Elementome of Endemic Dolomitic Flora: Pterocephalus spathulatus (Lag.) Coult
Abstract
1. Introduction
1.1. Historic Context
1.2. Properties and Characteristics of Dolomitic Soils
1.3. Dolomiticolous Flora and Vegetation
1.4. Ionomic Studies in Plants
1.5. Species under Study
- To determine the concentrations of the elements, both total and phytoavailable, found in the dolomitic soils where P. spathulatus is able to grow, as a reference system to which its ionomic composition may be related.
- To identify what elements, present in the soil (total) or in dissolution (phytoavailable), accumulate in different parts of the plant, mainly in leaves. In this way, this study sought to establish whether total or phytoavailable elements were the best predictors of the mineral composition of the plant, by calculating the Bioconcentration Factor (BCF).
- To determine the relationship between the elements accumulated in leaves, stems and roots by means of the Translocation Factor (TF), in order to establish whether they are likely to concentrate in stems, and especially in leaves.
- To relate the influence of the mineral composition of the soil to the richness and diversity of the plant communities where the diagnostic species P. spathulatus grows.
2. Materials and Methods
2.1. Study Area and Sampling
2.2. Soil Analysis
2.2.1. Analysis of Total Soil Elements
2.2.2. Analysis of the Phytoavailable Soil Elements
2.3. Analysis of Plant Tissues
2.4. Calculation of BCF and TF Parameters
2.5. P. spathulatus Plant Communities
2.6. Data Analysis
3. Results and Discussion
3.1. Analysis of the Mineral Contents of Soil Samples
3.1.1. Total Elements
3.1.2. Phytoavailable Elements
3.2. Analysis of Foliar Mineral Contents
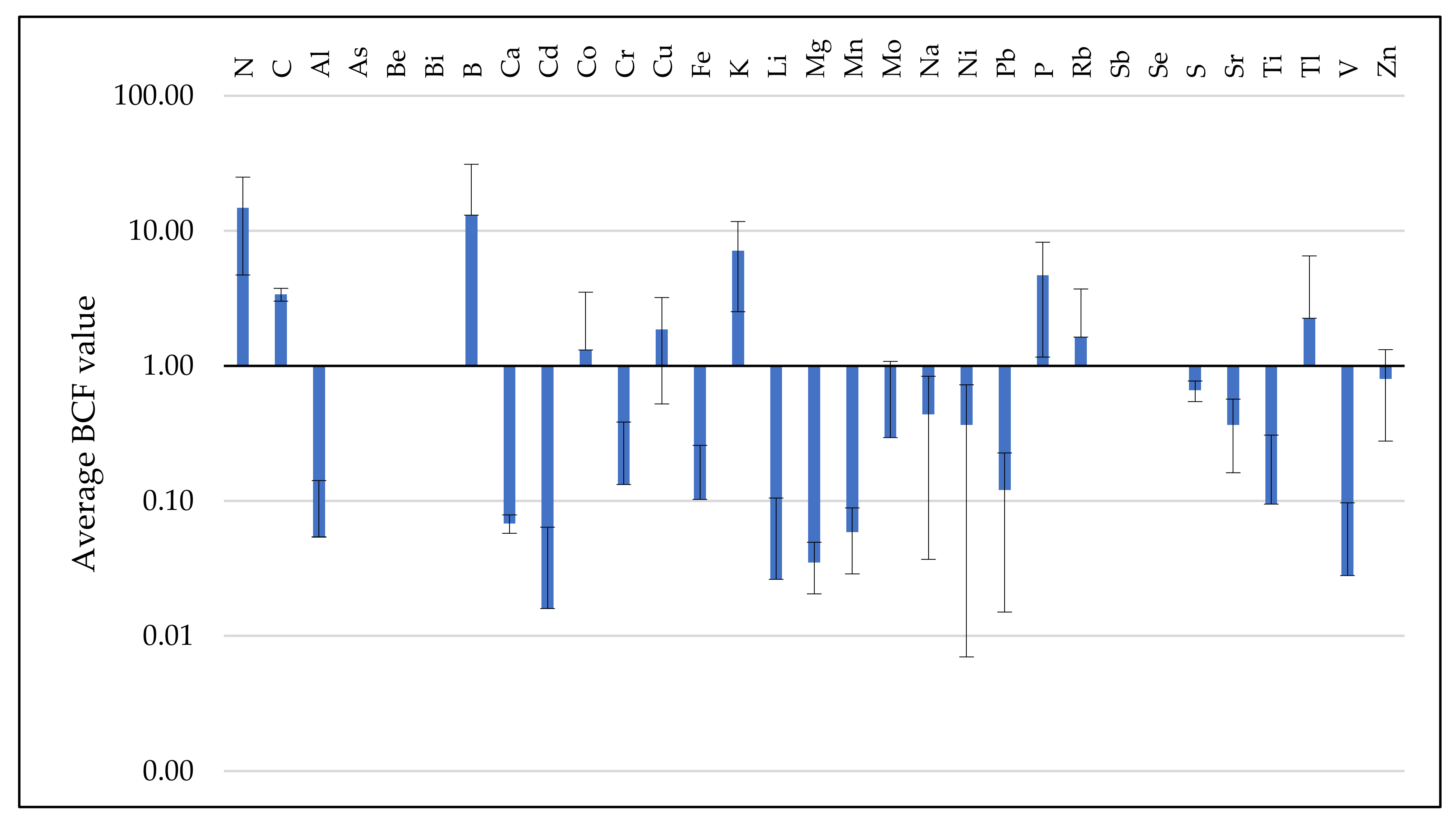
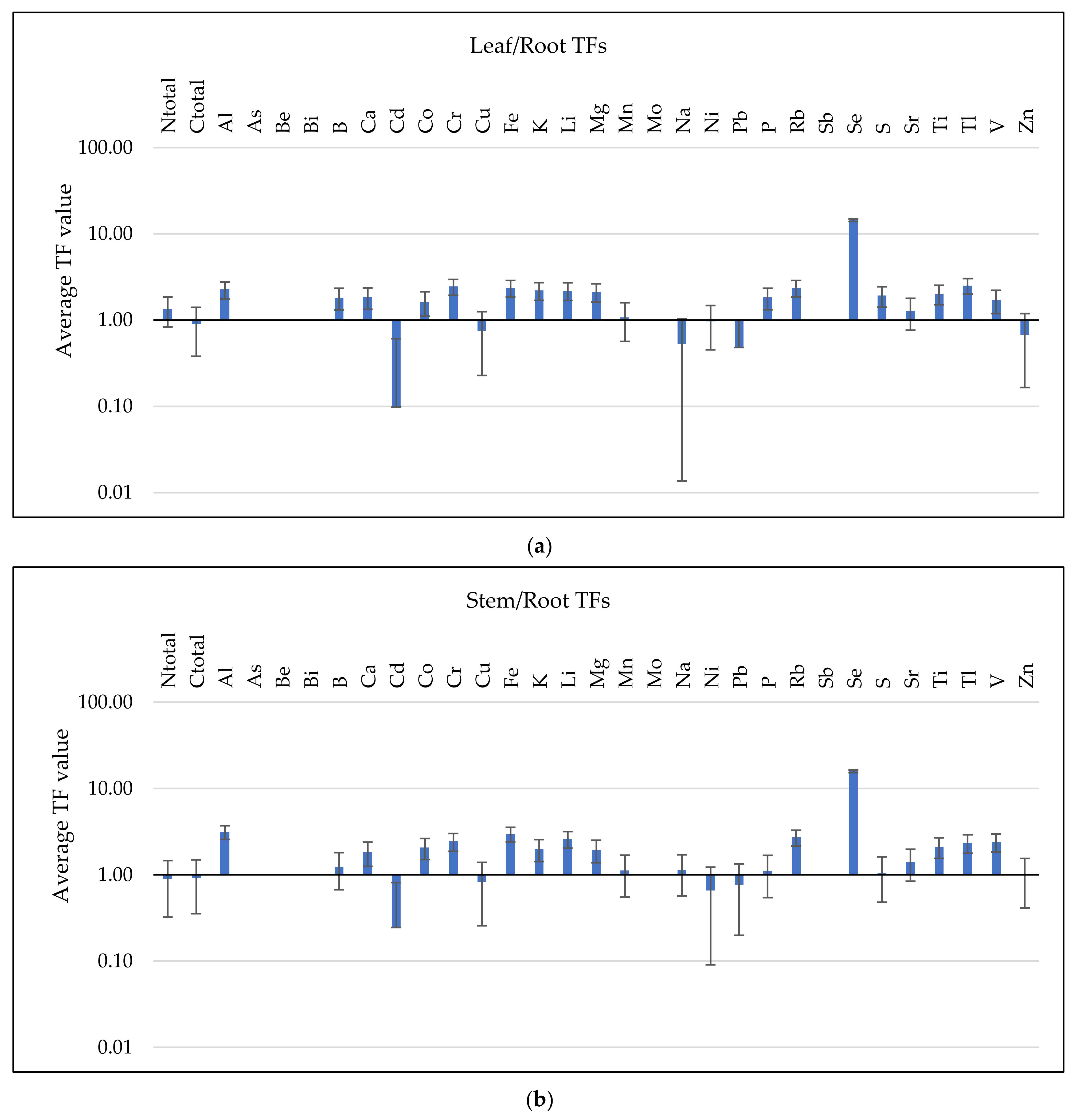
3.3. Analysis of BCF and TF Parameters
3.3.1. BCF Values Assessment
3.3.2. TF Values Assessment
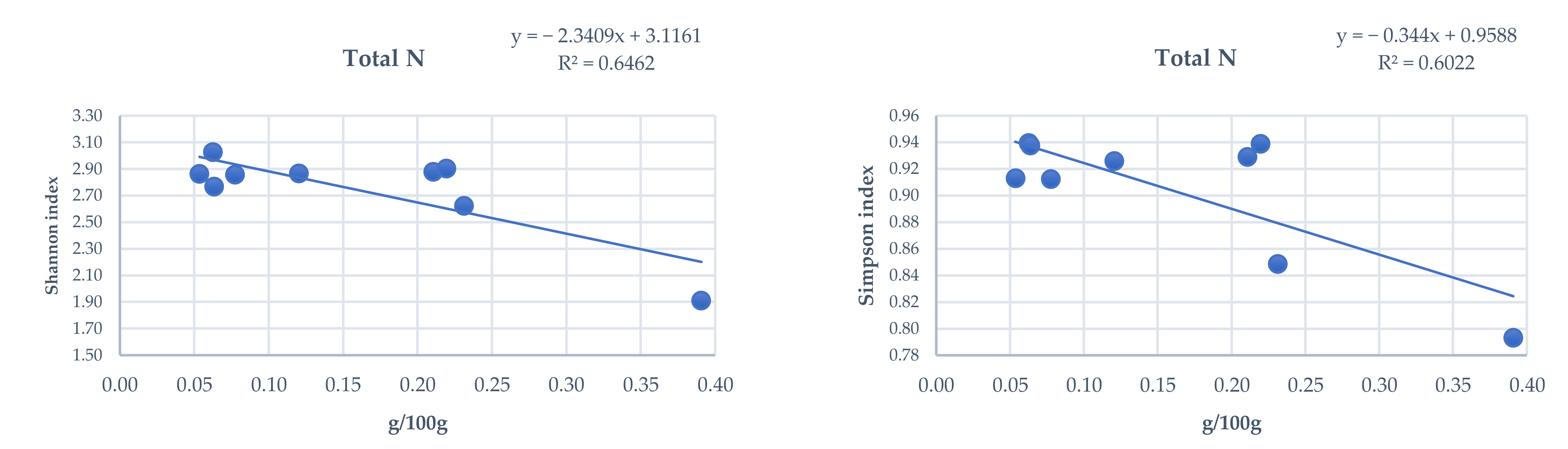
3.4. Influence of Total and Phytoavailable Soil Elements on P. spathulatus Communities
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Unger, F. Ueber den Einfluss des Bodens auf die Vertheilung der Gewächse: Nachgewiesen in der Vegetation des nordöstlichen Tirol’s; Bei Rohrmann und Schweigerd: Vienna, Austria, 1836. [Google Scholar] [CrossRef]
- Thurmann, J. Essai de Phytostatique Appliqué à la Chaîne du Jura et aux Contrées Voisines: Ou Étude de la Dispersion des Plantes Vasculaires Envisagée Principalement Quant à L’influence des Roches Soujacentes (Volume 1); Jent et Gassmann: Bern, Switzerland, 1849. [Google Scholar] [CrossRef]
- Kruckeberg, A.R. Geology and Plant Life: The Effects of Landforms and Rock Types on Plants; University of Washington Press: Washington, DC, USA, 2002. [Google Scholar]
- Mota, J.; Garrido-Becerra, J.; Merlo, M.; Medina-Cazorla, J.; Sánchez-Gómez, P. The edaphism: Gypsum, dolomite and serpentine flora and vegetation. In The Vegetation of the Iberian Peninsula; Loidi, J., Ed.; Springer Nature: Basel, Switzerland, 2017. [Google Scholar] [CrossRef]
- Schimper, A.F.W.; Fisher, W.R. Plant-Geography upon a Physiological Basis; Clarendon Press: Oxford, UK, 1902. [Google Scholar] [CrossRef]
- Rivas-Goday, S. Flora Serpentinícola Española: Nota Primera (Edafismos Endémicos del Reino de Granada); Real Academia de Farmacia: Madrid, Spain, 1969. [Google Scholar]
- Merlo, M.; Mota, J.; Cabello, J.; Alemán, M. La gipsofilia en plantas: Un apasionante edafismo. Investig. Gestión 1998, 3, 103–112. [Google Scholar]
- Mota, J.; Merlo, E.; Pérez-García, F.; Garrido-Becerra, J.; Martínez-Hernández, F.; Sánchez-Gómez, P.; Gil de Carrasco, C. Plants growing on unusual substrates: The edaphism. Notes from the antipodes (Iberian Peninsula). In Proceedings and Abstracts New Zealand: New home, New Habitat; Publishers Association of New Zealand Inc.: Palmerston North, New Zealand, 2007. [Google Scholar]
