Contamination of Water and Sediments of Harike Wetland with Phthalate Esters and Associated Risk Assessment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Study Area and Sample Collection
2.3. Water Sample Extraction
2.4. Sediment Sample Extraction
2.5. Instrumental Conditions
2.6. Risk Assessment
2.6.1. Human Health
2.6.2. Freshwater and Sediment
2.7. Statistical Analyses
3. Results
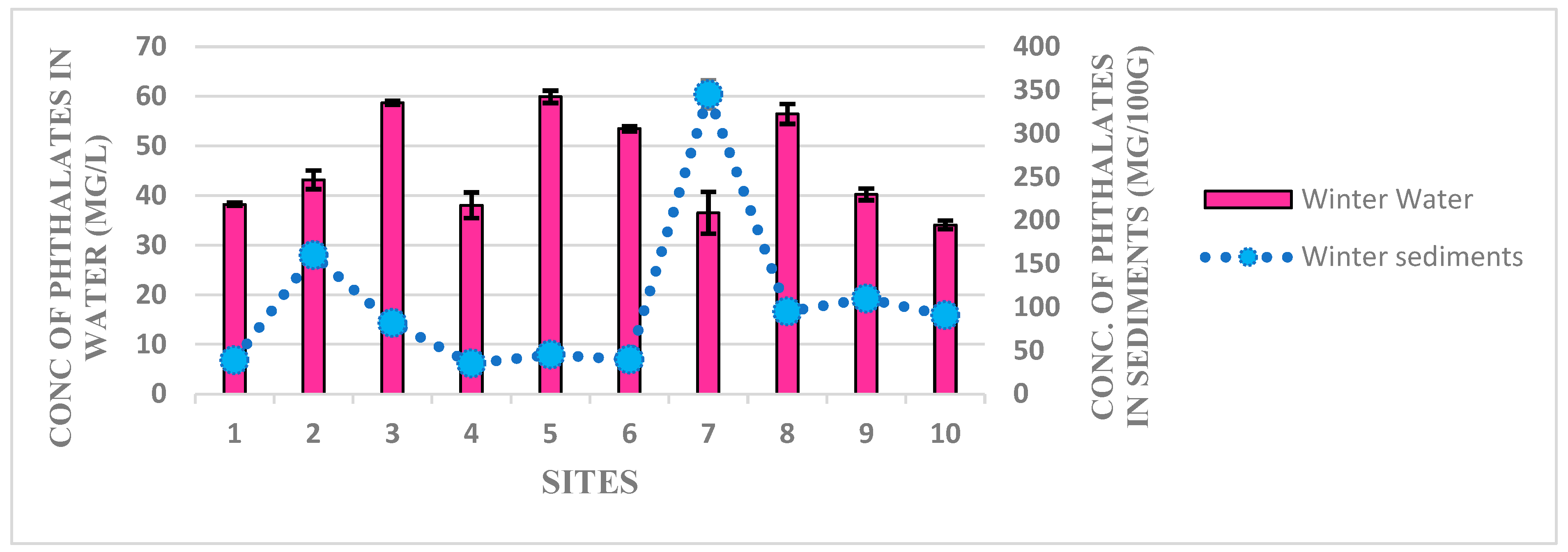
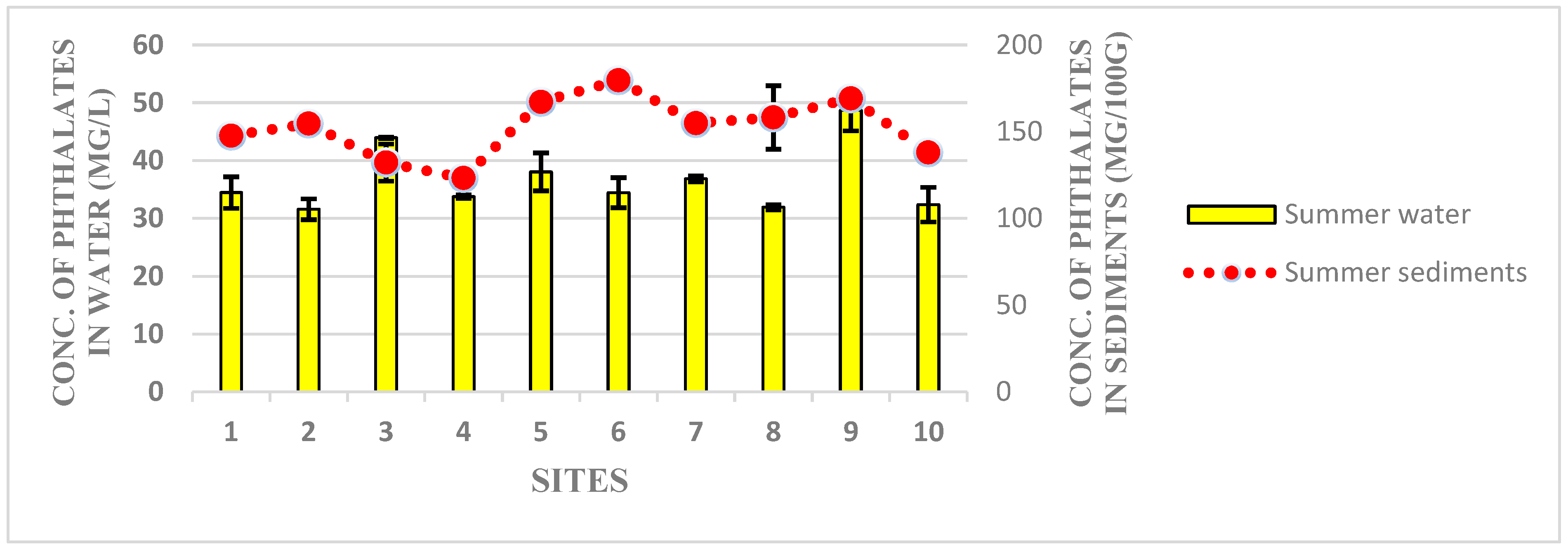
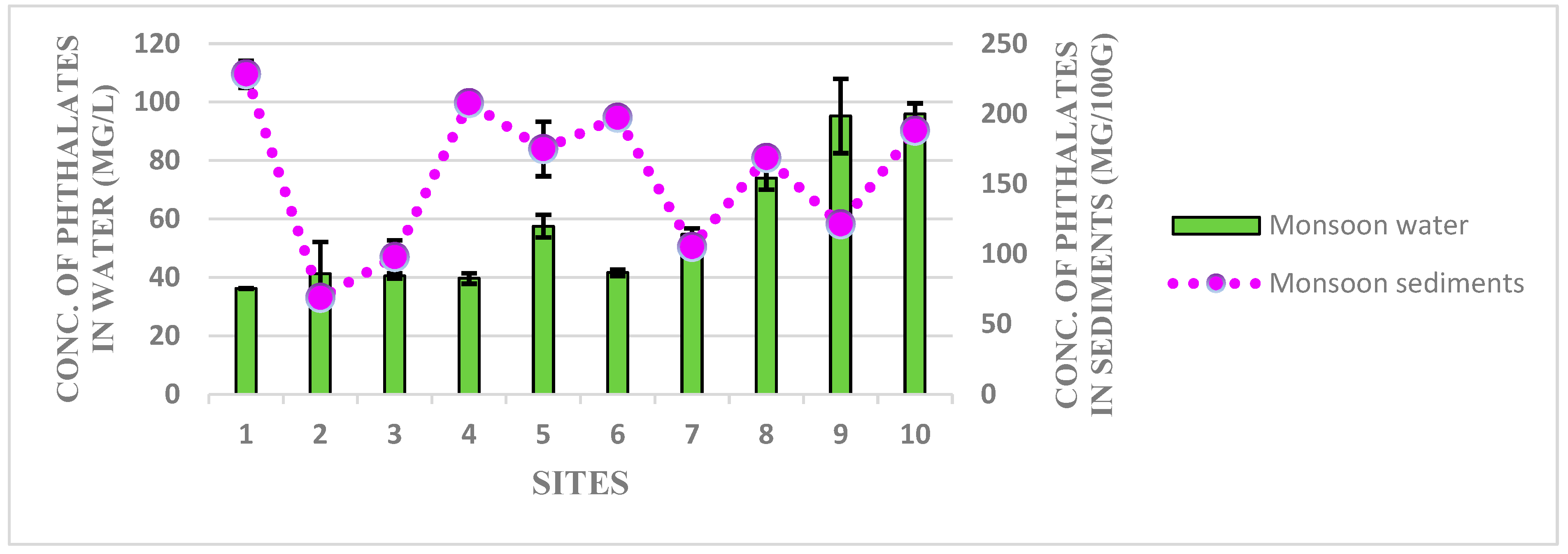
3.1. Detection of Phthalates Esters in Water and Sediment Samples of Harike Wetland
