Abstract
Stx1 toxin is one of the AB5 toxins of Shiga toxin-producing Escherichia coli (STEC) responsible for foodborne intoxication during outbreaks. The single-chain variable fragment (scFv) is the most common recombinant antibody format; it consists of both variable chains connected by a peptide linker with conserved specificity and affinity for antigen. The drawbacks of scFv production in bacteria are the heterologous expression, conformation and stability of the molecule, which could change the affinity for the antigen. In this work, we obtained a stable and functional scFv-Stx1 in bacteria, starting from IgG produced by hybridoma cells. After structural modifications, i.e., change in protein orientation, vector and linker, its solubility for expression in bacteria was increased as well as the affinity for its antigen, demonstrated by a scFv dissociation constant (KD) of 2.26 × 10−7 M. Also, it was able to recognize purified Stx1 and cross-reacted with Stx2 toxin by ELISA (Enzyme-Linked Immunosorbent Assay), and detected 88% of Stx1-producing strains using a rapid latex agglutination test. Thus, the scFv fragment obtained in the present work is a bacteria-produced tool for use in a rapid diagnosis test, providing an alternative for STEC diagnosis.
1. Introduction
The single-chain variable fragment (scFv) is a common format of recombinant antibody fragments, and consists of heavy (VH) and light (VL) variable domains, retaining the specificity of parental immunoglobulin [1]. Successful construction of scFvs depends on the choice of the peptide linker and connection of the VH and VL domains, which affect the stability and recognition properties of these antibody molecules [2,3]. Improvements in engineered recombinant antibody fragments could lead to ideal tools for therapy and diagnosis [1]. Indeed, it is possible to increase the antigen-binding affinity and specificity by mimicking somatic hypermutation during an immune response [4]. Moreover, with recombinant antibody technology, it is possible to replace animal immunization and hybridoma development with a bacterial system, which is capable of synthesizing and expressing practically unlimited quantities of antibodies, which would provide for a more cost-effective diagnostic tool [5].
Shiga toxin 1 (Stx1) is a potent bacterial toxin produced by Shiga toxin-producing Escherichia coli (STEC). It is a member of the AB5 bacterial toxin family, of which the B subunit (StxB) binds to globotriaosylceramide receptors (Gb3) on the host cell membrane and translocate the active A subunit (StxA) into the cytosol. StxA exhibits RNA N-glycosidase activity towards 28S rRNA, resulting in inhibition of protein synthesis in eukaryotic cells. This inhibition is associated with the ability of STEC bacteria to cause hemolytic uremic syndrome (HUS) in humans [6,7,8,9]. Even though the impact of foodborne illness on a global scale is difficult to estimate, the spread of disease through contaminated food still plays a major role in mortality, raising the importance of the standardization of rapid diagnostic methods, to minimize economic costs in terms of productivity loss, incomes, and health care [10].
Herein, we report the construction of a monoclonal scFv fragment targeting Stx1 by antibody engineering for diagnostic application. We demonstrated here the importance of testing different gene assembly and cloning strategies to obtain a functional scFv fragment, since a recombinant antibody can sometimes lose its affinity for the antigen after purification. The resulting molecule was able to bind purified antigen, and recognize toxin-producing strains using a rapid latex agglutination test, and thus it is considered a promising tool for STEC diagnosis.
2. Materials and Methods
2.1. scFvStx1(I) Gene Design, Expression and Purification
The scFvStx1(I) gene was constructed on the basis of murine hybridoma (mAb 3E2) secreting Stx1 IgG monoclonal antibodies [11], using the same protocol as described by Luz et al. [12]. The DNA encoding the scFvStx1(I) fragment was designed using the BioEdit program (www.bioedit.com) in a VH-linker-VL orientation, using a regular (Gly4Ser)3 linker type, and synthesized by GenScript (Jiangsu, China), (Figure 1). This gene was first cloned into the pAE vector through restriction enzymes BamHI/HindIII (Thermo Scientific, Waltham, MA, USA). Cloning was performed using T4 ligase (Invitrogen, Carlsbad, CA, USA), following the manufacturer’s recommendations and transformed into E. coli BL21 (DE3) (Promega, Madison, WI, USA) competent cells [13]. The recombinant vector was confirmed by plasmid sequencing. The scFvStx1(I) expression was induced by the addition of IPTG to 1 mM (Invitrogen). The purification was performed under denaturing conditions by the addition of 8 M urea by IMAC chromatography on AKTA Primeplus (GE Healthcare, Uppsala, Sweden), using a His-Trap HP Ni Sepharose column (GE Healthcare, Uppsala, Sweden). The first purified scFvStx1(I) was dialyzed to reestablish protein conformation with a Slide-A-Lyser Dialysis G2 (Thermo Scientific, USA) against decreasing concentrations of urea (5, 3, 2, 1 and 0.5 M). The purified protein was analyzed by SDS-PAGE and immunoblotting, detected by HRP-conjugated anti-His-tag monoclonal antibody (1:5000) (Sigma, St. Louis, MO, USA).
