A Methodology to Model Environmental Preferences of EPT Taxa in the Machangara River Basin (Ecuador)
Abstract
:1. Introduction
2. Materials and Methods
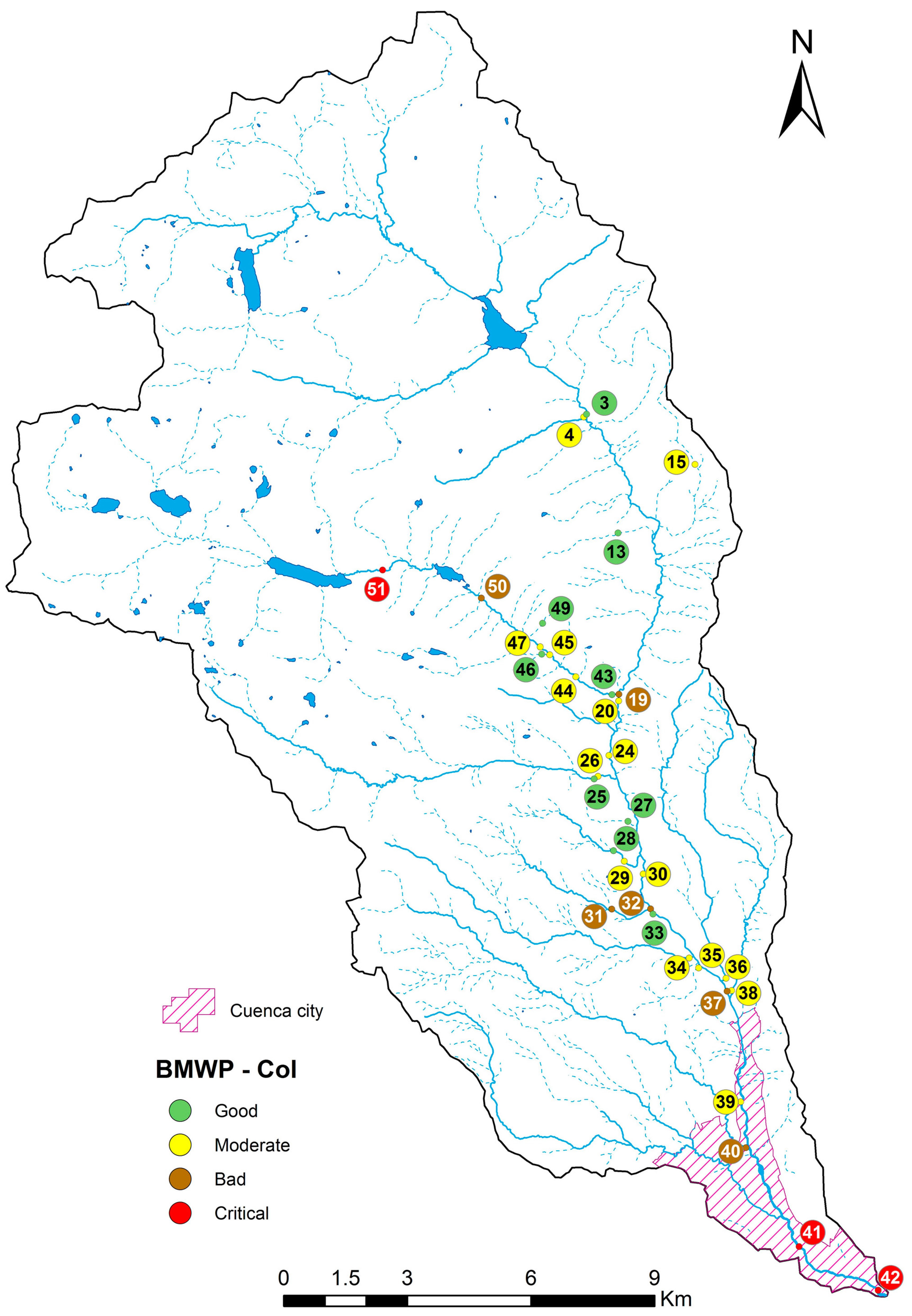
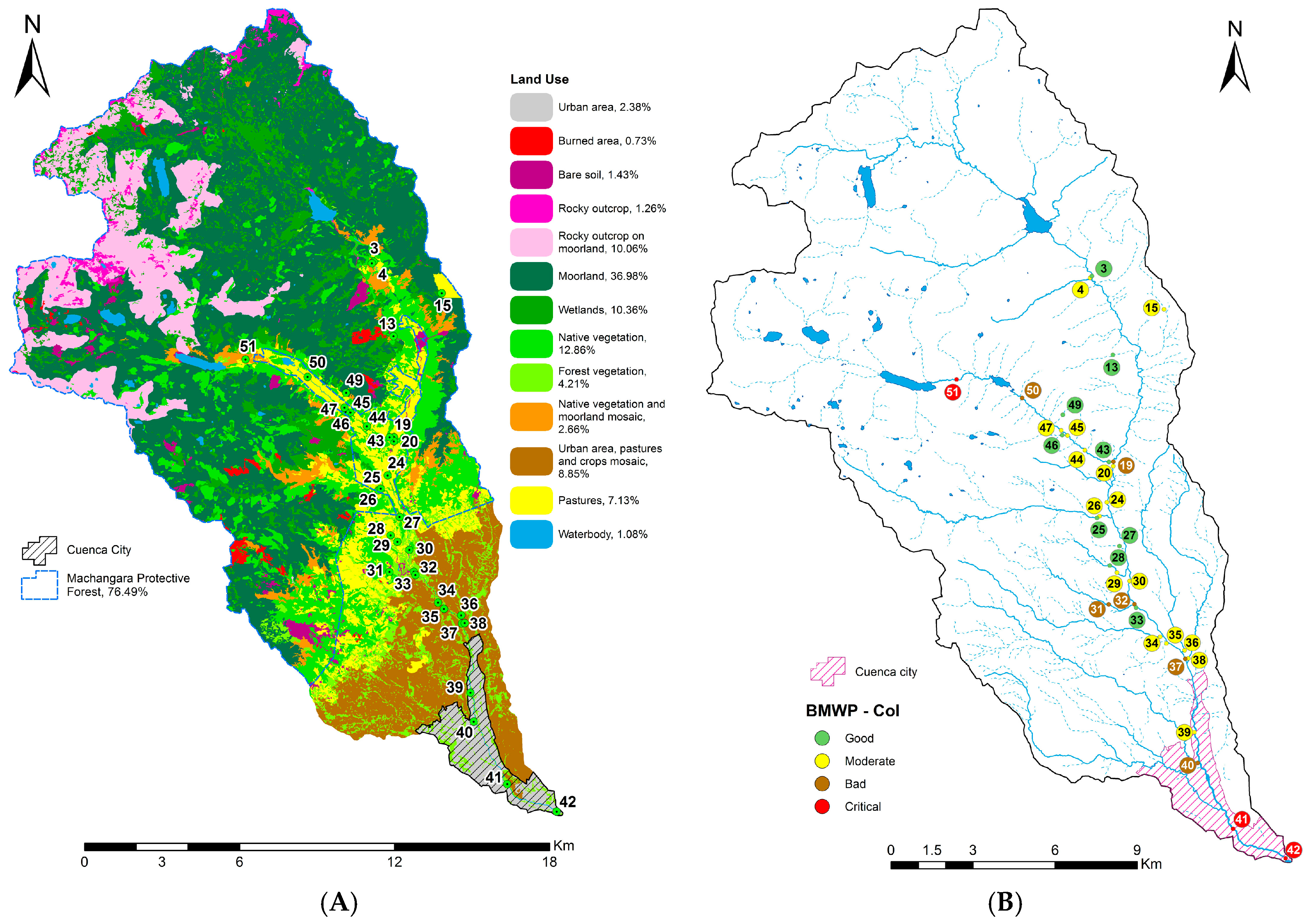
2.1. River Basin
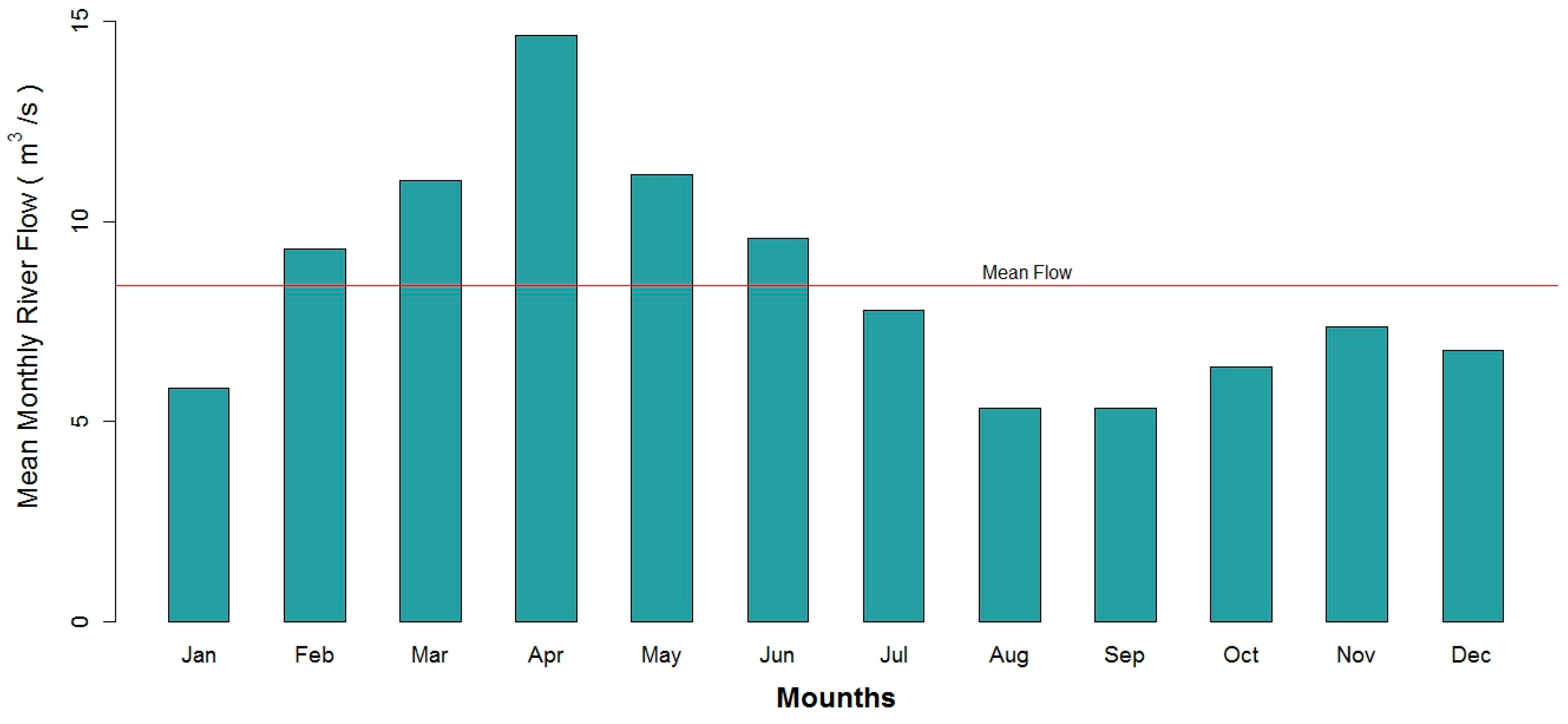
2.2. Data Collection
2.3. Model Species
2.4. Model Development, Selection, Validation and Optimization
3. Results
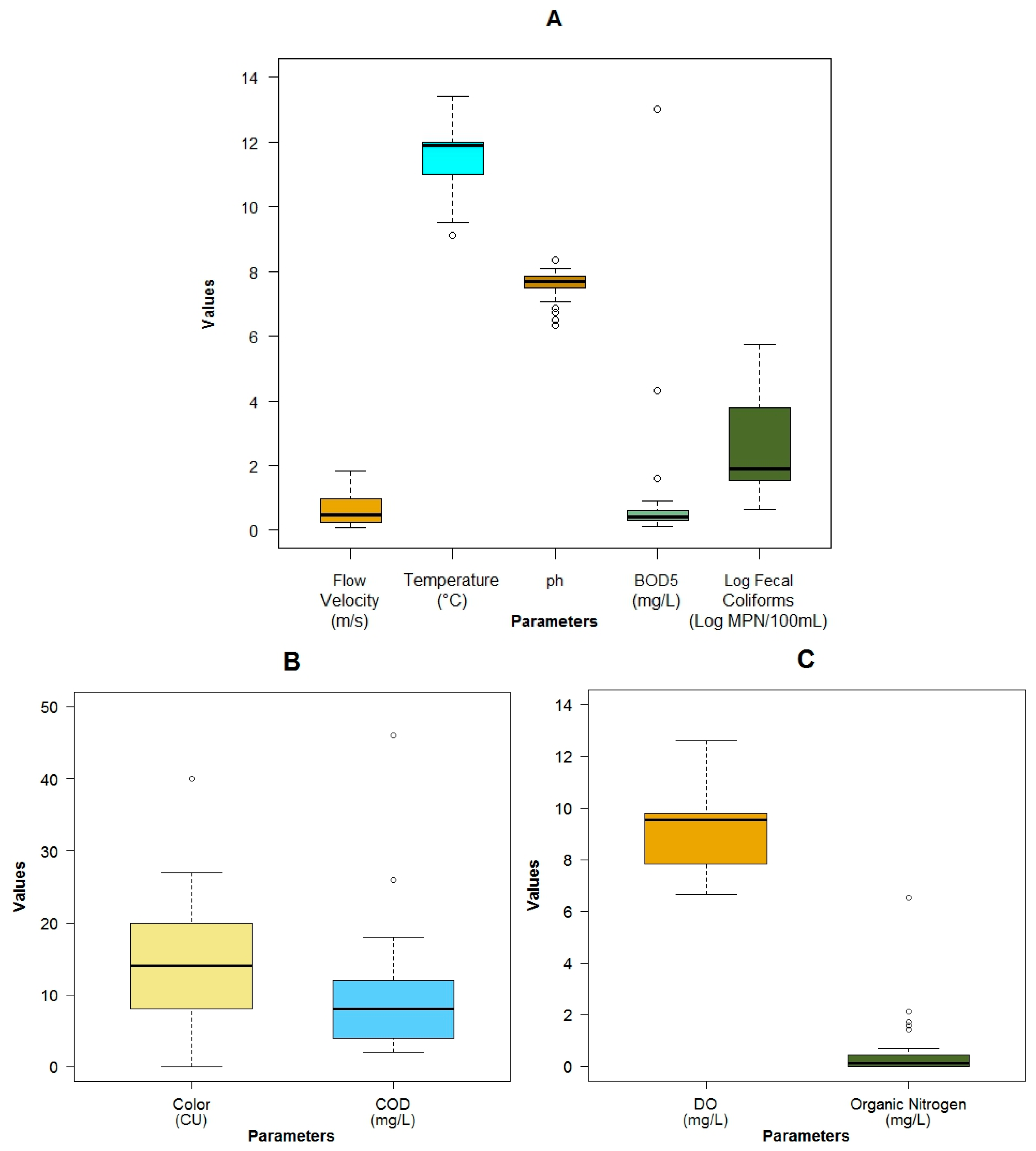
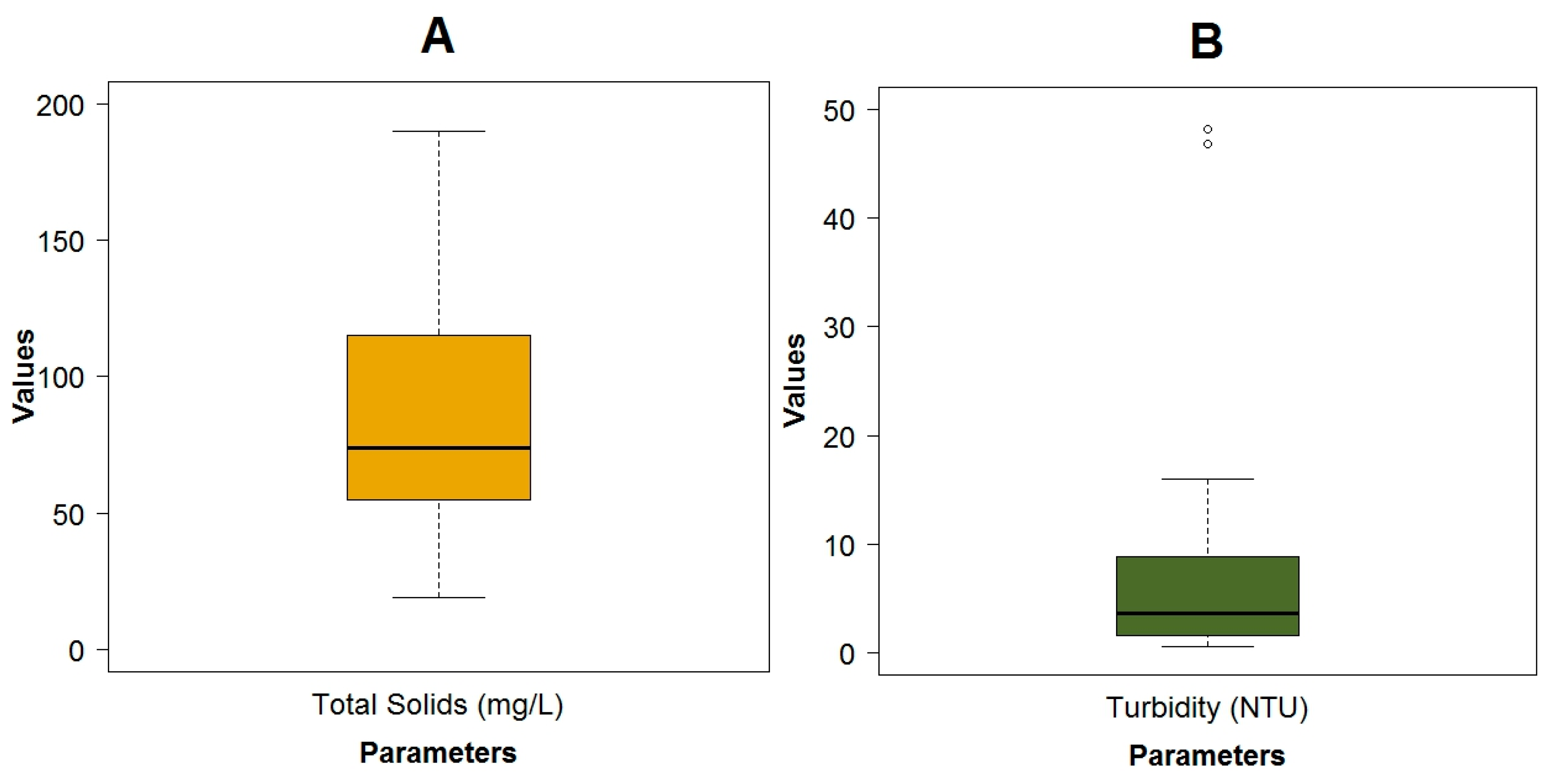
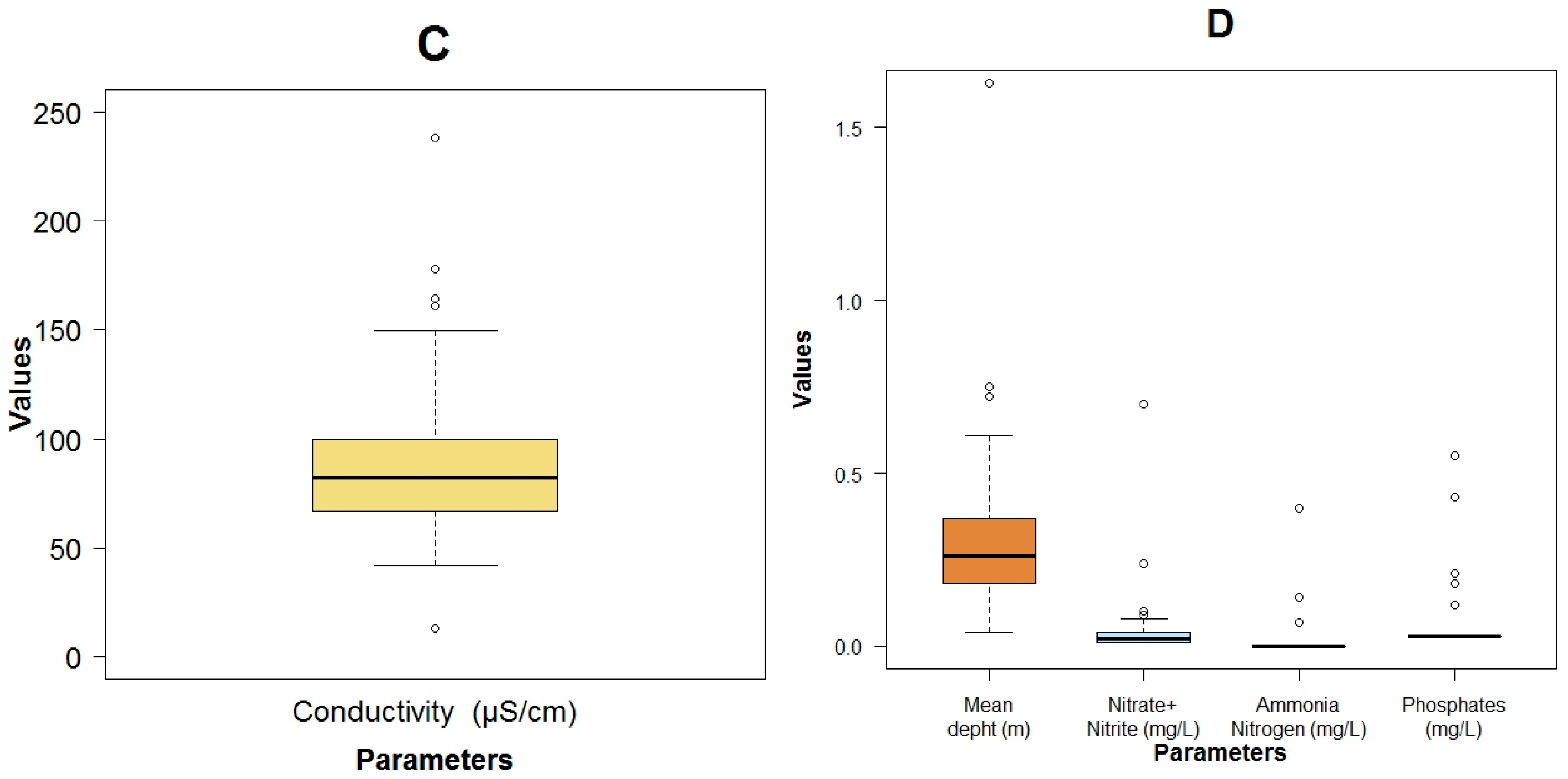
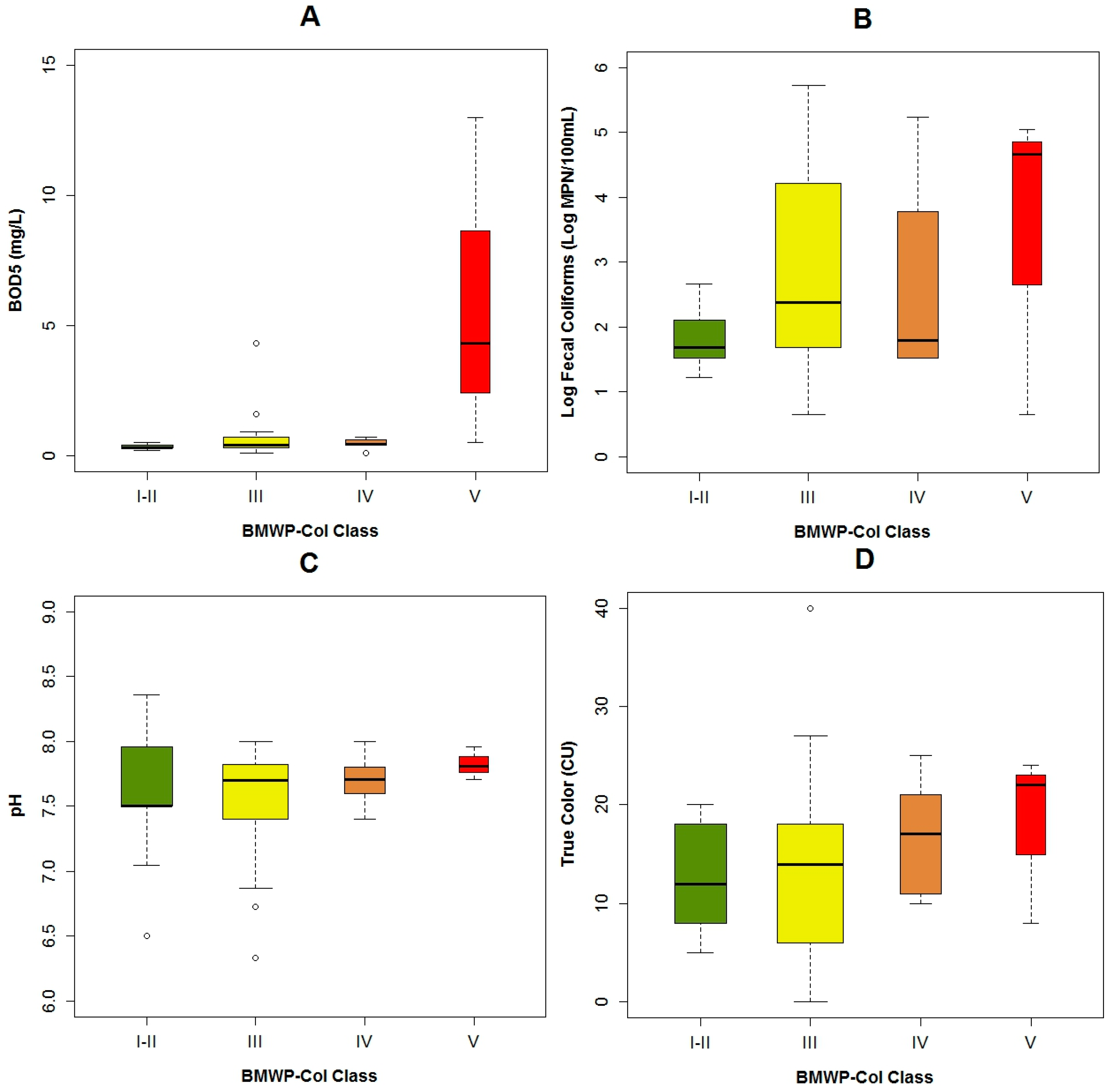
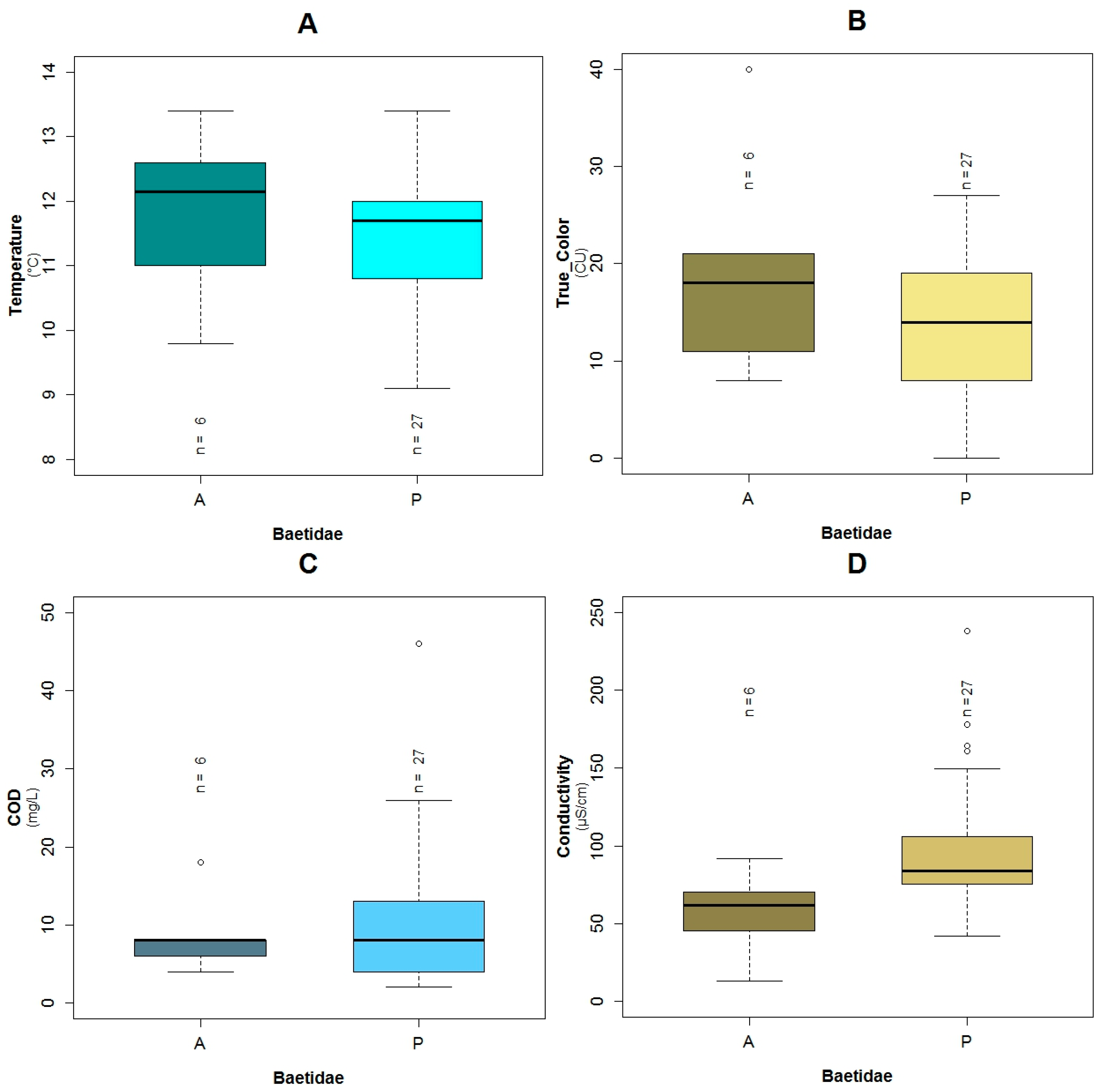
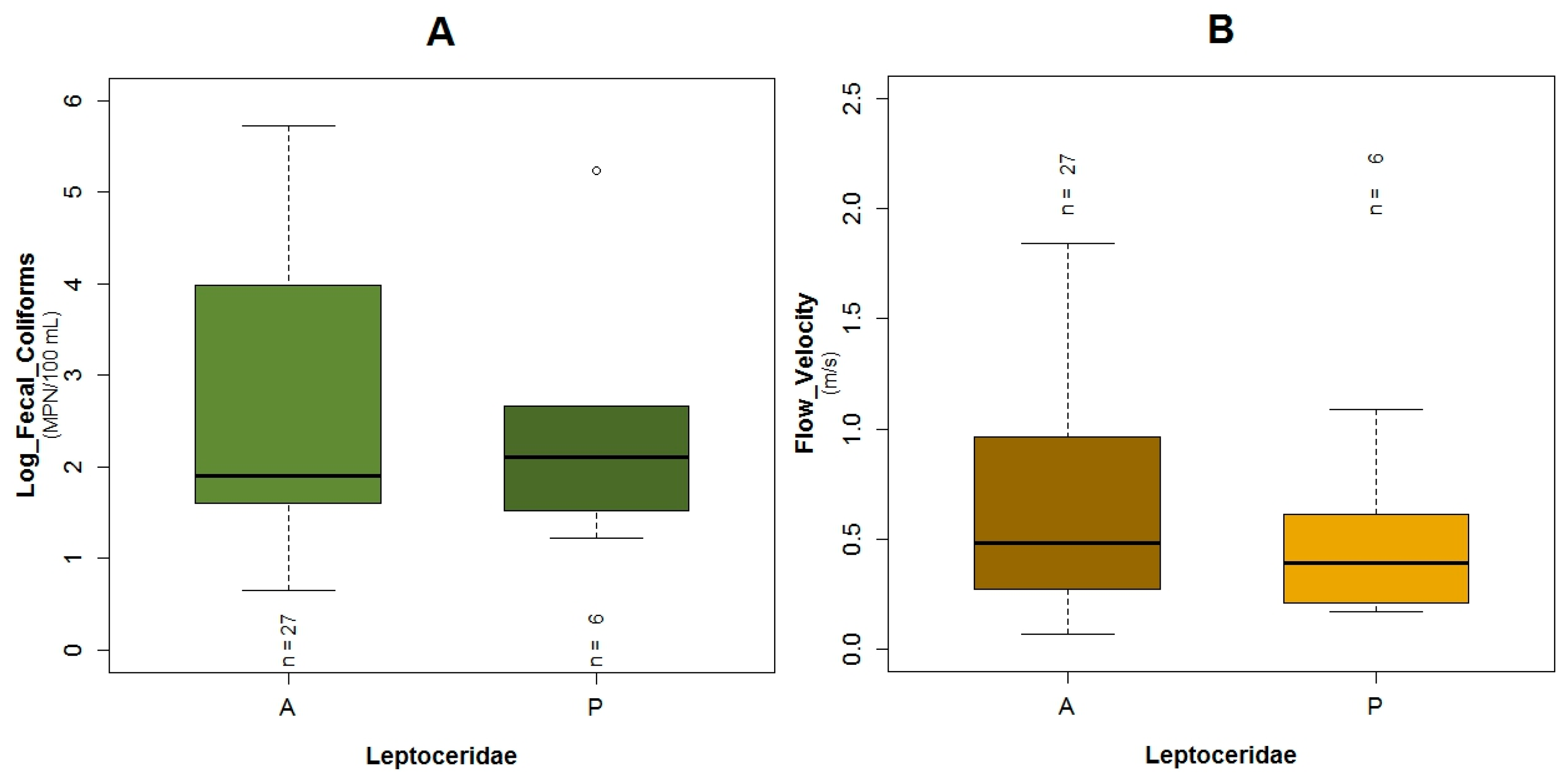
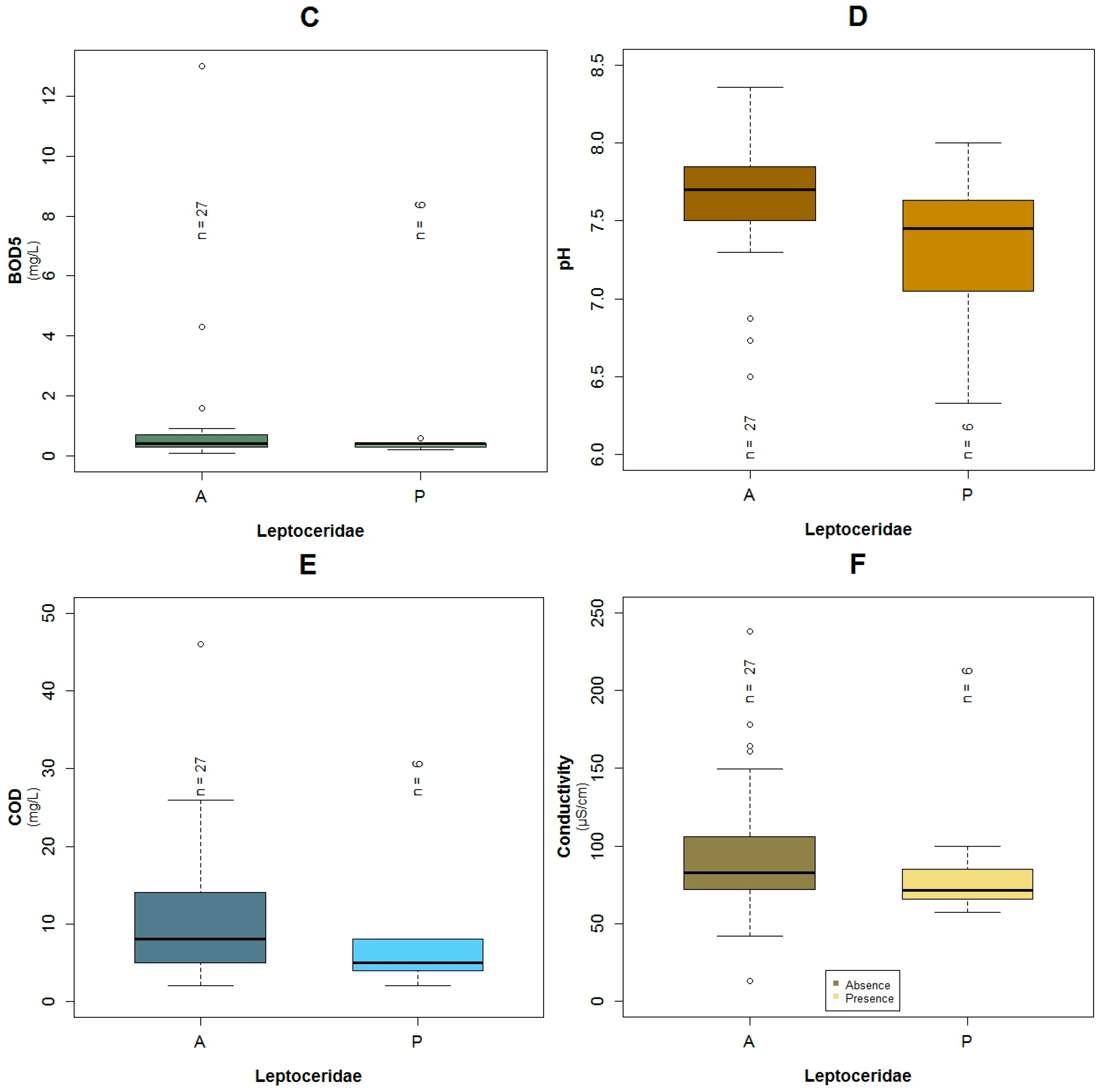
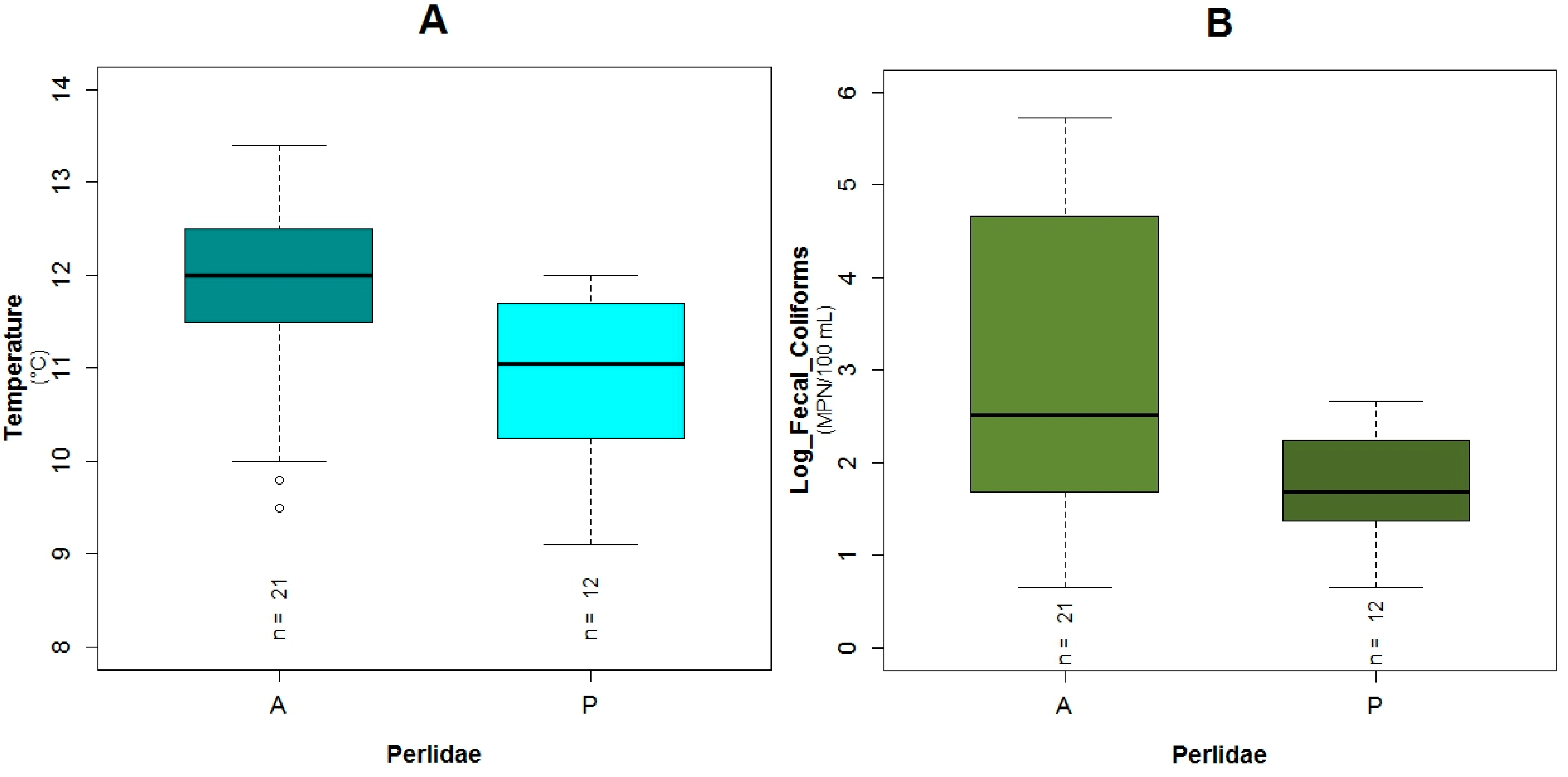
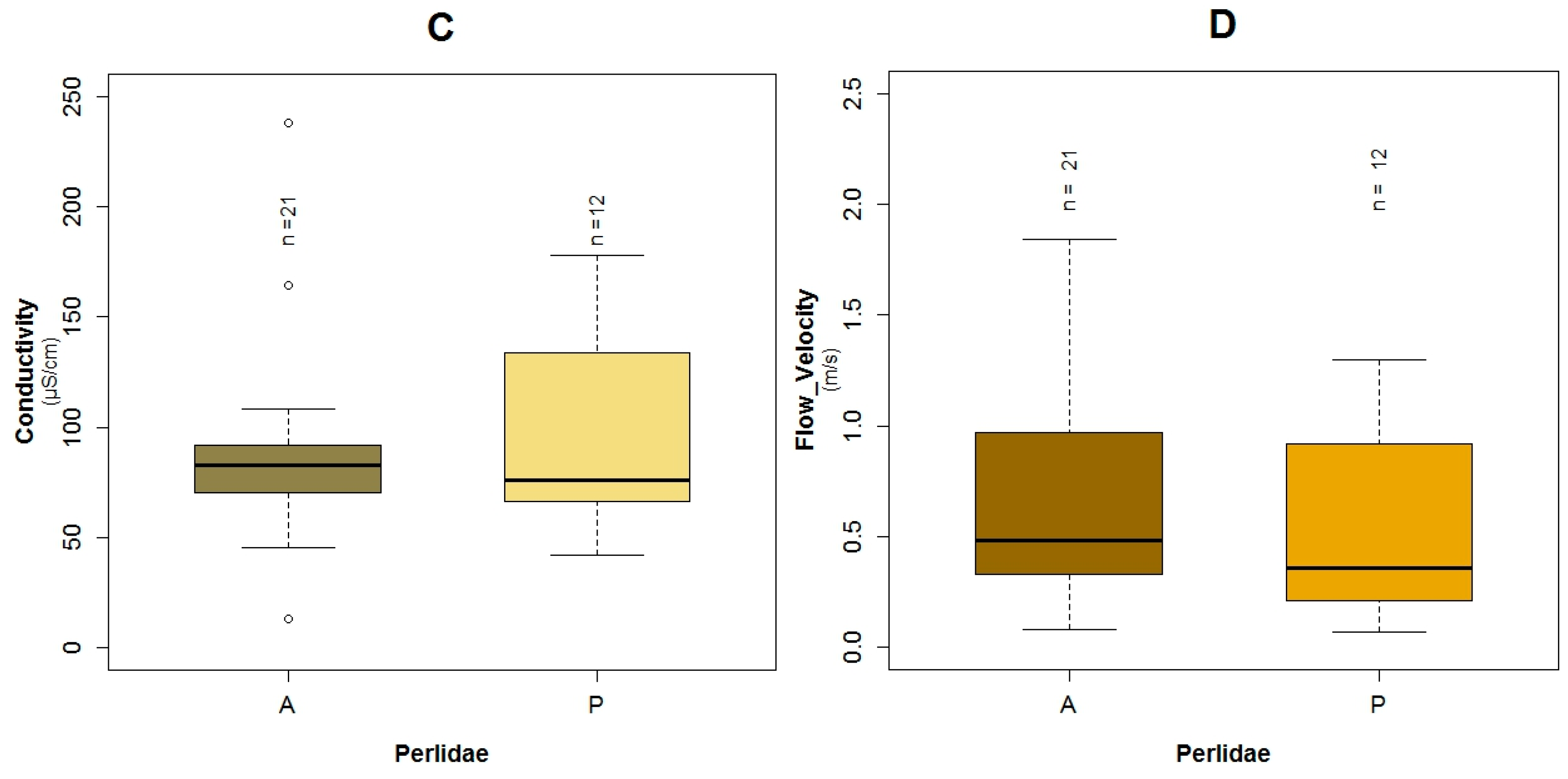
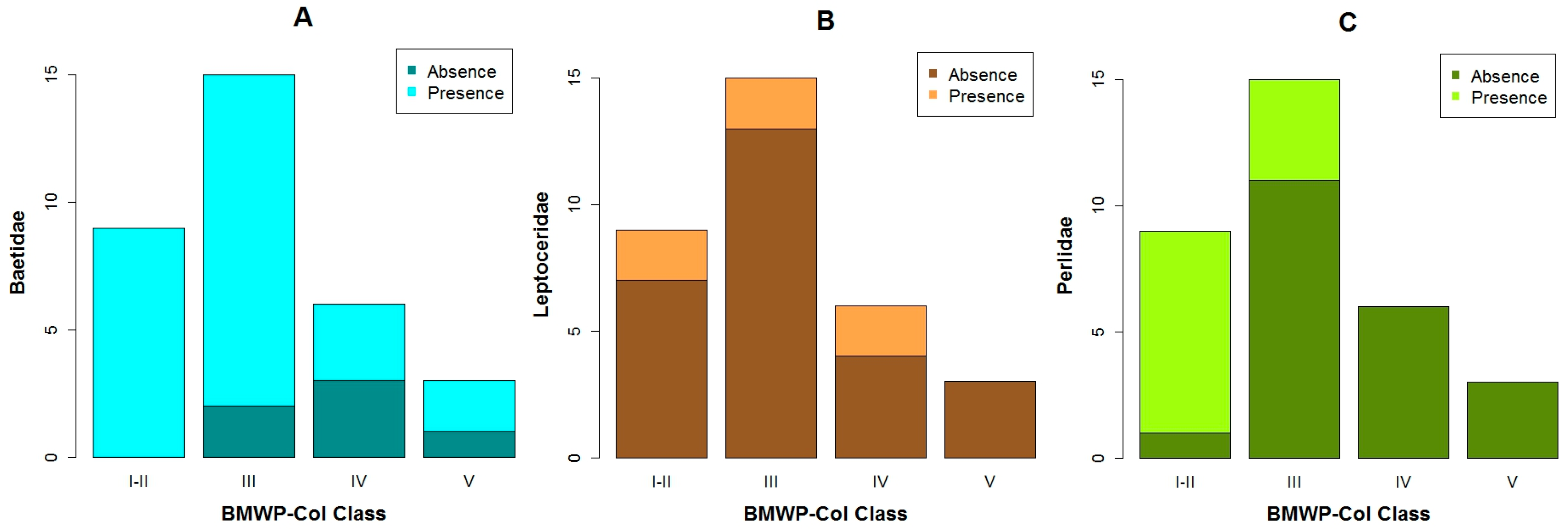
3.1. Data Exploration
3.2. Correlation Analysis
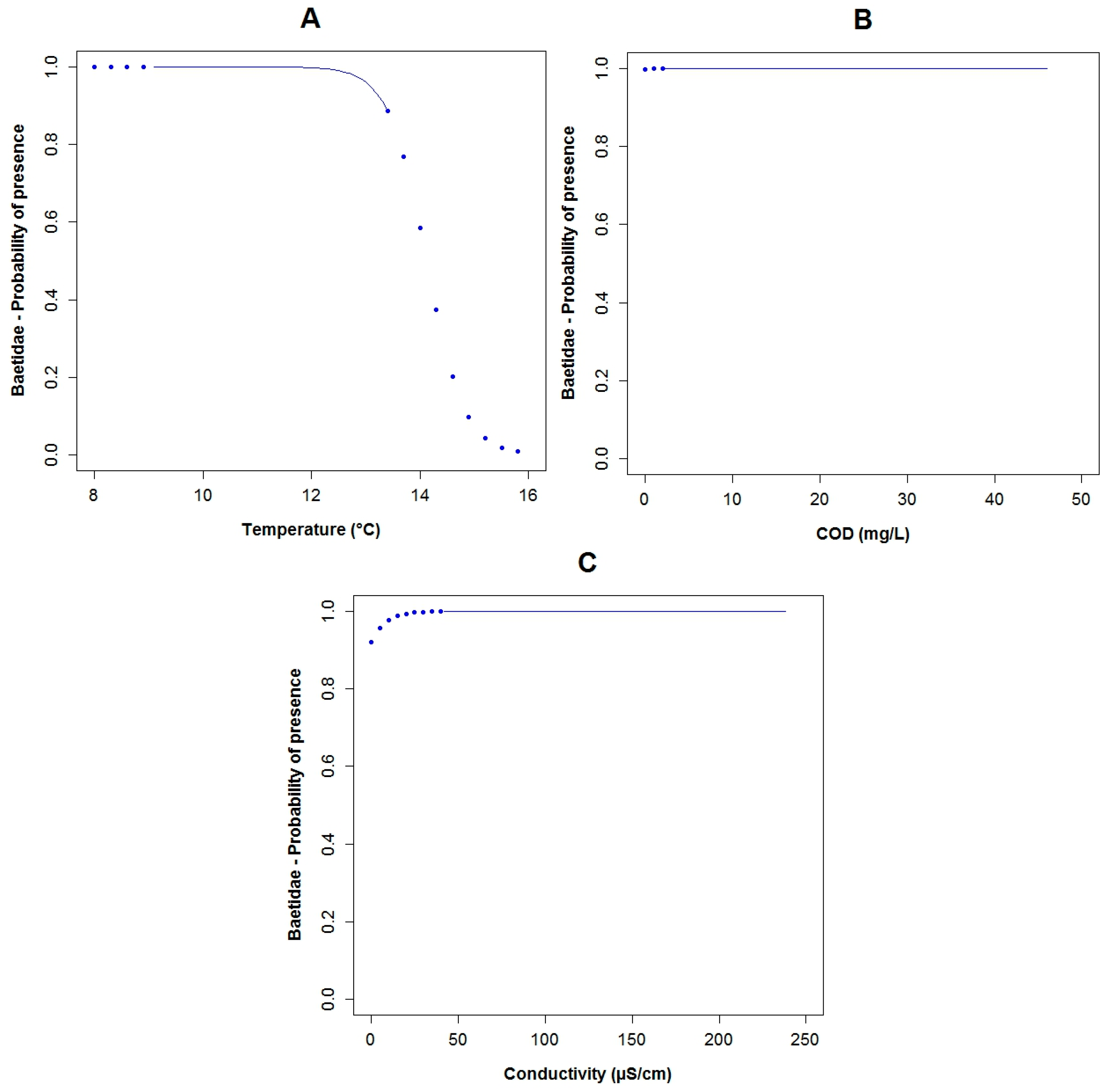
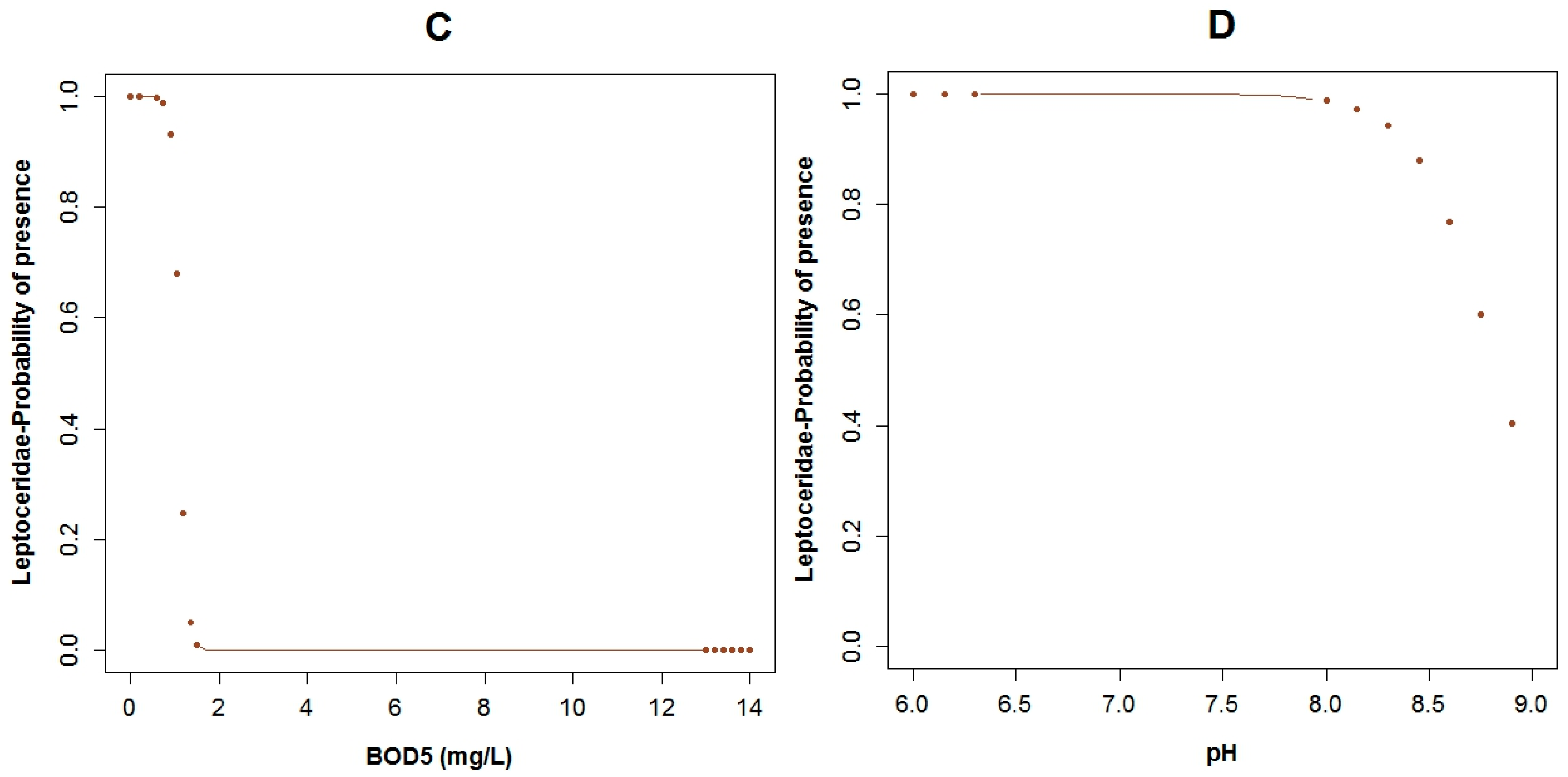
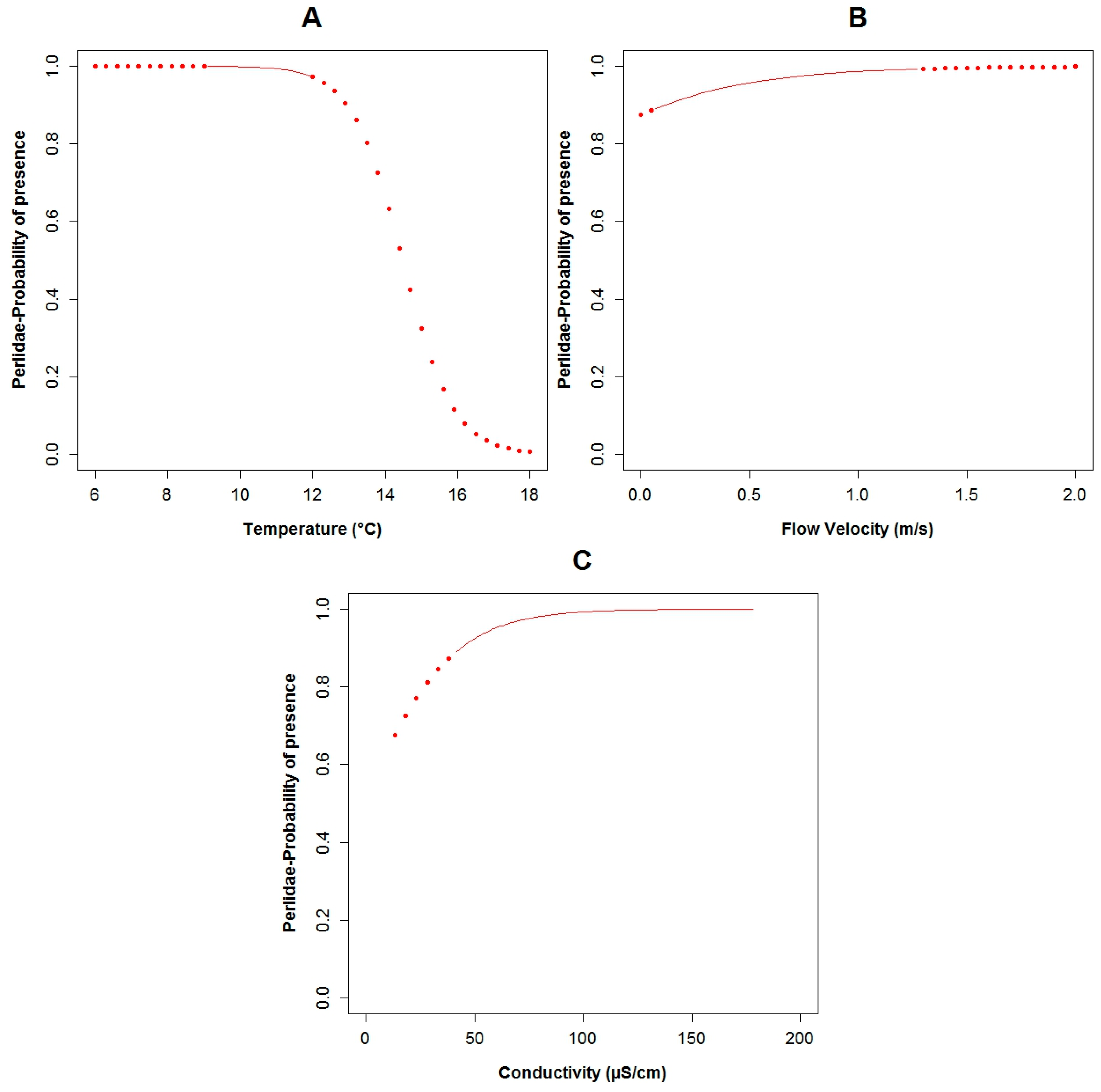
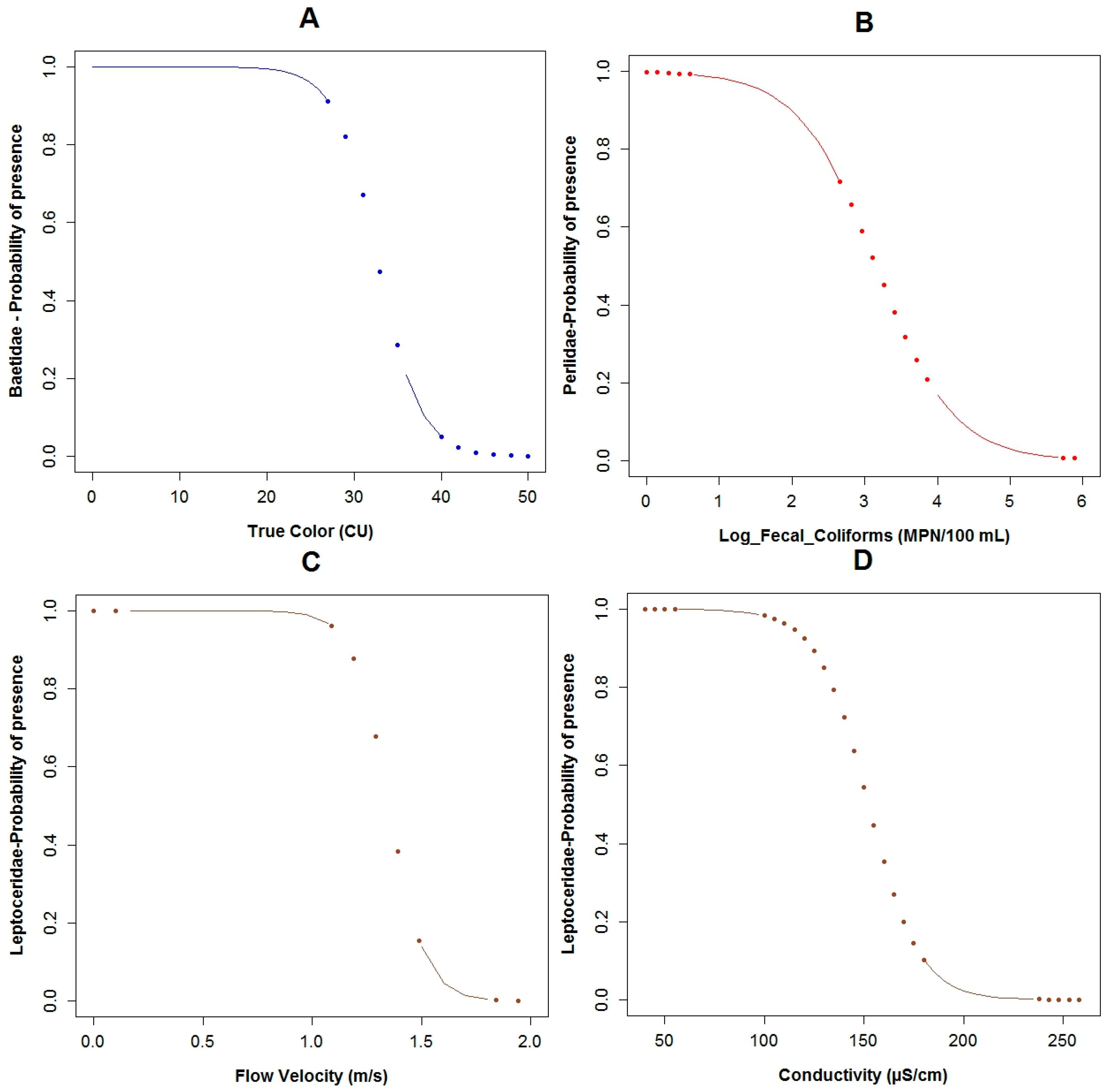
3.3. Logistic Regression Models
4. Discussion
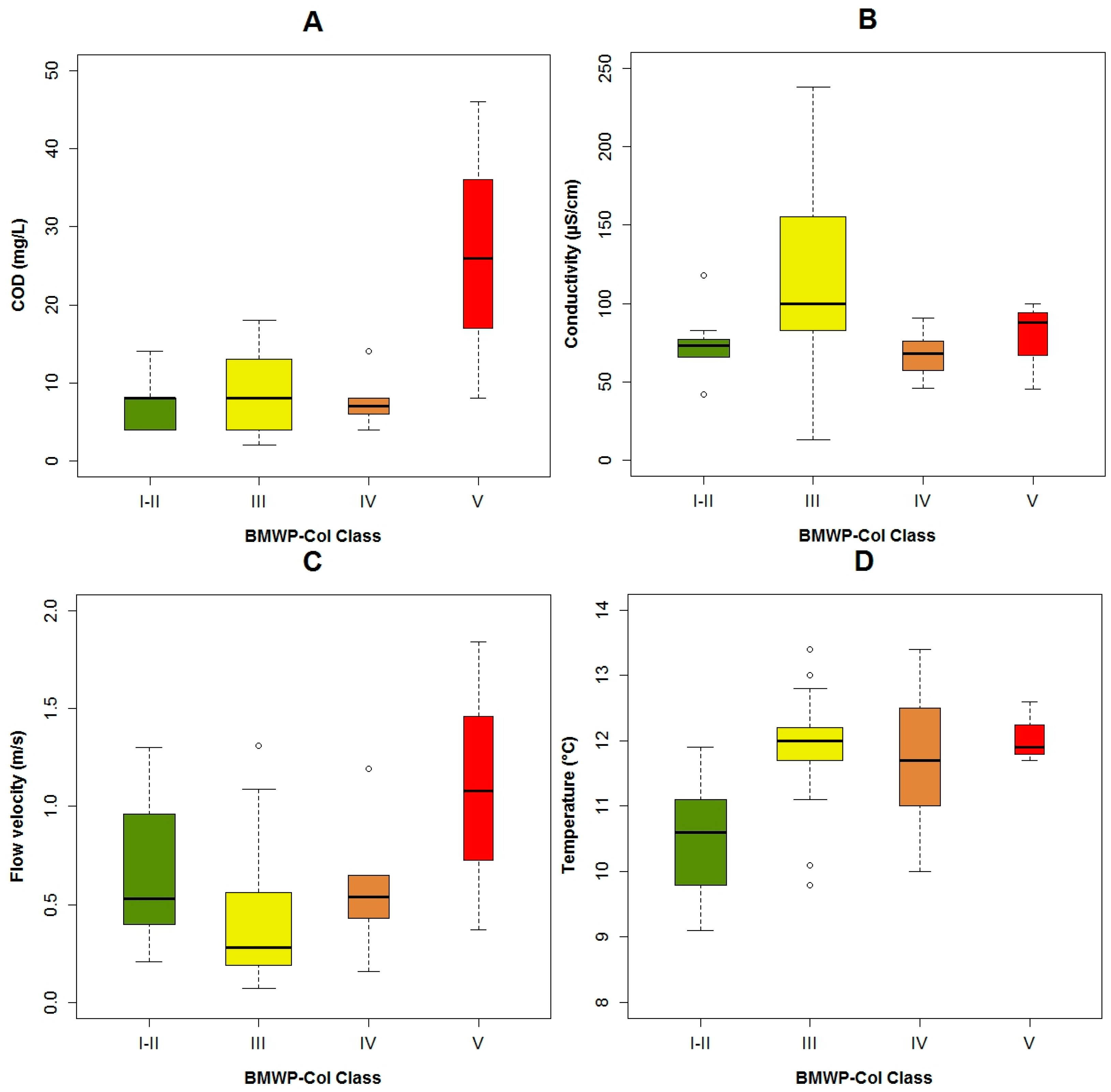
4.1. Analysis of the Chosen EPT Taxa in Relation with the BMWP-Col
4.2. Analysis of the Explanatory Variables in Relation to Response Variables
4.3. Model Performance
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AIC | Akaike information criterion |
| ANNs | artificial neural networks |
| BBNs | Bayesian belief networks |
| BMWP | Biological Monitoring Working Party |
| BMWP-Col | Biological Monitoring Working Party adapted to Colombia |
| BOD5 | Biochemical Oxygen Demand 5 d |
| color | True color |
| CSOs | combined sewer overflows |
| COD | chemical oxygen demand |
| CTs | classification trees |
| DO | Dissolved Oxygen |
| EPT | Ephemeroptera—Plecoptera—Trichoptera |
| GAs | genetic algorithms |
| GLM | generalized linear models |
| LRs | logistic regressions |
| MASL | meters above sea level |
| MPN.100 mL−1 | most probable number per 100 milliliters |
| RTs | regression trees |
| TS | tolerant score |
| TSol | Total Solids |
| SVMs | support-vector machines |
| SWO | surface water outfalls |
Appendix A
| Explanatory Variable | BOD5 | COD | Conductivity | Flow Velocity | Log Fecal Coliforms | pH | True Color (Color) | Water Temperature |
|---|---|---|---|---|---|---|---|---|
| BOD5 | ||||||||
| COD | <0.001 | |||||||
| Conductivity | 0.726 | 0.956 | ||||||
| Flow velocity | 0.531 | 0.151 | 0.097 | |||||
| Log fecal coliforms | <0.001 | 0.009 | 0.04 | 0.298 | ||||
| pH | 0.231 | 0.404 | 0.375 | 0.499 | 0.021 | |||
| True color (color) | 0.002 | <0.001 | 0.08 | 0.104 | 0.063 | 0.663 | ||
| Water temperature | 0.005 | 0.267 | 0.087 | 0.881 | 0.144 | 0.996 | 0.909 |
| Explanatory Variable | Baetidae | Leptoceridae | Perlidae | |||
|---|---|---|---|---|---|---|
| Std. Error | z Value | Std. Error | z Value | Std. Error | z Value | |
| 16.86 | 1.60 | 21.99 | 2.30 | 6.53 | 2.17 | |
| BOD5 | 6.59 | −1.88 | ||||
| COD | 0.46 | 1.63 | 0.57 | −1.95 | ||
| Conductivity | 0.06 | 2.06 | 0.04 | −1.85 | 0.02 | 2.10 |
| Flow velocity | 5.80 | −2.11 | 1.67 | 1.39 | ||
| Log fecal coliforms | 3.07 | 2.09 | 0.83 | −2.28 | ||
| pH | 2.42 | −2.19 | ||||
| Temperature | 1.55 | −1.84 | 0.64 | −2.24 | ||
| True color (color) | 0.25 | −1.66 | ||||
| Model | (Intercept) | p-Value | AIC | R2 (%) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Flow Velocity | BOD5 | Turbidity | Mean Depth | Temperature | pH | DO | Color | Conductivity | COD | Nitrate. Nitrite | Ammonia Nitrogen | Organic Nitrogen | Total Solids | Log Fecal Coliforms | Phosphates | ||||
| m1301 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 40 | 100 |
| m1302 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 38 | 100 |
| m1303 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | - | 36 | 100 |
