Effects of N/S Molar Ratio on Product Formation in Psychrophilic Autotrophic Biological Removal of Sulfide
Abstract
:1. Introduction
2. Materials and Methods
2.1. Inoculum and Enrichment
2.2. Synthetic Wastewater
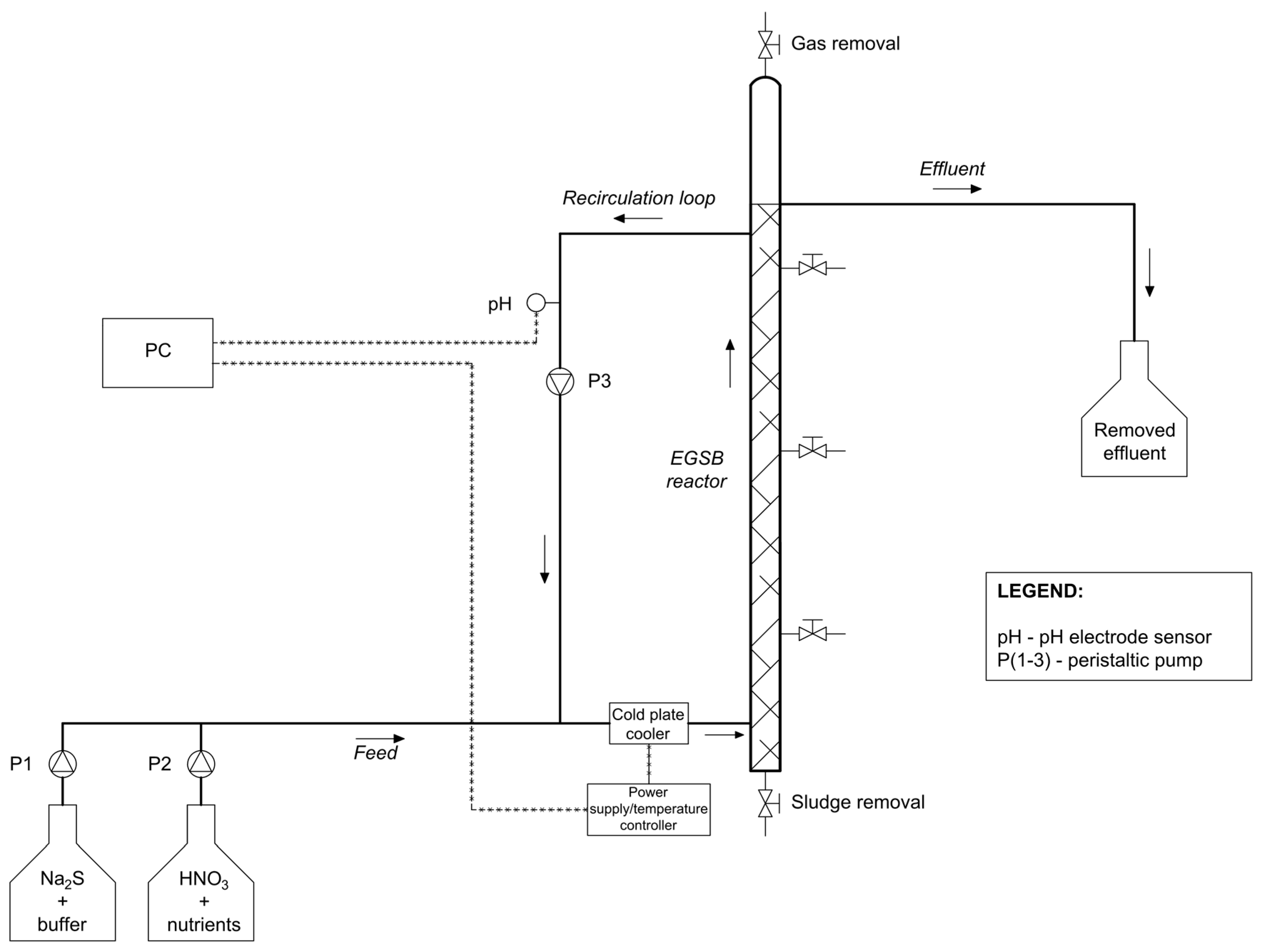
2.3. Experimental Setup
2.4. Analytical Procedure
2.5. Elemental Sulfur Measurements
3. Results and Discussion
3.1. Reactor Performance
3.2. Sulfur Components at Different N/S Ratios
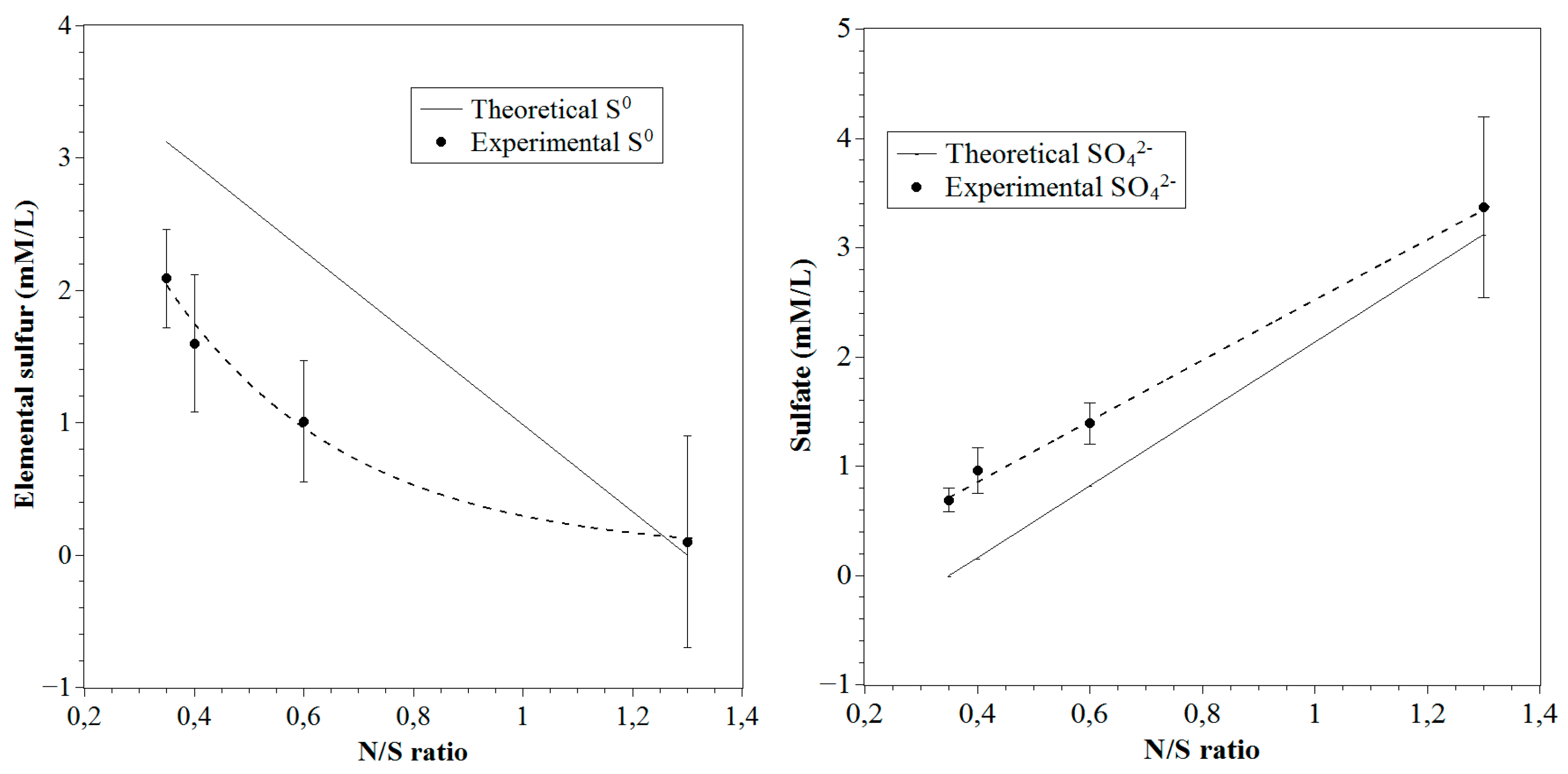
3.3. Relation between Experimental and Theoretical Products Distribution
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pokorna, D.; Zabranska, J. Sulfur-oxidizing bacteria in environmental technology. Biotechnol. Adv. 2015, 33, 1246–1259. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Cheng, J.J.; Creamer, K.S. Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 2008, 99, 4044–4064. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Yu, Z.; Meng, Q. Effects of nitrate on methane production, fermentation, and microbial populations in in vitro ruminal cultures. Bioresour. Technol. 