Simulating Climate Change Induced Thermal Stress in Coldwater Fish Habitat Using SWAT Model
Abstract
:1. Introduction
2. Materials and Methods
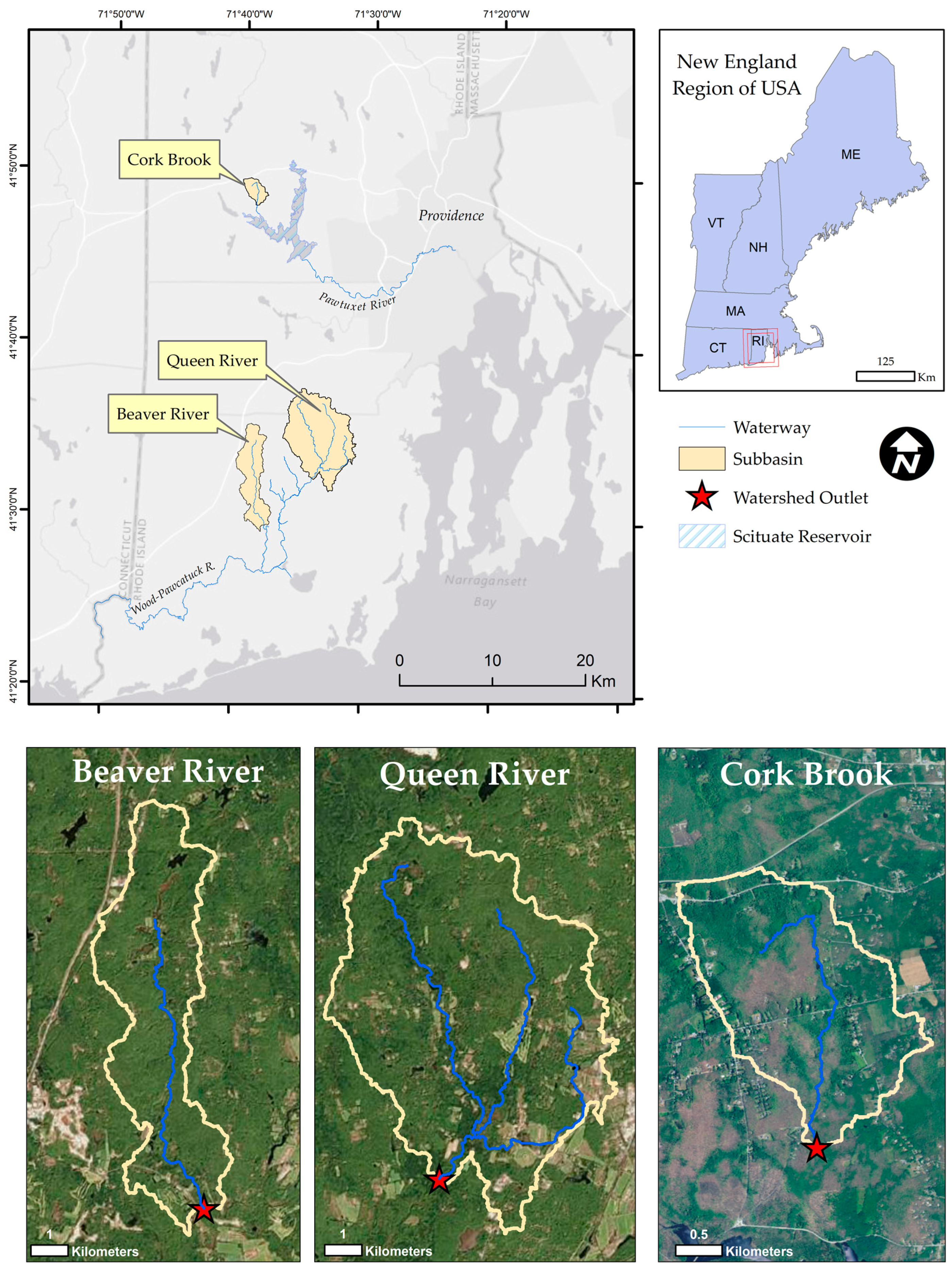
2.1. Study Sites
2.2. Model Setup
2.3. Model Calibration & Validation
2.4. Climate Change Variables
2.5. Stressful Event Identification
3. Results and Discussion
3.1. Historical Conditions
3.2. Future Projections
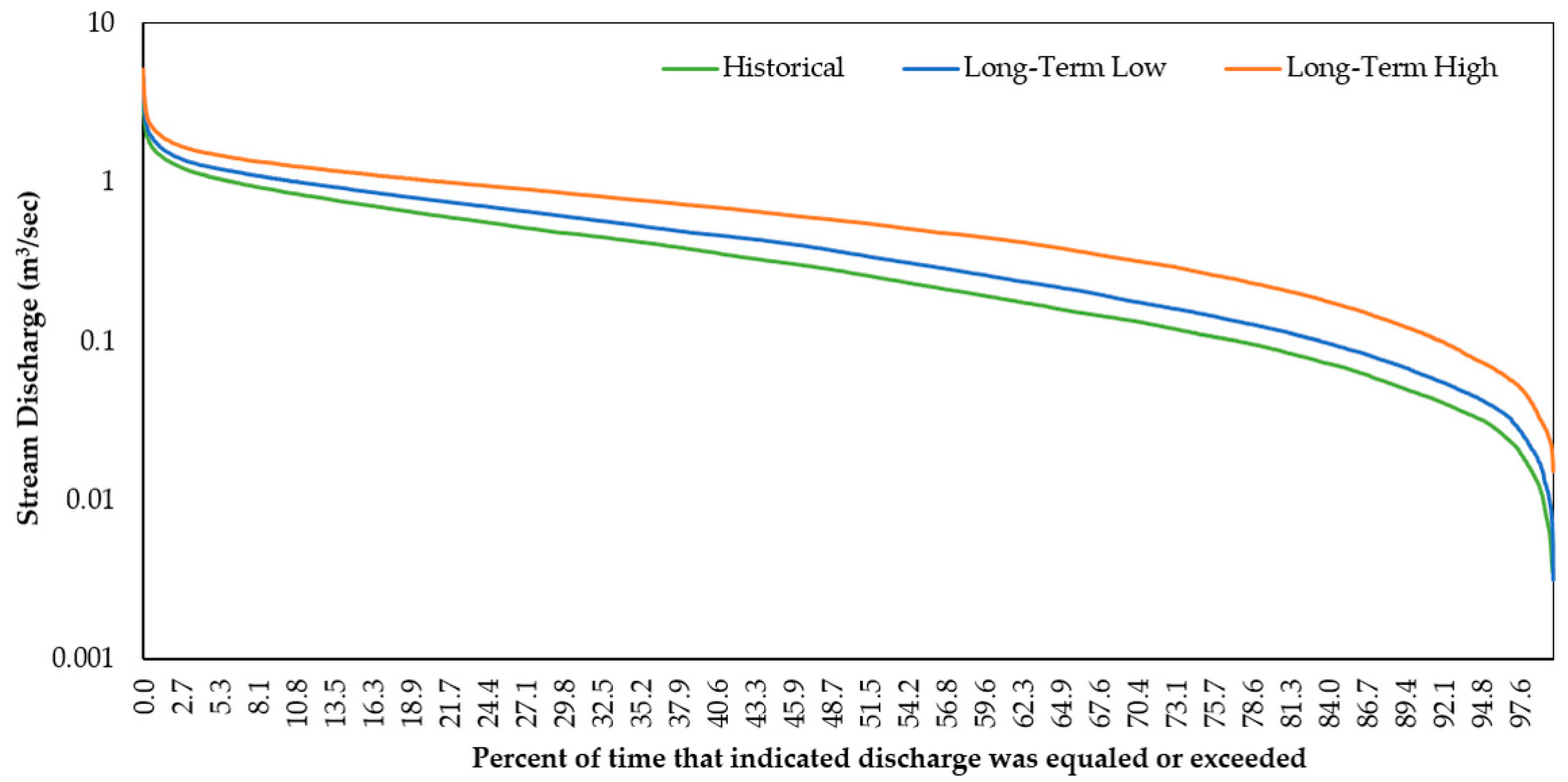
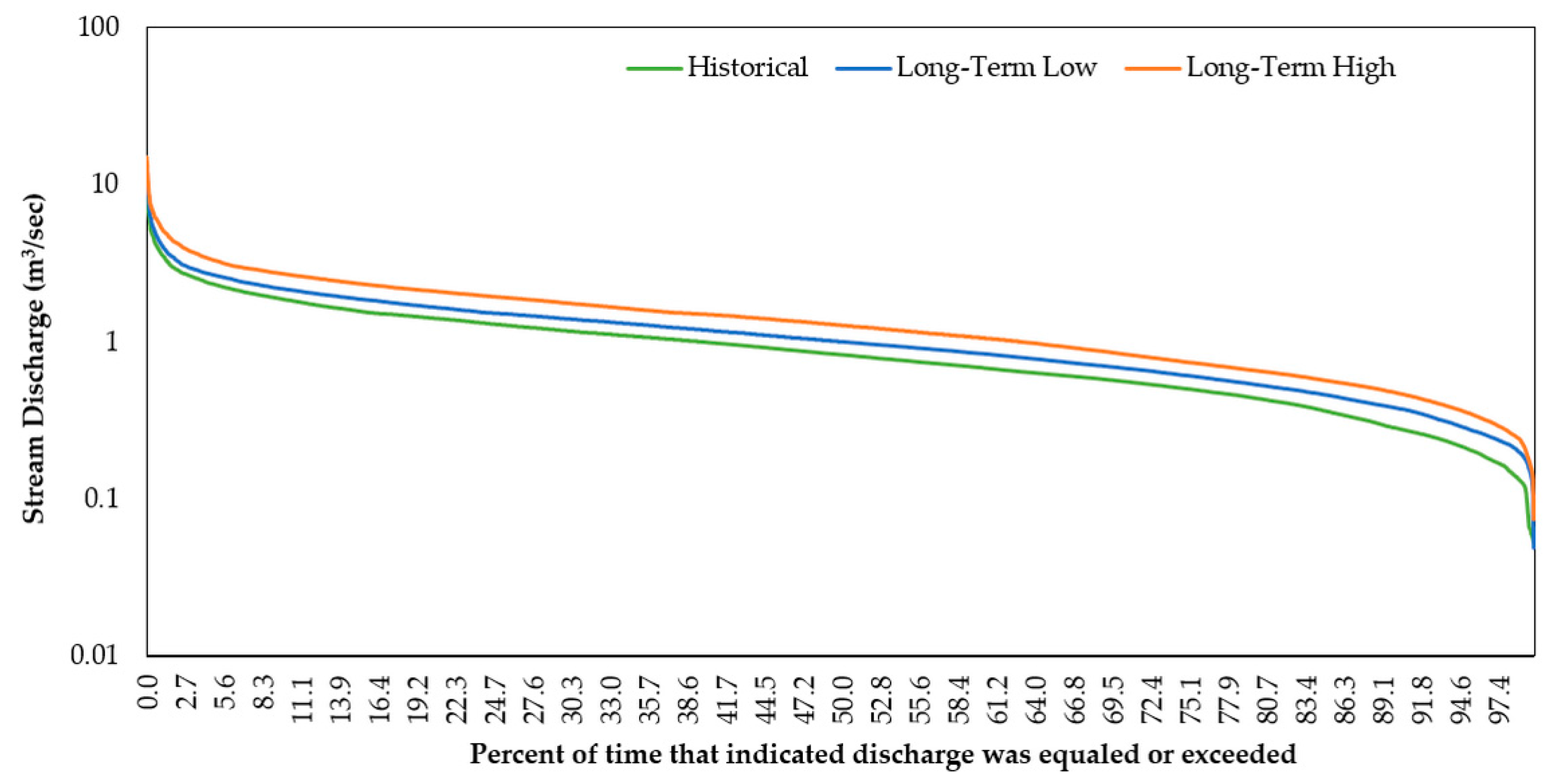
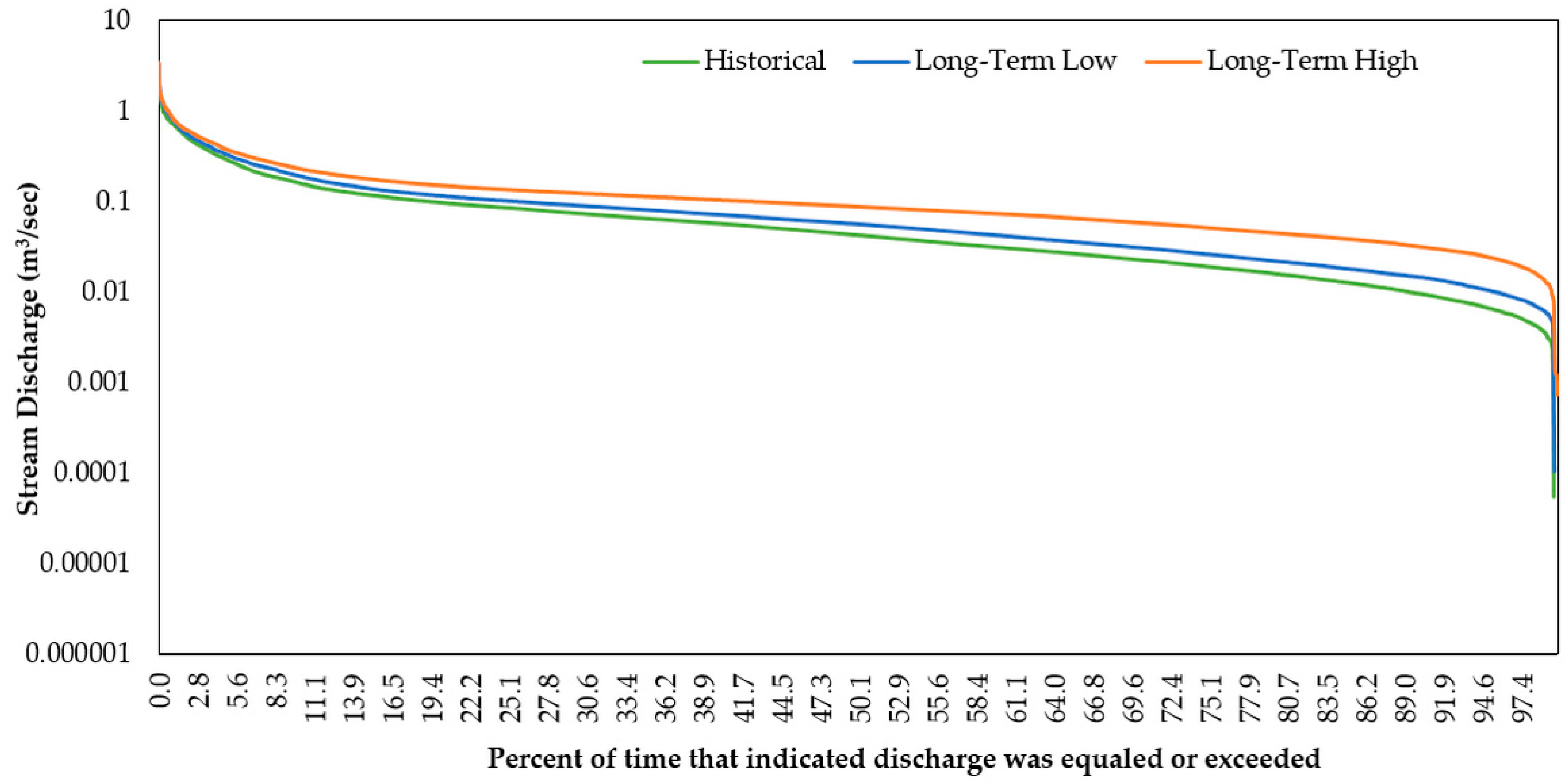
3.2.1. Stream Discharge and Stream Temperature
3.2.2. Flow Regime
3.2.3. Timing of Stream Temperatures
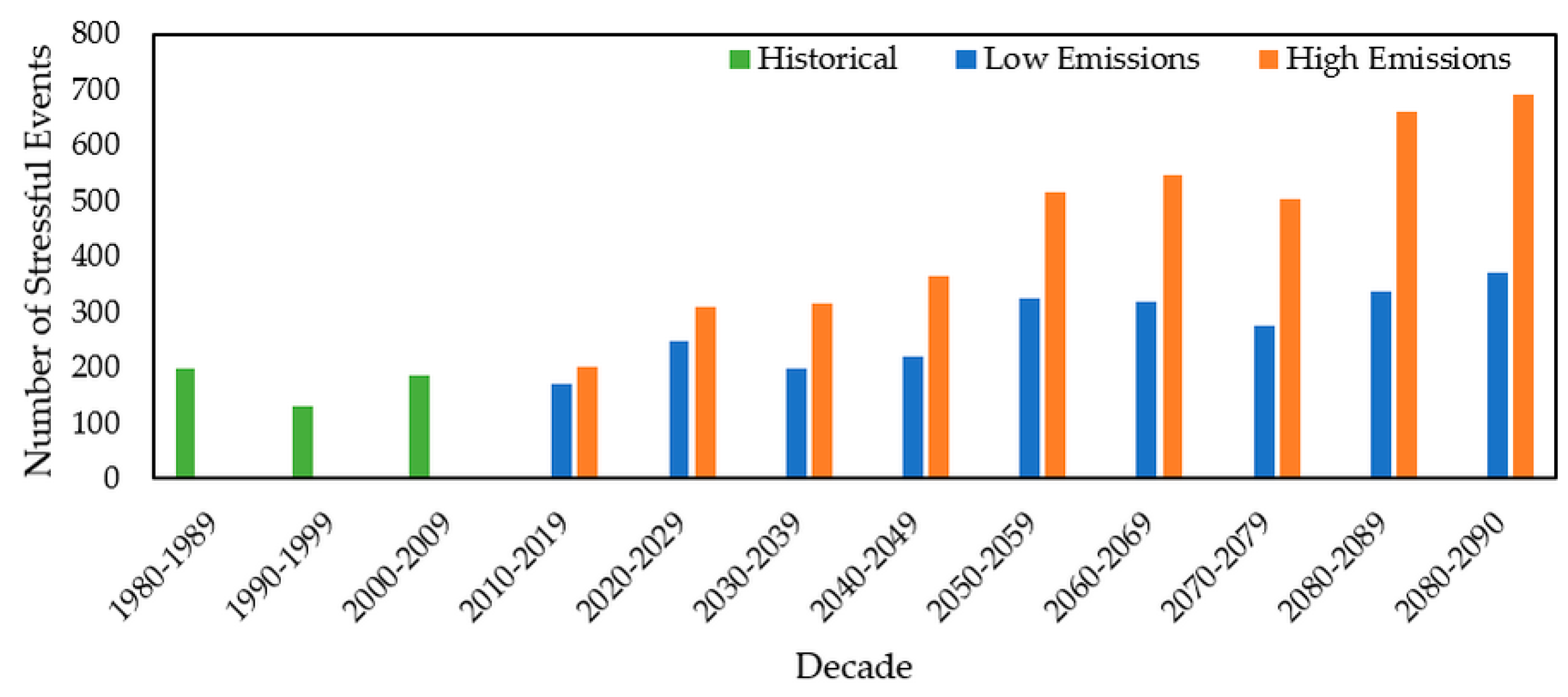
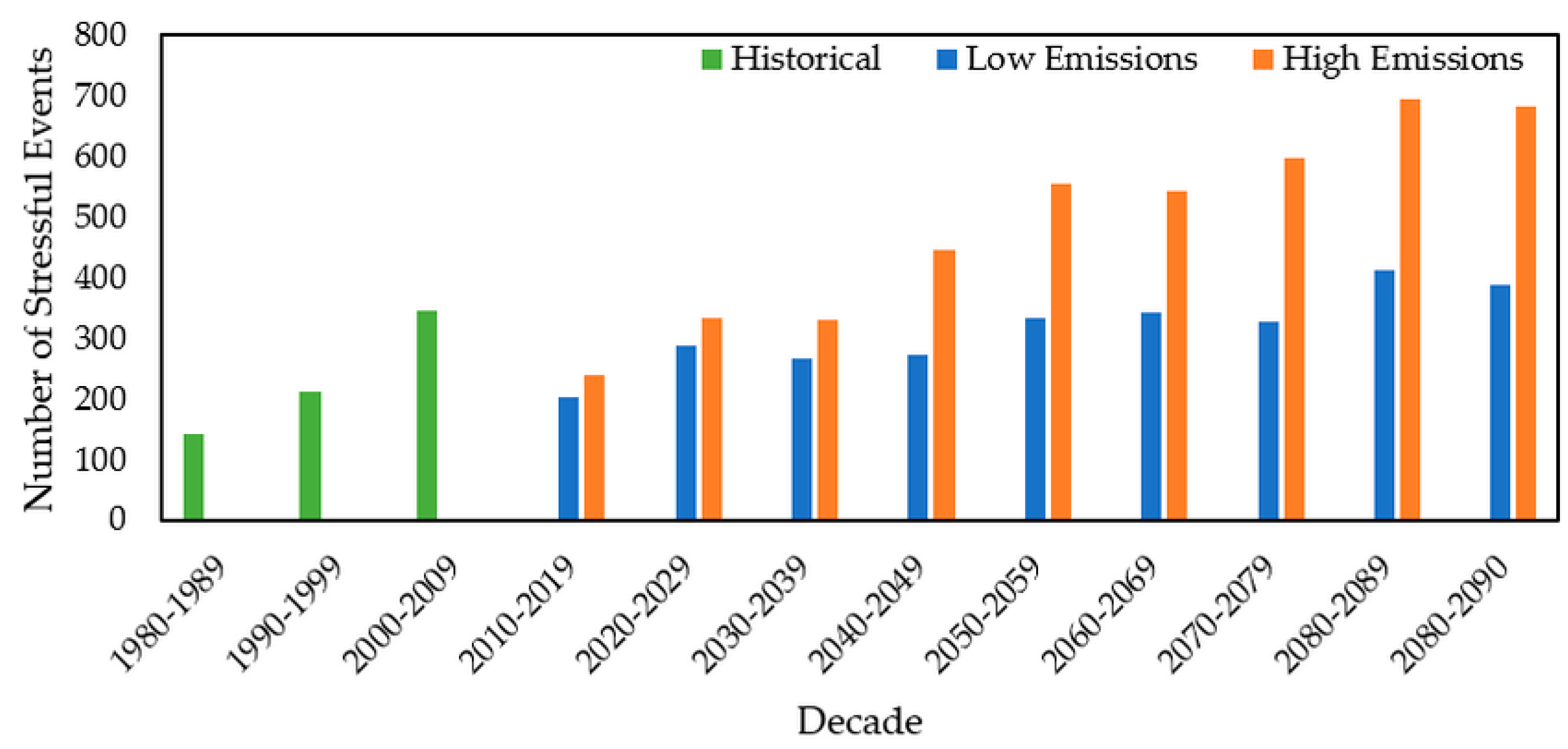
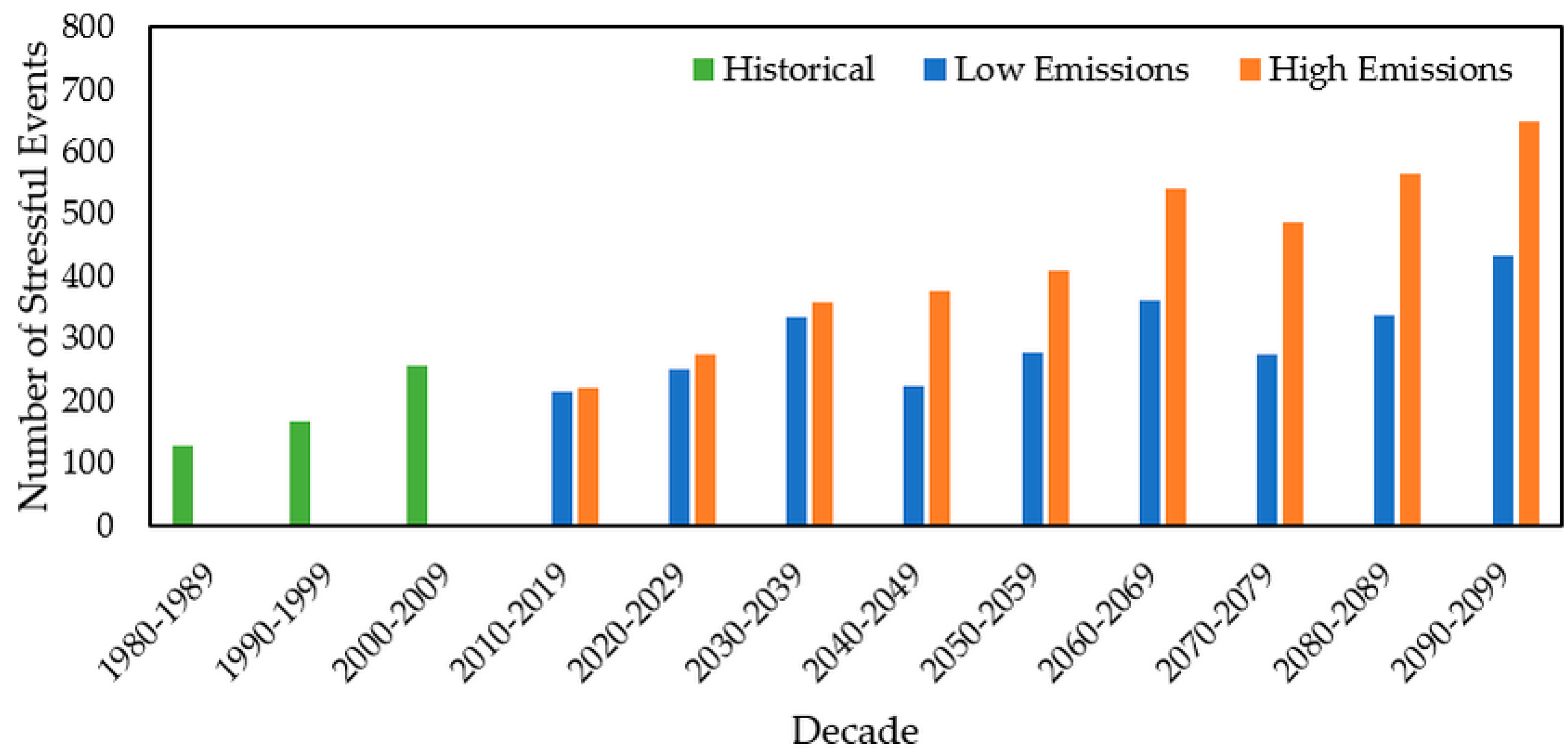
3.2.4. Stressful Event Analysis
4. Conclusions and Future Work
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| Date | Emission Scenario | Unit | Beaver | Queen | Cork |
|---|---|---|---|---|---|
| 1980–1989 | Low | Days | 200 | 141 | 127 |
| % Chance | 5.5% | 3.9% | 3.5% | ||
| High | Days | 200 | 141 | 127 | |
| % Chance | 5.5% | 3.9% | 3.5% | ||
| 1990–1999 | Low | Days | 130 | 213 | 168 |
| % Chance | 3.6% | 5.8% | 4.6% | ||
| High | Days | 130 | 213 | 168 | |
| % Chance | 3.6% | 5.8% | 4.6% | ||
| 2000–2009 | Low | Days | 185 | 346 | 256 |
| % Chance | 5.1% | 9.5% | 7.0% | ||
| High | Days | 185 | 346 | 256 | |
| % Chance | 5.1% | 9.5% | 7.0% | ||
| 2010–2019 | Low | Days | 172 | 141 | 216 |
| % Chance | 4.7% | 3.9% | 5.9% | ||
| High | Days | 203 | 238 | 221 | |
| % Chance | 5.6% | 6.5% | 6.0% | ||
| 2020–2029 | Low | Days | 249 | 213 | 252 |
| % Chance | 6.8% | 5.8% | 6.9% | ||
| High | Days | 308 | 334 | 276 | |
| % Chance | 8.4% | 9.1% | 7.6% | ||
| 2030–2039 | Low | Days | 200 | 346 | 335 |
| % Chance | 5.5% | 9.5% | 9.2% | ||
| High | Days | 317 | 330 | 358 | |
| % Chance | 8.7% | 9.0% | 9.8% | ||
| 2040–2049 | Low | Days | 221 | 273 | 223 |
| % Chance | 6.0% | 7.5% | 6.1% | ||
| High | Days | 364 | 445 | 375 | |
| % Chance | 10.0% | 12.2% | 10.0% | ||
| 2050–2059 | Low | Days | 325 | 334 | 278 |
| % Chance | 8.9% | 9.1% | 7.6% | ||
| High | Days | 516 | 555 | 410 | |
| % Chance | 14.1% | 15.2% | 11.0% | ||
| 2060–2069 | Low | Days | 320 | 343 | 363 |
| % Chance | 8.8% | 9.4% | 9.9% | ||
| High | Days | 547 | 543 | 540 | |
| % Chance | 15.0% | 14.9% | 14.8% | ||
| 2070–2079 | Low | Days | 276 | 326 | 276 |
| % Chance | 7.6% | 8.9% | 7.6% | ||
| High | Days | 502 | 597 | 487 | |
| % Chance | 13.7% | 16.3% | 13.3% | ||
| 2080–2089 | Low | Days | 337 | 412 | 338 |
| % Chance | 9.2% | 11.3% | 9.3% | ||
| High | Days | 662 | 694 | 566 | |
| % Chance | 18.1% | 19.0% | 15.5% | ||
| 2090–2099 | Low | Days | 370 | 389 | 433 |
| % Chance | 10.1% | 10.6% | 11.9% | ||
| High | Days | 692 | 682 | 649 | |
| % Chance | 18.9% | 18.7% | 17.8% |
References
- Hodgkins, G.A.; Dudley, R.W.; Huntington, T.G. Changes in the timing of high river flows in New England over the 20th Century. J. Hydrol. 2003, 278, 244–252. [Google Scholar] [CrossRef]
