Variation in Leaf Size and Fluctuating Asymmetry of Mountain Birch (Betula pubescens var. pumila) in Space and Time: Implications for Global Change Research
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Object
2.2. Study Region
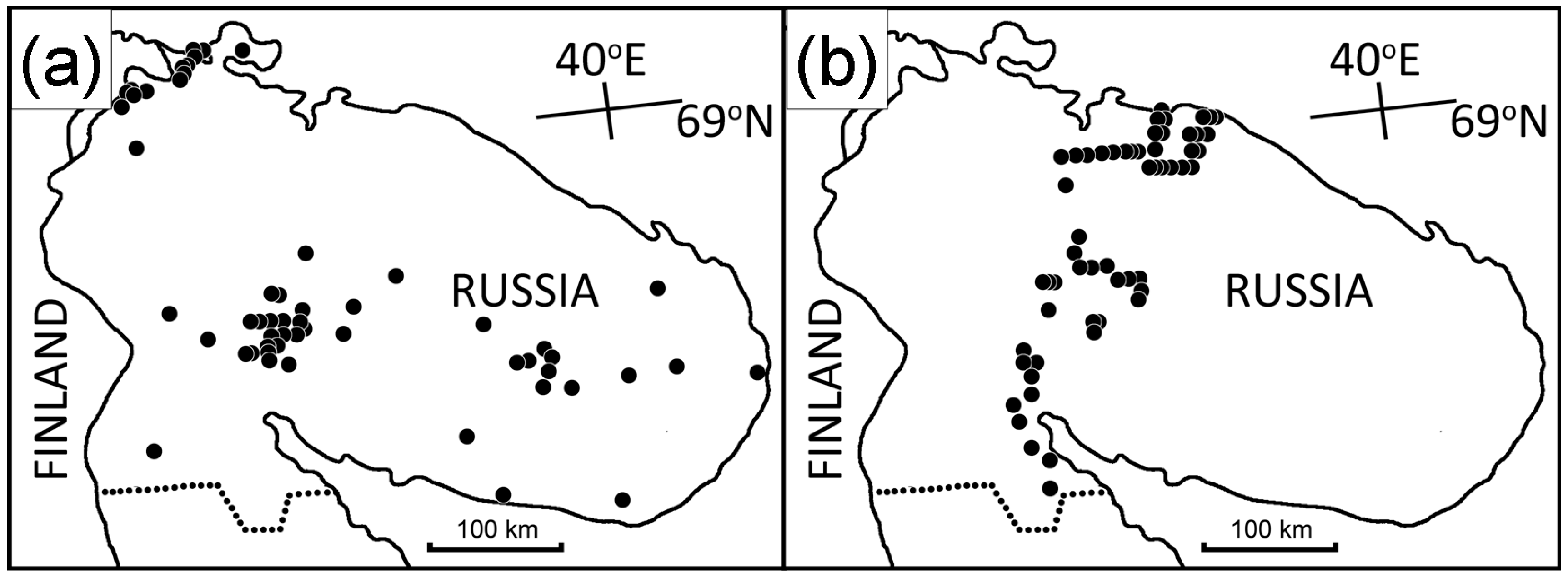
2.3. Historical Samples
2.4. Latitudinal Samples
2.5. Measurements
2.6. Calculation of FA and Testing for Reproducibility
2.7. Data Analysis
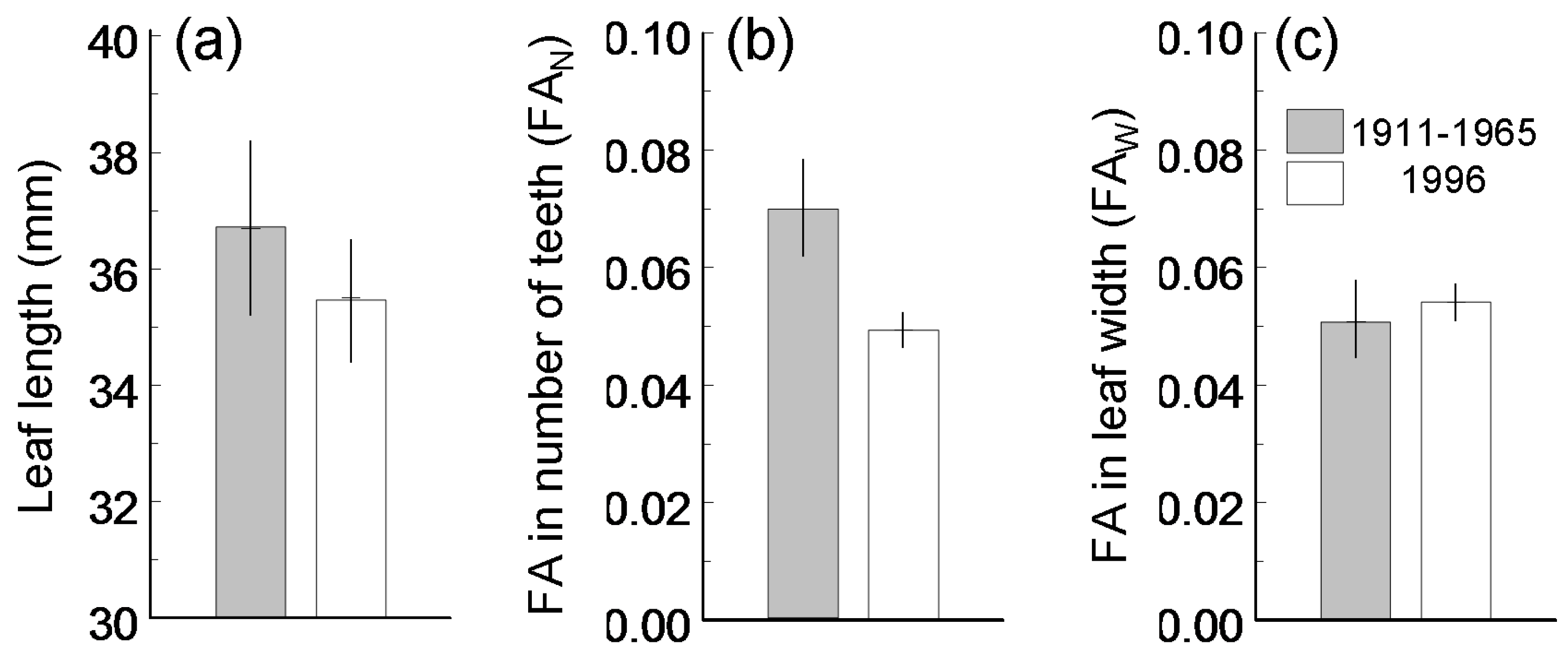
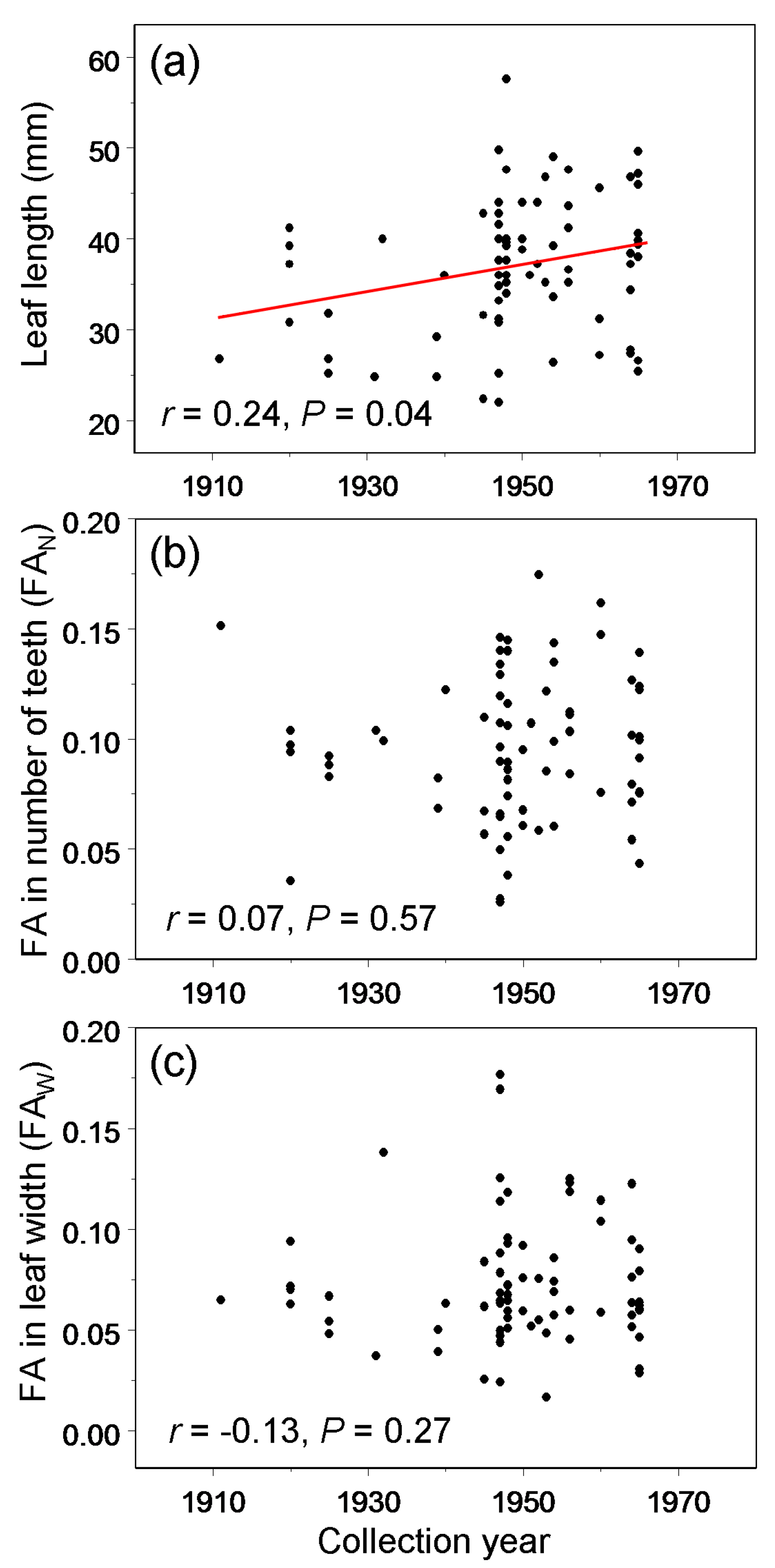
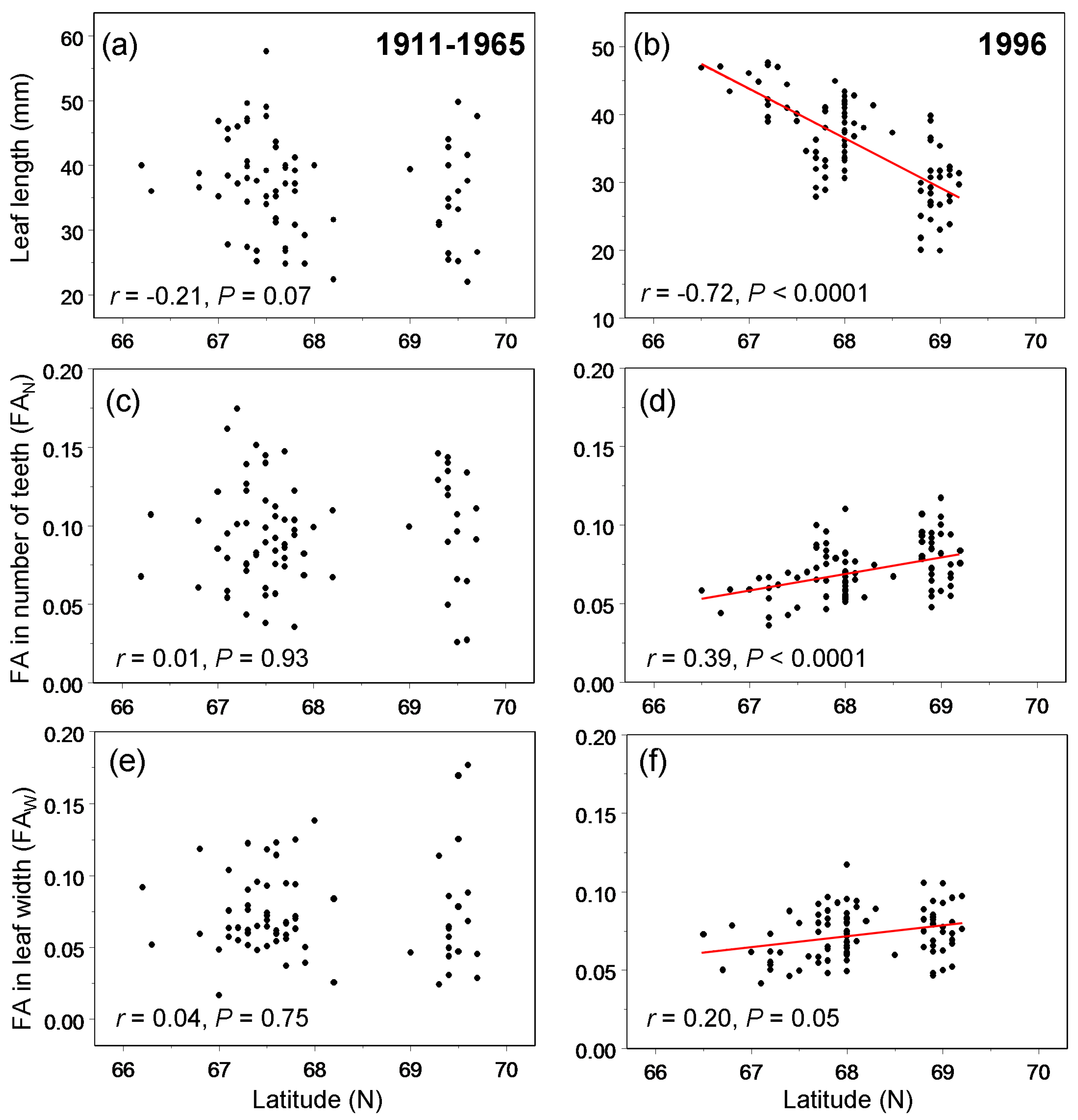
3. Results
4. Discussion
4.1. Temporal Variation
4.2. Spatial Variation
4.3. Importance of FA Measurements in Plants
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Intergovernmental Panel on Climate Change (IPCC). Climate Change 2014: Impacts, Adaptation, and Vulnerability; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014. [Google Scholar]
- Curtis, P.S.; Wang, X.Z. A meta-analysis of elevated CO2 effects on woody plant mass, form, and physiology. Oecologia 1998, 113, 299–313. [Google Scholar] [CrossRef] [PubMed]
- Ainsworth, E.A.; Long, S.P. What have we learned from 15 years of free-air CO2 enrichment (FACE)? A meta-analytic review of the responses of photosynthesis, canopy properties and plant production to rising CO2. New Phytol. 2005, 165, 351–372. [Google Scholar] [CrossRef] [PubMed]
- Zvereva, E.L.; Kozlov, M.V. Consequences of simultaneous elevation of carbon dioxide and temperature for plant-herbivore interactions: A meta-analysis. Glob. Chang. Biol. 2006, 12, 27–41. [Google Scholar] [CrossRef]
- Tylianakis, J.M.; Didham, R.K.; Bascompte, J.; Wardle, D.A. Global change and species interactions in terrestrial ecosystems. Ecol. Lett. 2008, 11, 1351–1363. [Google Scholar] [CrossRef]
- De Frenne, P.; Graae, B.J.; Rodríguez-Sánchez, F.; Kolb, A.; Chabrerie, O.; Decocq, G.; De Kort, H.; De Schrijver, A.; Diekmann, M.; Eriksson, O.; et al. Latitudinal gradients as natural laboratories to infer species’ responses to temperature. J. Ecol. 2013, 101, 784–795. [Google Scholar] [CrossRef]
