NF-RO Membrane Performance for Treating the Effluent of an Organized Industrial Zone Wastewater Treatment Plant: Effect of Different UF Types
Abstract
:1. Introduction
2. Materials and Methods
2.1. Kayseri Organized Industrial Zone (KOIZ) Wastewater Treatment Plant (WWTP)
2.2. Lab-Scale Filtration System
2.3. Experimental Methods
2.4. Membrane Characterization
3. Results and Discussion
3.1. UF Filtration of KOIZ Effluent
3.2. Characterization of UF Filters
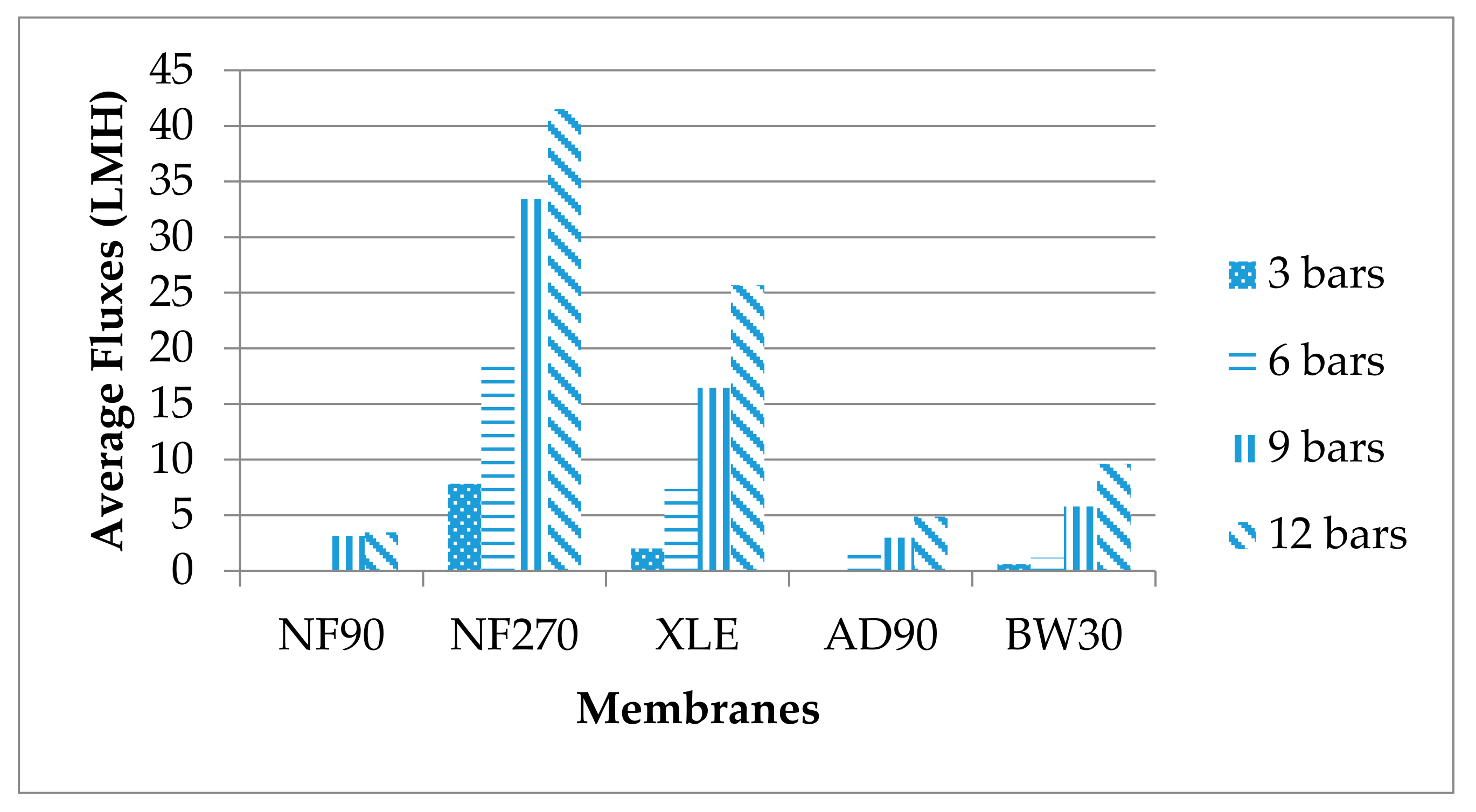
3.3. NF/RO Treatment of UF Effluents
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brown, A.; Matlock, M.D. A review of water scarcity indices and methodologies. White Pap. 2011, 106, 19. [Google Scholar]
- Into, M.; Jönsson, A.-S.; Lengdén, G. Reuse of industrial wastewater following treatment with reverse osmosis. J. Membr. Sci. 2004, 242, 21–25. [Google Scholar] [CrossRef]
- Turkish Republic, Ministry of Science, and Technology. Organized Industrial Zones (Organize Sanayi Bölgeleri). Available online: https://osbbs.sanayi.gov.tr/citylist.aspx (accessed on 5 January 2017). (In Turkish)
- Fane, A.G. Sustainability and membrane processing of wastewater for reuse. Desalination 2007, 202, 53–58. [Google Scholar] [CrossRef]
- Alturki, A.A.; Tadkaew, N.; McDonald, J.A.; Khan, S.J.; Price, W.E.; Nghiem, L.D. Combining mbr and nf/ro membrane filtration for the removal of trace organics in indirect potable water reuse applications. J. Membr. Sci. 2010, 365, 206–215. [Google Scholar] [CrossRef]
