Photocatalytic Decolorization of Direct Red16 from an Aqueous Solution Using B-ZnO/TiO2 Nano Photocatalyst: Synthesis, Characterization, Process Modeling, and Optimization
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Synthesis of TiO2, B-TiO2, ZnO/TiO2, and B-ZnO/TiO2
2.2.1. TiO2
2.2.2. B-TiO2
2.2.3. ZnO/TiO2
2.2.4. B-ZnO/TiO2
2.3. Instruments for Analysis and Characterizations
2.4. Adsorption Activity Experiments
2.5. Photocatalytic Activity Experiments
2.6. Experimental Design Methodology
3. Results and Discussion
3.1. Determination of B and ZnO Amounts in B-ZnO/TiO2 Nanocomposite
- An ability to absorb visible light;
- B-doped TiO2 forms two new chemical bonds B–O–Ti and B–O–B, which are beneficial for photodegradation;
- A decrease in particle size and increase in the surface area and active sites by doping B into TiO2;
- Formation of a mesoporous structure;
- Improving the microstructure properties of TiO2 due to occupying of interstitial positions of TiO2 by B;
- Formation of Ti (III), with its beneficial effect in reducing the recombination of photoexcited electrons and holes [23].
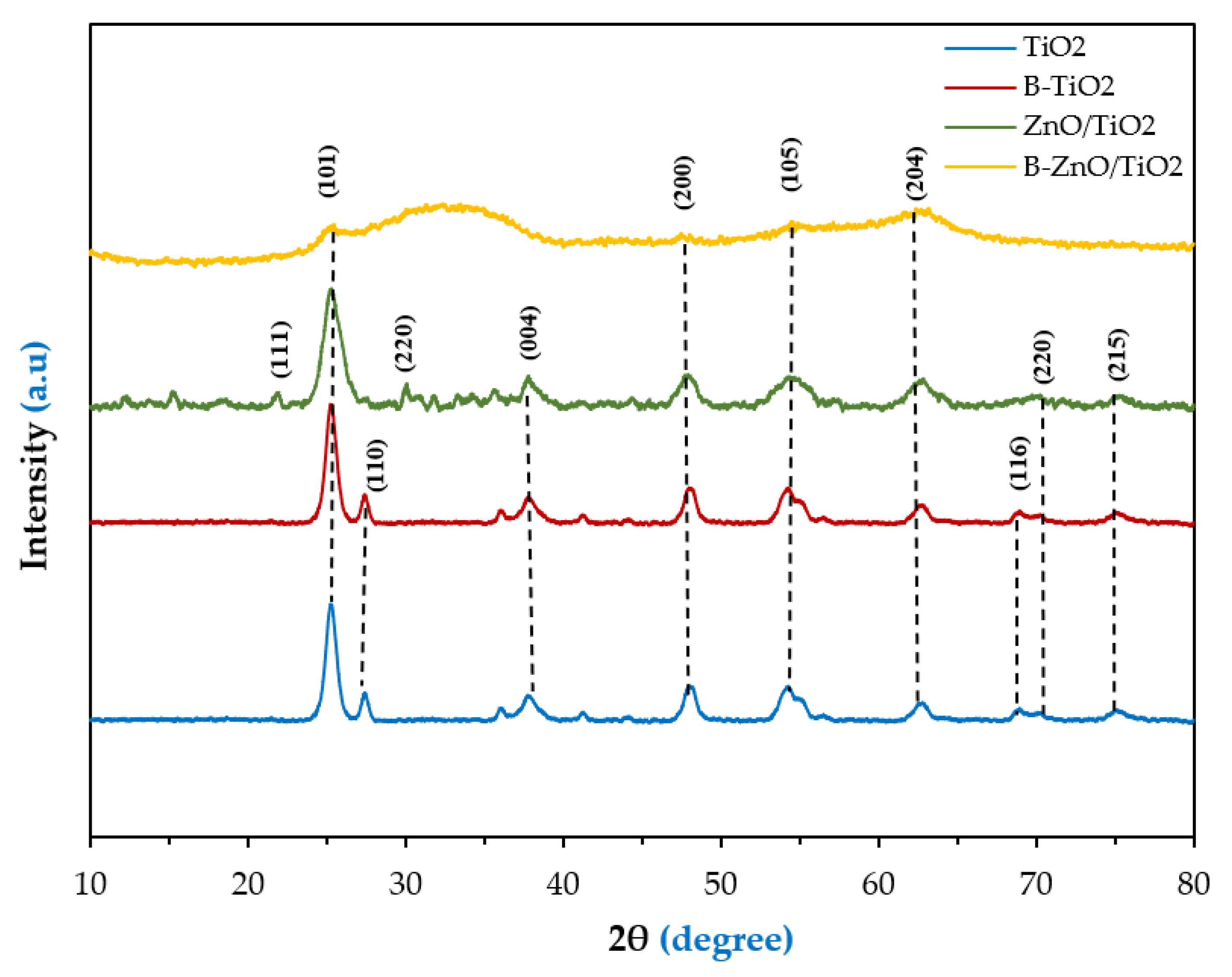
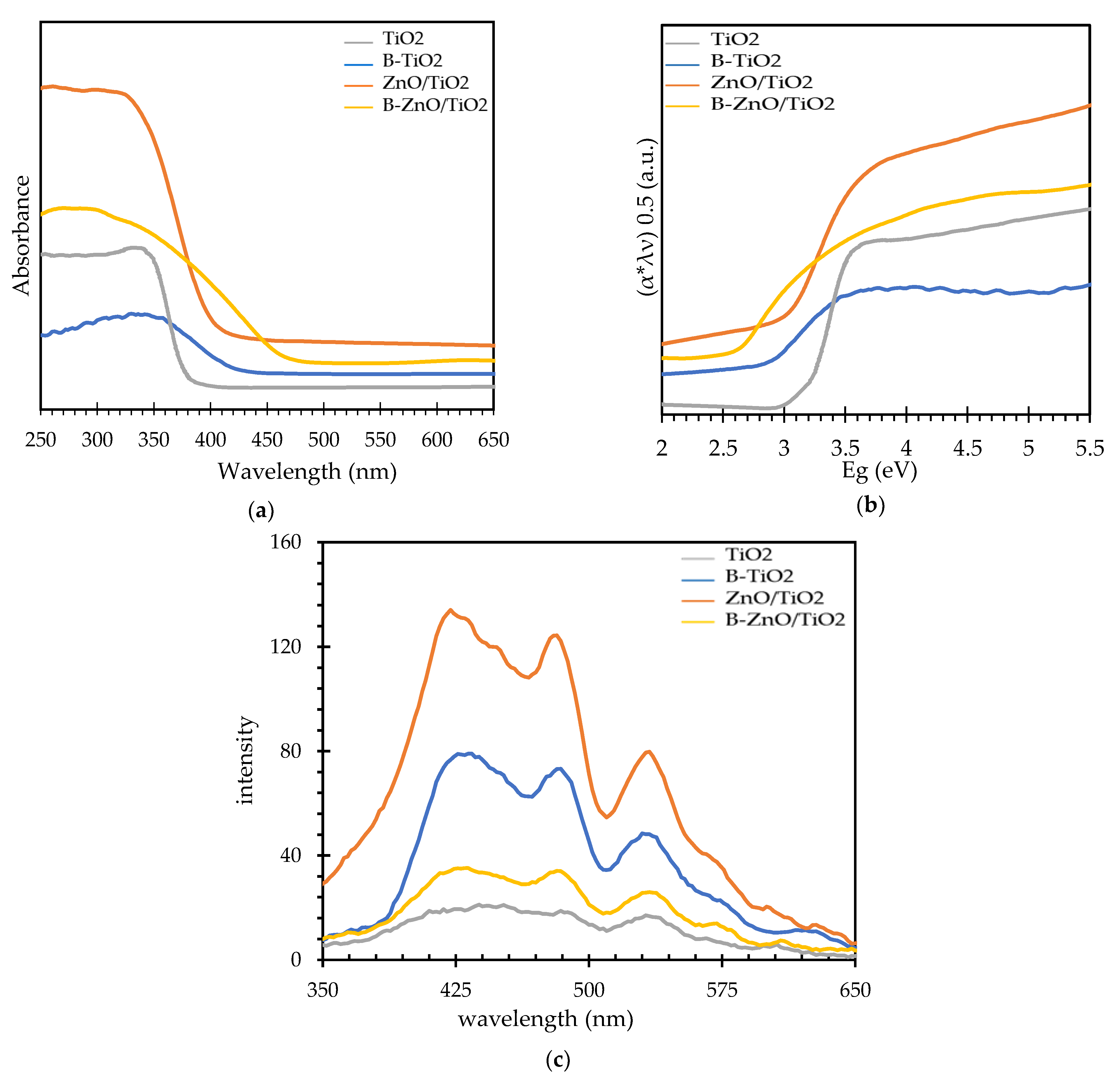
3.2. Characterization of the Synthesized B-ZnO/TiO2
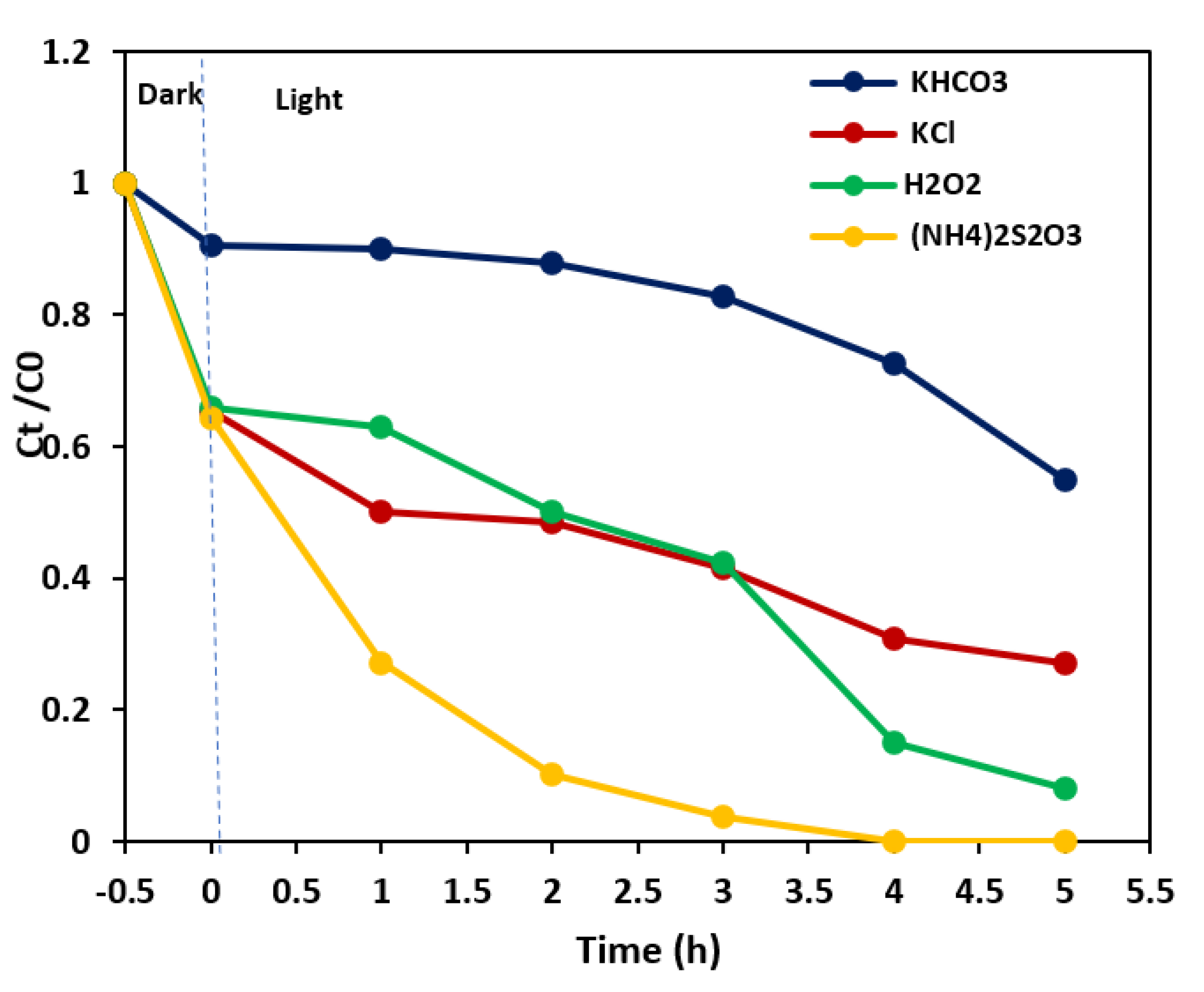
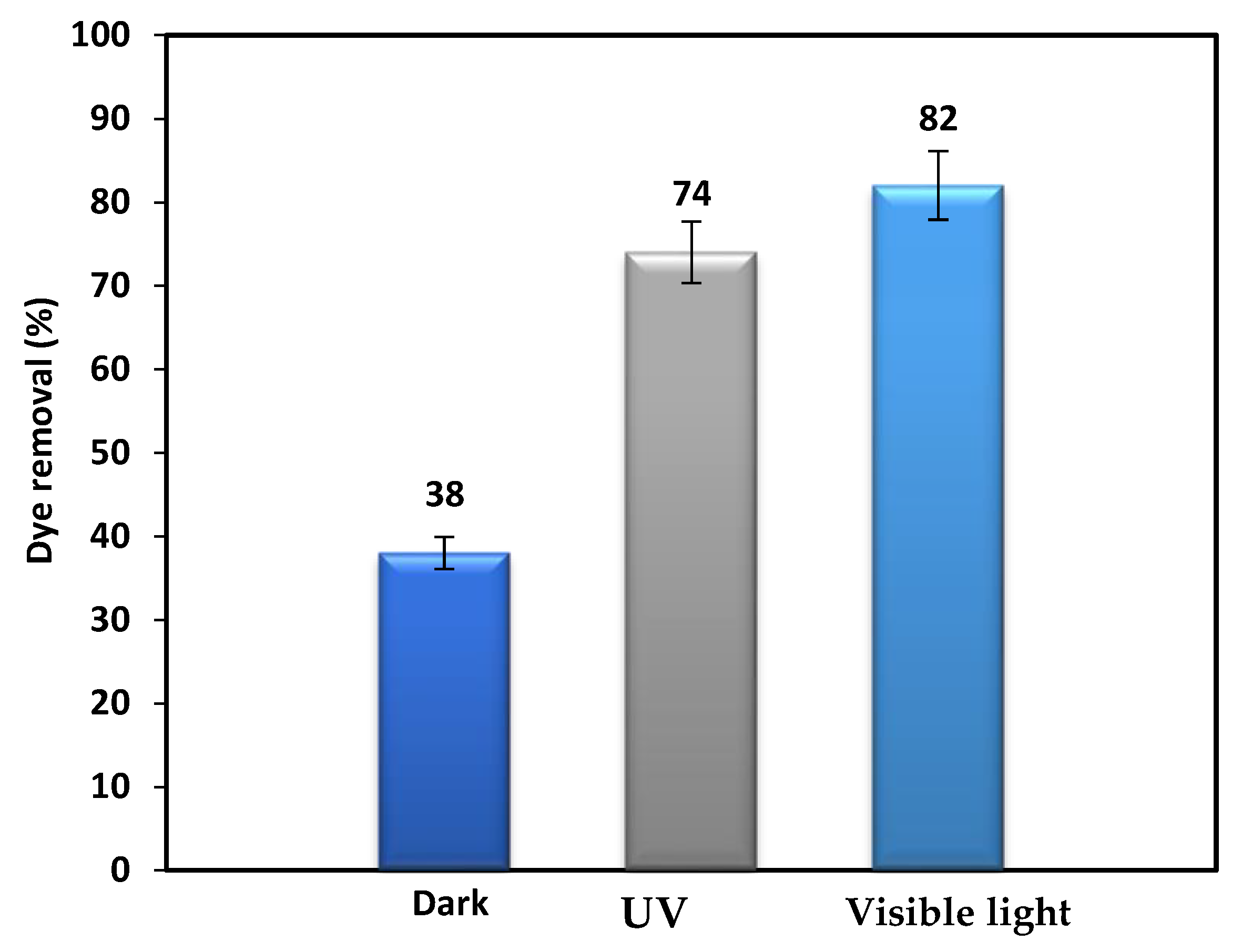
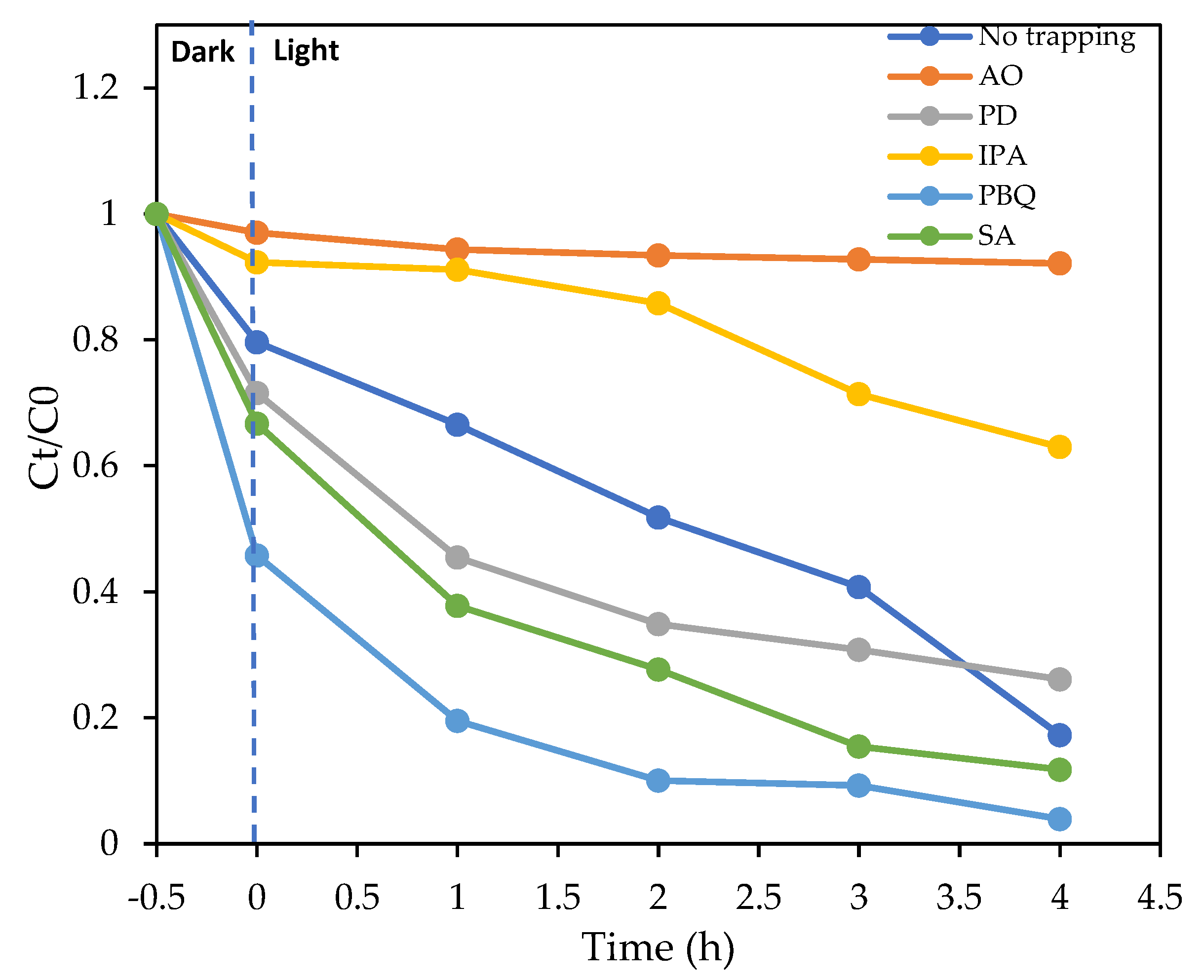
3.3. Effect of Electron Acceptors and Type of Irradiation on the Photodegradation Process
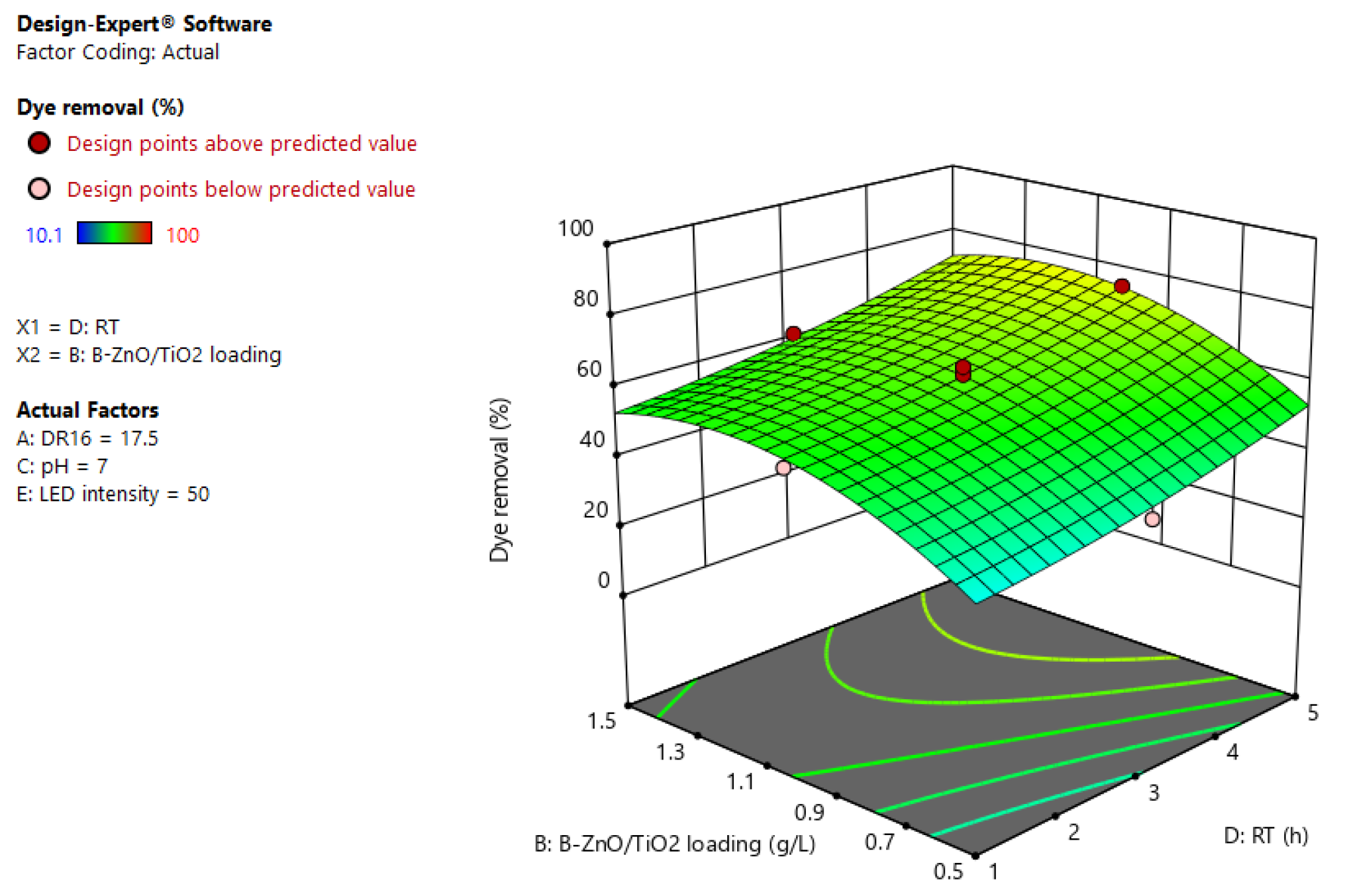
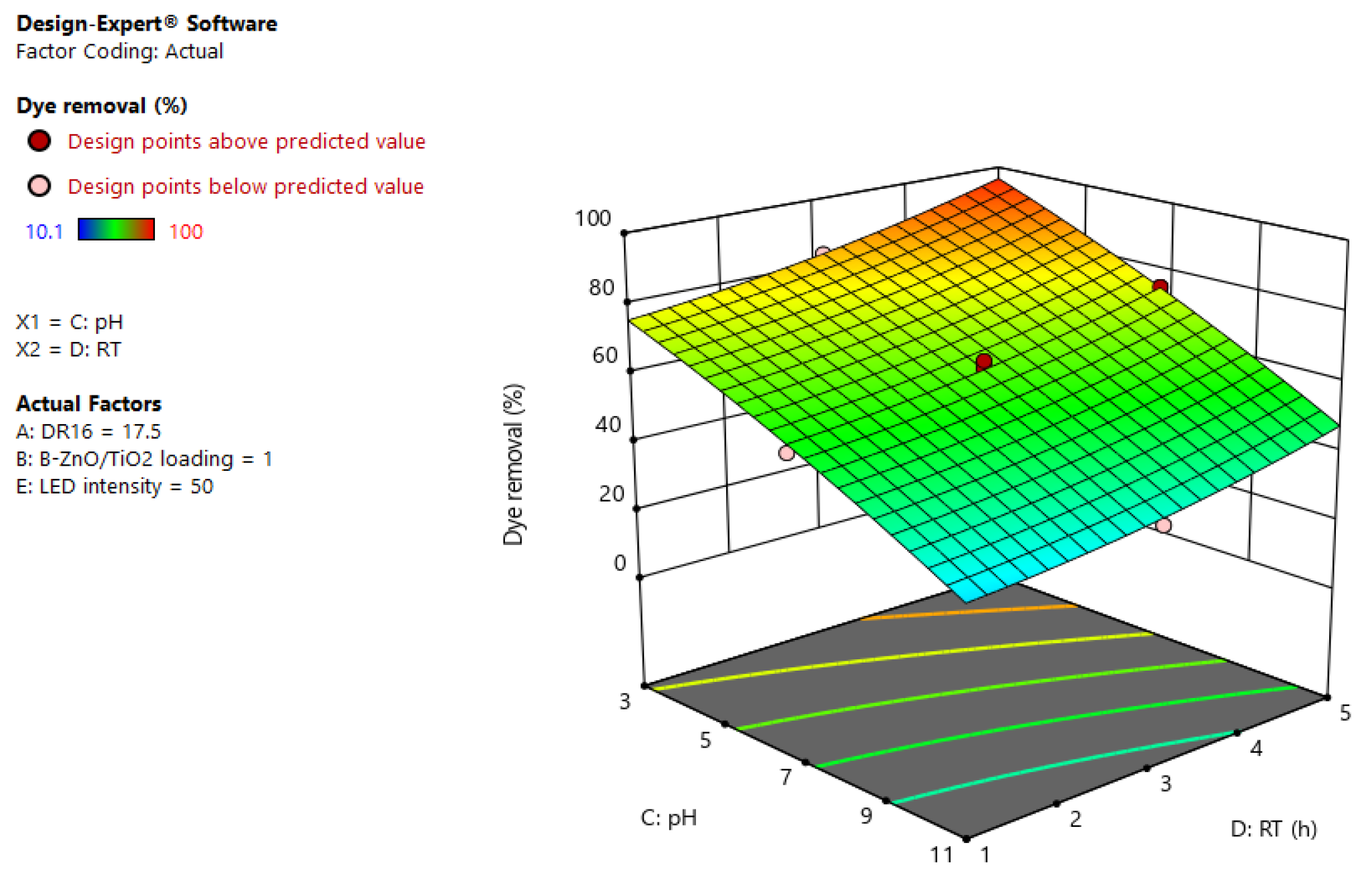
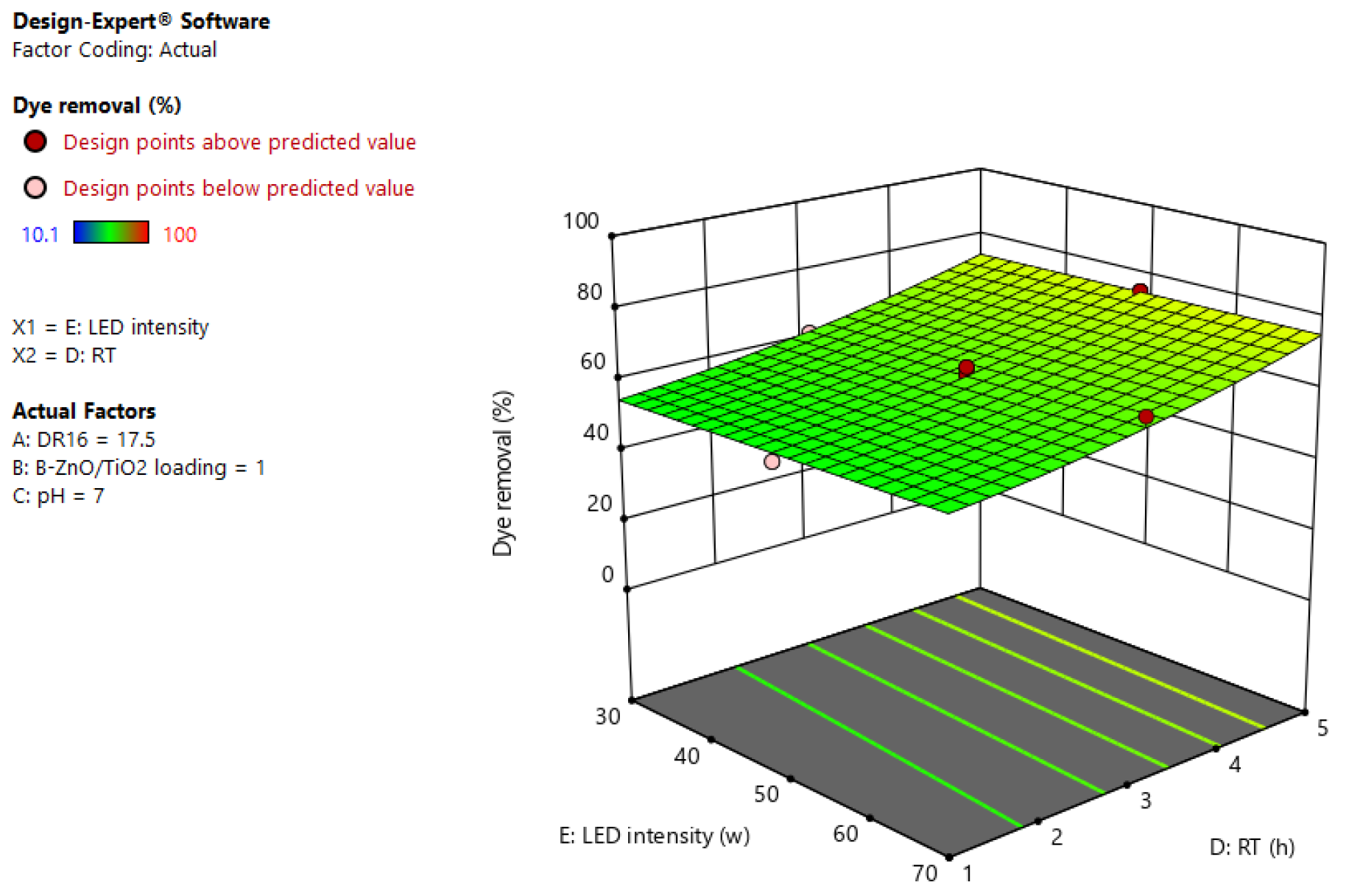
3.4. Photodegradation Process Modeling and Optimization
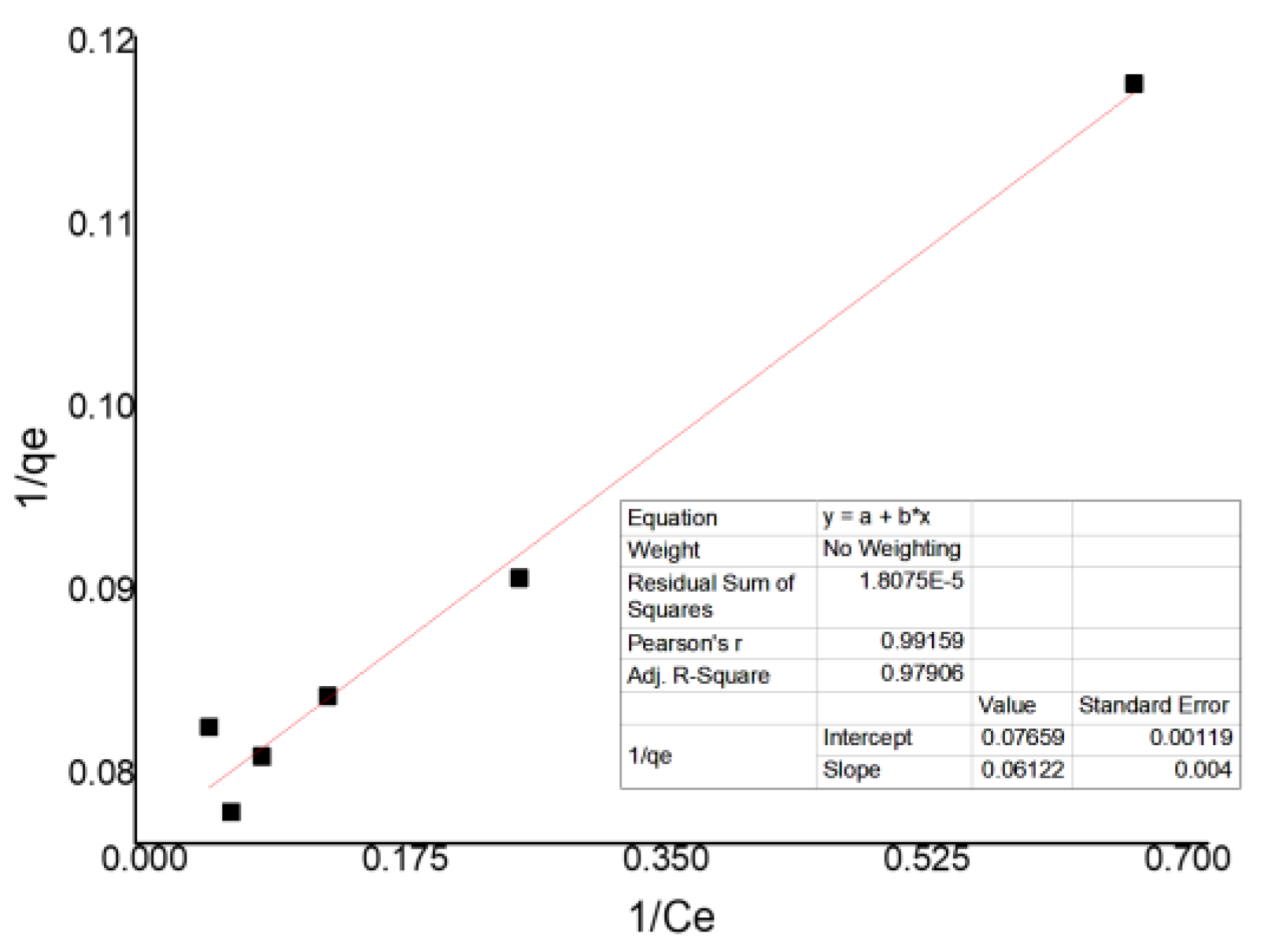
3.5. Evaluation of B-ZnO/TiO2 Isothermal Adsorption
3.6. The Mechanism of Photodegradation of DR16 under Visible Light
3.7. Reusability of B-ZnO/TiO2
3.8. Comparative Study
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boumaza, S.; Kaouah, F.; Hamane, D.; Trari, M.; Omeiri, S.; Bendjama, Z. Visible light assisted decolorization of azo dyes: Direct Red 16 and Direct Blue 71 in aqueous solution on the p-CuFeO2/n-ZnO system. J. Mol. Catal. A Chem. 2014, 393, 156–165. [Google Scholar] [CrossRef]
