Distribution Characteristics and Influencing Factors of Uranium Isotopes in Saline Lake Waters in the Northeast of Qaidam Basin
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Locations
2.2. Sampling Design and Methods
2.3. Analytical Methods
3. Results
3.1. Water Chemistry Characteristics in the Northeast of Qaidam Basin
3.2. Distribution Characteristics of Uranium Isotopes in These Lake Waters in the Northeast of Qaidam Basin
3.3. Influence of Water Sources Supply on the Uranium Isotopes of Lake
3.4. Seasonal Distribution Characteristics of Uranium Isotopes
4. Discussion
4.1. Factors Influencing the Concentration Distribution Characteristics of 238U in These Lake Waters in the Northeast of Qaidam Basin
4.1.1. Response Relationship of 238U Concentration to TDS and Salinity in These Lake Waters in the Northeast of Qaidam Basin
4.1.2. Response Relationship of 238U Concentration to pH in These Lake Waters in the Northeast of Qaidam Basin
4.1.3. Response Relationship of 238U Concentration to Organic Matter in These Lake Waters in the Northeast of Qaidam Basin
4.1.4. Response Relationship of 238U Concentration to Water Sources Supply in Open Lake Waters
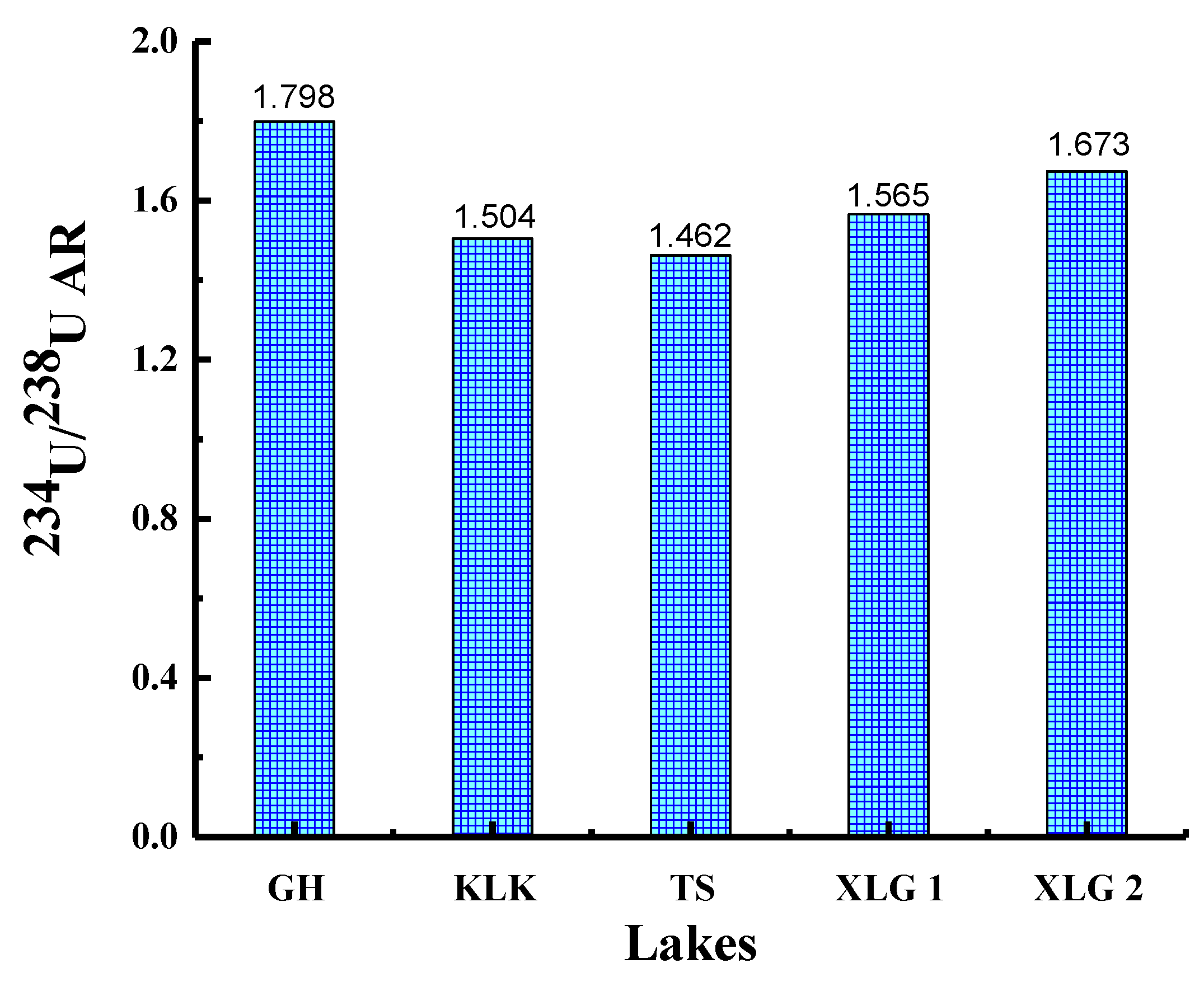
4.2. Distribution Characteristics and Influencing Factors of the [234U/238U]AR
4.3. Seasonal Effects on 238U Concentration and the [234U/238U]AR in Lakes and Their Potential Environmental Impact
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cheng, H.; Edwards, R.L.; Shen, C.C.; Polyak, V.J.; Asmerom, Y.; Woodhead, J.; Hellstrom, J.; Wang, Y.; Kong, X.; Spötl, C.; et al. Improvements in 230Th dating, 230Th and 234U half-life values, and U–Th isotopic measurements by multi-collector inductively coupled plasma mass spectrometry. Earth Planet. Sci. Lett. 2013, 371, 82–91. [Google Scholar] [CrossRef]
- Wang, L.; Ma, Z.; Sun, Z.; Wang, Y.; Wang, X.; Cheng, H.; Xiao, J. U concentration and 234U/238U of seawater from the Okinawa Trough and Indian Ocean using MC-ICP-MS with SEM protocols. Mar. Chem. 2017, 196, 71–80. [Google Scholar] [CrossRef]
- Broecker, W.S.; Peng, T.H. Reactive metals and the great particulate sweep. In Tracers in the Sea; Broecker, W.S., Peng, T.H., Eds.; Lamont-Doherty Geol Obs: Palisades, NY, USA, 1982; pp. 166–235. [Google Scholar]
- Andersen, M.B.; Stirling, C.H.; Porcelli, D.; Halliday, A.N.; Andersson, P.S.; Baskaran, M. The tracing of riverine U in Arctic seawater with very precise 234U/238U measurements. Earth Planet. Sci. Lett. 2007, 259, 171–185. [Google Scholar] [CrossRef]
- Schmidt, S.; Saari, H. Residence time of suspended particles in the Garonne River (SW France): Indications derived from Th-234, Be-7 and Pb-210. In Proceedings of the EGU General Assembly Conference Abstracts, Vienna, Austria, 7–12 April 2013. [Google Scholar]
- Chabaux, F.; Bourdon, B.; Riotte, J. U-series geochemistry in weathering profiles, river waters and lakes. In U/Th Series Radionuclides in Aquatic Systems; Krishnaswami, S., Cochran, J.K., Eds.; Elsevier: Edinburgh, UK, 2008; Volume 13, pp. 49–104. [Google Scholar]
