Effect of the Sodium Silicate Modulus and Slag Content on Fresh and Hardened Properties of Alkali-Activated Fly Ash/Slag
Abstract
1. Introduction
2. Materials and Methods
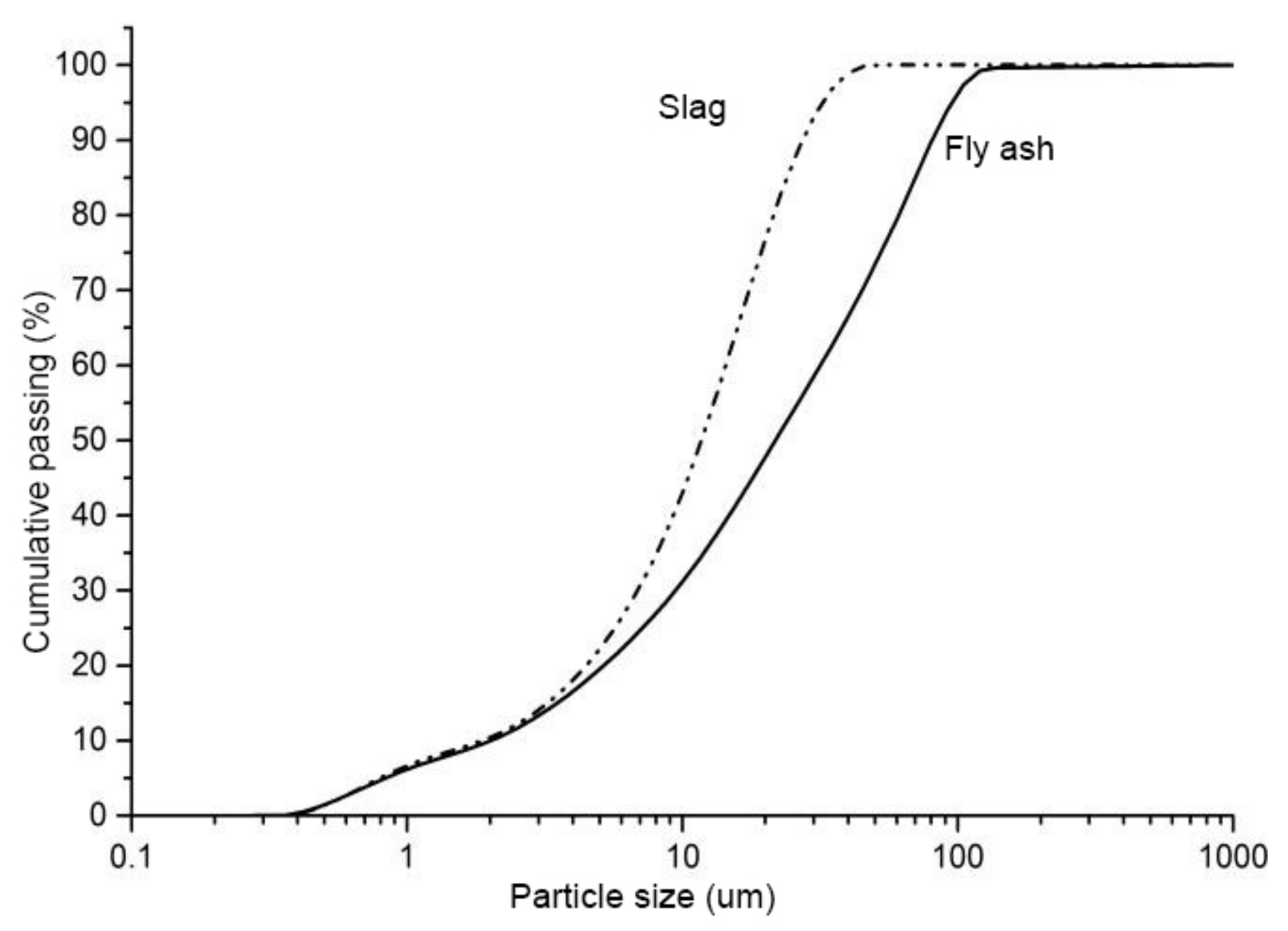
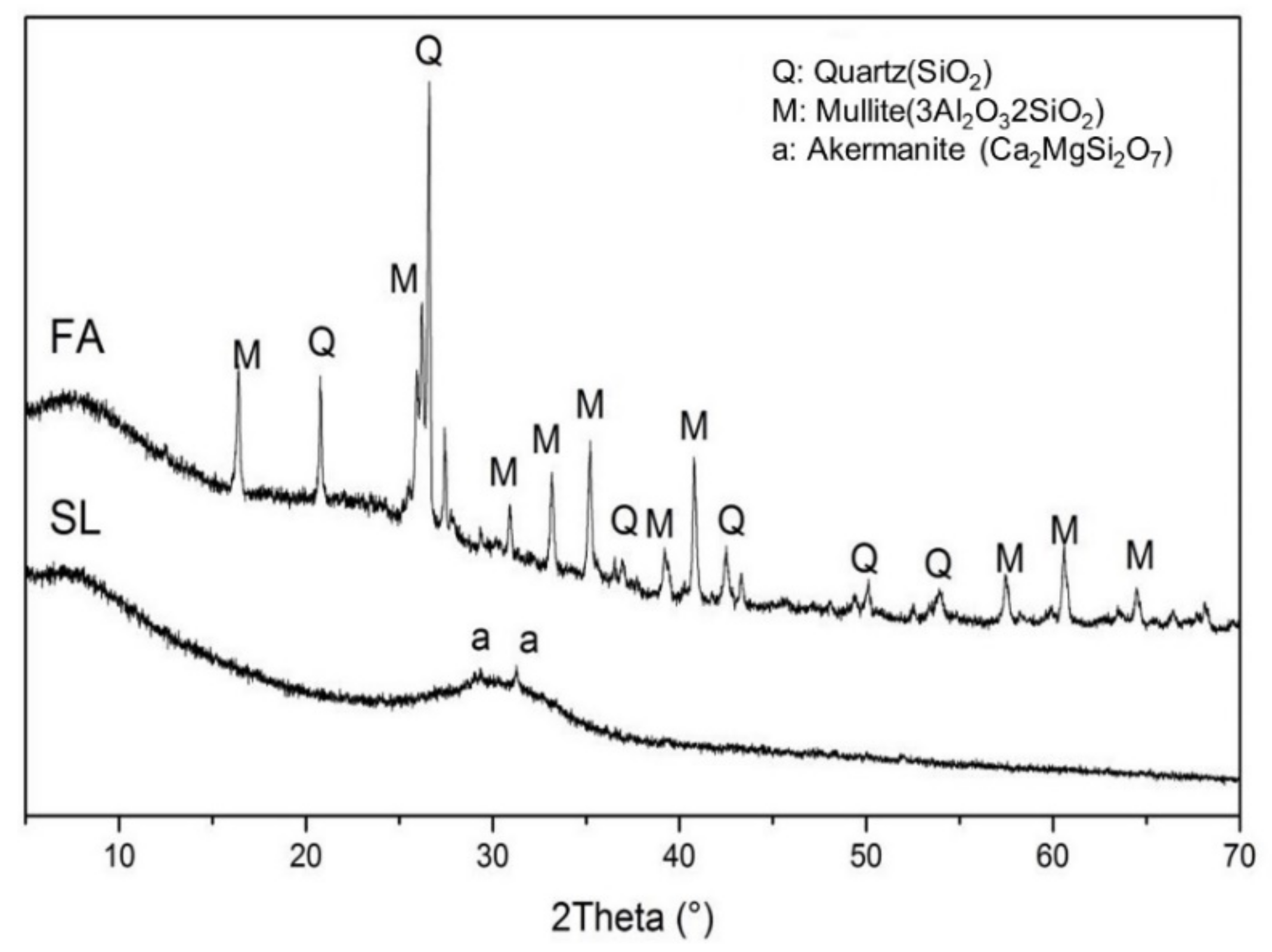
2.1. Materials
2.2. Methods
2.2.1. Setting Time
2.2.2. Workability
2.2.3. Heat of Hydration
2.2.4. Compressive Strength
2.2.5. Porosity, SEM/EDS Analysis
3. Results and Discussion
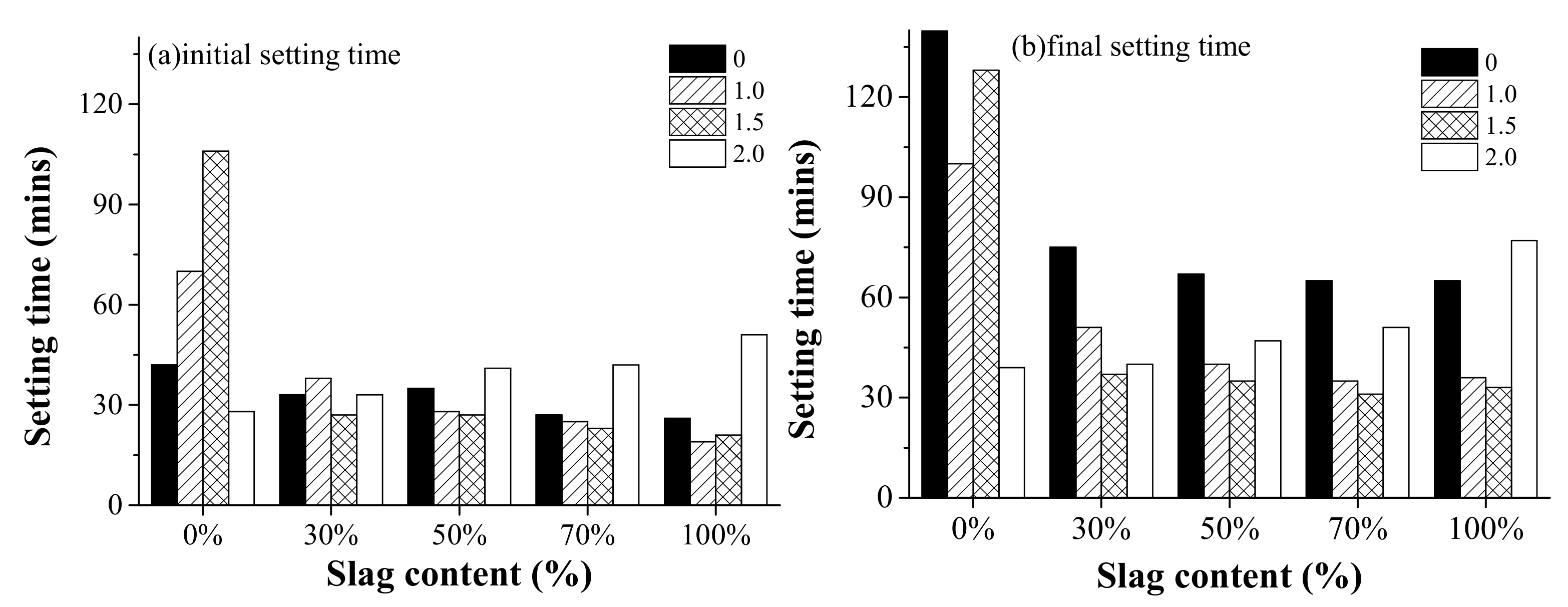
3.1. Setting Time
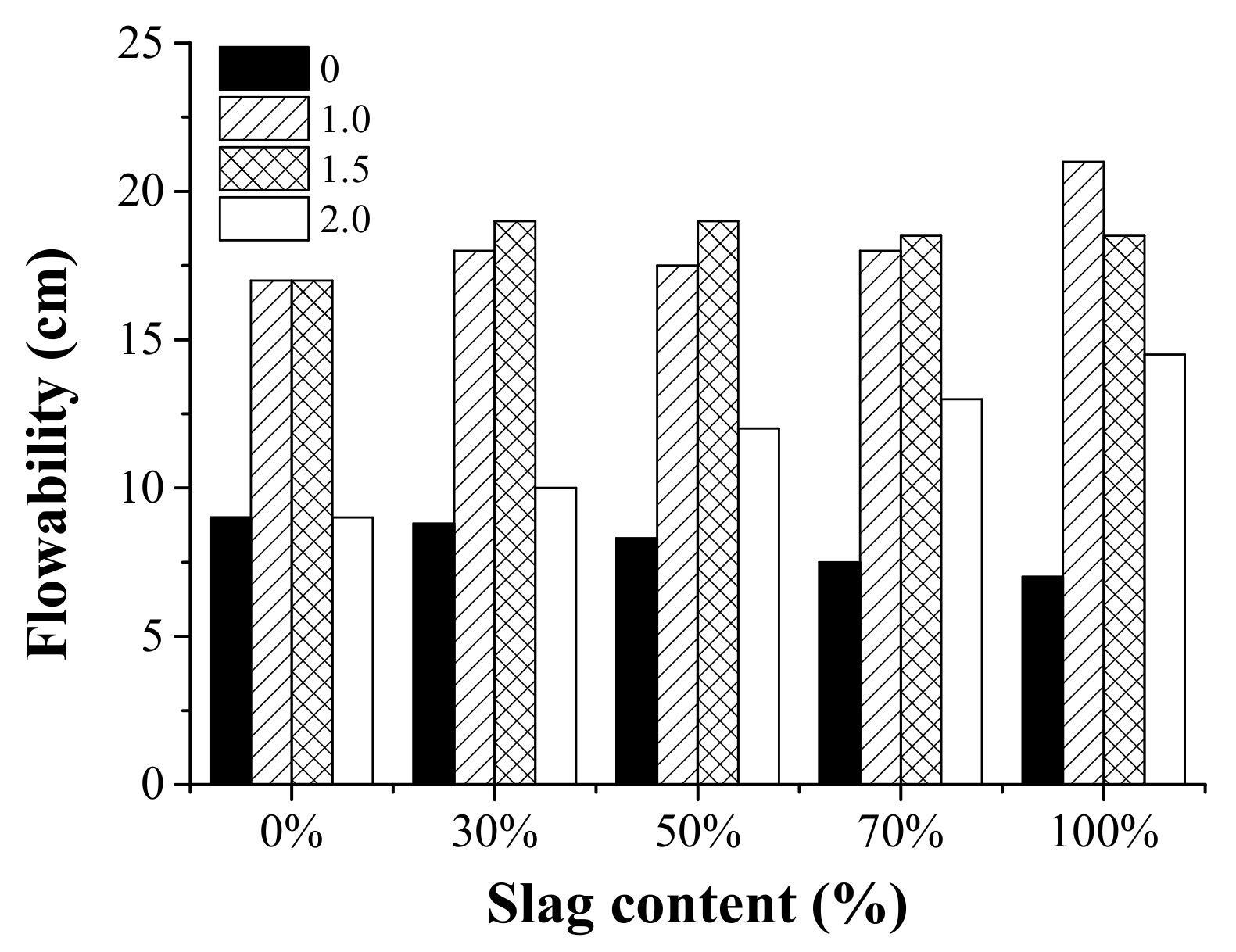
3.2. Flowability
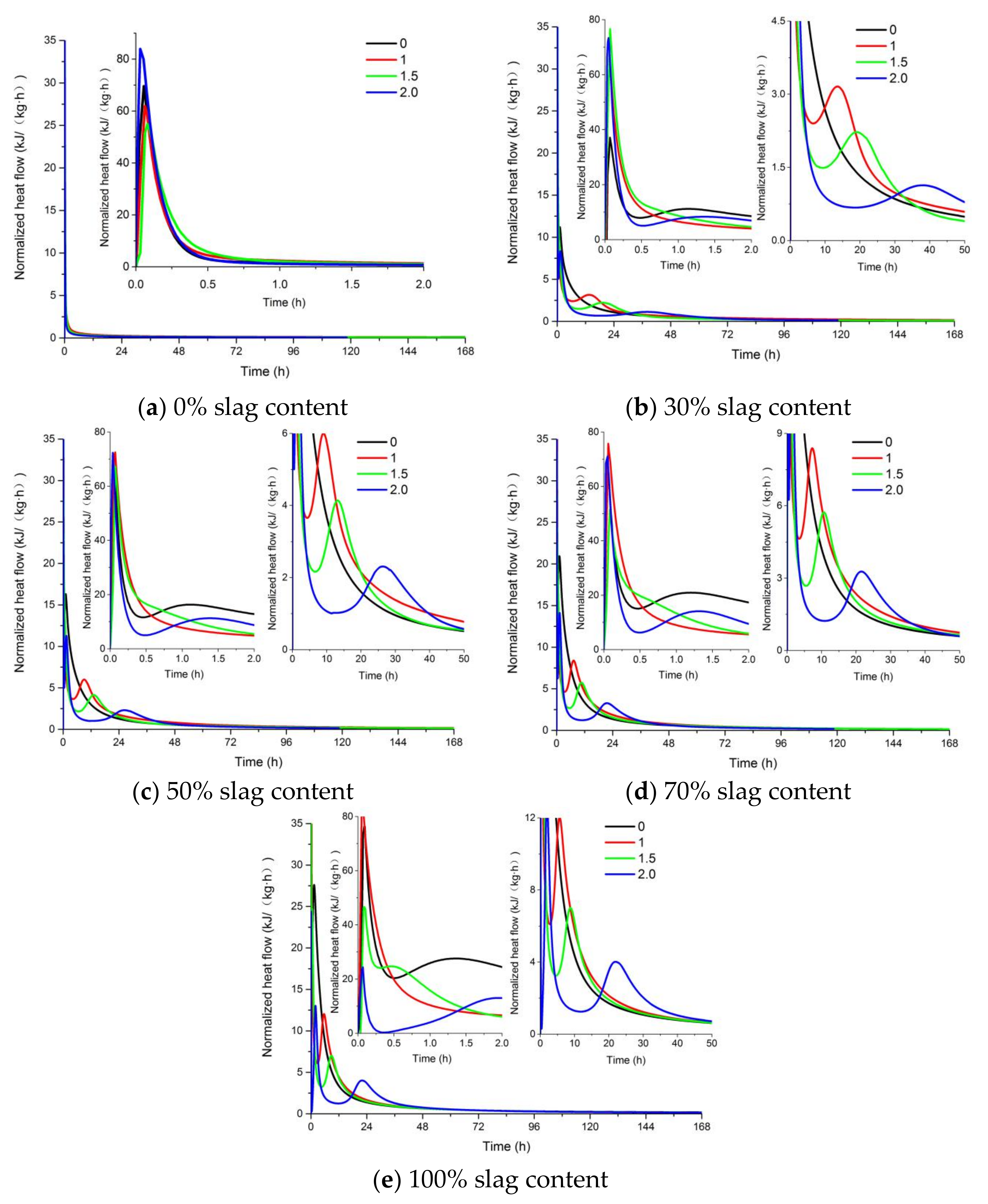
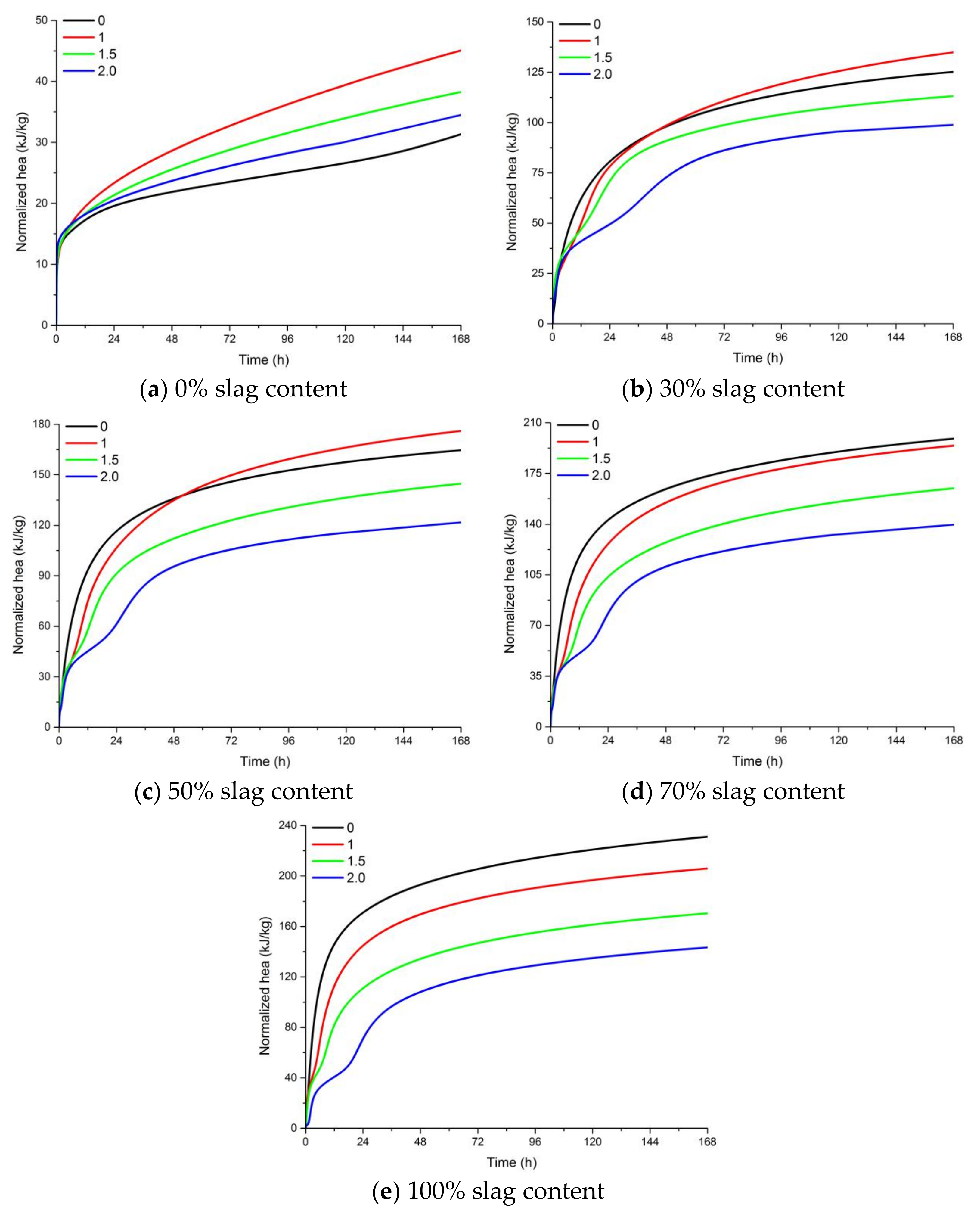
3.3. Heat Evolution of AAFS Pastes
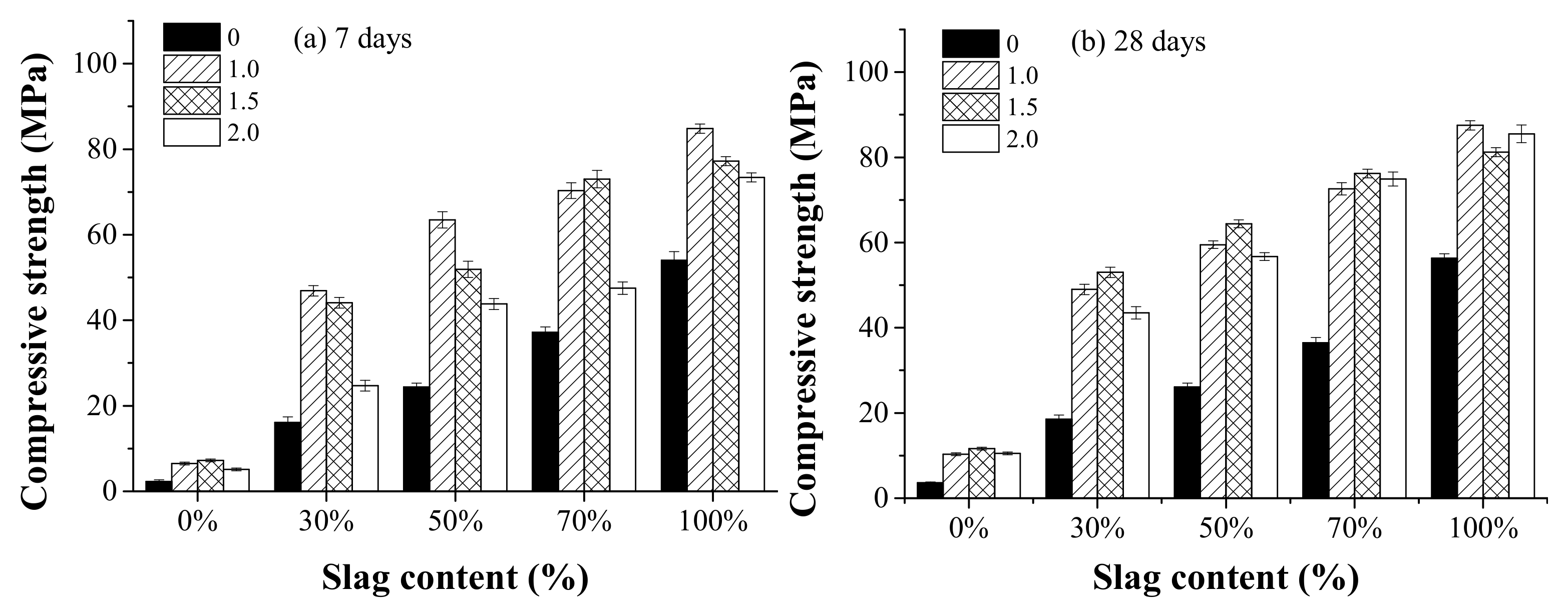
3.4. Compressive Strength
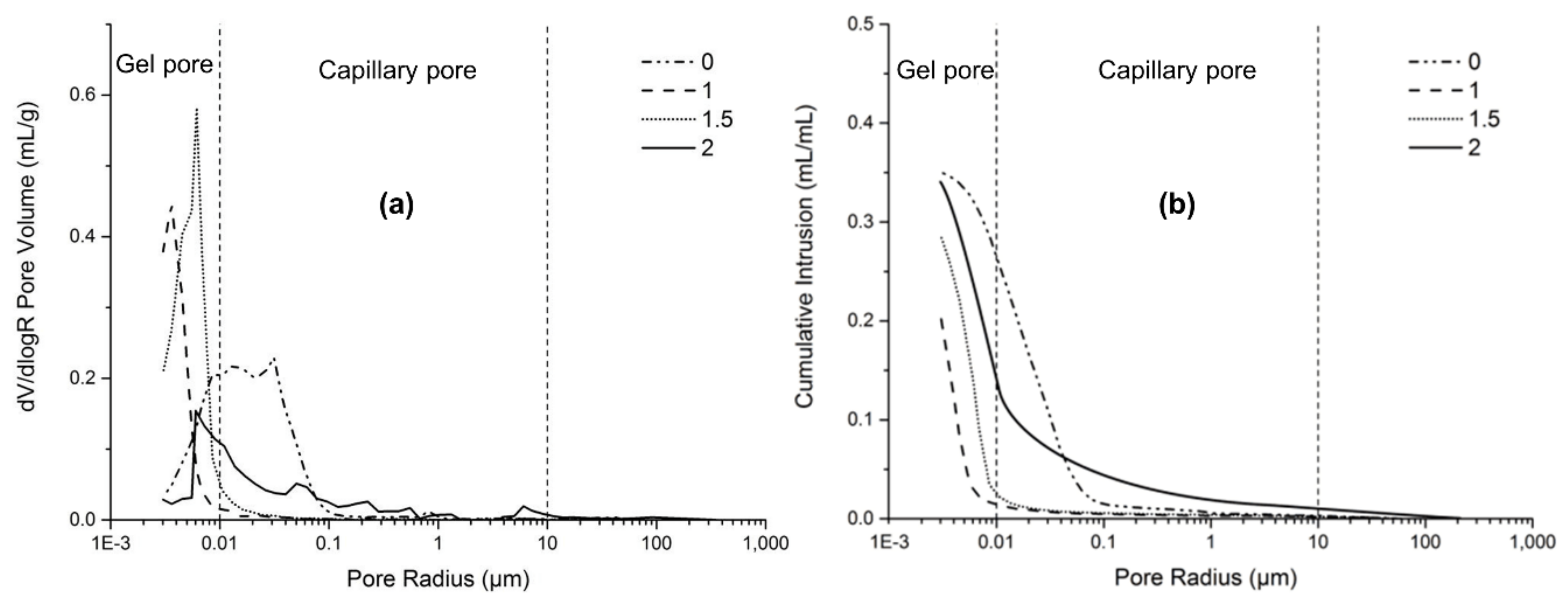
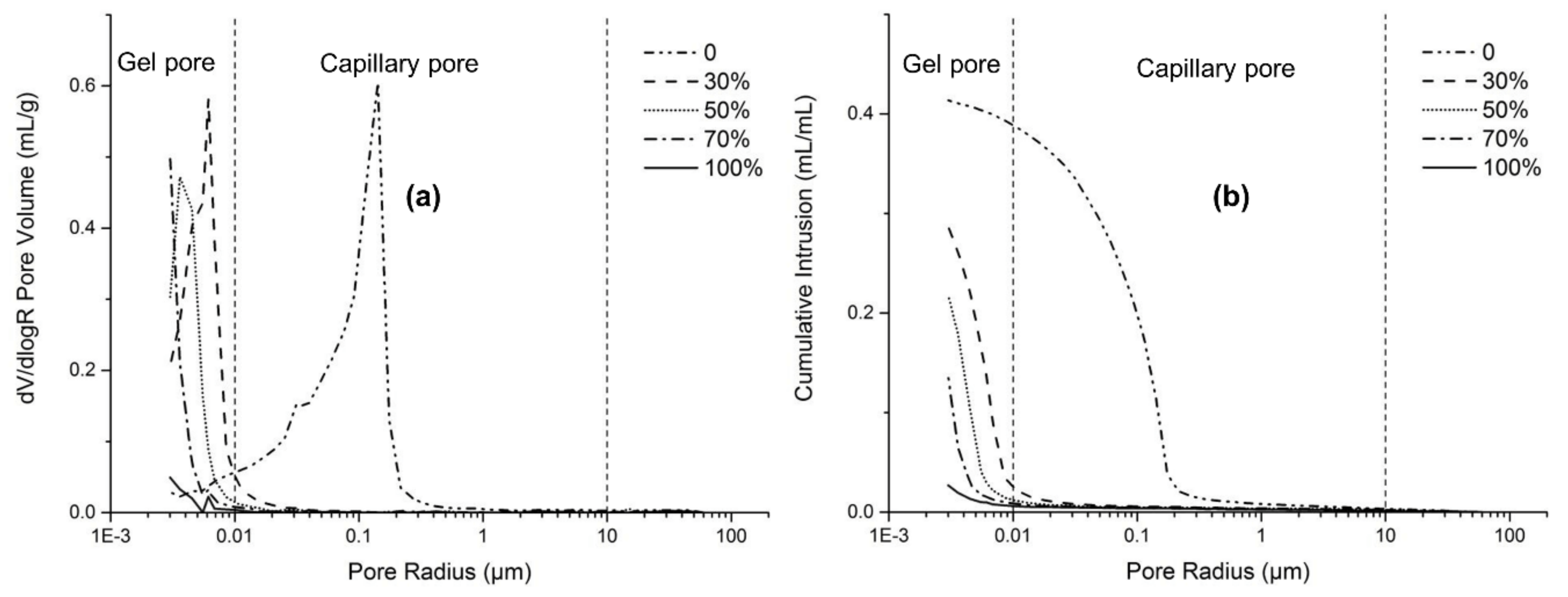
3.5. Porosity and Pore Size Distribution
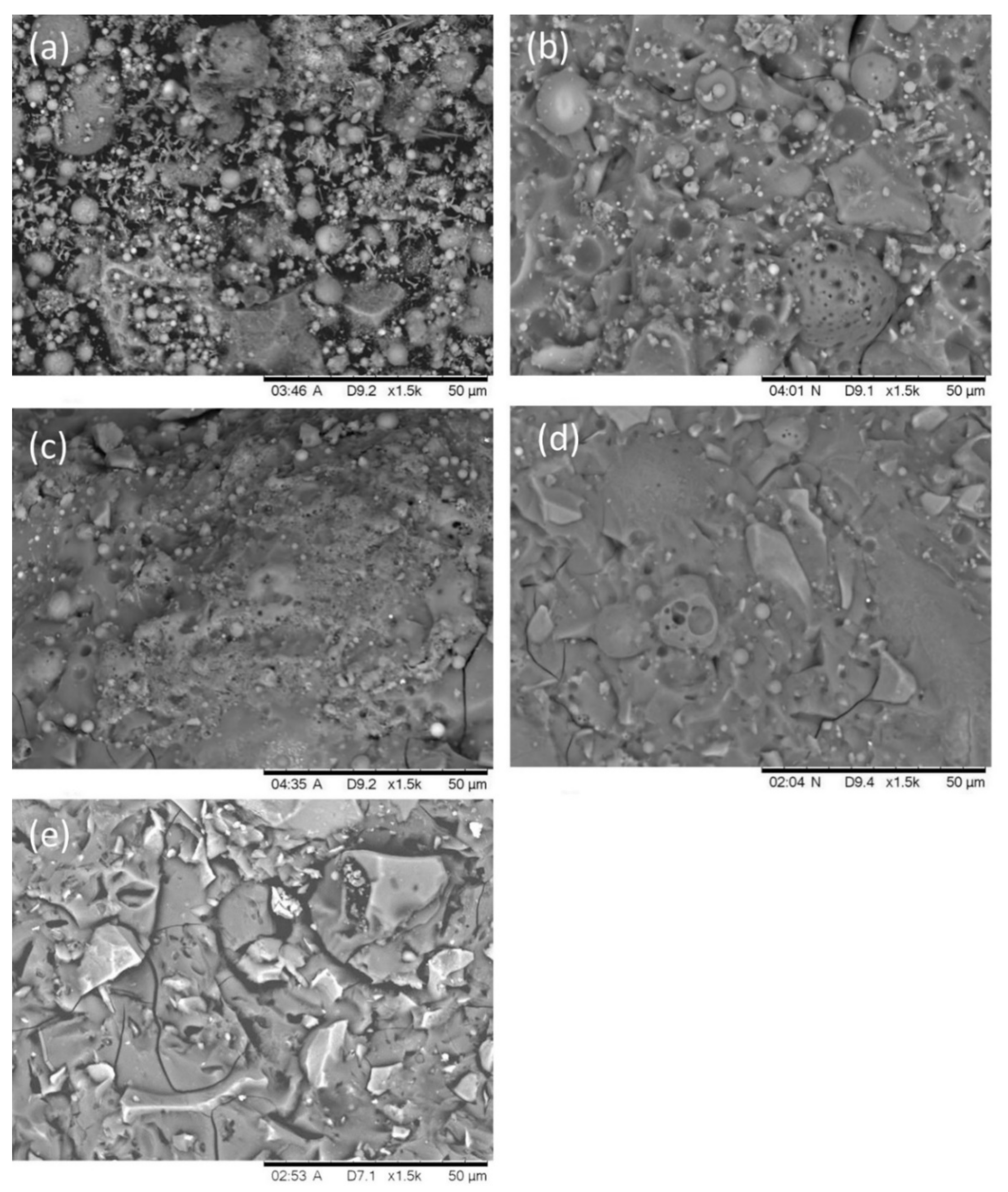
3.6. SEM Analysis
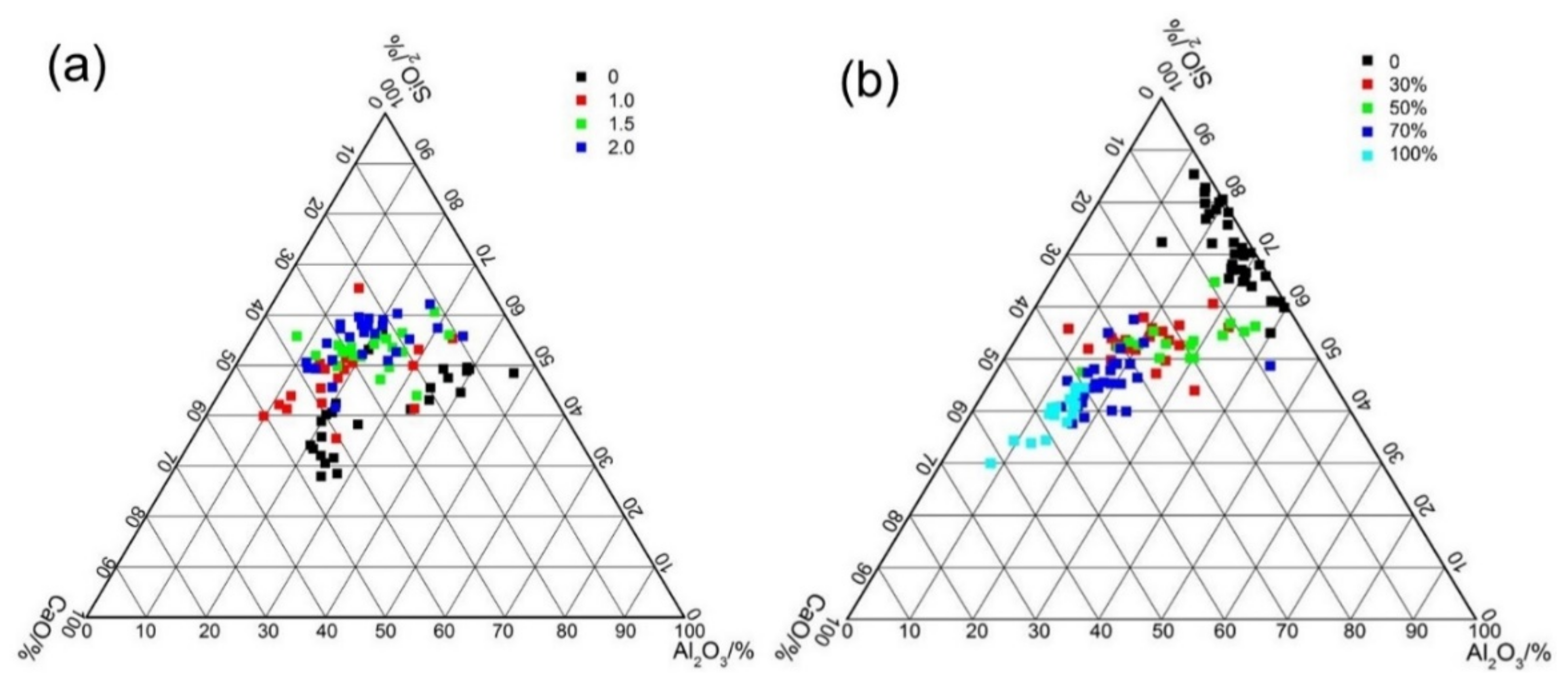
3.7. EDS Analysis
4. Conclusions
- (1)
- Both the activator modulus and slag content affected the setting time and workability of AAFS pastes. When the activator modulus was different, the effect of slag content on setting time and workability showed different trends. It indicated that a combined effect existed as the raw material and activator modulus varied.
- (2)
- AAFS mixtures with higher slag content had a higher rate of heat release and higher extent of the reaction, leading to an increased compressive strength both at 7 d and 28 d. The increase of the activator modulus from 0 to 1.0 resulted in a higher heat release and compressive strength, while further increasing the activator modulus to 2.0 resulted in a low extent of reaction, and was unfavorable to the development of the strength. The dissolution of slag was more pronounced when NaOH was applied as the activator.
- (3)
