Carbon Sequestration by Native Tree Species around the Industrial Areas of Southern Punjab, Pakistan
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Location and Sampling Methodology
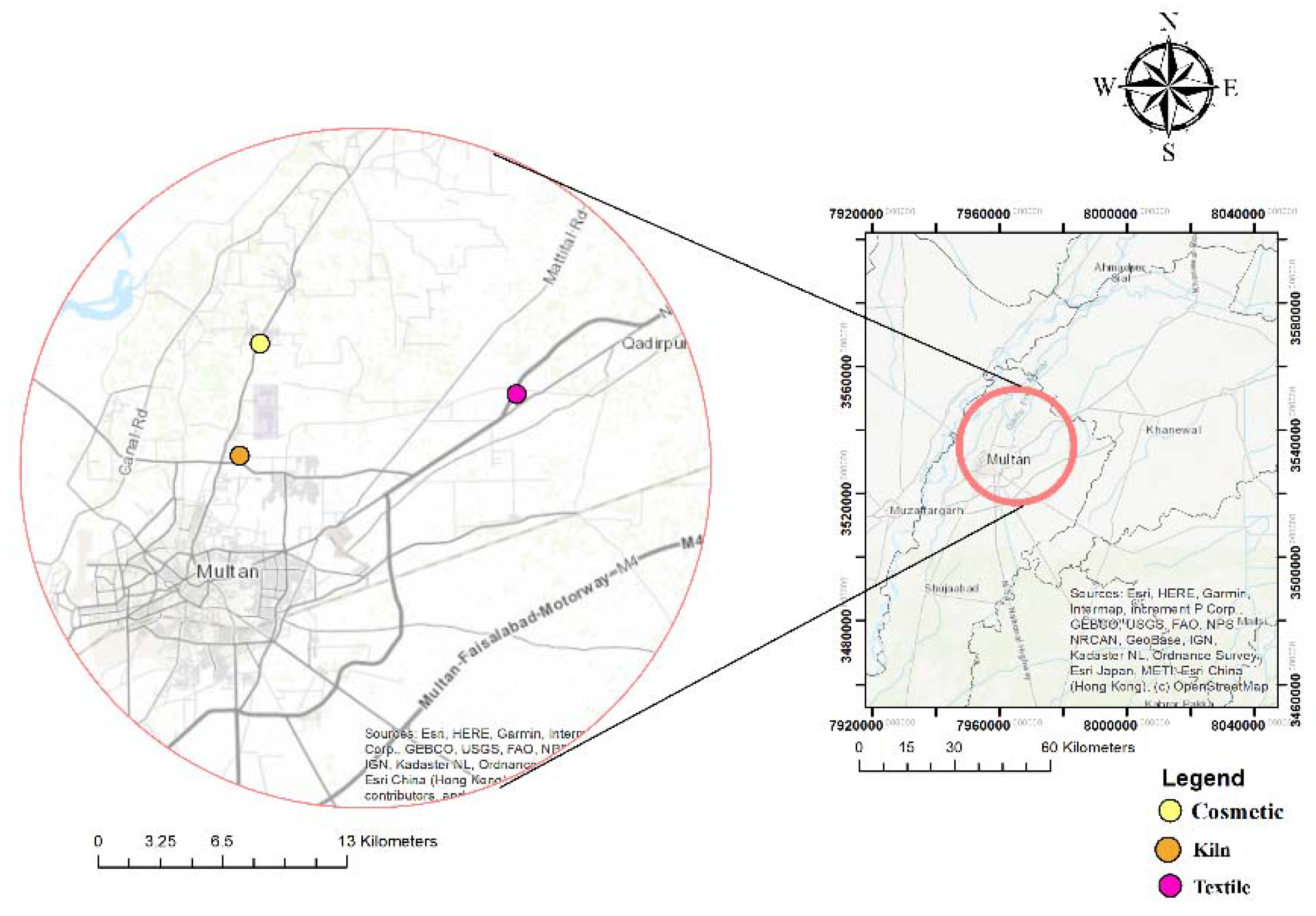
2.1.1. Background Information on Selected Industries
2.1.2. Sampling Methodology
2.2. Carbon Estimation of Above- and Belowground Biomass
2.3. Soil Sampling and Analysis
2.4. Statistical Analysis
3. Results
3.1. Growth Parameters
3.2. Tree Biomass, Carbon Stock, and CO2 Sequestration Rate
3.3. Soil Carbon stock
4. Discussion
5. Policy Implications and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- El Zein, A.L.; Chehayeb, N.A. The effect of greenhouse gases on earth’s temperature. Int. J. Environ. Monit. Anal. 2015, 3, 74. [Google Scholar] [CrossRef]
- Białecki, R.; Stanek, W. Cumulative Green-House Gasses (GHG) Emissions as Total Measure of Global Warming Potential. In Thermodynamics for Sustainable Management of Natural Resources; Springer: Berlin, Germany, 2017; pp. 473–488. [Google Scholar]
- Zhou, T.; Roorda, M.J.; MacLean, H.L.; Luk, J. Life cycle GHG emissions and lifetime costs of medium-duty diesel and battery electric trucks in Toronto, Canada. Transp. Res. Part D Transp. Environ. 2017, 55, 91–98. [Google Scholar] [CrossRef]
- Sinha, R.K.; Chaturvedi, N.D. A review on carbon emission reduction in industries and planning emission limits. Renew. Sustain. Energy Rev. 2019, 114, 109304. [Google Scholar] [CrossRef]
- Mohajerani, A.; Bakaric, J.; Jeffrey-Bailey, T. The urban heat island effect, its causes, and mitigation, with reference to the thermal properties of asphalt concrete. J. Environ. Manag. 2017, 197, 522–538. [Google Scholar] [CrossRef] [PubMed]
- Vidale, S.; Campana, C. Ambient air pollution and cardiovascular diseases: From bench to bedside. Eur. J. Prev. Cardiol. 2018, 25, 818–825. [Google Scholar] [CrossRef] [PubMed]
- Raza, A.; Dahlquist, M.; Jonsson, M.; Hollenberg, J.; Svensson, L.; Lind, T.; Ljungman, P.L. Ozone and cardiac arrest: The role of previous hospitalizations. Environ. Pollut. 2019, 245, 1–8. [Google Scholar] [CrossRef]
- Tabinda, A.B.; Anjum, Z.; Yasar, A.; Rasheed, R.; Mahmood, A.; Iqbal, A. Determination and dispersion of pollutants from different fuel types used in brick kilns by using Gaussian’s plume model. Energy Sources Part A Recovery Util. Environ. Eff. 2019, 41, 1022–1028. [Google Scholar] [CrossRef]
- Shakir, S.K.; Azizullah, A.; Murad, W.; Daud, M.K.; Nabeela, F.; Rahman, H.; ur Rehman, S.; Häder, D.-P. Toxic metal pollution in Pakistan and its possible risks to public health. Rev. Environ. Contam. Toxicol. 2016, 242, 1–60. [Google Scholar]
- Alam, K.; Mukhtar, A.; Shahid, I.; Blaschke, T.; Majid, H.; Rahman, S.; Khan, R.; Rahman, N. Source apportionment and characterization of particulate matter (PM10) in urban environment of Lahore. Aerosol Air Qual. Res. 2014, 14, 1851–1861. [Google Scholar] [CrossRef]