2012, 103, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Knoblauch, C.; Sahm, K.; Jørgensen, B.B. Psychrophilic sulfate-reducing bacteria isolated from permanently cold Arctic marine sediments: Description of Desulfofrigus oceanense gen. nov., sp. nov., Desulfofrigus fragile sp. nov., Desulfofaba gelida gen. nov., sp. nov., Desulfotalea psychrophila gen. nov., sp. nov. and Desulfotalea arctica sp. nov. Int. J. Syst. Evol. Microbiol. 1999, 49, 1631–1643. [Google Scholar]
- Auguet, O.; Pijuan, M.; Borrego, C.M.; Gutierrez, O. Control of sulfide and methane production in anaerobic sewer systems by means of Downstream Nitrite Dosage. Sci. Total Environ. 2016, 550, 1116–1125. [Google Scholar] [CrossRef] [PubMed]
- Kleerebezem, R.; Mendez, R. Autotrophic denitrification for combined hydrogen sulfide removal from biogas and post-denitrification. Water Sci. Technol. 2002, 45, 349–356. [Google Scholar] [PubMed]
- Cai, J.; Zheng, P.; Mahmood, Q. Effect of sulfide to nitrate ratios on the simultaneous anaerobic sulfide and nitrate removal. Bioresour. Technol. 2008, 99, 5520–5527. [Google Scholar] [CrossRef] [PubMed]
