Contrasting Fluids in the Svetlinsk Gold-Telluride Hydrothermal System, South Urals
Abstract
1. Introduction
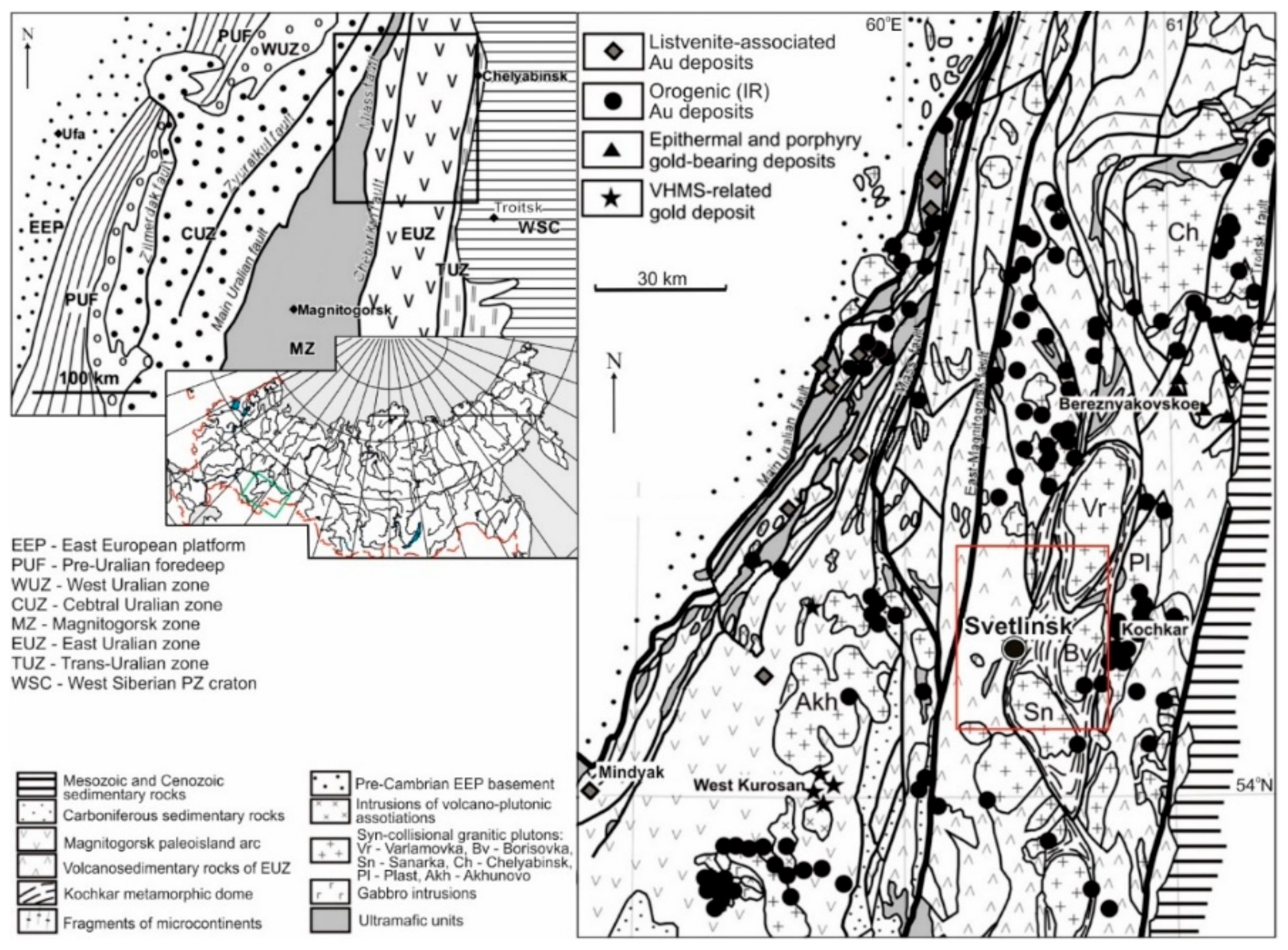
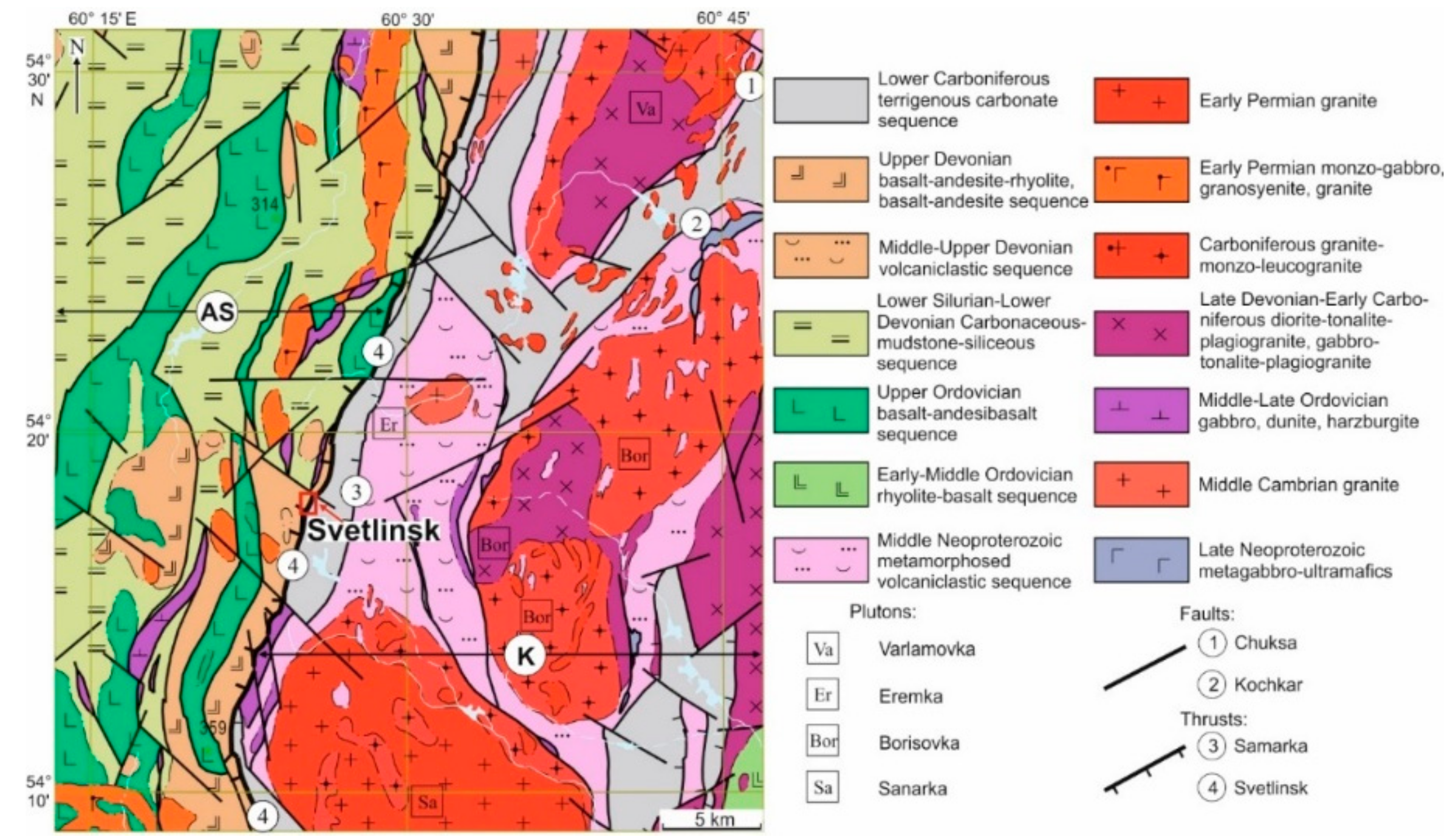
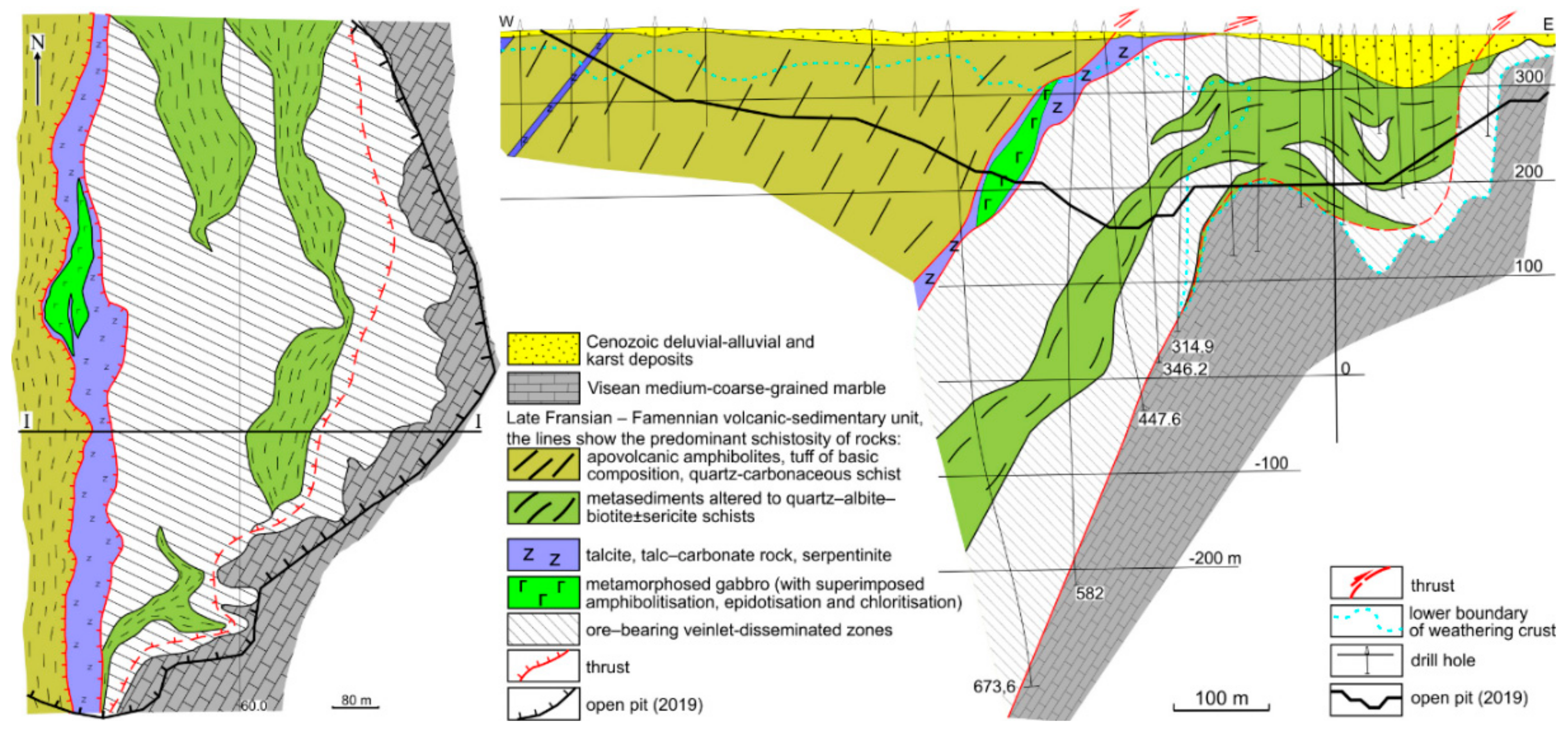
2. Geological Setting
3. Materials and Methods
4. Fluid Inclusion Study
4.1. Fluid Inclusion Petrography
- -
- type I (LH2O + VCO2 ± LCO2) is two-phase or three-phase aqueous-carbonic FIs containing liquid water as well as liquid and gaseous CO2;
- -
- type II (VCO2 ± CH4,N2) is vapour-rich CO2 one-phase or two-phase FIs, with minor CH4 and N2, sometimes with a thin liquid rim;
- -
- type III (LH2O + VH2O) is two-phase liquid–vapour aqueous inclusions;
- -
- type IV (LH2O + VH2O + S) is three-phase or multi-phase FIs, consisting of H2O liquid, gas bubble, and one or more daughter minerals. The cubic crystals in such inclusions are usually halite, based on a refractive index. Rarely the solid phase is represented by calcite and solid phases of complex composition (chalcoalumite group) based on Raman spectroscopy data. Additionally, a spherical native sulphur phase was detected in some FIs of I, II, and IV types (subtypes Ia, IIa, and IVa, respectively).
