Coexistence and Succession of Spontaneous and Planted Vegetation on Extensive Mediterranean Green Roofs: Impacts on Soil, Seed Banks, and Mesofauna
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site and Experimental Setup
2.2. Soil Analysis
2.3. Soil Seed Banks
2.4. Vegetation Survey
2.5. Collembola and Mite Survey
2.6. Data Analysis
3. Results
3.1. Effect of Substrate and Exposure on Soil Parameters
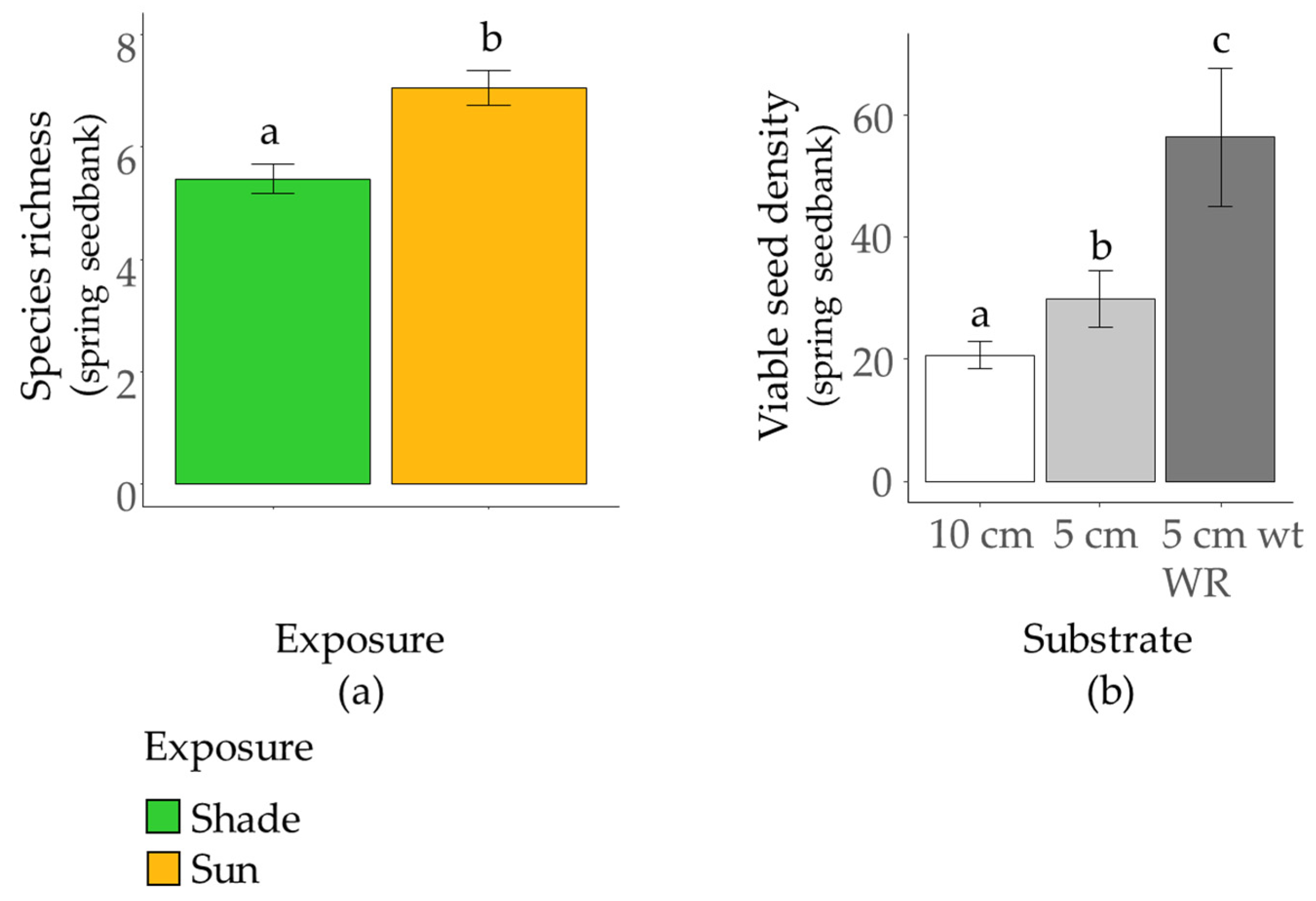
3.2. Effect of Substrate and Exposure on Seedbanks
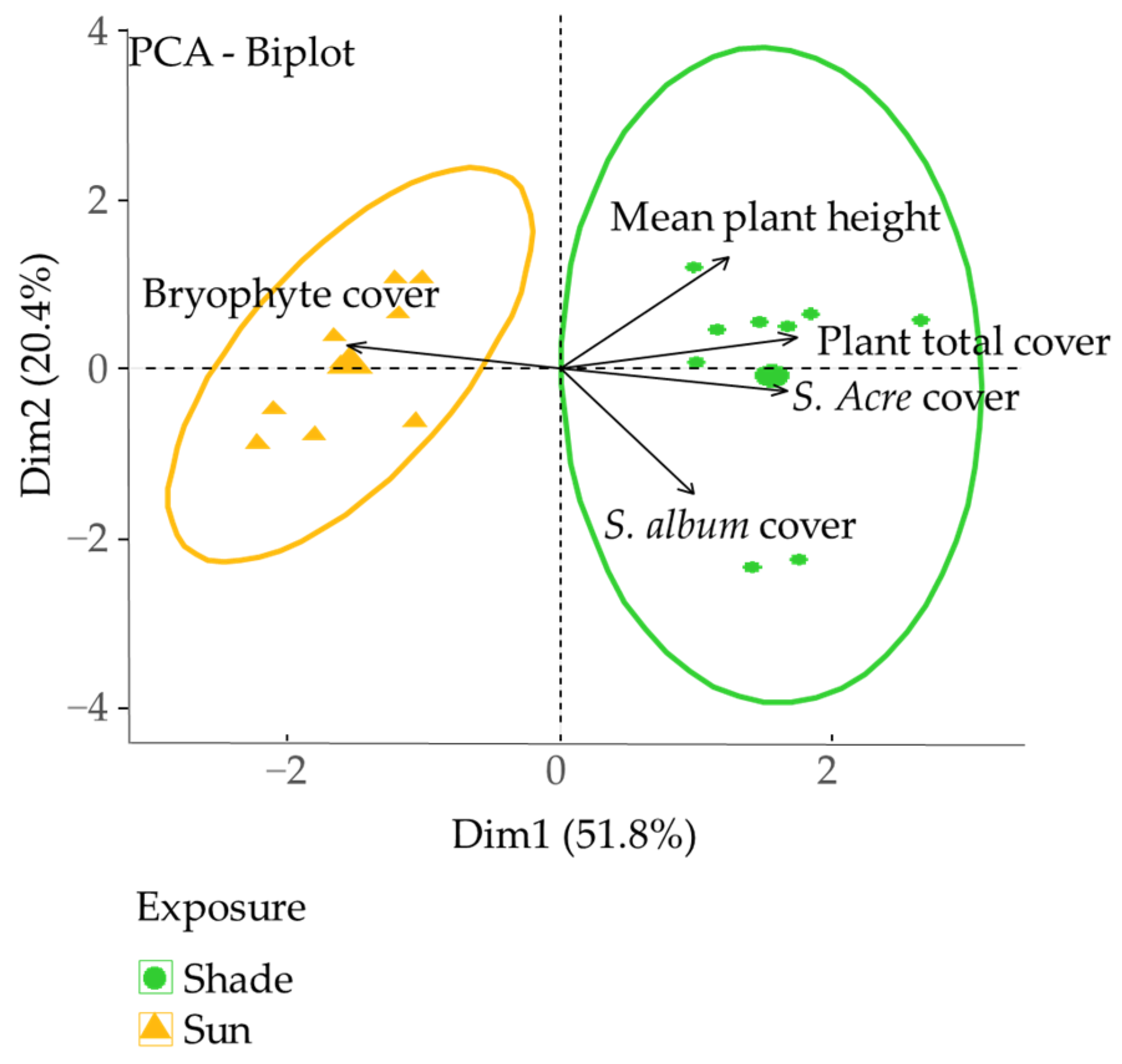
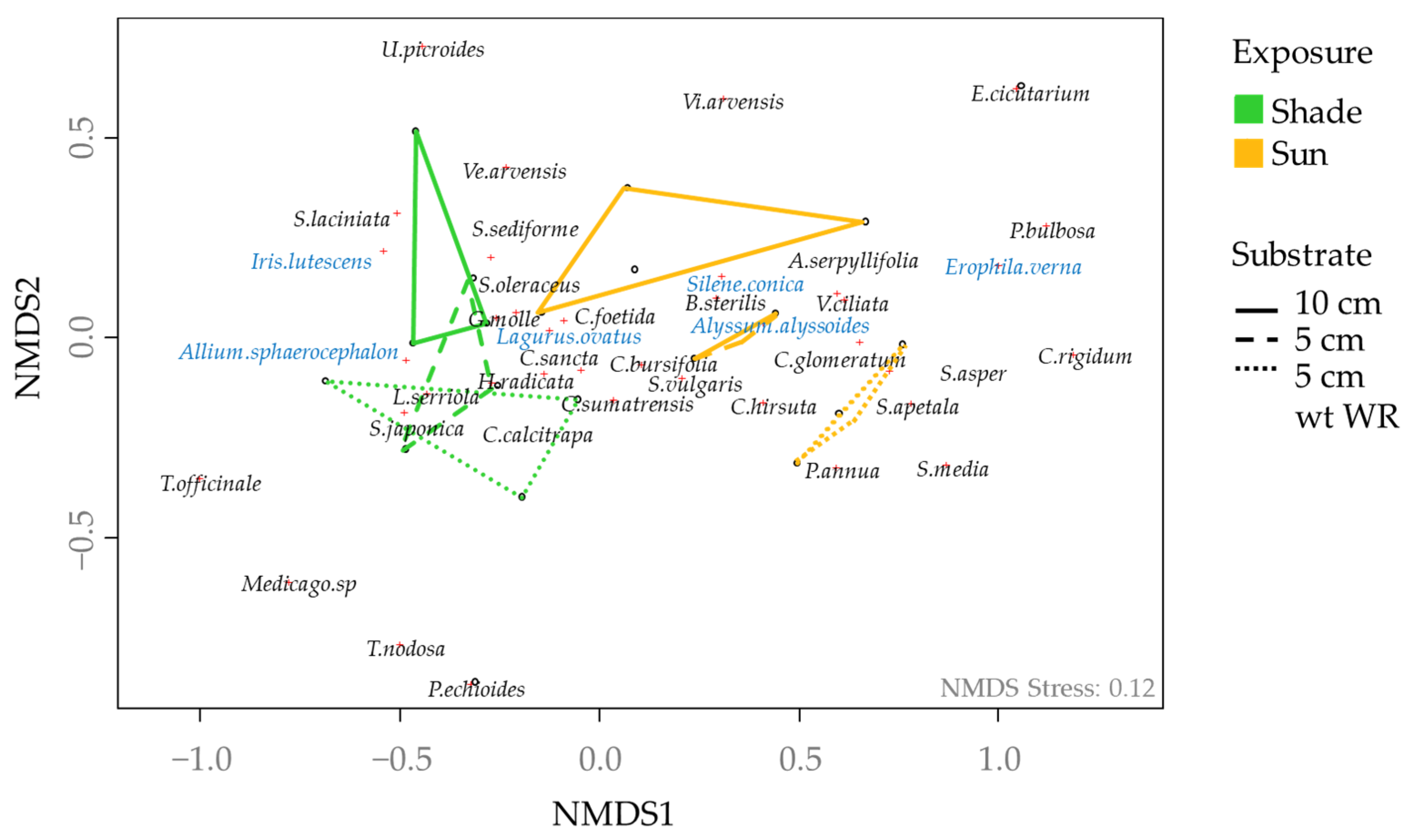
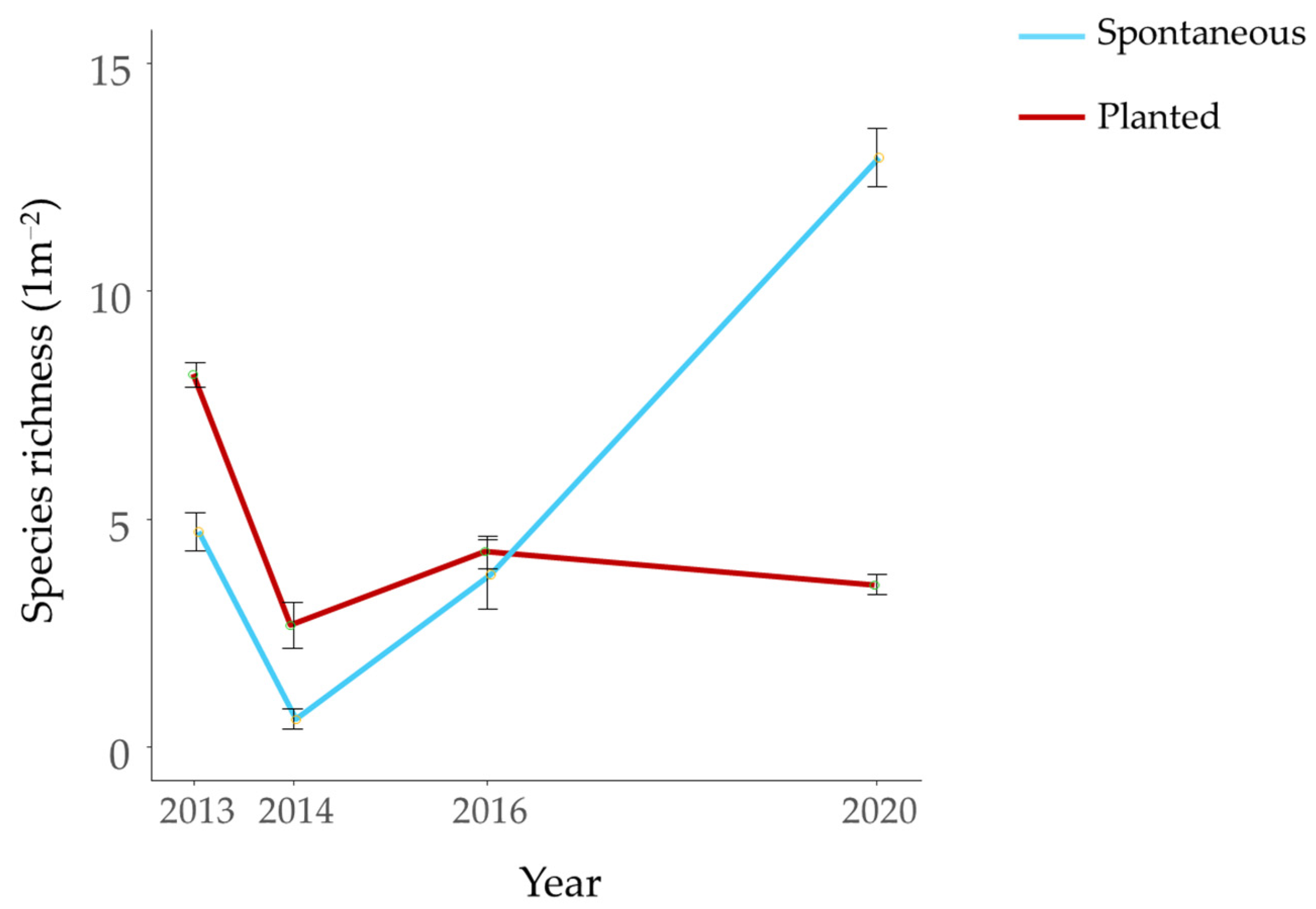
3.3. Effect of Substrate and Exposure on Mesological Data, Planted and Spontaneous Plant Community
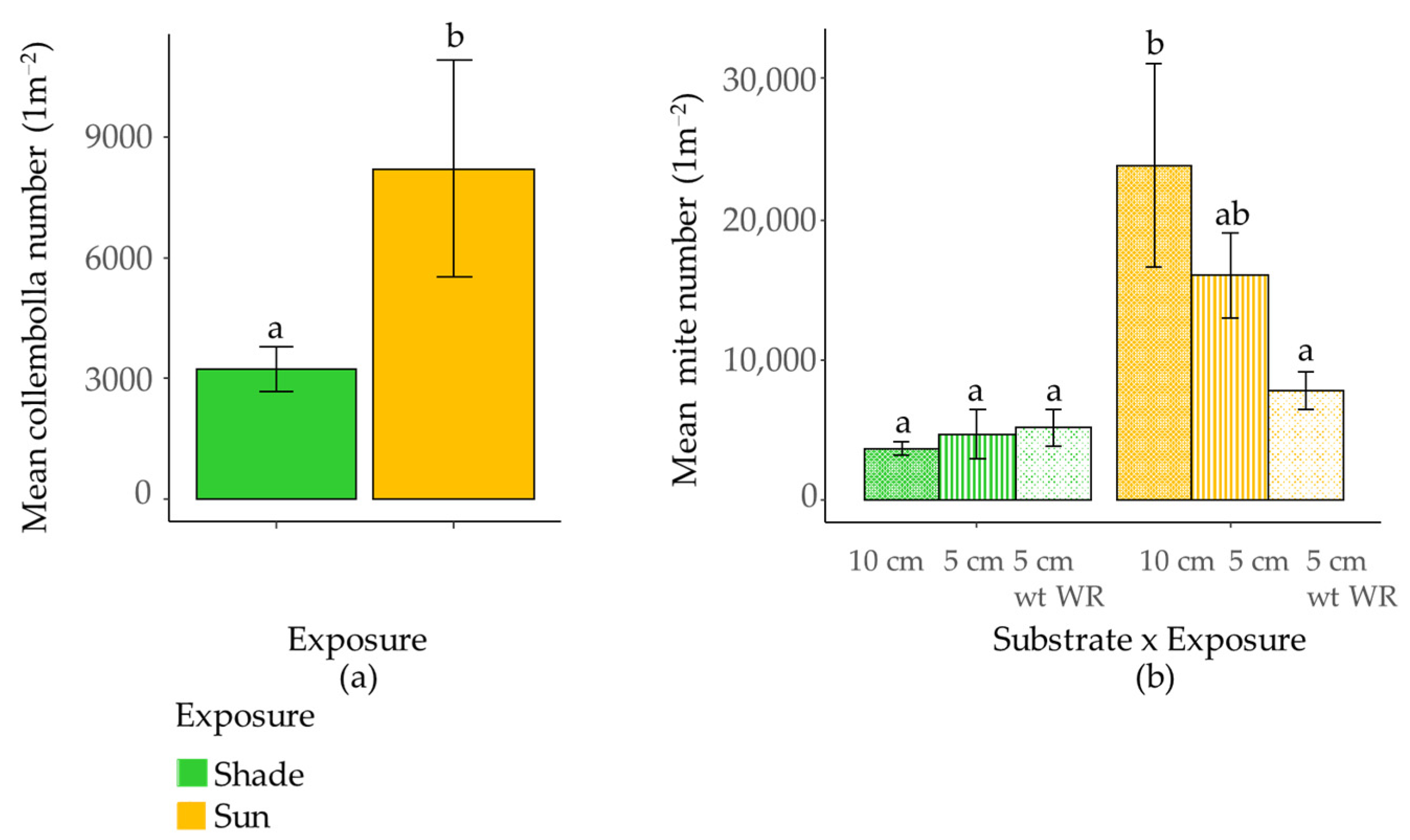
3.4. Effect of Substrate and Exposure on Collembola and Mite Density
3.5. Interactions between Studied Compartments in 2020
4. Discussion
4.1. Effect of Substrate on Physico-Chemical Characteristics of the Soil, Winter, and Spring Seed Banks, Mesofauna and Vegetation in the Medium-Term
4.2. Effect of Exposure on Physico-Chemical Characteristics of the Soil, Transient and Permanent Seed Banks, Mesofauna, and Vegetation in the Medium Term
4.3. Interactive Effect of Substrate and Exposure on Physico-Chemical Characteristics of the Soil, Winter and Spring Seed Banks, Mesofauna, and Vegetation in the Medium Term
4.4. Implications for Extensive Green Roof Installation, Management, and Sustainability under Mediterranean Climate Conditions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| (a) Planted species | ||||||||
| 2013 | 2014 | 2016 | 2020 | Spring seed bank | Winter seed bank | Life cycle | Biological form | |
| Allium sphaerocephalon | 1 | 1 | 1 | 1 | 0 | 0 | Perennial | Geophyte |
| Alyssum alyssoides | 1 | 0 | 1 | 1 | 0 | 0 | Annual | Therophyte/Hemicryptophyte |
| Carduus arvensis | 1 | 0 | 1 | 0 | 0 | 0 | Perennial | Geophyte |
| Clinopodium acinos | 0 | 0 | 0 | 0 | 0 | 0 | Annual | Therophyte/Hemicryptophyte |
| Dianthus superbus | 1 | 0 | 1 | 0 | 0 | 0 | Perennial | Hemicryptophyte/Geophyte |
| Erophila verna | 0 | 0 | 0 | 1 | 1 | 1 | Annual | Therophyte |
| Euphorbia cyparissias | 0 | 1 | 1 | 0 | 0 | 0 | Perennial | Hemicryptophyte/Geophyte |
| Helianthemum nummularium | 1 | 0 | 0 | 0 | 0 | 0 | Perennial | Phanérophyte |
| Iris lutescens | 1 | 1 | 1 | 1 | 0 | 0 | Perennial | Geophyte |
| Lagurus ovatus | 1 | 1 | 1 | 1 | 1 | 1 | Annual | Therophyte/Hemicryptophyte |
| Linum bienne | 0 | 1 | 1 | 0 | 0 | 0 | Biennal | Hemicryptophyte |
| Lobularia maritima | 1 | 1 | 1 | 0 | 1 | 1 | Perennial | Hemicryptophyte |
| Petrorhagia prolifera | 0 | 0 | 0 | 0 | 0 | 0 | Annual | Therophyte |
| Plantago afra | 1 | 1 | 0 | 0 | 0 | 0 | Annual | Therophyte |
| Sedum acre | 1 | 1 | 1 | 1 | 1 | 1 | Perennial | Chamephyte |
| Sedum album | 1 | 1 | 1 | 1 | 1 | 1 | Perennial | Chamephyte |
| Sideritis hyssopifolia | 1 | 0 | 0 | 0 | 0 | 0 | Annual-Perennial | T/H |
| Silene conica | 1 | 1 | 1 | 1 | 1 | 1 | Annual | Therophyte |
| (b) Spontaneous species | ||||||||
| 2013 | 2014 | 2016 | 2020 | Spring seed bank | Winter seed bank | Life cycle | Biological form | |
| Arenaria leptoclados | 1 | 0 | 1 | 0 | 0 | 0 | Annual | Therophyte |