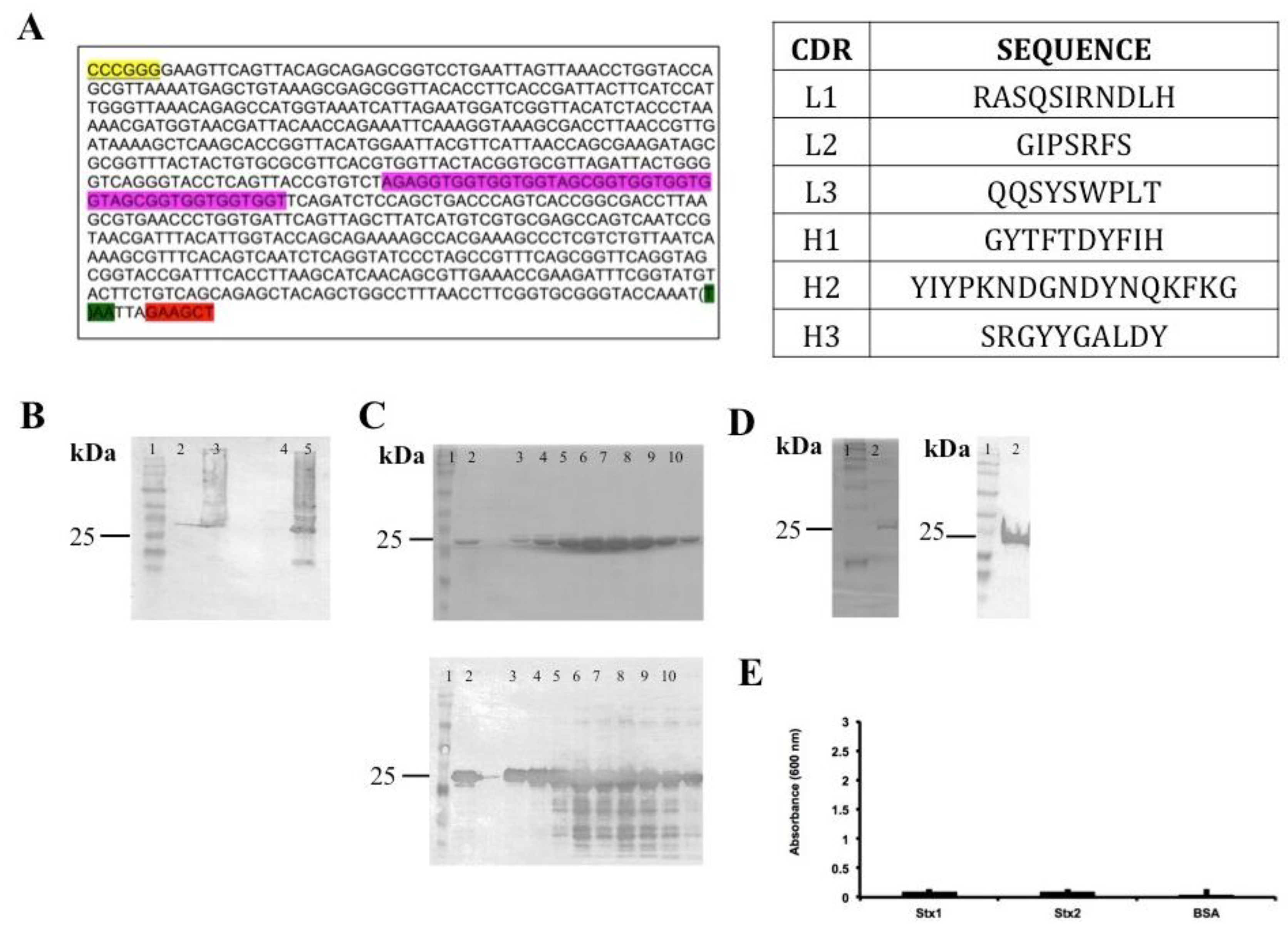
Figure 1.
Summary of scFvStx1(I) cloning and procurement. (A) Gene sequence and identification of variable chain CDR (complementarity-determining regions). Restriction enzyme BamHI site is highlighted in yellow, HindIII site is highlighted in red, linker sequence is highlighted in pink and a stop codon is highlighted in green; (B) Immunoblotting membrane with anti-6XHis-tag analysis of scFvStx1(I) recombinant protein expression induction: molecular marker (lane 1); fraction before induction (lane 2); fraction post-induction (lane 3); soluble fraction (lane 4); insoluble fraction (lane 5); (C) 12% SDS-PAGE analysis (above) of scFvStx1(I) recombinant protein affinity purification and mirror immunoblotting membrane with anti-6XHis-tag (below): molecular marker (lane 1); sample flow through (lane 2); imidazole-eluted fractions (lanes 3 to 10); (D) 12% SDS-PAGE analysis (left) of scFvStx1(I) recombinant protein after dialysis, and mirror immunoblotting membrane with anti-6XHis-tag (right): molecular marker (lane 1); post-dialysis sample (lane 2); (E) ELISA for cross-reaction employing 2 μg of each toxin and 0.2% BSA (bovine serum albumin) as control. scFvStx1 was used at 30 μg/mL concentration and the detection antibody was peroxidase-conjugated anti-His-tag (SIGMA). The assay was performed in triplicate.
