Comparison of Three Systems for Biological Greywater Treatment
Abstract
:1. Introduction
2. Experimental Section
2.1. Greywater source
2.2. Experimental setup
| SBR12 | SBR6 | |
| Cycle duration (h) | 6 | 4 |
| Filling (min) | 15 | 10 |
| Reaction time (h) | 4.8 | 3.2 |
| Settling time (min) | 45 | 30 |
| Emptying time (min) | 15 | 10 |

2.3. Chemical analysis
2.4. Determination of sludge yield
3. Results and Discussion
3.1. Characteristics of greywater
| days 1–168 | days 84–168 | |
| average | average | |
| COD total | 833 ± 188 | 827 ± 204 |
| COD suspended | 411 ± 151 | 385 ± 167 |
| COD colloidal | 204 ± 58 | 227 ± 65 |
| COD dissolved | 224 ± 59 | 196 ± 52 |
| Anionic surfactants | 43.5 ± 6.5 | |
| Total N | 41.2 ± 27.2 | 29.9 ± 11.0 |
| NH-N | 1.0 ± 0.7 | 0.6 ± 0.4 |
| NO-N | 0.12 ± 0.08 | 0.12 ± 0.07 |
| Total P | 6.6 ± 2.7 | 5.8 ± 1.4 |
3.2. General systems performance
3.3. Removal of different COD fractions
| SBR 12 | SBR6 | UASB12 | UASB7 | UASB7 + SBR6 | |
| (aerobic system) | (anaerobic system) | (anaerobic-aerobic system) | |||
| HRT (h) | 11.7 ± 1.1 | 6.1 ± 0.8 | 12.3 ± 1.8 | 7.0 ± 2.0 | 13.17 ± 2.03 |
| VLR (kg COD/md) | 1.6 ± 0.5 | 1.9 ± 0.4 | 1.7 ± 0.4 | 2.7 ± 0.8 | 1.5 ± 0.6 |
