Heavy Metal Contamination in the Surface Layer of Bottom Sediments in a Flow-Through Lake: A Case Study of Lake Symsar in Northern Poland
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area and Sampling Sites
2.2. Sampling Regime and Analytical Procedure
- Igeo—geochemical index,
- Cm—concentration of the analysed metal (mg·kg−1),
- GB—geochemical background (mg·kg−1) [17].
- Cm—concentration of the analysed metal (mg·kg−1)
- GB—geochemical background (mg·kg−1) [17].
2.3. Statistical Analyses
3. Results
3.1. Sediment Texture and Relationships between Fractions and Concentrations of Metals
3.2. Heavy Metal Content
3.3. Contamination Classes of Bottom Sediments (Igeo, CF, PLI)
3.4. Multivariate Analyses
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Glińska-Lewczuk, K. Effect of land use and lake presence on chemical diversity of the Łyna river system. Pol. J. Environ. Stud. 2006, 15, 259–269. [Google Scholar]
- Iwuoha, P.O.; Adiela, P.U.; Nwannah, C.C.; Okeke, O.C. Sediment source and transport in river channels: Implications for river structures. Int. J. Eng. Sci. 2016, 5, 19–26. [Google Scholar]
- Obolewski, K.; Glińska-Lewczuk, K. Distribution of heavy metals in bottom sediments of floodplain lakes and their partent river—A case study of the Słupia. J. Elementol. 2013, 18, 673–682. [Google Scholar]
- Sparks, D.L. Toxic metals in the environment: the role of surfaces. Elements 2005, 1, 193–197. [Google Scholar] [CrossRef]
- Pintilie, S.; Brânză, L.; Bețianu, C.; Pavel, L.V.; Ungureanu, F.; Gavrilescu, M. Modelling and simulation of heavy metals transport in water and sediments. Environ. Eng. Manag. J. 2007, 6, 153–161. [Google Scholar]
- Zhang, C.; Yu, Z.; Zeng, G.; Jiang, M.; Yang, Z.; Cui, F.; Zhu, M.; Shen, L.; Hu, L. Effect of sediment geochemical properties on heavy metal bioavailibility. Environ. Int. 2014, 73, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Saeedi, M.; Li, L.Y.; Karbassi, A.R.; Zanjani, A.J. Sorbed metals fractionation and assessment of release in river sediment and particulate matter. Environ. Monit. Assess. 2013, 185, 1737–1754. [Google Scholar] [CrossRef] [PubMed]
- Connell, D.W.; Miller, G.J. Chemistry and Ecotoxicology of Pollution; John Wiley & Sons: New York, NY, USA, 1984. [Google Scholar]
- Selvam, G.G.; Baskaran, R.; Mohen, P.M. Microbial diveristy and bioremediation or distillenes effluent. J. Res. Biol. 2011, 3, 153–162. [Google Scholar]
- Branza, L.; Dring, M.; Gavrilescu, M. Biosorption of Cu2+ ions from aqueous solution by Enteromorpha sp. Environ. Eng. Manag. J. 2005, 4, 41–50. [Google Scholar]
- Gavrilescu, M. Removal of heavy metals from environment by biosorption. Eng. Life Sci. 2004, 4, 219–232. [Google Scholar] [CrossRef]
- Oyarzun, R.; Lillo, J.; Higueras, P.; Oyarzún, J.; Maturana, H. Strong arsenic enrichment in sediments from the Elqui watershed, northern Chile: industrial (gold mining at El Indio-Tambo district) versus geologic processes. J. Geochem. Explor. 2004, 84, 53–64. [Google Scholar] [CrossRef]
- Ruhman, M.S.; Saha, N.; Molla, A.H.; Al-Reza, S.M. Assessment of Anthropogenic Influence on Heavy Metal Contamination in the Aquatic Ecosystem Components: Water, Sediment and Fish. Soil Sediment Contam. 2014, 23, 353–373. [Google Scholar] [CrossRef]
- Glińska-Lewczuk, K. Komentarz do Mapy Hydrograficznej w Skali 1:50 000 Arkusz Lidzbark Warmiński (N-34-66-C); Główny Urząd Geodezji i Kartografii, GEPOL: Poznań, Poland, 2011. (In Polish) [Google Scholar]