- Kappel, C.; Kemperman, A.; Temmink, H.; Zwijnenburg, A.; Rijnaarts, H.; Nijmeijer, K. Impacts of nf concentrate recirculation on membrane performance in an integrated mbr and nf membrane process for wastewater treatment. J. Membr. Sci. 2014, 453, 359–368. [Google Scholar] [CrossRef]
- Cartagena, P.; El Kaddouri, M.; Cases, V.; Trapote, A.; Prats, D. Reduction of emerging micropollutants, organic matter, nutrients and salinity from real wastewater by combined mbr-nf/ro treatment. Sep. Purif. Technol. 2013, 110, 132–143. [Google Scholar] [CrossRef]
- Sert, G.; Bunani, S.; Kabay, N.; Egemen, Ö.; Arda, M.; Pek, T.; Yüksel, M. Investigation of mini pilot scale mbr-nf and mbr-ro integrated systems performance—Preliminary field tests. J. Water Process Eng. 2016, 12, 72–77. [Google Scholar] [CrossRef]
- Kurt, E.; Koseoglu-Imer, D.Y.; Dizge, N.; Chellam, S.; Koyuncu, I. Pilot-scale evaluation of nanofiltration and reverse osmosis for process reuse of segregated textile dyewash wastewater. Desalination 2012, 302, 24–32. [Google Scholar] [CrossRef]
- Jegatheesan, V.; Pramanik, B.K.; Chen, J.; Navaratna, D.; Chang, C.-Y.; Shu, L. Treatment of textile wastewater with membrane bioreactor: A critical review. Bioresour. Technol. 2016, 204, 202–212. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Liu, L.; Liu, R.; Chen, J. Textile wastewater reuse as an alternative water source for dyeing and finishing processes: A case study. Desalination 2010, 258, 229–232. [Google Scholar] [CrossRef]