- Eaton, J.G.; Scheller, R.M. Effects of climate warming on fish thermal habitat in streams of the United States. Limnol. Oceanogr. 1996, 41, 1109–1115. [Google Scholar] [CrossRef]
- Hayhoe, K.; Wake, C.; Anderson, B.; Liang, X.-Z.; Maurer, E.; Zhu, J.; Bradbury, J.; DeGaetano, A.; Stoner, A.M.; Wuebbles, D. Regional climate change projections for the Northeast USA. Mitig. Adapt. Strateg. Glob. Chang. 2008, 13, 425–436. [Google Scholar] [CrossRef]
- Hayhoe, K.; Wake, C.P.; Huntington, T.G.; Luo, L.; Schwartz, M.D.; Sheffield, J.; Wood, E.; Anderson, B.; Bradbury, J.; DeGaetano, A.; et al. Past and future changes in climate and hydrological indicators in the US Northeast. Clim. Dyn. 2007, 28, 381–407. [Google Scholar] [CrossRef]
- Mohseni, O.; Erickson, T.R.; Stefan, H.G. Sensitivity of stream temperatures in the United States to air temperatures projected under a global warming scenario. Water Resour. Res. 1999, 35, 3723–3733. [Google Scholar] [CrossRef]
- Woodward, G.; Perkins, D.M.; Brown, L.E. Climate change and freshwater ecosystems: Impacts across multiple levels of organization. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 2093–2106. [Google Scholar] [CrossRef] [PubMed]
- Whitney, J.E.; Al-Chokhachy, R.; Bunnell, D.B.; Caldwell, C.A.; Cooke, S.J.; Eliason, E.J.; Rogers, M.; Lynch, A.J.; Paukert, C.P. Physiological basis of climate change impacts on North American Inland Fishes. Fisheries 2016, 41, 332–345. [Google Scholar] [CrossRef]
- Van Vliet, M.T.H.; Franssen, W.H.P.; Yearsley, J.R.; Ludwig, F.; Haddeland, I.; Lettenmaier, D.P.; Kabat, P. Global river discharge and water temperature under climate change. Glob. Environ. Chang. 2013, 23, 450–464. [Google Scholar] [CrossRef]
- Jiménez Cisneros, B.E.; Oki, T.; Arnell, N.W.; Benito, G.; Cogley, J.G.; Doll, P.; Jiang, T.; Mwakalila, S.S. 2014: Freshwater resources. In Climate Change 2014: Impacts, Adaptation and Vulnerability. Part A: Global and Sectoral Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Kundzewicz, Z., Ed.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014; Volume 40. [Google Scholar]
- Brett, J.R. Some principles in the thermal requirements of fishes. Q. Rev. Biol. 1956, 31, 75–87. [Google Scholar] [CrossRef]
- Fry, F.E.J. The effect of environmental factors on the physiology of fish. In Fish Physiology; Hoar, W.S., Randall, D.J., Eds.; Academic Press: Cambridge, MA, USA, 1971; pp. 1–98. [Google Scholar]
- Raleigh, R.F. Habitat Suitability Index Models: Brook trout. In FWS/OBS; United States Fish and Wildlife Service: Washington, DC, USA, 1982. [Google Scholar]
- Hokanson, K.E.F.; McCormick, J.H.; Jones, B.R.; Tucker, J.H. Thermal requirements for maturation, spawning, and embryo survival of the brook trout, salvelinus fontinalis. J. Fish. Res. Board Can. 1973, 30, 975–984. [Google Scholar] [CrossRef]
