An fcu Th-MOF Constructed from In Situ Coupling of Monovalent Ligands
Abstract
:1. Introduction
2. Materials and Methods
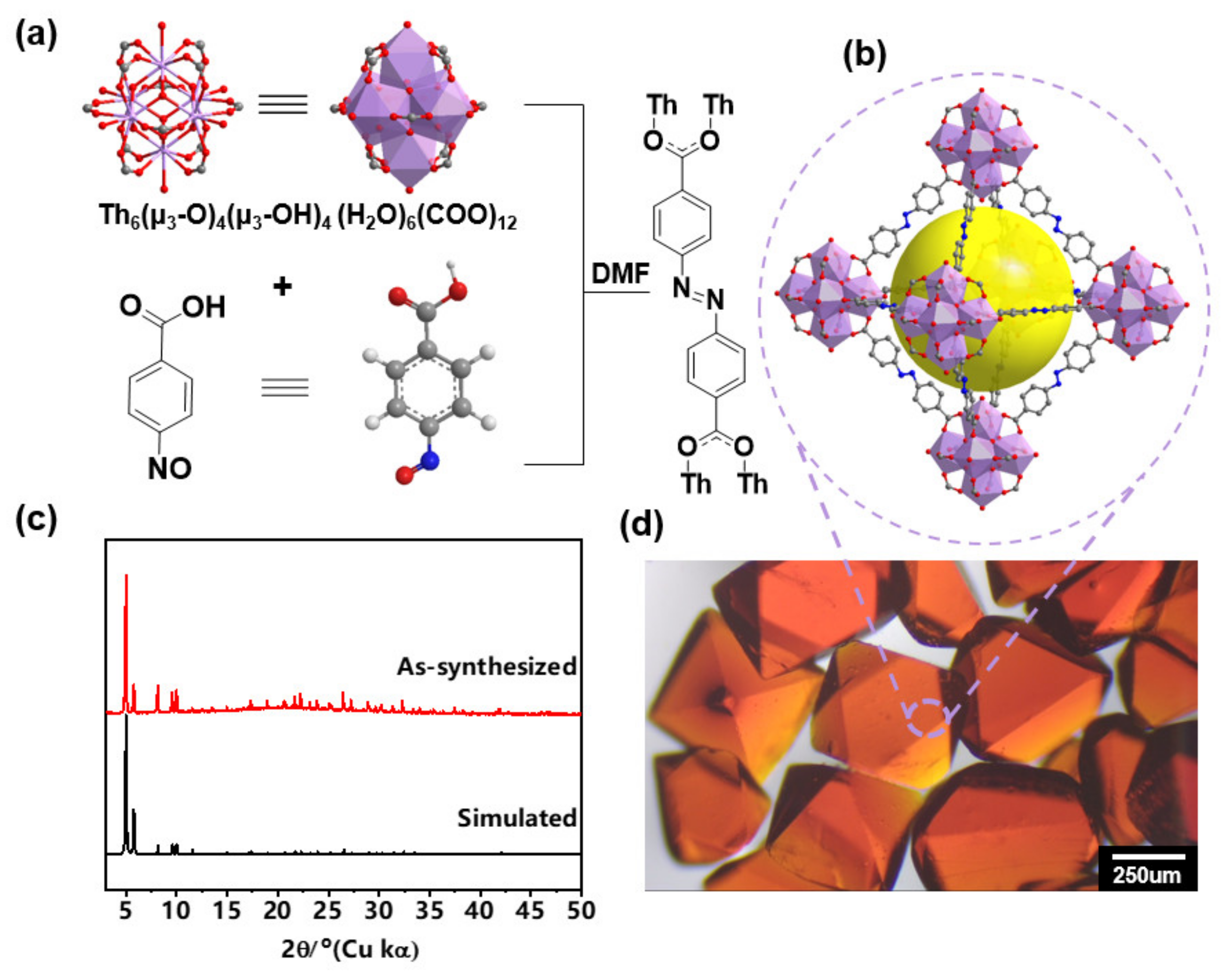
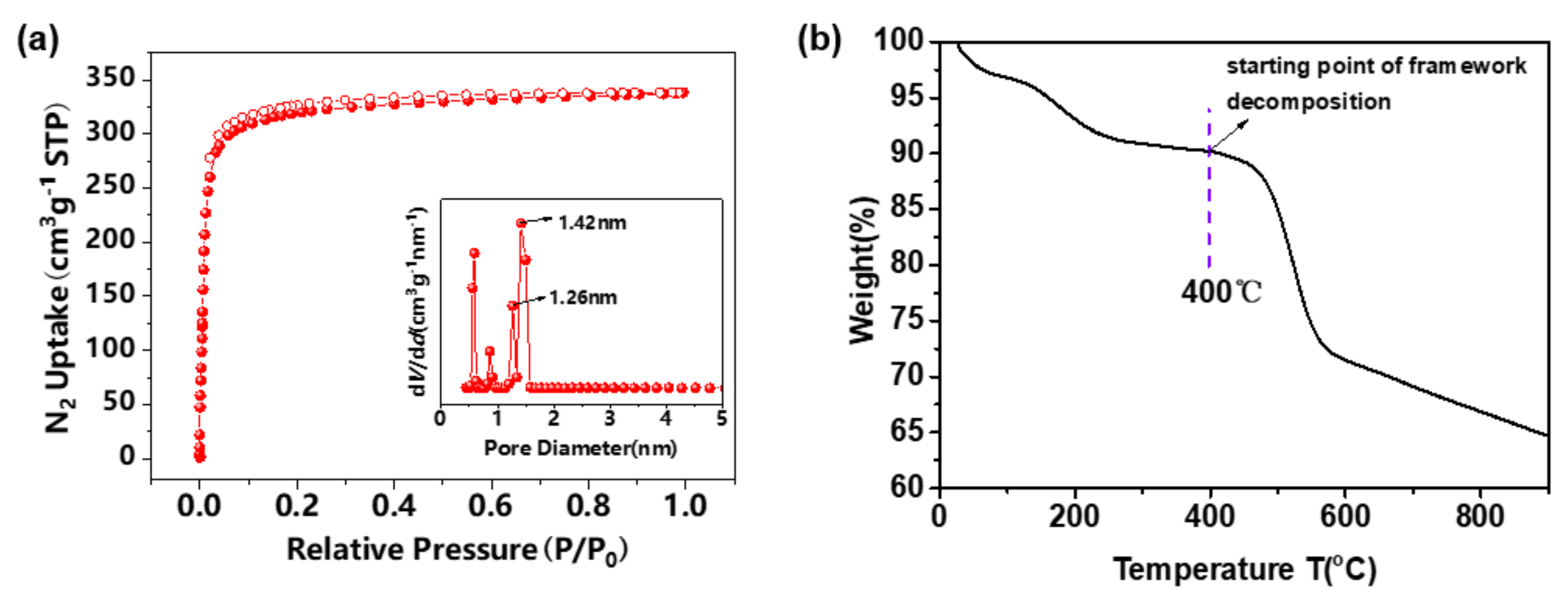
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maurin, G.; Serre, C.; Cooper, A.; Férey, G. The new age of MOFs and of their porous-related solids. Chem. Soc. Rev. 2017, 46, 3104–3107. [Google Scholar] [CrossRef]
- Dincǎ, M.; Long, J.R. Introduction: Porous Framework Chemistry. Chem. Rev. 2020, 120, 8037–8038. [Google Scholar] [CrossRef]
- O’Keeffe, M.; Peskov, M.A.; Ramsden, S.J.; Yaghi, O.M. The Reticular Chemistry Structure Resource (RCSR) database of, and symbols for, crystal nets. Acc. Chem. Res. 2008, 41, 1782–1789. [Google Scholar] [CrossRef]
- According to the Cambridge Crystallographic Data Centre Statistics. Available online: https://www.ccdc.cam.ac.uk/Community/blog/MOF-classification-search-screen/ (accessed on 15 June 2021).
