Therapeutic Efficacy of Anti-Bestrophin Antibodies against Experimental Filariasis: Immunological, Immune-Informatics and Immune Simulation Investigations
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Clearance
2.2. Antibodies and Consumables
2.3. Parasite
2.4. Development of Antibody
2.5. Experimental Infection Model of Filariasis
2.6. Treatment Schedule
2.7. Determination of the Inflammatory Parameters
2.8. Statistical Analysis
2.9. Immuno-Informatics and Immune Simulation Studies
2.10. Prediction of Linear B-Cell Epitopes
2.11. Prediction of T-Cell Epitopes
2.12. Prediction of Antigenicity
2.13. Design of Multi-Epitope Construct
2.14. Immune Simulation
3. Results
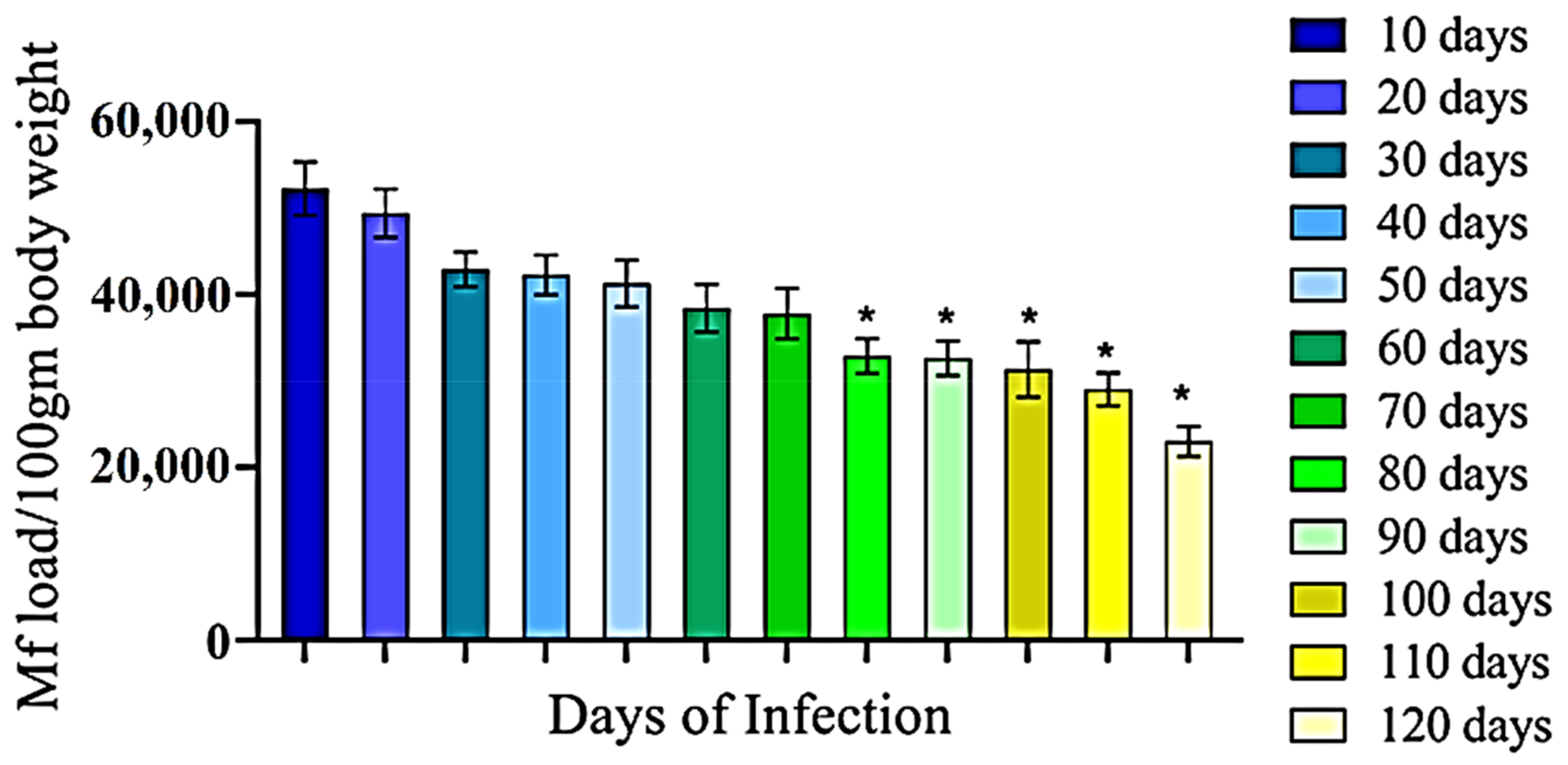
3.1. Development of Filarial Infection in Rat Model
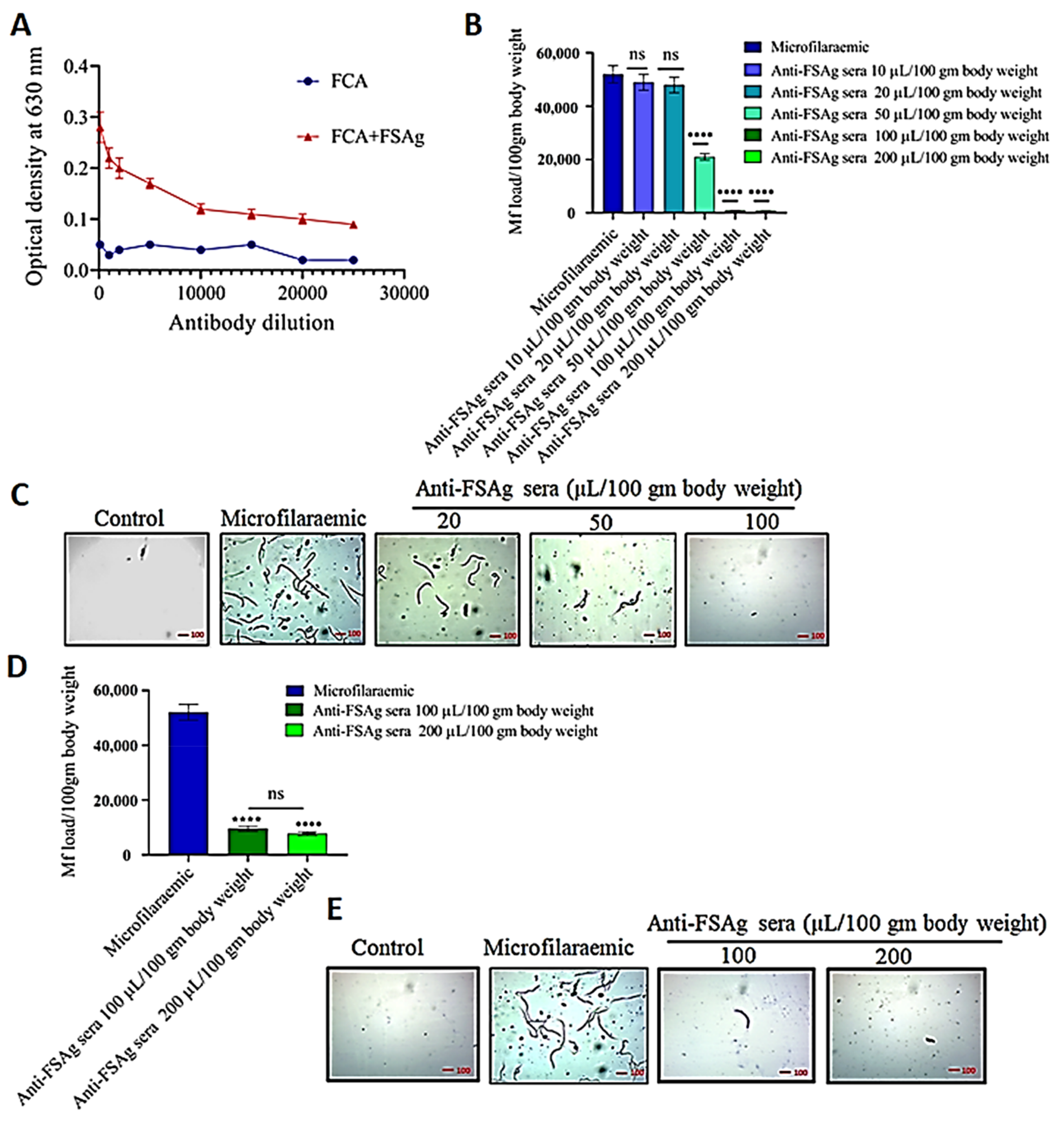
