Shelterbelts Planted on Cultivated Fields Are Not Solutions for the Recovery of Former Forest-Related Herbaceous Vegetation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Sites
2.2. Sampling
2.3. Diversity and Species Composition Analysis
3. Results
3.1. Tree and Shrub Species Composition
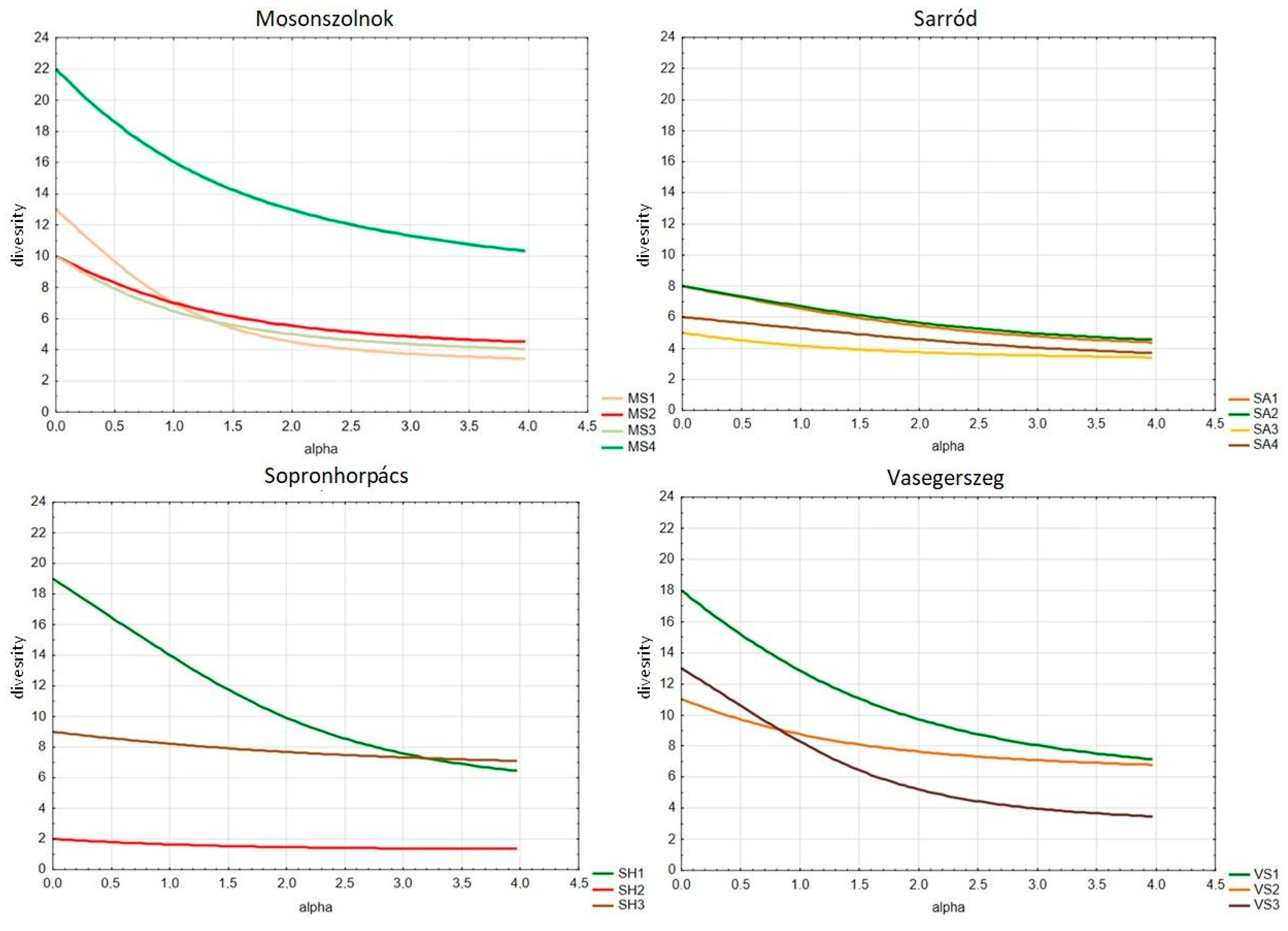
3.2. Herbaceous Diversity
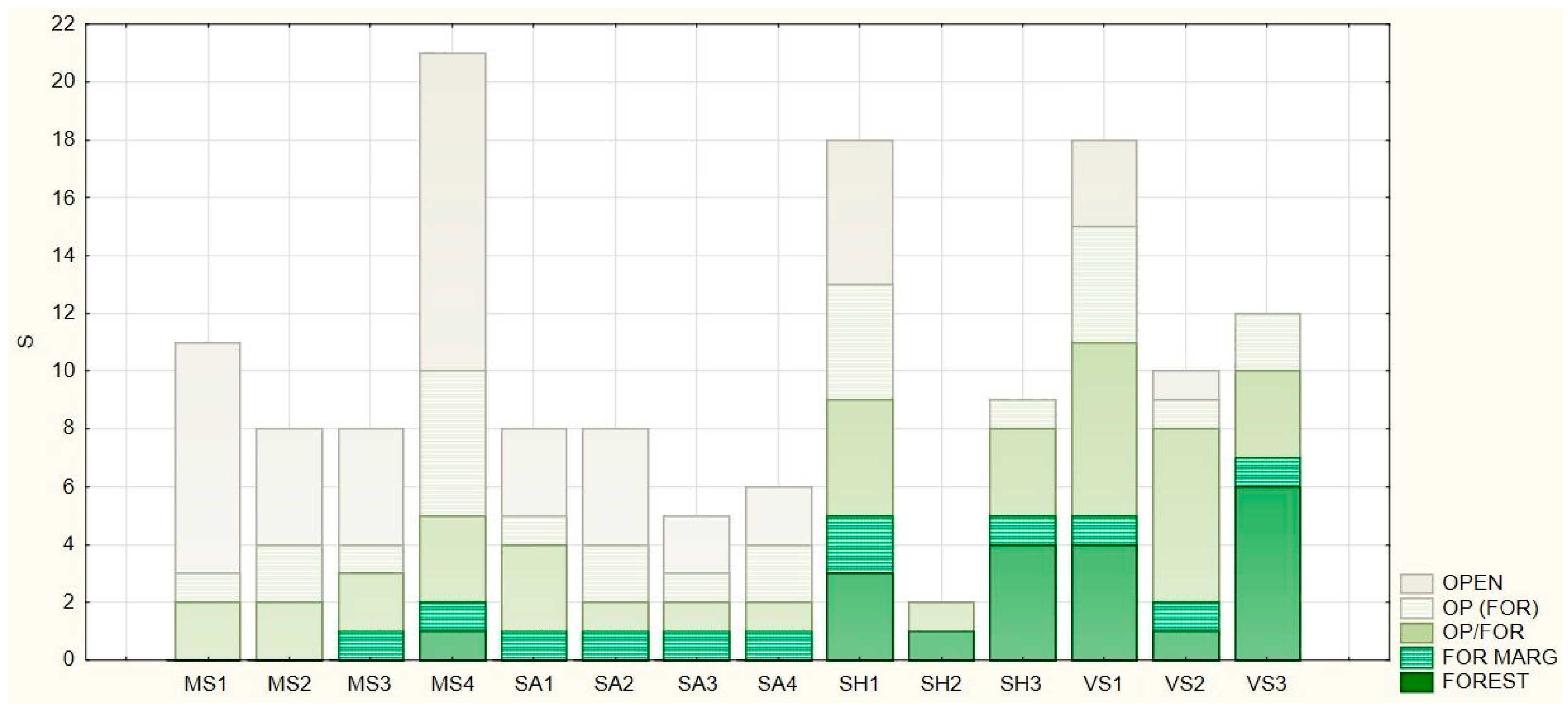
3.3. Naturalness Value and Forest Affinity of the Herbaceous Vegetation
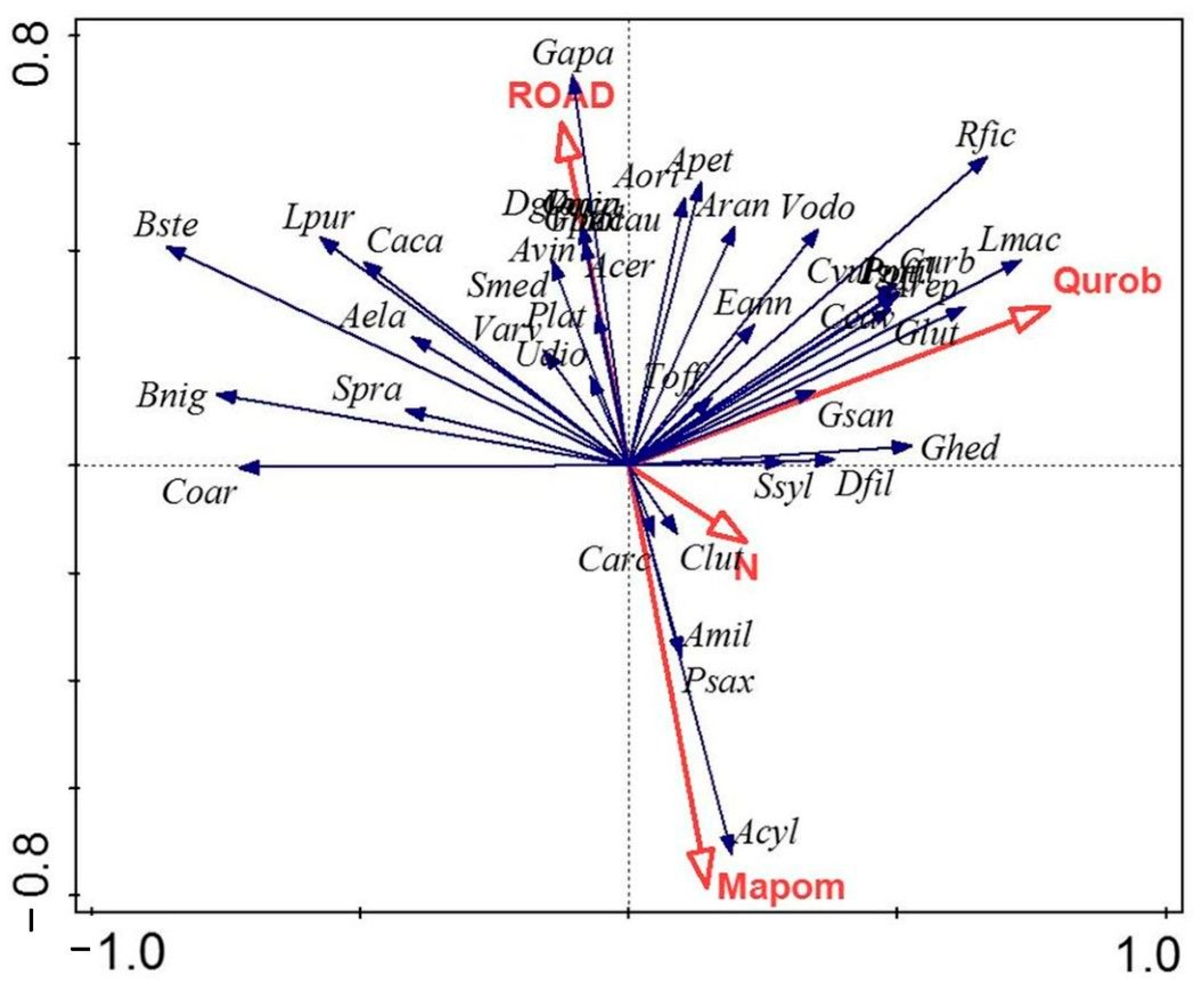
3.4. Redundancy Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Appendix A
| Mosonszolnok | ||||
| SHELT I | SHELT II | SHELT III | SHELT IV | |
| A | 47.85429, 17.14683 | 47.86501, 17.12457 | 47.86219, 17.19912 | 47.82253, 17.2079 |
| B | 47.85525, 17.14708 | 47.86484, 17.1254 | 47.86277, 17.20043 | 47.82196, 17.20875 |
| C | 47.85653, 17.14742 | 47.86467, 17.12718 | 47.86324, 17.20153 | 47.82114, 17.20992 |
| Sarród | ||||
| SHELT I | SHELT II | SHELT III | SHELT IV | |
| A | 47.65314, 16.87423 | 47.65546, 16.87111 | 47.65484, 16.87037 | 47.65232, 16.87603 |
| B | 47.65392, 16.87264 | 47.6558, 16.87156 | 47.65463, 16.87015 | 47.65223, 16.87625 |
| C | 47.65438, 16.87161 | 47.65665, 16.87261 | 47.65414, 16.86954 | 47.65197, 16.87679 |
| Sopronhorpács | ||||
| SHELT I | SHELT II | SHELT III | ||
| A | 47.493, 16.74852 | 47.47178, 16.72377 | 47.46881, 16.71774 | |
| B | 47.49838, 16.74481 | 47.4732, 16.72307 | 47.46984, 16.71738 | |
| C | 47.50114, 16.74311 | 47.4745, 16.72259 | 47.47036, 16.71707 | |
| Vasegerszeg | ||||
| SHELT I | SHELT II | SHELT III | ||
| A | 47.37375, 16.89984 | 47.3728, 16.90045 | 47.37294, 16.89763 | |
| B | 47.3736, 16.89995 | 47.37279, 16.90053 | 47.37259, 16.89771 | |
| C | 47.37346, 16.9 | 47.37262, 16.90068 | 47.37243, 16.89771 | |
| Plot | pH (KCl) | Viscosity | CaCO3 % | SOM % | NH4+ + NO3− N (mg/kg) | AL P (mg P2O5/kg) | AL K (mg K2O/kg) |
|---|---|---|---|---|---|---|---|
| MS1 | 7.11 ± 0.06 | 47.33 ± 1.25 | 5.99 ± 0.90 | 2.10 ± 0.22 | 8.90 ± 0.96 | 132.33 ± 14.34 | 513.00 ± 42.81 |
| MS2 | 6.98 ± 0.08 | 52.00 ± 1.41 | 3.33 ± 0.80 | 2.10 ± 0.16 | 9.70 ± 0.37 | 125.33 ± 7.72 | 615.67 ± 64.79 |
| MS3 | 6.88 ± 0.07 | 57.67 ± 2.87 | 5.99 ± 0.54 | 1.97 ± 0.17 | 10.07 ± 0.29 | 133.67 ± 5.31 | 513.67 ± 46.33 |
| MS4 | 7.19 ± 0.04 | 59.00 ± 4.90 | 7.99 ± 0.77 | 2.17 ± 0.17 | 9.47 ± 0.42 | 618.67 ± 24.96 | 747.67 ± 37.04 |
| SA1 | 7.29 ± 0.03 | 61.00 ± 6.68 | 22.25 ± 4.16 | 5.32 ± 0.15 | 8.06 ± 4.88 | 193.90 ± 11.49 | 532.00 ± 130.43 |
| SA2 | 7.26 ± 0.04 | 64.00 ± 7.87 | 26.22 ± 1.21 | 5.36 ± 0.03 | 2.32 ± 1.09 | 273.25 ± 163.27 | 769.33 ± 131.11 |
| SA3 | 7.22 ± 0.06 | 77.67 ± 12.26 | 16.15 ± 2.82 | 7.27 ± 1.40 | 6.85 ± 2.40 | 229.20 ± 58.24 | 880.33 ± 286.15 |
| SA4 | 7.45 ± 0.08 | 50.00 ± 4.90 | 13.74 ± 0.16 | 4.66 ± 0.40 | 3.52 ± 0.43 | 198.07 ± 15.34 | 543.33 ± 24.31 |
