Diagnosis and Treatment of Paraneoplastic Neurologic Syndromes
Abstract
1. Introduction
1.1. Overview and History
1.2. Epidemiology
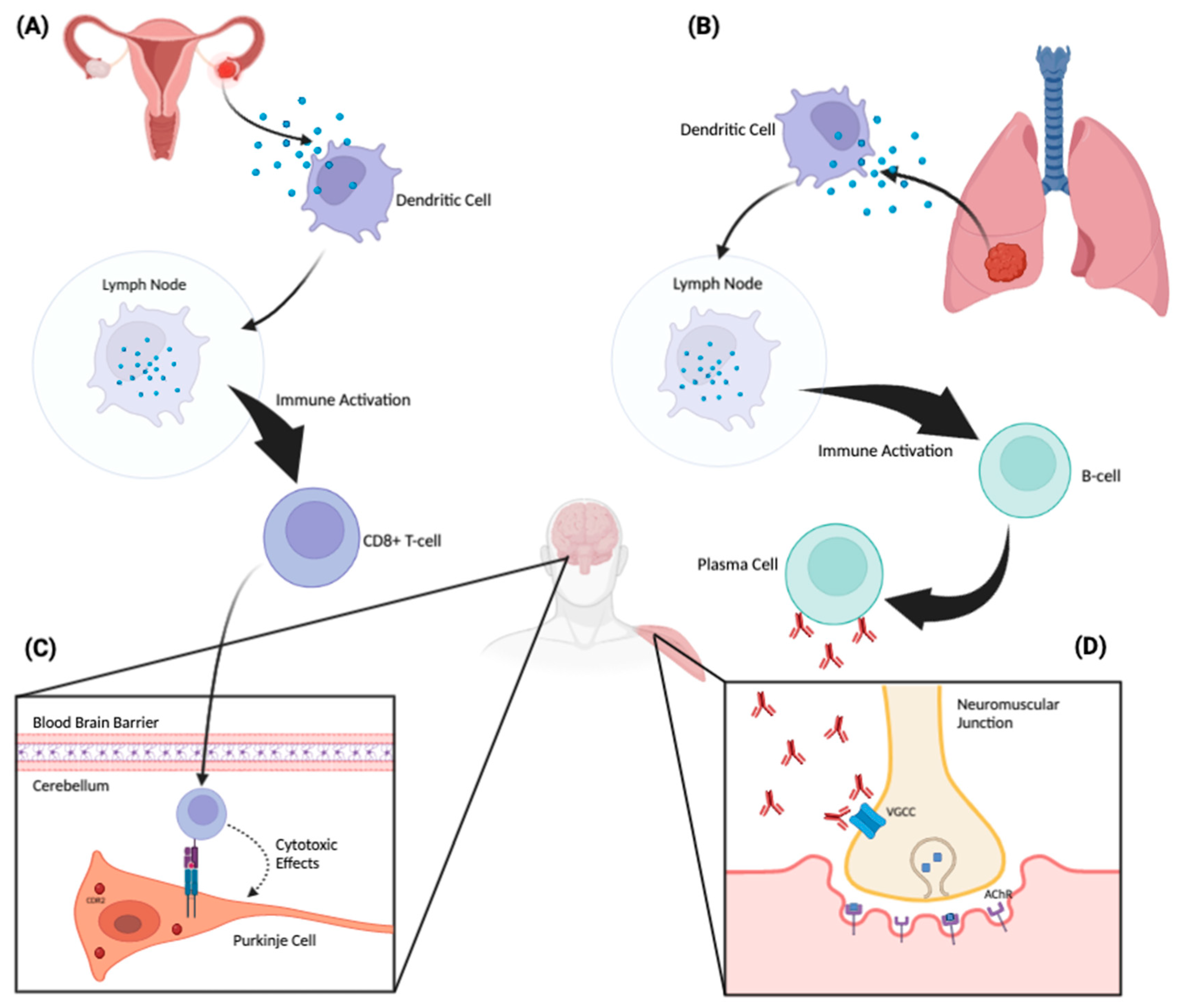
1.3. Pathophysiology
2. Approach to Diagnosis
3. Approach to the Selection of Paraneoplastic Neurologic Syndromes
3.1. Syndromes Affecting the Brain and Spinal Cord
3.1.1. Paraneoplastic Limbic Encephalitis
3.1.2. Paraneoplastic Encephalomyelitis
3.1.3. Paraneoplastic Cerebellar Degeneration
3.1.4. Paraneoplastic Opsoclonus-Myoclonus Syndrome
3.2. Syndromes Affecting Spinal Ganglia or Peripheral Nerves
3.2.1. Paraneoplastic Sensory Neuronopathy
3.2.2. Autonomic Neuropathy
3.3. Syndromes Affecting the Neuromuscular Junction (NMJ)
3.3.1. Myasthenia Gravis (MG)
3.3.2. Lambert–Eaton Myasthenic Syndrome (LEMS)
3.4. Syndromes Affecting Muscle Tissues
Paraneoplastic Myopathy
3.5. General Approach to Treatment
3.5.1. Immunosuppression
3.5.2. Prognosis
4. Conclusions/Areas of Uncertainty
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Oppenheim, H. Über Hirnsymptome bei Carcinomatose ohne nachweisbare Veränderungen im Gehirn. Charité-Annalen 1888, 13, 335–344. [Google Scholar]
- Schulz, P.; Prüss, H. “Hirnsymptome bei Carcinomatose”—Hermann Oppenheim an Early Description of a Paraneoplastic Neurological Syndrome. J. Hist. Neurosci. 2015, 24, 371–377. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Auché, M. Des névrites périphériques chez les cancéreux. Rev. Méd. 1890, 10, 785–807. [Google Scholar]
- Denny-Brown, D. Primary sensory neuropathy with muscular changes associated with carcinoma. J. Neurol. Neurosurg. Psychiatry 1948, 11, 73–87. [Google Scholar] [CrossRef]
- Wilkinson, P.C.; Zeromski, J. Immunofluorescent detection of antibodies against neurones in sensory carcinomatous neuropathy. Brain J. Neurol. 1965, 88, 529–583. [Google Scholar] [CrossRef]
- Graus, F.; Cordon-Cardo, C.; Posner, J.B. Neuronal antinuclear antibody in sensory neuronopathy from lung cancer. Neurology 1985, 35, 538–543. [Google Scholar] [CrossRef]
- Pelosof, L.C.; Gerber, D.E. Paraneoplastic syndromes: An approach to diagnosis and treatment. Mayo Clin. Proc. 2010, 85, 838–854. [Google Scholar] [CrossRef]
- Rosenfeld, M.R.; Dalmau, J. Update on Paraneoplastic Neurologic Disorders. Oncologist 2010, 15, 603–617. [Google Scholar] [CrossRef]
- Gozzard, P.; Woodhall, M.; Chapman, C.; Nibber, A.; Waters, P.; Vincent, A.; Lang, B.; Maddison, P. Paraneoplastic neurologic disorders in small cell lung carcinoma: A prospective study. Neurology 2015, 85, 235–239. [Google Scholar] [CrossRef]
