Ramifications of the HLA-I Allelic Reactivity of Anti-HLA-E*01:01 and Anti-HLA-E*01:03 Heavy Chain Monoclonal Antibodies in Comparison with Anti-HLA-I IgG Reactivity in Non-Alloimmunized Males, Melanoma-Vaccine Recipients, and End-Stage Renal Disease Patients
Abstract
1. Introduction
2. Material and Methods
2.1. Production of Murine Monoclonal Antibodies against HCs of HLA-ER107 and HLA-EG107
2.2. Single Antigen Beads Assay with Single-HLA Antigen-Coated Microbeads
2.3. Anti-HLA IgG Purified from the Sera of Non-Alloimmunized Males
2.4. Sera Anti-HLA IgG Antibodies in Melanoma Patients before and after Autologous Vaccine
2.5. Sera Anti-HLA IgG Antibodies in HLA-Sensitized End-Stage Kidney Disease Patients Waiting for Transplantation
3. Results
3.1. Classification of Monoclonal Antibodies Formed against HLA-E*01:01 (HLA-ER) and HLA-E*01:03 (HLA-EG)
3.2. The Monospecific mAbs Formed against HLA-E*01:01 and HLA-E*01:03
3.3. The HLA-Ib Specific but HLA-Ia Non-Reactive mAbs
3.4. Expanding Diversity of the HLA-Ia Reactivities of HLA-E*01:01 (HLA-ER) mAbs
3.4.1. HLA-B*4006 But Not HLA-A, HLA-C, HLA-F, and HLA-G Reactive (Group R-III)
3.4.2. HLA-B and HLA-C Reactive But Not HLA-A, HLA-F, or HLA-G Reactive (Group R-IV)
3.4.3. HLA-B and HLA-C Reactive but HLA-F, HLA-G Non-Reactive While Having Unique Affinity for HLA-A*1101 (Group R-V)
3.4.4. HLA-Ia Reactive but HLA-F, and HLA-G Non-Reactive (Group R-VI)
3.5. Expanding Diversity HLA-Ia Reactivity of HLA-EG mAbs
3.5.1. HLA-B Reactive but HLA-A, HLA-C, HLA-F, and HLA-G Non-Reactive (Group G-V)
3.5.2. HLA-Ia Reactive but HLA-F, and HLA-G Non-Reactive (Group G-VI)
3.6. Emergence of Highly Polyreactive mAbs against HLA-E*01:01 and HLA-E*01:03
HLA-Ib Specific mAbs Also Eact with all HLA-Ia Isoform Alleles
3.7. HLA-I Reactivity in Human Sera
3.7.1. Non-Alloimmunized Males’ Sera Reactivity to HLA-I Alleles Reflects the Profiles of Some of the HLA-E mAbs
3.7.2. Pattern of HLA-I Reactivity in Melanoma Patients before and after Immunizing with Autologous IFN-gamma Activated Tumor Cells Reflects HLA-E mAb Profiles
3.7.3. Pattern of HLA-I Reactivity of HLA-Sensitized ESKD Patients Waiting for Transplantation
4. Discussion
4.1. Immunogenicity of HLA HCs (Open Conformers) versus Intact HLA (Closed Conformers)
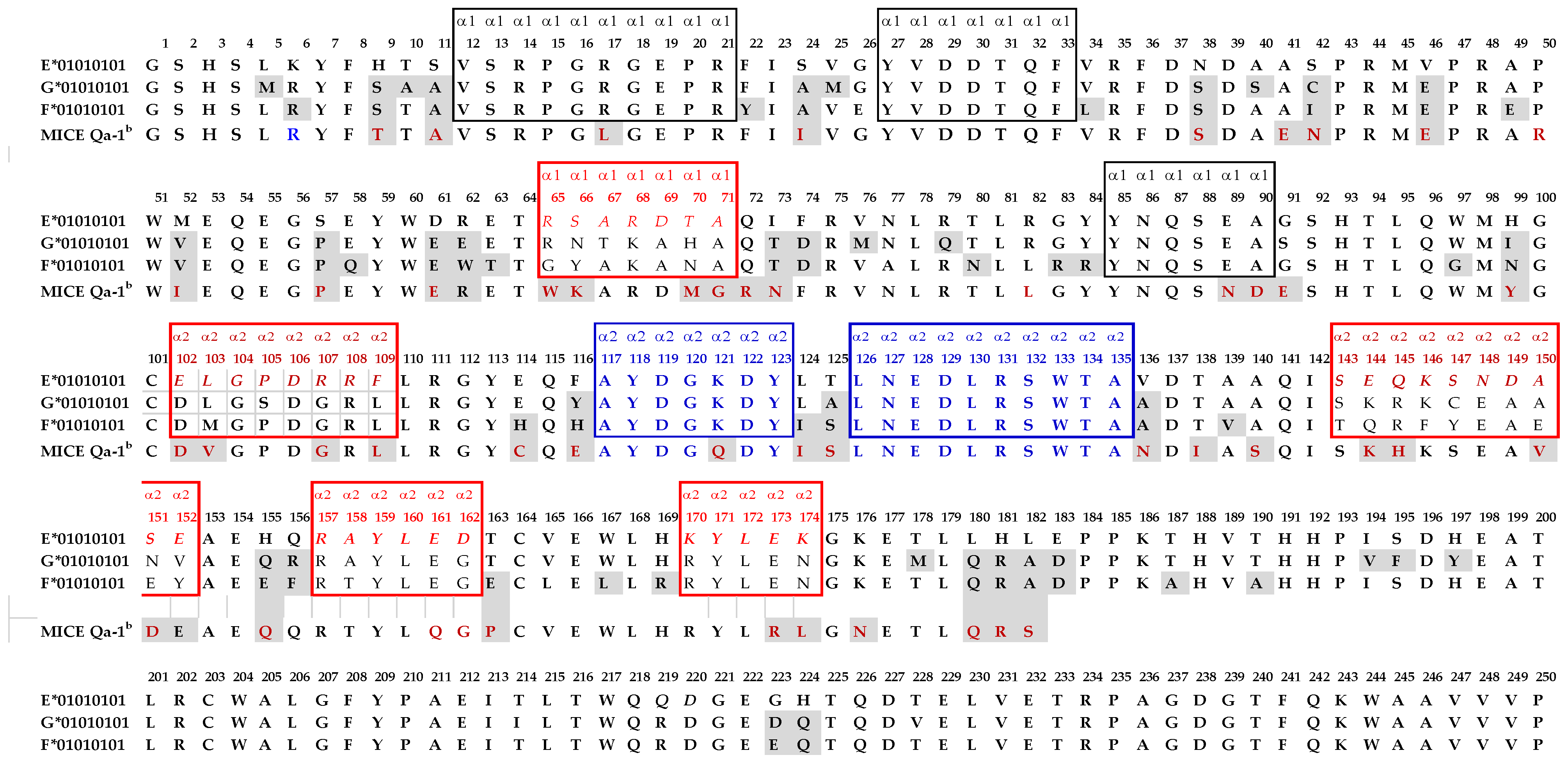
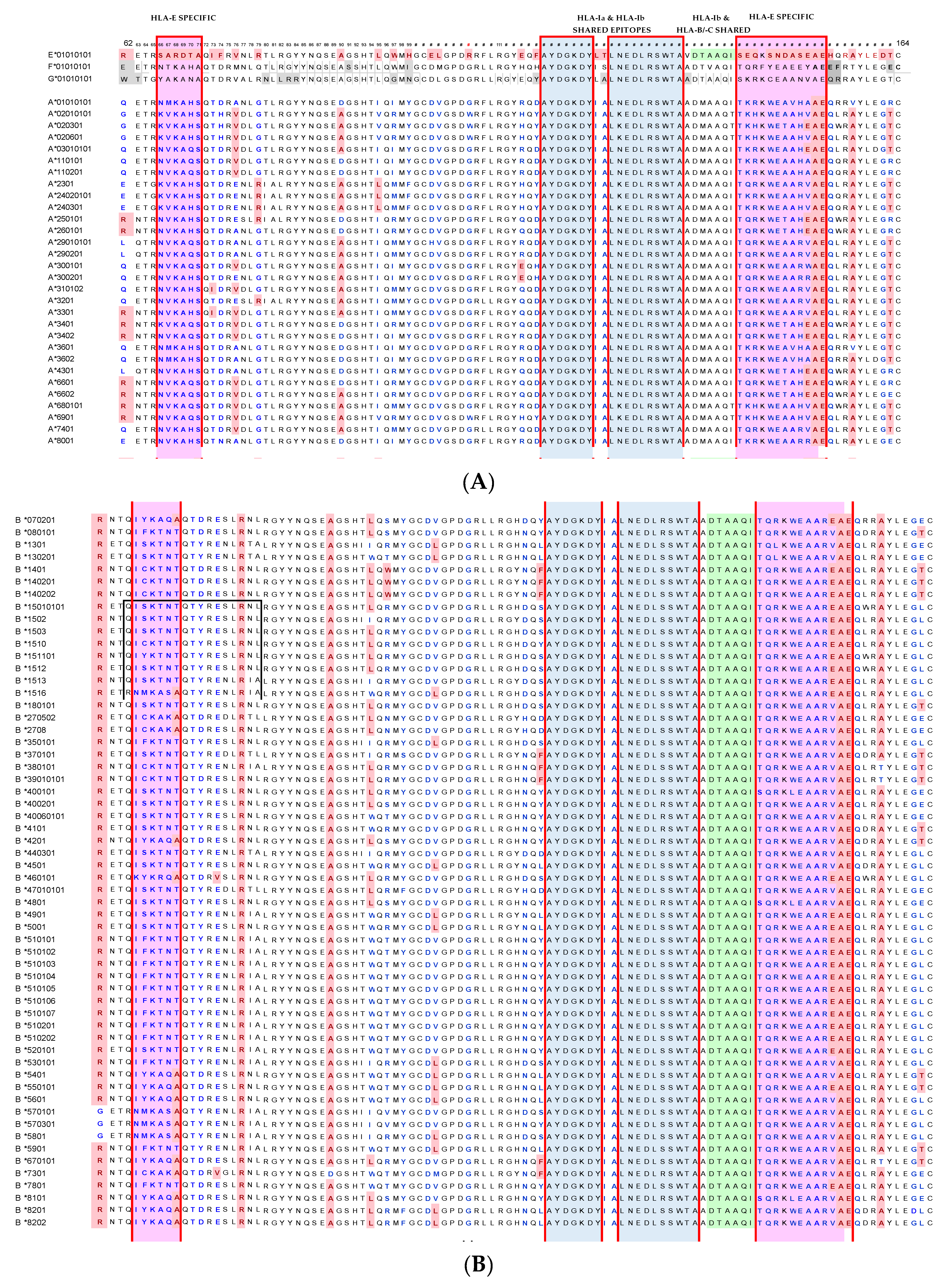
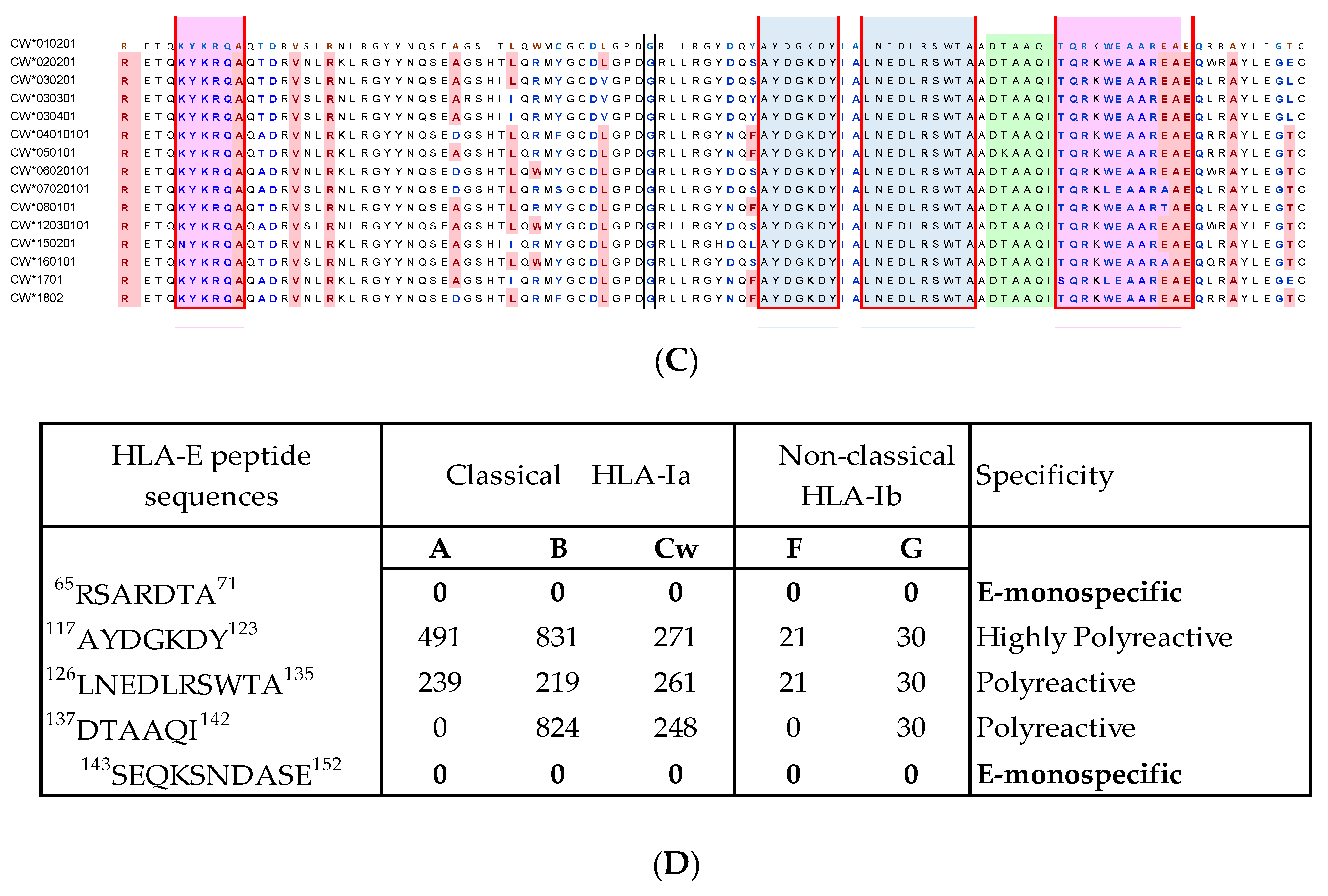
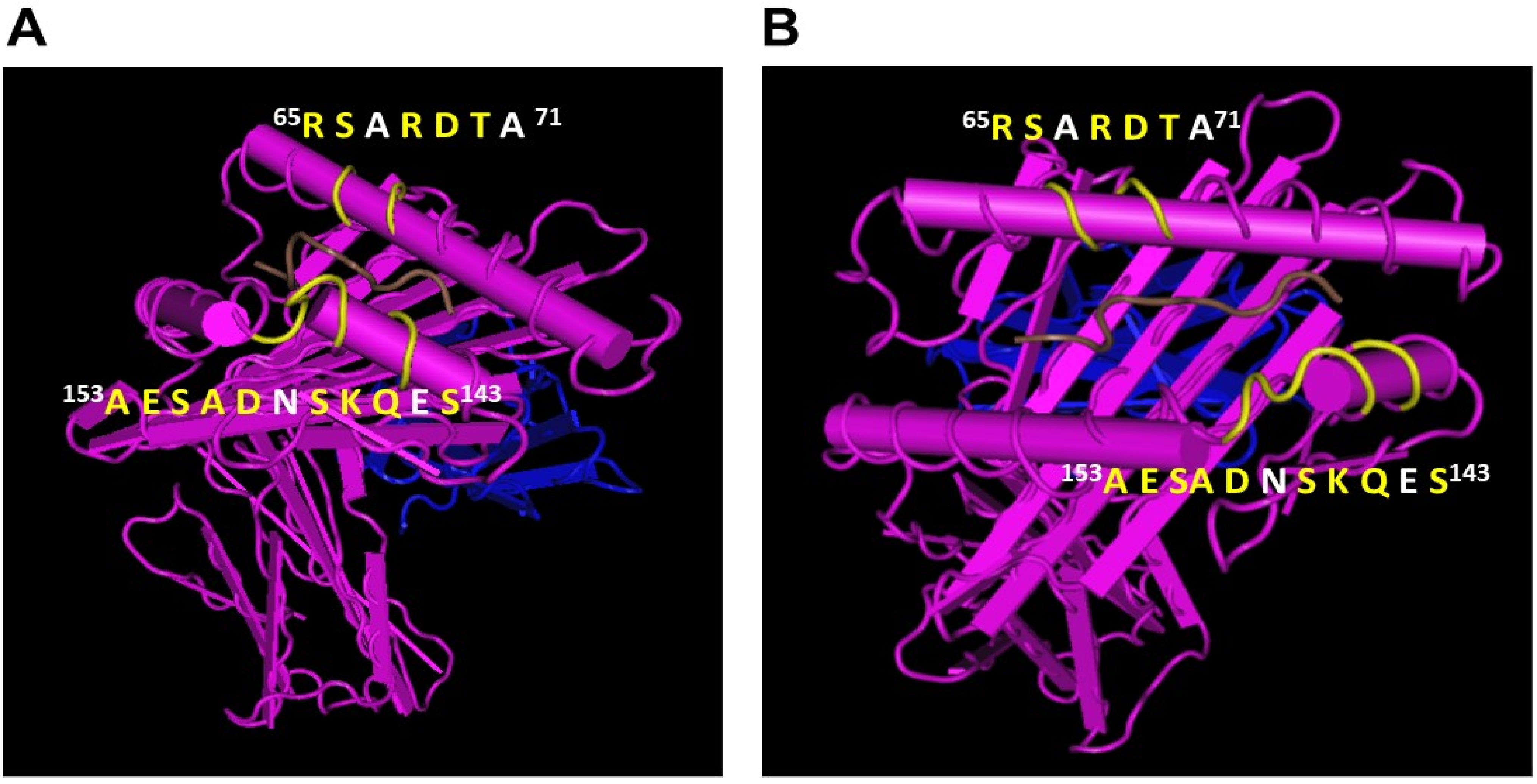
4.2. HLA-E mAbs Recognizing Private Epitopes
4.3. HLA-E mAbs Recognizing Public Epitopes
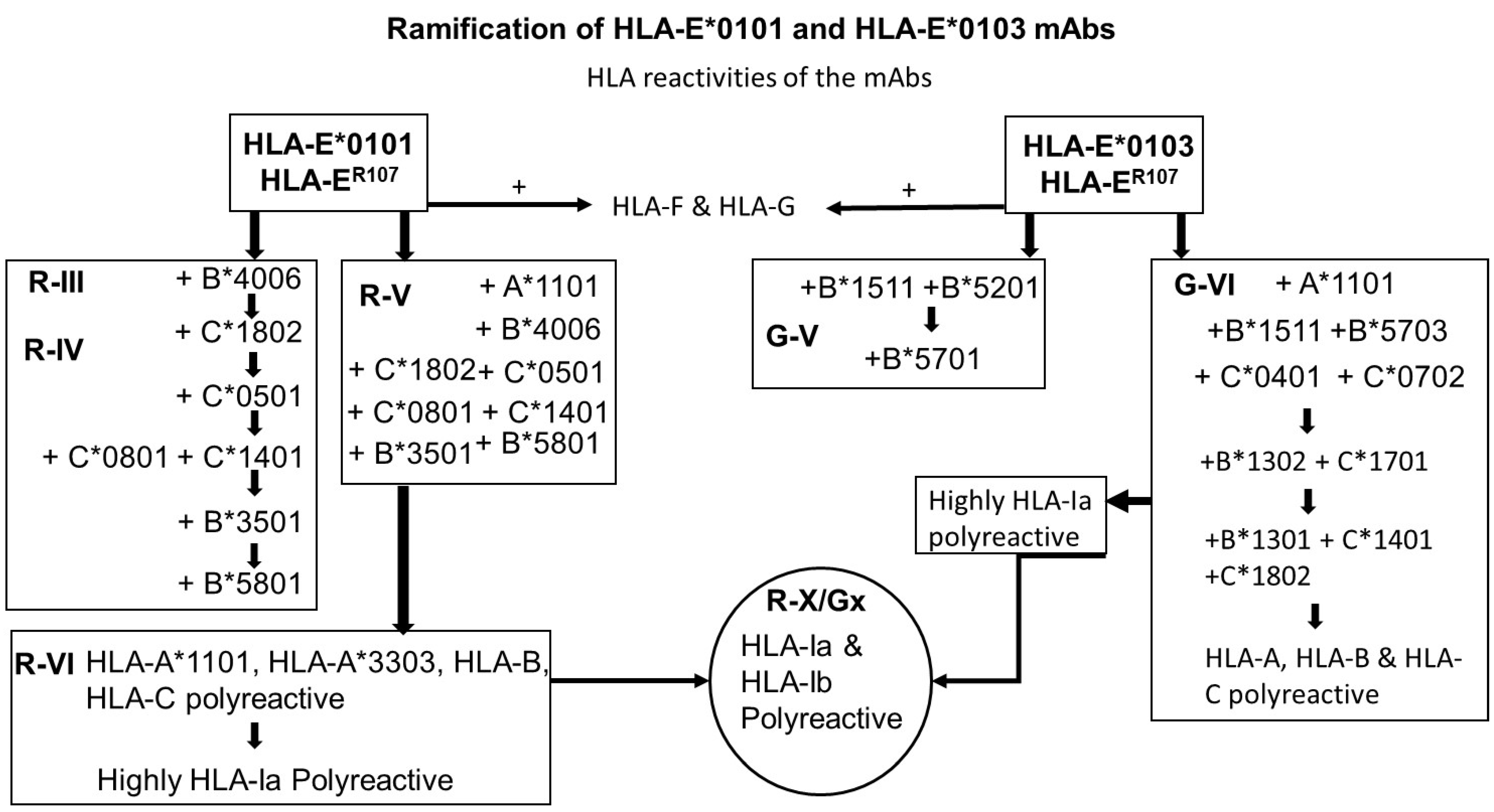
4.4. Ramifications of the HLA-I Allelic Reactivity of Anti-HLA-E*01:01 and Anti-HLA-E*01:03 mAbs
4.5. Many HLA-E mAbs May Recognize Conformationally Altered Sequences
- (a)
- The only difference in the amino acid sequences of HLA-EG107 and HLA-ER107 is the presence of glycine in the former, and arginine in the later, at position 107. Interestingly, of the total mAbs obtained after immunizing a single mouse with HLA-EG107 or two mice with HLA-ER107, eight mAbs of HLA-EG recognized HLA-F, while only one mAb of HLA-ER recognized HLA-F. While eight mAbs of HLA-EG recognized HLA-G, only one mAb of HLA-ER recognized HLA-G. These observations suggest that the presence of glycine at 107 in HLA-E may impact the conformational orientation of the HLA-E to elicit mAbs reactive to HLA-F and HLA-G.