- Fajardo, C.; Mora, M.; Fernández, I.; Mosquera-Corral, A.; Campos, J.L.; Méndez, R. Cross effect of temperature, pH and free ammonia on autotrophic denitrification process with sulphide as electron donor. Chemosphere 2014, 97, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Beristain-Cardoso, R.; Sierra-Alvarez, R.; Rowlette, P.; Flores, E.R.; Gómez, J.; Field, J.A. Sulfide oxidation under chemolithoautotrophic denitrifying conditions. Biotechnol. Bioeng. 2006, 95, 1148–1157. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Li, Z.; Chen, F.; Liu, Q.; Zhao, Y.; Gao, L.; Chen, C.; Zhou, J.; Wang, A. Efficient regulation of elemental sulfur recovery through optimizing working height of upflow anaerobic sludge blanket reactor during denitrifying sulfide removal process. Bioresour. Technol. 2016, 200, 1019–1023. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, Q.; Zheng, P.; Cai, J.; Wu, D.; Hu, B.; Li, J. Anoxic sulfide biooxidation using nitrite as electron acceptor. J. Hazard. Mater. 2007, 147, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Chen, N.; Feng, C.; Hao, C.; Peng, T. Sulfur-based autotrophic denitrification with eggshell for nitrate-contaminated synthetic groundwater treatment. Environ. Technol. 2016, 37, 3094–3103. [Google Scholar] [CrossRef] [PubMed]