| m1304 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | - | 34 | 100 |
| m1305 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | - | 32 | 100 |
| m1306 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | - | 30 | 100 |
| m1307 | 1.00 | 1.00 | 1.00 | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | - | 28 | 100 |
| m1308 | 1.00 | 1.00 | 1.00 | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | - | - | 1.00 | 1.00 | 1.00 | - | 26 | 100 |
| m1309 | 1.00 | - | 1.00 | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | - | - | 1.00 | 1.00 | 1.00 | - | 24 | 100 |
| m1310 | 1.00 | - | 1.00 | 1.00 | - | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | - | - | 1.00 | 1.00 | 1.00 | - | 22 | 100 |
| m1311 | 1.00 | - | 1.00 | 1.00 | - | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | - | - | - | 1.00 | 1.00 | - | 20 | 100 |
| m1312 | 0.97 | - | 0.98 | 0.97 | - | 0.97 | - | 0.97 | 0.97 | 0.97 | 0.97 | - | - | - | 0.97 | - | - | 18 | 100 |
| m1313 | 0.65 | - | - | 0.74 | - | 0.65 | - | 0.67 | 0.70 | 0.97 | 0.70 | - | - | - | 0.70 | - | - | 24 | 75 |
| m1314 | 0.31 | - | - | 0.41 | - | 0.34 | - | 0.31 | 0.38 | - | 0.42 | - | - | - | 0.33 | - | - | 22 | 75 |
| m1315 | 0.08 | - | - | 0.18 | - | 0.10 | - | 0.08 | 0.12 | - | - | - | - | - | 0.34 | - | - | 26 | 56 |
| m1316 | 0.04 | - | - | 0.09 | - | 0.08 | - | 0.06 | 0.05 | - | - | - | - | - | - | - | - | 25 | 52 |
| m1317 | 0.04 | - | - | - | - | 0.11 | - | 0.06 | 0.06 | - | - | - | - | - | - | - | - | 31 | 26 |
| m1318 | 0.04 | - | - | - | - | - | - | 0.16 | 0.11 | - | - | - | - | - | - | - | - | 33 | 15 |
| m1319 | 0.09 | - | - | - | - | - | - | 0.20 | 0.00 | - | - | - | - | - | - | - | - | 34 | 6 |
| m1320 | 0.08 | - | - | 0.26 | - | 0.08 | - | 0.14 | 0.25 | 0.27 | - | - | - | - | - | - | - | 25 | 58 |
| m1321 | 0.05 | - | - | 0.08 | - | 0.07 | - | 0.10 | 0.07 | - | 0.30 | - | - | - | - | - | - | 25 | 57 |
| m1322 | 0.07 | - | 0.85 | 0.12 | - | 0.14 | - | 0.07 | 0.07 | - | - | - | - | - | - | - | - | 27 | 53 |
| m1323 | 0.21 | - | - | 0.20 | - | 0.06 | 0.39 | 0.13 | 0.08 | - | - | - | - | - | - | - | - | 26 | 55 |
| m1324 | 0.98 | - | - | 0.98 | - | 0.98 | - | 0.98 | 0.98 | - | - | - | - | - | - | 0.98 | - | 12 | 100 |
| m1325 | 0.99 | - | - | 0.11 | - | 0.08 | - | 0.08 | 0.06 | - | - | - | - | - | - | - | 1.00 | 27 | 54 |
| m1326 | 0.05 | - | - | 0.10 | - | 0.09 | - | 0.06 | 0.05 | - | - | 1.0 | - | - | - | - | - | 27 | 52 |
| m1327 | 0.05 | - | - | 0.10 | - | 0.09 | - | 0.06 | 0.05 | - | - | - | 1.00 | - | - | - | - | 27 | 53 |
| m1328 | 0.05 | - | - | 0.10 | - | 0.09 | - | 0.06 | 0.06 | - | - | 0.83 | - | - | - | - | - | 27 | 53 |
| m1329 | 0.09 | - | - | 0.30 | - | 0.11 | - | 0.11 | 0.26 | - | - | - | - | 0.33 | - | - | - | 25 | 58 |
| m1330 | 0.08 | - | - | 0.18 | - | 0.10 | - | 0.08 | 0.12 | - | - | - | - | - | 0.34 | - | - | 26 | 56 |
| m1331 | 1.00 | - | - | 1.00 | - | 1.00 | - | 1.00 | 1.00 | - | - | - | - | - | - | - | - | 12 | 100 |
| m1332 | 0.99 | - | - | 0.11 | - | 0.08 | - | 0.08 | 0.06 | - | - | - | - | - | - | - | - | 27 | 54 |
| m1333 | 0.05 | - | - | 0.07 | 0.23 | 0.07 | - | 0.08 | 0.07 | - | - | - | - | - | - | - | - | 25 | 59 |
| m1334 | 0.15 | 0.12 | - | 0.13 | - | 0.17 | - | 0.15 | 0.07 | - | - | - | - | - | - | - | - | 21 | 72 |
| m1335 | 0.12 | 0.29 | - | 0.21 | - | 0.00 | - | 0.18 | 0.07 | - | - | - | - | - | - | - | - | 30 | 38 |
| m1336 | 0.07 | - | - | 0.15 | - | 0.00 | - | 0.17 | 0.07 | - | - | - | - | - | - | - | - | 29 | 33 |
| m1337 | 1.00 | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | - | - | 1.00 | 1.00 | - | - | 24 | 100 |
| m1338 | 1.00 | 1.00 | - | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | - | - | 1.00 | 1.00 | - | - | 22 | 100 |
| m1339 | 1.00 | 1.00 | - | 1.00 | - | 1.00 | - | 1.00 | 1.00 | 1.00 | 1.00 | - | - | 1.00 | 1.00 | - | - | 20 | 100 |