- Amin, M.M.; Heidari, M.; Momeni, S.A.R.; Ebrahimi, H. Performance evaluation of membrane bioreactor for treating industrial wastewater: A case study in isfahan mourchekhurt industrial estate. Int. J. Environ. Health Eng. 2016, 5, 12. [Google Scholar]
- Sánchez-Avila, J.; Bonet, J.; Velasco, G.; Lacorte, S. Determination and occurrence of phthalates, alkylphenols, bisphenol a, pbdes, pcbs and pahs in an industrial sewage grid discharging to a municipal wastewater treatment plant. Sci. Total Environ. 2009, 407, 4157–4167. [Google Scholar] [CrossRef] [PubMed]
- Sartor, M.; Kaschek, M.; Mavrov, V. Feasibility study for evaluating the client application of membrane bioreactor (mbr) technology for decentralised municipal wastewater treatment in Vietnam. Desalination 2008, 224, 172–177. [Google Scholar] [CrossRef]
- Juang, L.-C.; Tseng, D.-H.; Lin, H.-Y. Membrane processes for water reuse from the effluent of industrial park wastewater treatment plant: A study on flux and fouling of membrane. Desalination 2007, 202, 302–309. [Google Scholar] [CrossRef]
- Comerton, A.M.; Andrews, R.C.; Bagley, D.M.; Hao, C. The rejection of endocrine disrupting and pharmaceutically active compounds by nf and ro membranes as a function of compound and water matrix properties. J. Membr. Sci. 2008, 313, 323–335. [Google Scholar] [CrossRef]