- Farooq, U.; Chaudhary, P.; Ingole, P.P.; Kalam, A.; Ahmad, T. Development of cuboidal KNbO3@α-Fe2O3hybrid nanostructures for improved photocatalytic and photoelectrocatalytic applications. ACS Omega 2020, 5, 20491–20505. [Google Scholar] [CrossRef] [PubMed]
- Abdolmohammad-Zadeh, H.; Nejati, K.; Ghorbani, E. Synthesis, Characterization, and Application of Zn-Al Layered Double Hydroxide as a Nano-Sorbent for the Removal of Direct Red 16 from Industrial Wastewater Effluents. Chem. Eng. Commun. 2015, 202, 1349–1359. [Google Scholar] [CrossRef]
- Khan, I.; Saeed, K.; Zekker, I.; Zhang, B.; Hendi, A.H.; Ahmad, A.; Ahmad, S.; Zada, N.; Ahmad, H.; Shah, L.A.; et al. Review on Methylene Blue: Its Properties, Uses, Toxicity and Photodegradation. Water 2022, 14, 242. [Google Scholar] [CrossRef]
- Jamil, N.; Khan, M.; Ahsan, N.; Anwar, J.; Qadir, A.; Zameer, M.; Shafique, U. Removal of direct red 16 (Textile Dye) from industrial effluent by using feldspar. J. Chem. Soc. Pakistan 2014, 36, 191–197. [Google Scholar]
- Soleymani, A.R.; Saien, J.; Chin, S.; Le, H.A.; Park, E.; Jurng, J. Modeling and optimization of a sono-assisted photocatalytic water treatment process via central composite design methodology. Process Saf. Environ. Prot. 2015, 94, 307–314. [Google Scholar] [CrossRef]
- Fard, B.H.; Khojasteh, R.R.; Gharbani, P. Photocatalytic degradation of direct red 16 dye using Ag/Ag3VO4/AgVO3/GO nanocomposite. S. Afr. J. Chem. 2020, 73, 1–8. [Google Scholar] [CrossRef]
- Naeem, H.T.; Hassan, A.A.; Al-Khateeb, R.T. Wastewater-(Direct red dye) treatment-using solar fenton process. J. Pharm. Sci. Res. 2018, 10, 2309–2313. [Google Scholar]
- Araujo, F.P.; Trigueiro, P.; Honório, L.M.C.; Oliveira, D.M.; Almeida, L.C.; Garcia, R.P.; Lobo, A.O.; Cantanhêde, W.; Silva-Filho, E.C.; Osajima, J.A. Eco-friendly synthesis and photocatalytic application of flowers-like ZnO structures using Arabic and Karaya Gums. Int. J. Biol. Macromol. 2020, 165, 2813–2822. [Google Scholar] [CrossRef]
- Farooq, U.; Naz, F.; Phul, R.; Pandit, N.A.; Jain, S.K.; Ahmad, T. Development of Heterostructured Ferroelectric SrZrO3/CdS Photocatalysts with Enhanced Surface Area and Photocatalytic Activity. J. Nanosci. Nanotechnol. 2019, 20, 3770–3779. [Google Scholar] [CrossRef]