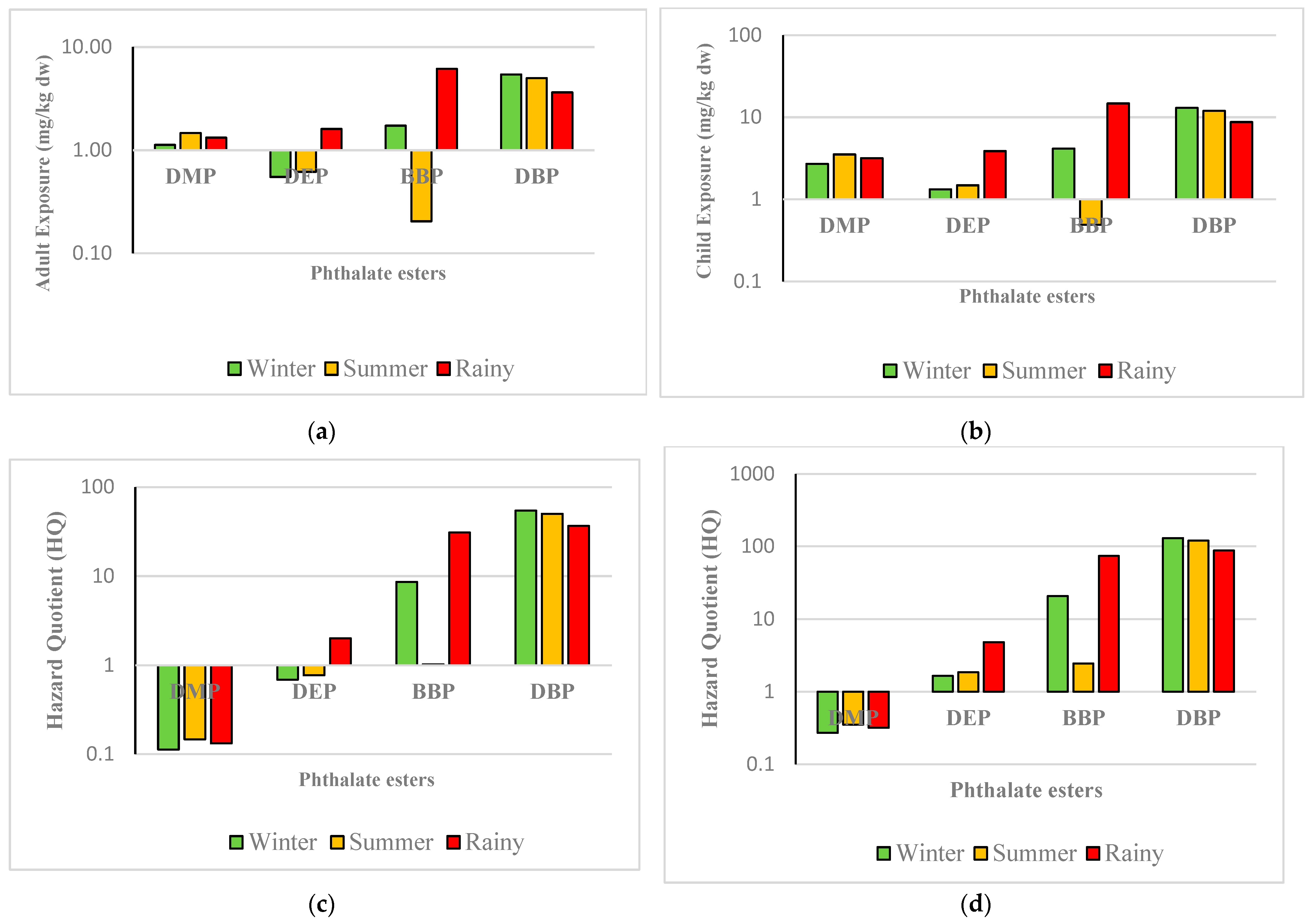
3.2. Human Heath Drinking Water Risk Assessment
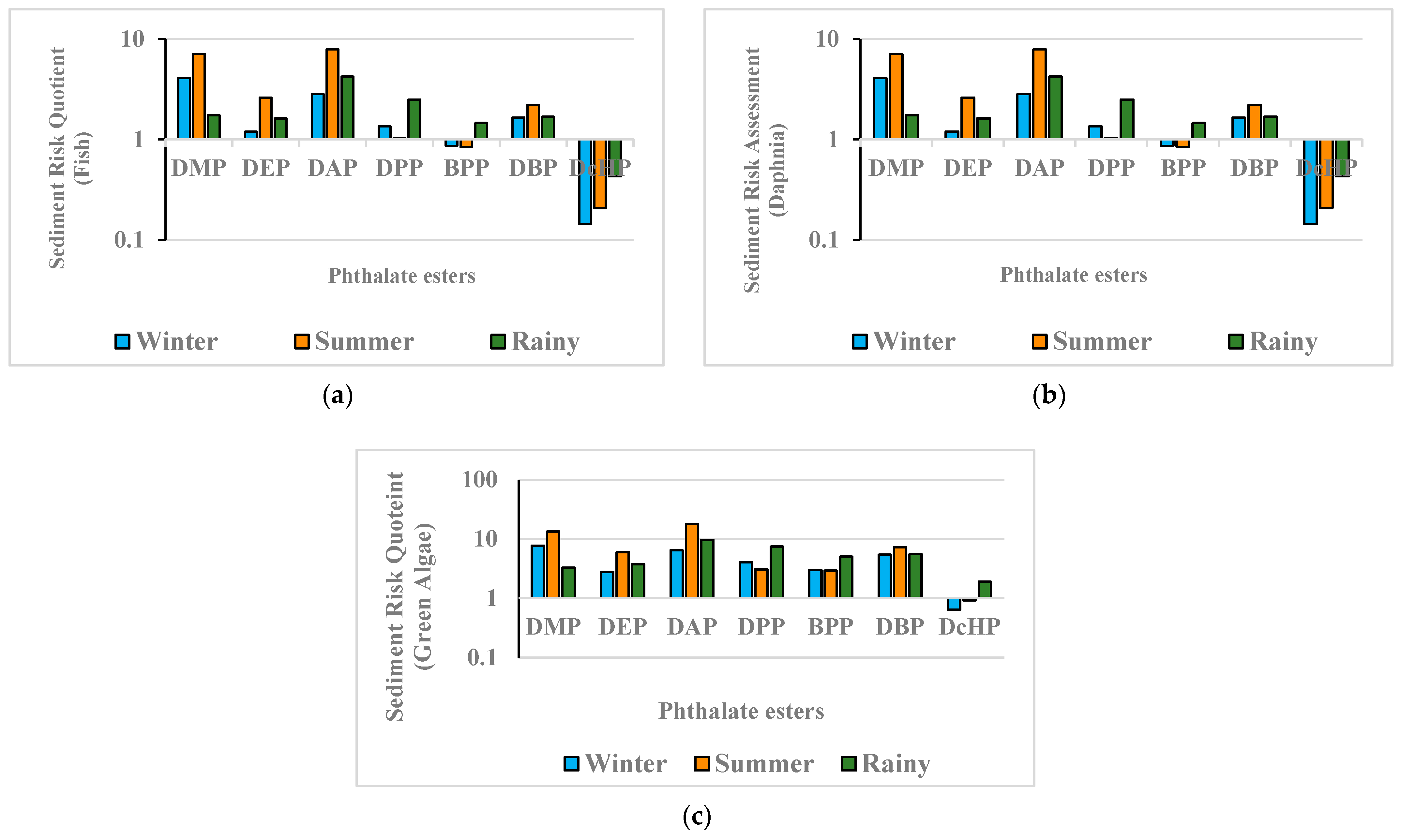
3.3. Environmental Risk Assessment
- i.
- Aquatic Risk Assessment
- ii.