- Shams, T.; Khwaja, M.A. Assessment of Pakistan National Ambient Air Quality Standards (NAAQS’s) with Selected Asian Countries and WHO; Sustainable Development Policy Institute: Islamabad, Pakistan, 2019. [Google Scholar]
- WHO. Estimated Deaths & DALYs Attributable to Selected Environmental Risk Factors, by WHO Member State, 2002; Department of Public Health & Environnment: Denver, CO, USA, 2007.
- Kondo, R.; Kinoshita, Y.; Yamada, T. Green procurement decisions with carbon leakage by global suppliers and order quantities under different carbon tax. Sustainability 2019, 11, 3710. [Google Scholar] [CrossRef]
- Lau, W.W. Beyond carbon: Conceptualizing payments for ecosystem services in blue forests on carbon and other marine and coastal ecosystem services. Ocean. Coast. Manag. 2013, 83, 5–14. [Google Scholar] [CrossRef]
- Cohen-Shacham, E.; Walters, G.; Janzen, C.; Maginnis, S. Nature-Based Solutions to Address Global Societal Challenges; IUCN: Gland, Switzerland, 2016; Volume 97, pp. 2016–2036. [Google Scholar]
- Abson, D.J.; Von Wehrden, H.; Baumgärtner, S.; Fischer, J.; Hanspach, J.; Härdtle, W.; Heinrichs, H.; Klein, A.; Lang, D.; Martens, P. Ecosystem services as a boundary object for sustainability. Ecol. Econ. 2014, 103, 29–37. [Google Scholar] [CrossRef]
- Bridgewater, P. Whose nature? What solutions? Linking Ecohydrology to Nature-based solutions. Ecohydrol. Hydrobiol. 2018, 18, 311–316. [Google Scholar] [CrossRef]
- Kabisch, N.; Frantzeskaki, N.; Pauleit, S.; Naumann, S.; Davis, M.; Artmann, M.; Haase, D.; Knapp, S.; Korn, H.; Stadler, J. Nature-based solutions to climate change mitigation and adaptation in urban areas: Perspectives on indicators, knowledge gaps, barriers, and opportunities for action. Ecol. Soc. 2016, 21, 15. [Google Scholar] [CrossRef]
- Hewitt, C.N.; Ashworth, K.; MacKenzie, A.R. Using green infrastructure to improve urban air quality (GI4AQ). Ambio 2020, 49, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Carstens, D.; Amer, R. Spatio-temporal analysis of urban changes and surface water quality. J. Hydrol. 2019, 569, 720–734. [Google Scholar] [CrossRef]
- Sæbø, A.; Janhäll, S.; Gawronski, S.W.; Hanslin, H.M. Urban Forestry and Pollution Mitigation; Routledge: London, UK, 2017. [Google Scholar]
- Bealey, W.; McDonald, A.; Nemitz, E.; Donovan, R.; Dragosits, U.; Duffy, T.; Fowler, D. Estimating the reduction of urban PM10 concentrations by trees within an environmental information system for planners. J. Environ. Manag. 2007, 85, 44–58. [Google Scholar] [CrossRef] [PubMed]
- Escobedo, F.J.; Nowak, D.J. Spatial heterogeneity and air pollution removal by an urban forest. Landsc. Urban Plan. 2009, 90, 102–110. [Google Scholar] [CrossRef]
- Nowak, D.J. Institutionalizing urban forestry as a “biotechnology” to improve environmental quality. Urban For. Urban Green. 2006, 5, 93–100. [Google Scholar] [CrossRef]
- Gopalakrishnan, V.; Ziv, G.; Hirabayashi, S.; Bakshi, B.R. Nature-based solutions can compete with technology for mitigating air emissions across the United States. Environ. Sci. Technol. 2019, 53, 13228–13237. [Google Scholar] [CrossRef]
