Nitrogen Removal for Low Concentration Ammonium Wastewater by Adsorption, Shortcut Simultaneous Nitrification and Denitrification Process in MBBR
Abstract
:1. Introduction
2. Materials and Methods
2.1. Wastewater Composition and Carrier
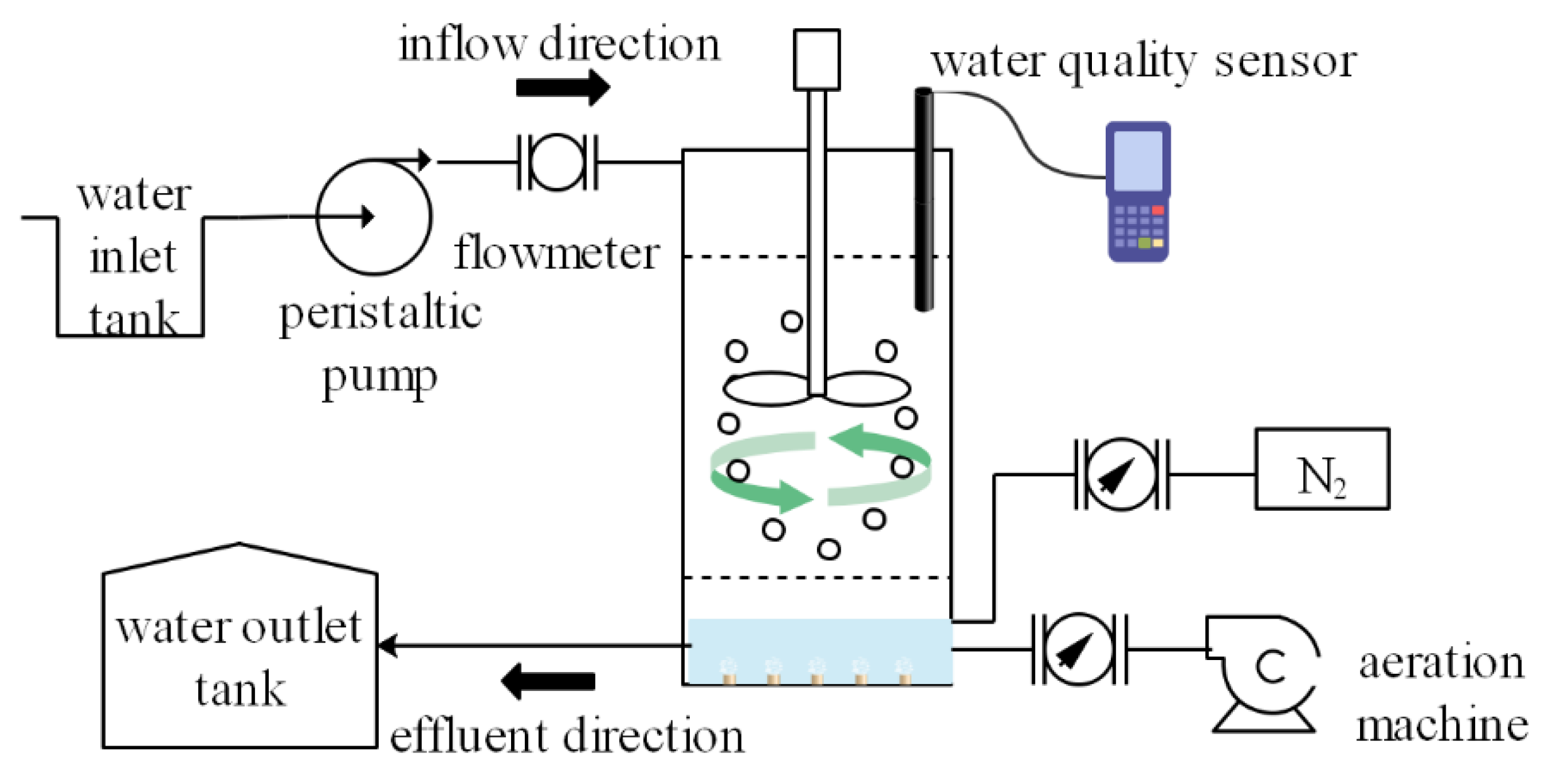
2.2. Experimental Set-Up and Operation of the MBBR
2.3. Technological Process
2.4. Experimental Design
2.4.1. Study on the Operational Performance at the MBBR Adsorption Stage
- (1)
- Effect of hydraulic retention time (HRT) on the treatment effect of the MBBR in the adsorption stage.
- (2)
- Effect of stirring speed on the adsorption of ammonia and nitrogen by ceramsite.
2.4.2. Study on the Operational Performance at the MBBR Shortcut SND Denitrification and Regeneration Stage
- (1)
- Effect of alkalinity dosage ratio on the MBBR shortcut SND denitrification and regeneration.
- (2)
- Effect of DO on the MBBR shortcut SND denitrification and regeneration.
2.5. Water Quality Analysis Method
2.6. Detection and Calculation of Ceramsite Desorption Amount
2.7. Calculation of Relevant Indicators for Each Stage
2.7.1. Adsorption Stage
- (1)
- Integral value of adsorption curvewhere I is the integral value of the adsorption curve, mg·h/L; is the concentration of ammonia nitrogen in the effluent at time t, mg/L; is the concentration of ammonia nitrogen in the effluent at the initial time, mg/L; t is the operating time of adsorption stage, h.
- (2)
- Accumulated ammonia nitrogen effluent
- (3)
- Accumulated ammonia nitrogen influentwhere is the total volume of wastewater treated in the MBBR system, L; V is the reactor volume, which is the volume of wastewater added to the reactor at the initial time, 6 L; is the accumulated ammonia nitrogen influent to the reactor, mg; is the input ammonia nitrogen concentration, 26.2 mg/L (in N).
- (4)
- Ammonia volume loadwhere AVL is ammonia volume load, mg/(L·h).