- Wolkovich, E.M.; Cook, B.I.; McLauchlan, K.K.; Davies, T.J. Temporal ecology in the Anthropocene. Ecol. Lett. 2014, 17, 1365–1379. [Google Scholar] [CrossRef]
- Stips, A.; Macias, D.; Coughlan, C.; Garcia-Gorriz, E.; San Liang, X. On the causal structure between CO2 and global temperature. Sci. Rep. 2016, 6, 21691. [Google Scholar] [CrossRef]
- Kozlov, M.V.; Berlina, N.G. Decline in length of the summer season on the Kola Peninsula, Russia. Clim. Chang. 2002, 54, 387–398. [Google Scholar] [CrossRef]
- Guerin, G.R.; Wen, H.; Lowe, A.J. Leaf morphology shift linked to climate change. Biol. Lett. 2012, 8, 882–886. [Google Scholar] [CrossRef]
- Li, Y.; Zou, D.; Shrestha, N.; Xu, X.; Wang, Q.; Jia, W.; Wang, Z. Spatiotemporal variation in leaf size and shape in response to climate. J. Plant Ecol. 2020, 13, 87–96. [Google Scholar] [CrossRef]
- Poorter, L.; Rozendaal, D.M.A. Leaf size and leaf display of thirty-eight tropical tree species. Oecologia 2008, 158, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Price, P.W. The plant vigour hypothesis and herbivore attack. Oikos 1991, 62, 244–251. [Google Scholar] [CrossRef]
- De Bruyn, L.; Scheirs, J.; Verhagen, R. Nutrient stress, host plant quality and herbivore performance of a leaf-mining fly on grass. Oecologia 2002, 130, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Migalina, S.V.; Ivanova, L.A.; Makhnev, A.K. Size of the leaf as a marker of birch productivity at a distance from the climatic optimum. Russ. J. Plant Physiol. 2009, 56, 858–862. [Google Scholar] [CrossRef]
- Zakharov, V.M. Analysis of fluctuating asymmetry as a method of biomonitoring at the population level. In Bioindications of Chemical and Radioactive Pollution; Krivolutsky, D., Ed.; Mir Publishers: Moscow, Russia, 1990; pp. 187–198. [Google Scholar]