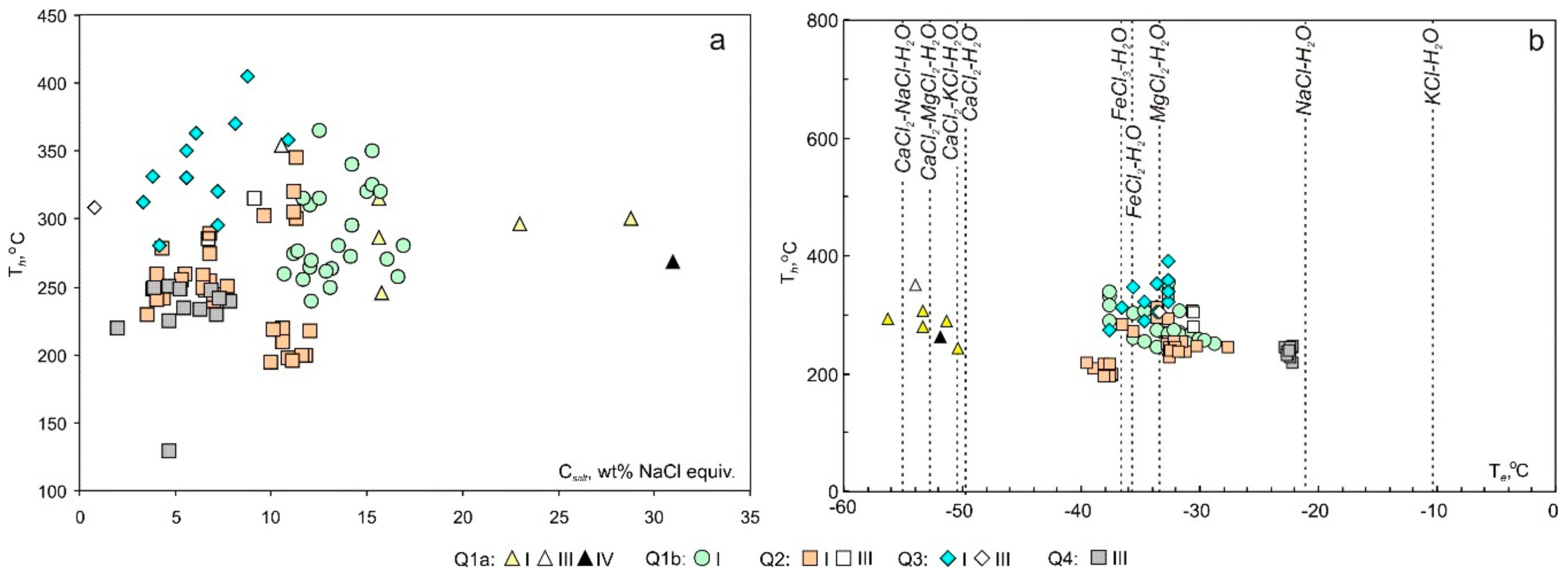
4.2. Microthermometry
4.2.1. Q1a (in the Footwall Marble)
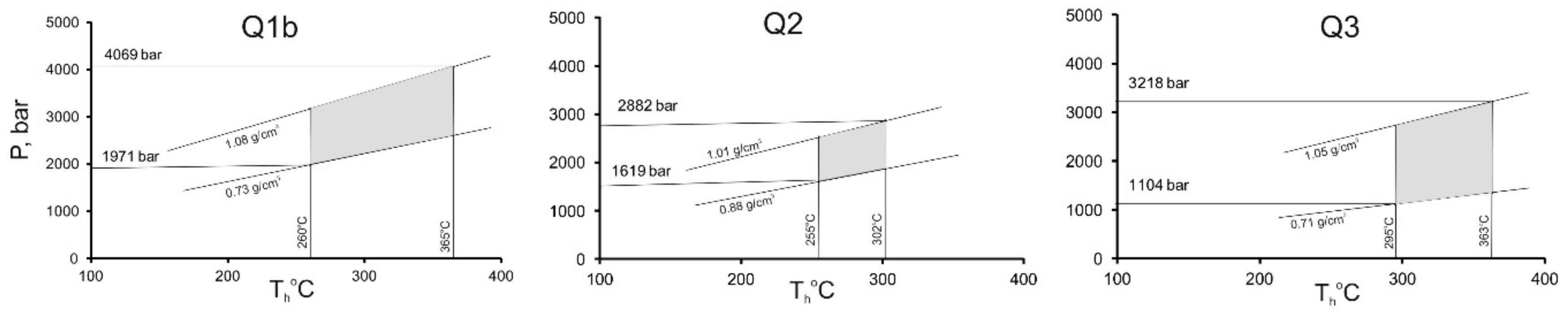
4.2.2. Q1b (in Metaterrigenic Rocks)
4.2.3. Q2
4.2.4. Q3
4.2.5. Q4
4.3. Crush-Leach Analysis
4.4. Raman Spectroscopy
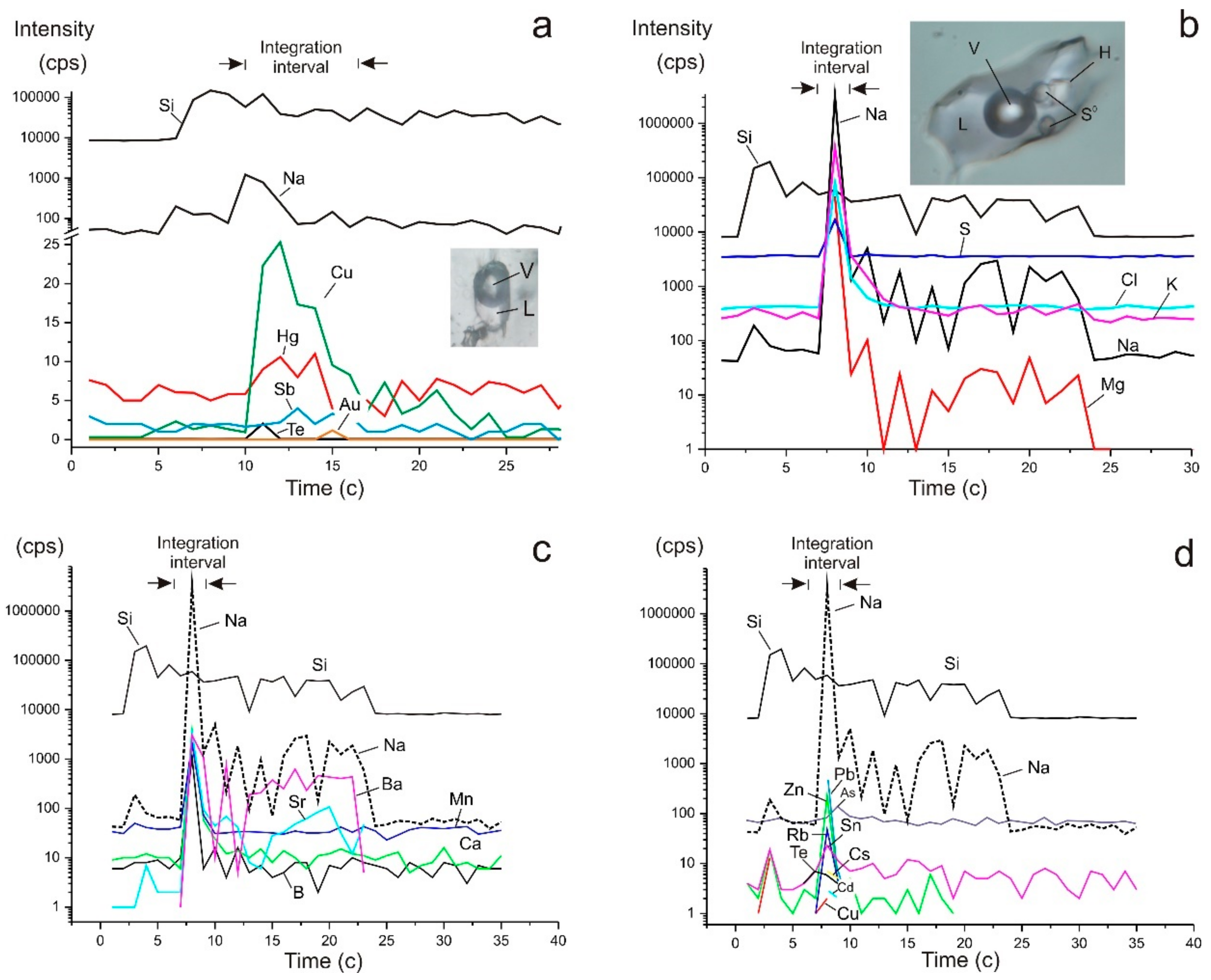
4.5. LA-ICP-MS of Fluid Inclusions
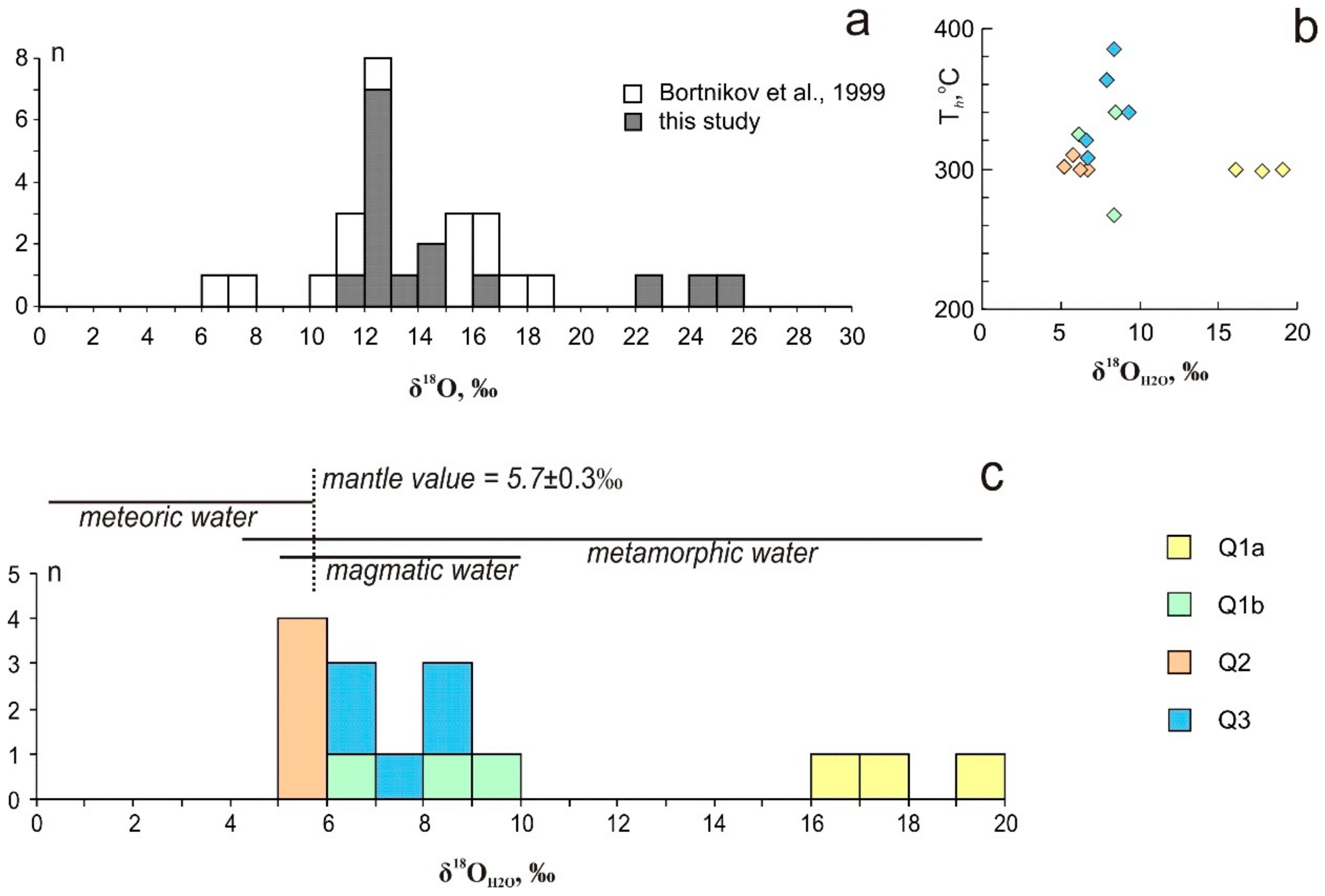
5. Oxygen Isotopes
6. Discussion
6.1. Fluid Immiscibility or Fluid Mixing
6.2. PTX Parameters of Ore Formation
6.2.1. Temperature
6.2.2. Pressure
6.2.3. Fluid Composition
6.2.4. Sulphur in Fluid Inclusions
6.2.5. Oxidation State of Ore Forming Fluid
6.3. Sources of Hydrothermal Fluids
6.4. Model of the Svetlinsk Hydrothermal System
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Component | Content | ±σ | Component | Content | ±σ | Component | Content | ±σ |
|---|---|---|---|---|---|---|---|---|
| Na2O, % | 13.7 | 0.3 | Ni | 38.8 | 0.2 | Pr | 37.9 | 1 |
| Al2O3 | 2.03 | 0.04 | Cu | 37.8 | 1.5 | Nd | 35.5 | 0.7 |
| SiO2 | 72.1 | 0.6 | Zn | 39.1 | 1.7 | Sm | 37.7 | 0.8 |
| CaO | 11.9 | 0.1 | Ga | 36.9 | 1.5 | Eu | 35.6 | 0.8 |
| Li, ppm | 40.2 | 1.3 | Ge | 36.1 | 3.8 | Gd | 37.3 | 0.9 |
| Be | 37.5 | 1.5 | As | 35.7 | 5.5 | Tb | 37.6 | 1.1 |
| B | 34.3 | 1.7 | Se | 16.3 | 1.9 | Dy | 35.5 | 0.7 |
| F | 80 | 89 | Rb | 31.4 | 0.4 | Ho | 38.3 | 0.8 |
| Na | 101,645 | 2226 | Sr | 78.4 | 0.2 | Er | 38 | 0.9 |
| Mg | 68 | 5.1 | Y | 38.3 | 1.4 | Tm | 36.8 | 0.6 |
| Al | 10,747 | 212 | Zr | 37.9 | 1.2 | Yb | 39.2 | 0.9 |
| Si | 336,467 | 2800 | Nb | 38.9 | 2.1 | Lu | 37 | 0.9 |
| P | 46.6 | 6.9 | Mo | 37.4 | 1.5 | Hf | 36.7 | 1.2 |
| S | 377 | 70 | Rh | 0.91 | 0.02 | Ta | 37.6 | 1.9 |
| Cl | 142 | 58 | Pd | 1.05 | 0.1 | W | 38 | 1.1 |
| K | 62.3 | 2.4 | Ag | 22 | 0.3 | Re | 6.63 | 0.61 |
| Ca | 85,000 | 714 | Cd | 28.1 | 1.1 | Ir | 0.0045 | |
| Sc | 39.9 | 2.5 | In | 38.9 | 2.1 | Pt | 2.51 | 0.1 |
| Ti | 44 | 2.3 | Sn | 38.6 | 1.3 | Au | 4.77 | 0.31 |
| V | 38.8 | 1.2 | Sb | 34.7 | 1.8 | Tl | 14.9 | 0.5 |
| Cr | 36.4 | 1.5 | Cs | 42.7 | 1.8 | Pb | 38.6 | 0.2 |
| Mn | 38.7 | 0.9 | Ba | 39.3 | 0.9 | Bi | 30.2 | 2.3 |
| Fe | 51 | 2 | La | 36 | 0.7 | Th | 37.79 | 0.08 |
| Co | 35.5 | 1 | Ce | 38.4 | 0.7 | U | 37.38 | 0.08 |
References
- Shackleton, J.M.; Spry, P.G.; Bateman, R. Telluride mineralogy of the Golden Mile deposit, Kalgoorlie, Western Australia. Can. Mineral. 2003, 41, 1503–1524. [Google Scholar] [CrossRef]
- Spry, P.; Foster, F.; Truckle, J.; Chadwick, T. The mineralogy of the Golden Sunlight gold–silver telluride deposit, Whitehall, Montana, USA. Mineral. Petrol. 1997, 59, 143–164. [Google Scholar] [CrossRef]
- Kelley, K.D.; Romberger, S.B.; Beaty, D.W.; Pontius, J.A.; Snee, L.W.; Stein, H.J.; Thompson, T.B. Geochemical and geochronological constraints on the genesis of Au-Te Deposits at Cripple Creek, Colorado. Econ. Geol. 1998, 93, 981–1012. [Google Scholar] [CrossRef]
- Ahmad, H.; Solomon, M.; Walshe, J.L. Mineralogical and geochemical studies of the Emperor gold telluride deposit, Fiji. Econ. Geol. 1987, 82, 345–370. [Google Scholar] [CrossRef]
- Pals, D.; Spry, P. Telluride mineralogy of the low-sulfidation epithermal Emperor gold deposit, Vatukoula, Fiji. Mineral. Petrol. 2003, 79, 285–307. [Google Scholar] [CrossRef]
- Cooke, D.R.; McPhail, D. Epithermal Au–Ag–Te mineralization, Acupan, Baguio district, Philippines: Numerical simulations of mineral deposition. Econ. Geol. 2001, 96, 109–131. [Google Scholar]
- Ciobanu, C.L.; Cook, N.J.; Capraru, N.; Damian, G.; Cristea, P. Mineral assemblages from the vein salband at Sacarimb, Golden Quadrilateral, Romania: I. Sulphides and sulphosalts. Geokh. Mineral. Petrol. 2005, 43, 47–55. [Google Scholar]
- Cook, N.J.; Ciobanu, C.L.; Spry, P.G.; Voudouris, P. Understanding gold-(silver)-telluride-(selenide) mineral deposits. Episodes 2009, 32, 249–263. [Google Scholar] [CrossRef]
- Jensen, E.P.; Barton, M.D. Geology of the Cripple Creek district Part I.; General summary of geologic features and regional setting, and Part II. Time-space synthesis of magmatic and hydrothermal activity. In Geology of the Cripple Creek Gold-Telluride Deposit, Colorado: Guidebook Prepared for the SEG and IGCP-486 Pre-Meeting Field Trip #411; Jensen, E.P., Spry, P.G., Eds.; GSA Annual Meeting and Exposition: Phoenix, AZ, USA, 2007. [Google Scholar]
- Mikucki, E.J. Hydrothermal transport and depositional processes in Archean lode-gold systems: A review. Ore Geol. Rev. 1998, 13, 307–321. [Google Scholar] [CrossRef]
- Pak, S.J.; Choi, S.-G.; Oh, C.-W.; Heo, C.-H.; Choi, S.-H.; Kim, S.-W. Genetic environment of the intrusion-related Yuryang Au-Te Deposit in the Cheonan Metallogenic Province, Korea. Resour. Geol. 2006, 56, 117–132. [Google Scholar] [CrossRef]
- Hu, H.B.; Mao, J.W.; Niu, S.Y.; Li, Y.F.; Li, M.W. Geology and geochemistry of telluride–Au deposits in the Pingyi area, western Shandong, China. Mineral. Petrol. 2006, 87, 209–240. [Google Scholar] [CrossRef]