2.7.2. Shortcut SND Denitrification and Regeneration Stage
- (1)
- Theoretical alkalinity dosage
- (2)
- Simultaneous nitrification and denitrification rate
- (3)
- Nitrite accumulation ratio
- (4)
- Total nitrogen removal rate
- (5)
- Ammonia removal rate
- (6)
- Regeneration rate of bio-ceramsite
- (7)
- Average regeneration speed of bio-ceramsite at 12 h
- (8)
- FA concentration
2.8. Statistical Analysis
3. Results and Discussion
3.1. Study on the Operational Performance at the MBBR Adsorption Stage
3.1.1. Effect of Hydraulic Retention Time on the Adsorption Performance of the MBBR
3.1.2. Effect of Stirring Speed on the Adsorption Performance of the MBBR
3.2. Study on the Operational Performance at MBBR Shortcut SND Denitrification and Regeneration Stage
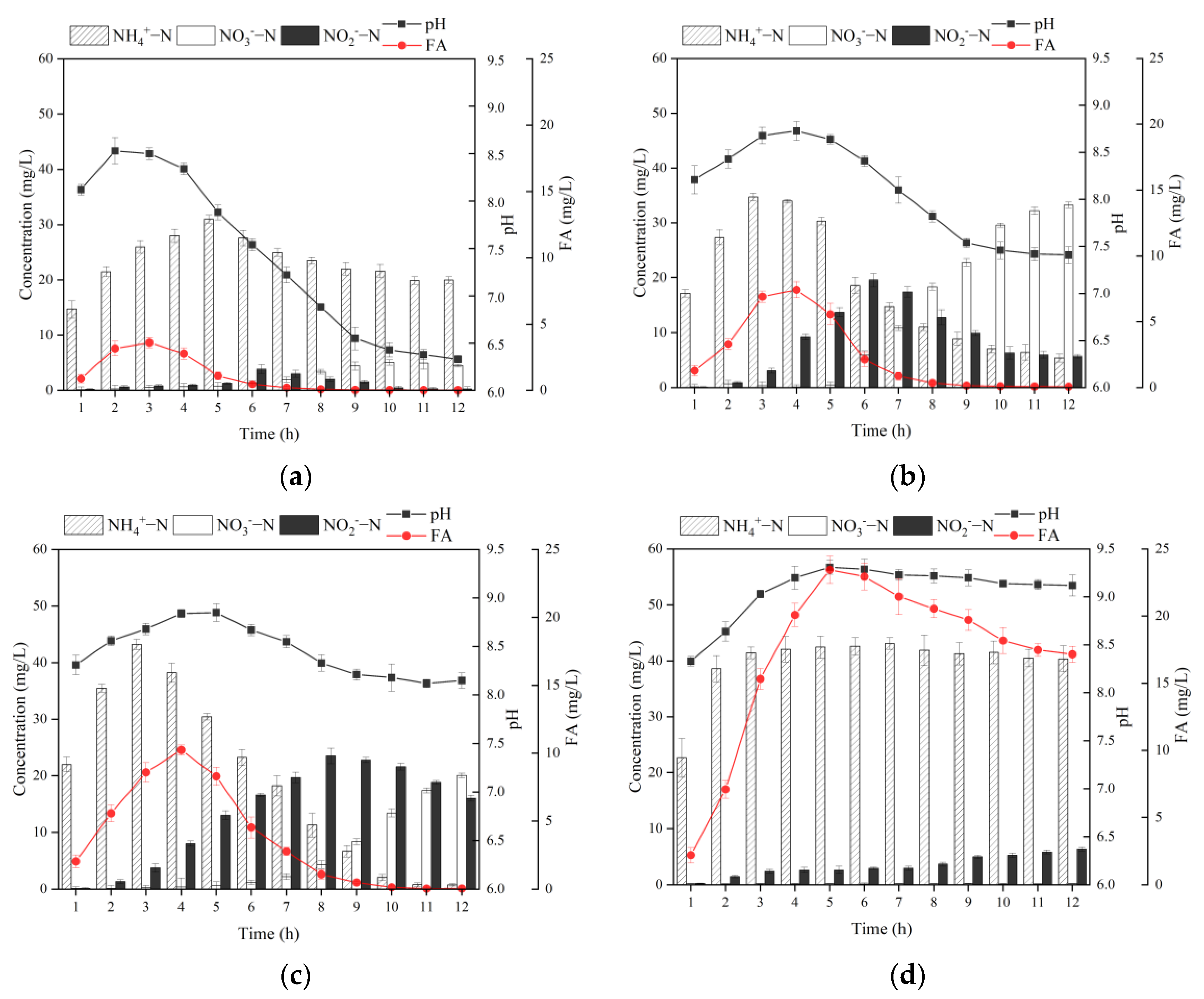
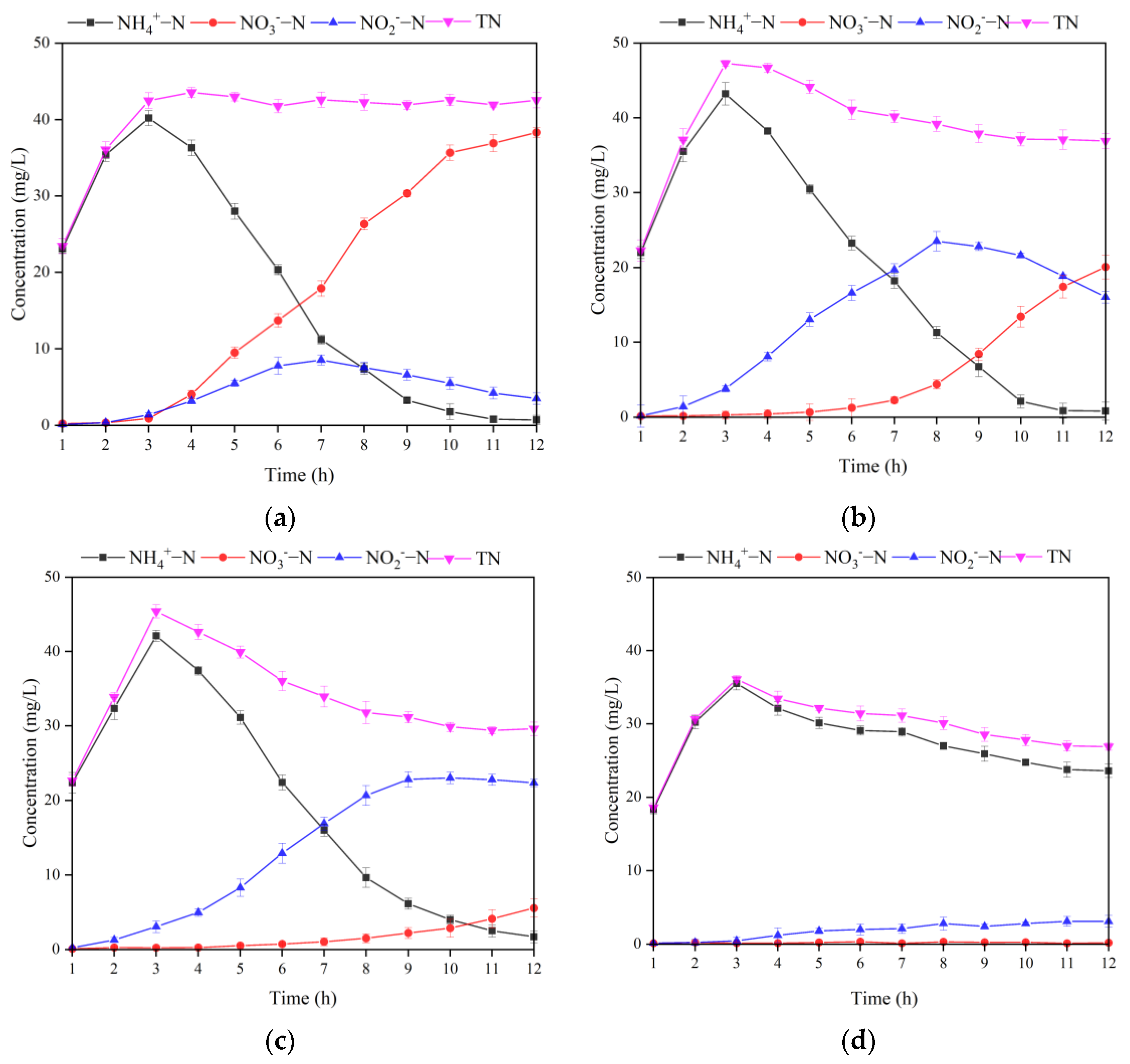
3.2.1. Effect of Alkalinity Dosage Ratio on MBBR Shortcut SND Denitrification and Regeneration
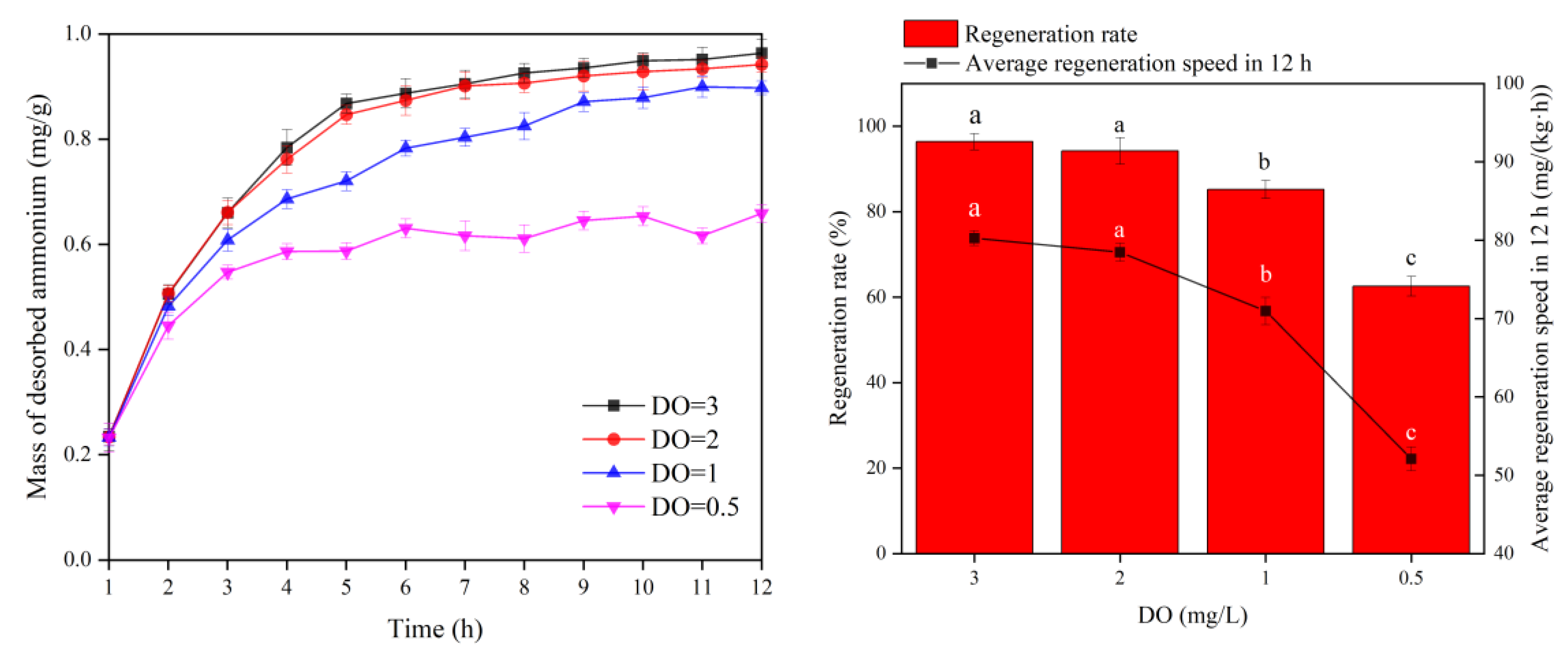
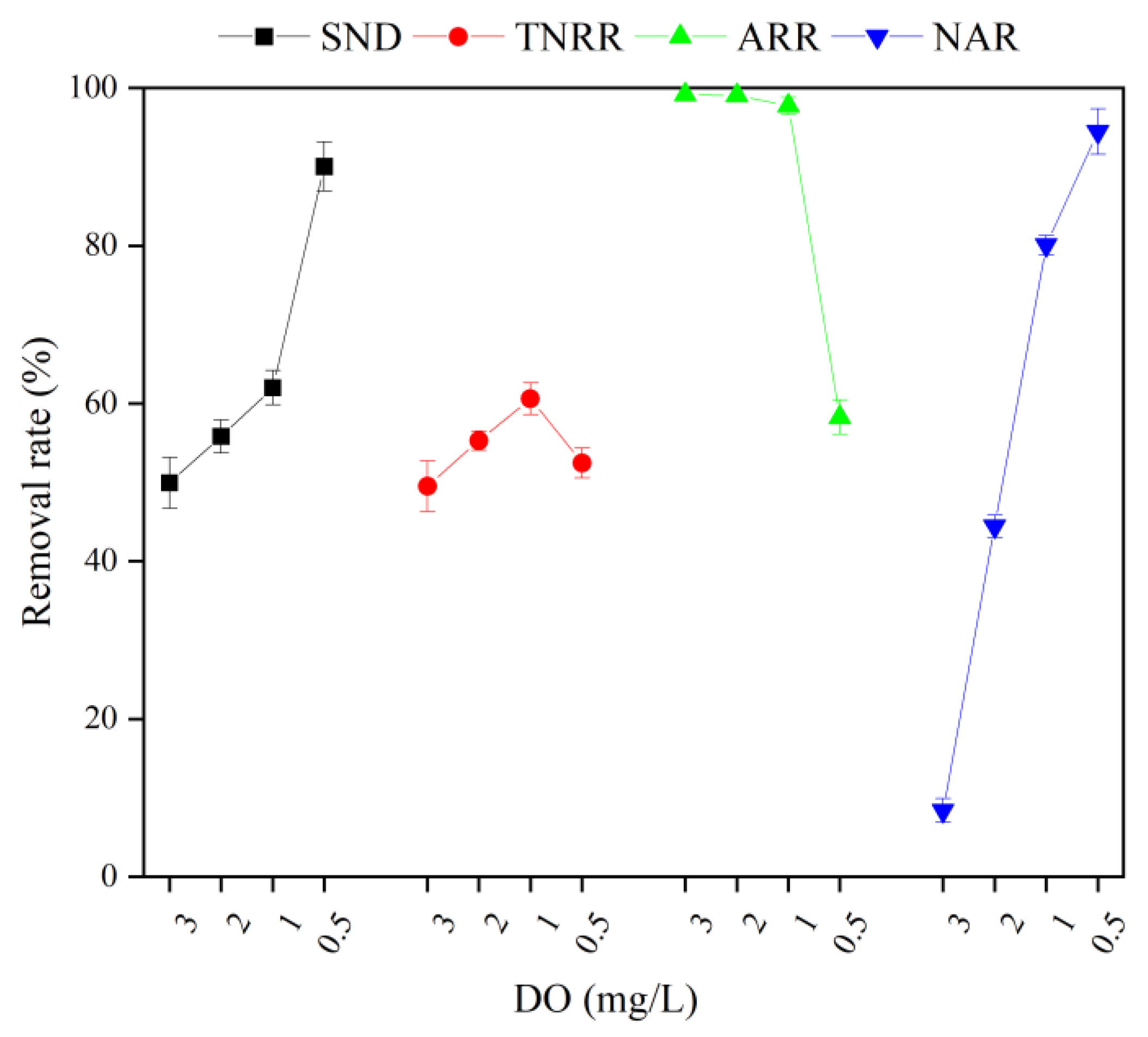
3.2.2. Effect of DO on the MBBR Shortcut SND Denitrification and Regeneration
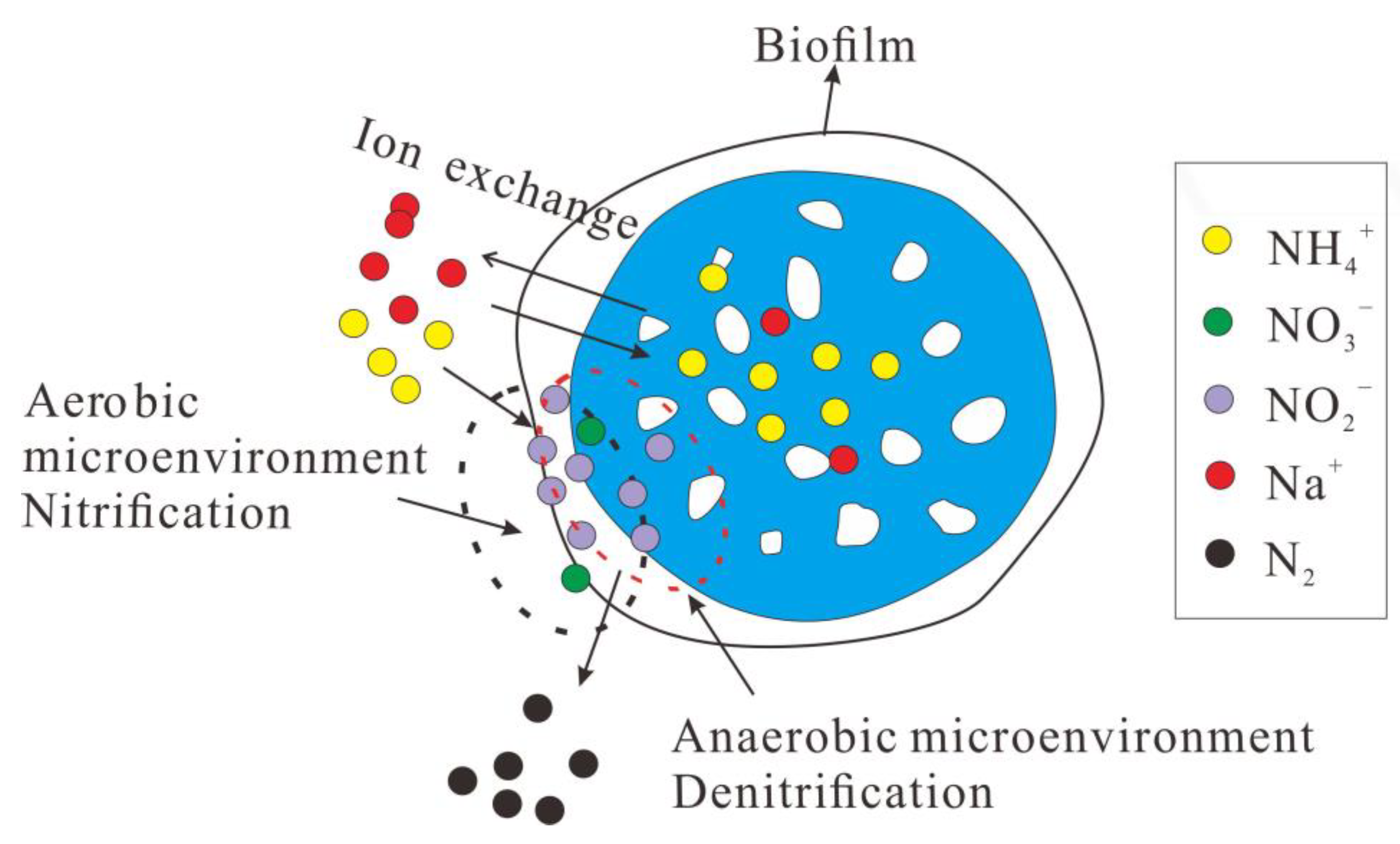
3.3. The Optimum Conditions and the Mechanism of the MBBR Adsorption-Shortcut SND Process
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ibrahim, L.A.; Abu-hashim, M.; Shaghaleh, H.; Elsadek, E.; Hamad, A.A.A.; Alhaj Hamoud, Y. A Comprehensive Review of the Multiple Uses of Water in Aquaculture-Integrated Agriculture Based on International and National Experiences. Water 2023, 15, 367. [Google Scholar] [CrossRef]
- Ramirez, M.; Gomez, J.M.; Aroca, G.; Cantero, D. Removal of ammonia by immobilized Nitrosomonas europaea in a biotrickling filter packed with polyurethane foam. Chemosphere 2009, 74, 1385–1390. [Google Scholar] [CrossRef] [PubMed]
- Faskol, A.S.; Racovieanu, G. Effect of DO, Alkalinity and pH on Nitrification Using Three Different Sunken Materials Types in Biological Aerated Filter BAFs. IOP Conf. Ser. Earth Environ. Sci. 2021, 664, 012079. [Google Scholar] [CrossRef]
- Zulkifli, M.; Abu Hasan, H.; Sheikh Abdullah, S.R.; Muhamad, M.H. A review of ammonia removal using a biofilm-based reactor and its challenges. J. Environ. Manag. 2022, 315, 115162. [Google Scholar] [CrossRef]
- Iovino, P.; Fenti, A.; Galoppo, S.; Najafinejad, M.S.; Chianese, S.; Musmarra, D. Electrochemical Removal of Nitrogen Compounds from a Simulated Saline Wastewater. Molecules 2023, 28, 1306. [Google Scholar] [CrossRef]
- Dong, Y.; Lin, H.; Zhang, X. Simultaneous ammonia nitrogen and phosphorus removal from micro-polluted water by biological aerated filters with different media. Water Air Soil Pollut. 2020, 231, 1–15. [Google Scholar] [CrossRef]