- (b)
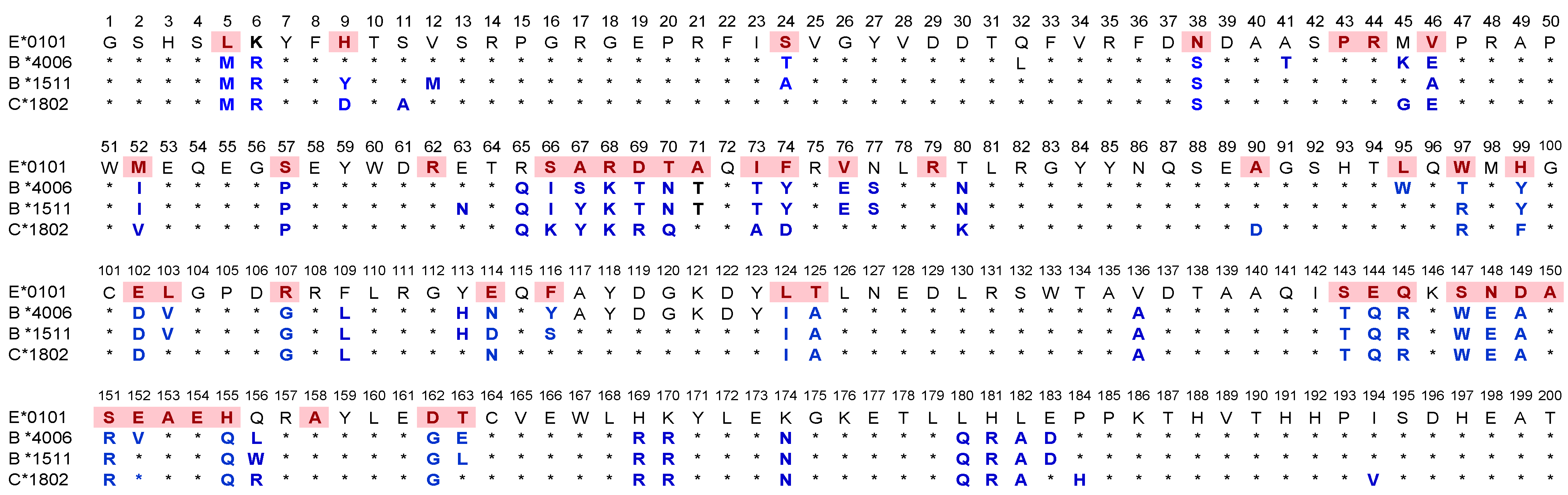
- The most striking aspect in their allelic recognition is that almost all mAbs (132 out of 133) developed against HLA-E*01:01 (HLA-ER) recognized HLA-B*4006 (Figure 6 and Figure 7). Only 17 anti- HLA-E*01:03 (HLA-EG) mAbs showed low reactivity (<4K) to B*4006. Specific recognition of B*4006 by mAbs belonging to groups R-III and R-IV (Table 4) clearly points out the unique exposure of the conformation of B*4006 comparable to HLA-ER107, that is not observed with Group G, which is possibly due to the presence of glycine at position 107.
- (c)
- Similarly, HLA-B*1511 is better recognized by the mAbs of HLA-E*01:03 (HLA-EG) than those developed from HLA-ER. The specific recognition of B*1511 by mAbs belonging to groups G-V and G-VI (Table 7) points out the unique exposure of the conformation of B*5011 is comparable to HLA-EG107, in contrast to that of HLA-ER107.
- (d)
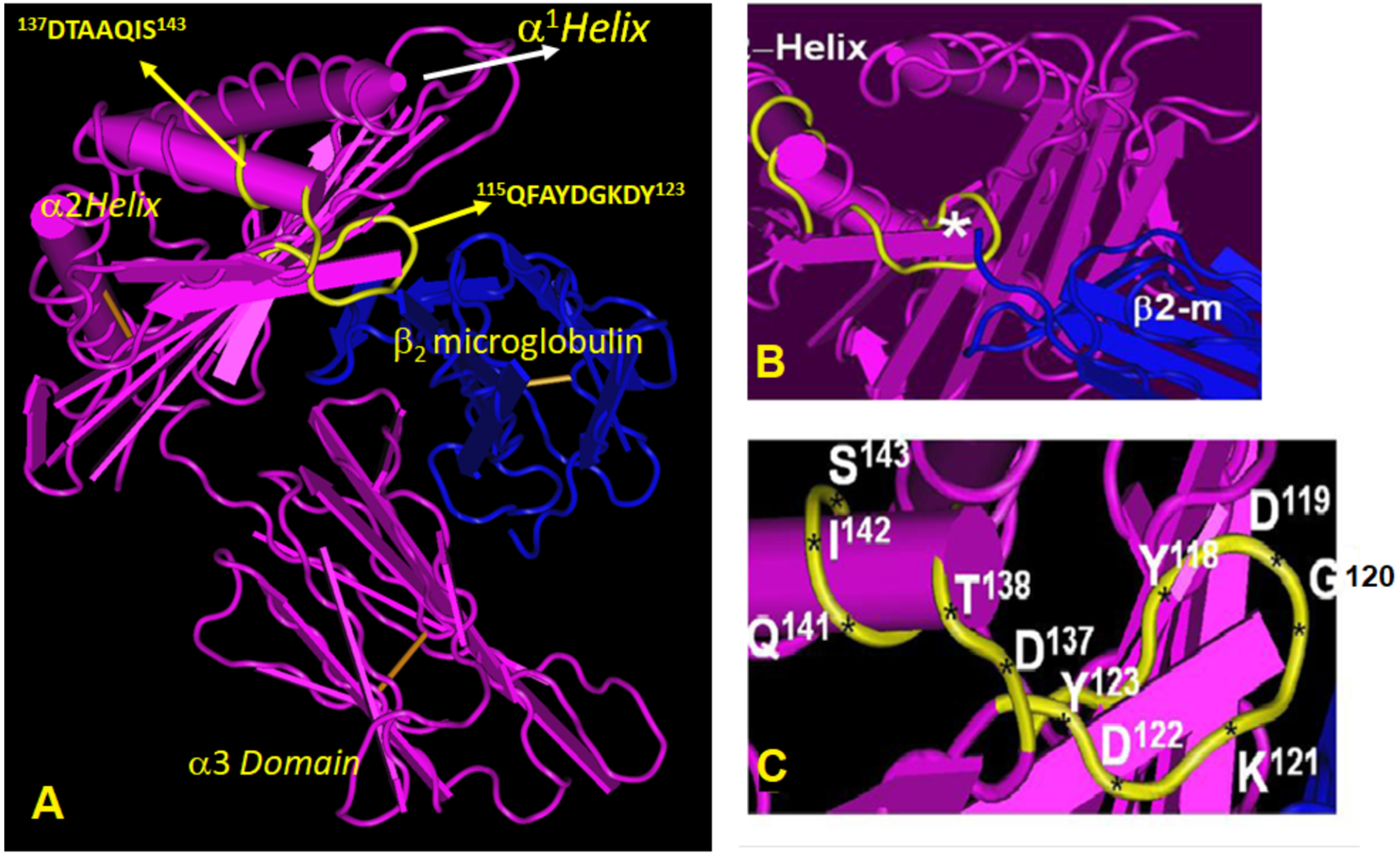
- HLA-C*1802 was recognized by all mAbs of HLA-ER, whereas many mAbs raised against HLA-EG failed to recognize the same allele. Figure 7 diagrammatically illustrates the trends of HLA-I reactivity of the anti-HLA-E*0101 and anti-HLA-E*0103 mAbs.
- (e)
- Group R-IV (Table 4) anti-HLA-E mAbs bind to HLA-B and C but do not bind to any of the HLA-A alleles, possibly due to the presence of 137DTAAQI142 found in HLA-E/-G/-B and –C), whereas in HLA-A, it is 137DTMAAQI142 (Figure 2). The T138 is replaced by M138. Figure 4 clearly shows the location of DTAAQI, in which only DT and QI are exposed during folding. Replacement of “T’ with “M” alters the immune recognition by the 30 mAbs in R-V.
- (f)
- Group R-IV mAbs do not bind to HLA-F, for DTAAQI is modified as 137DTVAQI142 in HLA-F.
- (g)
- Group R-IV mAbs also do not bind to HLA-G due to change in the closely associated neighboring amino acid of DTAAQI, as follows: in HLA-E, the amino acid preceding DTAAQI is V, whereas in HLA-F and HLA-G, the V is replaced with A (for details see Figure 1).
- (h)
- In 137DTAAQI142, the amino acid in the 136thposition and in the 143rdposition vary markedly, as seen in Figure 2. These changes may impact the exposure of the sequences. Such specific alterations can impact immune recognition by the mAbs.
4.6. Relevance of mAbs to Serum Antibodies of Normal Healthy Humans
4.7. HLA-E Polyreactive mAbs (TFL-006 and TFL007) Mimic Immunoreactivity of IVIg
4.8. Profiles of HLA-Ia Reactivity of Anti-HLA-E IgG in Melanoma Patients Reflect the HLA-I Reactivity of Some of the Anti-HLA-E mAbs
4.9. Profiles of HLA-Ia Reactivity of Sensitized Sera of ESKD Patients Reflect the HLA-I Reactivity of the Anti-HLA-E mAbs
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Townsend, A.; Bodmer, H. Antigen recognition by class I-restricted T lymphocytes. Ann. Rev. Lmmunol. 1989, 7, 601–624. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, R.; Trentini, A.; Bortolotti, D.; Manfrinato, M.C.; Rotola, A.; Castellazzi, M.; Melchiorri, L.; Di Luca, D.; Dallocchio, F.; Fainardi, E.; et al. Matrix metalloproteinase-2 (MMP-2) generates soluble HLA-G1 by cell surface proteolytic shedding. Mol. Cell. Biochem. 2013, 381, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Silva, Z.; Ferro, T.; Almeida, D.; Soares, H.; Ferreira, J.A.; Deschepper, F.M.; Hensbergen, P.J.; Pirro, M.; van Vliet, S.J.; Springer, S.; et al. MHC Class I Stability is Modulated by Cell Surface Sialylation in Human Dendritic Cells. Pharmaceutics 2020, 12, 249. [Google Scholar] [CrossRef] [PubMed]
- Ladasky, J.J.; Shum, B.P.; Canavez, F.; Seuanez, H.N.; Parham, P. Residue 3 of β2- microglobulin affects binding of class I MHC molecules by the W6/32 antibody. Immunogenetics 1999, 49, 312–320. [Google Scholar] [CrossRef]
- Giacomini, P.; Beretta, A.; Nicotra, M.R.; Ciccarelli, G.; Martayan, A.; Cerboni, C.; Lopalco, L.; Bini, D.; Delfino, L.; Ferrara, G.B.; et al. HLA-C heavy chains free of beta2-microglobulin: Distribution in normal tissues and neoplastic lesions of non-lymphoid origin and interferon-y responsiveness. Tissue Antigens 1997, 50, 555–566. [Google Scholar] [CrossRef]
- Raine, T.; Brown, D.; Bowness, P.; Gaston, J.S.H.; Moffett, A.; Trowsdale, J.; Allen, R. Consistent patterns of expression of HLA class I free heavy chains in healthy individuals and raised expression in spondyloarthropathy patients point to physiological and pathological roles. Rheumatology 2006, 45, 1338–1344. [Google Scholar] [CrossRef]
- Marozzi, A.; Meneveri, R.; Bunone, G.; De Santis, C.; Lopalco, L.; Beretta, A.; Agresti, A.; Siccardi, A.G.; Della Valle, G.; Ginelli, E. Expression of beta 2m-free HLA class I heavy chains in neuroblastoma cell lines. Scand. J. Immunol. 1993, 37, 661–667. [Google Scholar]
- Schnabl, E.; Stockinger, H.; Majdic, O.; Gaugitsch, H.; Lindley, I.J.; Maurer, D.; Hajek-Rosenmayr, A.; Knapp, W. Activated human T lymphocytes express MHC class I heavy chains not associated with beta 2-microglobulin. J. Exp. Med. 1990, 171, 1431–1442. [Google Scholar] [CrossRef]
- Madrigal, J.A.; Belich, M.P.; Benjamin, R.J.; Little, A.M.; Hildebrand, W.H.; Mann, D.L.; Parham, P. Molecular definition of a polymorphic antigen (LA45) of free HLA-A and -B heavy chains found on the surfaces of activated B and T cells. J. Exp. Med. 1991, 174, 1085–1095. [Google Scholar] [CrossRef]
- Demaria, S.; Schwab, R.; Bushkin, Y. The origin and fate of beta 2m-free MHC class I molecules induced on activated T cells. Cell. Immunol. 1992, 142, 103–113. [Google Scholar] [CrossRef]
- Pickl, W.F.; Holter, W.; Stöckl, J.; Majdic, O.; Knapp, W. Expression of beta 2-microglobulin-free HLA class I alpha-chains on activated T cells requires internalization of HLA class I heterodimers. Immunology 1996, 88, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Martayan, A.; Fiscella, M.; Setini, A.; Ciccarelli, G.; Gambari, R.; Feriotto, G.; Beretta, A.; Siccardi, A.G.; Appella, E.; Giacomini, P. Conformation and surface expression of free HLA-CW1 heavy chains in the absence of beta 2-microglobulin. Hum. Immunol. 1997, 53, 23–33. [Google Scholar] [CrossRef]
- Tsai, W.C.; Chen, C.J.; Yen, J.H.; Ou, T.T.; Tsai, J.J.; Liu, C.S.; Liu, H.W. Free HLA class I heavy chain-carrying monocytes--a potential role in the pathogenesis of spondyloarthropathies. J. Rheumatol. 2002, 29, 966–972. [Google Scholar] [PubMed]
- Goodridge, J.P.; Burian, A.; Lee, N.; Geraghty, D.E. HLA-F and MHC Class I Open Conformers Are Ligands for NK Cell Ig-like Receptors. J. Immunol. 2013, 191, 3553–3562. [Google Scholar] [CrossRef] [PubMed]
- Burian, A.; Wang, K.L.; Finton, K.A.; Lee, N.; Ishitani, A.; Strong, R.K.; Geraghty, D.E. HLA-F and MHC-I Open Conformers Bind Natural Killer Cell Ig-Like Receptor KIR3DS1. PLoS ONE 2016, 11, e0163297. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Beltran, W.F.; Hölzemer, A.; Martrus, G.; Chung, A.; Pacheco, Y.; Simoneau, C.R.; Rucevic, M.; Lamothe-Molina, P.A.; Pertel, T.; Kim, T.E.; et al. Open conformers of HLA-F are high-affinity ligands of the activating NK-cell receptor KIR3DS1. Nat. Immunol. 2016, 17, 1067–1074. [Google Scholar] [CrossRef]
- Geng, J.; Altman, J.D.; Krishnakumar, S.; Raghavan, M. Empty conformers of HLA-B preferentially bind CD8 and regulate CD8(+) T cell function. Elife 2018, 7, e36341. [Google Scholar] [CrossRef]
- Lin, A.; Yan, W.-H. The emerging roles of human leukocyte antigen-F in immune modulation and viral infection. Front. Immunol. 2019, 10, 964. [Google Scholar] [CrossRef]
- Arosa, F.A.; Esgalhado, A.J.; Reste-Ferreira, D.; Cardoso, E.M. Open MHC Class I Conformers: A Look through the Looking Glass. Int. J. Mol. Sci. 2021, 22, 9738. [Google Scholar] [CrossRef]
- Perosa, F.; Luccarelli, G.; Prete, M.; Favoino, E.; Ferrone, S.; Dammacco, F. Beta 2-microglobulin-freeHLA class Iheavy chain epitope mimicry by monoclonal antibody HC-10-specificpeptide. J. Immunol. 2003, 171, 1918–1926. [Google Scholar] [CrossRef]
- Ametani, A.; Apple, R.; Bhardwaj, V.; Gammon, G.; Miller, A.; Sercarz, E. Examining the crypticity of antigenic determinants. Cold Spring Harb. Symp. Quant. Biol. 1989, 54, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Gammon, G.; Sercarz, E.E.; Benichou, G. The dominant self and the cryptic self: Shaping the autoreactive T-cell repertoire. Immunol. Today 1991, 12, 193–195. [Google Scholar] [CrossRef]
- Sercarz, E.E.; Lehmann, P.V.; Ametani, A.; Benichou, G.; Miller, A.; Moudgil, K. Dominance and crypticity of T cell antigenic determinants. Ann. Rev. Immunol. 1993, 11, 729–766. [Google Scholar] [CrossRef]
- Tongio, M.M.; Falkenrodt, A.; Mitsuishi, Y.; Urlacher, A.; Bergerat, J.P.; North, M.L.; Mayer, S. Natural HLA Abs. Tissue Antigens 1985, 26, 271–285. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Saito, S.; Nakazawa, Y.; Kobayashi, N.; Matsuda, M.; Matsumoto, Y.; Hosoyama, T.; Koike, K. Existence of an immunoglobulin G component of naturally-occurring HLA Class I Abs that are not directed against self-Ags in human serum. Tissue Antigens 2008, 72, 98–104. [Google Scholar] [CrossRef]
- Luscher, M.A.; Choy, G.; Embree, J.E.; Nagelkerke, N.J.; Bwayo, J.J.; Njenga, S.; Plummer, F.A.; Barber, B.H.; Macdonald, K.S. Anti-HLA alloAb is found in children but does not correlate with a lack of HIV type 1 transmission from infected mothers. AIDS Res. Hum. Retrovir. 1998, 14, 99–107. [Google Scholar] [CrossRef]
- Májský, A. Natural HLA-A, B and DR Abs in the serum of nonallo-immunized men. Vnitr. Lek. 1989, 35, 467–471. [Google Scholar]
- Ravindranath, M.H.; Kaneku, H.; El-Awar, N.; Morales-Buenrostro, L.E.; Terasaki, P.I. Antibodies to HLA-E in non-alloimmunized Males: Pattern of HLA-Ia-reactivity of anti–HLA-E–positive sera. J. Immunol. 2010, 185, 1935–1948. [Google Scholar] [CrossRef]
- Morales-Buenrostro, L.E.; Terasaki, P.I.; Marino-Vázquez, L.A.; Lee, J.H.; El-Awar, N.; Alberú, J. “Natural” human leukocyte Ag Abs found in non-allo-immunized healthy males. Transplantation 2008, 86, 1111–1115. [Google Scholar] [CrossRef]
- Jucaud, V.; Ravindranath, M.H.; Terasaki, P.I. Conformational Variants of the Individual HLA-I Antigens on Luminex Single Antigen Beads Used in Monitoring HLA Antibodies: Problems and Solutions. Transplantation 2017, 101, 764–777. [Google Scholar] [CrossRef]
- Ravindranath, M.H.; Jucaud, V.; Ferrone, S. Monitoring native HLA-I trimer specific antibodies in Luminex multiplex single antigen bead assay: Evaluation of beadsets from different manufacturers. J. Immunol. Methods 2017, 450, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Ravindranath, M.H.; Taniguchi, M.; Chen, C.W.; Ozawa, M.; Kaneku, H.; El-Awar, N.; Cai, J.; Terasaki, P.I. HLA-E monoclonal antibodies recognize shared peptide sequences on classical HLA class Ia: Relevance to human natural HLA antibodies. Mol. Immunol. 2010, 47, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Ravindranath, M.H.; Pham, T.; El-Awar, N.; Kaneku, H.; Terasaki, P.I. Anti-HLA-E mAb 3D12 mimics MEM-E/02 in binding to HLA-B and HLA-C alleles: Web-tools validate the immunogenic epitopes of HLA-E recognized by the Abs. Mol. Immunol. 2011, 48, 423–430. [Google Scholar] [CrossRef]
- Ward, P.A.; Adams, J.; Faustman, D.; Gebhart, G.F.; Geistfeld, J.G.; Imbaratto, J.W.; Peterson, N.C.; Quimbey, F.; Marshak-Rothstein, A.; Rowan, A.N.; et al. Monoclonal Antibody Production; National Academy Press: Washington, DC, USA, 1999; pp. 1–47. [Google Scholar]
- Ravindranath, M.H.; Terasaki, P.I. Diagnostic and Therapeutic Potential of HLA-E Monospecific Monoclonal IgG Antibodies Directed against Tumor Cell Surface and Soluble HLA-E. U.S. Patent 10656156, 5 July 2012. [Google Scholar]
- Ravindranath, M.H.; Terasaki, P.I. Anti-HLA Class-IB Antibodies Mimic Immunoreactivity and Immunomodulatory Functions of Intravenous Immunoglobulin (IVIG) Useful as Therapeutic IVIG Mimetics and Methods of Their Use. International Patent WO 2013/106586 A2, 19 July 2013. [Google Scholar]
- Ravindranath, M.H.; Terasaki, P.I.; Pham, T.; Jucad, V. The monospecificity of novel anti-HLA-E monoclonal antibodies enables reliable immunodiagnosis, immunomodulation of HLA-E and upregulation of CD8+ T lymphocytes. Monoclon. Antibodies Immunodiag. Immunother. 2015, 34, 134–153. [Google Scholar] [CrossRef]