- Kroeger, T.; Escobedo, F.J.; Hernandez, J.L.; Varela, S.; Delphin, S.; Fisher, J.R.; Waldron, J. Reforestation as a novel abatement and compliance measure for ground-level ozone. Proc. Natl. Acad. Sci. USA 2014, 111, E4204–E4213. [Google Scholar] [CrossRef] [PubMed]
- Alonso, R.; Vivanco, M.G.; González-Fernández, I.; Bermejo, V.; Palomino, I.; Garrido, J.L.; Elvira, S.; Salvador, P.; Artíñano, B. Modelling the influence of peri-urban trees in the air quality of Madrid region (Spain). Environ. Pollut. 2011, 159, 2138–2147. [Google Scholar] [CrossRef]
- McHale, M.R.; McPherson, E.G.; Burke, I.C. The potential of urban tree plantings to be cost effective in carbon credit markets. Urban For. Urban Green. 2007, 6, 49–60. [Google Scholar] [CrossRef]
- Akbar, K.; Ashraf, I.; Shakoor, S. Analysis of urban forest structure, distribution and amenity value: A case study. J. Anim. Plant Sci. 2014, 24, 1636–1642. [Google Scholar]
- Cairns, M.A.; Brown, S.; Helmer, E.H.; Baumgardner, G.A. Root biomass allocation in the world’s upland forests. Oecologia 1997, 111, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.C.; Martin, A.R. Carbon content of tree tissues: A synthesis. Forests 2012, 3, 332–352. [Google Scholar] [CrossRef]
- Afzal, M.; Aqeela, M.A. Factors affecting carbon sequestration in trees. J. Agric. Res. 2013, 51, 6168. [Google Scholar]
- Ravindranath, N.H.; Ostwald, M. Carbon Inventory Methods: Handbook for Greenhouse Gas Inventory, Carbon Mitigation and Roundwood Production Projects; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007; Volume 29. [Google Scholar]
- Brown, S.; Gillespie, A.J.; Lugo, A.E. Biomass estimation methods for tropical forests with applications to forest inventory data. For. Sci. 1989, 35, 881–902. [Google Scholar]
- Rai, S. Above ground biomass in tropical rain forests of Western Ghats, India. Indian For. 1984, 110, 754–764. [Google Scholar]
- Hawkins, T. Biomass and Volume Tables for Eucalyptus camaldulensis, Dalbergia sissoo, Acacia auriculiformis and Cassia siamea in the Central Bhabar-Terai of Nepal; University of Oxford: Oxford, UK, 1987. [Google Scholar]
- de M. Sá, J.C.; Cerri, C.C.; Dick, W.A.; Lal, R.; Filho, S.P.V.; Piccolo, M.C.; Feigl, B.E. Organic matter dynamics and carbon sequestration rates for a tillage chronosequence in a Brazilian Oxisol. Soil Sci. Soc. Am. J. 2001, 65, 1486–1499. [Google Scholar]
- JHA, K.K. Root Carbon Sequestration and Its Efficacy in Forestry and Agroforestry Systems: A Case of Populus euramericana I-214 Cultivated in Mediterranean condition. Not. Sci. Biol. 2018, 10, 68–78. [Google Scholar] [CrossRef]
- Yasin, G.; Nawaz, M.; Siddiqui, M.; Niazi, N. Biomass, carbon stocks and CO2 sequestration in three different aged irrigated populus deltoides bartr. Ex marsh. Bund planting agroforestry systems. Appl. Ecol. Environ. Res. 2018, 16, 6239–6252. [Google Scholar] [CrossRef]