| SH1 | 6.83 ± 0.07 | 68.00 ± 2.16 | 0.68 ± 0.00 | 5.36 ± 0.08 | 3.66 ± 0.92 | 34.41 ± 13.20 | 384.00 ± 6.98 |
| SH2 | 6.48 ± 0.17 | 62.00 ± 1.41 | <0.10 | 4.86 ± 0.15 | 8.34 ± 4.42 | 81.57 ± 61.85 | 502.67 ± 64.22 |
| SH3 | 5.06 ± 0.54 | 52.33 ± 4.19 | <0.10 | 4.67 ± 0.69 | 27.12 ± 31.97 | 688.26 ± 927.21 | 589.67 ± 486.93 |
| VS1 | 6.23 ± 0.37 | 79.33 ± 7.54 | <0.10 | 5.31 ± 0.06 | 8.30 ± 1.05 | 268.19 ± 153.00 | 357.00 ± 99.03 |
| VS2 | 5.62 ± 0.65 | 73.33 ± 8.38 | <0.10 | 4.94 ± 0.58 | 14.43 ± 6.79 | 178.08 ± 106.25 | 399.00 ± 111.72 |
| VS3 | 5.86 ± 0.05 | 70.67 ± 11.12 | <0.10 | 5.32 ± 0.02 | 12.22 ± 1.14 | 208.04 ± 52.15 | 368.67 ± 21.70 |
| Species/Cover (%) | MS1 | MS2 | MS3 | MS4 | SA1 | SA2 | SA3 | SA4 | SH1 | SH2 | SH3 | VS1 | VS2 | VS3 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Achillea millefolium | 0 | 0 | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Aegilops cylindrica | 0 | 0 | 0 | 0 | 0 | 10 | 13 | 7 | 0 | 0 | 0 | 0 | 0 | 0 |
| Ajuga reptans | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 | 17 | 0 |
| Alliaria petiolata | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 0 | 3 | 0 | 0 |
| Allium vineale | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 13 | 0 | 0 | 0 | 0 | 0 |
| Amaranthus retroflexus | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 | 0 | 0 |
| Anagallis arvensis | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Anemone ranunculoides | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 |
| Anthriscus caucalis | 0 | 0 | 0 | 20 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Anthriscus cerefolium | 0 | 0 | 0 | 10 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Arrhenatherum elatius | 0 | 33 | 0 | 7 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Arum orientale | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 12 | 0 | 0 | 0 | 0 | 3 |
| Ballota nigra | 20 | 13 | 30 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Brachypodium sylvaticum | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 |
| Bromus sterilis | 30 | 33 | 40 | 7 | 2 | 0 | 0 | 0 | 7 | 0 | 0 | 0 | 0 | 0 |
| Carduus acanthoides | 3 | 8 | 0 | 13 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Carex hirta | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 0 | 0 | 0 | 0 |
| Chenopodium album | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Circaea lutetiana | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 0 | 0 |
| Cirsium arvense | 0 | 0 | 0 | 3 | 0 | 0 | 8 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Cirsium vulgare | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 3 |
| Convolvulus arvensis | 2 | 2 | 7 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Corydalis cava | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 12 | 0 | 5 |
