Atmospheric and Surface-Condition Effects on CO2 Exchange in the Liaohe Delta Wetland, China
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description
2.2. EC Measurements
2.3. Abiotic Measurements
2.4. Meteorological Data
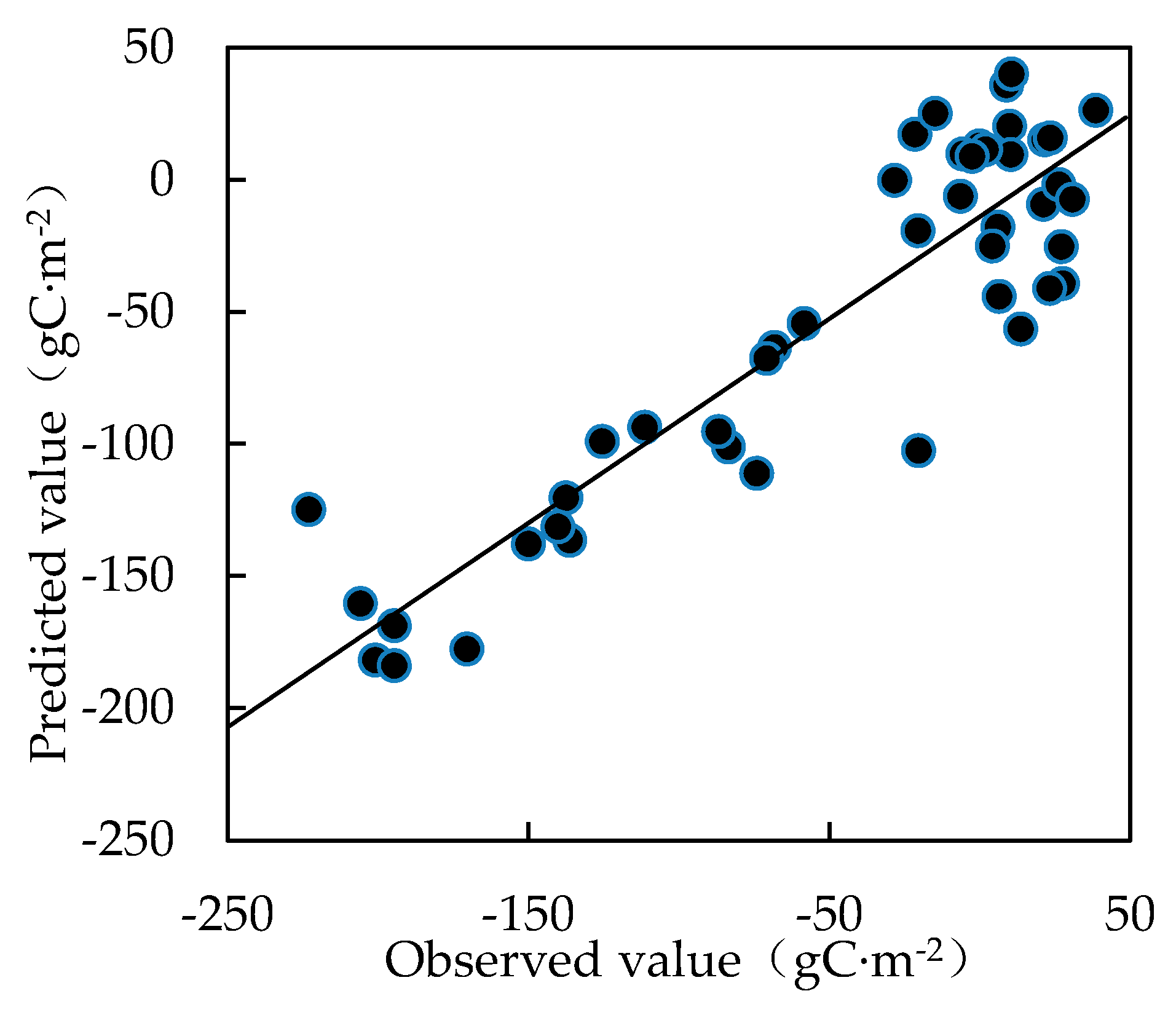
2.5. Partial Least Squares Regression Analysis
3. Results
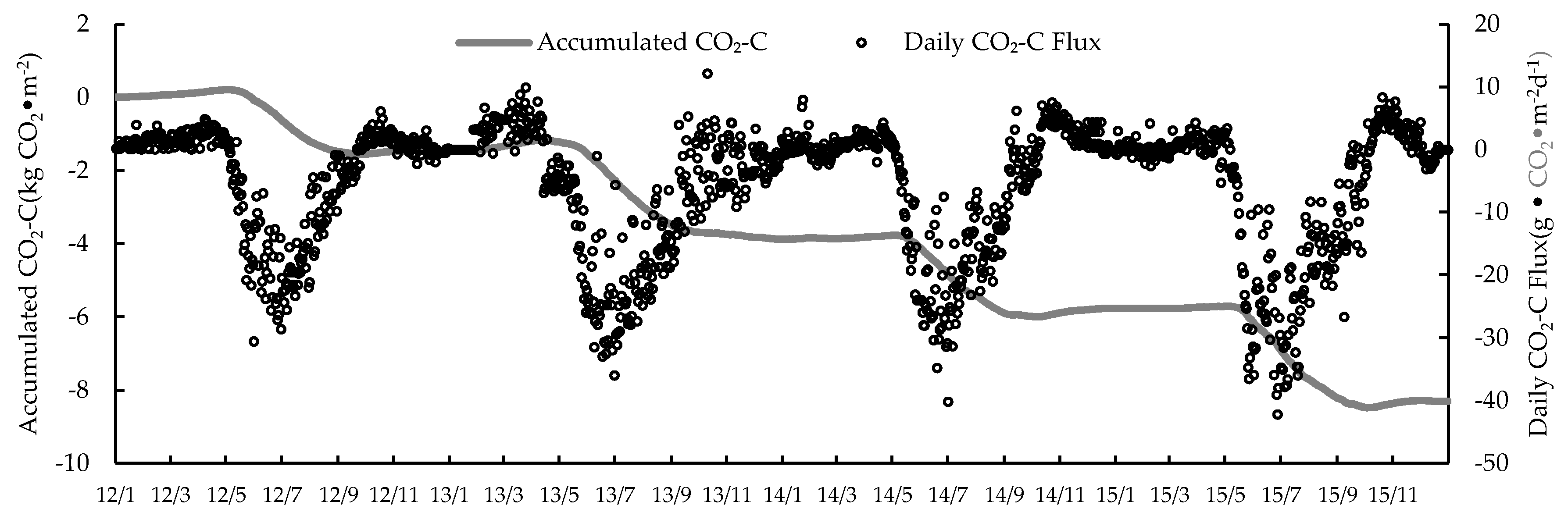
3.1. Seasonal and Inter-Annual Variation of the CO2 Exchange
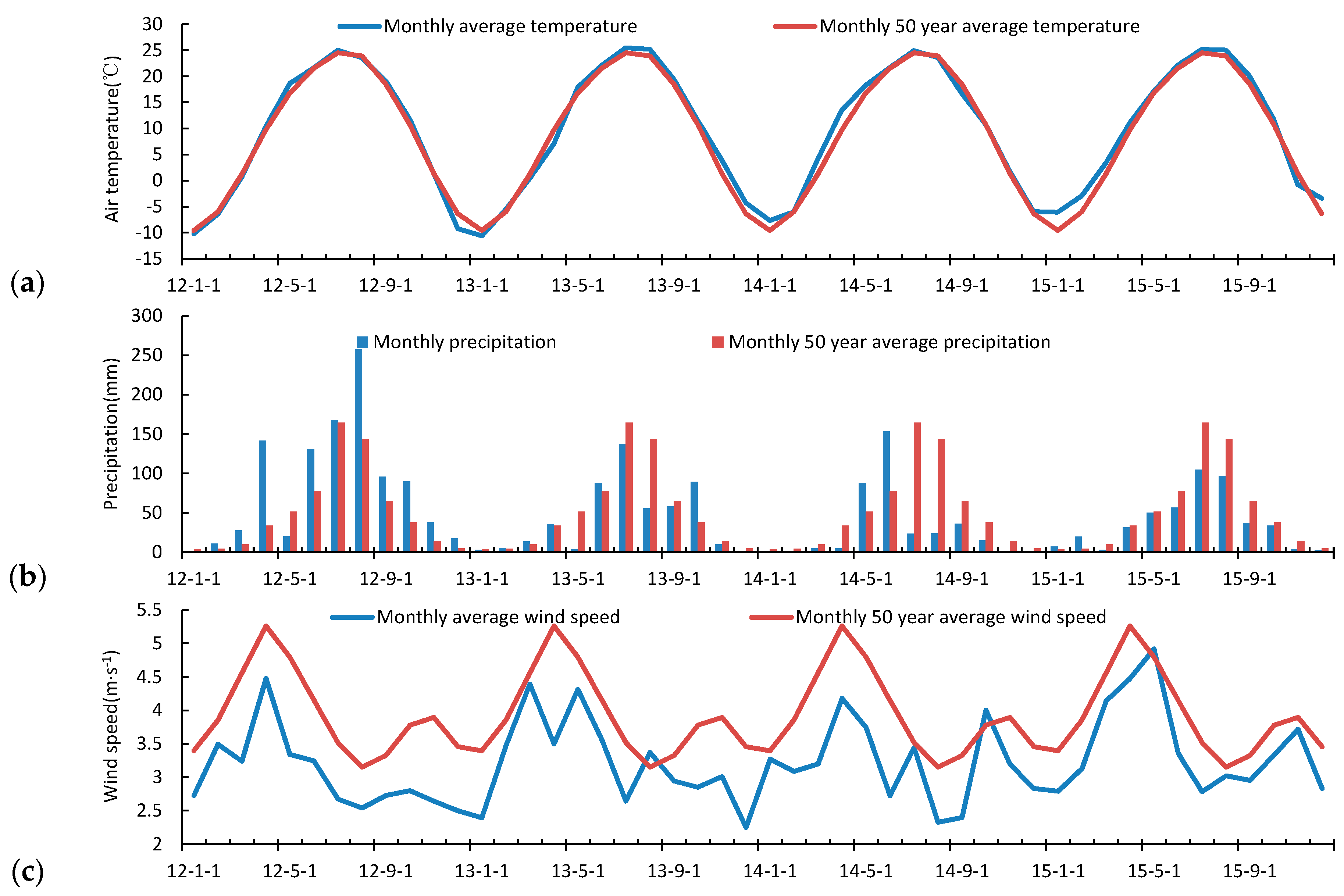
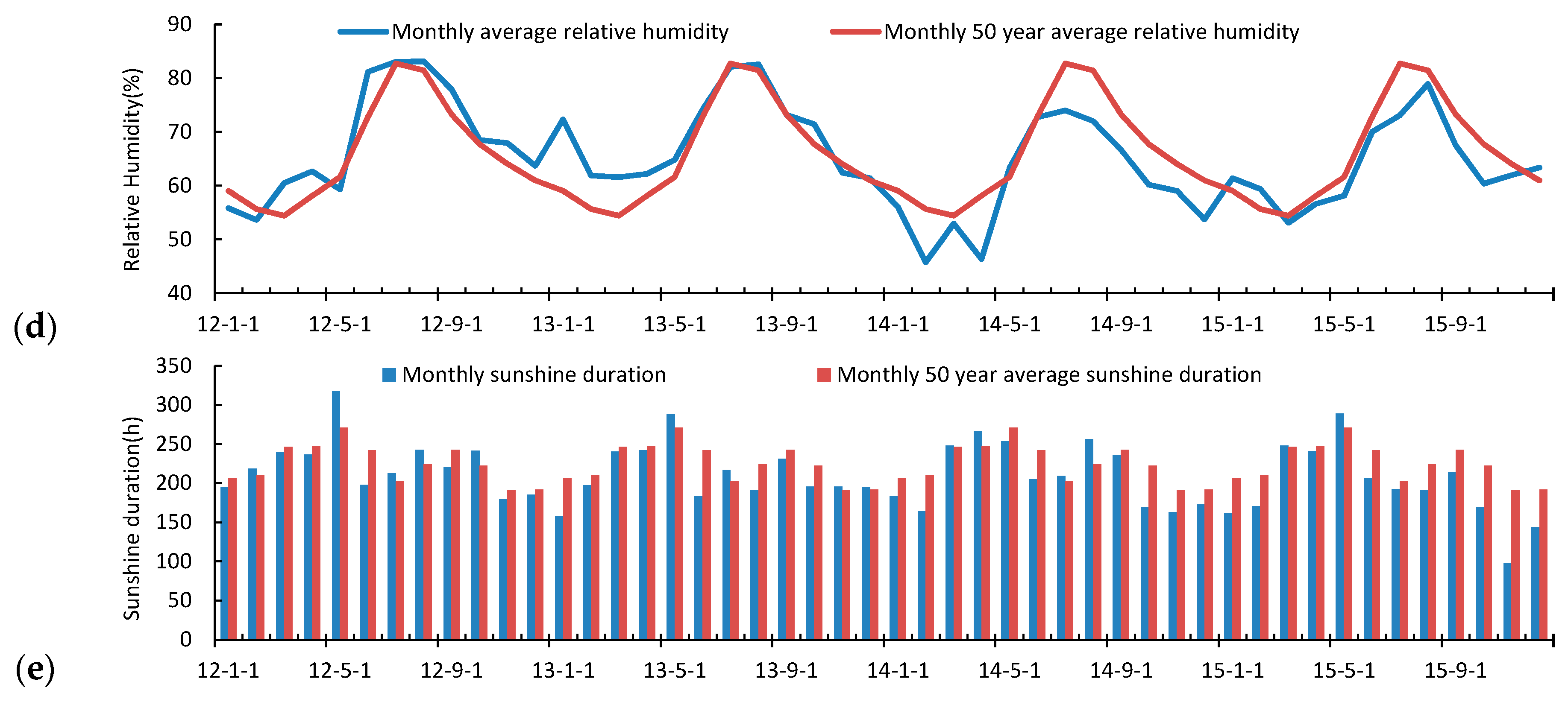
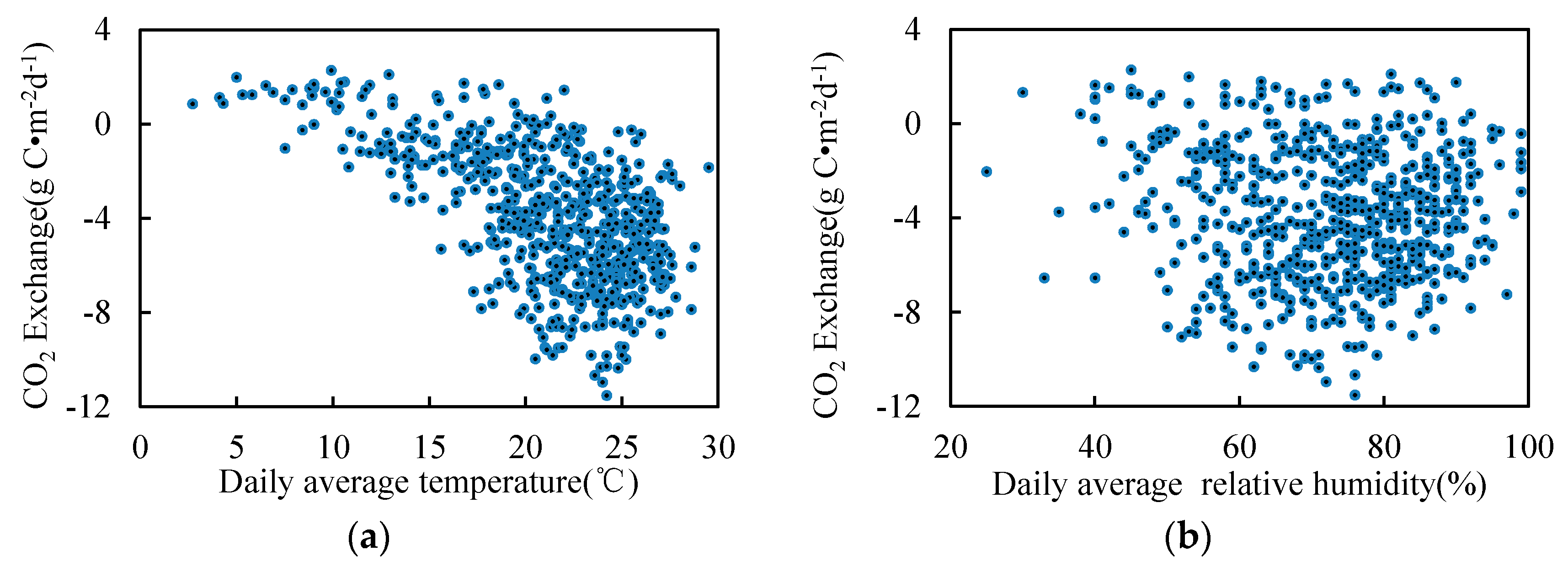
3.2. Seasonal and Inter-Annual Variation of the Atmospheric Measurements
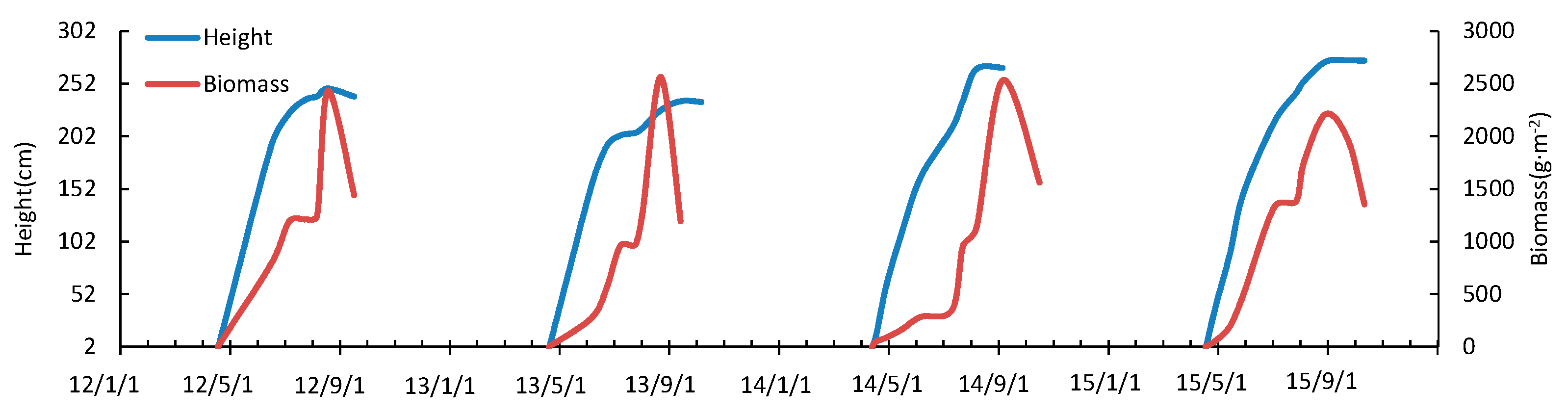
3.3. Seasonal and Inter-Annual Variation of Phragmites Communis
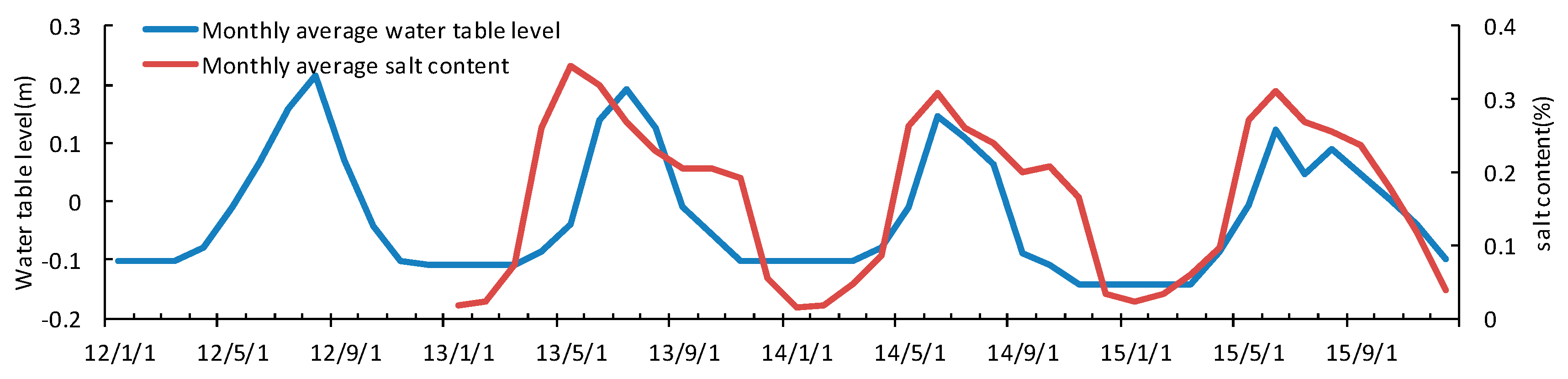
3.4. Seasonal and Inter-Annual Variation of Soil Water and Salinity
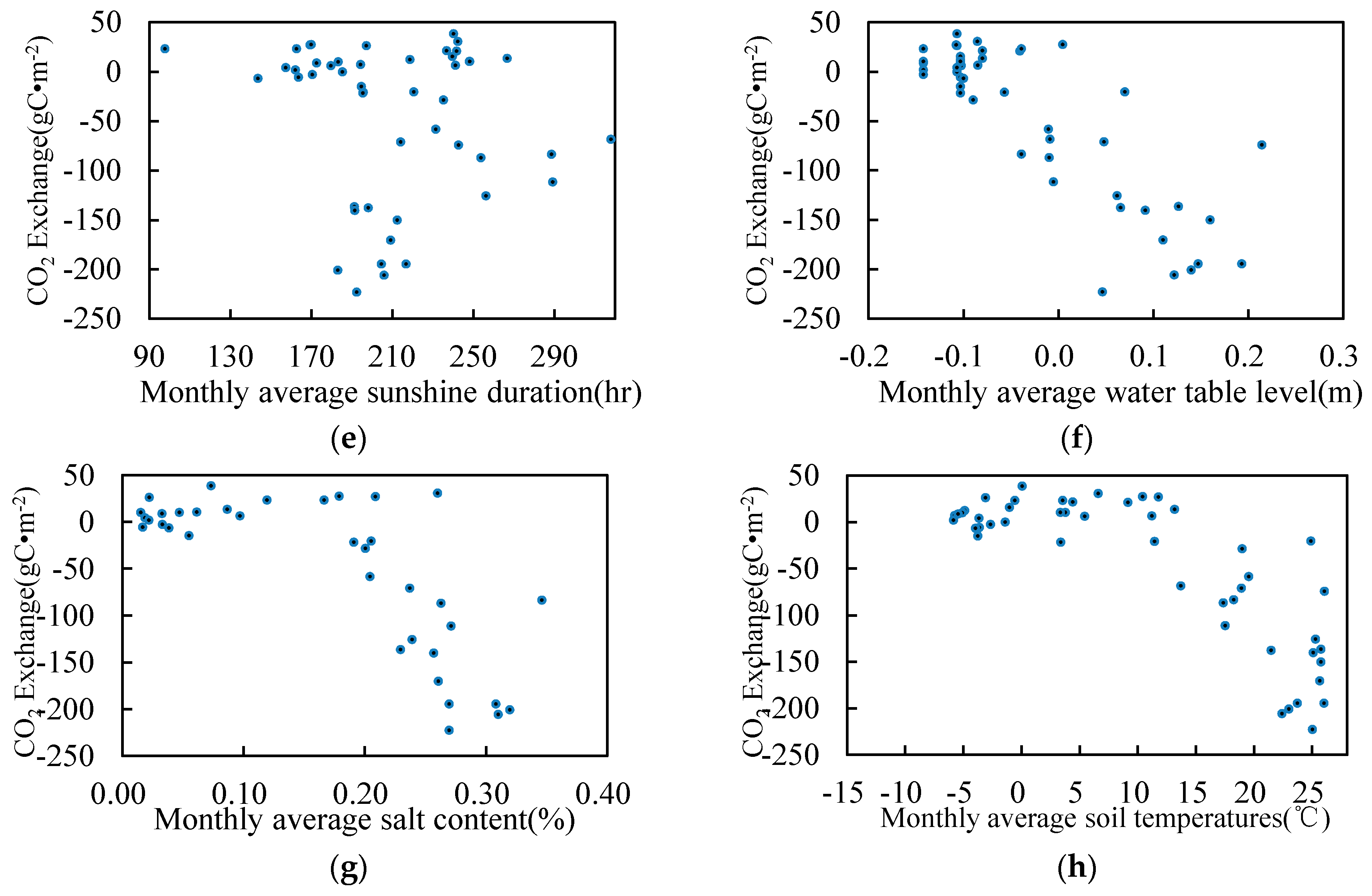
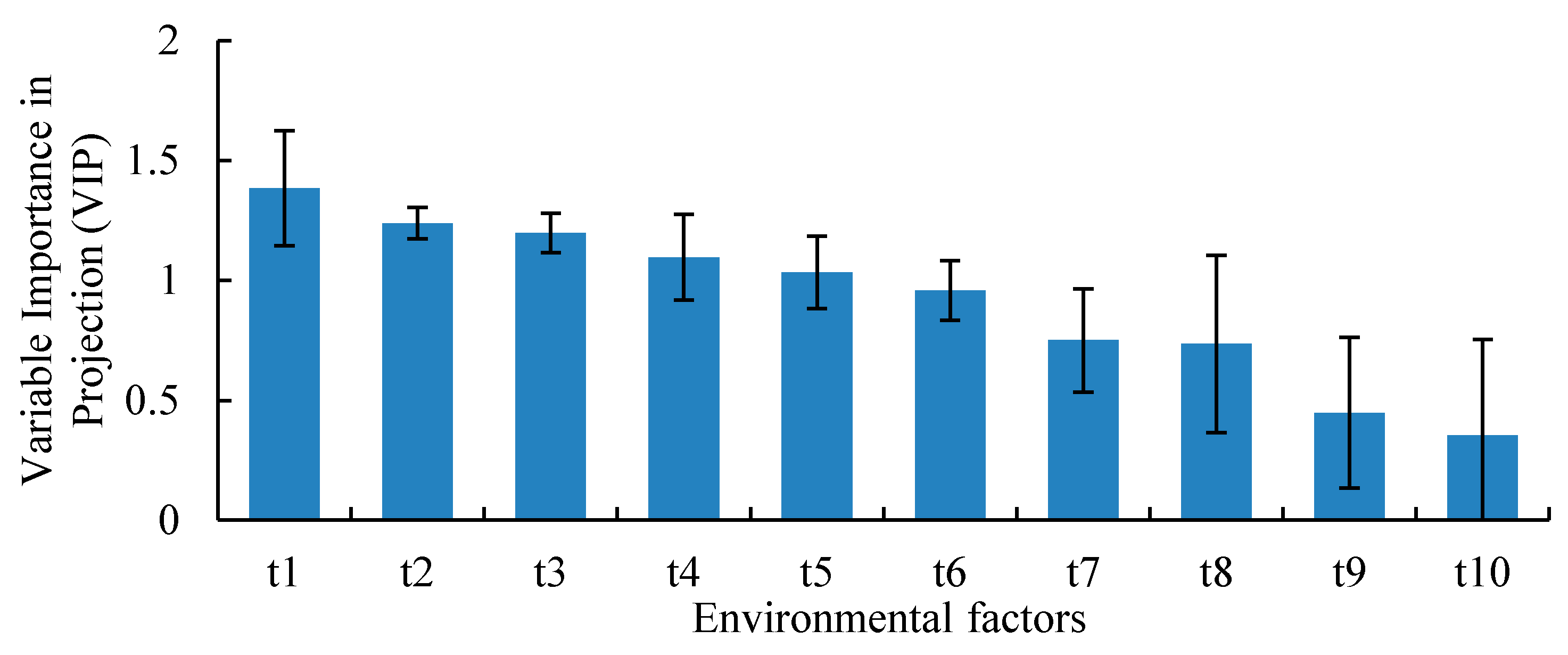
3.5. Relationship between Environmental Factors and CO2
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Davidson, N.C. How much wetland has the world lost? Long-term and recent trends in global wetland area. Mar. Freshw. Res. 2014, 65, 934–941. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Bernal, B.; Nahlik, A.M.; Mander, Ü.; Zhang, L.; Anderson, C.J. Wetlands, carbon, and climate change. Landsc. Ecol. 2012, 28, 1–15. [Google Scholar] [CrossRef]
- Solomon, S.; Qin, D.H.; Manning, M. Climate Change 2007: The Physical Science Basis; Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2007. [Google Scholar]
- Yu, Z. Holocene carbon flux histories of the world’s peatlands: Global carbon-cycle implications. Holocene 2011, 21, 761–774. [Google Scholar] [CrossRef]