2.2. scFvStx1(I) Gene Modifications
For the second scFv arrangement gene, the DNA fragments encoding the corresponding VL and VH domains were amplified from the previous synthetic gene, using as primers: VLFw (5′ CCT ATG CAT CCG ATT ACA AAG ATG ACG ATG ACA AAG GCG GTG ATA TCC AGC TGA CCC AGA G 3′), VLRv (5′ CTG CCA CCA CTA CTA CCA CTA GCG GCA GTA GTA CCC TTC AGT TCT AAT TTG GTA CC 3′), VHFw (5′ GTG GTA GTA GTG GTG GCA GTA GCA GTG GTG CCG AAG TTC AGT TAC AGC AGA GC 3′) and VHRv (5′ TTG TCG GCC GAA GAC ACG GTA ACT GAG GTA C 3′). The resulting gene was designated scFvStx1, and for this construction, the orientation was VL-Linker-VH, while the linker was also changed (Figure 2). Both scFvStx1 DNA and the pscFvHis-MBP [14] vector were double-digested with NsiI and EagI (NEB, Knowl Piece, Wilbury Way, Hitchin, UK) and purified with Qiaquick PCR purification (Qiagen, Hilden, Germany). Cloning was performed using T4 ligase (Invitrogen), following the manufacturer’s recommendations and transformed into E. coli BL21 (DE3) pLysS (Promega, Madison, WI, USA) competent cells [13]. The recombinant vector was confirmed by plasmid sequencing, and the final construction was designated pscfvStx1.
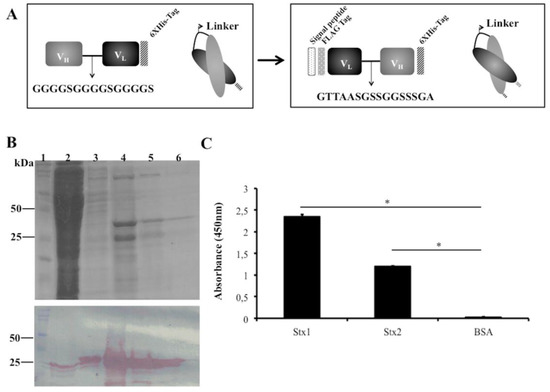
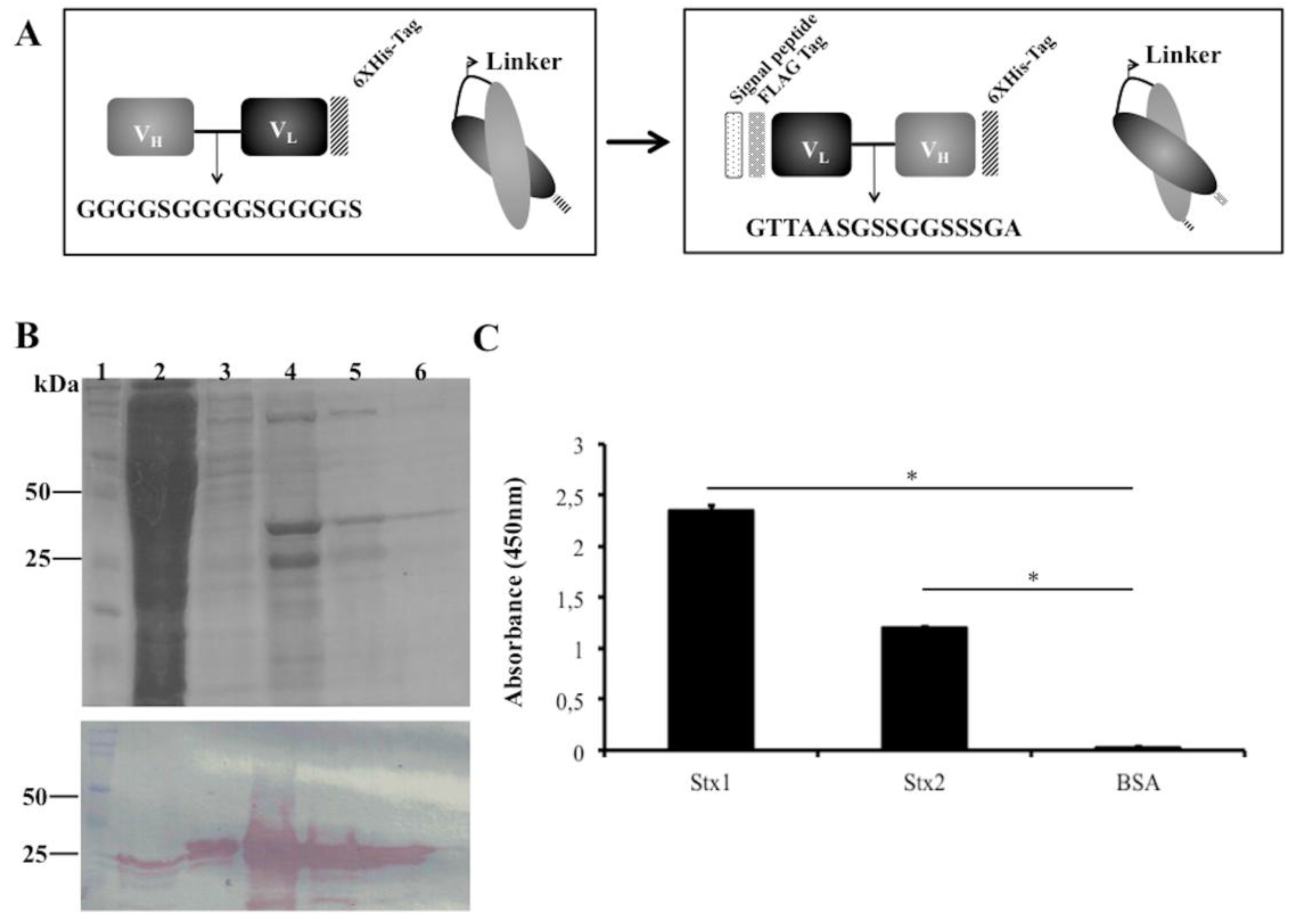
Figure 2.