| COD removal rate (kg COD/md) | 1.5 ± 0.4 | 1.5 ± 0.4 | 0.8 ± 0.3 | 1.1 ± 0.6 | 1.4 ± 0.5 |
| SLR (kg COD/kg VSS d) | 0.29 ± 0.07 | 0.6 ± 0.3 | 0.12 ± 0.04 | 0.23 ± 0.08 | ** |
| Sludge concentration (g VSS/L) | 5.5 ± 1.1 | 3.3 ± 1.1 | 12.5 ± 2.4 | 12.7 ± 4.3 | ** |
| SRT (d) | 15 | 379 | 392 | 97 | ** |
| Yield (kg VSS/kg COD) | 0.12 | 0.06 | 0.08 | 0.18 | 0.18 |
| COD removal (%) | 90 ± 7 | 82 ± 06 | 51 ± 13 | 39 ± 15 | 89 ± 3 |
| COD effluent (mg/L) | 82 ± 47 | 100 ± 33 | 392 ± 85 | 528 ± 180 | 100 ± 33 |
| Anionic surfactants (mg/L) | 1.4 ± 1.2 | 1.3 ± 1.5 | 33.4 ± 4.1 | 35.9 ± 5.3 | 1.3 ± 1.5 |
| Effluent total N (mg/L) | 31 ± 20 | 26 ± 13 | 34 ± 17 | 32 ± 10 | 26 ± 13 |
| Effluent NH4-N (mg/L) | 0.35 ± 0.20 | 0.4 ± 0.1 | 4.7 ± 2.1 | 5.4 ± 2.4 | 0.4 ± 0.1 |
| Effluent NO3-N (mg/L) | 1.5 ± 1.4 | 22.6 ± 13.5 | 0.2 ± 0.1 | 0.2 ± 0.1 | 22.6 ± 13.5 |
| Effluent total P (mg/L) | 4.4 ± 2.4 | 5.8 ± 1.7 | 5.3 ± 1.5 | 6.1 ± 1.7 | 5.8 ± 1.7 |
| Effluent VSS (mg/L) | 45 ± 61 | 30 ± 26 | 7 ± 9 | 21 ± 23 | 30 ± 26 |
| Removal total N (%) | 35 ± 37 | 26 ± 27 | 15 ± 33 | –1 ± 63 | 2 ± 56 |
| NH4-N removal (%) | 51 ± 47 | 92 ± 4 | ** | ** | 7 ± 86 |
| Total P removal (%) | 28 ± 50 | 31 ± 11 | 11 ± 28 | 1 ± 36 | 3 ± 44 |
| Methane flow (NL/d) | ** | ** | 0.76 | 0.8 | 0.8 |
