Suitability of a Coupled Hydrodynamic Water Quality Model to Predict Changes in Water Quality from Altered Meteorological Boundary Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Happy Valley Reservoir
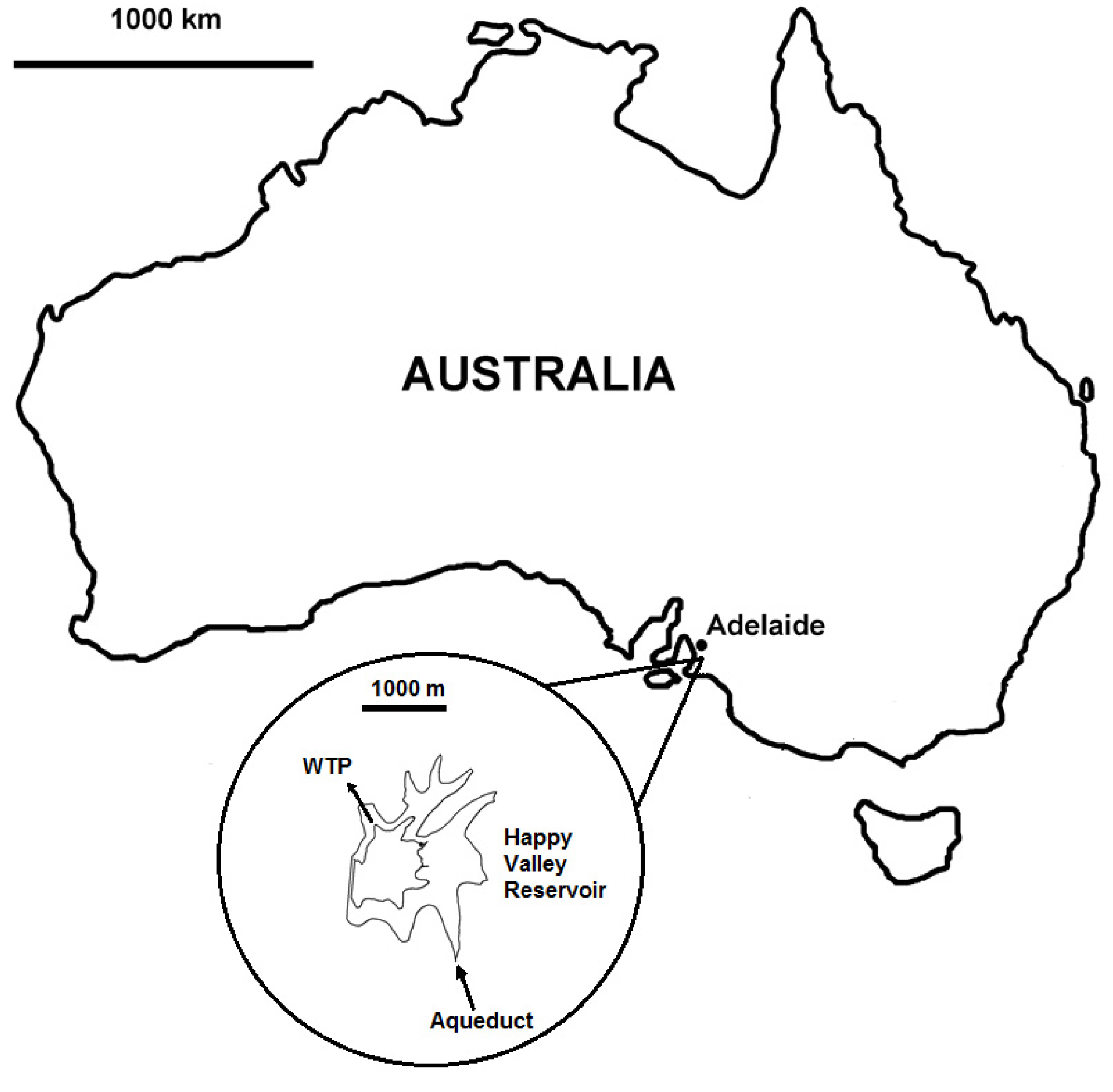
2.2. Model Description
| Parameter | Cyanophyte Value | Chlorophyte Value | Description | Reference |
|---|---|---|---|---|
| µGTH | 0.8 | 1.2 | Maximum growth rate (d−1) | [24] |
| ϑAg | 1.09 | 1.07 | Temperature multiplier for growth (-) | [25,26] |
| µRES | 0.09 | 0.10 | Respiration, mortality and excretion (d−1) | [27] |
| KP | 0.009 | 0.008 | P ½ saturation constant (mg L−1) | Calibrated |
| IK | 130 | 100 | Light ½ saturation constant (µE m−2 s−1) | [28] |
| TSTD | 24 | 20 | Standard temperature for algal growth (°C) | [29] |
| TOPT | 30 | 22 | Optimum temperature for algal growth (°C) | [29,30] |
| TMAX | 39 | 35 | Maximum temperature for algal growth (°C) | [29] |
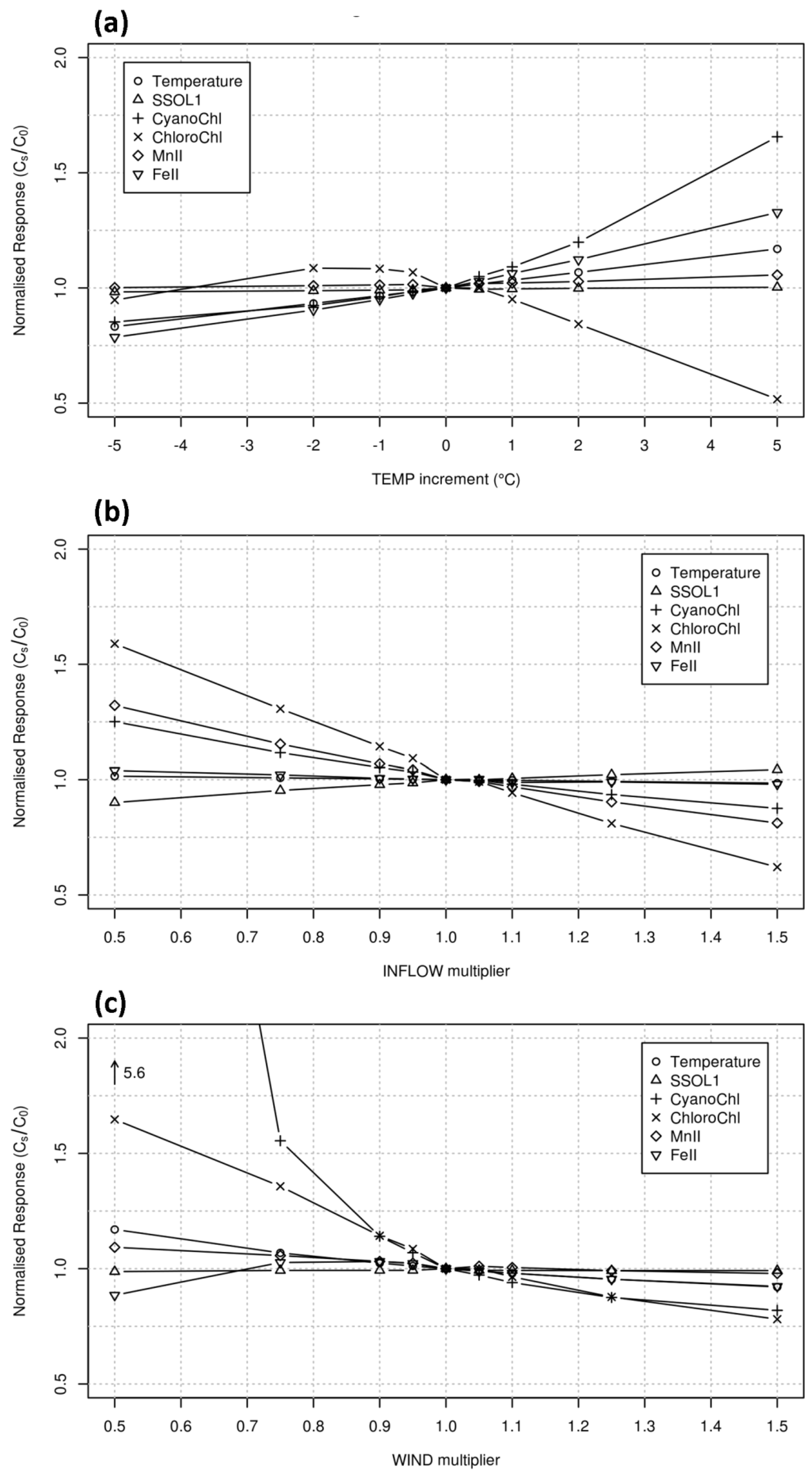
2.3. Scenarios for Analysis of ELCOM-CAEDYM Climatic Sensitivity
| Temperature (TEMP) [Increment] | Precipitation (FLOW) [Multiplier] | Wind Speed (WIND) [Multiplier] |
|---|---|---|
| −5.0 | 0.50 | 0.50 |
| −2.0 | 0.75 | 0.75 |
| −1.0 | 0.90 | 0.90 |
| −0.5 | 0.95 | 0.95 |
| 0.5 | 1.05 | 1.05 |
| 1.0 | 1.10 | 1.10 |
| 2.0 | 1.25 | 1.25 |
| 5.0 | 1.50 | 1.50 |
2.4. An Empirical Analysis of the Climatic Sensitivity of Chlorophyll-a to Temperature
3. Results and Discussion
3.1. Lake Physical Characteristics
3.2. Water Quality
| Factor | Increment/ Multiplier | g' (/s2) | Temperature Mean (°C) | Temperature Max (°C) | Temperature Min (°C) |
|---|---|---|---|---|---|