scFvStx1 molecule designs and purified protein. (A) Schematic representation of scFvStx1 assembly and modifications; (B) 12% SDS-PAGE analysis of scFvStx1 recombinant fragment affinity purification. Molecular marker (lane 1); flow-through fraction (lane 2); wash fraction (lane 3); elution fractions with 20 to 40 mM imidazole (lanes 4–6) showing a 25-kDa protein. Below, immunoblotting detected by peroxidase-conjugated anti-Flag antibody (1:5000), showing detection of a 25-kDa protein; (C) ELISA for cross-reaction employing 2 μg of each toxin and 0.2% BSA as control. scFvStx1 was used at EC50 concentration and peroxidase-conjugated anti-Flag antibody (SIGMA) was used for detection. The assay was performed in triplicate and considered positive when p > 0.05 by Student’s t-test versus control (*).
2.3. scFvStx1 Fragment Procurement
The scFvStx1 fragment was obtained from pscFvStx1 construction. Gene expression and purification was performed as described by Luz et al. [12]. Briefly, gene expression was induced by the addition of IPTG to 0.01 mM, and purification performed by IMAC chromatography. The purified protein was analyzed by SDS-PAGE and immunoblotting, with detection by HRP-conjugated anti-His-tag monoclonal antibody (1:5000) (Sigma, St. Louis, MO, USA). Affinity was determined by surface plasmon resonance (BIAcore T200, GE Healthcare, Uppsala, Sweden) following the manufacturer’s recommendations. Briefly, Stx1 (purchased from Tufts University School of Medicine, Boston, MA, USA) at 5 µg/mL was immobilized in 10 mM sodium acetate buffer, pH 5.5 (152 RU) on CM5 sensor chips activated by mixing equal amounts of N-ethyl-N′-(dimethylaminopropyl) carbodiimide (EDC) and N-hydroxysuccinimide (NHS). Analyses were done using the BIAcore T200 (GE Healthcare, Uppsala, Sweden) instrument. The running buffer used was HBS-EP buffer, pH 7.4, containing 10 mM HEPES, 150 mM NaCl, 3 mM EDTA, and 0.05% Tween 20. All samples were prepared in HBS-EP buffer (0–7.4 µM, twofold dilutions), and the kinetic study was carried out in a multicycle model at 25 °C and a flow rate of 30 µL/min (contact of 120 s and dissociation of 600 s). Between cycles the sensor chip was regenerated by a 15 µL pulse of 100 mM glycine containing 2 mM MgCl2, pH 2. The kinetic affinity constant (KD) was calculated using BIAevaluation version 3.0 (GE Healthcare, Uppsala, Sweden), using the Langmuir 1:1 binding model. The experiments were performed in duplicate. For the effective binding concentration EC50, the antibody concentration required to reach half of the maximal absorbance in ELISA [12] was determined. This concentration was used to perform the cross-reactive ELISA using both Stx toxins (2 μg/mL) immobilized in a 96-well plate using 0.2% BSA as control. For ELISA assays, 0.2% BSA was used as blocking solution for 1 h at room temperature, and the same conditions were used in the following incubations, such as the scFvStx1 and the HRP-conjugated anti-His-tag monoclonal antibody (1:5000, diluted in blocking solution). Between each step, the plates were washed three times with PBS-0.05% Tween 20. Student’s t-test was used for statistical analysis, where the differences were considered statistically significant when p ≤ 0.05.
