Does Habitat Restoration Increase Coexistence of Native Stream Fishes with Introduced Brown Trout: A Case Study on the Middle Provo River, Utah, USA
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Survey Methods
2.3. Analysis
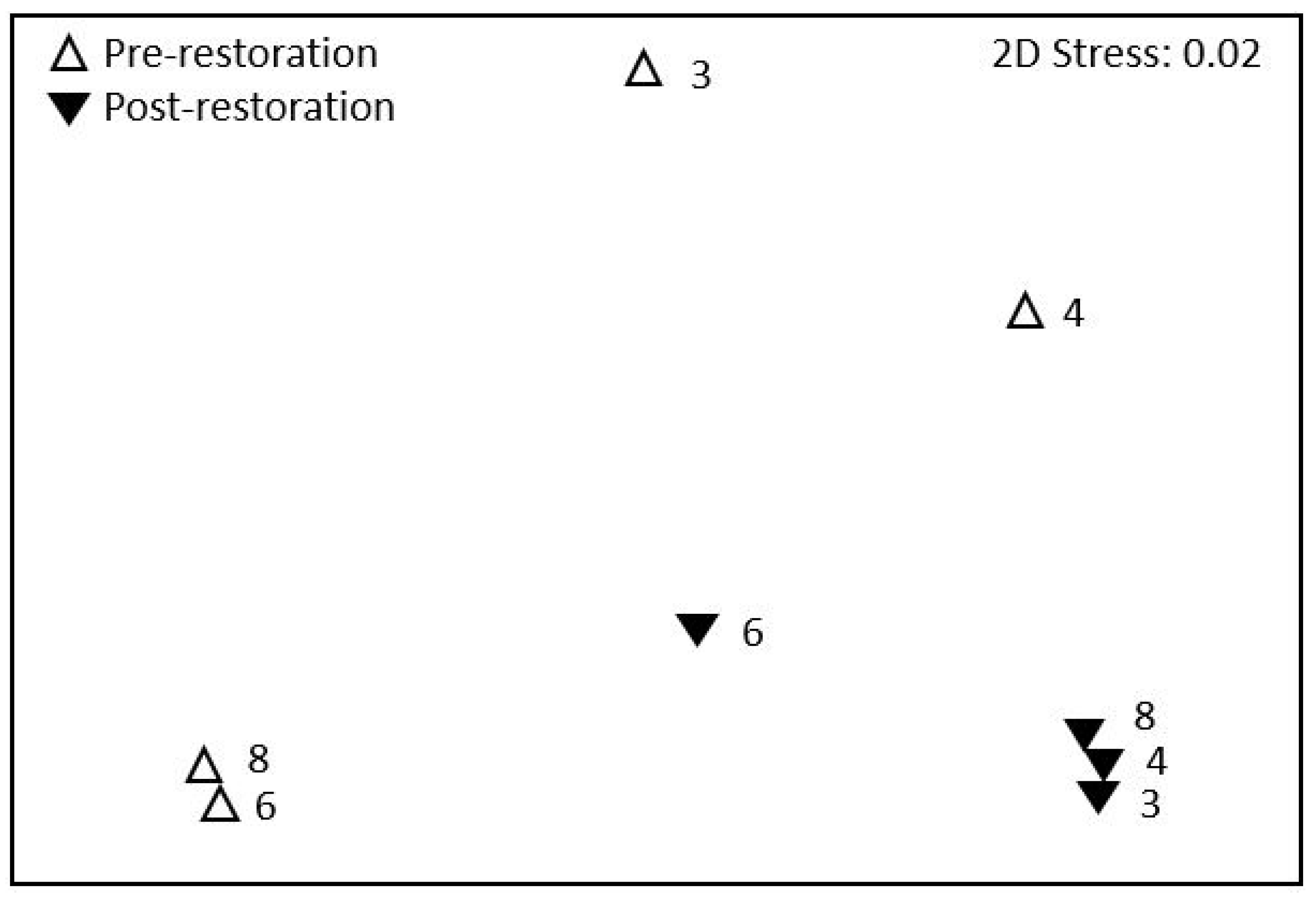
3. Results
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ANOSIM | analysis of similarity |
| NMDS | non-metric multidimensional scaling |
References
- Bernhardt, E.S.; Palmer, M.A.; Allan, J.D.; Alexander, G.; Barnas, K.; Brooks, S.; Carr, J.; Clayton, S.; Dahm, C.; Follstad-Shah, J.; et al. Synthesizing U.S. river restoration efforts. Science 2005, 308, 636–637. [Google Scholar] [CrossRef] [PubMed]
- Shields, F.D.; Cooper, C.M.; Knight, S.S.; Moore, M.T. Stream corridor restoration research: A long and winding road. Ecol. Eng. 2003, 20, 441–454. [Google Scholar] [CrossRef]
- Sondergard, M.; Jeppesen, E. Anthropogenic impacts on lake and stream ecosystems, and approaches to restoration. J. Appl. Ecol. 2007, 44, 1089–1094. [Google Scholar] [CrossRef]
- Rubin, J.F.; Glimsäter, C.; Jarvi, T. Characteristics and rehabilitation of the spawning habitats of the sea trout, Salmo trutta, in Gotland (Sweden). Fish. Manag. Ecol. 2004, 11, 15–22. [Google Scholar] [CrossRef]
- Meyer, E.I.; Niepagenkemper, O.; Molls, F.; Spänhoff, B. An experimental assessment of the effectiveness of gravel cleaning operations in improving hyporheic water quality in potential salmonid spawning areas. River Res. Appl. 2008, 24, 119–131. [Google Scholar] [CrossRef]
- Merz, J.E.; Chan, L.K. Effects of gravel augmentation on macroinvertebrate assemblages in a regulated California river. River Res. Appl. 2005, 21, 61–74. [Google Scholar] [CrossRef]
- Kail, J.; Brabec, K.; Poppe, M.; Januschke, K. The effect of river restoration on fish, macroinvertebrates and aquatic macrophytes: A meta-analysis. Ecol. Indic. 2015, 58, 311–321. [Google Scholar] [CrossRef]
- Lodge, D.M. Biological invasions: Lessons from ecology. Trends Ecol. Evol. 1993, 8, 133–137. [Google Scholar] [CrossRef]
- Townsend, C.R. Invasion biology and ecological impacts of brown trout Salmo trutta in New Zealand. Biol. Conserv. 1996, 78, 13–22. [Google Scholar] [CrossRef]