- Safieddine, N.; Liu, G.; Cuningham, K.; Ming, T.; Hwang, D.; Brade, A.; Bezjak, A.; Fischer, S.; Xu, W.; Azad, S.; et al. Prognostic Factors for Cure, Recurrence and Long-Term Survival After Surgical Resection of Thymoma. J. Thorac. Oncol. 2014, 9, 1018–1022. [Google Scholar] [CrossRef]
- Rudnicki, S.A.; Dalmau, J. Paraneoplastic syndromes of the spinal cord, nerve, and muscle. Muscle Nerve 2000, 23, 1800–1818. [Google Scholar] [CrossRef] [PubMed]
- Dumitru, D.; Amato, A.A.; Zwarts, M.J. Electrodiagnostic Medicine, 2nd ed.; Hanley & Belfus: Philadelphia, PA, USA, 2002. [Google Scholar]
- Darnell, R.B.; Darnell, R.; Posner, J.B. Paraneoplastic Syndromes, Contemporary Neurology Series; Oxford University Press: Los Angeles, CA, USA, 2011. [Google Scholar]
- Darnell, R.B.; Posner, J.B. Paraneoplastic syndromes involving the nervous system. N. Engl. J. Med. 2003, 349, 1543–1554. [Google Scholar] [CrossRef] [PubMed]
- Graus, F.; Vogrig, A.; Muñiz-Castrillo, S.; Antoine, J.-C.G.; Desestret, V.; Dubey, D.; Giometto, B.; Irani, S.R.; Joubert, B.; Leypoldt, F.; et al. Updated Diagnostic Criteria for Paraneoplastic Neurologic Syndromes. Neurol.—Neuroimmunol. Neuroinflammation 2021, 8, e1014. [Google Scholar] [CrossRef] [PubMed]
- Blachère, N.E.; Orange, D.E.; Santomasso, B.D.; Doerner, J.; Foo, P.K.; Herre, M.; Fak, J.; Monette, S.; Gantman, E.C.; Frank, M.O.; et al. T cells targeting a neuronal paraneoplastic antigen mediate tumor rejection and trigger CNS autoimmunity with humoral activation. Eur. J. Immunol. 2014, 44, 3240–3251. [Google Scholar] [CrossRef]
- Dalmau, J.; Rosenfeld, M.R. Update on paraneoplastic neurologic disorders. Community Oncol. 2010, 7, 219–224. [Google Scholar] [CrossRef]
- Posner, J.B. Neurologic Complications of Cancer; F.A. Davis: Philadelphia, PA, USA, 1995. [Google Scholar]
- Graus, F.; Delattre, J.Y.; Antoine, J.C.; Dalmau, J.; Giometto, B.; Grisold, W.; Honnorat, J.; Smitt, P.S.; Vedeler, C.; Verschuuren, J.J.G.M.; et al. Recommended diagnostic criteria for paraneoplastic neurological syndromes. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1135–1140. [Google Scholar] [CrossRef]
- Binks, S.; Uy, C.; Honnorat, J.; Irani, S.R. Paraneoplastic neurological syndromes: A practical approach to diagnosis and management. Pract. Neurol. 2022, 22, 19–31. [Google Scholar] [CrossRef]
- Rosenfeld, M.R.; Dalmau, J. Paraneoplastic Neurologic Syndromes. Neurol. Clin. Neuro-Oncol. 2018, 36, 675–685. [Google Scholar] [CrossRef]
- Madhavan, A.A.; Carr, C.M.; Morris, P.P.; Flanagan, E.P.; Kotsenas, A.L.; Hunt, C.H.; Eckel, L.J.; Lindell, E.P.; Diehn, F.E. Imaging Review of Paraneoplastic Neurologic Syndromes. AJNR Am. J. Neuroradiol. 2020, 41, 2176–2187. [Google Scholar] [CrossRef]