- Proctor, J. Vegetation and soil and plant chemistry on ultramafic rocks in the tropical Far East. Perspect. Plant Ecol. Evol. Syst. 2003, 6, 105–124. [Google Scholar] [CrossRef]
- Rajakaruna, N. The edaphic factor in the origin of plant species. Int. Geol. Rev. 2004, 46, 471–478. [Google Scholar] [CrossRef]
- Mota, J.; Medina-Cazorla, J.; Navarro, F.; Pérez-García, F.; Pérez-Latorre, A.; Sánchez-Gómez, P.; Merlo, M. Dolomite flora of the baetic ranges glades (South Spain). Flora Morphol. Distrib. Funct. Ecol. Plants 2008, 203, 359–375. [Google Scholar] [CrossRef]
- Merlo, M.; Mota, J.; Sánchez-Gómez, P. Ecofisiología y adaptaciones de las plantas vasculares a las características físicas y químicas de sustratos especiales. In Diversidad Vegetal de las Yeseras Ibéricas; Guirado Romero, J.S., Mota, J., Sánchez Gómez, P., Eds.; ADIF—Mediterráneo Asesores Consultores: Almería, Spain, 2011. [Google Scholar]
- Merlo, M.E.; Garrido-Becerra, J.A.; Mota, J.F.; Salmerón-Sánchez, E.; Martínez-Hernández, F.; Mendoza-Fernández, A.; Pérez-García, F.J. Threshold ionic contents for defining the nutritional strategies of gypsophile flora. Ecol. Indic. 2019, 97, 247–259. [Google Scholar] [CrossRef]
- Mota, J.; Merlo, E.; Martínez-Hernández, F.; Mendoza-Fernández, A.; Pérez-García, F.; Salmerón-Sánchez, E. Plants on Rich-Magnesium Dolomite Barrens: A Global Phenomenon. Biology 2021, 10, 38. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Fernández, A.J.; Mota, J.F. Red Lists versus nature protection Acts: New analytical and numerical method to test threat trends. Biodiv. Conserv. 2016, 25, 239–260. [Google Scholar] [CrossRef]
- Nunes, L.J.R.; Raposo, M.A.M.; Gomes, C.J.P. The Impact of Tourism Activity on Coastal Biodiversity: A Case Study at Praia da Cova Redonda (Algarve—Portugal). Environments 2020, 7, 88. [Google Scholar] [CrossRef]
- Salmerón-Sánchez, E. Diversidad Genética, Estructura Poblacional, Filogeografía y Filogenia de Especies Dolomitícolas Presentes en las Cordilleras Béticas; Universidad Almería: Almería, Spain, 2018. [Google Scholar]
- Chilingar, G.; Bissell, H.; Wolf, K. Diagenesis of carbonate rocks. Dev. Sedimentol. 1967, 8, 179–322. [Google Scholar] [CrossRef]
- Pérez-García, F.J.; Medina-Cazorla, J.M.; Martínez-Hernández, F.; Garrido-Becerra, J.A.; Mendoza-Fernández, A.J.; Salmerón-Sánchez, E.; Mota, J.F. Iberian Baetic endemic flora and the implications for a conservation policy. Ann. Bot. Fenn. 2012, 49, 43–54. [Google Scholar] [CrossRef]
- Pérez-García, F.J.; Merlo, M.E.; Martínez-Hernández, F.; Medina-Cazorla, J.M.; Mendoza-Fernández, A.J.; Garrido-Becerra, J.A.; Salmerón-Sánchez, E.; Mota, J.F. Syntaxa-area relationships, lessons from the vegetation of the Betic high mountain ranges (southern Spain). Phytocoenologia 2014, 44, 309–323. [Google Scholar] [CrossRef]
- Parolly, G.; Hein, P. Arabis lycia (Cruciferae), a new chasmophyte from the Taurus Mts, Turkey, and notes on related species. Willdenowia 2014, 30, 293–304. [Google Scholar] [CrossRef]
- Ritter-Studnička, H. Flora and vegetation on dolomites of Bosnia and Herzegovina (I-Konjic). God. Biol. Inst. Sarajevu 1956, 9, 73–122. [Google Scholar]
- Brullo, C.; Brullo, S.; Giusso, G. Considerations on the endemic flora of Sicily. Islands and Plants: Preservation and Understanding of Flora on Mediterranean Islands. In Proceedings of the 2nd Botanical Conference in Menorca, Proceedings and Abstracts, Menorca, Spain, 26–30 April 2013. [Google Scholar]
- Vogiatzakis, I.; Griffiths, G. A GIS-based empirical model for vegetation prediction in Lefka Ori, Crete. Plant Ecol. 2006, 184, 311–323. [Google Scholar] [CrossRef]