- Sediment Risk Assessment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Selvaraj, K.K.; Sundaramoorthy, G.; Ravichandran, P.K.; Girijan, G.K.; Sampath, S.; Ramaswamy, B.R. Phthalate esters in water and sediments of the Kaveri River, India: Environmental levels and ecotoxicological evaluations. Environ. Geochem. Health 2015, 37, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, P.; Shappell, N.W.; Mukhopadhyay, M.; Onanong, S.; Rex, K.R.; Snow, D. Surveillance of plasticizers, bisphenol A, steroids and caffeine in surface water of River Ganga and Sundarban wetland along the Bay of Bengal: Occurrence, sources, estrogenicity screening and ecotoxicological risk assessment. Water Res. 2021, 190, 116668. [Google Scholar] [CrossRef]
- Ogunwole, G.A.; Saliu, J.K.; Osuala, F.I. Seasonal Occurrences and Risk Assessment of Phthalate Esters in Sediment, Water and Biota of Two Sub-Saharan Rivers. Bull. Environ. Contam. Toxicol. 2021, 106, 832–838. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.Z.; Wang, Y.; Xiang, N.; Chen, L.; Liu, Z.G.; Wu, B. Flow of sewage sludge-borne phthalate esters (PAEs) from human release to human intake: Implication for risk assessment of sludge applied to soil. Sci. Total Environ. 2014, 476, 242–249. [Google Scholar] [CrossRef]
- Net, S.; Delmont, A.; Sempéré, R.; Paluselli, A.; Baghdad, O. Occurrence, fate, behavior and ecotoxicological state of phthalates in different environmental matrices. Environ. Sci. Technol. 2015, 49, 4019–4035. [Google Scholar] [CrossRef] [PubMed]
- Shariati, S.; Pourbabaee, A.A.; Alikhani, H.A.; Rezaei, K.A. Assessment of phthalic acid esters pollution in Anzali wetland, north of Iran. Int. J. Environ. Sci. Technol. 2019, 16, 7025–7036. [Google Scholar] [CrossRef]
- National Institutes of Health. National Library of Medicine. Tox Town, Phthalates. 2006. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1480516/ (accessed on 2 February 2006).
- Ternes, T.A.; Meisenheimer, M.; Mcdowell, D.; Brauch, H.J.; Brigitte, H.G.; Preuss, G.; William, U.; Zulei-Seibert, N. Removal of Pharmaceuticals during Drinking Water Treatment. Environ. Sci. Technol. 2002, 36, 3855–3863. [Google Scholar] [CrossRef]
- Mondal, S.; Mukherjee, S. Long-term dietary administration of diethyl phthalate triggers loss of insulin sensitivity in two key insulin target tissues of mice. Hum. Exp. Toxicol. 2020, 39, 984–993. [Google Scholar] [CrossRef]
- Xu, S.; Zhang, H.; Pao, P.C.; Lee, A.; Wang, J.; Chan, Y.S.; Chen, X. Exposure to phthalates impaired neurodevelopment through estrogenic effects and induced DNA damage in neurons. Aquat. Toxicol. 2020, 222, 105469. [Google Scholar] [CrossRef]
- Li, J.; Wang, Z.; Wang, Q.; Guo, L.; Wang, C.; Wang, Z. Construction of Hypercrosslinked Polymers for High-Performance Solid Phase Microextraction of Phthalate Esters from Water Samples. J. Chromatogr. 2021, 1641, 461972. [Google Scholar] [CrossRef]
- Gao, X.; Li, J.; Wang, X.; Zhou, J.; Fan, B.; Li, W.; Liu, Z. Exposure and ecological risk of phthalate esters in the Taihu Lake basin, China. Ecotoxicol. Environ. Safe 2019, 171, 564–570. [Google Scholar] [CrossRef]
- Li, R.; Liang, J.; Duan, H.; Gong, Z. Spatial distribution and seasonal variation of phthalate esters in the Jiulong River estuary, Southeast China. Mar. Pollut. Bull. 2017, 122, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Net, S.; Rabodonirina, S.; Sghaier, R.B.; Dumoulin, D.; Chbib, C.; Tlili, I.; Ouddane, B. Distribution of phthalates, pesticides and drug residues in the dissolved, particulate and sedimentary phases from transboundary rivers (France–Belgium). Sci. Total Environ. 2015, 521–522, 152–159. [Google Scholar] [CrossRef]