- The MIP results indicated that the addition of slag greatly decreased the capillary pores and increased the gel pores in AAFS, leading to lower total porosity and smaller pores. The use of Na2SiO3 as activator significantly refined the pores in AAFS, while further increasing the modulus from 1.0 to 2.0 resulted in a coarser pore structure.
- (4)
- The SEM/EDS results indicated that both the activator modulus and fly ash/slag content affected the types of reaction products in AAFS. The coexistence of N–A–S–H gel and C–A–S–H gel was identified in the AAFS activated with high pH but low SiO2 content. The C–A–S–H gel had higher space filling ability than N–A–S–H gel, resulting in a denser microstructure.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bernal, S.A.; Provis, J.L. Durability of alkali-activated materials: Progress and perspectives. J. Am. Ceram. Soc. 2014, 97, 997–1008. [Google Scholar] [CrossRef]
- Shi, C.; Qu, B.; Provis, J.L. Recent progress in low-carbon binders. Cem. Concr. Res. 2019, 122, 227–250. [Google Scholar] [CrossRef]
- Davidovits, J. Geopolymers: Inorganic polymeric new materials. J. Therm. Anal. Calorim. 1991, 37, 1633–1656. [Google Scholar] [CrossRef]
- Chindaprasirt, P.; De Silva, P.; Sagoe-Crentsil, K.; Hanjitsuwan, S. Effect of SiO2 and Al2O3 on the setting and hardening of high calcium fly ash-based geopolymer systems. J. Mater. Sci. 2012, 47, 4876–4883. [Google Scholar] [CrossRef]
- Ma, Y.; Ye, G.; Hu, J. Micro-mechanical properties of alkali-activated fly ash evaluated by nanoindentation. Constr. Build. Mater. 2017, 147, 407–416. [Google Scholar] [CrossRef]
- Duxson, P.; Fernández-Jiménez, A.; Provis, J.L.; Lukey, G.C.; Palomo, A.; van Deventer, J.S. Geopolymer technology: The current state of the art. J. Mater. Sci. 2006, 42, 2917–2933. [Google Scholar] [CrossRef]
- Hajimohammadi, A.; Ngo, T.; Vongsvivut, J. Interfacial chemistry of a fly ash geopolymer and aggregates. J. Clean. Prod. 2019, 231, 980–989. [Google Scholar] [CrossRef]