2.4. Rapid Latex Agglutination Test (RALT)
RALT was performed as described by Ristori et al. [15], with modifications. Briefly, the latex suspension was incubated with scFvStx1 (500 μg/mL) for 18 h at room temperature, followed by two blocking steps (with 0.2 M ethanolamine and with 1% BSA) for conjugation. After the blocking steps, the sample was maintained in stock buffer. As samples for agglutination tests, logarithmic phase (OD 0.5–0.8) lysates of 23 STEC strains were used as positive controls [11] and lysates of enteropathogenic E. coli, enterotoxigenic E. coli, and Shigella flexneri as negative controls. The bacterial cells were cultured in E. coli broth enriched with 0.5 ng/mL ciprofloxacin for 4 h at 37 °C, and then lysed with Triton X-100 for 1 h at 37 °C. The assay was performed on a glass slide, using a mixture of 20 μL of scFvStx1-latex and 20 μL of bacterial lysate, checking for agglutination after 1 min of gentle mixing. Three independent experiments were performed.
3. Results
The DNA encoding the variable domains of both heavy and light chains were isolated from mRNA extracted from hybridoma cells (mAb 3E2) [11] and used for scFv fragment design (Figure 1A). The first scFv design/construct was the VH-linker-VL, using (Gly4Ser)3 as the linker for the scFvStx1(I) gene. Even after several attempts, this construction resulted in inclusion body expression and non-functional protein afterwards (Figure 1).
As an alternative to increasing scFv stability during production, we changed the scFvStx1(I) design by adopting another linker (GTTAASGSSGGSSSGA), a linker identified from a phage library designed to optimize the linker between VL and VH of an anti-MBP scFv. In addition, the orientation of the variable domains was reversed as VL-linker-VH. A Ptac-driven expression vector that harbors a signal peptide sequence for periplasmic expression was used for scFv expression, the new gene was designated scFvStx1 (Figure 2A).
scFvStx1 was purified using a nickel affinity column (IMAC), yielding 2 mg/L, resulting in a 25-kDa protein on SDS-PAGE and immunoblotting (Figure 2B). The affinity constant (KD) was determined by SPR as 2.26 × 10−7 M. The half-maximal effective concentration (EC50) was determined to be 600 nM by ELISA, and this concentration was used to test cross-reactivity with the homologous toxin Stx2. scFvStx1 was able to bind to both toxins with significant difference compared to the control (Figure 2C). Compared with the parental IgG monoclonal antibody, scFvStx1 was more rapidly obtained in a bacterial system, remaining with the same yield and cross-reactivity (Table 1).

Table 1.
Comparison of scFvstx1 and IgG parental antibody. IgG characteristics were obtained from Rocha et al. (2012).
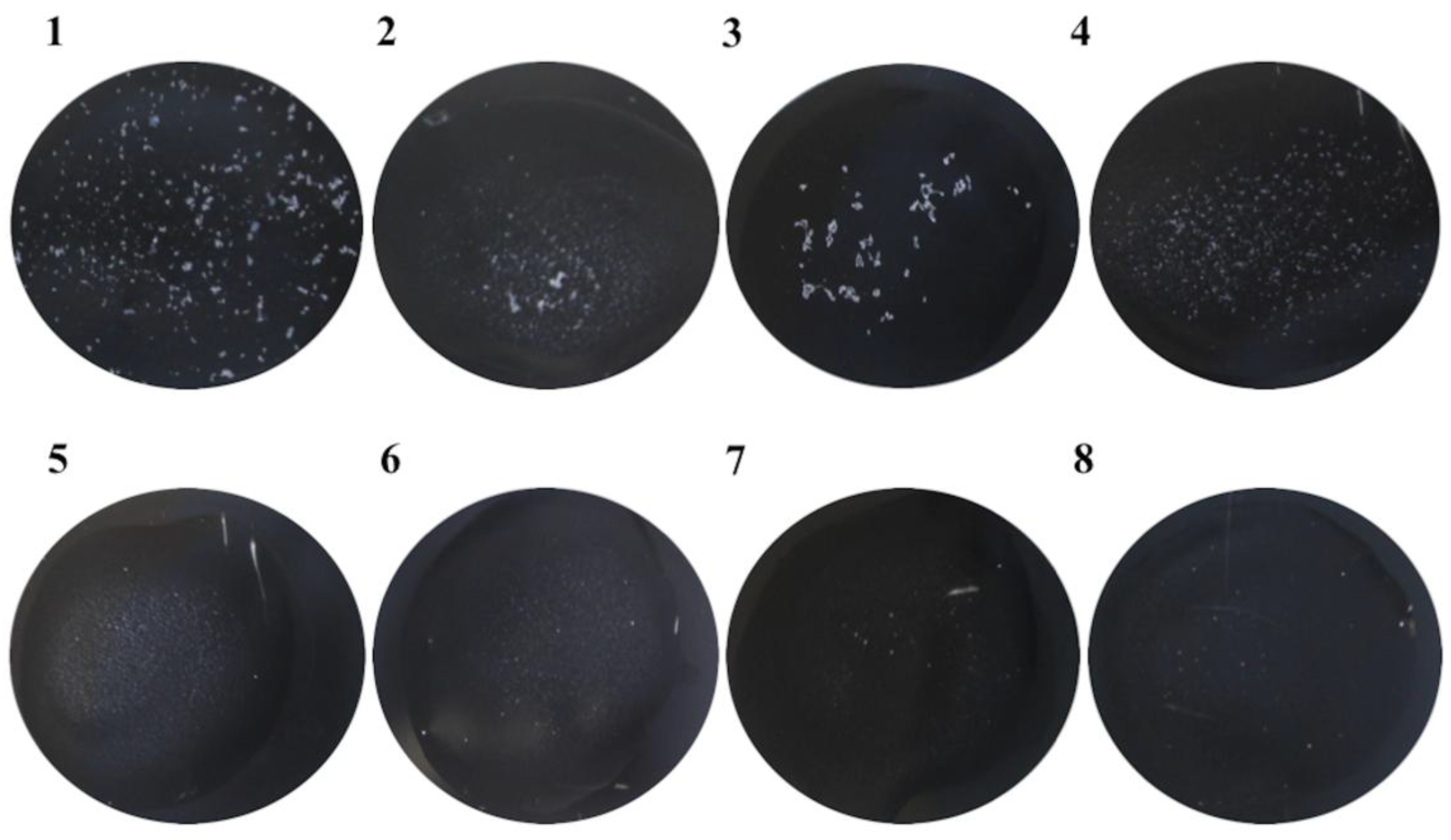
Moreover, scFvStx1 was conjugated with latex particles and employed in a rapid latex agglutination test. After a 1-min reaction, the recombinant fragment recognized 88% of the 23 Stx1-producing isolates (STEC) tested. Figure 3 shows a representative agglutination assay. In addition, the scFvStx1-latex complex was stable even after 2 months, giving the same percentage of detection.

Figure 3.
Representative example of the rapid latex agglutination test, with scFvStx1, against positive and negative isolates. The figure represents a 1-min reaction. (1) STEC O111:NM (human origin); (2) STEC O26:H11 (animal origin); (3) STEC O111:H8 (human origin); (4) STEC O157:H7 (human origin); (5) enterotoxigenic Escherichia coli (ETEC); (6) enteropathogenic Escherichia coli (EPEC); (7) Shigella flexneri; (8) E. coli broth.
