Landscape Fire, Biodiversity Decline and a Rapidly Changing Milieu: A Microcosm of Global Issues in an Australian Biodiversity Hotspot
Abstract
:1. Introduction
- (i)
- people are the dominant influence on fires and biodiversity, i.e., in the city of Adelaide and its suburbs on the coastal plain,
- (ii)
- agriculture is prominent, in grasslands and grassy woodlands, and tree-species regeneration is a conservation issue;
- (iii)
- the hilly topography has major effects on bushfire behaviour and can have serious consequences for social and economic assets, including human fatalities;
- (iv)
- swamp-vegetation is rare and “critically endangered” [24], but little conserved, and fires there can be in peat, a fuel of considerable significance in tropical, as well as temperate zones; and,
- (v)
- threatened species, such as orchids and bandicoots, particularly in shrub and shrub woodland communities face inappropriate fire regimes and other threats.
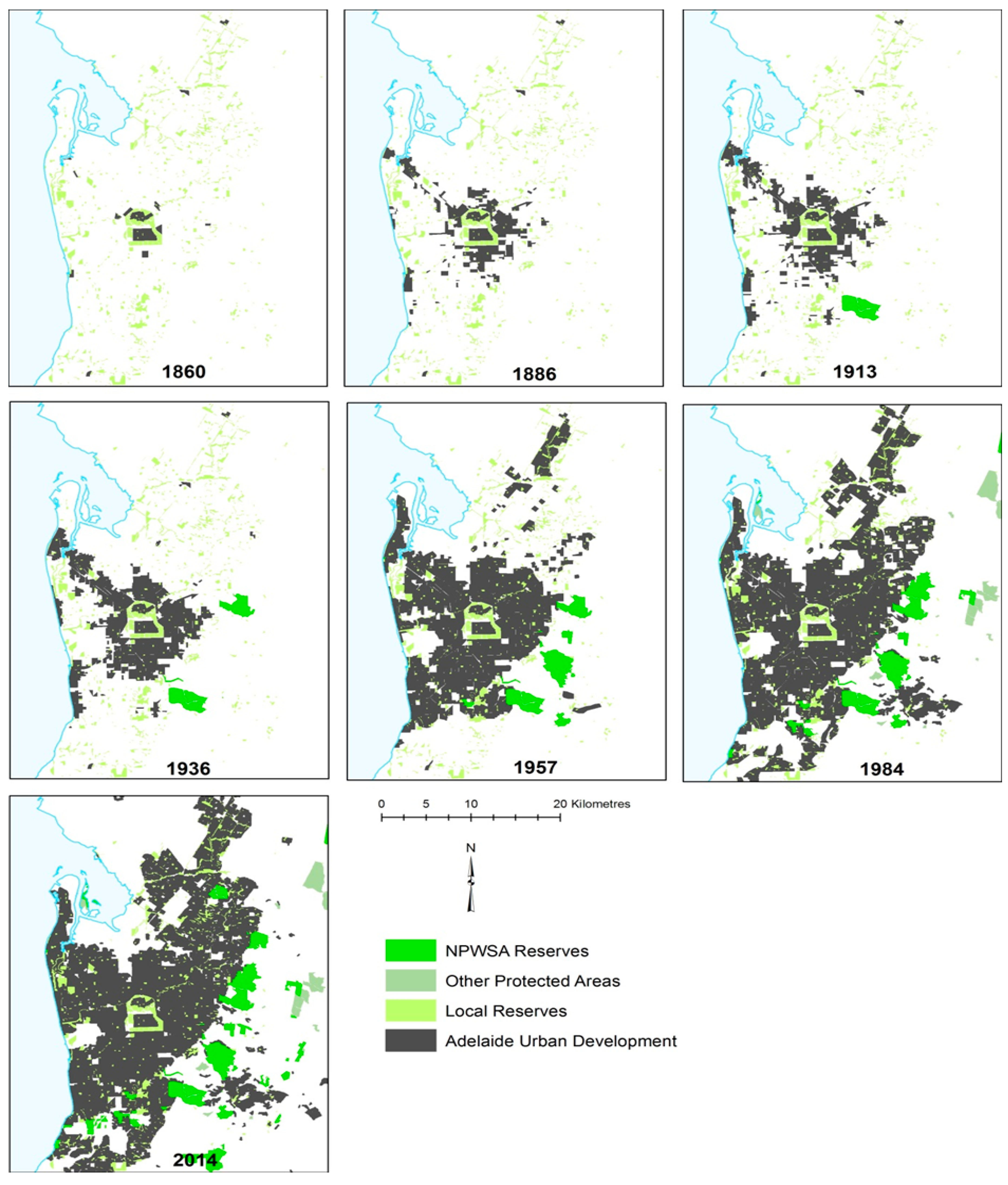
2. The Study Area



| Group | Total Species | Number Extinct | Number Threatened | Number Threatened by Fire Management Activities (Above Threat-Level “Low”) | Number Threatened by Inappropriate Fire Regimes (Above Threat-Level “Low”) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Flora | 1500 | 0 | 130 | 8 | 43 | ||||||
| Fauna | 450 | 29 | 64 (plus 9 fish) | 4 | 25 (plus 5 fish) | ||||||
| (Total: 194) | (Total: 12) | (Total: 68) | |||||||||
| Mammals | 9 | 2 | |||||||||
| Reptiles | 3 | 11 | |||||||||
| Birds | 17 | 50 | |||||||||
| Amphibians | - | 1 | |||||||||
| (Total: 29) | (Total: 64) |
3. Populous Adelaide, Fires and Biodiversity
“…with man’s progress (from settlement in 1836 to 1850), vast numbers of quail which used to frequent these parts have disappeared”.([27], p. 16)
4. Grassy Woodlands and Grasslands
“The grass was very high. We had been for the last 3 miles going through what looked like high corn, but was really kangaroo grass, now seldom seen, and when the fire was lit to show us the way to the camp the grass took fire, burning miles of the country, fortunately to the north. Even now I can remember what a glorious sight it was.”(16 February 1839) [50]

4.1. Effects of Fire Regimes
4.2. Livestock Grazing Regimes and Revegetation
5. Forest Fires, Woody Weeds and Hills’ Assets
“As there was an abundance of stringybark, broom [woody weed] and blackberry (woody weed), the roar of the fire was terrifying”.([77], p. 6)
“The fire roared through the funnel formed by gullies, shooting out one end to incinerate forests, property, houses, stock and fences.”[78]

6. Critically Endangered Swamp Vegetation: Land Use, Peat Fires and Biodiversity
“…a week later, underground fires fed by 120 hectares of peat a metre deep, were still smouldering, sending up smoke visible for thirty kilometres”.(in South-East South Australia) [114]

- their smoke and ash is a health issue especially, perhaps, if it contains silica, a known carcinogen [130];
- their smoke persists for extensive periods and causes local nuisance effects on road visibility.
- they may cause the felling of stands of mature trees, as occurred in western Victoria in 2012 due to substrate combustion [131];
- they may cause dramatic changes in plant-species assemblages, as in western Victoria, where the plant community became a “eucalypt monoculture with practically no understorey” [132];
- they may cause changed hydrological conditions, such as in an area that burned for three months, “the ground sank, the creek bank collapsed, and large areas of burnt peat were washed away” [132]; and,
- depletion of peat deposits represents a loss of “geohistorical and biotic information of the evolution of the wetlands” [130].
- the predicted warming and drying of the climate in South Australia [60];
- the increasing human population, which is likely to lead to an increase in ground-water extraction, due to the higher demand for drinking water and/or irrigating;
- the growth of plantations, such as those with eucalypts or pines [136]; and,
- an increasing intensity of land use, including the draining of wetlands.
7. Species Conservation and Fires: Orchids and Bandicoots
“The major threats to the Southern Brown Bandicoot (eastern) [include] inappropriate fire regimes and extensive wildfires …”.[142]

- the killing of the population by high intensity fire;
- the lack of unburnt habitat in the immediate area in which a remnant population could have persisted and recolonised the area [155].