| m1340 | 0.99 | - | - | 0.99 | - | 0.99 | - | 0.99 | 0.99 | 0.99 | 0.99 | - | - | 0.99 | 1.00 | - | - | 18 | 100 |
| m1341 | 0.99 | - | - | 0.99 | - | 0.99 | - | 0.99 | 0.99 | 0.99 | 0.99 | - | - | 0.99 | - | - | - | 16 | 100 |
| m1342 | 0.10 | - | - | 0.35 | - | 0.12 | - | 0.40 | 0.17 | 0.17 | 0.31 | - | - | - | - | - | - | 24 | 67 |
| m1343 | 0.14 | - | - | 0.36 | - | 0.12 | - | - | 0.14 | 0.09 | 0.20 | - | - | - | - | - | - | 23 | 65 |
| m1344 | 0.11 | - | - | - | - | 0.07 | - | - | 0.10 | 0.04 | 0.10 | - | - | - | - | - | - | 22 | 60 |
| Model | (Intercept) | p-Value | AIC | R2 (%) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Flow Velocity | BOD5 | Turbidity | Mean Depth | Temperature | pH | DO | Color | Conductivity | COD | Nitrate. Nitrite | Ammonia Nitrogen | Organic Nitrogen | Total Solids | Log Fecal Coliforms | Phosphates | ||||
| m15 | 0.69 | - | - | - | 0.05 | - | - | - | - | 0.80 | - | 0.11 | - | 0.50 | - | 0.11 | - | 45 | 25 |
| m150 | 0.82 | - | - | - | 0.05 | - | - | - | - | - | - | 0.11 | - | - | - | 0.11 | - | 37 | 24 |
| m152 | 0.30 | - | - | - | 0.07 | - | - | - | - | - | - | 0.26 | - | - | - | - | - | 38 | 16 |
| m153 | 0.86 | - | - | - | 0.13 | - | - | - | - | - | - | - | - | - | - | 0.65 | - | 41 | 10 |
| m154 | 0.86 | - | - | - | 0.14 | - | - | - | - | - | - | - | - | - | - | - | - | 39 | 9 |
| m155 | 0.51 | 0.12 | - | - | 0.15 | - | - | - | - | - | - | 0.07 | - | - | - | 0.07 | - | 36 | 32 |
| m156 | 0.88 | 0.04 | - | - | - | - | - | - | - | - | - | 0.11 | - | - | - | 0.08 | - | 38 | 23 |
| m157 | 0.63 | 0.17 | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.66 | - | 42 | 6 |
| m158 | 0.52 | 0.04 | 0.10 | - | - | - | - | - | - | - | - | 0.12 | - | - | - | 0.03 | - | 34 | 38 |
| m159 | 0.89 | 0.12 | 0.07 | - | - | - | - | - | - | - | - | - | - | - | - | 0.04 | - | 37 | 26 |
| m1590 | 1.00 | 0.24 | 0.10 | - | 0.69 | - | - | - | - | - | - | - | - | - | - | 0.05 | - | 39 | 26 |
| m1591 | 0.93 | 0.10 | 0.06 | 0.19 | - | - | - | - | - | - | - | - | - | - | - | 0.02 | - | 36 | 32 |
| m1592 | 1.00 | 0.07 | 0.22 | 0.19 | - | - | - | - | - | - | - | - | - | - | - | 0.03 | - | 37 | 36 |
| m1593 | 0.63 | 0.08 | 0.05 | 0.26 | - | - | - | - | - | 0.55 | - | - | - | - | - | 0.02 | - | 38 | 33 |
| m1594 | 0.85 | 0.10 | 0.06 | 0.20 | - | - | - | 0.87 | - | - | - | - | - | - | - | 0.03 | - | 38 | 32 |
| m1595 | 0.95 | 0.11 | 0.07 | 0.20 | - | 0.97 | - | - | - | - | - | - | - | - | - | 0.02 | - | 38 | 32 |
| m1596 | 0.71 | 0.10 | 0.05 | 0.25 | - | - | - | - | - | - | - | - | - | - | 0.63 | 0.02 | - | 38 | 32 |
| m1597 | 0.17 | 0.08 | 0.06 | 0.48 | - | - | 0.17 | - | - | - | - | - | - | - | - | 0.02 | - | 36 | 38 |
| m1598 | 0.08 | 0.07 | 0.06 | - | - | - | 0.07 | - | - | - | - | - | - | - | - | 0.03 | - | 35 | 36 |
| m1599 | 0.04 | 0.05 | 0.15 | - | - | - | 0.04 | - | - | - | 0.11 | - | - | - | - | 0.02 | - | 32 | 48 |
| m1580 | 0.05 | 0.12 | - | - | - | - | 0.06 | - | - | - | 0.03 | - | - | - | - | 0.04 | - | 35 | 36 |
| m1581 | 0.13 | - | - | - | - | - | 0.12 | - | - | - | 0.03 | - | - | - | - | 0.05 | - | 36 | 28 |
| m1582 | 0.75 | - | - | - | - | - | - | - | - | - | 0.04 | - | - | - | - | 0.09 | - | 37 | 20 |
| m1583 | 0.04 | 0.09 | 0.16 | - | 0.60 | - | 0.04 | - | - | - | 0.10 | - | - | - | - | 0.02 | - | 34 | 49 |
| m1584 | 0.02 | 0.03 | 0.06 | - | - | - | 0.03 | - | - | 0.06 | 0.05 | - | - | - | - | 0.04 | - | 27 | 68 |
| m1585 | 0.07 | 0.04 | 0.05 | 0.57 | - | - | 0.09 | - | - | 0.07 | 0.11 | - | - | - | - | 0.04 | - | 28 | 69 |
| m1586 | 0.04 | 0.03 | 0.07 | - | - | - | 0.05 | - | - | 0.09 | 0.09 | 0.90 | - | - | - | 0.05 | - | 29 | 68 |
| m1575 | 0.02 | 0.03 | 0.07 | - | 0.43 | - | 0.03 | - | - | 0.08 | 0.09 | - | - | - | - | 0.05 | - | 28 | 70 |
