Bacteria Removal from Stormwater Runoff Using Tree Filters: A Comparison of a Conventional and an Innovative System
Abstract
:1. Introduction
2. Methods
2.1. Site Description
2.2. ITF Preparation
2.3. Sampling and Analysis
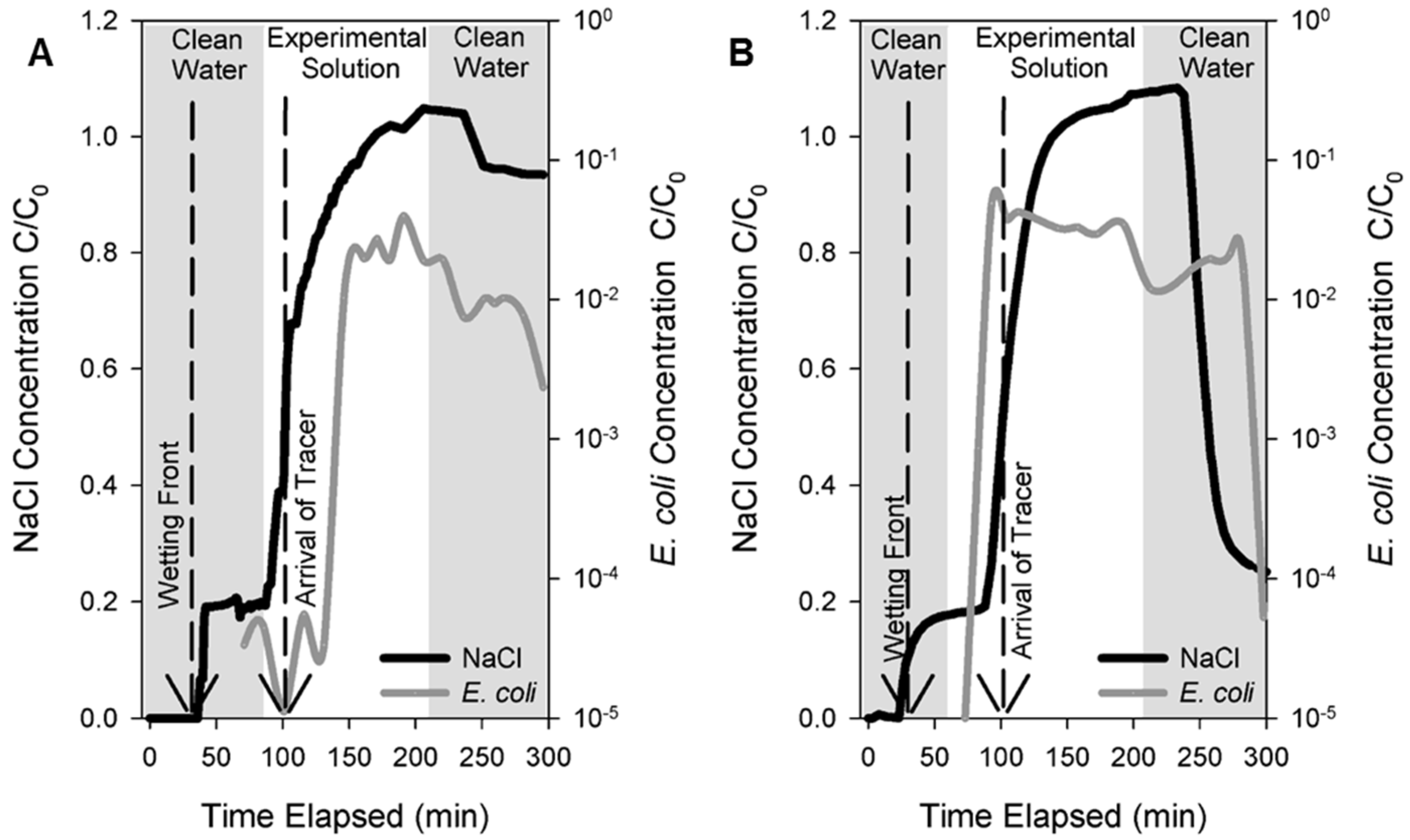
2.4. Controlled Field Tracer Experiments
3. Results and Discussion
3.1. Field Tracer Experiment
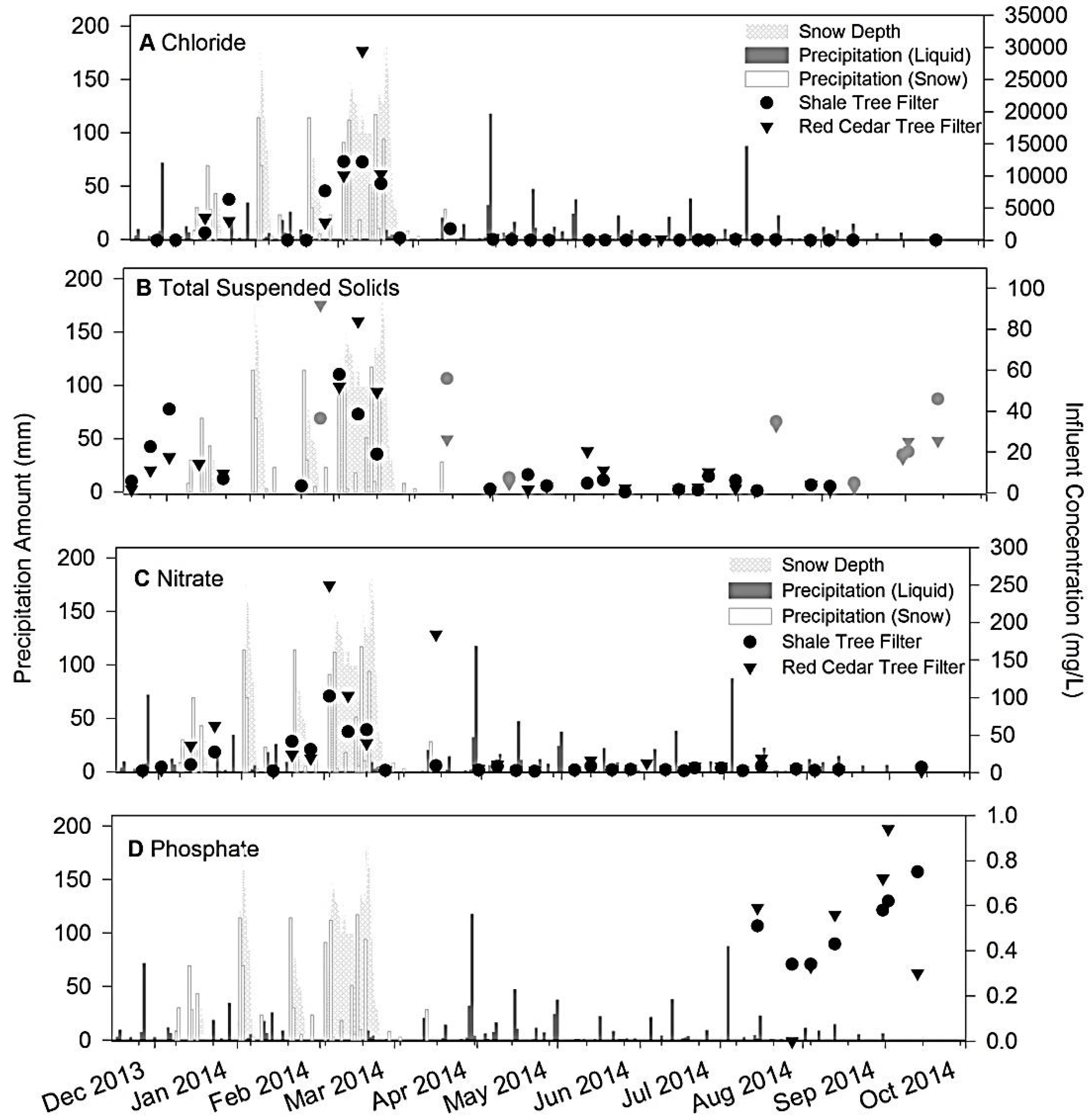
3.2. Field Monitoring Results
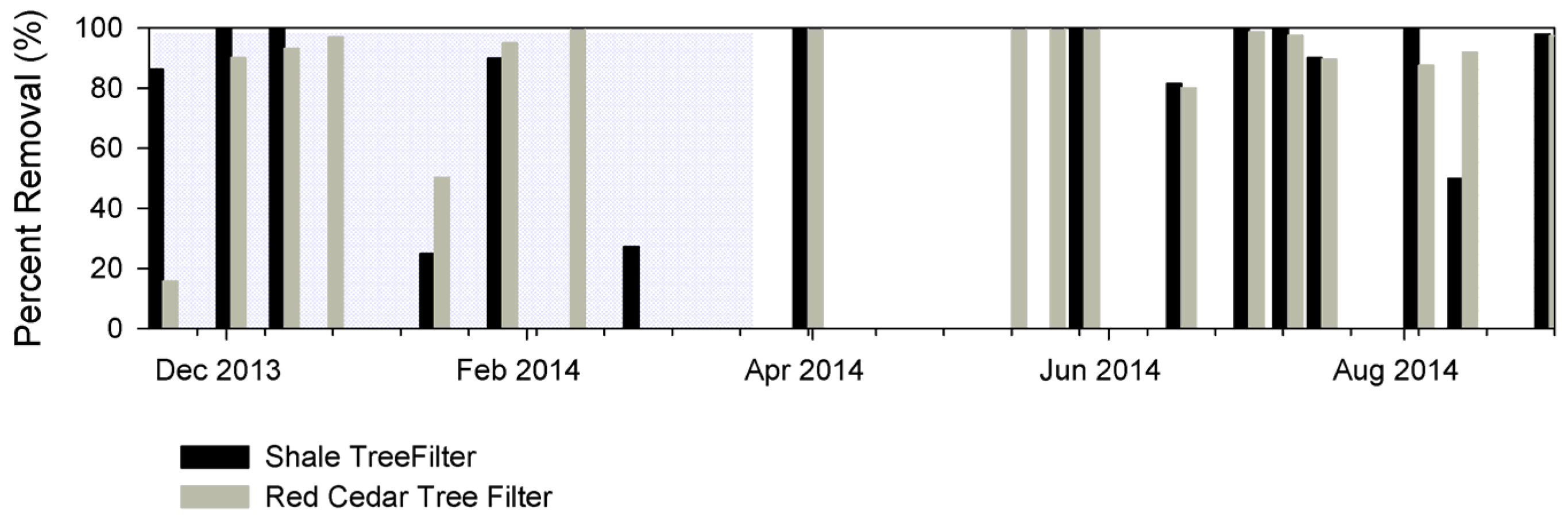
3.3. Contaminant Removal
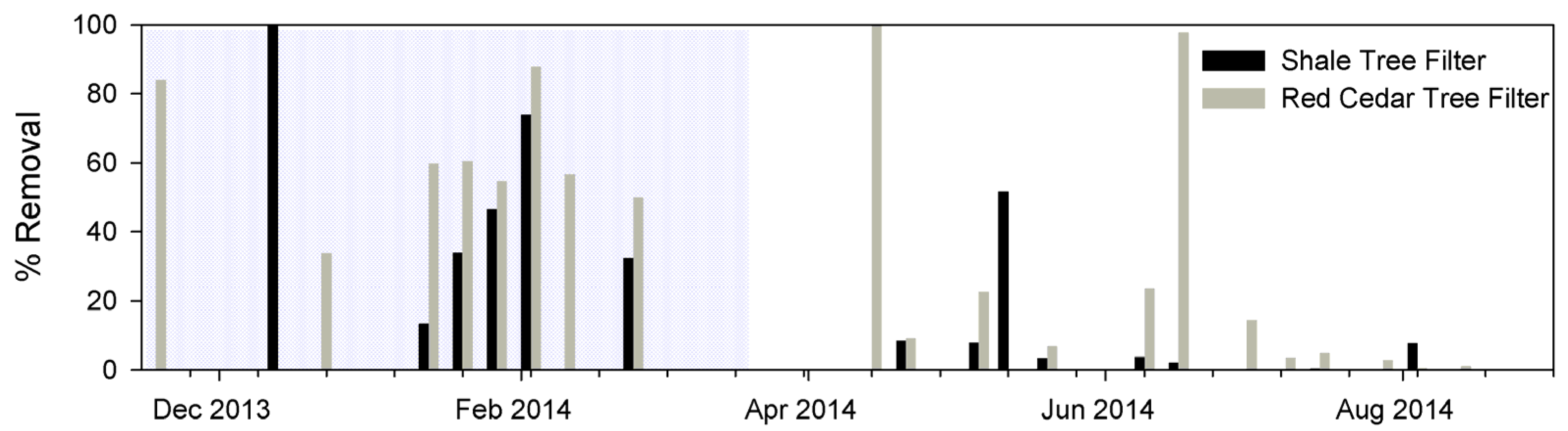
3.3.1. E. coli
3.3.2. Polycyclic Aromatic Hydrocarbons
3.3.3. Nutrients and TSS
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ahn, J.H.; Grant, S.B.; Surbeck, C.Q.; DiGiacomo, P.M.; Nezlin, N.P.; Jiang, S. Coastal water quality impact of stormwater runoff from an urban watershed in southern California. Environ. Sci. Technol. 2005, 39, 5940–5953. [Google Scholar] [CrossRef] [PubMed]
- Dorfman, M.H.; Stoner, N. Testing the Waters: A Guide to Water Quality at Vacation Beaches; Natural Resources Defense Council: New York, NY, USA, 2007; pp. 1–377. [Google Scholar]
- Lee, C.M.; Lin, T.Y.; Lin, C.C.; Kohbodi, G.A.; Bhatt, A.; Lee, R.; Jay, J.A. Persistence of fecal indicator bacteria in Santa Monica Bay beach sediments. Water Res. 2006, 40, 2593–2602. [Google Scholar] [CrossRef] [PubMed]