- Lee, S.; Lueptow, R.M. Reverse osmosis filtration for space mission wastewater: Membrane properties and operating conditions. J. Membr. Sci. 2001, 182, 77–90. [Google Scholar] [CrossRef]
- Yuliwati, E.; Ismail, A.F. Effect of additives concentration on the surface properties and performance of pvdf ultrafiltration membranes for refinery produced wastewater treatment. Desalination 2011, 273, 226–234. [Google Scholar] [CrossRef]
- Tang, C.Y.; Fu, Q.S.; Criddle, C.S.; Leckie, J.O. Effect of flux (transmembrane pressure) and membrane properties on fouling and rejection of reverse osmosis and nanofiltration membranes treating perfluorooctane sulfonate containing wastewater. Environ. Sci. Technol. 2007, 41, 2008–2014. [Google Scholar] [CrossRef] [PubMed]
- Gozálvez-Zafrilla, J.M.; Sanz-Escribano, D.; Lora-García, J.; León Hidalgo, M.C. Nanofiltration of secondary effluent for wastewater reuse in the textile industry. Desalination 2008, 222, 272–279. [Google Scholar] [CrossRef]
- Sioutopoulos, D.C.; Yiantsios, S.G.; Karabelas, A.J. Relation between fouling characteristics of ro and uf membranes in experiments with colloidal organic and inorganic species. J. Membr. Sci. 2010, 350, 62–82. [Google Scholar] [CrossRef]
