Agricultural Best Management Practice Abundance and Location does not Influence Stream Ecosystem Function or Water Quality in the Summer Season
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
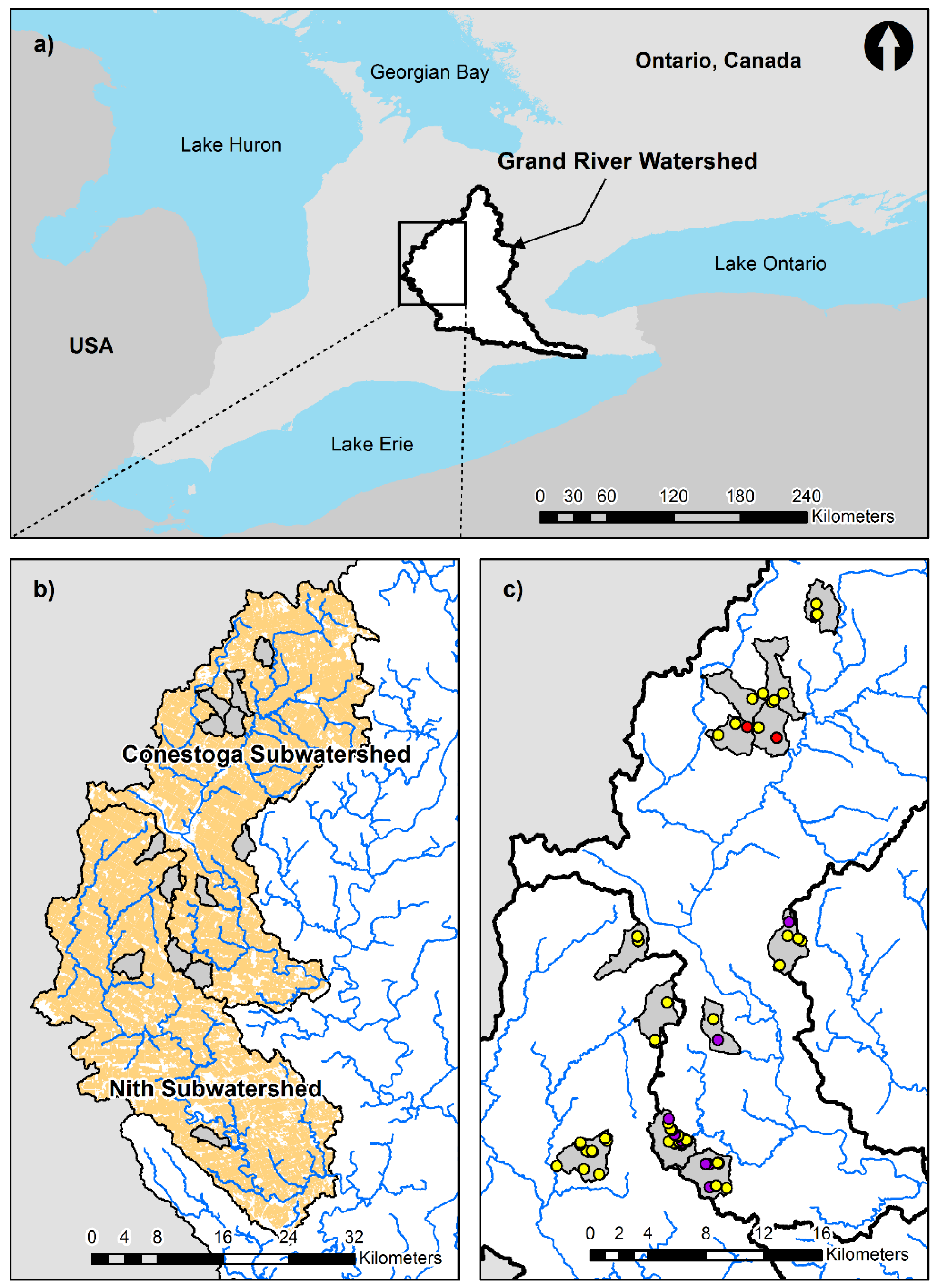
Site Selection and BMP Description
| Name | Acronym | Description | Expected Benefits |
|---|---|---|---|
| Manure Storage | MS | Impermeable structures designed to store livestock manure | Reduction in non-point organic matter and nutrient pollution |
| Livestock Access Restriction | LAR | Fences constructed adjacent to waterways preventing livestock from disturbing stream channels | Reduction in organic matter, nutrient, and sediment inputs |
| Erosion Control | EC | Structures preventing soil erosion by wind and/or water | Reduction in sediment and particulate nutrient inputs |
| Riparian Buffer Area | RBA | Area of vegetation either planted or left intact within a 30 m buffer on either side of a watercourse | Reduction in non-point organic matter, nutrient, sediment, and other contaminant inputs |
| Metric Type | Metric Name | Description | Applicable BMPs |
|---|---|---|---|
| BMP Abundance | Structural BMP(s) per farm | Ratio of BMP abundance to the number of farms within a catchment | MS, LAR, EC, BMP summary |
| % Riparian vegetation | Percentage of riparian vegetation present in a 30 m buffer area around the stream network | RBA | |
| Length of LAR | Length of LAR adjacent to the stream | LAR | |
| % River with LAR | Proportion of the stream length with LAR adjacent | LAR | |
| % River protected | Proportion of the stream with either LAR and/or riparian vegetation adjacent | RBA & LAR | |
| BMP Location | Hydrologic distance to stream | Overland distance from BMP to stream channel | MS, LAR, EC, BMP summary |
| Hydrologic distance to sampling point | Combined overland and instream distances water must travel from BMP to sampling site | MS, LAR, EC, BMP summary | |
| Flow accumulation | Catchment area that contributed surface runoff to BMP | MS, LAR, EC, BMP summary | |
| % Flow accumulation | The summed area contributing surface runoff to BMPs divided by the total catchment area | MS, LAR, EC, BMP summary |
2.2. Field Sampling
2.3. Data Analysis
3. Results
| Site Characteristic | Mean | Min. | Max. | Med. | St. Dev. | CV |
|---|---|---|---|---|---|---|
| Average stream depth (m) | 0.14 | 0.06 | 0.27 | 0.12 | 0.06 | 0.42 |
| Average stream width (m) | 1.65 | 0.85 | 2.86 | 1.59 | 0.56 | 0.34 |
| Flow velocity (m/s) | 0.05 | <0.01 | 0.13 | 0.03 | 0.06 | 1.07 |
| Discharge (m3/s) | 0.02 | <0.01 | 0.05 | 0.01 | 0.02 | 1.04 |