- Mallin, M.A.; Williams, K.E.; Esham, E.C.; Lowe, R.P. Effect of human development on bacteriological water quality in coastal watersheds. Ecol. Appl. 2000, 10, 1047–1056. [Google Scholar] [CrossRef]
- McLellan, S.; Salmore, A. Evidence for localized bacterial loading as the cause of chronic beach closings in a freshwater marina. Water Res. 2003, 37, 2700–2708. [Google Scholar] [CrossRef]
- Parker, J.; McIntyre, D.; Noble, R. Characterizing fecal contamination in stormwater runoff in coastal North Carolina, USA. Water Res. 2010, 44, 4186–4194. [Google Scholar] [CrossRef] [PubMed]
- Environmental Protection Agency (EPA). Performance Standards for Discharges from Newly Developed and Redeveloped Sites; EPA: Washington, DC, USA, 2001.
- Rhode Island Department of Environmental Management (RIDEM). Rhode Island Stormwater Design and Installation Standards Manual; RIDEM: Providence, RI, USA, 2010.
- Coffman, L.S.; Ruby, M.; Beach, C. Bacterra by Filterra® Advanced Bioretention System: Discussion of the Benefits, Mechanisms and Efficiencies for Bacteria Removal. In Proceedings of the Low Impact Development for Urban Ecosystem and Habitat Protection, Seattle, WA, USA, 16–19 November 2008; pp. 1–13.