| Methane production (NL/m) | ** | ** | 123 | 71.5 | 71.5 |
- * No ammonium removal
- ** Not applicable

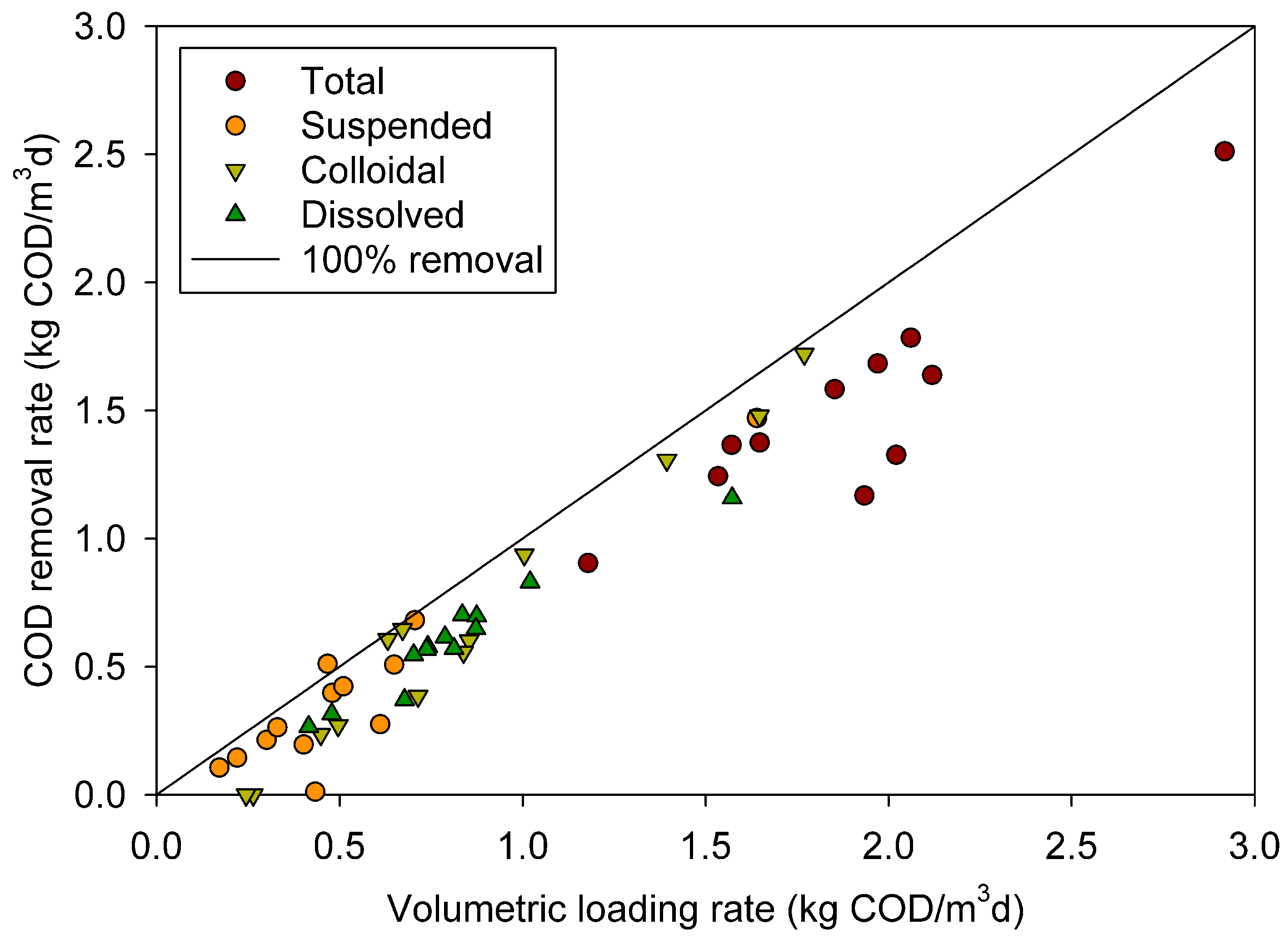
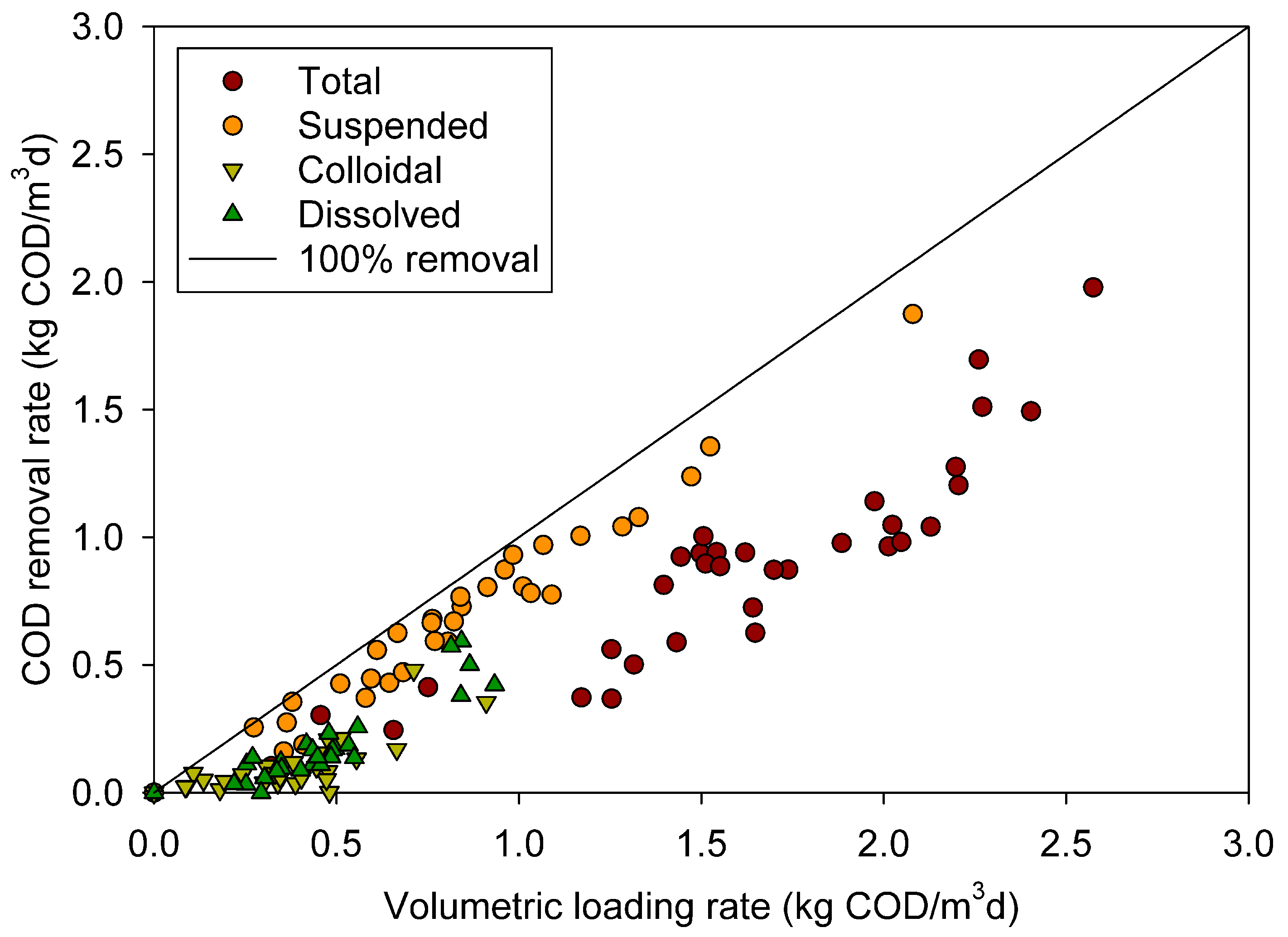
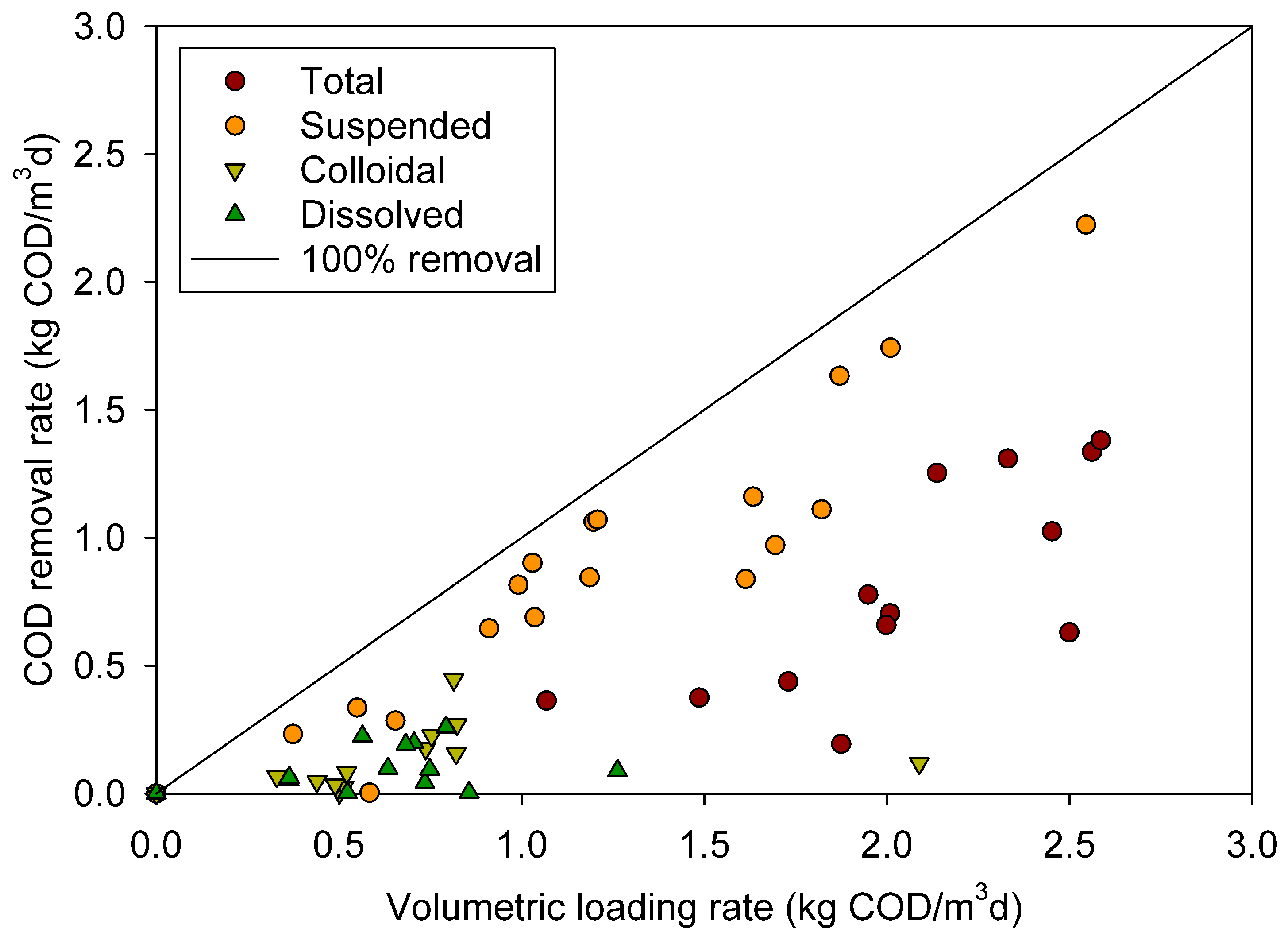
3.4. Surfactant effect

3.5. COD mass balances
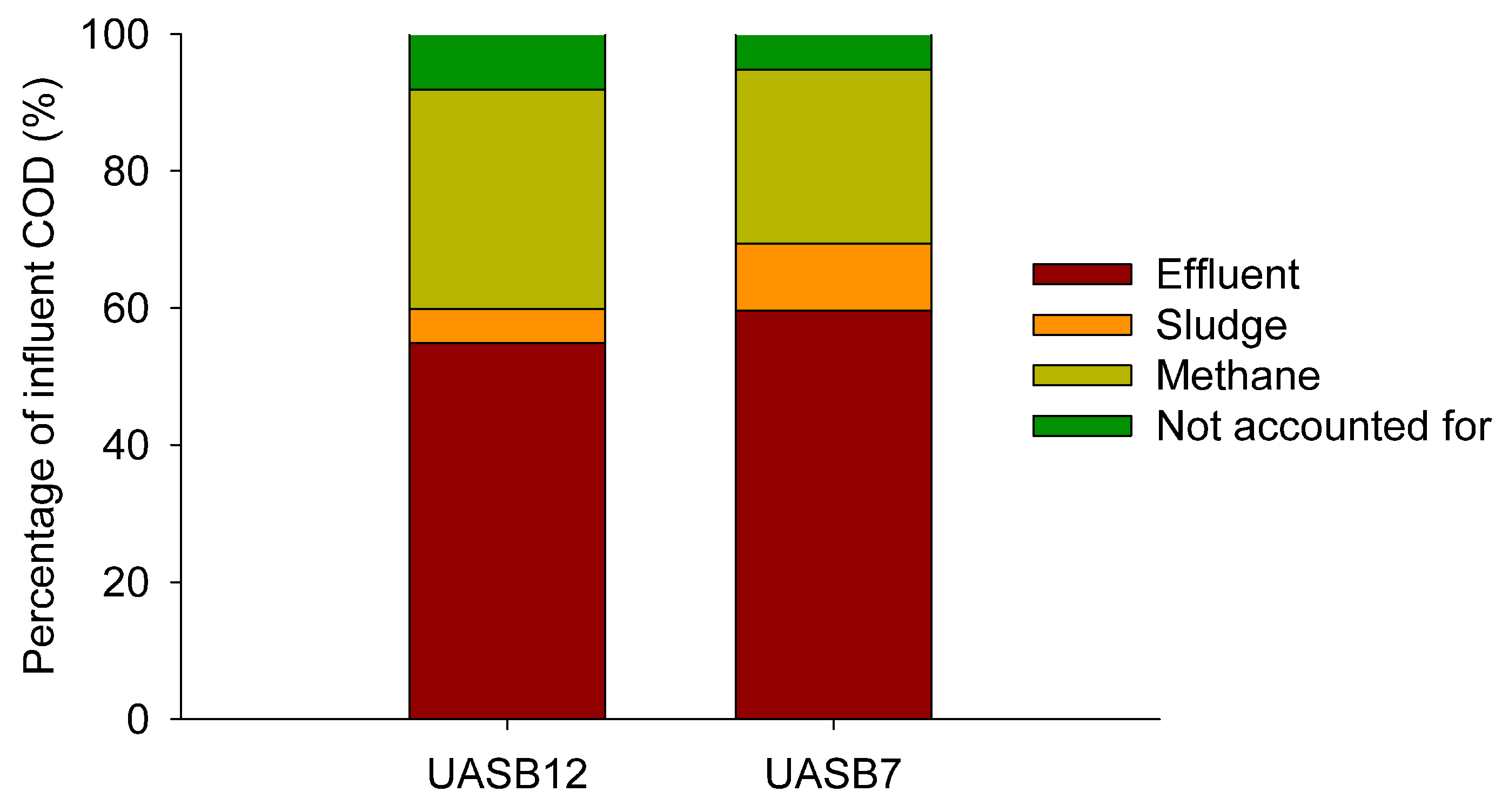
3.6. Sludge yield

3.7. Nutrient removal
| mass of N | mass of N | mass of P | mass of P | |
| removed (g) | in sludge | removed (g) | in sludge (g) | |
| SBR12 | 9.88 | 8.48 | 1.15 | 1.41 |
| SBR6 | 10.35 | 3.08 | 1.02 | 0.51 |
3.8. Energy production
4. Conclusions
- Aerobic treatment of greywater achieved 90% of COD removal and 97% removal of anionic surfactants at a HRT of 12 hours and a temperature of 32 ± 3 C.
- The low sludge yield of the aerobic SBR (0.12 g VSS/g COD) operated at 32 ± 3 C and an HRT or 12 hours makes it an attractive process for the treatment of greywater.
- Anaerobic treatment of greywater at an HRT of 12 hours and a temperature of 32 ± 3 C reaches 51% removal of COD, with a greywater methanization rate of 32% and with a poor removal (24%) of anionic surfactants.