- Aciego, S.M.; Aarons, S.M.; Sims, K.W. The uranium-isotopic composition of Saharan dust collected over the central Atlantic Ocean. Aeolian Res. 2015, 17, 61–66. [Google Scholar] [CrossRef]
- Chutcharavan, P.M.; Dutton, A.; Ellwood, M.J. Seawater 234U/238U recorded by modern and fossil corals. Geochim. Cosmochim. Acta 2018, 224, 1–17. [Google Scholar] [CrossRef]
- Aciego, S.M.; Aarons, S.M.; Sims, K.W. Behaviour of uranium during mixing in the delaware and chesapeake estuaries. Estuar. Coast. Shelf Sci. 1994, 39, 619–631. [Google Scholar]
- Moran, S.B.; Ellis, K.M.; Smith, J.N. 234Th/238U disequilibrium in the central Arctic Ocean: Implications for particulate organic carbon export. Deep Sea Res. Part II Top. Stud. Oceanogr. 1997, 44, 1593–1606. [Google Scholar] [CrossRef]
- Rengarajan, R.; Sarin, M.M.; Krishnaswami, S. Uranium in the Arabian Sea: Role of denitrification in controlling its distribution. Oceanol. Acta 2003, 26, 687–693. [Google Scholar] [CrossRef][Green Version]
- Schmidt, S. Impact of the Mediterranean Outflow Water on particle dynamics in intermediate waters of the Northeast Atlantic, as revealed by 234Th and 228Th. Mar. Chem. 2006, 100, 289–298. [Google Scholar] [CrossRef]
- Li, P.; Zhu, P.; Wang, X.; Yu, Y. Distribution of uranium in seawater on the continental shelf of the East China sea. Oceanol. Limnol. Sin. 1982, 13, 514–552. (In Chinese) [Google Scholar]
- Li, G.; Li, L.; Li, L.; Li, T. Controling mechanism of riverine uranium isotope and its implication for weathering limitation theory. Bull. Mineral. Petrol. Geochem. 2019, 38, 11–17. (In Chinese) [Google Scholar]
- Xu, Y.; Zhang, F.; Jin, Z. Uranium isotopic compositions of fine detrital particles in a glacial catchment of the Pamir Plateau and its implication for sediment transfer. J. Earth Environ. 2019, 10, 116–127. (In Chinese) [Google Scholar]
- Peng, A.; Huang, Y.; Chen, M.; Yang, W.; Qu, Y. Depth distribution of uranium isotopes of different grades in Xiamen Bay seawater. J. Appl. Oceanogr. 2012, 31, 3–11. (In Chinese) [Google Scholar]
- Francois, C.; Matheiu, G.; Philippe, L.; Jean, R.; Eugeni, V.; Olga, S.; Ludmilla, A. Geochemical and isotopic (Sr, U) variations of lake waters in the Ol’khon Region, Siberia, Russia: Origin and paleoenvironmental implications. Comptes Rendus Geosci. 2011, 343, 462–470. [Google Scholar]
- Osmond, J.K.; Kaufman, M.I.; Cowart, J.B. Mixing volume calculations, sources and aging trends of Floridan aquifer water by uranium isotopic methods. Geochim. Cosmochim. Acta 1974, 38, 1083–1100. [Google Scholar] [CrossRef]
- Yadav, D.N.; Sarin, M.M. Geo-chemical behavior of uranium in the Sambhar Salt Lake, Rajasthan (India): Implications to “Source” of salt and uranium “Sink”. Aquat. Geochem. 2009, 15, 529–545. [Google Scholar] [CrossRef]
- Borole, D.V.; Krishnaswami, S.; Somayajljlu, B.L.K. Uranium isotopes in rivers, estuaries and adjacent coastal sediments of western India: Their weathering, transport and oceanic budget. Geochim. Cosmochim. Acta 1982, 46, 125–137. [Google Scholar] [CrossRef]