- Dickoré, W.; Nüsser, M. Flora of Nanga Parbat (NW Himalaya, Pakistan): An annotated inventory of vascular plants with remarks on vegetation dynamics. Englera 2000, 19, 3–253. [Google Scholar] [CrossRef]
- Reid, J.; Hill, R.; Brown, M.; Hovenden, M. Vegetation of Tasmania. Nord. J. Bot. 1999, 19, 622. [Google Scholar] [CrossRef]
- Larson, D.; Matthes, U.; Kelly, P. Cliff ecology: Pattern and process in cliff ecosystems; Cambridge University Press: Cambridge, UK, 2005. [Google Scholar]
- Mota, J.; Peñas, J.; Cabello, J. Scree and ruderal weed vegetation of Andalusian highlands (South Spain). Fitosociologia 1997, 32, 229–237. [Google Scholar]
- Rivas Goday, S.; Mayor López, M. Aspectos de la vegetación y flora orófila del Reino de Granada. An. Real Acad. Farm. 1966, 31, 345–400. [Google Scholar]
- Mota, J.F.; Valle, F.; Cabello, J. Dolomitic vegetation of South Spain. Vegetatio 1993, 109, 29–45. [Google Scholar] [CrossRef]
- Pérez-Latorre, A. Restauración vegetal de extracciones de áridos dolomíticos en la Costa del Sol occidental (Sierra de Mijas, Málaga). Ecología 1998, 12, 123–134. [Google Scholar]
- Medina-Cazorla, J.; Pérez-García, F.; Garrido-Becerra, J.; Martínez-Hernández, F.; Mendoza, A.; Romera, M.; Mota, J. Análisis biogeográfico de la flora de dolomías de Andalucía: Ensayo preliminar. In Ambientes Mediterráneos. Funcionamiento, Biodiversidad y Conservación de los Ecosistemas Mediterráneos; Colección Medio Ambiente: Almería, Spain, 2007. [Google Scholar]
- Medina-Cazorla, J.M.; de Carrasco, C.G.; Merlo, M.E.; Martínez-Hernández, F.; Garrido-Becerra, J.A.; Salmerón, E.; Mendoza, A.J.; Pérez-García, F.J.; Mota, J.F. The dolomite shrublands of the Convolvuletalia boissieri order and their preservation by means of the Habitats Directive. Acta Bot. Gall. 2010, 157, 611–625. [Google Scholar] [CrossRef]
- Medina-Cazorla, J.M.; Garrido-Becerra, J.A.; Fernández, A.M.; Pérez-García, F.J.; Salmerón, E.; Gil, C.; Poveda, J.F.M. Biogeography of the Baetic Ranges (SE Spain): A historical approach using cluster and parsimony analyses of endemic dolomitophytes. Plant Biosyst. 2010, 144, 111–120. [Google Scholar] [CrossRef]
- Navarro-Fernández, C.M.; Aroca, R.; Barea, J.M. Influence of arbuscular mycorrhizal fungi and water regime on the development of endemic Thymus species in dolomitic soils. Appl. Soil Ecol. 2011, 48, 31–37. [Google Scholar] [CrossRef]
- Alonso, C.; García-Sevilla, M. Strong inbreeding depression and individually variable mating system in the narrow endemic Erodium cazorlanum (Geraniaceae). An. Jard. Bot. Madr. 2013, 70, 72–80. [Google Scholar] [CrossRef]
- Salmerón-Sánchez, E.; Fuertes-Aguilar, J.; Španiel, S.; Pérez-García, F.J.; Merlo, E.; Garrido-Becerra, J.A.; Mota, J. Plant evolution in alkaline magnesium-rich soils: A phylogenetic study of the mediterranean genus Hormathophylla (Cruciferae: Alysseae) based on nuclear and plastid sequences. PLoS ONE 2018, 13, e0208307. [Google Scholar] [CrossRef]
- Salmerón-Sánchez, E.; Merlo, M.E.; Medina-Cazorla, J.M.; Pérez-García, F.J.; Martínez-Hernández, F.; Garrido-Becerra, J.A.; Mendoza-Fernández, A.J.; Valle, F.; Mota, J.F. Variability, genetic structure and phylogeography of the dolomitophilous species Convolvulus boissieri (Convolvulaceae) in the Baetic ranges, inferred from AFLPs, plastid DNA and ITS sequences. Bot. J. Linn. 2014, 176, 506–523. [Google Scholar] [CrossRef]
- Mota, J.; Salmerón-Sánchez, E.; Pérez-García, F.; Martínez-Hernández, F.; Mendoza-Fernández, A.; Medina-Cazorla, J.; Merlo, M. Catálogo Delphi de la flora edafoendémica de los blanquizales dolomíticos béticos: Bases para su conocimiento y conservación. In Biología de la Conservación de Plantas en Sierra Nevada: Principios y Retos Para su Preservación; Peñas de Giles, J., Lorite, J., Eds.; Editorial Universidad de Granada: Granada, Spain, 2019. [Google Scholar]