| m1587 | 0.04 | 0.12 | 0.13 | - | - | 0.63 | 0.05 | - | - | 0.15 | 0.08 | - | - | - | - | 0.10 | - | 28 | 68 |
| m1588 | 0.02 | 0.03 | 0.05 | - | - | - | 0.12 | 0.61 | - | 0.06 | 0.10 | - | - | - | - | 0.03 | - | 28 | 68 |
| m1589 | 0.97 | 0.05 | 0.20 | - | - | - | 0.04 | - | - | 0.07 | 0.07 | - | - | - | - | 0.05 | - | 28 | 69 |
| m1570 | 0.06 | 0.06 | 0.08 | - | - | - | 0.07 | - | - | 0.09 | 0.12 | - | - | - | 0.36 | 0.07 | - | 27 | 71 |
| m1571 | 0.03 | 0.04 | 0.08 | - | - | - | 0.04 | - | 0.45 | 0.07 | 0.09 | - | - | - | - | 0.04 | - | 29 | 69 |
| m1572 | 0.03 | 0.04 | 0.16 | - | - | - | 0.04 | - | - | 0.06 | 0.06 | - | 1.00 | - | - | 0.04 | - | 28 | 68 |
| m1573 | 0.04 | 0.04 | 0.09 | - | - | - | 0.05 | - | - | 0.10 | 0.09 | 0.75 | - | - | - | 0.05 | - | 28 | 68 |
| m1574 | 0.97 | 0.05 | 0.20 | - | - | - | 0.04 | - | - | 0.07 | 0.07 | - | - | - | - | 0.05 | 1.00 | 28 | 69 |
| Model | (Intercept) | p-Value | AIC | R2 (%) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Flow Velocity | BOD5 | Turbidity | Mean Depth | Temperature | pH | DO | Color | Conductivity | COD | Nitrate. Nitrite | Ammonia Nitrogen | Organic Nitrogen | Total Solids | Log Fecal Coliforms | Phosphates | ||||
| m10 | 0.90 | - | - | 0.31 | - | 0.06 | 0.38 | 0.38 | 0.24 | 0.40 | 0.51 | 0.10 | 0.98 | 0.29 | - | 0.45 | - | 39 | 66 |
| m11 | 0.90 | - | - | 0.31 | - | 0.06 | 0.38 | 0.38 | 0.24 | 0.40 | 0.51 | 0.10 | - | 0.29 | - | 0.45 | - | 37 | 66 |
| m12 | 0.84 | - | - | 0.21 | - | 0.04 | 0.49 | 0.53 | 0.24 | 0.44 | - | 0.09 | - | 0.17 | - | 0.39 | - | 35 | 65 |
| m13 | 0.79 | - | - | 0.22 | - | 0.05 | 0.63 | - | 0.21 | 0.42 | - | 0.10 | - | 0.17 | - | 0.53 | - | 34 | 64 |
| m14 | 0.07 | - | 0.51 | 0.20 | - | 0.03 | - | - | 0.32 | 0.49 | - | 0.18 | - | 0.20 | - | 0.31 | - | 34 | 64 |
| m15 | 0.02 | - | - | 0.88 | - | 0.02 | - | - | 0.42 | - | - | 0.52 | - | 0.49 | - | - | - | 43 | 29 |
| m16 | 0.05 | 0.75 | 0.57 | - | 0.88 | 0.07 | - | - | 0.68 | - | - | - | - | 0.46 | - | - | - | 44 | 31 |
| m160 | 0.05 | 0.78 | 0.54 | - | - | 0.07 | - | - | 0.70 | - | - | - | - | 0.45 | - | - | - | 42 | 31 |
| m161 | 0.05 | - | 0.53 | - | - | 0.07 | - | - | 0.75 | - | - | - | - | 0.43 | - | - | - | 40 | 31 |
| m162 | 0.05 | - | 0.43 | - | - | 0.07 | - | - | - | - | - | - | - | 0.29 | - | - | - | 38 | 30 |
| m163 | 0.02 | - | - | - | - | 0.02 | - | - | - | - | - | - | - | 0.23 | - | - | - | 39 | 24 |
| m164 | 0.52 | - | - | - | - | - | - | - | - | - | - | - | - | 0.33 | - | - | - | 46 | 4 |
| m165 | 0.05 | - | - | - | - | 0.07 | - | 0.52 | - | - | - | - | - | - | - | 0.08 | - | 40 | 26 |
| m166 | 0.02 | - | - | 0.52 | - | 0.02 | - | - | - | - | - | - | - | 0.23 | - | 0.00 | - | 41 | 25 |
| m167 | 0.03 | - | - | - | - | 0.05 | - | - | - | - | - | - | - | 0.27 | - | 0.11 | - | 37 | 32 |
| m168 | 0.03 | - | - | - | - | 0.04 | - | 0.51 | - | - | - | - | - | 0.26 | - | 0.10 | - | 39 | 33 |
| m169 | 0.16 | 0.83 | 0.06 | - | - | - | - | - | - | - | - | - | - | - | - | 0.00 | - | 41 | 19 |
| m170 | 0.14 | - | 0.06 | - | - | - | - | - | - | - | - | - | - | - | - | 0.00 | - | 39 | 19 |
| m171 | 0.08 | - | 0.20 | - | - | 0.14 | - | - | - | - | - | - | - | - | - | 0.00 | - | 39 | 25 |
| m172 | 0.10 | - | 0.21 | - | - | 0.00 | - | - | - | - | - | - | - | - | - | 0.32 | - | 40 | 22 |
| m173 | 0.07 | - | 0.48 | - | - | 0.15 | - | - | - | - | - | - | - | - | - | 0.34 | - | 40 | 27 |
| m174 | 0.04 | - | 0.72 | - | - | 0.07 | - | - | - | - | - | - | - | 0.28 | - | 0.32 | - | 39 | 33 |
| m175 | 0.03 | - | - | - | - | 0.02 | - | - | - | - | - | - | - | - | - | - | - | 41 | 15 |
| m176 | 0.99 | - | - | 0.10 | - | - | - | - | - | - | 0.17 | - | - | - | - | 0.09 | 1.00 | 40 | 30 |
| m177 | 0.13 | - | - | 0.18 | - | - | - | - | - | - | 0.14 | - | - | - | 0.56 | 0.09 | - | 41 | 29 |
| m178 | 0.42 | - | - | 0.17 | - | - | - | - | - | 0.28 | 0.22 | - | - | - | - | 0.07 | - | 40 | 31 |