| Temperature (°C) | 17.96 | 15.80 | 19.88 | 17.99 | 1.42 | 0.08 |
| Canopy cover (%) | 29.27 | 6.92 | 85.75 | 25.08 | 22.50 | 0.77 |
| pH | 7.87 | 7.52 | 8.37 | 7.86 | 0.27 | 0.03 |
| Conductivity (µS/cm) | 575.84 | 472.00 | 753.00 | 551.00 | 87.74 | 0.15 |
| TN (N mg/L) | 1.99 | 0.75 | 4.68 | 1.87 | 1.11 | 0.56 |
| 3.39 | 1.56 | 5.80 | 3.38 | 1.12 | 0.33 | |
| NH4+ (N mg/L) | 0.07 | 0.01 | 0.46 | 0.03 | 0.12 | 1.73 |
| 0.30 | <0.01 | 2.62 | 0.04 | 0.73 | 2.41 | |
| NO2–NO3 (N mg/L) | 1.12 | 0.09 | 4.00 | 0.52 | 1.20 | 1.07 |
| 2.20 | 0.06 | 4.08 | 2.33 | 1.36 | 0.62 | |
| TP (P mg/L) | 0.22 | 0.02 | 0.92 | 0.07 | 0.30 | 1.37 |
| 0.32 | 0.02 | 1.70 | 0.08 | 0.46 | 1.47 | |
| SRP (P mg/L) | 0.15 | <0.01 | 0.71 | 0.02 | 0.25 | 1.62 |
| 0.21 | 0.01 | 1.24 | 0.05 | 0.35 | 1.63 | |
| TDP (P mg/L) | 0.16 | 0.01 | 0.68 | 0.03 | 0.23 | 1.50 |
| 0.22 | 0.01 | 1.13 | 0.06 | 0.32 | 1.49 | |
| Turbidity (NTU) | 7.75 | 1.53 | 27.46 | 3.97 | 7.70 | 0.99 |
| 7.57 | 1.10 | 24.27 | 4.93 | 7.68 | 1.02 | |
| TSS (mg/L) | 7.53 | 2.40 | 25.60 | 4.40 | 6.60 | 0.88 |
| 10.16 | 1.60 | 28.53 | 5.20 | 9.38 | 0.92 |
3.1. VIF Analysis
| BMP Metric | Count | Mean | Min. | Max. | Med. | SD | CV |
|---|---|---|---|---|---|---|---|
| BMP Summary | |||||||
| # of BMPs (w/o RBA) | 60 | 4.62 | 1.00 | 16.00 | 3.00 | 4.37 | 0.95 |
| BMP per farm | 0.41 | 0.12 | 0.94 | 0.35 | 0.24 | 0.59 | |
| BMP dist. sampling point (m) | 3457 | 1883 | 5234 | 3466 | 999 | 0.29 | |
| BMP dist. river (m) | 448 | 106 | 1020 | 337 | 330 | 0.74 | |
| BMP flow accumulation (m2) | Mean | 2803 | 113 | 9917 | 933 | 3306 | 1.18 |
| % | 0.12 | 9.0×10−5 | 0.81 | 2.0×10−3 | 0.26 | 2.16 | |
| BMP Type | |||||||
| # of MS | 40 | 3.08 | 1.00 | 8.00 | 2.00 | 2.40 | 0.78 |
| MS per farm | 0.29 | 0.08 | 0.62 | 0.29 | 0.15 | 0.52 | |
| MS dist. sampling point (m) | Mean | 3562 | 1940 | 5234 | 3466 | 1074 | 0.30 |
| Med. | 3657 | 1755 | 5234 | 3544 | 1063 | 0.29 | |
| MS dist. river (m) | Mean | 556 | 122 | 1694 | 302 | 469 | 0.84 |
| Med. | 534 | 109 | 1694 | 271 | 474 | 0.89 | |
| MS flow accumulation (m2) | Mean | 724 | 44 | 2921 | 400 | 803 | 1.11 |
| Med. | 564 | 33 | 2408 | 308 | 675 | 1.20 | |
| % | 1.2×10−3 | 1.9×10−5 | 0.01 | 5.3×10−4 | 1.7×10−3 | 1.43 | |
| # of LAR | 18 | 1.38 | 0.00 | 8.00 | 0.00 | 2.36 | 1.71 |
| LAR per farm | 0.09 | 0.00 | 0.47 | 0.08 | 0.14 | 1.49 | |
| LAR dist. sampling point (m) | Mean | 2720 | 1827 | 4651 | 2173 | 1102 | 0.41 |
| Med. | 2697 | 1753 | 4651 | 2149 | 1123 | 0.42 | |
| LAR dist. river (m) | Mean | 73 | 45 | 136 | 62 | 33 | 0.46 |
| Med. | 68 | 43 | 136 | 57 | 35 | 0.51 | |