- Clarke, G.M. Fluctuating asymmetry: A technique for measuring developmental stress of genetic and environmental origin. Acta Zool. Fenn. 1992, 191, 31–35. [Google Scholar]
- Parsons, P.A. Fluctuating asymmetry—a biological monitor of environmental and genomic stress. Heredity 1992, 68, 361–364. [Google Scholar] [CrossRef]
- Graham, J.H.; Freeman, D.C.; Emlen, J.M. Antisymmetry, directional asymmetry, and dynamic morphogenesis. Genetica 1993, 89, 121–137. [Google Scholar] [CrossRef]
- Freeman, D.C.; Graham, J.H.; Emlen, J.M. Developmental stability in plants—symmetries, stress and epigenesis. Genetica 1993, 89, 97–119. [Google Scholar] [CrossRef]
- Palmer, A.R. Waltzing with asymmetry. Bioscience 1996, 46, 518–532. [Google Scholar] [CrossRef]
- Anne, P.; Mawri, F.; Gladstone, S.; Freeman, D.C. Is fluctuating asymmetry a reliable biomonitor of stress? A test using life history parameters in soybean. Intern. J. Plant Sci. 1998, 159, 559–565. [Google Scholar] [CrossRef]
- Graham, J.H.; Raz, S.; Hel-Or, H.; Nevo, E. Fluctuating asymmetry: Methods, theory, and applications. Symmetry 2010, 2, 466–540. [Google Scholar] [CrossRef]
- Kozlov, M.V. Plant studies on fluctuating asymmetry in Russia: Mythology and methodology. Russ. J. Ecol. 2017, 48, 1–9. [Google Scholar] [CrossRef]
- Sandner, T.M.; Matthies, D. Fluctuating asymmetry of leaves is a poor indicator of environmental stress and genetic stress by inbreeding in Silene vulgaris. Ecol. Indic. 2017, 79, 247–253. [Google Scholar] [CrossRef]
- Mortensen, L.M. Effects of elevated CO2 concentration on growth of Betula pubescens Ehrh. in different climatic conditions. Scand. J. For. Res. 1998, 13, 197–203. [Google Scholar] [CrossRef]
- Grau, O.; Ninot, J.M.; Blanco-Moreno, J.M.; van Logtestijn, R.S.P.; Cornelissen, J.H.C.; Callaghan, T.V. Shrub—tree interactions and environmental changes drive treeline dynamics in the Subarctic. Oikos 2012, 121, 1680–1690. [Google Scholar] [CrossRef]