3.2. Anti-Bestrophin Antibody Reduces Microfilarial Load
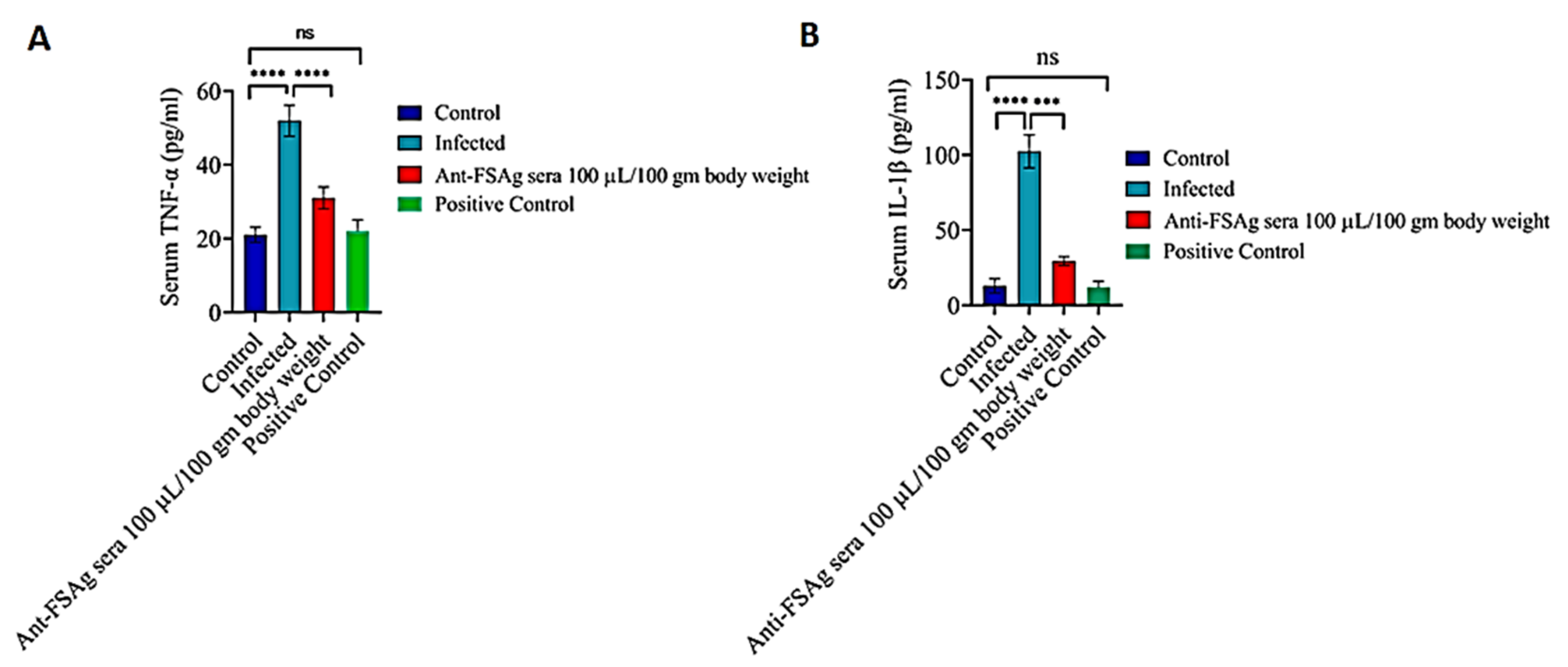
3.3. Anti-Bestrophin Antibody Reduces Microfilaria-Induced Inflammation
3.4. FSAg Possesses Both B-Cell and T-Cell Epitopes
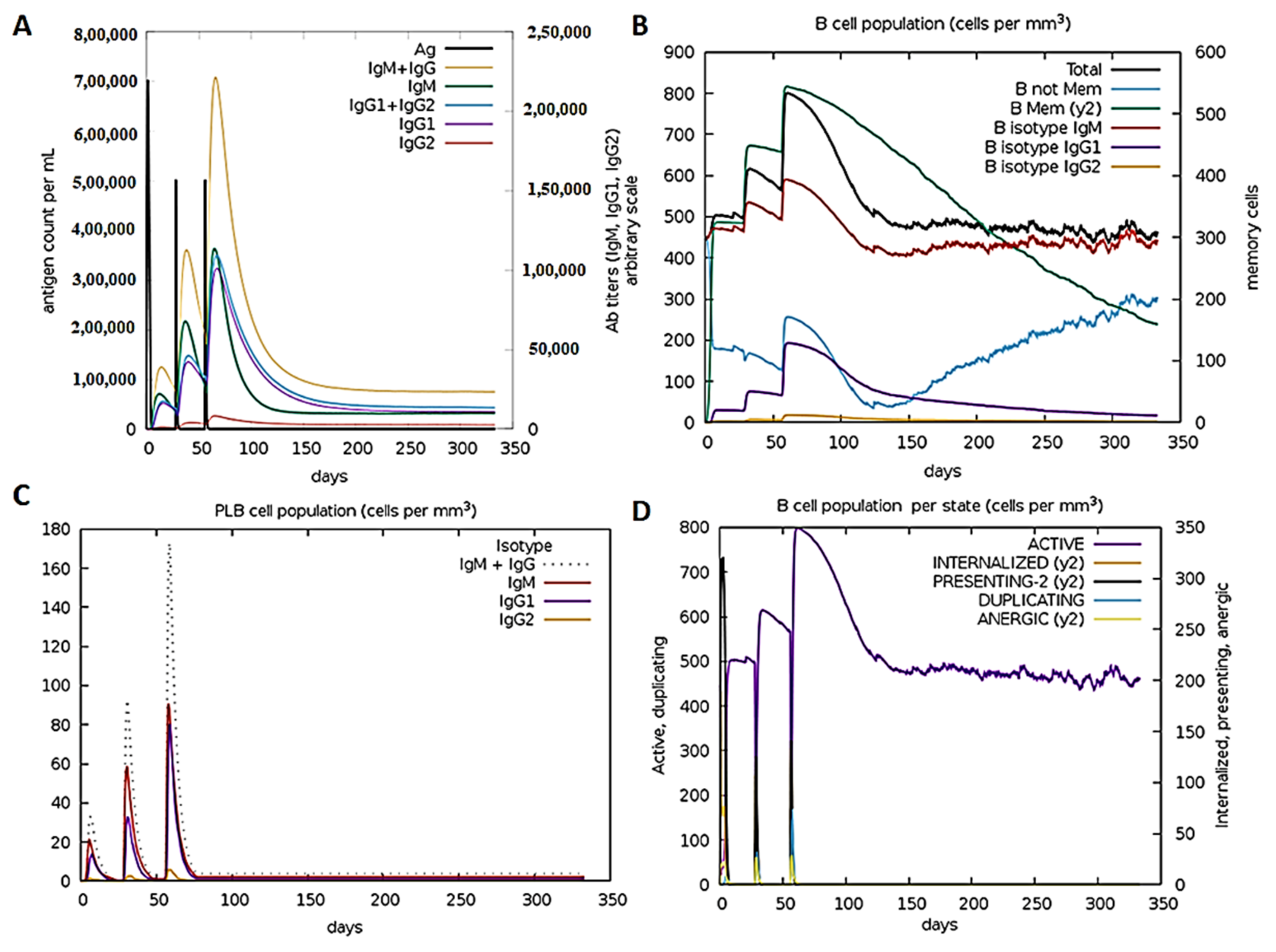
3.5. Anti-FSAg Antibody-Mediated Immune Response Is Mediated by IgG and IgM
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mukherjee, S.; Mukherjee, N.; Saini, P.; Gayen, P.; Roy, P.; Sinha Babu, S.P. Molecular evidence on the occurrence of co-infection with Pichia guilliermondii and Wuchereria bancrofti in two filarial endemic districts of India. Infect. Dis. Poverty 2014, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Lymphatic Filariasis. Available online: https://www.who.int/news-room/fact-sheets/detail/lymphatic-filariasis (accessed on 10 December 2020).
