Mining Leachates Effect on the Hydraulic Performance of Geosynthetic Clay Liners under Different Temperatures
Abstract
:1. Introduction
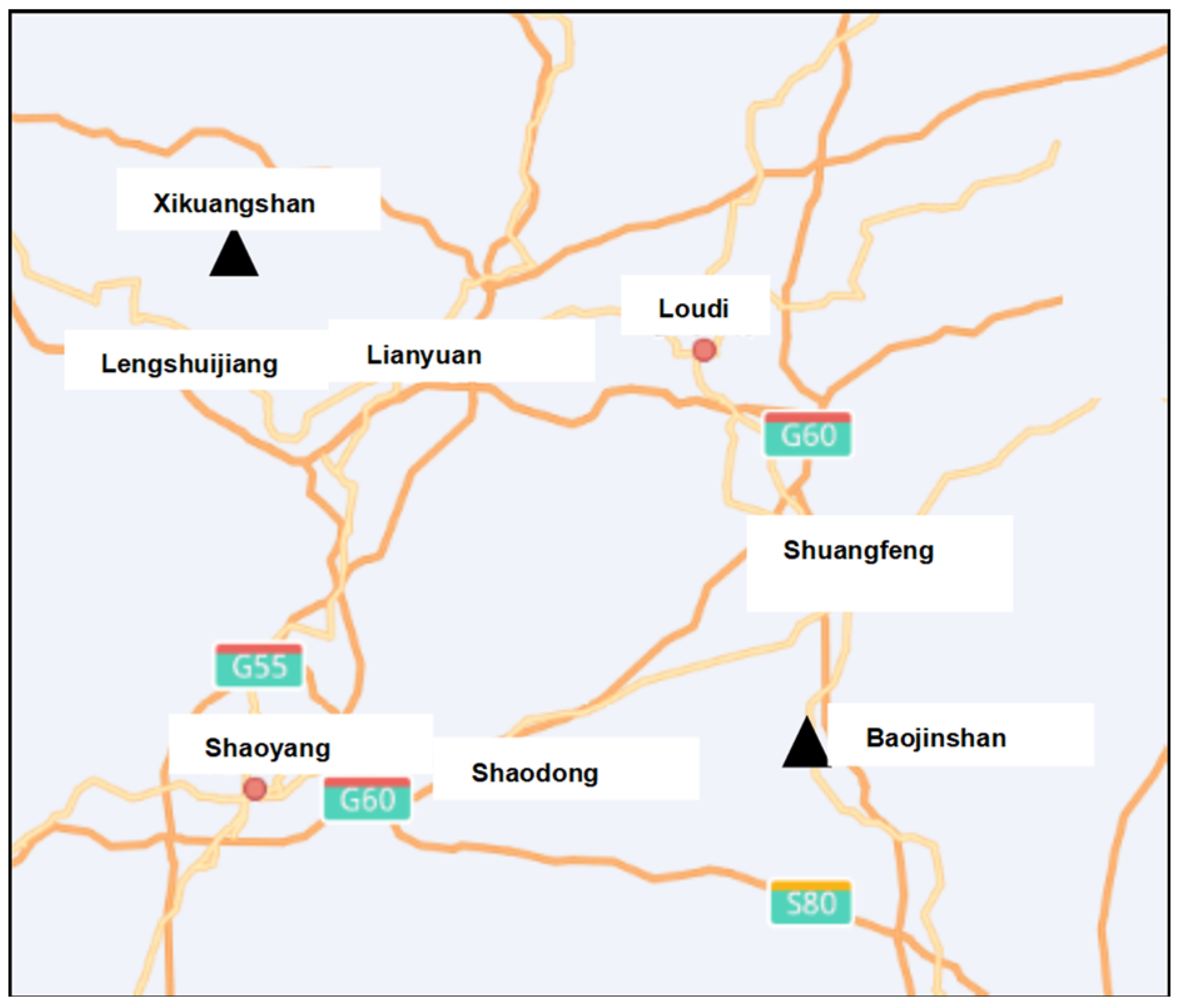
2. Materials and Methods
2.1. Materials
2.1.1. Geosynthetic Clay Liners
2.1.2. Test Solutions
2.2. Conducted Tests
2.2.1. Fluid Loss Test
2.2.2. Free Swell Index Test
2.2.3. X-ray Observation
2.2.4. Scanning Electron Microscope
3. Results and Discussion
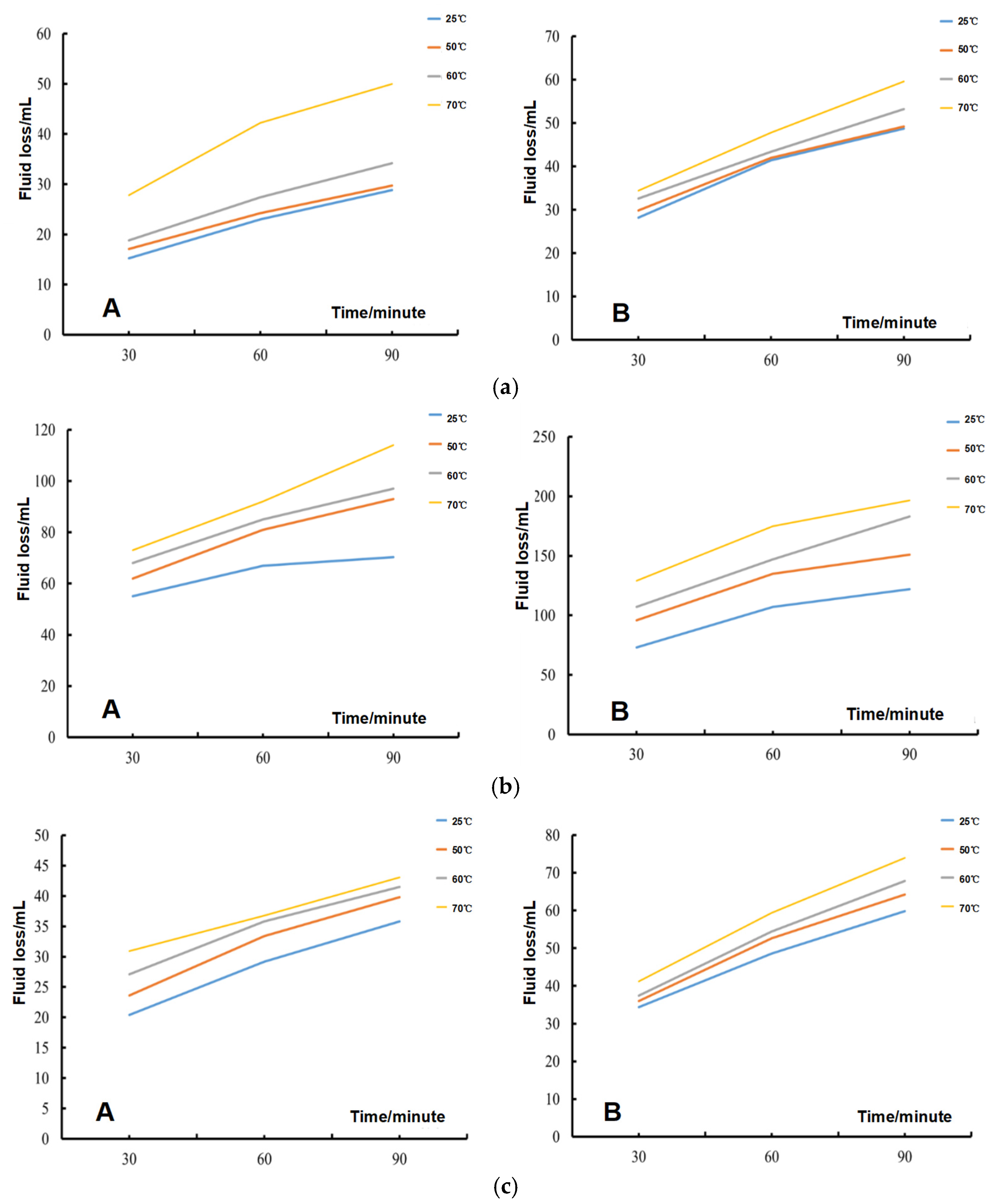
3.1. Fluid Loss Test
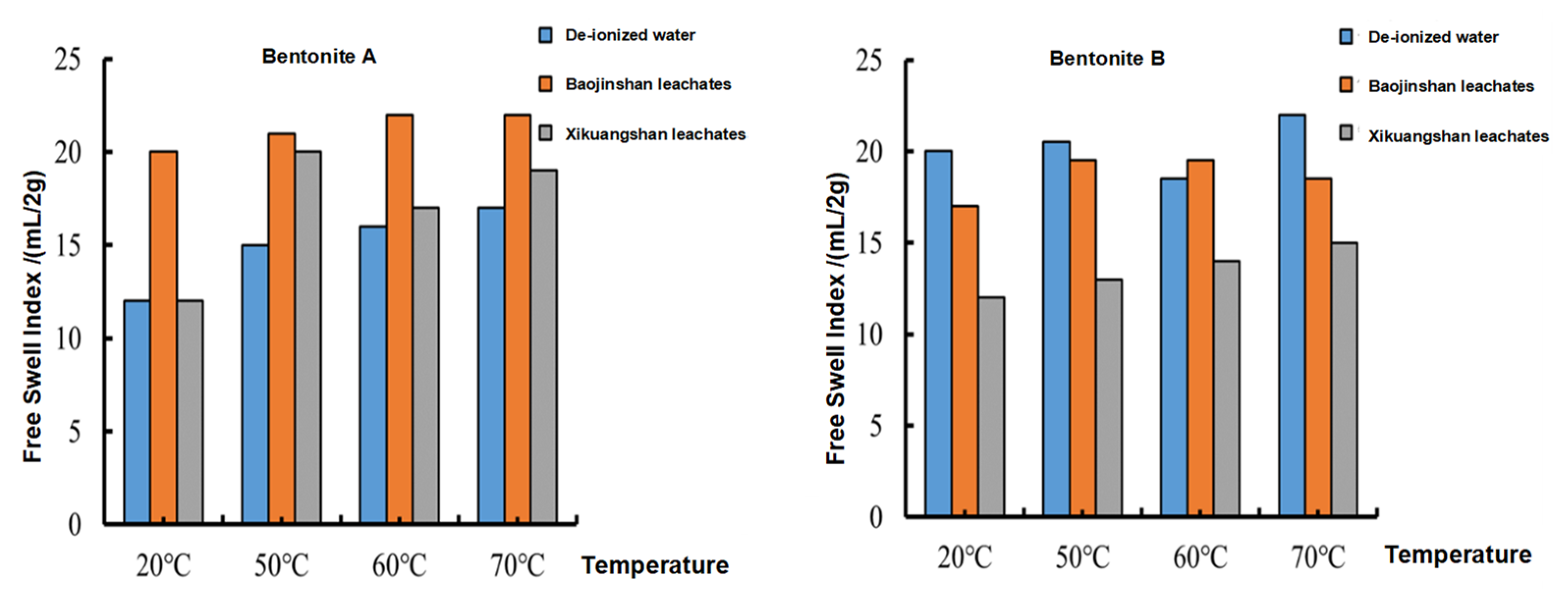
3.2. Free Swell Index Test
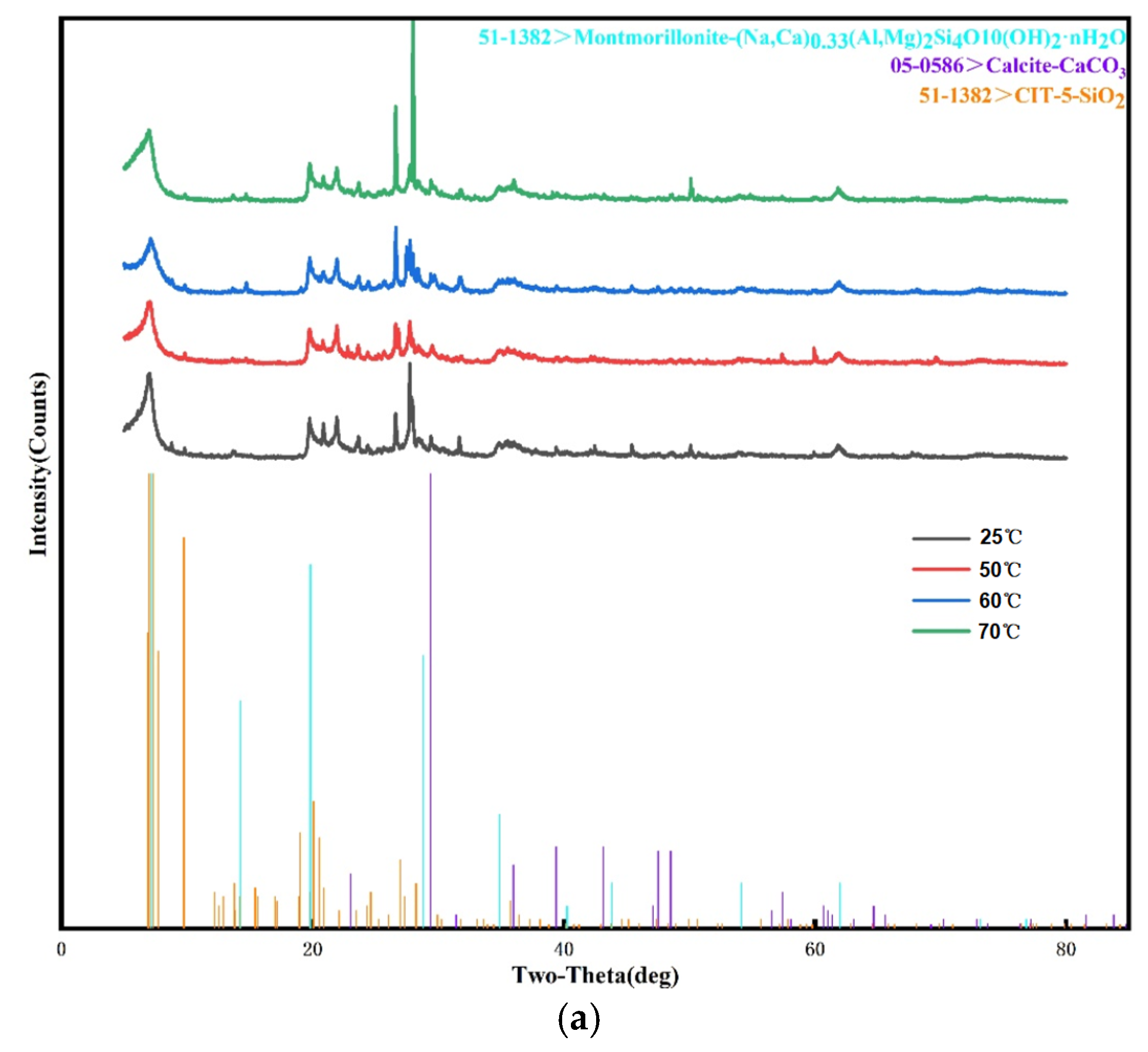
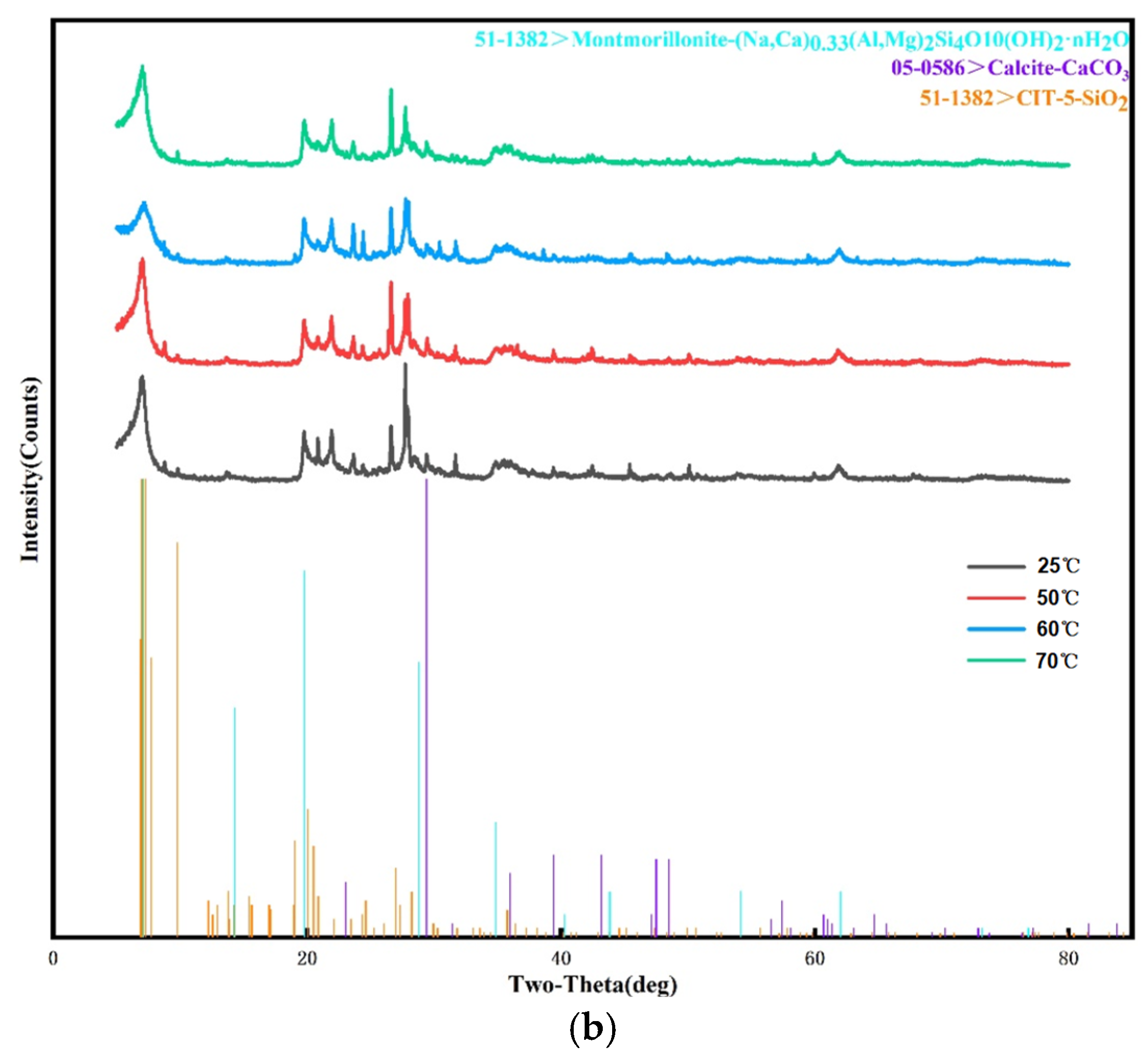
3.3. X-ray Diffraction and X-ray Fluorescence Spectrum
3.4. Scanning Electron Microscope
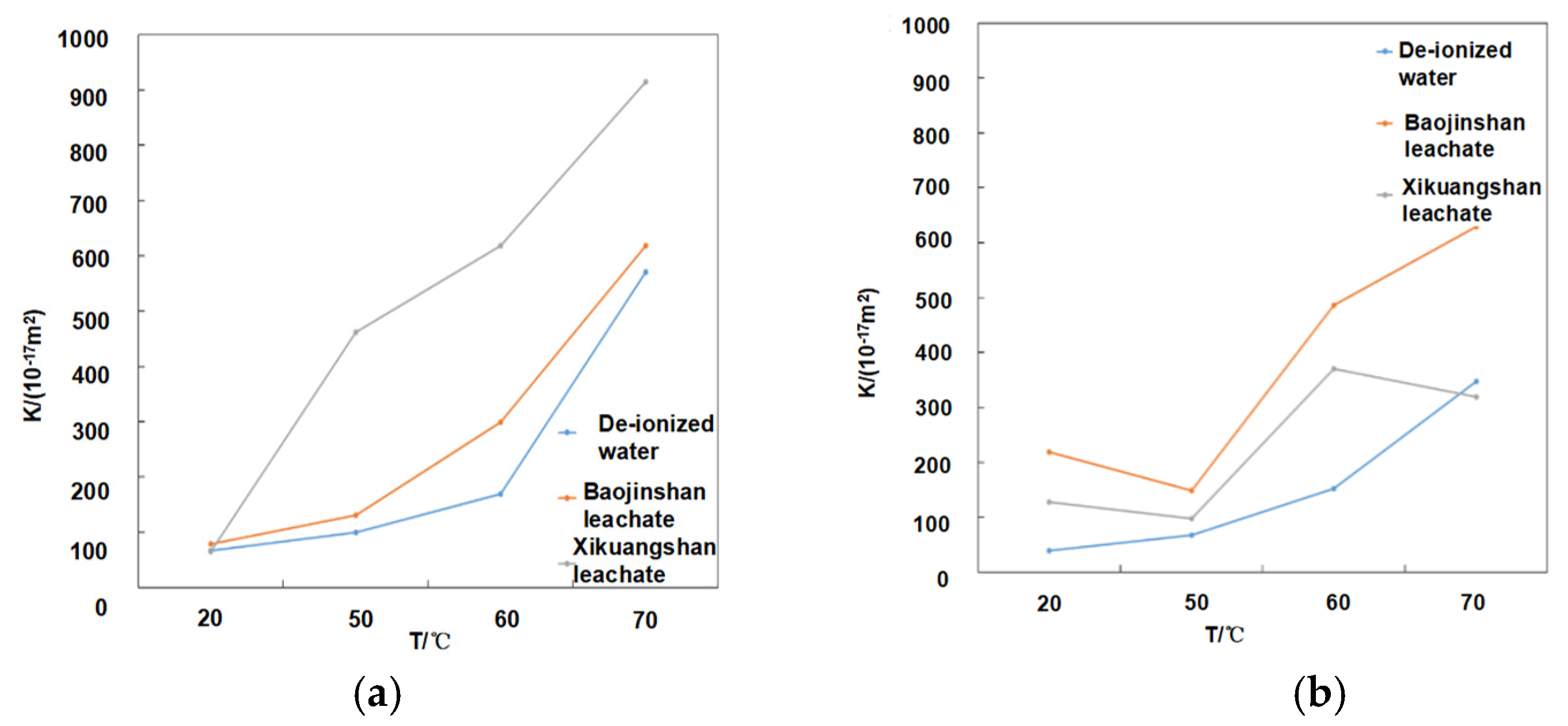
3.5. Hydraulic Conductivity Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Komnitsas, K.; Kontopoulos, A.; Lazar, I. Risk assessment and proposed remedial actions in coastal tailings disposal sites in Romania. Miner. Eng. 1998, 11, 1179–1190. [Google Scholar] [CrossRef]
- Li, Y.C.; Xu, Z.; Ma, H.; Hursthouse, A.S. Removal of Manganese(II) from Acid Mine Wastewater: A Review of the Challenges and Opportunities with Special Emphasis on Mn-Oxidizing Bacteria and Microalgae. Water 2019, 11, 2493. [Google Scholar] [CrossRef] [Green Version]
- Meng, D.; Li, J.; Liu, T.; Liu, Y.; Yan, M.; Hu, J.; Li, H.; Liu, X.; Liang, Y.; Liu, H.; et al. Effects of redox potential on soil cadmium solubility:Insight into microbial community. J. Environ. Sci. 2019, 75, 224–232. [Google Scholar] [CrossRef]
- Deng, R.J.; Jin, C.S.; Ren, B.Z.; Hou, B.L.; Hursthouse, A.S. The Potential for the Treatment of Antimony-Containing Wastewater by Iron-Based Adsorbents. Water 2017, 9, 794. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Liao, L.; Hursthouse, A.; Song, N.; Ren, B. Sepiolite-based adsorbents for the removal of potentially toxic elements from water: A strategic review for the case of environmental contamination in Hunan, China. Int. J. Environ. Res. Public Health 2018, 15, 1653. [Google Scholar] [CrossRef]
- Liu, Y.; Bouazza, A.; Gates, W.P.; Rowe, R.K. Hydraulic performance of geosynthetic clay liners to sulfuric acid solutions. Geotext. Geomembr. 2015, 43, 14–23. [Google Scholar] [CrossRef]