- Xu, Z.; Hu, T.; Zhang, Y. Effects of experimental warming on phenology, growth and gas exchange of treeline birch (Betula utilis) saplings, Eastern Tibetan Plateau, China. Eur. J. For. Res. 2012, 131, 811–819. [Google Scholar] [CrossRef]
- Hartikainen, K.; Kivimäenpää, M.; Nerg, A.M.; Mäenpää, M.; Oksanen, E.; Rousi, M.; Holopainen, T. Elevated temperature and ozone modify structural characteristics of silver birch (Betula pendula) leaves. Tree Physiol. 2020, 40, 467–483. [Google Scholar] [CrossRef] [PubMed]
- Riikonen, J.; Lindsberg, M.M.; Holopainen, T.; Oksanen, E.; Lappi, J.; Peltonen, P.; Vapaavuori, E. Silver birch and climate change: Variable growth and carbon allocation responses to elevated concentrations of carbon dioxide and ozone. Tree Physiol. 2004, 24, 1227–1237. [Google Scholar] [CrossRef][Green Version]
- Riikonen, J.; Percy, K.E.; Kivimäenpää, M.; Kubiske, M.E.; Nelson, N.D.; Vapaavuori, E.; Karnosky, D.F. Leaf size and surface characteristics of Betula papyrifera exposed to elevated CO2 and O3. Environ. Poll. 2010, 158, 1029–1035. [Google Scholar] [CrossRef]
- Llorens, L.; Penuelas, J.; Emmett, B. Developmental instability and gas exchange responses of a heathland shrub to experimental drought and warming. Intern. J. Plant Sci. 2002, 163, 959–967. [Google Scholar] [CrossRef]
- Cornelissen, T.; Stiling, P.; Drake, B. Elevated CO2 decreases leaf fluctuating asymmetry and herbivory by leaf miners on two oak species. Glob. Chang. Biol. 2003, 10, 27–36. [Google Scholar] [CrossRef]
- Kaligarič, M.; Tognetti, R.; Janžekovič, F.; Raschi, A. Leaf fluctuating asymmetry of Myrtus communis L., affected by increases in atmospheric CO2 concentration: Evidence from a natural CO2 spring. Pol. J. Environ. Stud. 2008, 17, 503–508. [Google Scholar]