- Ravindranath, M.H.; Flippone, E.J.; Mahowald, G.; Callender, C.; Babu, A.; Saidman, S.; Ferrone, S. Significance of the intraindividual variability of HLA IgG antibodies in renal disease patients observed with different SABs monitored with two different secondary antibodies on a Luminex platform. Immunol. Res. 2018, 66, 584–604. [Google Scholar] [CrossRef]
- Ravindranath, M.H.; Ravindranath, N.M.; Amato-Menker, C.J. Luminex multiplex bead assay monitoring HLA IgG antibodies in sensitized pre- and post-transplant patients: Clonality of the detection antibody impacts specificity and sensitivity. Appl. Sci 2021, 11, 6430. [Google Scholar] [CrossRef]
- Selvan, S.R.; Dillman, D.O.; Fowler, A.W.; Carbonell, D.J.; Ravindranath, M.H. Monitoring response to treatment in melanoma patients: Potential of a serum glycomic marker. Int. J. Cancer 2008, 122, 1374–1383. [Google Scholar] [CrossRef]
- Ravindranath, M.H.; Selvan, S.R.; Terasaki, P.I. Augmentation of anti-HLA-E antibodies with concomitant HLA-Ia reactivity in IFNγ-treated autologous melanoma cell vaccine recipients. J. Immunotoxicol. 2012, 9, 282–291. [Google Scholar] [CrossRef]
- Derré, L.; Corvaisier, M.; Charreau, B.; Moreau, A.; Godefroy, E.; Moreau-Aubry, A.; Jotereau, F.; Gervois, N. Expression and release of HLA-E by melanoma cells and melanocytes: Potential impact on the response of cytotoxic effector cells. J. Immunol. 2006, 177, 3100–3107. [Google Scholar] [CrossRef]
- Coupel, S.; Moreau, A.; Hamidou, M.; Horejsi, V.; Soulillou, J.P.; Charreau, B. Expression and release of sHLA-E is an immunoregulatory feature of EC activation. Blood 2007, 109, 2806–2814. [Google Scholar] [CrossRef]
- Jordan, S.C.; Tyan, D.; Stablein, D.; McIntosh, M.; Rose, S.; Vo, A.; Toyoda, M.; Davis, C.; Shapiro, R.; Adey, D.; et al. Evaluation of intravenous immunoglobulin as an agent to lower allosensitization and improve transplantation in highly sensitized adult patients with end-stage renal disease: Report of the NIH IGO2 trial. J. Am. Soc. Nephrol. 2004, 15, 3256–3262. [Google Scholar] [CrossRef]
- Meier-Kriesche, H.U.; Port, F.K.; Ojo, A.O.; Rudich, S.M.; Hanson, J.A.; Cibrik, D.M.; Leichtman, A.B.; Kaplan, B. Effect of waiting time on renal transplant outcome. Kidney Int. 2000, 58, 1311–1317. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, B.; Schold, J.; Meier-Kriesche, H.U. Overview of large database analysis in renal transplantation. Am. J. Transpl. 2003, 3, 1052–1056. [Google Scholar] [CrossRef] [PubMed]
- Jordan, S.; Cunningham-Rundles, C.; McEwan, R. Utility of intravenous immune globulin in kidney transplantation: Efficacy, safety, and cost implications. Am. J. Transpl. 2003, 3, 653–664. [Google Scholar] [CrossRef]
- Arosa, F.A.; Santos, S.G.; Powis, S.J. Open conformers: The hidden Face of MHC-I molecules. Trends Immunol. 2007, 28, 115–123. [Google Scholar] [CrossRef]
- Ravindranath, M.H.; EL Hilali, F.; Filippone, E.J. The Impact of Inflammation on the Immune Responses to Transplantation: Tolerance or Rejection? Front. Immunol. 2021, 12, 667834. [Google Scholar] [CrossRef]
- Ravindranath, M.H.; Ravindranath, N.M.; Selvan, S.R.; Filippone, E.J.; Amato-Menker, C.J.; El Hilali, F. Four Faces of cell-surface HLA Class-I: Their antigenic and immunogenic divergence generating novel targets for Vaccines. Vaccines 2022, in press. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Sullivan, L.C.; Vivian, J.P.; Walpole, N.G.; Harpur, C.M.; Rossjohn, J.; Clements, C.S.; Brooks, A.G. A structural basis of antigen presentation by the MHC class Ib molecule, Qa1. J. Immunol. 2012, 188, 302–310. [Google Scholar] [CrossRef]
- Tambur, A.R.; Claas, F.H.J. HLA Epitopes as Viewed by Antibodies: What is it All About? Am. J. Transplant. 2015, 15, 1148–1154. [Google Scholar] [CrossRef] [PubMed]
- Kanevskiy, L.; Erokhina, S.; Kobyzeva, P.; Streltsova, M.; Sapozhnikov, A.; Kovalenko, E. Dimorphism of HLA-E and Its Disease Association. Int. J. Mol. Sci. 2019, 20, 5496. [Google Scholar] [CrossRef] [PubMed]
- Ravindranath, M.H.; Terasaki, P.I.; Pham, T.; Jucaud, V.; Kawakita, S. Therapeutic preparations of IVIg contain naturally occurring anti-HLA-E Abs that react with HLA-Ia (HLA-A/-B/-Cw) alleles. Blood 2013, 121, 2013–2028. [Google Scholar] [CrossRef] [PubMed]
- EL Hilali, F.; Jucaud, V.; EL Hilali, H.; Bhuiyan, M.H.; Mancuso, A.; LiuSullivan, N.; Elidrissi, A.; Mazouz, H. Characterization of the Anti-HLA Class I and II IgG Antibodies in Moroccan IVIg Using Regular Beads and Ibeads in Luminex Multiplex Single Antigen Immunoassay. Int. J. Immunol. 2017, 5, 53–65. [Google Scholar] [CrossRef][Green Version]
- Ravindranath, M.H.; EL Hilali, F.; Filippone, E.J. Therapeutic Potential of HLA-I Polyreactive mAbs Mimicking the HLA-I Polyreactivity and Immunoregulatory Functions of IVIg. Vaccines 2021, 9, 680. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Ravindranath, M.H.; Terasaki, P.I.; Miyazaki, T.; Pham, T.; Jucaud, V. Suppression of allo-human leucocyte antigen (HLA) antibodies secreted by B memory cells in vitro: Intravenous immunoglobulin (IVIg) versus a monoclonal anti-HLA-E IgG that mimics HLA-I reactivities of IVIg. Clin. Exp. Immunol. 2014, 177, 464–477. [Google Scholar] [CrossRef] [PubMed]
- Ravindranath, M.H.; Terasaki, P.I.; Pham, T.; Jucaud, V.; Kawakita, S. Suppression of blastogenesis and proliferation of activated CD4(+) T cells: Intravenous immunoglobulin (IVIg) versus novel anti-human leucocyte antigen (HLA)-E monoclonal antibodies mimicking HLA-I reactivity of IVIg. Clin. Exp. Immunol. 2014, 178, 154–177. [Google Scholar] [CrossRef] [PubMed]
| Immunogen | Groups | Reactivity of Monoclonal Antibodies with HLA-I Alleles | Number of mAbs | |||||
|---|---|---|---|---|---|---|---|---|
| HLA-Ia | HLA-Ib | |||||||
| HLA-A | HLA-B | HLA-C | HLA-E | HLA-F | HLA-G | |||
| HLA-ER107 | R-I | Negative | Negative | Negative | Positive | Negative | Negative | 20 |
| R-II | Negative | Negative | Negative | Positive | Positive | Positive | 1 | |
| R-III | Negative | B*4006+ | Negative | Positive | Negative | Negative | 1 | |
| R-IV | Negative | Positive | Positive | Positive | Negative | Negative | 30 | |
| R-V | A*1101+ | Positive | Positive | Positive | Negative | Negative | 68 | |
| R-VI | Positive | Positive | Positive | Positive | Negative | Negative | 29 | |
| R-VII | A*1101+ | Positive | Positive | Positive | Positive | Negative | 1 | |
| R-VIII | Positive | Positive | Positive | Positive | Negative | Positive | 2 | |
| R-IX | A*1101+ | Positive | Positive | Positive | Positive | Positive | 1 | |
| R-X | Positive | Positive | Positive | Positive | Positive | Positive | 2 | |
| HLA-EG107 | G-I | Negative | Negative | Negative | Positive | Negative | Negative | 5 |
| G-II | Negative | Negative | Negative | Positive | Positive | Positive | 7 | |
| G-III | Negative | Negative | Negative | Positive | Positive | Negative | 1 | |
| G-IV | Negative | Negative | Negative | Positive | Negative | Positive | 1 | |
| G-V | Negative | Positive | Negative | Positive | Negative | Negative | 4 | |
| G-VI | A*1101+ | Positive | Positive | Positive | Negative | Negative | 15 | |
| G-VII | Positive | Positive | Positive | Positive | Negative | Negative | 25 | |
| G-VIII | Positive | Positive | Positive | Positive | Positive | Negative | 10 | |
| G-IX | Positive | Positive | Positive | Positive | Negative | Positive | 16 | |
| G-X | Positive | Positive | Positive | Positive | Positive | Positive | 4 | |
| Groups | Number of mAbs | mAb Nomenclature | Hybridoma ID | Antigen (Heavy Chain only) Tested on Beads | Subclass | HLA-E Reactivity in MFI (Shorter Version of MFI) | HLA-F Reactivity (MFI) | HLA-G Reactivity (MFI) | HLA-A Reactivity (MFI) | HLA-B Reactivity (MFI) | HLA-C Reactivity (MFI) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| R-I | 1 | TFL-147 | FA5746-30F1C8 | HLA-ER | IgG1 | 22,829 (22+) | 0 | 0 | 0 | 0 | 0 |
| 2 | TFL-148 | FA5746-30F1D10 | HLA-ER | IgG1 | 16,644 (16+) | 0 | 0 | 0 | 0 | 0 | |
| 3 | TFL-110 | FT5742-14B1G2 | HLA-ER | NT | 16,226 (16) | 0 | 0 | 0 | 0 | 0 | |
| 4 | TFL-034 | FT5742-8C6F6 | HLA-ER | IgG1 | 13,272 (13) | 0 | 0 | 0 | 0 | 0 | |
| 5 | TFL-125 | FA5746-14A5E9 | HLA-ER | IgG1 | 13,204 (13) | 0 | 0 | 0 | 0 | 0 | |
| 6 | TFL-033 | FT5742-8C6F4 | HLA-ER | IgG1 | 13,025 (13) | 0 | 0 | 0 | 0 | 0 | |
| 7 | TFL-126 | FA5746-14A5F12 | HLA-ER | IgG1 | 12,397 (12) | 0 | 0 | 0 | 0 | 0 | |
| 8 | TFL-074 | FT5742-20E2E9 | HLA-ER | IgG1 | 10,269 (10) | 0 | 0 | 0 | 0 | 0 | |
| 9 | TFL-073 | FT5742-20E2G7 | HLA-ER | IgG1 | 10,088 (10) | 0 | 0 | 0 | 0 | 0 | |
| 10 | TFL-144 | FA5746-28D10C6 | HLA-ER | IgG1 | 9180 (9) | 0 | 0 | 0 | 0 | 0 | |
| 11 | TFL-041 | FT5742-11B4A2 | HLA-ER | IgG1 | 8914 (8+) | 0 | 0 | 0 | 0 | 0 | |
| 12 | TFL-043 | FT5742-11B7F12 | HLA-ER | IgG1 | 8448 (8) | 0 | 0 | 0 | 0 | 0 | |
| 13 | TFL-145 | FA5746-29B1C10 | HLA-ER | IgG1 | 7622 (7+) | 0 | 0 | 0 | 0 | 0 | |
| 14 | TFL-042 | FT5742-11B4A6 | HLA-ER | IgG1 | 7452 (7) | 0 | 0 | 0 | 0 | 0 | |
| 15 | TFL-182 | FA5746-32D10D2 | HLA-ER | IgG1 | 7280 (7) | 0 | 0 | 0 | 0 | 0 | |
| 16 | TFL-001 | FT5742-1A1E2 | HLA-ER | IgG2a | 4691 (4+) | 0 | 0 | 0 | 0 | 0 | |
| 17 | TFL-081 | FT5742-23F8C12 | HLA-ER | IgG1 | 4261 (4) | 0 | 0 | 0 | 0 | 0 | |
| 18 | TFL-016 | FT5742-4F8F12 | HLA-ER | IgG2a | 1349 (1) | 0 | 0 | 0 | 0 | 0 | |
| 19 | TFL-047 | FT5742-12B4E10 | HLA-ER | NK | 1044 (1) | 0 | 0 | 0 | 0 | 0 | |
| 20 | TFL-013 | FT5742-4B3D8 | HLA-ER | IgG2a | 943 (0+) | 0 | 0 | 0 | 0 | 0 | |
| G-I | 1 | TFL-185 | FA5850-1E1E6 | HLA-EG | IgG1 | 19,644 (19+) | 0 | 0 | 0 | 0 | 0 |
| 2 | TFL-184 | FA5850-1E1C2 | HLA-EG | IgG1 | 19,227 (19) | 0 | 0 | 0 | 0 | 0 | |
| 3 | TFL-186 | FA5850-1E1C3 | HLA-EG | IgG1 | 19,075 (19) | 0 | 0 | 0 | 0 | 0 | |
| 4 | TFL-226 | FA5850-9C3B4 | HLA-EG HLA-ER | IgG1 | 18,865 (18+) 18,979 (18+) | 0 | 0 | 0 | 0 | 0 | |
| 5 | TFL-254 | FA5850-14C7F1 | HLA-EG HLA-ER | IgG1 | 1604 (1+) 1972 (1+) | 0 | 0 | 0 | 0 | 0 |
| Groups | Subgroups | Number of mAbs | mAb Nomenclature | Hybridoma ID | Antigen (Heavy Chain only) Tested on Beads | Subclass | HLA-E Reactivity in MFI | HLA-F Reactivity (MFI) | HLA-G Reactivity (MFI) | HLA-A Reactivity (MFI) | HLA-B Reactivity (MFI) | HLA-C Reactivity (MFI) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R-II | 1 | TFL-050 | FT5742-12B6F3 | HLA-ER | IgG2b | 4876 (4+) | 3292 (3) | 2683 (2+) | 0 | 0 | 0 | |
| G-II | 1 | TFL-209 | FA5850-5C4F3 | HLA-EG HLA-ER | IgG1 | 21,532 (21+) 21,452 (21) | 8744 (8+) | 20,303 (20) | 0 | 0 | 0 | |
| 2 | TFL-208 | FA5850-5C4E2 | HLA-EG HLA-ER | IgG1 | 21,294 (21) 21,030 (21) | 8605 (8+) | 20,562 (20+) | 0 | 0 | 0 | ||
| 3 | TFL-223 | FA5850-8D3A4 | HLA-EG HLA-ER | IgG1 | 18,447 (18) 21,086 (21) | 8587 (8+) | 20,587 (20+) | 0 | 0 | 0 | ||
| 4 | TFL-164 | FT5765-1C1B6 | HLA-EG | IgG2b | 15,370 (15) | 8230 (8) | 24,765 (24+) | 0 | 0 | 0 | ||
| 5 | TFL-165 | FT5765-1C1G5 | HLA-EG | IgG2b | 15,192 (15) | 8254 (8) | 25,812 (25+) | 0 | 0 | 0 | ||
| 6 | TFL-162 | FT5765-1C1B4 | HLA-EG | IgG2b | 14,667 (14+) | 9015 (9) | 25,085 (25) | 0 | 0 | 0 | ||
| 7 | TFL-161 | FT5765-1C1B1 | HLA-EG | IgG2b | 14,444 (14) | 8607 (8+) | 24,714 (24+) | 0 | 0 | 0 | ||
| G-III | 1 | TFL-228 | FA5850-9C3B3 | HLA-EG | IgG1 | 19,581 (19+) 19,919 (19+) | 1134 (1) | 0 | 0 | 0 | 0 | |
| G-IV | 1 | TFL-191 | FA5850-2C8C10 | HLA-EG | NK | 1000 (1) | 0 | 1016 (1) | 0 | 0 | 0 |
| MFI Expressed in Thousands | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Groups | Subgroups | number of mAbs | mAb Nomenclature | Subclass | HLA-E Reactivity (MFI) | HLA-F Reactivity (MFI) | HLA-G Reactivity (MFI) | HLA-A Reactivity (MFI) | HLA-B Reactivity | HLA-C Reactivity | ||||||||||||||||||||||
| B*0801 | B*1301 | B*1302 | B*1401 | B*1501 | B*1502 | B*1511 | B*1512 | B*1513 | B*1516 | B*3501 | B*3701 | B*4006 | B*4601 | B*5701 | B*5703 | B*5801 | C*0202 | C.*0401 | C*0501 | C*0602 | C*0702 | C*0801 | C*1802 | |||||||||
| R-III | 1 | TFL-114 | IgG2a | 14+ | 1 | |||||||||||||||||||||||||||
| R-IV | [A] | 1 | TFL-004 | IgG2a | 17+ | 1 | 1 | |||||||||||||||||||||||||
| 2 | TFL-005 | IgG2a | 17 | 1 | 1 | |||||||||||||||||||||||||||
| 3 | TFL-019 | IgG1 | 13+ | 1+ | 1+ | |||||||||||||||||||||||||||
| 4 | TFL-020 | IgG1 | 12+ | 1 | 1+ | |||||||||||||||||||||||||||
| 5 | TFL-099 | IgG2a | 14 | 1+ | 1+ | |||||||||||||||||||||||||||
| 6 | TFL-108 | IgG2a | 11 | 1 | 1 | |||||||||||||||||||||||||||
