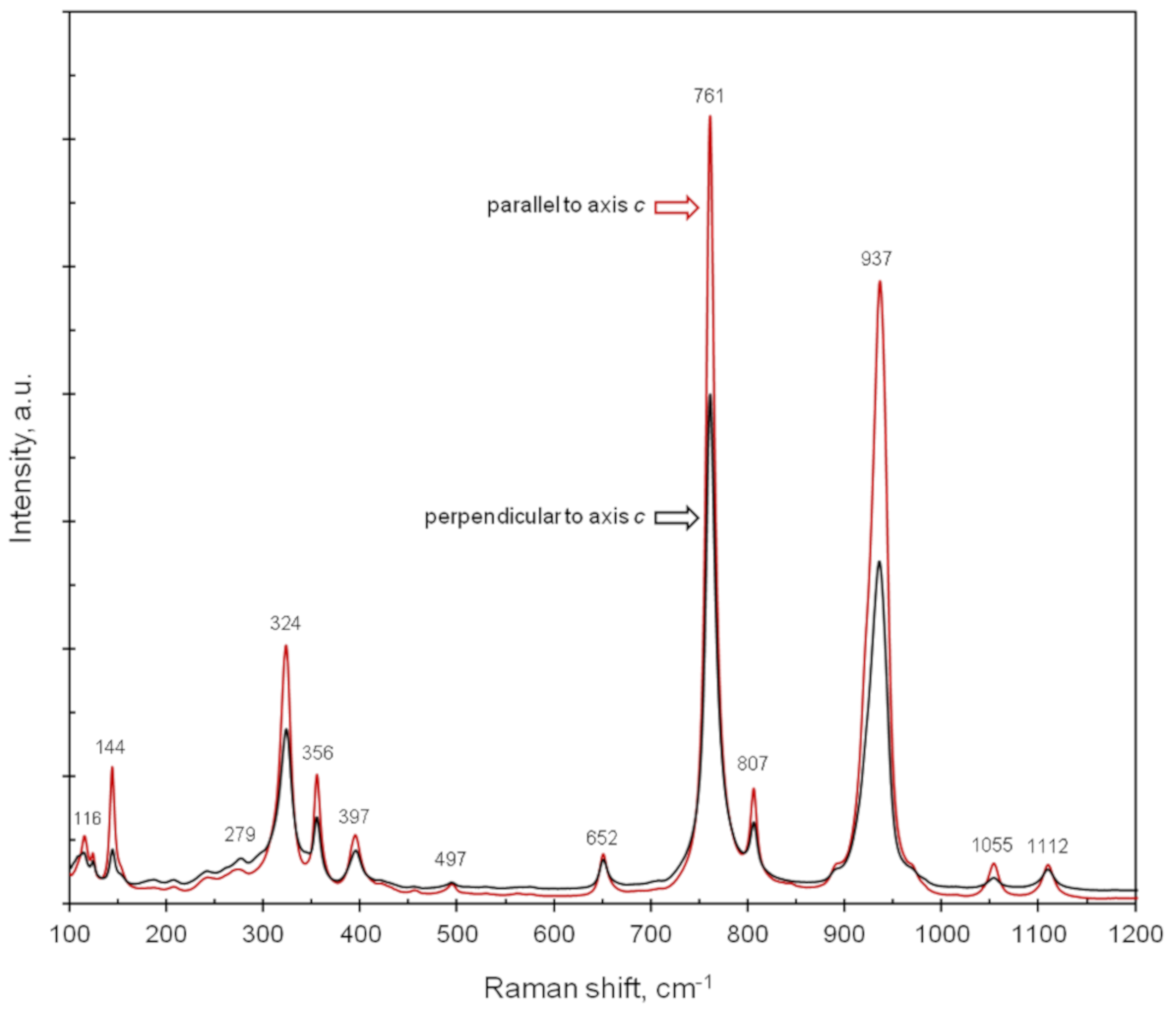
7.3.2. Raman Spectra
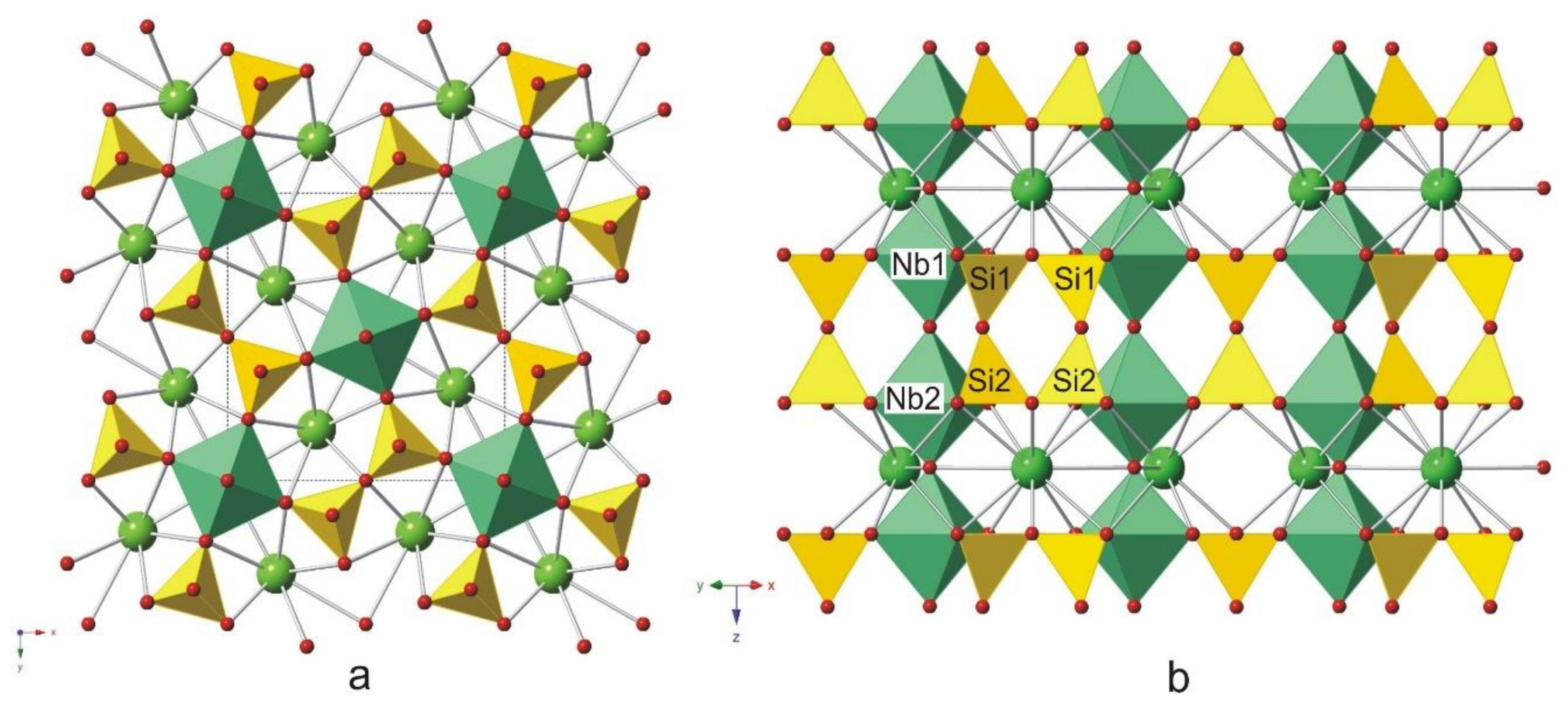
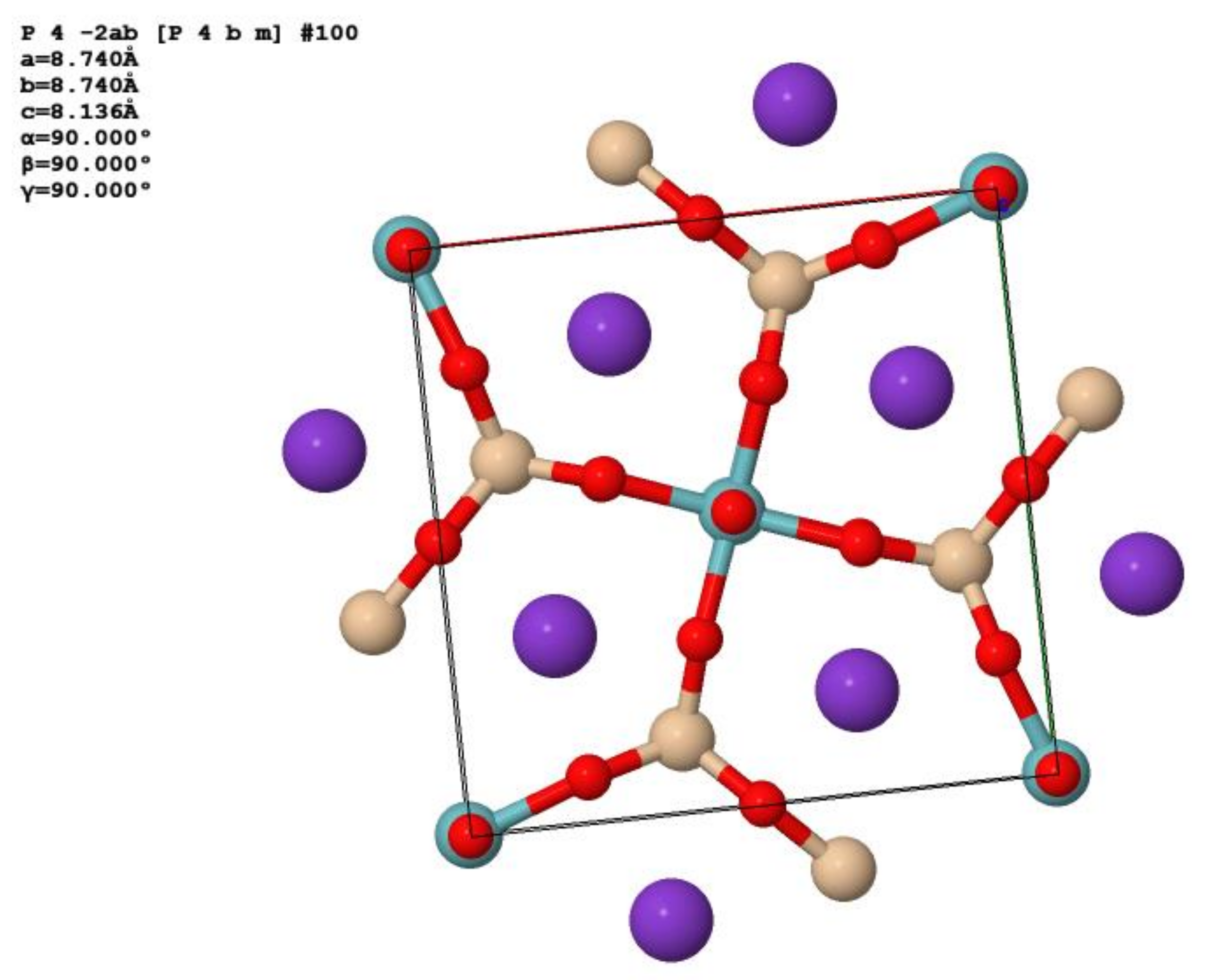
8. X-ray Crystallography and Crystal Structure of Rippite
9. Discussion and Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Doroshkevich, A.G.; Sharygin, V.V.; Seryotkin, Y.V.; Karmanov, N.S.; Belogub, E.V.; Moroz, T.N.; Nigmatulina, E.N.; Eliseev, A.P.; Vedenyapin, V.N.; Kupriyanov, I.N. Rippite, IMA 2016-025. CNMNC Newsletter no. 32, August 2016, page 919. Mineral. Mag. 2016, 80, 915–922. [Google Scholar]
- Sharygin, V.V.; Doroshkevich, A.G.; Seryotkin, Y.V.; Karmanov, N.S.; Belogub, E.V.; Moroz, T.N. A new K-Nb-cyclosilicate K2(Nb,Ti)2(Si4O12)O(O,F) from Chuktukon carbonatite massif, Chadobets upland, Russia. In Proceedings of the Abstract Volume of Second European Mineralogical Conference, Rimini, Italy, 11–15 September 2016; p. 421. [Google Scholar]
- Crosnier, M.P.; Guyomard, D.; Verbaere, A.; Piffard, Y.; Tournoux, M. K2(NbO)2Si4O12: A new material for non-linear optics. Ferroelectrics 1991, 124, 61–66. [Google Scholar] [CrossRef]
- Foster, M.C.; Arbogast, D.J.; Photinos, P.; Nielson, R.M.; Abrahams, S.C. K2(NbO)2Si4O12: A new ferroelectric. J. Appl. Crystallogr. 1999, 32, 421–425. [Google Scholar] [CrossRef]
- Crosnier, M.P.; Guyomard, D.; Verbaere, A.; Piffard, Y.; Tournoux, M. The potassium niobyl cyclotetrasilicate K2(NbO)2Si4O12. J. Solid State Chem. 1992, 98, 128–132. [Google Scholar] [CrossRef]
- Tanaka, H.; Yamamoto, M.; Takahashi, Y.; Benino, Y.; Fujiwara, T.; Komatsu, T. Crystalline phases and second harmonic intensities in potassium niobium silicate crystallized glasses. Opt. Mater. 2003, 22, 71–79. [Google Scholar] [CrossRef]
- Pernice, P.; Aronne, A.; Sigaev, V.N.; Sarkisov, P.D.; Molev, V.I.; Stefanovich, S.Y. Crystallization behavior of potassium niobium silicate glasses. J. Am. Ceram. Soc. 1999, 82, 3447–3452. [Google Scholar] [CrossRef]
- Sigaev, V.N.; Lotarev, S.V.; Ryzhenkov, V.S.; Golubev, N.V.; Stefanovich, S.Y.; Champagnon, B.; Vouagner, D.; Nardou, E.; Porcar, L.; Paleari, A.; et al. Nano-heterogeneous structure of (1-x)KNbO3–xSiO2 glasses in the low glass-forming oxide content range 0.05 ≤ x ≤ 0.3. J. Non-Cryst. Solids 2011, 357, 3136–3142. [Google Scholar] [CrossRef]
- Sigaev, V.N.; Ryzhenkov, V.S.; Lotarev, S.V.; Golubev, N.V.; Stefanovich, S.Y.; Okada, A. Glasses and their crystallization in the (1-x)KNbO3—xSiO2 system at low glass-forming oxide contents, 0 ≤ x ≤ 0.35. J. Non-Cryst. Solids 2010, 356, 958–965. [Google Scholar] [CrossRef]
- Sigaev, V.N.; Stefanovich, S.Y.; Champagnon, B.; Gregora, I.; Pernice, P.; Aronne, A.; LeParc, R.; Sarkisov, P.D.; Dewhurst, C. Amorphous nanostructuring in potassium niobium silicate glasses by SANS and SHG: A new mechanism for second-order optical non-linearity of glasses. J. Non-Cryst. Solids 2002, 306, 238–248. [Google Scholar] [CrossRef]
- Aronne, A.; Sigaev, V.N.; Pernice, P.; Fanelli, E.; Usmanova, L.Z. Non-isothermal crystallization and nanostructuring in potassium niobium silicate glasses. J. Non-Cryst. Solids 2004, 337, 121–129. [Google Scholar] [CrossRef]
- Santos, R.; Santos, L.F.; Almeida, R.M.; Deubener, J.; Wondraczek, L. Crystallization of niobium germanosilicate glasses. J. Solid State Chem. 2010, 183, 128–135. [Google Scholar] [CrossRef]
- Vernacotola, D.E.; Shelby, J.E. Potassium niobium silicate glasses. Phys. Chem. Glasses 1994, 35, 153–159. [Google Scholar]
- Sahashi, A.; Hoshina, T.; Takeda, H.; Tsurumi, T. Fabrication of ferroelectric silicate KNbSi2O7 single crystal. J. Ceram. Soc. Jpn. 2014, 122, 389–392. [Google Scholar] [CrossRef][Green Version]
- Gopalakrishnan, J.; Ramesha, K.; Rangan, K.K.; Pandey, S. In Search of Inorganic Nonlinear Optical Materials for Second Harmonic Generation. J. Solid State Chem. 1999, 148, 75–80. [Google Scholar] [CrossRef]
- Lee, J.-G.; Höhn, P.; Greenblatt, M. A potassium tantalum (V) tetrasilicate KTaSi2O7. J. Solid State Chem. 1996, 123, 123–128. [Google Scholar] [CrossRef]
