Evaluating the Ecological Status of Fluvial Networks of Tropical Andean Catchments of Ecuador
Abstract
:1. Introduction
2. Materials and Methods
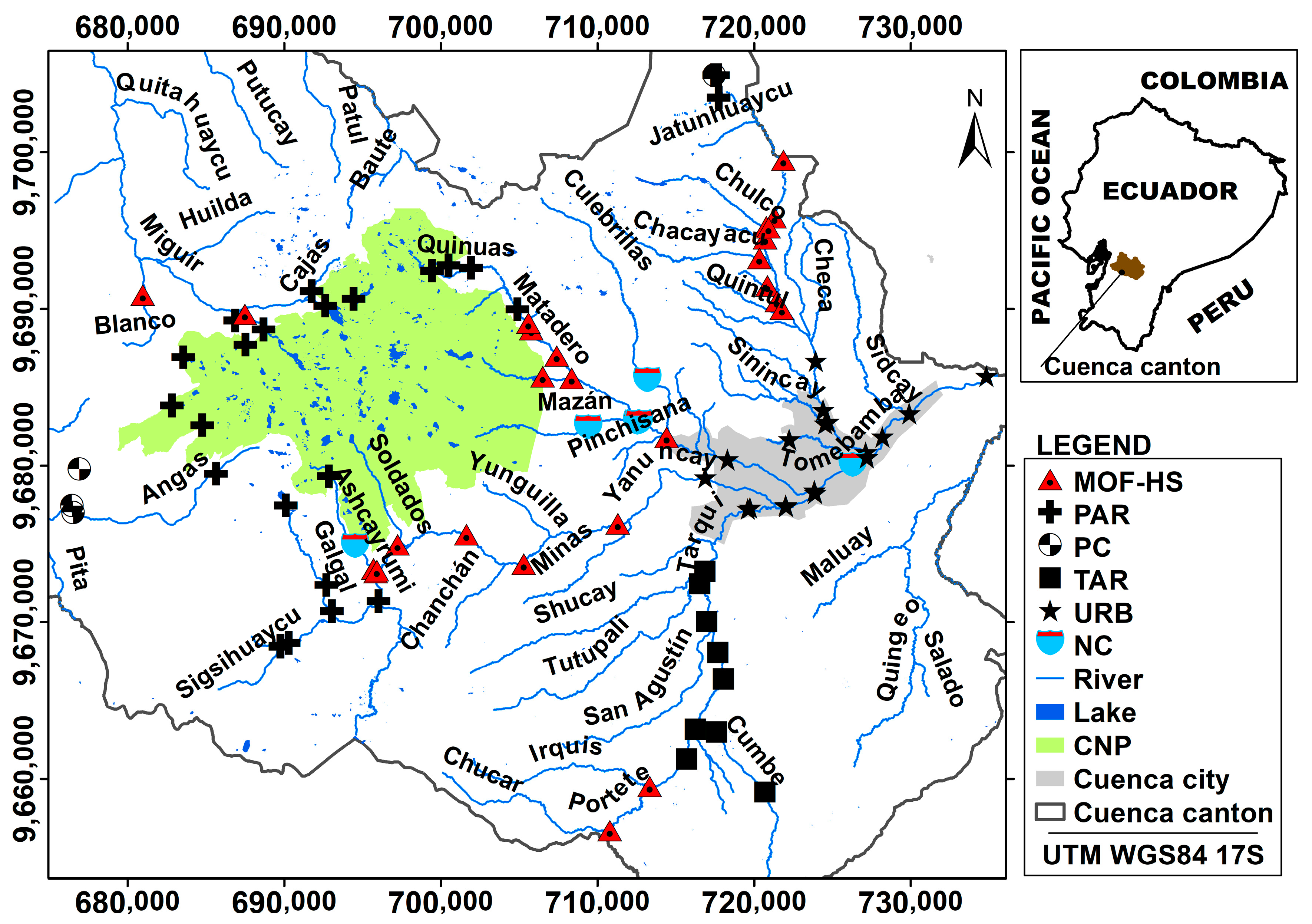
2.1. Study Area and Location of Sampling Stations
2.2. Biotic and Abiotic Monitoring
2.3. Analyzing Water Quality Parameters and Benthic Macroinvertebrates
2.4. Statistical Analyses
3. Results
3.1. Diversity of Macroinvertebrate Community at the Sampled Stations
3.2. Definition of River Typology
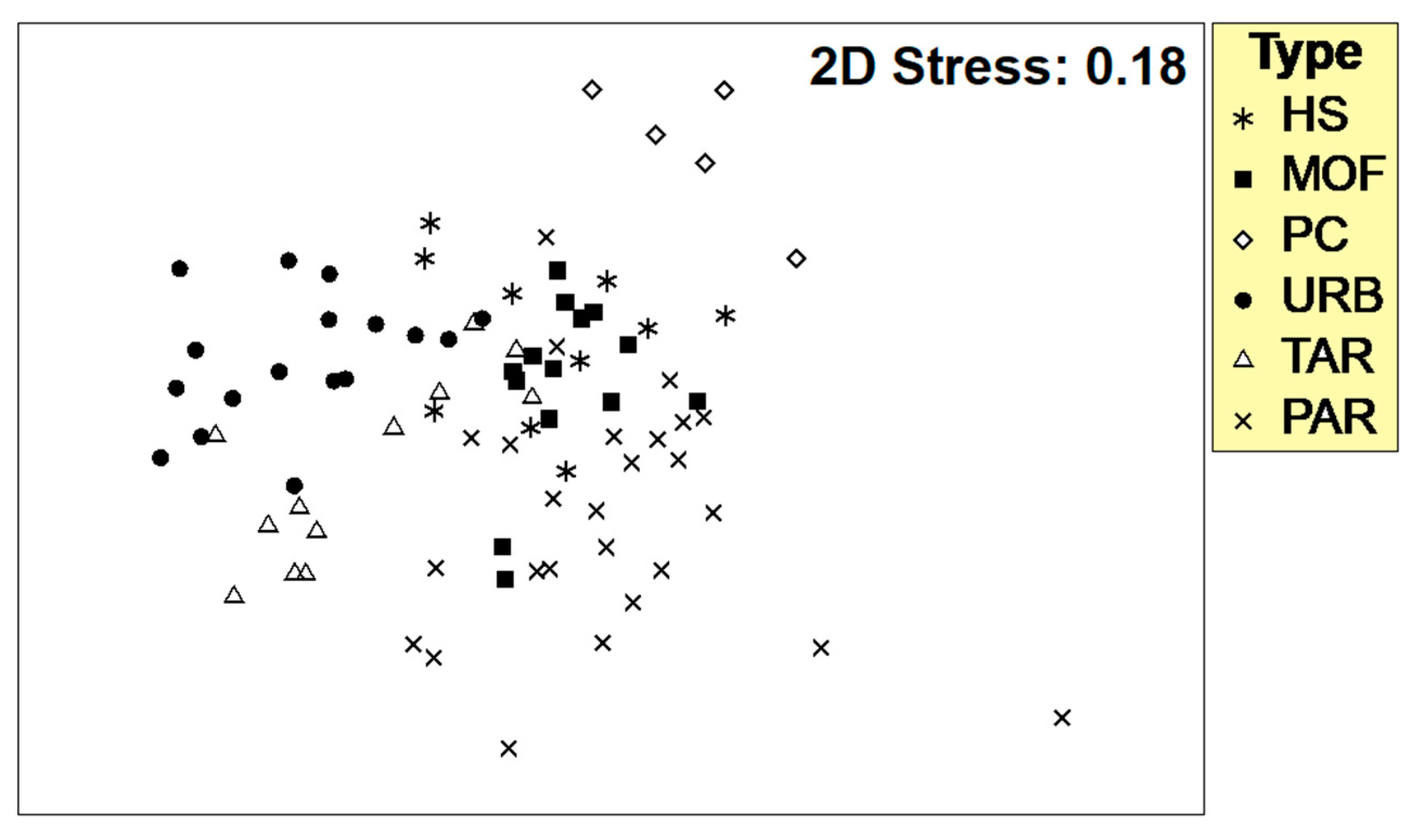
3.3. Validation of River Typology Using Macroinvertebrate Communities
3.4. Anthropogenic Pressure Gradient of the Different River Types
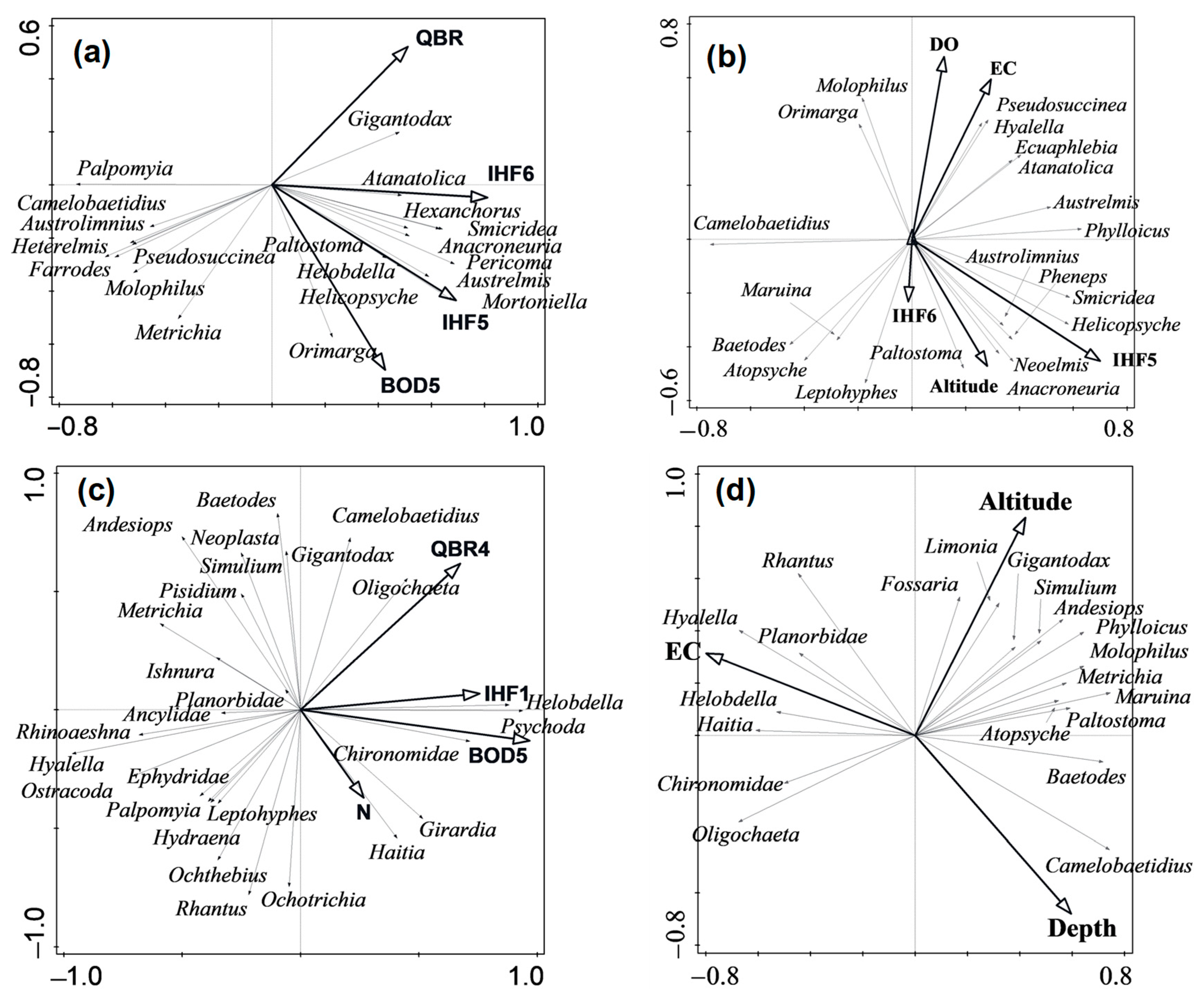
3.5. Taxa Present in the Different River Types and Identification of the Important Environmental Variables for Benthic Communities
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Viviroli, D.; Archer, D.R.; Buytaert, W.; Fowler, H.J.; Greenwood, G.B.; Hamlet, A.F.; Huang, Y.; Koboltschnig, G.; Litaor, M.I.; López-Moreno, J.I.; et al. Climate change and mountain water resources: Overview and recommendations for research, management and policy. Hydrol. Earth Syst. Sci. 2011, 15, 471–504. [Google Scholar] [CrossRef]
- Sotomayor, G.; Hampel, H.; Vázquez, R.F. Water quality assessment with emphasis in parameter optimisation using pattern recognition methods and genetic algorithm. Water Res. 2018, 130, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Arumí, J.L.; Muñoz, E.; Oyarzún, R. Andean Mountain Groundwater, Drinking Water Sources, and Vulnerability: A Case Study in Central Chile. In Groundwater; Modreck, G., Ed.; IntechOpen: Rijeka, Croatia, 2019. [Google Scholar]
- Grayson, R.; Blöschl, G. Spatial Patterns in Catchment Hydrology Observations and Modelling; Cambridge University Press: Cambridge, UK, 2000; p. 404. [Google Scholar]
- Mosquera, P.V.; Hampel, H.; Vázquez, R.F.; Alonso, M.; Catalan, J. Abundance and morphometry changes across the high-mountain lake-size gradient in the tropical Andes of Southern Ecuador. Water Resour. Res. 2017, 53, 7269–7280. [Google Scholar] [CrossRef]
- Minaya, V.; Camacho Suarez, V.; Wenninger, J.; Mynett, A. Runoff generation from a combined glacier and páramo catchment within the Antisana Reserve in Ecuador. J. Ecohydraul. 2021. [Google Scholar] [CrossRef]
- Mena-Vásconez, P.; Hofstede, R. Los Páramos Ecuatorianos. In Botánica Económica de los Andes Centrales; Moraes, M.R., Øllgaard, B., Kvist, L.P., Borchsenius, F., Balslev, H., Eds.; Universidad Mayor de San Andres: La Paz, Bolivia, 2006; pp. 91–109. [Google Scholar]
- Peters, M.K.; Hemp, A.; Appelhans, T.; Becker, J.N.; Behler, C.; Classen, A.; Detsch, F.; Ensslin, A.; Ferger, S.W.; Frederiksen, S.B.; et al. Climate–land-use interactions shape tropical mountain biodiversity and ecosystem functions. Nature 2019, 568, 88–92. [Google Scholar] [CrossRef]
- Baudino, O.M.; Suero, E.A.; Augusto, M.; Gimenez, M.E.; Flores, N. Monitoring Organochlorine Pesticides in Surface and Ground Water in San Juan Argentina. J. Chil. Chem. Soc. 2003, 48, 7–12. [Google Scholar] [CrossRef]
- Kim, M.; Boithias, L.; Cho, K.H.; Silvera, N.; Thammahacksa, C.; Latsachack, K.; Rochelle-Newall, E.; Sengtaheuanghoung, O.; Pierret, A.; Pachepsky, Y.A.; et al. Hydrological modeling of Fecal Indicator Bacteria in a tropical mountain catchment. Water Res. 2017, 119, 102–113. [Google Scholar] [CrossRef]
- Sotomayor, G.; Hampel, H.; Vázquez, R.F.; Forio, M.A.E.; Goethals, P.L.M. Implications of macroinvertebrate taxonomic resolution for freshwater assessments using functional traits: The Paute River Basin (Ecuador) case. Divers. Distrib. 2022, 28, 1735–1747. [Google Scholar] [CrossRef]