| m179 | 0.79 | - | - | 0.13 | - | - | 0.90 | - | - | 0.00 | 0.09 | - | - | - | - | 0.09 | - | 41 | 28 |
| m1601 | 0.04 | - | - | 0.11 | - | - | - | - | - | 0.00 | 0.08 | - | - | - | - | 0.09 | - | 39 | 28 |
| m1602 | 0.03 | - | - | 0.12 | - | 0.08 | - | - | - | 0.00 | 0.07 | - | - | - | - | 0.16 | - | 37 | 37 |
| m1603 | 0.03 | - | - | 0.38 | - | 0.04 | - | - | - | 0.00 | 0.04 | - | - | - | - | - | - | 39 | 29 |
| m1604 | 0.03 | - | - | - | - | 0.05 | - | - | - | 0.00 | 0.06 | - | - | - | - | - | - | 38 | 27 |
| m1605 | 0.04 | - | - | - | - | 0.08 | - | - | - | 0.00 | 0.15 | - | - | - | - | 0.28 | - | 38 | 31 |
| m1606 | 0.03 | - | - | - | - | 0.06 | - | 0.24 | - | 0.00 | 0.10 | - | - | - | - | 0.23 | - | 39 | 34 |
| m1607 | 0.03 | - | - | - | - | 0.04 | - | 0.32 | - | 0.00 | 0.04 | - | - | - | - | - | - | 38 | 30 |
| m1608 | 0.20 | - | - | - | - | 0.05 | 0.98 | - | - | 0.00 | 0.06 | - | - | - | - | - | - | 40 | 27 |
| m1609 | 0.03 | - | - | - | - | 0.04 | - | - | - | 0.00 | 0.08 | 0.52 | - | - | - | - | - | 39 | 29 |
| m1610 | 0.99 | - | - | - | - | 0.06 | - | - | - | 0.00 | 0.12 | - | - | - | - | - | 1.00 | 39 | 29 |
| m1611 | 0.04 | - | - | - | - | 0.04 | - | - | - | 0.22 | 0.06 | - | - | - | - | - | - | 38 | 31 |
| m1612 | 0.03 | 0.54 | - | - | - | 0.04 | - | - | - | - | 0.05 | - | - | - | - | - | - | 39 | 28 |
| m1613 | 0.03 | - | - | - | 0.89 | 0.06 | - | - | - | - | 0.06 | - | - | - | - | - | - | 39 | 27 |
| m1614 | 0.04 | - | - | - | - | 0.04 | - | - | - | - | 0.09 | - | - | - | 0.60 | - | - | 39 | 28 |
| m1615 | 0.03 | - | - | - | - | 0.04 | - | - | 0.54 | - | 0.28 | - | - | - | - | - | - | 39 | 28 |
| m1615’ | 0.06 | - | 0.51 | - | - | 0.12 | - | - | - | - | 0.19 | - | - | - | - | - | - | 39 | 29 |
| m1616 | 0.04 | 0.23 | 0.96 | 0.22 | - | 0.03 | - | 0.22 | - | 0.19 | 0.25 | 0.88 | - | - | - | 0.12 | - | 40 | 54 |
| m1617 | 0.03 | 0.21 | - | 0.19 | - | 0.03 | - | 0.19 | - | 0.18 | 0.21 | 0.89 | - | - | - | 0.09 | - | 38 | 54 |
| m1618 | 0.03 | 0.14 | - | 0.18 | - | 0.03 | - | 0.18 | - | 0.17 | 0.19 | - | - | - | - | 0.07 | - | 36 | 53 |
| m1619 | 0.04 | 0.25 | - | 0.66 | - | 0.03 | - | 0.57 | - | 0.04 | - | - | - | - | - | 0.02 | - | 38 | 45 |
| m1620 | 0.04 | 0.16 | - | - | - | 0.02 | - | 0.59 | - | 0.04 | - | - | - | - | - | 0.02 | - | 36 | 44 |
| m1621 | 0.03 | 0.17 | - | - | - | 0.02 | - | - | - | 0.04 | - | - | - | - | - | 0.02 | - | 35 | 44 |
| m1622 | 0.04 | - | - | - | - | 0.04 | - | - | - | 0.06 | - | - | - | - | - | 0.04 | - | 35 | 38 |
| m1623 | 0.04 | - | - | - | - | 0.08 | - | - | - | - | - | - | - | - | - | 0.09 | - | 38 | 25 |
| m1624 | 0.14 | - | 0.06 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 39 | 19 |
| m1625 | 0.12 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.04 | - | 40 | 17 |


References
- Ambelu, A.; Lock, K.; Goethals, P. Comparison of modelling techniques to predict macroinvertebrate community composition in rivers of Ethiopia. Ecol. Inform. 2010, 5, 147–152. [Google Scholar] [CrossRef]
- Džeroski, S.; Demšar, D.; Grbović, J. Predicting chemical parameters of river water quality from bioindicator data. Appl. Intell. 2000, 13, 7–17. [Google Scholar]
- De Pauw, N.; Gabriels, W.; Goethals, P.L. Goethals, River monitoring and assessment methods based on macroinvertebrates. In Biological Monitoring of Rivers; John Wiley & Sons: Chichester, UK, 2006; pp. 113–134. [Google Scholar]
- Rosenberg, D.M.; Resh, V.H. Freshwater Biomonitoring and Benthic Macroinvertebrates; Chapman & Hall: London, UK, 1993. [Google Scholar]
- Junqueira, V.; Campos, S. Adaptation of the “BMWP” method for water quality evaluation to Rio das Velhas watershed (Minas Gerais, Brazil). Acta Limnol. Bras. 1998, 10, 125–135. [Google Scholar]