| LAR flow accumulation (m2) | Mean | 7606 | 489 | 19789 | 6553 | 7265 | 0.96 |
| Med. | 3750 | 489 | 10838 | 1958 | 3992 | 1.06 | |
| % | 0.26 | 1.5×10−3 | 0.81 | 0.08 | 0.35 | 1.34 | |
| Length of LAR (m) | 1078 | 63 | 2846 | 871 | 1109 | 1.03 | |
| % river protected—LAR | 0.04 | 0.00 | 0.20 | 0.00 | 0.06 | 1.65 | |
| # of EC | 2 | 0.15 | 0.00 | 1.00 | 0.00 | 0.38 | 2.44 |
| EC per farm | 0.02 | 0.00 | 0.17 | 0.00 | 0.06 | 2.45 | |
| EC dist. sampling point (m) | Mean | 3226 | 1482 | 4969 | 3226 | 2466 | 0.76 |
| Med. | 3226 | 1482 | 4969 | 3226 | 2466 | 0.76 | |
| EC dist. river (m) | Mean | 717 | 53 | 1381 | 717 | 939 | 1.31 |
| Med. | 717 | 53 | 1381 | 717 | 939 | 1.31 | |
| EC flow accumulation (m2) | Mean | 7117 | 300 | 13933 | 71.17 | 96.40 | 1.35 |
| Med. | 7117 | 300 | 13933 | 71.17 | 96.40 | 1.35 | |
| % | 0.31 | 0.01 | 0.60 | 0.31 | 0.42 | 1.38 | |
| % RBA | 0.20 | 0.00 | 0.42 | 0.17 | 0.15 | 0.75 | |
| % river protected—LAR + RBA | 0.29 | 0.05 | 0.61 | 0.22 | 0.17 | 0.59 |
3.2. Stream Ecosystem Metabolism
| Metabolism Parameter | Mean | Min. | Max. | Median | St. Dev. | CV |
|---|---|---|---|---|---|---|
| k (day−1) | 21.99 | 6.41 | 44.84 | 17.77 | 11.75 | 0.53 |
| GPP (gO2/m2/day) | 11.75 | 0.88 | 35.93 | 8.45 | 10.67 | 0.91 |
| ER (gO2/m2/day) | 11.11 | 0.73 | 26.78 | 9.02 | 8.95 | 0.81 |
| NEM (gO2/m2/day) | 0.64 | −5.82 | 9.16 | 1.22 | 4.20 | 6.53 |
| P/R | 1.48 | 0.39 | 6.49 | 1.18 | 1.62 | 1.09 |
4. Discussion
Author Contributions
Conflicts of Interest
References
- Allan, J.D. Landscapes and riverscapes: The influence of land use on stream ecosystems. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 257–284. [Google Scholar] [CrossRef]
- Carpenter, S.R.; Caraco, N.F.; Correll, D.L.; Howarth, R.W.; Sharpley, A.N.; Smith, V.H. Nonpoint pollution of surface waters with phosphorus and nitrogen. Ecol. Appl. 1998, 83, 559–568. [Google Scholar] [CrossRef]
- Belsky, A.J.; Matzke, A.; Uselman, S. Survey of livestock influences on stream and riparian ecosystems in the western United States. J. Soil Water Conserv. 1999, 541, 419–431. [Google Scholar]
- Voora, V.A.; McCandless, M.; Roy, D.; Venema, H.D.; Oborne, B. Water quality trading in the lake winnipeg basin: A multilevel architecture. J. Gt. Lakes Res. 2012, 38, 183–192. [Google Scholar] [CrossRef]
- Young, R.G.; Huryn, A.D. Effects of land use on stream metabolism and organic matter turnover. Ecol. Appl. 1999, 94, 1359–1376. [Google Scholar] [CrossRef]
- Riseng, C.M.; Wiley, M.J.; Black, R.W.; Munn, M.D. Impacts of agricultural land use on biological integrity: A causal analysis. Ecol. Appl. 2011, 218, 3128–3146. [Google Scholar] [CrossRef]