- Weih, M.; Karlsson, P.S. Growth response of mountain birch to air and soil temperature: Is increasing leaf-nitrogen content an acclimation to lower air temperature? New Phytol. 2001, 150, 147–155. [Google Scholar] [CrossRef]
- Wiegolaski, F.E. Plant Ecology, Herbivory, and Human Impact in Nordic Mountain Birch Forests; Springer: Berlin, Germany, 2005. [Google Scholar]
- Kozlov, M.V.; Zverev, V. Temperature and herbivory, but not pollution, affect fluctuating asymmetry of mountain birch leaves: Results of 25-year monitoring around the copper-nickel smelter in Monchegorsk, northwestern Russia. Sci. Total Environ. 2018, 640–641, 678–687. [Google Scholar] [CrossRef] [PubMed]
- Lister, A.A. Climate Change Research Group. Natural history collections as sources of long-term datasets. Trends Ecol. Evol. 2011, 26, 153–154. [Google Scholar] [CrossRef]
- Kozlov, M.V.; Cornelissen, T.; Gavrikov, D.E.; Kunavin, M.A.; Lama, A.D.; Milligan, J.R.; Zverev, V.; Zvereva, E.L. Reproducibility of fluctuating asymmetry measurements in plants: Sources of variation and implications for study design. Ecol. Indic. 2017, 73, 733–740. [Google Scholar] [CrossRef]
- Palmer, A.R.; Strobeck, C. Fluctuating asymmetry analyses revisited. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: Oxford, UK, 2003; pp. 279–319. [Google Scholar]
- Cornelissen, T.; Stiling, P. Small variations over large scales: Fluctuating asymmetry over the range of two oak species. Intern. J. Plant Sci. 2010, 171, 303–309. [Google Scholar] [CrossRef]
- SAS Institute. SAS/Stat. User’s Guide, Version 9.2; SAS Institute: Cary, NC, USA, 2009. [Google Scholar]
- Kenward, M.G.; Roger, J.H. An improved approximation to the precision of fixed effects from restricted maximum likelihood. Computat. Stat. Data Anal. 2009, 53, 2583–2595. [Google Scholar] [CrossRef]
- Littell, R.C.; Milliken, G.A.; Stroup, W.W.; Wolfinger, R.D.; Schabenberger, O. SAS for Mixed Models, 2nd ed.; SAS Institute: Cary, NC, USA, 2006. [Google Scholar]