- Di Capua, F.; Milone, I.; Lakaniemi, A.M.; Lens, P.N.L.; Esposito, G. High-rate autotrophic denitrification in a fluidized-bed reactor at psychrophilic temperatures. Chem. Eng. J. 2017, 313, 591–598. [Google Scholar] [CrossRef]
- Yamamoto-Ikemoto, R.; Komori, T.; Nomuri, M.; Ide, Y.; Matsukami, T. Nitrogen removal from hydroponic culture wastewater by autotrophic denitrification using thiosulfate. Water Sci. Technol. 2000, 42, 369–376. [Google Scholar]
- Sposob, M.; Bakke, R.; Dinamarca, C. Metabolic divergence in simultaneous biological removal of nitrate and sulfide for elemental sulfur production under temperature stress. Bioresour. Technol. 2017, 233, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Wolin, E.A.; Wolin, M.J.; Wolfe, R.S. Formation of methane by bacterial extracts. J. Biol. Chem. 1963, 238, 2882–2886. [Google Scholar] [PubMed]
- Oh, S.E.; Kim, K.S.; Choi, H.C.; Cho, J.; Kim, I.S. Kinetics and physiological characteristics of autotrophic dentrification by denitrifying sulfur bacteria. Water Sci. Technol. 2000, 42, 59–68. [Google Scholar]
- Carroll, J.J.; Mather, A.E. The solubility of hydrogen sulphide in water from 0 to 90 °C and pressures to 1 MPa. Geochim. Cosmochim. Acta 1989, 53, 1163–1170. [Google Scholar] [CrossRef]
