Abstract
This work presents a selective overview of natural fogs in terms of fog types, forms and states of occurrence, physical, micro-physical, chemical and dynamic properties, basic characterizing parameters, etc. In focus are related achievements and contributions reported mainly during the last decade and a half, as a result of both laboratory studies and field observations. Processes of homogeneous and heterogeneous nucleation are analyzed in the aspects of condensation, nuclei diversity and specifics, as related to the activation, growth and deposition of fog droplets. The effect is highlighted of the water vapor’s partial pressure on the surface tension of the liquid water–air interface and the freezing point of the water droplets. Some problems and aspects of fog modeling, parameterization, and forecasting are outlined and discussed on the examples of newly developed relevant 1D/3D theoretical models. Important issues of fog impacts on the air quality, ecosystems, water basins, societal life, and human health are also addressed and discussed, particularly in cases of anthropogenically modified (chemical, radioactive, etc.) fogs. In view of reducing the possible negative effects of fogs, conclusions are drawn concerning the new demands and challenges to fog characterization imposed by the changing natural and social environment and the needs for new data on and approaches to more adequate observations of fog-related events.
1. Introduction
Fogs consist of a large amount of small liquid water droplets or ice crystals suspended in a certain air volume near the ground. The physical nature of fogs and clouds is the same. One can state that fog is a cloud touching the ground’s surface. Fogs reduce the visibility in the surrounding area—according to its meteorological definition [1], it is a state of the atmospheric air with visibility lower than 1 km, as measured near the surface.
Besides the hydrosphere, fogs present a form of liquid water in the atmosphere of the Earth. Generally, fog formation and existence are under the strong influence of local orographic factors, the actual synoptic situation, and the atmospheric circulations. It can be formed everywhere—over land and water surfaces. Although it is not a climate-forming factor, it is of great meteorological importance, particularly due to its local character of formation, its capability of reducing the temperature amplitude, and the fact that it is directly related to the humidity parameters.
Natural fog, as a form of condensed water existing in the atmosphere, has significant impacts on many components of the environment, such as the global and regional climate, the atmosphere’s thermal and radiative budget, air quality, waters, flora and fauna, air-surface interactions, etc. [2,3,4]. At the same time, as resulting in reduced visibility, fogs can perturb and affect severely the societal life and functionality (e.g., air-, surface-, and water transport) causing an impressive number of injures and fatalities [5]. Depending on the physical and chemical nature and composition of the droplets, fogs can also have direct or indirect adverse effects on human health (respiratory and radiation diseases, skin and eye damages, secondary health effects, etc.) [6,7].
Because of the various and some even severe influences of fogs on the environment and human activities and health, fog research and studies have a long and rich history. In a comprehensive review paper, Gultepe et al. [5] made a profound analysis of the experimental and theoretical contributions and achievements of fog-related research, including a detailed historical overview of the subject and highlighting problems of fog modeling and forecasting. Nowadays, scientific reports devoted to measurement, characterization and applications of fogs continue to appear in large numbers (e.g., [4,8,9]).
The present review considers important aspects of natural fogs. It is not aimed to survey comprehensively the fog-related scientific literature. Instead, the main purpose of the work is to summarize recent (mostly published during the last decade and a half) research contributions and achievements concerning selected aspects of the formation, collection, characterization, classification, and impacts of natural fogs.
The contents of this publication are structured as follows: in Section 2, basic characteristics of water and fog are discussed, in particular, specifics of the nucleation and condensation processes. Fog’s formation, conditioning, and classification are explored in Section 3. The important effect of water vapor’s partial pressure on the surface tension of the liquid water–air interface is handled in Section 4. Section 5 is devoted to fog modeling, parameterization, and forecasting. Fog’s impact on the environment, human health, and societal activities is analyzed in Section 6. Basic conclusions of the work are summarized in Section 7.
2. Fog—Physical Basis, General Characterization, and Classification
2.1. Physical Basis of the Fog
More than 70% of Earth’s surface is covered by water. Its molecule is composed of two hydrogen atoms and a single oxygen atom linked by covalent bonds. Due to the unique physical and chemical properties of these elements, as well as to its structural/phase variability and abundance, water is the base of the terrestrial organic life; its existence and balance is essential for all living species on Earth.
Water exists in all three phases of matter: solid (ice), liquid (liquid water), and gaseous (water vapor). Depending on the combination of the ambient temperature T and pressure P, transitions occur between the three phases of water. The pressure-temperature (P-T) diagram conditioning the phases’ existence, coexistence, and transitions is also known as the water phase diagram [10]. The discontinuous phase transitions between two water phases are characterized mathematically by the Clausius–Clapeyron relation:
where L is the specific latent heat and is the specific volume change accompanying the phase transition.
According to the Clausius–Clapeyron relation, the equilibrium vapor pressure of the atmosphere (known also as “water-holding capacity”) increases by about 7% per each 1 °C temperature rise. The water-holding capacity can be expressed approximately by the August–Roche–Magnus formula:
where is the equilibrium, or saturation, vapor pressure in hPa and the temperature T is in degrees Celsius. According to this formula, an increase of the atmospheric temperature (e.g., due to greenhouse effect) results in a corresponding exponential increase of the atmospheric absolute humidity for a constant relative humidity. It should be noted that the applicability of this deduction to atmospheric phenomena is debatable and could not be valid in cases of convective processes, which cause air drying, cloud formation, etc., due to relative humidity variations [11].
2.2. Water Condensation Process: Droplet Nucleation—Conditions and Analysis
Nucleation—the process of forming droplet nuclei—is of great significance for ultrafine-mode particle, cloud, and fog formation. Two types of nucleation can be distinguished—homogeneous and heterogeneous, depending on the absence or presence of foreign nuclei/substances, respectively [12].
Homogeneous nucleation occurs spontaneously and randomly, but it requires that the water-vapor critical supersaturation, or critical supercooling, be exceeded [13,14]. It is responsible for the formation of new particles, which subsequently can serve as cloud condensation nuclei (CCN, see below). Cases have been observed of the CCN number increasing by a factor of more than two over the course of a single day due to newly formed particles. Numerous very tiny particles (with diameters of 3–15 nm) have been detected near clouds and close to the tropopause. The formation of ultrafine particles (diameter around few nm) and their subsequent growth to about 100 nm in the next two days has been observed in the continental boundary layer [15].
Nucleation from a gas phase is the process responsible for the formation of a significant fraction of the total number of atmospheric particles. Four nucleation mechanisms are considered to be the most important ones, each of them taking place in a different part of the atmosphere [15,16,17]:
- In industrial plumes and the free troposphere, the main nucleation mechanism is homogeneous binary water-sulfuric acid nucleation;
- In the continental boundary layer, the most common one is homogeneous ternary water-sulfuric acid-ammonia nucleation;
- In the upper troposphere and lower stratosphere, ion-induced nucleation of binary or ternary inorganic vapors or of organic vapors take place;
- In coastal regions, the main mechanism is barrierless homogeneous nucleation of iodide species.
For drop formation in homogeneous water vapor (condensation of water vapor molecules in the absence of foreign condensation nuclei), supersaturations as high as several hundred percent would be necessary. Such high levels of supersaturation do not occur in the atmosphere. This is why cloud/fog droplets or ice particles do not form by homogeneous nucleation of supersaturated water vapor [13,18]. The typical supersaturations observed in the atmosphere remains below 10% and most often even below 1% [13].
An important factor facilitating water-vapor condensation into droplets is the presence of hydrophilic substances/particles in the atmosphere (heterogeneous nucleation). Droplets can grow to diameters of several micrometers to tens of micrometers. Condensation of supersaturated water vapor on soluble aerosol particles and on insoluble but wettable particles is responsible for fog/cloud droplets and ice particles formation. Particles having the potential to nucleate liquid cloud/fog droplets are called cloud condensation nuclei, whereas aerosol particles inducing formation of ice crystals are named ice nuclei [18,19]. The ability of a certain type of particles to act as CCN at a given supersaturation level depends on factors such as size, solute content, presence of surface-active substances, wettability and shape of insoluble particles, and the presence of soluble gases. CCN originate from all mechanisms that lead to the formation of atmospheric aerosols [13,18].
The different aerosol particle types are briefly presented below. The aerosol types can be classified according to their size, origin (natural or anthropogenic), physical properties (liquid, solid, organic), sources (primary or secondary), geographical origin (desert, polar, continental, marine, rural, urban). The atmospheric aerosol particles can be distinguished into different modes according to their diameters [20,21,22,23,24]:
- Fine particles:
- ○
- Nucleation mode: 0.001–0.01 µm,
- ○
- Aitken mode: 0.01–0.1 µm,
- ○
- Accumulation mode: 0.1–(1 to 2.5) µm,
- Coarse mode: >(1 to 2.5) µm,
- Giant particles: >10 µm.
The Aitken mode particles (also called Aitken nuclei) consist of sulfates, elemental carbon, metal compounds and organic compounds with a very low saturation vapor pressure at ambient temperature.
Accumulation mode particles encompass the coagulation of the smaller Aitken nuclei, sulfate (SO), nitrate (NO), ammonium (NH), and hydrogen (H+) ions, as well as elemental carbon, a large variety of organic compounds, and metal compounds of Pb, Cd, Ni, V, Cu, Zn, Mn, Fe, etc. [25].
The coarse mode is composed of suspended soil or street dust; ash and soot resulting from uncontrolled combustion of coal, oil and wood; nitrates and chlorides as HNO3 and HCl; oxides of crustal elements (Si, Al, Ti, Fe); CaCO3, NaCl and sea salts; pollen, mold and fungal spores, as well as plant and animal fragments [25].