- Rowe, R.K.; Mukunoki, T.; Bathurst, R.J. Hydraulic conductivity to Jet-A1 of GCLs after up to 100 freeze–thaw cycles. Géotechnique 2008, 58, 503–511. [Google Scholar] [CrossRef]
- Zhao, Y.L.; Zhang, C.S.; Wang, Y.X.; Lin, H. Shear-related roughness classification and strength model of natural rock joint based on fuzzy comprehensive evaluation. Int. J. Rock Mech. Min. Sci. 2021, 137, 104550. [Google Scholar] [CrossRef]
- Xie, Q.; Ren, B.Z.; Hursthouse, A.S.; Shi, X.Y. Effects of mining activities on the distribution, controlling factors, and sources of metals in soils from the Xikuangshan South Mine, Hunan Province. Integr. Environ. Assess. Manag. 2022, 18, 748–756. [Google Scholar] [CrossRef]
- Liu, Y.; He, B.; Xie, J.; Lu, Y.; Zhang, L. Compatibility of geosynthetic clay liners at different temperatures. J. Environ. Prot. Ecol. 2021, 22, 2295–2306. [Google Scholar]
- Feng, H.; Liu, F.; Luo, P.; Xie, G.; Xiao, R.; Hu, W.; Peng, J.; Wu, J. Performance of integrated ecological treatment system for decentralized rural wastewater and significance of plant harvest management. Ecol. Eng. 2018, 124, 69–76. [Google Scholar]
- Zhao, Y.L.; Zhang, L.Y.; Liao, J.; Wang, W.J.; Liu, Q.; Tang, L.M. Experimental study of fracture toughness and subcritical crack growth of three rocks under different environments. Int. J. Geomech. 2020, 20, 04020128. [Google Scholar] [CrossRef]
- Zhao, Y.L.; Tang, J.Z.; Chen, Y.; Zhang, L.Y.; Wang, W.J.; Liao, J.P. Hydromechanical coupling tests for mechanical and permeability characteristics of fractured limestone in complete stress-strain process. Environ. Earth Sci. 2017, 76, 24. [Google Scholar] [CrossRef]
- Tang, L.; Tang, X.W.; Liu, Y.; Qu, S.X. Prediction of pore size characteristics of woven slit-film geotextiles subjected to unequal biaxial tensile strains. Geotext. Geomembr. 2020, 48, 724–734. [Google Scholar] [CrossRef]
- Wang, T.; Huang, C.; Du, G.; Liu, Y.; Xie, J.; Li, H. Geochronology, geochemistry and zircon Hf-isotopes of the early Mesoproterozoic Yaopengzi dolerite in SW Yangtze block (Sichuan, SW China): Implications for the Columbia supercontinent breakup. Geosci. J. 2019, 23, 557–573. [Google Scholar] [CrossRef]