| Arenaria serpyllifolia | 0 | 0 | 1 | 1 | 1 | 0 | Annual | Therophyte/Chamephyte |
| Symphyotrichum subulatum | 0 | 0 | 1 | 0 | 0 | 0 | Annual | Therophyte |
| Avena barbata | 0 | 1 | 1 | 1 | 0 | 0 | Annual | Therophyte |
| Anisantha sterilis | 0 | 0 | 0 | 1 | 1 | 0 | Annual | Therophyte |
| Cardamine hirsuta | 0 | 0 | 1 | 1 | 1 | 1 | Biennal | Therophyte/Hemicryptophyte |
| Catapodium rigidum | 0 | 0 | 0 | 1 | 0 | 0 | Annual | Therophyte |
| Celtis australis | 1 | 0 | 0 | 0 | 0 | 0 | Perennial | Phanérophyte |
| Centranthus calcitrapa | 0 | 0 | 0 | 1 | 1 | 0 | Annual | Therophyte |
| Cerastium glomeratum | 0 | 0 | 1 | 1 | 1 | 1 | Annual | Therophyte |
| Chenopodium album | 0 | 0 | 0 | 0 | 1 | 0 | ||
| Erigeron canadensis | 0 | 0 | 1 | 0 | 0 | 1 | Annual | Therophyte/Hemicryptophyte |
| Erigeron sumatrensis | 0 | 0 | 0 | 1 | 1 | 1 | Annual | Therophyte |
| Crepis bursifolia | 0 | 0 | 0 | 1 | 1 | 0 | Biennal | Hemicryptophyte |
| Crepis foetida | 1 | 1 | 1 | 1 | 0 | 0 | Annual | Therophyte/Hemicryptophyte |
| Crepis sancta | 0 | 0 | 0 | 1 | 0 | 0 | ||
| Crepis vesicaria | 1 | 0 | 0 | 1 | 1 | 0 | Biennal | Hemicryptophyte |
| Dactylis glomerata | 0 | 0 | 0 | 0 | 1 | 0 | ||
| Epilobium hirsutum | 1 | 0 | 0 | 0 | 1 | 1 | Perennial | Hemicryptophyte |
| Erodium cicutarium | 0 | 0 | 0 | 1 | 0 | 0 | Annual | Therophyte/Hemicryptophyte |
| Euphorbia maculata | 0 | 0 | 1 | 0 | 1 | 1 | Annual | Therophyte |
| Geranium molle | 1 | 1 | 0 | 1 | 1 | 0 | Annual | Therophyte |
| Hordeum murinum | 1 | 0 | 0 | 0 | 0 | 0 | Annual | Therophyte |
| Hypochaeris radicata | 0 | 0 | 0 | 1 | 0 | 0 | Perennial | Hemicryptophyte |
| Lactuca serriola | 0 | 0 | 1 | 1 | 0 | 0 | Biennal | Therophyte/Hemicryptophyte |
| Medicago sativa | 1 | 0 | 0 | 1 | 0 | 0 | Perennial | Hemicryptophyte |
| Minuartia hybrida | 1 | 0 | 1 | 0 | 0 | 0 | Annual | Therophyte |
| Papaver argemone | 1 | 0 | 0 | 0 | 0 | 0 | Annual | Therophyte |
| Picris echioides | 0 | 0 | 0 | 1 | 0 | 0 | Annual | Therophyte/Hemicryptophyte |
| Poa annua | 1 | 0 | 1 | 1 | 1 | 0 | Annual | Therophyte/Hemicryptophyte |
| Poa bulbosa | 0 | 0 | 0 | 1 | 0 | 0 | Perennial | Hemicryptophyte |
| Populus alba | 0 | 0 | 0 | 0 | 0 | 1 | Perennial | Phanérophyte |
| Rostraria cristata | 0 | 0 | 1 | 0 | 0 | 0 | Annual | Therophyte |
| Rumex crispus | 1 | 0 | 0 | 0 | 0 | 1 | Perennial | Hemicryptophyte |
| Sagina apetala | 0 | 0 | 1 | 1 | 1 | 1 | Annual | Therophyte |
| Podospermum laciniatum | 0 | 0 | 0 | 1 | 0 | 0 | Biennal | Hemicryptophyte |
| Sedum sediforme | 0 | 0 | 0 | 1 | 0 | 0 | Perennial | Chamephyte |
| Senecio vulgaris | 0 | 0 | 1 | 1 | 1 | 1 | Annual | Therophyte/Hemicryptophyte |
| Sonchus asper | 0 | 0 | 0 | 1 | 1 | 0 | Annual | Therophyte |
| Sonchus oleraceus | 1 | 1 | 1 | 1 | 1 | 0 | Annual | Therophyte/Hemicryptophyte |
| Sophora japonica | 0 | 0 | 1 | 1 | 0 | 0 | Perennial | Phanérophyte |
| Stellaria media | 0 | 0 | 0 | 1 | 1 | 1 | Annual | Therophyte/Chamephyte |
| Taraxacum officinale | 0 | 0 | 0 | 1 | 1 | 1 | Perennial | Hemicryptophyte |
| Torilis nodosa | 0 | 0 | 0 | 1 | 0 | 0 | Annual | Therophyte |
| Trifolium campestre | 1 | 0 | 1 | 0 | 0 | 0 | Annual | Therophyte |
| Typha latifolia | 0 | 0 | 0 | 0 | 1 | 1 | Perennial | Geophyte/Hemicryptophyte |