- Mustow, S. Biological monitoring of rivers in Thailand: Use and adaptation of the BMWP score. Hydrobiologia 2002, 479, 191–229. [Google Scholar] [CrossRef]
- Roldán Pérez, G.A. Bioindicación de la Calidad del Agua en Colombia: Uso del Método BMWP/Col; Imprenta Universidad de Antioquia: Medellín, Colombia, 2003. [Google Scholar]
- Hvitved-Jacobsen, T. The impact of combined sewer overflows on the dissolved oxygen concentration of a river. Water Res. 1982, 16, 1099–1105. [Google Scholar] [CrossRef]
- Lenat, D.R.; Resh, V.H. Taxonomy and stream ecology—The benefits of genus-and species-level identifications. J. N. Am. Benthol. Soc. 2001, 20, 287–298. [Google Scholar] [CrossRef]
- Thorne, R.; Williams, P. The response of benthic macroinvertebrates to pollution in developing countries: A multimetric system of bioassessment. Freshw. Biol. 1997, 37, 671–686. [Google Scholar] [CrossRef]
- Ríos-Touma, B.; Encalada, A.C.; Prat Fornells, N. Macroinvertebrate Assemblages of an Andean High-Altitude Tropical Stream: The Importance of Season and Flow. Int. Rev. Hydrobiol. 2011, 96, 667–685. [Google Scholar]
- Jacobsen, D.; Marín, R. Bolivian Altiplano streams with low richness of macroinvertebrates and large diel fluctuations in temperature and dissolved oxygen. Aquat. Ecol. 2008, 42, 643–656. [Google Scholar] [CrossRef]
- Jacobsen, D.; Rostgaard, S.; Vásconez, J.J. Are macroinvertebrates in high altitude streams affected by oxygen deficiency? Freshw. Biol. 2003, 48, 2025–2032. [Google Scholar] [CrossRef]
- Jacobsen, D. The effect of organic pollution on the macroinvertebrate fauna of Ecuadorian highland streams. Arch. Hydrobiol. 1998, 143, 179–195. [Google Scholar]
- Degraer, S.; Verfaillie, E.; Willems, W.; Adriaens, E.; Vincx, M.; Van Lancker, V. Habitat suitability modelling as a mapping tool for macrobenthic communities: An example from the Belgian part of the North Sea. Cont. Shelf Res. 2008, 28, 369–379. [Google Scholar] [CrossRef]
- Zarkami, R. Habitat Suitability Modelling of Pike (Esox lucius) in Rivers; Ghent University: Ghent, Belgium, 2008. [Google Scholar]
- Dominguez-Granda, L.; Lock, K.; Goethals, P.L. Using multi-target clustering trees as a tool to predict biological water quality indices based on benthic macroinvertebrates and environmental parameters in the Chaguana watershed (Ecuador). Ecol. Inform. 2011, 6, 303–308. [Google Scholar] [CrossRef]
- Goethals, P. Data Driven Development of Predictive Ecological Models for Benthic Macroinvertebrates in Rivers; Ghent University: Ghent, Belgium, 2005. [Google Scholar]
- Fernández de Córdova, J.; González, H. Evoluación de la Calidad del Agua de los Tramos Bajos de los Ríos de la Ciudad de Cuenca; ETAPA-EP: Cuenca, Ecuador, 2012. [Google Scholar]
- Instituto Nacional de Estadísticas y Censos del Ecuador (INEC). Proyección de la Población Ecuatoriana, por años Calendario, Según Cantones 2010–2020; Instituto Nacional de Estadísticas y Censos del Ecuador: Quito, Ecuador, 2010.
- PROMAS-UCuenca. Información de la Red Meteorológica e Hidrológica; Programa para el Manejo del Agua y el Suelo; Universidad de Cuenca: Cuenca, Ecuador, 2010. [Google Scholar]
- Aeropuerto_Mariscal_Lamar. Información Meteorológica Aeropuerto Mariscal Lamar Cuenca; Dirección de Aviación Civil del Ecuador: Quito, Ecuador, 2012. [Google Scholar]
- Estrella, R.; Tobar, V. Hidrología y Climatología—Formulación del Plan de Manejo Integral de la Subcuenca del río Machangara; ACOTECNIC Cia. Ltda.—Consejo de Cuenca del Rio Machangara: Cuenca, Ecuador, 2013. [Google Scholar]
- Esquivel, J.C.; Verbeiren, B.; Alvarado, A.; Feyen, J.; Cisneros, F. Preliminary Statistical Analysis of the Water Quality Database of ETAPA; PROMAS—Universidad de Cuenca: Cuenca, Ecuador, 2008. [Google Scholar]
- Mulliss, R.; Revitt, D.M.; Shutes, R.B.E. The impacts of discharges from two combined sewer overflows on the water quality of an urban watercourse. Water Sci. Technol. 1997, 36, 195–199. [Google Scholar] [CrossRef]
