Riesite, a New High Pressure Polymorph of TiO2 from the Ries Impact Structure
Abstract
1. Introduction
2. Materials and Methods
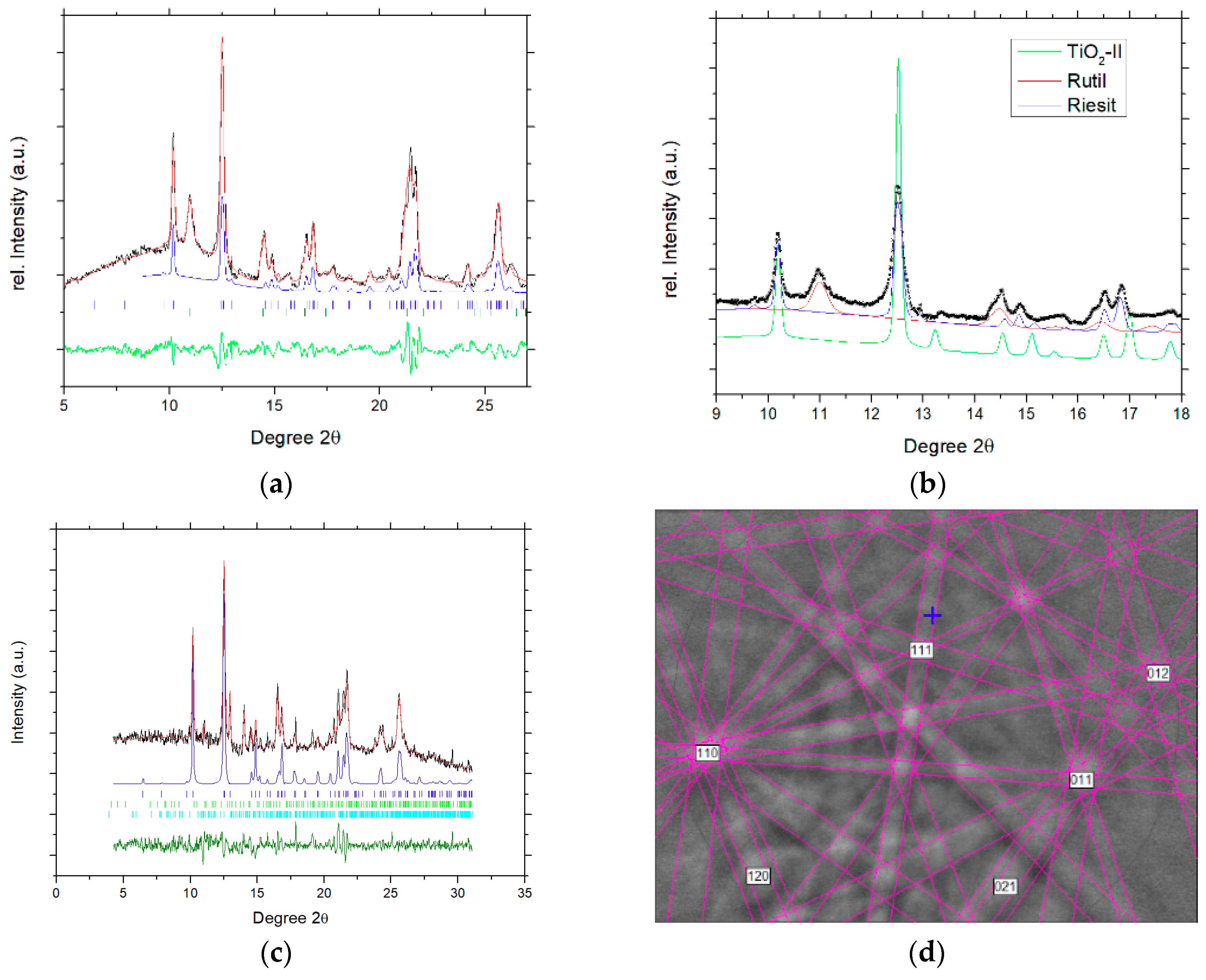
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jamieson, J.C.; Olinger, B. High-pressure polymorphism of titanium dioxide. Science 1968, 151, 893–895. [Google Scholar] [CrossRef] [PubMed]
- Withers, A.C.; Essene, E.J.; Jhang, Y. Rutile/TiO2II phase equilibria. Contrib. Mineral. Petrol. 2003, 145, 199–204. [Google Scholar] [CrossRef]
- Kojitani, H.; Yamazaki, M.; Kojima, M.; Inaguma, Y.; Mori, D.; Akaogi, M. Thermodynamic investigation of the phase equilibrium boundary between TiO2 rutile and its alpha-PbO2-type high-pressure polymorph. Phys. Chem. Min. 2018, 45, 963–980. [Google Scholar] [CrossRef]
- Mammone, J.F.; Nicol, M.; Sharma, S.K. Raman-spectra of TiO2-II, TiO2-III, SnO2, and GeO2 at high-pressure. J. Phys. Chem. Sol. 1981, 42, 379–384. [Google Scholar] [CrossRef]
- Sato, H.; Endo, S.; Sugiyama, M.; Kikegawa, T.; Shimomura, O.; Kusaba, K. Baddeleyite-type high-pressure phase of TiO2. Science 1991, 251, 786–788. [Google Scholar] [CrossRef] [PubMed]
- Dubrovinskaia, N.A.; Dubrovinsky, L.S.; Ahuja, R.; Prokopenko, V.B.; Dmitriev, V.; Weber, H.P.; Johansson, B. Experimental and theoretical identification of a new high-pressure TiO2 polymorph. Phys. Rev. Lett. 2001, 87, 275501. [Google Scholar] [CrossRef]
- Nishio-Hamane, D.; Shimizu, A.; Nakahira, R.; Niwa, K.; Sano-Furukawa, A.; Okada, T.; Kikegawa, T. The stability and equation of state for the cotunnite phase of TiO2 up to 70 GPa. Phys. Chem. Min. 2010, 37, 129–136. [Google Scholar] [CrossRef]
- El Goresy, A.; Dubrovinsky, L.; Gillet, P.; Graup, G.; Chen, M. Akaogiite: An ultra-dense polymorph of TiO2 with the baddeleyite-type structure, in shocked garnet gneiss from the Ries Crater, Germany. Am. Min. 2010, 95, 892–895. [Google Scholar] [CrossRef]
- Ferry, J.M.; Watson, E.B. New thermodynamic models and revised calibrations for the Ti-in-zircon and Zr-in-rutile thermometers. Contr. Min. Petr. 2007, 154, 429–437. [Google Scholar] [CrossRef]
- Akaogi, M.; Horiuchi, N.; Ishii, T.; Kojitani, H. High-pressure phase relations in the system TiO2-ZrO2 to 12 GPa: Stability of αPbO2-type srilankite solid solutions of (Ti1−x, Zrx)O2 (0 ≤ x ≤ 0.6). Phys. Chem. Min. 2012, 39, 797–802. [Google Scholar] [CrossRef]