| Crepis setosa | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 0 |
| Dactylis glomerata | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 0 | 0 | 0 | 0 |
| Daucus carota subsp. carota | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Dryopteris filix-mas | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 3 | 0 |
| Elymus repens | 0 | 7 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Erigeron annuus | 0 | 0 | 0 | 10 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 3 | 0 |
| Euphorbia virgata | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Falcaria vulgaris | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Festuca rupicola | 0 | 0 | 0 | 0 | 0 | 3 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Gagea lutea | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 5 |
| Galium aparine | 5 | 13 | 3 | 33 | 0 | 0 | 0 | 2 | 10 | 12 | 7 | 7 | 8 | 3 |
| Galium mollugo | 0 | 0 | 0 | 7 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Geranium robertianum | 0 | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Geranium sanguineum | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 3 | 0 | 0 |
| Geum urbanum | 0 | 0 | 17 | 12 | 3 | 7 | 10 | 3 | 15 | 0 | 3 | 12 | 17 | 8 |
| Glechoma hederacea | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 13 | 0 |
| Hypericum perforatum | 0 | 0 | 0 | 7 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Lamium maculatum | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 | 3 | 7 |
| Lamium purpureum | 2 | 5 | 7 | 23 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Onopordum acanthium | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 0 | 0 | 0 | 0 |
| Ornithogalum umbellatum | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 | 0 |
| Pimpinella saxifraga | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Poa pratensis | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Poa trivialis | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 0 | 0 | 0 | 0 |
| Polygonatum latifolium | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 50 | 3 | 5 | 0 | 0 | 0 |
| Polygonatum multiflorum | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 13 | 0 | 7 |
| Potentilla reptans | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Pulmonaria officinalis | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 | 0 | 2 |
| Ranunculus ficaria | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 10 | 0 | 0 | 33 | 15 | 33 |
| Salvia pratensis | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Setaria viridis | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 0 |
| Silene latifolia subsp. alba | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Solanum dulcamara | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 0 | 0 | 0 | 0 |
| Stachys sylvatica | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 0 | 3 |