- Zhao, C.; Pan, T.; Dou, T.; Liu, J.; Liu, C.; Ge, Y.; Zhang, Y.; Yu, X.; Mitrovic, S.; Lim, R. Making global river ecosystem health assessments objective, quantitative and comparable. Sci. Total Environ. 2019, 667, 500–510. [Google Scholar] [CrossRef]
- Munné, A.; Prat, N.; Solà, C.; Bonada, N.; Rieradevall, M. A simple field method for assessing the ecological quality of riparian habitat in rivers and streams: QBR index. Aquat. Conserv. 2003, 13, 147–163. [Google Scholar] [CrossRef]
- Acosta, R.; Ríos-Touma, B.; Rieradevall, M.; Prat, N. Propuesta de un protocolo de evaluación de la calidad ecológica de ríos Andinos (C.E.R.A) y su aplicación en dos cuencas en Ecuador y Perú. Limnetica 2009, 28, 35–64. [Google Scholar] [CrossRef]
- Pardo, I.; Álvarez, M.; Moreno, J.L.; Vivas, S.; Bonada, N.; Alba-Tercedor, J.; Jaimez-Cuellar, P.; Moya, G.; Prat, N.; Robles, N.; et al. El hábitat de los ríos mediterráneos. Diseño de un índice de diversidad de hábitat. Limnetica 2002, 21, 115–134. [Google Scholar] [CrossRef]
- Arias, P.A.; Bellouin, N.; Coppola, E.; Jones, R.G.; Krinner, G.; Marotzke, J.; Naik, V.; Palmer, M.D.; Plattner, G.K.; Rogelj, J.; et al. Technical Summary. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Masson-Delmotte, V., Zhai, P., Pirani, A., Connors, S.L., Péan, C., Berger, S., Caud, N., Chen, Y., Goldfarb, L., Gomis, M.I., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2021; pp. 33–144. [Google Scholar]
- Mosquera, P.V.; Hampel, H.; Vázquez, R.F.; Catalan, J. Water chemistry variation in tropical high-mountain lakes on old volcanic bedrocks. Limnol. Oceanogr. 2022, 67, 1522–1536. [Google Scholar] [CrossRef]
- Van Colen, W.; Mosquera, P.V.; Hampel, H.; Muylaert, K. Link between cattle and the trophic status of tropical high mountain lakes in páramo grasslands in Ecuador. Lakes Reserv. 2018, 23, 303–311. [Google Scholar] [CrossRef]
- ETAPA-EP. Diseños Definitivos de la Planta de Tratamiento de Aguas Residuales de Guangarcucho, Cantón Cuenca, Provincia del Azuay, Ecuador, Anexo 1: Informe de Condiciones de Tratabilidad; Empresa Pública Municipal de Telecomunicaciones, Agua Potable, Alcantarillado y Saneamiento ETAPA-EP: Cuenca, Ecuador, 2017; p. 40. [Google Scholar]
- G&H; ACSAN. Determinación del Caudal de Aguas no Residuales que Ingresa al Sistema de Saneamiento y Propuestas Para la Aplicación de Medidas Correctivas. Greeley and Hansen (G&H), ACSAN Consultores; Empresa Pública Municipal de Telecomunicaciones, Agua Potable, Alcantarillado y Saneamiento ETAPA-EP: Cuenca, Ecuador, 2023; p. 59. [Google Scholar]
- Ríos-Touma, B.; Acosta, R.; Prat, N. The Andean Biotic Index (ABI): Revised tolerance to pollution values for macroinvertebrate families and index performance evaluation. Rev. Biol. Trop. 2014, 62, 249–273. [Google Scholar] [CrossRef]
- Prat, N.; Ríos-Touma, B.; Acosta, R.; Rieradevall, M. Los macroinvertebrados como indicadores de la calidad del agua. In Macroinvertebrados Bentónicos Sudamericanos: Sistemática y Biología; Domínguez, E., Fernández, H.R., Eds.; Fundación Miguel Lillo: Tucumán, Argentine, 2009; pp. 631–654. [Google Scholar]
- EU-Parliament. Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 Establishing a Framework for Community Action in the Field of Water Policy; European Parliament: Brussels, Belgium, 2000. [Google Scholar]
- Segnini, S. El uso de los macroinvertebrados bentónicos como indicadores de la condición ecológica de los cuerpos de agua corriente. Ecotropicos 2003, 16, 45–63. [Google Scholar]