The Aitken mode particles and the accumulation particles belong to the group of secondary aerosol particles. Aitken nuclei are made up of particles that are by-products of combustion (mainly from anthropogenic sources), as well as particles that are products of the gas-to-particle conversion process, which is considered as a natural source if the precursor gases are not anthropogenic (terpenes emitted by plants, for example). The coarse mode particles have direct sources (mainly natural) and fall into the group of primary aerosol particles [16,25,26].
The sulfate aerosols are considered as being the most effective CCN, but, recently, the organic aerosols received particular attention and are now thought of as having activation capability comparable to that of sulfate aerosols. Organic CCN can arise from primary organic aerosols and secondary organic aerosols and be of both anthropogenic and natural origin [25,26]. Combustion products originating from vehicular exhaust and biomass burning, as well as biogenic emissions, are the main sources of primary organic CCN, while secondary biogenic aerosols, secondary anthropogenic organic aerosols, and the oxidation products of soot are recognized as being the main sources of secondary CCN. Aerosols containing whole organisms, reproductive material, fragments, decaying organic matter, and products of bubble-bursting processes generated in the sea represent primary biogenic organic aerosols. Oxidation processes of volatile organic compounds (VOCs) emitted from biological organisms form secondary biogenic organic aerosols [26]. Some of the organic aerosols able to act as CCN, are:
- Monocarboxylic acids (MCA) and dicarboxylic acids (DCA)—species observed predominantly in the gas phase, aerosols, precipitation-, cloud- and fog water, are the formic and acetic acids (belonging to MCA). In aerosol particles, DCA (oxalic acid, malonic, succinic acid) dominate, constituting only a small fraction of the total particulate water-soluble organic components in the atmosphere [26].
- Humic-like substances (HULIS)—fine particles with good water solubility consisting of polysaccharide and aliphatic substructures. Major components of continental organic aerosols are HULIS, which can also be of biogenic origin. They are considered as affecting the aerosol hygroscopicity, as well as the formation of CCN. HULIS have also been identified in fog droplets with a scavenging ratio similar to that of inorganic ions [26].
- Bacteria (0.25–8 µm in diameter)—a group of very metabolically diverse, prokaryotic, unicellular microorganisms. Sources of bacteria are found to be temperate vegetation zones, as raw crop areas (high primary production) and desert areas (relatively low production). Living and dead bacteria have been observed in clouds and fog, raindrops and hailstones, as well as in different parts of the atmosphere—boundary layer, upper troposphere, stratosphere (up to 41 km above sea level). Among the bacteria able to act as CCN at low saturation ratios (from 0.07% to 1%) are plant pathogenic bacteria Erwinia carotovora, as well as Gram-positive and Gram-negative bacteria—Micrococcus agilis, Mycoplana bullata, and Brevundimonas diminuta [26].
- Total and dissolved organic carbon (TOC and DOC)—significant amounts of organic carbon (C) have been found in fog and clouds. Its concentration varies from 1 mg C/L (in remote marine environments) to 100 mg C/L (in polluted radiation fogs). Very high concentrations, between 100 and 200 mg C/L, have been measured in biomass burning impacted clouds. In fog droplets, most of the organic matter is considered to be DOC [27].
CCN with dry particle diameters >5 µm are often defined as Giant CCN (GCCN). They represent a small fraction of all particles and can be activated at very low supersaturations (≤0.02%). Before droplet activation, these particles are able to grow to diameters larger than 10 µm and act as collector drops. GCCN reduce the peak supersaturation in clouds—due to their large surface, they inhibit the activation of smaller particles, thus preventing the latter from taking part in cloud shaping [18].
3. Formation and Types of Fog
Fog and clouds are a natural state of water condensates. The main difference between them is their place in altitude and the fact that the fog is closely near the ground surface. The difference in the size of droplets is also significant. In clouds, they may reach diameters of up to 6 mm. However, in mist and haze—other forms of water condensates in the free atmosphere very similar to fog—their diameters are less than 150 µm. Usually, the size of droplets in mist ranges between 50 µm and 150 µm. It is considered to be below 100 µm in fog and well below 1 µm in haze. In meteorological terms, they are classified with respect to their visibility—in cases of haze and mist, it is higher than 1 km. In mist, a high value of relative humidity should be present, while it should be below 80% in the case of haze.
The natural formation of fog starts when an air mass of relatively high humidity content comes in contact with a colder earth surface and thus cools down to the dew point.
3.1. Fog Types by Phase of Droplet
If the particles composing the fog have a hydro-meteorological origin—water vapor or ice—the fog is classified as a wetfog or as an icefog, respectively [5,28]. At temperatures from 0 °C down to −12 °C, the fog can be in a liquid state, depending on the humidity [29]. Below −30 °C, it is composed of ice crystals and is called an ice fog [28,30].
3.2. Fog Types by the Kind of Particles: Radioactive and Chemical Fogs
Upon fog formation, with all the condensation conditions present (high relative humidity, low temperature, mild wind, etc.), its droplets collect various aerosols spread in the air. In some particular cases, radioactive aerosol pollutants can serve as condensation nuclei. Some of the airborne radioactive isotopes are positively charged (e.g., 7Be and 210Pb) [31]. The positive charge increases their hygroscopicity and, hence, the possibility of being associated with the polar water molecules, thus enhancing the fog formation efficiency. Radioactive aerosols can be of either natural (e.g., volcanic) or artificial origin. The latter could appear in the atmosphere as a result of imperfect treatment or storage of radioactive products in nuclear technologies, past nuclear tests, or, rarely, as a result of accidental reactor releases (e.g., Chernobyl, Fukushima) [32]. Natural or artificial biomass-burning can also increase the radioactive aerosols content in fogs and clouds. Bourcier et al. [33] have provided experimental evidence of biomass burning as a source of atmospheric 137Cs present in cloud water. Fog droplets capturing radio-nuclides could either prolong or shorten the residence time of the radioactive atmospheric pollutants, by preventing fast deposition or enhancing it by scavenging [34]. Thus, fogs can play an important role in spreading, keeping close to the surface, and in wet deposition of radioactive particles, with direct impacts on the human health and ecosystems. However, difficulties arise in reliably detecting and quantifying the amounts of radio-nuclides in fog/cloud water because of their low absolute concentrations being below the activity detection thresholds. Thus, large amounts of fog water should be sampled to obtain reliable measurements. This is why a few works only have been published dealing with radioactive fog. Gibb et al. [35] have studied radionuclide deposition velocities. Presence of 7Be and 210Pb radio-nuclides in cloud water and rain water has been reported by Su and Huh [36]. They have observed specific activities of 7Be and 210Pb in cloud water samples spanning three orders of magnitude, in strong correlation with the samples’ acidity. Bourcier et al. [33] were the first to determine the 137Cs background level in cloud water and calculate the radionuclides’ scavenging efficiencies for 7Be, 210Pb and 137Cs.
3.3. Fog Types by Dynamical Origin
Radiation fogs are formed as a result of the nocturnal radiative cooling of the earth surface. The temperature of the air layer closest to the ground decreases via heat-transfer and, if the dew point is reached, fog generation starts from the ground rising up with the altitude [8]. Decisive conditions for such fog formation are temperature inversion and moderate wind speeds between 1 and 3 m/s (around 0.5–1.5 knots), while stronger winds would prevent the appearance of fog. Radiation fogs are usually formed in autumn, when the ground is wet, abundant evaporation takes place, and the nights are long enough for the temperature to fall significantly. Their formation can be stimulated by the orography—in valleys, due to the exchange with the free atmosphere, temperatures can decrease easily. After sunrise, the ground slowly warms up, the temperature inversion starts vanishing and fog becomes progressively less dense [37].
Advection fogs are formed when a warm moist air mass moves to a cold surface, e.g., from a warm lake over a cold ground. As a result of the heat exchange with the cold surface, the air layers closest to the ground cool faster than the upper ones and, if the air mass is sufficiently moist, condensation starts.
Mixed type fogs—advection-radiation fogs are generally formed over continents during the cold seasons; they are very stable and dense.
Fogs of evaporation appear when a large source of water vapor from a warm surface is present in a cold air mass. This type of fog formation is often encountered in sea regions at high latitudes, e.g., Norway, or around lakes, rivers and swamps [4].
Examples of other fog types are: inversion fog, frontal/precipitation fog (pre-frontal-, post-frontal-, frontal passage fog), orographic fog (up-slope fog), and cloud-base lowering fog.
4. The Effect of the Water-Vapor Partial Pressure on the Surface Tension and Freezing Point of the Liquid Water-Air Interface
The effective surface tension of a water droplet has been demonstrated to decrease as the air humidity increases [38,39], as shown in Figure 1.
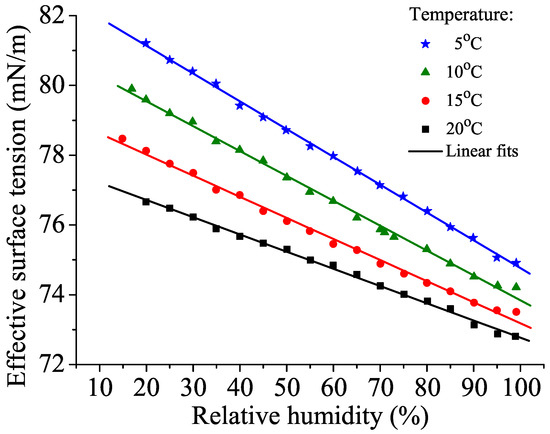
Figure 1.
Effective surface tension vs. relative humidity in a 2.8-mm diameter pure water drop.
A net flow of momentum associated with the ejection of molecules produces a “reaction pressure” on the liquid due to the law of action and reaction; this changes the effective or apparent surface tension. From a practical point of view, the dependence of the effective surface tension on air humidity may help explain the apparent discrepancies between the surface tension values obtained by different methods [40], as some of them have not been performed in air with 100% relative humidity.