- Hajimohammadi, A.; Ngo, T.; Kashani, A. Glass waste versus sand as aggregates: The characteristics of the evolving geopolymer binders. J. Clean. Prod. 2018, 193, 593–603. [Google Scholar] [CrossRef]
- Lee, W.K.W.; Van Deventer, J.S.J. The effect of ionic contaminants on the early-age properties of alkali-activated fly ash-based cements. Cem. Concr. Res. 2002, 32, 577–584. [Google Scholar] [CrossRef]
- Chindaprasirt, P.; Chareerat, T.; Sirivivatnanon, V. Workability and strength of coarse high calcium fly ash geopolymer. Cem. Concr. Compos. 2007, 29, 224–229. [Google Scholar] [CrossRef]
- Guo, X.; Shi, H.; Dick, W.A. Compressive strength and microstructural characteristics of class C fly ash geopolymer. Cem. Concr. Compos. 2010, 32, 142–147. [Google Scholar] [CrossRef]
- Zaharaki, D.; Komnitsas, K.; Perdikatsis, V. Use of analytical techniques for identification of inorganic polymer gel composition. J. Mater. Sci. 2010, 45, 2715–2724. [Google Scholar] [CrossRef]
- Škvára, F.; Jílek, T.; Kopecky, L. Geopolymer materials based on fly ash. Ceram.Silik. 2005, 49, 195–204. [Google Scholar]
- Sata, V.; Sathonsaowaphak, A.; Chindaprasirt, P. Resistance of lignite bottom ash geopolymer mortar to sulfate and sulfuric acid attack. Cem. Concr. Compos. 2012, 34, 700–708. [Google Scholar] [CrossRef]
- Lyon, R.E.; Balaguru, P.N.; Foden, A.; Sorathia, U.; Davidovits, J.; Davidovics, M. Fire-resistant aluminosilicate composites. Fire Mater. 1997, 21, 67–73. [Google Scholar] [CrossRef]
- García-Lodeiro, I.; Palomo, A.Y.; Fernández-Jiménez, A. Alkali–aggregate reaction in activated fly ash systems. Cem. Concr. Res. 2007, 37, 175–183. [Google Scholar] [CrossRef]
- Gifford, P.M.; Gillott, J.E. Freeze thaw durability of activated blast furnace slag cement concrete. ACI Mater. J. 1996, 93, 242–245. [Google Scholar]
- Soultana, A.; Valouma, A.; Bartzas, G.; Komnitsas, K. Properties of Inorganic Polymers Produced from Brick Waste and Metallurgical Slag. Minerals 2019, 9, 551. [Google Scholar] [CrossRef]