- Médail, F.; Quézel, P. Biodiversity hotspots in the Mediterranean basin: Setting global conservation priorities. Conserv. Biol. 1999, 13, 1510–1513. [Google Scholar] [CrossRef]
- Mendoza-Fernández, A.J.; Pérez-García, F.J.; Martínez-Hernández, F.; Salmerón-Sánchez, E.; Lahora, A.; Merlo, M.E.; Mota, J.F. Red List Index application for vascular flora along an altitudinal gradient. Biodivers. Conserv. 2019, 28, 1029–1048. [Google Scholar] [CrossRef]
- Salmerón-Sánchez, E.; Mendoza-Fernández, A.J.; Lorite, J.; Mota, J.F.; Peñas, J. Plant conservation in Mediterranean-type ecosystems. Mediterr. Bot. 2021, 42, e71333. [Google Scholar] [CrossRef]
- Anonymous. Council Directive 92/43/EEC of 21 May 1992 on the conservation of natural habitats and of wild fauna and flora. DOCE 1992, 206, 7–50. [Google Scholar]
- Bobo-Pinilla, J.; Salmerón-Sánchez, E.; Mota, J.F.; Peñas, J. Genetic conservation strategies of endemic plants from edaphic habitat islands: The case of Jacobaea auricula (Asteraceae). J. Nat. Conserv. 2021, 61, 126004. [Google Scholar] [CrossRef]
- Watanabe, T.; Maejima, E.; Yoshimura, T.; Urayama, M.; Yamauchi, A.; Owadano, M.; Shinano, T. The ionomic study of vegetable crops. PLoS ONE 2016, 11, e0160273. [Google Scholar] [CrossRef]
- Baxter, I.R.; Vitek, O.; Lahner, B.; Muthukumar, B.; Borghi, M.; Morrissey, J.; Salt, D.E. The leaf ionome as a multivariable system to detect a plant’s physiological status. Proc. Natl. Acad. Sci. USA 2008, 105, 12081–12086. [Google Scholar] [CrossRef] [PubMed]
- Chao, D.; Gable, K.; Chen, M.; Baxter, I.; Dietrich, C.; Cahoon, E.; Salt, D. Sphingolipids in the root play an important role in regulating the leaf ionome in Arabidopsis thaliana. Plant Cell 2011, 23, 1061–1081. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Broadley, M.; Jansen, S.; White, P.; Takada, J.; Satake, K.; Osaki, M. Evolutionary control of leaf element composition in plants. New Phytol. 2007, 174, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Sha, Z.; Oka, N.; Watanabe, T.; Tampubolon, B.; Okazaki, K.; Osaki, M.; Shinano, T. Ionome of soybean seed affected by previous cropping with mycorrhizal plant and manure application. J. Agric. Food Chem. 2012, 60, 9543–9552. [Google Scholar] [CrossRef]
- White, P.; Broadley, M.; Thompson, J.; McNicol, J.; Crawley, M.; Poulton, P.; Johnston, A. Testing the distinctness of shoot ionomes of angiosperm families using the Rothamsted Park Grass Continuous Hay Experiment. New Phytol. 2012, 196, 101–109. [Google Scholar] [CrossRef]
- Salt, D.E.; Baxter, I.; Lahner, B. Ionomics and the study of the plant ionome. Annu. Rev. Plant Biol. 2008, 59, 709–733. [Google Scholar] [CrossRef]
- Kattge, J.; Diaz, S.; Lavorel, S.; Prentice, I.C.; Leadley, P.; Bönisch, G.; Wirth, C. TRY-a global database of plant traits. Glob. Chang. Biol. 2011, 17, 2905–2935. [Google Scholar] [CrossRef]
- Peñuelas, J.; Fernández-Martínez, M.; Ciais, P.; Jou, D.; Piao, S.; Obersteiner, M.; Sardans, J. The bioelements, the elementome, and the biogeochemical niche. Ecology 2019, 100, e02652. [Google Scholar] [CrossRef]
- Müllerová, V.; Hejcman, M.; Hejcmanová, P.; Pavlů, V. Effect of fertilizer application on Urtica dioica and its element concentrations in a cut grassland. Acta Oecol. 2014, 59, 1–6. [Google Scholar] [CrossRef]
- Wang, M.; Moore, T. Carbon, nitrogen, phosphorus, and potassium stoichiometry in an ombrotrophic peatland reflects plant functional type. Ecosystems 2014, 17, 673–684. [Google Scholar] [CrossRef]
- Carreira, J.; Niell, F.; Lajtha, K. Soil nitrogen availability and nitrification in Mediterranean shrublands of varying fire history and successional stage. Biogeochemistry 1994, 26, 189–209. [Google Scholar] [CrossRef]