- Charman, D.J.; Beilman, D.W.; Blaauw, M.; Booth, R.K.; Brewer, S.; Chambers, F.M. Climate-related changes in peatland carbon accumulation during the last millennium. Biogeosci. Discuss. 2012, 9, 929–944. [Google Scholar] [CrossRef]
- Bu, Z.; Hans, J.; Li, H.; Zhao, G.; Zheng, X.; Ma, J.; Zeng, J. The response of peatlands to climate warming: A review. Acta Ecol. Sin. 2011, 31, 157–162. [Google Scholar] [CrossRef]
- Nilsson, M.; Sagerfors, J.; Buffam, I.; Laudon, H.; Eriksson, T.; Grelle, A.; Klemedtsson, L.; Weslien, P.; Lindroth, A. Contemporary carbon accumulation in a boreal oligotrophic minerogenic mire a significant sink after accounting for all C-fluxes. Glob. Chang. Biol. 2008, 14, 2317–2332. [Google Scholar] [CrossRef]
- Koehler, A.-K.; Sottocornola, M.; Kiely, G. How strong is the current carbon sequestration of an Atlantic blanket bog? Glob. Chang. Biol. 2015, 17, 309–319. [Google Scholar] [CrossRef]
- Kennedy, H.; Alongi, D.M.; Karim, A.; Chen, G.; Chmura, G.L.; Crooks, S. Chapter 4 coastal wetlands. In 2013 Supplement to the 2006 IPCC Guidelines for National Greenhouse Gas Inventories: Wetlands; IPCC: Geneva, Switzerland, 2014. [Google Scholar]
- Baldocchi, D.D. Assessing the eddy covariance technique for evaluating carbon dioxide exchange rates of ecosystems: Past, present and future. Glob. Chang. Biol. 2003, 9, 479–492. [Google Scholar] [CrossRef]
- Lund, M.; Lafleur, P.M.; Roulet, N.T.; Lindroth, A.; Christensen, T.R.; Aurela, M. Variability in exchange of CO2, across 12 northern peatland and tundra sites. Glob. Chang. Biol. 2010, 16, 2436–2448. [Google Scholar] [CrossRef]
- Kerkhoff, A.; Enquist, B.; Elser, J.; Fagan, W. Plant allometry, stoichiometry and the temperature-dependence of primary productivity. Glob. Ecol. Biogeogr. 2005, 14, 585–598. [Google Scholar] [CrossRef]