- Ripp, G.S.; Khodanovich, P.Y.; Smirnova, O.K. New data on the carbonatite nature of the Khalyuta deposit. Dokl. Earth Sci. 1999, 365, 351–353. [Google Scholar]
- Ripp, G.S.; Khodanovich, P.Y.; Smirnova, O.K. A new carbonatite province in West Transbaikalia. Geologiya i Geofizika 1999, 40, 73–81. [Google Scholar]
- Ripp, G.S.; Badmatsyrenov, M.V.; Skulyberdin, A.A. A new carbonatite occurrence in Northern Transbaikalia. Petrology 2002, 10, 391–394. [Google Scholar]
- Ripp, G.S.; Badmatsyrenov, M.V.; Doroshkevich, A.G.; Izbrodin, I.A. New carbonatite-bearing area in northern Transbaikalia. Petrology 2005, 13, 489–498. [Google Scholar]
- Ripp, G.S.; Doroshkevich, A.G.; Karmanov, N.S.; Kanakin, S.V. Micas from the Khaluta carbonatite deposit, western Transbaikal region. Geol. Ore Depos. 2009, 51, 812–821. [Google Scholar] [CrossRef]
- Ripp, G.S.; Doroshkevich, A.G.; Posokhov, V.F. Age of carbonatite magmatism in Transbaikalia. Petrology 2009, 17, 73–89. [Google Scholar] [CrossRef]
- Ripp, G.S.; Doroshkevich, A.G.; Posokhov, V.F.; Izbrodin, I.A.; Konopel’ko, D.L.; Sergeev, S.A. The age of carbonatites and mafic rocks (SHRIMP II and Rb-Sr dating) from the Oshurkovo apatite-bearing pluton (western Transbaikalia). Russ. Geol. Geophys. 2011, 52, 517–525. [Google Scholar] [CrossRef]
- Doroshkevich, A.G.; Ripp, G.S. Estimation of the conditions of formation of REE-carbonatites in western Transbaikalia. Geologiya i Geofizika 2004, 45, 492–500. [Google Scholar]
- Doroshkevich, A.G.; Wall, F.; Ripp, G.S. Calcite-bearing dolomite carbonatite dykes from Veseloe, North Transbaikalia, Russia and possible Cr-rich mantle xenoliths. Mineral. Petrol. 2007, 90, 19–49. [Google Scholar] [CrossRef]
- Doroshkevich, A.G.; Wall, F.; Ripp, G.S. Magmatic graphite in dolomite carbonatite at Pogranichnoe, North Transbaikalia, Russia. Contrib. Mineral. Petrol. 2007, 153, 339–353. [Google Scholar] [CrossRef]
- Doroshkevich, A.G.; Ripp, G.S.; Viladkar, S.G.; Vladykin, N.V. The Arshan REE carbonatites, southwestern Transbaikalia, Russia: Mineralogy, paragenesis and evolution. Can. Mineral. 2008, 46, 807–823. [Google Scholar] [CrossRef]
- Doroshkevich, A.G.; Ripp, G.S.; Moore, K.R. Genesis of the Khaluta alkaline-basic Ba-Sr carbonatite complex (West Transbaikalia, Russia). Mineral. Petrol. 2010, 98, 245–268. [Google Scholar] [CrossRef]
- Doroshkevich, A.G.; Chebotarev, D.A.; Sharygin, V.V.; Prokopyev, I.R.; Nikolenko, A.M. Petrology of alkaline silicate rocks and carbonatites of the Chuktukon massif, Chadobets upland, Russia: Sources, evolution and relation to the Triassic Siberian LIP. Lithos 2019, 332–333, 245–260. [Google Scholar] [CrossRef]
- Doroshkevich, A.G.; Sharygin, V.V.; Belousova, E.A.; Izbrodin, I.A.; Prokopyev, I.R. Zircon from the Chuktukon alkaline ultramafic carbonatite complex (Chadobets uplift, Siberian craton) as evidence of source heterogeneity. Lithos 2020. submitted. [Google Scholar]
- Chebotarev, D.A.; Doroshkevich, A.G.; Sharygin, V.V.; Yudin, D.S.; Ponomarchuk, A.V.; Sergeev, S.A. Geochronology of the Chuktukon carbonatite massif, Chadobets uplift (Krasnoyarsk Territory). Russ. Geol. Geophys. 2017, 58, 1222–1231. [Google Scholar] [CrossRef]
- Chebotarev, D.A.; Doroshkevich, A.G.; Klemd, R.; Karmanov, N.S. Evolution of Nb-mineralization in the Chuktukon carbonatite massif, Chadobets upland (Krasnoyarsk Territory, Russia). Periodico di Mineralogia 2017, 86, 99–118. [Google Scholar] [CrossRef]
- Sharygin, V.V. Tainiolite from Chuktukon Carbonatite Massif, Chadobets Upland, Russia. In Proceedings of the Abstract Volume of XXXIV International Conference “Magmatism of the Earth and Related Strategic Metal Deposits”, Miass, Russia, 4–9 August 2017; pp. 242–244. [Google Scholar]
- Sharygin, V.V.; Doroshkevich, A.G. Multiphase inclusions in zircons from Chuktukon carbonatite massif, Chadobets upland, Russia. In Proceedings of the Abstract Volume of XXXIV International Conference “Magmatism of the Earth and Related Strategic Metal Deposits”, Miass, Russia, 4–9 August 2017; pp. 244–247. [Google Scholar]