- Brady, K.; Kruckeberg, A.; Bradshaw, H., Jr. Evolutionary ecology of plant adaptation to serpentine soils. Annu. Rev. Ecol. Evol. Syst. 2005, 36, 243–266. [Google Scholar] [CrossRef]
- Oze, C.; Skinner, C.; Schroth, A.; Coleman, R. Growing up green on serpentine soils: Biogeochemistry of serpentine vegetation in the Central Coast Range of California. Appl. Geochem. 2008, 23, 3391–3403. [Google Scholar] [CrossRef]
- Visscher, A.; Paul, A.; Kirst, M.; Guy, C.; Schuerger, A.; Ferl, R. Growth performance and root transcriptome remodeling of Arabidopsis in response to Mars-like levels of magnesium sulfate. PLoS ONE 2010, 5, e12348. [Google Scholar] [CrossRef]
- Guo, W. Magnesium homeostasis mechanisms and magnesium use efficiency in plants. In Plant Macronutrient Use Efficiency: Molecular and Genomic Perspectives in Crop Plants; Hossain, M.A., Kamiya, T., Burritt, D., Tran, L.S.P., Fujiwara, T., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 197–213. [Google Scholar] [CrossRef]
- Cunningham, S.; Berti, W.; Huang, J. Phytoremediation of contaminated soils. Trends Biotechnol. 1995, 13, 393–397. [Google Scholar] [CrossRef]
- Thangavel, P.; Subbhuraam, C. Phytoextraction: Role of hyperaccumulators in metal contaminated soils. Proc. Indian Natl. Sci. Acad. 2004, 70, 109–130. [Google Scholar]
- Delgadillo-López, A.; González-Ramírez, C.; Prieto-García, F.; Villagómez-Ibarra, J.; Acevedo-Sandoval, O. Fitorremediación: Una alternativa para eliminar la contaminación. Trop. Subtrop. Agroecosyst. 2011, 14, 597–612. [Google Scholar]
- Brooks, R. Plants That Hyperaccumulate Heavy Metals, Their Role in Phytoremediation, Microbiology, Archaeology, Mineral Exploration and Phytomining; Cab International: New York, NY, USA, 1998. [Google Scholar]
- Buscaroli, A. An overview of indexes to evaluate terrestrial plants for phytoremediation purposes. Ecol. Indic. 2017, 82, 367–380. [Google Scholar] [CrossRef]
- Baker, A. Accumulators and excluders-strategies in the response of plants to heavy metals. J. Plant Nutr. 1981, 3, 643–654. [Google Scholar] [CrossRef]
- Mota-Merlo, M.; Martos, V. Use of machine learning to establish limits in the classification of hyperaccumulator plants growing on serpentine, gypsum and dolomite soils. Mediterr. Bot. 2021, 42, e67609. [Google Scholar] [CrossRef]
- Castroviejo, S. Flora Iberica. Plantas Vasculares de la Península Ibérica e Islas Baleares; Real Jardín Botánico de Madrid: Madrid, Spain, 2012. [Google Scholar]
- Heinert, M.; Endara, A.; Solórzano, H. Plasma acoplado inductivamente en espectroscopia de emisión óptica (ICP-OES). Reci-mundo 2020, 4, 4–12. [Google Scholar] [CrossRef]
- Abedin, J.; Beckett, P.; Spiers, G. An evaluation of extractants for assessment of metal phytoavailability to guide reclamation practices in acidic soilscapes in northern regions. Can. J. Soil Sci. 2012, 92, 253–268. [Google Scholar] [CrossRef]
- Quevauviller, P.; Lachica, M.; Barahona, E.; Gomez, A.; Rauret, G.; Ure, A.; Muntau, H. Certified reference material for the quality control of EDTA-and DTPA-extractable trace metal contents in calcareous soil (CRM 600). Fresenius J. Anal. Chem. 1998, 360, 505–511. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.; Ryan, P. PAST: Paleontological statistics software package for education and data analysis. Paleontol. Electron. 2001, 4, 9. [Google Scholar]
- IBM Corporation. Released. SPSS Software Version 26.0; IBM: Armonk, NY, USA, 2019. [Google Scholar]
- Jiménez Terrón, I. El patrimonio minero. In Sierra de Gádor: Patrimonio Natural e Infraestructura Verde de Almería; CAESCG: Almería, Spain, 2016. [Google Scholar]
- Marschner, H. Marschner’s Mineral Nutrition of Higher Plants; Academic Press: Adelaide, Australia, 2011. [Google Scholar]