- Neglected Tropical Diseases: Impact of COVID-19 and WHO’s Response. Available online: https://www.who.int/publications/i/item/who-wer9539-461-468 (accessed on 10 December 2020).
- Mukherjee, S.; Joardar, N.; Mondal, S.; Schiefer, A.; Hoerauf, A.; Pfarr, K.; Babu, S.P.S. Quinolone-fused cyclic sulfonamide as a novel benign antifilarial agent. Sci. Rep. 2018, 8, 12073. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, R.K. Clinical and pathological aspects of filarial lymphedema and its management. Korean J. Parasitol. 2008, 46, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Karnam, A.; Das, M.; Babu, S.P.S.; Bayry, J. Wuchereria bancrofti filaria activates human dendritic cells and polarizes T helper 1 and regulatory T cells via toll-like receptor 4. Commun. Biol. 2019, 2, 169. [Google Scholar] [CrossRef]
- Mukherjee, S.; Mukherjee, S.; Maiti, T.K.; Bhattacharya, S.; Sinha Babu, S.P. A novel ligand of toll-like receptor 4 from the sheath of Wuchereria bancrofti microfilaria induces proinflammatory response in macrophages. J. Infect. Dis. 2017, 215, 954–965. [Google Scholar] [CrossRef]
- Mukherjee, S.; Joardar, N.; Sinha Babu, S.P. Exploring the homolog of a novel proinflammatory microfilarial sheath protein (MfP) of Wuchereria bancrofti in the adult-stage bovine filarial parasite Setaria cervi. J. Helminthol. 2020, 94, e15. [Google Scholar] [CrossRef]
- Mukherjee, S.; Mukherjee, S.; Bhattacharya, S.; Sinha Babu, S.P. Surface proteins of Setaria cervi induce inflammation in macrophage through Toll-like receptor 4 (TLR 4)-mediated signalling pathway. Parasite Immunol. 2017, 39, e12389. [Google Scholar] [CrossRef]
- Sanchez-Trincado, J.L.; Gomez-Perosanz, M.; Reche, P.A. Fundamentals and methods for T- and B-Cell epitope prediction. J. Immunol. Res. 2017, 2017, 2680160. [Google Scholar] [CrossRef]
- Saha, S.; Raghava, G.P.S. Prediction of continuous B-cell epitopes in an antigen using recurrent neural network. Proteins 2006, 65, 40–48. [Google Scholar] [CrossRef]
- Chen, J.; Liu, H.; Yang, J.; Chou, K.-C. Prediction of linear B-cell epitopes using amino acid pair antigenicity scale. Amino Acids 2007, 33, 423–428. [Google Scholar] [CrossRef]
- Yao, B.; Zhang, L.; Liang, S.; Zhang, C. SVMTriP: A method to predict antigenic epitopes using support vector machine to integrate tri-peptide similarity and propensity. PLoS ONE 2012, 7, e45152. [Google Scholar] [CrossRef]
- Jespersen, M.C.; Peters, B.; Nielsen, M.; Marcatili, P. BepiPred-2.0: Improving sequence-based B-cell epitope prediction using conformational epitopes. Nucleic Acids Res. 2017, 45, W24–W29. [Google Scholar] [CrossRef]
- Ahmed, R.K.S.; Maeurer, M.J. T-cell epitope mapping. Methods Mol. Biol. 2009, 524, 427–438. [Google Scholar] [CrossRef]