- Staehr, P.A.; Sand-Jensen, K. Seasonal changes in temperature and nutrient control of photosynthesis, respiration and growth of natural phytoplankton communities. Fresh Water Biol. 2006, 51, 249–262. [Google Scholar] [CrossRef]
- Flanagan, L.B. Stimulation of both photosynthesis and respiration in response to warmer and drier conditions in a boreal peatland ecosystem. Glob. Chang. Biol. 2011, 17, 2271–2287. [Google Scholar] [CrossRef]
- Gedan, K.B.; Bertness, M.D. How will warming affect the salt marsh foundation species Spartina patens and its ecological role? Oecologia 2010, 164, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Kirwan, M.L.; Blum, L.K. Enhanced decomposition offsets enhanced productivity and soil carbon accumulation in coastal wetlands responding to climate change. Biogeosciences 2011, 8, 987–993. [Google Scholar] [CrossRef]
- Charles, H.; Dukes, J.S. Effects of warming and altered precipitation on plant and nutrient dynamics of a New England salt marsh. Ecol. Appl. Publ. Ecol. Soc. Am. 2009, 19, 1758. [Google Scholar] [CrossRef]
- Anna, M.A.; Jianjian, L.U. The Progress of research on carbon flux in wetland ecosystems. Wetl. Sci. 2008, 6, 116–123. [Google Scholar]
- Zhu, X.; Wang, S.; Zhang, C. Responses of different ecotypes of reed growing in the Hexi corridor to natural drought and salinity. Plant Physiol. J. 2003, 39, 371–376. [Google Scholar]
- Saenger, C.; Cronin, T.; Thunell, R.; Vann, C. Modelling river discharge and precipitation from estuarine salinity in the northern Chesapeake Bay: Application to Holocene palaeoclimate. Holocene 2006, 16, 467–477. [Google Scholar] [CrossRef]