- Dadparvar, S.; Anderson, G.S.; Bhargava, P.; Guan, L.; Reich, P.; Alavi, A.; Zhuang, H. Paraneoplastic encephalitis associated with cystic teratoma is detected by fluorodeoxyglucose positron emission tomography with negative magnetic resonance image findings. Clin. Nucl. Med. 2003, 28, 893–896. [Google Scholar] [CrossRef]
- Graus, F.; Dalmau, J. Paraneoplastic neurological syndromes. Curr. Opin. Neurol. 2012, 25, 795–801. [Google Scholar] [CrossRef]
- Muñiz-Castrillo, S.; Joubert, B.; Elsensohn, M.-H.; Pinto, A.-L.; Saint-Martin, M.; Vogrig, A.; Picard, G.; Rogemond, V.; Dubois, V.; Tamouza, R.; et al. Anti-CASPR2 clinical phenotypes correlate with HLA and immunological features. J. Neurol. Neurosurg. Psychiatry 2020, 91, 1076–1084. [Google Scholar] [CrossRef]
- Mathew, R.M.; Cohen, A.B.; Galetta, S.L.; Alavi, A.; Dalmau, J. Paraneoplastic cerebellar degeneration: Yo-expressing tumor revealed after a 5-year follow-up with FDG-PET. J. Neurol. Sci. 2006, 250, 153–155. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.R.; Subramaniam, R.M.; Mandrekar, J.N.; Hammack, J.E.; Lowe, V.J.; Jett, J.R. Occult malignancy in patients with suspected paraneoplastic neurologic syndromes: Value of positron emission tomography in diagnosis. Mayo Clin. Proc. 2008, 83, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Titulaer, M.J.; Soffietti, R.; Dalmau, J.; Gilhus, N.E.; Giometto, B.; Graus, F.; Grisold, W.; Honnorat, J.; Sillevis Smitt, P.E.; Tanasescu, R.; et al. European Federation of Neurological Societies, Screening for tumours in paraneoplastic syndromes: Report of an EFNS task force. Eur. J. Neurol. 2011, 18, 19-e3. [Google Scholar] [CrossRef] [PubMed]
- Gultekin, S.H.; Rosenfeld, M.R.; Voltz, R.; Eichen, J.; Posner, J.B.; Dalmau, J. Paraneoplastic limbic encephalitis: Neurological symptoms, immunological findings and tumour association in 50 patients. Brain J. Neurol. 2000, 123 Pt 7, 1481–1494. [Google Scholar] [CrossRef]
- Rosenfeld, M.R.; Eichen, J.G.; Wade, D.F.; Posner, J.B.; Dalmau, J. Molecular and clinical diversity in paraneoplastic immunity to Ma proteins. Ann. Neurol. 2001, 50, 339–348. [Google Scholar] [CrossRef]
- Voltz, R.; Gultekin, S.H.; Rosenfeld, M.R.; Gerstner, E.; Eichen, J.; Posner, J.B.; Dalmau, J. A Serologic Marker of Paraneoplastic Limbic and Brain-Stem Encephalitis in Patients with Testicular Cancer. N. Engl. J. Med. 1999, 340, 1788–1795. [Google Scholar] [CrossRef]