- An, S.; Tang, K.; Nemati, M. Simultaneous biodesulphurization and denitrification using an oil reservoir microbial culture: Effects of sulphide loading rate and sulphide to nitrate loading ratio. Water Res. 2010, 44, 1531–1541. [Google Scholar] [CrossRef] [PubMed]
- Sposob, M.; Dinamarca, C.; Bakke, R. Short-term temperature impact on simultaneous biological nitrogen-sulphur treatment in EGSB reactor. Water Sci. Technol. 2016, 74, 1610–1618. [Google Scholar] [CrossRef] [PubMed]
- Schedel, M.; Trüper, H.G. Anaerobic oxidation of thiosulfate and elemental sulfur in Thiobacillus denitrificans. Arch. Microbiol. 1980, 124, 205–210. [Google Scholar] [CrossRef]
- Chen, C.; Xu, X.J.; Xie, P.; Yuan, Y.; Zhou, X.; Wang, A.J.; Lee, D.J.; Ren, N.Q. Pyrosequencing reveals microbial community dynamics in integrated simultaneous desulfurization and denitrification process at different influent nitrate concentrations. Chemosphere 2017, 171, 294–301. [Google Scholar] [CrossRef] [PubMed]
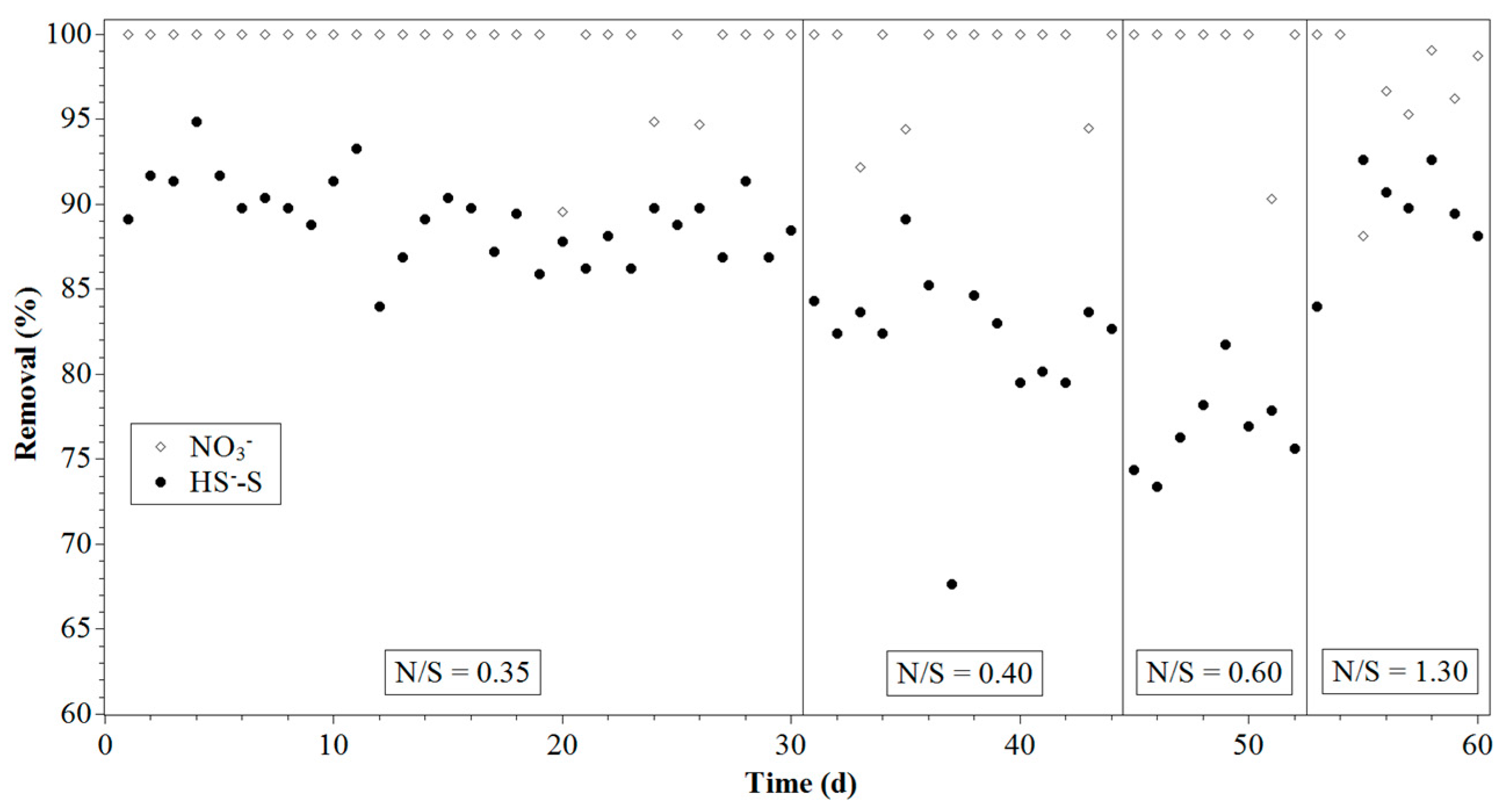
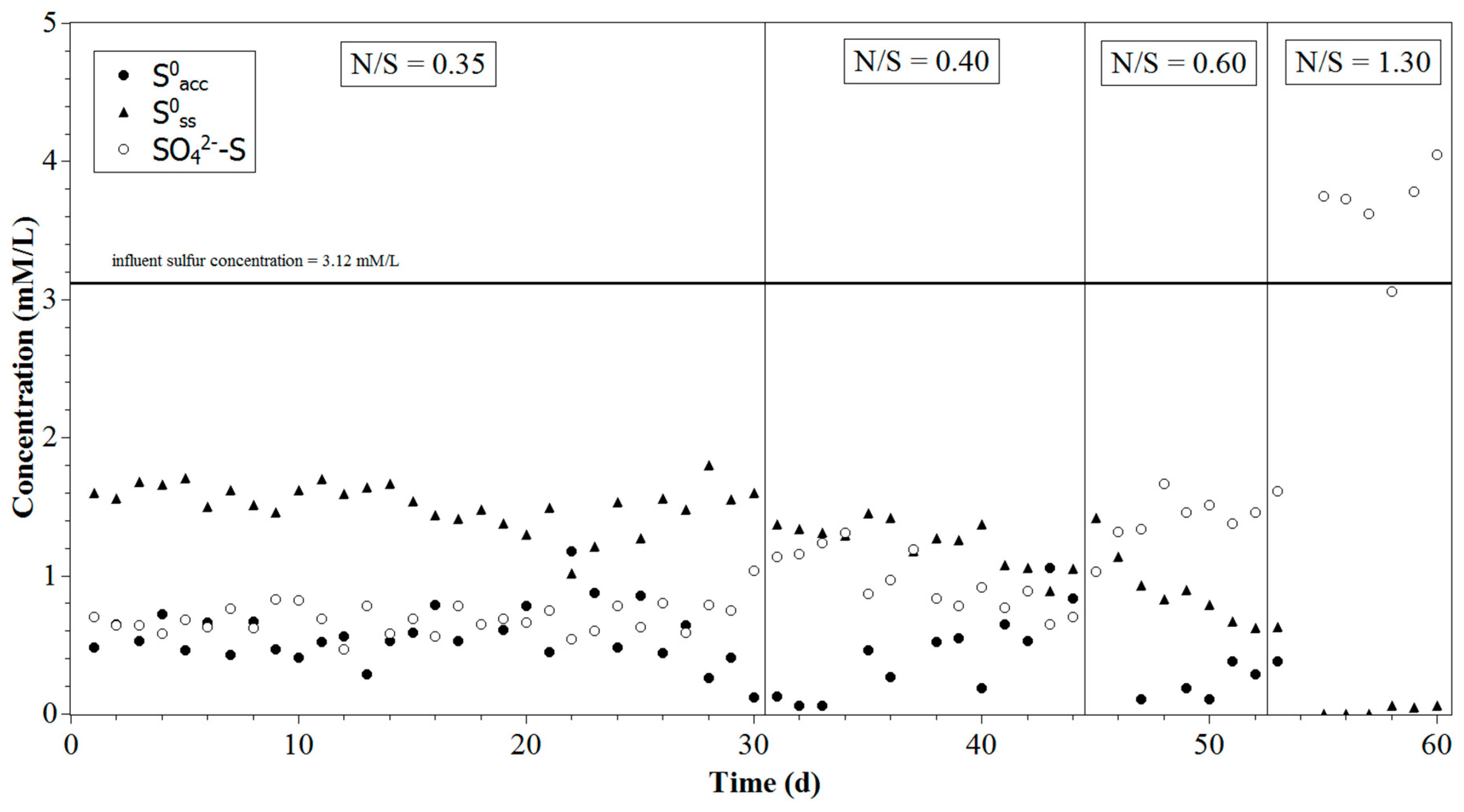
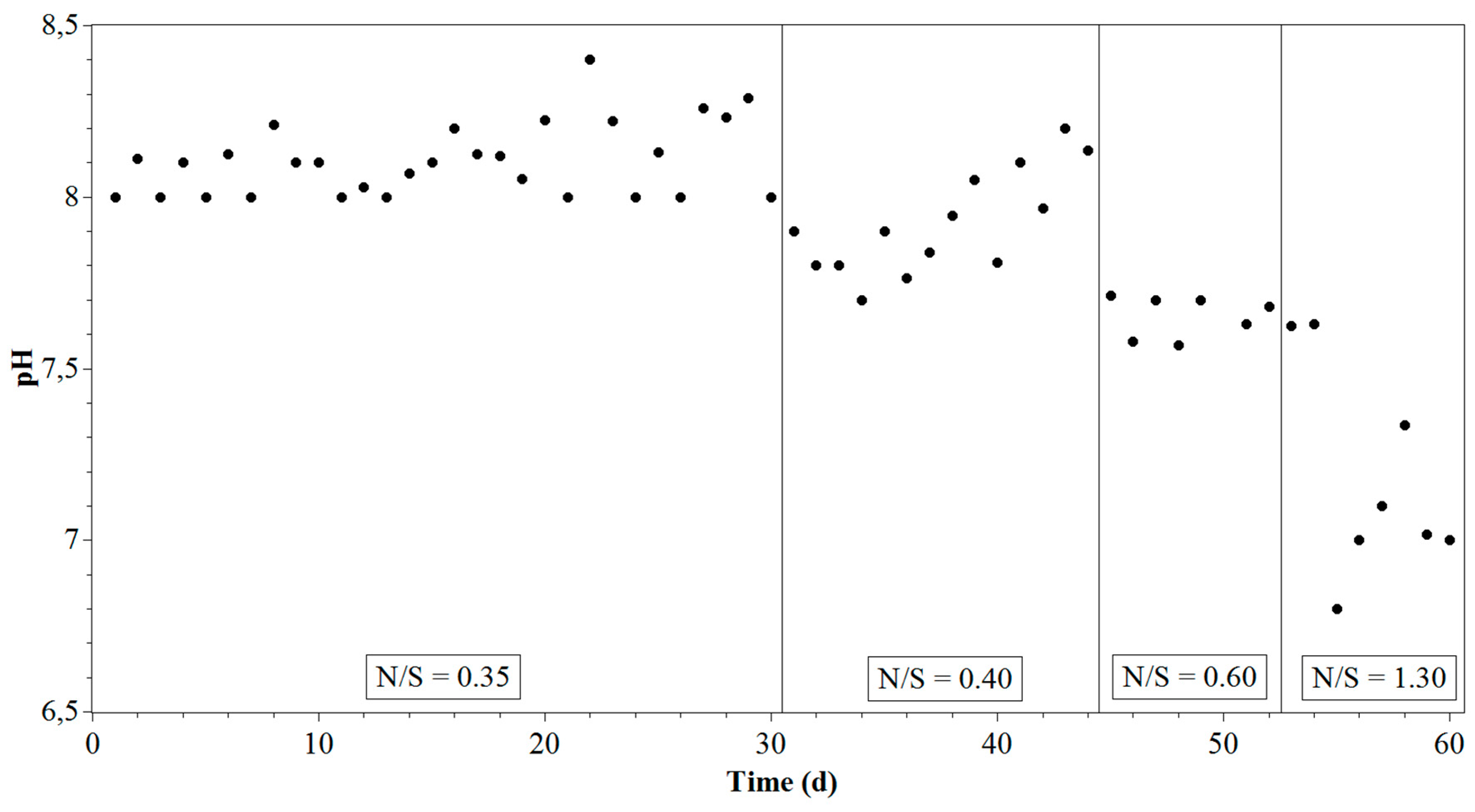