- Zhang, J.; Deng, R.; Ren, B.; Hou, B.; Hursthouse, A. Preparation of a novel Fe3O4/HCO composite adsorbent and the mechanism for the removal of antimony (III) from aqueous solution. Sci. Rep. 2019, 9, 13021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, Z.; Deng, R.; Zhang, J.; Ren, B.; Hursthouse, A. Regional distribution characteristics and ecological risk assessment of heavy metal pollution of different land use in an antimony mining area-Xikuangshan, China. Hum. Ecol. Risk Assess. 2020, 26, 1779–1794. [Google Scholar] [CrossRef]
- Liu, Y.; Hao, Y.; Lu, Y. Improved design of risk assessment model for PPP project under the development of marine architecture. J. Coast. Res. 2018, 83, 74–80. [Google Scholar] [CrossRef]
- Zhu, G.; Wang, C.; Dong, X. Fluorescence excitation-emission matrix spectroscopy analysis of landfill leachate DOM in coagulation-flocculation process. Environ. Technol. 2017, 9, 1489–1497. [Google Scholar] [CrossRef]
- Zhou, L.; Liu, X.; Li, J.; Liao, J. Robust AVO inversion for the fluid factor and shear modulus. Geophysics 2021, 86, 471–483. [Google Scholar] [CrossRef]
- Liu, Y.; Gates, W.P.; Bouazza, A. Acid induced degradation of the bentonite component used in geosynthetic clay liners. Geotext. Geomembr. 2013, 36, 71–80. [Google Scholar] [CrossRef]
- Li, Y.; Hu, X.; Ren, B. Treatment of antimony mine drainage: Challenges and opportunities with special emphasis on mineral adsorption and sulfate reducing bacteria. Water Sci. Technol. 2016, 73, 2039–2051. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Ren, B.; Hursthouse, A.S.; Thacker, R.M.; Wang, Z. Soil from an Abandoned Manganese Mining Area (Hunan, China): Significance of Health Risk from Potentially Toxic Element Pollution and Its Spatial Context. Int. J. Environ. Res. Public Health 2020, 17, 6554. [Google Scholar] [CrossRef]
- Zhou, Y.; Ren, B.; Hursthouse, A.S.; Zhou, S. Antimony Ore Tailings: Heavy Metals, Chemical Speciation, and Leaching Characteristics. Pol. J. Environ. Stud. 2019, 28, 485–495. [Google Scholar] [CrossRef]
- Liu, Y.; Gates, W.P.; Bouazza, A. Impact of acid leachates on microtexture of bentonites used in geosynthetic clay liners. Geosynth. Int. 2019, 26, 136–145. [Google Scholar] [CrossRef]
