Changes in the Occurrence of Five Invasive Plant Species in Different Ecosystem Types between 2009–2018 in Hungary
Abstract
:1. Introduction
- What has been the trend in the level of invasion of different types of land cover (ecosystems) in Hungary between 2006 and 2018?
- Which types of ecosystems of conservation importance are most threatened by the biological invasion of the studied species?
2. Materials and Methods
2.1. Studied Invasive Species
2.2. Used Databases
2.2.1. National Geospatial Database of Invasive Plants (NGDIP) of Hungary
2.2.2. Ecosystem Map of Hungary (EMH)
2.3. GIS and Statistical Methods
3. Results
3.1. SpatioTemporal Characterestics of Occurence of Ailanthus altissima in Different Ecosystem Types
3.2. SpatioTemporal Characteristics of Occurrence Asclepias syriaca in Different Ecosystem Types
3.3. Spatiotemporal Characteristics of the Occurrence of Elaeagnus angustifolia in Different Ecosystem Types
3.4. Spatiotemporal Characteristics of Occurrence of Robinia pseudoacacia in Different Ecosystem Types
3.5. Spatiotemporal Characteristics of the Occurrence of Solidago spp. in Different Ecosystem Types
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Appendix A
| L2 Code | Level 2 (Ecosystem Type) | L3 Code | Level 3 (Ecosystem Sub-Type) |
|---|---|---|---|
| 11 | Buildings | 1110 | Low buildings |
| 1120 | High buildings | ||
| 12 | Roads and railways | 1210 | Paved roads |
| 1220 | Dirt roads | ||
| 1230 | Railways | ||
| 13 | Other paved or nonpaved artificial areas | 1310 | Other paved or nonpaved artificial areas |
| 14 | Green urban areas | 1410 | Green urban areas with trees |
| 1420 | Green urban areas without trees | ||
| 21 | Arable land | 2100 | Arable land |
| 22 | Permanent crops | 2210 | Vineyards |
| 2220 | Fruit and berry, and other plantations | ||
| 2230 | Energy crops | ||
| 23 | Complex cultivation pattern | 2310 | Complex cultivation patterns with scattered buildings |
| 2320 | Complex cultivation patterns without scattered buildings | ||
| 30 | Grasslands | 3110 | Open sand steppes |
| 3120 | Closed sand steppes | ||
| 3200 | Salt steppes and meadows (grasslands affected by salinisation included) | ||
| 3310 | Calcareous open rocky grasslands | ||
| 3320 | Siliceous open rocky grasslands | ||
| 3400 | Closed grasslands in hills and mountains or on cohesive soil | ||
| 35 | Other herbaceous vegetation | 3500 | Other herbaceous vegetation |
| 41 | Forests without excess water | 4101 | Beech forests |
| 4102 | Sessile oak-hornbeam forests | ||
| 4103 | Turkey oak forests | ||
| 4104 | Downy oak forests | ||
| 4105 | Scots pine stands of Western Transdanubia | ||
| 4106 | Deciduous stands of Western Transdanubia mixed with Scots pine | ||
| 4107 | Native poplar-dominated forests | ||
| 4108 | Pioneer forests of hilly and mountainous regions | ||
| 4109 | Pedunculate oak-hornbeam forests | ||
| 4110 | Pedunculate oak forests, monospecific or mixed with ash | ||
| 4111 | Forests dominated by other native tree species without excess water | ||
| 4112 | Other mixed deciduous forests | ||
| 42 | Natural riverine (gallery) forests | 4201 | Riverine willow-poplar woodlands |
| 4202 | Riverine hardwood forests | ||
| 43 | Other forests with excess water | 4301 | Pedunculate oak forests, monospecific or mixed with ash |
| 4302 | Alder forests | ||
| 4304 | Willow woods outside the floodplain | ||
| 4305 | Poplar woods outside the floodplain | ||
| 4306 | Birch woodland | ||
| 4307 | Turkey oak forests with excess water | ||
| 4308 | Forests dominated by other native tree species (WEW) | ||
| 4309 | Other mixed deciduous forests with excess water | ||
| 44 | Plantations | 4401 | Conifer-dominated plantations |
| 4402 | Black locust-dominated mixed plantations | ||
| 4403 | Plantations dominated by non-native poplar and willow species | ||
| 4404 | Plantations of other nonnative tree species | ||
| 45 | Nonwooded areas registered as forest or areas under reforestation | 4501 | Clearcut |
| 4502 | Forest stand under regeneration | ||
| 46 | Other ligneous vegetation, woodlands | 4600 | Other ligneous vegetation, woodlands |
| 50 | Herbaceous or woodland-dominated wetlands | 5110 | Tall-herb vegetation of marshes and fens standing in water |
| 5120 | Fens and mesotrophic wet meadows, grasslands with periodic water effect | ||
| 5200 | Swamp woodlands | ||
| 60 | Water bodies or courses | 6100 | Water bodies |
| 6200 | Watercourses |
| EMH Ecosystem Types | Numbers and Percentages of LUCAS Points Invaded with Ailanthus altissima | National Average of Ailanthus altissima Invasion | Regression Values = Invasion Percentages − National Average | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2009 | 2012 | 2015 | 2018 | 2009 | 2012 | 2015 | 2018 | 2009 | 2012 | 2015 | 2018 | |||||
| Num. | Per. | Num. | Per. | Num. | Per. | Num. | Per. | |||||||||
| Plantations | 12 | 3.02% | 8 | 2.17% | 50 | 13.93% | 8 | 2.86% | 1.64% | 1.05% | 1.56% | 1.96% | 1.38% | 1.12% | 12.37% | 0.90% |