- Sharygin, V.V.; Doroshkevich, A.G.; Chebotarev, D.A. Na-Sr-Ba-REE-carbonates and phosphates in minerals of Chuktukon massif carbonatites, Chadobets upland, Krasnoyarsk territory. In Proceedings of the Abstract Volume of XVII Russian Fluid Inclusion Conference, Ulan-Ude, Russia, 12–17 September 2016; pp. 180–182. (In Russian). [Google Scholar]
- Kirichenko, V.T.; Zuev, V.K.; Perfilova, O.Y.; Sosnovskaya, O.V.; Smokotina, I.; Markovich, L.A.; Borodin, M.E.; Mironyuk, E. State Geological Map of Russian Federation, Scale 1:1000000 (Third Generation); Ser. Angaro-Yeniseysk. Sheet O-47 Bratsk. Explanatory Note; Cartografic Factory of VSEGEI: St. Petersburg, Russia, 2012; pp. 163–179. (In Russian) [Google Scholar]
- Dashkevich, N.N. Regional prediction of kimberlite magmatism in the southwestern Siberian Platform. In Geology and Mineral Resources of Krasnoyarsk Territory; Krasnoyarsk, Russia, 1999; pp. 31–42. (In Russian) [Google Scholar]
- Starosel’tsev, V.S. Identifying paleorifts as promising tectonic elements for active oil and gas generation. Russ. Geol. Geophys. 2009, 50, 358–364. [Google Scholar] [CrossRef]
- Prokopyev, I.; Starikova, A.; Doroshkevich, A.; Nugumanova, Y.; Potapov, V. Petrogenesis of ultramafic lamprophyres from the Terina complex (Chadobets upland, Russia): Mineralogy and melt inclusion composition. Minerals 2020, 10, 419. [Google Scholar] [CrossRef]
- Tsikyna, S.V. The Nb-REE Chuktukon Deposit: Modelling, Ore Typification and Assessment of Prospects. Ph.D. Thesis, The Krasnoyarsk Research Institute of Geology and Mineral Resources, Krasnoyarsk, Russia, 2004. (In Russian). [Google Scholar]
- Lomayev, V.G.; Serdyuk, S.S. The Chuktukon Nb-REE deposit—The priority object for modernization of the Russian rare-earth industry. J. Sib. Fed. Univ. Eng. Technol. 2011, 4, 132–154. (In Russian) [Google Scholar]
- Serdyuk, S.S.; Lomayev, V.G.; Kuzmin, V.I.; Flett, D.S.; Gudkova, N.V.; Kuzmin, D.V.; Mulagaleeva, M.A.; Kuzmina, V.N.; Kalyakin, S.N.; Logutenko, O.A. The Chuktukon niobium-rare earth metals deposit: Geology and investigation into the processing options of the ores. Miner. Eng. 2017, 113, 8–14. [Google Scholar] [CrossRef]
- Nosova, A.A.; Kargin, A.V.; Sazonova, L.V.; Dubinina, E.O.; Chugaev, A.V.; Lebedeva, N.M.; Yudin, D.S.; Larionova, Y.O.; Abersteiner, A.; Gareev, B.I.; et al. Sr-Nd-Pb isotopic systematic and geochronology of ultramafic alkaline magmatism of the southwestern margin of the Siberian Craton: Metasomatism of the sub-continental lithospheric mantle related to subduction and plume events. Lithos 2020, 364, 105509. [Google Scholar] [CrossRef]
- Kargin, A.V.; Nosova, A.A.; Postnikov, A.V.; Chugaev, A.V.; Postnikova, O.V.; Popova, L.P.; Poshibaev, V.V.; Sazonova, L.V.; Dokuchaev, A.Y.; Smirnova, M.D. Devonian ultramafic lamprophyre in the Irkineeva–Chadobets trough in the southwest of the Siberian Platform: Age, composition, and implications for diamond potential prediction. Geol. Ore Depos. 2016, 58, 383–403. [Google Scholar] [CrossRef]
- Nosova, A.A.; Sazonova, L.V.; Kargin, A.V.; Smirnova, M.D.; Lapin, A.V.; Shcherbakov, V.D. Olivine in ultramafic lamprophyres: Chemistry, crystallisation, and melt sources of Siberian pre- and post-trap aillikites. Contrib. Mineral. Petrol. 2018, 173, 55. [Google Scholar] [CrossRef]
- Lapin, A.V. About kimberlites of the Chadobets upland in connection with a problem of the formational-metallogeny analysis of the platform alkaline ultrabasic magmatic rocks. Otechestvennaya Geol. 2001, 4, 30–35. (In Russian) [Google Scholar]
- Lapin, A.V.; Lisitsyn, D.V. About mineralogical typomorphism of alkaline ultrabasic magmatic rocks of Chadobets upland. Otechestvennaya Geol. 2004, 6, 83–92. (In Russian) [Google Scholar]
- Jochum, K.P.; Dingwell, D.B.; Rocholl, A.; Stoll, B.; Hofmann, A.W.; Becker, S.; Besmehn, A.; Bessette, D.; Dietze, H.-J.; Dulski, P.; et al. The preparation and preliminary characterisation of eight geological MPI-DING reference glasses for in-situ microanalysis. Geostand. Newsl. J. Geostand. Geoanal. 2000, 24, 87–133. [Google Scholar] [CrossRef]