| Original | - | 0.0502 | 20.5 | 21.8 | 16.5 |
| INFLOW | 0.50 | 0.0481 | 20.9 | 22.2 | 16.6 |
| INFLOW | 0.75 | 0.0490 | 20.8 | 22.0 | 16.6 |
| INFLOW | 0.90 | 0.0496 | 20.6 | 21.9 | 16.5 |
| INFLOW | 0.95 | 0.0498 | 20.6 | 21.9 | 16.5 |
| INFLOW | 1.05 | 0.0503 | 20.5 | 21.8 | 16.5 |
| INFLOW | 1.10 | 0.0505 | 20.5 | 21.8 | 16.6 |
| INFLOW | 1.25 | 0.0510 | 20.3 | 21.7 | 16.6 |
| INFLOW | 1.50 | 0.0513 | 20.2 | 21.5 | 16.6 |
| TEMP | −5.0 | 0.0454 | 17.0 | 18.3 | 13.4 |
| TEMP | −2.0 | 0.0481 | 19.1 | 20.4 | 15.9 |
| TEMP | −1.0 | 0.0490 | 19.8 | 21.1 | 16.2 |
| TEMP | −0.5 | 0.0495 | 20.2 | 21.5 | 16.4 |
| TEMP | +0.5 | 0.0505 | 20.9 | 22.2 | 16.7 |
| TEMP | +1.0 | 0.0511 | 21.3 | 22.5 | 17.0 |
| TEMP | +2.0 | 0.0524 | 22.0 | 23.2 | 17.3 |
| TEMP | +5.0 | 0.0571 | 24.1 | 25.4 | 17.5 |
| WIND | 0.50 | 0.0984 | 22.7 | 25.9 | 17.0 |
| WIND | 0.75 | 0.0681 | 21.5 | 23.4 | 17.0 |
| WIND | 0.90 | 0.0560 | 20.9 | 22.4 | 16.7 |
| WIND | 0.95 | 0.0528 | 20.7 | 22.1 | 16.6 |
| WIND | 1.05 | 0.0474 | 20.4 | 21.6 | 16.6 |
| WIND | 1.10 | 0.0452 | 20.2 | 21.4 | 17.2 |
| WIND | 1.25 | 0.0397 | 19.8 | 20.8 | 17.4 |
| WIND | 1.50 | 0.0334 | 19.3 | 20.1 | 17.3 |
| Scenario | Production | Respiration | Limitation by | ||
|---|---|---|---|---|---|
| (day−1) | (day−1) | Light | Phosphorus | Nitrogen | |
| Original | 0.080 | 0.093 | 0.099 | 0.915 | 0.890 |
| INFLOW by 0.5 | 0.079 | 0.096 | 0.095 | 0.916 | 0.883 |
| INFLOW by 1.5 | 0.081 | 0.091 | 0.102 | 0.916 | 0.890 |
| TEMP by −5 | 0.061 | 0.076 | 0.101 | 0.917 | 0.890 |
| TEMP by +5 | 0.108 | 0.115 | 0.106 | 0.909 | 0.884 |
| WIND by 0.5 | 0.083 | 0.106 | 0.086 | 0.923 | 0.899 |