- Mao, J.W.; Li, Y.Q.; Goldfarb, R.J.; He, Y. Fluid Inclusion of the Dongping telluride gold deposit, Hebei province: Implication of the contribution of mantle-derived fluids to metallogenesis. Econ. Geol. 2003, 98, 517–534. [Google Scholar]
- Voudouris, P.; Spry, P.G.; Melfos, V.; Alfieris, D. Tellurides and bismuth sulfosalts in gold occurrences of Greece: Mineralogy and genetic considerations. In Proceedings of the Field Workshop of IGCP-486, Espoo, Finland, 26–31August 2007; Volume 53, pp. 85–94, Geological Survey of Finland Opas. [Google Scholar]
- Fedoseev, V.V. Mineral resources of JSC “Yuzhuralzoloto” in the Chelyabinsk region. Mineral resources of Russia. Econ. Manag. 2007, 20–27. [Google Scholar]
- Berger, R.V.; Fedoseev, V.V.; Saraskin, A.V. State-of-the-art and prospects of mineral and raw materials supply base expansion at UGC Gold Mining Company in the South Ural. Gorn. Zhurnal 2017, 9, 6–11. [Google Scholar] [CrossRef]
- Gosselin, P.; Dubé, B. Gold Deposits of Canada: Distribution, Geological Parameters and Gold Content; Open File (Geological Survey of Canada), 4896; Geological Survey of Canada: Quebec, QC, Canada, 2005. [Google Scholar]
- Poulsen, K.H.; Robert, F.; Dubé, B. Geological Classification of Canadian Gold Deposits; Geological Survey of Canada Bulletin: Ottawa, ON, Canada, 2000. [Google Scholar]
- Groves, D.I.; Goldfarb, R.J.; Robert, F.; Hart, C.J.R. Gold deposits in metamorphic belts: Overview of current understanding, outstanding problems, future research, and exploration significance. Econ. Geol. 2003, 98, 1–29. [Google Scholar]
- Bortnikov, N.S.; Murzin, V.V.; Sazonov, V.N.; Prokof’ev, V.Y.; Stolyarov, M.I. The Svetlinsk gold-telluride deposit, Urals, Russia: Mineral paragenesis, fluid inclusions and stable isotope studies. In Mineral Deposits: Processes to Proceessing, Proceedings of the Fifth Biennial SGA Meeting and the Tenth Quadrennial JAGOD Symposium, London, UK, 22–25 August 1999; A.A.Balkema Publishers: Rotterdam, The Netherlands, 1999; Volume 1, pp. 21–24. [Google Scholar]
- Sazonov, V.N.; Popov, V.A.; Grigoryev, N.A.; Murzin, V.V.; Metsner, E.I. Crust-Mantle Mineralisation in the Salic Blocks of Eugeosyncline; UrO USSR AN: Sverdlovsk, Russia, 1989. (In Russian) [Google Scholar]
- Bortnikov, N.S. Geochemistry and origin of the ore-forming fluids in hydrothermal-magmatic systems in tectonically active zones. Geol. Ore Dep. 2006, 48, 1–22. [Google Scholar] [CrossRef]
- Vikent’eva, O.V.; Bortnikov, N.S. The large Svetlinsk Au-Te deposit, South Urals: Telluride mineralization for genetic reconstructions. In Proceedings of the 13th Biennial SGA Meeting, Nancy, France, 24–27 August 2015; pp. 851–854. [Google Scholar]
- Vikent’ev, I.V.; Sidorova, N.V.; Vikent’eva, O.V.; Bortnikov, N.S.; Su, S.G.; Luo, Z.H. Tellurides in the Laowan gold deposit (East China): New evidence for a magmatic source of hydrothermal fluids. Dokl. Earth Sci. 2015, 462, 581–585. [Google Scholar] [CrossRef]
- Fedoseev, V.V.; Legotkin, V.N.; Savinov, A.I. Svetlinsk gold deposit in the Southern Urals (Report on the detailed exploration conducted by Kochkar geological-prospecting team in 1985–1988 with the calculation of reserves as of 01.10.1988). Plast 1988. [Google Scholar]
- Kisters, A.F.M.; Meyer, F.M.; Seravkin, I.B.; Znamensky, S.E.; Kosarev, A.M.; Ertl, R.G.W. The geological setting of lode-gold deposits in the central Southern Urals: A review. Geol. Rundsch. 1999, 87, 603–616. [Google Scholar] [CrossRef]
- Tevelev, A.V.; Kosheleva, I.A. Geological Structure and History of the Southern Urals (East Ural Uplift and Trans-Urals); MSU Publishing House: Moscow, Russia, 2002. (In Russian) [Google Scholar]
- Kissin, A.Y.; Pritchin, M.E. Fault tectonics of the Svetlinskoye gold deposit (South Urals) and its role in ore control. Bull. Perm Univ. Geol. 2015, 3, 34–42. (In Russian) [Google Scholar] [CrossRef][Green Version]
- Kisin, A.Y.; Koroteev, V.A. Block Folding and Ore Genesis; IGG UB RAS: Yekaterinburg, Russia, 2017. (In Russian) [Google Scholar]
- Sazonov, V.N.; van Herk, A.H.; de Boorder, H. Spatial and temporal distribution of gold deposits in the Urals. Econ. Geol. 2001, 96, 685–703. [Google Scholar] [CrossRef]
- Ogorodnikov, V.N.; Sazonov, V.N.; Polenov, Y.A. Minerageny of the Suture Zones of the Urals. Part 1. Kochkarskiy Ore District (South Urals); UGGA Publ.: Ekaterinburg, Russia, 2004. (In Russian) [Google Scholar]
- Belogub, E.V.; Melekestseva, I.Y.; Novoselov, K.A.; Zabotina, M.V.; Tret’yakov, G.A.; Zaykov, V.V.; Yuminov, A.M. Listvenite-related gold deposits of the South Urals (Russia): A review. Ore Geol. Rev. 2017, 85, 247–270. [Google Scholar] [CrossRef]
- Puzhakov, B.A.; Shokh, V.D.; Schulkina, N.E.; Shulkin, E.P.; Tarelkina, E.A.; Dolgova, O.Y. State Geological Map of Russian Federation; Scale 1: 200 000. South Ural Series. Sheet N-41-XIII (Plast). Explanatory Note; Moscow Branch of VSEGEI: Moscow, Russia, 2018. [Google Scholar]
- Bortnikov, N.S.; Vikentyev, I.V. Endogenous metallogeny of the Urals. In Proceedings of the 12th Biennial SGA Meeting, Uppsala, Sweden, 12–15 August 2013; pp. 1508–1511. [Google Scholar]