- Goldberg, S. Interaction of aluminium and iron oxides and clay minerals and their effect on soil physical properties: A review. Commun. Soil Sci. Plant Anal. 1989, 20, 1181–1207. [Google Scholar] [CrossRef]
- Baker, A.; Reeves, R.; Hajar, A. Heavy metal accumulation and tolerance in British populations of the metallophyte Thlaspi caerulescens J. & C. Presl (Brassicaceae). New Phytol. 1994, 127, 61–68. [Google Scholar] [CrossRef]
- Poschenrieder, C.; Llugany, M.; Lombini, A.; Dinelli, E.; Bech, J.; Barceló, J. Smilax aspera L. an evergreen Mediterranean climber for phytoremediation. J. Geochem. Explor. 2012, 123, 41–44. [Google Scholar] [CrossRef]
- Camacho-Cristóbal, J.; Rexach, J.; González-Fontes, A. Boron in plants: Deficiency and toxicity. J. Integr. Plant Biol. 2008, 50, 1247–1255. [Google Scholar] [CrossRef]
- Abreu, C.; Raij, B.; Abreu, M.; Gonzalez, A.P. Routine soil testing to monitor heavy metals and boron in soils. Sci. Agric. 2005, 62, 1–6. [Google Scholar] [CrossRef]
- Babaoğlu, M.; Gezgın, S.; Topal, A.; Sade, B.; Dural, H. Gypsophila sphaerocephala Fenzl ex Tchihat.: A boron hyperaccumulator plant species that may phytoremediate soils with toxic B levels. Turk. J. Bot. 2004, 28, 273–278. [Google Scholar]
- Jiang, Y.; Xia, W.; Zhang, B.; Pan, X.; Liu, W.; Jin, S.; Li, Y. Predictors of thallium exposure and its relation with preterm birth. Environ. Pollut. 2018, 233, 971–976. [Google Scholar] [CrossRef]
- Vaněk, A.; Grösslová, Z.; Mihaljevič, M.; Ettler, V.; Trubač, J.; Chrastný, V.; Ash, C. Thallium isotopes in metallurgical wastes/contaminated soils: A novel tool to trace metal source and behavior. J. Hazard. Mater. 2018, 343, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Biagioni, C.; D’Orazio, M.; Lepore, G.O.; d’Acapito, F.; Vezzoni, S. Thallium-rich rust scales in drinkable water distribution systems: A case study from northern Tuscany, Italy. Sci. Total Environ. 2017, 587, 491–501. [Google Scholar] [CrossRef]
- Pavoni, E.; Covelli, S.; Adami, G.; Baracchini, E.; Cattelan, R.; Crosera, M.; Petranich, E. Mobility and fate of Thallium and other potentially harmful elements in drainage waters from a decommissioned Zn-Pb mine (North-Eastern Italian Alps). J. Geochem. Explor. 2018, 188, 1–10. [Google Scholar] [CrossRef]
- Karbowska, B.; Rębiś, T.; Milczarek, G. Electrode modified by reduced graphene oxide for monitoring of total thallium in grain products. Int. J. Environ. Res. Public Health 2018, 15, 653. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, R.; Upreti, R.; Seth, P.; Chaturvedi, U. Effects of chromium on the immune system. FEMS Immunol. Med. Microbiol. 2002, 34, 1–7. [Google Scholar] [CrossRef]
- Shanker, A.; Cervantes, C.; Loza-Tavera, H.; Avudainayagam, S. Chromium toxicity in plants. Environ. Int. 2005, 31, 739–753. [Google Scholar] [CrossRef]
- Zayed, A.; Terry, N. Chromium in the environment: Factors affecting biological remediation. Plant Soil 2003, 249, 139–156. [Google Scholar] [CrossRef]
- Boyd, R.; Martens, S. Nickel hyperaccumulated by Thlaspi montanum var. montanum is acutely toxic to an insect herbivore. Oikos 1994, 70, 21–25. [Google Scholar] [CrossRef]
- Boyd, R.; Moar, W. The defensive function of Ni in plants: Response of the polyphagous herbivore Spodoptera exigua (Lepidoptera: Noctuidae) to hyperaccumulator and accumulator species of Streptanthus (Brassicaceae). Oecologia 1999, 118, 218–224. [Google Scholar] [CrossRef]
- Boyd, R.; Shaw, J.; Martens, S. Nickel hyperaccumulation defends Streptanthus polygaloides (Brassicaceae) against pathogens. Am. J. Bot. 1994, 81, 294–300. [Google Scholar] [CrossRef]