| WIND by 1.5 | 0.075 | 0.087 | 0.103 | 0.917 | 0.889 |
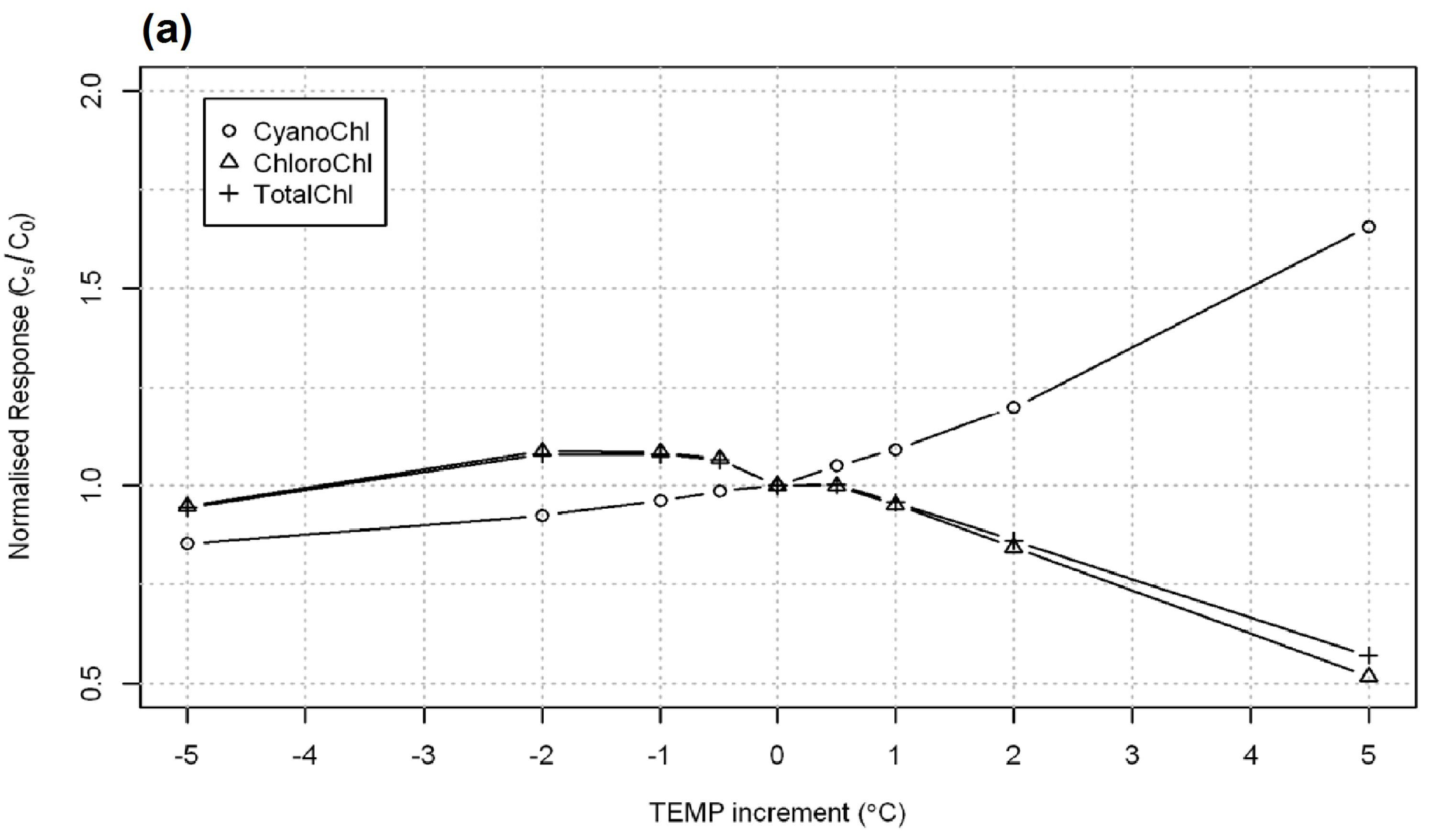
3.3. Implied Model Climatic Sensitivity
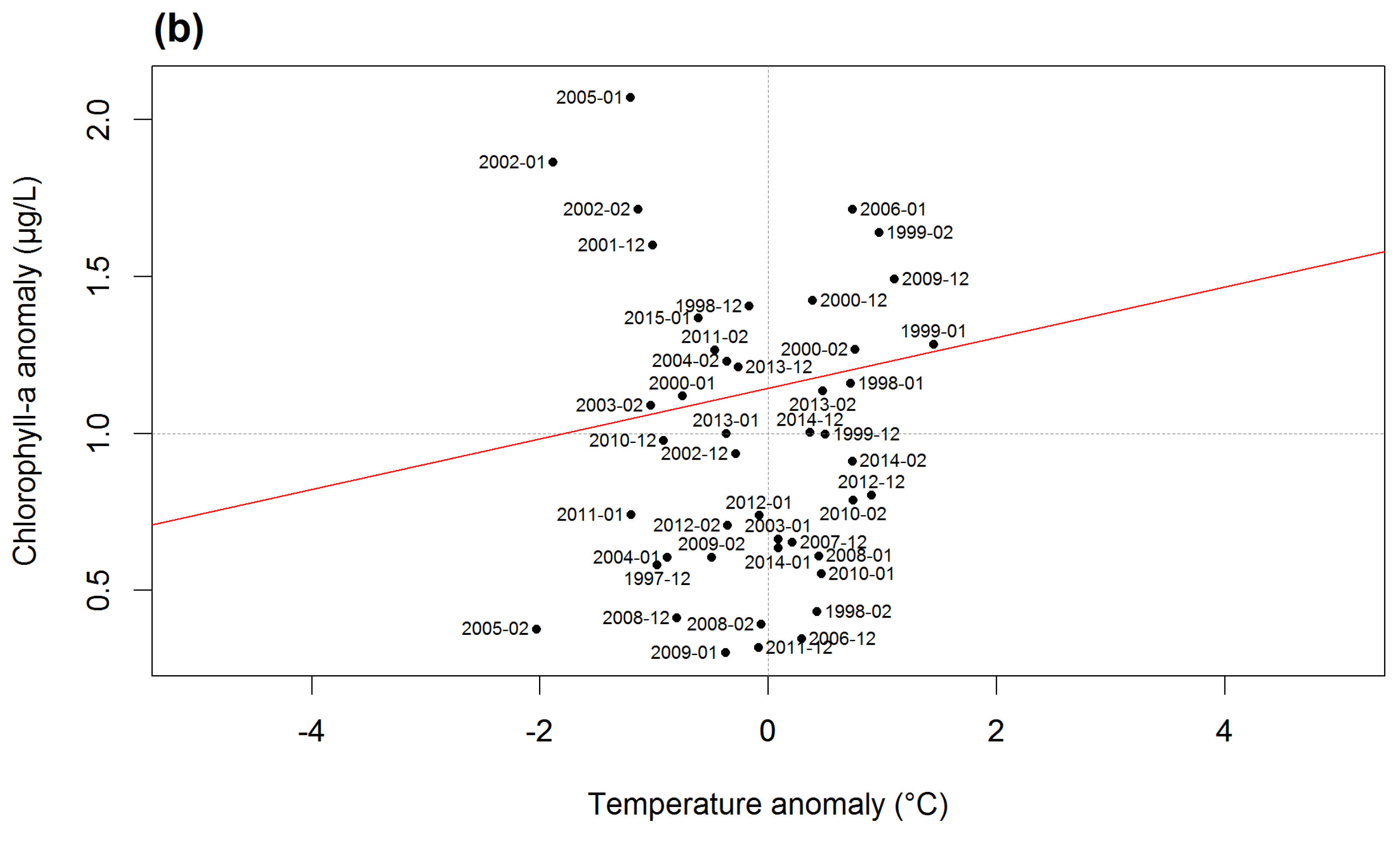
3.4. Empirical Reservoir Climatic Sensitivity
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- CSIRO. State of the Climate 2012; CSIRO and Bureau of Meteorology: Canberra, Australia, 2012; p. 12. [Google Scholar]
- CSIRO. Climate Change in Australia; CSIRO: Canberra, Australia, 2007. [Google Scholar]
- Heneker, T.; Cresswell, D. Potential Impact on Water Resource Availability in the Mount Lofty Ranges due to Climate Change; Science, Monitoring and Information Division, Department for Water: Adelaide, Australia, 2010. [Google Scholar]
- Charles, S.P.; Heneker, T.; Bates, B.C. Stochastically downscaled rainfall projections and modelled hydrological response for the Mount Lofty Ranges, South Australia. In Proceedings of Water Down Under 2008, Adelaide, Australia, 15–18 April 2008; Lambert, M., Daniell, T., Leonard, M., Eds.; Engineers Australia: Adelaide, Australia, 2008; pp. 428–438. [Google Scholar]
- Green, G.; Gibbs, M.; Wood, C. Impacts of Climate Change on Water Resources, Phase 3: Northern and Yorke Natural Resources Management Region DFW Technical Report 2011/03; Government of South Australia, Department for Water: Adelaide, Australia, 2011; Volume 1. [Google Scholar]
- Whitehead, P.G.; Wilby, R.L.; Battarbee, R.W.; Kernan, M.; Wade, A.J. A review of the potential impacts of climate change on surface water quality. Hydrol. Sci. J. 2009, 54, 101–123. [Google Scholar] [CrossRef]