- Lloyd, R.R.; Provis, J.L.; Smeaton, K.J.; van Deventer, J.S. Spatial distribution of pores in fly ash-based inorganic polymer gels visualised by Wood’s metal intrusion. Microporous Mesoporous Mater. 2009, 126, 32–39. [Google Scholar] [CrossRef]
- Ravikumar, D.; Neithalath, N. Effects of activator characteristics on the reaction product formation in slag binders activated using alkali silicate powder and NaOH. Cem. Concr. Compos. 2012, 34, 809–818. [Google Scholar] [CrossRef]
- Hajimohammadi, A.; van Deventer, J.S. Dissolution behaviour of source materials for synthesis of geopolymer binders: A kinetic approach. Int. J. Miner. Process. 2016, 153, 80–86. [Google Scholar] [CrossRef]
- Palomo, A.; Alonso, S.; Fernandez-Jiménez, A.; Sobrados, I.; Sanz, J. Alkaline activation of fly ashes: NMR study of the reaction products. J. Am. Ceram. Soc. 2004, 87, 1141–1145. [Google Scholar] [CrossRef]
- Phoo-ngernkham, T.; Maegawa, A.; Mishima, N.; Hatanaka, S.; Chindaprasirt, P. Effects of sodium hydroxide and sodium silicate solutions on compressive and shear bond strengths of FA–GBFS geopolymer. Constr. Build. Mater. 2015, 91, 1–8. [Google Scholar] [CrossRef]
- Criado, M.; Fernández-Jiménez, A.; De La Torre, A.; Aranda, M.; Palomo, A. An XRD study of the effect of the SiO2/Na2O ratio on the alkali activation of fly ash. Cem. Concr. Res. 2007, 37, 671–679. [Google Scholar] [CrossRef]
- Somna, K.; Jaturapitakkul, C.; Kajitvichyanukul, P.; Chindaprasirt, P. NaOH-activated ground fly ash geopolymer cured at ambient temperature. Fuel 2011, 90, 2118–2124. [Google Scholar] [CrossRef]
- Wu, X.; Jiang, W.; Roy, D.M. Early activation and properties of slag cement. Cem. Concr. Res. 1990, 20, 961–974. [Google Scholar] [CrossRef]
- Provis, J.L.; Myers, R.J.; White, C.E.; Rose, V.; Van Deventer, J.S. X-ray microtomography shows pore structure and tortuosity in alkali-activated binders. Cem. Concr. Res. 2012, 42, 855–864. [Google Scholar] [CrossRef]
