Effect of Ultrasound Pretreatment on Sludge Digestion and Dewatering Characteristics: Application of Particle Size Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Substrates and Digestion Tests
| Parameter | Inoculum | PS | WAS | SS |
|---|---|---|---|---|
| TS (g/L) | 9.4 ± 0.2 | 13.7 ± 0.5 | 20.3 ± 0.8 | 19.0 ± 0.6 |
| VS (g/L) | 7.4 ± 0.2 | 9.78 ± 0.3 | 15.6 ± 0.6 | 14.4 ± 0.5 |
| pH | 7.2 ± 0.1 | 7.6 ± 0.1 | 6.4 ± 0.1 | 6.7 ± 0.1 |
| O.M. (a) (%) | 0.57 ± 0.02 | 0.76 ± 0.09 | 1.37 ± 0.40 | 1.1 ± 0.06 |
| KN (b) (%) | 0.05 ± 0.01 | 0.06 ± 0.02 | 0.24 ± 0.04 | 0.17 ± 0.01 |
| NH4+ (mg/L) | 276 ± 8.2 | 874 ± 42 | 241 ± 16 | 400 ± 25 |
| C/N | 6.6 ± 0.3 | 6.4 ± 0.2 | 3.2 ± 0.2 | 4.0 ± 0.2 |
2.2. Ultrasound Pretreatment
2.3. Analytical Techniques
3. Results and Discussion
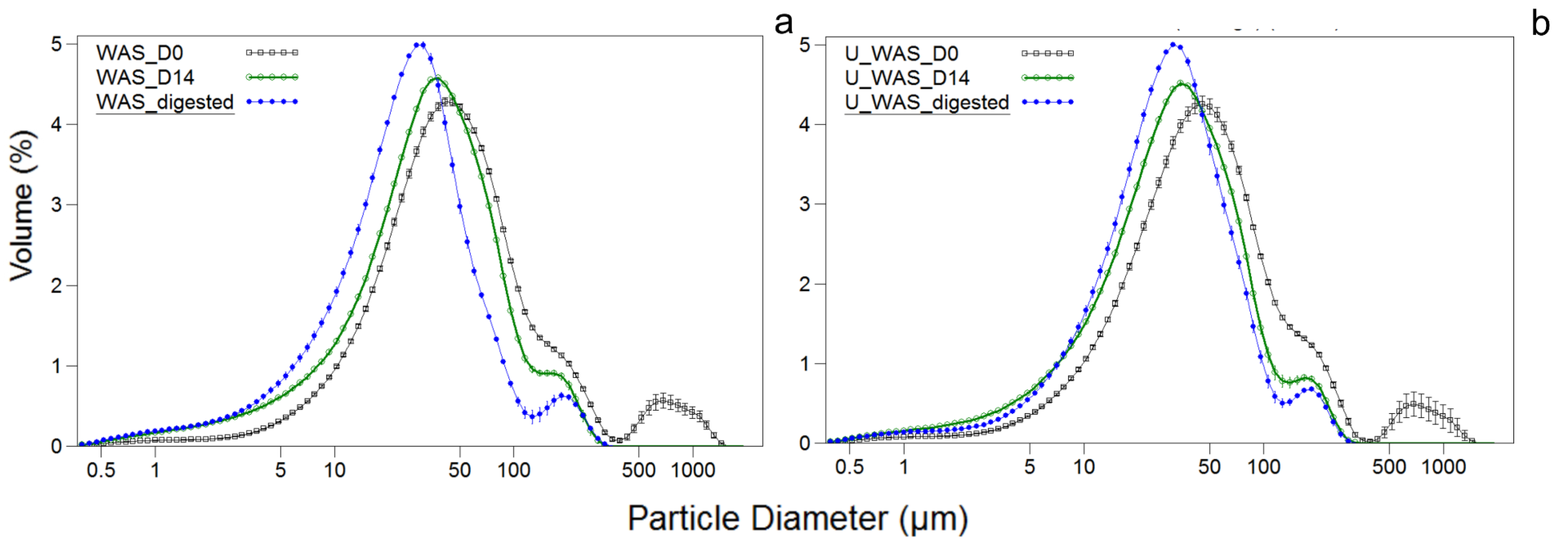
3.1. Effect of Pretreatment on Batch Digestion and Particle Size of the Sludge

| Substrate | Initial | Digested | ||
|---|---|---|---|---|
| Mean (μm) | SSA (cm2/mL) | Mean (μm) | SSA (cm2/mL) | |
| Inoculum | 29.5 ± 1.45 | 6122 ± 428 | n/a | n/a |
| PS | 59.9 ± 2.90 | 2594 ± 129 | 30.8 ± 1.52 | 4904 ± 245 |
| WAS | 61.0 ± 3.01 | 2672 ± 133 | 31.9 ± 1.50 | 4993 ± 249 |
| SS | 65.9 ± 3.25 | 2031 ± 101 | 34.8 ± 1.75 | 3664 ± 183 |
| U_WAS | 53.9 ± 2.62 | 2941 ± 147 | 31.3 ± 1.50 | 4695 ± 234 |
| U_SS | 60.9 ± 3.03 | 2203 ± 110 | 33.8 ± 1.74 | 3664 ± 183 |
3.2. Dewatering Parameters during the Digestion Process
| Sample | VSremoval (%) | TS (%) | TSS (%) | CST (s) | CSTs (s L/gTSS) | SRF (cm/g) |
|---|---|---|---|---|---|---|
| Inoculum | n/a | 3.7 ± 0.18 | 3.6 ± 0.10 | 332 ± 17 | 9.17 ± 0.27 | 2.62 × 1013 ± 1.31 × 1012 |
| WAS_D0 | n/a | 5.9 ± 0.29 | 5.1 ± 0.25 | 456 ± 27 | 8.89 ± 0.40 | 9.81 × 1013 ± 4.39 × 1012 |
| WAS_D14 | 22 ± 0.30 | 4.2 ± 0.21 | 3.5 ± 0.10 | 550 ± 38 | 15.33 ± 0.45 | 2.26 × 1014 ± 9.06 × 1012 |