- Potasznik, A.; Szymczyk, S. Magnesium and calcium concentrations in the surface water and bottom deposits of a river-lake system. J. Elementol. 2015, 20, 677–692. [Google Scholar] [CrossRef]
- Müller, G. Index of geo-accumulation in sediments of Rhine River. Gojound 1969, 2, 108–118. [Google Scholar]
- Bojakowska, I.; Sokołowska, G. Geochemiczne klasy czystości osadów wodnych. Prz. Geol. 1998, 46, 49–54. (In Polish) [Google Scholar]
- Bhuiyon, M.A.H.; Parvez, L.; Islam, M.A.; Dampare, S.B.; Suzuki, S. Heavy metal pollution of coal mine-affected agricultural soils in the northern part of Bangladesh. J. Hazard. Mater. 2010, 173, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Usero, I.; Garcia, A.; Fraidias, J. Calidad de Lasaquas y Sedimentos del Litoral Andaluz, inc Junta de Andalicia; Consejeria del Medio Ambiente: Sevilla, Spain, 2000; p. 164. [Google Scholar]
- Likuku, A.S.; Mmolawa, K.B.; Gaboutloeloe, G.K. Assessment of heavy metal enrichment and degree of contamination around the copper-nickel mine in the Selebi Phikwe region, Eastern Botswana. Environ. Ecol. Res. 2013, 1, 32–40. [Google Scholar]
- Ter Braak, C.J.F.; Smilauer, P. CANOCO Reference Manual and CanoDraw for Windows User's Guide: Software for Canonical Community Ordination (version 4.5); Microcomputer Power: Ithaca, NY, USA, 2002; p. 500. [Google Scholar]
- Thevenon, F.; Graham, N.D.; Chiaradia, M.; Arpagaus, P.; Wildi, W.; Poté, J. Local to regional scale industrial heavy metal pollution recorded in sediments of large freshwater lakes in central Europe (lakes Geneva and Lucerne) over the last centuries. Sci. Total Environ. 2011, 412, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Wunnemann, B. Late Quaternary Water depth changes in Hele Lake, northeastern Tibetan Plateau, delivered from ostracord assemblages and sediment properties in multiple sediment record. Quat. Sci. Rev. 2014, 95, 95–114. [Google Scholar] [CrossRef]
- Kondracki, J. Geografia Regionalna Polski; Polish Scientific Publishers (PWN): Warszawa, Poland, 2002; p. 445. [Google Scholar]
- Czajkowska, A. Stopień zanieczyszczenia związkami biogennymi płytkich wód podziemnych w zagospodarowanej rolniczo części zlewni Bierawki. Assessment of biogenic compounds pollution of shallow underground water in agricultural managed part of Bierawka river basin. Górnictwo Geol. 2010, 5, 91–103. [Google Scholar]
- Szalińska, E.; Graciak-Mannion, A.; Haffner, D.; Droullard, K.G. Assessment of decadal changes in sediment contamination in a large connecting channel (Detroit River, North America). Chemosphere 2013, 93, 1773–1781. [Google Scholar] [CrossRef] [PubMed]
- Skwierawski, A.; Sidoruk, M. Heavy metal concentrations in the sediment profiles of the antropogenically transformed Płociduga reservoir. Ecol. Chem. Eng. S 2014, 21, 79–88. [Google Scholar]
- Macintosh, K.A.; Griffiths, D. Catchment and in-stream influences on metal concentration and ochre deposit density in upland streams. Environ. Earth Sci. 2013, 70, 3023–3030. [Google Scholar] [CrossRef]
- Song, Y.; Ji, J.; Yang, Z.; Mao, C.; Frost, R.L.; Ayoko, G.A. Geochemical behavior assessment and apportionment of heavy metal contaminations in the bottom sediments of lower reach of Changjiang River. Catena 2011, 85, 73–81. [Google Scholar] [CrossRef]