- Hayatbakhsh, M.; Sadrzadeh, M.; Pernitsky, D.; Bhattacharjee, S.; Hajinasiri, J. Treatment of an in situ oil sands produced water by polymeric membranes. Desalin. Water Treat. 2016, 57, 14869–14887. [Google Scholar] [CrossRef]
- Clesceri, L.S.; Greenberg, A.E.; Trussell, R.R. Standard Methods for the Examination of Water and Wastewater, 17th ed.; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- Fang, H.H.; Shi, X. Pore fouling of microfiltration membranes by activated sludge. J. Membr. Sci. 2005, 264, 161–166. [Google Scholar] [CrossRef]
- Qu, F.; Liang, H.; Zhou, J.; Nan, J.; Shao, S.; Zhang, J.; Li, G. Ultrafiltration membrane fouling caused by extracellular organic matter (eom) from microcystis aeruginosa: Effects of membrane pore size and surface hydrophobicity. J. Membr. Sci. 2014, 449, 58–66. [Google Scholar] [CrossRef]
- Jeon, S.; Rajabzadeh, S.; Okamura, R.; Ishigami, T.; Hasegawa, S.; Kato, N.; Matsuyama, H. The effect of membrane material and surface pore size on the fouling properties of submerged membranes. Water 2016, 8, 602. [Google Scholar] [CrossRef]
- Zhao, C.; Xu, X.; Chen, J.; Wang, G.; Yang, F. Highly effective antifouling performance of pvdf/graphene oxide composite membrane in membrane bioreactor (mbr) system. Desalination 2014, 340, 59–66. [Google Scholar] [CrossRef]
- Boributh, S.; Chanachai, A.; Jiraratananon, R. Modification of pvdf membrane by chitosan solution for reducing protein fouling. J. Membr. Sci. 2009, 342, 97–104. [Google Scholar] [CrossRef]