- Henley, W.F.; Patterson, M.A.; Neves, R.J.; Lemly, A.D. Effects of sedimentation and turbidity on lotic food webs: A concise review for natural resource managers. Rev. Fish. Sci. 2000, 82, 125–139. [Google Scholar] [CrossRef]
- Kleinman, P.J.A.; Sharpley, A.N.; Withers, P.J.A.; Bergstrom, L.; Johnson, L.T.; Doody, D.G. Implementing agricultural phosphorus science and management to combat eutrophication. Ambio 2015, 44, 297–310. [Google Scholar] [CrossRef] [PubMed]
- Stuart, V.; Harker, D.B.; Scott, T.; Clearwater, R.L. Watershed Evaluation of Beneficial Management Practices (WEBs): Towards Enhanced Agricultural Landscape Planning—Four-Year Review (2004/5–2007/8); Agriculture and Agri-Food Canada: Ottawa, ON, Canada, 2010. [Google Scholar]
- Tomer, M.D.; Dosskey, M.G.; Burkart, M.R.; James, D.E.; Helmers, M.J.; Eisenhauer, D.E. Methods to prioritizing placement of riparian buffers for improved water quality. Agrofor. Syst. 2009, 75, 17–25. [Google Scholar] [CrossRef]
- Tamini, L.D.; Larue, B.; West, G. Technical and environmental efficiencies and best management practices in agriculture. Appl. Econ. 2012, 4413, 1659–1672. [Google Scholar] [CrossRef]
- Legge, J.T.; Doran, M.E.; Herbert, J.A.; O’Neil, G.; Mysorekar, S.; Sowa, S.; Hall, K.R. Prioritizing locations for implementing agricultural best management practices in a Midwestern watershed. J. Soil Water Conserv. 2013, 68, 22–33. [Google Scholar] [CrossRef]
- Giri, S.; Nejadhashemi, A.P.; Woznicki, S.; Zhang, Z. Analysis of best management practice effectiveness and spatiotemporal variability based on different targeting strategies. Hydrol. Process. 2014, 283, 431–445. [Google Scholar] [CrossRef]
- Owens, L.B.; Edwards, W.M.; van Keuren, R.W. Sediment losses from a pastured watershed before and after stream fencing. J. Soil Water Conserv. 1996, 511, 90–94. [Google Scholar]
- Vowell, J.L. Using stream bioassessment to monitor best management practice effectiveness. For. Ecol. Manag. 2001, 1431, 237–244. [Google Scholar] [CrossRef]
- Bagshaw, C.S.; Thorrold, B.; Davison, M.; Duncan, I.J.H.; Matthews, L.R. The influence of season and of providing a water trough on stream use by beef cattle grazing hill-country in New Zealand. Appl. Anim. Behav. Sci. 2008, 1092, 155–166. [Google Scholar] [CrossRef]
- Wang, L.; Lyons, J.; Kanehl, P. Effects of watershed best management practices on habitat and fish in Wisconsin streams. J. Am. Water Resour. Assoc. 2002, 38, 663–680. [Google Scholar] [CrossRef]
- Yates, A.G.; Bailey, R.C.; Schwindt, J.A. Effectiveness of best management practices in improving stream ecosystem quality. Hydrobiologia 2007, 5831, 331–344. [Google Scholar] [CrossRef]
