Competitive Removal of Antimony and Humic Acid by Ferric Chloride: Optimization of Coagulation Process Using Response Surface Methodology
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Stock Solutions and Test Samples
2.2. Experimental Procedure and Analytical Techniques
2.3. Experimental Design and Statistical Analysis
3. Results and Discussion
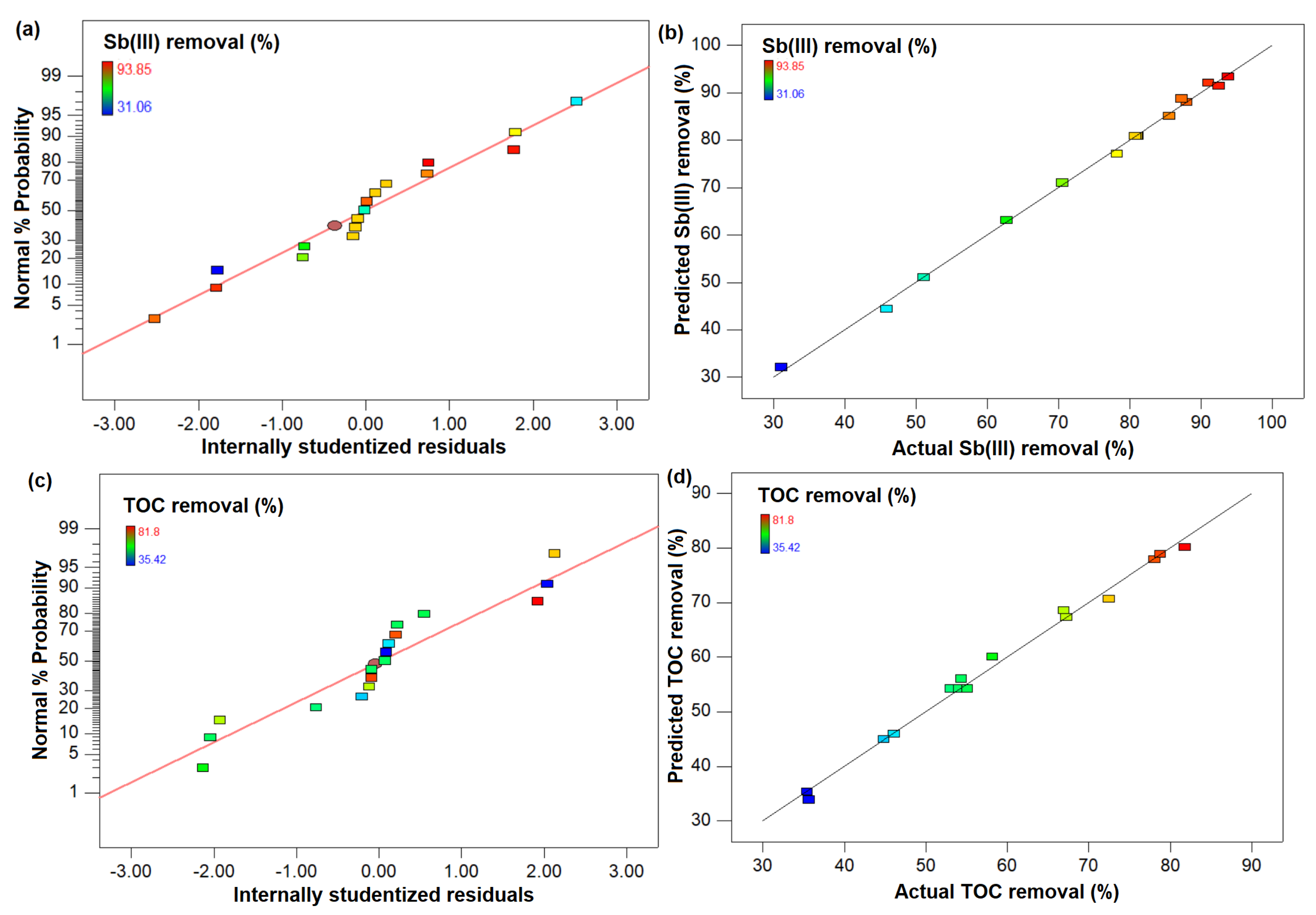
3.1. Identifying Best Response Function Using Statistical Analysis
3.2. Fitting the Models
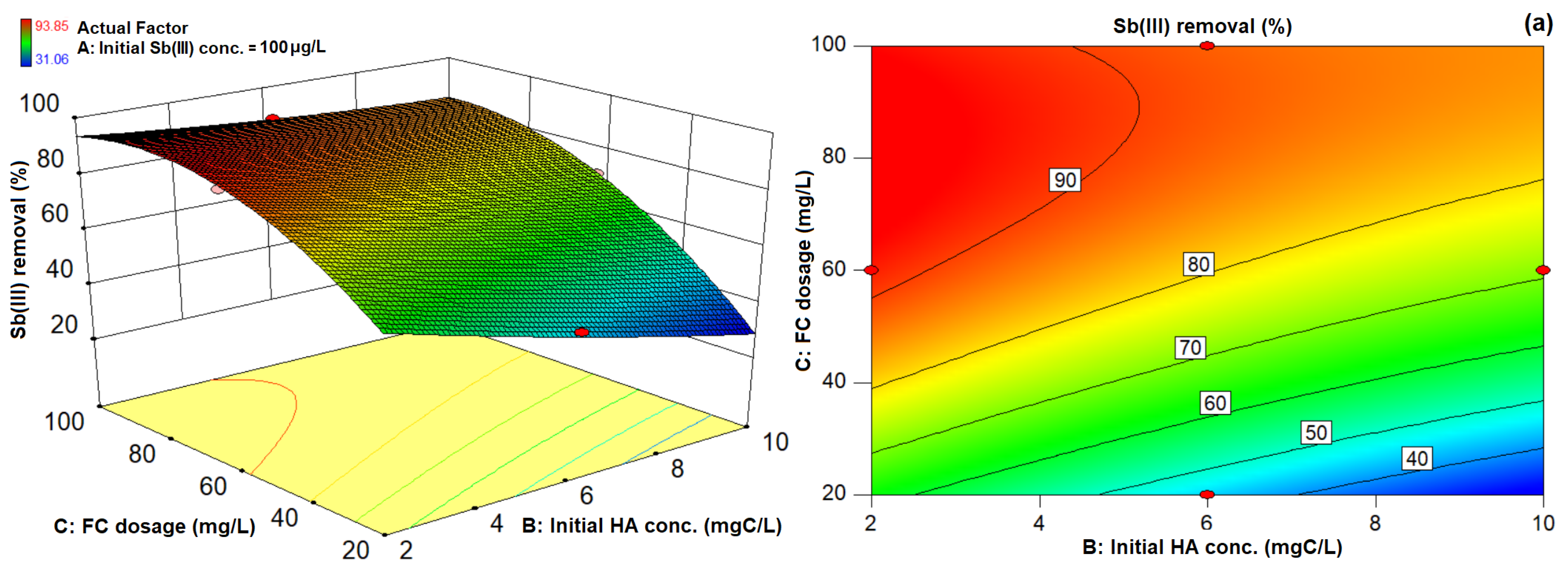
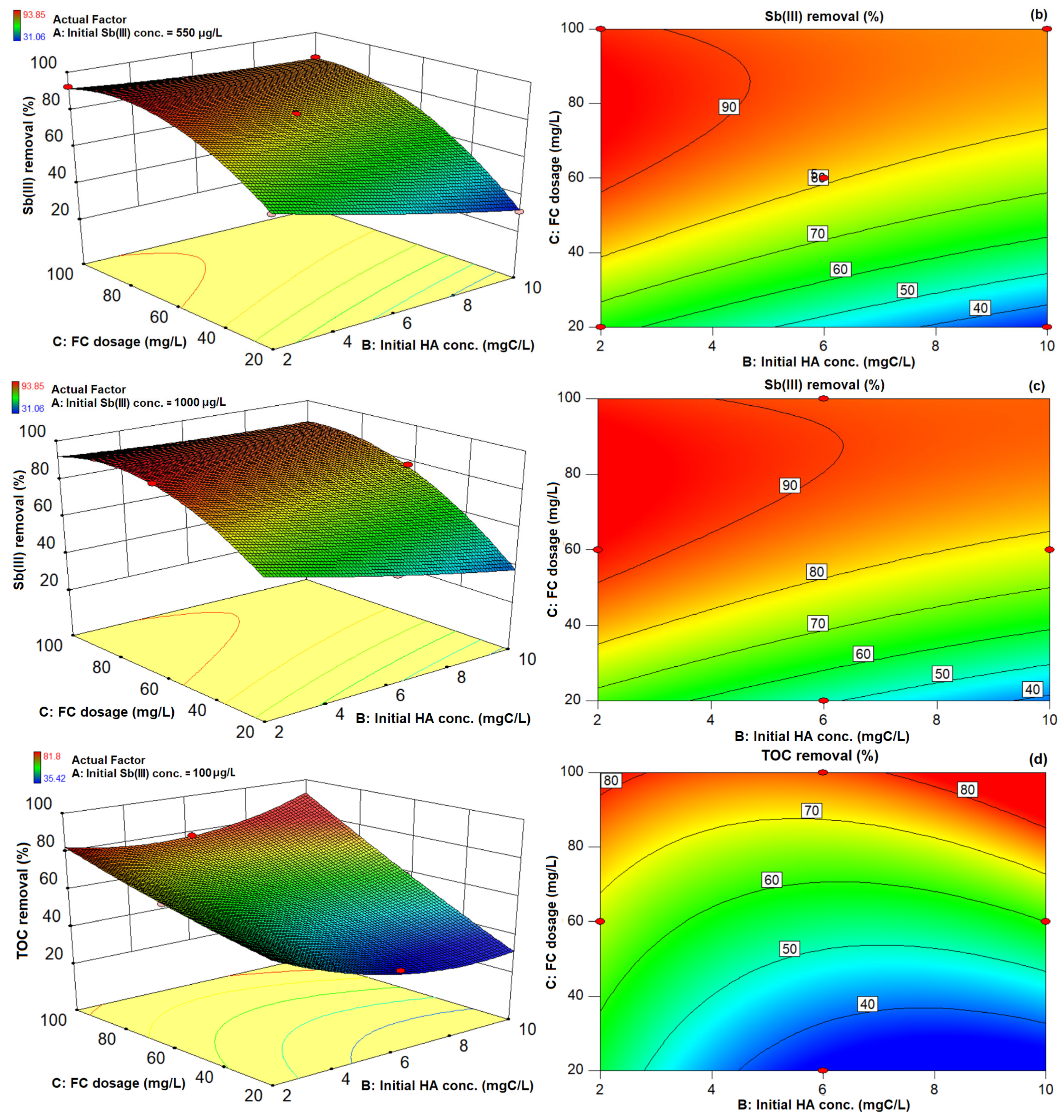
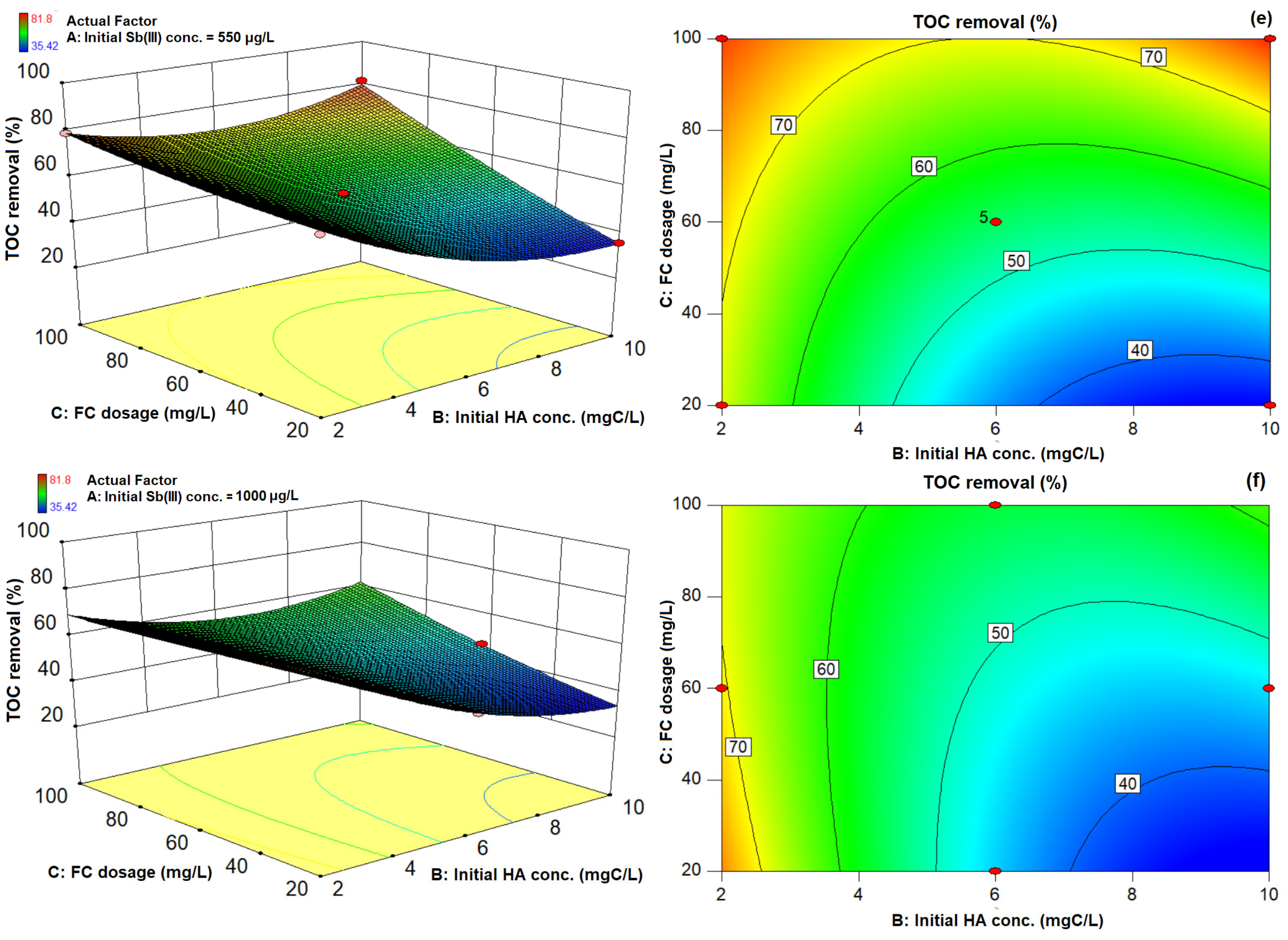
3.3. 3D Response Surface Plots and 2D Contour Plots of Sb(III) and TOC Removal
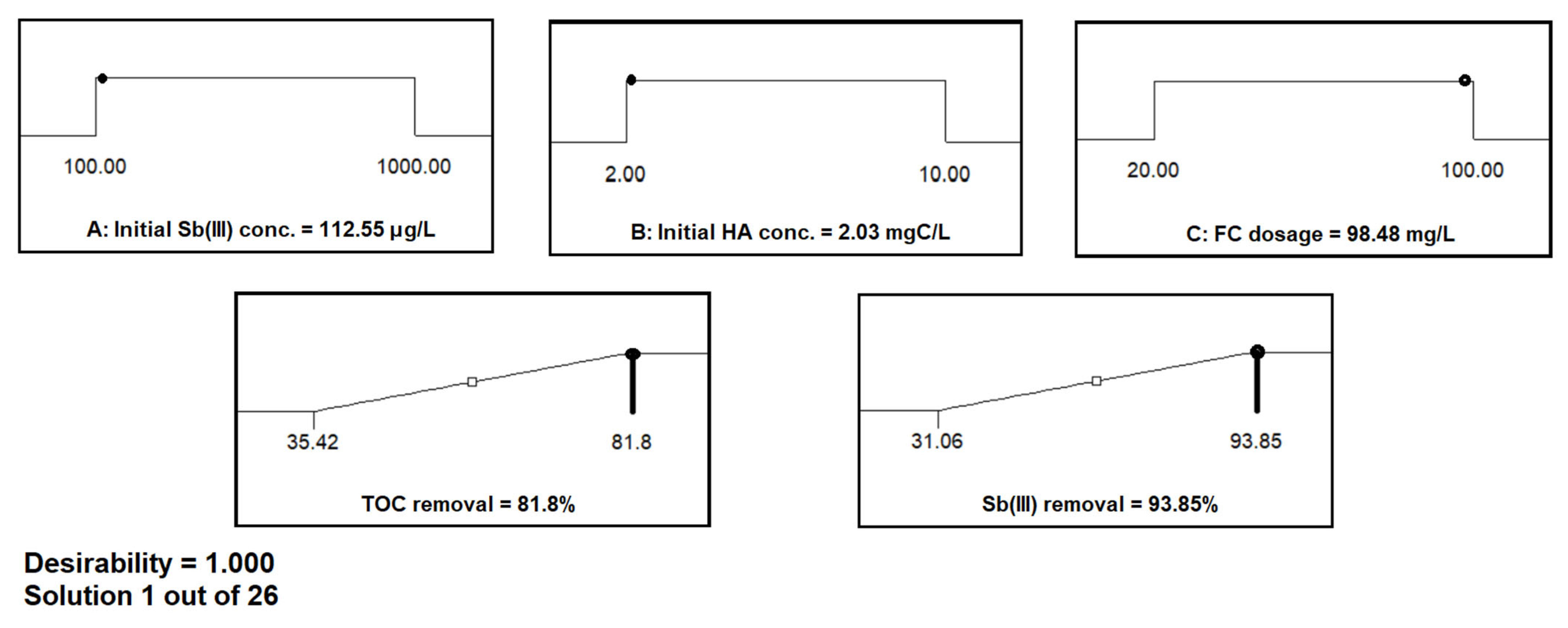
3.4. Optimum Experimental Design Conditions for Sb(III) and TOC Removal
3.5. Volumetric Analysis of Produced Sludge
3.6. Comparison of Sb(III) and TOC Removal with the Literature Studies
3.7. Future Prospects
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, D.; Sun, S.-P.; He, M.; Wu, Z.; Xiao, J.; Chen, X.D.; Wu, W.D. As (V) and Sb (V) co-adsorption onto ferrihydrite: Synergistic effect of Sb (V) on As (V) under competitive conditions. Environ. Sci. Pollut. Res. 2018, 25, 14585–14594. [Google Scholar] [CrossRef] [PubMed]
- Mubarak, H.; Chai, L.-Y.; Mirza, N.; Yang, Z.-H.; Pervez, A.; Tariq, M.; Shaheen, S.; Mahmood, Q. Antimony (Sb)–pollution and removal techniques–critical assessment of technologies. Toxicol. Environ. Chem. 2015, 97, 1296–1318. [Google Scholar] [CrossRef]
- Filella, M.; Belzile, N.; Chen, Y.W. Antimony in the environment: A review focused on natural waters I. Occurence. Earth-Sci. Rev. 2002, 57, 125–176. [Google Scholar] [CrossRef]
- Ober, J.A. Mineral Commodity Summaries 2017: US Geological Survey; U.S. Geological Survey: Reston, VA, USA, 2017. [Google Scholar] [CrossRef]