- Turkish Republic, Ministry of Forestry and Water Management. Atıksu Arıtma Tesisleri Teknik Usuller Tebliği. Available online: http://www.resmigazete.gov.tr/eskiler/2010/03/20100320-7.htm (accessed on 16 March 2017). (In Turkish)
- Luo, W.; Phan, H.V.; Hai, F.I.; Price, W.E.; Guo, W.; Ngo, H.H.; Yamamoto, K.; Nghiem, L.D. Effects of salinity build-up on the performance and bacterial community structure of a membrane bioreactor. Bioresour. Technol. 2016, 200, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Jang, D.; Hwang, Y.; Shin, H.; Lee, W. Effects of salinity on the characteristics of biomass and membrane fouling in membrane bioreactors. Bioresour. Technol. 2013, 141, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Lü, Z.; Chen, Z.; Yu, S.; Gao, C. Comparison of reverse osmosis and nanofiltration membranes in the treatment of biologically treated textile effluent for water reuse. Desalination 2011, 281, 372–378. [Google Scholar] [CrossRef]
- Bes-Piá, A.; Iborra-Clar, A.; García-Figueruelo, C.; Barredo-Damas, S.; Alcaina-Miranda, M.; Mendoza-Roca, J.; Iborra-Clar, M. Comparison of three nf membranes for the reuse of secondary textile effluents. Desalination 2009, 241, 1–7. [Google Scholar] [CrossRef]
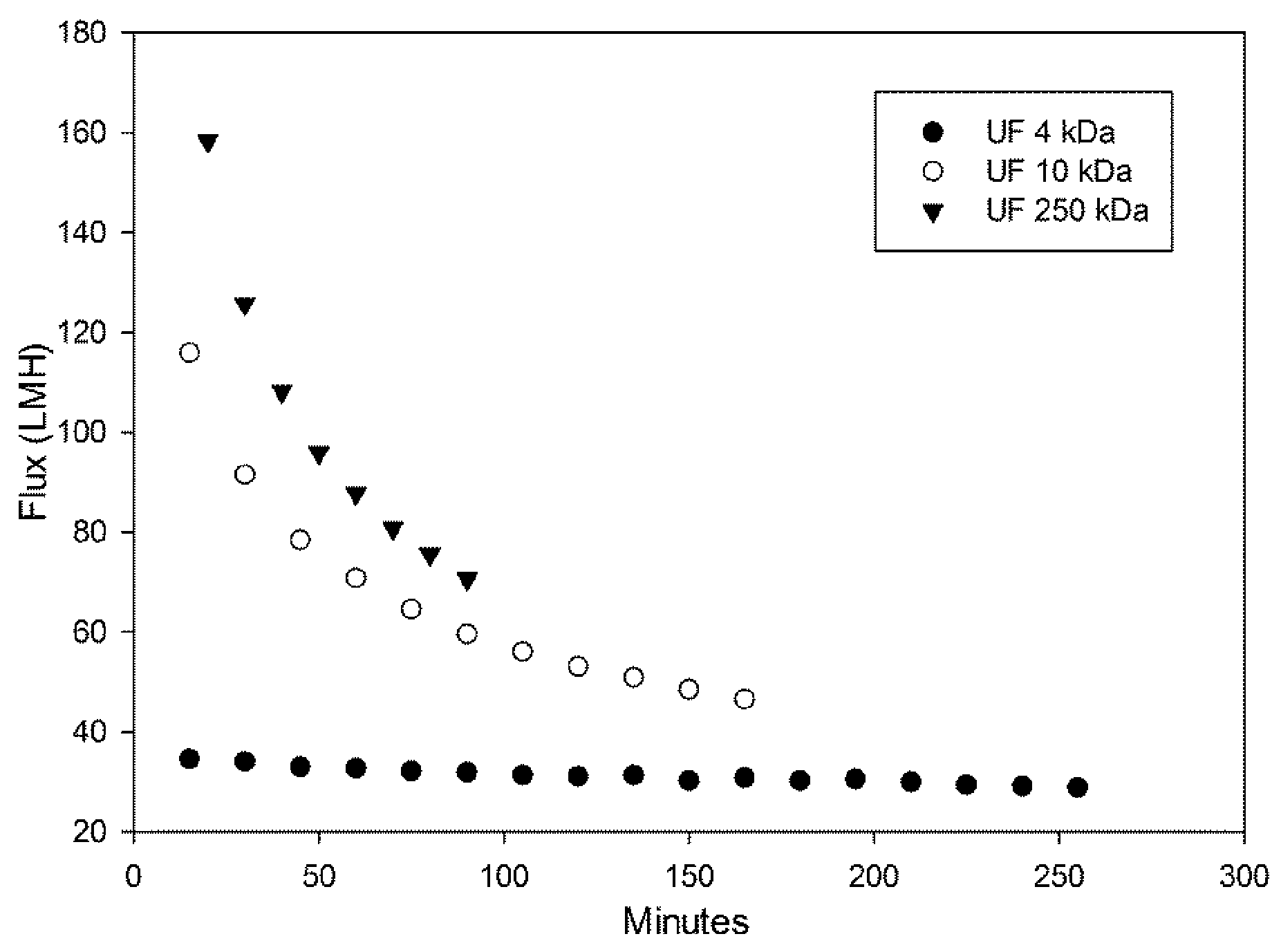
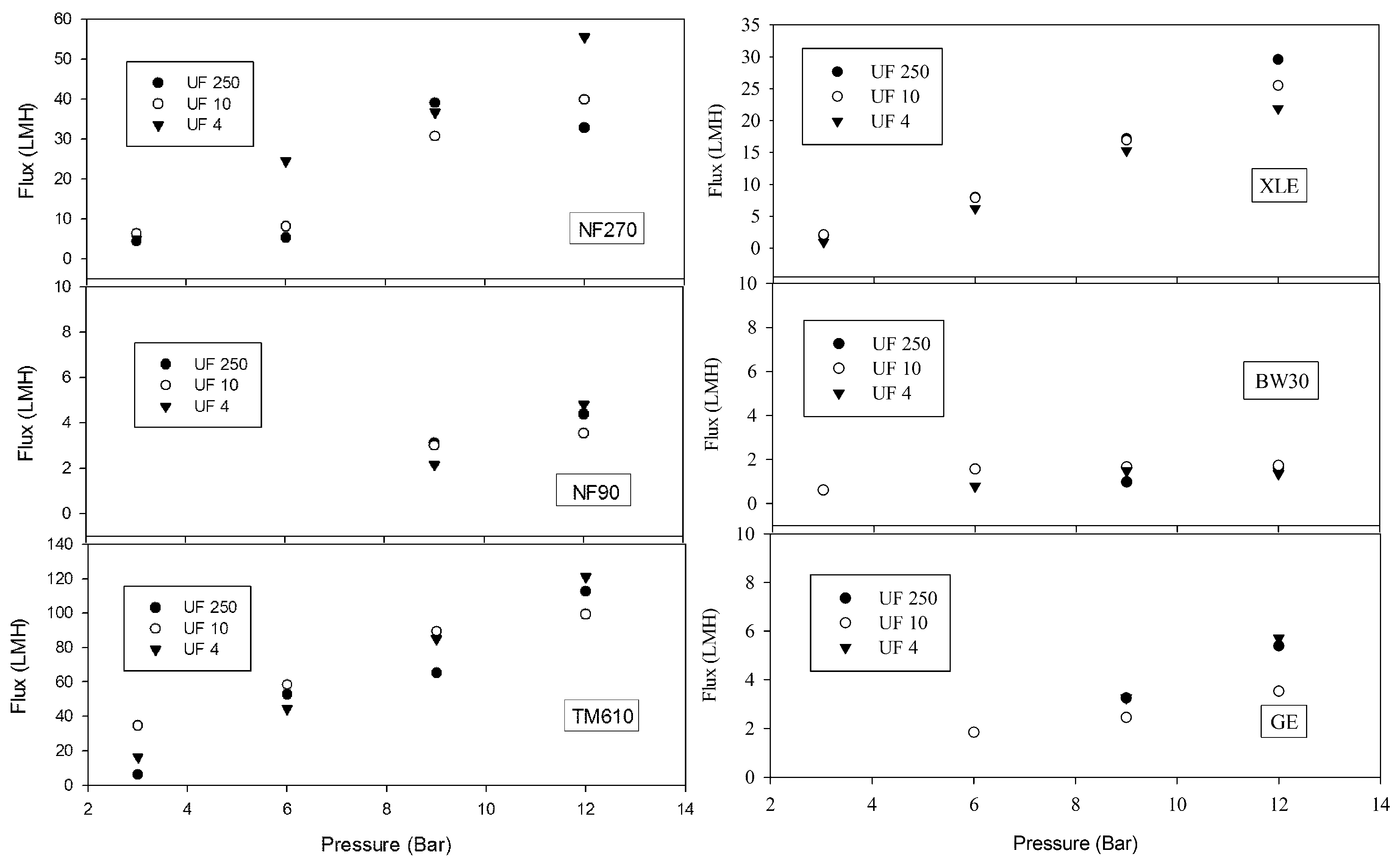
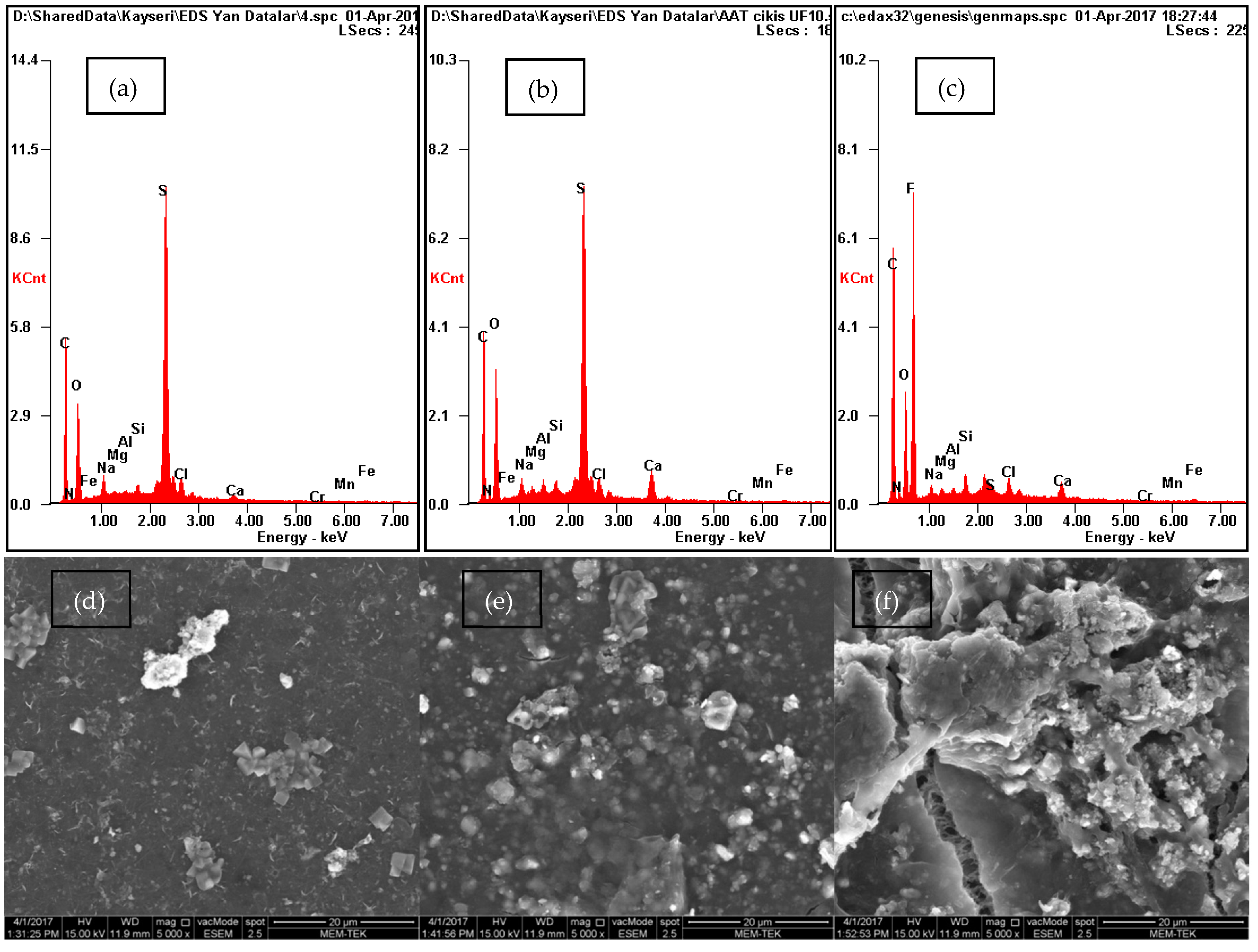