- Faucette, L.; Cardoso-Gendreau, F.; Codling, E.; Sadeghi, A.; Pachepsky, Y.; Shelton, D. Storm water pollutant removal performance of compost filter socks. J. Environ. Qual. 2009, 38, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.P.; Hunt, W.F.; Traver, R.G.; Clar, M. Bioretention Technology: Overview of Current Practice and Future Needs. J. Environ. Eng. 2009, 135, 109–117. [Google Scholar] [CrossRef]
- UNH Stormwater Center 2007 Annual Report; University of New Hampshire, Cooperative Institute for Coastal and Estuarine Environmental Technology: Durham, NH, USA; Available online: http://ciceet.unh.edu/unh_stormwater_report_2007/index.php (accessed on 17 April 2011).
- Stevik, T.K.; Aa, K.; Ausland, G.; Hanssen, J.F. Retention and removal of pathogenic bacteria in wastewater percolating through porous media: A review. Water Res. 2004, 38, 1355–1367. [Google Scholar]
- Zhang, X.; Lulla, M. Evaluation of pathogenic indicator bacteria in structural best management practices. J. Environ. Sci. Health Part A 2006, 41, 2483–2493. [Google Scholar] [CrossRef] [PubMed]
- Schifman, L.A.; Kasaraneni, V.; Sullivan, R.K.; Craver, V.O.; Boving, T.B. New antimicrobially amended media for improved non-point source bacterial pollution treatment. Environ. Sci. Technol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, S.K.; Torkelson, A.A.; Dodd, H.; Nelson, K.L.; Boehm, A.B. Engineering Solutions to Improve the Removal of Fecal Indicator Bacteria by Bioinfiltration Systems during Intermittent Flow of Stormwater. Environ. Sci. Technol. 