- Burmistrov, A.A. Structural conditions of localization of gold-quartz-sulphide mineralization of the Svetlinsk deposit (Yu. Ural). In Lomonosov Reading–2012; MSU; 2012; Available online: http://geo.web.ru/pubd//2012/06/01/0001186421/pdf/burmistrov_2012.pdf (accessed on 30 December 2019). (In Russian)
- Bodnar, R.J.; Vityk, M.O. Interpretation of microthermometric data for H2O–NaCl fluid inclusions. In Fluid Inclusions in Minerals: Methods and Applications; De Vivo, B., Frezzotti, M.L., Eds.; Fluids Research Laboratory, Department of Geological Sciences, Virginia Tech: Blacksburg, VA, USA, 1994. [Google Scholar]
- Collins, P.L.P. Gas hydrates in CO2 bearing fluid inclusions and the use of freezing data for estimation of salinity. Econ. Geol. 1979, 74, 1435–1444. [Google Scholar] [CrossRef]
- Borisenko, A.S. Cryometric study of salt composition of solutions in gas–liquid inclusions in minerals. Geol. Geofiz. 1977, 8, 16–28. (In Russian) [Google Scholar]
- Prokofyev, V.Y.; Naumov, V.B. Geochemical features of the mineralizing solutions at the Zyryanov sulfide-polymetallic deposit, Rudny Altay. Geochem. Int. 1987, 24, 50–74. [Google Scholar]
- Kalyuzhny, V.A. Principles of the Theory on Mineral-Forming Fluids; Naukova Dumka: Kiev, USSR, 1982. (In Russian) [Google Scholar]
- Kerkhof, A.M. The System CO2-CH4-N2 in Fluid Inclusions: Theoretical Modeling and Geological Applications; Free University Press: Amsterdam, The Netherlands, 1988. [Google Scholar]
- Thiery, R.; Kerkhof, A.M.; Dubessy, J. PTX properties of CH4-CO2 and CO2-N2 fluid inclusions: Modeling for T < 31 °C and P < 400 bars. Eur. J. Mineral. 1994, 6, 753–771. [Google Scholar]
- Brown, P.E. Flincor: A microcomputer program for the reduction and investigation of fluid inclusion data. Am. Min. 1989, 74, 1390–1393. [Google Scholar]
- Bakker, R.J. Package FLUIDS 1. Computer programs for analysis of fluid inclusions data and for modeling bulk fluid properties. Chem. Geol. 2003, 194, 3–23. [Google Scholar] [CrossRef]
- Roedder, E. Fluid inclusions. In Review in Mineralogy; Ribbe, P.H., Ed.; Mineralogical Society of America: Washington, DC, USA, 1984; Volume 12. [Google Scholar]
- Kryazhev, S.G.; Prokof’ev, V.Y.; Vasyuta, Y.V. Use of method ICP MS at the analysis of composition of ore-forming fluids. Vestn. MSU Geol. 2006, 4, 30–36. (In Russian) [Google Scholar]
- Jochum, K.; Weis, U.; Stoll, B.; Kuzmin, D.; Yang, Q.; Raczek, I.; Jacob, D.E.; Stracke, A.; Birbaum, K.; Frick, D.A.; et al. Determination of reference values for NIST SRM 610–617 glasses following ISO guidelines. Geostand. Geoanalytical Res. A 2011 Int. Assoc. Geoanal. 2011, 35, 397–429. [Google Scholar] [CrossRef]
- Longerich, H.P.; Jackson, S.E.; Günther, D. Laser ablation inductively coupled plasma mass spectrometric transient signal data acquisition and analyte concentration calculation. J. Anal. At. Spectrom. 1996, 11, 899–904. [Google Scholar] [CrossRef]
- Zhang, L.-G.; Liu, J.-X.; Zhou, H.B.; Chen, Z.-S. Oxygen isotope fractionation in the quartz-water-salt system. Econ. Geol. 1989, 89, 1643–1650. [Google Scholar] [CrossRef]
- Sheppard, S.M.F. Characterisation and isotopic variations in natural water. Rev. Mineral. 1986, 16, 165–183. [Google Scholar]
- Ramboz, C.; Pichavant, M.; Weisbrod, A. Fluid immiscibility in natural processes: Use and misuse of fluid inclusion data: II. Interpretation of fluid inclusion data in terms of immiscibility. Chem. Geol. 1982, 37, 29–48. [Google Scholar] [CrossRef]
- Bowers, T.S.; Helgeson, H.C. Calculation of the thermodynamic and geochemical consequences of nonideal mixing in the system H2O-CO2-NaCl on phase relations in geologic systems: Equation of state for H2O-CO2-NaCl fluids at high pressures and temperatures. Geochim. Cosmochim. Acta 1983, 47, 1247–1276. [Google Scholar] [CrossRef]
- Díaz-Gonzáles, L.; Santoyo, E. A new precise calibration of the Na/K geothermometer using a world database of geothermal fluids and improved geochemometric techniques. Geochim. Cosmochim. Acta 2008, 72, A215. [Google Scholar]
- Pester, N.J.; Rough, M.; Ding, K.; Seyfried, J.W.E. A new Fe/Mn geothermometer for hydrothermal systems: Implications for high-salinity fluids at 13°N on the East Pacific Rise. Geochim. Cosmochim. Acta 2011, 75, 7881–7892. [Google Scholar] [CrossRef]
- Sack, R.O.; Loucks, R.R. Thermodynamic properties of tetrahedrite-tennantites: Constraints on the interdependence of the Ag–Cu, Fe–Zn, Cu–Fe, and As–Sb exchange reactions. Am. Mineral. 1985, 70, 1270–1289. [Google Scholar]
- Sack, R.O.; Brackebusch, F.W. Fahlore as an indicator of mineralisation temperature and gold fineness. Can. Min. Metall. Bull. 2004, 97, 78–83. [Google Scholar]
- Sibson, R.H.; Robert, F.; Poulson, K.H. High-angle reverse faults, fluid pressure cycling and mesothermal gold-quartz deposits. Geology 1988, 16, 551–555. [Google Scholar] [CrossRef]
- Yardley, B.; Gleeson, S.; Bruce, S.; Banks, D. Origin of retrograde fluids in metamorphic rocks. J. Geoch. Explor. 2000, 69–70, 281–285. [Google Scholar] [CrossRef]