- Sheng, L.; Hao, C.; Guan, S.; Huang, Z. Spatial distribution, geochemical behaviors and risk assessment of antimony in rivers around the antimony mine of Xikuangshan, Hunan Province, China. Water Sci. Technol. 2022, 85, 1141–1154. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Song, X.; Zheng, Q.; Liu, C.; Li, K.; Luo, Q.; Chen, J.; Wang, Z.; Luo, J. Frontier materials for adsorption of antimony and arsenic in aqueous environments: A review. Int. J. Environ. Res. Public Health 2022, 19, 10824. [Google Scholar] [CrossRef]
- Xie, Q.; Ren, B. Pollution and risk assessment of heavy metals in rivers in the antimony capital of Xikuangshan. Sci. Rep. 2022, 12, 14393. [Google Scholar] [CrossRef]
- Gan, Y.; Ding, C.; Xu, B.; Liu, Z.; Zhang, S.; Cui, Y.; Wu, B.; Huang, W.; Song, X. Antimony (Sb) pollution control by coagulation and membrane filtration in water/wastewater treatment: A comprehensive review. J. Hazard. Mater. 2022, 442, 130072. [Google Scholar] [CrossRef]
- Arain, M.B.; Kazi, T.G.; Baig, J.A.; Jamali, M.K.; Afridi, H.I.; Shah, A.Q.; Jalbani, N.; Sarfraz, R.A. Determination of arsenic levels in lake water, sediment, and foodstuff from selected area of Sindh, Pakistan: Estimation of daily dietary intake. Food Chem. Toxicol. 2009, 47, 242–248. [Google Scholar] [CrossRef]
- Wang, X.; He, M.; Xi, J.; Lu, X. Antimony distribution and mobility in rivers around the world’s largest antimony mine of Xikuangshan, Hunan Province, China. Microchem. J. 2011, 97, 4–11. [Google Scholar] [CrossRef]
- Ritchie, V.J.; Ilgen, A.G.; Mueller, S.H.; Trainor, T.P.; Goldfarb, R.J. Mobility and chemical fate of antimony and arsenic in historic mining environments of the Kantishna Hills district, Denali National Park and Preserve, Alaska. Chem. Geol. 2013, 335, 172–188. [Google Scholar] [CrossRef]
- Ungureanu, G.; Santos, S.; Boaventura, R.; Botelho, C. Arsenic and antimony in water and wastewater: Overview of removal techniques with special reference to latest advances in adsorption. J. Environ. Manag. 2015, 151, 326–342. [Google Scholar] [CrossRef] [PubMed]