2013, 47, 10791–10798. [Google Scholar] [CrossRef] [PubMed]
- Davies, C.M.; Bavor, H.J. The fate of stormwater-associated bacteria in constructed wetland and water pollution control pond systems. J. Appl. Microbiol. 2000, 89, 349–360. [Google Scholar] [CrossRef]
- Morales, I.; Atoyan, J.A.; Amador, J.A.; Boving, T. Transport of Pathogen Surrogates in Soil Treatment Units: Numerical Modeling. Water 2014, 6, 818–838. [Google Scholar] [CrossRef]
- Hathaway, J.; Hunt, W.; Jadlocki, S. Indicator Bacteria Removal in Storm-Water Best Management Practices in Charlotte, North Carolina. J. Environ. Eng. 2009, 135, 1275–1285. [Google Scholar] [CrossRef]
- Karim, M.R.; Manshadi, F.D.; Karpiscak, M.M.; Gerba, C.P. The Persistence and Removal of Enteric Pathogens in Constructed Wetlands. Water Res. 2004, 38, 1831–1837. [Google Scholar] [CrossRef] [PubMed]
- Schueler, T.R.; Holland, H. Microbes and Urban Watersheds: Concentrations, Sources, and Pathways. The Pract. Watershed Prot. 2000, 74–84, 554–656. [Google Scholar]
- United States Environmental Protection Agency (EPA). Protocol for Developing Pathogen TMDLs; Office of Water (4503F), EPA: Washington, DC, USA, 2001; p. 132.