- Dalmau, J.; Graus, F.; Rosenblum, M.K.; Posner, J.B. Anti-Hu--associated paraneoplastic encephalomyelitis/sensory neuronopathy. A clinical study of 71 patients. Medicine 1992, 71, 59–72. [Google Scholar] [CrossRef]
- Lancaster, E.; Martinez-Hernandez, E.; Titulaer, M.J.; Boulos, M.; Weaver, S.; Antoine, J.-C.; Liebers, E.; Kornblum, C.; Bien, C.G.; Honnorat, J.; et al. Antibodies to metabotropic glutamate receptor 5 in the Ophelia syndrome. Neurology 2011, 77, 1698–1701. [Google Scholar] [CrossRef]
- Jitprapaikulsan, J.; Paul, P.; Thakolwiboon, S.; Mittal, S.O.; Pittock, S.J.; Dubey, D. Paraneoplastic neurological syndrome: An evolving story. Neuro-Oncol. Pract. 2021, 8, 362–374. [Google Scholar] [CrossRef] [PubMed]
- Honnorat, J.; Cartalat-Carel, S.; Ricard, D.; Camdessanche, J.P.; Carpentier, A.F.; Rogemond, V.; Chapuis, F.; Aguera, M.; Decullier, E.; Duchemin, A.M.; et al. Onco-neural antibodies and tumour type determine survival and neurological symptoms in paraneoplastic neurological syndromes with Hu or CV2/CRMP5 antibodies. J. Neurol. Neurosurg. Psychiatry 2009, 80, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Graus, F.; Keime-Guibert, F.; Reñe, R.; Benyahia, B.; Ribalta, T.; Ascaso, C.; Escaramis, G.; Delattre, J.Y. Anti-Hu-associated paraneoplastic encephalomyelitis: Analysis of 200 patients. Brain J. Neurol. 2001, 124, 1138–1148. [Google Scholar] [CrossRef] [PubMed]
- Shams’ili, S.; Grefkens, J.; de Leeuw, B.; van den Bent, M.; Hooijkaas, H.; van der Holt, B.; Vecht, C.; Sillevis Smitt, P. Paraneoplastic cerebellar degeneration associated with antineuronal antibodies: Analysis of 50 patients. Brain J. Neurol. 2003, 126, 1409–1418. [Google Scholar] [CrossRef]
- David, Y.B.; Warner, E.; Levitan, M.; Sutton, D.M.; Malkin, M.G.; Dalmau, J.O. Autoimmune paraneoplastic cerebellar degeneration in ovarian carcinoma patients treated with plasmapheresis and immunoglobulin. A case report. Cancer 1996, 78, 2153–2156. [Google Scholar] [CrossRef]
- Bhatia, P.; Heim, J.; Cornejo, P.; Kane, L.; Santiago, J.; Kruer, M.C. Opsoclonus-myoclonus-ataxia syndrome in children. J. Neurol. 2022, 269, 750–757. [Google Scholar] [CrossRef]
- Oh, S.-Y.; Kim, J.-S.; Dieterich, M. Update on opsoclonus-myoclonus syndrome in adults. J. Neurol. 2019, 266, 1541–1548. [Google Scholar] [CrossRef]
- Klaas, J.P.; Ahlskog, J.E.; Pittock, S.J.; Matsumoto, J.Y.; Aksamit, A.J.; Bartleson, J.D.; Kumar, R.; McEvoy, K.F.; McKeon, A. Adult-onset opsoclonus-myoclonus syndrome. Arch. Neurol. 2012, 69, 1598–1607. [Google Scholar] [CrossRef]
- Chalk, C.H.; Windebank, A.J.; Kimmel, D.W.; McManis, P.G. The distinctive clinical features of paraneoplastic sensory neuronopathy. Can. J. Neurol. Sci. J. Can. Sci. Neurol. 1992, 19, 346–351. [Google Scholar] [CrossRef]