The analysis of evaporation rates and this “mechanical reaction” effect suggested that the distribution of kinetic energy of water molecules ejected from the liquid phase into the gas phase is far from being thermal continuous. Only the fastest molecules are ejected or “evaporated” from the liquid surface. The actual distribution function remains to be experimentally measured.
It is interesting to note that a practical application can be developed using processes, in which wetting is essential. Since the wettability of a solid surface increases as the liquid–air surface tension decreases, the surfactant effect of steam, although dependent on the curvature, opens a possibility for developing a promising economic and eco-friendly method of modifying the surface tension in an aqueous solution–air interface by acting on the air rather than on the liquid. It is sufficient to simply increase the air humidity in order to reduce linearly the surface tension of the interface. Conversely, by reducing the air humidity, the surface tension will be increased—at least for large droplets. Thus, the simple use of common devices to humidify or dry the air can substitute for the use of chemical products. Ultimately, it would be just water that could replace, at least partially, harmful surfactants [41].
As the surface tension or the surface energy are critical factors in the thermodynamics of droplet’s growth, it is clear that air humidity plays a fundamental role in fog stability. In fact, recent experiments demonstrated that the freezing of droplets is strongly dependent on the humidity [29], as is shown in Figure 2. This means that, within an uncertainty of ±0.4 °C, freezing of supercooled water is triggered whenever the line is crossed by cooling or by increasing humidity. This explains the existence of stable liquid water droplets in mists well below 0 °C.
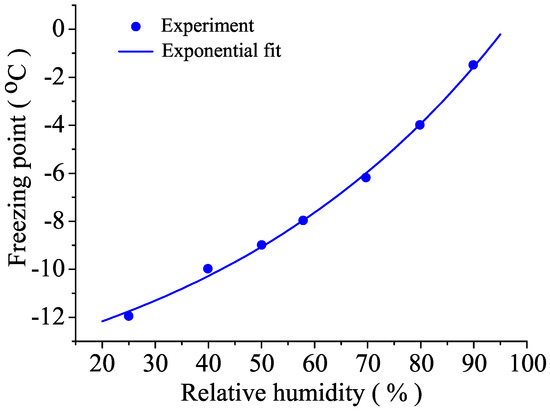
Figure 2.
Freezing point of deionized water as a function of humidity in air.
Additionally, these results were confirmed for water in macroscopic containers, demonstrating the existence of surface ice crystals whose structure depends on humidity [42]. Theoretical interpretation in terms of Gibbs free surface energy demonstrates that entropy transfer from the gas phase to the surface of ice determines the whole transition.
5. Fog Modeling and Forecasting
The processes of globalization, urbanization, and intensification of the economical and all other human activities, typical for the modern societal life, lead to considerable worldwide expansion of land-, water-, and air transportation. Due to the severe impacts of fogs on all types of transportation, the significance of fog/mist forecasting increases correspondingly. In order to provide reliable fog forecasting, adequate modeling of the processes of formation, presence, evolution, and dissipation/deposition of fogs becomes indispensable. As far as the distributions of water vapor and hydrometeors are highly variable in space and time, the precise analysis and forecasting are still challenging tasks [43]. In addition, few fog monitoring sites exist [44,45]. Therefore, studying fogs on a spatial scale requires numerical modeling and simulations. A wide variety of theoretical models have been reported, concerning different aspects of fog physics, microphysics, chemistry, dynamics, etc., ranging from simple local 1D-models [46,47] to large-scale complex 3D-models [48]. Mesoscale meteorological models have also been applied to regional forecasting of fog events [49,50,51], including the fifth-generation mesoscale model (MM5) [52].
The visibility is one of the basic characteristics directly related to the effects and possible damage caused by fogs. This is why the parameterization of fog visibility is an important issue and a subject of extensive modeling. Typically, the relationship between the extinction coefficient and the liquid water content is used in the visibility parameterization models [53], resulting in the following expression:
where are empirically determined numerical coefficients (in many operational forecast models: , [53,54]).
It has been shown [55,56] that for more adequate visibility parameterization, particularly under warm-fog conditions, it is necessary that the droplet number concentration be also taken into account as an independent variable along with the . According to the experimental relation of Jiusto [57], the visibility is directly related to the average cloud droplet radius (and hence to the number concentration) and is indirectly related to the LWC. A new visibility parameterization scheme has been offered in the case of warm fog [54,56], regarding visibility as a function not only of , but also of the droplet number concentration :
where is the so-called fog index.
The fog microphysics of the 1D Parameterized Fog (PAFOG) model [58] is incorporated into and fully coupled with the 3D NOAA Nonhydrostatic Mesoscale Model [59] and used in experiments for parameterization of fogs and low-level clouds in the planetary boundary layer.
In the work of Zhang et al. [60], parameterization of fog visibility and its relationship with other fog properties, particularly microphysical ones, have been investigated. Two parameterization schemes (one considering only, the other considering both and ) are applied to four fog cases, including dense and light fogs. The results obtained support the conclusions that the optimized parameterization scheme considering the two parameters ( and ) provides a better approximation to the observed visibility, yielding (in the case of dense fogs) a relative error of 5%, as compared to the one considering only, with a relative error of 20%.
It has been demonstrated that the use of the Global Positioning System (GPS) can be expanded into many fields, including the GPS meteorology, by using the delay of GPS signals passing through the atmosphere between GPS satellites and receivers [61,62]. An integrated Water Vapor (IWV) model based on GPS observations has been used in climatology along with the numerical weather prediction (NWP) model [63]. The GPS IWV model is also applied as a new approach to fog detection and assessments by studying (in time domain) the relationship between the GPS IWV and meteorological observations during formation, evolution, and dissipation of dense fog [64,65].
The rate of water-vapor phase transformations and their dynamics are described and studied by using the water-continuity equation (Euler equation):
where is the total water content, is the water vapor content, and is the cloud water content.
Four fog types, including radiation-, advection-, and mixed fog, as well as lack of fog, are analyzed on different time-scale series. The best results, in terms of predictability, are obtained for the radiation fog, while the worst ones concern the mixed fogs. The GPS IWV is assessed to be rather an auxiliary approach to analyzing fog formation and evolution dynamics.
The two-dimensional positive matrix factorization (PMF) model has been used to identify aerosol sources affecting fog formation [66]. Decomposing a time-series aerosol chemical data set, four fog formation factors are identified: secondary species, biomass burning aerosols, dust, and sea salt. The particle mass is predicted with a satisfactory fitting accuracy and relative standard deviations.
Along with the fog formation and evolution, the processes of fog–surface interactions and deposition are also subjected to extensive modeling, as an important factor for the water balance of ecosystems. Too little is known about the magnitude and the temporal and spatial variability of fog deposition and its driving forces for mountain ecosystems.
In order to analyze quantitatively and to understand the mechanisms and features of fog water deposition on the underlying surface, various approaches have previously been developed and used [46,67,68].
To study fog occurrence, acidification, and deposition in mountain forests, the scheme of fog deposition onto vegetation has been incorporated into the Weather Research and Forecasting (WRF) meteorological model in view of calculating the removal of cloud liquid water due to fog deposition [69,70,71]. By using a modified version of the model, better predictions concerning the liquid water content of fog than the original version of WRF are achieved [72].
The one-dimensional model of Lovett [46], on the rates and mechanisms of cloud water deposition to a subalpine balsam fir forest, has been tested experimentally on a monthly basis [47]. A relatively poor agreement between the model and measurements has been found, suggesting the limited applicability of the model for correct predictions, mainly to the fog deposition order of magnitude.
An important aspect of fog–surface interaction is the deposition of different chemical substances by fogs, when fog droplets intercept with vegetation. It is known that the concentration of ions in fog is much higher than in rain water [73], favoring the higher rates of chemical deposition on forests and canopy by fog, and affecting the ecosystems, particularly in mountainous areas characterized by more frequent fog occurrences [74].
In a series of works, Shimadera et al. [75,76,77] have studied the ionic concentrations in fog water of several ecologically important polluting ions. Fog deposition contribution rates of trans-boundary transported SO, NO, NH have been analyzed in [75]. A two-dimensional fog deposition model has been developed and used to predict the turbulent fog water flux, together with the Community Multiscale Air Quality (CMAQ) modeling system and meteorological fields produced by the MM5. By using the model, the amounts of sulfur and reactive nitrogen compounds NOx (NO, NO2, NO3, N2O5, HNO3, HONO, aerosol nitrate) deposition by fog have been estimated [76]. The annual deposition of sulfur (SO2 and SO) by fog in a mountain region has been studied, as compared to dry and wet deposition mechanisms, by using a combination of three numerical models, namely: the WRF model, the CMAQ model, and a fog deposition model [77]. It is ascertained that the sulfur deposition amount by fog is larger than that by dry deposition and comparable to the one by wet deposition. The accuracy of model predictions concerning deposition of polluting ions by fog water is critically analyzed.
The advection-diffusion equation of fog can be written as follows [77]:
where is the horizontal wind component (ms−1), is the eddy diffusivity of heat (m2 s−1), and are fog water deposition terms by inertial impaction and gravitational settling of fog droplets on leaves, respectively. Normally, the main deposition mechanism is inertial impaction. However, under low-speed wind conditions, the relative weight of gravitational settling could increase [46]. The deposition terms and are defined by
where is the one-sided leaf area density (m2 m−3), is the efficiency of inertial impaction, is the gravitational settling velocity of fog droplets, and are the portions of the effective leaf area for deposition of fog droplets by inertial impaction and gravitational settling, respectively. All coefficients present in Equations (3)–(8) can be determined empirically or from the literature (e.g., [69]). By using Equations (6)–(8), the processes of fog diffusion/advection and deposition could be analyzed.