- Ahmad, S.; Almehmadi, M.; Janjuhah, H.T.; Kontakiotis, G.; Abdulaziz, O.; Saeed, K.; Ahmad, H.; Allahyani, M.; Aljuaid, A.; Alsaiari, A.A.; et al. The Effect of Mineral Ions Present in Tap Water on Photodegradation of Organic Pollutants: Future Perspectives. Water 2023, 15, 175. [Google Scholar] [CrossRef]
- De Oliveira, W.V.; Morais, A.Í.S.; Honorio, L.M.C.; Trigueiro, P.A.; Almeida, L.C.; Garcia, R.R.P.; Viana, B.C.; Furtini, M.B.; Silva-Filho, E.C.; Osajima, J.A. TiO2 Immobilized on Fibrous Clay as Strategies to Photocatalytic Activity. Mat. Res. 2020, 23, 1439–1516. [Google Scholar] [CrossRef]
- Li, W.; Wu, D.; Yu, Y.; Zhang, P.; Yuan, J.; Cao, Y.; Cao, Y.; Xu, J. Investigation on a novel ZnO/TiO2-B photocatalyst with enhanced visible photocatalytic activity. Phys. E Low-Dimens. Syst. Nanostruct. 2014, 58, 118–123. [Google Scholar] [CrossRef]
- Yang, C.C.; Dao, K.C.; Lin, Y.S.; Cheng, T.Y.; Chen, K.F.; Tsai, Y.P. Impacts of mixing mode on photocatalytic reduction of hexavalent chromium over titanium dioxide nanomaterial under various environmental conditions. Water 2021, 13, 2291. [Google Scholar] [CrossRef]
- Freitas, W.A.; Soares, B.E.C.F.; Rodrigues, M.S.; Trigueiro, P.; Honorio, L.M.C.; Peña-Garcia, R.; Alcântara, A.C.S.; Silva-Filho, E.C.; Fonseca, M.G.; Furtini, M.B.; et al. Facile synthesis of ZnO-clay minerals composites using an ultrasonic approach for photocatalytic performance. J. Photochem. Photobiol. A Chem. 2022, 429, 113934. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, X.; Guo, L.; Shang, L.; Ge, S.; Song, G.; Naik, N.; Shao, Q.; Lin, J.; Guo, Z. Metal organic framework-derived C-doped ZnO/TiO2 nanocomposite catalysts for enhanced photodegradation of Rhodamine B. J. Colloid Interface Sci. 2021, 599, 566–576. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Wang, J.; Dai, J.; Wang, X.; Yin, Y. N-doped TiO2/ZnO composite powder and its photocatalytic performance for degradation of methyl orange. Surf. Coat. Technol. 2009, 204, 723–730. [Google Scholar] [CrossRef]
- Bai, N.; Liu, X.; Li, Z.; Ke, X.; Zhang, K.; Wu, Q. High-efficiency TiO2/ZnO nanocomposites photocatalysts by sol–gel and hydrothermal methods. J. Sol-Gel Sci. Technol. 2021, 99, 92–100. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, Y. Sol-gel Synthesis of Boron Doped TiO2/hollow Glass Bubbles Composite Powders for Photocatalytic Degradation of Azophloxine. Curr. Nanosci. 2021, 17, 475–483. [Google Scholar] [CrossRef]