- Milner, N.J.; Elliott, J.M.; Armstrong, J.D.; Gardiner, R.; Welton, J.S.; Ladle, M. The natural control of salmon and trout populations in streams. Fish. Res. 2003, 62, 111–125. [Google Scholar] [CrossRef]
- Peterson, J.T.; Kwak, T.J. Modeling the effects of land use and climate change on riverine smallmouth bass. Ecol. Appl. 1999, 9, 1391–1404. [Google Scholar] [CrossRef]
- Goniea, T.M.; Keefer, M.L.; Bjornn, T.C.; Peery, C.A.; Bennett, D.H.; Stuehrenberg, L.C. Behavioral thermoregulation and slowed migration by adult fall chinook salmon in response to high columbia river water temperatures. Trans. Am. Fish. Soc. 2006, 135, 408–419. [Google Scholar] [CrossRef]
- Arnold, J.G.; Srinivasan, R.; Muttiah, R.S.; Williams, J.R. Large area hydrologic modeling and assessment part I: Model development 1. JAWRA J. Am. Water Resour. Assoc. 1998, 34, 73–89. [Google Scholar] [CrossRef]
- Isaak, D.J.; Wollrab, S.; Horan, D.; Chandler, G. Climate change effects on stream and river temperatures across the northwest U.S. from 1980–2009 and implications for salmonid fishes. Clim. Chang. 2012, 113, 499–524. [Google Scholar] [CrossRef]
- Luo, Y.; Ficklin, D.L.; Liu, X.; Zhang, M. Assessment of climate change impacts on hydrology and water quality with a watershed modeling approach. Sci. Total Environ. 2013, 450, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Mohseni, O.; Stefan, H.G. Stream temperature/air temperature relationship: A physical interpretation. J. Hydrol. 1999, 218, 128–141. [Google Scholar] [CrossRef]
- Null, S.; Viers, J.; Deas, M.; Tanaka, S.; Mount, J. Stream temperature sensitivity to climate warming in California’s Sierra Nevada. In AGU Fall Meeting Abstracts; Utah State University: Logan, UT, USA, 2010. [Google Scholar]
- Preud’homme, E.B.; Stefan, H.G. Relationship between Water Temperatures and Air Temperatures for Central US Streams; St. Anthony Falls Hydraulic Lab, Minnesota University: Minneapolis, MN, USA, 1992; 146p. [Google Scholar]
- Anandhi, A.; Frei, A.; Pierson, D.C.; Schneiderman, E.M.; Zion, M.S.; Lounsbury, D.; Matonse, A.H. Examination of change factor methodologies for climate change impact assessment. Water Resour. Res. 2011, 47, 1–10. [Google Scholar] [CrossRef]
- Saila, S.; Cheeseman, M.; Poyer, D. Maximum Stream Temperature Estimation from Air Temperature Data and Its Relationship to Brook Trout (Salvelinus Fontinalis) Habitat Requirements in Rhode Island; Wood-Pawcatuck Watershed Association: Hope Valley, RI, USA, 2004. [Google Scholar]
- Mohseni, O.; Stefan, H.G.; Eaton, J.G. Global warming and potential changes in fish habitat in U.S. Streams. Clim. Chang. 2003, 59, 389–409. [Google Scholar] [CrossRef]
- Climate Solutions New England. Rhode Island’s Climate Past and Future Changes; Sustainability Institute at the University of New Hampshire: Durham, NH, USA, 2016; Available online: http://clim-map.csrcdev.com/files/Rhode%20Island%20(Total)%20Fact%20Sheet.pdf (accessed on 1 March 2017).