| Time (Day) | N/S Ratio | NO3− (mM/L) | HS− (mM/L) |
|---|---|---|---|
| 1–30 | 0.35 | 1.08 | 3.12 |
| 31–44 | 0.40 | 1.25 | |
| 45–52 | 0.60 | 1.87 | |
| 53–60 | 1.30 | 4.05 |
| N/S Ratio | S0acc 1 | SO42− | S0ss | HS−-S | NO3− | pH | Total Sulfur (Effluent) 2 |
|---|---|---|---|---|---|---|---|
| 0.35 | 0.57 ± 0.21 | 0.69 ± 0.11 | 1.52 ± 0.16 | 0.34 ± 0.07 | 0.01 ± 0.03 | 8.11 ± 0.11 | 2.55 ± 0.21 |
| 0.40 | 0.44 ± 0.32 | 0.96 ± 0.21 | 1.24 ± 0.16 | 0.56 ± 0.15 | 0.02 ± 0.03 | 7.92 ± 0.15 | 2.76 ± 0.36 |
| 0.60 | 0.09 ± 0.20 | 1.39 ± 0.19 | 0.91 ± 0.26 | 0.72 ± 0.08 | 0.02 ± 0.06 | 7.65 ± 0.06 | 3.03 ± 0.2 |
| 1.30 | −0.69 ± 0.58 | 3.37 ± 0.83 | 0.11 ± 0.23 | 0.32 ± 0.09 | 0.15 ± 0.16 | 7.19 ± 0.31 | 3.81 ± 0.58 |
| Reaction | ΔG° (1 M of Electron Donor) |
|---|---|
| HS− + 0.4NO3− + 1.4H+ → S0 + 0.2N2 + 1.2H2O | −252.13 |
| HS− + 0.8NO3− + 0.8H+ → 0.5S2O32− + 0.4N2 + 0.9H2O | −393.14 |
| HS− + 1.6NO3− + 0.6H+ → SO42− + 0.8N2 + 0.8H2O | −768.28 |
| S0 + 1.2NO3− + 0.4H2O → SO42− + 0.6N2 + 0.8H2O | −800.76 |
| N/S Ratio | Theoretical Share (%) | Experimental Share (%) | NO3− Uptake Share (%) | SO42− Produced by Another Electron Acceptor (mM/L) | |||