- Vretare, V.; Weisner, S.E.B.; Strand, J.A.; Granéli, W. Phenotypic plasticity in Phragmites australis as a functional response to water depth. Aquat. Bot. 2001, 69, 127–145. [Google Scholar] [CrossRef]
- Zhao, K.; Li, J. Effects of salinity on the contents of osmotica of monocotyledenous halophytes and their contribution to osmotic adjustment. Acta Bot. Sin. 2009, 41, 1287–1292. [Google Scholar]
- Zhang, S.; Guo, C.; Su, F. Effect of salinity on the growth of reed. J. Shenyang Agric. Univ. 2008, 39, 65–68. [Google Scholar]
- Weston, N.B.; Vile, M.A.; Neubauer, S.C.; Velinsky, D.J. Accelerated microbial organic matter mineralization following salt-water intrusion into tidal freshwater marsh soils. Biogeochemistry 2011, 102, 135–151. [Google Scholar] [CrossRef]
- Bubier, J.; Crill, P.; Mosedale, A.; Frolking, S.; Linder, E. Peatland responses to varying interannual misture conditions as measured by automatic CO2 chambers. Glob. Biogeochem. Cycles 2003, 17, 1066–1081. [Google Scholar] [CrossRef]
- Chen, G. Study on Marsh in Sanjiang Plain; Science Press: Beijing, China, 1996; pp. 165–168. [Google Scholar]
- Minchinton, T.E. Disturbance by wrack facilitates spread of Phragmites australis in a coastal marsh. J. Exp. Mar. Biol. Ecol. 2002, 281, 89–107. [Google Scholar] [CrossRef]
- Robroek, B.J.M.; Schouten, M.G.C.; Limpens, J.; Berendse, F.; Poorter, H. Interactive effects of water table and precipitation on net CO2 assimilation of three co-occurring sphagnum mosses differing in distribution above the water table. Glob. Chang. Biol. 2009, 15, 680–691. [Google Scholar] [CrossRef]