| Stellaria media | 2 | 5 | 10 | 20 | 7 | 0 | 0 | 3 | 0 | 0 | 0 | 7 | 10 | 0 |
| Symphytum officinale | 2 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Taraxacum officinale | 0 | 0 | 0 | 0 | 2 | 3 | 0 | 0 | 3 | 0 | 0 | 3 | 0 | 0 |
| Thlaspi perfoliatum | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Tripleurospermum inodorum | 3 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Urtica dioica | 0 | 3 | 0 | 0 | 10 | 0 | 0 | 0 | 7 | 0 | 3 | 0 | 3 | 0 |
| Verbascum lychnitis | 0 | 0 | 0 | 7 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Verbena officinalis | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 | 0 | 0 | 0 | 0 | 0 |
| Veronica arvensis | 2 | 0 | 5 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 10 | 0 | 0 |
| Veronica persica | 0 | 0 | 0 | 20 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Vinca minor | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 7 | 0 | 0 | 0 | 0 | 0 |
| Viola odorata | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 23 | 0 | 3 | 23 | 0 | 3 |
References
- Tiwari, P.; Kumar, R.; Thakur, L.; Singh, R. Soil Treatment through Agroforestry: A Review. Indian J. Pure Appl. Biosci. 2017, 5, 24–30. [Google Scholar] [CrossRef]
- Feliciano, D.; Ledo, A.; Hillier, J.; Nayak, D.R. Which agroforestry options give the greatest soil and above ground carbon benefits in different world regions? Agric. Ecosyst. Environ. 2018, 254, 117–129. [Google Scholar] [CrossRef]
- Jezeer, R.E.; Santos, M.J.; Verweija, P.A.; Boot, R.G.A.; Cough, Y. Benefits for multiple ecosystem services in Peruvian coffee agroforestry systems without reducing yield. Ecosyst. Serv. 2019, 40, 101033. [Google Scholar] [CrossRef]
- Elagib, N.A.; Al-Saidi, M. Balancing the benefits from the water–energy–land–food nexus through agroforestry in the Sahel. Sci. Total Environ. 2020, 742, 140509. [Google Scholar] [CrossRef]
- Tschora, H.; Cherubini, F. Co-benefits and trade-offs of agroforestry for climate change mitigation and other sustainability goals in West Africa. Glob. Ecol. Conserv. 2020, 22, e00919. [Google Scholar] [CrossRef]
- Winkler, D.; Traser, G. Collembola Diversity in Agricultural Environments (Lajta Project, Western Hungary). In Proceedings of the International Scientific Conference on Sustainable Development and Ecological Footprint, Sopron, Hungary, 26–27 March 2012. [Google Scholar]
- Gál, J. A mezőgazdasági terméshozamok növekedése az erdősávok védelmében. Erdészettudományi Közlemények 1963, 1–2, 41–83. (In Hungarian) [Google Scholar]
- Szarvas, P. Mezővédő Erdősávok, Fasorok Jellemzése, Ökológiai Feltárása, Kihatásai. Characterization, Ecological Exploration and Effects of Field Protection Forest Strips and Tree Lines. Ph.D. Dissertation, University of Debrecen, Debrecen, Hungary, 2010. (In Hungarian). [Google Scholar]