- Petrovic, M.; Eljarrat, E.; Lopez de Alda, M.J.; Barcelo, D. Analysis and environmental levels of endocrinedisrupting compounds in freshwater sediments. Trends Analyt. Chem. 2001, 20, 637–648. [Google Scholar] [CrossRef]
- Oehlmann, J.; Schulte-Oehlmann, U.; Kloas, W.; Jagnytsch, O.; Lutz, I.; Kusk, K.O. A critical analysis of the biological impacts of plasticizers on wildlife. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 2047–2062. [Google Scholar] [CrossRef] [Green Version]
- Chikae, M.; Ikeda, R.; Hatano, Y.; Hasan, Q.; Morita, Y.; Tamiya, E. Effects of bis (2-ethylhexyl) phthalate, γ-hexachlorocyclohexane, and 17β-estradiol on the fry stage of medaka (Oryzias latipes). Environ. Toxicol. Pharmacol. 2004, 18, 9–12. [Google Scholar] [CrossRef]
- Latif, M.; Faheem, M. Study of Oxidative Stress and Histo-Biochemical Biomarkers of Diethyl Phthalate Induced Toxicity in a Cultureable Fish, Labeo rohita. Pak. Vet. J. 2020, 40, 2. [Google Scholar] [CrossRef]
- Bisai, K.; Pati, M.K.; Behera, B.K. Hazards of phthalate exposure on fish reproduction—A review. South Asian J. Exp. Biol. 2022, 12, 184–191. [Google Scholar] [CrossRef]
- Poopal, R.K.; Zhang, J.; Zhao, R.; Ramesh, M.; Ren, Z. Biochemical and behavior effects induced by diheptyl phthalate (DHpP) and Diisodecyl phthalate (DIDP) exposed to zebrafish. Chemosphere 2020, 252, 126498. [Google Scholar] [CrossRef]
- Moon, H.B.; Yoon, S.P.; Jung, R.H.; Choi, M. Wastewater treatment plants (WWTPs) as a source of sediment contamination by toxic organic pollutants and fecal sterols in a semi-enclosed bay in Korea. Chemosphere 2008, 73, 880–889. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.F.; Chen, C.W.; Ju, Y.R.; Dong, C.D. Determination and assessment of phthalate esters content in sediments from Kaohsiung Harbor, Taiwan. Mar. Pollut. Bull. 2017, 124, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Colon, I.; Caro, D.; Bourdony, C.J.; Rosario, O. Identification of phthalate esters in the serum of young Puerto Rican girls with premature breast development. Environ. Health Perspect. 2000, 108, 895–900. [Google Scholar] [CrossRef] [Green Version]
- Casals-Casas, C.; Desvergne, B. Endocrine Disruptors: From Endocrine to Metabolic Disruption. Annu. Rev. Physiol. 2011, 73, 135–162. [Google Scholar] [CrossRef] [Green Version]
- Kruger, T.; Long, M.; Bonefeld-Jørgensen, E.C. Plastic components affect the activation of the aryl hydrocarbon and the androgen receptor. Toxicology 2008, 246, 112–123. [Google Scholar] [CrossRef]
- Gao, C.J.; Kannan, K. Phthalates, bisphenols, parabens, and triclocarban in feminine hygiene products from the United States and their implications for human exposure. Environ. Int. 2020, 136, 105465. [Google Scholar] [CrossRef]
- Johnson, S.; Saikia, N.; Sahu, R. Phthalates in toys available in Indian market. Bull. Environ. Contam. Toxicol. 2011, 86, 621–626. [Google Scholar] [CrossRef]
- DGOS. Final Findings of PAN Review. Directorate General of Safeguards, Customs and Central Excise, Government of India. 2013. Available online: http://www.dgsafeguards.gov.in/newversion/Final%20findings%20of%20pan%20review.pdf (accessed on 20 December 2013).