|---|---|---|---|---|---|---|---|
| S0 | SO42− | S0 | SO42− | S0 | SO42− | ||
| 0.35 | 100 | 0 | 67 | 22 | 67 | 33 | 0.41 (13%) 1 |
| 0.40 | 95 | 5 | 54 | 31 | 46 | 54 | 0.44 (14%) |
| 0.60 | 74 | 26 | 32 | 45 | 18 | 82 | 0.22 (7%) |
| 1.30 | 0 | 100 | 4 2 | 108 | 1 | 99 | 0.26 (8%) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sposob, M.; Bakke, R.; Dinamarca, C. Effects of N/S Molar Ratio on Product Formation in Psychrophilic Autotrophic Biological Removal of Sulfide. Water 2017, 9, 476. https://doi.org/10.3390/w9070476
Sposob M, Bakke R, Dinamarca C. Effects of N/S Molar Ratio on Product Formation in Psychrophilic Autotrophic Biological Removal of Sulfide. Water. 2017; 9(7):476. https://doi.org/10.3390/w9070476
Chicago/Turabian StyleSposob, Michal, Rune Bakke, and Carlos Dinamarca. 2017. "Effects of N/S Molar Ratio on Product Formation in Psychrophilic Autotrophic Biological Removal of Sulfide" Water 9, no. 7: 476. https://doi.org/10.3390/w9070476
APA StyleSposob, M., Bakke, R., & Dinamarca, C. (2017). Effects of N/S Molar Ratio on Product Formation in Psychrophilic Autotrophic Biological Removal of Sulfide. Water, 9(7), 476. https://doi.org/10.3390/w9070476