- Ismail, I.; Bernal, S.A.; Provis, J.L.; San Nicolas, R.; Hamdan, S.; van Deventer, J.S. Modification of phase evolution in alkali-activated blast furnace slag by the incorporation of fly ash. Cem. Concr. Compos. 2014, 45, 125–135. [Google Scholar] [CrossRef]
- ASTM. ASTM C191-13 Standard Test Methods for Time of Setting of Hydraulic Cement by Vicat Needle; ASTM International: West Conshohocken, PA, USA, 2013. [Google Scholar]
- ASTM. C305 Standard Practice for Mechanical Mixing of Hydraulic Cement Pastes and Mortars of Plastic Consistency. In Annual Book of ASTM Standards; ASTM International: West Conshohocken, PA, USA, 2006. [Google Scholar]
- Ma, Y.; Wang, G.; Ye, G.; Hu, J. A comparative study on the pore structure of alkali-activated fly ash evaluated by mercury intrusion porosimetry, N 2 adsorption and image analysis. J. Mater. Sci. 2018, 53, 5958–5972. [Google Scholar] [CrossRef]
- Li, N.; Shi, C.; Zhang, Z. Understanding the roles of activators towards setting and hardening control of alkali-activated slag cement. Compos. Part B Eng. 2019, 171, 34–45. [Google Scholar] [CrossRef]
- Ravikumar, D.; Neithalath, N. Reaction kinetics in sodium silicate powder and liquid activated slag binders evaluated using isothermal calorimetry. Thermochim. Acta 2012, 546, 32–43. [Google Scholar] [CrossRef]
- Li, N.; Shi, C.; Zhang, Z.; Zhu, D.; Hwang, H.-J.; Zhu, Y.; Sun, T. A mixture proportioning method for the development of performance-based alkali-activated slag-based concrete. Cem. Concr. Compos. 2018, 93, 163–174. [Google Scholar] [CrossRef]
- Provis, J.L.; Van Deventer, J.S. Alkali Activated Materials: State-of-the-Art Report, RILEM TC 224-AAM; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013; Volume 13. [Google Scholar]
- Palomo, A.; Grutzeck, M.; Blanco, M. Alkali-activated fly ashes: A cement for the future. Cem. Concr. Res. 1999, 29, 1323–1329. [Google Scholar] [CrossRef]