| 7 | TFL-128 | IgG3 | 13 | 1 | 1 | |||||||||||||||||||||||||||
| 8 | TFL-146 | IgG1 | 13 | 1 | 1 | |||||||||||||||||||||||||||
| 9 | TFL-084 | IgG2a | 16 | 2 | 1+ | |||||||||||||||||||||||||||
| 10 | TFL-028 | IgG1 | 12+ | 2 | 2 | |||||||||||||||||||||||||||
| [B] | 11 | TFL-116 | IgG1 | 8+ | 1+ | 1 | 2 | |||||||||||||||||||||||||
| 12 | TFL-156 | IgG1 | 12+ | 2 | 1 | 2+ | ||||||||||||||||||||||||||
| 13 | TFL-029 | IgG1 | 13+ | 2+ | 1 | 2+ | ||||||||||||||||||||||||||
| 14 | TFL-030 | IgG2a | 15 | 2+ | 1 | 2+ | ||||||||||||||||||||||||||
| 15 | TFL-060 | IgG2a | 18+ | 2+ | 1 | 3 | ||||||||||||||||||||||||||
| 16 | TFL-154 | IgG2b | 19+ | 3 | 1 | 3 | ||||||||||||||||||||||||||
| [C] | 17 | TFL-158 | IgG2b | 19 | 1 | 4 | 1+ | 1 | 6 | |||||||||||||||||||||||
| 18 | TFL-059 | IgG2a | 19+ | 1 | 1 | 3+ | 1+ | 4 | ||||||||||||||||||||||||
| 19 | TFL-079 | IgG1 | 11+ | 1 | 3+ | 1 | 1+ | 1 | 4 | |||||||||||||||||||||||
| 20 | TFL-093 | IgG1 | 13+ | 1+ | 1 | 1 | 1 | |||||||||||||||||||||||||
| 21 | TFL-143 | IgG2b | 20+ | 1 | 1 | 4+ | 1 | 1 | 5 | |||||||||||||||||||||||
| [D] | 22 | TFL-090 | IgG1 | 12 | 1 | 3 | 1 | 2 | 1 | 4 | ||||||||||||||||||||||
| 23 | TFL-052 | IgG2b | 10+ | 1+ | 1 | 2+ | 1 | 1+ | 1 | 3+ | ||||||||||||||||||||||
| 24 | TFL-062 | IgG1 | 15 | 1 | 1 | 3+ | 1 | 2 | 1 | 4 | ||||||||||||||||||||||
| 25 | TFL-076 | IgG2b | 20+ | 1 | 1 | 4 | 1 | 1+ | 1 | 4+ | ||||||||||||||||||||||
| 26 | TFL-076 | IgG2b | 20+ | 1 | 1 | 4 | 1 | 1+ | 1 | 4+ | ||||||||||||||||||||||
| 27 | TFL-091 | IgG1 | 16+ | 1 | 1 | 4 | 1 | 1+ | 1 | 5 | ||||||||||||||||||||||
| 28 | TFL-159 | IgG2b | 19+ | 1+ | 1 | 5 | 1 | 2 | 1 | 1+ | 7 | |||||||||||||||||||||
| [E] | 29 | TFL-155 | IgG2b | 19 | 8+ | 7+ | 9 | 9+ | 17 | 4+ | 15 | 16+ | 2+ | 16+ | 6+ | 8 | 3 | |||||||||||||||
| 30 | TFL-094 | IgG2b | 18+ | 12+ | 8 | 9 | 10+ | 18 | 5 | 16+ | 17+ | 2 | 17 | 6 | 8 | 2+ | ||||||||||||||||
| MFI expressed in Thousands | |||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Groups | Subgroups | number of mAbs | mAb Nomenclature | Subclass | HLA-E Reactivity (MFI) | HLA-F Reactivity (MFI) | HLA-G Reactivity (MFI) | HLA-A*1101 Reactivity (MFI) | HLA-B Reactivity | HLA-C Reactivity | |||||||||||||||||||||||||||||||||||||
| B*1301 | B*1302 | B*1401 | B*1501 | B*1502 | B*1511 | B*1512 | B*1513 | B*1516 | B*1801 | B*3501 | B*3701 | B*3901 | B*4001 | B*4002 | B*4006 | B*4101 | B*4403 | B*4501 | B*4601 | B*4701 | B*5301 | B*5401 | B*5701 | B*5703 | B*5801 | B*7801 | B*8201 | C*0202 | C*0303 | C*0304 | C*0501 | C*0602 | C*0801 | C*1402 | C*1502 | C*1601 | C*1701 | C*1802 | |||||||||
| R-V | [A] | 1 | TFL-035 | IgG1 | 11 | 1 | 1 | 1 | 3+ | 1 | 2 | 1+ | 4+ | ||||||||||||||||||||||||||||||||||
| 2 | TFL-064 | IgG2b | 21 | 1 | 1 | 1 | 4+ | 1 | 1 | 4 | |||||||||||||||||||||||||||||||||||||
| 3 | TFL-134 | IgG1 | 15+ | 1 | 1+ | 1 | 4 | 1 | 1+ | 1 | 5 | ||||||||||||||||||||||||||||||||||||
| 4 | TFL-075 | IgG1 | 16 | 1 | 1+ | 1 | 4+ | 1 | 2 | 1 | 5 | ||||||||||||||||||||||||||||||||||||
| 5 | TFL-011 | IgG2a | 17 | 1 | 1 | 1+ | 4 | 1 | 2 | 1 | 1 | ||||||||||||||||||||||||||||||||||||
| 6 | TFL-061 | IgG1 | 15 | 1 | 1+ | 1+ | 4+ | 1+ | 2+ | 1+ | 5 | ||||||||||||||||||||||||||||||||||||
| 7 | TFL-077 | IgG2b | 21+ | 1 | 1+ | 1+ | 6 | 1+ | 2+ | 1 | 6 | ||||||||||||||||||||||||||||||||||||
| [B] | 8 | TFL-036 | IgG1 | 11+ | 1 | 1 | 1 | 4 | 1 | 1 | 2 | 1+ | 4+ | ||||||||||||||||||||||||||||||||||
| 9 | TFL-078 | IgG1 | 12+ | 1 | 1 | 1 | 4 | 1 | 1 | 2+ | 1+ | 5 | |||||||||||||||||||||||||||||||||||
| 10 | TFL-096 | IgG1 | 12+ | 1 | 1+ | 1 | 3+ | 1 | 1 | 2 | 1+ | 4 | |||||||||||||||||||||||||||||||||||
| 11 | TFL-012 | IgG2a | 17+ | 1 | 1 | 1+ | 4+ | 1 | 1+ | 2 | 1+ | 4+ | |||||||||||||||||||||||||||||||||||
| 12 | TFL-045 | IgG2a | 16+ | 1+ | 1 | 1 | 5 | 1 | 1+ | 2+ | 1+ | 5 | |||||||||||||||||||||||||||||||||||
| 13 | TFL-160 | IgG3 | 15+ | 1+ | 1+ | 1+ | 5+ | 1 | 1+ | 2 | 1 | 5+ | |||||||||||||||||||||||||||||||||||
| 14 | TFL-105 | IgG2a | 19 | 1 | 2 | 1+ | 5 | 1 | 1+ | 2+ | 1+ | 5+ | |||||||||||||||||||||||||||||||||||
| 15 | TFL-120 | IgG1 | 16 | 1+ | 2 | 2 | 5+ | 1 | 1+ | 2+ | 1+ | 6 | |||||||||||||||||||||||||||||||||||
| 16 | TFL-087 | IgG2b | 21+ | 1+ | 2 | 2 | 6 | 1 | 1+ | 3+ | 2 | 7+ | |||||||||||||||||||||||||||||||||||
| [C] | 17 | TFL-106 | IgG2a | 19 | 1+ | 2 | 2 | 5+ | 1 | 1 | 1 | 2 | 3 | 2 | 6 | ||||||||||||||||||||||||||||||||
| 18 | TFL-032 | IgG2b | 21+ | 1+ | 2+ | 2 | 6 | 1 | 1 | 1 | 2 | 3+ | 2 | 7 | |||||||||||||||||||||||||||||||||
| 19 | TFL-068 | IgG2a | 20+ | 1+ | 2+ | 2 | 6+ | 1 | 1 | 1 | 2 | 3+ | 2 | 7+ | |||||||||||||||||||||||||||||||||
| 20 | TFL-088 | IgG2b | 21+ | 1+ | 2+ | 2 | 7 | 1 | 1 | 1+ | 2 | 4 | 2+ | 8+ | |||||||||||||||||||||||||||||||||
| 21 | TFL-065 | IgG1 | 18 | 2 | 3 | 2+ | 7 | 1 | 1 | 1+ | 2 | 3+ | 2 | 8 | |||||||||||||||||||||||||||||||||
| 22 | TFL-149 | IgG2b | 22 | 2+ | 3 | 2+ | 1 | 8 | 1 | 1 | 1+ | 2+ | 3+ | 2+ | 4 | ||||||||||||||||||||||||||||||||
| 23 | TFL-150 | IgG2b | 22+ | 2 | 2+ | 2+ | 1 | 7+ | 1 | 1 | 1+ | 2 | 3 | 2 | 8 | ||||||||||||||||||||||||||||||||
| 24 | TFL-066 | IgG1 | 16+ | 2 | 3 | 2+ | 1 | 7+ | 1 | 1+ | 1+ | 2+ | 4 | 2+ | 8+ | ||||||||||||||||||||||||||||||||
| 25 | TFL-067 | IgG2a | 20 | 1+ | 2+ | 2 | 6+ | 7 | 1 | 1 | 1 | 2 | 3+ | 2 | 7+ | ||||||||||||||||||||||||||||||||
| [D] | 26 | TFL-130 | IgG1 | 16 | 1+ | 2+ | 1 | 1 | 2 | 5+ | 1 | 1 | 2 | 2+ | 1+ | 6+ | |||||||||||||||||||||||||||||||
| 27 | TFL-117 | IgG2b | 22 | 2 | 3 | 1 | 1 | 2+ | 1 | 8 | 1 | 1 | 1+ | 2+ | 3+ | 2 | 8+ | ||||||||||||||||||||||||||||||
| 28 | TFL-151 | IgG1 | 16+ | 1+ | 2+ | 2 | 5+ | 1 | 1 | 1+ | 2+ | 1+ | 2 | 6+ | |||||||||||||||||||||||||||||||||
| 29 | TFL-136 | IgG2b | 20 | 1 | 2 | 1+ | 1 | 6 | 1 | 1 | 3 | 1+ | 2 | 8+ | |||||||||||||||||||||||||||||||||
| 30 | TFL-135 | IgG1 | 16 | 2 | 3 | 2 | 1 | 6 | 1 | 1+ | 1+ | 2 | 3 | 1+ | 2+ | 1 | 1 | 7 | |||||||||||||||||||||||||||||
| 31 | TFL-152 | IgG2b | 20 | 1 | 2 | 1+ | 1 | 6 | 1 | 1 | 3 | 2 | 2 | 1 | 1 | 8+ | |||||||||||||||||||||||||||||||
| 32 | TFL-100 | IgG1 | 16+ | 2+ | 2+ | 1 | 3 | 1 | 1 | 7+ | 1 | 1+ | 2 | 2+ | 3+ | 2+ | 7 | ||||||||||||||||||||||||||||||
| 33 | TFL-140 | IgG2b | 22+ | 2+ | 3+ | 1 | 3 | 1 | 8+ | 1 | 1+ | 1+ | 3 | 4+ | 2+ | 9+ | |||||||||||||||||||||||||||||||
| 34 | TFL-139 | IgG2b | 22+ | 3 | 3+ | 1 | 3+ | 1 | 1 | 9 | 1 | 1+ | 2 | 3 | 4+ | 3 | 10 | ||||||||||||||||||||||||||||||
| 35 | TFL-124 | IgG1 | 16 | 2+ | 3 | 1 | 2+ | 1 | 6+ | 1 | 1 | 1+ | 1+ | 2+ | 3+ | 2 | 2+ | 1 | 1 | 7+ | |||||||||||||||||||||||||||
| 36 | TFL-009 | IgG2a | 17+ | 2 | 2 | 1 | 1 | 2+ | 1 | 1 | 6 | 1 | 1+ | 1+ | 1+ | 2+ | 1 | 4 | 2+ | 7+ | |||||||||||||||||||||||||||
| 37 | TFL-053 | IgG2a | 20 | 2 | 3 | 1 | 2+ | 1 | 1 | 7+ | 1 | 1+ | 1+ | 1+ | 2+ | 4 | 2+ | 8 | |||||||||||||||||||||||||||||
| 38 | TFL-054 | IgG2a | 20+ | 2 | 3+ | 1 | 3 | 1 | 1 | 7+ | 1 | 1+ | 1+ | 1+ | 2+ | 4+ | 3 | 8 | |||||||||||||||||||||||||||||
| 39 | TFL-089 | IgG2a | 20+ | 2 | 3 | 1 | 2+ | 1 | 1 | 7+ | 1 | 1+ | 1+ | 2 | 2+ | 4+ | 3 | 8+ | |||||||||||||||||||||||||||||
| 40 | TFL-115 | IgG2a | 19 | 2 | 3 | 1 | 2+ | 1 | 1 | 7 | 1 | 1+ | 1+ | 1+ | 2+ | 4 | 2+ | 8 | |||||||||||||||||||||||||||||
| [E] | 41 | TFL-121 | IgG1 | 20+ | 2+ | 3 | 1 | 1 | 1 | 1 | 3 | 1 | 8 | 1 | 1+ | 1+ | 2+ | 4 | 2+ | 8+ | |||||||||||||||||||||||||||
| 42 | TFL-002 | IgG2a | 19+ | 2+ | 2+ | 1 | 1 | 1 | 3 | 1 | 1 | 7 | 1 | 1+ | 2 | 2 | 3 | 1 | 4 | 2+ | 7+ | ||||||||||||||||||||||||||
| 43 | TFL-003 | IgG2a | 19 | 2+ | 2+ | 1 | 1 | 3 | 1 | 1 | 7+ | 1 | 1+ | 2 | 2 | 3 | 1 | 4 | 3 | 7+ | |||||||||||||||||||||||||||
| 44 | TFL-021 | IgG1 | 18 | 3 | 4 | 1 | 1 | 1 | 3+ | 1 | 1+ | 8+ | 1 | 1+ | 2 | 2+ | 1 | 3 | 1 | 5 | 3+ | 9 | |||||||||||||||||||||||||
| 45 | TFL-022 | IgG1 | 18 | 3 | 4 | 1 | 1 | 1 | 3 | 1 | 1 | 8 | 1 | 1+ | 2 | 2 | 3 | 1 | 4+ | 3 | 8+ | ||||||||||||||||||||||||||
| 46 | TFL-023 | IgG2a | 21 | 2+ | 3+ | 1 | 1 | 1 | 3 | 1 | 1 | 8+ | 1 | 1+ | 2 | 2 | 1 | 3 | 1 | 5 | 3+ | 9 | |||||||||||||||||||||||||
| 47 | TFL-024 | IgG2a | 20+ | 2+ | 4 | 1 | 1 | 1 | 3 | 1 | 1 | 8 | 1 | 1+ | 2 | 2 | 3 | 1 | 5 | 3 | 0 | ||||||||||||||||||||||||||
| 48 | TFL-031 | IgG1 | 18+ | 3 | 3 | 1 | 1 | 1 | 4 | 1 | 1 | 9 | 1 | 1+ | 2 | 2+ | 3+ | 1 | 5 | 3 | 9+ | ||||||||||||||||||||||||||
| 49 | TFL-123 | IgG1 | 16+ | 2+ | 3 | 1 | 1 | 3 | 1 | 1 | 7 | 1 | 1+ | 1+ | 2 | 3 | 1 | 4 | 3 | 8 | |||||||||||||||||||||||||||
| 50 | TFL-122 | IgG1 | 17 | 3 | 3+ | 1 | 1 | 3 | 1 | 1 | 7+ | 1 | 1+ | 3 | 2 | 3 | 1 | 4 | 3 | 8+ | |||||||||||||||||||||||||||
| 51 | TFL-131 | IgG2a | 20+ | 3 | 4 | 1 | 1 | 1 | 1 | 3+ | 1 | 1 | 1+ | 7+ | 1+ | 2 | 2 | 2+ | 1 | 3+ | 1 | 1 | 5 | 3 | 8 | ||||||||||||||||||||||
| 52 | TFL-132 | IgG2a | 20 | 2+ | 3+ | 1 | 1 | 1 | 3 | 1 | 1 | 6+ | 1 | 1+ | 1+ | 2 | 1 | 3 | 1 | 4+ | 3 | 7+ | |||||||||||||||||||||||||
| [F] | 53 | TFL-138 | IgG2a | 22 | 3 | 1 | 4 | 1+ | 2+ | 2 | 1+ | 1 | 4 | 1 | 1+ | 9+ | 1 | 1+ | 2 | 1 | 2+ | 3+ | 1 | 5 | 3+ | 10+ | |||||||||||||||||||||
| 54 | TFL-083 | IgG1 | 17+ | 2+ | 1+ | 4 | 2 | 4+ | 3+ | 3 | 1 | 3+ | 1 | 1 | 8 | 1 | 1+ | 2 | 2+ | 2 | 3 | 1 | 4+ | 3 | 9 | ||||||||||||||||||||||
| 55 | TFL-157 | IgG1 | 16 | 2+ | 1 | 3 | 1+ | 3+ | 2+ | 2 | 1 | 3 | 1 | 1 | 7 | 1 | 1 | 1+ | 1+ | 2 | 2+ | 3+ | 2+ | 7+ | |||||||||||||||||||||||
| 56 | TFL-092 | IgG1 | 17+ | 2+ | 1+ | 3+ | 2 | 4+ | 3+ | 2+ | 1 | 3 | 1 | 1 | 1 | 7+ | 1 | 1 | 1+ | 2 | 2 | 2+ | 4 | 2+ | 8 | ||||||||||||||||||||||
| 57 | TFL-127 | IgG1 | 16+ | 3 | 1+ | 3+ | 2 | 4+ | 3+ | 3 | 1 | 3+ | 1 | 1+ | 7+ | 1 | 1+ | 2 | 2+ | 2 | 1 | 3 | 1 | 4+ | 3 | 8+ | |||||||||||||||||||||
| 58 | TFL-137 | IgG2b | 22 | 3+ | 1 | 4+ | 1+ | 3 | 2+ | 2 | 1 | 4 | 1 | 1+ | 9+ | 1 | 1+ | 2 | 1+ | 2+ | 1 | 3+ | 1 | 5 | 3+ | 10+ | |||||||||||||||||||||
| 59 | TFL-040 | IgG2a | 19 | 3+ | 1 | 3+ | 1 | 1 | 1+ | 4 | 1+ | 1+ | 8+ | 1+ | 2 | 2+ | 1 | 3 | 1 | 4 | 1 | 1 | 1 | 5 | 3+ | 9 | |||||||||||||||||||||
| 60 | TFL-080 | IgG1 | 14 | 2+ | 5 | 1 | 1+ | 1 | 2 | 3+ | 1 | 1+ | 1 | 1+ | 8 | 1+ | 2 | 2+ | 1 | 1 | 2+ | 1 | 3 | 1 | 1 | 1 | 1 | 1 | 6 | 4+ | 1 | 10 | |||||||||||||||
| 61 | TFL-026 | IgG1 | 17+ | 3 | 4 | 1 | 1+ | 1 | 1+ | 3 | 1+ | 1 | 1+ | 7+ | 1+ | 2 | 2+ | 1 | 1 | 2+ | 1 | 3 | 1 | 1 | 1 | 5 | 3+ | 1 | 1 | 8 | |||||||||||||||||
| [G] | 62 | TFL-119 | IgG2a | 18+ | 1+ | 5+ | 5 | 2+ | 7 | 6+ | 12+ | 3+ | 11+ | 9+ | 2 | 5+ | 1 | 1 | 9+ | 1 | 3+ | 5+ | 2 | 3 | 2 | 7 | |||||||||||||||||||||
| 63 | TFL-142 | IgG2b | 22+ | 3 | 9+ | 8+ | 4+ | 10+ | 11 | 17+ | 6 | 15+ | 15 | 1 | 3+ | 1 | 1 | 9 | 1 | 1+ | 2 | 13+ | 2 | 7 | 9 | 3+ | 1 | 5 | 3+ | 10 | |||||||||||||||||
| 64 | TFL-153 | IgG2b | 22 | 2+ | 10 | 8+ | 4 | 10+ | 11 | 18 | 6 | 16 | 14+ | 1 | 3+ | 1 | 1 | 8 | 1 | 1+ | 1+ | 14 | 2 | 7 | 9 | 3 | 1 | 5 | 3 | 10 | |||||||||||||||||
| 65 | TFL-118 | IgG2a | 20+ | 3+ | 10+ | 10 | 5 | 12+ | 12+ | 20 | 8 | 17+ | 17+ | 1+ | 4 | 1+ | 1+ | 9 | 1 | 2 | 2 | 16 | 2+ | 1 | 8+ | 10+ | 3+ | 1 | 1 | 5+ | 3+ | 10+ | |||||||||||||||
| 66 | TFL-133 | IgG2a | 22 | 3+ | 11+ | 10+ | 5 | 12+ | 13 | 20+ | 8 | 17+ | 17+ | 1+ | 4+ | 1+ | 1 | 1+ | 10 | 1+ | 2 | 2+ | 16 | 3 | 1 | 9 | 10+ | 4 | 1 | 1 | 6 | 4 | 11 | ||||||||||||||
| 67 | TFL-141 | IgG2b | 22+ | 3+ | 11+ | 11 | 5+ | 13 | 13+ | 21 | 8 | 18+ | 18 | 1+ | 4+ | 1+ | 1 | 1+ | 10+ | 1+ | 2 | 2+ | 17 | 3 | 1 | 9 | 11+ | 4 | 1 | 1 | 6+ | 4 | 12 | ||||||||||||||
| 68 | TFL-095 | IgG2b | 22+ | 4+ | 13 | 13+ | 6+ | 17 | 16+ | 24 | 12 | 21+ | 20+ | 2 | 5 | 1 | 2 | 1 | 2 | 11+ | 1+ | 2+ | 3 | 21 | 3+ | 1 | 12 | 13+ | 5 | 1+ | 1 | 1 | 7+ | 5 | 13 | ||||||||||||
| MFI Expressed in Thousands | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Groups | R-VI | |||||||||||||||||||||||||||||
| Subgroups | [A] | [B] | [C] | [E] | [F] | |||||||||||||||||||||||||
| Number of mAbs | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | 25 | 26 | 27 | 28 | 29 | |
| mAb Nomenclature | TFL-015 | TFL-027 | TFL-039 | TFL-111 | TFL-010 | TFL-014 | TFL-082 | TFL-102 | TFL-101 | TFL-072 | TFL-086 | TFL-113 | TFL-085 | TFL-008 | TFL-038 | TFL-057 | TFL-098 | TFL-025 | TFL-112 | TFL-037 | TFL-071 | TFL-097 | TFL-048 | TFL-058 | TFL-017 | TFL-018 | TFL-070 | TFL-051 | TFL-069 | |
| Subclass | IgG2a | IgG1 | IgG2a | IgG2a | IgG1 | IgG2a | IgG2a | IgG2b | IgG2b | NT | IgG3 | IgG2b | IgG3 | IgG1 | IgG2b | IgG1 | IgG1 | IgG1 | IgG2b | IgG2b | IgG2b | IgG2a | IgG1 | IgG1 | IgG1 | IgG1 | IgG2b | IgG2b | IgG2b | |