- Khan, R.; Kim, S.W.; Kim, T.J.; Nam, C.M. Comparative study of the photocatalytic performance of boron-iron Co-doped and boron-doped TiO2 nanoparticles. Mater. Chem. Phys. 2008, 112, 167–172. [Google Scholar] [CrossRef]
- Cano-Casanova, L.; Ansón-Casaos, A.; Hernández-Ferrer, J.; Benito, A.M.; Maser, W.K.; Garro, N.; Lillo-Ródenas, M.A.; Román-Martínez, M.C. Surface-Enriched Boron-Doped TiO2Nanoparticles as Photocatalysts for Propene Oxidation. ACS Appl. Nano Mater. 2022, 5, 12527–12539. [Google Scholar] [CrossRef] [PubMed]
- Moradi, S.; Aberoomand-Azar, P.; Raeis-Farshid, S.; Abedini-Khorrami, S.; Givianrad, M.H. The effect of different molar ratios of ZnO on characterization and photocatalytic activity of TiO2/ZnO nanocomposite. J. Saudi Chem. Soc. 2016, 20, 373–378. [Google Scholar] [CrossRef] [Green Version]
- Bakhtkhosh, P.; Mehrizad, A. Sonochemical synthesis of Sm-doped ZnS nanoparticles for photocatalytic degradation of Direct Blue 14: Experimental design by response surface methodology and development of a kinetics model. J. Mol. Liq. 2017, 240, 65–73. [Google Scholar] [CrossRef]
- Cavalcante, R.P.; Dantas, R.F.; Bayarri, B.; González, O.; Giménez, J.; Esplugas, S.; Machulek, A. Synthesis and characterization of B-doped TiO2 and their performance for the degradation of metoprolol. Catal. Today 2015, 252, 27–34. [Google Scholar] [CrossRef]
- Shathy, R.A.; Fahim, S.A.; Sarker, M.; Quddus, S.; Moniruzzaman, M.; Masum, S.M.; Molla, A.I. Natural Sunlight Driven Photocatalytic Removal of Toxic Textile Dyes in Water Using B-Doped ZnO/TiO2 Nanocomposites. Catalysts 2022, 12, 308. [Google Scholar] [CrossRef]
- Zou, X.; Dong, X.; Wang, L.; Ma, H.; Zhang, X.; Zhang, X. Preparation of ni doped ZnO-TiO2 composites and their enhanced photocatalytic activity. Int. J. Photoenergy 2014, 2014, 893158. [Google Scholar] [CrossRef] [Green Version]
- Ji, L.; Li, J.; Lei, J.; Ren, Y.; Zhou, S.; Liang, L. Preparation and characterization of Cu2+/ZnO/TiO2 nanocomposites for the treatment of typical benzene series in oilfield produced water. Catal. Commun. 2023, 174, 106572. [Google Scholar] [CrossRef]