- Patra, P.; Mondal, N.; Patra, B.C.; Bhattacharya, M. Epitope-based vaccine designing of Nocardia asteroides targeting the virulence factor mce-family protein by immunoinformatics approach. Int. J. Pept. Res. Ther. 2020, 26, 1165–1176. [Google Scholar] [CrossRef]
- Singh, H.; Raghava, G.P.S. ProPred1: Prediction of promiscuous MHC Class-I binding sites. Bioinformatics 2003, 19, 1009–1014. [Google Scholar] [CrossRef]
- Singh, H.; Raghava, G.P. ProPred: Prediction of HLA-DR binding sites. Bioinformatics 2001, 17, 1236–1237. [Google Scholar] [CrossRef]
- Doytchinova, I.A.; Flower, D.R. VaxiJen: A server for prediction of protective antigens, tumour antigens and subunit vaccines. BMC Bioinform. 2007, 8, 4. [Google Scholar] [CrossRef]
- Rapin, N.; Lund, O.; Bernaschi, M.; Castiglione, F. Computational immunology meets bioinformatics: The use of prediction tools for molecular binding in the simulation of the immune system. PLoS ONE 2010, 5, e9862. [Google Scholar] [CrossRef]
- Mukherjee, S.; Joardar, N.; Sinha Babu, S.P. Current Trends in Targeted Chemo- and Immunotherapy Against Bancroftian Filariasis: Biochemical, Molecular and Pharmacological Perspectives; Panima Publishing Corporation: New Delhi, India, 2018; pp. 397–414. [Google Scholar]
- Ansari, J.A. Studies on Setaria cervi (Nematoda: Filarioidea).II. Its peritoneal transplant and periodicity of the microfilariae in white rats. Z. Parasitenkd. 1964, 24, 105–111. [Google Scholar] [CrossRef]
- Zakai, H.A.; Khan, W. Effects of filaricidal drugs on longevity and enzyme activities of the microfilariae of Setaria cervi in white rats. Asian Pac. J. Trop. Biomed. 2015, 5, 714–719. [Google Scholar] [CrossRef]
- Mukherjee, N.; Parida, P.K.; Santra, A.; Ghosh, T.; Dutta, A.; Jana, K.; Misra, A.K.; Sinha Babu, S.P. Oxidative stress plays major role in mediating apoptosis in filarial nematode Setaria cervi in the presence of trans-stilbene derivatives. Free Radic. Biol. Med. 2016, 93, 130–144. [Google Scholar] [CrossRef]
- Kaushal, N.A.; Kaushal, D.C.; Ghatak, S. Identification of antigenic proteins of Setaria cervi by immunoblotting technique. Immunol. Investig. 1987, 16, 139–149. [Google Scholar] [CrossRef]
- Pokharel, D.R.; Rathaur, S. Purification and characterization of a leucine aminopeptidase from the bovine filarial parasite Setaria cervi. Acta Trop. 2008, 106, 1–8. [Google Scholar] [CrossRef]
- Mukherjee, N.; Mukherjee, S.; Saini, P.; Roy, P.; Sinha Babu, S.P. Antifilarial effects of polyphenol rich ethanolic extract from the leaves of Azadirachta indica through molecular and biochemical approaches describing reactive oxygen species (ROS) mediated apoptosis of Setaria cervi. Exp. Parasitol. 2014, 136, 41–58. [Google Scholar] [CrossRef]
- Das, N.C.; Patra, R.; Gupta, P.S.S.; Ghosh, P.; Bhattacharya, M.; Rana, M.K.; Mukherjee, S. Designing of a novel multi-epitope peptide-based vaccine against Brugia malayi: An in silico approach. Infect. Genet. Evol. 2020, 104633. [Google Scholar] [CrossRef]