- He, P.; Zhang, Y.; Zhang, X.; Chen, H. Diverse zeolites derived from a circulating fluidized bed fly ash based geopolymer for the adsorption of lead ions from wastewater. J. Clean Prod. 2021, 312, 127769. [Google Scholar] [CrossRef]
- Xiong, J.; Zheng, Z.; Yang, X.; Jian, H.; Luo, X.; Gao, B. Mature landfill leachate treatment by the MBBR inoculated with biocarriers from a municipal wastewater treatment plant. Process Saf. Environ. Prot. 2018, 119, 304–310. [Google Scholar] [CrossRef]
- Chaudhary, R.; Barwal, A. To study the performance of biocarriers in moving bed biofilm reactor (MBBR) technology and kinetics of biofilm for retrofitting the existing aerobic treatment systems: A review. Rev. Env. Sci. Biotechnol. 2014, 13, 285–299. [Google Scholar]
- Bakr, M.H.; Nasr, M.; Ashmawy, M.; Tawfik, A. Predictive performance of auto-aerated immobilized biomass reactor treating anaerobic effluent of cardboard wastewater enriched with bronopol (2-bromo-2-nitropropan-1,3-diol) via artificial neural network. Environ. Technol. Innov. 2021, 21, 101327. [Google Scholar] [CrossRef]
- Arabgol, R.; Vanrolleghem, P.A.; Delatolla, R. Influence of MBBR carrier geometrical properties and biofilm thickness restraint on biofilm properties, effluent particle size distribution, settling velocity distribution, and settling behaviour. J. Environ. Sci. 2022, 122, 138–149. [Google Scholar] [CrossRef]
- Zhu, J.; You, H.; Li, Z.; Ding, Y.; Ma, B. Impacts of bio-carriers on the characteristics of soluble microbial products in a hybrid membrane bioreactor for treating mariculture wastewater. Sci. Total Environ. 2020, 737, 140287. [Google Scholar] [CrossRef]
- Nasr, M.; Attia, M.; Ezz, H.; Ibrahim, M.G. Chapter 6—Recent applications of downflow hanging sponge technology for decentralized wastewater treatment. In Cost Effective Technologies for Solid Waste and Wastewater Treatment; Kathi, S., Devipriya, S., Thamaraiselvi, K., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 59–67. [Google Scholar]
- Ismail, S.; Nasr, M.; Abdelrazek, E.; Awad, H.M.; Zhaof, S.; Meng, F.; Tawfik, A. Techno-economic feasibility of energy-saving self-aerated sponge tower combined with up-flow anaerobic sludge blanket reactor for treatment of hazardous landfill leachate. J. Water Process. Eng. 2020, 37, 101415. [Google Scholar] [CrossRef]
- Dong, Z.; Lu, M.; Huang, W.; Xu, X. Treatment of oilfield wastewater in moving bed biofilm reactors using a novel suspended ceramic biocarrier. J. Hazard. Mater. 2011, 196, 123–130. [Google Scholar] [CrossRef]