- Herngren, L.; Goonetilleke, A.; Ayoko, G.A.; Mostert, M.M.M. Distribution of polycyclic aromatic hydrocarbons in urban stormwater in Queensland, Australia. Environ. Pollut. 2010, 158, 2848–2856. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.P. Field performance of bioretention: Water quality. Environ. Eng. Sci. 2007, 24, 1048–1064. [Google Scholar] [CrossRef]
- Dunne, E.J.; Coveney, M.F.; Marzolf, E.R.; Hoge, V.R.; Conrow, R.; Naleway, R.; Lowe, E.F.; Battoe, L.E. Efficacy of a large-scale constructed wetland to remove phosphorus and suspended solids from Lake Apopka, Florida. Ecol. Eng. 2012, 42, 90–100. [Google Scholar] [CrossRef]
- Hatt, B.E.; Fletcher, T.D.; Deletic, A. Hydrologic and pollutant removal performance of stormwater biofiltration systems at the field scale. J. Hydrol. 2009, 365, 310–321. [Google Scholar] [CrossRef]
- Zhang, H.; Oyanedel-Craver, V. Comparison of the bacterial removal performance of silver nanoparticles and a polymer based quaternary amine functionalized silsesquioxane coated point-of-use ceramic water filters. J. Hazard. Mater. 2013. [Google Scholar] [CrossRef]
- Li, Q.; Mahendra, S.; Lyon, D.Y.; Brunet, L.; Liga, M.V.; Li, D.; Alvarez, P.J. Antimicrobial nanomaterials for water disinfection and microbial control: Potential applications and implications. Water Res. 2008, 42, 4591–4602. [Google Scholar] [CrossRef] [PubMed]
- Kasaraneni, V.K.; Schifman, L.A.; Boving, T.B.; Oyanedel-Craver, V. Enhancement of Surface Runoff Quality Using Modified Sorbents. ACS Sustain. Chem. Eng. 2014, 2, 1609–1615. [Google Scholar] [CrossRef]
- Boving, T.; Klement, J.; Rowell, R.; Xing, B. Effectiveness of wood and bark in removing organic and inorganic contaminates from aqueous solution. Mol. Cryst. Liquid Cryst. 2008, 483, 339–347. [Google Scholar] [CrossRef]
- Boving, T.B.; Neary, K. Attenuation of polycyclic aromatic hydrocarbons from urban stormwater runoff by wood filters. J. Contam. Hydrol. 2007, 91, 43–57. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Boving, T.B.; Xing, B. Sorption of PAHs by Aspen Wood Fibers as Affected by Chemical Alterations. Environ. Sci. Technol. 2006, 40, 3279–3284. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Ou, Z.; Boving, T.B.; Tyson, J.; Xing, B. Sorption of copper by chemically modified aspen wood fibers. Chemosphere 2009, 76, 1056–1061. [Google Scholar] [CrossRef] [PubMed]
- Boving, T.B.; Rowell, R. Environmental Applications of Lignocellolusic Filter Materials. In Sustainable Development in the Forest Products Industry; Rowell, R., Ed.; UFP Editions: Lisbon, Portugal, 2010. [Google Scholar]
- Dickerman, D.C. Aquifer Tests in the Stratified Drift, Chipuxet River Basin, Rhode Island; Water-Resources Investigations Report; US Geological Survey: Reston, VA, USA, 1984.
- Boving, T.B.; Stolt, M.H.; Augenstern, J.; Brosnan, B. Potential for localized groundwater contamination in a porous pavement parking lot setting in Rhode Island. Environ. Geol. 2008, 55, 571–582. [Google Scholar] [CrossRef]
- National Oceanic and Atmospheric Administration (NOAA). Annual Climatological Summary; NOAA: Providence, RI, USA, 2014.