- Pearce, N.J.G.; Perkins, W.T.; Westgate, J.A.; Gorton, M.P.; Jackson, S.E.; Neal, C.R.; Chenery, S.P. A compilation of new and published major and trace element data for NIST SRM 610 and NIST SRM 612 glass reference materials. Geostand. Newsl. J. Geostand. Geoanal. 1997, 21, 115–144. [Google Scholar] [CrossRef]
- Van Achterbergh, E.; Ryan, C.G.; Jackson, S.E.; Griffin, W.L. Data reduction software for LA-ICP-MS: Appendix. In Laser Ablation-ICP-mass Spectrometry in the Earth Sciences: Principles and Applications; MAC Short Course Series; Sylvester, P.J., Ed.; Mineralog. Assoc. Canada (MAC): Ottawa, ON, Canada, 2001; Volume 29, pp. 239–243. [Google Scholar]
- Tauc, J. Optical properties and electronic structure of amorphous Ge and Si. Mater. Res. Bull. 1967, 3, 37–46. [Google Scholar] [CrossRef]
- Nikogosyan, D. Nonlinear optical crystals. In Complete Survey; Springer Science + Business Media Inc.: New York, NY, USA, 2005. [Google Scholar]
- Yelisseyev, A.P.; Isaenko, L.I.; Starikova, M.K. Optical study of defects in lithium iodate α-LiIO3. J. Opt. Soc. Am. B Opt. Phys. 2012, 29, 1430–1435. [Google Scholar] [CrossRef]
- Sirotin, Y.I.; Shaskolskaya, M.P. Fundamentals of Crystal Physics; Nauka: Moscow, Russia, 1975. (In Russian) [Google Scholar]
- Moroz, T.N.; Palchik, N.A. The uniqueness of determination of the space group symmetry by means of the vibrational spectroscopy methods. Crystallogr. Rep. 2009, 54, 734–737. [Google Scholar] [CrossRef]
- Rastsvetaeva, R.K.; Tamazyan, R.A.; Pushcharovsky, D.Y.; Nadezhina, T.N.; Voloshin, A.V. K-nenadkevichite, a new representative of the nenadkevichite-labuntsovite series. Crystallogr. Rep. 1994, 39, 908–914. [Google Scholar]
- Rastsvetaeva, R.K.; Tamazyan, R.A.; Pushcharovsky, D.Y.; Nadezhina, T.N. Crystal structure and microtwinning of K-rich nenadkevichite. Eur. J. Mineral. 1994, 6, 503–509. [Google Scholar] [CrossRef]
- Subbotin, V.V.; Voloshin, A.V.; Pakhomovskii, Y.A.; Bakhchisaraitsev, A.Y.; Pushcharovsky, D.Y.; Rastsvetaeva, R.K.; Nadezhina, T.N. Vuoriyarvite (K,Na)2(Nb,Ti)2Si4O12(O,OH)2·4H2O—A new mineral from carbonatites of the Vuoriyarvi massif, Kola Peninsula. Dokl. Akad. Nauk 1998, 358, 73–75. [Google Scholar]
- Chukanov, N.V.; Pekov, I.V.; Khomyakov, A.P. Recommended nomenclature for labuntsovite-group minerals. Eur. J. Mineral. 2002, 14, 165–173. [Google Scholar] [CrossRef]
- Chukanov, N.V.; Pekov, I.V.; Zadov, A.E.; Voloshin, A.V.; Subbotin, V.V.; Sorokhtina, N.V.; Rastsvetaeva, R.K.; Krivovichev, S.V. Minerals of the Labuntsovite Group; PH Nauka: Moscow, Russia, 2003; 323p. (In Russian) [Google Scholar]
- Rastsvetaeva, R.K.; Bolotina, N.B.; Pushcharovskii, D.Y.; Stefanovich, S.Y.; Nadezhina, T.N.; Dimitrova, O.V. K4(NbO)2[Si8O21]: Synthesis, structure, and properties. Crystallogr. Rep. 1994, 39, 915–922. [Google Scholar]
- Rastsvetaeva, R.K.; Bolotina, N.B.; Pushcharovskii, D.Y.; Nadezhina, T.N.; Dimitrova, O.V. Modulated crystal structure of K4(NbO)2[Si8O21]. J. Alloys Compd. 1994, 209, 145–150. [Google Scholar] [CrossRef]
- Pushcharovskii, D.Y. New approaches to the XRD studies of complicated crystal chemical phenomena in minerals. Croat. Chem. Acta 1999, 72, 135–145. [Google Scholar]
- Mitchell, R.H. Primary and secondary niobium mineral deposits associated with carbonatites. Ore Geol. Rev. 2015, 64, 626–641. [Google Scholar] [CrossRef]
- Traversa, G.; Gomes, C.B.; Brotzu, P.; Buraglini, N.; Morbidelli, L.; Principato, M.S.; Ronca, S.; Ruberti, E. Petrography and mineral chemistry of carbonatites and mica-rich rocks from the Araxá complex (Alto Paranaíba Province, Brazil). An. Acad. Bras. Ci. 2001, 73, 71–98. [Google Scholar] [CrossRef]



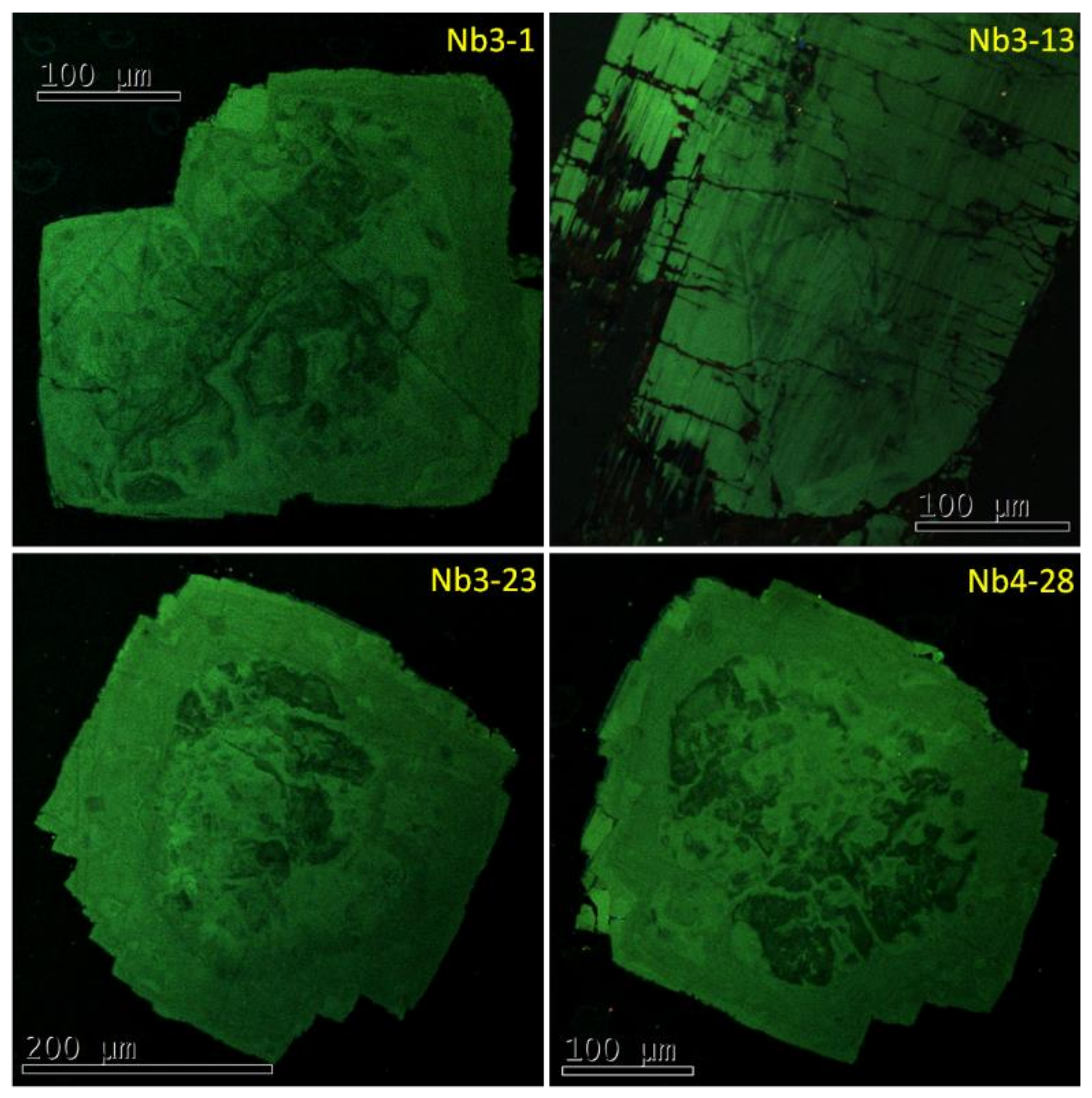

| Mineral | Formula | Mineral | Formula |
|---|---|---|---|
| Pyrite | FeS2 | Ancylite-(Ce) | (Ce,La,Nd)Sr(CO3)2(OH) H2O |
| Fluorite | CaF2 | Daqingshanite-(Ce) | (Sr,Ca,Ba)3(Ce,La)(CO3)3-x(PO4)(OH,F)2x |