- Alvites, C.; Battipaglia, G.; Santopuoli, G.; Hampel, H.; Vázquez, R.F.; Matteucci, G.; Tognetti, R.; De Micco, V. Dendrochronological analysis and growth patterns of Polylepis reticulata (Rosaceae) in the Ecuadorian Andes. IAWA J. 2019, 40, S331–S335. [Google Scholar] [CrossRef]
- Boiten, W. Hydrometry, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2008; p. 244. [Google Scholar]
- Strahler, A.N. Quantitative analysis of watershed geomorphology. Eos Trans. Am. Geophys. Union 1957, 38, 913–920. [Google Scholar] [CrossRef]
- Corral, L.R.; Montiel Olea, C.E. What Drives Take-up in Land Regularization: Ecuador’s Rural Land Regularization and Administration Program, Sigtierras. J. Econ. Race Policy 2020, 3, 60–75. [Google Scholar] [CrossRef]
- Sotomayor, G.; Romero, J.; Ballari, D.; Vázquez, R.F.; Ramírez-Morales, I.; Hampel, H.; Galarza, X.; Montesinos, B.; Forio, M.A.E.; Goethals, P.L.M. Occurrence Prediction of Riffle Beetles (Coleoptera: Elmidae) in a Tropical Andean Basin of Ecuador Using Species Distribution Models. Biology 2023, 12, 473. [Google Scholar] [CrossRef]
- Manzo, V. Key to the South America genera of Elmidae (Insecta: Coleoptera) with distributional data. Stud. Neotrop. Fauna Environ. 2005, 40, 201–208. [Google Scholar] [CrossRef]
- Domínguez, E.; Molineri, C.; Pescador, M.L.; Hubbard, M.D.; Nieto, M.C. Ephemeroptera of South America; Pensoft Publishers: Moscow, Russia, 2006; p. 646. [Google Scholar]
- Manzo, V.; Archangelsky, M. A key to the known larvae of South American Elmidae (Coleoptera: Byrrhoidea), with a description of the mature larva of Macrelmis saltensis Manzo. Ann. Limnol. Int. J. Lim. 2008, 44, 63–74. [Google Scholar] [CrossRef]
- Domínguez, E.; Fernández, H.R. Macroinvertebrados Bentónicos Sudamericanos: Sistemática y Biología; Fundación Miguel Lillo: Tucumán, Argentina, 2009; p. 656. [Google Scholar]
- APHA-AWWA-WEF. Standard Methods for Examination of Water and Wastewater, 23rd ed.; Baird, R.B., Eaton, A.D., Rice, E.W., Eds.; American Public Health Association (APHA), American Water Works Association (AWWA) and Water Environment Federation (WEF): Washington DC, USA, 2017. [Google Scholar]
- Legendre, P.; Legendre, L. Numerical Ecology, 2nd ed.; Elsevier Science B.V.: Amsterdam, The Netherlands, 1998; Volume 24, p. 852. [Google Scholar]
- Munné, A.; Prat, N. Defining river types in a Mediterranean area: A methodology for the implementation of the EU Water Framework Directive. Environ. Manag. 2004, 34, 711–729. [Google Scholar] [CrossRef] [PubMed]
- Sotomayor, G.; Hampel, H.; Vázquez, R.F.; Goethals, P.L.M. Multivariate-statistics based selection of a benthic macroinvertebrate index for assessing water quality in the Paute River basin (Ecuador). Ecol. Indic. 2020, 111, 106037. [Google Scholar] [CrossRef]
- Jain, A.K.; Dubes, R.C. Algorithms for Clustering Data; Prentice-Hall: Englewood Cliffs, NJ, USA, 1988; p. 320. [Google Scholar]
- Clarke, K.R.; Gorley, R.N.; Somerfield, P.J.; Warwick, R.M. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation, 3rd ed.; PRIMER-E: Plymouth, UK, 2014. [Google Scholar]
- Quinn, G.P.; Keough, M.J. Experimental Design and Data Analysis for Biologists; Cambridge University Press: Cambridge, UK, 2002; p. 537. [Google Scholar]
- Ter Braak, C.J.F.; Smilauer, P. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination; Version 4.5; Microcomputer Power: Ithaca, NY, USA, 2002. [Google Scholar]
- Bradley, R.S.; Vuille, M.; Diaz, H.F.; Vergara, W. Threats to Water Supplies in the Tropical Andes. Science 2006, 312, 1755–1756. [Google Scholar] [CrossRef]