- Nickel, E.H.; Rowland, J.F.; McAdam, R.C. Ixiolite—A columbite substructure. Am. Min. 1963, 48, 961–979. [Google Scholar]
- Willgallis, A.; Siegmann, E.; Hettiaratchi, T. Srilankite, a new Zr-Ti-oxide mineral. Neues Jahrbuch für Mineralogie Monatshefte 1983, 4, 151–157. [Google Scholar]
- Wang, L.P.; Essene, E.J.; Zhang, Y.X. Mineral inclusions in pyrope crystals from Garnet Ridge, Arizona, USA: Implications for processes in the upper mantle. Contr. Min. Petr. 1999, 135, 164–178. [Google Scholar] [CrossRef]
- Varlamov, D.A.; Garanin, V.K.; Kostrovitskii, S.I. The exotic high-titanium minerals as inclusions within granets from low crust and mantle xenoliths. Dokl. Akad. Nauk. 1995, 345, 364–366. [Google Scholar]
- El Goresy, A.; Chen, M.; Gillet, P.; Dubrovinsky, L.; Graup, G.; Ahuja, R. A natural shock-induced dense polymorph of rutile with alpha-PbO2 structure in the suevite from the Ries crater in Germany. Earth Planet. Sci. Lett. 2001, 192, 485–495. [Google Scholar] [CrossRef]
- Chen, M.; Gu, X.P.; Xie, X.D.; Yin, F. High-pressure polymorph of TiO2-II from the Xiuyan crater of China. Chin. Sci. Bul. 2013, 58, 4655–4662. [Google Scholar] [CrossRef]
- Zhang, R.Y.; Liou, J.G.; Ernst, W.G. The Dabie-Sulu continental collision zone: A comprehensive review. Gondw. Res. 2009, 16, 1–26. [Google Scholar] [CrossRef]
- Tschauner, O. High-pressure minerals. Am. Mineral. 2019, 104, 1701–1731. [Google Scholar] [CrossRef]
- Cavosie, A.J.; Erickson, T.M.; Timms, N.E. Nanoscale records of ancient shock deformation: Reidite (ZrSiO4) in sandstone at the Ordovician Rock Elm impact crater. Geology 2015, 43, 315–318. [Google Scholar] [CrossRef]
- Tschauner, O.; Ma, C. Riesite, IMA 2015-110a. CNMNC Newsletter No. 35, February 2017: p 213. Min. Mag. 2017, 81, 209–213. [Google Scholar]
- Staehle, V.; Altherr, R.; Nasdala, L.; Ludwig, T. Ca-rich majorite derived from high-temperature melt and thermally stressed hornblende in shock veins of crustal rocks from the Ries impact crater (Germany). Contr. Min. Petr. 2011, 161, 275–291. [Google Scholar] [CrossRef]