| WAS_digested | 34 ± 0.69 | 3.5 ± 0.17 | 3.0 ± 0.9 | >2000 | 66.28 ± 1.98 | 3.79 × 1014 ± 1.89 × 1013 |
| U_WAS_D0 | n/a | 5.8 ± 0.29 | 4.4 ± 0.13 | 608 ± 30 | 13.67 ± 0.42 | 5.52 × 1014 ± 2.20 × 1013 |
| U_WAS_D14 | 18 ± 0.28 | 4.7 ± 0.23 | 3.6 ± 0.10 | 535 ± 32 | 14.69 ± 0.44 | 1.32 × 1014 ± 6.60 × 1012 |
| U_WAS_digested | 39 ± 0.34 | 3.5 ± 0.17 | 2.6 ± 0.07 | 267 ± 13 | 9.89 ± 0.49 | 8.04 × 1013 ± 4.02 × 1012 |

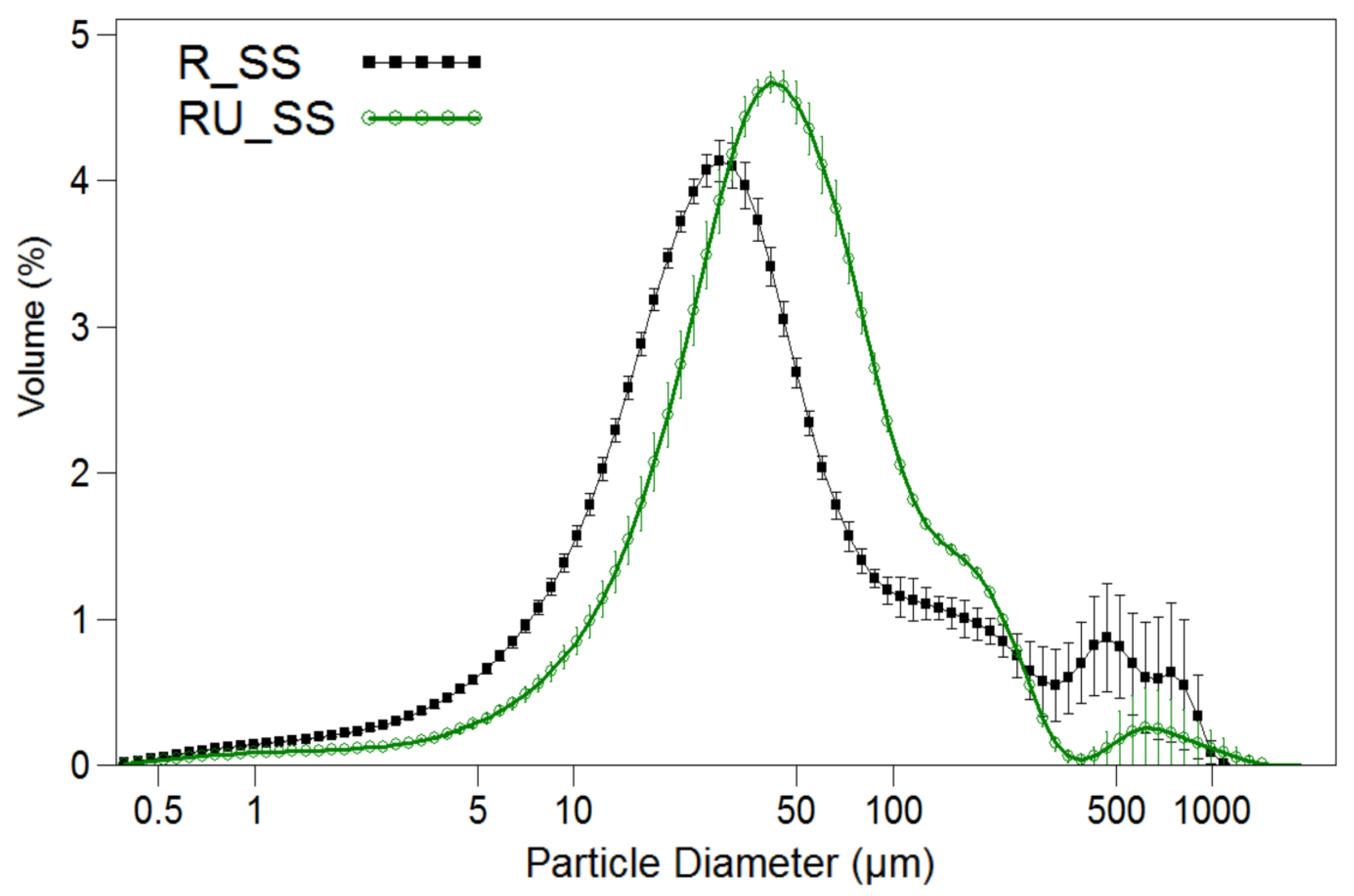
3.3. Digestion under Semi-Continuous Operation
| Performance Parameter | R_SS | RU_SS |
|---|---|---|
| OLR (a) (g VS/L day) | 1.80 ± 0.05 | 1.80 ± 0.09 |
| SMP (b) (mL/g VS ) | 120 ± 24 | 224 ± 20 |
| Gas Prod. (c) (L/day) | 0.92 ± 0.12 | 1.92 ± 0.12 |
| TVFA (mg/L) | 6711 ± 822 | n.d (d) |
| Acetic acid (mg/L) | 1938 ± 344 | n.d (d) |
| Propionic acid (mg/L) | 2733 ± 250 | n.d (d) |
| %VS removal | 6.0 ± 0.3 | 28.9 ± 0.86 |
| Mean particle size (μm) | 87.30 ± 4.36 | 76.10 ± 3.80 |
| Mode (μm) | 28.5 ± 1.14 | 55.7 ± 3.32 |
| Dp90 (μm) | 230 ± 11.0 | 144 ± 8.60 |
| SSA(cm2/mL) | 4191 ± 250 | 2487 ± 149 |
| CST(s) | 1765 ± 88 | 1737 ± 69 |
| SRF(cm/g) | 1.9552 × 1013 ± 7.8208 × 1011 | 6.6430 × 1012 ± 3.3215 × 1011 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dewil, R.; Baeyens, J.; Goutvrind, R. Ultrasonic treatment of waste activated sludge. Environ. Prog. 2006, 25, 121–128. [Google Scholar] [CrossRef]
- Lafitte-Trouqué, S.; Forster, C.F. The use of ultrasound and gamma-irradiation as pre-treatments for the anaerobic digestion of waste activated sludge at mesophilic and thermophilic temperatures. Bioresour. Technol. 2002, 84, 113–118. [Google Scholar] [CrossRef]
- Eastman, J.A.; Ferguson, J.F. Solubilization of particulate organic carbon during the acid phase of anaerobic digestion. J. Water Pollut. Control Fed. 1981, 53, 352–366. [Google Scholar]