- A combined anaerobic-aerobic system operated at 32 ± 3 C and at an HRT of 12 hours did not give an advantage, compared to aerobic treatment, in regard to the removal of COD from greywater and sludge yield. However, the benefits of this configuration depend on factors such as gas use and energy input.
- Based on COD removal, sludge yield and energy considerations, treatment of greywater in an aerobic system is preferred over an anaerobic system and a combined anaerobic-aerobic system.
Acknowledgements
References
- Eriksson, E.; Auffarth, K.; Henze, M.; Ledin, A. Characteristics of grey wastewater. Urban Water 2002, 4, 85–104. [Google Scholar] [CrossRef]
- Otterpohl, R. Options for alternative types of sewerage and treatment systems directed to improvement of the overall performance. Water Sci. Technol. 2002, 45, 149–158. [Google Scholar] [PubMed]
- Jefferson, B.; Judd, S.; Diaper, C. Treatment methods for grey water. In Decentralised Sanitation and Reuse: Concepts, Systems and Implementation; Lens, P., Zeeman, G., Lettinga, G., Eds.; IWA: Wageningen, The Netherlands, 2001; pp. 334–353. [Google Scholar]
- Seghezzo, L.; Zeeman, G.; van Lier, J.B.; Hamelers, H.V.M.; Lettinga, G. A review: The anaerobic treatment of sewage in UASB and EGSB reactors. Bioresour. Technol. 1998, 65, 175–190. [Google Scholar] [CrossRef]