- Schaate, A.; Roy, P.; Godt, A.; Lippke, J.; Waltz, F.; Wiebcke, M.; Behrens, P. Modulated Synthesis of Zr-Based Metal-Organic Frameworks: From Nano to Single Crystals. Chem. A Eur. J. 2011, 17, 6643–6651. [Google Scholar] [CrossRef]
- Feng, D.; Gu, Z.; Li, J.; Jiang, H.; Wei, Z.; Zhou, H.C. Zirconium-Metalloporphyrin PCN-222: Mesoporous Metal-Organic Frameworks with Ultrahigh Stability as Biomimetic Catalysts. Angew. Chem. Int. Ed. 2012, 51, 10307–10310. [Google Scholar] [CrossRef]
- Morris, W.; Volosskiy, B.; Demir, S.; Gándara, F.; McGrier, P.L.; Furukawa, H.; Cascio, D.; Stoddart, J.F.; Yaghi, O.M. Synthesis, Structure, and Metalation of Two New Highly Porous Zirconium Metal–Organic Frameworks. Inorg. Chem. 2012, 51, 6443–6445. [Google Scholar] [CrossRef]
- Mondloch, J.E.; Bury, W.; Fairen-Jimenez, D.; Kwon, S.; DeMarco, E.J.; Weston, M.H.; Sarjeant, A.A.; Nguyen, S.; Stair, P.C.; Snurr, R.; et al. Vapor-Phase Metalation by Atomic Layer Deposition in a Metal–Organic Framework. J. Am. Chem. Soc. 2013, 135, 10294–10297. [Google Scholar] [CrossRef]
- Xue, D.-X.; Cairns, A.J.; Belmabkhout, Y.; Wojtas, L.; Liu, Y.; Alkordi, M.; Eddaoudi, M. Tunable Rare-Earth fcu-MOFs: A Platform for Systematic Enhancement of CO2 Adsorption Energetics and Uptake. J. Am. Chem. Soc. 2013, 135, 7660–7667. [Google Scholar] [CrossRef]
- Nguyen, H.; Gándara, F.; Furukawa, H.; Doan, T.L.H.; Cordova, K.; Yaghi, O. A Titanium–Organic Framework as an Exemplar of Combining the Chemistry of Metal– and Covalent–Organic Frameworks. J. Am. Chem. Soc. 2016, 138, 4330–4333. [Google Scholar] [CrossRef]
- Feng, L.; Yuan, S.; Qin, J.S.; Wang, Y.; Kirchon, A.; Qiu, D.; Cheng, L.; Madrahimov, S.T.; Zhou, H.C. Lattice Expansion and Contraction in Metal-Organic Frameworks by Sequential Linker Reinstallation. Matter 2019, 1, 156–167. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Wang, J.; Xue, F.; Wu, Y.; Xu, H.; Yi, T.; Li, Q. An Imine-Linked Metal–Organic Framework as a Reactive Oxygen Species Generator. Angew. Chem. Int. Ed. 2021, 60, 2534–2540. [Google Scholar] [CrossRef]
- Xu, H.S.; Luo, Y.; See, P.Z.; Li, X.; Chen, Z.; Zhou, Y.; Zhao, X.; Leng, K.; Park, I.H.; Li, R.; et al. Divergent Chemistry Paths for 3D and 1D Metallo-Covalent Organic Frameworks (COFs). Angew. Chem. Int. Ed. 2020, 59, 11527–11532. [Google Scholar] [CrossRef]
- Beaudoin, D.; Maris, T.; Wuest, J.D. Constructing monocrystalline covalent organic networks by polymerization. Nat. Chem. 2013, 5, 830–834. [Google Scholar] [CrossRef]
- Ma, T.; Kapustin, E.A.; Yin, S.X.; Liang, L.; Zhou, Z.; Niu, J.; Li, L.-H.; Wang, Y.; Su, J.; Li, J.; et al. Single-crystal x-ray diffraction structures of covalent organic frameworks. Science 2018, 361, 48–52. [Google Scholar] [CrossRef] [Green Version]
- Edelstein, N.M.; Fuger, J.; Morss, L.R. The Chemistry of the Actinide and Transactinide Elements, 5th ed.; Springer: Dordrecht, The Netherlands, 2010; pp. 3472–3474. [Google Scholar]