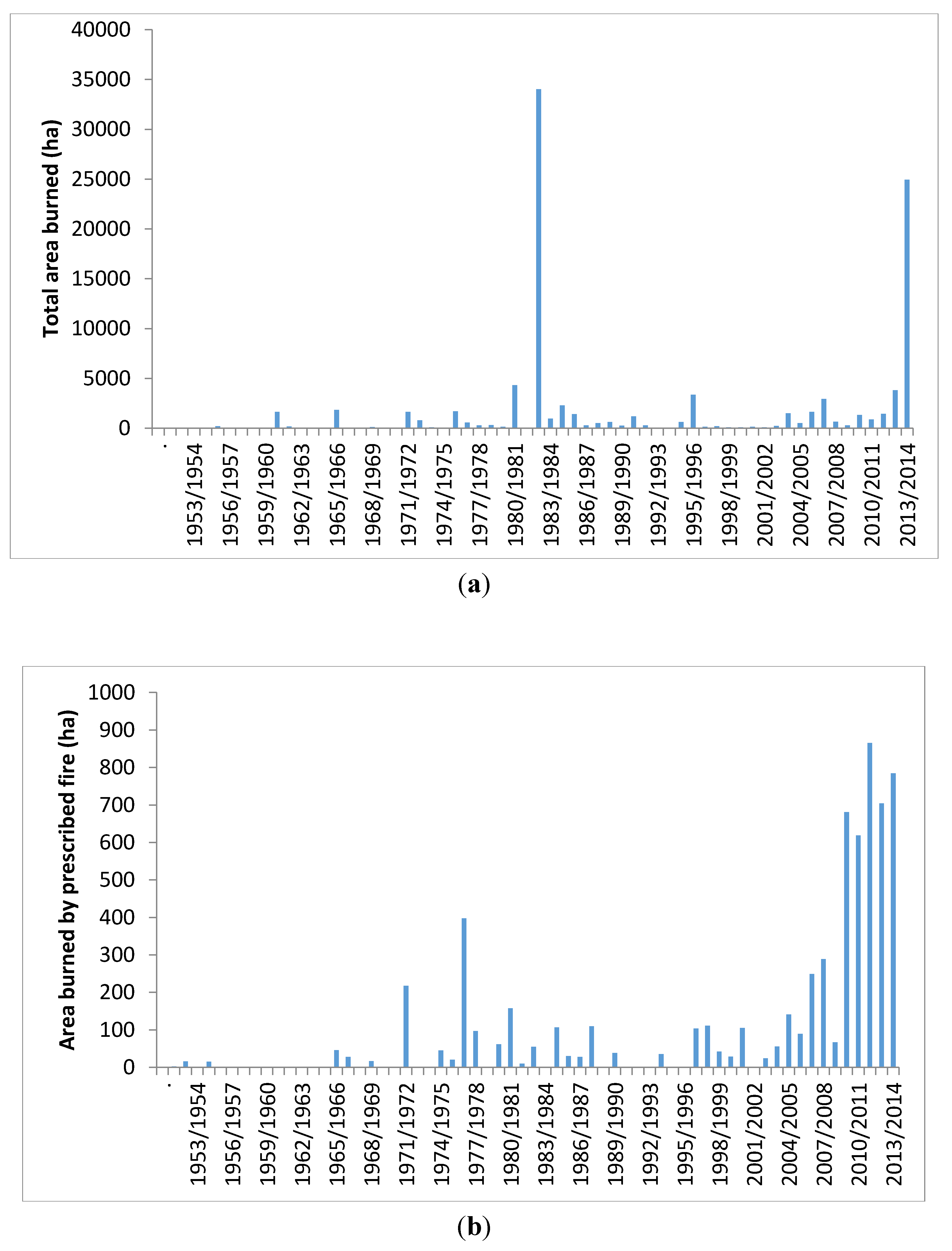
8. Discussion
“The settled areas of South Australia are within a natural environment which is bushfire-prone. This is a basic truth around which all else must revolve.”.[168]
“No Species Loss” policy: “… the aim is to lose no more species in South Australia …”.[169]
“Nature today is more about cities and farms than wilderness.”.([170], p. 47)
- many species have become extinct at local, state and national levels;
- many species are formally listed as threatened, endangered or vulnerable at state or national levels; and, similarly,
- a number of vegetation communities and their dependent fauna are under threat.
- effective and demonstrable connectivity through bio-links between all public reserves and all private remnant sites;
- strategic habitat restoration;
- enhanced conservation management across tenures;
- promotion of the “big picture” concepts, like all-tenure, whole-of-landscape [23] and whole-of-community approaches; and,
- fire regime management in all conservation management planning and actions’.
- anticipate the nature of more densely populated landscapes under climate change, land use change, soil change (e.g., peaty substrates) and new aggregations of indigenous and introduced species forming an increased range of “novel ecosystems” [178], a “new nature” [170], managed with a range of synthetic fire regimes [82];
- address species-based conservation in private urban, peri-urban and rural areas generally, with a view toward the continuity of conservation achievement through time, despite changes of land ownership;
- determine the responses of indigenous species to fire regimes [143], including responses to variation in fire interval and intensities and times of year (and peat fires where relevant);
- understand and respond to lags in tree-population change (and fire regimes) and its consequences for the habitat—the “extinction debt” [59] and barriers to species migration that could cause species’ population decline;
- measure and record the status of biodiversity across landscapes from cities and towns to far-flung areas and respond adaptively and comprehensively; and,
- record the occurrence of fires across the entire landscape more comprehensively, including the locations of fires in crop residues and the passage of unplanned fires across private land, along with the locations of all ignition points.
9. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix
| Year, Month | Location | Extent | Notes | Sources | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1837, February | “the hills” (Mt Lofty area) | “... a mass of flame” | [37] (p. 268) | ||||||||||||||||
| 1873, January | Salisbury, Echunga | small | [179] | ||||||||||||||||
| 1878, January | Echunga, Hahndorf [180] Uraidla, Carey’s Gully, Greenhill Rd, Crafers [181] | Losses of “miles of fencing, tons of timber and bark, besides feed”. “Hahndorf has not been in such peril from fire for 20 or 30 years” [180] | [180] and [181] (p. 45) | ||||||||||||||||
| 1882, February | Adelaide Hills including Marble Hill | [182] | |||||||||||||||||
| 1884, March | Adelaide Hills | Fifty acres of scrub, 400 acres of grass | Near Marble Hill | [183] | |||||||||||||||
| 1886, March | Lenswood | 60 square miles | “catastrophic fire” | [184] | |||||||||||||||
| 1897, February | Near Marble Hill | “… as the first hot months of the year go by pillars of smoke rising here and there from the Mt Lofty Range become normal”. | [90] | ||||||||||||||||
| 1900, February | Adelaide Hills | “… two big bush fires raged in the ranges near the hills residence of the Governor” | [185] | ||||||||||||||||
| 1901, February | Marble Hill | “Lord and Lady Tennyson spent the summer of 1901 at Marble Hill amid great heat, drought and bushfires” [186]. Lady Tennyson wrote that these [bushfires] were a “most wonderful sight, all the hills bursting into great volumes of smoke and great clouds of smoke rolling along the gullies.” Fire entered their “own gully garden” on February 7 [187]. | [186] and [187] (p. 139) | ||||||||||||||||
| 1902, March | Echunga-Meadows | 600 acres [188] | [189] | ||||||||||||||||
| 1905, February | Adelaide Hills: from Marble Hill to Belair | “Hollow trees were converted into furnaces”. “No words can portray the magnificence of the spectacle … at midnight”. “A large area to the north of the park [Belair] has been swept by previous fires, thus minimising the risks incurred three years ago, when 600 acres of the park ... were burnt.” | [188] | ||||||||||||||||
| 1906, January | Echunga | “The fire has extended north and south five miles in each direction” | “About 3am on Monday, strong winds sprang up, and made the fire more vigorous than ever” | [102] | |||||||||||||||
| 1907, March | Norton’s Summit | “As a rule a bush fire if confined to the bush does more good than harm, for a lot of rubbish and undergrowth is destroyed, allowing the feed to grow for the cattle during the ensuing year” | [190] | ||||||||||||||||
| 1909, February | Near Cherryville | “... regarded as the worst experienced in the neighbourhood for four years” | [191] | ||||||||||||||||
| 1910, February | Marble Hill | “... settlers … beat down the flames” “A fire swept up the valley towards (Norton Summit). Marble Hill: “The flames surged from hilltop to hilltop”. “The flames swept up a gully to the side of the Bungalow (at Marble Hill)” | [192] | ||||||||||||||||
| 1912, January | Marble Hill | “... vice-regal residence at Marble Hill narrowly escaped destruction”; “the stringybark and thick undergrowth provided excellent fuel”; “.. happily the conflagration had confined itself to the scrub”; “strong wind from the west” “pace estimated at fully 10 miles an hour” “sparks falling from the burning trees on to the roofs” | [193] | ||||||||||||||||
| 1912, February | Adelaide Hills | “… most appalling blaze that has ever been experienced in this portion of the State” [194]. “This was without a doubt a Black Thursday for Hahndorf and never before was such a terrible scene witnessed” (A map of the fires is presented.) [195] | [194,195] | ||||||||||||||||
| 1914, January | Adelaide Hills | “An extensive bush fire in the hills was visible from the city.” | [196] | ||||||||||||||||
| 1916, March | Adelaide Hills | “It (the fire) assumed serious dimensions” | [197] | ||||||||||||||||
| 1918, February | Montacute | “... extended over a distance of from 10 to 15 miles.” | “A devastating bush fire swept the Adelaide Hills” | [198] | |||||||||||||||
| 1920, February | Adelaide Hills | “.. large area of scrub” [199] | “... burned fiercely” [199] “... failure of the settlers to protect their homes ... ”. “Why, then, sleep on in a foolish insecurity?” [200] | [199,200] | |||||||||||||||
| 1923, February | Adelaide Hills: Morialta to Marble Hill | “about 1000 acres of rough country were laid bare” | “...bright glow above the hills” | [201] | |||||||||||||||
| 1926, February | Gawler, Port Gawler, Mt Crawford, Pewsey Vale | “Black Sunday” | [202] | ||||||||||||||||
| 1929, March | Mt Lofty Ranges | “Thousands of acres” | [203] | ||||||||||||||||
| 1930, February | Marble Hill | “The fire destroyed about 100 acres ..” | “... stringybark country adjoining the house” | [204] | |||||||||||||||
| 1931, February | Adelaide Hills | “Nearly 300 acres of scrub country ..” | “... near the Governor’s residence at Marble Hill” | [205] | |||||||||||||||
| 1933, February | Adelaide Hills | “Dense volumes of smoke” | [206] | ||||||||||||||||
| 1934, March | Mt Lofty Ranges | “At 8.30 a.m., the flames were roaring in the gully ...” | [207] | ||||||||||||||||
| 1935 | Mt Lofty region | [108] | |||||||||||||||||