- Rao, N.S.; Easton, Z.M.; Schneiderman, E.M.; Zion, M.S.; Lee, D.R.; Steenhuis, T.S. Modeling watershed-scale effectiveness of agricultural best management practices to reduce phosphorus loading. J. Environ. Manag. 2009, 903, 1385–1395. [Google Scholar] [CrossRef] [PubMed]
- Chaubey, I.; Chiang, L.; Gitau, M.; Mohamed, S. Effectiveness of best management practices in improving water quality in a pasture-dominated watershed. J. Soil Water Conserv. 2010, 656, 424–437. [Google Scholar] [CrossRef]
- Gabel, K.; Wehr, J.; Truhn, K. Assessment of the effectiveness of best management practices for streams draining agricultural landscapes using diatoms and macroinvertebrates. Hydrobiologia 2012, 6801, 247–264. [Google Scholar] [CrossRef]
- Olden, J.D.; Poff, N.L. Ecological processes driving biotic homogenization: Testing a mechanistic model using fish faunas. Ecology 2004, 85, 1867–1875. [Google Scholar] [CrossRef]
- Donohue, I.; Jackson, A.L.; Pusch, M.T.; Irvine, K. Nutrient enrichment homogenizes lake benthic assemblages at local and regional scales. Ecology 2009, 90, 3470–3477. [Google Scholar] [CrossRef] [PubMed]
- Maloney, K.O.; Munguia, P.; Mitchell, R.M. Anthropogenic disturbance and landscape patterns affect diversity patterns of aquatic benthic macroinvertebrates. J. N. Am. Benthol. Soc. 2011, 30, 284–295. [Google Scholar] [CrossRef]
- Palmer, M.A.; Hondula, K.L.; Koch, B.J. Ecological restoration of streams and rivers: Shifting strategies and shifting goals. Annu. Rev. Ecol. Evol. Syst. 2014, 45, 247–269. [Google Scholar] [CrossRef]
- Meyer, J.L. Can P/R ratio be used to assess the food base of stream ecosystems? Oikos 1989, 54, 119–121. [Google Scholar] [CrossRef]
- Young, R.G.; Matthaei, C.D.; Townsend, C.R. Organic matter breakdown and ecosystem metabolism: Functional indicators for assessing river ecosystem health. J. N. Am. Benthol. Soc. 2008, 273, 605–625. [Google Scholar] [CrossRef]
- Fellows, C.S.; Clapcott, J.E.; Udy, J.W.; Bunn, S.E.; Harch, B.D.; Smith, M.J.; Davies, P.M. Benthic metabolism as an indicator of stream ecosystem health. Hydrobiologia 2006, 5721, 71–87. [Google Scholar] [CrossRef]
- Yates, A.G.; Brua, R.B.; Culp, J.M.; Chambers, P.A. Multi-scaled drivers of rural prairie stream metabolism along human activity gradients. Freshw. Biol. 2012, 58, 675–689. [Google Scholar] [CrossRef]
- Grand River Conservation Authority (GRCA). About the Grand River Watershed. The Geography of the Grand River. Available online: http://www.grandriver.ca/index/document.cfm?Sec= 74&Sub1=7 (accessed on 23 February 2015).