- Camdessanché, J.-P.; Jousserand, G.; Ferraud, K.; Vial, C.; Petiot, P.; Honnorat, J.; Antoine, J.-C. The pattern and diagnostic criteria of sensory neuronopathy: A case-control study. Brain J. Neurol. 2009, 132, 1723–1733. [Google Scholar] [CrossRef]
- Sheikh, S.I.; Amato, A.A. The dorsal root ganglion under attack: The acquired sensory ganglionopathies. Pract. Neurol. 2010, 10, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Vernino, S.; O’Neill, B.P.; Marks, R.S.; O’Fallon, J.R.; Kimmel, D.W. Immunomodulatory treatment trial for paraneoplastic neurological disorders. Neuro-Oncology 2004, 6, 55–62. [Google Scholar] [CrossRef] [PubMed]
- McKeon, A.; Lennon, V.A.; Lachance, D.H.; Fealey, R.D.; Pittock, S.J. Ganglionic Acetylcholine Receptor Autoantibody. Arch. Neurol. 2009, 66, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Gilhus, N.E. Myasthenia Gravis. N. Engl. J. Med. 2016, 375, 2570–2581. [Google Scholar] [CrossRef]
- Nicolle, M.W. Myasthenia Gravis and Lambert-Eaton Myasthenic Syndrome. Contin. Minneap. Minn 2016, 22, 1978–2005. [Google Scholar] [CrossRef]
- Motomura, M.; Nakata, R.; Shiraishi, H. Lambert–Eaton myasthenic syndrome: Clinical review. Clin. Exp. Neuroimmunol. 2016, 7, 238–245. [Google Scholar] [CrossRef]
- Kassardjian, C.D.; Lennon, V.A.; Alfugham, N.B.; Mahler, M.; Milone, M. Clinical Features and Treatment Outcomes of Necrotizing Autoimmune Myopathy. JAMA Neurol. 2015, 72, 996–1003. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Peng, Q.; Yin, L.; Li, S.; Shi, J.; Zhang, Y.; Lu, X.; Shu, X.; Zhang, S.; Wang, G. Identification of multiple cancer-associated myositis-specific autoantibodies in idiopathic inflammatory myopathies: A large longitudinal cohort study. Arthritis Res. Ther. 2017, 19, 259. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, M.C.; Feldman, D.; Stevens, M.B. Adult onset polymyositis/dermatomyositis: An analysis of clinical and laboratory features and survival in 76 patients with a review of the literature. Semin. Arthritis Rheum. 1986, 15, 168–178. [Google Scholar] [CrossRef]
- Rotta, F.T.; Bradley, W.G. Marked improvement of severe polyneuropathy associated with multifocal osteosclerotic myeloma following surgery, radiation, and chemotherapy. Muscle Nerve 1997, 20, 1035–1037. [Google Scholar] [CrossRef]