- Herath, I.; Vithanage, M.; Bundschuh, J. Antimony as a global dilemma: Geochemistry, mobility, fate and transport Title. Environ. Pollut. 2017, 223, 545–559. [Google Scholar] [CrossRef] [PubMed]
- Daud, M.K.; Nafees, M.; Ali, S.; Rizwan, M.; Bajwa, R.A.; Shakoor, M.B.; Arshad, M.U.; Chatha, S.A.S.; Deeba, F.; Murad, W.; et al. Drinking Water Quality Status and Contamination in Pakistan. BioMed Res. Int. 2017, 7908183. [Google Scholar] [CrossRef]
- Jo, M.; Kim, T.; Choi, S.; Jung, J.; Song, H.; Lee, H.; Park, G.; Lim, S.; Sung, Y.; Oh, J. Investigation of Antimony in Natural Water and Leaching from Polyethylene Terephthalate (PET) Bottled Water. In Proceedings of the 3rd World Congress on New Technologies (NewTech’17), Rome, Italy, 6–8 June 2017; pp. 6–8. [Google Scholar]
- Guo, W.; Fu, Z.; Wang, H.; Liu, S.; Wu, F.; Giesy, J.P. Removal of antimonate (Sb (V)) and antimonite (Sb (III)) from aqueous solutions by coagulation-flocculation-sedimentation (CFS): Dependence on influencing factors and insights into removal mechanisms. Sci. Total Environ. 2018, 644, 1277–1285. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.; Fang, Y.; Li, H.; Yang, Z. Review of recently used adsorbents for antimony removal from contaminated water. Environ. Sci. Pollut. Res. 2022, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Ang, W.L.; Mohammad, A.W. State of the art and sustainability of natural coagulants in water and wastewater treatment. J. Clean. Prod. 2020, 262, 121267. [Google Scholar] [CrossRef]
- Cheng, K.; Wang, H.; Li, J.; Li, F. An effective method to remove antimony in water by using iron-based coagulants. Water 2020, 12, 66. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manage. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Wu, Z.; He, M. Removal of antimony(V) and antimony(III) from drinking water by coagulation-flocculation-sedimentation (CFS). Water Res. 2009, 43, 4327–4335. [Google Scholar] [CrossRef]