- Puertas, F.; Varga, C.; Alonso, M. Rheology of alkali-activated slag pastes. Effect of the nature and concentration of the activating solution. Cem. Concr. Compos. 2014, 53, 279–288. [Google Scholar] [CrossRef]
- Ouyang, X.; Koleva, D.A.; Ye, G.; van Breugel, K. Insights into the mechanisms of nucleation and growth of C-S-H on fillers. Mater. Struct. 2017, 50, 213. [Google Scholar] [CrossRef]
- Ouyang, X.; Koleva, D.A.; Ye, G.; van Breugel, K. Understanding the adhesion mechanisms between C–S–H and fillers. Cem. Concr. Res. 2017, 100, 275–283. [Google Scholar] [CrossRef]
- Wang, L.; Liu, Z.; Xu, S.; Ouyang, X.; Ouyang, D.; Jiao, C.; Zhang, Y. A study on the effect of ceramic polishing powder on the nucleation and growth of hydrates in cement paste. Crystals 2019, 9, 545. [Google Scholar] [CrossRef]
- Ouyang, X.; Pan, Z.; Qian, Z.; Ma, Y.; Ye, G.; van Breugel, K. Numerical Modelling of the Effect of Filler/Matrix Interfacial Strength on the Fracture of Cementitious Composites. Materials 2018, 11, 1362. [Google Scholar] [CrossRef]
- Ouyang, X.; Ye, G.; van Breugel, K. Experimental and numerical evaluation of mechanical properties of interface between filler and hydration products. Constr. Build. Mater. 2017, 135, 538–549. [Google Scholar] [CrossRef]
- Ouyang, X.; Gao, P.; Ye, G.; van Breugel, K. Effect of Filler-Hydrates Adhesion Properties on Cement Paste Strength. ACI Mater. J. 2018, 115, 437–447. [Google Scholar] [CrossRef]
- Ouyang, X. Filler-Hydrates Adhesion Properties in Cement Paste System: Development of Sustainable Building Materials. Ph.D. Thesis, Delft University of Technology, Delft, The Netherlands, 2017. [Google Scholar]
- Zhang, Y.M.; Sun, W.; Yan, H.D. Hydration of high-volume fly ash cement pastes. Cem. Concr. Compos. 2000, 22, 445–452. [Google Scholar] [CrossRef]