- Updawatta, R.P.; Rankoth, L.; Jose, S. Agroforestry and Biodiversity. Sustainability 2019, 11, 2879. [Google Scholar] [CrossRef] [Green Version]
- Pierce, R.A., II; Farrand, D.T.; Kurtz, W.B. Projecting the bird community response resulting from the adoption of shelterbelt agroforestry practices in Eastern Nebraska. Agrofor. Syst. 2001, 53, 333–350. [Google Scholar] [CrossRef]
- Bonifacio, R.S.; Kinross, C.M.; Gurr, G.M.; Nicol, H. The effect of woody plant diversity and other stand and landscape factors on the diversity and abundance of birds using farm shelterbelts. Pac. Conserv. Biol. 2011, 17, 22–35. [Google Scholar] [CrossRef]
- Leles, B.; Xiao, X.; Pasion, B.O.; Nakamura, A.; Tomlinson, K.W. Does plant diversity increase top-down control of herbivorous insects in tropical forest? Oikos 2017, 126, 1142–1149. [Google Scholar] [CrossRef]
- Pardon, P.; Reheul, D.; Mertens, J.; Reubens, B.; De Frenne, P.; De Smedt, P.; Proesmans, W.; Van Vooren, L.; Verheyen, C. Gradients in abundance and diversity of ground dwelling arthropods as a function of distance to tree rows in temperate arable agroforestry systems. Agric. Ecosyst. Environ. 2019, 270–271, 114–128. [Google Scholar] [CrossRef]
- Todd, J.H.; Poulton, J.; Richards, K.; Malone, L.A. Effect of orchard management, neighbouring land-use and shelterbelt tree composition on the parasitism of pest leafroller (Lepidoptera: Tortricidae) larvae in kiwifruit orchard shelterbelts. Agric. Ecosyst. Environ. 2018, 260, 27–35. [Google Scholar] [CrossRef]
- Gontijo, L.M. Engineering natural enemy shelters to enhance conservation biological control in field crops. Biol. Control 2019, 130, 155–163. [Google Scholar] [CrossRef]
- Mize, C.W.; Brandle, J.R.; Schoeneberger, M.M.; Bentrup, G. Ecological Development and Function of Shelterbelts in Temperate North America. In Toward Agroforestry Design; Jose, S., Gordon, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2008; Volume 4, pp. 27–54. [Google Scholar]
- Hass, A.L.; Liese, B.; Heong, K.L.; Settele, J.; Tscharntke, T.; Westphal, C. Plant-pollinator interactions and bee functional diversity are driven by agroforests in rice-dominated landscapes. Agric. Ecosyst. Environ. 2018, 253, 140–147. [Google Scholar] [CrossRef]
- Donkersley, P. Trees for bees. Agric. Ecosyst. Environ. 2019, 270–271, 79–83. [Google Scholar] [CrossRef]
- Damschen, E.I. Landscape corridors. In Encyclopedia of Biodiversity, 2nd ed.; Levin, S.A., Ed.; Academic Press: Cambridge, MA, USA, 2013; pp. 467–475. [Google Scholar] [CrossRef]
- Damschen, E.I.; Haddad, N.M.; Orrock, J.L.; Tewksbury, J.J.; Levey, D.J. Corridors Increase Plant Species Richness at Large Scales. Science 2006, 313, 1284–1286. [Google Scholar] [CrossRef] [Green Version]