- Yates, A.G.; Bailey, R.C. Selecting objectively defined reference sites for stream bioassessment programs. Environ. Monit. Assess. 2010, 170, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Government of Canada. Canadian Climate Normals 1981–2010 Station Data. Waterloo Wellington-A Ontario Canada Climate. Available online: http://climateweathergcca/climate_normals/results_1981_2010_ehtml?stnID=4832&lang=e&dCode=4&dispBack=1 (accessed on 6 February 15).
- Holysh, S.; Pitcher, J.; Boyd, D. Regional Ground Water Mapping: An Assessment Tool For Incorporating Groundwater into the Planning Process; Grand River Conservation Authority: Cambridge, ON, Canada, 2000. [Google Scholar]
- Ontario Ministry of Natural Resources (OMNR). Southwestern Ontario Orthophotography Project (2006): 1 km Coverage, Scholars Portal, Ontario Council of University Libraries, 2012. Available online: http://geo2scholarsportalinfo/#r/search/_queries@=SWOOP;&fields@=;&sort=relevance&limit=entitled (accessed on 17 November 2014).
- Hall, C.A. Migration and metabolism in a temperate stream ecosystem. Ecology 1972, 53, 585–604. [Google Scholar] [CrossRef]
- Grace, M.R.; Imberger, S.J. Stream Metabolism, Performing and Interpreting Measurements; Water Studies Centre Monash University, Murray Darling Basin Commission, New South Wales Department of Environment and Climate Change: Melbourne, Australia, 2006. [Google Scholar]
- Young, R.G.; Townsend, C.R.; Matthaei, C. Functional indicators of river ecosystem health—An interim guide for use in New Zealand. Cawthron Rep. 2004, 870, 495–523. [Google Scholar]
- Microsoft Excel, Version 1446; Microsoft Corporation: Redmond, WA, USA, 2010.
- Young, R.G.; Collier, K.J. Contrasting responses to catchment modification among a range of functional and structural indicators of river ecosystem health. Freshw. Biol. 2009, 54, 2155–2170. [Google Scholar] [CrossRef]
- Reynoldson, T.; Logan, C.; Pascoe, T.; Thompson, S.; Strachan, S.; Mackinlay, C.; McDermott, H.; Paull, T. Canadian Aquatic Biomonitoring Network (CABIN) Field Manual: Wadeable Streams; Environment Canada: Dartmouth, NS, Canada, 2012. [Google Scholar]
- Rice, E.W.; Baird, R.B.; Eaton, A.D.; Clesceri, L.S. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; American Public Health Association, American Water Works Association, Water Environment Federation: Washington, DC, USA, 2012. [Google Scholar]
- Hill, W.R.; Mulholland, P.J.; Marzolf, E.R. Stream ecosystem responses to forest leaf emergence in spring. Ecology 2001, 82, 2306–2319. [Google Scholar] [CrossRef]
- SYSTAT 13, Version 130005; Systat Software: San Jose, CA, USA, 2008.