4. Discussion
STECs are foodborne pathogens responsible for 90% of HUS cases [16]. The Shiga toxins produced by these strains are the main targets for detection [17]. Although commercial tests are available for Stx detection, none of them use a recombinant antibody produced in bacteria [18], which could decrease the cost of the final product, since this product is costly which makes it difficult to use in every routine laboratory. Here, we developed a recombinant scFv antibody fragment against Stx1 produced in bacteria, which has a potential for a rapid STEC diagnosis.
The most important challenges in protein engineering are to determine which factors influence recombinant protein stability and functionality [19]. The first strategy used to obtain the scFvStx1(I) fragment resulted in a protein occurring as inclusion bodies, and consequently, the purification was performed under denaturing conditions. The refolding process resulted in many aggregates, which interfered with functionality; this could have been due to the fact that under cytoplasmic reducing conditions, the intra-domain disulfide bonds of conserved antibody domains cannot form, which can interfere with the stability of purified scFv [20,21]. As reviewed by Worn [19], the highly conserved intra-domain disulfide bonds are critical for the stability of scFv fragments [22,23], where only intrinsically very stable scFv fragments will be able to fold correctly in sufficient amounts to be active as cytoplasmic intrabodies. This is also consistent with the finding that many cytoplasmic intrabodies show low expression levels and short half-lives [24].
Thus, to overcome the non-functional insoluble molecule, we made some changes in the scFvStx1(I) gene (then designated scFvStx1), in which the orientation was changed to VL-linker-VH. This arrangement allowed the CDH3 to be free at the C-terminal end, since this CDR is likely the most important contributor to antigen binding for most natural antibodies [25]. In addition, based on the fact that the linker could also interfere with molecule stability during expression or storage [26,27], the linker that connects the VL and VH domains was optimized for a common framework by phage display technology (data not shown). Compared with the classic Gly4Ser linker, this linker, GTTAASGSSGGSSSGA, was less flexible and more hydrophilic. Less flexible linkers with proline, threonine and alanine increase stability and the biological activity of recombinant proteins [27].
The scFvStx1 gene was also cloned into a more specific expression vector for scFv expression [14], which contained besides the histidine tag (6XHis) for nickel affinity chromatography, a FLAG® tag (Thermo Scientific, Waltham, MA, USA), which is composed of 8 hydrophilic amino acids (N-Y-K-N-N-N-K). FLAG® tag (Thermo Scientific, Waltham, MA, USA) is a small tag that does not interfere with protein conformation and activity, but it does increase detection sensitivity 20- to 200-fold compared with His-tag. The expression of this recombinant antibody was driven to host cell periplasm. Unlike the cytoplasm, the periplasm is an oxidative environment, and thus, it is more suitable for disulfide bond formation, which improves the stability of the molecule [28].