- Singh, S.; Bhardwaj, A.; Verma, V.K. Remote sensing and GIS based analysis of temporal land use/land cover and water quality changes in Harike wetland ecosystem, Punjab, Insdia. J. Environ. Manag. 2020, 262, 110355. [Google Scholar] [CrossRef] [PubMed]
- Brraich, O.S.; Jangu, S. Evaluation of water quality pollution indices for heavy metal contamination monitoring in the water of Harike Wetland (Ramsar Site), India. Int. J. Sci. Res. 2015, 5, 1–6. [Google Scholar]
- Sarkar, A.; Jain, S.K. Using Remote Sensing data to study wetland dynamics–A case study of Harike Wetland. In Proceedings of the Taal2007: The 12th World Lake Conference, Kusatsu, Japan, 28 October–2 November 2007; Volume 680, p. 684. [Google Scholar]
- Tiwana, N.S.; Jerath, N.; Saxena, S.K.; Sharma, V. 69-Conservation and Management of Wetlands in Punjab-Challenges & Opportunities. In Proceedings of the National Seminar on Conservation and Restoration of Lakes (CAROL-08), Nagpur, India, 16–17 October 2008. [Google Scholar]
- Kaur, J.; Chaudhary, A.; Kaur, R.; Arora, S. Assessment of mutagenic, genotoxic, and cytotoxic potential of water samples of Harike wetland: A Ramsar site in India using different ex vivo biological systems. Ecotoxicology 2014, 23, 967–977. [Google Scholar] [CrossRef]
- Kaur, H.; Datta, S.N.; Hassan, S.S.; Bedi, J.S.; Ajeet, S. Assessment of heavy metal concentration in water, sediment and fish flesh at Harike wetland-a Ramsar site, India. J. Exp. Zool. India 2019, 22, 375–381. [Google Scholar]
- Ladhar, S.S. Status of ecological health of wetlands in Punjab, India. Aquat. Ecosyst. Health Manag. 2002, 5, 457–465. [Google Scholar] [CrossRef]
- Wu, B.; Zhang, Y.; Zhang, X.; Cheng, S. Health risk from exposure of organic pollutants through drinking water consumption in Nanjing, China. Bull. Environ. Contam. Toxicol. 2010, 84, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Kaur, R.; Kumari, A.; Sharma, G.; Singh, D.; Kaur, R. Biodegradation of endocrine disrupting chemicals benzyl butyl phthalate and dimethyl phthalate by Bacillus marisflavi RR014. J. Appl. Microbiol. 2021, 131, 1274–1288. [Google Scholar] [CrossRef] [PubMed]
- Kumari, A.; Arora, S.; Kaur, R. Comparative cytotoxic and genotoxic potential of benzyl-butyl phthalate and di-n-butyl phthalate using Allium cepa assay. Energy Ecol. Environ. 2021, 6, 244–257. [Google Scholar] [CrossRef]
- USEPA. Risk Assessment Guidance for Superfund Volume I Human Health Evaluation Manual (Part A). EPA/540/1-89/ 002. Office of Emergency and Remedial Response; U.S. Environmental Protection Agency: Washington, DC, USA, 1989. Available online: http://www.epa.gov/oswer/riskassessment/ragsa/pdf/rags-vol1-pta_complete.pdf (accessed on 20 December 2013).
- Selvaraj, K.K.; Murugasamy, M.; Nikhil, N.P.; Elaiyaraja, A.; Sampath, S.; Krishnamoorthi, V.; Ramaswamy, B.R. Investigation of distribution, sources and flux of perfluorinated compounds in major southern Indian rivers and their risk assessment. Chemosphere 2021, 277, 130228. [Google Scholar] [CrossRef]
- United States. Hazardous Site Control Division, United States. Environmental Protection Agency. Office of Solid Waste & Emergency Response. Soil Screening Guidance: User’s Guide. United States, Environmental Protection Agency, Office of Solid Waste and Emergency Response. Available online: https://archive.epa.gov/region9/superfund/web/pdf/ssg_nonrad_technical-2.pdf (accessed on May 1996).
- USEPA. Freshwater and Marine Sediment Screening Benchmark Values. 2006. Available online: http://www.epa.gov/reg3hwmd/risk/eco/index.htm (accessed on 20 December 2013).
- ECHA. Guidance on Information Requirements and Chemical Safety Assessment—Chapter R.10: Characterisation of Dose [Concentration]-Response for Environment; European Chemicals Agency: Helsinki, Finland, 2008. Available online: http://echa.europa.eu/documents/10162/13632/information_requirements_r10_en.pdf (accessed on May 2008).