- Kang, M.; Kamei, T.; Magara, Y. Comparing polyaluminum chloride and ferric chloride for antimony removal. Water Res. 2003, 37, 4171–4179. [Google Scholar] [CrossRef]
- Wu, Z.; He, M.; Guo, X.; Zhou, R. Removal of antimony (III) and antimony (V) from drinking water by ferric chloride coagulation: Competing ion effect and the mechanism analysis. Sep. Purif. Technol. 2010, 76, 184–190. [Google Scholar] [CrossRef]
- Mustereț, C.P.; Morosanu, I.; Ciobanu, R.; Plavan, O.; Gherghel, A.; Al-Refai, M.; Roman, I.; Teodosiu, C. Assessment of coagulation–flocculation process efficiency for the natural organic matter removal in drinking water treatment. Water 2021, 13, 3073. [Google Scholar] [CrossRef]
- Dayarathne, H.N.P.; Angove, M.J.; Aryal, R.; Abuel-Naga, H.; Mainali, B. Removal of natural organic matter from source water: Review on coagulants, dual coagulation, alternative coagulants, and mechanisms. J. Water Process Eng. 2021, 40, 101820. [Google Scholar] [CrossRef]
- Tang, W.-W.; Zeng, G.-M.; Gong, J.-L.; Liang, J.; Xu, P.; Zhang, C.; Huang, B.-B. Impact of humic/fulvic acid on the removal of heavy metals from aqueous solutions using nanomaterials: A review. Sci. Total Environ. 2014, 468, 1014–1027. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Duan, J.; Liu, S.; Li, W.; van Leeuwen, J.; Mulcahy, D. Removal of As (III) and As (V) by ferric salts coagulation–Implications of particle size and zeta potential of precipitates. Sep. Purif. Technol. 2014, 135, 64–71. [Google Scholar] [CrossRef]
- Inam, M.A.; Khan, R.; Park, D.R.; Khan, S.; Uddin, A.; Yeom, I.T. Complexation of Antimony with Natural Organic Matter: Performance Evaluation during Coagulation-Flocculation Process. Int. J. Environ. Res. Public Health 2019, 16, 1092. [Google Scholar] [CrossRef]