| 1936, April | Adelaide Hills | “threatened the Governor’s residence at Marble Hill” | [208] | ||||||||||||||||
| 1938, April | Adelaide Hills | “...Tuesday morning. Fanned by a strong east wind and coming down the ranges, it appeared to threaten Governor’s summer residence”. | [209] | ||||||||||||||||
| 1939, January 10 | Adelaide Hills | “several hundred square miles” [210] | “some of the worst in the State’s history” [210]; 90 houses were lost [12] | [12,210] | |||||||||||||||
| 1940, March | Mt Lofty Ranges | “An entire gully leaped into blaze with a sudden change of wind” | [211] | ||||||||||||||||
| 1942, January, February | Mt Lofty, Waterfall Gully | [212] | |||||||||||||||||
| January, 1943 | Rowland’s Flat, Lyndoch, Mount Pleasant, Eden Valley and Springton areas | “More than 15 square miles of scrub country ..” | Large flocks of sheep lost. | [213] | |||||||||||||||
| 1944, January | Adelaide Hills | About 500 acres. | [214] | ||||||||||||||||
| 1948, January | “… from the north, through the Mt Lofty ranges to Victor Harbor ...” | [215] | |||||||||||||||||
| 1950, February [114], April [216] | Near Eagle on the Hill, Mt Lofty region | 39 square km [216] | “A scrub fire threatened the viceregal residence at Marble Hill at 1 a.m. today” “... strong breeze ...” [216] | [114] (p. 54) and [216] | |||||||||||||||
| 1951, January | “Upper Sturt Estate” (Mt Lofty Ranges?) | Three policemen died. | [114] (p. 54) | ||||||||||||||||
| 1952 | My Lofty region | [108] | |||||||||||||||||
| 1955, January | Gawler to Strathalbyn; Marble Hill | 600 square miles | “most devastating bushfire” | [217] | |||||||||||||||
| 1957, February | Horsnell’s Gully | >2500 acres | “Fire sped up Horsnell’s Gully”. Burned “orchards, gardens, houses, scrub and forest” | [100] | |||||||||||||||
| 1958, 1959, 1960, 1961 | Mt Lofty Region | [108] | |||||||||||||||||
| 1980, February | Adelaide Hills | “numerous large bushfires” | Fifty one homes lost | [80] | |||||||||||||||
| 1983, February | Many fires including Cudlee Creek, Mt Lofty, Kuitpo-Ashbourne | - | Death of people; loss of plantations, orchards; and grazing land burned [79]; 28 killed, 300 houses lost [80] | [79,80] | |||||||||||||||
| 1985, January | Black Hill | 1500 ha | [218] | ||||||||||||||||
| 1986, March | Adelaide and Adelaide Hills | “40 reports of bushfires in and around the Adelaide area” | [80] | ||||||||||||||||
| 1987, November | Strathalbyn | 6000 ha | [218] | ||||||||||||||||
| 1988, January 7 | Morialta, Cleland National Park | [114] (p. 230) | |||||||||||||||||
| 1995, January | Adelaide Hills | “hundreds of thousands of hectares burnt” | [80] | ||||||||||||||||
| 1996, January | Adelaide Hills | 5000 people affected | [80] | ||||||||||||||||
| 1998, March | Adelaide Hills | [80] | |||||||||||||||||
| 1999, May | Mt Lofty Ranges | “scrub fire after a burn-off got out of control” | [114] (p. 279) | ||||||||||||||||
| 2000, June [sic] | Brownhill Creek | 1000 ha | [218] | ||||||||||||||||
| 2001, December | Hillbank | 350 ha | [218] | ||||||||||||||||
| 2005, January | Mt Osmond | 120 ha | [218] | ||||||||||||||||
| 2007, January | Mt Bold reservoir area | [88] | |||||||||||||||||
| 2013, May | Cherryville, Basket Range, Mount Lofty | 730 ha | 1 house burned | [219] | |||||||||||||||
| 2014, January | Eden Valley | 25,000 ha (including area outside AMLR) | [218] | ||||||||||||||||
References
- Barnosky, A.D.; Matzke, N.; Tomiya, S.; Wogan, G.O.U.; Swartz, B.; Quental, T.B.; Marshall, C.; McGuire, J.L.; Lindsey, E.L.; Maguire, K.C.; et al. Has the Earth’s sixth mass extinction already arrived? Nature 2011, 471, 51–57. [Google Scholar]
- Ceballos, G.; Ehlich, P.R. Mammal population losses and the extinction crisis. Science 2002, 296, 904–907. [Google Scholar] [CrossRef]
- Department of the Environment and Heritage, Australian Government. Threatened Species and Communities Fact Sheet. 2004. Available online: http://www.environment.gov.au/resource/threatened-australian-plants-0 (accessed on 10 April 2014).
- Worboys, G.L.; Francis, W.L.; Lockwood, M. (Eds.) Connectivity Conservation Management. A Global Guide; Earthscan: London, UK, 2010.
- Gill, A.M. Fire and the Australian flora: A review. Aust. For. 1975, 38, 4–25. [Google Scholar] [CrossRef]
- Gill, A.M. Adaptive responses of Australian vascular plant species to fires. In Fire and the Australian Biota; Gill, A.M., Groves, R.H., Noble, I.R., Eds.; Australian Academy of Science: Canberra, ACT, Australia, 1981; pp. 243–272. [Google Scholar]
- Cary, G.J.; Bradstock, R.A.; Gill, A.M.; Williams, R.J. Global change and fire regimes in Australia. In Flammable Australia: Fire Regimes, Biodiversity and Ecosystems in a Changing World; Bradstock, R.A., Gill, A.M., Williams, R.J., Eds.; CSIRO Publishing: Melbourne, VIC, Australia, 2012; pp. 149–169. [Google Scholar]
- Steffen, W.; Hughes, L. The Critical Decade 2013. Climate Change Science, Risks and Responses; Climate Commission Secretariat, Commonwealth of Australia: Canberra, ACT, Australia, 2013. [Google Scholar]
- Gill, A.M.; Williams, R.J.; Woinarski, J.C.Z. Fires in Australia’s tropical savannas: Interactions with biodiversity, global warming and exotic biota. In Tropical Fire Ecology: Climate Change, Land Use and Ecosystem Dynamics; Cochrane, M., Ed.; Springer: Berlin, Germany, 2009; pp. 113–141. [Google Scholar]
- Pearce, K.; Holper, P.; Hopkins, M.; Bouma, W.; Whetton, P.; Hennessy, K.; Power, S. Climate Change in Australia; CSIRO: Melbourne, VIC, Australia, 2007. [Google Scholar]
- Steffen, W. The Angry Summer; Climate Commission Secretariat, Commonwealth of Australia: Canberra, ACT, Australia, 2013. [Google Scholar]
- Bryant, C. Understanding Bushfire: Trends in Deliberate Vegetation Fires in Australia; Technical Report 27; Australian Institute of Criminology: Canberra, ACT, Australia, 2008. [Google Scholar]
- Shu, L.; Tian, X. Fire situation in China. Int. For. Fire News 2002, 26, 9–14. [Google Scholar]
- Romero-Calcerrada, R.; Novillo, C.J.; Millington, J.D.A.; Gomez-Jimenez, I. GIS analysis of spatial patterns of human-caused wildfire ignition risk in the SW of Madrid (Central Spain). Landscape Ecol. 2008, 14. [Google Scholar] [CrossRef]
- Penman, T.D.; Bradstock, R.A.; Price, O. Modelling the determinants of ignition in the Sydney Basin, Australia: implications for future management. Int. J. Wildland Fire 2013, 22, 469–478. [Google Scholar]
- Jollands, M.; Morris, J.; Moffat, A.J. Wildfires in Wales; Report to Forestry Commission Wales. Forest Research: Farnham, Wales, 2011. Available online: http://www.forestry.gov.uk/fr/wildfiresinwales#finalreport (accessed on 10 April 2014).
- Curt, T.; Delcros, P. Managing road corridors to limit fire hazard. A simulation approach in southern France. Ecol. Eng. 2010, 36, 457–465. [Google Scholar] [CrossRef]
- Cardille, J.A.; Ventura, S.J.; Turner, M.G. Environmental and social factors influencing wildfires in the upper Midwest, United States. Ecol. Appl. 2001, 11, 111–127. [Google Scholar]
- Minnich, R.A. California climate and fire weather. In Fire in California’s Ecosystems; Sugihara, N.G., van Wagtendonk, J.W., Shaffer, K.E., Fites-Kaufman, J., Thode, A.E., Eds.; University of California Press: Berkeley, CA, USA, 2006; pp. 13–37. [Google Scholar]
- Gill, A.M.; Stephens, S.L.; Cary, G.J. The worldwide “wildfire” problem. Ecol. Appl. 2013, 23, 438–454. [Google Scholar]
- Department of the Environment, Australian Government. Australia’s 15 National Biodiversity Hotspots, Australian Government, Canberra, ACT, Australia; 2006. Available online: http://www.environment.gov.au/topics/biodiversity/biodiversity-conservation/biodiversity-hotspots/national-biodiversity-hotspots (accessed on 10 April 2014).