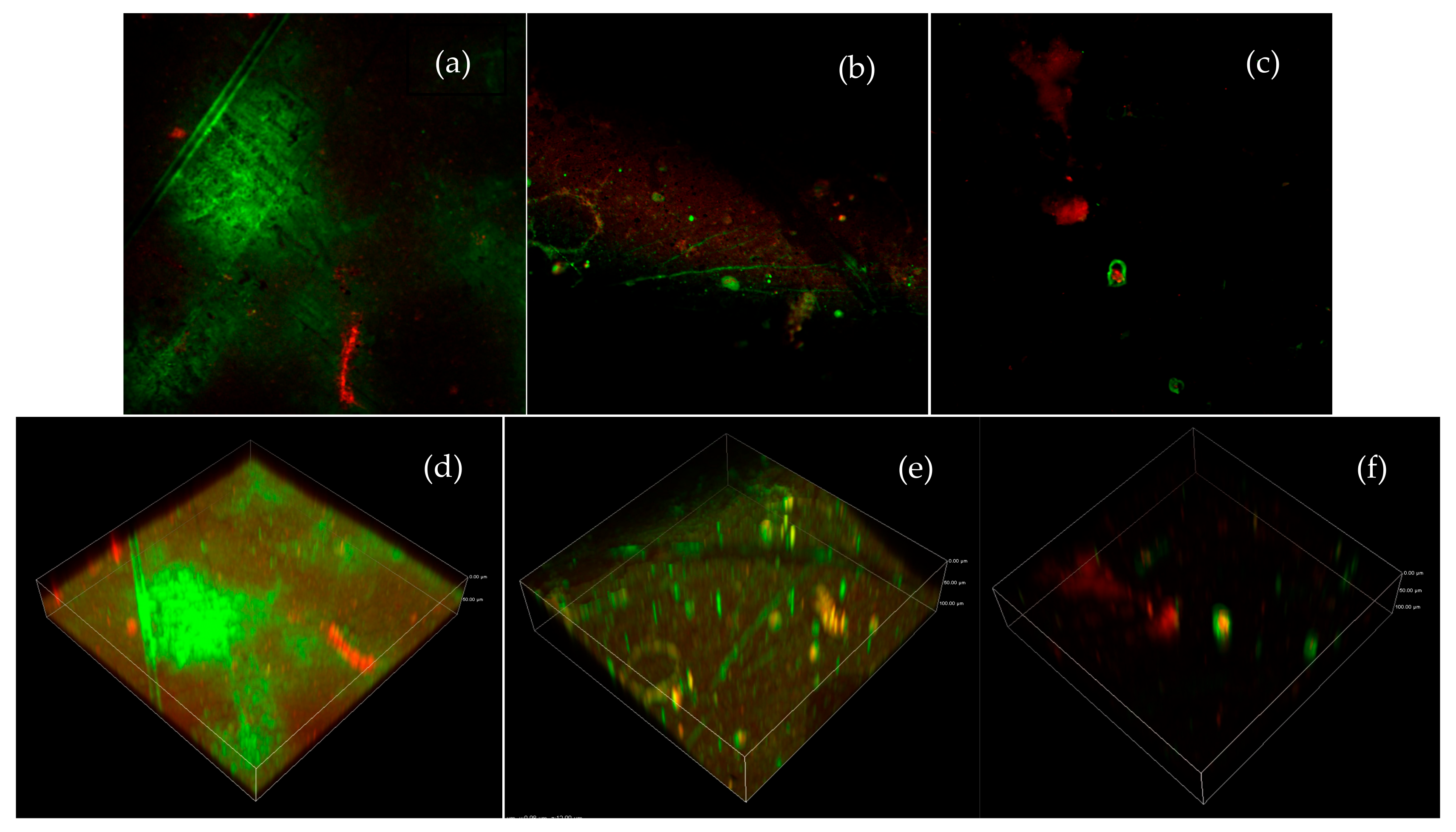
| Parameter | Influent | Effluent |
|---|---|---|
| COD (mg/L) | 1233 ± 67.4 | 81.9 ± 7.4 |
| SS (mg/L) | 1189 ± 90.2 | 28.6 ± 3.1 |
| TN (mg/L) | 27.1 ± 9.3 | 2.9 ± 1.0 |
| TP (mg/L) | 4.4 ± 1.1 | 0.1 ± 0.08 |
| EC (ms/cm) | 6.05 ± 0.9 | 5.44 ± 1.3 |
| Cu (mg/L) | 4.8 ± 0.79 | 0.2 ± 0.03 |
| Zn (mg/L) | 1.3 ± 0.31 | 0.2 ± 0.03 |
| Pb (mg/L) | 0.9 ±0.25 | 0.2 ± 0.06 |
| Fe (mg/L) | 13.9 ± 2.96 | 0.7 ± 0.23 |
| Cr6+ (mg/L) | 1.0 ± 0.22 | 0.0 ± 0.00 |
| Total Cr (mg/L) | 1.6 ± 0.22 | 0.1 ± 0.22 |
| Cd (mg/L) | 0.0 ± 0.00 | 0.0 ± 0.00 |
| Membrane Name | Pore Size (µ) | MWCO (kDa) | Material | Brand |
|---|---|---|---|---|
| UF 4 kDa | 0.07 | 4 | PES | Philos |
| UF 10 kDa | 0.10 | 10 | PES | Philos |
| UF 250 kDa | 0.44 | 250 | PVDF | Philos |
| Membranes | Membrane Material | Max. Oper. Temp. (°C) | Max. Oper. Pres. (Bar) | pH Range | MWCO (Da) | Salt Rejection (%) |
|---|---|---|---|---|---|---|
| NF90 | PA | 35 | 41 | 4–11 | 201 ± 25 | 85–95 |
| NF270 | PA | 45 | 35 | 3–10 | 330 ± 48 | 40–60 |
| TM610 | PA | 45 | 60 | 2–11 | 100 | 80–97 |
| BW30 | PA | 45 | 41 | 2–11 | 116 ± 30 | 99.5 |
| XLE | PA | 45 | 41 | - | - | 95 |
| AD90 | PA | 50 | 82 | 2–11 | - | 99.75 |
| Membrane | pH | COD (mg/L) | EC (ms/cm) | Turbidity (NTU) | Flux (LMH) |
|---|---|---|---|---|---|
| UF 4 kDa | 8.61 ± 0.36 | 73.5 ± 16.4 | 4.21 ± 0.25 | 0.85 ± 0.12 | 35.1 ± 11.5 |