- Zhou, A.; Jin, N.; Wei, Z.; Jiang, N.; Xiang, H. Experimental research onnitrogen removal by simultaneous nitrification and denitrificationin bio-ceramic filled MBBR. Ind. Water Wastewater 2010, 41, 30–34. [Google Scholar]
- Huang, W.; She, Z.; Gao, M.; Wang, Q.; Jin, C.; Zhao, Y.; Guo, L. Effect of anaerobic/aerobic duration on nitrogen removal and microbial community in a simultaneous partial nitrification and denitrification system under low salinity. Sci. Total Environ. 2019, 651, 859–870. [Google Scholar] [CrossRef]
- Vadivelu, V.M.; Keller, J.; Yuan, Z. Free ammonia and free nitrous acid inhibition on the anabolic and catabolic processes of Nitrosomonas and Nitrobacter. Water Sci. Technol. 2007, 56, 89–97. [Google Scholar] [CrossRef]
- Lai, C.; Guo, Y.; Cai, Q.; Yang, P. Enhanced nitrogen removal by simultaneous nitrification-denitrification and further denitrification (SND-DN) in a moving bed and constructed wetland (MBCW) integrated bioreactor. Chemosphere 2020, 261, 127744. [Google Scholar] [CrossRef]
- Yu, H.; Tian, Z.; Zuo, J.; Song, Y. Enhanced nitrite accumulation under mainstream conditions by a combination of free ammonia-based sludge treatment and low dissolved oxygen: Reactor performance and microbiome analysis. RSC Adv. 2020, 10, 2049–2059. [Google Scholar] [CrossRef] [Green Version]
- Al-Hazmi, H.E.; Lu, X.; Grubba, D.; Majtacz, J.; Badawi, M.; Mąkinia, J. Sustainable nitrogen removal in anammox-mediated systems: Microbial metabolic pathways, operational conditions and mathematical modelling. Sci. Total Environ. 2023, 868, 161633. [Google Scholar] [CrossRef]
- Chen, J.; Xie, Y.; Sun, S.; Zhang, M.; Yan, P.; Xu, F.; Tang, L.; He, S. Efficient nitrogen removal through coupling biochar with zero-valent iron by different packing modes in bioretention system. Environ. Res. 2023, 223, 115375. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, X.J.; Zhou, S.W.; Chen, Z.G. Effect of alkalinity on bio-zeolite regeneration in treating cold low-strength ammonium wastewater via adsorption and enhanced regeneration. Environ. Sci. Pollut. R. 2019, 26, 23–38. [Google Scholar] [CrossRef] [PubMed]
- Miladinovic, N.; Weatherley, L.R. Intensification of ammonia removal in a combined ion-exchange and nitrification column. Chem. Eng. J. 2008, 135, 15–24. [Google Scholar] [CrossRef]
- Wang, L.; Shao, Y.; Zhao, Z.; Chen, S.; Shao, X. Optimized utilization studies of dredging sediment for making water treatment ceramsite based on an extreme vertex design. J. Water Process. Eng. 2020, 38, 101603. [Google Scholar] [CrossRef]
- Furumai, H.; Tagui, H.; Fujita, K. Effects of pH and alkalinity on sulfur-denitrification in a biological granular filter. Water Sci. Technol. 1996, 34, 355–362. [Google Scholar] [CrossRef]