- Guerin, G.R.; Lowe, A.J. Multi-species distribution modelling highlights the Adelaide Geosyncline, South Australia, an important continental-scale arid-zone refugium. Austral Ecol. 2013, 38, 427–435. [Google Scholar]
- Lindenmayer, D.B.; Fischer, J. Habitat Fragmentation and Landscape Change: An Ecological and Conservation Synthesis; Island Press: Washington, DC, USA, 2006. [Google Scholar]
- Australian Government. Caring for our Country Business Plan 2012–13. Site Investment Guide. Protecting Critical Aquatic Ecosystems. Site: Fleurieu Peninsula Swamps Complex. 2013. Available online: http://www.nrm.gov.au/resources/publications/bp-2012-13/pubs/sig-hevae-fleurieu-peninsula-sa.pdf (accessed on 10 April 2014).
- Willson, A.; Bignall, J. Regional Recovery Plan for Threatened Species and Ecological Communities of Adelaide and the Mount Lofty Ranges, South Australia 2009–2014; Department of Environment and Heritage: Adelaide, SA, Australia, 2009. [Google Scholar]
- Australian Bureau of Statistics. Estimated Resident Population, States and Territories—Greater Capital City Statistical Areas. 2013. Available online: http://www.abs.gov.au/ausstats/abs@.nsf/Products/3218.0~2012~Main+Features~Main+Features?OpenDocument#PARALINK1 (accessed on 10 April 2014).
- Yelland, E.M. (Ed.) Colonists, Copper and Corn in the Colony of South Australia 1850–51 by an Old Colonist; The Hawthorn Press: Melbourne, VIC, Australia, 1970.
- Bardsley, D. There is a change on the way—An Initial Assessment of Projected Climate Change Impacts and Adaptation Options for Natural resource Management in the Adelaide and Mt Lofty Ranges Region; Government of South Australia, Department of Water, Land and Biodiversity Conservation: Adelaide, SA, Australia, 2006. [Google Scholar]
- Department of Planning and Local Government. The 30-Year Plan for Greater Adelaide. A Volume of the South Australian Planning Strategy; Government of South Australia, Adelaide, SA, Australia, 2010.
- Bryan, B.A. Reserve selection for nature conservation in South Australia: Past, present and future. Aust. Geog. Stud. 2002, 40, 196–209. [Google Scholar]
- Simeon Telfer; South Australian Department of Environment, Water and Natural Resources, Adelaide, SA, Australia. Personal communication, 2014.
- Williams, M. The Making of the South Australian Landscape. A Study in the Historical Geography of Australia; Academic Press: London, UK, 1974. [Google Scholar]
- Australian Bureau of Statistics. Population Projections, Australia, 2012 (base) to 2101. 2013. Available online: http://www.abs.gov.au/ausstats/abs@.nsf/Lookup/3222.0main+features102012%20(base)%20to%202101 (accessed on 10 April 2014).
- Sands, D.P.A.; New, T.R. The Action Plan for Australian Butterflies; Environment Australia: Canberra, ACT, Australia, 2002. [Google Scholar]
- Tait, C.J.; Daniels, C.B.; Hill, R.S. Changes in species assemblages within the Adelaide Metropolitan area, Australia, 1836–2002. Ecol. Appl. 2005, 15, 346–359. [Google Scholar] [CrossRef]
- Niesingh, M.; Shackley, A. Revegetation Action Plan. Clonlea Park. A Report for the Town of Gawler Council and the Adelaide and Mount Lofty Ranges Natural Resource Management Board. 2009. Available online: http://www.gawler.sa.gov.au/webdata/resources/files/Revegetation_Project_2009_-_2010.pdf (accessed on 10 April 2014).
- Gammage, B. The Biggest Estate on Earth. How Aborigines Made Australia; Allen and Unwin: Sydney, NSW, Australia, 2011. [Google Scholar]
- Gammage, B. The Adelaide District in 1836. In Turning Points: Chapters in South Australian History; Foster, R., Sendziuk, P., Eds.; Wakefield Press: Adelaide, SA, Australia, 2012; pp. 7–23. [Google Scholar]
- Paton, D.C.; Prescott, A.M.; Davies, R.J.-P.; Heard, L.M. The distribution, status and threats to temperate woodlands in South Australia. In Temperate Eucalypt Woodlands in Australia: Biology, Conservation, Management and Restoration; Hobbs, R.J., Yates, C.J., Eds.; Surrey Beatty & Sons: Chipping Norton, NSW, Australia, 2000; pp. 57–85. [Google Scholar]
- Davies, R.J.-P.; Whalen, M.A.; Mackay, D.A.; Taylor, D.A.; Pisanu, P. Does soil seed bank diversity limit post-fire regeneration in small, fragmented, long-unburnt remnants of fire-adapted vegetation? Biol. Cons. 2013, 158, 287–295. [Google Scholar] [CrossRef]
- City of Salisbury. Urban Forest Biodiversity Program (Undated). Available online: http://www.salisbury.sa.gov.au/Our_City/Environment/Sustainability_and_Climate_Change/Biodiversity_and_Open_Space/Urban_Forest_Biodiversity_Program (accessed on 10 April 2014).
- Cape Borda to Barossa NatureLink Plan; Government of South Australia: Adelaide, SA, Australia, 2009. Available online: http://www.environment.sa.gov.au/naturelinks/corridors/Cape_Borda_to_Barossa (accessed on 10 April 2014).
- Department of Sustainability, Environment, Water, Population and Communities, Australian Government. National Wildlife Corridors Plan: A Framework for Landscape-Scale Conservation; Commonwealth of Australia: Canberra, ACT, Australia, 2012. [Google Scholar]
- Adelaide Cemeteries Authority. West Terrace Plan of Management 2013–2018. Enfield Plaza, South Australia, 2013. Available online: http://www.aca.sa.gov.au/Portals/0/Documents/Annual%20reports/ACEM%20997%20West%20Terrace%20Report_full.w.pdf (accessed on 10 April 2014).
- Shackley, A. Understanding pre-European Adelaide Plains and Foothills Vegetation Associations–Managing Remnants and Recreating Grasslands. 2012. Available online: Understanding_pre-European_Adelaide_plains_and_foothills_vegetation_associations.pdf (accessed on 10 April 2014).
- Gill, A.M.; Sharples, J.J.; Johnstone, G. Edge effects on between-fire interval in landscape fragments such as fire-prone terrestrial conservation reserves. Biol. Cons. 2014, 169, 54–59. [Google Scholar] [CrossRef]
- Sumerling, P. The Adelaide Parklands, a Social History; Wakefield Press: Adelaide, SA, Australia, 2011. [Google Scholar]
- Long, M. A Biodiversity Survey of the Adelaide Parklands South Australia in 2003; Department of Environment and Heritage: Adelaide, SA, Australia, 2003. [Google Scholar]
- Urban Forests Biodiversity Program, Adelaide, Urban Biodiversity Unit, Backyards for Wildlife. Available online: http://www.backyards4wildlife.com.au/ (accessed on 10 April 2014).
- Mahony, E. The first settlers at Gawler. Proc. R. Geog. Soc. Australasia (South Australian Branch) 1926–1927, 28, 53–82. [Google Scholar]
- Ellis, R.W. The Aboriginal inhabitants and their environment. In Natural History of the Adelaide Region; Twidale, C.R., Tyler, M.J., Webb, B.P., Eds.; Royal Society of South Australia: Adelaide, SA, Australia, 1976; pp. 113–120, (From Clarke et al., 2005). [Google Scholar]
- Nicholson, P.H. Fire and the Australian Aborigine—An enigma. In Fire and the Australian Biota; Gill, A.M., Groves, R.H., Noble, I.R., Eds.; Australian Academy of Science: Canberra, ACT, Australia, 1981; pp. 55–76. [Google Scholar]
- Benson, J.S.; Redpath, P.A. The nature of pre-European native vegetation in Australia: A critique of Ryan, D.G., Ryan, J.R. and Starr, B.J. (1995) The Australian Landscape—Observations of explorers and early settlers. Cunninghamia 1997, 5, 285–328. [Google Scholar]
- Enright, N.J.; Thomas, I. Pre-European fire regimes in Australian ecosystems. Geog. Compass 2008, 2/4, 979–1011. [Google Scholar] [CrossRef]
- Hately, R. The Victorian Bush. Its “Original and Natural” Condition; Polybractea Press: South Melbourne, VIC, Australia, 2010. [Google Scholar]
- Lunt, I.D. Management of remnant lowland grasslands and grassy woodlands for nature conservation: A review. Victor. Nat. 1991, 108, 56–66. [Google Scholar]
- Lindenmayer, D.B.; Crane, M.; Michael, D. Woodlands. A Disappearing Landscape; CSIRO Publishing: Melbourne, VIC, Australia, 2005. [Google Scholar]
- Garnett, S.T.; Crowley, G.M. The Action Plan for Australian Birds, 2000; Environment Australia: Canberra, ACT, Australia, 2000. [Google Scholar]
- Szabo, J.K.; Vesk, P.; Baxter, P.W.J.; Possingham, H.P. Paying the extinction debt: Woodland birds in the Mount Lofty Ranges, South Australia. Emu 2011, 111, 59–70. [Google Scholar] [CrossRef]
- Suppiah, R.; Preston, B.; Whetton, P.H.; McInnes, K.L.; Jones, R.N.; Macadam, I.; Bathols, J.; Kirono, D. Climate Change under Enhanced Greenhouse Conditions in South Australia. An Updated Report on: Assessment of Climate Change, Impacts and Risk Management Strategies Relevant to South Australia; CSIRO Marine and Atmospheric Research: Melbourne, VIC, Australia, 2006. [Google Scholar]
- Mac Nally, R. The lag daemon: Hysteresis in rebuilding landscapes and implications for biodiversity futures. J. Environ. Manag. 2008, 88, 1202–1211. [Google Scholar] [CrossRef]
- Vesk, P.A.; Nolan, R.; Thomson, J.R.; Dorrough, J.W.; Mac Nally, R. Time lags in provision of habitat resources through revegetation. Biol. Cons. 2008, 141, 174–186. [Google Scholar] [CrossRef]
- Randall Johnson; South Australian Department of Environment, Water and Natural Resources, Adelaide, SA, Australia. Personal communication, 2014.