| UF 10 kDa | 8.75 ± 0.47 | 83.0 ± 12.7 | 4.33 ± 0.21 | 0.88 ± 0.08 | 52.9 ± 4.1 |
| UF 250 kDa | 8.72 ± 0.25 | 46.9 ± 21.3 | 4.02 ± 0.33 | 1.10 ± 0.14 | 74.2 ± 5.1 |
| Membranes | EC (ms/cm) |
|---|---|
| NF90 | 1.77 ± 0.17 |
| NF270 | 3.73 ± 0.27 |
| NF-TM610 | 4.20 ± 0.23 |
| RO-XLE | 0.83 ± 0.47 |
| RO-AD90 | 1.17 ± 0.47 |
| RO-BW30 | 1.13 ± 0.57 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uyanık, İ.; Özkan, O.; Koyuncu, İ. NF-RO Membrane Performance for Treating the Effluent of an Organized Industrial Zone Wastewater Treatment Plant: Effect of Different UF Types. Water 2017, 9, 506. https://doi.org/10.3390/w9070506
Uyanık İ, Özkan O, Koyuncu İ. NF-RO Membrane Performance for Treating the Effluent of an Organized Industrial Zone Wastewater Treatment Plant: Effect of Different UF Types. Water. 2017; 9(7):506. https://doi.org/10.3390/w9070506
Chicago/Turabian StyleUyanık, İbrahim, Oktay Özkan, and İsmail Koyuncu. 2017. "NF-RO Membrane Performance for Treating the Effluent of an Organized Industrial Zone Wastewater Treatment Plant: Effect of Different UF Types" Water 9, no. 7: 506. https://doi.org/10.3390/w9070506
APA StyleUyanık, İ., Özkan, O., & Koyuncu, İ. (2017). NF-RO Membrane Performance for Treating the Effluent of an Organized Industrial Zone Wastewater Treatment Plant: Effect of Different UF Types. Water, 9(7), 506. https://doi.org/10.3390/w9070506