- Leyden, E.; Cook, F.; Hamilton, B.; Zammit, B.; Barnett, Z.; Lush, A.M.; Stone, D.; Mosley, L. Near shore groundwater acidification during and after a hydrological drought in the Lower Lakes, South Australia. J. Contam. Hydrol. 2016, 189, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Mohiuddin, K.M.; Zakir, H.M.; Otomo, K.; Sharmin, S.; Shikazono, N. Geochemical distribution of trace metal pollutants in water and sediments of downstream of an urban river. Int. J. Environ. Sci. Technol. 2009, 7, 1728–2010. [Google Scholar] [CrossRef]
- Sondergaard, M.; Jeppesen, E.; Lauridsen, T.L.; Skov, C.; Van Nes, E.H.; Roijackers, R.; Lammens, E.; Portielje, R. Lake restoration: Successes, failures and long-term effects. J. Appl. Ecol. 2007, 44, 1095–1105. [Google Scholar] [CrossRef]
- Madison, A.S.; Tebo, B.M.; Mucci, A.; Sundby, B.; Luther, G.W. Abundant pore water Mn (III) is a major component of the sedimentary redox system. Science 2013, 341, 875–878. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Cheng, J. Speciation of Heavy Metal in the Sediment from different Eutrophic Lakes of China. Procedia Eng. 2011, 18, 318–323. [Google Scholar]
- Canfield, D.E.; Kristensen, E.; Thamdrup, B. The Iron and Manganese Cycles. Adv. Mar. Biol. 2005, 48, 269–312. [Google Scholar]
- Abualhaija, M.M.; Whitby, H.; van den Berg, C.M.G. Competition between cooper or iron for humic ligands in estuarine waters. Mar. Chem. 2015, 172, 46–56. [Google Scholar] [CrossRef]
- Marin, E.; Vaccaro, C.; Di Giuse, D.; Procesi, M.; Sciarra, A.; Zarlenga, F. Enrichment in Heavy metal (HM) and Rare Earth Element (REE) in fluvial placer deposits: Case study of Zambesi River (Mozambique). Int. J. Sci. Res. 2016, 5, NOV163251. [Google Scholar]
- Kabata-Pendias, A.; Szteka, A. Trace Elements in Abiotic and Biotic Environments; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Ikem, A.; Adisa, S. Runoff effect on eutrophic lake water quality and heavy metal distribution in recent littoral sediment. Chemosphere 2011, 82, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Zerbe, J.; Sobczyński, T.; Elbanowska, H.; Siepak, J. Speciation of heavy metals in bottom sediments of lakes. Pol. J. Environ. Stud. 1999, 8, 331–339. [Google Scholar]
- Wojtkowska, M.; Karwowska, E.; Chmielewska, I.; Bekenova, K.; Wanot, E. Copper and cadmium in bottom sediments dredged from Wyścigi Pond, Warsaw, Poland—Contamination and bioaccumulation study. Environ. Monit. Assess. 2015, 187, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Skorbiłowicz, E.; Skorbiłowicz, M. Metals in grain fractions of bottom sediments from selected rivers in north-eastern Poland. Phys. Chem. Earth A/B/C/ 2011, 36, 567–578. [Google Scholar] [CrossRef]
- Hakanson, L. Ecological risk index for aquatic pollutant control, a sedimentalogical approch. Water Res. 1980, 14, 975–1001. [Google Scholar] [CrossRef]
- Yuan, X.; Zhang, L.; Wang, C.; Ji, J. Sediment properties and heavy metal pollution assessment in the river, estuary and lake environments of a fluvial plain, China. Catena 2014, 119, 52–60. [Google Scholar] [CrossRef]
- Bojakowska, I.; Gliwicz, T.; Szatkowska-Konon, H. Osady Denne w Wodach Powierzchniowych. Biblioteka Monitoringu Środowiska. Available online: http://orka.sejm.gov.pl/druki4ka.nsf/($vAllByUnid)/A0C171F9B88533F1C1256DC60044F9C9/$file/2115_09.pdf (accessed on 16 August 2016).