- Erickson, T.M.; Pearce, M.A.; Reddy, S.M.; Timms, N.E.; Cavosie, A.J.; Bourdet, J.; Nemchin, A.A. Microstructural constraints on the mechanisms of the transformation to reidite in naturally shocked zircon. Contr. Min. Petr. 2017, 172, 6. [Google Scholar] [CrossRef]
- Stähle, V.; Altherr, R.; Nasdala, L.; Trieloff, M.; Varychev, A. Majoritic garnet grains within shock-induced melt veins in amphibolites from the Ries impact crater suggest ultrahigh crystallization pressures between 18 and 9 GPa. Contr. Min. Petr. 2017, 172, 86. [Google Scholar] [CrossRef]
- Armstrong, J.T. CITAF—A package of correction programs for the quantitative electron microbeam X-ray analysis of thick polished materials, thin-films, and particles. Microbeam Anal. 1995, 4, 177–200. [Google Scholar]
- Hammersley, A.P.; Svensson, S.O.; Hanfland, M.; Fitch, A.N.; Hausermann, D. Two-dimensional detector software: From real detector to idealised image or two-theta scan. High. Pres. Res. 1996, 14, 235–248. [Google Scholar] [CrossRef]
- Dera, P.; Zhuravlev, K.; Prakapenka, V.; Rivers, M.L.; Finkelstein, G.J.; Grubor-Urosevic, O.; Downs, R.T. High pressure single-crystal micro X-ray diffraction analysis with GSE_ADA/RSV software. High. Pres. Res. 2013, 33, 466–484. [Google Scholar] [CrossRef]
- Putz, H.; Schon, J.C.; Jansen, M. Combined method for ab initio structure solution from powder diffraction data. J. Appl. Cryst. 1999, 32, 864–870. [Google Scholar] [CrossRef]
- Altomare, A.; Camalli, M.; Cuocci, C.; Giacovazzo, C.; Moliterni, A.; Rizzi, R. EXPO2009: Structure solution by powder data in direct and reciprocal space. J. Appl. Cryst. 2009, 42, 1197–1202. [Google Scholar] [CrossRef]
- Kraus, W.; Nolze, G. POWDER CELL—A program for the representation and manipulation of crystal structures and calculation of the resulting X-ray powder patterns. J. Appl. Cryst. 1996, 29, 301–303. [Google Scholar] [CrossRef]
- von Dreele, R.B.; Larson, A.C. General Structure Analysis System (GSAS); Los Alamos National Laboratory Report LAUR; Los Alamos National Laboratory: Los Alamos, NM, USA, 2004. [Google Scholar]
- Grey, I.E.; Li, C.; Madsen, I.C.; Braunshausen, G. TiO2-II. Ambient pressure and structure refinement. Mat. Res. Bull. 1988, 23, 743–753. [Google Scholar] [CrossRef]
- Timms, N.E.; Erickson, T.M.; Pearce, M.A.; Cavosie, A.J.; Schmieder, M.; Tohver, E.; Wittmann, A. A pressure-temperature phase diagram for zircon at extreme conditions. Earth-Science Rev. 2017, 165, 185–202. [Google Scholar] [CrossRef]