- Daulat, W.E.; Clymo, R.S. Effects of temperature and watertable on the efflux of methane from peatland surface cores. Atmos. Environ. 1998, 32, 3207–3218. [Google Scholar] [CrossRef]
- Frolking, S.; Roulet, N.T. Holocene radiative forcing impact of northern peatland carbon accumulation and methane emissions. Glob. Chang. Biol. 2010, 13, 1079–1088. [Google Scholar] [CrossRef]
- Heinsch, F.A.; Heilman, J.L.; Mcinnes, K.J.; Cobos, D.R.; Zuberer, D.A. Carbon dioxide exchange in a high marsh on the Texas Gulf Coast: Effects of freshwater availability. Agric. For. Meteorol. 2004, 125, 159–172. [Google Scholar] [CrossRef]
- Fenner, N.; Freeman, C. Drought-induced carbon loss in peatlands. Nat. Geosci. 2011, 4, 895–900. [Google Scholar] [CrossRef]
- Han, G.; Yang, L.; Yu, J.; Wang, G.; Mao, P.; Gao, Y. Environmental Controls on Net Ecosystem CO2 Exchange Over a Reed (Phragmites australis) Wetland in the Yellow River Delta, China. Estuaries Coasts 2013, 36, 401–413. [Google Scholar] [CrossRef]
- Thomas, K.L.; Benstead, J.; Davies, K.L.; Lloyd, D. Role of wetland plants in the diurnal control of CH4 and CO2 fluxes in peat. Soil Biol. Biochem. 1996, 28, 17–23. [Google Scholar] [CrossRef]
- Mei, X.; Zhang, X. Carbon storage and fixation by a typical wetland vegetation in Changjiang River Estuary—A case study of Phragmites australis in east beach of Chongming Island. Chin. J. Eco-Agric. 2008, 16, 269–272. [Google Scholar] [CrossRef]
- Li, B.; Liu, C.-Q.; Wang, J.-X.; Zhang, Y.-J. Carbon Storage and Fixation Function by Phragmites australis, a Typical Vegetation in Baiyangdian Lake. J. Agro-Environ. Sci. 2009, 28, 2603–2607. [Google Scholar]
- Qi, Y. Study on the allocation scheme of multi-source ecological water supply of Panjin reed wetland. Water Resour. Hydropower Northeast China 2015, 33, 19–21. [Google Scholar]