| Nonwooded areas registered as forest or areas under reforestation | 2 | 5.41% | 2 | 5.41% | 4 | 14.29% | 1 | 3.57% | 1.64% | 1.05% | 1.56% | 1.96% | 3.77% | 4.36% | 12.73% | 1.61% |
| EMH L2 Codes | EMH Ecosystem Types | Rate of Deviation of Sampling Points Over the National Average Infection Level Per Year | Coefficient of Determination for Linear Trend | Coefficient of Determination for Logarithmic Trend | Deviation Due to Normalization | Coefficient of Determination for Exponential Trend | Coefficient of Determination for Binomial Trend | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 2009 | 2012 | 2015 | 2018 | |||||||
| 11 | Buildings | 6.69% | 2.23% | 0.40% | 2.26% | 0.53 | 0.72 | 0% | 0.73 | 0.86 |
| 12 | Roads and railways | 1.25% | 1.44% | 1.94% | 2.83% | 0.92 | 0.78 | 0% | 0.97 | 0.86 |
| 13 | Other paved or nonpaved artificial areas | 2.21% | 11.45% | −1.56% | 1.07% | 0.14 | 0.07 | 1.56% | 0.02 | 0 |
| 14 | Green urban areas | 1.33% | 0.79% | 1.58% | 2.10% | 0.54 | 0.36 | 0% | 0.62 | 0.42 |
| 21 | Arable land | −1.25% | −0.70% | 0.22% | −1.50% | 0 | 0.03 | 1.50% | 0.13 | 0.13 |
| 22 | Permanent crops | −1.64% | 0.40% | 10.35% | 0.04% | 0.13 | 0.2 | 1.64% | 0.04 | 0.01 |
| 23 | Complex cultivation pattern | 2.44% | −1.05% | 7.14% | 2.12% | 0.08 | 0.07 | 1.05% | 0.03 | 0.08 |
| 30 | Grasslands | −0.04% | −0.59% | 4.37% | −0.94% | 0.01 | 0.04 | 0.94% | 0 | 0.05 |
| 35 | Other herbaceous vegetation | 0.16% | −1.05% | 0.88% | 2.74% | 0.62 | 0.43 | 1.05% | 0.84 | 0.67 |
| 41 | Forests without excess water | −0.01% | −0.25% | 1.85% | 0.83% | 0.39 | 0.4 | 0.25% | 0.15 | 0.33 |
| 42 | Natural riverine (gallery) forests | −1.64% | 7.29% | −1.56% | 2.80% | 0.02 | 0.05 | 1.64% | 0.02 | 0.01 |
| 43 | Other forests with excess water | −0.34% | −1.05% | 2.73% | −0.37% | 0.08 | 0.1 | 1.05% | 0.02 | 0.08 |
| 44 | Plantations | 1.38% | 1.12% | 12.37% | 0.90% | 0.05 | 0.09 | 0% | 0.04 | 0.08 |
| 45 | Nonwooded areas registered as forest or areas under reforestation | 3.77% | 4.36% | 12.73% | 1.6% | 0 | 0.02 | 0% | 0.01 | 0.03 |
| 46 | Other ligneous vegetation woodlands | 3.36% | 1.82% | 7.46% | 1.12% | 0 | 0 | 0% | 0 | 0 |
| 50 | Herbaceous or woodland-dominated wetlands | −1.16% | −1.05% | 0.24% | −0.76% | 0.25 | 0.32 | 1.16% | 0 | 0.02 |
| 60 | Water bodies or courses | −1.64% | −1.05% | −1.56% | −1.96% | 0.25 | 0.11 | 1.96% | 0 | 0 |
| EMH L2 Codes | EMH Ecosystem Types | Rate of Deviation of Sampling Points over the National Average Infection Level Per Year | Correlation Coefficient Value for Linear Trend | Correlation Coefficient Value for Logarithmic Trend | Deviation Due to Normalization | Correlation Coefficient Value for Exponential Trend | Correlation Coefficient Value for Binomial Trend | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 2009 | 2012 | 2015 | 2018 | |||||||
| 11 | Buildings | −0.75% | −1.29% | −2.62% | −4.50% | 0.95 | 0.82 | 4.50% | 0.45 | 0.41 |
| 12 | Roads and railways | −2.35% | −1.94% | −0.94% | −2.45% | 0.02 | 0.07 | 2.45% | 0.15 | 0.15 |
| 13 | Other paved or nonpaved artificial areas | −4.92% | −2.93% | −4.40% | −4.50% | 0 | 0.02 | 4.92% | 0.07 | 0.07 |
| 14 | Green urban areas | −2.25% | −2.32% | −1.24% | −2.06% | 0.18 | 0.23 | 2.32% | 0.03 | 0.13 |
| 21 | Arable land | −2.75% | −2.34% | −2.59% | −1.73% | 0.65 | 0.57 | 2.76% | 0.84 | 0.80 |
| 22 | Permanent crops | 6.71% | 11.56% | 8.93% | −0.50% | 0.37 | 0.19 | 0.50% | 0 | 0 |
| 23 | Complex cultivation pattern | −0.84% | 4.76% | 4.69% | 5.70% | 0.72 | 0.86 | 0.84% | 0.27 | 0.31 |
| 30 | Grasslands | 0.28% | 1.90% | 1.85% | 4.43% | 0.87 | 0.80 | 0% | 0.91 | 0.89 |
| 35 | Other herbaceous vegetation | 4.09% | 2.82% | −1.93% | 0.21% | 0.62 | 0.68 | 1.93% | 0.67 | 0.56 |
| 41 | Forests without excess water | −0.72% | 0.25% | −0.93% | −0.91% | 0.16 | 0.07 | 0.93% | 0 | 0 |
| 42 | Natural riverine (gallery) forests | 6.85% | 5.40% | −4.40% | −4.50% | 0.85 | 0.82 | 4.50% | 0.47 | 0.44 |
| 43 | Other forests with excess water | −4.92% | −2.93% | 0.08% | −2.91% | 0.32 | 0.46 | 4.92% | 0 | 0.01 |
| 44 | Plantations | 12.71% | 8.72% | 11.17% | 10.86% | 0.06 | 0.15 | 0% | 0.06 | 0.16 |
| 45 | Nonwooded areas registered as forest or areas under reforestation | 11.30% | −0.23% | 10.98% | 2.64% | 0.11 | 0.15 | 0.23% | 0.09 | 0.15 |
| 46 | Other ligneous vegetation woodlands | 4.25% | 2.80% | 4.97% | 0.88% | 0.32 | 0.25 | 0% | 0.24 | 0.21 |
| 50 | Herbaceous or woodland-dominated wetlands | −3.01% | −1.33% | −2.57% | −3.90% | 0.22 | 0.08 | 3.90% | 0.01 | 0.02 |
| 60 | Water bodies or courses | −4.92% | −2.93% | −4.40% | −4.50% | 0 | 0.02 | 4.92% | 0.07 | 0.07 |
| EMH L2 Codes | EMH Ecosystem Types | Rate of Deviation of Sampling Points over the National Average Infection Level Per Year | Coefficient of Determination for Linear Trend | Coefficient of Determination for Logarithmic Trend | Deviation Due to Normalization | Coefficient of Determination for Exponential Trend | Coefficient of Determination for Binomial Trend | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 2009 | 2012 | 2015 | 2018 | |||||||