- Wake, C.; Large, S. Climate Grids for Rhode Island; Sustainability Institute at the University of New Hampshire: Durham, NH, USA, 2014; Available online: clim-map.csrcdev.com/files/Rhode%20Island%20(Total)%20Climate%20Grids.pdf (accessed on 1 March 2017).
- Wake, C.P.; Keeley, C.; Burakowski, E.; Wilkinson, P.; Hayhoe, K.; Stoner, A.; LaBrance, J. Climate Change in Northern New Hampshire: Past, Present and Future; Sustainability Institute at the University of New Hampshire: Durham, NH, USA, 2014; Available online: http://scholars.unh.edu/sustainability (accessed on 1 March 2017).
- Erkan, D.E. Strategic Plan for the Restoration of Anadromous Fishes to Rhode Island Coastal Streams; Rhode Island Department of Environmental Management, Division of Fish and Wildlife: Wakefield, RI, USA, 2002. [Google Scholar]
- WPWA; Saila, S.; Cheeseman, M.; Poyer, D. Assessing Habitat Requirements for Brook Trout (Salvelinus fontinalis) in Low Order Streams; Wood-Pawcatuck Watershed Association: Hope Valley, RI, USA, 2004. [Google Scholar]
- Hakala, J.P.; Hartman, K.J. Drought effect on stream morphology and brook trout (Salvelinus fontinalis) populations in forested headwater streams. Hydrobiologia 2004, 515, 203–213. [Google Scholar] [CrossRef]
- Bjornn, T.; Reiser, D. Habitat requirements of salmonids in streams. Am. Fish. Soc. Spec. Publ. 1991, 19, 138. [Google Scholar]
- Chadwick, J.J.G.; Nislow, K.H.; McCormick, S.D. Thermal onset of cellular and endocrine stress responses correspond to ecological limits in brook trout, an iconic cold-water fish. Conserv. Physiol. 2015, 3, cov017. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.M.; Rinne, J.N. Critical thermal maxima of five trout species in the southwestern United States. Trans. Am. Fish. Soc. 1980, 109, 632–635. [Google Scholar] [CrossRef]
- Letcher, B.H.; Nislow, K.H.; Coombs, J.A.; O’Donnell, M.J.; Dubreuil, T.L. Population response to habitat fragmentation in a stream-dwelling brook trout population. PLoS ONE 2007, 2, e1139. [Google Scholar] [CrossRef] [PubMed]
- Kling, G.W.; Hayhoe, K.; Johnson, L.B.; Magnuson, J.J.; Polasky, S.; Robinson, S.K.; Shuter, B.J.; Wander, M.M.; Wuebbles, D.J.; Zak, D.R. Confronting Climate Change in the Great Lakes Region: Impacts on Our Communities and Ecosystems; Union of Concerned Scientists: Cambridge, MA, USA; Ecological Society of America: Washington, DC, USA, 2003; p. 92. [Google Scholar]
- Magnuson, J.J.; Crowder, L.B.; Medvick, P.A. Temperature as an ecological resource. Am. Zool. 1979, 19, 331–343. [Google Scholar] [CrossRef]
- Vannote, R.L.; Minshall, G.W.; Cummins, K.W.; Sedell, J.R.; Cushing, C.E. The river continuum concept. Can. J. Fish. Aquat. Sci. 1980, 37, 130–137. [Google Scholar] [CrossRef]
- Bunn, S.E.; Arthington, A.H. Basic principles and ecological consequences of altered flow regimes for aquatic biodiversity. Environ. Manag. 2002, 30, 492–507. [Google Scholar] [CrossRef]
- Freeman, M.C.; Pringle, C.M.; Jackson, C.R. Hydrologic connectivity and the contribution of stream headwaters to ecological integrity at regional scales1. JAWRA J. Am. Water Resour. Assoc. 2007, 43, 5–14. [Google Scholar] [CrossRef]
- Poff, N.L.; Allan, J.D. Functional organization of stream fish assemblages in relation to hydrological variability. Ecology 1995, 76, 606–627. [Google Scholar] [CrossRef]
- Poff, N.L.; Allan, J.D.; Bain, M.B.; Karr, J.R.; Prestegaard, K.L.; Richter, B.D.; Sparks, R.E.; Stromberg, J.C. The natural flow regime. Bioscience 1997, 47, 769–784. [Google Scholar] [CrossRef]
- Bassar, R.D.; Letcher, B.H.; Nislow, K.H.; Whiteley, A.R. Changes in seasonal climate outpace compensatory density-dependence in eastern brook trout. Glob. Chang. Biol. 2016, 22, 577–593. [Google Scholar] [CrossRef] [PubMed]
- DePhilip, M.; Moberg, T. Ecosystem Flow Recommendations for the Susquehanna River Basin; The Nature Conservancy: Harrisburg, PA, USA, 2010. [Google Scholar]
- Nuhfer, A.J.; Zorn, T.G.; Wills, T.C. Effects of reduced summer flows on the brook trout population and temperatures of a groundwater-influenced stream. Ecol. Freshw. Fish 2017, 26, 108–119. [Google Scholar] [CrossRef]
- Walters, A.W.; Post, D.M. An experimental disturbance alters fish size structure but not food chain length in streams. Ecology 2008, 89, 3261–3267. [Google Scholar] [CrossRef] [PubMed]
- Saila, S.; Burgess, D.; Cheeseman, M.; Fisher, K.; Clark, B. Interspecific Association, Diversity, and Population Analysis of Fish Species in the Wood-Pawcatuck Watershed; Wood-Pawcatuck Watershed Association: Hope Valley, RI, USA, 2003. [Google Scholar]
- Fulweiler, R.W.; Nixon, S.W. Export of nitrogen, phosphorus, and suspended solids from a southern New England watershed to little Narragansett bay. Biogeochemistry 2005, 76, 567–593. [Google Scholar] [CrossRef]
- Poyer, D.; Hetu, M. Study of Maximum Daily Stream Temperature of Select Streams in the Pawcatuck Watershed Summer 2005; Wood-Pawcatuck Watershed Association: Hope Valley, RI, USA, 2005. [Google Scholar]
- Dickerman, D.C.; Ozbilgin, M.M. Hydrogeology, water quality, and ground-water development alternatives in the Beaver-Pasquiset ground-water reservoir, Rhode Island. In Water-Resources Investigations Report; US Geological Survey: Reston, VA, USA, 1985. [Google Scholar]
- Kliever, J.D. Hydrologic data for the Usquepaug-Queen river basin, Rhode Island. In Open-File Report; US Geological Survey: Reston, VA, USA, 1995. [Google Scholar]
- Liu, T.; Merrill, N.H.; Gold, A.J.; Kellogg, D.Q.; Uchida, E. Modeling the production of multiple ecosystem services from agricultural and forest landscapes in Rhode Island. Agric. Resour. Econ. Rev. 2013, 42, 251–274. [Google Scholar] [CrossRef]
- Poyer, D.; Hetu, M. Maximum Daily Stream Temperature in the Queen River Watershed and Mastuxet Brook Summer 2006; Wood-Pawcatuck Watershed Association: Hope Valley, RI, USA, 2006. [Google Scholar]
- The Nature Conservancy (TNC). Beaver River Preserve. Places We Protect 2017; The Nature Conservancy Is a Nonprofit, Charitable Organization under Section 501(c)(3). Available online: https://www.nature.org/ourinitiatives/regions/northamerica/unitedstates/rhodeisland/placesweprotect/beaver-river-preserve.xml (accessed on 1 May 2017).
- Armstrong, D.S.; Parker, G.W. Assessment of Habitat and Streamflow Requirements for Habitat Protection, Usquepaug-Queen River, Rhode Island, 1999–2000; USGS, Ed.; DTIC Document; DTIC: Denver, CO, USA, 2003.
- Tefft, E. Factors affecting the distribution of brook trout (salvelinus fontinalis) in the wood-pawcatuck watershed of Rhode Island. In Department of Natural Resources; University of Rhode Island: Kingston, RI, USA, 2013. [Google Scholar]
- The Nature Conservancy (TNC). Queen’s River Preserve. Places We Protect 2017; The Nature Conservancy Is a Nonprofit, Charitable Organization under Section 501(c)(3). Available online: https://www.nature.org/ourinitiatives/regions/northamerica/unitedstates/rhodeisland/placesweprotect/queens-river-preserve.xml (accessed on 1 May 2017).