- Zeeman, G.; Lettinga, G. The role of anaerobic digestion of domestic sewage in closing the water and nutrient cycle at community level. Water Sci. Technol. 1999, 39, 187–194. [Google Scholar] [CrossRef]
- Elmitwalli, T.A.; Otterpohl, R. Anaerobic biodegradability and treatment of grey water in upflow anaerobic sludge blanket (UASB) reactor. Water Res. 2007, 41, 1379–1387. [Google Scholar] [CrossRef] [PubMed]
- Abu Ghunmi, L.; Zeeman, G.; Fayyad, M.; van Lier, J.B. Anaerobic-aerobic treatment of grey water, continuous and batch operations. In Sanitation Challenge: New Sanitation Concepts and Models of Governance; Proceedings of the International IWA Conference Sanitation Challenge; Wageningen, The Netherlands, 19–21 May 2008, Van Eeckert, M., Ed.; IWA: Wageningen, The Netherlands, 2008. [Google Scholar]
- Hernandez Leal, L.; Zeeman, G.; Temmink, H.; Buisman, C. Characterisation and biological treatment of greywater. Water Sci. Technol. 2007, 56, 193–200. [Google Scholar] [CrossRef] [PubMed]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater; Clesceri, L.S., Ed.; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- Nolde, E. Greywater recycling systems in Germany—results, experiences and guidelines. Water Sci. Technol. 2005, 51, 203–210. [Google Scholar] [PubMed]