| Urospermum picroides | 0 | 0 | 0 | 1 | 0 | 0 | Annual | Therophyte |
| Verbena officinalis | 0 | 0 | 0 | 0 | 0 | 1 | Perennial | Hemicryptophyte/Therophyte |
| Veronica arvensis | 1 | 0 | 1 | 1 | 1 | 0 | Annual | Therophyte |
| Viola arvensis | 1 | 0 | 0 | 1 | 1 | 0 | Annual | Therophyte |
| Viola tricolor | 0 | 0 | 1 | 0 | 0 | 0 | Annual | Therophyte/Hemicryptophyte |
| Vulpia ciliata | 0 | 0 | 1 | 1 | 1 | 0 | Annual | Therophyte |
Appendix B. Analysis Protocols for Soil Parameters
References
- Grimm, N.B.; Faeth, S.H.; Golubiewski, N.E.; Redman, C.L.; Wu, J.; Bai, X.; Briggs, J.M. Global Change and the Ecology of Cities. Science 2008, 319, 756–760. [Google Scholar] [CrossRef]
- Wu, J. Urban Ecology and Sustainability: The State-of-the-Science and Future Directions. Landsc. Urban Plan. 2014, 125, 209–221. [Google Scholar] [CrossRef]
- Grimm, N.B.; Foster, D.; Groffman, P.; Grove, J.M.; Hopkinson, C.S.; Nadelhoffer, K.J.; Pataki, D.E.; Peters, D.P. The Changing Landscape: Ecosystem Responses to Urbanization and Pollution across Climatic and Societal Gradients. Front. Ecol. Environ. 2008, 6, 264–272. [Google Scholar] [CrossRef]
- Raudsepp-Hearne, C.; Peterson, G.D.; Bennett, E.M. Ecosystem Service Bundles for Analyzing Tradeoffs in Diverse Landscapes. Proc. Natl. Acad. Sci. USA 2010, 107, 5242–5247. [Google Scholar] [CrossRef] [PubMed]
- Millennium Ecosystem Assessment (Program) (Ed.) Ecosystems and Human Well-Being: Synthesis; Island Press: Washington, DC, USA, 2005; ISBN 978-1-59726-040-4. [Google Scholar]
- Frazer, L. Paving Paradise: The Peril of Impervious Surfaces. Environ. Health Perspect. 2005, 113, A456–A462. [Google Scholar] [CrossRef] [PubMed]
- Oberndorfer, E.; Lundholm, J.; Bass, B.; Coffman, R.R.; Doshi, H.; Dunnett, N.; Gaffin, S.; Köhler, M.; Liu, K.K.Y.; Rowe, B. Green Roofs as Urban Ecosystems: Ecological Structures, Functions, and Services. BioScience 2007, 57, 823–833. [Google Scholar] [CrossRef]
- Getter, K.L.; Rowe, D.B. The Role of Extensive Green Roofs in Sustainable Development. HortScience 2006, 41, 1276–1285. [Google Scholar] [CrossRef]
- Schrader, S.; Böning, M. Soil Formation on Green Roofs and Its Contribution to Urban Biodiversity with Emphasis on Collembolans. Pedobiologia 2006, 50, 347–356. [Google Scholar] [CrossRef]
- Hobbs, R.J.; Higgs, E.S.; Hall, C. Novel Ecosystems: Intervening in the New Ecological World Order; John Wiley & Sons: Hoboken, NJ, USA, 2013; ISBN 978-1-118-35420-9. [Google Scholar]
- De-Ville, S.; Menon, M.; Stovin, V. Temporal Variations in the Potential Hydrological Performance of Extensive Green Roof Systems. J. Hydrol. 2018, 558, 564–578. [Google Scholar] [CrossRef]
- Shao, H.; Kim, G. A Comprehensive Review of Different Types of Green Infrastructure to Mitigate Urban Heat Islands: Progress, Functions, and Benefits. Land 2022, 11, 1792. [Google Scholar] [CrossRef]
- Semeraro, T.; Scarano, A.; Buccolieri, R.; Santino, A.; Aarrevaara, E. Planning of Urban Green Spaces: An Ecological Perspective on Human Benefits. Land 2021, 10, 105. [Google Scholar] [CrossRef]
- Kim, J.; Kang, W. Assessing Green Roof Contributions to Tree Canopy Ecosystem Services and Connectivity in a Highly Urbanized Area. Land 2022, 11, 1281. [Google Scholar] [CrossRef]