- Zheng, X. China Salt Lake; Science Press: Beijing, China, 2002. (In Chinese) [Google Scholar]
- Zhang, P. Qaidam Basin Salt Lake; Science Press: Beijing, China, 1987. (In Chinese) [Google Scholar]
- Benjamin, L.; Philip, C.B.; Tamir, P.; Gerel, O. Geochemical evolution of uraniferous soda lakes in Eastern Mongolia. Environ. Earth Sci. 2011, 62, 171–183. [Google Scholar]
- Lin, H.; Fengqing, H. Salt Lake Evolution and Paleoclimate Fluctuations in Qaidam Basin; Science Press: Beijing, China, 2007. (In Chinese) [Google Scholar]
- Zhang, P.; Cheng, H.; Liu, W.; Mo, L.; Li, X.; Ning, Y.; Ji, M.; Zong, B.; Zhao, C. Geochemical and isotopic (U, Th) variations in lake waters in the Qinghai Lake Basin, Northeast Qinghai-Tibet Plateau, China: Origin and paleoenvironmental implications. Arab. J. Geosci. 2019, 12, 92. [Google Scholar] [CrossRef]
- Isupov, V.P.; Vladimirov, A.G.; Lyakhov, N.Z.; Shvartsev, S.L.; Ariunbileg, S.; Kolpakova, M.N.; Shatskaya, S.S.; Chupakhina, L.E.; Kuibida, L.V.; Moroz, E.N. Uranium in saline lakes of Northwestern Mongolia. Dokl. Earth Sci. 2011, 437, 352–356. [Google Scholar] [CrossRef]
- Strakhovenko, V.D.; Gas’kovaet, O.L. Thermodynamic model of formation of carbonates and uranium mineral phases in lakes Namshi-Nur and Tsagan-Tyrm (Cisbaikalia). Russ. Geol. Geophys. 2018, 59, 374–385. [Google Scholar] [CrossRef]
- Andersson, P.S.; Porcelli, D.; Gustafsson, Ö.; Ingri, J.; Wasserburg, G.J. The importance of colloids for the behavior of uranium isotopes in the low-salinity zone of a stable estuary. Geochim. Cosmochim. Acta 2001, 65, 13–25. [Google Scholar] [CrossRef]
- Koch-Steindl, H.; Pröhl, G. Considerations on the behaviour of long-lived radionuclides in the soil. Radiat. Environ. Biophys. 2001, 40, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Fredrickson, J.K.; Zachara, J.M.; Kennedy, D.W.; Liu, C.; Duff, M.C.; Hunter, D.B.; Dohnalkova, A. Influence of Mn oxides on the reduction of uranium (VI) by the metal-reducing bacterium Shewanella Putrefaciens. Geochim. Cosmochim. Acta 2002, 66, 3247–3262. [Google Scholar] [CrossRef]
- Belli, K.M.; DiChristina, T.J.; Van Cappellen, P.; Taillefert, M. Effects of aqueous uranyl speciation on the kinetics of microbial uranium reduction. Geochim. Cosmochim. Acta 2015, 157, 109–124. [Google Scholar] [CrossRef]
- Han, T.; QI, D.; Chen, H.; Xiang, S.; Shi, S.; Liu, C.; Li, N.; Wu, S. Spatial and Temporal Distribution of Precipitation in Qaidam Basin. Desert Oasis Meteorol. 2019, 29, 18–24. (In Chinese) [Google Scholar]
- Li, J.; Li, M.H.; Fang, X.M.; Wu, F.L.; Meng, Q.Q.; Zhang, Z.G.; Liu, X.M. Hydrochemical characteristics of the Hurleg Lake. Arid Land Geogr. 2015, 38, 43–51. (In Chinese) [Google Scholar]
- Chen, B.; Wang, W.; Duan, L.; Gu, X.; Liu, C.; Song, R. Spatial variability of hydraulic conductivity of riverbed sediment in the lower reaches of Bayin River. Water Resour. Hydropower Eng. 2019, 50, 52–57. (In Chinese) [Google Scholar]