- Luo, Z.; Zuo, Q.; Shao, Q. A new framework for assessing river ecosystem health with consideration of human service demand. Sci. Total Environ. 2018, 640–641, 442–453. [Google Scholar] [CrossRef]
- Tadaki, M.; Brierley, G.; Cullum, C. River classification: Theory, practice, politics. WIREs Water 2014, 1, 349–367. [Google Scholar] [CrossRef]
- Rinaldi, M.; Surian, N.; Comiti, F.; Bussettini, M. A method for the assessment and analysis of the hydromorphological condition of Italian streams: The Morphological Quality Index (MQI). Geomorphology 2013, 180–181, 96–108. [Google Scholar] [CrossRef]
- Rinaldi, M.; Gurnell, A.M.; del Tánago, M.G.; Bussettini, M.; Hendriks, D. Classification of river morphology and hydrology to support management and restoration. Aquat. Sci. 2016, 78, 17–33. [Google Scholar] [CrossRef]
- Olden, J.D.; Kennard, M.J.; Pusey, B.J. A framework for hydrologic classification with a review of methodologies and applications in ecohydrology. Ecohydrology 2012, 5, 503–518. [Google Scholar] [CrossRef]
- Orr, H.G.; Large, A.R.G.; Newson, M.D.; Walsh, C.L. A predictive typology for characterising hydromorphology. Geomorphology 2008, 100, 32–40. [Google Scholar] [CrossRef]
- Villamarín, C.; Rieradevall, M.; Paul, M.J.; Barbour, M.T.; Prat, N. A tool to assess the ecological condition of tropical high Andean streams in Ecuador and Peru: The IMEERA index. Ecol. Indic. 2013, 29, 79–92. [Google Scholar] [CrossRef]
- Álvarez-Cabria, M.; Barquín, J.; Antonio Juanes, J. Spatial and seasonal variability of macroinvertebrate metrics: Do macroinvertebrate communities track river health? Ecol. Indic. 2010, 10, 370–379. [Google Scholar] [CrossRef]
- Liu, W.; Xu, M.; Zhao, N.; Zhou, X.; Pan, B.; Tian, S.; Lei, F. River health assessment of the Yellow River source region, Qinghai-Tibetan Plateau, China, based on tolerance values of macroinvertebrates. Environ. Sci. Pollut. Res. Int. 2019, 26, 10251–10262. [Google Scholar] [CrossRef]
- Herman, M.R.; Nejadhashemi, A.P. A review of macroinvertebrate- and fish-based stream health indices. Int. J. Ecohydrol. Hydrobiol. 2015, 15, 53–67. [Google Scholar] [CrossRef]
- Rahman, A.; Jahanara, I.; Jolly, Y.N. Assessment of physicochemical properties of water and their seasonal variation in an urban river in Bangladesh. Water Sci. Eng. 2021, 14, 139–148. [Google Scholar] [CrossRef]
- Nasrabadi, T.; Ruegner, H.; Sirdari, Z.Z.; Schwientek, M.; Grathwohl, P. Using total suspended solids (TSS) and turbidity as proxies for evaluation of metal transport in river water. Appl. Geochem. 2016, 68, 1–9. [Google Scholar] [CrossRef]
- Wilson, J.L.; Everard, M. Real-time consequences of riparian cattle trampling for mobilization of sediment, nutrients and bacteria in a British lowland river. Int. J. River Basin Manag. 2018, 16, 231–244. [Google Scholar] [CrossRef]
- Reder, K.; Flörke, M.; Alcamo, J. Modeling historical fecal coliform loadings to large European rivers and resulting in-stream concentrations. Environ. Model. Softw. 2015, 63, 251–263. [Google Scholar] [CrossRef]
- Jayakody, P.; Parajuli, P.B.; Brooks, J.P. Evaluating Spatial and Temporal Variability of Fecal Coliform Bacteria Loads at the Pelahatchie Watershed in Mississippi. Hum. Ecol. Risk Assess. 2014, 20, 1023–1041. [Google Scholar] [CrossRef]
- Hiraishi, A.; Saheki, K.; Horie, S. Relationships of Total Coliform, Fecal Coliform, and Organic Pollution Levels in the Tamagawa River. Nippon Suisan Gakk. 1984, 50, 991–997. [Google Scholar] [CrossRef]
- Fernández de Córdova, J.; González, H. Evolución de la Calidad del Agua de los Tramos Bajos de los Ríos de la Ciudad de Cuenca; ETAPA-EP: Cuenca, Ecuador, 2013; p. 99. [Google Scholar]