| Neighborite | NaMgF3 | Quartz | SiO2 |
| Parascandolaite | KMgF3 | Rutile | TiO2 |
| Fluorapatite | Ca5(PO4)3F | Baddeleyite | ZrO2 |
| Carbonate-fluorapatite | (Ca,Sr,Na)5(PO4,CO3)3F | Thorianite | ThO2 |
| Monazite-(Ce) | (Ce,La,Nd)PO4 | Hematite | Fe2O3 |
| Ferrohagendorfite | NaCaFe2+Fe2+2(PO4)3 | Laachite | Ca2Zr2Nb2TiFe2+O14 |
| Florencite-(Ce) | (Ce,La.Nd)Al3(PO4)2(OH)6 | “Nb-Fe3+-zirconolite” | Ca2Zr2NbTi2Fe3+O14 |
| “Sr-Ba-Ca-Na-REE- phosphate” | (Na,REE)Ca2.5(Sr,Ba)3 (PO4)4(OH) | Fluorcalcio- pyrochlore | (Ca,Na)2(Nb,Ti)2O6F |
| “Na-Ca-Fe-Mn- phosphate” | Na4.8Ca0.6(Fe,Mn)3(PO4)4 | Hydroxykeno- pyrochlore | (□,Ce,Ba)2(Nb,Ti)2O6(OH,F) |
| “Na-REE-Ca-Sr-Ba- phosphate” | Na1.6REE0.6(Ba,Sr,Ca)0.8(PO4)O | Hydropyrochlore | (H2O,Ba,Sr,□)2Nb2(O,OH)6(H2O) |
| “Ba-Sr-Ca-REE-H2O- aluminophosphate” | (Ba,Sr,Ca,REE)Al(PO4) nH2O | Hydroxyplumbo- pyrochlore | (Pb1.5□0.5)Nb2O6(OH) |
| Churchite-(Y) | Y(PO4) 2H2O | Goethite | α-FeOOH |
| Anhydrite | CaSO4 | “Hydrogoethite” | 3Fe2O3·4H2O |
| Barite | BaSO4 | Bernalite | Fe(OH)3 nH2O |
| Celestine | SrSO4 | Ferrihydrite | Fe3+10O14(OH) |
| Calcite | CaCO3 | Hollandite | Ba(Mn4+6Mn3+2)O16 |
| Strontianite | SrCO3 | Romanèchite | (Ba,H2O)2(Mn4+,Mn3+)5O10 |
| Dolomite | CaMg(CO3)2 | K-feldspar | KAlSi3O8 |
| Ankerite | Ca(Fe2+,Mg,Mn)(CO3)2 | Diopside | CaMgSi2O6 |
| Olekminskite | Sr(Sr,Ca,Ba)(CO3)2 | Aegirine | NaFe3+Si2O6 |
| Nyerereite | Na2Ca(CO3)2 | Tainiolite | KMg2Li(Si3O10)F2 |
| Shortite | Na2Ca2(CO3)3 | Rippite | K2(Nb,Ti)2(Si4O12)O(O,F) |
| Synchysite-(Ce) | Ca(Ce,La,Nd)(CO3)2F | Zircon | ZrSiO4 |
| Parisite-(Ce) | Ca(Ce,La,Nd)2(CO3)3F2 | Thorite | ThSiO4 |
| Burbankite | (Na,Ca)3(Sr,Ba,Ce)3(CO3)5 | Kaolinite | Al2(Si2O5)(OH)4 |
| Khanneshite | (Na,Ca)3(Ba,Sr,Ce,Ca)3(CO3)5 | “K-Ca-zeolite” | K3Ca5(Al3Si9O29)∙7–8H2O |
| Component | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grain | Nb3-1 | Nb3-18 | Nb3-13 | Nb3-23 | Nb4-28 | ||||||||||||||
| Position | c | or | c | m | r | or | or | c | r | c | r | c | r | inc | Ideal | ||||
| n | 120 | sd | Range | 22 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 5 | |
| SiO2 | 40.28 | 0.23 | 40.07–41.02 | 40.17 | 40.07 | 40.92 | 40.17 | 40.26 | 40.54 | 40.63 | 41.02 | 40.25 | 40.34 | 40.09 | 40.21 | 40.13 | 40.23 | 40.27 | 40.03 |
| TiO2 | 1.11 | 1.06 | 0.15–4.41 | 0.52 | 0.15 | 4.05 | 0.58 | 1.18 | 2.01 | 3.02 | 4.41 | 0.45 | 1.39 | 0.26 | 0.74 | 0.57 | 0.82 | 0.91 | 0.00 |
| ZrO2 | 0.37 | 0.26 | 0.05–1.28 | 0.17 | 0.26 | 0.35 | 0.85 | 0.68 | 0.82 | 0.61 | 0.14 | 0.42 | 0.96 | 0.26 | 0.63 | 0.33 | 0.80 | 0.26 | |
| Nb2O5 | 42.30 | 1.61 | 38.23–44.00 | 43.37 | 43.81 | 38.20 | 42.51 | 41.81 | 40.61 | 39.28 | 37.87 | 43.30 | 41.29 | 43.61 | 42.55 | 43.06 | 42.25 | 42.72 | 44.28 |
| Na2O | 0.01 | 0.02 | 0.00–0.06 | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.04 | 0.00 | 0.03 | 0.00 | 0.00 | 0.00 | 0.02 | 0.02 | 0.03 | |
| K2O | 15.77 | 0.09 | 15.68–16.07 | 15.72 | 15.71 | 16.03 | 15.74 | 15.77 | 15.89 | 15.86 | 16.07 | 15.74 | 15.80 | 15.71 | 15.76 | 15.71 | 15.74 | 15.74 | 15.69 |
| F | 0.32 | 0.22 | 0.09–0.95 | 0.13 | 0.15 | 0.91 | 0.25 | 0.37 | 0.56 | 0.69 | 0.95 | 0.15 | 0.50 | 0.19 | 0.38 | 0.30 | 0.32 | 0.28 | 0.00 |
| H2O | 0.18 | 0.05 | 0.12–0.26 | 0.18 | |||||||||||||||
| Sum | 100.34 | 100.27 | 100.15 | 100.46 | 100.10 | 100.07 | 100.43 | 100.13 | 100.46 | 100.33 | 100.28 | 100.12 | 100.27 | 100.12 | 100.18 | 100.20 | 100.00 | ||
| O-F2 | 0.14 | 0.05 | 0.06 | 0.38 | 0.11 | 0.16 | 0.24 | 0.29 | 0.40 | 0.06 | 0.21 | 0.08 | 0.16 | 0.13 | 0.13 | 0.12 | 0.00 | ||
| Sum | 100.20 | 100.22 | 100.09 | 100.07 | 99.99 | 99.92 | 100.19 | 99.84 | 100.06 | 100.27 | 100.07 | 100.04 | 100.11 | 99.99 | 99.99 | 100.08 | 100.00 | ||
| Formula based on 8 cations | |||||||||||||||||||
| Si | 4.000 | 4.000 | 3.999 | 3.999 | 4.001 | 4.001 | 4.000 | 4.001 | 4.001 | 4.000 | 4.000 | 4.001 | 4.000 | 4.000 | 4.000 | 4.001 | 4.000 | ||
| Ti | 0.083 | 0.039 | 0.011 | 0.298 | 0.043 | 0.088 | 0.149 | 0.224 | 0.324 | 0.033 | 0.104 | 0.019 | 0.055 | 0.042 | 0.061 | 0.068 | 0.000 | ||