- Lunt, I.D.; Morgan, J.W. The role of fire regimes in temperate lowland grasslands of south-eastern Australia. In Flammable Australia—The Fire Regimes and Biodiversity of a Continent; Bradstock, R.A., Williams, J.E., Gill, A.M., Eds.; Cambridge University Press: Cambridge, UK, 2002; pp. 177–196. [Google Scholar]
- Bradstock, R.A.; Williams, J.E.; Gill, A.M. (Eds.) Flammable Australia. The Fire Regimes and Biodiversity of a Continent; Cambridge University Press: Cambridge, UK, 2002.
- Hobbs, R. Fire regimes and their effects in Australian temperate woodlands. In Flammable Australia. The Fire Regimes and Biodiversity of a Continent; Bradstock, R.A., Williams, J.E., Gill, A.M., Eds.; Cambridge University Press: Cambridge, UK, 2002; pp. 305–326. [Google Scholar]
- Lunt, I.D.; Prober, S.M.; Morgan, J.W. How do fire regimes affect ecosystem structure, function and diversity in grasslands and grassy woodlands of southern Australia. In Flammable Australia. Fire Regimes, Biodiversity and Ecosystems in a Changing World; Bradstock, R.A., Gill, A.M., Williams, R.J., Eds.; CSIRO Publishing: Melbourne, VIC, Australia, 2012; pp. 253–270. [Google Scholar]
- Davies, R.J.-P. Description, distribution, conservation status and weed management of box and buloke grassy woodlands in South Australia. In Temperate Eucalypt Woodlands in Australia: Biology, Conservation, Management and Restoration; Hobbs, R.J., Yates, C.J., Eds.; Surrey Beatty & Sons: Chipping Norton, NSW, Australia, 2000; pp. 167–189. [Google Scholar]
- Fischer, J.; Stott, J.; Zerger, A.; Warren, G.; Sherren, K.; Forrester, R.I. Reversing a tree regeneration crisis in an endangered ecoregion. Proc. Nat. Acad. Sci. USA 2009, 106, 10386–10391. [Google Scholar] [CrossRef]
- Williams, P.R. Contrasting demographics of tropical savanna and temperate forest eucalypts provide insight into how savannas and forests function—A case study using Corymbia clarksoniana from north-eastern Australia. Austral Ecol. 2009, 34, 120–131. [Google Scholar]
- Werner, P.A.; Prior, L.D. Demography and growth of subadult savannah trees: Interactions of life history, size, fire season, and grassy understorey. Ecol. Monog. 2013, 83, 67–93. [Google Scholar] [CrossRef]
- Williams, R.J.; Cook, G.D.; Gill, A.M.; Moore, P.H.R. Fire regime, fire intensity and tree survival in a tropical savanna in northern Australia. Aust. J. Ecol. 1999, 24, 50–59. [Google Scholar]
- Gibbons, P.; Lindenmayer, D.B. Tree Hollows and Wildlife Conservation in Australia; CSIRO Publishing: Melbourne, VIC, Australia, 2002. [Google Scholar]
- Gill, A.M. Underpinnings of Fire Management for the Conservation of Biodiversity in Reserves, Revised Edition; Victorian Department of Sustainability and Environment: Melbourne, VIC, Australia, 2009. [Google Scholar]
- Lunt, I.D.; Winsemius, L.M.; McDonald, S.P.; Morgan, J.W.; Dehaan, R.L. How widespread is woody plant encroachment in temperate Australia? Changes in woody vegetation cover in lowland woodland and coastal ecosystems in Victoria from 1989 to 2005. J. Biogeog. 2010, 37, 722–732. [Google Scholar] [CrossRef]
- Bennett, V.A.; Doerr, V.A.J.; Doerr, E.D.; Manning, A.D.; Lindenmayer, D.B; Yoon, H.-J. Causes of reintroduction failure of the brown treecreeper: Implications for ecosystem restoration. Austral Ecol. 2013, 38, 700–712. [Google Scholar] [CrossRef]
- Anonymous. Black Thursday. The Adelaide Register, 2 February 1912; 6. [Google Scholar]
- Country Fire Services. The Meadow/Kuitpo fire. The Volunteer Off. J. Ctry. Fire Serv. S. Aust. 1983, 17, 10–13. [Google Scholar]
- Bardsley, K.L.; Fraser, A.S.; Heathcote, R.L. The second Ash Wednesday: 16 February 1983. Aust. Geog. Stud. 1983, 21, 129–141. [Google Scholar] [CrossRef]
- Tait, C. Text Box 107: Bushfires in Adelaide. In Adelaide, Nature of a City; Daniels, C.B., Tait, C.J., Eds.; University of Adelaide: Adelaide, SA, Australia, 2005; pp. 425–427. [Google Scholar]
- Teague, B.; McLeod, R.; Pascoe, S. 2009 Victorian Bushfires Royal Commission; Final Report. Victorian Government: Melbourne, VIC, Australia, 2010. Available online: http://www.royalcommission.vic.gov.au/ (accessed on 10 April 2014).
- Gill, A.M. Fire regimes, biodiversity conservation and prescribed burning regimes. Proc. R. Soc. Vic. 2012, 124, 7–12. [Google Scholar]
- Marble Hill. Available online: http://marblehill.com.au/planningandconstruction.html (accessed on 10 April 2014).
- Adelaide Hills Council. Community Profile. Available online: http://profile.id.com.au/adelaide-hills/about (accessed on 10 April 2014).
- District Council of Mount Barker. Community Profile. Available online: http://profile.id.com.au/mount-barker/population (accessed on 10 April 2014).
- Adelaide Hills Council and District Council of Mount Barker. Bushfire Mitigation Plan. Final Plan; Self Published: Adelaide, SA, Australia, 2008. [Google Scholar]
- Clarke, P.A.; Gepp, B.; Vickery, F.; Wouters, M.; Henderson, M. Chapter 18. The urban forest 4: Fire, biology and city living. In Adelaide, Nature of a City; Daniels, C.B., Tait, C.J., Eds.; University of Adelaide: Adelaide, SA, Australia, 2005; pp. 422–453. [Google Scholar]
- Buckman, S.; Brownlie, K.C.; Bourman, R.P.; Murray-Wallace, C.V.; Morris, R.H.; Lachlan, T.J.; Roberts, R.G.; Arnold, L.J.; Cann, J.H. Holocene palaeofire records in a high-level, proximal valley-fill (Wilson Bog), Mount Lofty Ranges, South Australia. The Holocene 2009, 19, 1017–1029. [Google Scholar] [CrossRef]
- Naturae Amator. The mountains. South Australian Register, 27 March 1841; 4. [Google Scholar]
- Anonymous. Bush fire near Marble Hill. South Australian Register, 3 February 1897; 5. [Google Scholar]
- Clarke, H.; Lucas, C.; Smith, P. Changes in Australian fire weather between 1973 and 2010. Int. J. Clim. 2013, 33, 931–944. [Google Scholar] [CrossRef]
- Lucas, C.; Hennessy, K.; Mills, G.; Bathols, J. Bushfire Weather in Southeast Australia: Recent Trends and Projected Climate Change Impacts; Bushfire Cooperative Research Centre and CSIRO: Melbourne, VIC, Australia, 2007. [Google Scholar]
- Berry, S.L.; Roderick, M.L. Gross primary productivity and transpiration flux of the Australian vegetation from 1788 to 1988 AD: Effects of CO2 and land use change. Glob. Change Biol. 2004, 10, 1884–1898. [Google Scholar] [CrossRef]
- King, K.; Cary, G.J.; Gill, A.M.; Moore, A. Implications of changing climate and atmospheric CO2 for grassland fire in South East Australia: Insights using the GRAZPLAN grassland simulation model. Int. J. Wildland Fire 2012, 21, 695–708. [Google Scholar] [CrossRef]
- Gill, A.M. Fire, science and society at the rural-urban interface. Proc. R. Soc. Qld. 2009, 115, 153–160. [Google Scholar]
- McFarlane, A.C.; Raphael, B. Ash Wednesday: The effects of a fire. Aust. N. Zeal. J. Psychiatry 1984, 18, 341–351. [Google Scholar] [CrossRef]
- Menadue, B. Tea Tree/Anstey Hill/Gumeracha fire. The Volunteer Off. J. Ctry. Fire Serv. S. Aust. 1983, 17, 14–16. [Google Scholar]
- Brodison, K. Effect of Smoke in Grape and Wine Production Bulletin. 2013, 4847.