- Shimizu, T.; Kudo, K.; Nasu, Y. Anaerobic waste-activated sludge digestion-a bioconversion mechanism and kinetic model. Biotechnol. Bioeng. 1993, 41, 1082–1091. [Google Scholar] [CrossRef] [PubMed]
- Khanal, S.K.; Grewell, D.; Sung, S.; van Leeuwen, J. Ultrasound Applications in Wastewater Sludge Pretreatment: A Review. Crit. Rev. Environ. Sci. Technol. 2007, 277–313. [Google Scholar] [CrossRef]
- Hogan, F.; Mormede, S.; Clark, P.; Crane, M. Ultrasonic sludge treatment for enhanced anaerobic digestion. Water Sci. Technol. 2004, 50, 25–32. [Google Scholar] [PubMed]
- Kim, D.J.; Lee, J. Ultrasonic sludge disintegration for enhanced methane production in anaerobic digestion: Effects of sludge hydrolysis efficiency and hydraulic retention time. Bioprocess. Biosyst. Eng. 2012, 35, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Hernando, M.; Martín-Díaz, J.; Labanda, J.; Mata-Alvarez, J.; Llorens, J.; Lucena, F.; Astals, S. Effect of ultrasound, low-temperature thermal and alkali pre-treatments on waste activated sludge rheology, hygienization and methane potential. Water Res. 2014, 61, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Zhen, G.; Lu, X.; Li, Y.Y.; Zhao, Y. Combined electrical-alkali pretreatment to increase the anaerobic hydrolysis rate of waste activated sludge during anaerobic digestion. Appl. Energy 2014, 128, 93–102. [Google Scholar] [CrossRef]
- Tiehm, A.; Nickel, K.; Zellhorn, M.; Neis, U. Ultrasonic waste activated sludge disintegration for improving anaerobic stabilization. Water Res. 2001, 35, 2003–2009. [Google Scholar] [CrossRef]
- Braguglia, C.M.; Gianico, A.; Mininni, G. Effect of ultrasound on particle surface charge and filterability during sludge anaerobic digestion. Water Sci. Technol. 2009, 60, 2025–2033. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.P.; Lee, D.J.; Chang, B.V.; You, C.S.; Tay, J.H. “Weak” ultrasonic pre-treatment on anaerobic digestion of flocculated activated biosolids. Water Res. 2002, 36, 2681–2688. [Google Scholar] [CrossRef]