- Sagerfors, J.; Lindroth, A.; Grelle, A.; Klemedtsson, L.; Weslien, P.; Nilsson, M. Annual CO2 exchange between a nutrient-poor, minerotrophic, boreal mire and the atmosphere. J. Geophys. Res. 2008, 113, G01001. [Google Scholar] [CrossRef]
- Lv, X. Wetland Ecosystem Observation Method; China Environmental Science Press: Beijing, China, 2005; pp. 94–99. [Google Scholar]
- Krishnan, A.; Williams, L.J.; Mcintosh, A.R.; Abdi, H. Partial least squares (PLS) methods for neuroimaging: A tutorial and review. Neuroimage 2011, 56, 455–475. [Google Scholar] [CrossRef] [PubMed]
- Wold, S.; Sjöström, M.; Eriksson, L. PLS-regression: A basic tool of chemometrics. Chemom. Intell. Lab. Syst. 2001, 58, 109–130. [Google Scholar] [CrossRef]
- Giuseppe, P.; Paolo, P.; Hans-Dieter, Z. Performance of PLS regression coefficients in selecting variables for each response of a multivariate PLS for omics-type data. Adv. Appl. Bioinform. Chem. 2009, 2, 57–70. [Google Scholar]
- Johnson, R.A.; Wichern, D.W. Applied Multivariate Statistical Analysis; Prentice Hall: New Jersey, NJ, USA, 2002. [Google Scholar]
- Sagerfors, J. Land-Atmosphere Exchange of CO2, Water and Energy at a Boreal Minerotrophic Mire. Ph.D. Thesis, Swedish University of Agricultural Sciences, Umea, Sweden, January 2007. [Google Scholar]
- Peichl, M.; Sonnentag, O.; Nilsson, M.B. Bringing color into the picture: Using digital repeat photography to investigate phenology controls of the carbon dioxide exchange in a boreal mire. Ecosystems 2014, 18, 115–131. [Google Scholar] [CrossRef]
- Sun, B.; Xie, J.C.; Wang, N.; Li, S.-Q.; Li, C.-J. Effect of reeds on salt enrichment and improvement of saline-alkali land. J. Soil Water Conserv. 2012, 26, 92–101. [Google Scholar]