- Marsh, L. Towards an Understanding of the Adelaide Gully Wind, 4th ed.; Meteorological Notes 175; Bureau of Meteorology: Melbourne, VIC, Australia, 1987; p. 37. [Google Scholar]
- Cochrane, G.R.; Burnard, S.; Philpott, J.M. Land use and forest fires in the Mount Lofty Ranges, South Australia. Aust. Geogr. 1962, 8, 143–160. [Google Scholar]
- Grace, W.; Holton, I. A Mechanism for Downslope Winds with Special Reference to the Adelaide Gully Wind; Meteorological Notes 179; Bureau of Meteorology: Melbourne, VIC, Australia, 1988. [Google Scholar]
- Anonymous. Bush fire in the hills. The Adelaide Advertiser, 24 January 1906; 5. [Google Scholar]
- Sharples, J.J.; Gill, A.M.; Dold, J.W. The trench effect and eruptive wildfires: lessons from the King’s Cross underground disaster. In Proceedings of the Australasian Fire Authorities Council/Co-Operative Research Centre Conference, Darwin, NT, Australia, 10 September 2010.
- Viegas, D.X.; Pita, L.P.; Ribeiro, L.; Palheiro, P. Eruptive fire behaviour in past fatal accidents. In Proceedings of the Eighth International Wildland Fire Safety Summit, Missoula, MT, USA, 26–28 April 2005.
- Sharples, J.J.; McRae, R.H.D.; Wilkes, S.R. Wind-terrain effects on the propagation of wildfires in rugged terrain: fire channelling. Int. J. Wildland Fire 2012, 21, 282–296. [Google Scholar] [CrossRef]
- Viegas, D.X.; Simeoni, A. Eruptive behaviour of forest fires. Fire Technol. 2011, 47, 303–320. [Google Scholar] [CrossRef]
- Australian Broadcasting Commission. Flames Approach a Hills Property. 2013. Available online: http://www.abc.net.au/news/2013-05-10/flames-approach-a-hills-propertyjpg/4682470 (accessed on 10 April 2014).
- Cochrane, G.R. Vegetation studies in forest-fire areas of the Mount Lofty Ranges, South Australia. Ecology 1963, 44, 41–52. [Google Scholar] [CrossRef]
- Marsden-Smedley, J.B. Prescribed Burning in South Australia: Review of Operational Prescriptions; South Australian Department of Environment and Natural Resources: Adelaide, SA, Australia, 2011. [Google Scholar]
- Department for Environment and Heritage, South Australia. Fire Management Plan. Reserves of the Hill Face Zone, Mount Lofty Ranges. 2009–2019; Government of South Australia: Adelaide, SA, Australia, 2009. [Google Scholar]
- Heddle, E.M. South African Daisy in the national parks of South Australia. Environ. Cons. 1974, 1, 152. [Google Scholar] [CrossRef]
- Richards, N.W. Prescribed burning in the southern Mt. Lofty Ranges: How and why is the decision to burn made? Proc. R. Soc. Qld. 2009, 115, 29–35. [Google Scholar]
- Department of Environment and Natural Resources, South Australia. Corporate Plan 2012–2014; Government of South Australia: Adelaide, SA, Australia, 2012. [Google Scholar]
- Ellis, J-A. Tried by Fire. The Story of the South Australian Country Fire Service; South Australian Country Fire Service: Adelaide, SA, Australia, 2001. [Google Scholar]
- Australian Government. Swamps of the Fleurieu Peninsula, South Australia. 2003. Available online: http://www.environment.gov.au/node/14507 (accessed on 10 April 2014).
- Australian Government. Approved Conservation Advice for the Swamps of the Fleurieu Peninsula Ecological Community. 2013. Available online: http://www.environment.gov.au/biodiversity/threatened/communities/pubs/031-conservation-advice.pdf (accessed on 10 April 2014).
- Rebecca Duffield; Conservation Council of South Australia, Adelaide, SA, Australia. Personal communication, 2014.
- Joosten, H.; Clarke, D. Wise Use of Mires and Peatlands-Background and Principles Including a Framework for Decision-Making; International Mire Conservation Group and International Peat Society; Saarijärven Offset Oy: Saarijärvi, Finland, 2002. Available online: http://www. gret-perg.ulaval.ca/fileadmin/fichiers/fichiersGRET/pdf/Doc_generale/WUMP_Wise_Use_of_ Mires_and_Peatlands_book.pdf (accessed on 10 April 2014).
- Wein, R.W. Fire behaviour and ecological effects in organic terrain. In The Role of Fire in Northern Circumpolar Ecosystems; Wein, R.W., MacLean, D.A., Eds.; John Wiley and Sons: Toronto, ON, Canada, 1983; pp. 81–95. [Google Scholar]
- Albini, F.A. Wildland fires. Am. Sci. 1984, 72, 590–597. [Google Scholar]
- Rein, G.; Cleaver, N.; Ashton, C.; Pironi, P.; Torero, J.L. The severity of smouldering peat fires and damage to the forest soil. Catena 2008, 74, 304–309. [Google Scholar] [CrossRef]
- Chistjakov, V.I.; Kuprijanov, A.I.; Gorshkov, V.V.; Artsybashev, E.S. Measures for fire prevention on peat deposits. In The Role of Fire in Northern Circumpolar Ecosystems; Wein, R.W., MacLean, D.A., Eds.; John Wiley and Sons: Toronto, ON, Canada, 1983; pp. 259–271. [Google Scholar]
- Hungerford, R.D.; Frandsen, W.H.; Ryan, K.C. Ignition and burning characteristics of organic soils. In Fire in Wetlands: A Management Perspective, Proceedings of the 19th Tall Timbers Fire Ecology Conference, Tallahassee, FL, USA, 3–6 November 1993; Cerulean, S.I., Engstrom, R.T., Eds.; Tall Timbers Research Station: Tallahassee, FL, USA, 1995; pp. 78–91. [Google Scholar]
- Armstrong, J. Spontaneous Combustion of Forest Fuels: A Review; Information Report FF-X-42; Forest Fire Research Institute: Ottawa, ON, Canada, 1973. [Google Scholar]
- Tacconi, L.; Moore, P.F.; Kaimowitz, D. Fires in tropical forests—What is really the problem? Lessons from Indonesia. Mitig. Adapt. Strate. Glob. Change 2007, 12, 55–66. [Google Scholar] [CrossRef]
- The Guardian. Indonesian President Apologises to Singapore and Malaysia for Forest Fires. 2013. Available online: http://www.guardian.co.uk/world/2013/jun/24/indonesian-president-singapore-malaysia-fires (accessed on 10 April 2014).
- Chubarova, N.Y.; Prilepsky, N.G.; Rublev, A.N.; Riebau, A.R. A mega-fire event in Central Russia: Fire weather, radiative, and optical properties of the atmosphere, and consequences of subboreal forest plants. In Wildland Fires and Air Pollution. Developments in Environmental Science; Bytnerowicz, A., Arbaugh, M., Riebau, A., Anderson, C., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; Volume 8, pp. 247–264. [Google Scholar]
- Global Fire Monitoring Center. Preliminary assessment of the fire situation in western Russia in 2010. Int. For. Fire News 2010, 40, 20–42. [Google Scholar]
- Clement, B.M.; Javier, J.; Sah, J.P.; Ross, M.S. The effects of wildfires on the magnetic properties of soils in the Everglades. Earth Surface Process. Landf. 2010, 7. [Google Scholar] [CrossRef]
- Semeniuk, V.; Semeniuk, C.A. Wetland sediments and soils on the Swan Coastal Plain, southwestern Australia: Types, distribution, susceptibility to combustion, and implications for fire management. J. R. Soc. W. Aust. 2005, 88, 91–120. [Google Scholar]
- Malcolm Gill; Fenner School of Environment and Society, Australian National University, Canberra, ACT, Australia. Personal communication, 2012.
- Wark, M.C. Regeneration of some forest and gully communities in the Angahook-Lorne State Park (North-Eastern Otway Ranges) 1–10 years after the wildfire of February 1983. Proc. R. Soc. Victoria 1997, 109, 7–36. [Google Scholar]
- Department of Environment and Primary Industries, Victoria. Otway Fire District Planned Burn Program Autumn 2013. Planned Burn: Aireys Inlet-Breakfast Creek 1122 Hectares; Victorian Government: Melbourne, VIC, Australia, 2013.
- Malcolm Gill; Fenner School of Environment and Society, Australian National University, Canberra, ACT, Australia. Personal communication, 2009.