- Carlier, J.; Moran, J. Hedgerow typology and condition analysis to inform greenway design in rural landscapes. J. Environ. Manag. 2019, 247, 790–803. [Google Scholar] [CrossRef]
- Wilson, P.J. Restoring pollinator communities and pollination services in hedgerows in intensively managed agricultural landscapes. In The Ecology of Hedgerows and Field Margins; Dover, J.W., Ed.; Routledge: New York, NY, USA, 2019; pp. 163–185. [Google Scholar]
- Milberg, P.; Bergman, K.; Jonason, D.; Karlsson, J.; Westerberg, L. Land-use history influence the vegetation in coniferous production forests in southern Sweden. For. Ecol. Manag. 2019, 440, 23–30. [Google Scholar] [CrossRef]
- Harta, I.; Simon, B.; Vinogradov, S.; Winkler, D. Collembola communities and soil conditions in forest plantations established in an intensively managed agricultural area. J. For. Res. 2020, 29, 1291–1297. [Google Scholar] [CrossRef]
- Bihari, Z.; Babolcsai, G.; Bartholdy, J.; Ferenczi, Z.; Gerhát-Kerényi, J.; Haszpra, L.; Homoki-Ijváry, K.; Kovács, T.; Lakatos, M.; Németh, Á.; et al. National Atlas of Hungary: Natural Environment; Kocsis, K., Ed.; MTA CSFK Geographical Institute: Budapest, Hungary, 2018; pp. 58–69. [Google Scholar]
- Zepner, L.; Karrasch, P.; Wiemann, F.; Bernard, L. ClimateCharts.net—An interactive climate analysis web platform. Int. J. Digit. Earth 2020, 4, 338–356. [Google Scholar] [CrossRef]
- Simon, T. A Kisalföld természetes növénytakarója. Földrajzi Közlemények 1962, 86, 183–193. (In Hungarian) [Google Scholar]
- Dövényi, Z.; Ambrózy, P.; Juhász, Á.; Marosi, S.; Mezősi, G.; Michalkó, G.; Szalai, Z.; Tiner, T. Magyarország Kistájainak Katasztere (Inventory of Microregions in Hungary); MTA Földrajztudományi Kutatóintézet: Budapest, Hungary, 2010. (In Hungarian) [Google Scholar]
- Takács, V. A Sopronhorpácsi Mezővédő Erdősávrendszer Hasznosítási Lehetősége. Possibility of utilization of the Sopronhorpács shelterbelt system. Erdészeti Lapok 2004, 139, 127–130. (In Hungarian) [Google Scholar]
- Horváth, F.; Dobolyi, K.; Morschauer, T.; Lőkös, L.; Karas, L.; Szerdahelyi, T. Flóra Adatbázis [Flora Database]; MTA ÖBKI: Vácrátót, Hungary, 1995. (In Hungarian) [Google Scholar]
- Tóthmérész, B. Diversity; University of Debrecen: Debrecen, Hungary, 2013; p. 290. [Google Scholar]
- Grime, J.P. Plant Strategies, Vegetation Processes, and Ecosystem Properties; Wiley: New York, NY, USA, 1979. [Google Scholar]
- Borhidi, A. Social behaviour types, the naturalness and relative ecological indicator values of the higher plants in the Hungarian flora. Acta Bot Hung 1995, 39, 97–181. [Google Scholar]
- Legendre, P.; Gallagher, E.D. Ecologically meaningful transformations for ordination of species data. Oecologia 2001, 129, 271–280. [Google Scholar] [CrossRef]
- Rose, F. Indicators of ancient woodland. Br. Wildl. 1999, 10, 241–251. [Google Scholar]