- Lu, Y.; Cao, C.; Liu, Y.; Liu, Y. Study on Application of Comprehensive Geophysical Prospecting Method in Urban Geological Survey—Taking Concealed Bedrock Detection as an Example in Dingcheng District, Changde City, Hunan Province, China. Appl. Sci. 2023, 13, 417. [Google Scholar] [CrossRef]
- Song, N.; Hursthouse, A.; McLellan, I.; Wang, Z. Treatment of environmental contamination using sepiolite: Current approaches and future potential. Environ. Geochem. Health 2021, 43, 2679–2697. [Google Scholar] [CrossRef]
- He, Z.; Ren, B.; Hursthouse, A.; Wang, Z. Efficient removal of Cd(II) using SiO2-Mg(OH)2 nanocomposites derived from sepiolite. Int. J. Environ. Res. Public Health 2020, 17, 2223. [Google Scholar] [CrossRef] [Green Version]
- Ling, J.; Dai, S.; Zhou, Y.; Chen, Q.; Zhang, Y.; Li, K. Three-Dimensional DC Anisotropic Resistivity Modeling Using a Method in the Mixed Space-Wavenumber Domain. Pure Appl. Geophys. 2022, 179, 2183–2200. [Google Scholar] [CrossRef]
- Song, K.; Ren, X.; Mohanmod, A.; Liu, J.; Wang, F. Research on drinking groundwater source safety management based on numerical simulation. Sci. Rep. 2020, 10, 15481. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.; Zhang, C.; Feng, Y. Research progress on temperature effect of impervious clay liners in sanitary landfills. In Proceedings of the Second National Geotechnical and Engineering, Taipei, Taiwan, 28 October–1 November 2006; Chinese Society of Rock Mechanics and Engineering: Wuhan, China, 2006; Volume 2, p. 8. [Google Scholar]
- Arden, B.; Mohammad, C. Effect of temperature on hydration of geosynthetic clay liners in landfills. Waste Manag. Res. 2013, 22, 58–71. [Google Scholar]
- Zhao, Y.L.; Luo, S.L.; Wang, Y.X.; Wang, W.J.; Zhang, L.Y.; Wan, W. Numerical analysis of karst water inrush and a criterion for establishing the width of water-resistant rock pillars. Mine Water Environ. 2017, 36, 508–519. [Google Scholar] [CrossRef]
- Yuan, Q.; Zhu, G. A review on metal organic frameworks (MOFs) modified membrane for remediation of water pollution. Environ. Eng. Res. 2021, 26, 190435.1–190435.2. [Google Scholar] [CrossRef]