- Dvorak, B.; Volder, A. Green Roof Vegetation for North American Ecoregions: A Literature Review. Landsc. Urban Plan. 2010, 96, 197–213. [Google Scholar] [CrossRef]
- Köhler, M.; Keeley, M. The Green Roof Tradition in Germany: The Example of Berlin. In Green Roofs: Ecological Design and Construction; Schiffer: New York, NY, USA, 2005; pp. 108–112. [Google Scholar]
- Fioretti, R.; Palla, A.; Lanza, L.G.; Principi, P. Green Roof Energy and Water Related Performance in the Mediterranean Climate. Build. Environ. 2010, 45, 1890–1904. [Google Scholar] [CrossRef]
- Kotsiris, G.; Nektarios, P.A.; Ntoulas, N.; Kargas, G. An Adaptive Approach to Intensive Green Roofs in the Mediterranean Climatic Region. Urban For. Urban Green. 2013, 12, 380–392. [Google Scholar] [CrossRef]
- Zakeri, S.M.H.; Mahdiyar, A. The Hindrances to Green Roof Adoption in a Semi-Arid Climate Condition. Sustainability 2020, 12, 9542. [Google Scholar] [CrossRef]
- Benvenuti, S.; Bacci, D. Initial Agronomic Performances of Mediterranean Xerophytes in Simulated Dry Green Roofs. Urban Ecosyst 2010, 13, 349–363. [Google Scholar] [CrossRef]
- Williams, N.S.G.; Rayner, J.P.; Raynor, K.J. Green Roofs for a Wide Brown Land: Opportunities and Barriers for Rooftop Greening in Australia. Urban For. Urban Green. 2010, 9, 245–251. [Google Scholar] [CrossRef]
- Agra, H.; Klein, T.; Vasl, A.; Shalom, H.; Kadas, G.; Blaustein, L. Sedum-Dominated Green-Roofs in a Semi-Arid Region Increase CO2 Concentrations during the Dry Season. Sci. Total Environ. 2017, 584–585, 1147–1151. [Google Scholar] [CrossRef]
- Vasl, A.; Shalom, H.; Kadas, G.J.; Blaustein, L. Sedum—Annual Plant Interactions on Green Roofs: Facilitation, Competition and Exclusion. Ecol. Eng. 2017, 108, 318–329. [Google Scholar] [CrossRef]
- Nektarios, P.A.; Kokkinou, I.; Ntoulas, N. The Effects of Substrate Depth and Irrigation Regime, on Seeded Sedum Species Grown on Urban Extensive Green Roof Systems under Semi-Arid Μediterranean Climatic Conditions. J. Environ. Manag. 2021, 279, 111607. [Google Scholar] [CrossRef] [PubMed]
- Van Mechelen, C.; Dutoit, T.; Hermy, M. Vegetation Development on Different Extensive Green Roof Types in a Mediterranean and Temperate Maritime Climate. Ecol. Eng. 2015, 82, 571–582. [Google Scholar] [CrossRef]
- Getter, K.L.; Rowe, D.B. Substrate Depth Influences Sedum Plant Community on a Green Roof. HortScience 2009, 44, 401–407. [Google Scholar] [CrossRef]
- Brown, C.; Lundholm, J. Microclimate and Substrate Depth Influence Green Roof Plant Community Dynamics. Landsc. Urban Plan. 2015, 143, 134–142. [Google Scholar] [CrossRef]
- Getter, K.; Rowe, D.; Cregg, B. Solar Radiation Intensity Influences Extensive Green Roof Plant Communities. Urban For. Urban Green. 2009, 8, 269–281. [Google Scholar] [CrossRef]
- Van Mechelen, C.; Dutoit, T.; Hermy, M. Adapting Green Roof Irrigation Practices for a Sustainable Future: A Review. Sustain. Cities Soc. 2015, 19, 74–90. [Google Scholar] [CrossRef]
- Dunnett, N.; Kingsbury, N. Planting Green Roofs and Living Walls; Timber Press: Portland, OR, USA, 2008; ISBN 0-88192-911-5. [Google Scholar]
- Van Mechelen, C.; Dutoit, T.; Hermy, M. Mediterranean Open Habitat Vegetation Offers Great Potential for Extensive Green Roof Design. Landsc. Urban Plan. 2014, 121, 81–91. [Google Scholar] [CrossRef]