- Blanchet, F.G.; Legendre, P.; Borcard, B. Forward selection of explanatory variables. Ecology 2008, 89, 2623–2632. [Google Scholar] [CrossRef] [PubMed]
- Wiley, M.J.; Osborne, L.L.; Larimore, R.W. Longitudinal structure of an agricultural prairie river system and its relationship to current stream ecosystem theory. Can. J. Fish. Aquat. Sci. 1990, 47, 373–384. [Google Scholar] [CrossRef]
- Bernot, M.J.; Sobota, D.J.; Hall, R.O.; Mulholland, P.J.; Dodds, W.K.; Webster, J.R.; Tank, J.L.; Ashkenas, L.R.; Cooper, L.W.; Dahm, C.N.; et al. Inter-regional comparison of land-use effects on stream metabolism. Freshw. Biol. 2010, 55, 1874–1890. [Google Scholar] [CrossRef]
- Frankforter, J.; Weyers, H.; Bales, J.; Moran, P.; Calhoun, D. The relative influence of nutrients and habitat on stream metabolism in agricultural streams. Environ. Monit. Assess. 2010, 1681, 461–479. [Google Scholar] [CrossRef] [PubMed]
- Young, R.G.; Huryn, A.D. Interannual variation in discharge controls ecosystem metabolism along a grassland river continuum. Can. J. Fish. Aquat. Sci. 1996, 53, 2199–2211. [Google Scholar] [CrossRef]
- Acuña, V.; Giorgi, A.; Munoz, I.; Uehlinger, U.; Sabater, S. Flow extremes and benthic organic matter shape the metabolism of a headwater Mediterranean stream. Freshw. Biol. 2004, 49, 960–971. [Google Scholar] [CrossRef]
- Griffiths, N.A.; Tank, J.L.; Royer, T.V.; Roley, S.S.; Rosi-Marshall, E.J.; Whiles, M.R.; Beaulieu, J.J.; Johnson, L.T. Agricultural land use alters the seasonality and magnitude of stream metabolism. Limnol. Oceanogr. 2013, 58, 1513–1529. [Google Scholar]
- Ontario Ministry of Environment and Energy. Water Management—Policies, Guidelines, and Provincial Water Quality Objectives of the Ministry of Environment and Energy; PIBS 3303E; Queen’s Printer for Ontario: Toronto, ON, Canada, 1994.
- Canadian Council of Ministers of the Environment. Canadian water quality guidelines for the protection of aquatic life: Phosphorus: Canadian Guidance Framework for the Management of Freshwater Systems. In Canadian Environmental Quality Guidelines; Canadian Council of Ministers of the Environment: Winnipeg, MB, Canada, 2004. [Google Scholar]
- Canadian Council of Ministers of the Environment. Canadian water quality guidelines for the protection of aquatic life: Ammonia. In Canadian Environmental Quality Guidelines; Canadian Council of Ministers of the Environment: Winnipeg, MB, Canada, 1999. [Google Scholar]
- Chambers, P.A.; McGoldrick, D.J.; Brua, R.B.; Vis, C.; Culp, J.M.; Benoy, G.A. Development of environmental thresholds for nitrogen and phosphorus in streams. J. Environ. Qual. 2012, 41, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Groffman, P.M.; Baron, J.S.; Blett, T.; Gold, A.J.; Goodman, I.; Gunderson, L.H.; Levinson, B.M.; Palmer, M.A.; Paerl, H.W.; Peterson, G.D.; et al. Ecological thresholds: The key to successful environmental management or an important concept with no practical application? Ecosystems 2006, 9, 1–13. [Google Scholar] [CrossRef]
- Bosch, N.S.; Allan, J.D.; Selegean, J.P.; Scavia, D. Scenario-testing of agricultural best management practices in Lake Erie watersheds. J. Gt. Lakes Res. 2013, 39, 429–436. [Google Scholar] [CrossRef]
- Clausen, J.C.; Meals, D.W. Water quality achievable with agricultural best management practices. J. Soil Water Conserv. 1989, 44, 593–596. [Google Scholar]
- Walker, J.; Graczyk, D. Preliminary evaluation of effects of best management practices in the black earth creek, wisconsin, priority watershed. Water Sci. Technol. 1993, 283, 539–548. [Google Scholar]
- Park, S.; Cooke, R.; Mcclellan, P.; Mostaghimi, S. Bmp impacts on watershed runoff, sediment, and nutrient yields. Water Resour. Bull. 1994, 30, 1011–1023. [Google Scholar] [CrossRef]
- Mayer, P.M.; Reynolds, S.K.; McCutchen, M.D.; Canfield, T.J. Meta-analysis of nitrogen removal in riparian buffers. J. Environ. Qual. 2007, 36, 1172–1180. [Google Scholar] [CrossRef] [PubMed]
- Bishop, P.L.; Hively, W.D.; Stedinger, J.R.; Rafferty, M.R.; Lojpersberger, J.L.; Bloomfield, J.A. Multivariate analysis of paired watershed data to evaluate agricultural best management practice effects on stream water phosphorus. J. Environ. Qual. 2005, 34, 1087–1101. [Google Scholar] [CrossRef] [PubMed]