- Chen, Z.L.; Megharaj, M.; Naidu, R. Determination of Bromate and Bromide in Seawater by Ion Chromatography, with an Ammonium Salt Solution as Mobile Phase, and Inductively Coupled Plasma Mass Spectrometry. Chromatographia 2006, 65, 115–118. [Google Scholar] [CrossRef]
- Thermo Scientific. Dionex Aquion Ion Chromatography System Installation Instructions; Thermo Scientific Press: Waltham, MA, USA, 2016. [Google Scholar]
- Cheng, H.; Edwards, R.L.; Hoff, J.; Gallup, C.D.; Richards, D.A.; Asmerom, Y. The half-lives of uranium-234 and thorium-230. Chem. Geol. 2000, 169, 17–33. [Google Scholar] [CrossRef]
- Shen, C.C.; Edwards, R.L.; Cheng, H.; Dorale, J.A.; Thomas, R.B.; Moran, S.B.; Weinstein, S.; Edmonds, H.N. Uranium and thorium isotopic and concentration measurements by magnetic sector inductively coupled plasma mass spectrometry. Chem. Geol. 2002, 185, 165–178. [Google Scholar] [CrossRef]
- Shen, C.C.; Wu, C.C.; Cheng, H.; Edwards, R.L.; Hsieh, Y.T.; Gallet, S.; Chang, C.C.; Li, T.Y.; Lam, D.D.; Kano, A.; et al. High precision and high-resolution carbonate Th-230 dating by MCICP-MS with SEM protocols. Geochim. Acta 2012, 99, 71–86. [Google Scholar] [CrossRef]
- Shugui, D. Environmental Chemistry; Higher Education Press: Beijing, China, 2006. (In Chinese) [Google Scholar]
- Shi, W. The Principle of Water Geochemistry of Uranium Atomic; Energy Press: Beijing, China, 1990. (In Chinese) [Google Scholar]
- Langmuir, D. Uranium-solution-mineral equilibria at low temperatures with applications to sedimentary ore deposits. Geochim. Cosmochim. Acta 1978, 42, 547–569. [Google Scholar] [CrossRef]
- Langmuir, D.; Herman, J.S. The mobility of thorium in natural waters at low temperatures. Geochim. Cosmochim. Acta 1980, 44, 1753–1766. [Google Scholar] [CrossRef]
- Owens, S.A.; Buesseler, K.O.; Sims, K.W.W. Re-evaluating the 238U salinity relationship in seawater: Implications for the238U-234Th disequilibrium method. Mar. Chem. 2011, 127, 31–39. [Google Scholar] [CrossRef]
- Lü, E.; Lei, Z.; Min, C.; Yusheng, Q.; Na, X.; Weifeng, Y.; Yanping, L.; Yipu, H. Size-fractionated uranium isotopes in surface waters in the Jiu-long River Estuary in China. Acta Oceanol. Sin. 2007, 29, 59–68. (In Chinese) [Google Scholar]
- Mochizuki, A.; Hosoda, K.; Sugiyama, M. Characteristic seasonal variation in dissolved uranium concentration induced by the change of lake water pH in Lake Biwa, Japan. Limnology 2016, 17, 127–142. [Google Scholar] [CrossRef]
- Newsome, L.; Morris, K.; Lloyd, J.R. The biogeochemistry and bioremediation of uranium and other priority radionuclides. Chem. Geol. 2014, 363, 164–184. [Google Scholar] [CrossRef]
- Newsome, L.; Morris, K.; Shaw, S.; Trivedi, D.; Lloyd, J.R. The stability of microbially reduced U (IV); impact of residual electron donor and sediment ageing. Chem. Geol. 2015, 409, 125–135. [Google Scholar] [CrossRef]