The new scFvStx1 was expressed with a yield of 2 mg/L, in a soluble manner, with a KD of 2.26 × 10−7 M, lower than its precursor mAb 3E2 [11]. Differences in the affinity constant of mono or dimeric antibodies are due to avidity [29]. The recombinant antibody affinity constant is usually lower compared to IgG molecules, which was previously demonstrated by our group with the scFv anti-Stx2 [12]. Moreover, the scFv purification process resulted in a less expensive and faster protocol compared to that for parental IgG (Table 1). scFv-Stx1 also showed cross-reactivity with both toxins, as observed with the scFvStx2 [12] constructed on the basis of Stx2 mAb, which showed cross-reactivity as well [11]. Additionally, scFv was able to detect Stx1-producing isolates with the rapid latex agglutination test. This test is a possible alternative for the current commercial tests.
Taken together, these results strengthen the importance of using structural and expression strategies to improve the functionality and stability of scFv molecules, which contributes to the development of scFv fragments for use in identifying the agent of a worldwide foodborne outbreak, such as STEC.
Acknowledgments
This work was supported by São Paulo Research Foundation (FAPESP—2011/12928-2). RMFP and AMM received a fellowship from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq—301302/2013-8 and 307636/2016-0, respectively). D.L. is a recipient of a FAPESP fellowship (2017/17006-2) also was recipient FAPESP and PNPD-CAPES fellowship (2010/201484; 2013/03160-9; PNPD20132071); E.A.S. is a recipient of a Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) fellowship. A. Leyva helped with English editing of the manuscript.
Author Contributions
Conceived and designed the experiments: D.L., E.A.S., G.C., A.Q.M., W.Q., M.A.S., R.M.F.P.; Performed the experiments: D.L., W.Q., E.A.S., F.B.A., T.M., B.A.C.; Analyzed the data: D.L., E.A.S., G.C., S.S.S, M.A.S., W.Q., A.M.M., R.M.F.P.; Contributed reagents/materials/analysis tools: S.S.S, L.B.R., A.M.M., R.M.F.P.; Wrote the paper: D.L., E.A.S., R.M.F.P.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Holliger, P.; Hudson, P.J. Engineered antibody fragments and the rise of single domains. Nat. Biotechnol. 2005, 23, 1126–1136. [Google Scholar] [CrossRef] [PubMed]
- Weatherill, E.E.; Cain, K.L.; Heywood, S.P.; Heywood, S.P.; Compson, J.E.; Heads, J.T.; Adams, R.; Humphreys, D.P. Towards a universal disulphide stabilised single chain Fv format: Importance of interchain disulphide bond location and vL-vH orientation. Protein Eng. Des. Sel. 2012, 25, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Jia, X.; Feng, J.; Shen, B.; Huang, Y.; Geng, S.; Sun, Y.; Wang, Y.; Li, Y.; Long, M. Molecular modeling and affinity determination of scFv antibody: Proper linker peptide enhances its activity. Ann. Biomed. Eng. 2010, 38, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Gram, H.; Marconi, L.A.; Barbas, C.F.; Collet, T.A.; Lerner, R.A.; Kang, A.S. In vitro selection and affinity maturation of antibodies from a naive combinatorial immunoglobulin library. Proc. Natl. Acad. Sci. USA 1992, 89, 3576–3580. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.Z.; Yeap, S.K.; Ali, A.M.; Ho, W.Y.; Alitheen, N.B.; Hamid, M. scFv Antibody: Principles and Clinical Application. Clin. Dev. Immunol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Melton-Celsa, A.R. Shiga toxin (Stx) classification, structure, and function. Microbiol. Spectr. 2014. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, A.; Arondel, J.; Sansonetti, P.J. Role of Shiga toxin in the pathogenesis of bacillary dysentery, studied by using a Tox-mutant of Shigella dysenteriae 1. Infect. Immun. 1988, 56, 3099–3109. [Google Scholar] [PubMed]
- Thompson, G.S.; Shimizu, H.; Homans, S.W.; Donohue-Rolfe, A. Localization of the binding site for the oligosaccharide moiety of Gb3 on verotoxin 1 using NMR residual dipolar coupling measurements. Biochemistry 2000, 39, 13153–13156. [Google Scholar] [CrossRef] [PubMed]
- Ibarra, C.; Amaral, M.M.; Palermo, M.S. Advances in pathogenesis and therapy of hemolytic uremic syndrome caused by Shiga toxin-2. IUBMB Life 2013, 65, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Anderson, K.M.; Abbott, J.; Zhao, S.; Liu, E.; Himathongkham, S. Molecular subtyping of Shiga toxin-producing Escherichia coli using a commercial repetitive sequence-based PCR assay. J. Food Prot. 2015, 78, 902–911. [Google Scholar] [CrossRef] [PubMed]