- Yang, X.; He, J.; Jiang, T. Effects of aeration volume and alkalinity on simultaneous nitrification and denitrification (SND) of aerobic granular sludge sequencing batch reactor. J. Biotechnol. 2008, 136, S664–S665. [Google Scholar] [CrossRef]
- Subtil, E.L.; Silva, M.V.; Lotto, B.A.; Moretto, M.R.D.; Mierzwa, J.C. Pilot-scale investigation on the feasibility of simultaneous nitrification and denitrification (SND) in a continuous flow single-stage membrane bioreactor. J. Water Process Eng. 2019, 32, 145–160. [Google Scholar] [CrossRef]
- Markou, G.; Vandamme, D.; Muylaert, K. Using natural zeolite for ammonia sorption from wastewater and as nitrogen releaser for the cultivation of Arthrospira platensis. Bioresour. Technol. 2014, 155, 373–378. [Google Scholar] [CrossRef] [Green Version]
- Wang, M.; Xie, R.; Chen, Y.; Pu, X.; Jiang, W.; Yao, L. A novel mesoporous zeolite-activated carbon composite as an effective adsorbent for removal of ammonia-nitrogen and methylene blue from aqueous solution. Bioresour. Technol. 2018, 268, 726–732. [Google Scholar] [CrossRef]
- Cao, X.; Jiang, L.; Zheng, H.; Liao, Y.; Zhang, Q.; Shen, Q.; Mao, Y.; Ji, F.; Shi, D. Constructed wetlands for rural domestic wastewater treatment: A coupling of tidal strategy, in-situ bio-regeneration of zeolite and Fe(Ⅱ)-oxygen denitrification. Bioresour. Technol. 2022, 344, 126185. [Google Scholar] [CrossRef]
- Han, Z.; Dong, J.; Shen, Z.; Mou, R.; Zhou, Y.; Chen, X.; Fu, X.; Yang, C. Nitrogen removal of anaerobically digested swine wastewater by pilot-scale tidal flow constructed wetland based on in-situ biological regeneration of zeolite. Chemosphere 2019, 217, 364–373. [Google Scholar] [CrossRef]
- Castro, C.J.; Shyu, H.Y.; Xaba, L.; Bair, R.; Yeh, D.H. Performance and onsite regeneration of natural zeolite for ammonium removal in a field-scale non-sewered sanitation system. Sci. Total Environ. 2021, 776, 145938. [Google Scholar] [CrossRef]
- Muscarella, S.M.; Badalucco, L.; Cano, B.; Laudicina, V.A.; Mannina, G. Ammonium adsorption, desorption and recovery by acid and alkaline treated zeolite. Bioresour. Technol. 2021, 341, 125812. [Google Scholar] [CrossRef]
- Liu, J.; Yuan, Y.; Li, B.; Zhang, Q.; Wu, L.; Li, X.; Peng, Y. Enhanced nitrogen and phosphorus removal from municipal wastewater in an anaerobic-aerobic-anoxic sequencing batch reactor with sludge fermentation products as carbon source. Bioresour. Technol. 2017, 244, 1158–1165. [Google Scholar] [CrossRef]