- Burgos, W.D.; Senko, J.M.; Dempsey, B.A.; Roden, E.E.; Stone, J.J.; Kemner, K.M.; Kelly, S.D. Soil humic acid decreases biological uranium(VI) reduction by Shewanella putrefaciens CN32. Environ. Eng. Sci. 2007, 24, 755–761. [Google Scholar] [CrossRef]
- Bogdanov, R.V.; Timofeev, S.A.; Selivanov, S.S. Radiogenic 234U and 210Po in humus acids of dyctionemic shale. Radiochemistry 2006, 48, 409–418. [Google Scholar] [CrossRef]
- Xu, M.; Cao, H.; Jia, Q.; Gao, Y.; Chen, S. Preliminary study of plankton community diversity of the Gahai Salt Lake in the Qaidam Basin of the Qaidam Basin of the Qinghai-Tibet Plateau. Biodivers. Sci. 2002, 10, 38–43. (In Chinese) [Google Scholar]
- Shu, L. Protection and Development of the artemia in the salt lakes of Qinghai Province—A case of Ga Hai lake. Resour. Dev. Mark 2003, 19, 311–313. (In Chinese) [Google Scholar]
- Zhao, S.; Chen, L. Institutional selection and analysis on the development of halogen resources in Gahai lake. Chin. Fish. 2007, 9, 71–72. (In Chinese) [Google Scholar]
- Li, X.; Liu, W.; Xu, L. Stable oxygen isotope of ostracods in recent sediments of Lake Gahai in the Qaidam Basin, northwest China: The implications for paleoclimatic reconstruction. Glob. Planet. Chang. 2012, 94–95, 13–19. [Google Scholar] [CrossRef]
- Jin, Y.; Qiang, M.; Liu, Y.; Li, Y.; Li, H.; Li, F. Variations in carbon and oxygen isotopes of carbonate and water environments: Genggahai Lake, northeastern Qinghai-Tibetan Plateau. Chin. Sci. Bull. 2015, 60, 847–856. (In Chinese) [Google Scholar]
- Liu, W.G.; Xiao, Y.K.; Wang, Q.Z.; Qi, H.P.; Wang, Y.H.; Zhou, Y.M.; Shirodkar, P.V. Chlorine isotopic geochemistry of salt lakes in the Qaidam Basin, China. Chem. Geol. 1997, 136, 271–279. [Google Scholar] [CrossRef]
- Bourdon B, Turner S, Henderson G M, Introduction to U-series Geochemistry. Rev. Mineral. Geochem. 2003, 52, 1–21. [CrossRef]
- Scott, M.R. The chemistry of U and Th series nuclides in rivers. In Uranium Series Disequilibrium: Applications to Environmental Problems; Ivanovich, M., Harmon, R.S., Eds.; Oxford Clarendon Press: Oxford, UK, 1982; pp. 181–201. [Google Scholar]
- Chabaux, F.; Riotte, J.; Clauer, N.; FranceLanord, G. Isotopic tracing of the dissolved U fluxes of Himalayan rivers: Implications for present and past U budgets of the Ganges-Brahmaputra system. Geochim. Cosmochim. Acta 2001, 65, 3201–3217. [Google Scholar] [CrossRef]
- Osmond, J.K.; Rydell, H.S.; Kaufman, M.I. Uranium disequilibrium in groundwater: An isotope dilution approach in hydrologic investigations. Science 1968, 162, 997–999. [Google Scholar] [CrossRef]
- Kigoshi, K. Alpha-recoil thorium-234: Dissolution into water and the Uranium-234/Uranium-238 disequilibrium in nature. Science 1971, 173, 47–48. [Google Scholar] [CrossRef] [PubMed]
- Fleischer, R.L. Isotopic disequilibrium of uranium: Alpha-recoil damage and preferential solution effects. Science 1980, 207, 979–981. [Google Scholar] [CrossRef] [PubMed]
- Chabaux, F.; Riotte, J.; Dequincey, O. U-Th-Ra fractionation during weathering and river transport. Rev. Mineral. Geochem. 2003, 52, 533–576. [Google Scholar] [CrossRef]