- Högberg, P.; Näsholm, T.; Franklin, O.; Högberg, M.N. Tamm review: On the nature of the nitrogen limitation to plant growth in Fennoscandian boreal forests. Forest Ecol. Manag. 2017, 403, 161–185. [Google Scholar] [CrossRef]
- Paakkonen, E.; Holopainen, T. 1995. Influence of nitrogen supply on the response of clones of birch (Betula pendula Roth) to ozone. New Phytol. 1995, 129, 595–603. [Google Scholar] [CrossRef]
- Engardt, M.; Simpson, D.; Schwikowski, M.; Granat, L. Deposition of sulphur and nitrogen in Europe 1900–2050. Model calculations and comparison to historical observations. Tellus B Chem. Phys. Meteorol. 2017, 69, 1328945. [Google Scholar] [CrossRef]
- Zverev, V.E. Impact of Industrial Contamination on Ecology of Mountain Birch (Betula pubescens ssp. czerepanovii (Orlova) Hämet-Ahti) in the Kola Peninsula. Ph.D. Thesis, Institute of Plant and Animal Ecology, Yekaterinburg, Russia, March 2012. [Google Scholar]
- Zvereva, E.L.; Hunter, M.D.; Zverev, V.; Kozlov, M.V. Factors affecting population dynamics of leaf beetles in a subarctic region: The interplay between climate warming and pollution decline. Sci. Total Environ. 2016, 566–567, 1277–1288. [Google Scholar] [CrossRef] [PubMed]
- Klingenberg, C.P. Phenotypic plasticity, developmental instability, and robustness: The concepts and how they are connected. Front. Ecol. Evol. 2019, 7, 56. [Google Scholar] [CrossRef]
- Hagen, S.B.; Ims, R.A.; Yoccoz, N.G.; Sørlibråten, O. Fluctuating asymmetry as an indicator of elevation stress and distribution limits in mountain birch (Betula pubescens). Plant Ecol. 2008, 195, 157–163. [Google Scholar] [CrossRef]
- Murphy, H.T.; Lovett-Doust, J. 2004. Landscape-level effects on developmental instability: Fluctuating asymmetry across the range of honey locust, Gleditsia triacanthos (Fabaceae). Intern. J. Plant Sci. 2004, 165, 795–803. [Google Scholar] [CrossRef]