- Deng, R.J.; Shao, R.; Ren, B.Z.; Hou, B.L.; Tang, Z.E.; Hursthouse, A. Adsorption of Antimony(III) onto Fe(III)-Treated Humus Sludge Adsorbent: Behavior and Mechanism Insights. Pol. J. Environ. Stud. 2019, 28, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Liu, Y.; Yang, C. Research on the slurry for long-distance large-diameter pipe jacking in expansive soil. Adv. Civ. Eng. 2018, 9, 47–54. [Google Scholar] [CrossRef]
- Zhao, Y.L.; Liu, Q.; Zhang, C.; Liao, J.; Lin, H.; Wang, Y. Coupled seepage-damage effect in fractured rock masses: Model development and a case study. Int. J. Rock Mech. Min. Sci. 2021, 144, 104822. [Google Scholar] [CrossRef]
- Zhang, Y.; Ren, B.; Hursthouse, A.; Deng, R.; Hou, B. Study on the migration rules of Sb in antimony ore soil based on HYDRUS-1D. Pol. J. Environ. Stud. 2018, 28, 965–972. [Google Scholar] [CrossRef]
- Zhang, Y.; Ren, B.; Hursthouse, A.; Deng, R.; Hou, B. An Improved SWAT for predicting manganese pollution load at the soil-water interface in a manganese mine area. Pol. J. Environ. Stud. 2018, 27, 2357–2365. [Google Scholar] [CrossRef]
- Xie, J.; Liu, Y.; Lu, Y.; Zhang, L. Application of the high-density resistivity method in detecting a mined-out area of a quarry in Xiangtan City, Hunan Province. Front. Environ. Sci. 2022, 10, 1068956. [Google Scholar]
- Hou, B.L.; Liu, X.; Li, Z.; Ren, B.Z.; Kuang, Y. Heterogeneous fenton oxidation of butyl xanthate catalyzed by iron-loaded sewage sludge. Fresenius Environ. Bull. 2022, 31, 4125–4131. [Google Scholar]
- Zhao, Y.L.; Zhang, L.; Wang, W.; Tang, J.; Lin, H.; Wan, W. Transient pulse test and morphological analysis of single rock fractures. Int. J. Rock Mech. Min. Sci. 2017, 91, 139–154. [Google Scholar] [CrossRef]
- Hou, B.; Li, Z.; Deng, R.; Ren, B. Advanced treatment of coal chemical industry wastewater by expansive flow biological aerated filter. Fresenius Environ. Bull. 2017, 26, 4517–4521. [Google Scholar]
- Shi, X.; Ren, B.; Hursthouse, A. Source identification and groundwater health risk assessment of PTEs in the stormwater runoff in an abandoned mining area. Environ. Geochem. Health 2022, 44, 3555–3570. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huang, F. Indicative significance of the magnetic susceptibility of substrate sludge to heavy metal pollution of urban lakes. Sci. Asia 2021, 47, 374. [Google Scholar] [CrossRef]