- Thomas, I.; Cullen, P.; Fletcher, M.-S. Ecological drift or stable fire cycles in Tasmania: A resolution? In Altered Technologies. Fire, Climate and Human Influence on Terrestrial Landscapes; Stevenson, J., Prebble, M., Eds.; Australian National University E-Press: Canberra, ACT, Australia, 2010; pp. 341–352. [Google Scholar]
- Whinam, J.; Copley, P. A survey of Sphagnum moss in South Australia. Trans. R. Soc. S. Aust. 2011, 135, 55–61. [Google Scholar]
- Gott, B. Cumbungi, Typha species: A staple aboriginal food in southern Australia. Aust. Aborig. Stud. 1999, 1, 33–50. [Google Scholar]
- Burbidge, A.A.; Roberts, J.D. Sunset Frog Recovery Plan. June 2001–June 2006; Department of Conservation and Land Management Western Australian Threatened Species and Communities Unit: Wanneroo, WA, Australia, 2002. [Google Scholar]
- Horizon, C.; O’Connor, N.R.M. A Study of the Mount Lofty Ranges Southern Emu-wren and Fleurieu Peninsula Swamps Recovery Program and how It Contributed to Biodiversity Outcomes in the Adelaide and Mount Lofty Resources Management Region; Performance Story Report; Commonwealth of Australia: Canberra, ACT, Australia, 2008. [Google Scholar]
- Abley, A.; Cantono, D.; Pickett, M.; Smith, G.; Stevens, A.; Vale, T. Protecting Fleurieu Peninsula Swamps and the Mount Lofty Ranges Southern Emu-Wren. Conservation Council of South Australia: Adelaide, SA, Australia. Available online: http://www.ccsa.asn.au/files/emuwren/Protecting%20FPS%20MLRSEW%20booklet%20100307.pdf (accessed on 5 September 2014).
- Armstrong, D.M.; Brandle, R. Birds. In A Biological Survey of the Southern Mount Lofty Ranges, South Australia 2000–2001; Armstrong, D.M., Croft, S., Foulkes, J.N., Eds.; South Australian Department of Environment and Heritage: Adelaide, SA, Australia, 2003; pp. 157–176. [Google Scholar]
- Department of the Environment, Australian Government. Isoodon obselus obselus—Southern Brown Bandicoot (Eastern). Species Profile and Threats Database; Australian Government: Canberra, ACT, Australia, 2013. Available online: http://www.environment.gov.au/cgi-bin/sprat/public/publicspecies.pl?taxon_id=68050 (accessed on 10 April 2014).
- Gill, A.M.; Stephens, S.L. Scientific and social challenges for the management of fire-prone wildland-urban interfaces. Environ. Res. Lett. 2009, 4, 1–10. [Google Scholar]
- Duncan, M. Response of Orchids to Bushfire: Black Saturday Victoria 2009—Natural Values Fire Recovery Program; Department of Sustainability and Environment: Heidelberg, VIC, Australia, 2012. [Google Scholar]
- Jones, D.L. Native Orchids of Australia; Reed Books: Sydney, NSW, Australia, 1988. [Google Scholar]
- Joe Quarmby; South Australian Department of Environment, Water and Natural Resources. Adelaide, SA, Australia, 2009.
- Quarmby, J.P. Recovery Plan for Twelve Threatened Orchids in the Lofty Block Region of South Australia 2010; Department of Environment and Natural Resources, South Australia: Adelaide, SA, Australia, 2010. [Google Scholar]
- Tremblay, R.L.; Perez, M-E.; Larcombe, M.; Brown, A.; Quarmby, J.; Bickerton, D.; French, G.; Bould, A. Dormancy in Caladenia: A Bayesian approach to evaluating latency. Aust. J. Bot. 2009, 57, 340–350. [Google Scholar] [CrossRef]
- Coates, F.; Lunt, I.D.; Tremblay, R.L. Effects of disturbance on population dynamics of the threatened orchid Prasophyllum correctum D.L. Jones and implications for grassland management in South-Eastern Australia. Biol. Cons. 2006, 129, 59–69. [Google Scholar]
- Department of the Environment, Australian Government. Caladenia ovata. Species Profile and Threats Database; Australian Government: Canberra, ACT, Australia, 2013. Available online: http://www.environment.gov.au/cgi-bin/sprat/public/spratlookupspecies.pl?name=caladenia+ovata&searchtype=Wildcard (accessed on 10 April 2014).
- Department for Environment and Heritage, South Australia. Adelaide and Mount Lofty Ranges Threatened Species Profile. Diuris brevifolia. In Short-Leaf Donkey Orchid; South Australian Government: Adelaide, SA, Australia, 2008. [Google Scholar]
- Department of the Environment, Australian Government. Thelymitra cyanapicata. Species Profile and Threats Database; Australian Government: Canberra, ACT, Australia, 2013. Available online: http://www.environment.gov.au/cgi-bin/sprat/public/publicspecies.pl?taxon_id=81872#management_documentation (accessed on 10 April 2014).
- Department of the Environment, Australian Government. Pterostylis bryophila. Species Profile and Threats Database; Australian Government: Canberra, ACT, Australia, 2013. Available online: http://www.environment.gov.au/cgi-bin/sprat/public/publicspecies.pl?taxon_id=75722 (accessed on 10 April 2014).
- Paull, D.J.; Mills, D.J.; Claridge, A.W. Fragmentation of the southern Brown Bandicoot Isoodon obesulus: Unraveling past climate change from vegetation clearing. Int. J. Ecol. 2013, 11, 536524. [Google Scholar]
- Long, K. Burrowing bandicoots—An adaptation to life in a fire-prone environment? Aust. Mammal. 2009, 31, 57–59. [Google Scholar] [CrossRef]
- Thompson, M.B.; Medlin, G.; Hutchinson, R.; West, N. Short-term effects of fuel reduction burning on populations of small terrestrial mammals. Aust. Wildl. Res. 1989, 16, 117–129. [Google Scholar] [CrossRef]
- Hope, B. Short-term response of the long-nosed bandicoot, Perameles nasuta, and the southern brown bandicoot, Isoodon obesulus obesulus, to low-intensity prescribed fire in heathland vegetation. Wildl. Res. 2012, 39, 731–744. [Google Scholar]
- Gepp, B. Box 114. Impact of fires on southern brown bandicoot populations in South Australia. In Adelaide, Nature of a City; Daniels, C.B., Tait, C.J., Eds.; University of Adelaide: Adelaide, SA, Australia, 2005; p. 447. [Google Scholar]
- Haby, N.; Long, K. Recovery Plan for the Southern Brown Bandicoot in the Mount Lofty Ranges, South Australia, 2004 to 2009; Department for Environment and Heritage: Adelaide, SA, Australia, 2005. [Google Scholar]
- Possingham, H.P.; Gepp, B. Assessment of fire regime options for the Southern Brown Bandicoot Isoodon obesulus in South Australia using population viability analysis. In Proceedings of the Conference on Fire and Biodiversity—The Effects and Effectiveness of Fire Management, Melbourne, VIC, Australia, 8–9 October 1994; Australian Department of the Environment, Sport and Territories: Canberra, ACT, Australia, 1996; pp. 149–153. [Google Scholar]
- Gill, A.M.; Allan, G.; Yates, C. Fire-created patchiness in Australian savannas. Int. J. Wildl. Fire 2003, 12, 323–331. [Google Scholar]
- Menkhorst, P.W.; Seebeck, J.H. Distribution and conservation status of bandicoots in Victoria. In Bandicoots and Bilbies; Seebeck, J.H., Brown, P.R., Wallis, R.L., Kemper, C.M., Eds.; Surrey Beatty & Sons: Chipping Norton, NSW, Australia, 1990; pp. 51–60. [Google Scholar]
- Rees, M.; Paull, P. Distribution of the southern brown bandicoot (Isoodon obesulus) in the Portland region of South-Western Victoria. Wildl. Res. 2000, 27, 539–545. [Google Scholar] [CrossRef]
- Bradstock, R.A.; Bedward, M.; Gill, A.M.; Cohn, J.S. Which mosaic? A landscape ecological approach for evaluating interactions between fire regimes, habitat and animals. Wildl. Res. 2005, 32, 409–423. [Google Scholar] [CrossRef]
- Singh, G.; Kershaw, A.P.; Clark, R. Quaternary vegetation and fire history in Australia. In Fire and the Australian Biota; Gill, A.M., Groves, R.H., Noble, I.R., Eds.; Australian Academy of Science: Canberra, ACT, Australia, 1981; pp. 23–54. [Google Scholar]
- Aitken, P.F. Mammals. In Natural History of the South East; Tyler, M.J., Twidale, C.R., Ling, J.K., Holmes, J.W., Eds.; Royal Society of South Australia: Adelaide, SA, Australia, 1983; pp. 127–133. [Google Scholar]
- Clarke, M.F. Catering for the needs of fauna in fire management: Science or just wishful thinking. Wildl. Res. 2008, 35, 385–394. [Google Scholar]
- Douglas, D. Bushfire Disasters in South Australia with Special Reference to Fires on the Ash Wednesdays of 1980 and 1983; State Government Insurance Commission: Adelaide, SA, Australia, 1983. [Google Scholar]
- Department of Environment, Water and Natural Resources. No Species Loss; Government of South Australia: Adelaide, SA, Australia, 2014. Available online: http://www.environment.sa.gov.au/managing-natural-resources/Ecosystem_conservation/No_species_loss (accessed on 13 June 2014).