- Royer, D.L.; Mcelwain, J.C.; Adams, J.M.; Wilf, P. Sensitivity of leaf size and shape to climate within Acer rubrum and Quercus kelloggii. New Phytol. 2008, 179, 808–817. [Google Scholar] [CrossRef]
- Maya-Garcia, R.; Torres-Miranda, A.; Cuevas-Reyes, P.; Oyama, K. Morphological differentiation among populations of Quercus elliptica Nee (Fagaceae) along an environmental gradient in Mexico and Central America. Bot. Sci. 2020, 98, 50–65. [Google Scholar] [CrossRef]
- Waddington, C.H. Canalization of development and the inheritance of acquired characters. Nature 1942, 150, 563–565. [Google Scholar] [CrossRef]
- Zverev, V.; Lama, A.D.; Kozlov, M.V. Fluctuating asymmetry of birch leaves did not increase with pollution and drought stress in a controlled experiment. Ecol. Indic. 2018, 84, 283–289. [Google Scholar] [CrossRef]
- Debat, V. Symmetry is beauty—or is it? Grandeur et decadence de l’asymétrie fluctuante. Médecines Sci. 2016, 32, 774–780. [Google Scholar] [CrossRef][Green Version]
- Diaz-Gil, C.; Palmer, M.; Catalan, I.A.; Alos, J.; Fuiman, L.A.; Garcia, E.; Gil, M.D.; Grau, A.; Kang, A.; Maneja, R.H.; et al. Otolith fluctuating asymmetry: A misconception of its biological relevance? ICES J. Marine Sci. 2015, 72, 2079–2089. [Google Scholar] [CrossRef]
- Kozlov, M.V.; Zverev, V.; Sandner, T.M. Photosynthetic efficiency is higher in asymmetric leaves than in symmetric leaves of the same plant. Symmetry 2019, 11, 834. [Google Scholar] [CrossRef]
- Cornelissen, T.; Stiling, P. Perfect is best: Low leaf fluctuating asymmetry reduces herbivory by leaf miners. Oecologia 2005, 142, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Kozlov, M.V.; Zverev, V.; Zvereva, E.L. Do defoliating insects distinguish between symmetric and asymmetric leaves within a plant? Ecol. Entomol. 2018, 43, 656–664. [Google Scholar] [CrossRef]
- Grime, J.P. Plant Strategies and Vegetation Processes; Wiley: New York, NY, USA, 1979. [Google Scholar]