- Hanson, J.L.; Chrysovergis, T.S.; Yesiller, N. Temperature and moisture effects on GCL and textured geomembrane interface shear strength. Geotextile Geomembr. 2015, 22, 110–124. [Google Scholar] [CrossRef]
- Jiang, J. Experimental Study on Application Characteristics of GCL under Complex Conditions; Wenzhou University: Wenzhou, China, 2017. [Google Scholar]
- He, J.; Hu, X.; Yan, X.; Wan, J. Temperature effect test on clay permeability. Adv. Sci. Technol. Water Resour. 2017, 37, 55–60. [Google Scholar]
- Shao, Y.; Shi, B.; Liu, C.; Gu, K.; Tang, C. Study on temperature effect of hydrophysical properties of clay. J. Geotech. Eng. 2011, 33, 1576–1582. [Google Scholar]
- Ishimori, H.; Katsumi, T. Temperature effects on the swelling capacity and barrier performance of geosynthetic clay liners permeated with sodium chloride solutions. Geotext. Geomembr. 2012, 33, 25–33. [Google Scholar] [CrossRef]
- Ye, W.M.; Wan, M.; Chen, B.; Chen, Y.; Cui, J. Temperature effects on the unsaturated permeability of the densely compacted GMZ01 bentonite under confined conditions. Eng. Geol. 2012, 126, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Tang, C.; Shi, B. Swelling and shrinkage deformation characteristics of expansive soil during the dry-wet cycle. Chin. J. Geotech. Eng. 2011, 33, 1376–1384. [Google Scholar]
- Xie, Q.; Ren, B. Pollution and risk assessment of heavy metals in rivers in the antimony capital of Xikuangshan. Sci. Rep. 2022, 12, 14393. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Hursthouse, A. The impact of physical properties on the leaching of potentially toxic elements from antimony ore processing wastes. Int. J. Environ. Res. Public Health 2019, 16, 2355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, K.; Yang, G.; Wang, F.; Liu, J.; Liu, D. Application of Geophysical and Hydrogeochemical Methods to the Protection of Drinking Groundwater in Karst Regions. Int. J. Environ. Res. Public Health 2020, 17, 3627. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yang, T.; Abdollah, T.; Liu, Y. Fast Recognition on Shallow Groundwater and Anomaly Analysis Using Frequency Selection Sounding Method. Water 2023, 15, 96. [Google Scholar]