- Van Mechelen, C.; Dutoit, T.; Kattge, J.; Hermy, M. Plant Trait Analysis Delivers an Extensive List of Potential Green Roof Species for Mediterranean France. Ecol. Eng. 2014, 67, 48–59. [Google Scholar] [CrossRef]
- Lundholm, J.T. Green Roofs and Facades: A Habitat Template Approach. Urban Habitats 2006, 4, 87–101. [Google Scholar]
- Thompson, K.; Grime, J.P. Seasonal Variation in the Seed Banks of Herbaceous Species in Ten Contrasting Habitats. J. Ecol. 1979, 67, 893–921. [Google Scholar] [CrossRef]
- Heerdt, G.N.J.T.; Verweij, G.L.; Bekker, R.M.; Bakker, J.P. An Improved Method for Seed-Bank Analysis: Seedling Emergence After Removing the Soil by Sieving. Funct. Ecol. 1996, 10, 144–151. [Google Scholar] [CrossRef]
- Mamarot, J.; Rodriguez, A. Mauvaises Herbes Des Cultures, 4th ed.; ACTA Association de Coordination Technique Agricole: Paris, France, 2014; ISBN 978-2-85794-284-9. [Google Scholar]
- Pielou, E.C. An Introduction to Mathematical Ecology; Wiley-Interscience: Hoboken, NJ, USA, 1969. [Google Scholar]
- Macfadyen, A. Improved Funnel-Type Extractors for Soil Arthropods. J. Anim. Ecol. 1961, 30, 171–184. [Google Scholar] [CrossRef]
- Gisin, H. Okologie Und Levensgemenischaften Der Collembolen Im Schweizerischen Exkursionsgebiet Basels. Rev. Suisse De Zool. 1943, 50, 131–224. [Google Scholar]
- Borcard, D.; Gillet, F.; Legendre, P. Unconstrained Ordination. In Numerical Ecology with R; Use R; Springer: New York, NY, USA, 2011; pp. 115–151. ISBN 978-1-4419-7975-9. [Google Scholar]
- Legendre, P.; Anderson, M.J. Distance-Based Redundancy Analysis: Testing Multispecies Responses in Multifactorial Ecological Experiments. Ecol. Monogr. 1999, 69, 1–24. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Reyes, R.; Bustamante, W.; Gironás, J.; Pastén, P.A.; Rojas, V.; Suárez, F.; Vera, S.; Victorero, F.; Bonilla, C.A. Effect of Substrate Depth and Roof Layers on Green Roof Temperature and Water Requirements in a Semi-Arid Climate. Ecol. Eng. 2016, 97, 624–632. [Google Scholar] [CrossRef]
- Chenot, J.; Gaget, E.; Moinardeau, C.; Jaunatre, R.; Buisson, E.; Dutoit, T. Substrate Composition and Depth Affect Soil Moisture Behavior and Plant-Soil Relationship on Mediterranean Extensive Green Roofs. Water 2017, 9, 817. [Google Scholar] [CrossRef]
- Dunnett, N.; Nagase, A.; Hallam, A. The Dynamics of Planted and Colonising Species on a Green Roof over Six Growing Seasons 2001–2006: Influence of Substrate Depth. Urban Ecosyst. 2008, 11, 373–384. [Google Scholar] [CrossRef]
- Sutton, R.; Lambrinos, J. Green Roof Ecosystems: Summary and Synthesis; Springer: Berlin/Heidelberg, Germany, 2015; pp. 423–440. ISBN 978-3-319-14982-0. [Google Scholar]
- Rivière, L.; Delruelle, A.; Reniers, J.; Boisson, S.; Mahy, G. Disentangling Dynamics of Green Roof Vegetation Analogue to Dry Grassland over 3 Years: Plant and Substrate Response to Microenvironmental Variations. J. Living Archit. 2022, 9, 1–7. [Google Scholar]
- Carlisle, S.; Piana, M. Green Roof Plant Assemblage and Dynamics. In Green Roof Ecosystems; Sutton, R.K., Ed.; Ecological Studies; Springer International Publishing: Cham, Switzerland, 2015; pp. 285–310. ISBN 978-3-319-14983-7. [Google Scholar]