- Cox, S. Injection-driven swarm seismicity and permeability enhancement: Implications for the dynamics of hydrothermal ore systems in high fluid-flux, overpressured faulting regimes. Econ. Geol. 2016, 111, 559–587. [Google Scholar] [CrossRef]
- Bodnar, R.J.; Lecumberri-Sanchez, P.; Moncada, D.; Steele-MacInnis, M. Fluid inclusions in hydrothermal or deposits. In Treatise on Geochemistry, 2nd ed.; Holland, H.D., Turekian, K.K., Eds.; Elsevier: Oxford, UK, 2014; Volume 13, pp. 119–142. [Google Scholar]
- Lowenstern, J.B. Carbon dioxide in magmas and implications for hydrothermal systems. Mineral. Dep. 2001, 36, 490–502. [Google Scholar] [CrossRef]
- Bottrell, S.H.; Carr, L.P.; Dubessy, J. A nitrogen-rich metamorphic fluid and coexisting minerals in slates from North Wales. Mineral. Mag. 1988, 52, 451–457. [Google Scholar] [CrossRef]
- Baker, T.; Lang, R.J. Fluid inclusion characteristics of intrusion-related gold mineralisation, Tombstone-Tungsten magmatic belt, Yukon Territory, Canada. Miner. Dep. 2001, 36, 563–582. [Google Scholar] [CrossRef]
- Chicharro, E.; Boiron, M.-C.; López-García, J.Á.; Barfod, D.N.; Villaseca, C. Origin, ore forming fluid evolution and timing of the Logrosán Sn–(W) ore deposits (Central Iberian Zone, Spain). Ore Geol. Rev. 2016, 72, 896–913. [Google Scholar] [CrossRef]
- Moin, B.; Guillot, C.; Gibert, F. Controls of the composition of nitrogen-rich fluids originating from reaction with graphite and ammonium-bearing biotite. Geochim. Cosmochim. Acta 1994, 58, 5503–5523. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, K.; Konare, Y. N2-rich fluid in the vein-type Yangjingou scheelite deposit, Yanbian, NE China. Sci. Rep. 2018, 8, 5662. [Google Scholar] [CrossRef]
- Andersen, T.; Burke, E.A.J.; Neumann, E.R. Nitrogen-rich fluid in the upper mantle: Fluid inclusions in spinel dunite from Lanzarote, Canary Islands. Contr. Mineral. Petrol. 1995, 120, 20. [Google Scholar] [CrossRef]
- Lang, J.R.; Baker, T.; Hart, C.J.R.; Mortensen, J.K. An exploration model for intrusion-related gold systems. Soc. Econ. Geol. Newsl. 2000, 40, 1–15. [Google Scholar]
- McCoy, D.; Newberry, R.J.; Layer, P.; DiMarchi, J.J.; Bakke, A.; Masterman, S.; Minehane, D.L. Plutonic-Related Gold Deposits of Interior Alaska, Mineral Deposits of Alaska, In Mineral Deposits of Alaska; Goldfarb, R.J., Miller, L.D., Eds.; Society of Economic Geologists: Littleton, CO, USA, 1997; Volume 9, pp. 191–241. [Google Scholar]
- Vikent’eva, O.V.; Bortnikov, N.S.; Vikentyev, I.V.; Groznova, E.O.; Lyubimtseva, N.G.; Murzin, V.V. The Berezovsk giant intrusion-related gold-quartz deposit, Urals, Russia: Evidence for multiple magmatic and metamorphic fluid reservoirs. Ore Geol. Rev. 2017, 91, 837–863. [Google Scholar] [CrossRef]
- Vikent’eva, O.V.; Prokofiev, V.Y.; Gamyanin, G.N.; Goryachev, N.A.; Bortnikov, N.S. Intrusion-related gold-bismuth deposits of North-East Russia: PTX parameters and sources of hydrothermal fluids. Ore Geol. Rev. 2018, 102, 240–259. [Google Scholar] [CrossRef]
- Goryachev, N.A.; Berdnikov, N.V. Types of ore-bearing granites of south-eastern part of Mesozoids of North-East Russia and their fluid specialisation. J. Geol. Pac. Ocean 2006, 25, 40–52. [Google Scholar]
- Böhlke, J.K.; Irwin, J.J. Laser microprobe analyses of Cl, Br, I, and K in fluid inclusions: Implications for sources of salinity in some ancient hydrothermal fluids. Geochim. Cosmochim. Acta 1992, 56, 203–225. [Google Scholar] [CrossRef]
- Irwin, J.J.; Roedder, E. Diverse origins of fluid in magmatic inclusions at Bingham (Utah, USA), Butte (Montana, USA), St Austell (Cornwall, UK) and Ascension Island (mid-Atlantic, UK) indicated by laser microprobe analysis of Cl, K, Br, I, Ba+Te, U, Ar, Kr and Xe. Geochim. Cosmochim. Acta 1995, 59, 295–312. [Google Scholar] [CrossRef]
- Campbell, A.R.; Lundberg, S.A.W.; Dunbar, N.W. Solid inclusions of halite in quartz: Evidence for the halite trend. Chem. Geol. 2001, 173, 179–191. [Google Scholar] [CrossRef][Green Version]
- Prokof’ev, V.Y.; Baksheev, I.A.; Korytov, F.Y.; Touret, J. Immiscible fluids (CO2-brines) in optical fluorite, Nordvik-Taimyr, Russia. Comptes Rendus Geosci. 2006, 338, 617–623. [Google Scholar] [CrossRef]
- Bény, C.; Guilhaumou, N.; Touray, J.C. Native-sulphur-bearing fluid inclusions in the CO2-H2S-H2O-S system-microthermometry and Raman microprobe (mole) analysis—Thermochemical interpretations. Chem. Geol. 1982, 37, 113–127. [Google Scholar] [CrossRef]
- Grishina, S.; Dubessy, J.; Kontorovich, A.; Pironon, J. Inclusions in salt beds resulting from thermal metamorphism by dolerite sills (Eastern Siberia, Russia). Eur. J. Mineral. 1992, 4, 1187–1202. [Google Scholar] [CrossRef]
- Giuliani, G.; Dubessy, J.; Banks, D.; Vinh, H.Q.; Lhomme, T.; Pironon, J.; Garnier, V.; Trinh, P.T.; Long, P.V.; Ohnenstetter, D.; et al. CO2-H2S-COS-S8-Al(OH)-bearing fluid inclusions in ruby from marble-hosted deposits in Luc Yen area, North Vietnam. Chem. Geol. 2003, 194, 167–185. [Google Scholar] [CrossRef]