- US Geological Survey (USGS). National Water Information System Web Interface; US Geological Survey: Reston, VA, USA, 2017.
- Arnold, J.G.; Allen, P.M. Automated methods for estimating baseflow and ground water recharge from streamflow records. JAWRA J. Am. Water Resour. Assoc. 1999, 35, 411–424. [Google Scholar] [CrossRef]
- Douglas-Mankin, K.R.; Srinivasan, R.; Arnold, J.G. Soil and Water Assessment Tool (SWAT) Model: Current developments and applications. Trans. ASABE 2010, 53, 1423–1431. [Google Scholar] [CrossRef]
- Gassman, P.W.; Reyes, M.R.; Green, C.H.; Arnold, J.G. The soil and water assessment tool: Historical development, applications, and future research directions. Trans. ASABE 2007, 50, 1211–1250. [Google Scholar] [CrossRef]
- Neitsch, S.L.; Arnold, J.G.; Kiniry, J.R.; Williams, J.R. Soil and Water Assessment Tool Theoretical Documentation Version 2009; Texas Water Resources Institute: College Station, TX, USA, 2011. [Google Scholar]
- Penman, H.L. Estimating evaporation. EOS Trans. Am. Geophys. Union 1956, 37, 43–50. [Google Scholar] [CrossRef]
- Monteith, J.L. Evaporation and environment. Symp. Soc. Exp. Biol. 1965, 19, 4. [Google Scholar]
- Stefan, H.G.; Preud’homme, E.B. Stream Temperature estimation from air temperature. JAWRA J. Am. Water Resour. Assoc. 1993, 29, 27–45. [Google Scholar] [CrossRef]
- RIGIS. Rhode Island Geographic Information System; University of Rhode Island: Kingston, RI, USA, 2016. [Google Scholar]
- Texas A&M University. Arcswat Software; Texas A&M University: College Station, TX, USA, 2012; Available online: http://swat.tamu.edu/ (accessed on 19 Octorber 2012).
- Homer, C.G.; Dewitz, J.A.; Yang, L.; Jin, S.; Danielson, P.; Xian, G.; Coulston, J.; Herold, N.D.; Wickham, J.; Megown, K. Completion of the 2011 National Land Cover Database for the conterminous United States-Representing a decade of land cover change information. Photogramm. Eng. Remote Sens. 2015, 81, 345–354. [Google Scholar]
- Rhode Island Geographic Information System (RIGIS) Data Distribution System SOIL_Soils. Available online: http://www.rigis.org/geodata/soil/Soils16.zip (accessed on 5 September 2016).
- Saha, S.; Moorthi, S.; Wu, X.; Wang, J.; Nadiga, S.; Tripp, P.; Behringer, D.; Hou, Y.-T.; Chuang, H.-Y.; Iredell, M.; et al. The NCEP climate forecast system version 2. J. Clim. 2014, 27, 2185–2208. [Google Scholar] [CrossRef]
- Texas A&M, U. NCEP Global Weather Data for Swat. Available online: https://globalweather.tamu.edu (accessed on 3 January 2016).
- Abbaspour, K.C. SWAT-CUP 2012; SWAT Calibration and Uncertainty Program—A User Manual; Swiss Federal Institute of Aquatic Science and Technology Eawag: Duebendorf, Switzerland, 2013. [Google Scholar]
- Abbaspour, K. User Manual for SWAT-CUP, SWAT Calibration and Uncertainty Analysis Programs; Swiss Federal Institute of Aquatic Science and Technology Eawag: Duebendorf, Switzerland, 2007. [Google Scholar]
- Nash, J.E.; Sutcliffe, J.V. River flow forecasting through conceptual models part I—A discussion of principles. J. Hydrol. 1970, 10, 282–290. [Google Scholar] [CrossRef]
- Moriasi, D.N.; Arnold, J.G.; Liew, M.W.V.; Bingner, R.L.; Harmel, R.D.; Veith, T.L. Model evaluation guidelines for systematic quantification of accuracy in watershed simulations. Trans. ASABE 2007, 50, 885–900. [Google Scholar] [CrossRef]
- Pradhanang, S.M.; Mukundan, R.; Schneiderman, E.M.; Zion, M.S.; Anandhi, A.; Pierson, D.C.; Frei, A.; Easton, Z.M.; Fuka, D.; Steenhuis, T.S. Streamflow responses to climate change: Analysis of hydrologic indicators in a New York City water supply watershed. J. Am. Water Resour. Assoc. 2013, 49, 1308–1326. [Google Scholar] [CrossRef]