- Hair, J.; Black, W.; Babin, B.; Anderson, R. Multivariate Data Analysis: Pearson New International Edition PDF eBook; Pearson: Zug, Switzerland, 2013; Volume 3, pp. 128–134. [Google Scholar]
- Li, Y.; Cui, L.; Pan, X.; Ning, Y.; Li, W.; Kang, X. Spatial distribution of plant diversity and functional groups in the Liaohe estuary. Biodivers. Sci. 2015, 23, 471–478. [Google Scholar] [CrossRef]
- Kang, X.M.; Cui, L.J.; Yue, X.L.; Li, W.; Zhang, M.Y.; Zhao, X.S.; Hao, Y.B.; Lei, Y.R.; Gao, Q. Valuation of Air Regulation by the Reed (Phragmites australis) Wetland in the Yellow River Delta. Wetl. Sci. Manag. 2015, 11, 23–25. [Google Scholar]





| Depth (cm) | pH | Organic Matter (g·kg−1) | Total N (g·kg−1) | Nitrate N (mg·kg−1) | Ammonium N (mg·kg−1) | Alkali-Hydrolysis N (mg·kg−1) | Total P (mg·kg−1) | Available P (mg·kg−1) | Total K (g·kg−1) | Available K (g·kg−1) | C/N |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0–20 | 8.80 ± 0.2 | 19.4 ± 10 | 1.55 ± 0.1 | 4.21 ± 1.4 | 14.75 ± 6.0 | 67.2 ± 26 | 35 ± 6 | 14.50 ± 0.7 | 2.86 ± 0.3 | 0.75 ± 0.05 | 6.8 ± 1.6 |
| 20–40 | 9.27 ± 0.1 | 9.8 ± 3 | 1.65 ± 0.1 | 6.24 ± 0.9 | 4.33 ± 0.5 | 30.8 ± 14 | 22.62 ± 1.4 | 0.83 ± 0.32 |
| Year | Reed Wetland (ppm) | Standard Deviation SD |
|---|---|---|
| 2012 | 375.14 | 24.67 |
| 2013 | 366.78 | 21.76 |
| 2014 | 383.65 | 11.90 |
| 2015 | 397.84 | 14.77 |
| Year | Ta (°C) | PPT (mm) | RH (%) | Sunshine Duration (h) | WS (m·s−1) |
|---|---|---|---|---|---|
| 2012 | 8.88 | 999 | 68.13 | 2688.2 | 3.03 |
| 2013 | 9.48 | 499.8 | 69.22 | 2534.8 | 3.22 |
| 2014 | 9.71 | 350.4 | 60.32 | 2525.9 | 3.20 |
| 2015 | 10.26 | 447.1 | 63.67 | 2326.8 | 3.45 |