- Makarewicz, J.C.; Lewis, T.W.; Bosch, I.; Noll, M.R.; Herendeen, N.; Simon, R.D.; Zollweg, J.; Vodacek, A. The impact of agricultural best management practices on downstream systems: Soil loss and nutrient chemistry and flux to Conesus Lake, New York, USA. J. Gt. Lakes Res. 2009, 35, 23–36. [Google Scholar] [CrossRef]
- Mulholland, P.J.; Hill, W.R. Seasonal patterns in streamwater nutrient and dissolved organic carbon concentrations: Separating catchments flow path and in-stream effects. Water Resour. Res. 1997, 33, 1297–1306. [Google Scholar] [CrossRef]
- Yates, A.G.; Brua, R.B.; Corriveau, J.; Culp, J.M.; Chambers, P.A. Seasonally driven variation in spatial relationships between agricultural land use and in-stream nutrient concentrations. River Res. Appl. 2013, 30, 476–493. [Google Scholar] [CrossRef]
- Holmes, R. Determining the Association between the Structure of Stream Benthic Macroinvertebrate Communities and Agricultural Best Management Practices. Master’s Thesis, The University of Western Ontatio, London, ON, Canada, October 2014. [Google Scholar]
- Burcher, C.L.; Benfield, E.F. Physical and biological responses of streams to suburbanization of historically agricultural watersheds. J. N. Am. Benthol. Soc. 2006, 25, 356–369. [Google Scholar] [CrossRef]
- Harding, J.S.; Benfield, E.F.; Bolstad, P.V.; Helfman, G.S.; Jones, E.B.D., III. Stream biodiversity, the ghost of land use past. Ecology 1998, 95, 14843–14847. [Google Scholar] [CrossRef]
- Maloney, K.O.; Feminella, J.W.; Mitchell, R.M.; Miller, S.A.; Mulholland, P.J.; Houser, J.N. Landuse legacies and small streams: Identifying relationships between historical land use and contemporary stream conditions. J. N. Am. Benthol. Soc. 2008, 27, 280–294. [Google Scholar] [CrossRef]
- Violin, C.R.; Cada, P.; Sudduth, E.B.; Hassett, B.A.; Penrose, D.L.; Bernhardt, E.S. Effects of urbanization and urban stream restoration on the physical and biological structure of stream ecosystems. Ecol. Appl. 2011, 21, 1932–1949. [Google Scholar] [CrossRef] [PubMed]
- Meals, D.W.; Dressing, S.A.; Davenport, T.E. Lag time in water quality response to best management practices: A review. J. Environ. Qual. 2010, 39, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Tomer, M.D.; Burkart, M.R. Long-term effects of nitrogen fertilizer use on ground water nitrate in two small watersheds. J. Environ. Qual. 2003, 32, 2158–2171. [Google Scholar] [CrossRef] [PubMed]
- Giroux, M.; Royer, R. Long term effects of phosphate applications on yields, evolution of P soil test, saturation, and solubility in two very rich soils. Agrosolutions 2007, 18, 17–24. [Google Scholar]
- McCollum, R.E. Buildup and decline of soil phosphorus: 30-Year trends on a Typic Umprabuult. Agron. J. 1991, 83, 77–85. [Google Scholar] [CrossRef]
- Schnepf, M.; Cox, C. Environmental Benefits of Conservation on Cropland: The Status of Our Knowledge; Soil and Water Conservation Society: Ankeny, IA, USA, 2006. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pearce, N.J.T.; Yates, A.G. Agricultural Best Management Practice Abundance and Location does not Influence Stream Ecosystem Function or Water Quality in the Summer Season. Water 2015, 7, 6861-6876. https://doi.org/10.3390/w7126661
Pearce NJT, Yates AG. Agricultural Best Management Practice Abundance and Location does not Influence Stream Ecosystem Function or Water Quality in the Summer Season. Water. 2015; 7(12):6861-6876. https://doi.org/10.3390/w7126661
Chicago/Turabian StylePearce, Nolan J. T., and Adam G. Yates. 2015. "Agricultural Best Management Practice Abundance and Location does not Influence Stream Ecosystem Function or Water Quality in the Summer Season" Water 7, no. 12: 6861-6876. https://doi.org/10.3390/w7126661
APA StylePearce, N. J. T., & Yates, A. G. (2015). Agricultural Best Management Practice Abundance and Location does not Influence Stream Ecosystem Function or Water Quality in the Summer Season. Water, 7(12), 6861-6876. https://doi.org/10.3390/w7126661