Comprehensive studies on and analyses of fog modeling and forecasting/nowcasting, including method and model classification and characterization, can be found in the review of Gultepe et al. [5], as well as in the reports of various Actions of the European Cooperation in Science and Technology (COST Actions) (e.g., [78]).
Forecasting fogs in synoptic meteorology is a very difficult task. Even the currently most advanced numerical weather models have troubles in predicting precisely fog formation. This may be related to the strong influence of local conditions, in addition to the lack of consideration of the proper surface tension physics. However, as mentioned above, there are ways to predict fog bearing in mind the development of the atmospheric circulation. For example, during the movement of a cold front over a well-known warm surface with enough humidity, fog would often appear in the morning [79]. Another synoptic situation assisting in fog forecasting is the absence of wind in the eye of an anticyclone. This state of the atmosphere favors temperature inversions, particularly in the case of taking place over valleys with water basins or rivers [80,81].
Detection and measurement of ground fogs by satellites is a modern instrument for operational nowcasting applications and studies on the climate processes and changes [82,83].
6. Fog Impacts
6.1. Impacts on Air Quality
Urban air pollution is commonly known as smog (smoke + fog) [84] or, in some cases, as haze-fog [85,86]. Two types of smog exist: classical (London type) smog and photochemical (Los Angeles type) smog. Classical smog results from the interaction of particulate matter with sulfur dioxide and occurs near the ground, under windless conditions and temperatures around 0 °C—normally in autumn and winter. Photochemical smog is typical for the summer season, occurring usually near midday, since it is caused by the effect of NOx, CO, O3, and VOCs interacting with solar radiation [3,87,88].
As the smog/haze-fog contains polluting primary or secondary particulate matter and, possibly, irritable chemicals, it can have severe negative effects on the air quality and human health [89,90]. The smog/haze-fog formation basically depends on the atmospheric aerosol loading levels and meteorological state, particularly, on air humidity. Important factors defining or influencing the loadings of the atmospheric air with polluting aerosols or chemical compounds and, by this manner, the occurrence and evolution of smog/haze-fog, are the natural air circulations and solar irradiation [24,91,92], as well as some anthropogenic ones resulting from traffic or industrial fuel combustion, heating, fires, or of other industrial and construction human activities [93,94,95].
Improving the air quality in several major cities in Brazil, Argentina, Canada, the UK, and the USA is found to be among the reasons (along with urban heat island) for fewer dense-fog events [96]. In contrast, in China, increased aerosol loadings, along with the weakening of the East Asian winter monsoon circulations, have caused a doubling of the winter fog event frequency over the past three decades [97].
Fog droplets are found to be efficient in scavenging boundary layer pollutants and removing them from the atmospheric air. They also assist in the production of strong acids by aqueous-phase reactions. High ionic concentrations, especially of NH, NO, and SO have been observed in fog water. Deposition of these large droplets on surfaces leads to a decrease in fog water ionic concentration in the course of a fog event. Ammonia is found to neutralize high concentrations of acidic anions in fog water [98].
Weiss-Penzias et al. [99] have investigated total mercury (HgT) and monomethyl mercury (MMHg) concentrations in fog water. They reported mean HgT concentrations of ng/L and mean MMHg concentrations of ng/L. The MMHg concentrations measured in fog water were higher by about a factor of five than those found in rain water. Fog is recognized as being an important source of MMHg to coastal ecosystems, accounting for 7–42% of HgT and 61–99% of MMHg of the total atmospheric deposition.
6.2. Impacts of Fog on Human Health
Air pollutants, such as nitrogen oxides ( NOx), sulfur dioxides (SO2), ozone (O3), sulfuric acid (H2SO4), and nitric acid (HNO3), are among the constituents of acid fog; most of the fog acidity is attributable to the latter two [6]. The increase in respiratory mortality and morbidity, and of cardiopulmonary and lung cancer mortality is associated with exposure to fine aerial aerosol particles (in particular acidic ones), microbes, sulfur oxide and sulfur dioxide [6,7,100,101] (see Table 1).

Table 1.
Effects of air/fog pollutants on human health.
The exposure to sulfur dioxide tends to exacerbate airway constriction in exercising asthmatics at exposure concentrations attainable in ambient air. Nitrogen dioxide shows slight unfavorable respiratory effect at ambient concentrations. Sulfuric acid has been suggested to increase the bronchial reactivity and to change the mucociliary clearance at concentrations higher than the ambient range, but no significant effect has been observed at concentrations within the ambient range [102]. Further investigations have been carried out by Avol et al. [103]. They explored the short-term respiratory effects of sulfuric acid in fog by exposing 44 adult volunteers (normal and asthmatic) to light fogs (with different sulfuric acid concentrations) in a controlled-atmosphere chamber. Their results showed a slight effect on the pulmonary function, even at the highest sulfuric acid concentration, and a modest increase in respiratory symptoms with increasing acid concentrations. The authors concluded that no pulmonary dysfunctions and only slight respiratory symptoms are likely to occur in response to inhalation of fog containing sulfuric acid.
Tanaka et al. [6] investigated the association between asthma-related hospital visits and meteorological factors or air pollutants. They studied 102 adult asthmatic patients (44 nonatopic and 58 atopic) in a period of two years. The results showed significantly lower levels of gaseous pollutants on foggy days, compared to fog-free days, which could be due to the absorption of gaseous air pollutants by the fog, resulting in an increase of the acidity of fog water. A weak correlation was found between the mean pH on a foggy day and the number of hospital visits by nonatopic asthmatic patients. Concerning the meteorological factors, air pollutants and their relation to asthma-provoked hospital visits, it is reported that an increase of hospital visits was observed in the case of fog, high water vapor pressure and low concentrations of NO and NO2 (in nonatopic patients) and NO2 and SO2 (in atopic patients). High ozone concentrations and low day-to-day temperature differences also contributed to hospital visits in nonatopic subjects. The authors concluded that naturally occurring acid fog may have a weak bronchoconstrictive effect, especially on nonatopic asthmatic patients.
A recent investigation on fog-induced respiratory response, suggesting a possible way to attenuate it, has been carried out by Lavorini et al. [104] on 14 healthy subjects. They found that fog inhalation is capable of inducing cough and changes in breathing patterns in healthy subjects. The effects were evaluated of no drug, placebo, and 4- and 8-mg nedocromil sodium (NCS) administration on the cough threshold and changes in breathing patterns during fog inhalation. The results showed that NCS possesses antitussive effects and reduces changes in the pattern of breathing induced by fog.
Recently, N-nitrosodimethylamine (NDMA) in clouds and radiation fogs was suggested to be a potent carcinogen [105,106]. It is readily soluble in water and has been found mostly in industrial and manufacturing areas [106]. Hutchings et al. [105] have observed NDMA at substantial concentrations (7.5–397 ng/L) in fogs and clouds.
Numerous studies of the air pollution impact on human health have been carried out. We will mention here a few of them only, bearing in mind that naturally occurring acid fog, for example, is associated with some of the gaseous air pollutants and acidic aerosols, as well as that some of the aerosol particles present in the atmosphere act as CCN during cloud and fog formation.
In his study, Pope III [107] observed a correlation between elevated PM10 levels and the number of hospital admissions for pneumonia, pleurisy, bronchitis, and asthma. He reported that when:
- 24 h PM10 levels exceeded 150 µg/m3, the average number of admissions of children nearly tripled; in adults, the increase in admissions was 44%
- Mean PM10 levels were greater than or equal to 50 µg/m3, the average number of admissions for children and adults increased by 89% and 47%, respectively.
Strong correlation was observed between PM10 levels and hospital admissions for bronchitis and asthma, especially in the case of children.
Pope III et al. [100] explored the long-term exposure to fine aerosol particles and its relation to the observed cardiopulmonary and lung cancer mortality. Their results showed the following relation between fine aerosol particles (especially PM2.5), sulfur oxide—related pollutant and all-causes, lung cancer and cardiopulmonary mortality: the risk of all-causes, cardiopulmonary, and lung cancer mortality increased by 4%, 6%, and 8%, respectively, for each 10 µg/m3 increase in fine particulate air pollutants. Bigger particles (coarse mode) and total suspended matter were not consistently associated with mortality [90].
The effects of air/fog pollutants on human health and their mechanisms of action can be summarized as follows:
- NOx affects mainly the respiratory system, causing nose and throat irritation, bronchoconstriction, dyspnoea (especially in asthmatics), and respiratory infections. NO2 provokes emphysema-like lesions [7] and is also related to increased rates of infant mortality and cardiovascular diseases [87].
- SO2 influences the respiratory system in a way similar to nitrogen oxides.
- O3 causes lung inflammation and reduction of the lung function [7]. It is also recognized to initiate cardiovascular diseases and cause acute coronary events in middle-aged adults without heart disease [87].
- CO affects the cardiovascular system, binding hemoglobin to modify its conformation and reduce its capacity to transfer oxygen, which in turn affects both the brain and heart. CO causes impaired concentration, confusion, slow reflexes, hypoxia in the nervous system, affects the muscular system [7,87].
- Volatile organic components, such as benzene, for example, induce hematological problems [7] affect the central nervous system, cause anemia, lymphopenia, thrombocytopenia, pancytopenia [87]. Benzene causes also a certain type of leukemia [87,89]. Other effects caused by VOCs include eyes, nose, and throat irritation, headache, nausea, dizziness [87]. All types of gaseous pollutants could cause cancer [7,87].