- Dzwonko, Z.; Loster, S. Distribution of vascular plant species in small woodlands on the Western Carpathian foothills. Oikos 1989, 56, 77–86. [Google Scholar] [CrossRef]
- Hermy, A.; Honnay, O.; Firbank, L.; Grashof-Bokdam, C.; Lawesson, J.L. An ecological comparison between ancient and other forest plant species of Europe, and the implications for forest conservation. Biol. Conserv. 1999, 91, 9–22. [Google Scholar] [CrossRef]
- Wulf, M. Plant species as indicators of ancient woodland in northwestern Germany. J. Veg. Sci. 1997, 8, 635–642. [Google Scholar] [CrossRef]
- Reif, A.; Achtziger, R. Gebüsche, Hecken, Waldmäntel, Feldgehölze. In Handbuch Naturschutz und Landschaftspflege; Konold, W., Böcker, R., Hampicke, U., Eds.; Wiley: New York, NY, USA, 2014. [Google Scholar] [CrossRef]
- Sciama, D.; Augusto, L.; Dupouey, J.L.; Gonzalez, M.; Moares Domínguez, C. Floristic and ecological differences between recent and ancient forests growing on non-acidic soils. For. Ecol. Manag. 2009, 258, 600–608. [Google Scholar] [CrossRef]
- Peterken, G.F.; Game, M. Historical factors affecting the nuber and distribution of vascular plant species in the woodlands of Central Lincolnshire. J. Ecol. 1984, 72, 155–182. [Google Scholar] [CrossRef]
- Van Den Berge, S.; Baeten, L.; Vanhellemont, M.; Ampoorter, E.; Proesmans, W.; Earaerts, M.; Hermy, M.; Smagghe, G.; Vermeulen, I.; Verheyen, K. Species diversity, pollinator resource value and edibility potential of woody networks in the countryside in northern Belgium. Agric. Ecosyst. Environ. 2018, 259, 119–126. [Google Scholar] [CrossRef]
- Wehling, S.; Diekmann, M. Factors influencing the spatial distribution of forest plant species in hedgerows of North-western Germany. Biodivers. Conserv. 2008, 17, 2799–2813. [Google Scholar] [CrossRef]
- Aude, E.; Tybirk, K.; Bruus Pedersen, M. Vegetation diversity of conventional and organic hedgerows in Denmark. Agric. Ecosyst. Environ. 2003, 99, 135–147. [Google Scholar] [CrossRef] [Green Version]


| Habitat Type | Social Behavior Type | Abb. | Nat. Value |
|---|---|---|---|
| Natural habitats | Specialists | S | +6 |
| Competitors | C | +5 | |
| Generalists | G | +4 | |
| Natural pioneers | NP | +3 | |
| Disturbed, secondary, and artificial habitats | Disturbance-tolerant | DT | +2 |
| Weeds | W | +1 | |
| Introduced alien species | I | −1 | |
| Adventives | A | −1 | |
| Ruderal competitors | RC | −2 | |
| Aggressive alien species | AC | −3 |
| Forest Affinity Category | Abbreviation |
|---|---|
| Obligate forest species | FOREST |
| Species typical to forest edges and clearings | FOR MARG |
| Species that can be found both in woody habitats and open (non-woody) land | FOR/OP |
| Species appearing mainly in open land but also in forests | OP (FOR) |
| Species specific to open habitats | OPEN |