- Ghaderian, Y.; Lyon, A.; Baker, A. Seedling mortality of metal hyperaccumulator plants resulting from damping off by Pythium spp. New Phytol. 2000, 146, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Bhuyan, M.; Raza, A.; Hawrylak-Nowak, B.; Matraszek-Gawron, R.; Al Mahmud, J.; Fujita, M. Selenium in plants: Boon or bane? Environ. Exp. Bot. 2020, 104170. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E.; Moller, I.M.; Murphy, A. (Eds.) Plant Physiology and Development, 6th ed.; Sinauer Associates: Sunderland, CT, USA, 2015. [Google Scholar]


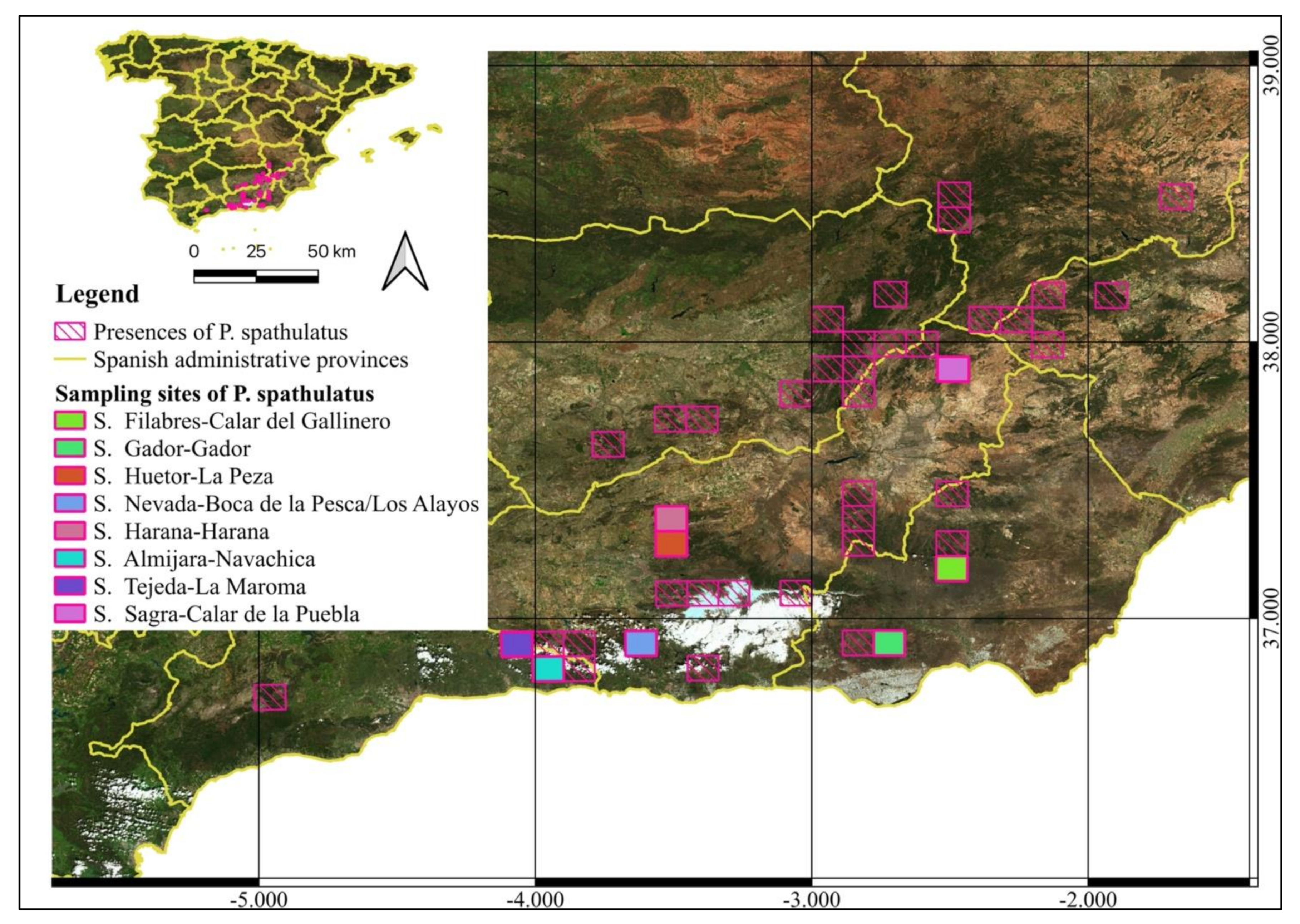

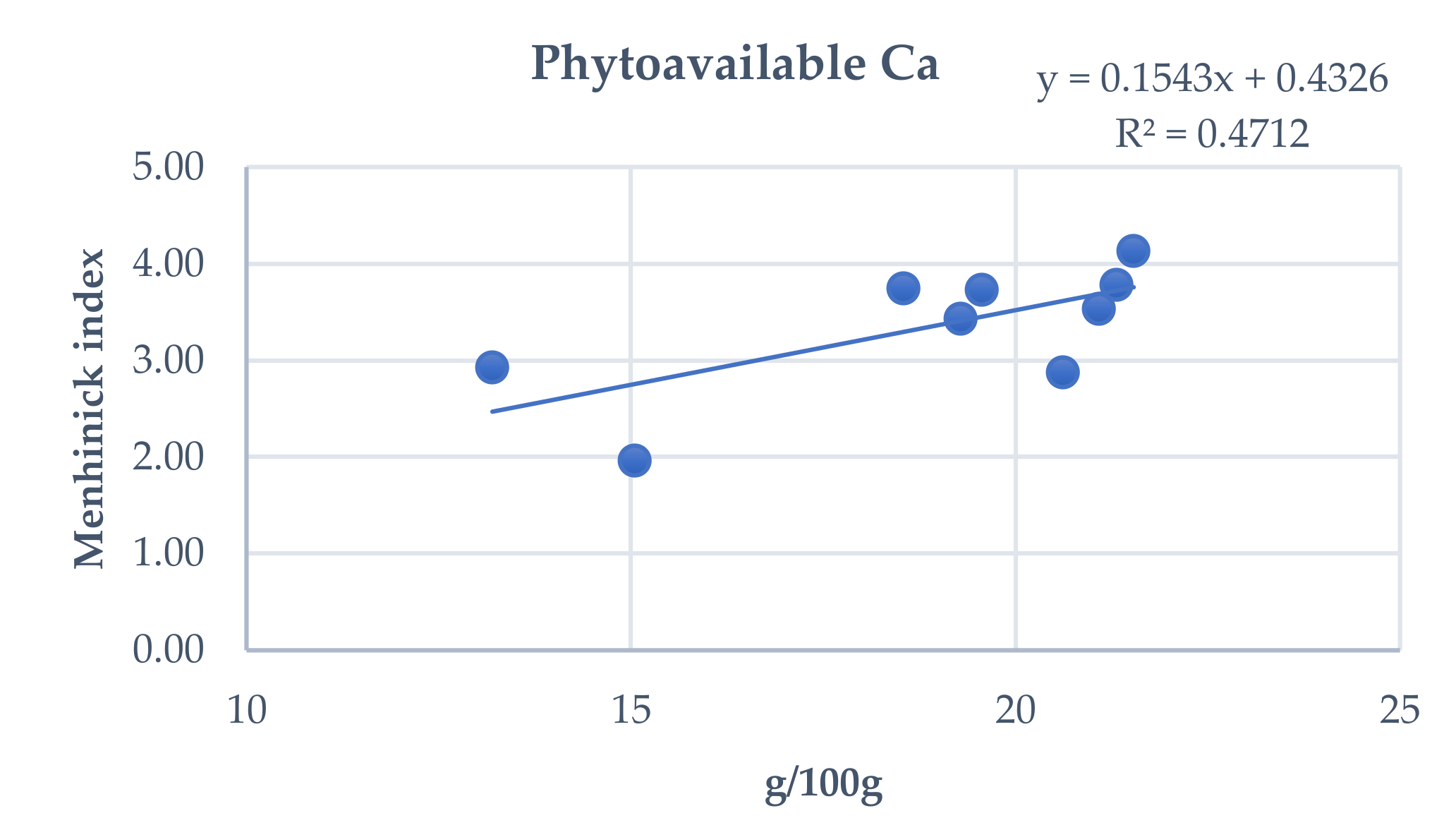
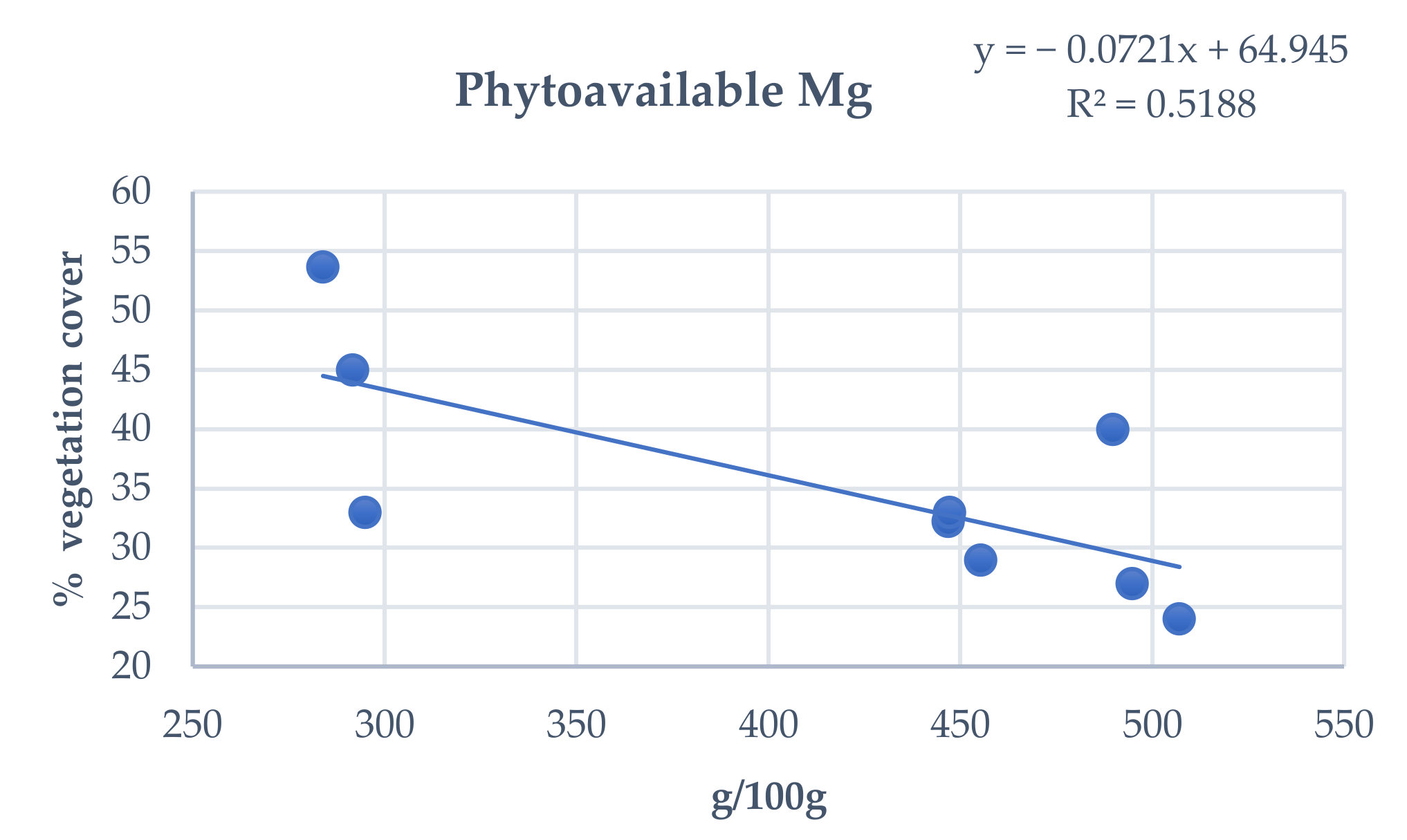
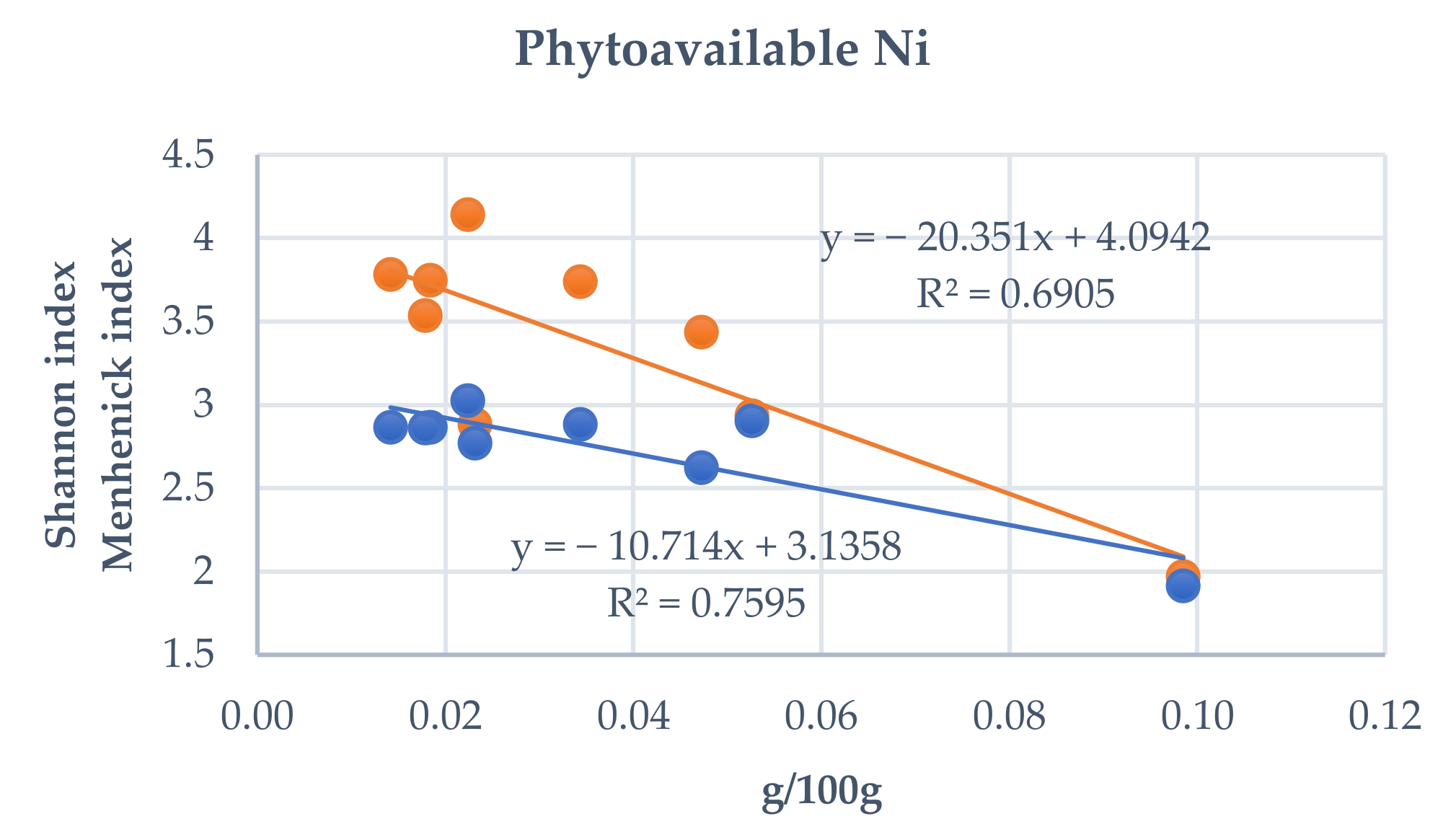
| Range | Study Area | Cod. | Altitude (m) | X | Y |
|---|---|---|---|---|---|
| Sierra de los Filabres | Calar del Gallinero | GAL | 2020 | 542733 | 4123047 |
| Sierra de Gador | Gador | GAD | 2180 | 515730 | 4084443 |
| Sierra de Huetor | La Peza | PEZ | 1085 | 461908 | 4123864 |
| Sierra Nevada | Boca de la Pesca | BOC | 1350 | 451598 | 4103580 |
| Sierra Nevada | Los Alayos | ALA | 1980 | 451598 | 4103580 |
| Sierra Harana | Harana | HAR | 2027 | 456418 | 4130782 |
| Sierra de Almijara | Navachica | NAV | 1832 | 429674 | 4082412 |
| Sierra de Tejeda | La Maroma | MAR | 2069 | 406846 | 4084642 |
| Sierra de la Sagra | Calar de la Puebla | PUE | 1800 | 547695 | 4199135 |
| Macronutrients (g/100 g) | Micronutrients (mg/kg) | Other Elements (mg/kg) | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | N | Ca | K | Mg | P | S | B | Cu | Fe | Mn | Mo | Ni | Zn | Al | Cr | Pb | Si | Tl | ||
| MAR | 14.32 | 0.21 | 19.57 | 0.14 | 11.49 | 0.04 | 0.16 | 2.68 | 3.61 | 3702.39 | 708.05 | 0.44 | 4.08 | 24.92 | 0.63 | 20.25 | 20.11 | 163.66 | 9.85 | |
| GAL | 13.13 | 0.39 | 15.05 | 0.47 | 8.75 | 0.05 | 0.16 | 11.60 | 10.95 | 15,290.77 | 1183.68 | 2.31 | 10.13 | 41.13 | 2.04 | 31.01 | 42.71 | 533.96 | 14.23 | |
| BOC | 13.45 | 0.06 | 21.54 | 0.08 | 14.07 | 0.01 | 0.16 | 1.09 | 1.70 | 1247.20 | 697.25 | 1.60 | 2.26 | 34.01 | 0.23 | 6.03 | 24.67 | 126.32 | 7.44 | |
| PEZ | 13.57 | 0.05 | 21.31 | 0.07 | 13.22 | 0.01 | 0.16 | 0.29 | 1.33 | 1009.78 | 682.71 | 2.24 | 1.54 | 8.80 | 0.19 | 4.64 | 7.75 | 129.76 | 7.46 | |
| ALA | 12.97 | 0.08 | 21.08 | 0.08 | 13.19 | 0.01 | 0.16 | 0.90 | 1.85 | 1446.81 | 713.33 | 0.73 | 2.09 | 26.77 | 0.28 | 13.21 | 11.51 | 247.00 | 8.22 | |
| GAD | 10.28 | 0.22 | 13.20 | 0.66 | 7.47 | 0.05 | 0.15 | 19.45 | 52.82 | 26,207.33 | 2029.63 | 1.90 | 14.36 | 236.86 | 2.40 | 37.79 | 410.92 | 526.98 | 18.18 | |
| NAV | 13.63 | 0.06 | 20.61 | 0.05 | 12.75 | 0.01 | 0.16 | 0.00 | 0.99 | 915.32 | 720.44 | 0.88 | 1.47 | 16.38 | 0.17 | 5.45 | 9.26 | 131.66 | 7.59 | |
| PUE | 13.84 | 0.23 | 19.29 | 0.20 | 9.09 | 0.02 | 0.19 | 6.76 | 3.84 | 3936.07 | 610.54 | 0.22 | 5.04 | 20.84 | 0.79 | 16.93 | 20.64 | 424.17 | 10.07 | |
| HAR | 13.76 | 0.12 | 18.54 | 0.07 | 10.44 | 0.01 | 0.17 | 2.43 | 1.37 | 947.60 | 658.31 | 0.25 | 1.54 | 5.04 | 0.21 | 6.39 | 2.47 | 164.93 | 7.55 | |
| 13.22 | 0.16 | 18.91 | 0.20 | 11.16 | 0.02 | 0.16 | 5.02 | 8.72 | 6078.14 | 889.33 | 1.17 | 4.72 | 46.08 | 0.77 | 15.74 | 61.12 | 272.05 | 10.07 | ||
| SD | 1.17 | 0.11 | 2.93 | 0.21 | 2.34 | 0.02 | 0.01 | 6.58 | 16.82 | 8829.94 | 459.61 | 0.85 | 4.56 | 72.44 | 0.86 | 12.03 | 131.71 | 173.84 | 3.75 | |
| Macronutrients (mg/L) | Micronutrients (mg/L) | Other Elements (mg/L) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ca | K | Mg | P | S | Fe | Mn | Zn | Al | Pb | Si | |
| MAR | 1391.90 | 16.47 | 507.10 | 3.86 | 15.77 | 10.18 | 6.95 | 0.53 | 12.41 | 1.13 | 10.31 |
| GAL | 1487.25 | 37.92 | 295.00 | 4.55 | 19.83 | 14.57 | 20.15 | 0.43 | 16.32 | 1.29 | 13.56 |
| BOC | 1081.00 | 10.35 | 494.85 | 1.37 | 16.99 | 3.74 | 4.37 | 0.80 | 5.02 | 0.71 | 5.36 |
| PEZ | 1897.12 | 19.76 | 893.60 | 2.94 | 21.28 | 9.21 | 8.05 | 0.32 | 11.48 | 0.86 | 12.56 |
| ALA | 2217.35 | 23.22 | 979.40 | 2.74 | 22.34 | 8.26 | 9.15 | 1.03 | 12.56 | 1.14 | 12.93 |
| GAD | 792.83 | 28.26 | 291.70 | 2.20 | 13.77 | 14.28 | 23.30 | 1.58 | 13.23 | 20.65 | 7.25 |