- Rocha, L.B.; Luz, D.E.; Moraes, C.T.P.; Caravelli, A.; Fernandes, I.; Guth, B.E.C.; Horton, D.S.P.Q.; Piazza, R.M.F. Interaction between Shiga toxin and monoclonal antibodies: Binding characteristics and in vitro neutralizing abilities. Toxins 2012, 4, 729–747. [Google Scholar] [CrossRef] [PubMed]
- Luz, D.; Chen, G.; Maranhão, A.Q.; Rocha, L.B.; Sidhu, S.; Piazza, R.M. Development and characterization of recombinant antibody fragments that recognize and neutralize in vitro Stx2 toxin from Shiga toxin-producing Escherichia coli. PLoS ONE 2015, 10, e0120481. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.T.; Niemela, S.L.; Miller, R.H. One-step preparation of competent Escherichia coli: Transformation and storage of bacterial cells in the same solution. Proc. Natl. Acad. Sci. USA 1989, 86, 2172–2175. [Google Scholar] [CrossRef] [PubMed]
- Persson, H.; Ye, W.; Wernimont, A.; Adams, J.J.; Koide, A.; Koide, S.; Lam, R.; Sidhu, S.S. CDR-H3 diversity is not required for antigen recognition by synthetic antibodies. J. Mol. Biol. 2013, 425, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Ristori, C.A.; Rowlands, R.E.G.; Jakabi, M.; Gelli, D.S.; Scola, M.C.G.; de Gaspari, E.N. Detecção de Vibrio cholerae O1 em ostras utilizando anticorpo monoclonal em ensaio de aglutinação. Rev. Inst. Adolfo Lutz 2006, 65, 127–132. (In Portuguese) [Google Scholar]
- Melli, L.J.; Ciocchini, A.E.; Caillava, A.J.; Vozza, N. Serogroup-specific bacterial engineered glycoproteins as novel antigenic targets for diagnosis of Shiga toxin-producing Escherichia coli-associated hemolytic uremic syndrome. J. Clin. Microbiol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Zoja, C.; Buelli, S.; Morigi, M. Shiga toxin-associated hemolytic uremic syndrome: Pathophysiology of endothelial dysfunction. Pediatr. Nephrol. 2010, 25, 2231–2240. [Google Scholar] [CrossRef] [PubMed]
- Gould, L.H.; Bopp, C.; Strockbine, N.; Atkinson, R.; Baselski, V.; Body, B.; Carey, R.; Crandall, C.; Hurd, S.; Kaplan, R.; et al. Recommendations for diagnosis of Shiga toxin producing Escherichia coli infections by clinical laboratories. MMWR Recomm. Rep. 2009, 58, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Worn, A.; Pluckthun, A. Stability engineering of antibody single-chain Fv fragments. J. Mol. Biol. 2001, 305, 989–1010. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, H.F. Molecular and cellular aspects of thiol-disulphide exchange. Adv. Enzymol. Relat. Areas Mol. Biol. 1990, 63, 69–172. [Google Scholar] [PubMed]
- Williams, A.F.; Barclay, A.N. The immunoglobulin superfamily, domains for cell surface recognition. Annu. Rev. Immunol. 1988, 6, 381–405. [Google Scholar] [CrossRef] [PubMed]
- Proba, K.; Honegger, A.; Pluckthun, A. A natural antibody missing a cysteine in VH: Consequences for thermodynamic stability and folding. J. Mol. Biol. 1997, 265, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Glockshuber, R.; Schmidt, T.; Plukthun, A. The disulphide bonds in antibody variable domains: Effects on stability, folding in vitro, and functional expression in Escherichia coli. Biocheimistry 1992, 31, 1270–1279. [Google Scholar] [CrossRef]
- Cattaneo, A.; Biocca, S. The selection of intracellular antibodies. Trends Biotechnol. 1999, 17, 115–121. [Google Scholar] [CrossRef]
- Cheng, Y.; Li, Z.; Xi, H.; Gu, T.; Yuan, R.; Chen, X.; Jiang, C.; Kong, W.; Wu, Y. A VL-linker-VH orientation dependent single chain variable antibody fragment against rabies virus G protein with enhanced neutralizing potency in vivo. Protein Pept. Lett. 2016, 23, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Gustavsson, M.; Lehtiö, J.; Denman, S.; Teeri, T.T.; Hult, K.; Martinelle, M. Stable linker peptides for a cellulose-binding domain-lipase fusion protein expressed in Pichia pastoris. Protein Eng. 2001, 14, 711–715. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.L.; Yao, X.Q.; Xue, C.; Wang, Y.; Xiong, X.H.; Liu, Z.M. Increasing the homogeneity, stability and activity of human serum albumin and interferon-alpha2b fusion protein by linker engineering. Protein Expr. Purif. 2008, 61, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Feije, M.J.; Buchner, J. Principles and engineering of antibody folding and assembly. Biochim. Biophys. Acta 2014, 1844, 2024–2031. [Google Scholar] [CrossRef] [PubMed]
- Andrade, E.V.; Freitas, S.M.; Ventura, M.M.; Maranhão, A.Q.; Brigido, M.M. Thermodynamic basis for antibody binding to Z-DNA: Comparison of a monoclonal antibody and its recombinant derivatives. Biochim. Biophys. Acta 2005, 1726, 293–301. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).