- Perera, M.K.; Englehardt, J.D.; Tchobanoglous, G.; Shamskhorzani, R. Control of nitrification/denitrification in an onsite two-chamber intermittently aerated membrane bioreactor with alkalinity and carbon addition: Model and experiment. Water Res. 2017, 115, 94–110. [Google Scholar] [CrossRef] [Green Version]
- Pan, D.; Shao, S.; Zhong, J.; Wang, M.; Wu, X. Performance and mechanism of simultaneous nitrification–denitrification and denitrifying phosphorus removal in long-term moving bed biofilm reactor (MBBR). Bioresour. Technol. 2022, 348, 126726. [Google Scholar] [CrossRef]
- Jiménez, E.; Giménez, J.B.; Seco, A.; Ferrer, J.; Serralta, J. Effect of pH, substrate and free nitrous acid concentrations on ammonium oxidation rate. Bioresour. Technol. 2012, 124, 12–24. [Google Scholar] [CrossRef]
- Wang, Z.; Zheng, M.; Xue, Y.; Xia, J.; Zhong, H.; Ni, G.; Liu, Y.; Yuan, Z.; Hu, S. Free ammonia shock treatment eliminates nitrite-oxidizing bacterial activity for mainstream biofilm nitritation process. Chem. Eng. J. 2019, 7, 666–670. [Google Scholar] [CrossRef]
- Anthonisen, A.C.; Loehr, R.C.; Prakasam, T.B.S.; Srinath, E.G. Inhibition of nitrification by ammonia and nitrous acid. Water Pollut. Control. Fed. 1976, 48, 10–25. [Google Scholar]
- Kent, T.R.; Sun, Y.W.; An, Z.H.; Bott, C.B.; Wang, Z.W. Mechanistic understanding of the NOB suppression by free ammonia inhibition in continuous flow aerobic granulation bioreactors. Environ. Int. 2019, 131, 51–63. [Google Scholar] [CrossRef]
- Torresi, E.; Casas, M.E.; Polesel, F.; Plósz, B.; Christensson, M.; Kai, B. Impact of external carbon dose on the removal of micropollutants using methanol and ethanol in post-denitrifying Moving Bed Biofilm Reactors. Water Res. 2017, 1, 124–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, Y.; Zhou, M.; Chen, Y.; Hu, Y.; Luo, J. Insight into short-cut of simultaneous nitrification and denitrification process in moving bed biofilm reactor: Effects of carbon to nitrogen ratio. Chem. Eng. J. 2020, 400, 125905. [Google Scholar] [CrossRef]
- Ming, F.; Jiao, H.; Xiao, Y.; Wen, J.; Xing, Y. Study on the effects of technological factors of shortcut nitrification-denitrification simultaneous phosphorus removal. Adv. Mater. Res. 2014, 3181, 156–171. [Google Scholar]
- Huang, R.; Meng, T.; Liu, G.; Gao, S.; Tian, J. Simultaneous nitrification and denitrification in membrane bioreactor: Effect of dissolved oxygen. J. Environ. Manag. 2022, 323, 116183. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Rong, H.; Cao, Y.; Zhang, C. Factors affecting simultaneous nitrification and denitrification (SND) in a moving bed sequencing batch reactor (MBSBR) system as revealed by microbial community structures. Bioprocess Biosyst. Eng. 2020, 12, 623–630. [Google Scholar] [CrossRef]