- Mora-Campos, M.R.; Tamay-Heras, A.A. Determinación Del íNdice de Calidad de Agua Mediante El Monitoreo de Macro Invertebrados, Parámetros Fisicoquímicos Y Microbiológicos en El Río Sinincay, Cuenca-Ecuador; Universidad Politécnica Salesiana Cuenca: Cuenca, Ecuador, 2022. [Google Scholar]
- Rowiński, P.M.; Västilä, K.; Aberle, J.; Järvelä, J.; Kalinowska, M.B. How vegetation can aid in coping with river management challenges: A brief review. Int. J. Ecohydrol. Hydrobiol. 2018, 18, 345–354. [Google Scholar] [CrossRef]
- Osterkamp, W.R.; Hupp, C.R.; Stoffel, M. The interactions between vegetation and erosion: New directions for research at the interface of ecology and geomorphology. Earth Surf. Process. Landf. 2012, 37, 23–36. [Google Scholar] [CrossRef]
- Rutherford, J.C.; Marsh, N.A.; Davies, P.M.; Bunn, S.E. Effects of patchy shade on stream water temperature: How quickly do small streams heat and cool? Mar. Freshw. Res. 2004, 55, 737–748. [Google Scholar] [CrossRef]
- Studholme, A.M.; Hampel, H.; Finn, D.S.; Vázquez, R.F. Secondary production of caddisflies reflects environmental heterogeneity among tropical Andean streams. Hydrobiologia 2017, 797, 231–246. [Google Scholar] [CrossRef]
- Gutiérrez-Fonseca, P.E.; Springer, M. Description of the final instar nymphs of seven species from Anacroneuria Klapálek (Plecoptera: Perlidae) in Costa Rica, and first record for an additional genus in Central America. Zootaxa 2011, 2965, 16–38. [Google Scholar] [CrossRef]
- Palatov, D.M.; Chertoprud, M.V.; Frolov, A.A. Fauna and types of soft-bottom macrozoobenthic assemblages in watercourses of mountainous regions on the eastern Black Sea coast. Inland. Water. Biol. 2016, 9, 150–159. [Google Scholar] [CrossRef]
- Vázquez, R.F.; Vimos-Lojano, D.; Hampel, H. Habitat Suitability Curves for Freshwater Macroinvertebrates of Tropical Andean Rivers. Water 2020, 12, 2703. [Google Scholar] [CrossRef]
- Ward, J.V. Ecology of alpine streams. Freshw. Biol. 1994, 32, 277–294. [Google Scholar] [CrossRef]
- Sajamí-Reymundo, J.I.; Huamantinco-Araujo, A.A. Distribución espacial de Ephemeroptera, Plecoptera, Trichoptera y Coleoptera (Insecta) en una quebrada de primer orden, bosque montano, Junín, Perú. Rev. Peru. Biol. 2016, 23, 95–102. [Google Scholar] [CrossRef]
- Braccia, A.; Voshell, J.R. Benthic Macroinvertebrate Fauna in Small Streams Used by Cattle in the Blue Ridge Mountains, Virginia. Northeast. Nat. 2006, 13, 269–286. [Google Scholar] [CrossRef]
- Sokol, E.R.; Hoch, J.M.; Gaiser, E.; Trexler, J.C. Metacommunity Structure Along Resource and Disturbance Gradients in Everglades Wetlands. Wetlands 2014, 34, 135–146. [Google Scholar] [CrossRef]
- Scheibler, E.E.; Claps, M.C.; Roig-Juñent, S.A. Temporal and altitudinal variations in benthic macroinvertebrate assemblages in an Andean river basin of Argentina. J. Limnol. 2014, 73. [Google Scholar] [CrossRef]
- Finn, D.S.; Encalada, A.C.; Hampel, H. Genetic isolation among mountains but not between stream types in a tropical high-altitude mayfly. Freshw. Biol. 2016, 61, 702–714. [Google Scholar] [CrossRef]
- Iñiguez-Armijos, C.; Hampel, H.; Breuer, L. Land-use effects on structural and functional composition of benthic and leaf-associated macroinvertebrates in four Andean streams. Aquat. Ecol. 2018, 52, 77–92. [Google Scholar] [CrossRef]
- Ríos-Touma, B.; Holzenthal, R.W.; Rázuri-Gonzales, E.; Heckenhauer, J.; Pauls, S.U.; Storer, C.G.; Frandsen, P.B. De Novo Genome Assembly and Annotation of an Andean Caddisfly, Atopsyche davidsoni Sykora, 1991, a Model for Genome Research of High-Elevation Adaptations. Genome Biol. Evol. 2021, 14, evab286. [Google Scholar] [CrossRef]