- Bray, C.J.; Spooner, E.T.C. Fluid inclusion volatile analysis by gas chromatography with photoionization/micro-thermal conductivity detectors: Applications to magmatic MoS2 and other H2O-CO2 and H2O-CH4 fluids. Geochim. Cosmochim. Acta 1992, 56, 261–272. [Google Scholar] [CrossRef]
- Binder, B.; Keppler, H. The oxidation state of sulfur in magmatic fluids. Earth Planet Sci. Lett. 2011, 301, 190–198. [Google Scholar] [CrossRef]
- Borisenko, A.S.; Borovikov, A.A.; Borisenko, I.D.; Bortnikov, N.S.; Prokofiev, V.Y.; Vikent’eva, O.V.; Gamyanin, G.N.; Gaskov, I.V. Sulfur forms in the hydrothermal fluids and their role in ore formation. In European Current Research on Fluid Inclusions (ECROFI-XXIII); Extended Abstracts’ Volume; University of Leeds: Leeds, UK, 2015; pp. 44–46. [Google Scholar]
- Pokrovski, G.; Dubessy, J. Stability and abundance of the trisulfur radical ion S3– in hydrothermal fluids. Earth Planet Sci. Lett. 2015, 411, 298–309. [Google Scholar] [CrossRef]
- Borovikov, A.A.; Guzhina, L.V.; Goverdovski, V.A.; Gimon, V.O. Inversion of redox potential of oxidized ore-forming fluids as a factor of ore formation of Kalgutinsky Deposit (Gorny Altai). In The Main Problems in the Doctrine of Endogenous Ore Deposits: New Horizons; IGEM RAS: Moscow, Russia, 2017; Extended Abstracts’ Volume. [Google Scholar]
- Afifi, A.M.; Kelly, W.C.; Essene, E.J. Phase relations among tellurides, sulphides and oxides: I. Thermochemical data and calculated equilibria. Econ. Geol. 1988, 83, 377–404. [Google Scholar] [CrossRef]
- Ciobanu, C.L.; Cook, N.J. Tellurides, selenides (and Bisulphosalts) in gold deposits. In Proceedings of the 11th IAGOD Symp-Geocongress, Windhoek, Namibia, 22–26 July 2002; Geol Surv: Windhoek, Namibia, 2004. [Google Scholar]
- Ohmoto, H. Stable isotope geochemistry of ore deposits. Rev. Mineral. 1986, 16, 491–560. [Google Scholar]
- Koroteev, V.A.; De Boorder, H.; Necheukhin, V.M.; Sazonov, V.N. Geodynamic setting of the mineral deposits of the Urals. Tectonophysics 1997, 276, 291–300. [Google Scholar] [CrossRef]
- Puchkov, V.N. General features relating to the occurrence of mineral deposits in the Urals: What, where, when and why. Ore Geol. Rev. 2017, 85, 4–29. [Google Scholar] [CrossRef]
- Vikentyev, I.V.; Belogub, E.V.; Novoselov, K.A.; Moloshag, V.P. Metamorphism of volcanogenic massive sulphide deposits in the Urals. Ore geology. Ore Geol. Rev. 2017, 85, 30–63. [Google Scholar] [CrossRef]


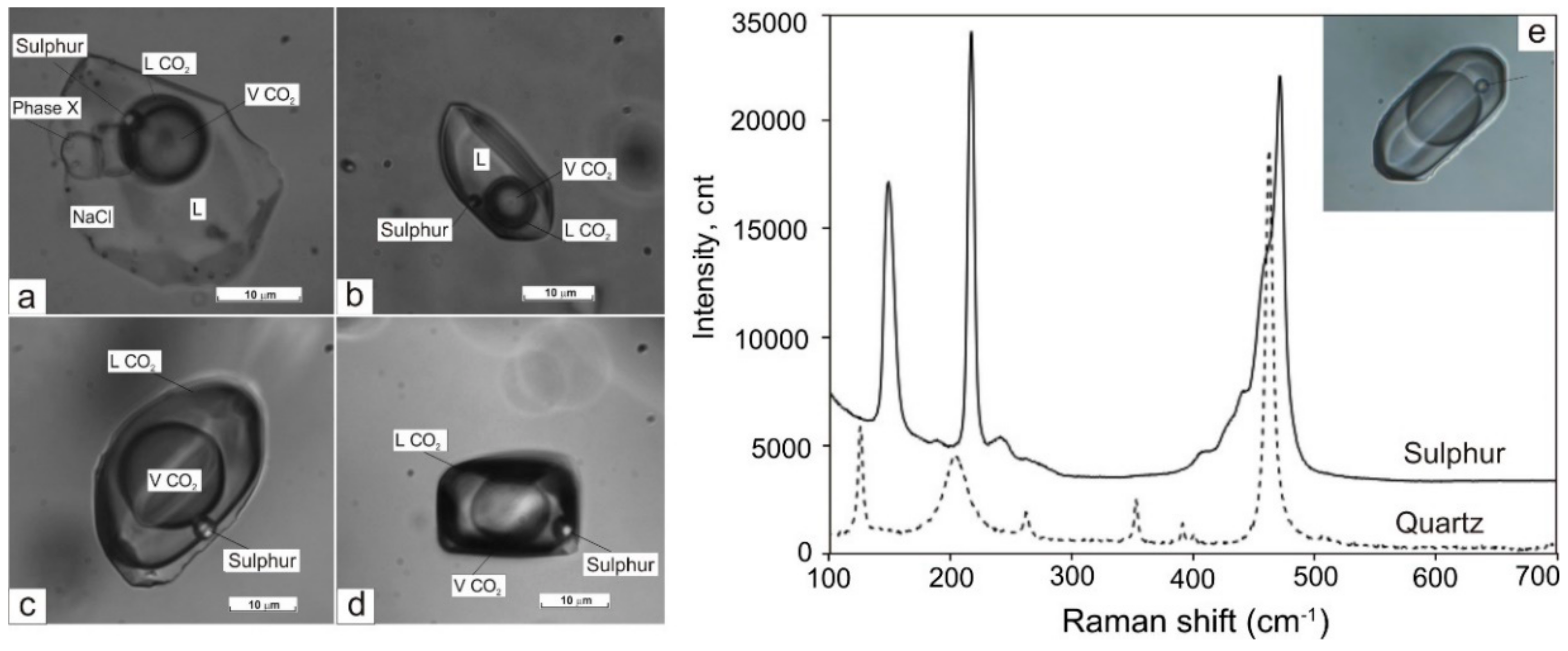
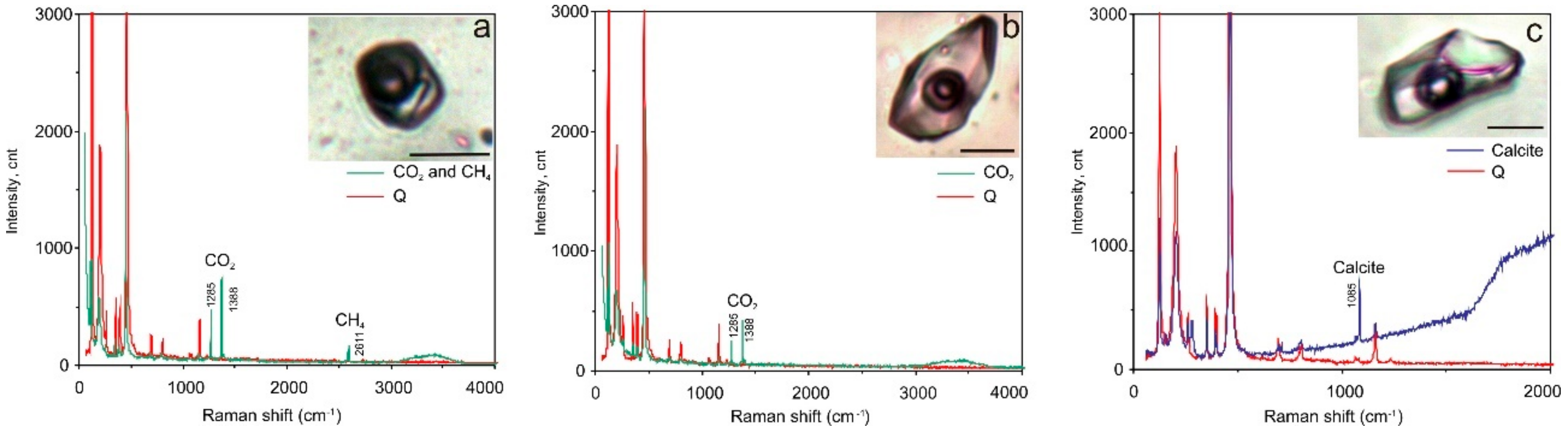
| Sample | FIs Type1 | n2 | Th, °C | Te, °C | Tm ice (NaCl) [S0], °C3 | Tm(CO2), °C | Th(CO2)(N2), °C | H.m.4 | Tm (clatrate), °C | Csalt, wt% NaCl Equiv. | CCO2, mol/kg Solution | CCH4, mol/kg Solution | d, g/cm3 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Q1a | |||||||||||||
| SV | I P | 17 | 286–315 | −53 | −20.5 (85)3 | −57.4…−58.6 | 33.1 | V | 0.0 | 15.6 | 1.4 | 0.1 | 1.11 |
| II P | 15 | - | - | - | −60.2…−60.8 | 16.6…32.8 | L | - | - | - | - | 0.47–0.81 | |
| III P | 5 | 354 | −54 | −7.0 (75)3 | - | - | - | - | 10.5 | - | - | 0.75 | |
| IV P | 18 | 269 | −52 | (181)3 | - | - | - | - | 31.0 | - | - | 1.05 | |
| I PS | 2 | 246 | −50 | −11.8 | −58.4 | 33.6 | V | −0.1 | 15.8 | 2.2 | 0.2 | 1.03 | |
| II PS | 2 | - | - | - | −60.1 | 14.5…24.0 | L | - | - | - | - | 0.73–0.83 | |
| SV62/18 | I PS | 32 | 296–300 | −51…−56 | −24.1…−28.6 (148)3 | −59.4…−62.2 | 33.5…34.1 | L | −2.3–3.2 | 23.0–28.8 | 2.1–2.4 | 0.1–0.4 | 0.91–0.95 |
| II PS | 54 | - | - | - | −59.7…−63.8 | 18.8…31.0 | L | - | - | - | 0.51–0.79 | ||
| S64/18 | II P | 9 | - | - | - | −61.7 | 9.4 | L | - | - | - | - | 0.87 |
| Q1b | |||||||||||||
| 216/148.5 | I P | 3 | 265–310 | −35 | −8.3 | −56.8 | −8.8 | L | 4.1 | 12.0 | 5.4 | - | 1.09 |
| II P | 32 | - | - | - | −56.6…−56.8 | −29.8…0.6 | L | - | - | - | - | 0.92–1.08 | |
| 216/409.6 | I P | 37 | 275–320 | −31…−33 | −7.6…−11.1 | −57.1…−57.2 | −6.5…15.6 | L | 1.3–7.9 | 11.2–15.0 | 4.2–7.6 | - | 1.06–1.10 |
| II P | 51 | - | - | - | −57.4…−57.5 | −13.6…7.0 | L | - | - | - | - | 0.88–0.97 | |
| 202/289.2 | I P | 8 | 315–365 | −31…−32 | −4.1…−4.7 | −57.4…−57.8 | −12.4 | L | 4.1 | 12.5 | 6.4–7.2 | - | 1.06 |
| II P | 47 | - | - | - | −58.1…−58.7 | −21.1…−1.7 | L | - | - | - | 0.94–1.04 | ||
| 202/318.8 | I P | 8 | 260–315 | −34 | −7.7 | −57.5 | 21.0…26.3 | L | 3.2–3.9 | 10.7–11.7 | 2.9–3.6 | 0.3 | 1.05–1.06 |
| II P | 25 | - | - | - | −57.2…−57.8 | −25.6…23.8 | L | - | - | - | 0.73–1.06 | ||
| III S | 13 | 275–300 | −31 | −5.8 | - | - | - | 8.9 | - | - | 0.82–0.85 | ||
| 202/601.2 | I P | 7 | 240–270 | −32 | −8.3 | −60.6 | −31.0 | L | 12.2 | 12.1 | 2.7 | 0.7 | 1.03 |
| II P | 32 | - | - | - | −59.3…-−61.0 | −37.6…−12.5 | L | - | - | - | - | 1.00–1.11 | |
| III S | 6 | 310 | −31 | −9.1 | - | - | 13.0 | - | - | 0.85 | |||
| II S | 4 | - | - | - | - | −131 | V | - | - | - | 0.16 | ||
| S12/96 | I P | 9 | 295–340 | −37 | −12.8 | −57.5 | 2.1…18.0 | L | 1.2 | 14.2 | 4.9–6.2 | 1.07–1.08 | |
| II P | 66 | - | - | - | −57.5…−60.2 | −35.7…13.1 | L | - | - | - | - | 0.84–1.10 | |