- Palmquist, H.; Hanæus, J. Hazardous substances in separately collected grey—and blackwater from ordinary Swedish households. Sci. Total Environ. 2005, 348, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Elmitwalli, T.A.; van Dun, M.; Bruning, H.; Zeeman, G.; Lettinga, G. The role of filter media in removing suspended and colloidal particles in an anaerobic reactor treating domestic sewage. Bioresour. Technol. 2000, 72, 235–242. [Google Scholar] [CrossRef]
- Ying, G.G. Fate, behavior and effects of surfactants and their degradation products in the environment. Environ. Int. 2002, 32, 417–431. [Google Scholar] [CrossRef] [PubMed]
- Garcia Morales, J.L.; Nebot, E.; Romero, L.I.; Sales, D. Comparison between acidogenic and methanogenic inhibition caused by linear alkylbenzene-sulfonate (LAS). Chem. Biochem. Eng. Q. 2001, 15, 13–19. [Google Scholar]
- Tchobanoglous, G.; Burton, F.L.; Stensel, H.D. Wastewater Engineering, Treatment and Reuse, 4th ed.; McGraw Hill: New York, NY, USA, 2003. [Google Scholar]
- Elmitwalli, T.A. Anaerobic Treatment of Domestic Sewage at Low Temperature; Wageningen University, Sub-department of Environmental Technology: Wageningen, The Netherlands, 2000; p. 3. [Google Scholar]