- Bourdon, B.; Bureau, S.; Andersen, M.B.; Pili, E.; Hubert, A. Weathering rates from top to bottom in a carbonate environment. Chem. Geol. 2009, 258, 275–287. [Google Scholar] [CrossRef]
- Vengosh, A.C.; Chivas, A.R.; Starinsky, A.; Kolodny, Y.; Baozhen, Z.; Pengxi, Z. Chemical and boron isotopic composition of non-marine brines from the Qaidam Basin, Qinghai, China. Chem. Geol. 1995, 120, 136–155. [Google Scholar] [CrossRef]
- Ivanovich, M.; Harmon, R.S. Uranium-Series Disequilibrium: Application to Earth, Marine, and Environmental Sciences; Oxford Clarendon Press: Oxford, UK, 1992. [Google Scholar]
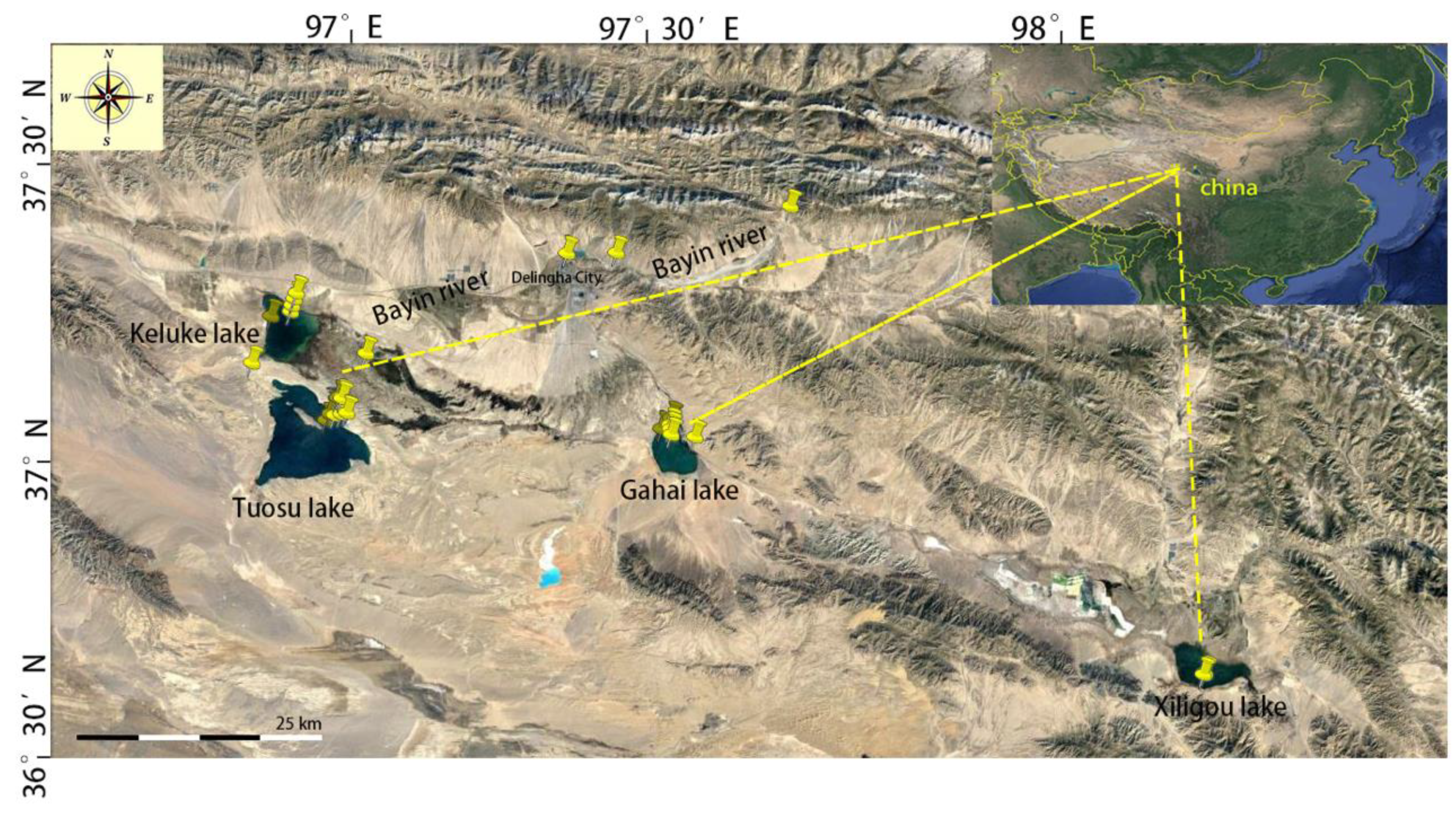
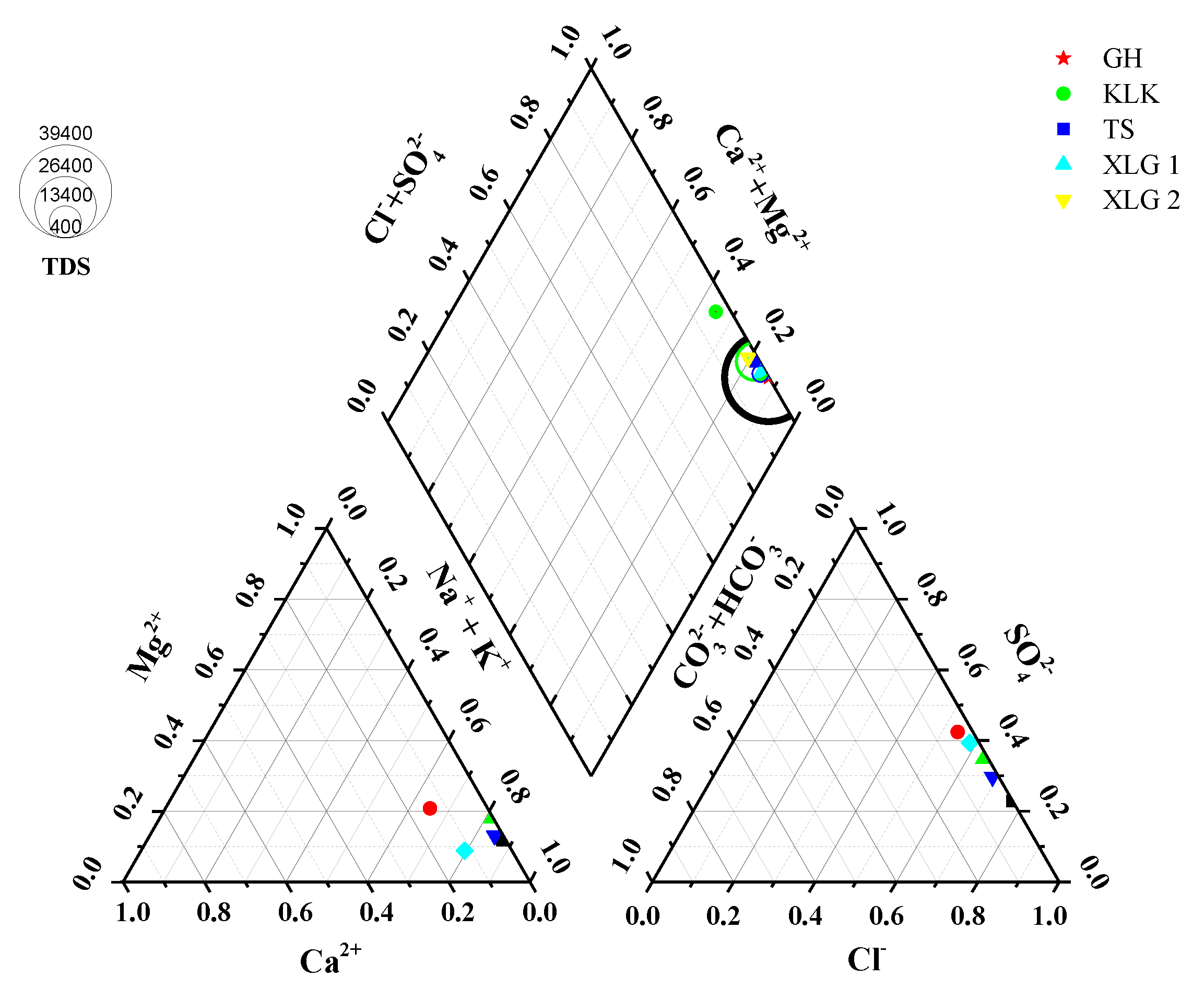
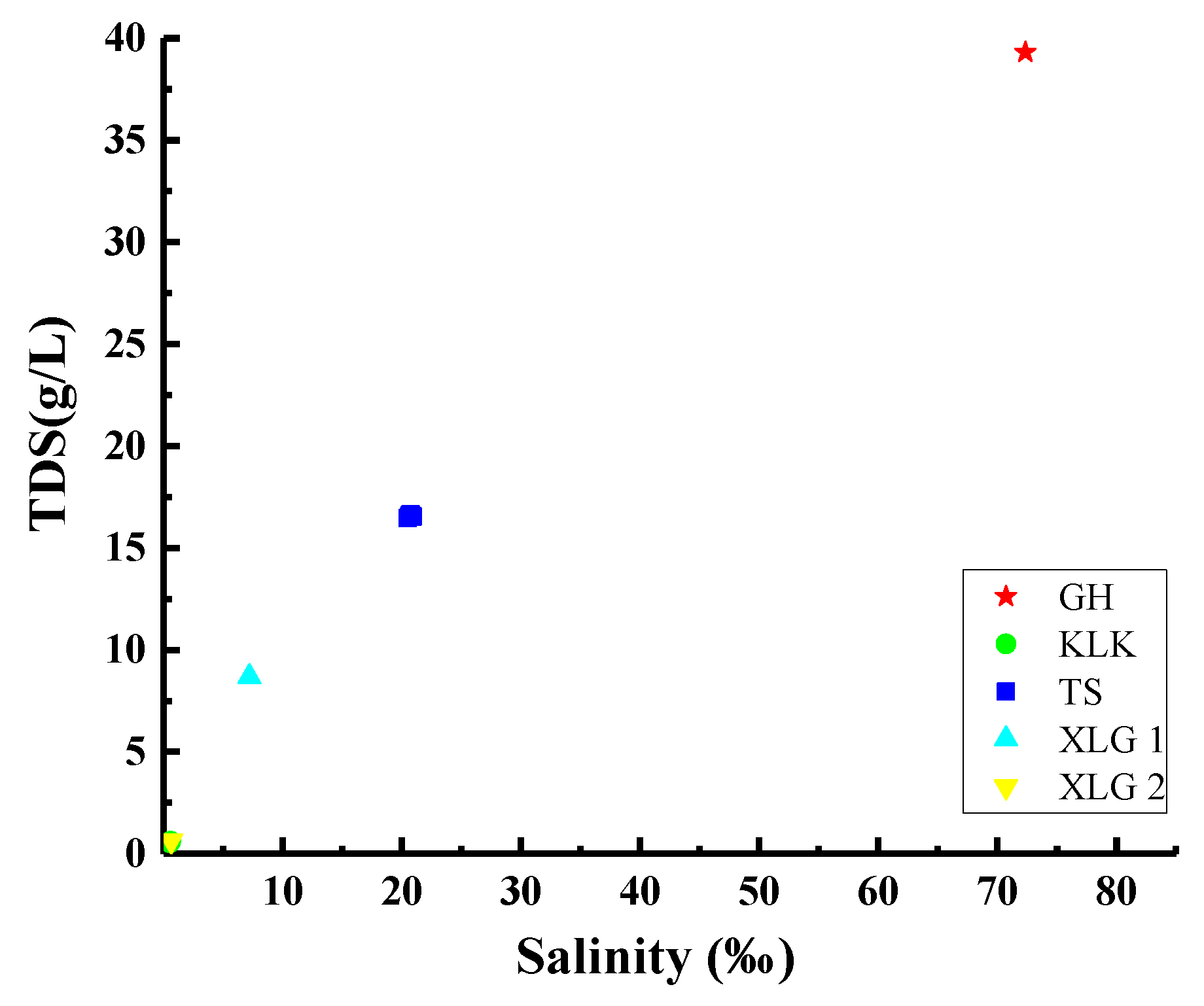


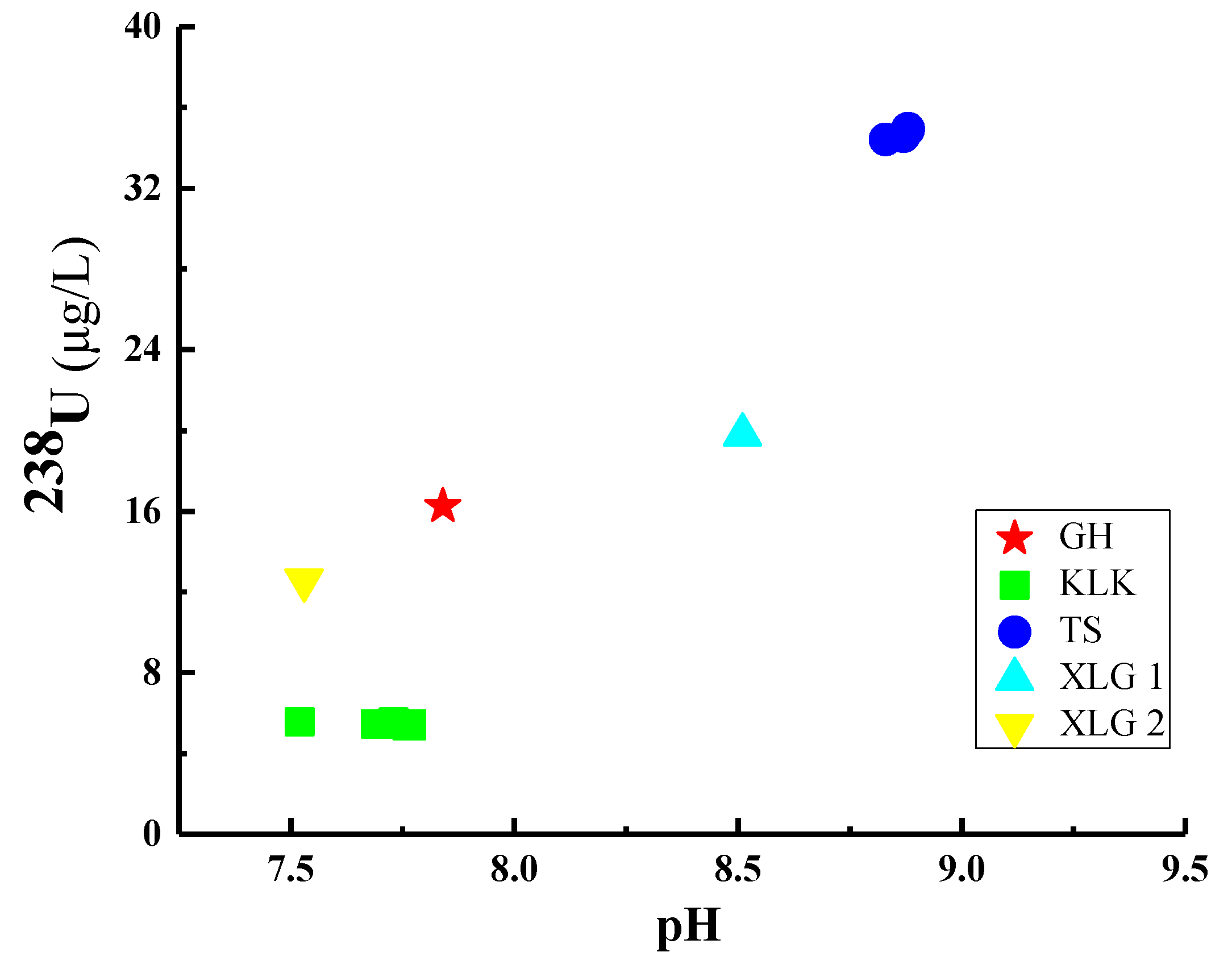
| Batch | Location | Sample No. | Depth (m) | Latitude (°N) | Longitude (°E) | Date |
|---|---|---|---|---|---|---|
| 1 | Tuosu lake | TS-1-1 | 0.5 | 37°08′40″ | 96°57′24″ | 9 December 2017 |
| TS-1-2 | 10 | |||||
| TS-1-3 | 21 | |||||
| TS-2 | 4 | 37°09′11″ | 96°57′49″ | |||
| TS-3-1 | 0.02 | 37°09′05″ | 96°58′18″ | |||
| TS-3-2 | 13 | |||||
| TS-4-1 | 0.02 | 37°09′11″ | 96°58′58″ | |||
| TS-4-2 | 14 | |||||
| TS-5 | 2 | 37°09′18″ | 96°59′31″ | |||
| Gahai lake | GH-1-1 | 0.02 | 37°08′28″ | 97°31′34″ | 10 December 2017 | |
| GH-1-2 | 8 | |||||
| GH-2-1 | 0.02 | 37°08′01″ | 97°32′34″ | |||
| GH-2-2 | 10 | |||||
| GH-3-1 | 0.02 | 37°08′27″ | 97°32′29″ | |||
| GH-3-2 | 9.98 | |||||
| GH-4-1 | 0.02 | 37°08′48″ | 97°32′26″ | |||
| GH-4-2 | 8.98 | |||||
| GH-5-1 | 0.02 | 37°09′19″ | 97°32′21″ | |||
| GH-5-2 | 7.48 | |||||
| 2 | Gahai lake | GH | 0.2 | 37°07′51″ | 97°34′58″ | 15 June 2018 |
| Keluke lake | KLK 1-T | 0.2 | 37°17′24″ | 96°53′34″ | 16 June 2018 | |
| KLK 1-M | 2.5 | |||||
| KLK 1-B | 4.5 | |||||
| KLK 2-T | 0.2 | 37°17′55″ | 96°53′42″ | |||
| KLK 2-M | 1.5 | |||||
| KLK 2-B | 3.5 | |||||
| KLK 3-T | 0.2 | 37°18′27″ | 96°53′55″ | |||
| KLK 3-M | 1 | |||||
| Tuosu lake | TS-1-T | 0.2 | 37°10′21″ | 96°58′34″ | 16 June 2018 | |
| TS-1-M | 8 | |||||
| TS-2-T | 0.2 | 37°10′27″ | 96°58′59″ | |||
| TS-2-M | 4.5 | |||||
| TS-3-T | 0.2 | 37°10′35″ | 96°58′57″ | |||
| TS-3-M | 3 | |||||
| Xiligou lake | XLG 1 | 0.2 | 36°49′10″ | 98°26′52″ | 17 June 2018 | |
| XLG 2 | 0.2 | 36°49′03″ | 98°26′41″ | 17 June 2018 | ||
| 3 | Keluke lake | KLK-W | 0.2 | 37°17′01″ | 96°51′35″ | 22 July 2019 |
| KLK-N | 0.2 | 37°18′56″ | 96°54′07″ | |||
| Bayin river | Upper reaches | 0.2 | 37°26′33″ | 97°44′19″ | 22 July 2019 | |
| Middle reaches | 0.2 | 37°22′37″ | 97°26′44″ | |||
| Delhi city | 0.2 | 37°22′30″ | 97°21′43″ | |||
| Lower reaches | 0.2 | 37°14′09″ | 97°1′22″ | |||
| KLK-TS-River | 0.2 | 37°13′11″ | 96°49′44″ |
| Sample | pH | Conductivity mS/cm | Cl− mg/L | mg/L | b mg/L | b mg/L | NO mg/L | mg/L | Na+ mg/L | K+ mg/L | Mg2+ mg/L | Ca2+ mg/L | TDS (g/L) | Salinity (‰) | 238U (μg/L) | [234U/238U]AR |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| GH | 7.84 | 78.62 | 31,865.59 | 9436.10 | 19.38 | 0.06 | 296.70 | 18.07 | 18,830.66 | 2102.96 | 2852.06 | 199.02 | 39.31 | 72.36a | 16.2 | 1.798 |