- Mills, M.D.; Rader, R.B.; Belk, M.C. Complex interactions between native and invasive fish: The simultaneous effects of multiple negative interactions. Oecologia 2004, 141, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Olsen, D.G.; Belk, M.C. Relationship of diurnal habitat use of native stream fishes of the eastern Great Basin to presence of introduced salmonids. West N. Am. Nat. 2005, 65, 501–506. [Google Scholar]
- McHugh, P.; Budy, P. Experimental effects of nonnative brown trout on the individual- and population-level performance of native Bonneville cutthroat trout. Trans. Am. Fish. Soc. 2006, 135, 1441–1455. [Google Scholar] [CrossRef]
- Billman, E.J.; Kreitzer, J.D.; Creighton, J.C.; Habit, E.; McMillan, B.; Belk, M.C. Habitat enhancement and native fish conservation: Can enhancement of channel complexity promote the coexistence of native and introduced fishes? Environ. Biol. Fish. 2013, 96, 555–565. [Google Scholar] [CrossRef]
- Utah Reclamation Mitigation and Conservation Commission; United States. Bureau of Land Management. Final Environmental Impact Statement on the Provo River Restoration Project; Utah Reclamation Mitigation and Conservation Commission: Salt Lake City, UT, USA, 1997. [Google Scholar]
- Heckmann, R.A.; Thompson, C.W.; White, D.A. Fishes of Utah Lake. Great Basin Nat. Mem. 1981, 5, 107–127. [Google Scholar]
- Shiozawa, D.K.; Rader, R.B. Great Basin Rivers. In Rivers of North America; Benke, A.C., Cushing, C.E., Eds.; Elsevier Academic Press: San Diego, CA, USA, 2005; pp. 655–694. [Google Scholar]
- Ellsworth, C.M. Response of the fish community and age class structure to channelization and habitat restoration in the Provo River between Deer Creek and Jordanelle Reservoirs. Master’s Thesis, Department of Integrative Biology, Brigham Young University, Provo, UT, USA, 23 March 2003. [Google Scholar]
- Sigler, W.F.; Miller, R.R. Fishes of Utah; Utah State Department of Fish and Game: Salt Lake City, UT, USA, 1963. [Google Scholar]
- Hepworth, R.; Utah Division of Wildlife Resources, Cedar City, Utah, USA. Personal communication, 2015.