- Luning, L. Sludge mineralization in The Netherlands. In Proceedings of the Workshop Saving Energy in RWZI, The Netherlands, October 2006.
- Kujawa-Roeleveld, K.; Elmitwalli, T.; Zeeman, G. Enhanced primary treatment of concentrated black water and kitchen residues within DESAR concept using two types of anaerobic digesters. Water Sci. Technol. 2006, 53, 159–168. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license http://creativecommons.org/licenses/by/3.0/.
Share and Cite
Hernández Leal, L.; Temmink, H.; Zeeman, G.; Buisman, C.J.N. Comparison of Three Systems for Biological Greywater Treatment. Water 2010, 2, 155-169. https://doi.org/10.3390/w2020155
Hernández Leal L, Temmink H, Zeeman G, Buisman CJN. Comparison of Three Systems for Biological Greywater Treatment. Water. 2010; 2(2):155-169. https://doi.org/10.3390/w2020155
Chicago/Turabian StyleHernández Leal, Lucía, Hardy Temmink, Grietje Zeeman, and Cees J. N. Buisman. 2010. "Comparison of Three Systems for Biological Greywater Treatment" Water 2, no. 2: 155-169. https://doi.org/10.3390/w2020155
APA StyleHernández Leal, L., Temmink, H., Zeeman, G., & Buisman, C. J. N. (2010). Comparison of Three Systems for Biological Greywater Treatment. Water, 2(2), 155-169. https://doi.org/10.3390/w2020155