- Huang, X.W.; Lee, P.H. Shortcut nitrification/denitrification through limited-oxygen supply with two extreme COD/N-and-ammonia active landfill leachates. Chem. Eng. J. 2021, 404, 124–131. [Google Scholar] [CrossRef]
- Zhu, G.C.; Lu, Y.Z.; Xu, L.R. Effects of the carbon/nitrogen (C/N) ratio on a system coupling simultaneous nitrification and denitrification (SND) and denitrifying phosphorus removal (DPR). Environ. Technol. 2020, 34, 1156–1162. [Google Scholar] [CrossRef]
- Luan, Y.; Yin, Y.; An, Y.; Zhang, F.; Wang, X.; Zhao, F.; Xiao, Y.; Liu, C. Investigation of an intermittently-aerated moving bed biofilm reactor in rural wastewater treatment under low dissolved oxygen and C/N condition. Bioresour. Technol. 2022, 358, 127405. [Google Scholar] [CrossRef]
- Ashkanani, A.; Almomani, F.; Khraisheh, M.; Bhosale, R.; Tawalbeh, M.; AlJaml, K. Bio-carrier and operating temperature effect on ammonia removal from secondary wastewater effluents using moving bed biofilm reactor (MBBR). Sci. Total Environ. 2019, 693, 133425. [Google Scholar] [CrossRef]
- Zhang, H.; Li, A.; Zhang, W.; Shuang, C. Combination of Na-modified zeolite and anion exchange resin for advanced treatment of a high ammonia–nitrogen content municipal effluent. J. Colloid Interface Sci. 2016, 468, 128–135. [Google Scholar] [CrossRef]
- Song, Z.; Zhang, X.; Ngo, H.H.; Guo, W.; Song, P.; Zhang, Y.; Wen, H.; Guo, J. Zeolite powder based polyurethane sponges as biocarriers in moving bed biofilm reactor for improving nitrogen removal of municipal wastewater. Sci. Total Environ. 2019, 651, 1078–1086. [Google Scholar] [CrossRef]
- Singh, V.; Ormeci, B.; Mishra, S.; Hussain, A. Simultaneous partial Nitrification, ANAMMOX and denitrification (SNAD)—A review of critical operating parameters and reactor configurations. Chem. Eng. J. 2022, 433, 133677. [Google Scholar] [CrossRef]
- Di Capua, F.; Iannacone, F.; Sabba, F.; Esposito, G. Simultaneous nitrification–denitrification in biofilm systems for wastewater treatment: Key factors, potential routes, and engineered applications. Bioresour. Technol. 2022, 361, 127702. [Google Scholar] [CrossRef]
- Sun, H.; Jiang, T.; Zhang, F.; Zhang, P.; Zhang, H.; Yang, H.; Lu, J.; Ge, S.; Ma, B.; Ding, J.; et al. Understanding the effect of free ammonia on microbial nitrification mechanisms in suspended activated sludge bioreactors. Environ. Res. 2021, 200, 111737. [Google Scholar] [CrossRef]




| Primary Nutrients | Trace Elements | ||
|---|---|---|---|
| Component | Concentration (mg/L) | Component | Concentration (mg/L) |
| 100 | CuSO4 | 0.005 | |
| glucose | 167 | CoCl2·6H2O | 0.021 |
| NaHCO3 | 107 | Na2MoO4·2H2O | 0.016 |