- Delpla, I.; Jung, A.-V.; Baures, E.; Clement, M.; Thomas, O. Impacts of climate change on surface water quality in relation to drinking water production. Environ. Int. 2009, 35, 1225–1233. [Google Scholar] [CrossRef] [PubMed]
- Brookes, J.; Burch, M.; Hipsey, M.; Linden, L.; Antenucci, J.; Steffensen, D.; Hobson, P.; Thorne, O.; Lewis, D.; Rinck-Pfeiffer, S.; et al. A Practical Guide to Reservoir Management; Cooperative Research Centre for Water Quality and Treatment: Adelaide, Australia, 2008; p. 116. [Google Scholar]
- Arheimer, B.; Andréasson, J.; Fogelberg, S.; Johnsson, H.; Pers, C.B.; Persson, K. Climate change impact on water quality: Model results from southern Sweden. Ambio 2005, 34, 559–566. [Google Scholar] [PubMed]
- Mimikou, M.A.; Baltas, E.; Varanou, E.; Pantazis, K. Regional impacts of climate change on water resources quantity and quality indicators. J. Hydrol. 2000, 234, 95–109. [Google Scholar] [CrossRef]
- Saloranta, T.; Forsius, M.; Järvinen, M.; Arvola, L. Impacts of projected climate change on the thermodynamics of a shallow and a deep lake in Finland: Model simulations and Bayesian uncertainty analysis. Hydrol. Res. 2009, 40, 234–248. [Google Scholar] [CrossRef]
- Thorne, O.M.; Fenner, R.A. Modelling the impacts of climate change on a water treatment plant in South Australia. Water Sci. Technol. Water Supply 2008, 8, 305–312. [Google Scholar] [CrossRef]
- Elliott, J.A. Is the future blue-green? A review of the current model predictions of how climate change could affect pelagic freshwater cyanobacteria. Water Res. 2012, 46, 1364–1371. [Google Scholar] [CrossRef] [PubMed]
- Arhonditsis, G.B.; Perhar, G.; Zhang, W.; Massos, E.; Shi, M.; Das, A. Addressing equifinality and uncertainty in eutrophication models. Water Resour. Res. 2008, 44. [Google Scholar] [CrossRef]