| Zr | 0.018 | 0.008 | 0.013 | 0.016 | 0.041 | 0.033 | 0.039 | 0.029 | 0.007 | 0.020 | 0.047 | 0.013 | 0.031 | 0.016 | 0.039 | 0.012 | |||
| Nb | 1.899 | 1.953 | 1.977 | 1.688 | 1.914 | 1.878 | 1.811 | 1.748 | 1.670 | 1.945 | 1.851 | 1.967 | 1.914 | 1.940 | 1.899 | 1.919 | 2.000 | ||
| K + Na | 2.000 | 2.000 | 2.000 | 2.000 | 2.000 | 1.999 | 2.000 | 2.000 | 1.999 | 2.001 | 1.999 | 2.000 | 2.000 | 2.001 | 2.000 | 2.000 | 2.000 | ||
| F | 0.101 | 0.041 | 0.049 | 0.281 | 0.079 | 0.116 | 0.175 | 0.215 | 0.293 | 0.046 | 0.157 | 0.060 | 0.120 | 0.095 | 0.101 | 0.088 | 0.000 | ||
| OH | 0.118 | 0.119 | |||||||||||||||||
| End–members, mol.% | |||||||||||||||||||
| K2Nb2(Si4O12)O2 | 0.95 | 0.98 | 0.99 | 0.84 | 0.96 | 0.94 | 0.91 | 0.87 | 0.83 | 0.97 | 0.93 | 0.98 | 0.96 | 0.97 | 0.95 | 0.96 | 1.00 | ||
| K2Ti2(Si4O12)(F,OH)2 | 0.05 | 0.02 | 0.01 | 0.16 | 0.04 | 0.06 | 0.09 | 0.13 | 0.17 | 0.03 | 0.07 | 0.02 | 0.04 | 0.03 | 0.05 | 0.04 | 0.00 | ||
| Sample | 514-231 | 514-231 | 549-192 | 549-192 | 549-192 | 549-192 | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Grain | R8-5 | R8-4 | R9-Rip2 | R9-Rip3 | R5-7 | R5-3 | |||||
| Position | c | r | c | r | c | r | c | r | or | inc | inc |
| SiO2 | 40.20 | 40.30 | 40.31 | 40.43 | 40.19 | 40.44 | 40.10 | 40.35 | 41.04 | 40.17 | 40.75 |
| TiO2 | 0.37 | 0.91 | 1.10 | 1.56 | 0.30 | 1.81 | 0.46 | 1.69 | 4.27 | 0.80 | 3.39 |
| ZrO2 | 0.24 | 0.38 | 0.69 | 0.43 | 0.63 | 0.55 | 0.17 | 0.43 | 0.40 | 0.52 | 0.36 |
| Nb2O5 | 43.54 | 42.68 | 42.00 | 41.62 | 43.26 | 41.15 | 43.39 | 41.40 | 37.80 | 42.52 | 39.00 |
| Na2O | 0.07 | 0.00 | 0.00 | 0.04 | 0.05 | 0.05 | 0.04 | 0.03 | 0.03 | 0.03 | 0.06 |
| K2O | 15.64 | 15.81 | 15.80 | 15.78 | 15.67 | 15.76 | 15.67 | 15.78 | 16.04 | 15.71 | 15.89 |
| F | 0.11 | 0.12 | 0.50 | 0.29 | 0.23 | 0.51 | 0.34 | 0.57 | 0.74 | 0.55 | 0.84 |
| Sum | 100.17 | 100.20 | 100.41 | 100.15 | 100.33 | 100.27 | 100.17 | 100.25 | 100.31 | 100.31 | 100.29 |
| O-F2 | 0.05 | 0.05 | 0.21 | 0.12 | 0.10 | 0.22 | 0.14 | 0.24 | 0.31 | 0.23 | 0.35 |
| Sum | 100.12 | 100.15 | 100.20 | 100.03 | 100.23 | 100.05 | 100.02 | 100.01 | 100.00 | 100.07 | 99.94 |
| Formula based on 8 cations | |||||||||||
| Si | 4.002 | 3.998 | 4.000 | 4.001 | 4.001 | 4.000 | 4.000 | 3.999 | 4.001 | 4.000 | 4.000 |
| Ti | 0.027 | 0.068 | 0.082 | 0.116 | 0.022 | 0.134 | 0.034 | 0.126 | 0.313 | 0.060 | 0.250 |
| Zr | 0.011 | 0.019 | 0.034 | 0.021 | 0.031 | 0.027 | 0.008 | 0.021 | 0.019 | 0.025 | 0.017 |
| Nb | 1.959 | 1.914 | 1.884 | 1.862 | 1.947 | 1.840 | 1.957 | 1.855 | 1.666 | 1.914 | 1.731 |
| K + Na | 2.000 | 2.001 | 2.000 | 2.000 | 2.000 | 1.999 | 2.001 | 2.000 | 2.000 | 2.001 | 2.001 |
| F | 0.036 | 0.036 | 0.158 | 0.090 | 0.073 | 0.160 | 0.107 | 0.179 | 0.227 | 0.174 | 0.261 |
| End-members, mol.% | |||||||||||
| K2Nb2(Si4O12)O2 | 0.98 | 0.96 | 0.94 | 0.93 | 0.97 | 0.92 | 0.98 | 0.93 | 0.83 | 0.96 | 0.87 |
| K2Ti2(Si4O12)(F,OH)2 | 0.02 | 0.04 | 0.06 | 0.07 | 0.03 | 0.08 | 0.02 | 0.07 | 0.17 | 0.04 | 0.13 |
| Component | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
|---|---|---|---|---|---|---|---|---|---|---|
| Grain | 5-1Rp | 5-2Rp | Rp-1 | Rp-2 | Rp-3 | Rp-4 | Rp-5 | Rp-6-1 | Rp-6-2 | Rp-7 |
| n | 2 | 2 | 2 | 3 | 3 | 3 | 3 | 3 | 3 | 2 |
| SiO2, wt % | 40.31 | 40.32 | 40.16 | 40.19 | 40.15 | 40.17 | 40.20 | 40.19 | 40.15 | 40.11 |
| TiO2 | 0.65 | 0.83 | 0.87 | 0.62 | 0.44 | 0.45 | 0.49 | 0.69 | 0.39 | 0.23 |
| ZrO2 | 0.21 | 0.32 | 0.20 | 0.22 | 0.18 | 0.30 | 0.15 | 0.17 | 0.05 | 0.08 |
| Nb2O5 | 42.88 | 43.22 | 42.75 | 43.17 | 43.48 | 43.37 | 43.48 | 43.1 | 43.69 | 43.87 |
| Na2O | 0.00 | 0.04 | 0.00 | 0.03 | 0.00 | 0.00 | 0.02 | 0.00 | ||
| K2O | 15.57 | 15.67 | 15.74 | 15.70 | 15.73 | 15.70 | 15.75 | 15.75 | 15.70 | 15.71 |
| F | 0.23 | 0.11 | 0.16 | 0.10 | 0.12 | 0.09 | 0.15 | 0.10 | ||
| H2O | 0.16 | 0.17 | 0.25 | 0.17 | 0.14 | 0.13 | 0.26 | 0.12 | ||
| Sum | 99.61 | 100.34 | 100.10 | 100.21 | 100.39 | 100.29 | 100.33 | 100.12 | 100.40 | 100.21 |