- Rivière, L.; Sellier, A.; Dutoit, T.; Vidaller, C.; Buisson, E.; Mahy, G. The Contribution of Seedbank to the Green Roof Plant Community Dynamics Analogous to Semi-Natural Grasslands. Front. Ecol. Evol. 2023, 11, 1152319. [Google Scholar] [CrossRef]
- Bazzaz, F.A.; Chiariello, N.R.; Coley, P.D.; Pitelka, L.F. Allocating Resources to Reproduction and Defense: New Assessments of the Costs and Benefits of Allocation Patterns in Plants Are Relating Ecological Roles to Resource Use. BioScience 1987, 37, 58–67. [Google Scholar] [CrossRef]
- Tilman, D.; Wedin, D. Plant Traits and Resource Reduction For Five Grasses Growing on a Nitrogen Gradient. Ecology 1991, 72, 685–700. [Google Scholar] [CrossRef]
- Rowe, D.B.; Getter, K.L.; Durhman, A.K. Effect of Green Roof Media Depth on Crassulacean Plant Succession over Seven Years. Landsc. Urban Plan. 2012, 104, 310–319. [Google Scholar] [CrossRef]
- Heim, A.; Lundholm, J. The Effects of Substrate Depth Heterogeneity on Plant Species Coexistence on an Extensive Green Roof. Ecol. Eng. 2014, 68, 184–188. [Google Scholar] [CrossRef]
- Köhler, M.; Poll, P.H. Long-Term Performance of Selected Old Berlin Greenroofs in Comparison to Younger Extensive Greenroofs in Berlin. Ecol. Eng. 2010, 36, 722–729. [Google Scholar] [CrossRef]
- Thuring, C.E.; Dunnett, N.P. Persistence, Loss and Gain: Characterising Mature Green Roof Vegetation by Functional Composition. Landsc. Urban Plan. 2019, 185, 228–236. [Google Scholar] [CrossRef]
- Thuring, C.E.; Berghage, R.D.; Beattie, D.J. Green Roof Plant Responses to Different Substrate Types and Depths under Various Drought Conditions. HortTechnology 2010, 20, 395–401. [Google Scholar] [CrossRef]
- Koehler, H.H. Predatory Mites (Gamasina, Mesostigmata). Agric. Ecosyst. Environ. 1999, 74, 395–410. [Google Scholar] [CrossRef]
- Koehler, H.H. Mesostigmata (Gamasina, Uropodina), Efficient Predators in Agroecosystems. Agric. Ecosyst. Environ. 1997, 62, 105–117. [Google Scholar] [CrossRef]
- Manu, M.; Băncilă, R.I.; Bîrsan, C.C.; Mountford, O.; Onete, M. Soil Mite Communities (Acari: Mesostigmata) as Indicators of Urban Ecosystems in Bucharest, Romania. Sci. Rep. 2021, 11, 3794. [Google Scholar] [CrossRef]
- Seniczak, A.; Seniczak, S.; Iturrondobeitia, J.C.; Gwiazdowicz, D.J.; Waldon-Rudzionek, B.; Flatberg, K.I.; Bolger, T. Mites (Oribatida and Mesostigmata) and Vegetation as Complementary Bioindicators in Peatlands. Sci. Total Environ. 2022, 851, 158335. [Google Scholar] [CrossRef]
- Sławska, M.; Bruckner, A.; Sławski, M. Edaphic Collembola Assemblages of European Temperate Primeval Forests Gradually Change along a Forest-Type Gradient. Eur. J. Soil Biol. 2017, 80, 92–101. [Google Scholar] [CrossRef]
- Chauvat, M.; Wolters, V.; Dauber, J. Response of Collembolan Communities to Land-Use Change and Grassland Succession. Ecography 2007, 30, 183–192. [Google Scholar] [CrossRef]
- MooRE, J.C.; Snider, R.J.; Robertson, L.S. Effects of Different Management Practices on Collembola and Acarina in Corn Production Systems. I: The Effects of No-Tillage and Atrazine. Pedobiologia 1984, 26, 143–152. [Google Scholar] [CrossRef]
- Kobayashi, T.; Hori, Y.; Nomoto, N. Effects of Trampling and Vegetation Removal on Species Diversity and Micro-Environment under Different Shade Conditions. J. Veg. Sci. 1997, 8, 873–880. [Google Scholar] [CrossRef]