| 11 | Buildings | −2.86% | 1.77% | −3.77% | −1.74% | 0.01 | 0 | 3.77% | 0 | 0.03 |
| 12 | Roads and railways | −0.44% | −0.52% | −1.29% | −0.71% | 0.28 | 0.35 | 1.29% | 0.39 | 0.34 |
| 13 | Other paved or nonpaved artificial areas | −4.94% | −1.51% | 0.58% | −1.74% | 0.44 | 0.62 | 4.94% | 0 | 0.01 |
| 14 | Green urban areas | −0.19% | 0.94% | 2.24% | −0.38% | 0.01 | 0.06 | 0.38% | 0.18 | 0.18 |
| 21 | Arable land | −0.38% | −0.72% | −1.34% | −0.81% | 0.39 | 0.51 | 1.34% | 0.58 | 0.58 |
| 22 | Permanent crops | −0.29% | −1.51% | −3.77% | −1.74% | 0.35 | 0.47 | 3.77% | 0.56 | 0.55 |
| 23 | Complex cultivation pattern | −4.94% | −1.51% | −3.77% | −1.74% | 0.33 | 0.41 | 4.94% | 0.25 | 0.24 |
| 30 | Grasslands | 7.26% | 0.79% | 3.87% | 5.15% | 0.02 | 0.11 | 0% | 0.02 | 0.13 |
| 35 | Other herbaceous vegetation | −3.14% | −0.36% | 2.40% | −1.74% | 0.14 | 0.27 | 3.14% | 0.01 | 0 |
| 41 | Forests without excess water | −3.77% | −1.25% | −3.00% | −1.74% | 0.24 | 0.32 | 3.77% | 0.15 | 0.14 |
| 42 | Natural riverine (gallery) forests | −4.94% | −1.51% | 1.49% | −1.74% | 0.38 | 0.55 | 4.94% | 0 | 0 |
| 43 | Other forests with excess water | −4.94% | −0.32% | −3.77% | −1.74% | 0.15 | 0.23 | 4.94% | 0.09 | 0.08 |
| 44 | Plantations | −2.67% | 0.12% | −1.90% | −1.74% | 0.01 | 0.06 | 2.67% | 0.02 | 0.02 |
| 45 | Nonwooded areas registered as forest or areas under reforestation | −2.24% | −1.51% | 0.08% | −1.74% | 0.16 | 0.26 | 2.24% | 0.01 | 0 |
| 46 | Other ligneous vegetation, woodlands | −0.77% | 1.36% | 2.09% | −0.58% | 0.01 | 0.09 | 0.77% | 0.15 | 0.10 |
| 50 | Herbaceous or woodland-dominated wetlands | 2.68% | 3.28% | 9.65% | 7.84% | 0.68 | 0.69 | 0% | 0.58 | 0.69 |
| 60 | Water bodies or courses | 1.31% | 16.35% | 11.23% | 6.60% | 0.05 | 0.16 | 0% | 0 | 0.03 |
| EMH L2 Codes | EMH Ecosystem Types | Rate of Deviation of Sampling Points over the National Average Infection Level Per Year | Correlation Coefficient Value for Linear Trend | Correlation Coefficient Value for Logarithmic Trend | Deviation Due to Normalization | Correlation Coefficient Value for Exponential Trend | Correlation Coefficient Value for Binomial Trend | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 2009 | 2012 | 2015 | 2018 | |||||||
| 11 | Buildings | −0.38% | −0.17% | −1.48% | −8.80% | 0.70 | 0.52 | 8.80% | 0.17 | 0.16 |
| 12 | Roads and railways | 1.47% | 4.69% | −0.92% | 1.51% | 0.10 | 0.06 | 0.92% | 0.04 | 0.01 |
| 13 | Other paved or nonpaved artificial areas | −12.08% | −18.20% | −11.42% | −17.04% | 0.09 | 0.12 | 18.20% | 0.09 | 0.12 |
| 14 | Green urban areas | 3.10% | 6.65% | 5.75% | 1.07% | 0.13 | 0.03 | 0% | 0.05 | 0.01 |
| 21 | Arable land | −15.75% | −11.81% | −11.62% | −11.30% | 0.70 | 0.85 | 15.75% | 0.21 | 0.25 |
| 22 | Permanent crops | −11.19% | −10.95% | −5.10% | −14.07% | 0.01 | 0 | 14.07% | 0.03 | 0.02 |
| 23 | Complex cultivation pattern | −7.06% | 4.88% | 6.96% | 20.75% | 0.94 | 0.90 | 7.06% | 0.73 | 0.77 |
| 30 | Grasslands | −15.27% | −12.45% | −11.37% | −10.38% | 0.93 | 0.99 | 15.27% | 0.46 | 0.53 |
| 35 | Other herbaceous vegetation | 4.96% | 10.54% | 15.09% | 4.64% | 0.01 | 0.07 | 0% | 0.05 | 0.01 |
| 41 | Forests without excess water | −8.59% | −7.59% | −3.80% | −7.72% | 0.15 | 0.24 | 8.59% | 0.01 | 0 |
| 42 | Natural riverine (gallery) forests | −9.82% | −1.53% | 21.07% | −1.02% | 0.23 | 0.33 | 9.82% | 0.01 | 0 |
| 43 | Other forests with excess water | −6.69% | −0.34% | 8.11% | −10.55% | 0 | 0.01 | 10.55% | 0.11 | 0.11 |
| 44 | Plantations | 32.48% | 30.99% | 39.68% | 32.07% | 0.06 | 0.09 | 0% | 0.06 | 0.09 |
| 45 | Nonwooded areas registered as forest or areas under reforestation | 4.96% | 8.83% | 30.38% | 12.07% | 0.24 | 0.32 | 0% | 0.14 | 0.25 |
| 46 | Other ligneous vegetation, woodlands | 20.87% | 22.30% | 26.42% | 18.01% | 0.03 | 0 | 0% | 0.02 | 0 |
| 50 | Herbaceous or woodland-dominated wetlands | −16.99% | −7.56% | −8.45% | −11.69% | 0.21 | 0.39 | 16.99% | 0 | 0 |
| 60 | Water bodies or courses | −21.22% | −14.63% | −0.77% | −3.40% | 0.82 | 0.87 | 21.22% | 0.25 | 0.32 |
| EMH L2 Codes | EMH Ecosystem Types | Rate of Deviation of Sampling Points over the National Average Infection Level Per Year | Coefficient of Determination Value for Linear Trend | Coefficient of Determination for Logarithmic Trend | Deviation Due to Normalization | Coefficient of Determination for Exponential Trend | Coefficient of Determination for Binomial Trend | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 2009 | 2012 | 2015 | 2018 | |||||||
| 11 | Buildings | −8.40% | −6.89% | −5.72% | −7.69% | 0.14 | 0.27 | 8.40% | 0.02 | 0 |
| 12 | Roads and railways | −0.04% | −0.92% | 0.41% | −2.56% | 0.38 | 0.29 | 2.56% | 0.16 | 0.15 |
| 13 | Other paved or nonpaved artificial areas | −4.55% | −2.73% | −7.51% | −7.69% | 0.58 | 0.46 | 7.69% | 0.12 | 0.10 |
| 14 | Green urban areas | 1.69% | 2.00% | 4.20% | 0.98% | 0 | 0.02 | 0% | 0 | 0.02 |