- Rushton, A.; Ward, A.S.; Holdich, R.G. Solid-Liquid Filtration and Separation Technology, 2nd ed.; Wiley-VCH Verlag GmbH: Weinbeim, Germany, 2000. [Google Scholar]
- Chung, J.; Daniel, D.E. Modified fluid loss test as an improved measure of hydraulic conductivity for Bentonite. Geotech. Test. J. 2008, 31, 243–251. [Google Scholar]
- Katsumi, T.; Ishimori, H.; Onikata, M.; Fukagawa, R. Long-term barrier performance of modified bentonite materials against sodium and calcium permeant solutions. Geotext. Geomembr. 2008, 26, 14–30. [Google Scholar] [CrossRef]



| Bentonite A | Bentonite B | ||
|---|---|---|---|
| Mineral Constituents/% | Montmorillonite | 78.1 | 74.1 |
| Quartz | 7.6 | 5.3 | |
| Feldspar | 9.6 | 9.4 | |
| Calcite | 4.7 | 6.1 | |
| Mica | — | 5.1 | |
| Element Constituents/% | O | 47.5 | 45.4 |
| Na | 2.9 | 8.01 | |
| Mg | 1.59 | 1.05 | |
| Al | 8.67 | 8.18 | |
| Si | 29.37 | 28.61 | |
| K | 1.07 | 1.07 | |
| Ca | 3.48 | 2.85 | |
| Others | 5.42 | 4.38 | |
| Element | Al | Ca | K | Mg | Na | Pb | S | Si | Sb | Sr | W |
|---|---|---|---|---|---|---|---|---|---|---|---|
| mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | |
| Content | 0.03 | 37.1 | 19.9 | 2 | 480 | 0.02 | 192 | 9 | 0.02 | 0.07 | 1.8 |
| Element | Al | Ca | K | Mg | Li | Na | S | Si | Sb | Sr |
|---|---|---|---|---|---|---|---|---|---|---|
| mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | |
| Content | 0.26 | 20.9 | 22.0 | 3.9 | 0.124 | 441 | 316 | 8.4 | 0.023 | 0.208 |
| Sample | Element | |||||||
|---|---|---|---|---|---|---|---|---|
| O | Na | Mg | Al | Si | P | S | Cl | |
| A | 47.200 | 2.650 | 1.490 | 8.952 | 28.050 | 0.114 | 0.055 | 1.227 |
| A + 50 °C | 46.100 | 2.800 | 1.500 | 7.999 | 27.230 | 0.089 | 1.479 | 1.290 |
| A + 70 °C | 47.500 | 3.510 | 1.580 | 8.163 | 27.490 | 0.099 | 0.715 | 1.200 |
| K | Ca | Ti | Cr | Mn | Fe | Ni | Cu | |
| A | 2.060 | 4.151 | 0.445 | 0.005 | 0.064 | 3.409 | - | 0.007 |
| A + 50 °C | 1.050 | 3.838 | 0.435 | - | 0.087 | 3.103 | - | - |
| A + 70 °C | 0.958 | 3.709 | 0.447 | - | 0.077 | 3.156 | - | 0.003 |
| Zn | Ga | Rb | Sr | Zr | Nb | Ba | Others | |
| A | 0.010 | - | 0.005 | 0.011 | 0.009 | - | 0.073 | 0.010 |
| A + 50 °C | 0.009 | - | 0.004 | 0.027 | 0.016 | - | 0.065 | 2.154 |