- Dioxins affect the cardiovascular system, increasing mortality caused by ischemic heart disease; the nervous system, decreasing nerve conduction speed and impairing mental development of children; the digestive system, inducing cell damage, gastrointestinal and liver cancer; growth and development of the central nervous system of the fetus.
- Heavy metals influence the respiratory system, causing asthma, emphysema and lung cancer (arsenic, nickel, vanadium); the cardiovascular system, inducing tachycardia, increased blood pressure, anaemia (mercury, nickel, arsenic); the nervous system, causing memory disturbances, sleep disorders, anger, fatigue, hand tremors, blurred vision, slurred speech, and neurological cancer (arsenic, lead, mercury); the urinary system, inducing kidney damage, stone formation, nephrocalcinosis, and renal cancer. Lead exposure during pregnancy increases the risk of spontaneous abortion and reduced fetal growth, congenital malformations and lesions of developing nervous system.
- Particulate matter (especially, ultrafine and fine particle size modes) affects the respiratory system, inducing lung inflammation; the cardiovasculary system, leading to obstruction of blood vessels and myocardial infarction [7].
6.3. Impacts of Fog on Plants and Microorganisms
Investigations have been performed on the impacts of fog on plants, such as the red spruce [108] and beech [109], as well as on hypolithic microbial communities [101,110]. Percy et al. [108] reported that red spruce exposure to pH 4.2 acid fog increased wax quantity for a certain period, but a longer exposure decreased it; reduced the rate of de novo wax synthesis; changed the composition of wax reducing production of secondary alcohols (pH 3.0 fog). Acidic fogs also influence the cuticular membrane thickness and wettability of needles. Nara et al. [109] have investigated the decline of beech forest by observing precipitation, fog and stem flow. Seasonal variations in the pH of fog water (ranging from 4.2 to 6.4) were observed with higher values from May until mid-August. Higher concentrations of most chemical species were seen in fog samples, in comparison with precipitation samples. The pH of the stem flow ranged from 4.2 to 5.3. The authors reported also that more potassium and magnesium ions were leached by acidic precipitation from unhealthy trees, than from healthy trees. Based on model results, they suggested that the polluted air over the Tokyo Metropolitan Area has affected the fog acidity.
Most recently, Warren-Rhodes et al. [110] investigated the role of fog and other factors in physical ecology of hypolithic microbial communities in Namib Desert and showed that fog sustains and impacts the hypolithic ecology, effectively replacing the rainfall, and enables high hypolithic abundance; it is also responsible for smaller size-class rocks to be colonized at a higher proportion, due to the higher availability of water.
6.4. Impacts of Fog on Aviation and Public Transportation
A wide variety of human activities are affected by fog occurrence. In some cases, fog causes inconvenience and high costs, in other, death. Fog reduces the visibility, which impacts our ability to drive, sail, fly, etc. [2,111,112]. Gultepe et al. [5,113] pointed out that the total economic loss, associated with the impact of fog on all types of transportation (air, sea, land), can be comparable to those of tornadoes or, in some cases, winter storms and hurricanes. Croft [2] reported research on deadly accidents and financial losses due to fog occurrence. In fog-prone regions, fog has been cited up to 10% of the time as the primary cause of accidents, particularly, in multiple-vehicle crashes. The average claim for one vehicle is nearly $8000 and over one million dollars for a multiple-vehicle crash. Concerning sea transportation, fog may slow or stop shipping operations, if the visibility range is below 0.5 km. The financial losses may range from $ 10,000 to $ 25,000 per day per ship and into millions for even a moderately active harbor or port of call. In air transportation, visibility of at least 1.207 km is required for takeoff and landing for most flight operations [2]. Airports covered by fog often cancel take-offs and refuse to accept airplanes for security reasons [114]. Delayed or canceled flights may cost to the airlines between $5000 and $25,000 per flight [2]. The losses associated with heavy fog event in the pre-Christmas period of 2006 at seven British airports have been estimated to at least £25 million [113]. In Canada, approximately 50 people per year die because of motor vehicle accidents, in which fog was a contributing factor [5,113]. In the USA, 13,720 people died as a result of fog-related accidents in the period between 1995 and 2004 [111]. Many people have died all over the world in fog-related ship collisions and aviation accidents. Fog impacts also a variety of military rescue operations, space shuttle operations and contributes to supply delays, property damages, sporting events cancellation, affects adversely the operation of schools and colleges and travel time reliability, etc. [2,111,115].
Numerous studies have been carried out on developing methods for artificially eliminating fog from airports. One of the most recent ones, conducted by Czyż and Markowski [114], was related to the influence of an acoustic field on the dispersed phase of a fluid, and intended to use this innovative technology for airport fog precipitation. It has been shown that, if the air is saturated sufficiently with water vapor, an increase of the fog droplet size occurs under the influence of an acoustic field, so that fog precipitation is achieved under specific conditions.
7. Conclusions
Clouds and fogs are essential for the Earth’s climate. In spite of the abundant research on this topic, it is still an open field for research and development. Accordingly, important issues to be addressed still remain, such as how the atmospheric water cycle is affected by water droplet nucleation and condensation, or how to achieve better understanding and characterization of the mechanisms and conditions for cloud/fog formation, existence, and decay. This imposes new demands and challenges to the cloud/fog research, needing more and permanent monitoring and studies of fogs, due to their importance for the environment and societal life.
Fog forecasting remains a challenging task, necessitating updating of our knowledge on droplet- and water–air-interface physics. Existing 1D and 3D fog models should be updated regularly to keep their relevance or new ones should be developed, requiring new parameterization schemes and data. Since the meteorological models evolve faster because of the large number of stations and abundance of parameterization data, combining fog models with mesoscale and regional meteorological models appears to be effective and fruitful for more adequate fog parameterization and forecasting.
The intensification of the use of combustive and polluting technologies and power sources in industry and transportation affects the natural fog composition, provoking more frequent and longer-lasting smog occurrences in industrial and urban areas, with severe impacts on air quality and human health. In addition, increasing production and usage of artificial fogs, along with their usefulness and advantages, can also be a potential source of allergies and other side effects, as well as a subject of non-correct dangerous or damaging applications.
In summary, fogs are important factors influencing the environmental balance, air quality, ecosystems, transportation, and human health. Fog research has achieved remarkable progress in understanding the nature and mechanisms of fog formation, existence, and decay. Still, the constantly changing natural and social environment impose new demands and challenges, needing new efforts, data, and approaches, aimed to better and more adequate parameterization, modeling, and forecasting of fog events, in order to reduce their negative environmental, social, and medical effects.
Acknowledgments
This work has been funded by the European Commission FP7 Security Research Programme, under COUNTERFOG Grant No. 312804. Authors gratefully acknowledge the kind technical assistance of Iliana Mitova and Zhivko Stoyanov.
Author Contributions
J.L.P.-D.; O.I., A.V., and Z.P. developed the idea and outlines of the review; Z.P., M.A.Á.-V., I.V.-B., T.E., T.D., O.G., and P.V.T. contributed to the literature research; J.L.P.-D., Z.P., M.A.Á.-V., I.V.-B., O.G., and T.E. contributed to the manuscript preparation; J.L.P.-D., Z.P., O.I., A.V., and T.D. revised the manuscript and further proofread the text.
Conflicts of Interest
The authors declare no conflict of interest.