- Schock, M.R.; Neff, C.H. Trace metal contamination from brass fittings. J. American Water Works Assoc. 1988, 80, 47–56. [Google Scholar]
- Pinheiro, L.B.; Gibbs, M.D.; Vesey, G.; Smith, J.J.; Bergquist, P. Fluorescent reference strains of bacteria by chromosomal integration of a modified green fluorescent protein gene. Appl. Microbiol. Biotechnol. 2008, 77, 1287–1295. [Google Scholar] [CrossRef] [PubMed]
- Chandrasena, G.I.; Deletic, A.; McCarthy, D.T. Survival of Escherichia coli in stormwater biofilters. Environ. Sci. Pollut. Res. 2013. [Google Scholar] [CrossRef]
- DiBlasi, C.J.; Li, H.; Davis, A.P.; Ghosh, U. Removal and Fate of Polycyclic Aromatic Hydrocarbon Pollutants in an Urban Stormwater Bioretention Facility. Environ. Sci. Technol. 2008, 43, 494–502. [Google Scholar] [CrossRef]
- Schwartzenbach, R.P.; Gschwend, P.M.; Imboden, D.M. Environmental Organic Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2002. [Google Scholar]
- Kendall, C.; Silva, S.; Chang, C.; Burns, D.; Campbell, D.; Shanley, J. Use of the δ 18 O and δ 15 N of Nitrate to Determine Sources of Nitrate in Early Spring Runoff in Forested Catchments. In Isotopes in Water Resources Management. V. 1; International Atomic Energy Agency (IAEA): Wien, Austria, 1996. [Google Scholar]
- Kurian, L.M.; Lautz, L.K.; Mitchell, M.J. Winter Hydrology and NO3− Concentrations in a Forested Watershed: A Detailed Field Study in the Adirondack Mountains of New York1. JAWRA J. Am. Water Resour. Assoc. 2013, 49, 264–283. [Google Scholar] [CrossRef]
- Jasper, J.T.; Jones, Z.L.; Sharp, J.O.; Sedlak, D.L. Nitrate Removal in Shallow, Open-Water Treatment Wetlands. Environ. Sci. Technol. 2014, 48, 11512–11520. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.P.; Shokouhian, M.; Sharma, H.; Minami, C. Water quality improvement through bioretention media: Nitrogen and phosphorus removal. Water Environ. Res. 2006, 78, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Seagren, E.A.; Davis, A.P. Engineered bioretention for removal of nitrate from stormwater runoff. Water Environ. Res. 2003, 75, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Keeney, D. The nitrogen cycle in sediment-water systems. J. Environ. Qual. 1973, 2, 15–29. [Google Scholar] [CrossRef]