- Yasin, G.; Nawaz, M.F.; Martin, T.A.; Niazi, N.K.; Gul, S.; Yousaf, M.T.B. Evaluation of Agroforestry Carbon Storage Status and Potential in Irrigated Plains of Pakistan. Forests 2019, 10, 640. [Google Scholar] [CrossRef]
- Yasin, G.; Nawaz, M.F.; Yousaf, M.T.B.; Gul, S.; Qadir, I.; Niazi, N.K.; Sabir, M.A. Carbon stock and CO2 sequestration rate in linearly planted Vachellia nilotica farm trees. Pak. J. Agric. Sci. 2020, 57, 807–814. [Google Scholar]
- Kanime, N.; Kaushal, R.; Tewari, S.; Raverkar, K.; Chaturvedi, S.; Chaturvedi, O. Biomass production and carbon sequestration in different tree-based systems of Central Himalayan Tarai region. For. Trees Livelihoods 2013, 22, 38–50. [Google Scholar] [CrossRef]
- Faiz, M.; Singh, R.; Jattan, S.; Singh, K. Root studies in a Eucalyptus hybrid plantation at various ages. Indian For. 2000, 126, 1165–1174. [Google Scholar]
- Nawaz, M.F.; Mazhar, K.; Gul, S.; Ahmad, I.; Yasin, G.; Asif, M.; Tanvir, M. Comparing the early stage carbon sequestration rates and effects on soil physico-chemical properties after two years of planting agroforestry trees. J. Basic Appl. Sci. 2017, 13, 527–533. [Google Scholar] [CrossRef]
- Kaur, B.; Gupta, S.; Singh, G. Carbon storage and nitrogen cycling in silvopastoral systems on a sodic in northwestern India. Agrofor. Syst. 2002, 54, 21–29. [Google Scholar] [CrossRef]
- Takimoto, A.; Nair, P.R.; Nair, V.D. Carbon stock and sequestration potential of traditional and improved agroforestry systems in the West African Sahel. Agric. Ecosyst. Environ. 2008, 125, 159–166. [Google Scholar] [CrossRef]
- Arora, P.; Chaudhry, S. Vegetation and soil carbon pools of mixed plantation of Acacia nilotica and Dalbergia sissoo under social forestry scheme in Kurukshetra, India. J. Mater. Environ. Sci. 2017, 8, 4565–4572. [Google Scholar] [CrossRef][Green Version]
- Zabek, L.; Prescott, C. Biomass equations and carbon content of above-ground leafless biomass of hybrid poplar in Coastal British Columbia. For. Ecol. Manag. 2006, 223, 291–302. [Google Scholar] [CrossRef]
- Kaul, M.; Mohren, G.; Dadhwal, V. Carbon storage and sequestration potential of selected tree species in India. Mitig. Adapt. Strateg. Glob. Change 2010, 15, 489–510. [Google Scholar] [CrossRef]
- Lal, M.; Singh, R. Carbon sequestration potential of Indian forests. Environ. Monit. Assess. 2000, 60, 315–327. [Google Scholar] [CrossRef]
- Arora, G.; Chaturvedi, S.; Kaushal, R.; Nain, A.; Tewari, S.; Alam, N.M.; Chaturvedi, O.P. Growth, biomass, carbon stocks, and sequestration in an age series of Populus deltoides plantations in Tarai region of central Himalaya. Turk. J. Agric. For. 2014, 38, 550–560. [Google Scholar] [CrossRef]
- Nawaz, M.; Yousaf, M.; Yasin, G.; Gul, S.; Ahmed, I.; Abdullah, M.; Rafay, M.; Tanvir, M.; Asif, M.; Afzal, S. Agroforestry status and its role to sequester atmospheric CO2 under semi-arid climatic conditions in Pakistan. Appl. Ecol. Environ. Res. 2018, 16, 645–661. [Google Scholar] [CrossRef]