- Bray, J.R.; Curtis, J.T. An ordination of the upland forest communities of southern Wisconsin. Ecol. Monogr. 1957, 27, 326–349. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. Primer v6: User Manual/Tutorial; Primer-E: Plymouth, UK, 2006; p. 192. [Google Scholar]
- Clarke, K.R.; Gorley, R.N. Primer v6: User Manual/Tutorial; Primer-E: Plymouth, UK, 2006. [Google Scholar]
- Hanisch, J.R.; Tonn, W.M.; Paszkowski, C.A.; Scrimgeour, G.J. Complex littoral habitat influences the response of native minnows to stocked trout: Evidence from whole-lake comparisons and experimental predator enclosures. Can. J. Fish. Aquat. Sci. 2012, 69, 273–281. [Google Scholar] [CrossRef]
- Power, M.E. Depth distributions of armored catfish: Predator-induced resource avoidance? Ecology 1984, 65, 523–528. [Google Scholar] [CrossRef]
- Rosenberger, A.; Angermeier, P.L. Ontogenetic shifts in habitat use by the endangered Roanoke logperch (Percina Rex). Freshw. Biol. 2003, 48, 1563–1577. [Google Scholar] [CrossRef]
- McHugh, P.; Budy, P. An experimental evaluation of competitive and thermal effects on brown trout (Salmo trutta) and Bonneville cutthroat trout (Oncorhynchus clarkii utah) performance along an altitudinal gradient. Can. J. Fish. Aquat. Sci. 2005, 62, 2784–2795. [Google Scholar] [CrossRef]
- Rasmussen, J.E.; Belk, M.C.; Habit, E.; Shiozawa, D.K.; Hepworth, R.D.; Anthony, A. Variation in size-at-age between native cutthroat and introduced brown trout in allopatry and sympatry: Implications for competitive interaction. Aquat. Biol. 2011, 13, 285–292. [Google Scholar] [CrossRef]
- Budy, P.; Thiede, G.P.; McHugh, P. Quantification of the vital rates, abundance, and status of a critical, endemic population of bonneville cutthroat trout. N. Am. J. Fish. Manag. 2007, 27, 593–604. [Google Scholar] [CrossRef]
- Nannini, M.A.; Belk, M.C. Antipredator responses of two native stream fishes to an introduced predator: Does similarity in morphology predict similarity in behavioural response? Ecol. Freshw. Fish 2006, 15, 453–463. [Google Scholar] [CrossRef]
- Belk, M.C.; Johnson, J.B. Biological status of leatherside chub: A framework for conservation of western freshwater fishes. Am. Fish. Soc. Symp. 2007, 53, 67–76. [Google Scholar]
- Billman, E.J.; Tjarks, B.J.; Belk, M.C. Effect of predation and habitat quality on growth and reproduction of a stream fish. Ecol. Freshw. Fish 2011, 20, 102–113. [Google Scholar] [CrossRef]
- Wilson, K.W.; Belk, M.C. Habitat Characteristics of leatherside chub (Gila copei) at two spatial scales. Western N. Am. Nat. 2001, 61, 36–42. [Google Scholar]
- Nielson, J.; Wiley, D. Middle Provo River Population Estimates-2010; Utah Division of Wildlife Resources: Springville, UT, USA, 2011. [Google Scholar]

| Reach, Year | Presence/Absence | Pre-Post Difference | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Species | 8, 1998 | 8, 2007 | 6, 1998 | 6, 2009 | 4, 1998 | 4, 2007 | 3, 1998 | 3, 2007 | ||
| Brown trout-Juvenile | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | |
| Brown trout-Adult | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | |
| Rainbow Rrout-Juvenile | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | -3 | |
| Rainbow Trout-Adult | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 1 | -1 | |
| Redside Shiner-Juvenile | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | |
| Redside Shiner-Adult | 0 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | |
| Mottled Sculpin-Juvenile | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | |
| Mottled Sculpin-Adult | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | |
| Mountain Whitefish-Juvenile | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 3 | |
| Mountain Whitefish-Adult | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | |
| Utah Sucker-Juvenile | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 1 | 3 | |
| Utah Sucker-Adult | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | |
| Mountain Sucker-Juvenile | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 | 3 | |
| Mountain Sucker-Adult | 0 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 2 | |
| Speckled Dace-Juvenile | 0 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 2 | |
| Speckled Dace-Adult | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 4 | |
| Longnose Dace-Juvenile | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 2 | |
| Longnose Dace-Adult | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 2 | |
| S. Leatherside Chub-Juvenile | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | |
| S. Leatherside Chub-Adult | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belk, M.C.; Billman, E.J.; Ellsworth, C.; McMillan, B.R. Does Habitat Restoration Increase Coexistence of Native Stream Fishes with Introduced Brown Trout: A Case Study on the Middle Provo River, Utah, USA. Water 2016, 8, 121. https://doi.org/10.3390/w8040121
Belk MC, Billman EJ, Ellsworth C, McMillan BR. Does Habitat Restoration Increase Coexistence of Native Stream Fishes with Introduced Brown Trout: A Case Study on the Middle Provo River, Utah, USA. Water. 2016; 8(4):121. https://doi.org/10.3390/w8040121
Chicago/Turabian StyleBelk, Mark C., Eric J. Billman, Craig Ellsworth, and Brock R. McMillan. 2016. "Does Habitat Restoration Increase Coexistence of Native Stream Fishes with Introduced Brown Trout: A Case Study on the Middle Provo River, Utah, USA" Water 8, no. 4: 121. https://doi.org/10.3390/w8040121
APA StyleBelk, M. C., Billman, E. J., Ellsworth, C., & McMillan, B. R. (2016). Does Habitat Restoration Increase Coexistence of Native Stream Fishes with Introduced Brown Trout: A Case Study on the Middle Provo River, Utah, USA. Water, 8(4), 121. https://doi.org/10.3390/w8040121