- Weyrauch, P.; Matzinger, A.; Pawlowsky-Reusing, E.; Plume, S.; von Seggern, D.; Heinzmann, B.; Schroeder, K.; Rouault, P. Contribution of combined sewer overflows to trace contaminant loads in urban streams. Water Res. 2010, 44, 4451–4462. [Google Scholar] [CrossRef] [PubMed]
- Passerat, J.; Ouattara, N.K.; Mouchel, J.-M.; Vincent, R.; Servais, P. Impact of an intense combined sewer overflow event on the microbiological water quality of the Seine River. Water Res. 2011, 45, 893–903. [Google Scholar] [CrossRef] [PubMed]
- Novotny, V. Diffuse pollution from agriculture—A worldwide outlook. Water Sci. Technol. 1999, 39, 1–13. [Google Scholar] [CrossRef]
- Armitage, P.D.; Moss, D.; Wright, J.F.; Furse, M.T. The performance of a new biological water quality score system based on macroinvertebrates over a wide range of unpolluted running-water sites. Water Res. 1983, 17, 333–347. [Google Scholar] [CrossRef]
- Sutherland, W.J. Ecological Census Techniques: A Handbook; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Alba-Tecedor, J.; Pardo, I.; Prat, N.; Pujanta, A. Protocolos de Muestreo y Análisis para Invertebrados Bentónicos; Ministerio de Medio Ambiente, Confederación Hidrográfica del Ebro y URS, Metodología para el establecimiento del Estado Ecológico según la Directiva Marco del Aguas: Madrid, Spain, 2005. [Google Scholar]
- Roldán Pérez, G.A. Guía Para el Estudio de los Macroinvertebrados Acuáticos del Departamento de Antioquia; Fondo para la Protección del Medio Ambiente José Celestino Mutis: Bogotá, Colombia, 1988. [Google Scholar]
- Álvarez, L.F. Metodología Para la Utilización de los Macroinvertebrados Acuáticos Como Indicadores de la Calidad del Agua; Instituto Alexander von Humboldt: Bogotá, Colombia, 2006. [Google Scholar]
- Encalada, A.C. Protocolo Simplificado y Guía de Evaluación de la Calidad Ecológica de ríos Andinos (CERA-S): Text; 2. Làmines; Proyecto FUCARA: Quito, Ecuador, 2011. [Google Scholar]
- Zúñiga, M.D.C.; Cardona, W.; Cantera, J.; Carvajal, Y.; Castro, L. Bioindicadores de calidad de agua y caudal ambiental. In Caudal Ambiental: Conceptos Experiencias Desafíos; Programa Editorial, Universidad del Valle: Cali, Colombia, 2009; Volume 1, pp. 167–197. [Google Scholar]
- Zuur, A.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer: New York, NY, USA, 2009. [Google Scholar]
- Holguin-Gonzalez, J.E.; Everaert, G.; Boets, P.; Galvis, A.; Goethals, P.L. Development and application of an integrated ecological modelling framework to analyze the impact of wastewater discharges on the ecological water quality of rivers. Environ. Model. Softw. 2013, 48, 27–36. [Google Scholar] [CrossRef]
- Everaert, G.; de Neve, J.; Boets, P.; Dominguez-Granda, L.; Mereta, S.T.; Ambelu, A.; Hoang, T.H.; Goethals, P.L.M.; Thas, O. Comparison of the abiotic preferences of macroinvertebrates in tropical river basins. PLoS ONE 2014, 9, e108898. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.H.; Everaert, G.; Gabriels, W.; Hoang, T.H.; Goethals, P.L. A multimetric macroinvertebrate index for assessing the water quality of the Cau river basin in Vietnam. Limnol.-Ecol. Manag. Inland Waters 2014, 45, 16–23. [Google Scholar] [CrossRef]
- Hering, D.; Feld, C.K.; Moog, O.; Ofenböck, T. Cook book for the development of a Multimetric Index for biological condition of aquatic ecosystems: Experiences from the European AQEM and STAR projects and related initiatives. Hydrobiologia 2006, 566, 311–324. [Google Scholar] [CrossRef]
- Booth, G.D.; Niccolucci, M.J.; Schuster, E.G. Identifying Proxy Sets in Multiple Linear Regression: An Aid to Better Coefficient Interpretation; U.S. Dept. of Agriculture, Forest Service, Intermountain Research Station: Ogden, UT, USA, 1994.