| Sr. No | Weaving Industry | Brick Kiln Industry | Cosmetic Industry | |||
|---|---|---|---|---|---|---|
| Inside | Outside | Inside | Outside | Inside | Outside | |
| 1 | V. nilotica | V. nilotica | D. sissoo | D. sissoo | E. camaldulensis | E. camaldulensis |
| 2 | M. indica | M. indica | V. nilotica | V. nilotica | D. sissoo | D. sissoo |
| 3 | D. sissoo | D. sissoo | M. oleifera | M. oleifera | M. oleifera | M. oleifera |
| 4 | M. oleifera | M. oleifera | M. indica | M. indica | M. indica | M. indica |
| 5 | S. cumini | S. cumini | S. cumini | S. cumini | V. nilotica | V. nilotica |
| Species | Equation | Citation |
|---|---|---|
| Vachellia nilotica | 10−1.0646 ∗ D2H0.9098 | Ravindranath and Ostwald [33] |
| Mangifera indica | 2.87 ∗ D0.87 | |
| Dalbergia sissoo | e−3.1141 ∗ D2H0.9719 | Brown, et al. [34] |
| Moringa oleifera | e−3.1141 ∗ D2H0.9719 | Brown, et al. [34] |
| Syzygium cumini | 10−1.2066 ∗ D2H0.9872 | Rai [35] |
| Eucalyptus camaldulensis | e−2.2660 ∗ D2.4663 | Hawkins [36] |
| Industries | Species | Inside | Outside | ||||
|---|---|---|---|---|---|---|---|
| Height (m) | Diameter (cm) | Estimated Age Class | Height (m) | Diameter (cm) | Estimated Age Class | ||
| Weaving | Dalbergia sissoo | 8.99 ± 1.46 | 20.02 ± 5.42 | 8–13 | 9.53 ± 1.92 | 23.05 ± 8.50 | 9–14 |
| Mangifera indica | 12.27 ± 1.12 | 38.01 ± 17.70 | 8–13 | 9.14 ± 2.16 | 40.24 ± 9.94 | 10–15 | |
| Moringa oleifera | 12.19 ± 1.42 | 40.85 ± 14.66 | 5–10 | 5.03 ± 0.81 | 11.93 ± 0.77 | 1–5 | |
| Syzygium cumini | 10.97 ± 1.49 | 25.74 ± 15.42 | 8–13 | 9.75 ± 3.51 | 24.87 ± 10.00 | 7–12 | |
| Vachellia nilotica | 9.98 ± 1.10 | 32.76 ± 3.65 | 7–11 | 11.96 ± 2.98 | 25.48 ± 8.37 | 5–10 | |
| Brick kiln | Dalbergia sissoo | 11.51 ± 1.87 | 21.64 ± 4.87 | 11–16 | 9.98 ± 1.45 | 12.34 ± 4.91 | 3–7 |
| Mangifera indica | 11.28 ± 3.64 | 40.85 ± 9.92 | 5–10 | 10.82 ± 1.13 | 33.57 ± 12.78 | 3–7 | |
| Moringa oleifera | 9.22 ± 1.80 | 20.22 ± 6.60 | 1–5 | 11.74 ± 1.50 | 38.42 ± 23.49 | 7–13 | |
| Syzygium cumini | 12.80 ± 2.69 | 27.50 ± 17.76 | 8–13 | 9.60 ± 3.78 | 18.61 ± 12.61 | 5–10 | |
| Vachellia nilotica | 14.48 ± 1.60 | 27.91 ± 12.98 | 4–9 | 14.71 ± 4.51 | 23.46 ± 7.29 | 3–7 | |
| Cosmetic | Dalbergia sissoo | 8.76 ± 1.22 | 22.44 ± 9.84 | 10–15 | 7.47 ± 2.73 | 21.64 ± 12.13 | 7–12 |
| Eucalyptus camaldulensis | 19.05 ± 1.58 | 33.57 ± 9.98 | 5–10 | 8.38 ± 1.35 | 18.40 ± 2.59 | 1–5 | |
| Mangifera indica | 11.89 ± 3.85 | 43.89 ± 18.02 | 6–12 | 12.19 ± 3.59 | 50.96 ± 18.05 | 8–14 | |
| Moringa oleifera | 12.34 ± 3.08 | 46.92 ± 21.80 | 7–13 | 9.37 ± 1.71 | 37.61 ± 19.69 | 5–10 | |
| Vachellia nilotica | 8.15 ± 2.27 | 19.82 ± 9.67 | 3–7 | 12.57 ± 4.22 | 39.64 ± 14.67 | 10–15 | |
| Industries | Species | Inside | Outside | ||||
|---|---|---|---|---|---|---|---|
| AGB Mg ha−1 | BGB Mg ha−1 | TB Mg ha−1 | AGB Mg ha−1 | BGB Mg ha−1 | TB Mg ha−1 | ||
| Weaving | Dalbergia sissoo | 0.38 b ± 0.16 | 0.09 b ± 0.04 | 0.48 b ± 0.21 | 1.60 a ± 1.21 | 0.42 a ± 0.31 | 2.02 a ± 1.52 |