- Williams, K.; Caldwell, M.M.; Richards, J.H. The Influence of Shade and Clouds on Soil Water Potential: The Buffered Behavior of Hydraulic Lift. Plant Soil 1993, 157, 83–95. [Google Scholar] [CrossRef]
- Rixen, C.; Mulder, C.P.H. Improved Water Retention Links High Species Richness with Increased Productivity in Arctic Tundra Moss Communities. Oecologia 2005, 146, 287–299. [Google Scholar] [CrossRef]
- Cruz de Carvalho, R.; Varela, Z.; do Paço, T.A.; Branquinho, C. Selecting Potential Moss Species for Green Roofs in the Mediterranean Basin. Urban Sci. 2019, 3, 57. [Google Scholar] [CrossRef]
- Chahartaghi, M.; Langel, R.; Scheu, S.; Ruess, L. Feeding Guilds in Collembola Based on Nitrogen Stable Isotope Ratios. Soil Biol. Biochem. 2005, 37, 1718–1725. [Google Scholar] [CrossRef]
- Emilsson, T. Vegetation Development on Extensive Vegetated Green Roofs: Influence of Substrate Composition, Establishment Method and Species Mix. Ecol. Eng. 2008, 33, 265–277. [Google Scholar] [CrossRef]
- Schrieke, D.; Lönnqvist, J.; Blecken, G.-T.; Williams, N.S.G.; Farrell, C. Socio-Ecological Dimensions of Spontaneous Plants on Green Roofs. Front. Sustain. Cities 2021, 3, 777128. [Google Scholar] [CrossRef]
- Butler, C.; Orians, C.M. Sedum Cools Soil and Can Improve Neighboring Plant Performance during Water Deficit on a Green Roof. Ecol. Eng. 2011, 37, 1796–1803. [Google Scholar] [CrossRef]
- Aguiar, A.C.; Robinson, S.A.; French, K. Friends with Benefits: The Effects of Vegetative Shading on Plant Survival in a Green Roof Environment. PLoS ONE 2019, 14, e0225078. [Google Scholar] [CrossRef] [PubMed]


| (a) Soil granulometry | ||||||||||||
| df | Clays | Fine silt | Coarse silt | Fine sand | Coarse sand | |||||||
| Substrate | 2 | 1.22 NS | 2.78 NS | 2.72 NS | 7.37 * | 4.13 * | ||||||
| Exposure | 1 | 10.56 * | 0.01 NS | 0.04 NS | 0.01 NS | 0.01 NS | ||||||
| S × E | 2 | 7.95 * | 0.77 NS | 1.88 NS | 0.89 NS | 2.14 NS | ||||||
| (b) Soil chemistry | ||||||||||||
| df | CEC | pH | P2O5 | K2O | MgO | CaO | Na2O | Total N | Organic C | OM | C:N | |
| Substrate | 2 | 1.89 NS | 0.44 NS | 1.22 NS | 1.16 NS | 6.80 * | 5.19 * | 3.26 | 2.87 NS | 4.12 * | 4.02 | 0.73 NS |
| Exposure | 1 | 14.73 * | 0.64 NS | 28.87 ** | 37.49 ** | 106.60 *** | 42.27 ** | 180.20 *** | 78.74 *** | 139.50 *** | 139.50 *** | 0.01 NS |
| S × E | 2 | 1.32 NS | 0.80 NS | 0.82 NS | 0.76 NS | 0.63 NS | 1.92 NS | 1.22 NS | 0.29 NS | 0.45 NS | 0.45 NS | 0.09 NS |
| (a) Spring seed bank | ||||
| df | Viable seed density | Species richness | Evenness | |
| Substrate | 2 | 11.33 *** | 1.28 NS | 11.13 *** |
| Exposure | 1 | 30.25 ** | 6.49 * | 63.64 ** |
| S × E | 2 | 8.36 *** | 0.14 NS | 12.13 *** |
| (b) Winter seed bank | ||||
| df | Viable seed density | Species richness | Evenness | |
| Substrate | 2 | 1.86 NS | 2.18 NS | 4.41 * |
| Exposure | 1 | 0.90 NS | 0.03 NS | 0.02 NS |
| S × E | 2 | 0.79 NS | 2.82 | 1.17 NS |
| (a) Cover and height | ||||||
| df | Total plant cover | S. album cover | S. acre cover | Bryophyte cover | Mean plant height | |
| Substrate | 2 | 0.18 NS | 3.80 | 4.68 * | 0.30 NS | 2.33 NS |
| Exposure | 1 | 15.76 * | 2.38 NS | 20.54 * | 20.37 * | 4.78 NS |
| S × E | 2 | 0.16 NS | 3.64 | 0.61 NS | 0.98 NS | 0.11 NS |