- Inam, M.A.; Lee, K.H.; Soni, H.L.; Mangi, K.H.; Channa, A.S.; Khan, R.; Wie, Y.M.; Lee, K.G. Coagulation Behavior of Antimony Oxyanions in Water: Influence of pH, Inorganic and Organic Matter on the Physicochemical Characteristics of Iron Precipitates. Molecules 2022, 27, 1663. [Google Scholar] [CrossRef]
- Trinh, T.K.; Kang, L.S. Response surface methodological approach to optimize the coagulation–flocculation process in drinking water treatment. Chem. Eng. Res. Des. 2011, 89, 1126–1135. [Google Scholar] [CrossRef]
- Amiri, S.; Vatanpour, V.; He, T. Optimization of Coagulation-Flocculation Process in Efficient Arsenic Removal from Highly Contaminated Groundwater by Response Surface Methodology. Molecules 2022, 27, 7953. [Google Scholar] [CrossRef]
- Watson, M.A.; Tubić, A.; Agbaba, J.; Nikić, J.; Maletić, S.; Jazić, J.M.; Dalmacija, B. Response surface methodology investigation into the interactions between arsenic and humic acid in water during the coagulation process. J. Hazard. Mater. 2016, 312, 150–158. [Google Scholar] [CrossRef]
- Inam, M.A.; Khan, R.; Yeom, I.T.; Buller, A.S.; Akram, M.; Inam, M.W. Optimization of Antimony Removal by Coagulation-Flocculation-Sedimentation Process Using Response Surface Methodology. Processes 2021, 9, 117. [Google Scholar] [CrossRef]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 2008, 76, 965–977. [Google Scholar] [CrossRef] [PubMed]
- Inam, M.A.; Khan, R.; Park, D.R.; Lee, Y.W.; Yeom, I.T. Removal of Sb(III) and Sb(V) by ferric chloride coagulation: Implications of Fe solubility. Water 2018, 10, 418. [Google Scholar] [CrossRef]
- Baskan, M.B.; Pala, A. A statistical experiment design approach for arsenic removal by coagulation process using aluminum sulfate. Desalination 2010, 254, 42–48. [Google Scholar] [CrossRef]
- Inam, M.A.; Khan, R.; Inam, M.W.; Yeom, I.T. Kinetic and isothermal sorption of antimony oxyanions onto iron hydroxide during water treatment by coagulation process. J. Water Process Eng. 2021, 41, 102050. [Google Scholar] [CrossRef]