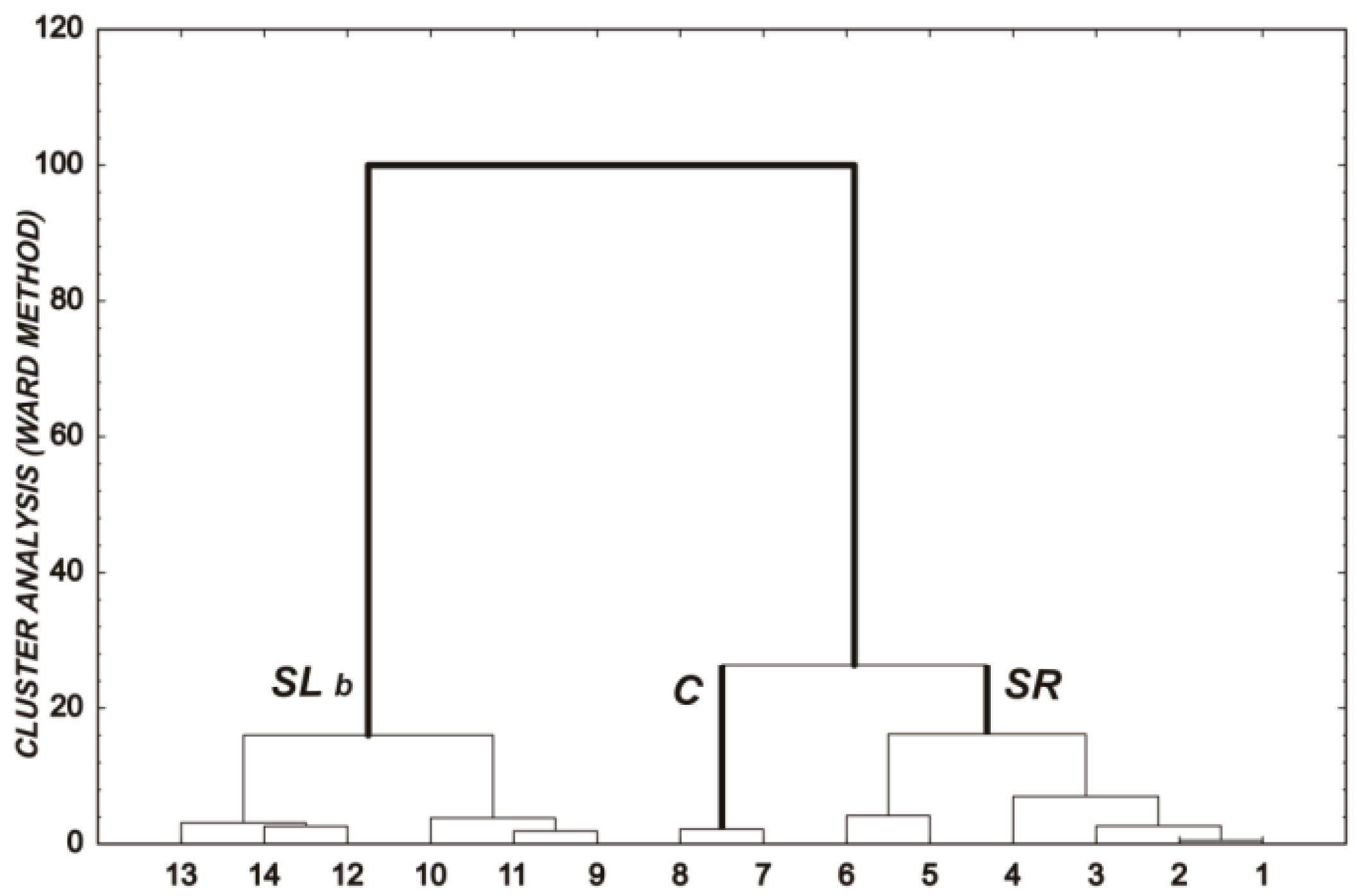


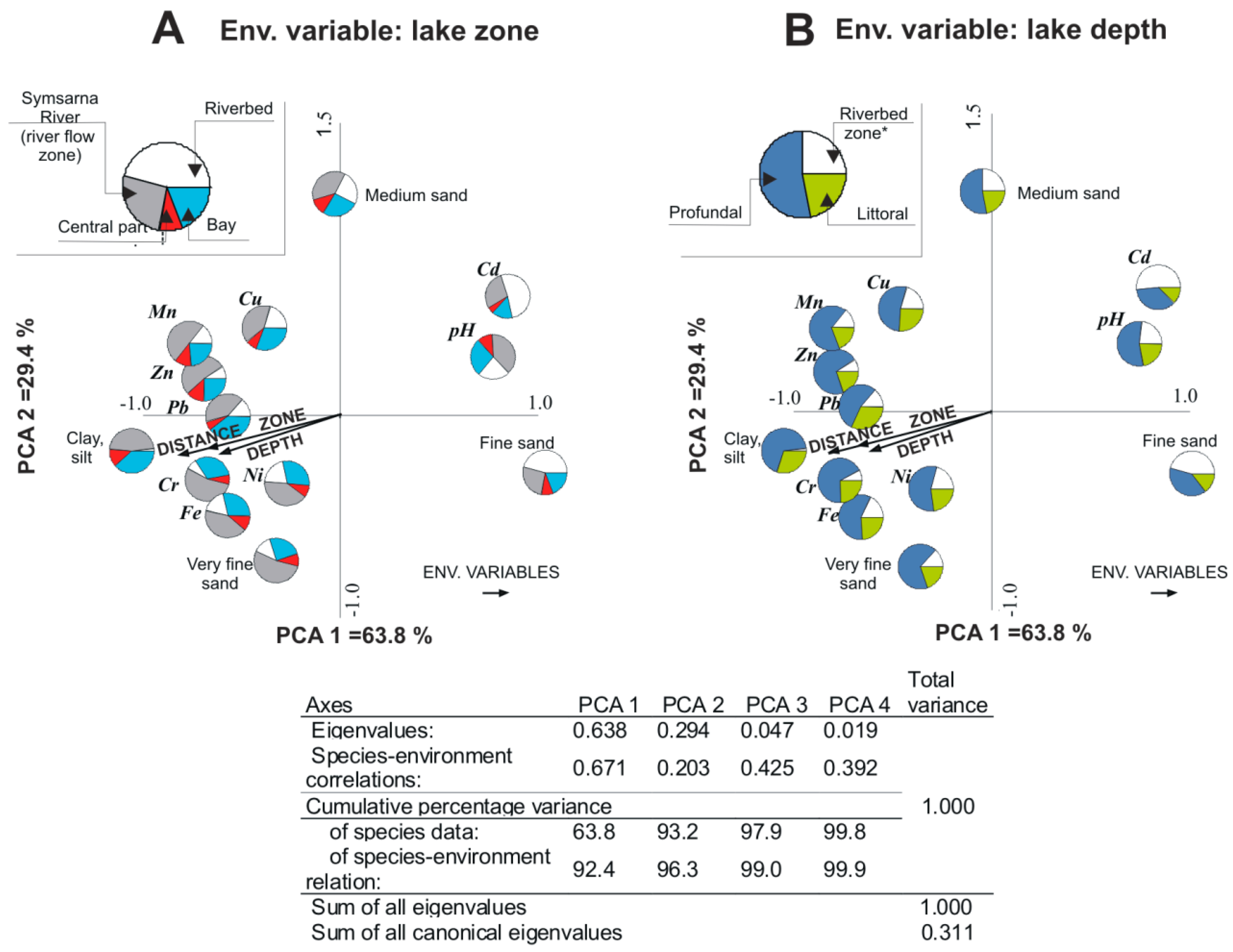
| Metals | Extraction in HNO3 + HClO4 [mg·kg−1] | Sewage Sludge Certified Reference Material [mg·kg−1] | Recovery [%] | Acceptable Deviation [%] |
|---|---|---|---|---|
| Mn | 677.5 | 693 | 97.8 | 15.6 |
| Cr | 310.7 | 289 | 107.3 | 10.5 |
| Cu | 503.3 | 482 | 104.4 | 10.4 |
| Ni | 174.9 | 163 | 107.3 | 8.3 |
| Cd | 63.1 | 60.6 | 104.0 | 4.9 |
| Pb | 152.5 | 154 | 99.0 | 8.1 |
| Zn | 1076.2 | 1240 | 86.8 | 14.6 |
| Fraction (%) | Lake’s Bottom Sediment (L) | River (R) | p | ||||
|---|---|---|---|---|---|---|---|
| SR (±SD) | C (±SD) | SLb (±SD) | AS (±SD) | SR-i | SR-o | ||
| Medium sand | 36.96 ± 7.63 | 41.51 ± 4.10 | 36.87 ± 7.19 | 43.92 ± 25.32 | 35.23 | 12.04 | 0.812 |
| Fine sand | 15.54 ± 6.10 | 17.98 ± 0.91 | 15.64 ± 0.99 | 47.93 ± 21.29 | 44.37 | 78.92 | 0.007 * |
| Very fine sand | 13.46 ± 7.45 | 8.02 ± 1.21 | 8.78 ± 3.27 | 5.84 ± 6.05 | 13.77 | 7.36 | 0.246 |
| Clay, silt | 34.04 ± 4.38 | 32.49 ± 3.79 | 38.71 ± 3.94 | 2.31 ± 2.94 | 6.63 | 1.68 | 0.008 * |
| Texture | Mn | Fe | Zn | Cu | Ni | Cd | Cr | Pb |
|---|---|---|---|---|---|---|---|---|
| Medium sand | 0.236 | −0.309 | −0.063 | 0.040 | −0.356 | 0.408 | −0.125 | 0.115 |