| O-F2 | 0.10 | 0.05 | 0.07 | 0.04 | 0.05 | 0.04 | 0.06 | 0.04 | ||
| Sum | 99.61 | 100.34 | 100.00 | 100.16 | 100.32 | 100.25 | 100.28 | 100.08 | 100.34 | 100.17 |
| Li, ppm | 0.36 | 0.37 | 0.22 | 0.31 | 0.11 | 0.05 | 0.11 | 0.13 | 1.23 | 0.03 |
| Be | 0.01 | 0.03 | 0.04 | 0.01 | 0.01 | 0.03 | 0.52 | 0.01 | ||
| B | 0.85 | 0.45 | 0.96 | 0.22 | 0.35 | 0.45 | 2.80 | 0.11 | ||
| P | 22 | 23 | ||||||||
| Ca | 16 | 16 | ||||||||
| Ba | 201 | 153 | ||||||||
| Sr | 1.31 | 0.70 | ||||||||
| Sum REE | 0.56 | 0.59 | ||||||||
| Y | 0.07 | 0.09 | ||||||||
| Sc | 7.1 | 9.4 | ||||||||
| Ga | 1.61 | 0.56 | ||||||||
| Cr | 0.95 | 1.00 | ||||||||
| V | 26 | 12 | ||||||||
| Mn | 0.42 | 0.24 | ||||||||
| Co | 0.03 | 0.03 | ||||||||
| Ni | 0.25 | 0.26 | ||||||||
| Hf | 22 | 36 | ||||||||
| Ta | 7.3 | 11.3 | ||||||||
| Th | 0.05 | 0.10 | ||||||||
| U | 0.61 | 0.35 |
| h | k | l | dcalc (Å) | dobs (Å) | I | h | k | l | dcalc (Å) | dobs (Å) | I |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 0 | 1 | 8.127 | 8.161 | 4 | 1 | 5 | 0 | 1.713 | 1.712 | 19 |
| 1 | 1 | 0 | 6.177 | 6.205 | 100 | 2 | 2 | 4 | 1.697 | ||
| 1 | 1 | 1 | 4.917 | 4.943 | 5 | 1 | 3 | 4 | 1.637 | 1.639 | 14 |
| 0 | 2 | 0 | 4.367 | 4.383 | 83 | 1 | 5 | 2 | 1.579 | 1.580 | 18 |
| 0 | 0 | 2 | 4.063 | 4.082 | 90 | 4 | 4 | 0 | 1.544 | 1.546 | 13 |
| 1 | 2 | 0 | 3.906 | 3.914 | 46 | 1 | 2 | 5 | 1.501 | 1.502 | 11 |
| 0 | 2 | 1 | 3.847 | 3.850 | 18 | 3 | 5 | 0 | 1.498 | ||
| 1 | 2 | 1 | 3.521 | 3.530 | 87 | 0 | 4 | 4 | 1.487 | 1.491 | 7 |
| 1 | 1 | 2 | 3.395 | 3.405 | 40 | 1 | 4 | 4 | 1.466 | 1.469 | 5 |
| 2 | 2 | 0 | 3.088 | 3.096 | 59 | 0 | 6 | 0 | 1.456 | 1.460 | 5 |
| 0 | 2 | 2 | 2.975 | 2.985 | 81 | 4 | 4 | 2 | 1.443 | 1.446 | 8 |
| 1 | 2 | 2 | 2.816 | 2.822 | 70 | 2 | 4 | 4 | 1.408 | 1.410 | 11 |
| 1 | 3 | 0 | 2.762 | 2.768 | 99 | 2 | 6 | 0 | 1.381 | 1.383 | 6 |
| 2 | 2 | 2 | 2.459 | 2.466 | 7 | 2 | 3 | 5 | 1.350 | 1.351 | 3 |
| 2 | 3 | 0 | 2.423 | 2.426 | 3 | 1 | 5 | 4 | 1.310 | 1.309 | 16 |
| 2 | 3 | 1 | 2.322 | 2.324 | 6 | 2 | 6 | 2 | 1.308 | ||
| 0 | 2 | 3 | 2.302 | 2.293 | 9 | 0 | 4 | 5 | 1.304 | ||
| 1 | 3 | 2 | 2.284 | 3 | 6 | 0 | 1.302 | 1.301 | 11 | ||
| 0 | 4 | 0 | 2.184 | 2.187 | 3 | 0 | 2 | 6 | 1.294 | ||
| 1 | 4 | 0 | 2.119 | 2.120 | 16 | 4 | 4 | 4 | 1.229 | 1.233 | 3 |
| 0 | 4 | 1 | 2.109 | 4 | 6 | 0 | 1.211 | 1.211 | 5 | ||
| 1 | 4 | 1 | 2.050 | 2.036 | 26 | 3 | 5 | 4 | 1.206 | ||
| 0 | 0 | 4 | 2.032 | 2 | 7 | 0 | 1.200 | 1.206 | 5 | ||
| 2 | 4 | 0 | 1.953 | 1.955 | 44 | 0 | 6 | 4 | 1.183 | 1.184 | 3 |
| 1 | 3 | 3 | 1.934 | 4 | 6 | 2 | 1.161 | 1.161 | 2 | ||
| 1 | 1 | 4 | 1.930 | 1.930 | 23 | 3 | 7 | 0 | 1.147 | 1.148 | 6 |
| 0 | 4 | 2 | 1.924 | 2 | 6 | 4 | 1.142 | ||||
| 0 | 2 | 4 | 1.842 | 1.841 | 13 | 3 | 7 | 2 | 1.104 | 1.104 | 3 |
| 3 | 3 | 2 | 1.837 | 3 | 5 | 5 | 1.102 | ||||
| 2 | 3 | 3 | 1.806 | 1.810 | 5 | 3 | 6 | 4 | 1.096 | 1.098 | 2 |
| 2 | 4 | 2 | 1.760 | 1.762 | 25 | 0 | 8 | 2 | 1.054 | 1.058 | 3 |
| Crystal Data | |
|---|---|
| Unit cell dimensions (Å) | a = 8.73885(16), c = 8.1277(2) |
| Space group | P4bm (No. 100) |
| Volume (Å3) | 620.69(2) |
| Z | 2 |
| Chemical formula | K2(Nb1.93Ti0.05Zr0.02)[Si4O12]O(O0.93F0.07) |
| Intensity Measurement | |
| Crystal shape | Fragment of prismatic crystal bounded by cleavage on (001) |
| Crystal size (mm) | 0.30 × 0.24 × 0.14 |
| Diffractometer | Oxford Diffraction Xcalibur Gemini |
| X-ray radiation | Mo Kα |
| X-ray power | 50 kV 30 mA |
| Monochromator | Graphite |
| Temperature | 293 K |
| Detector to sample distance | 85 mm |
| Measurement method | Omega scan |
| Radiation width | 1° |
| Time per frame | 5 s |
| Max. θ°-range for Data collection | 32.04 |
| Index ranges | −6 ≤ h ≤ 12 |
| −12 ≤ k ≤ 11 | |
| −11 ≤ l ≤ 9 | |
| No. of measured reflections | 3667 |
| No. of unique reflections | 949 |
| No. of observed reflections (I > 2σ(I)) | 834 |
| Refinement of the structure | |
| No. of parameters used in refinement | 63 |
| Rint | 0.0269 |
| Flack x parameter | 0.15(16) |
| R1, I > 2σ(I) | 0.0360 |