| 21 | Arable land | −4.71% | −4.81% | −4.29% | −4.86% | 0 | 0.01 | 4.86% | 0.03 | 0.02 |
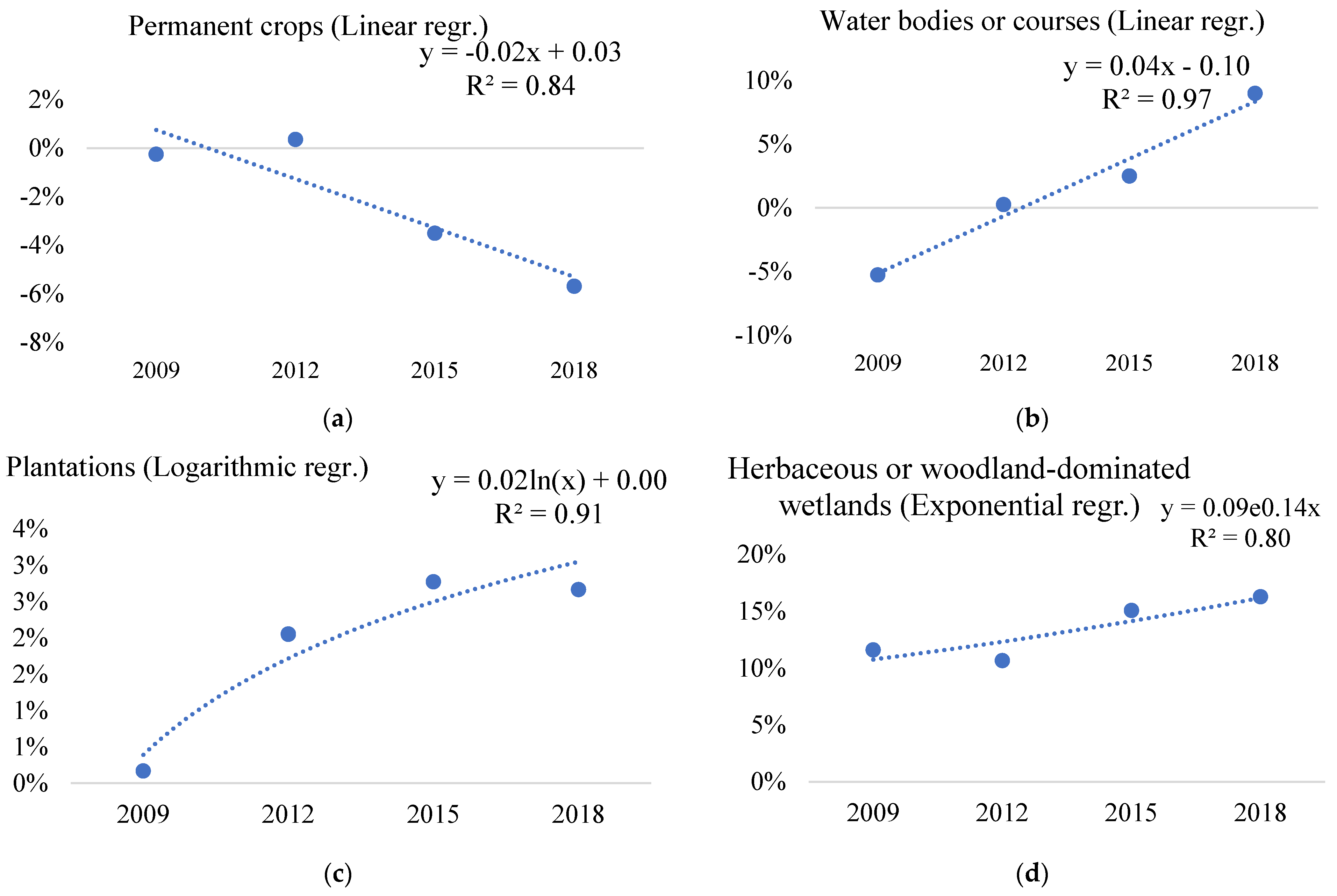
| 22 | Permanent crops | −0.26% | 0.35% | −3.51% | −5.69% | 0.84 | 0.69 | 5.69% | 0.28 | 0.25 |
| 23 | Complex cultivation pattern | 5.89% | 11.06% | 1.58% | 8.63% | 0 | 0 | 0% | 0 | 0 |
| 30 | Grasslands | 0.60% | 0.23% | −0.80% | 0.22% | 0.22 | 0.33 | 0.80% | 0.37 | 0.40 |
| 35 | Other herbaceous vegetation | 9.62% | 5.75% | 6.07% | 8.78% | 0.02 | 0.12 | 0% | 0.02 | 0.13 |
| 41 | Forests without excess water | −1.64% | 0% | −0.56% | −2.51% | 0.14 | 0.03 | 2.51% | 0.04 | 0.05 |
| 42 | Natural riverine (gallery) forests | 9.25% | 1.44% | 3.02% | 20.88% | 0.28 | 0.13 | 0% | 0.42 | 0.17 |
| 43 | Other forests with excess water | 9.79% | 10.96% | 13.39% | 9.77% | 0.03 | 0.10 | 0% | 0.03 | 0.09 |
| 44 | Plantations | 0.17% | 2.05% | 2.77% | 2.66% | 0.77 | 0.91 | 0% | 0.45 | 0.60 |
| 45 | Nonwooded areas registered as forest or areas under reforestation | −0.29% | 3.92% | −7.51% | 10.16% | 0.12 | 0.07 | 7.51% | 0.05 | 0.03 |
| 46 | Other ligneous vegetation, woodlands | 8.83% | 9.95% | 9.29% | 8.08% | 0.23 | 0.09 | 0% | 0.21 | 0.08 |
| 50 | Herbaceous or woodland-dominated wetlands | 11.60% | 10.66% | 15.05% | 16.26% | 0.78 | 0.65 | 0% | 0.80 | 0.69 |
| 60 | Water bodies or courses | −5.27% | 0.25% | 2.49% | 8.97% | 0.97 | 0.93 | 5.27% | 0.73 | 0.78 |
References
- Vilà, M.; Espinar, J.L.; Hejda, M.; Hulme, P.E.; Jarošík, V.; Maron, J.L.; Pergl, J.; Schaffner, U.; Sun, Y.; Pyšek, P. Ecological Impacts of Invasive Alien Plants: A Meta-Analysis of Their Effects on Species, Communities and Ecosystems. Ecol. Lett. 2011, 14, 702–708. [Google Scholar] [CrossRef]
- Lake, I.R.; Jones, N.R.; Agnew, M.; Goodess, C.M.; Giorgi, F.; Hamaoui-Laguel, L.; Semenov, M.A.; Solomon, F.; Storkey, J.; Vautard, R.; et al. Climate Change and Future Pollen Allergy in Europe. Environ. Health Perspect. 2017, 125, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Ehrenfeld, J.G.; Scott, N. Invasive Species and the Soil: Effects on Organisms and Ecosystem Processes. Ecol. Appl. 2001, 11, 1259–1260. [Google Scholar] [CrossRef]
- Bielecka, A.; Borkowska, L.; Królak, E. Environmental Changes Caused by the Clonal Invasive Plant Solidago Canadensis. Ann. Bot. Fenn. 2020, 57, 33. [Google Scholar] [CrossRef]
- Kitka, D.; Szilassi, P. Geographic Factors Influencing the Spreading of Invasive Species: A Gis-Based Case Study in the Southern Great Plain of Hungary. J. Landsc. Ecol. 2016, 14, 155–169. [Google Scholar]
- Szilassi, P.; Soóky, A.; Bátori, Z.; Hábenczyus, A.A.; Frei, K.; Tölgyesi, C.; van Leeuwen, B.; Tobak, Z.; Csikós, N. Natura 2000 Areas, Road, Railway, Water, and Ecological Networks May Provide Pathways for Biological Invasion: A Country Scale Analysis. Plants 2021, 10, 2670. [Google Scholar] [CrossRef]
- Chytrý, M.; Wild, J.; Pyšek, P.; Jarošík, V.; Dendoncker, N.; Reginster, I.; Pino, J.; Maskell, L.C.; Vilà, M.; Pergl, J.; et al. Projecting Trends in Plant Invasions in Europe under Different Scenarios of Future Land-Use Change. Glob. Ecol. Biogeogr. 2012, 21, 75–87. [Google Scholar] [CrossRef]
- Szilassi, P.; Visztra, G.V.; Soóky, A.; Bátori, Z.; Hábenczyus, A.A.; Frei, K.; Tölgyesi, C.; Balogh, M.B. Towards an Understanding of the Geographical Background of Plants Invasion as a Natural Hazard: A Case Study in Hungary. Geogr. Pannonica 2022, 26, 176–183. [Google Scholar] [CrossRef]
- Kovács-Hostyánszki, A.; Bereczki, K.; Czúcz, B.; Fabók, V.; Fodor, L.; Kalóczkai, Á.; Kiss, M.; Koncz, P.; Kovács, E.; Rezneki, R.; et al. Nemzeti Ökoszisztéma-Szolgáltatás Térképezés És Értékelés, Avagy a Természetvédelem Országos Programja. Természetvédelmi Közlemények 2019, 25, 80–90. [Google Scholar] [CrossRef]
- Tanács, E.; Belényesi, M.; Lehoczki, R.; Pataki, R.; Petrik, O.; Standovár, T.; Pásztor, L.; Laborczi, A.; Szatmári, G.; Molnár, Z.; et al. Országos, Nagyfelbontású Ökoszisztéma-Alaptérkép: Módszertan, Validáció És Felhasználási Lehetőségek. Természetvédelmi Közlemények 2019, 25, 34–58. [Google Scholar] [CrossRef]
- Tanács, E.; Bede-Fazekas, Á.; Csecserits, A.; Fodor, L.K.; Pásztor, L.; Somodi, I.; Varga, A.; Vári, Á. Assessing Ecosystem Condition at the National Level in Hungary-Indicators, Approaches, Challenges. One Ecosyst. 2022, 7, e81543. [Google Scholar] [CrossRef]
- Botta-Dukat, Z.; Balogh, L.; Szigetvári, C.; Bagi, I.; Dancza, I.; Udvardy, L. A Növényi Invázióhoz Kapcsolódó Fogalmak Áttekintése, Egyben Javaslat a Jövőben Használandó Fogalmakra És Azok Definícióira. In Biológiai inváziók Magyarországon: Özönnövények/[Biological Invasions in Hungary: Invasive Plants] Chapter: II; Botond, M., Botta-Dukat, Z., Eds.; A KvVM Természetvédelmi Hivatalának tanulmánykötetei 9, TermészetBÚVÁR Alapítvány Kiadó: Budapest, Hungary, 2004; pp. 35–59. ISBN 963 86107 5 1. [Google Scholar]
- European Alien Species Information Network—Species Explorer. Available online: https://easin.jrc.ec.europa.eu/spexplorer/search/ (accessed on 21 June 2023).