- Agresti, A.; Kateri, M. Categorical Data Analysis; Springer-Verlag: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Gabriels, W.; Goethals, P.L.; Dedecker, A.P.; Lek, S.; De Pauw, N. Analysis of macrobenthic communities in Flanders, Belgium, using a stepwise input variable selection procedure with artificial neural networks. Aquat. Ecol. 2007, 41, 427–441. [Google Scholar] [CrossRef]
- Hu, B.; Shao, J.; Palta, M. Pseudo-R 2 in logistic regression model. Stat. Sin. 2006, 16, 847–860. [Google Scholar]
- R-Core-Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2015. [Google Scholar]
- Kasangaki, A.; Chapman, L.J.; Balirwa, J. Land use and the ecology of benthic macroinvertebrate assemblages of high-altitude rainforest streams in Uganda. Freshw. Biol. 2008, 53, 681–697. [Google Scholar] [CrossRef]
- Beadle, L.C. The Inland Waters of Tropical Africa: An Introduction to Tropical Limnology; Longman Group Ltd. Publishers: London, UK, 1974. [Google Scholar]
- Acreman, M.; Ferguson, A. Environmental flows and the European water framework directive. Freshw. Biol. 2010, 55, 32–48. [Google Scholar] [CrossRef]
- Ogleni, N.; Topal, B. Water quality assessment of the Mudurnu River, Turkey, using biotic indices. Water Resour. Manag. 2011, 25, 2487. [Google Scholar] [CrossRef]
- Forio, M.A.E.; Van Echelpoel, W.; Dominguez-Granda, L.; Mereta, S.T.; Ambelu, A.; Hoang, T.H.; Boets, P.; Goethals, P.L. Analysing the effects of water quality on the occurrence of freshwater macroinvertebrate taxa among tropical river basins from different continents. AI Commun. 2016, 29, 665–685. [Google Scholar] [CrossRef]
- D’Heygere, T.; Goethals, P.L.M.; De Pauw, N. Use of genetic algorithms to select input variables in decision tree models for the prediction of benthic macroinvertebrates. Ecol. Model. 2003, 160, 291–300. [Google Scholar]
- Schwarzenbach, R.P.; Egli, T.; Hofstetter, T.B.; von Gunten, U.; Wehrli, B. Global water pollution and human health. Annu. Rev. Environ. Resour. 2010, 35, 109–136. [Google Scholar] [CrossRef]
- Lock, K.; Goethals, P.L. Habitat suitability modelling for mayflies (Ephemeroptera) in Flanders (Belgium). Ecol. Inform. 2013, 17, 30–35. [Google Scholar] [CrossRef]
- Lock, K.; Goethals, P.L. Distribution and ecology of the caddisflies (Trichoptera) of Flanders (Belgium). Ann. Limnol.-Int. J. Limnol. 2012, 48, 31–37. [Google Scholar] [CrossRef]
- Lock, K.; Goethals, P.L. Distribution and ecology of the stoneflies (Plecoptera) of Flanders (Belgium). Ann. Limnol.-Int. J. Limnol. 2008, 44, 203–213. [Google Scholar] [CrossRef]
- Dedecker, A.P.; Goethals, P.L.; Gabriels, W.; De Pauw, N. Optimization of Artificial Neural Network (ANN) model design for prediction of macroinvertebrates in the Zwalm river basin (Flanders, Belgium). Ecol. Model. 2004, 174, 161–173. [Google Scholar] [CrossRef]
- Holguin-Gonzalez, J.E.; Boets, P.; Alvarado, A.; Cisneros, F.; Carrasco, M.C.; Wyseure, G.; Nopens, I.; Goethals, P.L. Integrating hydraulic, physicochemical and ecological models to assess the effectiveness of water quality management strategies for the River Cuenca in Ecuador. Ecol. Model. 2013, 254, 1–14. [Google Scholar] [CrossRef]
- Connolly, N.; Crossland, M.; Pearson, R. Effect of low dissolved oxygen on survival, emergence, and drift of tropical stream macroinvertebrates. J. N. Am. Benthol. Soc. 2004, 23, 251–270. [Google Scholar] [CrossRef]
- Chakona, A.; Phiri, C.; Day, J.A. Potential for Trichoptera communities as biological indicators of morphological degradation in riverine systems. Hydrobiologia 2009, 621, 155–167. [Google Scholar] [CrossRef]
- Hawkins, C.P.; Hogue, J.N.; Decker, L.M.; Feminella, J.W. Channel morphology, water temperature, and assemblage structure of stream insects. J. N. Am. Benthol. Soc. 1997, 16, 728–749. [Google Scholar] [CrossRef]
- Hynes, H.B.N. The Biology of Polluted Waters; Liverpool UP: Liverpool, UK, 1960. [Google Scholar]
- Burneo, P.C.; Gunkel, G. Ecology of a high Andean stream, Rio Itambi, Otavalo, Ecuador. Limnol.-Ecol. Manag. Inland Waters 2003, 33, 29–43. [Google Scholar] [CrossRef]
- Bennett, L.E.; Drikas, M. The evaluation of colour in natural waters. Water Res. 1993, 27, 1209–1218. [Google Scholar] [CrossRef]
- Haaland, S.; Hongve, D.; Laudon, H.; Riise, G.; Vogt, R. Quantifying the drivers of the increasing colored organic matter in boreal surface waters. Environ. Sci. Technol. 2010, 44, 2975–2980. [Google Scholar] [CrossRef] [PubMed]
- Dallas, H.F.; Day, J.A. Natural variation in macroinvertebrate assemblages and the development of a biological banding system for interpreting bioassessment data—A preliminary evaluation using data from upland sites in the south-western Cape, South Africa. Hydrobiologia 2007, 575, 231–244. [Google Scholar] [CrossRef]
- Feldman, R.S.; Connor, E.F. The relationship between pH and community structure of invertebrates in streams of the Shenandoah National Park, Virginia, USA. Freshw. Biol. 1992, 27, 261–276. [Google Scholar] [CrossRef]
- Ambelu, A.; Mekonen, S.; Koch, M.; Addis, T.; Boets, P.; Everaert, G.; Goethals, P. The application of predictive modelling for determining bio-environmental factors affecting the distribution of blackflies (Diptera: Simuliidae) in the Gilgel Gibe watershed in Southwest Ethiopia. PLoS ONE 2014, 9, e112221. [Google Scholar] [CrossRef] [PubMed]
- Damanik-Ambarita, M.N.; Everaert, G.; Forio, M.A.E.; Nguyen, T.H.T.; Lock, K.; Musonge, P.L.S.; Suhareva, N.; Dominguez-Granda, L.; Bennetsen, E.; Boets, P.; et al. Generalized Linear Models to Identify Key Hydromorphological and Chemical Variables Determining the Occurrence of Macroinvertebrates in the Guayas River Basin (Ecuador). Water 2016, 8, 297. [Google Scholar] [CrossRef]
- Thuiller, W. BIOMOD—Optimizing predictions of species distributions and projecting potential future shifts under global change. Glob. Chang. Biol. 2003, 9, 1353–1362. [Google Scholar] [CrossRef]
- Vaughan, I.P.; Ormerod, S.J. The continuing challenges of testing species distribution models. J. Appl. Ecol. 2005, 42, 720–730. [Google Scholar] [CrossRef]
- Stockwell, D.R.; Peterson, A.T. Effects of sample size on accuracy of species distribution models. Ecol. Model. 2002, 148, 1–13. [Google Scholar] [CrossRef]
- Yu, R.; Abdel-Aty, M. Utilizing support vector machine in real-time crash risk evaluation. Accid. Anal. Prev. 2013, 51, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Pearce, J.; Ferrier, S. Evaluating the predictive performance of habitat models developed using logistic regression. Ecol. Model. 2000, 133, 225–245. [Google Scholar] [CrossRef]
- Manel, S.; Williams, H.C.; Ormerod, S.J. Evaluating presence–absence models in ecology: The need to account for prevalence. J. Appl. Ecol. 2001, 38, 921–931. [Google Scholar] [CrossRef]
- Mac Nally, R. Regression and model-building in conservation biology, biogeography and ecology: The distinction between–and reconciliation of ‘predictive’ and ‘explanatory’ models. Biodivers. Conserv. 2000, 9, 655–671. [Google Scholar] [CrossRef]

| Parameter | Units | Mean Value | Standard Deviation | Min Value | Max Value | Median Value |
|---|---|---|---|---|---|---|
| Mean depth | m | 0.33 ± 0.30 | 0.04 | 1.63 | 0.26 | |
| Flow velocity | m·s−1 | 0.59 ± 0.44 | 0.07 | 1.84 | 0.47 | |
| Temperature | °C | 11.50 ± 1.10 | 9.10 | 13.40 | 11.90 | |
| pH | 7.58 ± 0.45 | 6.33 | 8.36 | 7.70 | ||
| Dissolved oxygen (DO) | mg·L−1 | 9.08 ± 1.47 | 6.65 | 12.60 | 9.54 | |
| Total solids (TSol) | mg·L−1 | 89.09 ± 51.65 | 19.00 | 190.00 | 74.00 | |
| Turbidity | NTU | 7.68 ± 11.11 | 0.51 | 48.20 | 3.66 | |
| True color (color) | HU | 14.39 ± 8.52 | 0.00 | 40.00 | 14.00 | |
| Specific conductivity | μS·cm−1 | 91.64 ± 44.12 | 13.20 | 238.00 | 82.30 | |
| Phosphates | mg·L−1 | 0.07 ± 0.12 | 0.03 | 0.55 | 0.03 | |
| Nitrate + Nitrite | mg·N·L−1 | 0.05 ± 0.12 | BDL | 0.70 | 0.02 | |
| Ammonia nitrate | mg·L−1 | 0.02 ± 0.07 | 0.00 | 0.40 | 0.00 | |
| Organic nitrogen | mg·L−1 | 0.55 ± 1.21 | 0.00 | 6.55 | 0.14 | |
| Biochemical oxygen demand 5 day (BOD5) | mg·L−1 | 1.06 ± 2.35 | BDL | 13.00 | 0.40 | |
| Chemical oxygen demand (COD) | mg·L−1 | 9.94 ± 8.39 | 2.00 | 46.00 | 8.00 | |
| Fecal coliforms | MPN.100 mL−1 | 3.60 × 104 ± 1.02 × 105 | 4.5 × 100 | 5.4 × 105 | 7.9 × 101 | |
| Descriptive statistics of physicochemical and microbiological variables are given as mean values ± standard deviations, minimums and maximums | ||||||
| NTU = Nephelometric turbidity units | ||||||
| HU = Hazen units | ||||||
| MPN = Most probable number | ||||||
| BDL = Below Detection Limit | ||||||
| Explanatory Variable | BOD5 | COD | Conductivity | Flow Velocity | Log Fecal Coliforms | pH | True Color (Color) | Water Temperature |
|---|---|---|---|---|---|---|---|---|
| BOD5 | 1.00 | |||||||
| COD | 0.61 | 1.00 | ||||||
| Conductivity | 0.06 | 0.01 | 1.00 | |||||
| Flow velocity | 0.11 | 0.26 | −0.29 | 1.00 | ||||
| Log fecal coliforms | 0.57 | 0.45 | 0.36 | 0.19 | 1.00 | |||
| pH | 0.21 | 0.15 | 0.16 | 0.12 | 0.40 | 1.00 | ||
| True color (color) | 0.51 | 0.63 | −0.31 | 0.29 | 0.33 | −0.08 | 1.00 | |
| Water temperature | 0.48 | 0.20 | 0.30 | 0.03 | 0.26 | 0 | −0.02 | 1.00 |
| Explanatory Variable | Regression Parameters | Baetidae | Leptoceridae | Perlidae | |||
|---|---|---|---|---|---|---|---|
| Coefficient | p-Values | Coefficient | p-Values | Coefficient | p-Values | ||
| Α | 26.95 | 0.11 | 50.48 | 0.02 | 14.17 | 0.03 | |
| BOD5 | Β1 | −12.40 | 0.06 | ||||
| COD | Β2 | 0.75 | 0.10 | −1.12 | 0.05 | ||
| Conductivity | Β3 | 0.13 | 0.04 | −0.08 | 0.06 | 0.05 | 0.04 |
| Flow Velocity | Β5 | −12.27 | 0.04 | 2.31 | 0.17 | ||
| Log Fecal Coliforms | Β4 | 6.41 | 0.04 | −1.89 | 0.02 | ||
| pH | Β6 | −5.31 | 0.03 | ||||
| Temperature | Β7 | −2.86 | 0.07 | −1.43 | 0.03 | ||
| True Color (Color) | Β8 | −0.41 | 0.10 | ||||
| AIC: | 22.52 | 26.52 | 34.46 | ||||
| Adjusted R2: | 59.99% | 67.64% | 43.46% | ||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jerves-Cobo, R.; Everaert, G.; Iñiguez-Vela, X.; Córdova-Vela, G.; Díaz-Granda, C.; Cisneros, F.; Nopens, I.; Goethals, P.L.M. A Methodology to Model Environmental Preferences of EPT Taxa in the Machangara River Basin (Ecuador). Water 2017, 9, 195. https://doi.org/10.3390/w9030195
Jerves-Cobo R, Everaert G, Iñiguez-Vela X, Córdova-Vela G, Díaz-Granda C, Cisneros F, Nopens I, Goethals PLM. A Methodology to Model Environmental Preferences of EPT Taxa in the Machangara River Basin (Ecuador). Water. 2017; 9(3):195. https://doi.org/10.3390/w9030195
Chicago/Turabian StyleJerves-Cobo, Rubén, Gert Everaert, Xavier Iñiguez-Vela, Gonzalo Córdova-Vela, Catalina Díaz-Granda, Felipe Cisneros, Ingmar Nopens, and Peter L. M. Goethals. 2017. "A Methodology to Model Environmental Preferences of EPT Taxa in the Machangara River Basin (Ecuador)" Water 9, no. 3: 195. https://doi.org/10.3390/w9030195
APA StyleJerves-Cobo, R., Everaert, G., Iñiguez-Vela, X., Córdova-Vela, G., Díaz-Granda, C., Cisneros, F., Nopens, I., & Goethals, P. L. M. (2017). A Methodology to Model Environmental Preferences of EPT Taxa in the Machangara River Basin (Ecuador). Water, 9(3), 195. https://doi.org/10.3390/w9030195