- Nassar, M.Y.; Ali, A.A.; Amin, A.S. A facile Pechini sol-gel synthesis of TiO2/Zn2TiO2/ZnO/C nanocomposite: An efficient catalyst for the photocatalytic degradation of Orange G textile dye. RSC Adv. 2017, 7, 30411–30421. [Google Scholar] [CrossRef] [Green Version]
- Sienkiewicz, A.; Wanag, A.; Kusiak-Nejman, E.; Ekiert, E.; Rokicka-Konieczna, P.; Morawski, A.W. Effect of calcination on the photocatalytic activity and stability of TiO2 photocatalysts modified with APTES. J. Environ. Chem. Eng. 2021, 9, 104794. [Google Scholar] [CrossRef]
- Wanag, A.; Sienkiewicz, A.; Rokicka-Konieczna, P.; Kusiak-Nejman, E.; Morawski, A.W. Influence of modification of titanium dioxide by silane coupling agents on the photocatalytic activity and stability. J. Environ. Chem. Eng. 2020, 8, 103917. [Google Scholar] [CrossRef]
- Joseyphus, R.S.; Pushparajan, J.; Akansha, M.; Johnson, K.; Anjana, P.M.; Arish, D. Selective detection of Fe3+ ion using Zn2TiO4 through fluorescence quenching probes. Res. Sq. 2022. preprint. [Google Scholar] [CrossRef]
- Gebru, K.A.; Das, C. Removal of chromium (VI) ions from aqueous solutions using amine-impregnated TiO2 nanoparticles modified cellulose acetate membranes. Chemosphere 2018, 191, 673–684. [Google Scholar] [CrossRef]
- Liao, D.L.; Wu, G.S.; Liao, B.Q. Zeta potential of shape-controlled TiO2 nanoparticles with surfactants. Colloids Surfaces A Physicochem. Eng. Asp. 2009, 348, 270–275. [Google Scholar] [CrossRef]
- Pirinejad, L.; Maleki, A.; Shahmoradi, B.; Daraei, H.; Yang, J.K.; Lee, S.M. Synthesis and application of Fe-N-Cr-TiO2 nanocatalyst for photocatalytic degradation of Acid Black 1 under LED light irradiation. J. Mol. Liq. 2019, 279, 232–240. [Google Scholar] [CrossRef]
- Raguram, T.; Rajni, K.S. Influence of boron doping on the structural, spectral, optical and morphological properties of TiO2 nanoparticles synthesized by sol–gel technique for DSSC applications. Mater. Today Proc. 2019, 33, 2110–2115. [Google Scholar] [CrossRef]
- Jaiswal, R.; Patel, N.; Dashora, A.; Fernandes, R.; Yadav, M.; Edla, R.; Varma, R.; Kothari, D.; Ahuja, B.; Miotello, A. Efficient Co-B-codoped TiO2 photocatalyst for degradation of organic water pollutant under visible light. Appl. Catal. B Environ. 2016, 183, 242–253. [Google Scholar] [CrossRef]