| HLA-E Reactivity | 19+ | 17+ | 20 | 18+ | 18+ | 19+ | 18+ | 22 | 21+ | 15+ | 17 | 16 | 16 | 19 | 24 | 17 | 17+ | 17 | 23 | 24 | 23+ | 21 | 17+ | 17+ | 17+ | 17+ | 23 | 23+ | 22+ | |
| HLA-F Reactivity | ||||||||||||||||||||||||||||||
| HLA-G Reactivity | ||||||||||||||||||||||||||||||
| HLA-A Reactivity | A*1101 | 3+ | 3+ | 4 | 3+ | 4 | 4 | 4 | 5 | 5 | 3 | 4 | 3 | 4+ | 5+ | 6+ | 3+ | 4+ | 5+ | 7 | 7 | 8 | 7 | 4 | 4+ | 4+ | 4+ | 8+ | 8+ | 9 |
| A*2402 | 1 | 1 | 1+ | 2 | 1+ | 1 | 1+ | 2 | 1 | 1 | 1 | 2 | 2 | 2 | ||||||||||||||||
| A*2403 | 1 | 1+ | 1 | 1 | 1 | 1+ | 1 | 1 | 1 | 1+ | 1+ | 1+ | ||||||||||||||||||
| A*2601 | 1 | 1 | 1 | |||||||||||||||||||||||||||
| A*2901 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1 | 1+ | 2 | 1 | 1+ | 1+ | 1+ | 2 | 2 | 2 | |||||||||||||
| A*3001 | 1 | |||||||||||||||||||||||||||||
| A*3002 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | |||||||||||||||||||||||
| A*3201 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | |||||||||||||||||||||
| A*3301 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | |||||||||||||||||||||
| A*3303 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 1 | 1+ | 2 | 2 | 2+ | 3 | 2 | 2 | 2+ | 2 | 3+ | 3+ | 3+ | |
| A*3401 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1 | 1+ | 1+ | 1+ | 2 | 2 | 2 | 2 | 2 | 2+ | 2+ | 3 | |||||||||
| A*3601 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 2 | 2+ | 1 | 1+ | 1+ | 1+ | 2+ | 2+ | 3 | |||||||
| A*4301 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | |||||||||||||||||||||||
| A*6601 | 1 | |||||||||||||||||||||||||||||
| A*6802 | 1 | 1 | 1 | |||||||||||||||||||||||||||
| A*8001 | 1 | 1 | 1 | |||||||||||||||||||||||||||
| HLA-C Reactivity | C*0102 | 2+ | 1 | 1 | 1 | 2 | 1+ | 1+ | 1+ | 1 | 1+ | 1+ | ||||||||||||||||||
| C*0202 | 1+ | 1+ | 1 | 1 | 1 | 1+ | 1 | 1 | 1+ | 2 | 2+ | 2 | 1+ | 2+ | 3 | 1 | 2 | 2+ | 2+ | 3 | 3 | 4+ | 3+ | 3+ | 3+ | 4 | 4 | 4+ | ||
| C*0302 | 2 | 1 | 1+ | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 1+ | 1+ | 3 | 2 | 2+ | 2 | 2 | 2 | 2+ | |||||||||||
| C*0303 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 2 | 1+ | 1+ | 1+ | 2+ | 1 | 1+ | 2+ | 2 | 2+ | 3 | 3+ | 3 | 3 | 3 | 3+ | 3+ | 4 | |||||
| C*0304 | 1 | 1 | 1+ | 1+ | 1+ | 1 | 1+ | 2+ | 1 | 1 | 1+ | 1+ | 2 | 2+ | 3 | 3 | 3 | 2+ | 3 | 3 | 3+ | |||||||||
| C*0401 | 1 | 1+ | 1 | 1 | 1 | 1 | ||||||||||||||||||||||||
| C*0501 | 5+ | 5+ | 5+ | 5 | 5+ | 5+ | 6 | 7 | 7 | 6+ | 6 | 6+ | 6+ | 7+ | 9+ | 7 | 6 | 7+ | 10 | 10 | 10+ | 9 | 9+ | 8 | 7+ | 7+ | 11 | 11 | 11+ | |
| C*0602 | 1 | 1+ | 1 | 1 | 1+ | 1 | 1 | 1 | 1+ | 5 | 2 | 2+ | 2 | 2 | 2 | 2 | ||||||||||||||
| C*0702 | 1 | 1 | 1 | 1 | 3+ | 1+ | 1+ | 1+ | 1 | 1 | 1+ | |||||||||||||||||||
| C*0801 | 3+ | 4 | 4 | 3+ | 4 | 4 | 4 | 5 | 4+ | 5 | 5 | 4+ | 5 | 5+ | 7 | 6 | 4+ | 6 | 7+ | 7+ | 8 | 7+ | 7 | 7 | 6 | 6 | 9 | 9+ | 9+ | |
| C*1203 | 2 | 1 | 1 | 2 | 1 | 1+ | 1+ | 1 | 1 | 1 | ||||||||||||||||||||
| C*1402 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 2 | 1+ | 1+ | 1+ | 2+ | 1 | 1+ | 2 | 2 | 2 | 3 | 4 | 3 | 3 | 3 | 3+ | 3+ | 4 | ||
| C*1502 | 1 | 1 | 1+ | 1 | 1 | 1 | 1+ | 1 | 1+ | 1+ | 1+ | 2 | 2+ | 2 | 2+ | 2 | 2+ | 2+ | 2+ | |||||||||||
| C*1601 | 1 | 1 | 1+ | 1 | 1 | 1 | 1+ | 1 | 1 | 1+ | 1 | 1 | 1 | 1 | 1+ | 3 | 2 | 2 | 2 | 2 | 2 | 2 | ||||||||
| C*1701 | 1 | 1 | 1+ | 1 | 1+ | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 1+ | 2 | 3+ | 2+ | 2+ | 2+ | 2+ | 2+ | 2+ | |||||||||
| C*1802 | 8+ | 8+ | 9+ | 8+ | 10 | 9 | 9 | 11+ | 11+ | 9+ | 9+ | 9+ | 9+ | 11+ | 15 | 10+ | 10 | 11+ | 14+ | 15 | 15+ | 13 | 12 | 12 | 11 | 10+ | 15+ | 16 | 15+ | |
| HLA-B Reactivity | B*0801 | 1+ | 1 | |||||||||||||||||||||||||||
| B*1301 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 2 | 1+ | 1+ | 3 | 1+ | 1+ | 1+ | 2+ | 3+ | 2+ | 3+ | 2 | 2+ | 2+ | 2+ | 3+ | 3+ | 4 | |
| B*1302 | 1 | 1 | 1+ | 1 | 1 | 1 | 1+ | 1 | 1 | 1+ | 1+ | |||||||||||||||||||
| B*1401 | 3+ | 4+ | 4 | 3+ | 5 | 3+ | 4 | 4+ | 5 | 5+ | 5+ | 5+ | 6 | 6 | 8+ | 6 | 5 | 6+ | 8+ | 9 | 9 | 8+ | 8 | 8 | 7 | 7 | 10+ | 10+ | 11 | |
| B*1402 | 1 | 1 | 1 | 1 | 1 | 1+ | 1 | 1 | 1 | 1+ | 2 | 2+ | 2 | 2+ | 2 | 2+ | 2+ | 2+ | ||||||||||||
| B*1502 | 1 | 1+ | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 2 | 2 | 2 | 4+ | 2+ | 2 | 2+ | 3 | 5 | 3+ | 4 | 3 | 3 | 3 | 3 | 4+ | 4+ | 5 | |
| B*1503 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 2 | |||||||||||||||||
| B*1510 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | ||||||||||||||||||||||
| B*1511 | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 2+ | 2 | 2+ | 5 | 2+ | 2 | 2+ | 3+ | 5+ | 3+ | 4 | 3+ | 3+ | 3+ | 3+ | 4+ | 5 | 5 | |
| B*1512 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | |||||||||||||||||||||||
| B*1513 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 2 | 1+ | 1+ | 4 | 2 | 1 | 1+ | 2 | 4+ | 2+ | 2+ | 2+ | 2+ | 2+ | 2+ | 3+ | 3+ | 3+ | |
| B*1516 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1 | 3 | 1+ | 1 | 1+ | 2 | 3+ | 2 | 2+ | 2 | 2 | 2+ | 2 | 3 | 3 | 3 | ||||||
| B*1801 | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 2 | 2 | 2+ | 2+ | 3 | 3 | 2+ | 3+ | 3 | 2 | 3 | 4 | 4 | 4 | 4+ | 4 | 3+ | 4 | 3+ | 5 | 5+ | 5+ | |
| B*2705 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | ||||||||||||||||||||||
| B*2708 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1 | 1 | 1 | 1+ | 2 | 2 | 1+ | 2 | 2 | 2 | 2 | 2+ | |||||||||||
| B*3501 | 4 | 4 | 4+ | 4 | 4+ | 4 | 4+ | 5+ | 5+ | 3+ | 4+ | 4 | 5 | 5+ | 7+ | 4+ | 5 | 6 | 8 | 8 | 8+ | 7+ | 5+ | 5+ | 5+ | 5 | 9 | 9 | 9+ | |
| B*3701 | 1 | 1 | 2 | 2 | 2+ | 2 | 1+ | 0 | 2+ | 1 | 2 | 2+ | 3 | 2+ | 3+ | 3 | 4 | 3+ | 4 | 4 | 4+ | |||||||||
| B*3801 | 1 | 1 | 1 | 1 | ||||||||||||||||||||||||||
| B*3901 | 1+ | 1+ | 2 | 1+ | 2 | 1+ | 2 | 2 | 2 | 2 | 2+ | 2+ | 2+ | 2+ | 3+ | 2+ | 2 | 3 | 4 | 4 | 4+ | 4+ | 3+ | 3+ | 3+ | 3+ | 5+ | 4+ | 6 | |
| B*4001 | 1 | 1 | ‘ | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 2 | 2 | 1+ | 2 | 2 | 1+ | 2 | 2+ | 2+ | 2+ | 3 | 2+ | 3 | 3 | 3 | 3+ | 3+ | 4 | |
| B*4002 | 1+ | 1+ | 2 | 2 | 2 | 2 | 2 | 2 | 2+ | 2 | 2+ | 2 | 2+ | 2+ | 3+ | 2+ | 2 | 3 | 4 | 4 | 4 | 4 | 3 | 3+ | 3+ | 3 | 5 | 5 | 5+ | |
| B*4006 | 8 | 8 | 9+ | 8+ | 9 | 8+ | 9 | 11+ | 11+ | 8 | 9 | 8 | 9+ | 11 | 14 | 9 | 9 | 11 | 14 | 14 | 14+ | 12+ | 10 | 10+ | 9+ | 9+ | 14+ | 14+ | 15+ | |
| B*4101 | 1+ | 1+ | 2 | 1+ | 2 | 2 | 2 | 2 | 2 | 2+ | 2+ | 2+ | 2+ | 2+ | 3 | 3 | 2 | 2+ | 4 | 3+ | 4 | 4+ | 3+ | 3+ | 3+ | 3+ | 5 | 5 | 5+ | |
| B*4402 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1 | 1+ | 1 | 1+ | 2 | 2 | 2 | 2+ | 2 | 2+ | 2+ | 2+ | ||||||||||
| B*4403 | 2+ | 2+ | 2+ | 2 | 2+ | 2+ | 2+ | 2+ | 2+ | 2 | 3 | 2+ | 3 | 3+ | 4 | 3+ | 2+ | 3+ | 4+ | 4 | 5 | 5 | 4 | 4 | 4 | 4 | 6 | 6 | 6 | |
| B*4501 | 2+ | 3 | 3 | 2+ | 3 | 3 | 3 | 3+ | 3+ | 3 | 4 | 3+ | 4 | 4+ | 5 | 4 | 3+ | 4+ | 6+ | 5+ | 6+ | 6 | 5 | 5 | 5 | 5 | 7+ | 7+ | 8 | |
| B*4601 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 2 | 1+ | 1+ | 3+ | 2 | 1 | 1+ | 2+ | 3+ | 2+ | 3 | 2+ | 2+ | 2+ | 2+ | 3 | 3+ | 3+ | |
| B*4701 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 2 | 1+ | 1+ | 1+ | 2+ | 1 | 1+ | 2 | 2 | 2+ | 3 | 3 | 3 | 3 | 3 | 3+ | 3+ | 3+ | |||
| B*4801 | 1 | 1 | 1+ | 1 | 1 | 1+ | 1 | 1 | 1 | 1+ | 2 | 2 | 2 | 2 | 2 | 2 | 2 | |||||||||||||
| B*5101 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1 | 1+ | 1+ | 1+ | 2 | 2 | 2 | 2 | 2 | 2+ | 2+ | 2+ | |||||||||||
| B*5102 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1 | 1+ | 2 | 2 | 2 | 2+ | 2 | 2 | 2 | 2 | 3 | 3 | 3+ | ||||||||
| B*5201 | 1 | 1 | 1+ | 1 | 1 | 1 | 1+ | 1 | 1+ | 1+ | 1+ | 2 | 2+ | 2+ | 2+ | 2 | 2+ | 2+ | 2+ | |||||||||||
| B*5301 | 2 | 2+ | 3 | 3 | 3 | 2 | 3 | 4 | 4 | 3 | 3+ | 3+ | 4 | 4+ | 5+ | 3+ | 3+ | 4+ | 6+ | 6 | 6+ | 6+ | 4+ | 4+ | 5 | 4+ | 7+ | 7+ | 8 | |
| B*5401 | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 2 | 2 | 2+ | 2 | 2 | 2+ | 3+ | 3 | 3+ | 4 | 3 | 3 | 3 | 3 | 4+ | 4+ | 5 | |
| B*5501 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 2 | 1+ | 2 | 2 | 2+ | ||||||||||||||
| B*5801 | 3+ | 3+ | 4 | 3+ | 4 | 4 | 4 | 5 | 5 | 4 | 3+ | 4+ | 5+ | 7 | 4+ | 4+ | 5+ | 7 | 7+ | 7+ | 7+ | 4+ | 5 | 5 | 5 | 8+ | 9 | 9+ | ||
| B*5901 | 1 | 1 | 1 | 1 | 1+ | 1 | 1 | 1 | 1 | 1+ | 2 | 2 | 2 | 2 | 2 | 2 | 2+ | |||||||||||||
| B*7301 | 3 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 2 | ||||||||||||||
| B*7801 | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 1 | 2 | 2 | 2 | 2+ | 3 | 2 | 2 | 2+ | 3+ | 3 | 3+ | 4 | 3 | 3 | 3 | 3 | 4+ | 5 | 4 | |
| B*8101 | 1+ | 1 | ||||||||||||||||||||||||||||
| B*8201 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 1 | 1+ | 2+ | 2 | 2+ | 3 | 3 | 2+ | 3 | 2+ | 3+ | 3+ | 3+ | |
| MFI Expressed in Thousands | ||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Groups | Subgroups | number of mAbs | mAb Nomenclature | IgG Subclass | HLA-ER Reactivity (MFI) | HLA-EG Reactivity (MFI) | HLA-F Reactivity (MFI) | HLA-G Reactivity (MFI) | HLA-A*1101 Reactivity (MFI) | HLA-B Reactivity | HLA-C Reactivity | |||||||||||||||||||||||||||||||
| B*0702 | B*0801 | B*1301 | B*1302 | B*1402 | B*1501 | B*1510 | B*1511 | B*1512 | B*1513 | B*1516 | B*2708 | B*3501 | B*3901 | B*4001 | B*4002 | B*4201 | B*4402 | B*4501 | B*5001 | B*5201 | B*5301 | B*5401 | B*5601 | B*5701 | B*5703 | B*5801 | B*8101 | C*0401 | C*0702 | C*1402 | C*1701 | C*1802 | ||||||||||
| G-V | 1 | TFL-175 | IgG1 | 12 | 1+ | 1+ | ||||||||||||||||||||||||||||||||||||
| 2 | TFL-174 | IgG1 | 12 | 1+ | 1+ | 1 | ||||||||||||||||||||||||||||||||||||
| 3 | TFL-173 | IgG1 | 12 | 1+ | 1+ | 1 | ||||||||||||||||||||||||||||||||||||
| 4 | TFL-219 | IgG1 | 21 | 20+ | 2 | 2 | 1 | |||||||||||||||||||||||||||||||||||
| G-VI | [A] | 1 | TFL-222 | IgG1 | 19 | 18+ | 1 | 3+ | 2 | 1+ | 1 | |||||||||||||||||||||||||||||||
| 2 | TFL-220 | IgG1 | 19 | 19+ | 1 | 3+ | 2 | 1+ | 1 | |||||||||||||||||||||||||||||||||
| 3 | TFL-187 | IgG2a | 25 | 24+ | 1+ | 3+ | 2 | 1+ | 1 | |||||||||||||||||||||||||||||||||
| [B] | 4 | TFL-201 | IgG1 | 11 | 12 | 1+ | 1 | 3 | 1+ | 2+ | 1+ | 3 | ||||||||||||||||||||||||||||||
| 5 | TFL-193 | IgG1 | 14 | 15+ | 2 | 1 | 1 | 5 | 2+ | 3+ | 2+ | 1 | 4 | 1 | ||||||||||||||||||||||||||||
| 6 | TFL-194 | IgG1 | 21 | 21 | 2 | 1 | 1+ | 5+ | 3 | 3+ | 3 | 1 | 4+ | 1 | ||||||||||||||||||||||||||||
| 7 | TFL-199 | IgG1 | 15+ | 15+ | 2 | 1 | 1+ | 4+ | 2+ | 3+ | 2+ | 1 | 4 | 1 | ||||||||||||||||||||||||||||
| 8 | TFL-258 | IgG1 | 20+ | 20+ | 3+ | 0 | 1 | 7 | 1 | 1 | 4+ | 4 | 3 | 1 | ||||||||||||||||||||||||||||
| 9 | TFL-257 | IgG1 | 20+ | 21+ | 3+ | 1 | 1 | 7 | 1 | 1 | 4 | 4 | 3 | 5 | 1 | |||||||||||||||||||||||||||
| [C] | 10 | TFL-253 | IgG2b | 26+ | 26 | 6 | 1 | 2+ | 2+ | 1 | 11 | 1 | 1 | 1 | 2 | 1+ | 1 | 2 | 7+ | 1 | 1 | 7+ | 1+ | 1 | 7 | 5+ | 1+ | 8+ | 2 | |||||||||||||
| 11 | TFL-249 | IgG2b | 26+ | 25 | 6 | 1 | 2+ | 2+ | 1 | 11 | 1 | 1 | 1 | 2 | 2 | 1+ | 1 | 2 | 7+ | 1 | 1 | 1 | 1 | 7+ | 1+ | 1 | 7 | 5+ | 1+ | 9 | 2 | |||||||||||
| 12 | TFL-250 | IgG2b | 27 | 26 | 6 | 1 | 2+ | 2+ | 1 | 10+ | 1 | 2 | 1+ | 1 | 2 | 7+ | 1 | 1 | 1 | 7+ | 1 | 1 | 7 | 5+ | 1+ | 8+ | 2 | |||||||||||||||
| 13 | TFL-241 | IgG1 | 22 | 21 | 4 | 2+ | 13+ | 2 | 3 | 8+ | 1+ | 7+ | 14 | 12 | 1+ | 1 | 1 | 14 | 1 | 5 | 5+ | 11 | 5 | 4+ | 4 | 1 | 6 | 3+ | ||||||||||||||
| 14 | TFL-242 | IgG1 | 22 | 21+ | 4+ | 2+ | 14 | 2 | 3+ | 9 | 1+ | 7+ | 14 | 12+ | 1 | 1+ | 1 | 1+ | 14+ | 1 | 5 | 1 | 5+ | 11 | 5 | 5 | 4 | 1 | 6 | 4 | ||||||||||||
| 15 | TFL-247 | IgG1 | 21+ | 20+ | 4 | 2+ | 14 | 2 | 3 | 8+ | 1+ | 7+ | 14+ | 12+ | 1+ | 1 | 14+ | 1 | 5 | 1 | 5+ | 11+ | 5 | 1 | 4+ | 1 | 6 | 4 | ||||||||||||||
| Groups | G-VII | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MFI Expressed in Thousands | ||||||||||||||||||||||||||
| Subgroups | [A] | [B] | [C] | [D] | [E] | [F] | [G] | |||||||||||||||||||
| Number of mAbs | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | 25 | |
| mAb Nomenclature | TFL-221 | TFL-218 | TFL-217 | TFL-216 | TFL-196 | TFL-167 | TFL-231 | TFL-252 | TFL-170 | TFL-169 | TFL-166 | TFL-168 | TFL-251 | TFL-207 | TFL-206 | TFL-205 | TFL-243 | TFL-246 | TFL-244 | TFL-245 | TFL-189 | TFL-190 | TFL-188 | TFL-172 | TFL-171 | |
| IgG Subclass | NT | IgG1 | IgG1 | IgG1 | IgG2a | IgG1 | IgG2b | IgG2b | IgG1 | IgG1 | IgG1 | IgG1 | IgG2b | IgG1 | IgG1 | IgG1 | IgG2a | IgG2a | IgG2a | IgG2a | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | |
| HLA-ER Reactivity | 25 | 21 | 20 | 20 | 24 | 24+ | 25 | 26 | 18+ | 19 | 26 | 24+ | 25 | 25 | 13+ | 14+ | 14 | |||||||||
| HLA-EG Reactivity | 25 | 20 | 19+ | 20 | 24 | 15 | 24 | 25 | 17 | 17+ | 16+ | 15 | 25+ | 17+ | 19 | 18+ | 25 | 24+ | 24+ | 24+ | 15+ | 16+ | 16 | 14+ | 15 | |
| HLA-F Reactivity | ||||||||||||||||||||||||||
| HLA-G Reactivity | ||||||||||||||||||||||||||
| HLA-A Reactivity | A*0101 | 14 | 15 | |||||||||||||||||||||||
| A*0201 | 1 | 1 | ||||||||||||||||||||||||
| A*0203 | 1+ | 1+ | ||||||||||||||||||||||||
| A*0206 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | |||||||||||||||
| A*0301 | 12+ | 14 | ||||||||||||||||||||||||
| A*1101 | 6 | 5 | 5+ | 5+ | 4 | 5+ | 8+ | 9+ | 6+ | 6+ | 6+ | 6+ | 9+ | 8 | 8 | 8 | 12 | 12 | 12 | 12 | 1+ | 1+ | 1+ | 9+ | 10 | |
| A*1102 | 11 | 10+ | 11+ | 11 | 11 | 11+ | ||||||||||||||||||||
| A*2402 | 1 | 1 | 1 | 1 | 1 | 1+ | 2+ | 3 | 2 | 2 | 2 | 2 | 3 | 3 | 3 | 3 | 2+ | 2+ | 3 | 3 | 1 | 1 | ||||
| A*2403 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | ||||||||||||||||
| A*2601 | 1 | 1+ | 2 | 1+ | 2 | 2 | 2 | 2 | 2+ | 3 | 3 | 3 | 1 | 1 | ||||||||||||
| A*2901 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 2 | |||||||||||||||
| A*3001 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | |||||||||||||||||
| A*3002 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 2 | 2 | 2 | |||||||||||||||
| A*3201 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | |||||||||||||||
| A*3301 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | ||||||||||||||||
| A*3303 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 2 | 14+ | 15 | |||||||||||||
| A*3401 | 1 | 1 | 1 | 1 | 11 | 12 | ||||||||||||||||||||
| A*3402 | 1 | 1 | 1 | 11+ | 12 | |||||||||||||||||||||
| A*3601 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 11 | 12 | ||||||||||||||||
| A*4301 | 1 | 1 | 1 | 11+ | 12+ | |||||||||||||||||||||
| A*7401 | 1+ | 1+ | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2+ | 13+ | 14 | ||||||||||||
| A*8001 | 3 | 3+ | ||||||||||||||||||||||||
| HLA-B Reactivity | B*0702 | 1 | 1 | 1 | ||||||||||||||||||||||
| B*0801 | 15+ | 3 | 3 | 3 | 1+ | 2+ | 23 | 4 | 3 | 3 | 3 | 3 | 4 | 20+ | 19 | 20 | 1+ | 1+ | 1+ | 1+ | 11 | 11+ | ||||
| B*1301 | 16+ | 17 | 17+ | 17+ | 2 | 4 | 24 | 6 | 4+ | 4+ | 4+ | 4+ | 6 | 20+ | 19+ | 20 | 3 | 3 | 3 | 3 | <1 | 1 | 1 | 2 | 2+ | |
| B*1302 | 3 | 3 | 3 | 3 | 2 | 3+ | 6 | 6+ | 4+ | 4+ | 4+ | 4+ | 7 | 6 | 6 | 6 | 8 | 9 | 8 | 8+ | <1 | <1 | 1 | 2 | 2+ | |
| B*1401 | 1 | 1+ | 1 | 1 | 1 | 1 | 1+ | 2 | 2 | 2 | 7+ | 7 | 7+ | 8 | ||||||||||||
| B*1402 | 4 | 7+ | 8 | 8+ | 11 | 12+ | 12+ | 12+ | ||||||||||||||||||
| B*1501 | 10+ | 12+ | 12+ | 13 | 1 | 2 | 17+ | 3+ | 2+ | 2+ | 2+ | 3 | 3+ | 16+ | 16+ | 16+ | 6 | 6 | 6 | 6+ | 1 | 1 | ||||
| B*1502 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 15+ | 15 | 15+ | 15+ | 12 | 13 | |||||||||||
| B*1503 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 10 | 9+ | 10 | 10+ | ||||||||||||||||
| B*1510 | 1+ | 4 | 4+ | 5 | 5+ | 8 | 7+ | 8 | 1+ | 1+ | ||||||||||||||||
| B*1511 | 10+ | 8+ | 9 | 8+ | 6+ | 8 | 13+ | 14 | 9 | 9 | 9 | 9 | 14 | 11+ | 10+ | 11 | 10+ | 10+ | 10 | 10 | 4 | 4+ | 4+ | 11+ | 12 | |
| B*1512 | 17+ | 17+ | 17+ | 18 | 1 | 2+ | 23+ | 3+ | 3 | 3 | 3 | 3 | 3+ | 20 | 20 | 19+ | 8+ | 7+ | 8+ | 9 | 1 | 1 | ||||
| B*1513 | 12+ | 15+ | 16+ | 17 | 1 | 22 | 1 | 1 | 1 | 1 | 1 | 1 | 20 | 18+ | 19 | 7+ | 7 | 8 | 8+ | 14+ | 15+ | |||||
| B*1516 | 1+ | 1+ | 1+ | 1+ | 1 | 2 | 3 | 3+ | 2+ | 2+ | 2+ | 2+ | 3+ | 3+ | 3+ | 3+ | 1 | 1+ | 1+ | 1+ | 1 | 1 | ||||
| B*1801 | 1 | 1 | ||||||||||||||||||||||||
| B*2705 | 1 | 1 | 1 | 1 | ||||||||||||||||||||||
| B*2708 | 1 | 1 | 1+ | 1+ | 1 | 2 | 2+ | 3+ | 2+ | 2+ | 2+ | 2+ | 3+ | 3+ | 3 | 3+ | 1 | 1 | 1 | 1 | 1 | |||||
| B*3501 | 1 | 1+ | 1+ | 1+ | 1 | 2 | 3+ | 4 | 3 | 2+ | 3 | 3 | 4 | 4 | 4 | 4 | 1+ | 1 | 1+ | 1+ | 13 | 14+ | ||||
| B*3701 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | |||||||||||||||||||
| B*3901 | 2+ | 2 | 2+ | 2+ | 1+ | 3 | 4+ | 5+ | 3+ | 3+ | 3+ | 3+ | 5+ | 5 | 5 | 5 | 2+ | 2+ | 2+ | 2+ | 11 | 12+ | ||||
| B*4001 | 1 | 1 | 1 | 1 | 1+ | 2 | 2+ | 2 | 2 | 2 | 2 | 2+ | 3 | 3 | 3 | 1 | 1 | 1 | 1 | 7 | 7+ | |||||
| B*4002 | 2 | 1+ | 2 | 2 | 1+ | 2+ | 4 | 5+ | 3+ | 3 | 3+ | 3+ | 5 | 4+ | 4+ | 4+ | 2+ | 2+ | 2+ | 2+ | 9 | 9+ | ||||
| B*4006 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 2 | |||||||||||||||
| B*4201 | 1+ | 1 | 1+ | 1+ | 1 | 2 | 3 | 4 | 2+ | 2+ | 2+ | 2+ | 4 | 4 | 4 | 4 | 3 | 2+ | 3 | 3 | 12+ | 13+ | ||||
| B*4402 | 2+ | 2 | 2 | 2 | 1+ | 3 | 4+ | 5+ | 3+ | 3+ | 3+ | 3+ | 5+ | 5 | 5 | 4+ | 2+ | 2+ | 2+ | 2+ | 8+ | 9 | ||||
| B*4403 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 6 | 6 | |||||||||||
| B*4501 | 16+ | 18 | 18+ | 18+ | 1+ | 23+ | 2 | 2 | 1+ | 2 | 2 | 2 | 20 | 20 | 20+ | 13+ | 15 | |||||||||
| B*4601 | 1 | 1 | 1+ | 1 | 1 | 1 | 1 | 1+ | 2 | 2 | 2 | 10+ | 11+ | |||||||||||||
| B*4701 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 12 | 14 | ||||||||||||||
| B*4801 | 3+ | 2+ | 3+ | 4 | 9 | 9+ | ||||||||||||||||||||
| B*4901 | 4+ | 4 | 5 | 5 | 8+ | 9+ | ||||||||||||||||||||
| B*5001 | 1 | 1+ | 1+ | 1+ | 1 | 2 | 3+ | 4 | 2+ | 2+ | 2+ | 2+ | 4 | 4 | 4 | 4 | 6+ | 5+ | 6+ | 7 | 1 | 1 | ||||
| B*5101 | 1 | 1 | 1+ | 1 | 1 | 1 | 1 | 1+ | 2 | 2 | 2 | 5 | 4 | 5 | 5+ | |||||||||||
| B*5102 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 2 | 3+ | 3 | 4 | 4 | 11 | 12 | ||||||||||
| B*5201 | 7 | 6 | 6+ | 6+ | 4+ | 6+ | 10+ | 11 | 7+ | 7+ | 7 | 7+ | 11 | 9+ | 9+ | 9+ | 6+ | 7 | 6+ | 7 | 2 | 2+ | 2+ | 4+ | 5 | |
| B*5301 | 1 | 1 | 1+ | 1+ | 1 | 2 | 2 | 3+ | 2+ | 2+ | 2+ | 2+ | 3+ | 3 | 3 | 3 | 1 | 1 | 1 | 1+ | 1 | 1 | ||||
| B*5401 | 1 | 1 | 1 | 1 | 1 | 1+ | 2+ | 3+ | 2 | 2 | 2 | 2 | 3+ | 3 | 3 | 3+ | 15+ | 15+ | 15 | 15 | 1 | 1 | ||||
| B*5501 | 9+ | 1 | 1 | 1+ | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 0 | 0 | 0 | 0 | ||||||||||
| B*5601 | 7 | 9 | 9+ | 15 | 1 | 1+ | 12+ | 2+ | 2+ | 2 | 2+ | 2+ | 3 | 13 | 12+ | 12+ | <1 | 1 | 1 | 1 | 2 | 2 | ||||
| B*5701 | 13 | 14+ | 14+ | 6+ | 1 | 2+ | 20 | 4 | 3 | 3 | 3 | 3 | 4 | 17+ | 17+ | 18 | 1 | 1 | 1 | 1 | 4+ | 5 | ||||
| B*5703 | 7+ | 6+ | 6+ | 2 | 4+ | 6 | 10+ | 10+ | 7 | 7 | 7 | 7 | 10+ | 9 | 8 | 9 | 7 | 7 | 6+ | 7 | 2+ | 3 | 2+ | 7 | 7+ | |
| B*5801 | 1+ | 1+ | 2 | 1 | 1 | 2+ | 4 | 4+ | 3 | 3 | 3 | 3 | 4+ | 4+ | 4+ | 1+ | 1+ | 1+ | 1+ | 2+ | 3 | 3 | 1 | 1+ | ||
| B*6701 | 1 | 1 | 1 | 1+ | 1 | 1+ | 2 | 2+ | 2 | 2 | 2 | 2 | 2+ | 3 | 3 | 3 | 1 | |||||||||
| B*7301 | 1 | 1+ | 2+ | 1+ | 1+ | 1+ | 1+ | 2+ | 2+ | 2+ | 2+ | 12+ | 12 | 12 | 13 | |||||||||||
| B*7801 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | |||||||||||
| B*8101 | 1 | 1 | 1+ | 0 | 1 | 2 | 3 | 4 | 2+ | 2+ | 2+ | 2+ | 4 | 3+ | 3+ | 3+ | 1+ | 1+ | 1+ | 1+ | 1 | 1 | ||||
| B*8201 | 1 | 1 | 2 | 1+ | 1+ | 1+ | 1+ | 2+ | 2 | 2 | 2 | 12 | 13 | 13 | 14 | 16 | ||||||||||
| HLA-C Reactivity | C*0102 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 10+ | 11+ | 11+ | 14 | 15+ | ||||||||||
| C*0202 | 1+ | 1 | 2+ | 1+ | 1+ | 1+ | 1+ | 2+ | 2+ | 2+ | 2+ | 13+ | 11+ | 13+ | 13+ | 13+ | 13+ | 14+ | 13+ | 15 | ||||||
| C*0302 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 8 | 6+ | 8+ | 8 | 16+ | 16+ | 16+ | 14 | 15+ | ||||||||
| C*0303 | 1 | 1 | 1 | 6 | 1 | 2 | 2 | 3+ | 2+ | 2+ | 2+ | 2+ | 3+ | 3+ | 3 | 3+ | 9 | 7+ | 9 | 9 | 15 | 15 | 15 | 14 | 15+ | |
| C*0304 | 1 | 1 | 2 | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 2 | 2 | 5+ | 6+ | 6+ | 13+ | 15 | ||||||||||
| C*0401 | 6+ | 5+ | 6 | 5+ | 4+ | 6 | 9 | 10+ | 7 | 7 | 6+ | 7 | 10+ | 8+ | 8+ | 8 | 6+ | 6+ | 6+ | 6+ | 10 | 11+ | 11 | 13 | 14 | |
| C*0501 | 1 | 1 | 2 | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 1+ | 2 | 1 | 1 | 1 | 1 | 20 | 20 | 19 | 13 | 14+ | ||||||
| C*0602 | 1 | 1 | 1+ | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 11+ | 10 | 12 | 12 | 17+ | 17+ | 18 | 12 | 14 | ||||||
| C*0702 | 5+ | 5+ | 5+ | 1 | 9+ | 6+ | 6+ | 6+ | 6+ | 9+ | 8 | 7+ | 7+ | 5+ | 5+ | 5+ | 5+ | 11 | 12+ | 12+ | 13+ | 15 | ||||
| C*0801 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1 | 1 | 20 | 20+ | 20 | 15+ | 17 | ||||||||||||
| C*1203 | 1 | 1 | 2 | 1 | 1+ | 1+ | 3 | 2 | 2 | 2 | 2 | 3 | 2+ | 2+ | 2+ | 1 | 1 | 1 | 1 | 13 | 13+ | 14 | 13+ | 14+ | ||
| C*1402 | 1+ | 1+ | 2 | 7+ | 1+ | 2+ | 3+ | 4+ | 3 | 3 | 3 | 3 | 4+ | 4 | 3+ | 4 | 19+ | 19+ | 19+ | 19+ | 18+ | 18+ | 18 | 14+ | 16 | |
| C*1502 | 1 | 1 | 2 | 1 | 1 | 1 | 1 | 2 | 2 | 1+ | 2 | 24+ | 24+ | 24+ | 24+ | 18+ | 19 | 18+ | 13+ | 15 | ||||||
| C*1601 | 1 | 1 | 11 | 2 | 1+ | 1+ | 1+ | 1+ | 2+ | 2+ | 2+ | 2+ | 22 | 21+ | 21+ | 21+ | 16 | 16+ | 17 | 13 | 14+ | |||||
| C*1701 | 8+ | 7 | 7+ | 4+ | 5 | 7 | 24+ | 12 | 8 | 8 | 8 | 8 | 12 | 10 | 9+ | 9+ | 8 | 8 | 8 | 8 | 11+ | 12+ | 12+ | 14+ | 16 | |
| C*1802 | 18 | 4+ | 4+ | 0 | 1+ | 3+ | 5+ | 4 | 4 | 4 | 4+ | 6 | 20+ | 20 | 20+ | 2+ | 2+ | 2+ | 2+ | 1 | 1 | 1 | 13 | 14+ | ||
| Groups | R-VII | R-VIII | R-IX | R-X | G-VIII | G-IX | G-X | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MFI Expressed in Thousands | |||||||||||||||||||||||||||||||||||||
| Subgroups | [A] | [B] | [C] | [A] | [B] | ||||||||||||||||||||||||||||||||
| Number of mAbs | 1 | 1 | 2 | 1 | 1 | 2 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 1 | 2 | 3 | 4 | |
| mAb Nomenclature | TFL-063 | TFL-103 | TFL-104 | TFL-049 | TFL-006 | TFL-007 | TFL-227 | TFL-197 | TFL-198 | TFL-202 | TFL-203 | TFL-204 | TFL-210 | TFL-211 | TFL-212 | TFL-235 | TFL-230 | TFL-256 | TFL-224 | TFL-214 | TFL-213 | TFL-215 | TFL-225 | TFL-233 | TFL-240 | TFL-238 | TFL-236 | TFL-237 | TFL-239 | TFL-234 | TFL-248 | TFL-229 | TFL-232 | TFL-177 | TFL-176 | TFL-198 | |
| IgG Subclass | IgG2b | IgG1 | IgG1 | IgG2b | IgG2a | IgG2a | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG3 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG1 | IgG2b | IgG3 | IgG1 | IgG1 | IgG1 | |
| HLA-ER Reactivity | 22+ | 18 | 17+ | 15 | 22+ | 21+ | 18 | 17 | 16 | 18 | 18 | 17+ | 19 | 18 | 18 | 21 | 0 | 22+ | 22 | 20+ | 21 | 21 | 19 | 19+ | 18 | 18 | 19 | 20 | 19+ | 19 | 21+ | 30 | 22+ | 14+ | 13+ | 15 | |
| HLA-EG Reactivity | 18+ | 16 | 15 | 17+ | 16+ | 16+ | 17+ | 17+ | 18 | 19+ | 18+ | 22 | 20+ | 21+ | 20 | 20+ | 19+ | 18 | 19+ | 19+ | 18 | 21 | 27+ | 21 | |||||||||||||
| HLA-F Reactivity | 3+ | 8+ | 12+ | 11 | 5+ | 10+ | 10 | 11 | 10+ | 10+ | 11 | 11 | 11 | 9 | 2+ | 5 | 4+ | 10 | |||||||||||||||||||
| HLA-G Reactivity | 4+ | 4+ | 7+ | 7+ | 2+ | 1 | 1 | 1 | 2 | 2+ | 2+ | 1 | 1+ | 11+ | 14 | 13+ | 9+ | 8 | 10 | 2 | 18+ | 21+ | 11 | 11 | 0+ | ||||||||||||
| HLA-A Reactivity | A*0101 | 2 | 1 | 5+ | 13 | 1 | 1 | 1 | 4 | 0 | 0 | 0 | |||||||||||||||||||||||||
| A*0201 | 3 | ||||||||||||||||||||||||||||||||||||
| A*0203 | 1 | 2+ | 2 | 2+ | 2+ | 2+ | 2 | 2 | 2 | 4+ | 2 | ||||||||||||||||||||||||||
| A*0206 | 1 | 3+ | 2+ | 3+ | 3+ | 3+ | 3 | 3 | 3 | 4+ | 0 | 0 | 2+ | ||||||||||||||||||||||||
| A*0301 | 3 | 7 | 5+ | 8+ | 5 | 5 | 3+ | 3 | 3+ | 10 | 5+ | ||||||||||||||||||||||||||
| A*1101 | 2 | 6 | 6 | 1 | 10 | 8 | 4 | 11 | 10 | 11+ | 11+ | 11 | 11+ | 11 | 11 | 11 | 4+ | 4+ | 4+ | 5 | 5 | 5+ | 5+ | 5+ | 5+ | 5 | 5+ | 7 | 6+ | 7 | 7+ | 9+ | 3+ | 4 | 10 | ||
| A*1102 | 3 | 1 | 1 | 1+ | 1 | 1 | 1 | 1 | 1 | 9 | 1 | ||||||||||||||||||||||||||
| A*2301 | 3+ | 9+ | 8 | 11 | 10 | 10+ | 10 | 10 | 10 | 11 | 8 | ||||||||||||||||||||||||||
| A*2402 | 2+ | 3 | 3 | 2 | 4+ | 14+ | 13 | 15+ | 14+ | 14+ | 14 | 14 | 14+ | 12+ | 1 | 1+ | 1 | 1 | 1+ | 1+ | 1 | 1 | 2+ | 2+ | 3 | 1+ | 2+ | 1+ | 1+ | 13 | |||||||
| A*2403 | 2+ | 2+ | 3 | 1+ | 4+ | 12+ | 11 | 13+ | 13+ | 13 | 13+ | 12+ | 13 | 12 | 1 | 1 | 1+ | 1 | 1 | 1 | 11 | ||||||||||||||||
| A*2501 | 1 | 4 | |||||||||||||||||||||||||||||||||||
| A*2601 | 3 | 1+ | 1 | 1 | 1+ | 1 | 1 | 1 | 1 | 1 | 8 | 2 | 2+ | 2+ | 2+ | 2 | 2+ | 1+ | 2 | 2 | 2 | 1 | |||||||||||||||
| A*2901 | 1+ | 1+ | 3 | 2 | 5 | 4+ | 6+ | 5 | 5 | 4+ | 4 | 4+ | 6+ | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 4+ | ||||||||||||||||
| A*2902 | 2 | 1 | 6+ | 5 | 7+ | 6 | 5+ | 4+ | 4 | 4+ | 6 | 0 | 0 | 5 | |||||||||||||||||||||||
| A*3001 | 2 | 1 | 5 | 5 | 4 | 6+ | 4+ | 4+ | 4 | 3+ | 4 | 12 | 1+ | 1 | 1+ | 1 | 1 | 4 | |||||||||||||||||||
| A*3002 | 1 | 1 | 3 | 2 | 3 | 7 | 6 | 8 | 6+ | 6+ | 6+ | 6 | 6+ | 9+ | 1 | 1 | 1 | 1 | 6 | ||||||||||||||||||
| A*3101 | 4 | 3 | 5 | 3 | 3+ | 2+ | 2 | 2 | 3 | 3 | |||||||||||||||||||||||||||