- Low, T. The New Nature. Winners and Losers in Wild Australia; Viking: Melbourne, VIC, Australia, 2002. [Google Scholar]
- Town of Gawler. Draft Gawler Strategic Directions Report. 2013. Available online: http://www.gawler.sa.gov.au/webdata/resources/files/Draft%20Strategic%20Directions%20Report-2.pdf (accessed on 10 April 2014).
- Keeley, J.E.; Bond, W.J. Convergent seed germination in South African fynbos and Californian chaparral. Plant Ecol. 1997, 133, 153–167. [Google Scholar]
- Gill, A.M.; Zylstra, P. Flammability of Australian forests. Aust. For. 2005, 68, 88–94. [Google Scholar]
- Turner, M.S. Conserving Adelaide’s Biodiversity: Resources; Urban Forest Biodiversity Program: Adelaide, SA, Australia, 2001. [Google Scholar]
- Setterfield, S.A.; Rossiter-Rachor, N.A.; Hutley, L.B.; Douglas, M.M.; Williams, R.J. Turning up the heat: The impacts of Andropogon gayanus (gamba grass) invasion on fire behaviour in northern Australian savannas. Divers. Distrib. 2010, 16, 854–861. [Google Scholar] [CrossRef]
- Melzer, A.; Carrick, F.; Menkhorst, P.; Lunney, D.; St. John, B. Overview, critical assessment, and conservation implications of Koala distribution and abundance. Cons. Biol. 2000, 14, 619–628. [Google Scholar] [CrossRef]
- Department of the Environment, Australian Government. The Ramsar Convention on Wetlands. Available online: http://www.environment.gov.au/topics/water/water-our-environment/wetlands/ramsar-convention-wetlands (accessed on 13 June 2014).
- Hobbs, R.J.; Arico, S.; Aronson, J.; Baron, J.S.; Bridgewater, P.; Cramer, V.A.; Epstein, P.R.; Ewel, J.J.; Clink, C.A.; Lugo, A.E.; et al. Novel ecosystems: Theoretical and management aspects of the new ecological world order. Global Ecol. Biogeogr. 2006, 15, 1–7. [Google Scholar]
- Anonymous. Topics of the day. The South Australian Advertiser, 7 January 1873; 2. [Google Scholar]
- Anonymous. Further particulars. South Australian Register, 1 February 1878; 5. [Google Scholar]
- De Crespigny, M. The Adelaide Establishment: The 1878 Diary of a Provincial Lady of Mount Lofty and Adelaide, Gillingham Printers: Adelaide, SA, Australia, 1987.
- Anonymous. South Australia. The Hobart Mercury, 15 February 1882; 2. [Google Scholar]
- Anonymous. Fires in the hills. South Australian Register, 24 March 1884; 7. [Google Scholar]
- Bishop, G.C. Stringybark to Orchards. A History of Forest Range and Lenswood; Lenswood Cold Stores Co-Operative Society Ltd.: Lenswood, SA, Australia, 1984. [Google Scholar]
- Anonymous. Bush fires. South Australian Register, 26 February 1900; 4. [Google Scholar]
- Anonymous. Memories of Marble Hill. The Adelaide Register, 21 March 1922; 4. [Google Scholar]
- Hasluck, A. (Ed.) Audrey Tennyson’s Vice-Regal Days. The Australian letters of Audrey Lady Tennyson to her mother Zacyntha Boyle, 1899–1903; National Library of Australia: Canberra, ACT, Australia, 1978.
- Anonymous. The bush fires still raging. The Adelaide Advertiser, 11 February 1905; 7. [Google Scholar]
- Anonymous. Bushfires near Echunga. Adelaide Chronicle, 22 March 1902; 12. [Google Scholar]
- Anonymous. Bush fire at Norton’s summit. Adelaide Chronicle, 16 March 1907; 40. [Google Scholar]
- Anonymous. Bush fire at Marble Hill. The Adelaide Register, 25 February 1909; 5. [Google Scholar]
- Anonymous. Fighting the flames. The Adelaide Register, 22 February 1910; 7. [Google Scholar]
- Anonymous. Hot time at Marble Hill. The Adelaide Register, 15 January 1912; 6. [Google Scholar]
- Anonymous. A record blaze. The Adelaide Register, 3 February 1912; 16. [Google Scholar]
- Anonymous. An excited township. The Adelaide Register, 3 February 1912; 16. [Google Scholar]
- Anonymous. Bush fire near Marble Hill. The Adelaide Register, 24 January 1914; 17. [Google Scholar]
- Anonymous. A fire near Marble Hill. The Adelaide Advertiser, 8 March 1916; 5. [Google Scholar]
- Anonymous. Bush fires in the hills. The Adelaide Register, 12 February 1918; 5. [Google Scholar]
- Anonymous. Progress of hills fire. The Adelaide Register, 5 February 1920; 7. [Google Scholar]
- Anonymous. Hills bush fire. The Adelaide Register, 24 February 1920; 3. [Google Scholar]
- Anonymous. Bush fire above Morialta. Adelaide Daily Herald, 22 February 1923; 2. [Google Scholar]
- Anonymous. The district ablaze. The Gawler Bunyip, 18 February 1926; 2. [Google Scholar]
- Anonymous. Serious fires in Adelaide Hills. Port Pirie Recorder, 13 March 1929; 1. [Google Scholar]
- Anonymous. Governor combats flames—Summer residence threatened. The Melbourne Argus, 19 February 1930; 7. [Google Scholar]
- Anonymous. Bushfires in Adelaide Hills. The Adelaide Register News-Pictorial, 9 February 1931; 2. [Google Scholar]
- Anonymous. Fire near Marble Hill under control. Adelaide News, 27 February 1933; 5. [Google Scholar]
- Anonymous. Fourth day of hills fire. The Adelaide Advertiser, 6 March 1934; 15. [Google Scholar]
- Anonymous. £250 damage in hills fire. Adelaide News, 4 April 1936; 1. [Google Scholar]
- Anonymous. Scrub fire at Marble Hill. Governor’s residence threatened. Adelaide Chronicle, 7 April 1938; 47. [Google Scholar]
- Foley, J.C. A Study of Meteorological Conditions Associated with Bush and Grass Fires and Fire Protection Strategy in Australia; Commonwealth of Australia Bulletin No. 38; Bureau of Meteorology: Melbourne, VIC, Australia, 1947; p. 234.
- Anonymous. Bushfires. Gawler Bunyip, 22 March 1940; 1. [Google Scholar]
- Anonymous. Bushfire at Mount Lofty. The Adelaide Advertiser, 16 February 1942; 3. [Google Scholar]
- Anonymous. Bushfires kill sheep. Adelaide Chronicle, 14 January 1943; 21. [Google Scholar]
- Anonymous. Bushfire in hills. Adelaide Chronicle, 27 January 1944; 23. [Google Scholar]
- Anonymous. Worst bushfire toll for years. The Adelaide Advertiser, 23 January 1948; 1. [Google Scholar]
- Anonymous. Fire threat to viceregal home. Adelaide News, 22 April 1950; 1. [Google Scholar]
- Auhl, I. From Settlement to City. A History of Tea Tree Gully 1836–1976; Lynton Publications: Blackwood, SA, Australia, 1978. [Google Scholar]
- Country Fire Service, South Australia (CFS). Bushfire History. Available online: http://www.cfs.sa.gov.au/site/about/history/bushfire_history.jsp (accessed on 10 April 2014).
- Rice, S.; Hegarty, A.; Conlin, T.; Kelton, S. Adelaide Hills bushfire-blaze near Cherryville in Mount Lofty Ranges declared contained. The Adelaide Advertiser, 11 May 2013. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gill, A.M.; McKenna, D.J.; Wouters, M.A. Landscape Fire, Biodiversity Decline and a Rapidly Changing Milieu: A Microcosm of Global Issues in an Australian Biodiversity Hotspot. Land 2014, 3, 1091-1136. https://doi.org/10.3390/land3031091
Gill AM, McKenna DJ, Wouters MA. Landscape Fire, Biodiversity Decline and a Rapidly Changing Milieu: A Microcosm of Global Issues in an Australian Biodiversity Hotspot. Land. 2014; 3(3):1091-1136. https://doi.org/10.3390/land3031091
Chicago/Turabian StyleGill, A. Malcolm, David J. McKenna, and Michael A. Wouters. 2014. "Landscape Fire, Biodiversity Decline and a Rapidly Changing Milieu: A Microcosm of Global Issues in an Australian Biodiversity Hotspot" Land 3, no. 3: 1091-1136. https://doi.org/10.3390/land3031091
APA StyleGill, A. M., McKenna, D. J., & Wouters, M. A. (2014). Landscape Fire, Biodiversity Decline and a Rapidly Changing Milieu: A Microcosm of Global Issues in an Australian Biodiversity Hotspot. Land, 3(3), 1091-1136. https://doi.org/10.3390/land3031091