- Chang, N.; Wanielista, M.; Daranpob, A. Filter media for nutrient removal in natural systems and built environments: II—Design and application challenges. Environ. Eng. Sci. 2010, 27, 707–720. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Inyang, M.; Zimmerman, A.R.; Cao, X.; Pullammanappallil, P.; Yang, L. Removal of phosphate from aqueous solution by biochar derived from anaerobically digested sugar beet tailings. J. Hazard. Mater. 2011, 190, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.N.; Peake, B.M. Sources of heavy metals and polycyclic aromatic hydrocarbons in urban stormwater runoff. Sci. Total Environ. 2006, 359, 145–155. [Google Scholar] [CrossRef] [PubMed]
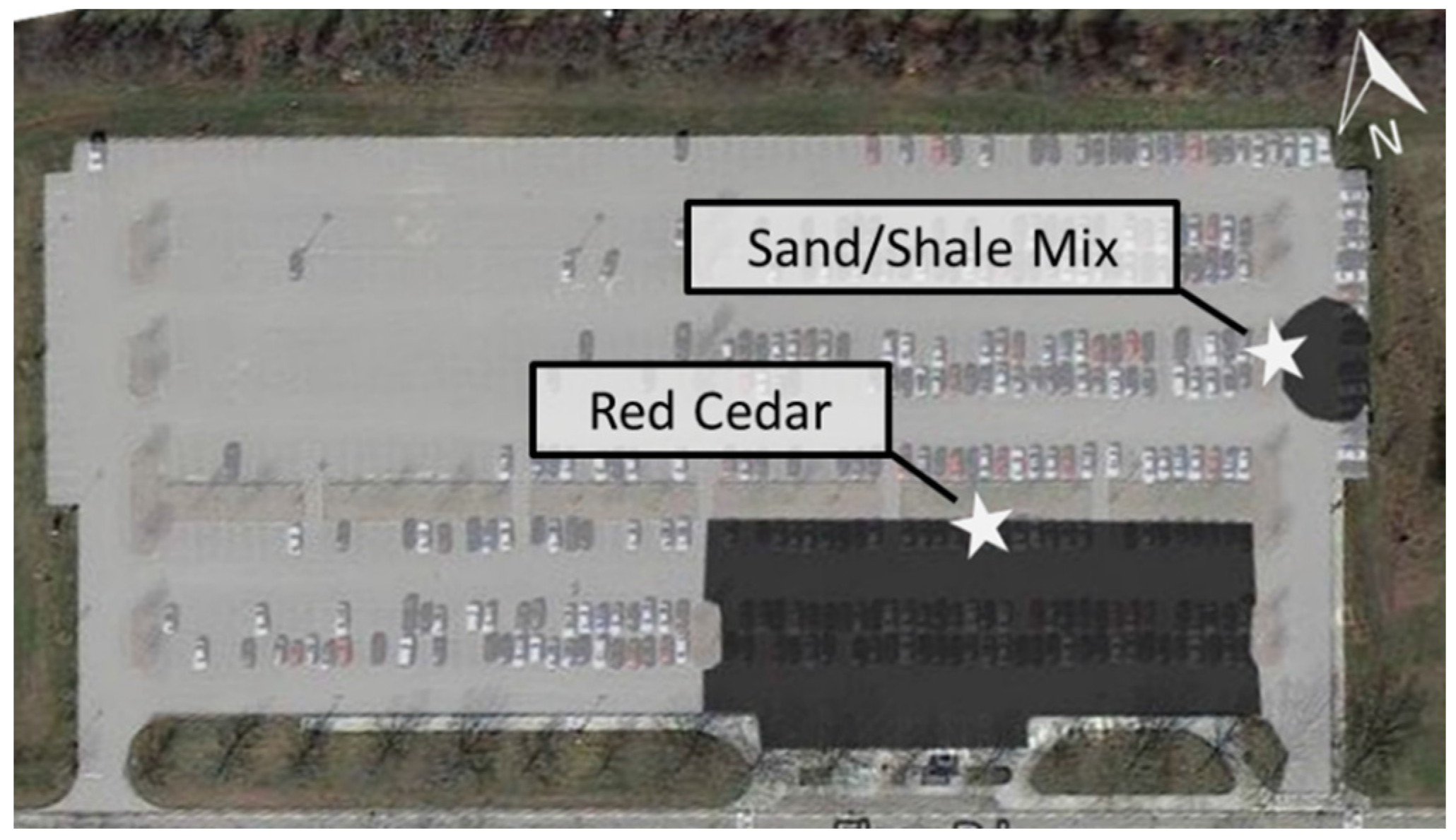
| Measure | Conventional Sand/Shale Tree Filter | Innovative Red Cedar Tree Filter * |
|---|---|---|
| Catch Basin Length (m) | 0.45 | 0.45 |
| Catch Basin Width (m) | 0.91 | 1.25 |
| Catch Basin Volume (L) | 409.5 | 542.6 |
| Catch Basin Depth (m) | 1.00 | 0.97 |
| Infiltration Area (m2) | 0.90 | 1.59 |
| Engineered treatment media Volume (m3) | 0.64 | 1.13 |
| Contributing Area (m2) | 238 | 2978 |
| Parameter | Conventional Sand/Shale Tree Filter (CTF) | Innovative Red Cedar Tree Filter (ITF) |
|---|---|---|
| Wetting Front (min) | 30 | 23 |
| Travel time of tracer (min) | 26 | 38 |
| NaCl Mass Recovered | 76% | 72.6% |
| Log10 Removal Value (LRV) | 2.09 | 2.02 |
| % Removal | 99.2 | 99.1 |
| Constituent | Units | WINTER | SUMMER | Literature Values | Reference |
|---|---|---|---|---|---|
| pH | - | 8.30 ± 1.62 | 7.69 ± 1.12 | 4.54–10.1 | a, b, c, l, o, s |
| (6.01–11.25) | (6.01–9.67) | ||||
| SC | µs/cm | 4286.0 ± 5616.0 | 91.9 ± 70.5 | 25–30,800 | a, d, e |
| (20.3–20,115.0) | (18.7–378.0) | ||||
| Chloride | mg/L | 4564.7 | 30.8 | 0.2–11,200 | a, d, e, l, s |
| (2.1–29,480.0) | (18.7–366.6) | ||||
| Nitrate | mg/L | 40.2 | 10.2 | 0.02–34.7 | a, b, c, f, g, h, n, s |
| (1.4–249.6) | (2.1–18.5) | ||||
| Phosphate | mg/L | - | 0.5 | 0.005–2.3 | a, b, c, g, n |
| (0.0–0.94) | |||||
| TSS* | mg/L | 44.9 | 9.2 | 1–46,000 | a, c, g, h, p, r |
| (26.3–92.0) | (2.5–56.0) | ||||
| Copper | µg/L | 85 | 2.4 | 6–1800 | a, g, h, q |
| (b.d.–1356) | (b.d.–36.3) | ||||
| Nickel | µg/L | 4.9 | 4–170 | a, g | |
| (b.d.–68) | b.d. | ||||
| Lead | µg/L | 63.1 | 7.9 | 2–525 | a, g, h, m, q |
| (b.d.–893.0) | (b.d.–118.0) | ||||
| Zinc | µg/L | 459.1 | 19.7 | 25–13,000 | a, g, h, q, r |
| (b.d.–5899.0) | (n.d.–118.0) | ||||