| R1 all Data | 0.0429 |
| wR2 on (F2) | 0.0980 |
| GooF | 1.109 |
| Δρ min (e Å−3) | −0.814 close to K1 |
| Δρ max (e Å−3) | 0.855 close to Nb1 |
| Atom | Occupancy | x | y | z | Ueq |
|---|---|---|---|---|---|
| Nb1 | Nb0.96Ti0.03Zr0.01 | 0.0 | 0.0 | 0.23217(13) | 0.0255(2) |
| Nb2 | Nb0.96Ti0.03Zr0.01 | 0.0 | 0.0 | 0.73373(12) | 0.0237(2) |
| K1 | 1 | 0.32254(8) | 0.82254(8) | 0.0003(3) | 0.0216(2) |
| Si1 | 1 | 0.12425(14) | 0.62425(14) | 0.3080(2) | 0.0139(3) |
| Si2 | 1 | 0.12401(13) | 0.62401(13) | 0.69906(18) | 0.0076(3) |
| O1 | 1 | 0.4240(4) | 0.2895(4) | 0.2361(4) | 0.0182(8) |
| O2 | 1 | 0.4229(5) | 0.2877(5) | 0.7680(3) | 0.0158(9) |
| O3 | 1 | 0.0 | 0.0 | −0.0020(14) | 0.0200(11) |
| O4 | 1 | 0.0 | 0.0 | 0.4943(17) | 0.0229(12) |
| O5 | 1 | 0.1204(3) | 0.6204(3) | 0.4974(8) | 0.0203(7) |
| O6 | 1 | 0.5 | 0.0 | 0.2356(8) | 0.0156(14) |
| O7 | 1 | 0.5 | 0.0 | 0.7679(7) | 0.0138(17) |
| Atom | U11 | U22 | U33 | U12 | U13 | U23 |
|---|---|---|---|---|---|---|
| Nb1 | 0.0105(2) | 0.0105(2) | 0.0555(5) | 0.0 | 0.0 | 0.0 |
| Nb2 | 0.0114(2) | 0.0114(2) | 0.0483(5) | 0.0 | 0.0 | 0.0 |
| K1 | 0.0237(3) | 0.0237(3) | 0.0176(4) | −0.0041(3) | 0.0004(6) | 0.0004(6) |
| Si1 | 0.0141(5) | 0.0141(5) | 0.0133(7) | −0.0007(6) | −0.0001(4) | −0.0001(4) |
| Si2 | 0.0098(4) | 0.0098(4) | 0.0032(6) | 0.0005(5) | −0.0002(4) | −0.0002(4) |
| O1 | 0.0189(16) | 0.0109(14) | 0.0247(15) | −0.0009(13) | −0.0029(12) | −0.0022(12) |
| O2 | 0.0217(19) | 0.0147(18) | 0.0110(14) | −0.0043(15) | −0.0001(11) | −0.0035(11) |
| O3 | 0.0161(12) | 0.0161(12) | 0.028(3) | 0.0 | 0.0 | 0.0 |
| O4 | 0.0194(13) | 0.0194(13) | 0.030(3) | 0.0 | 0.0 | 0.0 |
| O5 | 0.0264(9) | 0.0264(9) | 0.0080(14) | −0.0090(12) | −0.0054(18) | −0.0054(18) |
| O6 | 0.0139(18) | 0.0139(18) | 0.019(3) | 0.005(3) | 0.0 | 0.0 |
| O7 | 0.017(2) | 0.017(2) | 0.007(3) | 0.007(3) | 0.0 | 0.0 |
| Nb1–O3 | 1.903(12) | Nb2–O4 | 1.946(13) |
| Nb1–O1(4×) | 1.956(4) | Nb2–O2(4×) | 1.993(4) |
| Nb1–O4 | 2.131(13) | Nb2–O3 | 2.148(12) |
| mean | 1.976 | mean | 2.011 |
| K1–O2(2×) | 2.874(4) | Si1–O5 | 1.541(7) |
| K1–O7 | 2.894(4) | Si1–O1(2×) | 1.614(4) |
| K1–O1(2×) | 2.898(4) | Si1–O6 | 1.644(3) |
| K1–O6 | 2.910(5) | mean | 1.603 |
| K1–O2(2×) | 3.073(4) | ||
| K1–O1(2×) | 3.088(4) | Si2–O2(2×) | 1.590(4) |
| K1–O3(2×) | 3.2171(3) | Si2–O7 | 1.632(2) |
| mean | 3.009 | Si2–O5 | 1.639(7) |
| mean | 1.623 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharygin, V.V.; Doroshkevich, A.G.; Seryotkin, Y.V.; Karmanov, N.S.; Belogub, E.V.; Moroz, T.N.; Nigmatulina, E.N.; Yelisseyev, A.P.; Vedenyapin, V.N.; Kupriyanov, I.N. Rippite, K2(Nb,Ti)2(Si4O12)O(O,F), a New K-Nb-Cyclosilicate from Chuktukon Carbonatite Massif, Chadobets Upland, Krasnoyarsk Territory, Russia. Minerals 2020, 10, 1102. https://doi.org/10.3390/min10121102
Sharygin VV, Doroshkevich AG, Seryotkin YV, Karmanov NS, Belogub EV, Moroz TN, Nigmatulina EN, Yelisseyev AP, Vedenyapin VN, Kupriyanov IN. Rippite, K2(Nb,Ti)2(Si4O12)O(O,F), a New K-Nb-Cyclosilicate from Chuktukon Carbonatite Massif, Chadobets Upland, Krasnoyarsk Territory, Russia. Minerals. 2020; 10(12):1102. https://doi.org/10.3390/min10121102
Chicago/Turabian StyleSharygin, Victor V., Anna G. Doroshkevich, Yurii V. Seryotkin, Nikolai S. Karmanov, Elena V. Belogub, Tatyana N. Moroz, Elena N. Nigmatulina, Alexander P. Yelisseyev, Vitalii N. Vedenyapin, and Igor N. Kupriyanov. 2020. "Rippite, K2(Nb,Ti)2(Si4O12)O(O,F), a New K-Nb-Cyclosilicate from Chuktukon Carbonatite Massif, Chadobets Upland, Krasnoyarsk Territory, Russia" Minerals 10, no. 12: 1102. https://doi.org/10.3390/min10121102
APA StyleSharygin, V. V., Doroshkevich, A. G., Seryotkin, Y. V., Karmanov, N. S., Belogub, E. V., Moroz, T. N., Nigmatulina, E. N., Yelisseyev, A. P., Vedenyapin, V. N., & Kupriyanov, I. N. (2020). Rippite, K2(Nb,Ti)2(Si4O12)O(O,F), a New K-Nb-Cyclosilicate from Chuktukon Carbonatite Massif, Chadobets Upland, Krasnoyarsk Territory, Russia. Minerals, 10(12), 1102. https://doi.org/10.3390/min10121102