- Liew, M.W.V.; Veith, T.L.; Bosch, D.D.; Arnold, J.G. Suitability of SWAT for the conservation effects assessment project: Comparison on USDA agricultural research service watersheds. J. Hydrol. Eng. 2007, 12, 173–189. [Google Scholar] [CrossRef]
- Arnold, J.G.; Allen, P.M.; Muttiah, R.; Bernhardt, G. Automated base flow separation and recession analysis techniques. Ground Water 1995, 33, 1010–1018. [Google Scholar] [CrossRef]
- Singh, J.; Knapp, H.V.; Arnold, J.G.; Demissie, M. Hydrological modeling of the Iroquois river watershed using HSPF and SWAT. JAWRA J. Am. Water Resour. Assoc. 2005, 41, 343–360. [Google Scholar] [CrossRef]
- Pyrce, R. Hydrological Low Flow Indices and Their Uses; Watershed Science Centre (WSC) Report; Watershed Science Center, Trent University: Peterborough, ON, Canada, 2004. [Google Scholar]
- Smakhtin, V.U. Low flow hydrology: A review. J. Hydrol. 2001, 240, 147–186. [Google Scholar] [CrossRef]
- Ahearn, E.A. Flow Durations, Low-Flow Frequencies, and Monthly Median Flows for Selected Streams in Connecticut through 2005; US Department of the Interior, US Geological Survey: Reston, VA, USA, 2008. [Google Scholar]
- Demaria, E.M.C.; Palmer, R.N.; Roundy, J.K. Regional climate change projections of streamflow characteristics in the Northeast and Midwest U.S. J. Hydrol. Reg. Stud. 2016, 5, 309–323. [Google Scholar] [CrossRef]
- Douglas, E.M.; Vogel, R.M.; Kroll, C.N. Trends in floods and low flows in the United States: Impact of spatial correlation. J. Hydrol. 2000, 240, 90–105. [Google Scholar] [CrossRef]
- Ficklin, D.L. SWAT Stream Temperature Executable Code; Indiana State University: Terre Haute, IN, USA, 2012. [Google Scholar]
- Meisner, J.D.; Rosenfeld, J.S.; Regier, H.A. The role of groundwater in the impact of climate warming on stream salmonines. Fisheries 1988, 13, 2–8. [Google Scholar] [CrossRef]
- Moore, R.D.; Spittlehouse, D.L.; Story, A. Riparian microclimate and stream temperature response to forest harvesting: A Review. J. Am. Water Resour. Assoc. 2005, 41, 813–834. [Google Scholar] [CrossRef]
- Rishel, G.B.; Lynch, J.A.; Corbett, E.S. Seasonal stream temperature changes following forest harvesting. J. Environ. Qual. 1982, 11, 112–116. [Google Scholar] [CrossRef]
- Ficklin, D.L.; Luo, Y.; Stewart, I.T.; Maurer, E.P. Development and application of a hydroclimatological stream temperature model within the Soil and Water Assessment Tool. Water Resour. Res. 2012, 48. [Google Scholar] [CrossRef]
- Brennan, L. Stream Temperature Modeling: A modeling comparison for resource managers and climate change analysis. In Environmental and Water Resources Engineering; University of Massachusetts: Amherst, MA, USA, 2015. [Google Scholar]
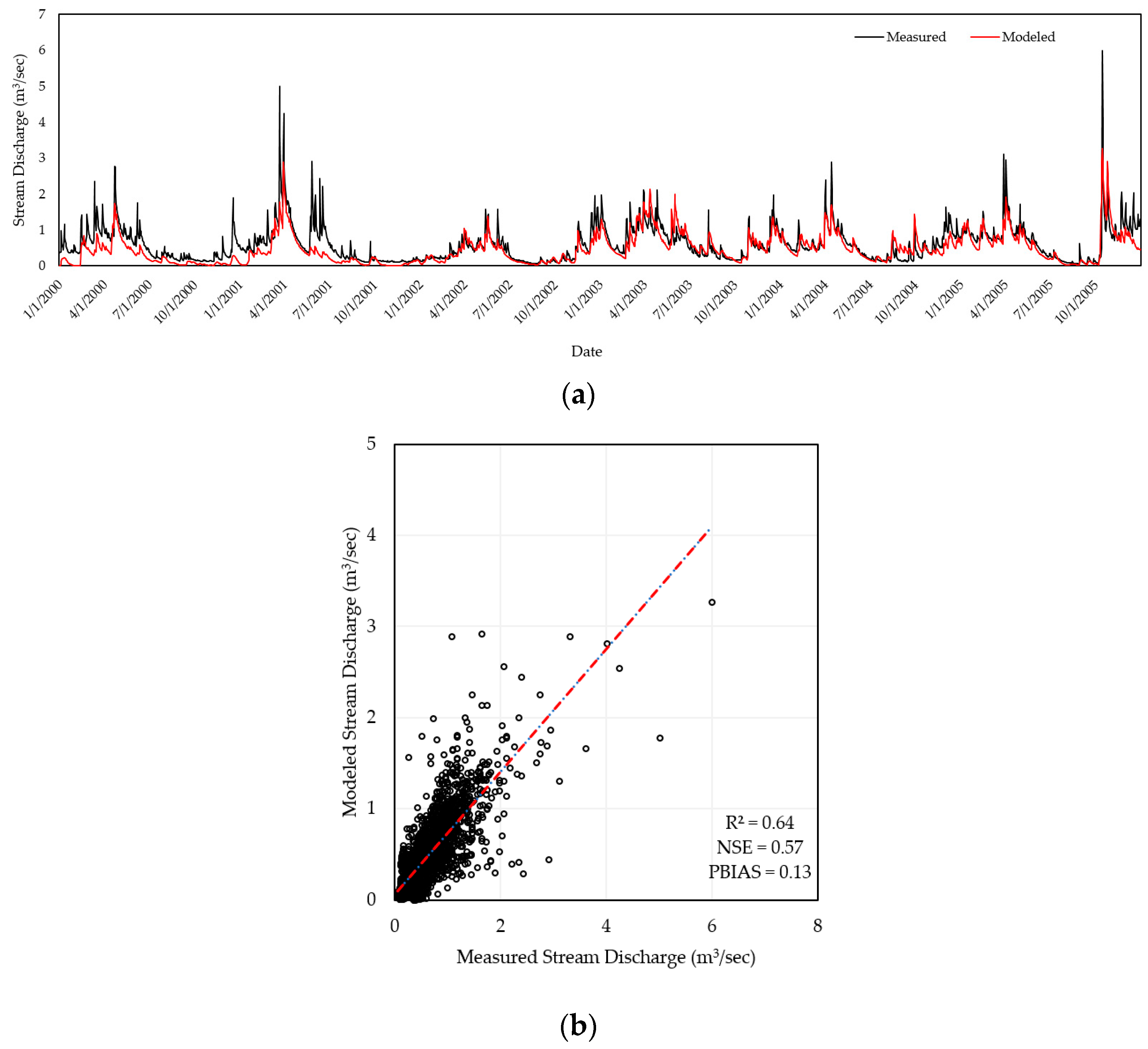
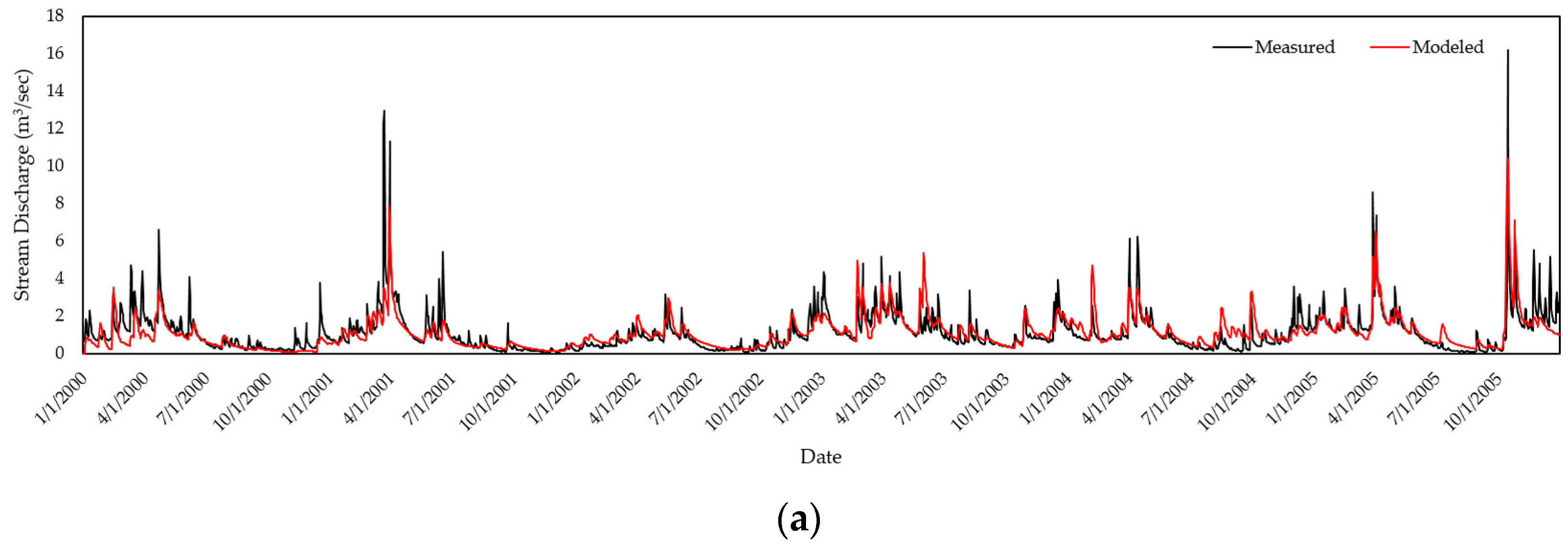
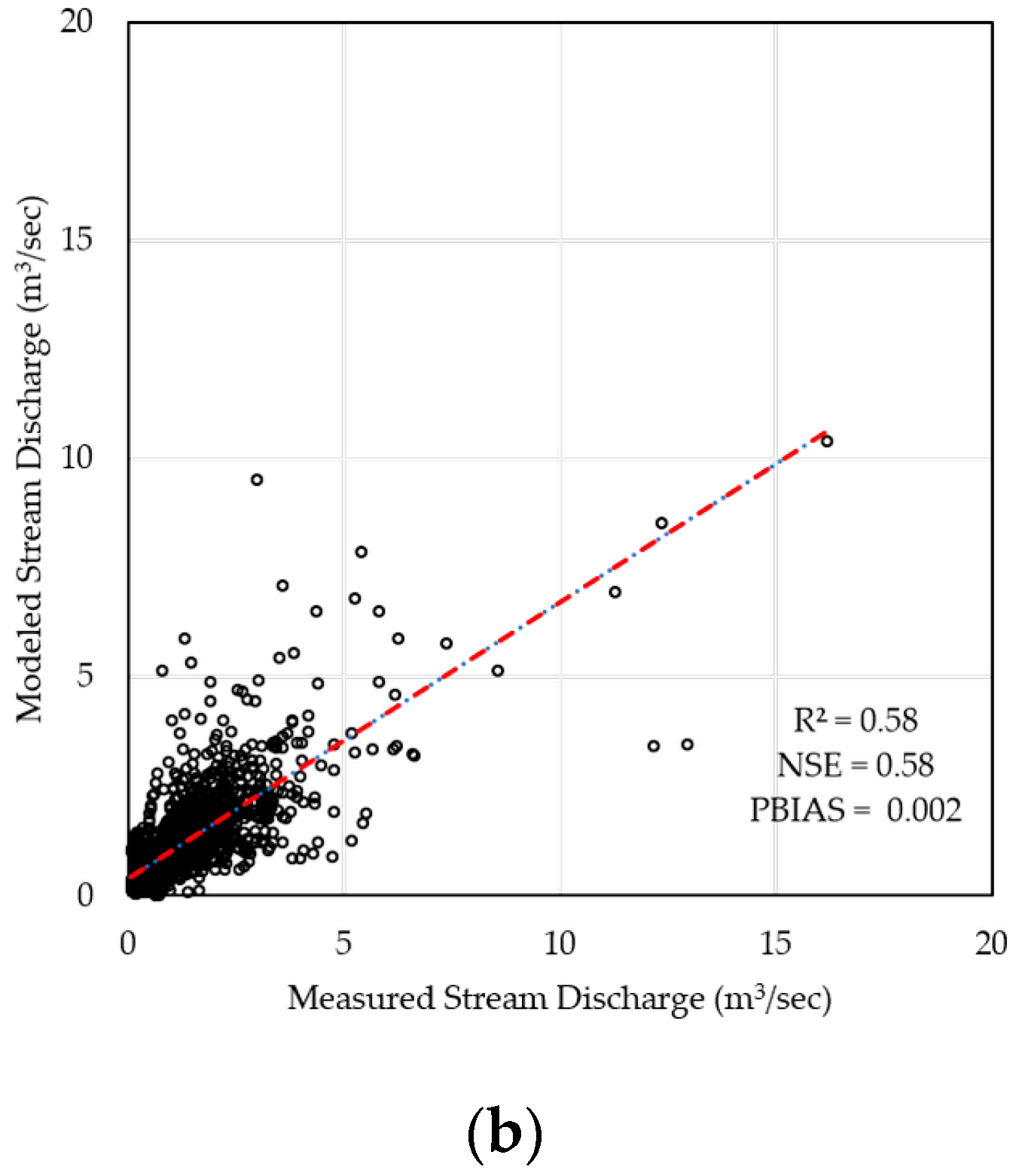
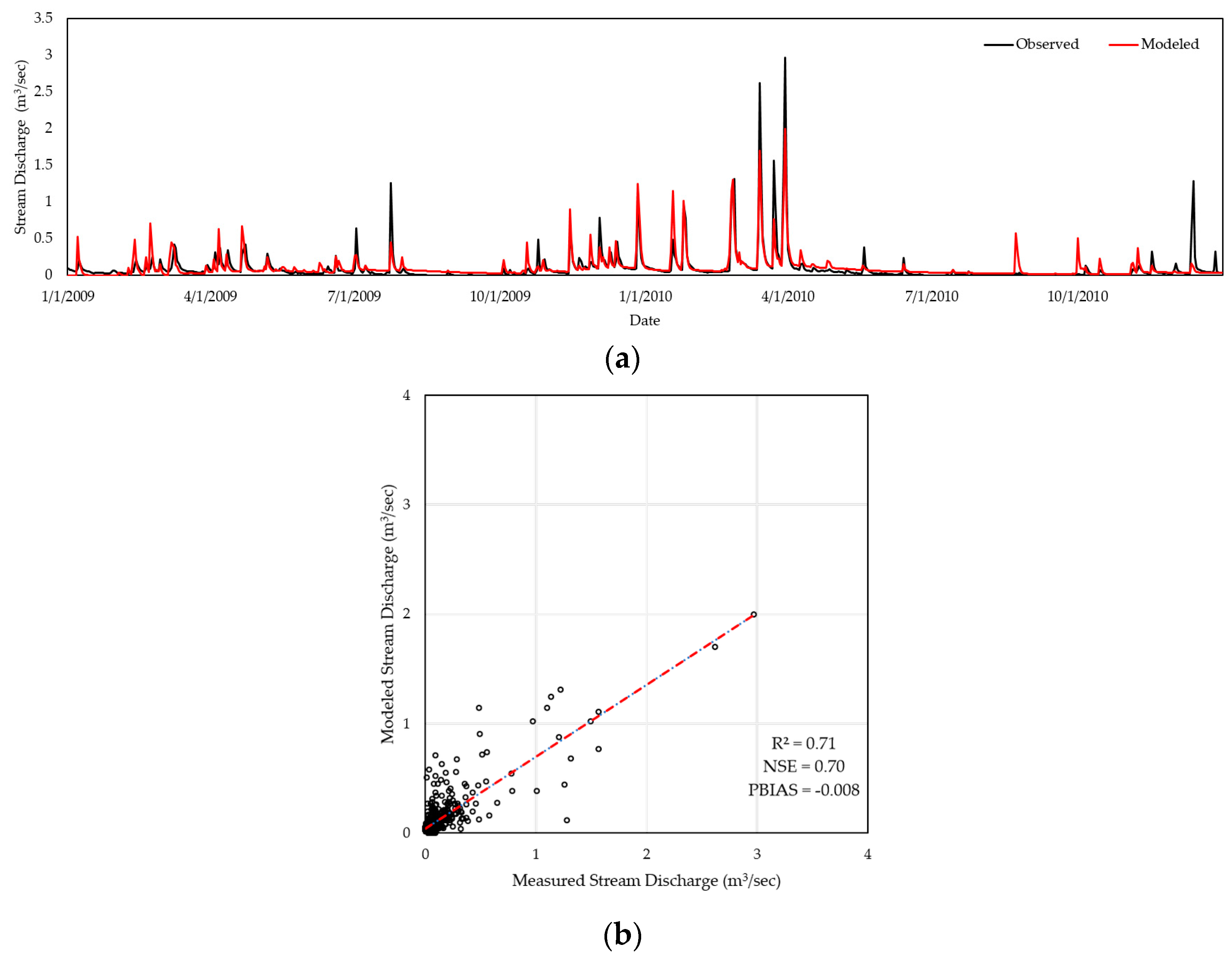
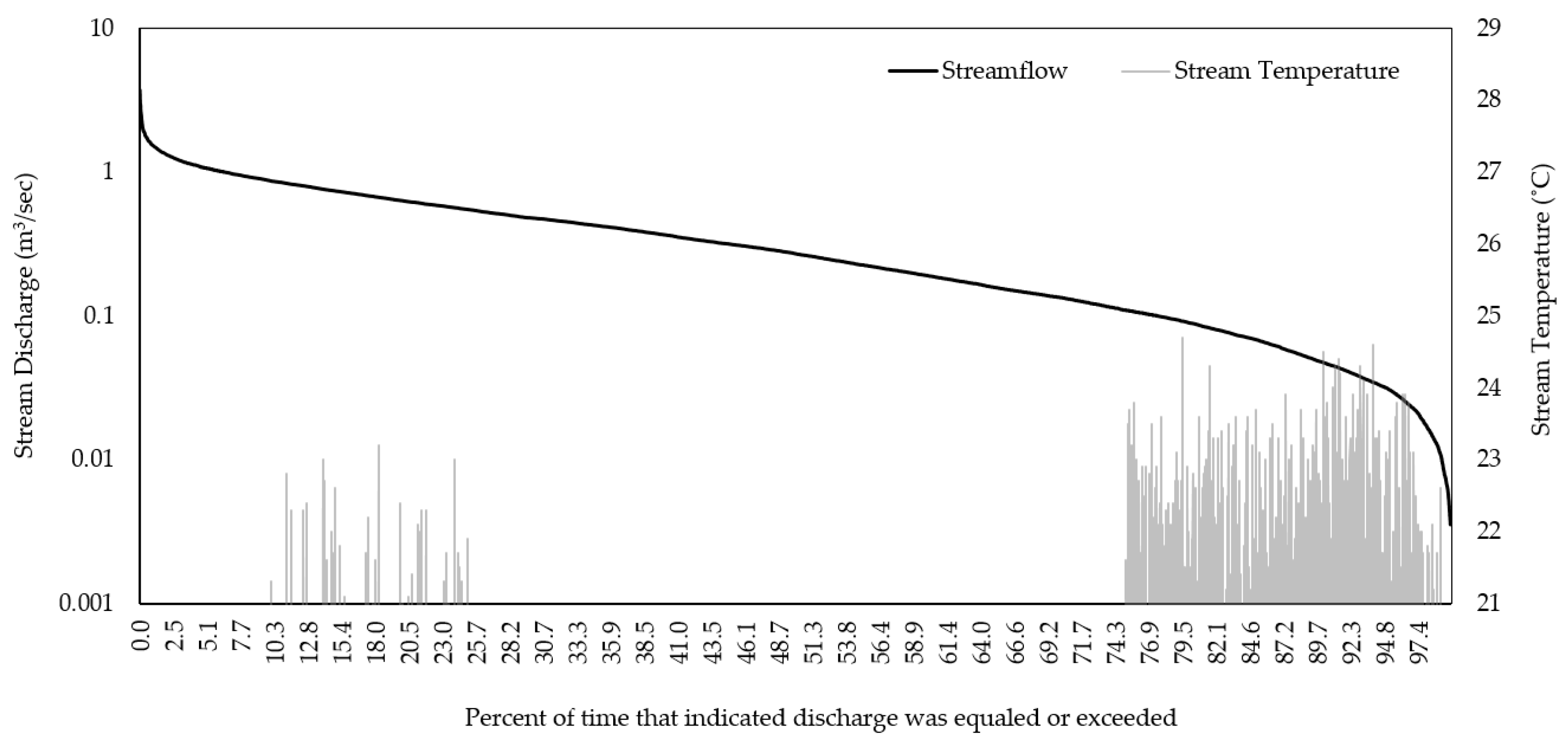
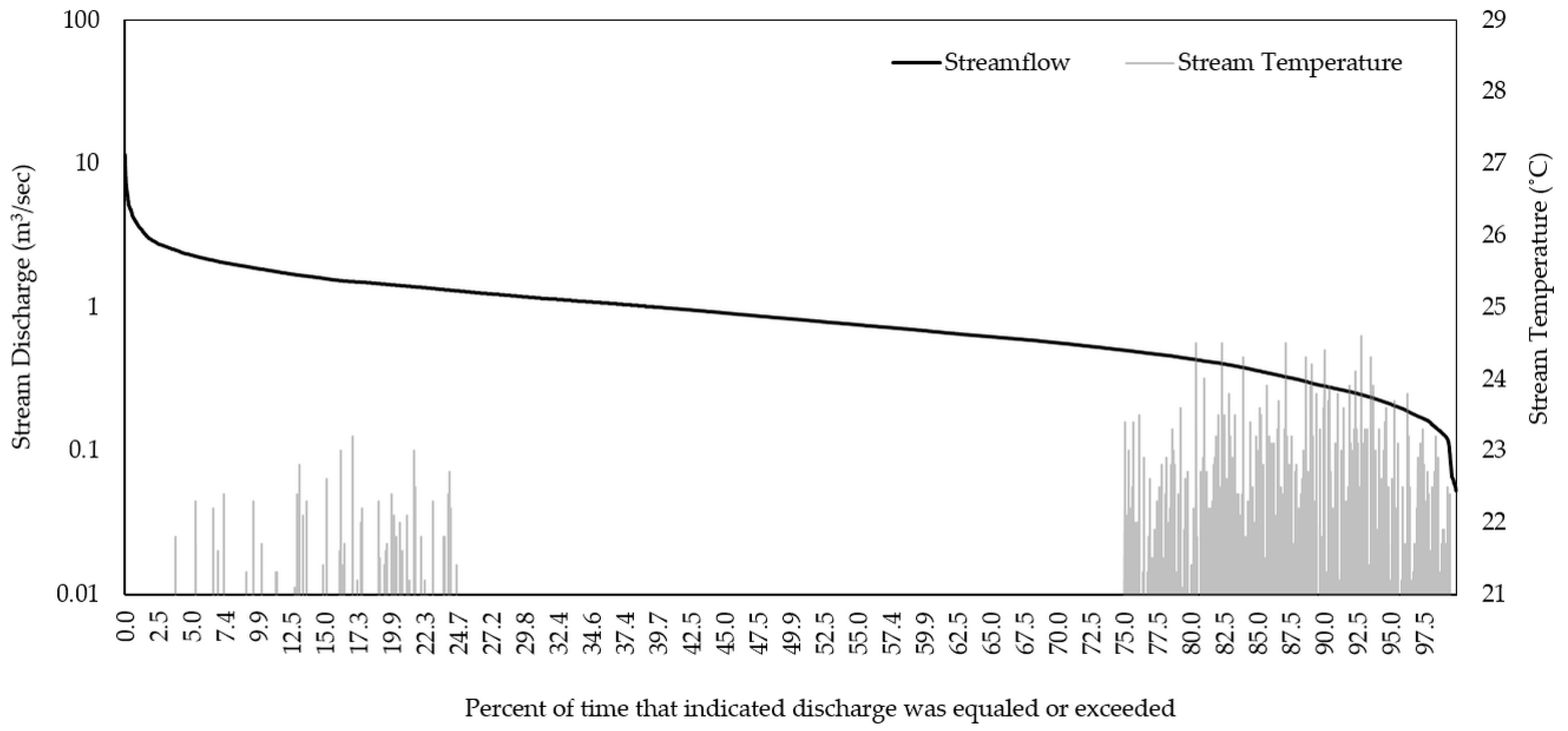
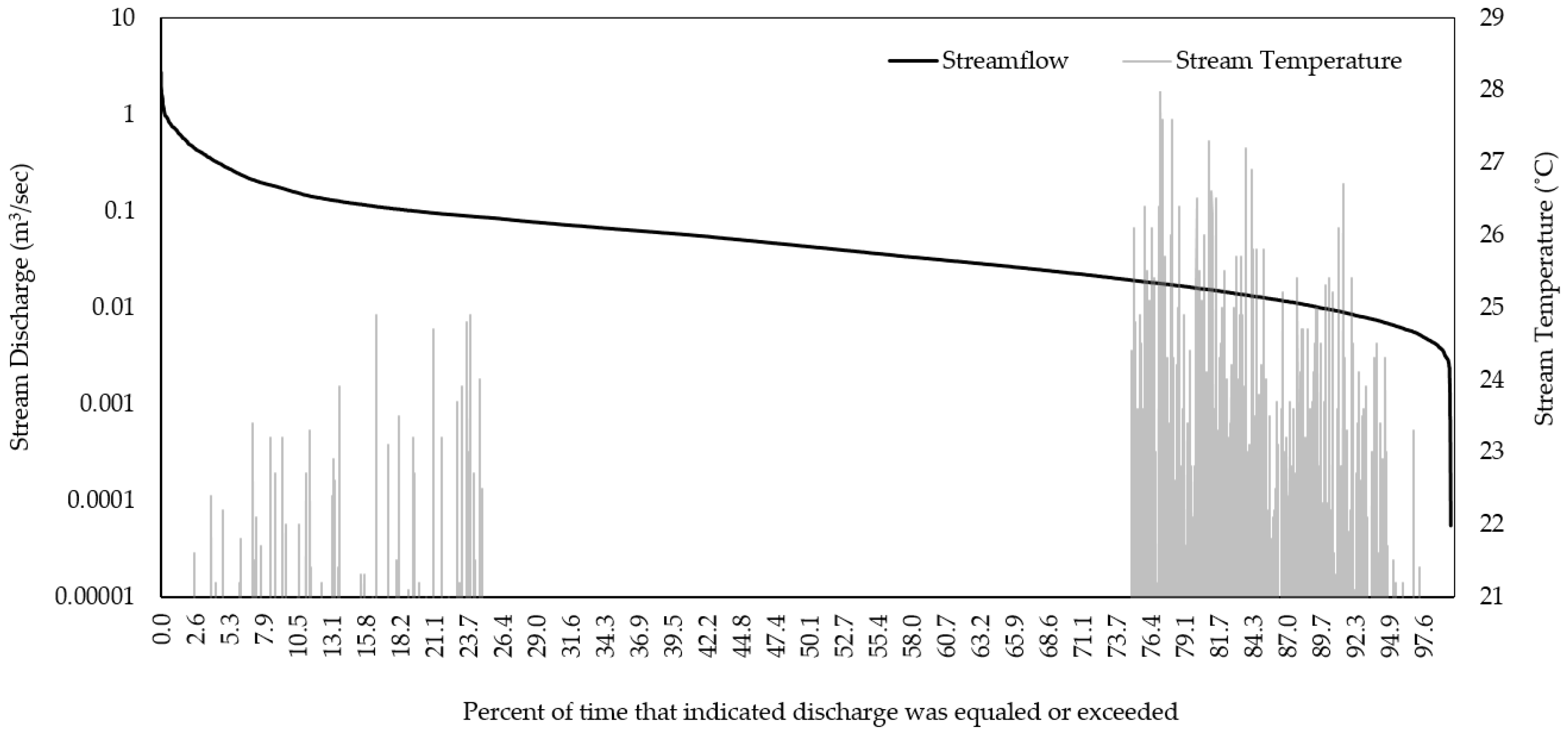

| Watershed | R2 | NSE | PBIAS |
|---|---|---|---|
| Beaver River | 0.64 | 0.57 | 0.13 |
| Queen River | 0.58 | 0.58 | 0.002 |
| Cork Brook | 0.70 | 0.71 | −0.01 |
| Streamflow | R2 | NSE | PBIAS |
|---|---|---|---|
| Beaver River | 0.66 | 0.60 | 0.13 |
| Queen River | 0.60 | 0.59 | 0.003 |
| Cork Brook | 0.54 | 0.50 | 0.03 |
| Parameter | Definition | Value Range | Units |
|---|---|---|---|
| (a) Beaver River parameters for daily streamflow calibration. | |||
| CN2.mgt | SCS runoff curve number | −60–75 | - |
| ALPHA_BF.gw | Baseflow alpha factor | 0.0–0.10 | 1/Days |