- Dong, C.-H.; Xie, X.-Q.; Wang, X.-L.; Zhan, Y.; Yao, Y.-J. Application of Box-Behnken design in optimisation for polysaccharides extraction from cultured mycelium of Cordyceps sinensis. Food Bioprod. Process. 2009, 87, 139–144. [Google Scholar] [CrossRef]
- Amuda, O.S.; Amoo, I.A.; Ajayi, O.O. Performance optimization of coagulant/flocculant in the treatment of wastewater from a beverage industry. J. Hazard. Mater. 2006, 129, 69–72. [Google Scholar] [CrossRef]
- Aguilar, M.I.; Saez, J.; Llorens, M.; Soler, A.; Ortuno, J.F. Nutrient removal and sludge production in the coagulation–flocculation process. Water Res. 2002, 36, 2910–2919. [Google Scholar] [CrossRef]
- Zhao, Y.X.; Gao, B.Y.; Zhang, G.Z.; Qi, Q.B.; Wang, Y.; Phuntsho, S.; Kim, J.-H.; Shon, H.K.; Yue, Q.Y.; Li, Q. Coagulation and sludge recovery using titanium tetrachloride as coagulant for real water treatment: A comparison against traditional aluminum and iron salts. Sep. Purif. Technol. 2014, 130, 19–27. [Google Scholar] [CrossRef]
- Sillanpää, M.; Ncibi, M.C.; Matilainen, A.; Vepsäläinen, M. Removal of natural organic matter in drinking water treatment by coagulation: A comprehensive review. Chemosphere 2018, 190, 54–71. [Google Scholar] [CrossRef]

| Components | Value/Concentration |
|---|---|
| pH | 6.85 |
| HCO3− (mg CaCO3/L) | 430 |
| PO43− (mg/L) | 0.476 |
| SO42− (mg/L) | 75.77 |
| Cl− (mg/L) | 70.97 |
| NO3− (mg/L) | 9.37 |
| As (mg/L) | 0.011 |
| Fe (mg/L) | 0.243 |
| Zn (mg/L) | 0.521 |
| Cr (mg/L) | 0.121 |
| Sb (mg/L) * | 0.1 to 1 * |
| TOC (mgC/L) * | 2 to 10 * |
| Variables | Units | Symbol | Coded Levels | |||
|---|---|---|---|---|---|---|
| Uncoded | Coded | −1 | 0 | +1 | ||
| A: Initial Sb(III) concentration | µg/L | X1 | A | 100 | 550 | 1000 |
| B: Initial HA concentration | mgC/L | X2 | B | 2 | 6 | 10 |
| C: FC dosage | mg/L | X3 | C | 20 | 60 | 100 |
| Source | Sequential p-Value | Lack of Fit p-Value | Std. Dev. | R2 | Adjusted R2 | Predicted R2 | Remarks |