- Makler-Pick, V.; Gal, G.; Gorfine, M.; Hipsey, M.R.; Carmel, Y. Sensitivity analysis for complex ecological models—A new approach. Environ. Model. Softw. 2011, 26, 124–134. [Google Scholar] [CrossRef]
- Saltelli, A. Making best use of model evaluations to compute sensitivity indices. Comput. Phys. Commun. 2002, 145, 280–297. [Google Scholar] [CrossRef]
- Campolongo, F.; Cariboni, J.; Saltelli, A. An effective screening design for sensitivity analysis of large models. Environ. Model. Softw. 2007, 22, 1509–1518. [Google Scholar] [CrossRef]
- Schlabing, D.; Frassl, M.A.; Eder, M.M.; Rinke, K.; Bárdossy, A. Use of a weather generator for simulating climate change effects on ecosystems: A case study on Lake Constance. Environ. Model. Softw. 2014, 61, 326–338. [Google Scholar] [CrossRef]
- Paerl, H.W.; Paul, V.J. Climate change: Links to global expansion of harmful cyanobacteria. Water Res. 2012, 46, 1349–1363. [Google Scholar] [CrossRef] [PubMed]
- Trolle, D.; Hamilton, D.P.; Pilditch, C.A.; Duggan, I.C.; Jeppesen, E. Predicting the effects of climate change on trophic status of three morphologically varying lakes: Implications for lake restoration and management. Environ. Model. Softw. 2011, 26, 354–370. [Google Scholar] [CrossRef]
- Hodges, B.; Dallimore, C. Estuary, Lake and Coastal Ocean Model: ELCOM v2.2 User Manual; Centre for Water Research, University of Western Australia: Crawley, Australia, 2007. [Google Scholar]
- Hipsey, M.R.; Romero, J.R.; Antenucci, J.P.; Hamilton, D.P. Computational Aquatic Ecosystem Dynamics Model CAEDYM v2.3 User Manual; Centre for Water Research, University of Western Australia: Crawley, Australia, 2006. [Google Scholar]