- Ma, Y.; Yang, X.; Hu, J.; Zhang, Z.; Wang, H. Accurate determination of the “time-zero” of autogenous shrinkage in alkali-activated fly ash/slag system. Compos. Part B Eng. 2019, 177, 107367. [Google Scholar] [CrossRef]
- Bernal, S.A. Effect of the activator dose on the compressive strength and accelerated carbonation resistance of alkali silicate-activated slag/metakaolin blended materials. Constr. Build. Mater. 2015, 98, 217–226. [Google Scholar] [CrossRef]
- Gao, X.; Yu, Q.L.; Brouwers, H.J.H. Reaction kinetics, gel character and strength of ambient temperature cured alkali activated slag–fly ash blends. Constr. Build. Mater. 2015, 80, 105–115. [Google Scholar] [CrossRef]
- Komnitsas, K.; Zaharaki, D.; Perdikatsis, V. Geopolymerisation of low calcium ferronickel slags. J. Mater. Sci. 2007, 42, 3073–3082. [Google Scholar] [CrossRef]
- Komnitsas, K.; Zaharaki, D.; Perdikatsis, V. Effect of synthesis parameters on the compressive strength of low-calcium ferronickel slag inorganic polymers. J. Hazard. Mater. 2009, 161, 760–768. [Google Scholar] [CrossRef]
- Wang, G.; Ma, Y. Drying shrinkage of alkali-activated fly ash/slag blended system. J. Sustain. Cem. Based Mater. 2018, 7, 203–213. [Google Scholar] [CrossRef]


| Oxide | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | Na2O | K2O | LOI* |
|---|---|---|---|---|---|---|---|---|
| Fly ash (wt %) | 49.1 | 34.8 | 4.5 | 4.9 | 0.4 | 0.4 | 1.3 | 2.3 |
| Slag (wt %) | 32.6 | 16.4 | 0.4 | 38.7 | 7.1 | 0.3 | 0.3 | 0.5 |
| Name | Fly Ash (g) | Slag (g) | The Activator Modulus (SiO2/Na2O) | SiO2 (g) | Na2O (g) | Water (g) |
|---|---|---|---|---|---|---|
| S0N0 | 100 | 0 | 0 | 0 | 4.89 | 35 |
| S3N0 | 70 | 30 | 0 | 0 | 4.89 | 35 |
| S5N0 | 50 | 50 | 0 | 0 | 4.89 | 35 |
| S7N0 | 30 | 70 | 0 | 0 | 4.89 | 35 |
| S10N0 | 0 | 100 | 0 | 0 | 4.89 | 35 |
| S0N10 | 100 | 0 | 1.0 | 5.90 | 6.10 | 35 |
| S3N10 | 70 | 30 | 1.0 | 5.90 | 6.10 | 35 |
| S5N10 | 50 | 50 | 1.0 | 5.90 | 6.10 | 35 |
| S7N10 | 30 | 70 | 1.0 | 5.90 | 6.10 | 35 |
| S10N10 | 0 | 100 | 1.0 | 5.90 | 6.10 | 35 |