|---|---|---|---|---|---|---|---|
| Sb(III) removal | |||||||
| Linear | <0.0001 | <0.0001 | 8.86 | 0.7982 | 0.7516 | 0.6058 | |
| 2FI | 0.5991 | <0.0001 | 9.24 | 0.8312 | 0.7300 | 0.2503 | |
| Quadratic | <0.0001 | 0.0004 | 1.18 | 0.9981 | 0.9956 | 0.9695 | Suggested |
| Cubic | 0.0004 | 0.18 | 1.0000 | 0.9999 | Aliased | ||
| TOC removal | |||||||
| Linear | 0.0025 | <0.0001 | 9.20 | 0.6546 | 0.5748 | 0.3024 | |
| 2FI | 0.0269 | 0.0001 | 6.76 | 0.8567 | 0.7708 | 0.3700 | |
| Quadratic | <0.0001 | 0.0212 | 1.72 | 0.9935 | 0.9851 | 0.9062 | Suggested |
| Cubic | 0.0212 | 0.75 | 0.9993 | 0.9972 | Aliased | ||
| Source | Sum of Squares | df | Mean Square | F Value | p-Values Prob > F |
|---|---|---|---|---|---|
| Sb(III) removal | |||||
| Model | 5044.74 | 9 | 560.53 | 402.40 | <0.0001 * |
| A-Initial Sb(III) concentration | 27.79 | 1 | 27.79 | 19.95 | 0.0029 * |
| B-Initial HA concentration | 698.45 | 1 | 698.45 | 501.42 | <0.0001 * |
| C-FC dosage | 3308.10 | 1 | 3308.10 | 2374.90 | <0.0001 * |
| AB | 5.93 | 1 | 5.93 | 4.26 | 0.0780 |
| AC | 8.88 | 1 | 8.88 | 6.38 | 0.0395 * |
| BC | 152.28 | 1 | 152.28 | 109.32 | <0.0001 * |
| A2 | 7.47 | 1 | 7.47 | 5.37 | 0.0537 |
| B2 | 6.45 | 1 | 6.45 | 4.63 | 0.0685 |
| C2 | 840.69 | 1 | 840.69 | 603.54 | <0.0001 * |
| Lack of fit | 9.62 | 3 | 3.21 | 95.34 | 0.0004 * |
| Pure error | 0.13 | 4 | 0.034 | ||
| R2 = 0.9981, R2Adj = 0.9956, R2Pre = 0.9695, adequate precision = 67.722 (>4) | |||||
| TOC removal | |||||
| Model | 3165.36 | 9 | 351.71 | 118.86 | <0.0001 * |
| A-Initial Sb(III) concentration | 57.46 | 1 | 57.46 | 19.42 | 0.0031 * |
| B-Initial HA concentration | 511.68 | 1 | 511.68 | 172.93 | <0.0001 * |
| C-FC dosage | 1516.35 | 1 | 1516.35 | 512.47 | <0.0001 * |
| AB | 75.26 | 1 | 75.26 | 25.43 | 0.0015 * |
| AC | 270.77 | 1 | 270.77 | 91.51 | <0.0001 * |
| BC | 298.08 | 1 | 298.08 | 100.74 | <0.0001 * |