- Patel, N.; Dashora, A.; Jaiswal, R.; Fernandes, R.; Yadav, M.; Kothari, D.C.; Ahuja, B.L.; Miotello, A. Experimental and Theoretical Investigations on the Activity and Stability of Substitutional and Interstitial Boron in TiO2 Photocatalyst. J. Phys. Chem. C 2015, 119, 18581–18590. [Google Scholar] [CrossRef]
- Pozan, G.S.; Kambur, A. Significant enhancement of photocatalytic activity over bifunctional ZnO-TiO2 catalysts for 4-chlorophenol degradation. Chemosphere 2014, 105, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Vučić, M.D.R.; Mitrović, J.Z.; Kostić, M.M.; Velinov, N.D.; Najdanović, S.M.; Bojić, D.V.; Bojić, A.L. Heterogeneous photocatalytic degradation of anthraquinone dye reactive blue 19: Optimization, comparison between processes and identification of intermediate products. Water SA 2020, 46, 291–299. [Google Scholar] [CrossRef]
- Zebardast, M.; Shojaei, A.F.; Tabatabaeian, K. Enhanced removal of methylene blue dye by bimetallic nano-sized MOF-5s. Iran. J. Catal. 2018, 8, 297–309. [Google Scholar]
- Bouanimba, N.; Laid, N.; Zouaghi, R.; Sehili, T. Effect of pH and inorganic salts on the photocatalytic decolorization of methyl orange in the presence of TiO2 P25 and PC500. Desalin. Water Treat. 2015, 53, 951–963. [Google Scholar] [CrossRef]
- Guillard, C.; Lachheb, H.; Houas, A.; Ksibi, M.; Elaloui, E.; Herrmann, J.M. Influence of chemical structure of dyes, of pH and of inorganic salts on their photocatalytic degradation by TiO2 comparison of the efficiency of powder and supported TiO2. J. Photochem. Photobiol. A Chem. 2003, 158, 27–36. [Google Scholar] [CrossRef]
- Zangeneh, H.; Farhadian, M.; Zinatizadeh, A.A. A reusable visible driven N and C-N doped TiO2 magnetic nanocomposites for photodegradation of direct red 16 azo dye in water and wastewater. Environ. Technol. 2022, 43, 1269–1284. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid Interface Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Ani, I.J.; Akpan, U.G.; Olutoye, M.A.; Hameed, B.H. Photocatalytic degradation of pollutants in petroleum refinery wastewater by TiO2- and ZnO-based photocatalysts: Recent development. J. Clean. Prod. 2018, 205, 930–954. [Google Scholar] [CrossRef]