| KLK 1-T | 7.73 | 1.09 | 205.23 | 164.40 | 15.04 | 0.04 | 11.17 | 0.50 | 117.45 | 22.75 | 44.86 | 30.69 | 0.54 | 0.54 | 5.5 | 1.505 |
| KLK 1-M | 7.73 | 1.10 | 204.37 | 163.91 | 15.04 | 0.04 | 11.15 | 0.86 | 117.35 | 23.09 | 44.85 | 30.78 | 0.55 | 0.55 | 5.5 | 1.506 |
| KLK 1-B | 7.52 | 1.14 | 204.90 | 164.79 | 9.28 | 0.01 | 10.98 | 1.29 | 117.49 | 22.87 | 44.45 | 30.70 | 0.57 | 0.57 | 5.6 | 1.503 |
| KLK 2-T | 7.69 | 1.11 | 203.59 | 162.11 | 13.72 | 0.03 | 10.85 | 0.75 | 115.90 | 22.90 | 44.34 | 28.74 | 0.55 | 0.55 | 5.5 | 1.504 |
| KLK 2-M | 7.71 | 1.12 | 203.09 | 162.05 | 14.37 | 0.03 | 10.90 | 0.66 | 116.22 | 23.04 | 44.51 | 28.85 | 0.56 | 0.55 | 5.4 | 1.506 |
| KLK 2-B | 7.77 | 1.07 | 198.50 | 159.99 | 16.49 | 0.04 | 10.96 | 0.45 | 116.53 | 23.64 | 44.71 | 28.92 | 0.54 | 0.53 | 5.5 | 1.504 |
| KLK 3-T | 7.76 | 1.22 | 217.37 | 161.77 | 16.12 | 0.04 | 11.95 | 1.08 | 127.04 | 22.98 | 48.29 | 34.47 | 0.61 | 0.60 | 5.4 | 1.502 |
| KLK 3-M | 7.77 | 1.21 | 225.53 | 166.76 | 16.49 | 0.04 | 12.31 | 1.35 | 126.77 | 23.50 | 48.18 | 34.24 | 0.60 | 0.60 | 5.4 | 1.504 |
| TS-1-T | 8.83 | 33.51 | 13,212.45 | 7187.91 | 189.38 | 5.89 | 192.07 | 13.61 | 8294.33 | 848.16 | 2027.81 | 30.91 | 16.54 | 20.91 | 34.4 | 1.462 |
| TS-1-M | 8.87 | 33.20 | 10,942.59 | 5936.21 | 207.65 | 7.08 | 164.27 | 7.29 | 6809.05 | 801.47 | 1675.98 | 29.30 | 16.60 | 20.68 | 34.6 | 1.462 |
| TS-2-T | 8.87 | 33.08 | 10,802.30 | 5880.24 | 207.65 | 7.08 | 160.38 | 5.76 | 6758.64 | 776.27 | 1659.81 | 23.62 | 16.54 | 20.60 | 34.6 | 1.462 |
| TS-2-M | 8.88 | 33.10 | 11,206.53 | 6075.39 | 212.49 | 7.42 | 173.93 | 11.48 | 6944.53 | 855.72 | 1713.14 | 27.32 | 16.55 | 20.62 | 35.0 | 1.461 |
| TS-3-T | 8.88 | 33.29 | 14,840.03 | 8097.02 | 212.49 | 7.42 | 208.02 | 7.03 | 9666.54 | 894.58 | 2353.41 | 29.93 | 16.66 | 20.75 | 34.9 | 1.460 |
| TS-3-M | 8.88 | 33.99 | 11,192.01 | 6104.67 | 212.49 | 7.42 | 168.63 | 5.87 | 7037.38 | 801.03 | 1723.72 | 23.51 | 16.46 | 20.50 | 34.9 | 1.462 |
| XLG 1 | 8.51 | 14.95 | 4030.92 | 1741.98 | 90.64 | 1.35 | 64.35 | 3.73 | 2129.13 | 361.96 | 385.49 | 62.99 | 7.20 | 8.67 | 19.8 | 1.565 |
| XLG 2 | 7.53 | 1.25 | 242.26 | 163.88 | 9.49 | 0.01 | 12.94 | 3.37 | 196.65 | 23.60 | 24.53 | 32.25 | 0.63 | 0.63 | 12.6 | 1.673 |
| Sample | 238U (μg/L) | Average Value | [234U/238U]AR | Average Value | |
|---|---|---|---|---|---|
| KLK-T | 1 | 5.5 | 5.4 | 1.505 | 1.504 |
| 2 | 5.5 | 1.504 | |||
| 3 | 5.4 | 1.502 | |||
| KLK-M | 1 | 5.5 | 5.5 | 1.506 | 1.505 |
| 2 | 5.4 | 1.506 | |||
| 3 | 5.4 | 1.504 | |||
| KLK-B | 1 | 5.6 | 5.5 | 1.503 | 1.503 |
| 2 | 5.5 | 1.504 | |||
| TS-T | 1 | 34.4 | 34.6 | 1.462 | 1.461 |
| 2 | 34.6 | 1.462 | |||
| 3 | 34.9 | 1.460 | |||
| TS-M | 1 | 34.6 | 34.8 | 1.462 | 1.462 |
| 2 | 35.0 | 1.461 | |||
| 3 | 34.9 | 1.462 | |||
| Sample | Depth (m) | 238U (μg/L) | Average Value | [234U/238U] AR | Average Value | |
|---|---|---|---|---|---|---|
| KLK-1 | T | 0.2 | 5.5 | 5.5 | 1.505 | 1.505 |