- Roy, D.; Alderman, D.; Anastasiu, P.; Arianoutsou, M.; Augustin, S.; Bacher, S.; Başnou, C.; Beisel, J.; Bertolino, S.; Bonesi, L.; et al. DAISIE-Inventory of Alien Invasive Species in Europe. 2020, Version 1.7. Research Institute for Nature and Forest (INBO). Available online: https://www.gbif.org/dataset/39f36f10-559b-427f-8c86-2d28afff68ca (accessed on 9 September 2023).
- Katsanevakis, S.; Deriu, I.; D’amico, F.; Nunes, A.L.; Sanchez, S.P.; Crocetta, F.; Arianoutsou, M.; Bazos, I.; Christopoulou, A.; Curto, G.; et al. European Alien Species Information Network (EASIN): Supporting European Policies and Scientific Research. Manag. Biol. Invasions 2015, 6, 147–157. [Google Scholar] [CrossRef]
- András, D.; Szilárd, C. A Mirigyes Bálványfa (Ailanthus altissima (Mill.) Swingle) Hazai Kutatásainak Áttekintése És Inváziójának Mértéke a Hazai Élőhelyeken. Természetvédelmi Közlemények 2016, 22, 20–32. [Google Scholar]
- Balogh, L.; Dancza, I.; Király, G. Preliminary Report on the Grid-Based Mapping of Invasive Plants in Hungary. Neobiota 2008, 7, 105–114. [Google Scholar]
- Sladonja, B.; Sušek, M.; Guillermic, J. Review on Invasive Tree of Heaven (Ailanthus altissima (Mill.) Swingle) Conflicting Values: Assessment of Its Ecosystem Services and Potential Biological Threat. Environ. Manag. 2015, 56, 1009–1034. [Google Scholar] [CrossRef] [PubMed]
- Kowarik, I.; Säumel, I. Biological Flora of Central Europe: Ailanthus altissima (Mill.) Swingle. Perspect. Plant Ecol. Evol. Syst. 2007, 8, 207–237. [Google Scholar] [CrossRef]
- Bhowmik, P.C.; Bandeen, J.D. The Bilogy of Canadian Weeds: 19. Asclepias syriaca L. Can. J. Plant Sci. 1976, 56, 579–589. [Google Scholar] [CrossRef]
- Bakacsy, L.; Bagi, I. Survival and Regeneration Ability of Clonal Common Milkweed (Asclepias syriaca L.) after a Single Herbicide Treatment in Natural Open Sand Grasslands. Sci. Rep. 2020, 10, 14222. [Google Scholar] [CrossRef]
- Follak, S.; Bakacsy, L.; Essl, F.; Hochfellner, L.; Lapin, K.; Schwarz, M.; Tokarska-Guzik, B.; Wołkowycki, D. Monograph of Invasive Plants in Europe N 6: Asclepias syriaca L. Bot. Lett. 2021, 168, 422–451. [Google Scholar] [CrossRef]
- Szilassi, P.; Szatmári, G.; Pásztor, L.; Árvai, M.; Szatmári, J.; Szitár, K.; Papp, L. Understanding the Environmental Background of an Invasive Plant Species (Asclepias syriaca) for the Future: An Application of LUCAS Field Photographs and Machine Learning Algorithm Methods. Plants 2019, 8, 593. [Google Scholar] [CrossRef]
- Papp, L.; Van Leeuwen, B.; Szilassi, P.; Tobak, Z.; Szatmári, J.; Árvai, M.; Mészáros, J.; Pásztor, L. Monitoring Invasive Plant Species Using Hyperspectral Remote Sensing Data. Land 2021, 10, 29. [Google Scholar] [CrossRef]
- Csiszár, Á.; Korda, M. Russian Olive (Elaeagnus angustifolia). In Practical Experiences in Invasive Alien Plant Control; Duna–Ipoly National Park Directorate: Esztergom, Hungary, 2015; pp. 214–215. [Google Scholar]
- Redei, K.; Osvath-Bujtas, Z.; Veperdi, I. Black Locust (Robinia pseudoacacia L.) Improvement in Hungary: A Review. Acta Silv. Et Lignaria Hung. 2008, 4, 127–132. [Google Scholar]
- Csiszár, Á.; Korda, M. Black Locust (Robinia pseudoacacia). In Practical Experiences in Invasive Alien Plant Control; Duna–Ipoly National Park Directorate: Esztergom, Hungary, 2015; pp. 208–210. [Google Scholar]
- Csiszár, Á.; Korda, M. Giant and Canadian Goldenrod (Solidago gigantea, Solidago canadensis). In Practical Experiences in Invasive Alien Plant Control; Duna–Ipoly National Park Directorate: Esztergom, Hungary, 2015; pp. 231–233. [Google Scholar]
- Botta-Dukát, Z.; Dancza, I. Giant and Canadian Goldenrod (Solidago gigantea Ait., S. canadensis L.). In The Most Important Invasive Plants in Hungary; Institute of Ecology and Botany, Hungarian Academy of Sciences: Vácrátót, Hungary, 2008; pp. 167–177. [Google Scholar]
- Botta-Dukát, Z.; Dancza, I. Aranyvessző Fajok (Solidago spp.). In Inváziós növényfajok Magyarországon; Csiszár, Á., Ed.; Nyugat-magyarországi Egyetemi Kiadó: Sopron, Hungary, 2012. [Google Scholar]
- Šutovská, M.; Capek, P.; Kocmálová, M.; Fraňová, S.; Pawlaczyk, I.; Gancarz, R. Characterization and Biological Activity of Solidago Canadensis Complex. Int. J. Biol. Macromol. 2013, 52, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Bölöni, J.; Molnár, Z.; Kun, A.; Biró, M. Általános Nemzeti Élőhely-Osztályozási Rendszer (Á-NÉR 2007). Kézirat MTA Öbki Vácrátót 2007, 184. [Google Scholar]
- Werner, P.A.; Bradbury, I.K.; Gross, R.S. The Biology of Canadian Weeds.: 45. Solidago canadensis L. Can. J. Plant Sci. 1980, 60, 1393–1409. [Google Scholar] [CrossRef]
- Fenesi, A.; Vágási, C.I.; Beldean, M.; Földesi, R.; Kolcsár, L.P.; Shapiro, J.T.; Török, E.; Kovács-Hostyánszki, A. Solidago Canadensis Impacts on Native Plant and Pollinator Communities in Different-Aged Old Fields. Basic. Appl. Ecol. 2015, 16, 335–346. [Google Scholar] [CrossRef]
- Varga, Z. A BIOLÓGIAI INVÁZIÓ ÁLTALÁNOS KÉRDÉSEI. Magy. Tudomány 2017, 178, 388–394. [Google Scholar]
- With, K.A. The Landscape Ecology of Invasive Spread. Conserv. Biol. 2002, 16, 1192–1203. [Google Scholar] [CrossRef]
- National Geospatial Database of Invasive Plants (NGDIP). Available online: http://www.geo.u-szeged.hu/invasive/ (accessed on 21 June 2023).