| ∑PAH16 | µg/L | 11.1 | 5.3 | 0–22.6 | a, l |
| (b.d.–21.1) | (b.d.–8.0) | ||||
| E. coli | CFU/100 mL | 2.3 * 104 | 6.44 * 102 | 110–106 | g, i, j, k, p |
| (0–2.0 * 105) | (0–2.3 * 104) |
| Shale Tree Filter | Red Cedar Tree Filter | |||||||
|---|---|---|---|---|---|---|---|---|
| Parameter | Unit | Winter | Summer | Winter | Summer | RISM Minimum Removal % | Literature Reported Removal % | Reference |
| % Change | % Change | % Change | % Change | |||||
| Chloride | mg/L | 70.7 ± 20.6 | −13.7 ± 12.9 | 46.0 ± 25.1 | 7.1 ± 5.9 | - | - | - |
| Nitrate | mg/L | 32.1 ± 26.8 | −8.0 ± 29.3 | 60.8 ± 53.8 | −8.3 ± 40.2 | 30 | 3–99 | h, r, s, t, u |
| Phosphate | mg/L | ~ | 45.3 ± 20.5 | ~ | 50.1 ± 26.9 | 30 | 65–80 | h, t |
| TSS* | mg/L | 63.4 ± 14.3 | 59.1 ± 10.3 | 85.4 ± 17.3 | 73.8 ± 10.0 | 85 | 54–99 | h, u, v |
| Copper | µg/L | ^ | ^ | ^ | ^ | - | 67–99 | h, y |
| Nickel | µg/L | ^ | ^ | ^ | ^ | - | + | z |
| Lead | µg/L | ^ | ^ | ^ | ^ | - | 80–99 | h, y |
| Zinc | µg/L | ^ | ^ | ^ | ^ | - | 84–99 | h, y |
| ∑PAH16 | µg/L | 46.5 ± 5.2 | 8.5 ± 2.4 | 54.7 ± 8.4 | 9.1 ± 3.6 | - | 18.5–99 | l, u |
| E. coli | CFU/100 mL | 71.3 ± 35.5 | 86.3 ± 20.3 | 77.3 ± 32.0 | 89.9 ± 14.5 | 60 | 0–96 | w, x |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schifman, L.A.; Kasaraneni, V.K.; Sullivan, R.K.; Oyanedel-Craver, V.; Boving, T.B. Bacteria Removal from Stormwater Runoff Using Tree Filters: A Comparison of a Conventional and an Innovative System. Water 2016, 8, 76. https://doi.org/10.3390/w8030076
Schifman LA, Kasaraneni VK, Sullivan RK, Oyanedel-Craver V, Boving TB. Bacteria Removal from Stormwater Runoff Using Tree Filters: A Comparison of a Conventional and an Innovative System. Water. 2016; 8(3):76. https://doi.org/10.3390/w8030076
Chicago/Turabian StyleSchifman, Laura A., Varun K. Kasaraneni, Ryan K. Sullivan, Vinka Oyanedel-Craver, and Thomas B. Boving. 2016. "Bacteria Removal from Stormwater Runoff Using Tree Filters: A Comparison of a Conventional and an Innovative System" Water 8, no. 3: 76. https://doi.org/10.3390/w8030076
APA StyleSchifman, L. A., Kasaraneni, V. K., Sullivan, R. K., Oyanedel-Craver, V., & Boving, T. B. (2016). Bacteria Removal from Stormwater Runoff Using Tree Filters: A Comparison of a Conventional and an Innovative System. Water, 8(3), 76. https://doi.org/10.3390/w8030076