| 470/293.5 | I P | 3 | 325–350 | −37 | −11.1 | −58.6 | −9.7 | L | 1.3 | 15.3 | 4.4–4.9 | 0.2–0.3 | 1.09 |
| II P | 40 | - | - | - | −57.7…−59.4 | −22.7…4.4 | L | - | - | - | 0.95–1.05 | ||
| 38-3a | I PS | 10 | 250–280 | −28…−33 | −8.0…−13.0 | −58.4…−60.0 | 17.0…32.0 | L | 7.0–12.0 | 11.4…17 | - | - | |
| II PS | 3 | - | - | - | −58.5…−59.1 | 19.1…24.0 | L | - | - | - | - | 0.73–0.78 | |
| Q2 | |||||||||||||
| 202/593.0 | III P | 15 | 285–315 | −30 | −4.2…−5.9 | - | - | - | - | 6.7… 9.1 | - | - | 0.80–0.81 |
| II P | 45 | - | - | - | −57.9…−58.7 | −26.8…0.9 | L | - | - | - | 0.92–1.06 | ||
| S1p/95 | I P | 9 | 300–345 | −32 | −5.5…−6.7 | −56.9 | −9.1 | L | 4.7 | 11.3 | 5.3–5.4 | - | 1.08 |
| II P | 31 | - | - | - | −56.9…−59.9 | −29.2…10.2 | L | 0.86–1.07 | |||||
| II S | 14 | - | - | - | - | −153 | V | 0.13 | |||||
| S3/95a | I P | 3 | 305–320 | −33 | −8.2 | −59.2 | −21.2 | L | 6.2 | 11.2 | 6.6 | - | 1.09 |
| II P | 16 | −58.6…−59.4 | −28.5…−7.5 | L | - | - | - | - | 0.97–1.07 | ||||
| II S | 2 | - | - | - | - | −149 | V | - | - | - | - | 0.17 | |
| 618/162 | I P | 6 | 255–275 | −32 | −7.4 | −57.7 | −6.8…5.3 | L | 6.6 | 6.8 | 4.0–5.0 | 0.4–0.5 | 1.08 |
| II P | 53 | - | - | - | −57.4…−57.7 | −15.8…1.8 | L | - | - | - | - | 0.92–1.01 | |
| S2/10 | I P | 5 | 302 | −33 | −5.0 | −57.8 | 17.5…25.1 | L | 4.6 | 9.6 | 4.6 | 0.4 | 1.02 |
| II P | 13 | - | - | - | −57.8 | −1.3…8.0 | L | - | - | - | - | 0.88–0.94 | |
| III S | 3 | 159 | −32 | −4.9 | - | - | 7.7 | 0.97 | |||||
| S14/10 | I P | 7 | 278–289 | −35…−36 | −4.1…−6.4 | −57.2…−59.6 | 3.5…20.4 | L | 6.7–7.8 | 4.3–6.8 | 5.2–5.3 | 0.5 | 1.02–1.04 |
| II P | 13 | - | - | - | −59.2…−59.6 | −7.8…12.6 | L | - | - | - | - | 0.97–0.84 | |
| SV66 | I PS | 13 | 230–260 | −27…−32 | −4.0 | −57.0…−57.8 | 11.0…23.0 | L | 5.8–8.3 | 3.5–7.5 | - | - | |
| II PS | 2 | −57.5…−57.8 | 19.5…22.0 | L | 0.74–0.78 | ||||||||
| SV51 | I PS | 9 | 195–220 | −37…−39 | −6.9…−8.0 | −58.1…−58.8 | 1.6…27.9 | L | 2.8–4.6 | 10–12.0 | - | - | |
| II PS | 2 | - | - | - | −58.5 | 22.0…25.2 | L | - | - | - | - | 0.71–0.75 | |
| Q3 | |||||||||||||
| 202/592.7 | I P | 17 | 295–320 | −34…−36 | −7.8…−10.1 | −56.6…−57.6 | −30.8…−24.1 | L | 9.4–12.6 | 7.2 | 5.9–6.0 | 0.6–0.9 | 1.11–1.12 |
| II P | 69 | - | - | - | −56.8…−57.6 | −24.3…22.1 | L | - | - | - | - | 0.75–1.05 | |
| III S | 23 | 190–230 | −30 | −4.9 | - | - | - | - | 7.7 | - | - | 0.90–0.94 | |
| S5/ 95b | I P | 17 | 370–405 | −32 | −5.2…−5.7 | −74.4 | −146.9…−147.5 | V | 9.3–10.5 | 8.1–8.8 | 0,8 | - | 0.83 |
| II P | 76 | - | - | - | −57.8…−60.8 | −38.8…6.3 | L | - | - | - | - | 0.89–1.11 | |
| III S | 29 | 265–305 | −32…−33 | −4.7…−6.2 | - | - | - | 7.4–9.5 | - | - | 0.81–0.84 | ||
| II S | 40 | - | - | - | −82.9 | −149…−152 | V | - | - | - | - | 0.14–0.17 | |
| 470/ 170 | I P | 7 | 330–350 | −32 | −8.1…−9.2 | −56.7 | 16.3…26.5 | L | 7.1 | 5.6 | 5.0–6.3 | 0.6–0.7 | 1.01–1.04 |
| II P | 26 | - | - | - | −56.8…−57.9 | 2.5…25.0 | L | - | - | - | - | 0.71–0.91 | |
| S1/10 | I P | 5 | 358 | −35 | −5.9 | −57.3 | −7.0 | L | 4.8 | 10,9 | 6.3 | 0.7 | 1.07 |
| II P | 22 | - | - | - | −57.2…−57.4 | −1.0…13.4 | L | - | - | - | - | 0.84–0.93 | |
| I PS | 3 | 280 | −37 | −5.4 | −57.2 | 2.9 | L | 8.3 | 4.2 | 4.6 | 0.4 | 1.06 | |
| II PS | 10 | - | - | - | −57.4 | −3.1…9.1 | L | - | - | - | - | 0.87–0.95 | |
| III S | 2 | 262 | −30 | −0.8 | - | - | 1.3 | - | - | 0.78 | |||
| S9/10 | I PS | 6 | 363 | −33 | −5.0 | −58.2 | 2.7 | L | 7.2 | 6.1 | 7.1 | 0.7 | 1.04 |
| II PS | 11 | - | - | - | −58.0 | −13.6…−0.2 | L | - | - | - | - | 0.93–1.00 | |
| III S | 3 | 347 | −35 | −0.3 | - | - | - | 0.5 | - | - | 0.58 | ||
| S35/10 | III P | 6 | 308 | −33 | −0.5 | - | - | - | - | 0.8 | - | - | 0.68 |
| S41/18 | I P | 6 | 312–331 | −33…−34 | −1.9…−2.7 | −58.6…−59.9 | 2.1…11.4 | L | 8.1–8.9 | 3.3–3.8 | 6.7–8.3 | 0.7–0.9 | 1.01–1.02 |
| II P | 43 | - | - | - | −58.3…−60.5 | −9.8…1.9 | L | - | - | - | - | 0.92–0.98 | |
| III S | 8 | 203 | −22 | −0.3 | - | - | - | - | 0.5 | - | - | 0.87 | |
| Q4 | |||||||||||||
| 63-1 | III P | 2 | 220–250 | −21…−22 | −1.0…−4.6 | - | - | - | - | 1.7–7.3 | - | - | 0.85–0.87 |
| S10/95 | III P | 28 | 130–230 | −23…−24 | −2.9 | - | - | - | - | 4.8 | - | - | 0.85–0.95 |
| Quartz | Q3 | Q3 | Q3 | Q2 | Q1a | Quartz | Q3 | Q3 | Q3 | Q2 | Q1a |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | S-1/10 | S-9/10 | S-35/10 | S-14/10 | SV | Sample | S-1/10 | S-9/10 | S-35/10 | S-14/10 | SV |
| CO2,×103 ppm | 49.05 | 293.29 | 1231.85 | 406.23 | 471.05 | Ge, ppm | 0.52 | 0.31 | 0.27 | 0.11 | 0.91 |
| CH4 | 0.683 | 0.652 | 2.566 | 1.955 | 0.026 | Cd | 1.96 | 0.53 | 0.30 | 0.18 | – |
| Cl− | 14.42 | <0.6 | <1 | 6.13 | 59.96 | Pb | 35.54 | – | 13.10 | – | – |
| SO42− | <0.8 | <0.8 | <1.5 | <0.5 | <1 | Au | 9.89 | – | – | – | – |
| HCO3− | 65.25 | 11.74 | 35.30 | 23.45 | 93.48 | Ag | 0.05 | – | 0.14 | – | – |
| Na | 25.31 | 2.99 | 8.31 | 11.43 | 31.27 | Bi | 0.07 | 0.09 | – | – | – |
| K | 2.25 | 0.42 | 0.78 | 0.72 | 3.07 | Mo | 0.19 | 0.02 | – | 0.05 | 0.13 |
| Ca | 3.92 | 0.70 | 2.38 | 0.65 | 31.83 | W | – | – | – | – | 1.67 |