| A + 70 °C | 0.009 | - | 0.004 | 0.029 | 0.014 | - | 0.069 | 1.962 |
| O | Na | Mg | Al | Si | P | S | Cl | |
| B | 42.200 | 5.460 | 1.350 | 8.877 | 28.960 | 0.087 | 0.398 | 0.886 |
| B + 50 °C | 47.900 | 3.380 | 1.500 | 8.240 | 28.260 | 0.101 | 0.551 | 1.280 |
| B + 70 °C | 47.400 | 3.830 | 1.480 | 8.079 | 30.150 | 0.091 | 0.362 | 1.240 |
| K | Ca | Ti | Cr | Mn | Fe | Ni | Cu | |
| B | 1.060 | 3.151 | 0.445 | 0.005 | 0.064 | 3.409 | - | 0.007 |
| B + 50 °C | 1.239 | 2.918 | 0.454 | - | 0.074 | 3.147 | - | - |
| B + 70 °C | 1.100 | 2.890 | 0.436 | - | 0.073 | 3.131 | - | - |
| Zn | Ga | Rb | Sr | Zr | Nb | Ba | Others | |
| B | 0.010 | - | 0.005 | 0.011 | 0.009 | - | 0.073 | 0.013 |
| B + 50 °C | 0.009 | - | 0.004 | 0.028 | 0.012 | - | 0.075 | 0.390 |
| B + 70 °C | 0.009 | - | 0.004 | 0.028 | 0.012 | - | 0.074 | 0.021 |
| Bentonite | A | B | ||||||
|---|---|---|---|---|---|---|---|---|
| Temperature( °C) | 0 | 50 | 60 | 70 | 0 | 50 | 60 | 70 |
| De-ionized water | 6.96 × 10−9 | 8.12 × 10−9 | 1.22 × 10−8 | 3.24 × 10−8 | 2.04 × 10−8 | 4.63 × 10−8 | 6.11 × 10−8 | 8.56 × 10−8 |
| Baojinshan leachates | 2.62 × 10−8 | 4.46 × 10−8 | 7.15 × 10−8 | 9.35 × 10−8 | 4.37 × 10−8 | 5.12 × 10−8 | 7.26 × 10−8 | 9.44 × 10−8 |
| Xikuangshan leachates | 9.18 × 10−9 | 6.04 × 10−7 | 8.81 × 10−7 | 2.12 × 10−6 | 4.14 × 10−7 | 4.78 × 10−7 | 6.88 × 10−7 | 8.66 × 10−7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Li, X.; Tu, Y.; Lu, Y. Mining Leachates Effect on the Hydraulic Performance of Geosynthetic Clay Liners under Different Temperatures. Water 2023, 15, 1132. https://doi.org/10.3390/w15061132
Liu Y, Li X, Tu Y, Lu Y. Mining Leachates Effect on the Hydraulic Performance of Geosynthetic Clay Liners under Different Temperatures. Water. 2023; 15(6):1132. https://doi.org/10.3390/w15061132
Chicago/Turabian StyleLiu, Yang, Xinxin Li, Yuanzhuo Tu, and Yulong Lu. 2023. "Mining Leachates Effect on the Hydraulic Performance of Geosynthetic Clay Liners under Different Temperatures" Water 15, no. 6: 1132. https://doi.org/10.3390/w15061132
APA StyleLiu, Y., Li, X., Tu, Y., & Lu, Y. (2023). Mining Leachates Effect on the Hydraulic Performance of Geosynthetic Clay Liners under Different Temperatures. Water, 15(6), 1132. https://doi.org/10.3390/w15061132