- Romero, J.; Antenucci, J.; Okley, P. Happy Valley Reservoir Modelling Study—Final Report; Centre for Water Research, University of Western Australia: Crawley, Australia, 2005; p. 41. [Google Scholar]
- USCE. CE-QUAL-R1: A Numerical One-Dimensional Model of Reservoir Water Quality; User’s Manual; Instruction Report E-82-1 (Revised Edition); Department of the Army, U.S. Corps Engineers: Washington, DC, USA, 1995; p. 427. [Google Scholar]
- Krüger, G.H.J.; Eloff, J.N. The influence of light intensity on the growth of different Microcystis isolates. J. Limnol. Soc. South. Afr. 2010, 3, 21–25. [Google Scholar] [CrossRef]
- Coles, J.F.; Jones, R.C. Effect of temperature on photosynthesis-light response and growth of four phytoplankton species isolated from a tidal freshwater river. J. Phycol. 2000, 36, 7–16. [Google Scholar] [CrossRef]
- Schladow, S.; Hamilton, D. Prediction of water quality in lakes and reservoirs: Part II—Model calibration, sensitivity analysis and application. Ecol. Modell. 1997, 96, 111–123. [Google Scholar] [CrossRef]
- Hamilton, D.P.; Schladow, S.G. Prediction of water quality in lakes and reservoirs. Part I—Model description. Ecol. Model. 1997, 96, 91–110. [Google Scholar]
- Griffin, S.L.; Herzfeld, M.; Hamilton, D.P. Modelling the impact of zooplankton grazing on phytoplankton biomass during a dinoflagellate bloom in the Swan River Estuary, Western Australia. Ecol. Eng. 2001, 16, 373–394. [Google Scholar] [CrossRef]