- Tutson, C.D.; Gorden, A.E. Thorium coordination: A comprehensive review based on coordination number. Coord. Chem. Rev. 2017, 333, 27–43. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Yang, Z.; Wang, Y.; Bai, Z.; Zheng, T.; Dai, X.; Liu, S.; Gui, D.; Liu, W.; Chen, M.; et al. A mesoporous cationic thorium-organic framework that rapidly traps anionic persistent organic pollutants. Nat. Commun. 2017, 8, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Andreo, J.; Priola, E.; Alberto, G.; Benzi, P.; Marabello, D.; Proserpio, D.M.; Lamberti, C.; Diana, E. Autoluminescent Metal-Organic Frameworks: Self-Photoemission of a Highly Stable Thorium MOF. J. Am. Chem. Soc. 2018, 140, 14144–14149. [Google Scholar] [CrossRef] [Green Version]
- Gilson, S.E.; Fairley, M.; Julien, P.; Oliver, A.G.; Hanna, G.A.; Farha, O.K.; LaVere, J.A.; Burns, P.C. Unprecedented Radiation Resistant Thorium-Binaphthol MOF. J. Am. Chem. Soc. 2020, 142, 13299–13304. [Google Scholar] [CrossRef]
- Takeuchi, T.; Mori, T.; Kuwahara, A.; Ohta, T.; Oshita, A.; Sunayama, H.; Kitayama, Y.; Ooya, T. Conjugated-Protein Mimics with Molecularly Imprinted Reconstructible and Transformable Regions that are Assembled Using Space-Filling Prosthetic Groups. Angew. Chem. Int. Ed. 2014, 53, 12765–12770. [Google Scholar] [CrossRef]
- Wei, R.-J.; Zhou, H.-G.; Zhang, Z.-Y.; Ning, G.-H.; Li, D. Copper (I)–Organic Frameworks for Catalysis: Networking Metal Clusters with Dynamic Covalent Chemistry. CCS Chem. 2020, 2, 2045–2053. [Google Scholar] [CrossRef]
- Li, Z.-J.; Ju, Y.; Yu, B.; Wu, X.; Lu, H.; Li, Y.; Zhou, J.; Guo, X.; Zhang, Z.-H.; Lin, J.; et al. Modulated synthesis and isoreticular expansion of Th-MOFs with record high pore volume and surface area for iodine adsorption. Chem. Commun. 2020, 56, 6715–6718. [Google Scholar] [CrossRef]
- Falaise, C.; Charles, J.-S.; Volkringer, C.; Loiseau, T. Thorium Terephthalates Coordination Polymers Synthesized in Solvothermal DMF/H2O System. Inorg. Chem. 2015, 54, 2235–2242. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, T.; Tang, W.; Bao, C.; Lai, Q.; Zhang, Z.; Feng, X.; Liu, C. An fcu Th-MOF Constructed from In Situ Coupling of Monovalent Ligands. Symmetry 2021, 13, 1332. https://doi.org/10.3390/sym13081332
Song T, Tang W, Bao C, Lai Q, Zhang Z, Feng X, Liu C. An fcu Th-MOF Constructed from In Situ Coupling of Monovalent Ligands. Symmetry. 2021; 13(8):1332. https://doi.org/10.3390/sym13081332
Chicago/Turabian StyleSong, Ting, Wenlei Tang, Chuer Bao, Qiuxue Lai, Zhiyuan Zhang, Xuan Feng, and Chong Liu. 2021. "An fcu Th-MOF Constructed from In Situ Coupling of Monovalent Ligands" Symmetry 13, no. 8: 1332. https://doi.org/10.3390/sym13081332
APA StyleSong, T., Tang, W., Bao, C., Lai, Q., Zhang, Z., Feng, X., & Liu, C. (2021). An fcu Th-MOF Constructed from In Situ Coupling of Monovalent Ligands. Symmetry, 13(8), 1332. https://doi.org/10.3390/sym13081332