| KH2PO4 | 10.2 | ZnSO4·7H2O | 0.041 |
| FeSO4·7H2O | 2.51 | H3BO3 | 0.15 |
| MgSO4·7H2O | 5.71 | MnCl·4H2O | 0.211 |
| CaCl2·7H2O | 1.54 | Vitamin D | 0.0002 |
| Parameters | HRT = 12 h | HRT = 8 h | HRT = 6 h |
|---|---|---|---|
| Influent -N concentration (mg/L) | 26.18 ± 0.11 | 26.20 ± 0.08 | 26.24 ± 0.12 |
| Adsorption breakthrough time (h) | 34 | 20 | 14 |
| Adsorption breakthrough concentration (mg/L) | 5.40 ± 0.65 | 5.01 ± 0.34 | 5.82 ± 0.44 |
| Integral value of adsorption curve (mg·h/L) | 65.46 | 26.49 | 17.91 |
| Input flow rate (L/h) | 0.5 | 0.75 | 1 |
| Cumulative -N effluent (mg) | 32.73 | 19.87 | 17.91 |
| Cumulative -N influent (mg) | 602.14 ± 1.72 | 550.20 ± 1.57 | 524.80 ± 1.13 |
| Unit adsorption quantity (mg/g) | 1.0910 ± 0.0023 a | 1.0143 ± 0.0016 b | 0.9566 ± 0.0011 c |
| -N volume load (mg/(L·h)) | 0.7706 ± 0.0019 a | 1.3100 ± 0.0023 b | 1.8714 ± 0.0035 c |
| Parameters | 60 r/min | 90 r/min | 120 r/min | 150 r/min |
|---|---|---|---|---|
| Influent -N concentration (mg/L) | 26.22 ± 0.07 | 26.19 ± 0.08 | 26.15 ± 0.11 | 26.16 ± 0.10 |
| HRT (h) | 8 | 8 | 8 | 8 |
| Adsorption breakthrough time (h) | 18 | 20 | 22 | 24 |
| Adsorption breakthrough concentration (mg/L) | 5.21 ± 0.17 | 5.03 ± 0.14 | 5.08 ± 0.20 | 5.89 ± 0.15 |
| Integral value of adsorption curve (mg·h/L) | 25.18 | 27.29 | 28.43 | 32.78 |
| Cumulative -N effluent (mg) | 18.89 | 20.47 | 21.32 | 24.59 |
| Cumulative -N influent (mg) | 511.29 ± 1.15 | 549.99 ± 1.46 | 588.38 ± 2.09 | 627.84 ± 2.07 |
| Unit adsorption quantity (mg/g) | 0.9354 ± 0.0016 a | 1.0143 ± 0.0024 b | 1.0912 ± 0.0038 c | 1.1546 ± 0.0045 d |
| -N volume load (mg/(L·h)) | 1.4556 ± 0.0032 a | 1.3100 ± 0.0028 b | 1.1909 ± 0.0019 c | 1.0917 ± 0.0015 d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Mao, X.; Hamoud, Y.A.; Zhu, N.; Shao, X.; Wang, Q.; Shaghaleh, H. Nitrogen Removal for Low Concentration Ammonium Wastewater by Adsorption, Shortcut Simultaneous Nitrification and Denitrification Process in MBBR. Water 2023, 15, 1334. https://doi.org/10.3390/w15071334
Wang L, Mao X, Hamoud YA, Zhu N, Shao X, Wang Q, Shaghaleh H. Nitrogen Removal for Low Concentration Ammonium Wastewater by Adsorption, Shortcut Simultaneous Nitrification and Denitrification Process in MBBR. Water. 2023; 15(7):1334. https://doi.org/10.3390/w15071334
Chicago/Turabian StyleWang, Liangkai, Xinyu Mao, Yousef Alhaj Hamoud, Ningyuan Zhu, Xiaohou Shao, Qilin Wang, and Hiba Shaghaleh. 2023. "Nitrogen Removal for Low Concentration Ammonium Wastewater by Adsorption, Shortcut Simultaneous Nitrification and Denitrification Process in MBBR" Water 15, no. 7: 1334. https://doi.org/10.3390/w15071334