| A*3201 | 1 | 1 | 2 | 1+ | 3+ | 2+ | 4+ | 3 | 3 | 2+ | 2+ | 2+ | 5 | 1 | 1 | 1 | 1 | 1 | 2+ | ||||||||||||||||||
| A*3301 | 1 | 1 | 2+ | 1+ | 6+ | 5+ | 7+ | 6+ | 6+ | 5+ | 5 | 5+ | 5+ | 1 | 1 | 1 | 1 | 1 | 1 | 5+ | |||||||||||||||||
| A*3303 | 2 | 2 | 4 | 2+ | 1+ | 7 | 6+ | 8 | 7 | 7+ | 7+ | 7+ | 7+ | 8+ | 1 | 1 | 1 | 1+ | 1 | 1 | 1 | 1+ | 6+ | ||||||||||||||
| A*3401 | 1+ | 1+ | 6 | 3+ | 1+ | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 3+ | 1 | |||||||||||||||||||||||
| A*3402 | 1 | 4 | 4+ | 4 | 5 | 4+ | 5 | 5 | 4+ | 5 | 11+ | 1 | 0 | 1 | 4 | ||||||||||||||||||||||
| A*3601 | 2 | 2 | 5+ | 3+ | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8+ | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | ||||||||||||||||
| A*4301 | 1 | 4 | 2 | 5 | |||||||||||||||||||||||||||||||||
| A*6601 | 3+ | 1+ | 6+ | 5+ | 7+ | 6+ | 6+ | 5+ | 5 | 5 | 4+ | 5+ | |||||||||||||||||||||||||
| A*6602 | 1 | 3+ | 3 | 4+ | 3+ | 3+ | 3 | 3 | 3 | 2+ | 3 | ||||||||||||||||||||||||||
| A*6801 | 1 | 5+ | 4+ | 6+ | 5+ | 5+ | 5 | 4 | 4+ | 5 | 4+ | ||||||||||||||||||||||||||
| A*6802 | 1 | 1 | 2 | 1 | 2 | 1+ | 2 | 2 | 2 | 1+ | 1+ | 1+ | 8 | 1+ | 2 | 1+ | |||||||||||||||||||||
| A*6901 | 1+ | 6+ | 5 | 7+ | 5+ | 5 | 3+ | 3+ | 4 | 4+ | 5 | ||||||||||||||||||||||||||
| A*7401 | 1 | 2+ | 2 | 2+ | 2+ | 2+ | 2+ | 2 | 2+ | 10 | 1 | 1 | 1+ | 1 | 1 | 1 | 2 | ||||||||||||||||||||
| A*8001 | 2+ | 1 | 2 | 13 | 13 | 13+ | 13 | 13 | 13 | 12+ | 12+ | 9 | 13 | ||||||||||||||||||||||||
| HLA-B Reactivity | B*0702 | 1 | 4+ | 18 | 17+ | 18+ | 18 | 18 | 17+ | 17 | 18 | 13+ | 2 | 1+ | 2 | 17+ | |||||||||||||||||||||
| B*0801 | 2 | 1 | 7 | 2+ | 2 | 3 | 2+ | 2+ | 2+ | 2+ | 2+ | 15 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 2 | 2 | 2 | 19+ | 19+ | 19+ | 2+ | 24+ | 1+ | 2 | 2 | 2 | |||||
| B*1301 | 2 | 2+ | 5+ | 3+ | 8+ | 18+ | 19 | 19_ | 19 | 18+ | 18+ | 18 | 18+ | 16 | 2 | 2 | 2 | 2+ | 2+ | 2+ | 2+ | 2+ | 3+ | 3 | 3 | 19 | 20+ | 19+ | 4+ | 24 | 3+ | 3+ | 4 | 18 | |||
| B*1302 | 1 | 2 | 1 | 6 | 13+ | 12+ | 14+ | 13+ | 14 | 13+ | 13+ | 13+ | 13+ | 2 | 2 | 2 | 2+ | 2+ | 2+ | 2+ | 3 | 3 | 3 | 3+ | 6 | 6 | 6 | 3+ | 6+ | 2+ | 2+ | 12+ | |||||
| B*1401 | 2 | 7 | 7 | 2 | 11 | 8+ | 6 | 15+ | 14+ | 16 | 16 | 15+ | 16 | 16 | 15+ | 14 | 3 | 2+ | 3 | 1+ | 1 | 14+ | |||||||||||||||
| B*1402 | 1+ | 1+ | 4 | 2+ | 2 | 9+ | 8 | 10 | 10 | 10+ | 9+ | 9 | 9+ | 8+ | 12+ | 12+ | 12+ | 11+ | 8 | ||||||||||||||||||
| B*1501 | 1 | 4 | 5+ | 4+ | 6 | 5+ | 6 | 5+ | 5+ | 5+ | 11+ | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 16 | 16 | 16 | 2 | 18+ | 1+ | 2 | 4+ | ||||||||
| B*1502 | 3 | 3 | 6 | 4 | 6+ | 19 | 18+ | 19+ | 19+ | 19 | 19 | 18 | 18+ | 15 | 1 | 1+ | 1+ | 2+ | 2 | 2+ | 5 | 8 | 1 | 1 | 18+ | ||||||||||||
| B*1503 | 1 | 1 | 2+ | 1+ | 6+ | 16+ | 16+ | 17+ | 17+ | 17 | 17 | 16+ | 18+ | 14 | 1+ | 1+ | 1+ | 1 | 1 | 1 | 16+ | ||||||||||||||||
| B*1510 | 2+ | 1 | 1 | 8 | 7 | 8+ | 8 | 8 | 7+ | 7+ | 7+ | 6+ | 8 | 7+ | 8 | 5 | 7 | ||||||||||||||||||||
| B*1511 | 3 | 3+ | 9 | 5+ | 9 | 15 | 14+ | 16 | 15+ | 15+ | 15+ | 15 | 15+ | 16+ | 7+ | 7+ | 8 | 8+ | 8+ | 9 | 9 | 9 | 9 | 9 | 9+ | 10+ | 10 | 10+ | 10+ | 16 | 1 | 6 | 7 | 14+ | |||
| B*1512 | 1+ | 7 | 3+ | 3 | 4+ | 4 | 4 | 3+ | 3 | 3+ | 15 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 9 | 19 | 19+ | 2 | 24 | 2+ | 2 | 2+ | 3 | ||||||
| B*1513 | 2 | 2 | 5 | 3 | 6+ | 1+ | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 15 | 19 | 19+ | 18+ | 22+ | 4 | 1 | 1 | 1 | |||||||||||||||
| B*1516 | 2 | 2 | 5+ | 3 | 4+ | 15 | 13+ | 15+ | 15+ | 15 | 15 | 14+ | 14+ | 12 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 3 | 3 | 3 | 2+ | 3 | 1+ | 2 | 13+ | ||||
| B*1801 | 3+ | 3+ | 6+ | 4+ | 2 | 10+ | 10 | 11+ | 11 | 10+ | 10+ | 10+ | 10+ | 7+ | 10 | ||||||||||||||||||||||
| B*2705 | 2+ | 1+ | 1 | 8+ | 7+ | 8+ | 8+ | 8+ | 8 | 7+ | 8 | 6+ | 7+ | ||||||||||||||||||||||||
| B*2708 | 1 | 1 | 4 | 2+ | 4+ | 5+ | 4+ | 6 | 5+ | 5+ | 5+ | 5 | 5+ | 11+ | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 2+ | 2 | 2+ | 2 | 2+ | 2 | 2 | 4+ | ||||||
| B*3501 | 2 | 6+ | 6+ | 1 | 10 | 8+ | 7+ | 19 | 19 | 20 | 19+ | 18+ | 18 | 18 | 18 | 16 | 1 | 1 | 1+ | 1 | 1 | 1 | 3 | 3+ | 3+ | 8+ | 8 | 9 | 2+ | 8 | 9 | 2 | 2 | 19 | |||
| B*3701 | 2+ | 3 | 6 | 4 | 6 | 15+ | 15 | 16 | 16 | 16+ | 15+ | 15+ | 15+ | 14 | 1 | 1 | 1 | 1 | 1 | 15 | |||||||||||||||||
| B*3801 | 3+ | 1+ | 3+ | 13+ | 12+ | 14 | 13+ | 13+ | 13 | 12+ | 13+ | 11 | 2 | 2 | 3 | 3 | 3 | 4 | 4 | 4 | 4 | 5 | 12+ | ||||||||||||||
| B*3901 | 3+ | 3+ | 7 | 5 | 7+ | 19+ | 19 | 20+ | 20 | 19+ | 19+ | 18+ | 19 | 15+ | 1+ | 2 | 1+ | 2 | 2+ | 2 | 2+ | 3 | 19 | ||||||||||||||
| B*4001 | 2+ | 2+ | 5+ | 3+ | 4+ | 15+ | 15 | 16+ | 16 | 15+ | 16 | 15 | 15+ | 12+ | 1 | 1 | 1 | 1 | 1 | 2 | 1+ | 2 | 2 | 2 | 1 | 1 | 15 | ||||||||||
| B*4002 | 3+ | 3+ | 6 | 4+ | 6 | 4 | 3+ | 4+ | 4 | 4+ | 4 | 4 | 4 | 14 | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 1+ | 2 | 2 | 2+ | 3 | 3 | 3+ | 3+ | 4 | 2 | 2+ | 3+ | ||||
| B*4006 | 7 | 11 | 11 | 3 | 15+ | 13+ | 4+ | 3+ | 5+ | 4+ | 4+ | 4 | 3+ | 4 | 3+ | 1 | 1 | 1 | 1 | 1 | 1 | 3+ | |||||||||||||||
| B*4101 | 3+ | 3+ | 7 | 5 | 3 | 15+ | 15 | 15+ | 15+ | 15+ | 15 | 15 | 15 | 10+ | 15 | ||||||||||||||||||||||
| B*4201 | 3 | 9+ | 8+ | 11 | 10 | 10 | 9+ | 9 | 9 | 14+ | 1 | 1 | 1 | 1 | 1+ | 1+ | 1 | 1 | 2+ | 2+ | 3 | 3+ | 3+ | 3+ | 2+ | 9+ | 12+ | 2+ | 3 | 8+ | |||||||
| B*4402 | 1+ | 1+ | 7 | 4 | 1+ | 6+ | 6 | 7+ | 7 | 7 | 6 | 6 | 6 | 10 | 1+ | 1+ | 1+ | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 3+ | 3+ | 3+ | 3+ | 4+ | 2 | 2+ | 6 | ||||
| B*4403 | 4 | 4 | 7 | 5+ | 3+ | 1 | 1 | 11 | 1 | ||||||||||||||||||||||||||||
| B*4501 | 1 | 5 | 5+ | 9+ | 7+ | 6+ | 19 | 19 | 20+ | 19+ | 19 | 19 | 19 | 19 | 16 | 1 | 19+ | 21 | 20 | 1+ | 25 | 2 | 2 | 2 | 19 | ||||||||||||
| B*4601 | 2+ | 2+ | 6 | 4 | 6+ | 17+ | 17 | 19 | 17+ | 18 | 17+ | 17 | 18 | 14+ | 1 | 1 | 1+ | 1 | 2 | 1 | 1 | 1 | 17 | ||||||||||||||
| B*4701 | 2 | 2 | 6+ | 3+ | 8+ | 20 | 20 | 21 | 19+ | 19+ | 19 | 19 | 19 | 16 | 1+ | 2 | 2+ | 3+ | 3 | 3+ | 2 | 2 | 1+ | 1+ | 20 | ||||||||||||
| B*4801 | 1 | 1 | 4 | 2+ | 7 | 14+ | |||||||||||||||||||||||||||||||
| B*4901 | 5+ | 13+ | |||||||||||||||||||||||||||||||||||
| B*5001 | 8 | 4 | 3+ | 4+ | 4+ | 4+ | 4+ | 4 | 4+ | 15+ | 1 | 1 | 1 | 1 | 1 | 1+ | 1 | 1 | 1+ | 1+ | 1+ | 5 | 4+ | 5 | 2+ | 3+ | 2+ | 2+ | 3+ | ||||||||
| B*5101 | 1+ | 1+ | 6 | 3+ | 7+ | 1+ | 1+ | 2 | 1+ | 1+ | 1_ | 1+ | 1+ | 15 | 2+ | 2 | 2+ | 1 | 1 | 1+ | 1+ | 1+ | |||||||||||||||
| B*5102 | 2 | 2 | 5 | 3+ | 7 | 2 | 1+ | 2 | 2 | 2 | 2 | 2 | 2 | 15+ | 3 | 2+ | 3+ | 1 | 1 | 1 | 1+ | ||||||||||||||||
| B*5201 | 1+ | 1+ | 4+ | 2+ | 9+ | 10 | 9 | 11 | 10+ | 10 | 10 | 9+ | 10 | 15+ | 5+ | 5+ | 5+ | 6 | 6 | 6+ | 6+ | 6+ | 6 | 6+ | 6+ | 8+ | 9 | 9 | 8 | 12 | 5+ | 6+ | 9 | ||||
| B*5301 | 1 | 5+ | 5+ | 8+ | 7 | 6+ | 2+ | 2+ | 3 | 2+ | 2+ | 2+ | 2+ | 2+ | 14 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2+ | 1+ | 2 | 2+ | |||
| B*5401 | 3 | 3+ | 5+ | 4 | 5 | 2+ | 2+ | 3 | 3 | 3 | 2+ | 2+ | 2+ | 12+ | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 3+ | 3 | 3+ | 1 | 3 | 1+ | 1+ | 2+ | ||||
| B*5501 | 2+ | 1+ | 2 | 2+ | 2 | 2+ | 2+ | 2+ | 2 | 2 | 2 | 8 | 1 | 1 | 1 | 1 | 1 | 2 | |||||||||||||||||||
| B*5601 | 1 | 2+ | 4+ | 3+ | 4+ | 4+ | 4+ | 4 | 4 | 4+ | 10 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 12+ | 12+ | 12+ | 2 | 13+ | 1 | 1 | 3+ | |||||||||||
| B*5701 | 5 | 5+ | 4+ | 6 | 5+ | 5+ | 5+ | 5+ | 5+ | 13 | 1 | 1 | 1 | 1 | 1 | 1+ | 1 | 1 | 1+ | 1+ | 1+ | 17+ | 18 | 17+ | 2+ | 21+ | 2 | 2 | 4+ | ||||||||
| B*5703 | 1 | 7 | 12+ | 11+ | 13 | 12+ | 12+ | 12+ | 12+ | 12+ | 13+ | 5 | 5+ | 5+ | 6 | 6 | 6+ | 6+ | 6 | 6+ | 6 | 6+ | 8 | 8+ | 8 | 8 | 12+ | 1 | 5 | 5+ | 11+ | ||||||
| B*5801 | 2 | 6 | 6+ | 1 | 10 | 8 | 9 | 2+ | 2 | 2+ | 2+ | 2+ | 2+ | 2+ | 2 | 17 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 2+ | 2+ | 2+ | 6 | 5+ | 6 | 2+ | 5 | 1 | 2+ | 3 | 2 | |
| B*5901 | 1 | 1 | 5+ | 3 | 2 | 9 | 8 | 10 | 9+ | 9+ | 9 | 9 | 9 | 8+ | 8 | ||||||||||||||||||||||
| B*6701 | 4+ | 4+ | 4 | 5 | 4+ | 4+ | 4+ | 4+ | 4+ | 11 | 2 | 2 | 2 | 2 | 2 | 1 | 1+ | 4 | |||||||||||||||||||
| B*7301 | 1 | 1 | 3 | 2 | 6+ | 4 | 3+ | 4 | 4 | 4 | 4 | 3+ | 4 | 14 | 1 | 1 | 1 | 3 | 3 | 3 | 1+ | 2 | 1+ | 1+ | 3+ | ||||||||||||
| B*7801 | 3 | 3+ | 6 | 4+ | 5 | 18+ | 17+ | 19 | 18+ | 18+ | 17+ | 17+ | 18 | 13+ | 1 | 1 | 1 | 17+ | |||||||||||||||||||
| B*8101 | 1 | 7+ | 19+ | 19+ | 20+ | 19+ | 19+ | 18+ | 19+ | 18+ | 16 | 1 | 1 | 1 | 1+ | 1+ | 1 | 1 | 2 | 2+ | 2+ | 4+ | 4+ | 5 | 2+ | 5+ | 4 | 2 | 2 | 19+ | |||||||
| B*8201 | 3 | 2+ | 4 | 3 | 13 | 20 | 19 | 20+ | 20 | 19+ | 19+ | 19+ | 19+ | 18+ | 1+ | 1+ | 1+ | 1 | 1 | 1 | 1+ | 2+ | 3 | 3 | 3+ | 19 | |||||||||||
| HLA-C Reactivity | Cw*0102 | 7 | 3 | 11+ | 20 | 29 | 20+ | 20+ | 20 | 20 | 19 | 19+ | 18+ | 1 | 1 | 1 | 2 | 2 | 2 | 2+ | 19 | ||||||||||||||||
| Cw*0202 | 2+ | 2+ | 10+ | 6 | 11 | 15+ | 14 | 16+ | 15+ | 15+ | 11 | 10+ | 11 | 16 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1+ | 2 | 2 | 14 | ||||||||||||
| Cw*0302 | 1+ | 1+ | 5+ | 3 | 10 | 15+ | 1+ | 2 | |||||||||||||||||||||||||||||
| Cw*0303 | 2 | 2+ | 7 | 4 | 9+ | 7+ | 6+ | 8+ | 8 | 7+ | 7 | 6+ | 7 | 15+ | 1 | 1 | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 1+ | 1+ | 2 | 2+ | 2+ | 3 | 6+ | |||||||
| Cw*0304 | 2 | 2 | 6+ | 3+ | 11 | 2 | 1+ | 2+ | 2+ | 2+ | 5+ | 5 | 5 | 17 | 1 | 1+ | 1+ | 1+ | 2+ | 3+ | 1+ | 2 | 1+ | ||||||||||||||
| Cw*0401 | 2+ | 1 | 9+ | 18 | 18 | 19+ | 19+ | 19 | 18 | 18+ | 18 | 15+ | 5 | 5 | 5+ | 5+ | 6 | 6 | 6+ | 6 | 6 | 6 | 6 | 6+ | 6+ | 6+ | 7+ | 12+ | 2 | 5+ | 6 | 18 | |||||
| Cw*0501 | 4 | 8 | 8+ | 2 | 16 | 13 | 13 | 7 | 6+ | 8 | 7+ | 7+ | 12 | 12 | 12+ | 18+ | 2+ | 3+ | 3+ | 1 | 1 | 1 | 1 | 5+ | 13+ | 2+ | 2+ | 6+ | |||||||||
| Cw*0602 | 1 | 1 | 9 | 4 | 16+ | 5 | 4+ | 6 | 5+ | 5 | 10 | 9+ | 10 | 18+ | 1+ | 2 | 2 | 1 | 4+ | 9+ | 6 | 7 | 4+ | ||||||||||||||
| Cw*0702 | 12 | 6+ | 10 | 19+ | 17+ | 19+ | 19 | 19+ | 18+ | 18+ | 19 | 17 | 4 | 5 | 5 | 5+ | 5+ | 5+ | 6 | 6 | 6+ | 6+ | 10+ | 1 | 5+ | 6 | 17+ | ||||||||||
| CW*0801 | 3 | 6 | 6+ | 1+ | 13 | 10+ | 11 | 21+ | 20 | 22 | 21 | 21 | 21 | 21 | 21 | 19 | 1 | 1+ | 1+ | 2+ | 5 | 1 | 1+ | 20+ | |||||||||||||
| Cw*1203 | 5 | 2 | 12 | 19+ | 19+ | 21 | 20 | 19+ | 19+ | 19 | 19 | 17 | 1 | 1 | 1+ | 2 | 2 | 1+ | 1 | 1+ | 2 | 3 | 2+ | 2+ | 3 | 19+ | |||||||||||
| Cw*1402 | 2 | 2 | 8+ | 4+ | 9 | 10+ | 9+ | 11 | 10+ | 10 | 9+ | 9+ | 9+ | 16+ | 1+ | 1+ | 2+ | 3 | 3 | 2 | 2 | 2+ | 3 | 5 | 2+ | 2 | 2+ | 9+ | |||||||||
| Cw*1502 | 1+ | 1+ | 6 | 3 | 11 | 20+ | 20+ | 22 | 20+ | 20 | 20 | 19+ | 19+ | 18+ | 1 | 1 | 1 | 1+ | 1+ | 1+ | 1+ | 2 | 2 | 1 | 1+ | 4 | 7+ | 2+ | 3 | 20+ | |||||||
| Cw*1601 | 1 | 1+ | 8 | 4 | 15+ | 19+ | 19+ | 21 | 19+ | 19 | 18+ | 19 | 18+ | 18 | 1 | 1+ | 1+ | 1+ | 1 | 1 | 1 | 2 | 2+ | 3+ | 6 | 7 | 19+ | ||||||||||
| Cw*1701 | 1+ | 1+ | 13+ | 9 | 11 | 13 | 13 | 14 | 13+ | 14 | 14 | 14+ | 14+ | 17 | 6 | 6 | 6 | 7 | 7 | 7+ | 7+ | 7+ | 7+ | 7+ | 7+ | 8 | 8 | 8 | 9 | 15 | 4 | 6 | 6+ | 13 | |||
| Cw*1802 | 8 | 11 | 11+ | 4 | 17+ | 15 | 8+ | 9+ | 8+ | 10 | 9+ | 9+ | 9 | 9 | 9 | 16+ | 1+ | 1+ | 2 | 2 | 2 | 2+ | 2 | 2 | 3+ | 3+ | 3+ | 19+ | 20+ | 10+ | 4 | 24+ | 3 | 3+ | 4 | 6+ | |
| HLA-I Reactivity with TFL-006 (MFI) at 20 mG/mL * | |||||||
|---|---|---|---|---|---|---|---|
| HLA-I | LABSCreen | LIFECODES | HLA-I | LABSCreen | LIFECODES | ||
| (Lot # 10) | (Lot # 11) | 3005613 | (Lot # 10) | (Lot # 11) | 3005613 | ||
| A*01:01 | 933 | 852 | 0 | B*07:02 | 862 | 670 | 0 |
| A*02:01 | 339 | 542 | 0 | B*08:01 | 1226 | 1204 | 0 |
| A*02:03 | 1018 | 156 | 0 | B*13:01 | n/a | 675 | |
| A*02:06 | n/a | 616 | B*13:02 | 2514 | 4923 | 0 | |
| A*03:01 | 193 | 210 | 0 | B*14:01 | 7805 | 1983 | 0 |
| A*11:01 | 4782 | 3900 | 0 | B*14:02 | 1831 | 197 | 0 |
| A*11:02 | 537 | 321 | 0 | B*15:01 | 335 | 2541 | 0 |
| A*23:01 | 133 | 125 | 0 | B*15:02 | 1935 | 809 | 0 |
| A*24:02 | 716 | 580 | 0 | B*15:03 | 1822 | 1146 | 0 |
| A*24:03 | 2516 | 1042 | 0 | B*15:10 | n/a | 579 | |
| A*25:01 | 194 | 349 | 0 | B*15:11 | n/a | 1658 | |
| A*26:01 | 2221 | 2273 | 0 | B*15:12 | 770 | 1875 | 0 |
| A*29:01 | 1017 | 1667 | 0 | B*15:13 | 3135 | 2437 | 0 |
| A*29:02 | 778 | 715 | 0 | B*15:16 | 3076 | 882 | 0 |
| A*30:01 | 1496 | 1243 | 0 | B*18:01 | 3096 | 2891 | 0 |
| A*30:02 | n/a | 728 | B*27:05 | 634 | 3252 | 0 | |
| A*31:01 | 396 | 687 | 0 | B*27:08 | 1659 | 2095 | 0 |