| NAV | 2378.25 | 24.05 | 844.60 | 3.27 | 28.24 | 11.21 | 9.84 | 0.56 | 16.69 | 0.78 | 11.31 |
| PUE | 1455.07 | 18.86 | 283.97 | 2.94 | 17.97 | 12.02 | 4.08 | 0.24 | 12.53 | 0.80 | 9.06 |
| HAR | 1051.55 | 9.07 | 455.35 | 1.66 | 12.91 | 3.56 | 4.26 | 0.11 | 4.97 | 0.10 | 4.59 |
| 1528.03 | 20.88 | 560.62 | 2.84 | 18.79 | 9.67 | 10.02 | 0.62 | 11.69 | 3.05 | 9.66 | |
| SD | 539.35 | 8.91 | 274.90 | 1.01 | 4.77 | 4.00 | 7.01 | 0.46 | 4.19 | 6.61 | 3.31 |
| Macronutrients (g/100 g) | Micronutrients (mg/kg) | Other Elements (mg/kg) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | N | Ca | K | Mg | P | S | B | Cu | Fe | Mn | Zn | Al | Cr | Pb | Tl | |
| MAR | 44.15 | 1.46 | 1.65 | 0.47 | 0.54 | 0.06 | 0.11 | 17.35 | 4.46 | 73.66 | 39.39 | 13.98 | 62.98 | 1.33 | 1.20 | 18.42 |
| GAL | 44.70 | 1.33 | 1.13 | 0.67 | 0.22 | 0.04 | 0.09 | 17.27 | 2.60 | 58.15 | 24.14 | 13.84 | 57.31 | 0.18 | 0.64 | 0.64 |
| BOC | 44.81 | 1.56 | 1.15 | 1.10 | 0.23 | 0.07 | 0.10 | 16.00 | 6.22 | 140.01 | 63.67 | 22.88 | 116.25 | 0.34 | 5.68 | 0.57 |
| PEZ | 44.34 | 1.50 | 1.40 | 0.80 | 0.24 | 0.05 | 0.13 | 16.01 | 4.16 | 117.07 | 50.47 | 13.09 | 111.54 | 0.40 | 0.99 | 2.59 |
| ALA | 42.25 | 1.30 | 1.45 | 0.64 | 0.67 | 0.05 | 0.11 | 15.32 | 3.83 | 722.50 | 67.84 | 11.56 | 780.95 | 10.53 | 2.45 | 110.80 |
| GAD | 44.57 | 1.57 | 0.83 | 0.83 | 0.23 | 0.05 | 0.09 | 11.92 | 3.66 | 39.64 | 20.70 | 16.32 | 32.50 | 0.19 | 0.56 | 2.68 |
| NAV | 45.29 | 1.84 | 1.09 | 0.64 | 0.36 | 0.09 | 0.14 | 9.72 | 2.94 | 77.04 | 34.86 | 17.65 | 69.78 | 0.16 | 0.69 | 9.30 |
| PUE | 44.49 | 1.48 | 1.47 | 0.94 | 0.49 | 0.07 | 0.11 | 11.56 | 2.70 | 90.11 | 50.53 | 19.77 | 104.71 | 0.71 | 1.25 | 15.22 |
| HAR | 44.40 | 1.32 | 1.38 | 0.60 | 0.47 | 0.06 | 0.09 | 18.10 | 3.71 | 60.48 | 33.88 | 8.07 | 56.54 | 0.65 | 0.75 | 11.80 |
| 44.33 | 1.48 | 1.28 | 0.74 | 0.38 | 0.06 | 0.11 | 14.81 | 3.81 | 153.18 | 42.83 | 15.24 | 154.73 | 1.61 | 1.58 | 19.11 | |
| SD | 0.85 | 0.17 | 0.25 | 0.19 | 0.17 | 0.01 | 0.02 | 2.99 | 1.11 | 215.70 | 16.48 | 4.46 | 236.57 | 3.36 | 1.64 | 35.00 |
| Foliar/Soil | C_s | Ca_s | K_s | Mg_s | P_s | B_s | Cu_s | Fe_s | Mn_s | Zn_s | Al_s | Cr_s | Pb_s | Tl_s |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ca_f | 0.769 * | 0.583 | −0.641 | 0.341 | −0.296 | −0.614 | −0.694 * | −0.680 * | −0.758 * | −0.705 * | −0.586 | −0.409 | −0.696 * | −0.589 |
| S_f | 0.472 | 0.741 * | −0.682 * | 0.679 * | −0.667 * | −0.707 * | −0.53 | −0.649 | −0.565 | −0.495 | −0.692 * | −0.695 * | −0.477 | −0.661 |
| Mn_f | 0.395 | 0.820 * | −0.670 * | 0.748 * | −0.701 * | −0.667 * | −0.579 | −0.682 * | −0.641 | −0.489 | −0.687 * | −0.608 | −0.520 | −0.677 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Merlo, E.; Mendoza-Fernández, A.J.; Salmerón-Sánchez, E.; Martínez-Hernández, F.; Ortiz-Úbeda, A.; Mota, J. Elementome of Endemic Dolomitic Flora: Pterocephalus spathulatus (Lag.) Coult. Land 2021, 10, 1253. https://doi.org/10.3390/land10111253
Merlo E, Mendoza-Fernández AJ, Salmerón-Sánchez E, Martínez-Hernández F, Ortiz-Úbeda A, Mota J. Elementome of Endemic Dolomitic Flora: Pterocephalus spathulatus (Lag.) Coult. Land. 2021; 10(11):1253. https://doi.org/10.3390/land10111253
Chicago/Turabian StyleMerlo, Encarna, Antonio J. Mendoza-Fernández, Esteban Salmerón-Sánchez, Fabián Martínez-Hernández, Andrea Ortiz-Úbeda, and Juan Mota. 2021. "Elementome of Endemic Dolomitic Flora: Pterocephalus spathulatus (Lag.) Coult" Land 10, no. 11: 1253. https://doi.org/10.3390/land10111253
APA StyleMerlo, E., Mendoza-Fernández, A. J., Salmerón-Sánchez, E., Martínez-Hernández, F., Ortiz-Úbeda, A., & Mota, J. (2021). Elementome of Endemic Dolomitic Flora: Pterocephalus spathulatus (Lag.) Coult. Land, 10(11), 1253. https://doi.org/10.3390/land10111253