| Parameters | Range and Levels | ||
|---|---|---|---|
| −1 | 0 | +1 | |
| A: DR16 concentration (ppm) | 10 | 17.5 | 25 |
| B: B-ZnO/TiO2 loading (g/L) | 0.5 | 1 | 1.5 |
| C: pH | 3 | 7 | 11 |
| D: Retention time (h) | 1 | 3 | 5 |
| E: Light (LED) intensity (W) | 30 | 50 | 70 |
| Catalyst | D, nm | Average, nm | Max., nm | Min., nm | Mode, nm | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| XRD | SEM | XRD | SEM | XRD | SEM | XRD | SEM | XRD | SEM | |
| TiO2 | 9.6 | >12 | 11.5 | 11.1 | 10.8 | 23.7 | 5.8 | 4.3 | 9.5 | 11 |
| B-TiO2 | 6.9 | >12 | 8.9 | 11.7 | 25.4 | 11.7 | 5 | 5.7 | 6.6 | 10.5 |
| ZnO-TiO2 | 5.8 | >12 | 11.4 | 11.4 | 31.7 | 28.5 | 3.5 | 1.5 | 4 | 12.9 |
| B-ZnO/TiO2 | 6.3 | >25 | 15.3 | 21.4 | 65.2 | 46.5 | 1.9 | 13.3 | 2.6 | 22 |
| Sample | Actual Zeta Values | Stability Terms |
|---|---|---|
| TiO2 | +40.7 | Moderate stability, without aggregation [18,19,20] |
| B-TiO2 | +64 | Excellent stability, without aggregation [19,20] |
| ZnO/TiO2 | +9.38 | Rapid coagulation or flocculation full [30] |
| B-ZnO/TiO2 | −6.99 | Rapid coagulation or flocculation full [30] |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 28,085.38 | 15 | 1872.36 | 498.75 | <0.0001 | Significant |
| A-DR16 | 497.06 | 1 | 497.06 | 132.41 | <0.0001 | |
| B-B-ZnO/TiO2 loading | 3072.30 | 1 | 3072.30 | 818.39 | <0.0001 | |
| C-pH | 18,771.80 | 1 | 18,771.80 | 5000.38 | <0.0001 | |
| D-RT | 3146.89 | 1 | 3146.89 | 838.26 | <0.0001 | |
| E-LED intensity | 13.47 | 1 | 13.47 | 3.59 | 0.0667 | |
| AC | 130.82 | 1 | 130.82 | 34.85 | <0.0001 | |
| AD | 127.60 | 1 | 127.60 | 33.99 | <0.0001 | |
| AE | 160.65 | 1 | 160.65 | 42.79 | <0.0001 | |
| BC | 320.68 | 1 | 320.68 | 85.42 | <0.0001 | |
| BE | 203.52 | 1 | 203.52 | 54.21 | <0.0001 | |
| CD | 25.38 | 1 | 25.38 | 6.76 | 0.0137 | |
| CE | 74.12 | 1 | 74.12 | 19.74 | <0.0001 | |
| B² | 401.82 | 1 | 401.82 | 107.04 | <0.0001 | |
| C² | 17.03 | 1 | 17.03 | 4.54 | 0.0405 | |
| D² | 16.37 | 1 | 16.37 | 4.36 | 0.0443 | |
| Residual | 127.64 | 34 | 3.75 | |||
| Lack of Fit | 116.71 | 27 | 4.32 | 2.77 | 0.0833 | Not significant |
| Response | Probability | R2 | Adj. R2 | Pre. R2 | S.D | CV | Lack of Fit |
|---|---|---|---|---|---|---|---|