- d’Andrimont, R.; Yordanov, M.; Martinez-Sanchez, L.; Eiselt, B.; Palmieri, A.; Dominici, P.; Gallego, J.; Reuter, H.I.; Joebges, C.; Lemoine, G.; et al. Harmonised LUCAS In-Situ Land Cover and Use Database for Field Surveys from 2006 to 2018 in the European Union. Sci. Data 2020, 7, 352. [Google Scholar] [CrossRef]
- Tanács, E.; Vári, Á.; Bede-Fazekas, Á.; Báldi, A.; Csákvári, E.; Endrédi, A.; Fabók, V.; Kisné Fodor, L.; Kiss, M.; Koncz, P.; et al. Finding the Green Grass in the Haystack? Integrated National Assessment of Ecosystem Services and Condition in Hungary, in Support of Conservation and Planning. Sustainability 2023, 15, 8489. [Google Scholar] [CrossRef]
- Agrárminisztérium Ökoszisztéma Alaptérkép És Adatmodell Kialakítása. 2019. Budapest. Available online: http://okoszisztema-miniszter.hu/Magyarorszag-Okoszisztema-alapterkepe (accessed on 9 September 2023).
- Maes, J.; Teller, A.; Erhard, M.; Liquete, C.; Braat, L.; Berry, P.; Egoh, B.; Puydarrieux, P.; Fiorina, C.; Santos, F.; et al. Mapping and Assessment of Ecosystems and Their Services: An Analytical Framework for Ecosystem Assessments under Action. 5 of the EU Biodiversity Strategy to 2020; Publications Office of the European Union: Luxembourg, 2013. [Google Scholar]
- Anscombe, F.J. Graphs in Statistical Analysis. Am. Stat. 1973, 27, 17. [Google Scholar] [CrossRef]
- Guo, Q.; Fei, S.; Dukes, J.S.; Oswalt, C.M.; III, B.V.I.; Potter, K.M. A Unified Approach for Quantifying Invasibility and Degree of Invasion. Ecology 2015, 96, 2613–2621. [Google Scholar] [CrossRef] [PubMed]
- Chytrý, M.; Jarošík, V.; Pyšek, P.; Hájek, O.; Knollová, I.; Tichý, L.; Danihelka, J. Separating Habitat Invasibility by Alien Plants from the Actual Level of Invasion. Ecology 2008, 89, 1541–1553. [Google Scholar] [CrossRef]
- Godoy, O. Coexistence Theory as a Tool to Understand Biological Invasions in Species Interaction Networks: Implications for the Study of Novel Ecosystems. Funct. Ecol. 2019, 33, 1190–1201. [Google Scholar] [CrossRef]
- Liu, D.; Semenchuk, P.; Essl, F.; Lenzner, B.; Moser, D.; Blackburn, T.M.; Cassey, P.; Biancolini, D.; Capinha, C.; Dawson, W.; et al. The Impact of Land Use on Non-Native Species Incidence and Number in Local Assemblages Worldwide. Nat. Commun. 2023, 14, 2090. [Google Scholar] [CrossRef] [PubMed]
- Turbelin, A.J.; Cuthbert, R.N.; Essl, F.; Haubrock, P.J.; Ricciardi, A.; Courchamp, F. Biological Invasions Are as Costly as Natural Hazards. Perspect. Ecol. Conserv. 2023, 21, 143–150. [Google Scholar] [CrossRef]
- Hobbs, R.J.; Huenneke, L.F. Disturbance, Diversity, and Invasion: Implications for Conservations. Conserv. Biol. 1992, 6, 324–337. [Google Scholar] [CrossRef]
- Nielson, K.G.; Gill, K.M.; Springer, A.E.; Ledbetter, J.D.; Stevens, L.E.; Rood, S.B. Springs Ecosystems: Vulnerable Ecological Islands Where Environmental Conditions, Life History Traits, and Human Disturbance Facilitate Non-Native Plant Invasions. Biol. Invasions 2019, 21, 2963–2981. [Google Scholar] [CrossRef]