| Mangifera indica | 0.17 b ± 0.07 | 0.04 a ± 0.01 | 0.20 b ± 0.09 | 0.18 b ± 0.04 | 0.05 b ± 0.01 | 0.22 b ± 0.05 | |
| Moringa oleifera | 2.24 a ± 1.42 | 0.58 a ± 0.37 | 2.58 b ± 1.79 | 0.07 b ± 0.02 | 0.02 b ± 0.00 | 0.09 b ± 0.02 | |
| Syzygium cumini | 1.62 a ± 0.63 | 0.42 b ± 0.16 | 2.04 a ± 0.79 | 1.61 ab ± 0.86 | 0.30 ab ± 0.22 | 1.46 ab ± 1.08 | |
| Vachellia nilotica | 1.89 a ± 0.56 | 0.49 a ± 0.15 | 2.38 a ± 0.70 | 1.08 ab ± 0.83 | 0.28 ab ± 0.22 | 1.36 ab ± 1.05 | |
| Brick Kiln | Dalbergia sissoo | 0.57 ab ± 0.24 | 0.15 ab ± 0.06 | 0.72 ab ± 0.31 | 0.17 b ± 0.12 | 0.04 b ± 0.03 | 0.21 b ± 0.15 |
| Mangifera indica | 0.18 b ± 0.04 | 0.05 b ± 0.01 | 0.22 b ± 0.05 | 0.14 b ± 0.05 | 0.03 b ± 0.01 | 0.19 b ± 0.06 | |
| Moringa oleifera | 0.44 ab ± 0.33 | 0.11 ab ± 0.86 | 0.56 ab ± 0.42 | 2.09 a ± 2.16 | 0.54 a ± 0.56 | 2.64 a ± 2.73 | |
| Syzygium cumini | 2.17 a ± 2.44 | 0.57 a ± 0.63 | 2.74 a ± 3.07 | 0.84 ab ± 0.90 | 0.22 ab ± 0.23 | 1.06 ab ± 1.13 | |
| Vachellia nilotica | 2.09 a ± 1.34 | 0.54 a ± 0.35 | 2.64 a ± 1.70 | 1.35 ab ± 0.74 | 0.35 ab ± 0.19 | 1.70 ab ± 0.92 | |
| Cosmetic | Dalbergia sissoo | 0.55 b ± 0.43 | 0.14 b ± 0.11 | 0.69 b ± 0.54 | 0.56 b ± 0.79 | 0.14 b ± 0.20 | 0.71 b ± 0.99 |
| Eucalyptus camueldenisis | 12.95 a ± 5.36 | 3.36 a ± 1.39 | 16.32 a ± 6.76 | 1.04 b± 0.41 | 0.27 b ± 0.10 | 1.31 b ± 0.52 | |
| Mangifera indica | 0.18 b ± 0.06 | 0.04 b ± 0.01 | 0.23 b ± 0.08 | 0.21 b ± 0.06 | 0.05 b ± 0.01 | 0.27 b ± 0.08 | |
| Moringa oleifera | 3.63 b ± 3.1 | 0.94 b ± 0.82 | 4.57 b ± 4.02 | 1.64 b ± 1.52 | 0.42 b ± 0.39 | 2.06 b ± 1.92 | |
| Vachellia nilotica | 0.76 b ± 0.79 | 0.19 b ± 0.20 | 0.96 b ± 1.00 | 4.15 a ± 3.06 | 1.07 a ± 0.79 | 5.22 a ± 3.85 | |
| Industries | Species | Inside | Outside | ||||||
|---|---|---|---|---|---|---|---|---|---|
| AGC Mg ha−1 | BGC Mg ha−1 | TC Mg ha−1 | CO2 Sequestration Mg ha−1 | AGC Mg ha−1 | BGC Mg ha−1 | TC Mg ha−1 | CO2 Sequestration Mg ha−1 | ||
| Weaving | Dalbergia sissoo | 0.18 b ± 0.08 | 0.05 b ± 0.02 | 0.23 cd ± 0.10 | 0.83 cd ± 0.36 | 0.77 a ± 0.58 | 0.20 a ± 0.15 | 0.97 a ± 0.72 | 3.54 a ± 2.67 |
| Mangifera indica | 0.08 b ± 0.03 | 0.02 b ± 0.01 | 0.10 d ± 0.04 | 0.36 d ± 0.15 | 0.08 b ± 0.02 | 0.02 b ± 0.00 | 0.10 b ± 0.02 | 0.38 b ± 0.08 | |
| Moringa oleifera | 1.08 a ± 0.69 | 0.28 a ± 0.18 | 1.08 bc ± 0.69 | 3.95 bc ± 2.50 | 0.03 b ± 0.01 | 0.01 b ± 0.00 | 0.04 b ± 0.01 | 0.15 b ± 0.03 | |
| Syzygium cumini | 0.78 a ± 0.30 | 0.20 a ± 0.08 | 2.82 a ± 1.10 | 10.32 a ± 4.03 | 0.56 ab ± 0.41 | 0.14 ab ± 0.11 | 0.70 ab ± 0.52 | 2.57 ab ± 1.89 | |
| Vachellia nilotica | 0.91 a ± 0.27 | 0.24 a ± 0.07 | 1.14 b ± 0.34 | 4.19 b ± 1.24 | 0.52 ab ± 0.40 | 0.13 ab ± 0.10 | 0.65 ab ± 0.51 | 2.38 ab ± 1.85 | |
| Brick Kiln | Dalbergia sissoo | 0.27 ab ± 0.12 | 0.07 ab ± 0.03 | 0.34 ab ± 0.15 | 1.25 b ± 0.54 | 0.08 b ± 0.06 | 0.02 b ± 0.02 | 0.10 b ± 0.07 | 0.37 b ± 0.26 |
| Mangifera indica | 0.09 b ± 0.02 | 0.02 b ± 0.00 | 0.10 b ± 0.02 | 0.39 b ± 0.08 | 0.07 b ± 0.02 | 0.01 b ± 0.01 | 0.09 b ± 0.03 | 0.33 b ± 0.11 | |