- Rosa, B.J.F.V.; Rodrigues, L.F.T.; de Oliveira, G.S.; da Gama Alves, R. Chironomidae and Oligochaeta for water quality evaluation in an urban river in southeastern Brazil. Environ. Monit. Assess. 2014, 186, 7771–7779. [Google Scholar] [CrossRef]
- Scheibler, E.E.; Roig-Juñent, S.A.; Claps, M.C. Chironomid (Insecta: Diptera) assemblages along an Andean altitudinal gradient. Aquat. Biol. 2014, 20, 169–184. [Google Scholar] [CrossRef]
| Components of River Habitat Index (IHF) | Definition | Explanation of Definition | Ecological Meaning of Score | Maximum Score |
|---|---|---|---|---|
| IHF1 | Substrate inclusion and limitation | The amount of compacted sand present between the larger blocks in the rapid zones | Compacted sand limits the colonization of macroinvertebrates; hence, lack of it represents a higher score | 10 |
| IHF2 | Frequency of riffles | Indicates the frequency of riffles in the riverbed | Riffles are particularly suitable habitats for macroinvertebrates; hence, their presence increases the score value | 10 |
| IHF3 | Substrate composition | Evaluates the composition of the mineral substrate in the riverbed | Macroinvertebrates benefit from the diverse substrate; hence, the assigned score is higher when more types are present | 20 |
| IHF4 | Speed/depth regimens | Evaluates the presence of four combinations of velocity (fast or slow) and depth (shallow or deep) of the water column | Maximum score is obtained when all the four combinations of velocity and depth are present in the river section | 10 |
| IHF5 | Shade on the riverbed | Assesses the coverage of shadow on the riverbed | When shadow is present with some sunny openings, the assigned score is the highest | 10 |
| IHF6 | Riverbed heterogeneity | Estimates the presence of leaf litter, exposed roots, trunks/branches, and natural dams | Presence of natural elements favors macroinvertebrates by creating physically more complex habitats; hence, the assigned score is higher | 10 |
| IHF7 | Aquatic vegetation cover | Estimates the presence of different types of aquatic vegetation (bryophytes, benthic stream algae, filamentous algae, macrophytes) | Presence of aquatic vegetation provide food sources and an increased surface for colonization; hence, the assigned score is higher | 30 |
| Components of Riparian Vegetation Quality Index (QBR) | Definition | Explanation of Definition | Explanation of the Score | Maximum Score |
|---|---|---|---|---|
| QBR1 | Coverage of the riparian zone | Evaluates the degree of vegetation cover on each of the riverbanks | Larger areas covered by vegetation on the riverbanks represent higher scores | 25 |
| QBR2 | Vegetation structure of the riparian zone | Assesses the type of species that form the riparian cover and the natural stratification of the plant community | Presence of trees and more developed vegetation stratification receive higher scores | 25 |
| QBR3 | Quality of the riparian cover | Evaluates whether the tree species are native or introduced and whether anthropogenic activities are present | Native vegetation receives the highest score, and presence of anthropogenic activities reduces the score | 25 |
| QBR4 | Degree of naturalness of the river channel | Evaluates if the river channel is natural or to what degree it has been modified | The natural river receives the maximum score, and the different channel modifications reduce the score | 25 |
| River Type | R Statistic | River Type | R Statistic |
|---|---|---|---|
| PAR/PC | 0.65 | PC/MOF | 0.76 |
| PAR/URB | 0.62 | HS/TAR | 0.73 |