| GW_DELAY.gw | Groundwater delay | 0.0–10 | Days |
| TIMP.bsn | Snowpack temperature lag factor | −1.5–2.0 | - |
| ALPHA_BNK.rte | Baseflow alpha factor for bank storage | 0.50–1.0 | Days |
| OV_N.hru | Manning’s (n) value for overland flow | 1.0–30 | - |
| SLSUBBSN.hru | Average slope length | 10–50 | m |
| (b) Queen River parameters for daily streamflow calibration. | |||
| CN2.mgt | SCS runoff curve number | 60–75 | - |
| ALPHA_BF.gw | Baseflow alpha factor | 0.0–0.10 | 1/Days |
| GW_REVAP.gw | Groundwater revap coefficient | 0.02–0.15 | Days |
| GW_DELAY.gw | Groundwater delay | 0.0–10.0 | Days |
| GWQMN.gw | Depth of water in shallow aquifer for return flow | 150–1000 | mm |
| TIMP.bsn | Snowpack temperature lag factor | 0.0–1.0 | - |
| ALPHA_BNK | Baseflow alpha factor for bank storage | 0.5–1.0 | Days |
| (c) Cork Brook parameters for daily streamflow calibration. | |||
| CN2.mgt | SCS runoff curve number | −60–75 | - |
| ALPHA_BF.gw | Baseflow alpha factor | 0.0–0.10 | 1/Days |
| GW_DELAY.gw | Groundwater delay | 0.0–7.0 | Days |
| GWQMN.gw | Depth of water in shallow aquifer for return flow | 200–1000 | mm |
| SMTMP.bsn | Snowmelt base temperature | −0.5–2.0 | °C |
| ESCO.hru | Soil evaporation compensation factor | 0.15–0.65 | - |
| EPCO.hru | Plant uptake compensation factor | 0.15–0.65 | - |
| SLSOIL.hru | Slope length for lateral subsurface flow | 0.0–150.0 | m |
| Indicator | January | February | March | April | May | June | July | August | September | October | November | December |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (a) Low Emissions–Kingston, RI | ||||||||||||
| Short-term Temp. | 0.97 | 0.97 | 1.42 | 1.42 | 1.42 | 0.83 | 0.83 | 0.83 | 0.36 | 0.36 | 0.36 | 0.97 |
| Med-term Temp. | 1.50 | 1.50 | 2.47 | 2.47 | 2.47 | 1.58 | 1.58 | 1.58 | 0.56 | 0.56 | 0.56 | 1.50 |
| Long-term Temp. | 2.17 | 2.17 | 3.25 | 3.25 | 3.25 | 1.97 | 1.97 | 1.97 | 0.83 | 0.83 | 0.83 | 2.17 |
| Short-term Precip. | 8.76 | 8.76 | 9.80 | 9.80 | 9.80 | 17.9 | 17.9 | 17.9 | 5.59 | 5.59 | 5.59 | 8.76 |
| Med-term Precip. | 14.3 | 14.3 | 10.3 | 10.3 | 10.3 | 17.9 | 17.9 | 17.9 | 6.90 | 6.90 | 6.90 | 14.3 |
| Long-term Precip. | 14.9 | 14.9 | 16.3 | 16.3 | 16.3 | 18.6 | 18.6 | 18.6 | 10.6 | 10.6 | 10.6 | 14.9 |
| (b) High Emissions–Kingston, RI | ||||||||||||
| Short-term Temp. | 0.97 | 0.97 | 0.83 | 0.83 | 0.83 | 1.11 | 1.11 | 1.11 | 1.00 | 1.00 | 1.00 | 0.97 |
| Med-term Temp. | 2.22 | 2.22 | 2.36 | 2.36 | 2.36 | 3.06 | 3.06 | 3.06 | 3.00 | 3.00 | 3.00 | 2.22 |
| Long-term Temp. | 3.83 | 3.83 | 4.28 | 4.28 | 4.28 | 5.22 | 5.22 | 5.22 | 4.92 | 4.92 | 4.92 | 3.83 |
| Short-term Precip. | 8.09 | 8.09 | 14.2 | 14.2 | 14.2 | 12.5 | 12.5 | 12.5 | 4.93 | 4.93 | 4.93 | 8.09 |
| Med-term Precip. | 10.0 | 10.0 | 15.8 | 15.8 | 15.8 | 12.5 | 12.5 | 12.5 | 6.2 | 6.2 | 6.2 | 10.0 |
| Long-term Precip. | 22.3 | 22.3 | 22.0 | 22.0 | 22.0 | 10.2 | 10.2 | 10.2 | 8.16 | 8.16 | 8.16 | 22.3 |
| (c) Low Emissions–North Foster, RI | ||||||||||||
| Short-term Temp. | 1.00 | 1.00 | 1.42 | 1.42 | 1.42 | 0.97 | 0.97 | 0.97 | 0.39 | 0.39 | 0.39 | 1.00 |
| Med-term Temp. | 1.58 | 1.58 | 2.53 | 2.53 | 2.53 | 1.81 | 1.81 | 1.81 | 0.58 | 0.58 | 0.58 | 2.22 |
| Long-term Temp. | 2.22 | 2.22 | 3.33 | 3.33 | 3.33 | 2.25 | 2.25 | 2.25 | 0.81 | 0.81 | 0.81 | 2.22 |
| Short-term Precip. | 10.6 | 10.6 | 11.3 | 11.3 | 11.3 | 16.9 | 16.9 | 16.9 | 6.62 | 6.62 | 6.62 | 10.6 |
| Med-term Precip. | 12.9 | 12.9 | 11.9 | 11.9 | 11.9 | 17.4 | 17.4 | 17.4 | 10.1 | 10.1 | 10.1 | 12.9 |
| Long-term Precip. | 16.2 | 16.2 | 15.6 | 15.6 | 15.6 | 17.4 | 17.4 | 17.4 | 11.8 | 11.8 | 11.8 | 16.2 |