| A2 | 34.88 | 1 | 34.88 | 11.79 | 0.0109 * |
| B2 | 391.02 | 1 | 391.02 | 132.15 | <0.0001 * |
| C2 | 14.44 | 1 | 14.44 | 4.88 | 0.0629 |
| Lack of fit | 18.47 | 3 | 6.16 | 10.97 | 0.0212 * |
| Pure error | 2.25 | 4 | 0.56 | ||
| R2 = 0.9935, R2Adj = 0.9851, R2Pre = 0.9062, adequate precision = 35.044 (>4) | |||||
| Coagulants | Initial Sb(III) (mg/L) | Initial HA (mgC/L) | Sb(III) Removal (%) | TOC Removal (%) | Ref. | |
|---|---|---|---|---|---|---|
| Type | Dosage (mg/L) | |||||
| Al2(SO4)3·18H2O | 66.64 | 0.05 | - | 18.5 | - | [21] |
| FeCl3·6H2O | 54.06 | 0.506 | - | 93.04 | - | |
| FeCl3·6H2O | 54.06 | 0.101 | 0 | 96 | - | [23] |
| 2 | 84 | - | ||||
| 4 | 58 | - | ||||
| Polymeric ferric sulfate | 6.04 | 0.100 | 0 | 96.5 | - | [16] |
| 1 | 90 | - | ||||
| 2 | 71.5 | - | ||||
| FeCl3·6H2O | 27.03 | 1 | 0 | 90.4 | 0 | [29] |
| 0.417 | 84.15 | 81.26 | ||||
| 0.834 | 82.53 | 71.61 | ||||
| 2.085 | 74.92 | 64.15 | ||||
| 4.17 | 62.48 | 65.86 | ||||
| Al2(SO4)3 | 30 | - | 3.9 | - | 43.2 | [41,42] |
| FeCl3 | 65 | - | 3.9 | - | 57.9 | |
| FeCl3·6H2O | 100 | 0.55 | 2 | 92.51 | 78.8 | This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inam, M.A.; Khan, R.; Lee, K.H.; Babar, Z.B.; Yeom, I.T. Competitive Removal of Antimony and Humic Acid by Ferric Chloride: Optimization of Coagulation Process Using Response Surface Methodology. Water 2023, 15, 1676. https://doi.org/10.3390/w15091676
Inam MA, Khan R, Lee KH, Babar ZB, Yeom IT. Competitive Removal of Antimony and Humic Acid by Ferric Chloride: Optimization of Coagulation Process Using Response Surface Methodology. Water. 2023; 15(9):1676. https://doi.org/10.3390/w15091676
Chicago/Turabian StyleInam, Muhammad Ali, Rizwan Khan, Kang Hoon Lee, Zaeem Bin Babar, and Ick Tae Yeom. 2023. "Competitive Removal of Antimony and Humic Acid by Ferric Chloride: Optimization of Coagulation Process Using Response Surface Methodology" Water 15, no. 9: 1676. https://doi.org/10.3390/w15091676