- Riera-Franco De Sarabia, E.; Gallego-Juárez, J.A.; Rodríguez-Corral, G.; Elvira-Segura, L.; González-Gómez, I. Application of high-power ultrasound to enhance fluid/solid particle separation processes. Ultrasonics 2000, 38, 642–646. [Google Scholar] [CrossRef]
- Feng, X.; Deng, J.; Lei, H.; Bai, T.; Fan, Q.; Li, Z. Dewaterability of waste activated sludge with ultrasound conditioning. Bioresour. Technol. 2009, 100, 1074–1081. [Google Scholar] [CrossRef] [PubMed]
- Martinez, E.J.; Fierro, J.; Moreno, R.; Gomez, X. Particle size analysis and ultrasound pre-treatment of sludge. J. Residuals Sci. Technol. 2013, 10, 93–98. [Google Scholar]
- American Public Health Association (APHA); American Water Works Association (AWWA); The World Economic Forum (WEF). Standard Methods for the Examination of Water and Wastewater; American Public Health Association (APHA): Washington, DC, USA, 1999. [Google Scholar]
- Martínez, E.J.; Fierro, J.; Sánchez, M.E.; Gómez, X. Anaerobic co-digestion of FOG and sewage sludge: Study of the process by Fourier transform infrared spectroscopy. Int. Biodeter. Biodegr. 2012, 75, 1–6. [Google Scholar] [CrossRef]
- Lo, I.M.C.; Lai, K.C.K.; Chen, G.H. Salinity effect on mechanical dewatering of sludge with and without chemical conditioning. Environ. Sci. Technol. 2001, 35, 4691–4696. [Google Scholar] [CrossRef] [PubMed]
- Kopp, J.; Dichtl, N. Prediction of full-scale dewatering results of sewage sludges by the physical water distribution. Water Sci. Technol. 2001, 43, 135–143. [Google Scholar] [PubMed]
- Na, S.; Kim, Y.U.; Khim, J. Physiochemical properties of digested sewage sludge with ultrasonic treatment. Ultrason. Sonochem. 2007, 14, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; He, P.; Yu, G.; Shao, L. Effect of ultrasonic pretreatment on anaerobic digestion and its sludge dewaterability. J. Environ. Sci. 2011, 23, 1472–1478. [Google Scholar] [CrossRef]