| M | 2.5 | 5.5 | 1.506 | |||
| B | 4.5 | 5.6 | 1.503 | |||
| KLK-2 | T | 0.2 | 5.5 | 5.5 | 1.504 | 1.505 |
| M | 1.5 | 5.4 | 1.506 | |||
| B | 3.5 | 5.5 | 1.504 | |||
| KLK-3 | T | 0.2 | 5.4 | 5.4 | 1.502 | 1.503 |
| M | 1 | 5.4 | 1.504 | |||
| TS-1 | T | 0.2 | 34.4 | 34.5 | 1.462 | 1.462 |
| M | 8 | 34.6 | 1.462 | |||
| TS-2 | T | 0.2 | 34.6 | 34.8 | 1.462 | 1.462 |
| M | 4.5 | 35.0 | 1.461 | |||
| TS-3 | T | 0.2 | 34.9 | 34.9 | 1.460 | 1.461 |
| M | 3 | 34.9 | 1.462 | |||
| Sample | 238U (μg/L) | [234U/238U]AR | |
|---|---|---|---|
| BY-River | upper reaches | 6.0 | 1.392 |
| middle reaches | 5.9 | 1.402 | |
| Delhi city | 6.2 | 1.408 | |
| lower reaches | 2.8 | 1.464 | |
| KLK-TS-River | 5.6 | 1.482 | |
| KLK | KLK-W | 5.6 | 1.476 |
| KLK-N | 5.3 | 1.451 | |
| Season | Sample | Depth (m) | 238U (μg/L) | Average Value | [234U/238U]AR | Average Value |
|---|---|---|---|---|---|---|
| December (Winter) | GH-1 | 0.02 | 18.5 | 18.4 | 1.791 | 1.792 |
| 8.0 | 18.5 | 1.792 | ||||
| GH-2 | 0.02 | 18.3 | 1.792 | |||
| 10.0 | 18.3 | 1.793 | ||||
| GH-3 | 0.02 | 18.2 | 1.793 | |||
| 9.98 | 18.4 | 1.791 | ||||
| GH-4 | 0.02 | 18.3 | 1.796 | |||
| 8.98 | 18.4 | 1.794 | ||||
| GH-5 | 0.02 | 18.3 | 1.793 | |||
| 7.48 | 18.3 | 1.794 | ||||
| June (Summer) | GH | 0.2 | 16.2 | 16.2 | 1.798 | 1.798 |
| December (Winter) | TS-1 | 0.5 | 35.4 | 35.5 | 1.464 | 1.463 |
| 10.0 | 35.8 | 1.462 | ||||
| 21.0 | 35.6 | 1.462 | ||||
| TS-2 | 4.0 | 35.2 | 1.462 | |||
| TS-3 | 0.02 | 35.2 | 1.463 | |||
| 13.0 | 35.3 | 1.46 | ||||
| TS-4 | 0.02 | 35.5 | 1.464 | |||
| 14.0 | 35.7 | 1.460 | ||||
| TS-5 | 2.0 | 35.8 | 1.461 | |||
| June (Summer) | TS-1 | 0.2 | 34.4 | 34.7 | 1.462 | 1.462 |
| 8.0 | 34.6 | 1.462 | ||||
| TS-2 | 0.2 | 34.6 | 1.462 | |||
| 4.5 | 35.0 | 1.461 | ||||
| TS-3 | 0.2 | 34.9 | 1.460 | |||
| 3.0 | 34.9 | 1.462 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, C.; Zhang, P.; Li, X.; Ning, Y.; Tan, L.; Edwards, R.L.; Yao, X.; Cheng, H. Distribution Characteristics and Influencing Factors of Uranium Isotopes in Saline Lake Waters in the Northeast of Qaidam Basin. Minerals 2020, 10, 74. https://doi.org/10.3390/min10010074
Zhao C, Zhang P, Li X, Ning Y, Tan L, Edwards RL, Yao X, Cheng H. Distribution Characteristics and Influencing Factors of Uranium Isotopes in Saline Lake Waters in the Northeast of Qaidam Basin. Minerals. 2020; 10(1):74. https://doi.org/10.3390/min10010074
Chicago/Turabian StyleZhao, Chen, Pu Zhang, Xiangzhong Li, Youfeng Ning, Liangcheng Tan, R. Lawrence Edwards, Xiunan Yao, and Hai Cheng. 2020. "Distribution Characteristics and Influencing Factors of Uranium Isotopes in Saline Lake Waters in the Northeast of Qaidam Basin" Minerals 10, no. 1: 74. https://doi.org/10.3390/min10010074
APA StyleZhao, C., Zhang, P., Li, X., Ning, Y., Tan, L., Edwards, R. L., Yao, X., & Cheng, H. (2020). Distribution Characteristics and Influencing Factors of Uranium Isotopes in Saline Lake Waters in the Northeast of Qaidam Basin. Minerals, 10(1), 74. https://doi.org/10.3390/min10010074