References
- World Meteorological Organization. International Meteorological Vocabulary; WMO Publication 182; World Meteorological Organization: Geneva, Switzerland, 1992; 782p. [Google Scholar]
- Croft, P.J. Fog. In Encyclopedia of Atmospheric Sciences; Holton, J.R., Pyle, J.A., Curry, J.A., Eds.; Academic Press: London, UK, 2003; pp. 777–792. ISBN 978-0-12-227090-1. [Google Scholar]
- Fu, H.; Chen, J. Formation, features and controlling strategies of severe hazefog pollutions in China. Sci. Total Environ. 2017, 578, 121–138. [Google Scholar] [CrossRef] [PubMed]
- Gultepe, I.; Mildbrandt, J.A.; Zhou, B. Marine fog: A review on microphysics and visibility prediction. In Marine Fog: Challenges and Advancements in Observations, Modeling, and Forecasting. Springer Atmospheric Sciences; Koracin, D., Dorman, C., Eds.; Springer: Cham, Switzerland, 2017; pp. 345–394. [Google Scholar] [CrossRef]
- Gultepe, I.; Tardif, R.; Michaelides, S.C.; Cermak, J.; Bott, A.; Bendix, J.; Müller, M.D.; Pagowski, M.; Hansen, B.; Ellrod, G.; et al. Fog research: A review of past achievements and future perspectives. Pure Appl. Geophys. 2007, 164, 1121–1159. [Google Scholar] [CrossRef]
- Tanaka, H.; Honma, S.; Nishi, M.; Igarashi, T.; Teramoto, S.; Nishio, F.; Abe, S. Acid fog and hospital visits for asthma: An epidemiological study. Eur. Respir. J. 1998, 11, 1301–1306. [Google Scholar] [CrossRef] [PubMed]
- Kampa, M.; Castanas, E. Human health effects of air pollution. Environ. Pollut. 2008, 151, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Maalick, Z.; Kühn, T.; Korhonen, H.; Kokkola, H.; Laaksonen, A.; Romakkaniemi, S. Effect of aerosol concentration and absorbing aerosol on the radiation fog life cycle. Atmos. Environ. 2016, 133, 26–33. [Google Scholar] [CrossRef]
- Ritter, A.; Regalado, C.M.; Guerra, J.C. Quantification of fog water collection in three locations of Tenerife (Canary Islands). Water 2015, 7, 3306–3319. [Google Scholar] [CrossRef]
- Lehr, J.H.; Keeley, J. (Eds.) Water Encyclopedia: Oceanography; Meteorology; Physics and Chemistry; Water Law; and Water History, Art, and Culture; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005; p. 344. ISBN 978-0-471-73684-4. [Google Scholar]
- Stoycheva, A.; Guerova, G. Study of fog in Bulgaria by using the GNSS tropospheric products and large scale dynamic analysis. J. Atmos. Sol.-Terr. Phys. 2015, 133, 87–97. [Google Scholar] [CrossRef]
- Nanev, C. Theory of nucleation. In Handbook of Crystal Growth (Fundamentals: Thermodynamics and Kinetics), 2nd ed.; Nishinaga, T., Ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 315–358. [Google Scholar] [CrossRef]
- Pruppacher, H.R.; Klett, J.D. Microphysics of Clouds and Precipitation, 2nd ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1997. [Google Scholar] [CrossRef]
- Haeffelin, M.; Laffineur, Q.; Bravo-Aranda, J.-A.; Drouin, M.-A.; Casquero-Vera, J.-A.; Dupont, J.-C.; De Backer, H. Radiation fog formation alerts using attenuated backscatter power from automatic lidars and ceilometers. Atmos. Meas. Tech. 2016, 9, 5347–5365. [Google Scholar] [CrossRef]
- Kulmala, M.; Vehkamäki, H.; Petäjä, T.; Dal Maso, M.; Lauri, A.; Kerminen, V.-M.; Birmili, W.; McMurry, P.H. Formation and growth rates of ultrafine atmospheric particles: A review of observations. J. Aerosol Sci. 2004, 35, 143–176. [Google Scholar] [CrossRef]
- Curtius, J. Nucleation of atmospheric aerosol particles. Comptes Rendus Phys. 2006, 7, 1027–1045. [Google Scholar] [CrossRef]
- Kulmala, M.; Kerminen, V.-M. On the formation and growth of atmospheric nanoparticles. Atmos. Res. 2008, 90, 132–150. [Google Scholar] [CrossRef]
- Andreae, M.O.; Rosenfeld, D. Aerosol-cloud-precipitation interactions. Part 1. The nature and sources of cloud-active aerosols. Earth-Sci. Rev. 2008, 89, 13–41. [Google Scholar] [CrossRef]
- Sun, Y.; Zhou, X.; Wang, W. Aerosol size distributions during haze episodes in winter in Jinan, China. Particuology 2016, 28, 77–85. [Google Scholar] [CrossRef]
- Noone, K.J.; Ogren, J.A.; Hallberg, A.; Heintzenberg, J.; Ström, J.; Hansson, H.C.; Svenningsson, B.; Wiedensohler, A.; Fuzzi, S.; Facchini, M.C.; et al. Changes in aerosol size- and phase distributions due to physical and chemical processes in fog. Tellus 1992, 44B, 489–504. [Google Scholar] [CrossRef]
- Jacob, D.J. Introduction to Atmospheric Chemistry; Princeton University Press: Princeton, NJ, USA, 1999; pp. 146–156, ISBN-13 978-0691001852, ISBN-10 0691001855. [Google Scholar]
- Brasseur, G.P.; Jacob, D.J. Modeling of Atmospheric Chemistry; Cambridge University Press: Cambridge, UK, 2017; p. 77, ISBN-13 9781107146969, ISBN-10 1107146968. [Google Scholar]
- Gillani, N.V.; Daum, P.H.; Schwartz, S.E.; Leaitch, W.R.; Strapp, J.W.; Isaac, G.A. Fractional activation of accumulation-mode particles in warm continental stratiform clouds. In Precipitation Scavenging and Atmosphere Surface Exchange Process; Schwartz, S.E., Slinn, W.G.N., Eds.; Hemisphere Publishing Corp., Taylor & Francis Group: Washington, DC, USA, 1992; Volume 1, pp. 345–358, ISBN-13 978-1560322634, ISBN-10 1560322632. [Google Scholar]
- Seinfeld, J.H.; Pandis, S.N. Atmospheric Chemistry and Physics: From Air Pollution to Climate Change, 2nd ed.; John Wiley Sons Inc.: Hoboken, NJ, USA, 2006; ISBN-13 978-0471720188, ISBN-10 0471720186. [Google Scholar]
- Wilson, W.E.; Suh, H.H. Fine particles and coarse particles: Concentration relationships relevant to epidemiologic studies. J. Air Waste Manag. 1997, 47, 1238–1249. [Google Scholar] [CrossRef]
- Sun, J.; Ariya, P.A. Atmospheric organic and bio-aerosols as cloud condensation nuclei (CCN): A review. Atmos. Environ. 2006, 40, 795–820. [Google Scholar] [CrossRef]
- Herckes, P.; Valsaraj, K.T.; Collett, J.L., Jr. A review of observations of organic matter in fogs and clouds: Origin, processing and fate. Atmos. Res. 2013, 132–133, 434–449. [Google Scholar] [CrossRef]
- Gultepe, I.; Zhou, B.; Milbrandt, J.; Bott, A.; Li, Y.; Heymsfield, A.J.; Ferrier, B.; Ware, R.; Pavolonis, M.; Kuhn, T.; et al. A review on ice fog measurements and modeling. Atmos. Res. 2015, 151, 2–19. [Google Scholar] [CrossRef]
- Pérez-Díaz, J.L.; Álvarez-Valenzuela, M.A.; Sánchez-García-Casarrubios, J.; Jiménez-López, S. Ice surface entropy induction by humidity or how humidity prompts freezing. JMEST 2016, 3, 3825–3828. [Google Scholar]
- Zhou, Y.; Niu, S.; Lü, J.; Zhou, Y. The effect of freezing drizzle, sleet and snow on microphysical characteristics of supercooled fog during the icing process in a mountainous area. Atmosphere 2016, 7, 143. [Google Scholar] [CrossRef]
- Ali, G.; Khan, E.U.; Ali, N.; Khan, H.A.; Waheed, A. The effect of radioactive aerosols on fog formation. Pak. J. Meteorol. 2011, 7, 1–7. [Google Scholar]
- Masson, O.; Ott, A.V.; Bourcier, L.; Paulat, P.; Ribeiro, M.; Pichon, J.-M.; Sellegri, K.; Gurriaran, R. Change of radioactive cesium (137Cs and 134Cs) content in cloud water at an elevated site in France, before and after the Fukushima nuclear accident: Comparison with radioactivity in rainwater and in aerosol particles. Atmos. Res. 2015, 151, 45–51. [Google Scholar] [CrossRef]
- Bourcier, L.; Masson, O.; Laj, P.; Paulat, P.; Pichon, J.-M.; Chausse, P.; Gurriaran, R.; Sellegri, K. 7Be, 210Pb and 137Cs concentrations in cloud water. J. Environ. Radioact. 2014, 128, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Decesari, S.; Sowlat, M.H.; Hasheminassab, S.; Sandrini, S.; Gilardoni, S.; Facchini, M.C.; Fuzzi, S.; Sioutas, C. Enhanced toxicity of aerosol in fog conditions in the Po Valley, Italy. Atmos. Chem. Phys. 2017, 17, 7721–7731. [Google Scholar] [CrossRef]
- Gibb, R.; Carson, P.; Thompson, W. The effect of fog on radionuclide deposition velocities. Proc. Annu. Conf. Can. Nucl. Assoc. 1997, 2, 1–14. [Google Scholar]
- Su, C.-C.; Huh, C.-A. Measurements of 7Be and 210Pb in cloudwaters: Toward a better understanding of aerosol transport and scavenging. Geophys. Res. Lett. 2006, 33, L04807. [Google Scholar] [CrossRef]
- Stolaki, S.; Haeffelin, M.; Lac, C.; Dupont, J.-C.; Elias, T.; Masson, V. Influence of aerosols on the life cycle of a radiation fog event. A numerical and observational study. Atmos. Res. 2015, 151, 146–161. [Google Scholar] [CrossRef]
- Pérez-Díaz, J.L.; Álvarez-Valenzuela, M.A.; García-Prada, J.C. The effect of the partial pressure of water vapor on the surface tension of the liquid water–air interface. J. Colloid Interface Sci. 2012, 381, 180–182. [Google Scholar] [CrossRef] [PubMed]