- Leroux, H.; Reimold, W.U.; Koeberl, C.; Hornemann, U.; Doukhan, J.C. Experimental shock deformation in zircon: A transmission electron microscopic study. Earth. Planet. Sci. Lett. 1999, 169, 291–301. [Google Scholar] [CrossRef]
- Ma, C.; Tschauner, O.; Beckett, J.R.; Rossman, G.R.; Prescher, C.; Prakapenka, V.B.; MacDowell, A. Liebermannite, KAlSi3O8, a new shock-metamorphic, high-pressure mineral from the Zagami Martian meteorite. Meteorit. Planet. Sci. 2018, 53, 50–61. [Google Scholar] [CrossRef]
- Artemieva, N.A.; Wuennemann, K.; Krien, F.; Reimold, W.U.; Stöffler, D. Ries crater and suevite revisited-Observations and modeling Part II: Modeling. Meteorit. Planet. Sci. 2013, 48, 590–627. [Google Scholar] [CrossRef]

| Constituent | Wt % | Range | SD | Probe Standard |
|---|---|---|---|---|
| TiO2 | 99.25 | 98.98–99.62 | 0.19 | TiO2 |
| FeO | 0.42 | 0.33–0.53 | 0.07 | Fayalite |
| CaO | 0.03 | 0.02–0.04 | 0.01 | Anorthite |
| Total | 99.70 | - | - | - |
| Atom | Wyckoff | x/a | y/b | z/c | SFO | Biso (Å2) |
|---|---|---|---|---|---|---|
| Ti1 | 4g | 0.041(2) | 0.142(4) | 0.268(2) | 0.47(3) | 0.6(1) |
| Ti2 | 4g | 0.51(1) | 0.311(7) | 0.78(1) | 0.53(1) | 0.7(1) |
| O1 | 4g | 0.28(1) | 0.36(1) | 0.436(7) | 1 1 | 0.9(2) |
| O2 | 4g | 0.224(1) | 0.086(6) | 0.937(7) | 1 1 | 0.9(2) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tschauner, O.; Ma, C.; Lanzirotti, A.; Newville, M.G. Riesite, a New High Pressure Polymorph of TiO2 from the Ries Impact Structure. Minerals 2020, 10, 78. https://doi.org/10.3390/min10010078
Tschauner O, Ma C, Lanzirotti A, Newville MG. Riesite, a New High Pressure Polymorph of TiO2 from the Ries Impact Structure. Minerals. 2020; 10(1):78. https://doi.org/10.3390/min10010078
Chicago/Turabian StyleTschauner, Oliver, Chi Ma, Antonio Lanzirotti, and Matthew G. Newville. 2020. "Riesite, a New High Pressure Polymorph of TiO2 from the Ries Impact Structure" Minerals 10, no. 1: 78. https://doi.org/10.3390/min10010078
APA StyleTschauner, O., Ma, C., Lanzirotti, A., & Newville, M. G. (2020). Riesite, a New High Pressure Polymorph of TiO2 from the Ries Impact Structure. Minerals, 10(1), 78. https://doi.org/10.3390/min10010078