| (b) Planted vegetation | ||||||
| df | Species richness | Simpson index | Evenness | Abundance | ||
| Substrate | 2 | 4.73 * | 3.22 | 1.52 NS | 11.12 ** | |
| Exposure | 1 | 1.25 NS | 6.26 | 2.15 NS | 65.51 ** | |
| S × E | 2 | 1.95 NS | 6.51 * | 18.70 *** | 12.35 ** | |
| (c) Spontaneous vegetation | ||||||
| df | Species richness | Simpson index | Evenness | Abundance | ||
| Substrate | 2 | 0.43 NS | 0.52 NS | 0.66 NS | 3.29 | |
| Exposure | 1 | 15.00 * | 0.42 NS | 3.15 NS | 87.05 ** | |
| S × E | 2 | 0.95 NS | 1.14 NS | 0.38 NS | 0.92 NS | |
| df | Mean Collembola Number | Mean Mite Number | |
|---|---|---|---|
| Substrate | 2 | 1.73 NS | 2.05 NS |
| Exposure | 1 | 51.69 * | 4.79 NS |
| S × E | 2 | 0.22 NS | 4.90 * |
| (a) Mean Collembola number | ||||
| df | Epedaphic | Hemiedaphic | Euedaphic | |
| Substrate | 2 | 0.07 NS | 2.88 | 0.87 NS |
| Exposure | 1 | 20.51 NS | 23.77 NS | 1.42 NS |
| Substrate × Exposure | 2 | 0.62 NS | 0.37 NS | 0.85 NS |
| (b) Mean Mite number | ||||
| df | Oribatida | Actinedida | Gamasida | |
| Substrate | 2 | 2.69 | 0.67 NS | 3.59 * |
| Exposure | 1 | 1.30 NS | 10.20 NS | 11.60 NS |
| Substrate × Exposure | 2 | 1.03 NS | 0.83 NS | 2.20 NS |
| Variable | F-Value | Explained Variation (%) |
| Mean plant height | 10.9 | 16.40 * |
| Fine sand | 7.31 | 11.00 * |
| Total collembola density | 7.13 | 10.70 * |
| CEC | 5.50 | 8.28 * |
| C:N | 5.49 | 8.26 * |
| Total plant cover | 4.44 | 6.67 * |
| Viable seed density (winter seed bank) | 3.47 | 5.22 NS |
| pH | 2.91 | 4.38 NS |
| Organic Matter | 2.82 | 4.24 NS |
| Clay | 2.81 | 4.23 NS |
| Fine silt | 2.79 | 4.19 NS |
| Total mite density | 2.67 | 4.02 NS |
| Viable seed density (spring seed bank) | 2.40 | 3.61 NS |
| Bryophyte cover | 2.36 | 3.55 NS |
| K2O | 1.50 | 2.25 NS |
| Coarse silt | 0.91 | 1.37 NS |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vidaller, C.; Jouet, A.; Van Mechelen, C.; De Almeida, T.; Cortet, J.; Rivière, L.; Mahy, G.; Hermy, M.; Dutoit, T. Coexistence and Succession of Spontaneous and Planted Vegetation on Extensive Mediterranean Green Roofs: Impacts on Soil, Seed Banks, and Mesofauna. Land 2023, 12, 1726. https://doi.org/10.3390/land12091726
Vidaller C, Jouet A, Van Mechelen C, De Almeida T, Cortet J, Rivière L, Mahy G, Hermy M, Dutoit T. Coexistence and Succession of Spontaneous and Planted Vegetation on Extensive Mediterranean Green Roofs: Impacts on Soil, Seed Banks, and Mesofauna. Land. 2023; 12(9):1726. https://doi.org/10.3390/land12091726
Chicago/Turabian StyleVidaller, Christel, Anaïs Jouet, Carmen Van Mechelen, Tania De Almeida, Jérôme Cortet, Lucie Rivière, Grégory Mahy, Martin Hermy, and Thierry Dutoit. 2023. "Coexistence and Succession of Spontaneous and Planted Vegetation on Extensive Mediterranean Green Roofs: Impacts on Soil, Seed Banks, and Mesofauna" Land 12, no. 9: 1726. https://doi.org/10.3390/land12091726
APA StyleVidaller, C., Jouet, A., Van Mechelen, C., De Almeida, T., Cortet, J., Rivière, L., Mahy, G., Hermy, M., & Dutoit, T. (2023). Coexistence and Succession of Spontaneous and Planted Vegetation on Extensive Mediterranean Green Roofs: Impacts on Soil, Seed Banks, and Mesofauna. Land, 12(9), 1726. https://doi.org/10.3390/land12091726