| S0N15 | 100 | 0 | 1.5 | 7.11 | 4.89 | 35 |
| S3N15 | 70 | 30 | 1.5 | 7.11 | 4.89 | 35 |
| S5N15 | 50 | 50 | 1.5 | 7.11 | 4.89 | 35 |
| S7N15 | 30 | 70 | 1.5 | 7.11 | 4.89 | 35 |
| S10N15 | 0 | 100 | 1.5 | 7.11 | 4.89 | 35 |
| S0N20 | 100 | 0 | 2.0 | 7.91 | 4.09 | 35 |
| S3N20 | 70 | 30 | 2.0 | 7.91 | 4.09 | 35 |
| S5N20 | 50 | 50 | 2.0 | 7.91 | 4.09 | 35 |
| S7N20 | 30 | 70 | 2.0 | 7.91 | 4.09 | 35 |
| S10N20 | 0 | 100 | 2.0 | 7.91 | 4.09 | 35 |
| Raw Materials | SiO2/Na2O Ratio | Temperature (°C) | Curing Age (Days) | Compressive Strength (MPa) | Reference |
|---|---|---|---|---|---|
| Low-calcium slag and kaolin (69:9) | 3.4 | 60 | 7 | 10.8 | [49] |
| High-calcium fly ash | 4.8 | 60 | 2 | 62.6 | [4] |
| Brick waste and metallurgical slag (1:1) | 1.16 | 90 | 7 | 48.9 | [18] |
| Fly ash and GBFS (1:1) | 2.56 | 23 | 7 | 84.9 | [23] |
| This study (FA/SL = 1:1) | 1.5 | 20 | 7 | 52 | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ouyang, X.; Ma, Y.; Liu, Z.; Liang, J.; Ye, G. Effect of the Sodium Silicate Modulus and Slag Content on Fresh and Hardened Properties of Alkali-Activated Fly Ash/Slag. Minerals 2020, 10, 15. https://doi.org/10.3390/min10010015
Ouyang X, Ma Y, Liu Z, Liang J, Ye G. Effect of the Sodium Silicate Modulus and Slag Content on Fresh and Hardened Properties of Alkali-Activated Fly Ash/Slag. Minerals. 2020; 10(1):15. https://doi.org/10.3390/min10010015
Chicago/Turabian StyleOuyang, Xiaowei, Yuwei Ma, Ziyang Liu, Jianjun Liang, and Guang Ye. 2020. "Effect of the Sodium Silicate Modulus and Slag Content on Fresh and Hardened Properties of Alkali-Activated Fly Ash/Slag" Minerals 10, no. 1: 15. https://doi.org/10.3390/min10010015
APA StyleOuyang, X., Ma, Y., Liu, Z., Liang, J., & Ye, G. (2020). Effect of the Sodium Silicate Modulus and Slag Content on Fresh and Hardened Properties of Alkali-Activated Fly Ash/Slag. Minerals, 10(1), 15. https://doi.org/10.3390/min10010015