- Chen, P.; Nirula, A.; Heller, B.; Gottlieb, R.L.; Boscia, J.; Morris, J.; Huhn, G.; Cardona, J.; Mocherla, B.; Stosor, V.; et al. SARS-CoV-2 neutralizing antibody LY-CoV555 in outpatients with Covid-19. N. Engl. J. Med. 2021, 384, 229–237. [Google Scholar] [CrossRef]
- Galeotti, C.; Kaveri, S.V.; Bayry, J. Intravenous immunoglobulin immunotherapy for coronavirus disease-19 (COVID-19). Clin. Transl. Immunol. 2020, 9, e1198. [Google Scholar] [CrossRef]
- Casadevall, A.; Dadachova, E.; Pirofski, L.A. Passive antibody therapy for infectious diseases. Nat. Rev. Microbiol. 2004, 2, 695–703. [Google Scholar] [CrossRef]
| Sl. No. | Peptide Sequence | Start Position | End Position |
|---|---|---|---|
| 1 | KGSIWRSVWREL | 17 | 28 |
| 2 | YTDPTDSKGYRKIFKVMCNE | 48 | 67 |
| 3 | NVVSR | 87 | 91 |
| 4 | LLTEKEYEKLQDINEPSPG | 159 | 177 |
| 5 | ITAGRGSVNYVSVATN | 195 | 210 |
| 6 | RISF | 216 | 219 |
| 7 | QDDDPPKFEEDVFWKHHNEQQQMHQSMFLPRVPT | 321 | 354 |
| 8 | HPPKLHTYLEMKNQDPEEYKNRKNN | 370 | 394 |
| Sl. No. | B-Cell Epitope | MHC-II Epitope | VaxiJen Score |
|---|---|---|---|
| 1 | LLTEKEYEKLQDINEPSPG | LLTEKEYEK | 1.1015 |
| 2 | QDDDPPKFEEDVFWKHHNEQQQMHQSMFLPRVPT | WKHHNEQQQ | 0.7647 |
| MHQSMFLPR | 0.7518 | ||
| 3 | HPPKLHTYLEMKNQDPEEYKNRKNN | MKNQDPEEY | 0.8284 |
| LHTYLEMKN | 0.9114 |
| Sequence | Length |
|---|---|
| KGSIWRSVWGPGPGLLTEKEYEKGPGPGQxQMHQSMFLGPGPG | 223 |
| PKFEEDVFWGPGPGKHHNEQQQMGPGPGDPPKFEEDVGPGPG | |
| PPKFEEDVFGPGPGPKLHTYLEMGPGPGDPEEYKNRKGPGPGP | |
| KLHTYLEMGPGPGKLHTYLEMKGPGPGEEYKNRKNNAAYLL | |
| TEKEYEKAAYWKHHNEQQQAAYMHQSMFLPRAAYMKNQDP | |
| EEYAAYLHTYLEMKN |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, N.C.; Ray, A.S.; Bayry, J.; Mukherjeee, S. Therapeutic Efficacy of Anti-Bestrophin Antibodies against Experimental Filariasis: Immunological, Immune-Informatics and Immune Simulation Investigations. Antibodies 2021, 10, 14. https://doi.org/10.3390/antib10020014
Das NC, Ray AS, Bayry J, Mukherjeee S. Therapeutic Efficacy of Anti-Bestrophin Antibodies against Experimental Filariasis: Immunological, Immune-Informatics and Immune Simulation Investigations. Antibodies. 2021; 10(2):14. https://doi.org/10.3390/antib10020014
Chicago/Turabian StyleDas, Nabarun Chandra, Anindya Sundar Ray, Jagadeesh Bayry, and Suprabhat Mukherjeee. 2021. "Therapeutic Efficacy of Anti-Bestrophin Antibodies against Experimental Filariasis: Immunological, Immune-Informatics and Immune Simulation Investigations" Antibodies 10, no. 2: 14. https://doi.org/10.3390/antib10020014
APA StyleDas, N. C., Ray, A. S., Bayry, J., & Mukherjeee, S. (2021). Therapeutic Efficacy of Anti-Bestrophin Antibodies against Experimental Filariasis: Immunological, Immune-Informatics and Immune Simulation Investigations. Antibodies, 10(2), 14. https://doi.org/10.3390/antib10020014