- Axmanová, I.; Kalusová, V.; Danihelka, J.; Dengler, J.; Pergl, J.; Pyšek, P.; Večeřa, M.; Attorre, F.; Biurrun, I.; Boch, S.; et al. Neophyte Invasions in European Grasslands. J. Veg. Sci. 2021, 32, e12994. [Google Scholar] [CrossRef]
- Ibáñez, I.; Petri, L.; Barnett, D.T.; Beaury, E.M.; Blumenthal, D.M.; Corbin, J.D.; Diez, J.; Dukes, J.S.; Early, R.; Pearse, I.S.; et al. Combining Local, Landscape, and Regional Geographies to Assess Plant Community Vulnerability to Invasion Impact. Ecol. Appl. 2023, 33, e2821. [Google Scholar] [CrossRef]
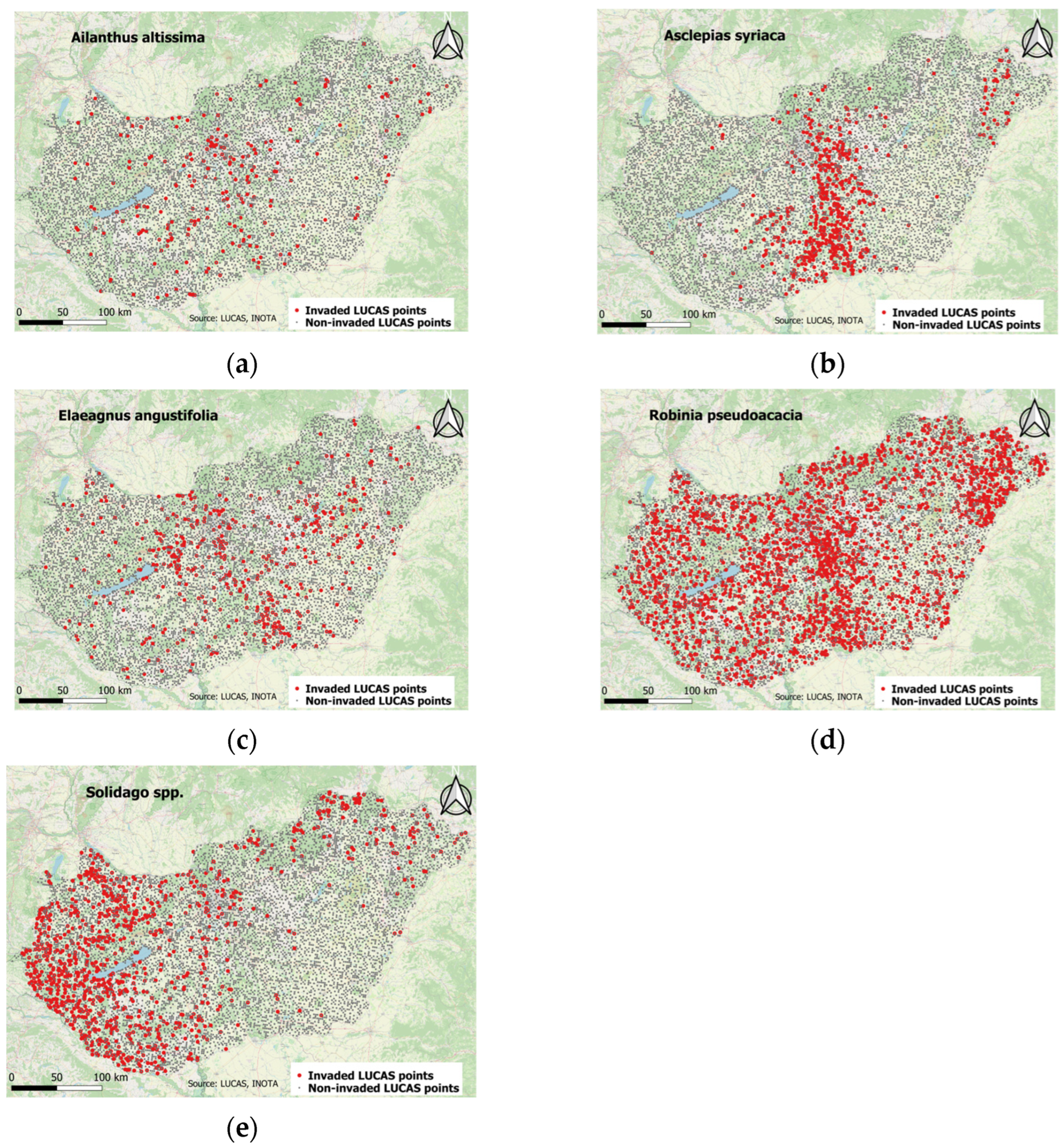

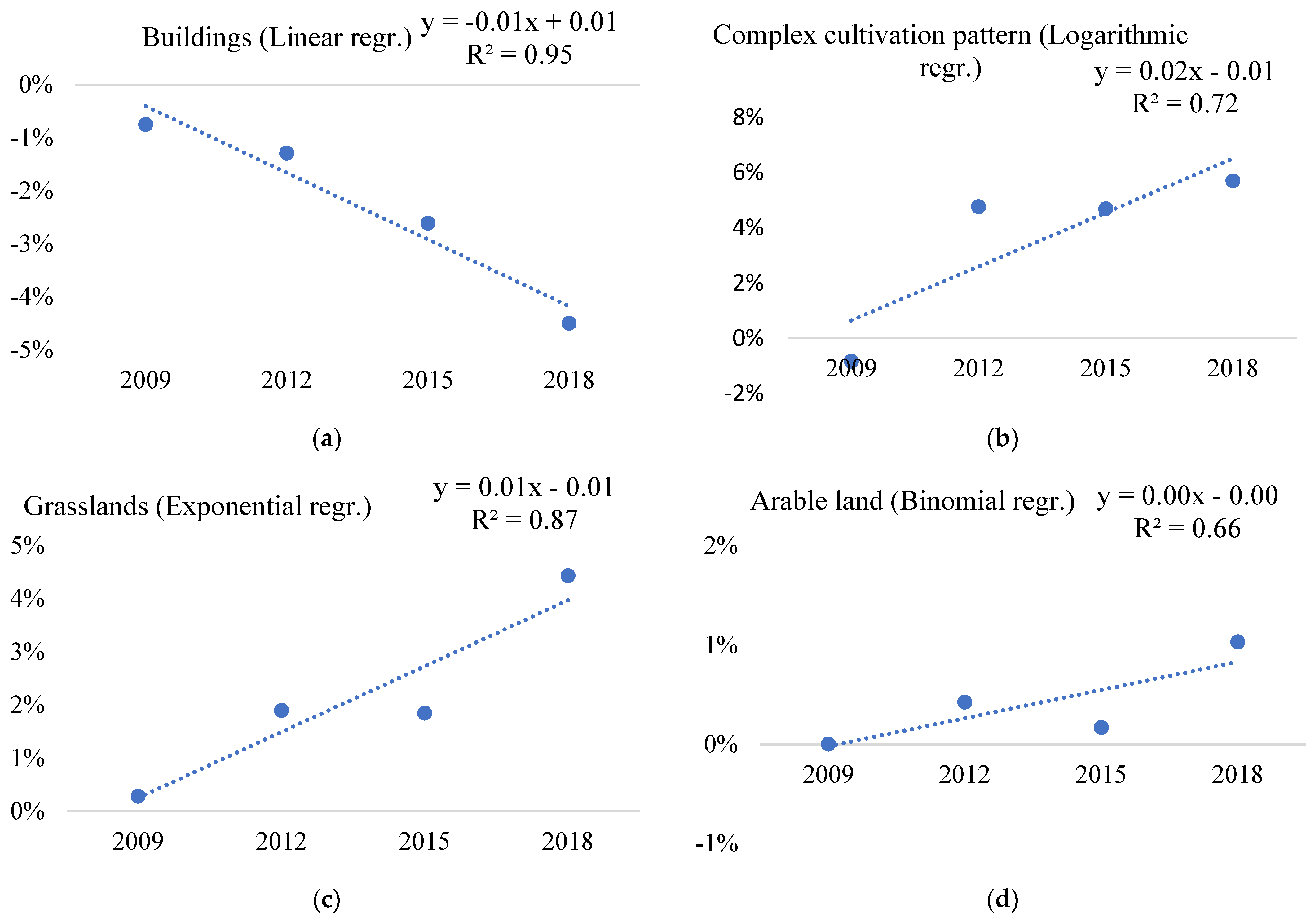
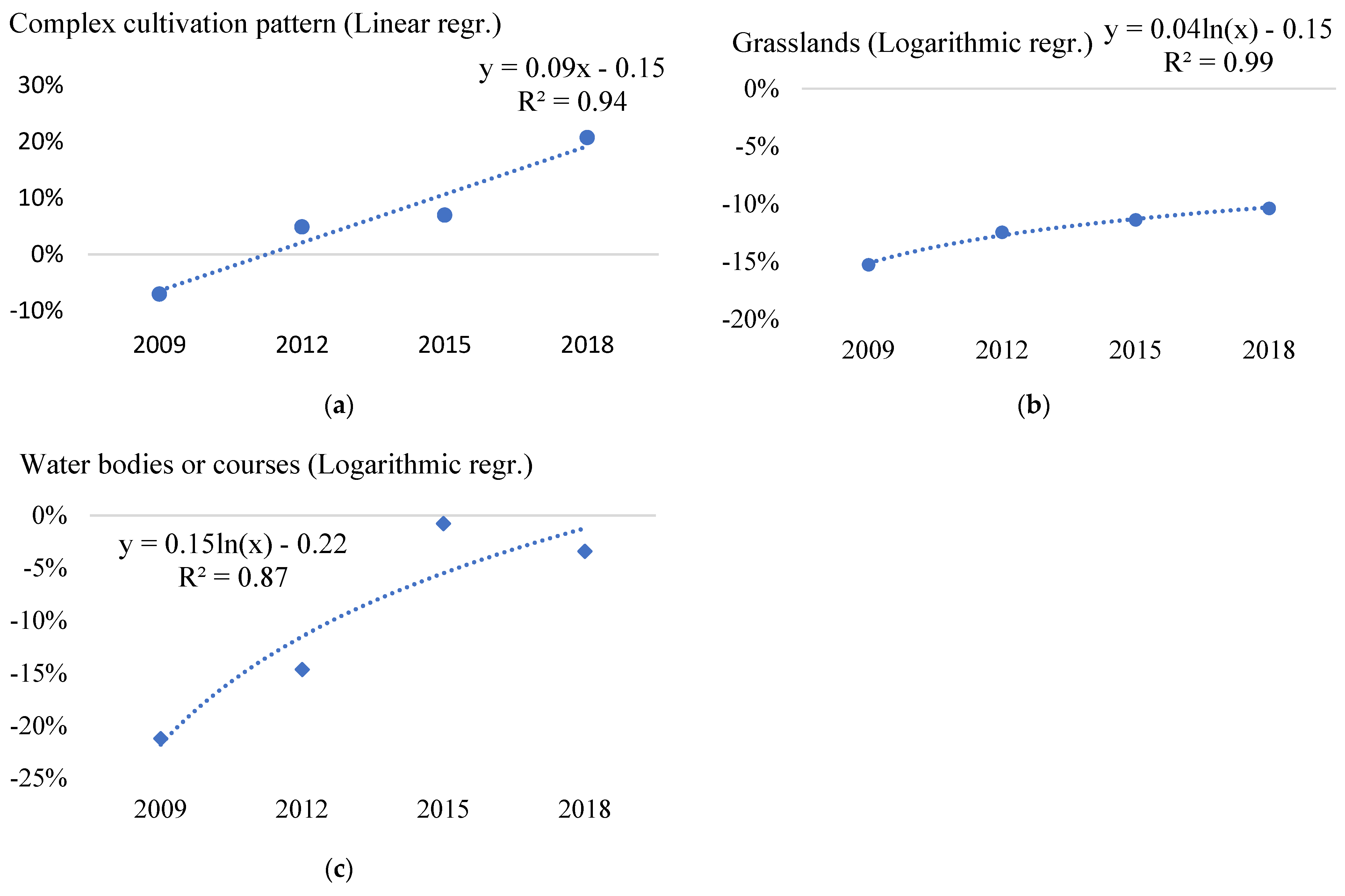
| Invasive Species | Invaded LUCAS Points in 2009 | Invaded LUCAS Points in 2012 | Invaded LUCAS Points in 2015 | Invaded LUCAS Points in 2018 | ||||
|---|---|---|---|---|---|---|---|---|
| (N) | Average Invasion | (N) | Average Invasion | (N) | Average Invasion | (N) | Average Invasion | |
| Ailanthus altissima | 86 | 1.64% | 48 | 1.05% | 71 | 1.56% | 80 | 1.96% |
| Asclepias syriaca | 250 | 4.92% | 132 | 2.93% | 195 | 4.40% | 175 | 4.50% |
| Eleaeagnus angustifolia | 251 | 4.94% | 69 | 1.51% | 168 | 3.77% | 71 | 1.74% |
| Robinia pseudoacacia | 1149 | 27.47% | 714 | 18.20% | 630 | 15.77% | 695 | 20.08% |
| Solidago spp. | 413 | 8.40% | 299 | 6.89% | 323 | 7.51% | 297 | 7.70% |
| All LUCAS points in Hungary | LUCAS points in 2009 | LUCAS points in 2012 | LUCAS points in 2015 | LUCAS points in 2018 | ||||
| 5332 = 100% | 4637 = 100% | 4625 = 100% | 4156 = 100% | |||||
| EMH Ecosystem Types | Rate of Deviation of Sampling Points over the National Average Infection Level Per Year | The Average Deviation of Invasion Rates among EMH Ecosystem Types | |||
|---|---|---|---|---|---|
| 2009 | 2012 | 2015 | 2018 | ||
| Nonwooded areas registered as forest or areas under reforestation | 3.77% | 4.36% | 12.73% | 1.61% | 5.62% |
| Plantations | 1.38% | 1.12% | 12.37% | 0.90% | 3.94% |
| Other ligneous vegetation, woodlands | 3.36% | 1.82% | 7.46% | 1.12% | 3.44% |
| Buildings | 6.69% | 2.23% | 0.40% | 2.26% | 2.90% |
| Roads and railways | 1.25% | 1.44% | 1.94% | 2.83% | 1.87% |
| Green urban areas | 1.33% | 0.79% | 1.58% | 2.10% | 1.45% |
| EMH Ecosystem Types | Rate of Deviation of Sampling Points over the National Average Infection Level Per Year | The Average Deviation of Invasion Rates among EMH Ecosystem Types | |||
|---|---|---|---|---|---|
| 2009 | 2012 | 2015 | 2018 | ||
| Plantations | 12.71% | 8.72% | 11.17% | 10.86% | 10.87% |
| Other ligneous vegetation, woodlands | 4.25% | 2.80% | 4.97% | 0.88% | 3.23% |
| Grasslands | 0.28% | 1.90% | 1.85% | 4.43% | 2.11% |
| EMH Ecosystem Types | Rate of Deviation of Sampling Points over the National Average Infection Level Per Year | The Average Deviation of Invasion Rates among EMH Ecosystem Types | |||