- Titulaer, M.J.; McCracken, L.; Gabilondo, I.; Armangué, T.; Glaser, C.; Iizuka, T.; Honig, L.S.; Benseler, S.M.; Kawachi, I.; Martinez-Hernandez, E.; et al. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: An observational cohort study. Lancet Neurol. 2013, 12, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Dalmau, J.; Gleichman, A.J.; Hughes, E.G.; Rossi, J.E.; Peng, X.; Lai, M.; Dessain, S.K.; Rosenfeld, M.R.; Balice-Gordon, R.; Lynch, D.R. Anti-NMDA-receptor encephalitis: Case series and analysis of the effects of antibodies. Lancet Neurol. 2008, 7, 1091–1098. [Google Scholar] [CrossRef]
- Orange, D.; Frank, M.; Tian, S.; Dousmanis, A.; Marmur, R.; Buckley, N.; Parveen, S.; Graber, J.J.; Blachère, N.; Darnell, R.B. Cellular immune suppression in paraneoplastic neurologic syndromes targeting intracellular antigens. Arch. Neurol. 2012, 69, 1132–1140. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hiyama, E.; Yokoyama, T.; Ichikawa, T.; Hiyama, K.; Kobayashi, M.; Tanaka, Y.; Ueda, K.; Tanaka, Y.; Yano, H. Poor outcome in patients with advanced stage neuroblastoma and coincident opsomyoclonus syndrome. Cancer 1994, 74, 1821–1826. [Google Scholar] [CrossRef] [PubMed]
- Altman, A.J.; Baehner, R.L. Favorable prognosis for survival in children with coincident opso-myoclonus and neuroblastoma. Cancer 1976, 37, 846–852. [Google Scholar] [CrossRef]
- Maddison, P.; Newsom-Davis, J.; Mills, K.R.; Souhami, R.L. Favourable prognosis in Lambert-Eaton myasthenic syndrome and small-cell lung carcinoma. Lancet Lond. Engl. 1999, 353, 117–118. [Google Scholar] [CrossRef]
- Rojas, I.; Graus, F.; Keime-Guibert, F.; Reñé, R.; Delattre, J.Y.; Ramón, J.M.; Dalmau, J.; Posner, J.B. Long-term clinical outcome of paraneoplastic cerebellar degeneration and anti-Yo antibodies. Neurology 2000, 55, 713–715. [Google Scholar] [CrossRef]
- Byrne, T.; Mason, W.P.; Posner, J.B.; Dalmau, J. Spontaneous neurological improvement in anti-Hu associated encephalomyelitis. J. Neurol. Neurosurg. Psychiatry 1997, 62, 276–278. [Google Scholar] [CrossRef]
- Douglas, C.A.; Ellershaw, J. Anti-Hu antibodies may indicate a positive response to chemotherapy in paraneoplastic syndrome secondary to small cell lung cancer. Palliat. Med. 2003, 17, 638–639. [Google Scholar] [CrossRef]
- Graus, F.; Dalmou, J.; Reñé, R.; Tora, M.; Malats, N.; Verschuuren, J.J.; Cardenal, F.; Viñolas, N.; Garcia del Muro, J.; Vadell, C.; et al. Anti-Hu antibodies in patients with small-cell lung cancer: Association with complete response to therapy and improved survival. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 1997, 15, 2866–2872. [Google Scholar] [CrossRef]