| Dye removal (%) | <0.0001 | 0.995 | 0.9935 | 0.9902 | 1.94 | 3.56 | 0.0833 |
| Intercept | Slope | qmax | KL | RL | R2 |
|---|---|---|---|---|---|
| 0.07659 | 0.06122 | 13.05653 | 1.251062 | 0.444235 | 0.97906 |
| Catalyst | Procedure | Waste Type | Optimum Condition | Efficiency % | Reusability | Ref. |
|---|---|---|---|---|---|---|
| B-ZnO/TiO2 | Sol-gel | DR16 | Cata. mass = 1 g/L Dye = 10 ppm pH = 3 Time = 200 min | 100 | 4 cycles with/without reactivation process | Present study |
| B-ZnO/TiO2 | Mechano-chemical–calcination | M.B. | Cata. mass = 30 mg/50 mL Dye = 15 mg/L pH = 11 Time = 30 min | 94.2 | 5 cycles | [37] |
| B-ZnO/TiO2 | Sol-gel | 4-chlorophenol | Cata. mass = 10 mg Waste = 5 × 10−5 mole pH = 5.38 Time = 480 min | 58.2 | - | [38] |
| Ni-ZnO/TiO2 | Sol—gel | Anthraquinone dye (reactive brilliant blue KN-R) | Cata. mass = 1.3 g/L Waste = 6 mg/L pH = 4 Time = 120 min | 69 | 4 cycles | [39] |
| C-ZnO/TiO2 | Microwave hydrothermal | Rhodamine B | Cata. mass = 50 mg/100 mL Waste = 10 mg/L Time = 45 min | 96 | 5 cycles | [26] |
| Cu2+/ZnO/TiO2 | Sol-gel | Aniline | Cata. mass = 2 g/L Waste = 125 mg/L pH = 11 Time = 7 h Temp. = 25 °C | COD = 77.8% | - | [27] |
| TiO2/Zn2TiO2/ZnO/C | Sol–gel | Orange G dye (OG) | Cata. mass = 50 mg/50 mL Waste = 20 mg/L Time = 50 min sunlight 180 min—UV | 100—sun light 40—UV | 5 cycles | [28] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Habeeb, S.A.; Zinatizadeh, A.A.; Zangeneh, H. Photocatalytic Decolorization of Direct Red16 from an Aqueous Solution Using B-ZnO/TiO2 Nano Photocatalyst: Synthesis, Characterization, Process Modeling, and Optimization. Water 2023, 15, 1203. https://doi.org/10.3390/w15061203
Habeeb SA, Zinatizadeh AA, Zangeneh H. Photocatalytic Decolorization of Direct Red16 from an Aqueous Solution Using B-ZnO/TiO2 Nano Photocatalyst: Synthesis, Characterization, Process Modeling, and Optimization. Water. 2023; 15(6):1203. https://doi.org/10.3390/w15061203
Chicago/Turabian StyleHabeeb, Saba Abdulmunem, Ali Akbar Zinatizadeh, and Hadis Zangeneh. 2023. "Photocatalytic Decolorization of Direct Red16 from an Aqueous Solution Using B-ZnO/TiO2 Nano Photocatalyst: Synthesis, Characterization, Process Modeling, and Optimization" Water 15, no. 6: 1203. https://doi.org/10.3390/w15061203