| Fine sand | −0.717 | −0.232 | −0.763 | −0.174 | −0.020 | 0.209 | −0.759 | −0.385 |
| Very fine sand | 0.172 | 0.317 | 0.253 | 0.154 | 0.488 | −0.496 | 0.251 | −0.079 |
| Clay, silt | 0.226 | 0.401 | 0.455 | 0.228 | 0.317 | −0.633 | 0.623 | 0.453 |
| Heavy Metal | Lake’s Bottom Sediment (L) | River (R) | p | ||||
|---|---|---|---|---|---|---|---|
| SR (±SD) | C (±SD) | SLb (±D) | AS (±SD) | SR-i | SR-o | ||
| Mn [g·kg−1] | 0.85 ± 0.1 | 0.74 ± 0.0 | 0.57 ± 0.0 | 0.44 ± 0.2 | 0.52 | 0.18 | 0.006 * |
| Fe [g·kg−1] | 32.83 ± 3.7 | 29.09 ± 3.9 | 31.65 ± 3.1 | 24.61 ± 6.6 | 20.35 | 29.94 | 0.285 |
| Zn [g·kg−1] | 0.13 ± 0.0 | 0.11 ± 0.0 | 0.13 ± 0.0 | 0.04 ± 0.0 | 0.05 | 0.02 | 0.122 |
| Cu [mg·kg−1] | 20.63 ± 2.9 | 14.56 ± 4.1 | 22.36 ± 3.2 | 18.97 ± 14.2 | 23.40 | 58.00 | 0.354 |
| Ni [mg·kg−1] | 33.88 ± 6.0 | 26.03 ± 12.6 | 34.75 ± 5.3 | 30.04 ± 8.1 | 39.80 | 25.50 | 0.618 |
| Cd [mg·kg−1] | 0.69 ± 0.3 | 0.36 ± 0.0 | 0.49 ± 0.6 | 2.14 ± 1.0 | 1.10 | 2.10 | 0.029 * |
| Cr [mg·kg−1] | 19.76 ± 4.6 | 8.68 ± 1.9 | 15.97 ± 4.5 | 5.06 ± 1.7 | 3.20 | 6.00 | 0.005 * |