|---|---|---|---|---|---|
| 2009 | 2012 | 2015 | 2018 | ||
| Water bodies or courses | 1.31% | 16.35% | 11.23% | 6.60% | 8.87% |
| Herbaceous or woodland-dominated wetlands | 2.68% | 3.28% | 9.65% | 7.84% | 5.86% |
| Grasslands | 7.26% | 0.79% | 3.87% | 5.15% | 4.27% |
| EMH Ecosystem Types | Rate of Deviation of Sampling Points over the National Average Infection Level Per Year | The Average Deviation of Invasion Rates among EMH Ecosystem Types | |||
|---|---|---|---|---|---|
| 2009 | 2012 | 2015 | 2018 | ||
| Plantations | 32.48% | 30.99% | 39.68% | 32.07% | 33.81% |
| Other ligneous vegetation, woodlands | 20.87% | 22.30% | 26.42% | 18.01% | 21.90% |
| Nonwooded areas registered as forest or areas under reforestation | 4.96% | 8.83% | 30.38% | 12.07% | 14.06% |
| Other herbaceous vegetation | 4.96% | 10.54% | 15.09% | 4.64% | 8.81% |
| Green urban areas | 3.10% | 6.65% | 5.75% | 1.07% | 4.14% |
| EMH Ecosystem Types | Rate of Deviation of Sampling Points over the National Average Infection Level Per Year | The Average Deviation of INVASION Rates among EMH Ecosystem Types | |||
|---|---|---|---|---|---|
| 2009 | 2012 | 2015 | 2018 | ||
| Herbaceous or woodland-dominated wetlands | 11.60% | 10.66% | 15.05% | 16.26% | 13.39% |
| Other forests with excess water | 9.79% | 10.96% | 13.39% | 9.77% | 10.98% |
| Other ligneous vegetation, woodlands | 8.83% | 9.95% | 9.29% | 8.08% | 9.04% |
| Natural riverine (gallery) forests | 9.25% | 1.44% | 3.02% | 20.88% | 8.65% |
| Other herbaceous vegetation | 9.62% | 5.75% | 6.07% | 8.78% | 7.56% |
| Complex cultivation pattern | 5.89% | 11.06% | 1.58% | 8.63% | 6.79% |
| Green urban areas | 1.69% | 2.00% | 4.20% | 0.98% | 2.22% |
| Plantations | 0.17% | 2.05% | 2.77% | 2.66% | 1.91% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balogh, M.B.; Kertész, M.; Török, K.; Visztra, G.V.; Szilassi, P. Changes in the Occurrence of Five Invasive Plant Species in Different Ecosystem Types between 2009–2018 in Hungary. Land 2023, 12, 1784. https://doi.org/10.3390/land12091784
Balogh MB, Kertész M, Török K, Visztra GV, Szilassi P. Changes in the Occurrence of Five Invasive Plant Species in Different Ecosystem Types between 2009–2018 in Hungary. Land. 2023; 12(9):1784. https://doi.org/10.3390/land12091784
Chicago/Turabian StyleBalogh, Márton Bence, Miklós Kertész, Katalin Török, Georgina Veronika Visztra, and Péter Szilassi. 2023. "Changes in the Occurrence of Five Invasive Plant Species in Different Ecosystem Types between 2009–2018 in Hungary" Land 12, no. 9: 1784. https://doi.org/10.3390/land12091784
APA StyleBalogh, M. B., Kertész, M., Török, K., Visztra, G. V., & Szilassi, P. (2023). Changes in the Occurrence of Five Invasive Plant Species in Different Ecosystem Types between 2009–2018 in Hungary. Land, 12(9), 1784. https://doi.org/10.3390/land12091784