| Mg | 1.53 | 0.20 | 0.94 | 0.14 | 2.59 | Sn | 0.30 | – | – | – | – |
| Br, ppm | – | – | – | – | 2824.5 | Hg | 0.02 | – | – | – | – |
| B | 2015.8 | 408.43 | 661.34 | 592.13 | 596.48 | Tl | 0.12 | – | 1.47 | 0.02 | 0.17 |
| Li | 1.04 | 0.30 | 0.48 | 0.11 | 10.68 | Co | 0.10 | 0.06 | 0.07 | 0.04 | 20.06 |
| Rb | 3.28 | 1.10 | 2.78 | 2.06 | 7.00 | Ni | 5.38 | 0.64 | 12.75 | 1.80 | 174.94 |
| Cs | 0.28 | 0.23 | 1.17 | 1.13 | 2.23 | Cr | 0.82 | 0.38 | – | – | – |
| Sr | 30.29 | 7.20 | 6.79 | 27.55 | 1166.0 | V | 0.21 | – | – | – | 0.55 |
| Ba | 12.19 | 0.92 | – | 9.44 | 3951.1 | U | – | – | – | – | – |
| Cu | 263.86 | – | 236.34 | 8.92 | – | Mn | 34.29 | 4.74 | 13.54 | 32.37 | 511.06 |
| Zn | 1630 | 54.05 | 1699.9 | 65.48 | 164.87 | Fe | 126.15 | 2.69 | – | – | – |
| As | 72.51 | 42.13 | 68.67 | 19.76 | 21.26 | I | – | – | – | – | 116.22 |
| Sb | 27.37 | 52.99 | 51.53 | 2.96 | 19.22 | Si | 40,127 | 35,623 | 31,309 | – | – |
| Sample 1 | Q Generation | FIs Type 2 | CO2 | N2 | H2S | CH4 |
|---|---|---|---|---|---|---|
| SV | Q1a | II | 96.9 | 1.23 | 1.82 | − |
| SV | Q1a | IV | 98.2 | −3 | 1.81 | − |
| SV | Q1a | IV | 94.2 | 1.64 | 4.12 | − |
| SV | Q1a | IV | 98.8 | 0.65 | 0.53 | − |
| S1/10 | Q3 | II | 98.4 | 1.62 | − | − |
| S1/10 | Q3 | II | 98 | 2.01 | − | − |
| S1/10 | Q3 | II | 89.6 | 10.41 | − | − |
| S1/10 | Q3 | II | 95 | 4.98 | − | − |
| S1/10 | Q3 | I | 73.3 | 26.70 | − | − |
| 38-3a | Q1b | I | 50 | − | − | 50 |
| 38-3a | Q1b | I | 80 | − | − | 20 |
| 38-3a | Q1b | I | 70 | − | − | 30 |
| 38-3a | Q1b | I | 96 | − | 4 | − |
| 38-3a | Q1b | I | 98 | − | 2 | − |
| 38-3a | Q1b | I | 100 | − | − | − |
| SV-51 | Q2 | I | 100 | − | − | − |
| SV-66 | Q2 | I | 100 | − | − | − |
| 63-1 | Q4 | III | − | − | − | − |
| Quartz | Q1a | Q3 (Main Ore Stage) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | SV (1) | SV (2) | SV (3) | SV (4) | SV (5) | SV (6) | S1/10 (1) | S1/10 (2) | S1/10 (3) | S1/10 (4) | S1/10 (5) |
| FI type 1 | IV | IV | IV | IV | IV | II | I | I | I | II | II |
 |  |  |  |  |  |  |  |  |  |  | |
| B, ppm | 140 | 100 | 50 | 14 | 39 | 0 | 1350 | 460 | 350 | 2580 | 560 |
| Na | 58,900 | 58,900 | 58,900 | 58,900 | 58,900 | 13,000 | 47,200 | 19,700 | 19,700 | 19,700 | 19,700 |
| Mg | 1170 | 3770 | 3160 | 1750 | 1680 | 20 | 4360 | 0 | 0 | 58,500 | 45 |
| Al | 0 | 2390 | 520 | 560 | 36 | 3830 | 90 | 0 | 3680 | 1170 | 1390 |
| S | 260 | 2370 | 960 | 530 | 890 | 0 | 0 | 0 | 0 | 0 | 1290 |
| K | 1870 | 1250 | 4770 | 1320 | 740 | 970 | 5550 | 5480 | 930 | 5820 | 8320 |
| Ca | 79,800 | 45,400 | 124,000 | 32,700 | 23,500 | 0 | 0 | 0 | 0 | 0 | 0 |
| Mn | 160 | 26 | 140 | 54 | 30 | 8 | 0 | 0 | 0 | 0 | 110 |
| Fe | 0 | 0 | 8 | 380 | 9 | 0 | 0 | 0 | 0 | 0 | 0 |
| Ni | 0 | 8 | 4 | 25 | 19 | 0 | 1200 | 1900 | 1480 | 0 | 2000 |
| Co | 0 | 0 | 0 | 0.1 | 0.2 | 0 | 0 | 0 | 88 | 0 | 18 |
| Cu | 2 | 14 | 0.1 | 0.3 | 0.5 | 12 | 510 | 5890 | 0 | 150 | 0 |
| Zn | 210 | 0 | 39 | 1130 | 18 | 44 | 400 | 27 | 310 | 830 | 1220 |
| As | 5 | 0 | 7 | 235 | 0 | 5150 | 0 | 0 | 3970 | 0 | 270 |
| Rb | 15 | 3 | 2 | 290 | 1 | 0 | 0 | 140 | 0 | 0 | 40 |
| Sr | 2200 | 320 | 220 | 49,000 | 100 | 0 | 15 | 0 | 0 | 510 | 100 |
| Mo | 0 | 7 | 0.2 | 0 | 1 | 0 | 0 | 250 | 0 | 0 | 0 |
| Ag | 1 | 0 | 0 | 2 | 0.4 | 0 | 30 | 0 | 130 | 60 | 0 |
| Cd | 8 | 2 | 1 | 25 | 0 | 480 | 0 | 0 | 0 | 0 | 0 |
| Sn | 20 | 0 | 2 | 4 | 35 | 560 | 0 | 0 | 0 | 0 | 28 |
| Te2 | 4 | 17 | 1 | 10 | 1 | 1050 | 65 | 560 | 2300 | 80 | 330 |
| Sb | 0.4 | 1.1 | 0 | 1.2 | 0.0 | 2504 | 50 | 240 | 0 | 0 | 140 |
| Cs | 55 | 11 | 0.2 | 4 | 0.5 | 0 | 0 | 0 | 0 | 0 | 25 |
| Ba | 14,000 | 17,000 | 160 | 3900 | 290 | 0 | 0 | 0 | 0 | 3000 | 110 |
| Au | 0 | 0 | 0 | 1.5 | 0 | 0 | 0 | 110 | 0 | 0 | 0 |
| Hg 2 | 7 | 25 | 1 | 0 | 2 | 1800 | 0 | 410 | 890 | 620 | 50 |
| Pb | 180 | 12 | 20 | 12 | 0.2 | 44 | 0 | 0 | 210 | 610 | 340 |
| Bi | 0 | 0 | 0 | 0 | 0 | 0 | 12 | 22 | 0 | 80 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vikent’eva, O.; Prokofiev, V.; Borovikov, A.; Kryazhev, S.; Groznova, E.; Pritchin, M.; Vikentyev, I.; Bortnikov, N. Contrasting Fluids in the Svetlinsk Gold-Telluride Hydrothermal System, South Urals. Minerals 2020, 10, 37. https://doi.org/10.3390/min10010037
Vikent’eva O, Prokofiev V, Borovikov A, Kryazhev S, Groznova E, Pritchin M, Vikentyev I, Bortnikov N. Contrasting Fluids in the Svetlinsk Gold-Telluride Hydrothermal System, South Urals. Minerals. 2020; 10(1):37. https://doi.org/10.3390/min10010037
Chicago/Turabian StyleVikent’eva, Olga, Vsevolod Prokofiev, Andrey Borovikov, Sergey Kryazhev, Elena Groznova, Mikhail Pritchin, Ilya Vikentyev, and Nikolay Bortnikov. 2020. "Contrasting Fluids in the Svetlinsk Gold-Telluride Hydrothermal System, South Urals" Minerals 10, no. 1: 37. https://doi.org/10.3390/min10010037
APA StyleVikent’eva, O., Prokofiev, V., Borovikov, A., Kryazhev, S., Groznova, E., Pritchin, M., Vikentyev, I., & Bortnikov, N. (2020). Contrasting Fluids in the Svetlinsk Gold-Telluride Hydrothermal System, South Urals. Minerals, 10(1), 37. https://doi.org/10.3390/min10010037