| Layer/Species | MS1 | MS2 | MS3 | MS4 | SA1 | SA2 | SA3 | SA4 | SH1 | SH2 | SH3 | VS1 | VS2 | VS3 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Canopy layer cover | 70 | 80 | 80 | 90 | 90 | 80 | 90 | 95 | 90 | 95 | 80 | 80 | 90 | 80 |
| Acer campestre | 50 | 70 | 80 | 50 | 30 | 40 | ||||||||
| Acer platanoides | 5 | 5 | 10 | 30 | 30 | 20 | 10 | 80 | 10 | |||||
| Cerasus avium | 10 | |||||||||||||
| Fraxinus excelsior | 5 | 5 | ||||||||||||
| Fraxinus pennsylvanica | 60 | 20 | 10 | 20 | ||||||||||
| Gleditsia triacanthos | 5 | |||||||||||||
| Koelreuteria paniculata | 1 | |||||||||||||
| Larix decidua | 5 | |||||||||||||
| Maclura pomifera | 5 | 10 | 20 | 20 | 20 | |||||||||
| Pinus sylvestris | 30 | |||||||||||||
| Populus × canescens | 20 | 10 | ||||||||||||
| Quercus cerris | 10 | 10 | 10 | |||||||||||
| Quercus robur | 20 | 30 | 50 | 60 | 50 | |||||||||
| Quercus rubra | 10 | |||||||||||||
| Robinia pseudoacacia | 30 | 60 | 10 | 20 | 10 | |||||||||
| Salix alba | 10 | |||||||||||||
| Ulmus minor | 10 | 30 | 50 | 40 | 50 | 50 | 50 | 50 |
| Layer/Species | MS1 | MS2 | MS3 | MS4 | SA1 | SA2 | SA3 | SA4 | SH1 | SH2 | SH3 | VS1 | VS2 | VS3 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Shrub layer cover | 30 | 40 | 20 | 30 | 20 | 30 | 40 | 30 | 50 | 15 | 30 | 20 | ||
| Cornus mas | 20 | |||||||||||||
| Cornus sanguinea | 10 | |||||||||||||
| Crataegus monogyna | 10 | |||||||||||||
| Elaeagnus angustifolia | 10 | |||||||||||||
| Euonymus verrucosus | 20 | |||||||||||||
| Ligustrum vulgare | 20 | 20 | 20 | 20 | 20 | |||||||||
| Maclura pomifera | 5 | 20 | ||||||||||||
| Prunus spinosa | 10 | 10 | 15 | 10 | 20 | 20 | 5 | 20 | 10 | |||||
| Sambucus nigra | 10 | 10 | 5 | 10 | 10 | 5 | 5 | |||||||
| Ulmus minor | 10 | 20 | ||||||||||||
| Viburnum lantana | 20 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szigeti, N.; Berki, I.; Vityi, A.; Rasran, L. Shelterbelts Planted on Cultivated Fields Are Not Solutions for the Recovery of Former Forest-Related Herbaceous Vegetation. Land 2021, 10, 930. https://doi.org/10.3390/land10090930
Szigeti N, Berki I, Vityi A, Rasran L. Shelterbelts Planted on Cultivated Fields Are Not Solutions for the Recovery of Former Forest-Related Herbaceous Vegetation. Land. 2021; 10(9):930. https://doi.org/10.3390/land10090930
Chicago/Turabian StyleSzigeti, Nóra, Imre Berki, Andrea Vityi, and Leonid Rasran. 2021. "Shelterbelts Planted on Cultivated Fields Are Not Solutions for the Recovery of Former Forest-Related Herbaceous Vegetation" Land 10, no. 9: 930. https://doi.org/10.3390/land10090930
APA StyleSzigeti, N., Berki, I., Vityi, A., & Rasran, L. (2021). Shelterbelts Planted on Cultivated Fields Are Not Solutions for the Recovery of Former Forest-Related Herbaceous Vegetation. Land, 10(9), 930. https://doi.org/10.3390/land10090930