- Portuguez, E.; Alzina, A.; Nichaud, P.; Oudjedi, M.; Smith, A. Evolution of a water pendant droplet: Effect of temperature and relative humidity. Nat. Sci. 2017, 9, 1–20. [Google Scholar] [CrossRef]
- Vargaftik, N.B.; Volkov, B.N.; Voljak, L.D. International tables of the surface tension of water. J. Phys. Chem. Ref. Data 1983, 12, 817–820. [Google Scholar] [CrossRef]
- Pérez-Díaz, J.L.; Álvarez-Valenzuela, M.A.; García-Prada, J.C. Method for Modifying the Surface Tension in Liquid-Air Interfaces. ES Patent P201132074, 24 July 2013. [Google Scholar]
- Pérez-Díaz, J.L.; Álvarez-Valenzuela, M.A.; Rodríguez-Celis, F. Surface freezing of water. SpringerPlus 2016, 5, 629. [Google Scholar] [CrossRef] [PubMed]
- Bendix, J.; Thies, B.; Cermak, J.; Nauß, T. Ground fog detection from space based on MODIS daytime data—A feasibility study. Weather Forecast 2005, 20, 989–1005. [Google Scholar] [CrossRef]
- Rudiger, J.J.; Book, K.; Baker, B.; deGrassie, J.S.; Hammel, S. A model for predicting fog aerosol size distributions. Proc. SPIE 2016, 9979, 99790V. [Google Scholar] [CrossRef]
- Tonttila, J.; Maalick, Z.; Raatikainen, T.; Kokkola, H.; Kühn, T.; Romakkaniemi, S. UCLALES-SALSA v1.0: A large-eddy model with interactive sectional microphysics for aerosol, clouds and precipitation. Geosci. Model Dev. 2017, 10, 169–188. [Google Scholar] [CrossRef]
- Lovett, G. Rates and mechanisms of cloud water deposition to a subalpine balsam fir forest. Atmos. Environ. 1984, 18, 361–371. [Google Scholar] [CrossRef]
- Klemm, O.; Wrzesinsky, T.; Scheer, C. Fog water flux at a canopy top: Direct measurement versus one-dimensional model. Atmos. Environ. 2005, 39, 5375–5386. [Google Scholar] [CrossRef]
- Müller, M.D.; Masbou, M.; Bott, A. Three-dimensional fog forecasting in complex terrain. Q. J. R. Meteorol. Soc. 2010, 136, 2189–2202. [Google Scholar] [CrossRef]
- Ballard, S.P.; Golding, B.W.; Smith, R.N.B. Mesoscale model experimental forecasts of the haar of northeast Scotland. Mon. Weather Rev. 1991, 119, 2107–2123. [Google Scholar] [CrossRef]
- Pagowski, M.; Gultepe, I.; King, P. Analysis and modeling of an extremely dense fog event in southern Ontario. J. Appl. Meteorol. 2004, 43, 3–16. [Google Scholar] [CrossRef]
- Shimadera, H.; Shrestha, K.L.; Kondo, A.; Kaga, A.; Inoue, Y. Fog simulation using a mesoscale model in and around the Yodo River Basin, Japan. J. Environ. Sci. 2008, 20, 838–845. [Google Scholar]
- Grell, G.A.; Dudhia, J.; Stauffer, D.R. A Description of the Fifth-Generation Penn State/NCAR Mesoscale Model (MM5). In NCAR Technical Note NCAR/TN-398 + STR; National Center for Atmospheric Research: Boulder, CO, USA, 1994. [Google Scholar]
- Stoelinga, M.T.; Warner, T.T. Nonhydrostatic, mesobeta-scale model simulations of cloud ceiling and visibility for an East Coast winter precipitation event. J. Appl. Meteorol. 1999, 38, 385–404. [Google Scholar] [CrossRef]
- Gultepe, I.; Müller, M.D.; Boybeyi, Z. A new visibility parameterization for warm-fog applications in numerical weather prediction models. J. Appl. Meteorol. Clim. 2006, 45, 1469–1480. [Google Scholar] [CrossRef]
- Meyer, M.B.; Jiusto, J.E.; Lala, G.G. Measurement of visual range and radiation-fog (haze) microphysics. J. Atmos. Sci. 1980, 37, 622–629. [Google Scholar] [CrossRef]
- Gultepe, I.; Isaac, G. An analysis of cloud droplet number concentration (Nd) for climate studies: Emphasis on constant Nd. Q. J. R. Meteorol. Soc. 2004, 130, 2377–2390. [Google Scholar] [CrossRef]
- Jiusto, J.E. Fog structure. In Clouds, Their Formation, Optical Properties, and Effects; Hobbs, P.V., Deepak, A., Eds.; Academic Press: New York, NY, USA, 1981; pp. 187–239. [Google Scholar] [CrossRef]
- Bott, A.; Trautmann, T. PAFOG—A new efficient forecast model of radiation fog and low-level stratiform clouds. Atmos. Res. 2002, 64, 191–203. [Google Scholar] [CrossRef]
- Müller, M.D.; Bott, A.; Masbou, M.; Janjic, Z. Fog prediction in a 3D Model with Parameterized Microphysics. In Proceedings of the World Weather Reasearch Programme Symposium on Nowcasting and Very Short Forecasting, Toulouse, France, 5–9 September 2005. [Google Scholar]
- Zhang, J.; Xue, H.; Deng, Z.; Ma, N.; Zhao, C.; Zhan, Q. A comparison of the parameterization schemes of fog visibility using the in-situ measurements in the North China Plain. Atmos. Environ. 2014, 92, 44–50. [Google Scholar] [CrossRef]
- Anthes, R.A.; Bernhardt, P.A.; Chen, Y.; Cucurull, L.; Dymond, K.F.; Ector, D.; Healy, S.B.; Ho, S.P.; Hunt, D.C.; Kuo, Y.H.; et al. The COSMIC/FORMOSAT-3 mission: Early results. Bull. Am. Meteorol. Soc. 2008, 89, 313–333. [Google Scholar] [CrossRef]
- Jin, S.G.; Li, Z.; Cho, J. Integrated water vapour field and multiscale variations over China from GPS measurements. J. Appl. Meteorol. Climatol. 2008, 47, 3008–3015. [Google Scholar] [CrossRef]
- Smith, T.L.; Benjamin, S.G.; Gutman, S.I.; Sahm, S. Short-range forecast impact from assimilation of GPS-IPW observations into the rapid update cycle. Mon. Weather Rev. 2007, 135, 2914–2930. [Google Scholar] [CrossRef]
- Lee, J.; Park, J.-U.; Cho, J.; Baek, J.; Kim, H.W. A characteristic analysis of fog using GPS-derived integrated water vapour. Meteorol. Appl. 2010, 17, 463–473. [Google Scholar] [CrossRef]
- Abdullah, E.M.; Habib, F.; Mohamed-Slim, A. Outdoor FSO communications under fog: Attenuation modeling and performance evaluation. IEEE Photonics J. 2016, 8, 7905507. [Google Scholar] [CrossRef]
- Mehta, B.; Venkataraman, C.; Bhushan, M.; Tripathi, S.N. Identification of sources affecting fog formation using receptor modeling approaches and inventory estimates of sectoral emissions. Atmos. Environ. 2009, 43, 1288–1295. [Google Scholar] [CrossRef]
- Fowler, D.; Morse, A.P.; Gallagher, M.; Choularton, T. Measurements of cloud water deposition on vegetation using a lysimeter and a fluxgradient technique. Tellus 1990, 42, 285–293. [Google Scholar] [CrossRef]
- Mueller, S. Estimating cloud water deposition to subalpine spruce-fir forests I. Modifications to an existing model. Atmos. Environ. 1991, 25, 1093–1104. [Google Scholar] [CrossRef]
- Katata, G.; Nagai, H.; Wrzesinsky, T.; Klemm, O.; Eugster, W.; Burkard, R. Development of a land surface model including cloud water deposition on vegetation. J. Appl. Meteorol. Climatol. 2008, 47, 2129–2146. [Google Scholar] [CrossRef]
- Katata, G.; Nagai, H.; Kajino, M.; Ueda, H.; Hozumi, Y. Numerical study of fog deposition on vegetation for atmosphere-land interactionsin semi-arid and arid regions. Agric. For. Meteorol. 2010, 150, 340–353. [Google Scholar] [CrossRef]
- Katata, G.; Kajino, M.; Hiraki, T.; Aikawa, M.; Kobayashi, T.; Nagai, H. A method for simple and accurate estimation of fog deposition in a mountain forest using a meteorological model. J. Geophys. Res. 2011, 116, D20102. [Google Scholar] [CrossRef]
- Chaouch, N.; Temimi, M.; Weston, M.; Ghedira, H. Sensitivity of the meteorological model WRF-ARW to planetary boundary layer schemes during fog conditions in a coastal arid region. Atmos. Res. 2017, 187, 106–127. [Google Scholar] [CrossRef]
- Igawa, M.; Tsutsumi, Y.; Mori, T.; Okochi, H. Fog water chemistry at a mountainside forest and the estimation of the air pollutant deposition via fog droplets based on the atmospheric quality at the mountain base. Environ. Sci. Technol. 1998, 32, 1566–1572. [Google Scholar] [CrossRef]
- Dawson, T.E. Fog in the California redwood forest: Ecosystem inputs and use by plants. Oecologia 1998, 117, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Shimadera, H.; Kondo, A.; Kaga, A.; Shrestha, K.L.; Inoue, Y. Contribution of transboundary air pollution to ionic concentrations in fog in the Kinki Region of Japan. Atmos. Environ. 2009, 43, 5894–5907. [Google Scholar] [CrossRef]
- Shimadera, H.; Kondo, A.; Kaga, A.; Shrestha, K.L.; Inoue, Y. Numerical predictions of sulfur and nitrogen depositions through fog in forest areas. J. Jpn. Soc. Atmos. Environ. 2010, 45, 247–255. [Google Scholar]
- Shimadera, H.; Kondo, A.; Shrestha, K.L.; Kaga, A.; Inoue, Y. Annual sulfur deposition through fog, wet and dry deposition in the Kinki Region of Japan. Atmos. Environ. 2011, 45, 6299–6308. [Google Scholar] [CrossRef]
- Jacobs, W.; Nietosvaara, V.; Bott, A.; Bendix, J.; Cermak, J.; Michaelides, S.; Gultepe, I. (Eds.) Short Range Forecasting Methods of Fog, Visibility and Low Clouds; ESSEM COST Action 722 Final Report; Office for Official Publications of the European Communities: Luxembourg, 2008. [Google Scholar]