| PAR/TAR | 0.51 | HS/URB | 0.60 |
| PAR/HS | 0.20 | HS/MOF | 0.05 |
| PAR/MOF | 0.08 | MOF/TAR | 0.88 |
| PC/TAR | 1.00 | MOF/URB | 0.71 |
| PC/URB | 0.95 | URB/TAR | 0.57 |
| PC/HS | 0.83 |
| PAR | MOF-HS | TAR | URB | |||||
|---|---|---|---|---|---|---|---|---|
| Environmental Variables | Mean | STD | Mean | STD | Mean | STD | Mean | STD |
| Temperature (°C) | 8.3 | 2.1 | 10.7 | 1.9 | 13.8 | 1.2 | 14.5 | 2.4 |
| Conductivity (µS cm−1) | 70.5 | 28.2 | 81.1 | 25.8 | 131.4 | 76.5 | 127.5 | 46.3 |
| Dissolved oxygen (mg L−1) | 8.0 | 0.4 | 8.1 | 0.4 | 7.2 | 1.4 | 7.5 | 0.5 |
| Oxygen saturation (%) | 104.9 | 3.0 | 102.2 | 9.4 | 93.6 | 17.3 | 98.5 | 4.1 |
| pH | 7.8 | 0.4 | 7.6 | 0.4 | 7.7 | 0.2 | 7.9 | 0.1 |
| Total dissolved solids (mg L−1) | 73.6 | 20.2 | 80.3 | 16.3 | 133.9 | 60.4 | 125.5 | 38.3 |
| BOD5 (mg L−1) | 0.7 | 0.5 | 0.6 | 0.3 | 2.2 | 1.6 | 5.9 | 5.4 |
| Nitrates (mg L−1) | 12.3 | 11.9 | 20.3 | 18.4 | 0.2 | 0.2 | 1.9 | 6.9 |
| Total phosphorus (mg L−1) | 8.1 | 5.4 | 12.7 | 11.1 | 0.2 | 0.1 | 0.9 | 2.7 |
| Turbidity (NTU) | 1.5 | 0.9 | 3.2 | 2.4 | 8.8 | 2.9 | 12.8 | 11.8 |
| Fecal coliforms (MPN (100 mL)−1) | 49.0 | 68.8 | 1317.9 | 3428.4 | 187,073.8 | 241,871.0 | 423,954.9 | 893,572.2 |
| IHF1 | 6.7 | 4.2 | 7.1 | 2.9 | 2.9 | 3.5 | 3.8 | 3.0 |
| IHF2 | 9.6 | 1.3 | 9.7 | 0.9 | 6.5 | 2.1 | 9.4 | 1.2 |
| IHF3 | 10.4 | 3.5 | 8.8 | 2.0 | 6.4 | 3.5 | 6.6 | 3.1 |
| IHF4 | 8.3 | 1.7 | 9.1 | 1.3 | 8.3 | 3.6 | 7.5 | 1.5 |
| IHF5 | 4.8 | 2.8 | 5.3 | 2.4 | 3.3 | 0.7 | 3.9 | 1.7 |
| IHF6 | 6.8 | 2.5 | 7.3 | 1.7 | 4.3 | 2.0 | 6.0 | 1.9 |
| IHF7 | 13.7 | 5.8 | 12.0 | 5.3 | 7.5 | 4.6 | 3.8 | 4.9 |
| IHF | 60.3 | 8.2 | 58.9 | 7.0 | 39.0 | 4.7 | 41.1 | 7.9 |
| QBR1 | 19.2 | 9.2 | 6.4 | 10.0 | 0.0 | 0.0 | 0.3 | 1.2 |
| QBR3 | 20.4 | 8.9 | 9.2 | 10.4 | 0.0 | 0.0 | 0.7 | 2.1 |
| QBR4 | 24.0 | 3.2 | 14.5 | 10.4 | 1.3 | 3.5 | 0.3 | 1.2 |
| QBR | 82.4 | 25.2 | 18.4 | 6.5 | 10.0 | 8.0 | 8.8 | 8.8 |
| Parameter | River Type | |||||||
|---|---|---|---|---|---|---|---|---|
| Páramo | Mountain Forest and Humid Shrub | Tarqui | Urban | |||||
| Axis 1 | Axis 2 | Axis 1 | Axis 2 | Axis 1 | Axis 2 | Axis 1 | Axis 2 | |
| Conductivity | 0.11 | −0.46 | 0.47 | 0.28 | - | - | 0.59 | 0.11 |
| BOD5 | 0.31 | 0.39 | −0.48 | −0.05 | - | - | - | - |
| N | 0.39 | −0.38 | 0.43 | 0.34 | 0.25 | −0.43 | −0.19 | 0.42 |
| P | −0.05 | 0.14 | −0.06 | 0.39 | 0.38 | 0.29 | - | - |
| Turbidity | −0.07 | 0.15 | −0.29 | 0.45 | 0.48 | −0.07 | 0.55 | −0.15 |
| FC | 0.51 | −0.15 | −0.31 | −0.11 | 0.03 | 0.77 | - | - |
| IHF1 | 0.21 | 0.50 | - | - | - | - | - | - |
| IHF | 0.32 | 0.22 | 0.10 | −0.50 | 0.33 | −0.28 | −0.54 | 0.06 |
| QBR4 | −0.41 | −0.24 | 0.28 | −0.03 | 0.49 | −0.00 | - | - |
| QBR | −0.41 | 0.25 | 0.30 | −0.43 | 0.45 | 0.23 | 0.14 | 0.89 |
| Class | Family | Genus | PAR | MOF-HS | TAR | URB | PC | Class | Family | Genus | PAR | MOF-HS | TAR | URB | PC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Arachnida | Clade: Hydracarina | x | x | x | x | Insecta | Hydraenidae | Hydraena | x | x | |||||
| Bivalvia | Sphaeriidae | Pisidium | x | x | x | x | Ochthebius | x | x | x | |||||
| Clitellata | Glossophonidae | Helobdella | x | x | x | x | x | Hydrobiosidae | Atopsyche | x | x | x | x | ||
| Subclass: Oligochaeta | x | x | x | x | x | Cailloma | x | x | |||||||
| Gastropoda | Ancylidae | x | x | Hydrophilidae | Enochrus | x | |||||||||
| Lymnaeidae | Fossaria | x | x | x | Tropisternus | x | |||||||||
| Pseudosuccinea | x | x | x | Hydropsychidae | Leptonema | x | |||||||||
| Physidae | Haitia | x | x | x | x | x | Smicridea | x | x | x | |||||
| Planorbidae | x | x | x | Hydroptilidae | Leucotrichia | x | x | x | x | ||||||