| Moringa oleifera | 0.21 ab ± 0.16 | 0.05 ab± 0.04 | 0.26 ab ± 0.20 | 0.98 b ± 0.73 | 1.00 a ± 1.04 | 0.26 a ± 0.27 | 1.26 a ± 1.31 | 4.63 a ± 4.80 | |
| Syzygium cumini | 1.04 a ± 1.16 | 1.32 a ± 1.47 | 3.78 a ± 4.24 | 13.85 a ± 15.53 | 0.40 ab ± 0.43 | 0.10 ab ± 0.11 | 0.50 ab ± 0.54 | 1.85 ab ± 1.99 | |
| Vachellia nilotica | 1.00 a ± 0.65 | 1.27 a ± 0.81 | 0.05 a ± 0.04 | 8.31 ab ± 5.35 | 0.65 ab ± 0.35 | 0.16 ab ± 0.09 | 0.82 ab ± 0.44 | 2.99 ab ± 1.62 | |
| Cosmetic | Dalbergia sissoo | 0.26 b ± 0.20 | 0.06 b ± 0.05 | 0.33 b ± 0.26 | 1.23 b ± 0.95 | 0.27 b ± 0.37 | 0.07 b ± 0.09 | 0.34 a ± 0.47 | 1.25 b ± 1.75 |
| Eucalyptus camaldulensis | 6.22 a ± 2.57 | 1.61 a ± 0.67 | 7.83 a ± 3.24 | 28.70 a ± 11.89 | 0.50 b ± 0.20 | 0.13 b ± 0.05 | 0.63 b ± 0.25 | 2.30 b ± 0.92 | |
| Mangifera indica | 0.09 b ± 0.03 | 0.02 b ± 0.01 | 0.11 b ± 0.04 | 0.41 b ± 0,15 | 0.10 b ± 0.03 | 0.02 b ± 0.01 | 0.13 b ± 0.04 | 0.47 b ± 0.18 | |
| Moringa oleifera | 1.74 b ± 1.53 | 0.45 b ± 0.39 | 2.19 b ± 1.93 | 8.04 b ± 7.07 | 0.78 b ± 0.73 | 0.20 b ± 0.19 | 0.99 b ± 0.92 | 3.63 b ± 3.37 | |
| Vachellia nilotica | 0.36 b ± 0.38 | 0.09 b ± 0.38 | 0.46 b ± 0.48 | 1.70 b ± 1.76 | 1.99 a ± 1.46 | 0.51 a ± 0.38 | 2.51 a ± 1.85 | 9.19 a ± 6.78 | |
| Industries | Species | Inside | Outside | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OC% (0–20) | SOC Mg ha−1 (0–20) | OC% (20–40) | SOC Mg ha−1 (20–40) | TOC (0–40) (%) | TSOC Mg ha−1 (0–40) | OC% (0–20) | SOC Mg ha−1 (0–20) | OC% (20–40) | SOC Mg ha−1 (20–40) | TOC (0–40) (%) | TSOC Mg ha−1 (0–40) | ||
| Weaving | Dalbergia sissoo | 1.90 b ± 0.07 | 40.81 b ± 3.32 | 1.57 b ± 0.02 | 34.73 b ± 2.01 | 2.685 ± 0.045 | 58.175 ± 4.325 | 2.64 b ± 0.04 | 56.72 b ± 2.53 | 1.78 b ± 0.06 | 38.62 b ± 2.15 | 3.53 ± 0.07 | 76.03 ± 3.605 |
| Mangifera indica | 1.49 d ± 0.03 | 29.43 d ± 1.11 | 1.32 d ± 0.02 | 27.17 d ± 0.38 | 2.15 ± 0.04 | 43.015 ± 1.3 | 2.41 d ± 0.03 | 47.45 d ± 1.25 | 1.65 a ± 0.05 | 33.31 c ± 1.16 | 3.23 ± 0.055 | 64.10 ± 1.83 | |
| Moringa oleifera | 1.39 d ± 0.07 | 28.05 d ± 0.63 | 1.2 e ± 0.04 | 24.69 e ± 0.83 | 1.295 ± 0.09 | 26.37 ± 0.73 | 1.94 e ± 0.05 | 38.20 e ± 2.53 | 1.39 c ± 0.03 | 28.54 d ± 0.77 | 1.66 ± 0.065 | 52.47 ± 2.91 | |
| Syzygium cumini | 1.75 c ± 0.08 | 35.53 c ± 2.38 | 1.46 c ± 0.07 | 30.46 c ± 0.87 | 2.48 ± 0.115 | 32.9 ± 2.815 | 2.51 c ± 0.05 | 52.19 c ± 1.38 | 1.70 b ± 0.01 | 35.21 c ± 1.19 | 3.36 ± 0.05 | 69.75 ± 1.97 | |
| Vachellia nilotica | 2.31 a ± 0.07 | 48.16 a ± 2.55 | 1.9 a ± 0.01 | 40.53 a ± 1.23 | 3.26 ± 0.075 | 68.42 ± 3.16 | 3.08 a ± 0.11 | 63.16 a ± 2.86 | 2.04 a ± 0.14 | 42.31 a ± 3.75 | 4.1 ± 0.18 | 84.31 ± 4.73 | |
| Brick kiln | Dalbergia sissoo | 2.6 a ± 0.10 | 53.05 a ± 2.86 | 2.01 a ± 0.06 | 43.11 a ± 0.73 | 3.605 ± 0.08 | 74.60 ± 3.22 | 3.11 a ± 0.10 | 66.16 a ± 2.40 | 2.20 a ± 0.14 | 47.51 a ± 2.68 | 4.21 ± 0.17 | 89.915 ± 3.74 |