- Tardif, R.; Rasmussen, R.M. Event-based climatology and typology of fog in the New York City region. J. Appl. Meteorol. Clim. 2007, 46, 1141–1168. [Google Scholar] [CrossRef]
- Wobrock, W.; Schell, D.; Maser, R.; Kessel, M.; Jaeschke, W.; Fuzzi, S.; Facchini, M.C.; Orsi, G.; Marzorati, A.; Winkler, P.; et al. Meteorological characteristics of the Po Valley fog. Tellus B 1992, 44, 469–488. [Google Scholar] [CrossRef]
- Roach, W. Back to basics: Fog: Part 2—The formation and dissipation of land fog. Weather 1995, 50, 7–11. [Google Scholar] [CrossRef]
- Cermak, J.; Bendix, J. Detecting ground fog from space—A microphysics-based approach. Int. J. Remote Sens. 2011, 32, 3345–3371. [Google Scholar] [CrossRef]
- Marchetti, M.; Boucher, V.; Dumoulin, J.; Colomb, M. Retrieving visibility distance in fog combining infrared thermography, Principal Components Analysis and Partial Least-Square regression. Infrared Phys. Technol. 2015, 71, 289–297. [Google Scholar] [CrossRef]
- Gaffney, J.S.; Marley, N.A.; Frederick, J.E. Formation and effects of smog. In Environmental and Ecological Chemistry; Sabljic, A., Ed.; Eolss Publishers Co., Ltd.: Oxford, UK, 2009; Volume 2, pp. 25–51. ISBN 9781848266940. [Google Scholar]
- Zhang, Y.W.; Zhang, X.Y.; Zhang, Y.M.; Shen, X.J.; Sun, J.Y.; Ma, Q.L.; Yu, X.M.; Zhu, J.L.; Zhang, L.; Che, H.C. Significant concentration changes of chemical components of PM1 in the Yangtze River Delta area of China and the implications for the formation mechanism of heavy haze-fog pollution. Sci. Total Environ. 2015, 538, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Wu, L. A new perspective on formation of haze-fog: The fuzzy cognitive map and its approaches to data mining. Sustainability 2017, 9, 352. [Google Scholar] [CrossRef]
- Sancini, A.; Tomei, F.; Tomei, G.; Caciari, T.; Di Giorgio, V.; André, J.-C.; Palermo, P.; Andreozzi, G.; Nardone, N.; Schifano, M.P.; et al. Urban pollution. G. Ital. Med. Lav. Erg. 2012, 34, 187–196. [Google Scholar]
- Hu, Y.; Lin, J.; Zhang, S.; Kong, L.; Fu, H.; Chen, J. Identification of the typical metal particles among haze, fog, and clear episodes in the Beijing atmosphere. Sci. Total Environ. 2015, 511, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Allaby, M. Fog, Smog and Poisoned Rain; Facts on File Inc.: New York, NY, USA, 2003; ISBN 10 0816047898. [Google Scholar]
- Li, H.; Wu, H.; Wang, Q.; Yang, M.; Li, F.; Sun, Y.; Qian, X.; Wang, J.; Wang, C. Chemical partitioning of fine particle-bound metals on haze-fog and non-haze-fog days in Nanjing, China and its contribution to human health risks. Atmos. Res. 2017, 183, 142–150. [Google Scholar] [CrossRef]
- Ganguly, N.D.; Tzanis, C. Study of stratosphere-troposphere exchange events of ozone in India and Greece using ozonesonde ascents. Meteorol. Appl. 2011, 18, 467–474. [Google Scholar] [CrossRef]
- Tzanis, C. Ground-based observations of ozone at Athens, Greece during the solar eclipse of 1999. Int. J. Remote Sens. 2005, 26, 3585–3596. [Google Scholar] [CrossRef]
- Le, G.E.; Breysse, P.N.; McDermott, A.; Eftim, S.E.; Geyh, A.; Berman, J.D.; Curriero, F.C. Canadian forest fires and the effects of long-range transboundary air pollution on hospitalizations among the elderly. ISPRS Int. J. Geo-Inf. 2014, 3, 713–731. [Google Scholar] [CrossRef]
- Miranda, A.I.; Monteiro, A.; Martins, V.; Carvalho, A.; Schaap, M.; Builtjes, P.; Borrego, C. Forest fires impact on air quality over Portugal. In Air Pollution Modeling and Its Application XIX; Borrego, C., Miranda, A.I., Eds.; NATO Science for Peace and Security Series Series C: Environmental Security; Springer: Dordrecht, The Netherlands, 2008; pp. 190–198. [Google Scholar] [CrossRef]
- Chan, C.K.; Yao, X.H. Air pollution in megacities in China. Atmos. Environ. 2008, 42, 1–42. [Google Scholar] [CrossRef]
- LaDochy, S. The disappearance of dense fog in Los Angeles: Another urban impact? Phys. Geogr. 2005, 26, 177–191. [Google Scholar] [CrossRef]
- Niu, F.; Li, Z.; Li, C.; Lee, K.-H.; Wang, M. Increase of wintertime fog in China: Potential impacts of weakening of the Eastern Asian monsoon circulation and increasing aerosol loading. J. Geophys. Res. 2010, 115, D00K20. [Google Scholar] [CrossRef]
- Jacob, D.J.; Waldman, J.M.; Munger, J.W.; Hoffmann, M.R. A field investigation of physical and chemical mechanisms affecting pollutant concentrations in fog droplets. Tellus B 1984, 36, 272–285. [Google Scholar] [CrossRef]
- Weiss-Penzias, P.S.; Ortiz, C., Jr.; Acosta, R.P.; Heim, W.; Ryan, J.P.; Fernandez, D.; Collett, J.L., Jr.; Flegal, A.R. Total and monomethyl mercury in fog water from the central California coast. Geophys. Res. Lett. 2012, 39, L03804. [Google Scholar] [CrossRef]
- Pope, C.A., III; Burnett, R.; Thun, M.; Calle, E.; Krewski, D.; Ito, K.; Thurston, G. Lung cancer, cardiopulmonary mortality and long-term exposure to fine particulate air pollution. J. Am. Med. Assoc. 2002, 287, 1132–1141. [Google Scholar] [CrossRef]
- Dong, L.; Qi, J.; Shao, C.; Zhong, X.; Gao, D.; Cao, W.; Gao, J.; Bai, R.; Long, G.; Chu, C. Concentration and size distribution of total airborne microbes in hazy and foggy weather. Sci. Total Environ. 2016, 541, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Hackney, J.D.; Linn, W.S.; Avol, E.L. Potential risks to human respiratory health from “Acid fog”: Evidence from experimental studies of volunteers. Environ. Health Perspect. 1985, 63, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Avol, E.L.; Linn, W.S.; Wightman, L.H.; Whynot, J.D.; Anderson, K.R.; Hackney, J.D. Short-term respiratory effects of sulfuric acid in fog: A laboratory study of healthy and asthmatic volunteers. JAPCA 1988, 38, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Lavorini, F.; Fontana, G.; Pantaleo, T.; Camiciottoli, G.; Castellani, W.; Maluccio, N.; Pistolesi, M. Fog-induced respiratory responses are attenuated by nedocromil sodium in humans. Am. J. Respir. Crit. Care 2001, 163, 1117–1120. [Google Scholar] [CrossRef] [PubMed]
- Hutchings, J.; Herckes, P.; Ervens, B. N-nitrosodimethylamine (NDMA): A potent carcinogen in clouds and fogs. In Proceedings of the AAAR International Conference on Air Pollution and Health: Bridging the Gap from Sources to Heath Outcomes, San Diego, CA, USA, 22–26 March 2010. [Google Scholar]
- Hutchings, J.W.; Ervens, B.; Straub, D.; Herckes, P. N-nitrosodimethylamine occurrence, formation and cycling in clouds and fogs. Environ. Sci. Technol. 2010, 44, 8128–8133. [Google Scholar] [CrossRef] [PubMed]
- Pope III, C.A. Respiratory disease associated with community air pollution and a steel mill, Utah Valley. Am. J. Public Health 1989, 79, 623–628. [Google Scholar] [CrossRef]
- Percy, K.E.; Jensen, K.F.; McQuattie, C.J. Effects of ozone and acidic fog on red spruce needle epicuticular wax production, chemical composition, cuticular membrane ultrastructure and needle wettability. New Phytol. 1992, 122, 71–80. [Google Scholar] [CrossRef]
- Nara, C.; Dokiya, Y.; Usui, N.; Maruta, E.; Fa-Hua, L.; Sagi, T. Acid fog damage of beech at Hinokiboramaru-Tanzawa, Japan. J. Environ. Sci. Health A 2001, 36, 355–366. [Google Scholar] [CrossRef]
- Warren-Rhodes, K.A.; McKay, C.P.; Boyle, L.N.; Wing, M.R.; Kiekebusch, E.M.; Cowan, D.A.; Stomeo, F.; Pointing, S.B.; Kaseke, K.F.; Eckardt, F. et al. Physical ecology of hypolithic communities in the central Namib Desert: The role of fog, rain, rock habitat, and light. J. Geophys. Res. Biogeosci. 2013, 118, 1451–1460. [Google Scholar] [CrossRef]
- Forthun, G.M.; Johnson, M.B.; Schmitz, W.G.; Blume, J.; Caldwell, R.J. Trends in fog frequency and duration in the southeast United States. Phys. Geogr. 2006, 27, 206–222. [Google Scholar] [CrossRef]
- Bullough, J.D.; Rea, M.S. Impacts of fog characteristics, forward illumination, and warning beacon intensity distribution on roadway hazard visibility. Sci. World J. 2016, 2016, 4687816. [Google Scholar] [CrossRef] [PubMed]
- Gultepe, I.; Pearson, G.; Milbrandt, J.A.; Hansen, B.; Platnick, S.; Taylor, P.; Gordon, M.; Oakley, J.P.; Cober, S.G. The fog remote sensing and modeling field project. Bull. Am. Meteorol. Soc. 2009, 90, 341–359. [Google Scholar] [CrossRef]
- Czyż, H.; Markowski, T. Acoustic method of airport fog precipitation. Aviation 2007, 11, 26–30. [Google Scholar]
- Koetse, M.J.; Rietveld, P. The impact of climate change and weather on transport: An overview of empirical findings. Transp. Res. Part D 2009, 14, 205–221. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).