- Robarts, R.D.; Zohary, T. Temperature effects on photosynthetic capacity, respiration, and growth-rates of bloom-forming cyanobacteria. N. Z. J. Mar. Freshw. Res. 1987, 21, 391–399. [Google Scholar] [CrossRef]
- Elliott, J.A.; Jones, I.D.; Thackeray, S.J. Testing the sensitivity of phytoplankton communities to changes in water temperature and nutrient load, in a temperate lake. Hydrobiologia 2006, 559, 401–411. [Google Scholar] [CrossRef]
- Mooij, W.M.; Janse, J.H.; Senerpont Domis, L.N.; Hülsmann, S.; Ibelings, B.W. Predicting the effect of climate change on temperate shallow lakes with the ecosystem model PCLake. Hydrobiologia 2007, 584, 443–454. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van der Linden, L.; Daly, R.I.; Burch, M.D. Suitability of a Coupled Hydrodynamic Water Quality Model to Predict Changes in Water Quality from Altered Meteorological Boundary Conditions. Water 2015, 7, 348-361. https://doi.org/10.3390/w7010348
Van der Linden L, Daly RI, Burch MD. Suitability of a Coupled Hydrodynamic Water Quality Model to Predict Changes in Water Quality from Altered Meteorological Boundary Conditions. Water. 2015; 7(1):348-361. https://doi.org/10.3390/w7010348
Chicago/Turabian StyleVan der Linden, Leon, Robert I. Daly, and Mike D. Burch. 2015. "Suitability of a Coupled Hydrodynamic Water Quality Model to Predict Changes in Water Quality from Altered Meteorological Boundary Conditions" Water 7, no. 1: 348-361. https://doi.org/10.3390/w7010348
APA StyleVan der Linden, L., Daly, R. I., & Burch, M. D. (2015). Suitability of a Coupled Hydrodynamic Water Quality Model to Predict Changes in Water Quality from Altered Meteorological Boundary Conditions. Water, 7(1), 348-361. https://doi.org/10.3390/w7010348