- Rosenblum, M.K. Paraneoplasia and autoimmunologic injury of the nervous system: The anti-Hu syndrome. Brain Pathol. Zur. Switz. 1993, 3, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Keime-Guibert, F.; Graus, F.; Fleury, A.; René, R.; Honnorat, J.; Broet, P.; Delattre, J.Y. Treatment of paraneoplastic neurological syndromes with antineuronal antibodies (Anti-Hu, anti-Yo) with a combination of immunoglobulins, cyclophosphamide, and methylprednisolone. J. Neurol. Neurosurg. Psychiatry 2000, 68, 479–482. [Google Scholar] [CrossRef] [PubMed]
- Gresa-Arribas, N.; Titulaer, M.J.; Torrents, A.; Aguilar, E.; McCracken, L.; Leypoldt, F.; Gleichman, A.J.; Balice-Gordon, R.; Rosenfeld, M.R.; Lynch, D.; et al. Antibody titres at diagnosis and during follow-up of anti-NMDA receptor encephalitis: A retrospective study. Lancet Neurol. 2014, 13, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Furneaux, H.F.; Reich, L.; Posner, J.B. Autoantibody synthesis in the central nervous system of patients with paraneoplastic syndromes. Neurology 1990, 40, 1085–1091. [Google Scholar] [CrossRef]
| Use the History and Neurologic Exam to Define the Syndrome and Clinical Phenotype |
|---|
| Perform targeted antibody testing (in both blood and CSF). |
| Evaluate for systemic malignancy. Employ additional adjunctive testing, as needed (e.g., MRI, EEG and EMG/NCS). |
| Antibody | Antigen | Antigen Type | Associated Cancer | Syndrome(s) |
|---|---|---|---|---|
| Anti-Hu (ANNA-1) | HuD and related nuclear proteins | Intracellular | SCLC | Encephalitis, myelitis, encephalomyelitis, sensory neuronopathy, peripheral neuropathy |
| Anti-Yo (PCA-1) | CDR2 | Intracellular | Ovarian, breast | Cerebellar degeneration |
| Anti-Ri (ANNA-2) | NOVA proteins | Intracellular | Breast, ovarian, SCLC | Cerebellar ataxia, opsoclonus, brainstem encephalitis |
| Anti-Tr (DNER) | DNER | Intracellular | Hodgkin lymphoma | Cerebellar degeneration |
| Anti-CV2/CRMP5 | CRMP5 | Intracellular | SCLC | Encephalitis, myelitis, encephalomyelitis, cerebellar degeneration, optic and peripheral neuropathy |
| Anti-Ma1, Anti-Ma2 (Ta) | PNMA1, PNMA2 | Intracellular | Testicular germ cell tumors | Limbic encephalitis, brainstem encephalitis, cerebellar degeneration |
| Anti-Recoverin | Recoverin | Intracellular | SCLC, gynecologic cancer | Retinopathy |
| Anti-Hu2 (ANNA-1); Anti-Ri (ANNA-2); Others | Various | Intracellular | Neuroblastoma | Opsoclonus-myoclonus syndrome (most common pediatric paraneoplastic syndrome) |
| Anti-GAD65 | GAD65 (enzyme that synthesizes GABA) | Intracellular | Usually none | Cerebellar degeneration, Stiff person syndrome |
| Anti-Amphiphysin | Amphiphysin (synaptic antigen) | Intracellular | Breast, SCLC | Stiff person syndrome, encephalomyelitis |