| (d) High Emissions–North Foster, RI | ||||||||||||
| Short-term Temp. | 0.97 | 0.97 | 0.89 | 0.89 | 0.89 | 1.22 | 1.22 | 1.22 | 0.89 | 0.89 | 0.89 | 0.97 |
| Med-term Temp. | 2.22 | 2.22 | 2.50 | 2.50 | 2.50 | 3.28 | 3.28 | 3.28 | 2.78 | 2.78 | 2.78 | 2.22 |
| Long-term Temp. | 3.86 | 3.86 | 4.47 | 4.47 | 4.47 | 5.50 | 5.50 | 5.50 | 4.64 | 4.64 | 4.64 | 3.86 |
| Short-term Precip. | 6.29 | 6.29 | 10.8 | 10.8 | 10.8 | 15.7 | 15.7 | 15.7 | 2.08 | 2.08 | 2.08 | 6.29 |
| Med-term Precip. | 8.84 | 8.84 | 11.3 | 11.3 | 11.3 | 18.0 | 18.0 | 18.0 | 2.76 | 2.76 | 2.76 | 8.84 |
| Long-term Precip. | 17.7 | 17.7 | 20.0 | 20.0 | 20.0 | 17.4 | 17.4 | 17.4 | 5.37 | 5.37 | 5.37 | 17.7 |
| Watershed | Average Daily Stream Temp. (°C) | Average Daily Discharge (m3/s) |
|---|---|---|
| Beaver River | 13.0 | 0.38 |
| Queen River | 13.0 | 1.0 |
| Cork Brook | 12.5 | 0.081 |
| Date | Watershed | Indicator | Any Type of Stress | Stream Temp. > 21 °C | Q25 or Q75 Flow | Stressful Event |
|---|---|---|---|---|---|---|
| 1980–2009 | Beaver River | Days | 6416 | 959 | 5457 | 511 |
| % Chance | 58.6% | 8.8% | 49.8% | 4.7% | ||
| Queen River | Days | 6506 | 959 | 5547 | 700 | |
| % Chance | 59.4% | 8.8% | 50.6% | 5.5% | ||
| Cork Brook | Days | 6875 | 1409 | 5466 | 551 | |
| % Chance | 62.7% | 12.9% | 49.9% | 4.4% |
| Scenario | Date | Average Daily Stream Temp. (°C) | Average Daily Discharge (m3/s) |
|---|---|---|---|
| (a) | |||
| Beaver River Historical | 1980–2009 | 13.0 | 0.38 |
| Beaver River Low Emissions | 2010–2039 | 13.6 | 0.44 |
| 2040–2069 | 14.2 | 0.45 | |
| 2070–2099 | 14.6 | 0.47 | |
| Beaver River High Emissions | 2010–2039 | 13.7 | 0.49 |
| 2040–2069 | 15.0 | 0.53 | |
| 2070–2099 | 16.4 | 0.65 | |
| (b) | |||
| Queen River Historical | 1980–2009 | 13.0 | 1.0 |
| Queen River Low Emissions | 2010–2039 | 13.6 | 1.14 |
| 2040–2069 | 14.2 | 1.16 | |
| 2070–2099 | 14.6 | 1.19 | |
| Queen River High Emissions | 2010–2039 | 13.7 | 1.20 |
| 2040–2069 | 15.0 | 1.27 | |
| 2070–2099 | 16.4 | 1.49 | |
| (c) | |||
| Cork Brook Historical | 1980–2009 | 12.5 | 0.081 |
| Cork Brook Low Emissions | 2010–2039 | 13.2 | 0.09 |
| 2040–2069 | 13.25 | 0.10 | |
| 2070–2099 | 14.11 | 0.10 | |
| Cork Brook High Emissions | 2010–2039 | 13.25 | 0.10 |
| 2040–2069 | 14.52 | 0.10 | |
| 2070–2099 | 15.97 | 0.13 | |
| Date | Emission Scenario | Unit | Beaver | Queen | Cork |
|---|---|---|---|---|---|
| 1980–2009 | Historical | % Chance | 4.7 | 5.5 | 4.4 |
| Historical | 4.7 | 5.5 | 4.4 | ||
| 2010–2039 | Low | % Chance | 6.2 | 6.9 | 6.5 |
| High | 7.2 | 7.9 | 7.2 | ||
| 2040–2069 | Low | % Chance | 7.9 | 8.5 | 7.1 |
| High | 12.4 | 13.1 | 11.3 | ||
| 2079–2099 | Low | % Chance | 9.0 | 9.8 | 8.6 |
| High | 16.1 | 16.8 | 15.2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chambers, B.M.; Pradhanang, S.M.; Gold, A.J. Simulating Climate Change Induced Thermal Stress in Coldwater Fish Habitat Using SWAT Model. Water 2017, 9, 732. https://doi.org/10.3390/w9100732
Chambers BM, Pradhanang SM, Gold AJ. Simulating Climate Change Induced Thermal Stress in Coldwater Fish Habitat Using SWAT Model. Water. 2017; 9(10):732. https://doi.org/10.3390/w9100732
Chicago/Turabian StyleChambers, Britta M., Soni M. Pradhanang, and Arthur J. Gold. 2017. "Simulating Climate Change Induced Thermal Stress in Coldwater Fish Habitat Using SWAT Model" Water 9, no. 10: 732. https://doi.org/10.3390/w9100732
APA StyleChambers, B. M., Pradhanang, S. M., & Gold, A. J. (2017). Simulating Climate Change Induced Thermal Stress in Coldwater Fish Habitat Using SWAT Model. Water, 9(10), 732. https://doi.org/10.3390/w9100732