| Mangifera indica | 1.51 d ± 0.12 | 31.38 d ± 2.26 | 1.32 b ± 0.04 | 28.19 c ± 1.93 | 2.17 ± 0.14 | 45.47 ± 3.22 | 2.01 e ± 0.05 | 41.54 e ± 1.98 | 1.72 c ± 0.05 | 36.12 d ± 1.38 | 2.87 ± 0.075 | 59.66 ± 1.68 | |
| Moringa oleifera | 2.31 b ± 0.07 | 49.69 a ± 1.78 | 1.91 b ± 0.05 | 42.57 a ± 0.78 | 3.26 ± 0.09 | 70.97 ± 2.17 | 2.88 b ± 0.08 | 61.99 b ± 4.29 | 2.00 b ± 0.07 | 44.08 b ± 2.10 | 3.88 ± 0.15 | 84.03 ± 6.39 | |
| Syzygium cumini | 1.93 c ± 0.03 | 40.40 b ± 0.15 | 1.72 c ± 0.02 | 36.82 b ± 1.05 | 2.79 ± 0.04 | 58.81 ± 0.67 | 2.72 c ± 0.05 | 55.76 c ± 2.49 | 1.91 b ± 0.02 | 39.26 c ± 0.94 | 3.675 ± 0.07 | 75.39 ± 2.96 | |
| Vachellia nilotica | 1.83 c ± 0.19 | 36.80 c ± 3.27 | 1.43 d ± 0.02 | 30.03 c ± 1.30 | 2.54 ± 0.21 | 51.8 ± 3.92 | 2.45 d ± 0.295 | 49.51 d ± 0.59 | 1.95 b ± 0.05 | 40.73 c ± 1.53 | 3.42 ± 0.17 | 69.87 ± 1.35 | |
| Cosmetic | Dalbergia sissoo | 1.85 b ± 0.06 | 36.49 b ± 1.15 | 1.72 c ± 0.05 | 35.39 b ± 1.41 | 2.71 ± 0.11 | 54.18 ± 2.56 | 2.02 c ± 0.05 | 39.74 b ± 1.15 | 1.36 c ± 0.05 | 37.69 b ± 1.25 | 2.27 ± 0.075 | 58.58 ± 1.775 |
| Eucalyptus camaldulensis | 2.06 a ± 0.04 | 42.78 a ± 1.23 | 1.8 b ± 0.02 | 38.07 a ± 1.32 | 2.96 ± 0.05 | 62.18 ± 1.89 | 2.19 b ± 0.03 | 45.56 a ± 2.06 | 1.89 a ± 0.06 | 40.01 ab ± 1.58 | 3.13 ± 0.06 | 65.5 ± 2.84 | |
| Mangifera indica | 1.46 c ± 0.06 | 29.42 c ± 2.25 | 1.39 d ± 0.01 | 28.76 c ± 0.77 | 2.155 ± 0.06 | 43.17 ± 1.89 | 1.64 d ± 0.09 | 33.05 c ± 2.67 | 1.37 c ± 0.06 | 30.44 c ± 2.19 | 2.32 ± 0.12 | 48.27 ± 3.76 | |
| Moringa oleifera | 1.36 d ± 0.03 | 29.25 c ± 2.15 | 1.25 e ± 0.04 | 27.28 c ± 1.78 | 1.98 ± 0.05 | 42.89 ± 3.04 | 1.45 e ± 0.04 | 31.05 c ± 2.06 | 1.45 b ± 0.01 | 28.71 c ± 1.81 | 2.17 ± 0.045 | 45.40 ± 2.08 | |
| Vachellia nilotica | 2.11 a ± 0.01 | 43.08 a ± 1.16 | 1.86 a ± 0.04 | 38.95 a ± 1.48 | 3.04 ± 0.03 | 62.55 ± 1.9 | 2.30 a ± 0.02 | 47.07 a ± 1.75 | 1.95 a ± 0.04 | 40.78 a ± 1.42 | 3.27 ± 0.04 | 67.46 ± 2.46 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zubair, M.; Yasin, G.; Qazlbash, S.K.; Ul Haq, A.; Jamil, A.; Yaseen, M.; Rahman, S.U.; Guo, W. Carbon Sequestration by Native Tree Species around the Industrial Areas of Southern Punjab, Pakistan. Land 2022, 11, 1577. https://doi.org/10.3390/land11091577
Zubair M, Yasin G, Qazlbash SK, Ul Haq A, Jamil A, Yaseen M, Rahman SU, Guo W. Carbon Sequestration by Native Tree Species around the Industrial Areas of Southern Punjab, Pakistan. Land. 2022; 11(9):1577. https://doi.org/10.3390/land11091577
Chicago/Turabian StyleZubair, Muhammad, Ghulam Yasin, Sehrish Khan Qazlbash, Ahsan Ul Haq, Akash Jamil, Muhammad Yaseen, Shafeeq Ur Rahman, and Wei Guo. 2022. "Carbon Sequestration by Native Tree Species around the Industrial Areas of Southern Punjab, Pakistan" Land 11, no. 9: 1577. https://doi.org/10.3390/land11091577
APA StyleZubair, M., Yasin, G., Qazlbash, S. K., Ul Haq, A., Jamil, A., Yaseen, M., Rahman, S. U., & Guo, W. (2022). Carbon Sequestration by Native Tree Species around the Industrial Areas of Southern Punjab, Pakistan. Land, 11(9), 1577. https://doi.org/10.3390/land11091577