| 1961–2010 | 8.92 ± 0.73(±SD) | 611.2 ± 158.3(±SD) | 65.97 ± 2.47(±SD) | 2697.6 ± 192.3(±SD) | 3.93 ± 0.65(±SD) |
| Factors | Tolerance | VIF |
|---|---|---|
| t1 | 0.150 | 6.673 |
| t2 | 0.017 | 57.506 |
| t3 | 0.017 | 59.054 |
| t4 | 0.405 | 2.471 |
| t5 | 0.104 | 9.629 |
| t6 | 0.451 | 2.215 |
| t7 | 0.087 | 11.540 |
| t8 | 0.361 | 2.770 |
| t9 | 0.070 | 14.303 |
| t10 | 0.274 | 3.653 |
| Factor | R2X (cum) | R2Y (cum) | Q2 (cum) |
|---|---|---|---|
| The first group (Total factors) | 0.567 | 0.647 | 0.629 |
| The second group (Tair, P, RH) | 0.9 | 0.621 | 0.574 |
| The third group (WTL, SAL, TSoil) | 0.9 | 0.673 | 0.661 |
| The fourth group (H, Biomass) | 0.907 | 0.162 | 0.091 |
| The fifth group (Tair, P, RH, WTL, SAL, Tsoil, Biomass) | 0.885 | 0.781 | 0.711 |
| The sixth group (Tair, P, RH, WTL, SAL, Biomass) | 0.0898 | 0.781 | 0.725 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, Q.; Yu, W.; Zhou, L.; Liang, C. Atmospheric and Surface-Condition Effects on CO2 Exchange in the Liaohe Delta Wetland, China. Water 2017, 9, 806. https://doi.org/10.3390/w9100806
Jia Q, Yu W, Zhou L, Liang C. Atmospheric and Surface-Condition Effects on CO2 Exchange in the Liaohe Delta Wetland, China. Water. 2017; 9(10):806. https://doi.org/10.3390/w9100806
Chicago/Turabian StyleJia, Qingyu, Wenying Yu, Li Zhou, and Chenghua Liang. 2017. "Atmospheric and Surface-Condition Effects on CO2 Exchange in the Liaohe Delta Wetland, China" Water 9, no. 10: 806. https://doi.org/10.3390/w9100806
APA StyleJia, Q., Yu, W., Zhou, L., & Liang, C. (2017). Atmospheric and Surface-Condition Effects on CO2 Exchange in the Liaohe Delta Wetland, China. Water, 9(10), 806. https://doi.org/10.3390/w9100806