| Pb [mg·kg−1] | 87.32 ± 41.8 | 45.65 ± 21.9 | 118.96 ± 12.6 | 52.47 ± 28.3 | 51.4 | 36.20 | 0.070 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuriata-Potasznik, A.; Szymczyk, S.; Skwierawski, A.; Glińska-Lewczuk, K.; Cymes, I. Heavy Metal Contamination in the Surface Layer of Bottom Sediments in a Flow-Through Lake: A Case Study of Lake Symsar in Northern Poland. Water 2016, 8, 358. https://doi.org/10.3390/w8080358
Kuriata-Potasznik A, Szymczyk S, Skwierawski A, Glińska-Lewczuk K, Cymes I. Heavy Metal Contamination in the Surface Layer of Bottom Sediments in a Flow-Through Lake: A Case Study of Lake Symsar in Northern Poland. Water. 2016; 8(8):358. https://doi.org/10.3390/w8080358
Chicago/Turabian StyleKuriata-Potasznik, Angela, Sławomir Szymczyk, Andrzej Skwierawski, Katarzyna Glińska-Lewczuk, and Ireneusz Cymes. 2016. "Heavy Metal Contamination in the Surface Layer of Bottom Sediments in a Flow-Through Lake: A Case Study of Lake Symsar in Northern Poland" Water 8, no. 8: 358. https://doi.org/10.3390/w8080358
APA StyleKuriata-Potasznik, A., Szymczyk, S., Skwierawski, A., Glińska-Lewczuk, K., & Cymes, I. (2016). Heavy Metal Contamination in the Surface Layer of Bottom Sediments in a Flow-Through Lake: A Case Study of Lake Symsar in Northern Poland. Water, 8(8), 358. https://doi.org/10.3390/w8080358