| Insecta | Aeshnidae | Rhinoaeshna | x | x | x | Metrichia | x | x | x | x | |||||
| Anomalopsychidae | Contulma | x | x | x | Neotrichia | x | x | x | |||||||
| Baetidae | Andesiops | x | x | x | x | x | Ochotrichia | x | x | x | x | ||||
| Blephariceridae | Baetodes | x | x | x | x | x | Oxyethira | x | x | ||||||
| Camelobaetidius | x | x | x | x | x | Leptoceridae | Grumichella | x | |||||||
| Limonicola | x | x | x | Nectopsyche | x | x | x | ||||||||
| Mayobaetis | x | x | Leptohyphidae | Ecuaphlebia | x | x | x | ||||||||
| Nanomis | x | x | Farrodes | x | x | x | |||||||||
| Paltostoma | x | x | x | x | Leptohyphes | x | x | x | x | x | |||||
| Prebaetodes | x | x | Tricorythodes | x | x | x | |||||||||
| Calamoceratidae | Phylloicus | x | x | x | x | Thraulodes | x | x | x | ||||||
| Ceratopogonidae | Bezzia | x | x | Limnephilidae | Anomalocosmoecus | x | x | ||||||||
| Palpomyia | x | x | x | x | x | Limoniidae | Hexatoma | x | x | x | |||||
| Chironomidae | x | x | x | x | x | Tribe: Hexatomini | x | x | x | x | |||||
| Coenagrionidae | Ishnura | x | x | x | Limonia | x | x | x | x | ||||||
| Corydalidae | Corydalus | x | x | Molophilus | x | x | x | x | |||||||
| Dixidae | Dixa | x | Orimarga | x | x | x | |||||||||
| Dytiscidae | Liodessus | x | x | Polymera | x | x | x | ||||||||
| Rhantus | x | x | x | Muscidae | Limnophora | x | x | x | x | ||||||
| Elmidae | Austrelmis | x | x | x | x | Perlidae | Anacroneuria | x | x | x | |||||
| Austrolimnius | x | x | x | x | Claudioperla | x | x | x | |||||||
| Heterelmis | x | x | x | x | x | Polycentropodidae | Polycentropus | x | x | ||||||
| Hexanchorus | x | x | x | x | Psephenidae | Pheneps | x | x | x | ||||||
| Huleechius | x | x | x | x | x | Psychodidae | Maruina | x | x | x | x | ||||
| Macrelmis | x | x | x | Pericoma | x | x | x | ||||||||
| Microcylloepus | x | x | Psychoda | x | x | x | x | ||||||||
| Neoelmis | x | x | x | x | Scirtidae | Cyphon | x | x | x | x | |||||
| Notelmis | x | x | Simuliidae | Gigantodax | x | x | x | x | x | ||||||
| Onychelmis | x | x | Simulium | x | x | x | x | x | |||||||
| Pharceonus | x | x | Tabanidae | Tabanus | x | x | x | ||||||||
| Pseudodisersus | x | x | Tipulidae | Tipula | x | x | x | ||||||||
| Empididae | Neoplasta | x | x | x | x | x | Xiphocentronidae | x | x | x | |||||
| Ephydridae | x | x | x | Malacostraca | Hyalellidae | Hyalella | x | x | x | x | x | ||||
| Glossosomatidae | Mortoniella | x | x | Ostracoda | x | x | x | ||||||||
| Grumichellinae | Atanatolica | x | x | x | Turbellaria | Dugesiidae | Girardia | x | x | x | x | x | |||
| Helicopsychidae | Helicopsyche | x | x | x |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hampel, H.; Vázquez, R.F.; González, H.; Acosta, R. Evaluating the Ecological Status of Fluvial Networks of Tropical Andean Catchments of Ecuador. Water 2023, 15, 1742. https://doi.org/10.3390/w15091742
Hampel H, Vázquez RF, González H, Acosta R. Evaluating the Ecological Status of Fluvial Networks of Tropical Andean Catchments of Ecuador. Water. 2023; 15(9):1742. https://doi.org/10.3390/w15091742
Chicago/Turabian StyleHampel, Henrietta, Raúl F. Vázquez, Hari González, and Raúl Acosta. 2023. "Evaluating the Ecological Status of Fluvial Networks of Tropical Andean Catchments of Ecuador" Water 15, no. 9: 1742. https://doi.org/10.3390/w15091742
APA StyleHampel, H., Vázquez, R. F., González, H., & Acosta, R. (2023). Evaluating the Ecological Status of Fluvial Networks of Tropical Andean Catchments of Ecuador. Water, 15(9), 1742. https://doi.org/10.3390/w15091742