| A*32:01 | 515 | 341 | 0 | B*35:01 | 6128 | 517 | 0 |
| A*33:01 | 1038 | 574 | 0 | B*37:01 | 2650 | 4352 | 0 |
| A*33:03 | 554 | 1039 | 0 | B*38:01 | 2521 | 2425 | 0 |
| A*34:01 | n/a | 3002 | B*39:01 | 704 | 4447 | 0 | |
| A*34:02 | 1535 | 1165 | 0 | B*40:01 | 3429 | 330 | 0 |
| A*36:01 | 1353 | 941 | 0 | B*40:02 | 2697 | 3354 | 0 |
| A*43:01 | 2479 | 2768 | 0 | B*40:06 | n/a | 1869 | |
| A*66:01 | 1886 | 2145 | 0 | B*41:01 | 3739 | 1364 | 0 |
| A*66:02 | 1454 | 1327 | 0 | B*42:01 | 347 | 957 | 0 |
| A*68:01 | 713 | 454 | 0 | B*44:02 | 3650 | 892 | 0 |
| A*68:02 | 1185 | 578 | 0 | B*44:03 | 1829 | 2516 | 0 |
| A*69:01 | 3128 | 1786 | 0 | B*45:01 | 1736 | 2568 | 0 |
| A*74:01 | 652 | 603 | 0 | B*46:01 | 3572 | 1415 | 0 |
| A*80:01 | 3132 | 3758 | 0 | B*47:01 | 2152 | 1859 | 0 |
| B*48:01 | 3262 | 2418 | 0 | ||||
| B*49:01 | 1554 | 3042 | 0 | ||||
| 0 | B*50:01 | 1799 | 4246 | 0 | |||
| C*01:02 | 4066 | 2820 | 0 | B*51:01 | 2461 | 2413 | 0 |
| C*02:02 | 7446 | 3403 | B*51:02 | n/a | 696 | ||
| C*03:02 | n/a | 6614 | 0 | B*52:01 | 2146 | 1745 | 0 |
| C*03:03 | 2458 | 3071 | 0 | B*53:01 | 5442 | 695 | 0 |
| C*03:04 | 4504 | 8167 | 0 | B*54:01 | 1662 | 2427 | 0 |
| C*04:01 | 3337 | 4182 | 0 | B*55:01 | 2519 | 4867 | 0 |
| C*05:01 | 9124 | 2567 | 0 | B*56:01 | 3662 | 3084 | 0 |
| C*06:02 | 5644 | 5794 | 0 | B*57:01 | 2089 | 141 | 0 |
| C*07:02 | 8702 | 1689 | 0 | B*57:03 | n/a | 1878 | |
| C*08:01 | 6090 | 4479 | B*58:01 | 5268 | 3190 | 0 | |
| C*12:03 | n/a | 6731 | 0 | B*59:01 | 3553 | 1278 | 0 |
| C*14:02 | 3937 | 3627 | 0 | B*67:01 | 406 | 3520 | 0 |
| C*15:02 | 4465 | 4651 | 0 | B*73:01 | 1423 | 3565 | 0 |
| C*16:01 | 4648 | 2706 | 0 | B*78:01 | 2996 | 5271 | 0 |
| C*17:01 | 8296 | 5760 | 0 | B*81:01 | 1525 | 2446 | 0 |
| C*18:02 | n/a | 829 | B*82:02 | n/a | 1959 | ||
| HLA Alleles | ID of Non-Alloimmunized Healthy Males | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Values Expressed as Mean Fluorescent Intensity (MFI) | ||||||||||||
| TJ | HR | ME | RC | TP | CM | MR | TS | NR | VJ | HO | ||
| HLA-A Reactivity | A*1101 | 769 | 898 | |||||||||
| A*2301 | 609 | 972 | ||||||||||
| A*2402 | 616 | 726 | 869 | 632 | 1099 | 738 | 784 | 1021 | ||||
| A*2403 | 557 | 599 | ||||||||||
| A*2501 | 906 | |||||||||||
| A*2601 | 835 | 613 | 1094 | |||||||||
| A*2901 | 1013 | |||||||||||
| A*2902 | 715 | 2224 | ||||||||||
| A*3001 | 540 | |||||||||||
| A*3101 | 578 | |||||||||||
| A*3201 | 539 | 1511 | ||||||||||
| A*3301 | 502 | 1260 | ||||||||||
| A*3303 | 712 | |||||||||||
| A*3401 | 958 | 710 | 1867 | 749 | 2070 | |||||||
| A*3601 | ||||||||||||
| A*4301 | 779 | 562 | 1660 | |||||||||
| A*6601 | 2535 | 1387 | ||||||||||
| A*6602 | 1830 | 1382 | ||||||||||
| A*6802 | 818 | 946 | ||||||||||
| A*6901 | 628 | 618 | 581 | 523 | ||||||||
| A*7401 | 716 | |||||||||||
| A*8001 | 595 | 919 | ||||||||||
| HLA-B Reactivity | B*1301 | 556 | 564 | 969 | 737 | 722 | 1025 | |||||
| B*1302 | 582 | 594 | ||||||||||
| B*1401 | 524 | 518 | 704 | 506 | 602 | 976 | 830 | 944 | 911 | |||
| B*1402 | 552 | 556 | 648 | 624 | ||||||||
| B*1502 | 618 | 523 | 573 | |||||||||
| B*1503 | 535 | |||||||||||
| B*1510 | 578 | 1089 | ||||||||||
| B*1511 | 592 | 634 | 703 | 667 | 862 | 1113 | 882 | 1014 | 1268 | |||
| B*1512 | 562 | 1923 | 564 | 2484 | 501 | |||||||
| B*1513 | 507 | 745 | 757 | 713 | 808 | |||||||
| B*1516 | 596 | 572 | 571 | 1009 | 750 | 1341 | 2200 | |||||
| B*3501 | 727 | |||||||||||
| B*3701 | 748 | 550 | 1374 | 881 | 1135 | 725 | 5103 | 1203 | 1215 | 1901 | ||
| B*3801 | 559 | |||||||||||
| B*4001 | 739 | |||||||||||
| B*4006 | 746 | 976 | 932 | 578 | 1196 | 1586 | 1288 | 1211 | 1749 | |||
| B*4402 | 576 | 2027 | 546 | 1375 | 713 | 688 | 779 | |||||
| B*4403 | 1777 | |||||||||||
| B*4501 | 673 | 2044 | 506 | |||||||||
| B*4601 | 2821 | 442 | 941 | 741 | 549 | 558 | 871 | 842 | ||||
| B*4701 | 929 | |||||||||||
| B*4801 | 697 | 609 | 648 | 537 | ||||||||
| B*5101 | 626 | 658 | 687 | 718 | ||||||||
| B*5102 | 525 | 656 | 748 | |||||||||
| B*5201 | 836 | 565 | 672 | 1281 | ||||||||
| B*5301 | 816 | 693 | 748 | 842 | ||||||||
| B*5401 | 565 | 627 | ||||||||||
| B*5501 | 548 | 541 | 604 | |||||||||
| B*5601 | 537 | 1197 | ||||||||||
| B*5701 | ||||||||||||
| B*5703 | 632 | 829 | ||||||||||
| B*5801 | 659 | 669 | 653 | 960 | 885 | 934 | 1193 | |||||
| B*5901 | 591 | 552 | 658 | 675 | 887 | 900 | ||||||
| B*6701 | ||||||||||||
| B*7301 | 583 | |||||||||||
| B*7801 | 812 | 574 | 680 | 648 | ||||||||
| B*8101 | 807 | 543 | ||||||||||
| B*8201 | 799 | 942 | 3259 | 989 | 1041 | 994 | 993 | |||||
| HLA-C Reactivity | C*0102 | 619 | 1301 | 676 | 1421 | 1588 | 1451 | 1682 | 1796 | 1876 | 1774 | 2602 |
| C*0202 | 680 | 1314 | 877 | 1286 | 1823 | 1032 | 1758 | 2080 | 1833 | 1824 | 2508 | |
| C*0302 | 549 | 1397 | 759 | 1226 | 1280 | 886 | 1291 | 1913 | 1897 | 1351 | 1893 | |
| C*0303 | 1442 | 607 | 937 | 1073 | 977 | 1376 | 1417 | 1151 | 1619 | |||
| C*0304 | 620 | 1438 | 689 | 1211 | 1395 | 668 | 1309 | 2017 | 1743 | 1487 | 2209 | |
| C*0401 | 907 | 1602 | 1299 | 2038 | 2427 | 1113 | 2130 | 2815 | 2610 | 2148 | 4036 | |
| C*0501 | 628 | 1149 | 1078 | 1268 | 1406 | 659 | 1814 | 2123 | 1851 | 1529 | 1762 | |
| C*0602 | 1326 | 2657 | 1445 | 2520 | 3017 | 1572 | 2675 | 3410 | 2743 | 2945 | 2819 | |
| C*0702 | 1120 | 1649 | 1520 | 2276 | 2748 | 1426 | 2801 | 3562 | 3441 | 2760 | 4098 | |
| C*0801 | 855 | 926 | 1027 | 1117 | 1423 | 1274 | 1281 | 1514 | ||||
| C*1203 | 550 | 920 | 672 | 1187 | 1402 | 1207 | 1430 | 1631 | 1502 | 1590 | 1820 | |
| C*1402 | 792 | 1609 | 1100 | 1778 | 2045 | 1155 | 2019 | 2669 | 2386 | 2023 | 3444 | |
| C*1502 | 701 | 509 | 915 | 1119 | 1086 | 1025 | 1348 | 1122 | 1364 | 1489 | ||
| C*1601 | 652 | 1249 | 1064 | 1362 | 1596 | 877 | 1510 | 2044 | 1731 | 2038 | 2093 | |
| C*1701 | 720 | 1396 | 1043 | 1719 | 2197 | 763 | 1735 | 2683 | 2120 | 1792 | 1885 | |
| C*1802 | 1175 | 1878 | 1634 | 2135 | 2386 | 1549 | 2605 | 3014 | 2932 | 2463 | 2695 | |
| Mean Fluorescent Intensity (MFI) of Anti-HLA-Ia IgG Antibodies in Melanoma Patients before and 4 Weeks after Autologus Whole Cell Cancer Vaccine | |||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Patient ID | Before and after 4 Wks Post vaccine | ER*0101 | HLA-B Reactivity | HLA-C Reactivity | |||||||||||||||||||||||||||||||||||||
| B*0702 | B*1301 | B*1401 | B*1512 | B*1516 | B*2705 | B*2705 | B*2708 | B*3701 | B*4001 | B*4002 | B*4006 | B*4201 | B*4402 | B*4701 | B*5401 | B*5501 | B*5601 | B*5701 | B*5703 | B*6701 | B*8101 | B*8201 | C*0101 | C*0202 | C*0302 | C*0303 | C*0304 | C*0401 | C*0501 | C*0602 | C*0702 | C*0801 | C*1203 | C*1402 | C*1502 | C*1601 | C*1701 | C*1801 | |||
| HCC-1763 | PreVax | 550 | 590 | ||||||||||||||||||||||||||||||||||||||
| PostVax | 830 | 1330 | |||||||||||||||||||||||||||||||||||||||
| HCC-1766 | PreVax | 3140 | |||||||||||||||||||||||||||||||||||||||
| PostVax | 3830 | 700 | |||||||||||||||||||||||||||||||||||||||
| HCC-1937 | PreVax | 3710 | |||||||||||||||||||||||||||||||||||||||
| PostVax | 4040 | 570 | 940 | 650 | 550 | 670 | 1107 | ||||||||||||||||||||||||||||||||||
| HCC-1896 | PreVax | 3710 | 3340 | 1490 | 1740 | 1080 | 980 | 1430 | 1150 | 820 | 1210 | 3250 | 1170 | ||||||||||||||||||||||||||||
| PostVax | 4520 | 3610 | 1720 | 1820 | 990 | 1030 | 1480 | 1190 | 830 | 1240 | 3460 | 1210 | |||||||||||||||||||||||||||||
| HCC-2030 | PreVax | 1650 | 540 | 12,330 | 1180 | 540 | 530 | 8760 | 540 | 570 | 3460 | 6180 | 540 | 6140 | 750 | 750 | 930 | 500 | 700 | 2180 | 950 | 1500 | 660 | 600 | 1830 | 910 | |||||||||||||||
| PostVax | 8620 | 640 | 1230 | 12,700 | 2240 | 790 | 760 | 9040 | 830 | 950 | 3190 | 5520 | 670 | 5340 | 1580 | 1340 | 1901 | 1030 | 690 | 1330 | 2560 | 1690 | 2940 | 650 | 1240 | 630 | 2730 | 1300 | |||||||||||||
| HCC-1957 | PreVax | 3870 | 540 | 640 | 1250 | 570 | 1080 | 1020 | 540 | 770 | 1150 | 580 | 560 | ||||||||||||||||||||||||||||
| PostVax | 4990 | 1050 | 1370 | 2550 | 1170 | 2250 | 1933 | 700 | 1620 | 880 | 1370 | 2040 | 850 | 850 | |||||||||||||||||||||||||||
| Patient ID | 10DM | 17LE | 12DM | 20HN | 5DM | 19HN | 1DM | 11DM | |
|---|---|---|---|---|---|---|---|---|---|
| No of Alleles | HLA-A | 1 | 9 | 3 | 6 | 10 | 2 | 10 | 22 |
| HLA-B | 11 | 4 | 15 | 14 | 10 | 16 | 12 | 4 | |
| HLA-C | 2 | 1 | 1 | 0 | 0 | 2 | 2 | 0 | |
| HLA-A Reactivity | A*01:01 | 9470 | 1694 | 1255 | 14,961 | 3937 | |||
| A*02:01 | 1364 | 1894 | 872 | ||||||
| A*02:03 | 1034 | 1809 | 690 | ||||||
| A*03:01 | 5139 | ||||||||
| A*11:01 | 4621 | ||||||||
| A*11:02 | 4519 | ||||||||
| A*23:01 | 1783 | 1521 | 13,902 | 15,118 | |||||
| A*24:02 | 1985 | 1527 | 13,190 | 14,810 | |||||
| A*24:03 | 754 | 1395 | 11,895 | 689 | |||||
| A*25:01 | 1010 | 4204 | |||||||
| A*26:01 | 4826 | ||||||||
| A*29:02 | 1634 | 3543 | 1090 | 3014 | 1596 | 4622 | |||
| A*30:01 | 4679 | ||||||||
| A*31:01 | 3733 | ||||||||
| A*32:01 | 4744 | ||||||||
| A*33:01 | 1831 | 4431 | |||||||
| A*33:03 | 5485 | ||||||||
| A*34:02 | 7463 | ||||||||
| A*36:01 | 2587 | 5234 | |||||||
| A*43:01 | 661 | 3826 | |||||||
| A*66:01 | 6275 | ||||||||
| A*66:02 | 6318 | ||||||||
| A*68:01 | 1980 | 537 | 5555 | ||||||
| A*68:02 | 823 | 2054 | 619 | 6220 | |||||
| A*69:01 | 639 | 1557 | 5415 | ||||||
| A*74:01 | 5083 | ||||||||
| A*80:01 | 537 | 2209 | 3355 | 1010 | |||||
| HLA-B Reactivity | B*07:02 | 2638 | 5808 | 2593 | |||||
| B*08:01 | 1460 | ||||||||
| B*15:12 | 517 | 1509 | 1304 | ||||||
| B*15:16 | 2057 | 4253 | 3698 | ||||||
| B*27:05 | 932 | 1650 | 591 | 2757 | |||||
| B*27:08 | 1459 | 1232 | |||||||
| B*37:01 | 808 | 1495 | 2849 | 1095 | |||||
| B*38:01 | 1073 | 1405 | 2923 | 1204 | |||||
| B*39:01 | 917 | 1559 | |||||||
| B*40:01 | 2573 | 4726 | 583 | 549 | |||||
| B*40:02 | 841 | 710 | 982 | 17,106 | 17,617 | ||||
| B*41:01 | 1288 | 1142 | 15,827 | 17,864 | |||||
| B*42:01 | 1807 | 3532 | 776 | 1469 | |||||
| B*44:02 | 796 | 549 | 1373 | 2117 | |||||
| B*44:03 | 1888 | 1532 | 2212 | ||||||
| B*46:01 | 613 | 998 | |||||||
| B*47:01 | 1447 | 3431 | |||||||
| B*48:01 | 1612 | 3014 | |||||||
| B*49:01 | 1248 | 2768 | |||||||
| B*50:01 | 1488 | 1351 | |||||||
| B*51:01 | 589 | 2013 | |||||||
| B*52:01 | 1606 | 3103 | |||||||
| B*53:01 | 611 | 1740 | 1235 | ||||||
| B*54:01 | 760 | 1909 | 1375 | ||||||
| B*55:01 | 552 | 1666 | 1493 | ||||||
| B*56:01 | 1260 | 1578 | |||||||
| B*57:01 | 2252 | 4064 | 503 | ||||||
| B*59:01 | 2527 | 5685 | 2546 | ||||||
| B*67:01 | 1198 | 1015 | 2872 | 17,248 | 17,353 | ||||
| HLA-C | C*03:03 | 611 | |||||||
| C*15:02 | 1283 | 1630 | 1300 | ||||||
| C*17:01 | 1048 | 1365 | 2695 | 997 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravindranath, M.H.; Ravindranath, N.M.; El Hilali, F.; Selvan, S.R.; Filippone, E.J. Ramifications of the HLA-I Allelic Reactivity of Anti-HLA-E*01:01 and Anti-HLA-E*01:03 Heavy Chain Monoclonal Antibodies in Comparison with Anti-HLA-I IgG Reactivity in Non-Alloimmunized Males, Melanoma-Vaccine Recipients, and End-Stage Renal Disease Patients. Antibodies 2022, 11, 18. https://doi.org/10.3390/antib11010018
Ravindranath MH, Ravindranath NM, El Hilali F, Selvan SR, Filippone EJ. Ramifications of the HLA-I Allelic Reactivity of Anti-HLA-E*01:01 and Anti-HLA-E*01:03 Heavy Chain Monoclonal Antibodies in Comparison with Anti-HLA-I IgG Reactivity in Non-Alloimmunized Males, Melanoma-Vaccine Recipients, and End-Stage Renal Disease Patients. Antibodies. 2022; 11(1):18. https://doi.org/10.3390/antib11010018
Chicago/Turabian StyleRavindranath, Mepur H., Narendranath M. Ravindranath, Fatiha El Hilali, Senthamil R. Selvan, and Edward J. Filippone. 2022. "Ramifications of the HLA-I Allelic Reactivity of Anti-HLA-E*01:01 and Anti-HLA-E*01:03 Heavy Chain Monoclonal Antibodies in Comparison with Anti-HLA-I IgG Reactivity in Non-Alloimmunized Males, Melanoma-Vaccine Recipients, and End-Stage Renal Disease Patients" Antibodies 11, no. 1: 18. https://doi.org/10.3390/antib11010018
APA StyleRavindranath, M. H., Ravindranath, N. M., El Hilali, F., Selvan, S. R., & Filippone, E. J. (2022). Ramifications of the HLA-I Allelic Reactivity of Anti-HLA-E*01:01 and Anti-HLA-E*01:03 Heavy Chain Monoclonal Antibodies in Comparison with Anti-HLA-I IgG Reactivity in Non-Alloimmunized Males, Melanoma-Vaccine Recipients, and End-Stage Renal Disease Patients. Antibodies, 11(1), 18. https://doi.org/10.3390/antib11010018