- Shao, L.; He, P.; Yu, G.; He, P. Effect of proteins, polysaccharides, and particle sizes on sludge dewaterability. J. Environ. Sci. 2009, 21, 83–88. [Google Scholar] [CrossRef]
- Kopp, J.; Dichtl, N. Influence of the free water content on the dewaterability of sewage sludges. Water Sci. Technol. 2001, 44, 177–183. [Google Scholar] [PubMed]
- Lawler, D.F.; Chung, Y.J.; Hwang, S.-J.; Hull, B.A. Anaerobic digestion: Effects on particle size and dewaterability. J. Water Pollut. Control Fed. 1986, 58, 1107–1117. [Google Scholar]
- Mahmoud, N.; Zeeman, G.; Gijzen, H.; Lettinga, G. Interaction between digestion conditions and sludge physical characteristics and behaviour for anaerobically digested primary sludge. Biochem. Eng. J. 2006, 28, 196–200. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez, E.J.; Rosas, J.G.; Morán, A.; Gómez, X. Effect of Ultrasound Pretreatment on Sludge Digestion and Dewatering Characteristics: Application of Particle Size Analysis. Water 2015, 7, 6483-6495. https://doi.org/10.3390/w7116483
Martínez EJ, Rosas JG, Morán A, Gómez X. Effect of Ultrasound Pretreatment on Sludge Digestion and Dewatering Characteristics: Application of Particle Size Analysis. Water. 2015; 7(11):6483-6495. https://doi.org/10.3390/w7116483
Chicago/Turabian StyleMartínez, Elia Judith, Jose Guillermo Rosas, Antonio Morán, and Xiomar Gómez. 2015. "Effect of Ultrasound Pretreatment on Sludge Digestion and Dewatering Characteristics: Application of Particle Size Analysis" Water 7, no. 11: 6483-6495. https://doi.org/10.3390/w7116483
APA StyleMartínez, E. J., Rosas, J. G., Morán, A., & Gómez, X. (2015). Effect of Ultrasound Pretreatment on Sludge Digestion and Dewatering Characteristics: Application of Particle Size Analysis. Water, 7(11), 6483-6495. https://doi.org/10.3390/w7116483