| Source of Variation | Leaf Width | Teeth Number | ||
|---|---|---|---|---|
| Test Statistics | p | Test Statistics | p | |
| Side | F1,200 = 0.42 | 0.52 | F1,200 = 25.5 | <0.0001 |
| Individual | F99,200 = 189.4 | <0.0001 | F99,200 = 238.0 | <0.0001 |
| Side × Individual | F99,200 = 4.85 | <0.0001 | F99,200 = 7.93 | <0.0001 |
| Source of Variation | Leaf Length | FAN † | FAW ‡ | |||
|---|---|---|---|---|---|---|
| Test Statistics | p | Test Statistics | p | Test Statistics | p | |
| Date * | F1,167.6 = 23.6 | <0.0001 | F1,402.2 = 4.73 | 0.0301 | F1,361.6 = 0.58 | 0.45 |
| Latitude | F1,167.8 = 62.3 | <0.0001 | F1,403.1 = 5.05 | 0.0251 | F1,362.5 = 1.80 | 0.18 |
| Date × Latitude | F1,167.8 = 23.7 | <0.0001 | F1,403.1 = 4.45 | 0.0355 | F1,362.5 = 0.59 | 0.44 |
| Site | χ21 = 159.9 | <0.0001 | χ21 = 2.53 | 0.0560 | χ21 = 5.37 | 0.0103 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zverev, V.; Kozlov, M.V. Variation in Leaf Size and Fluctuating Asymmetry of Mountain Birch (Betula pubescens var. pumila) in Space and Time: Implications for Global Change Research. Symmetry 2020, 12, 1703. https://doi.org/10.3390/sym12101703
Zverev V, Kozlov MV. Variation in Leaf Size and Fluctuating Asymmetry of Mountain Birch (Betula pubescens var. pumila) in Space and Time: Implications for Global Change Research. Symmetry. 2020; 12(10):1703. https://doi.org/10.3390/sym12101703
Chicago/Turabian StyleZverev, Vitali, and Mikhail V. Kozlov. 2020. "Variation in Leaf Size and Fluctuating Asymmetry of Mountain Birch (Betula pubescens var. pumila) in Space and Time: Implications for Global Change Research" Symmetry 12, no. 10: 1703. https://doi.org/10.3390/sym12101703
APA StyleZverev, V., & Kozlov, M. V. (2020). Variation in Leaf Size and Fluctuating Asymmetry of Mountain Birch (Betula pubescens var. pumila) in Space and Time: Implications for Global Change Research. Symmetry, 12(10), 1703. https://doi.org/10.3390/sym12101703