| Anti-Caspr2 | Caspr2 | Intracellular | Thymoma | Neuromyotonia, encephalitis, Morvan syndrome (neuromyotonia + insomnia) |
| Anti-LGI1 | LGI1 | Intracellular | Usually none | Faciobrachial dystonic seizures, encephalitis, myoclonus |
| Anti-NMDAR | NMDAR (ionotropic Glu receptor) | Extracellular | Ovarian teratoma, testicular germ cell tumors | Limbic encephalitis |
| Anti-AMPA | AMPA (ionotropic Glu receptor) | Extracellular | Lung, breast, thymus | Limbic encephalitis |
| Anti-GABA-A | GABA-A (ionotropic inhibitory receptor) | Extracellular | Hodgkin lymphoma | Refractory status epilepticus |
| Anti-GABA-B | GABA-B (metabotropic inhibitory receptor) | Extracellular | SCLC | Limbic encephalitis with seizures, opsoclonus, ataxia |
| Anti-mGluR1 | mGluR1 (cerebellar metabotropic GluR) | Extracellular | Hodgkin lymphoma, prostate | Cerebellar degeneration |
| Anti-VGCC | VGCC at NMJ | Extracellular | SCLC | LEMS, cerebellar degeneration |
| Anti-AChR | AChR at NMJ | Extracellular | Thymoma | Myasthenia gravis, autonomic neuropathy |
| Criteria | Points |
|---|---|
| Clinical Phenotype Risk Level | |
| High-risk phenotype (syndrome often triggered by cancer) | 3 |
| Intermediate-risk phenotype (can occur with or without cancer) | 2 |
| Low-risk phenotype (weaker association with cancer) | 1 |
| Laboratory level | |
| High-risk antibody (>70% cancer association) | 3 |
| Intermediate-risk antibody (30–70% cancer association) | 2 |
| Low-risk antibody (<30% cancer association) | 0 |
| Cancer | |
| Found, consistent with phenotype and antibody | 4 |
| Not found or not consistent with phenotype, with follow-up <2 years | 1 |
| Not found, and follow up ≥2 years | 0 |
| Score | Diagnostic Level of Confidence |
| ≥8 | Definite PNS |
| 6–7 | Probable PNS |
| 4–5 | Possible PNS |
| <4 | Not PNS |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiu, D.; Rhee, J.; Gonzalez Castro, L.N. Diagnosis and Treatment of Paraneoplastic Neurologic Syndromes. Antibodies 2023, 12, 50. https://doi.org/10.3390/antib12030050
Chiu D, Rhee J, Gonzalez Castro LN. Diagnosis and Treatment of Paraneoplastic Neurologic Syndromes. Antibodies. 2023; 12(3):50. https://doi.org/10.3390/antib12030050
Chicago/Turabian StyleChiu, Daniel, John Rhee, and L. Nicolas Gonzalez Castro. 2023. "Diagnosis and Treatment of Paraneoplastic Neurologic Syndromes" Antibodies 12, no. 3: 50. https://doi.org/10.3390/antib12030050
APA StyleChiu, D., Rhee, J., & Gonzalez Castro, L. N. (2023). Diagnosis and Treatment of Paraneoplastic Neurologic Syndromes. Antibodies, 12(3), 50. https://doi.org/10.3390/antib12030050