- USEPA. Ecological Structure Activity Relationships (ECOSAR) Class Program, Version 1.11. 2011. Available online: http://www.epa.gov/oppt/newchems/tools/21ecosar.htm (accessed on 12 September 2013).
- Ramaswamy, B.R.; Shanmugam, G.; Velu, G.; Rengarajan, B.; Larsson, D.G. GC–MS analysis and ecotoxicological risk assessment of triclosan, carbamazepine and parabens in Indian rivers. J. Hazard. Mater. 2011, 186, 1586–1593. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, G.; Sampath, S.; Selvaraj, K.K.; Larsson, D.J.; Ramaswamy, B.R. Non-steroidal anti-inflammatory drugs in Indian rivers. Environ. Sci. Pollut. Res. 2014, 21, 921–931. [Google Scholar] [CrossRef]
- Gao, D.W.; Li, Z.; Wang, H.; Liang, H. An overview of phthalate acid ester pollution in China over the last decade: Environmental occurrence and human exposure. Sci. Total Environ. 2018, 645, 1400–1409. [Google Scholar] [CrossRef]
- Zhou, L.J.; Zhang, Q.; Shi, L.L. The formulation methods of priority water pollutants and environmental quality standards in the EU and their reference significance to China. Environ. Monit. Forewarn. 2019, 11, 1–9. [Google Scholar] [CrossRef]
- Zang, Y.; Jiao, Y.; Li, Z.; Tao, Y.; Yang, Y. Hazards of phthalates (PAEs) exposure: A review of aquatic animal toxicology studies. Sci. Total Environ. 2021, 771, 145418. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Qin, N.; Kong, X.Z.; Liu, W.X.; He, Q.S.; Wang, Q.M.; Xu, F.L. Water quality benchmarking (WQB) and priority control screening (PCS) of persistent toxic substances (PTSs) in China: Necessity, method and a case study. Sci. Total Environ. 2014, 472, 1108–1120. [Google Scholar] [CrossRef] [PubMed]
- Elaiyaraja, A.; Mayilsamy, M.; Vimalkumar, K.; Nikhil, N.P.; Noorani, P.M.; Bommuraj, V.; Rajendran, R.B. Aquatic and human health risk assessment of Humanogenic Emerging Contaminants (HECs), Phthalate Esters from the Indian Rivers. Chemosphere 2022, 306, 135624. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, J.; Liu, H.; Wan, G.; Zhang, S. The occurrence and ecological risk assessment of phthalate esters (PAEs) in urban aquatic environments of China. Ecotoxicology 2015, 24, 967–984. [Google Scholar] [CrossRef]
- Lee, Y.M.; Lee, J.E.; Choe, W.; Kim, T.; Lee, J.Y.; Kho, Y.; Zoh, K.D. Distribution of phthalate esters in air, water, sediments, and fish in the Asan Lake of Korea. Environ. Int. 2019, 126, 635–643. [Google Scholar] [CrossRef]
- Zhao, X.; Shen, J.M.; Zhang, H.; Li, X.; Chen, Z.L.; Wang, X.C. The occurrence and spatial distribution of phthalate esters (PAEs) in the Lanzhou section of the Yellow River. Environ. Sci. Pollut. Res. 2020, 27, 19724–19735. [Google Scholar] [CrossRef] [PubMed]
- Muhamad, A.; Karim, A.T.A.; Zakaria, N.F.; Rapok, K.J.A. Determination of phthalate ester compounds in Sembrong river sediment. J. Techno-Soc. 2016, 8, 2. [Google Scholar]
- CPCB. River Stretches for Restoration for Water Quality. 2015. Available online: https://cpcb.nic.in/annual-report.php (accessed on February 2015).
- Subedi, B.; Balakrishna, K.; Sinha, R.K.; Yamashita, N.; Balasubramanian, V.G.; Kannan, K. Mass loading and removal of pharmaceuticals and personal care products, including psychoactive and illicit drugs and artificial sweeteners, in five sewage treatment plants in India. J. Environ. Chem. Eng. 2015, 3, 2882–2891. [Google Scholar] [CrossRef]
- Kumar, V.; Sharma, A.; Kumar, R.; Bhardwaj, R.; Kumar Thukral, A.; Rodrigo-Comino, J. Assessment of heavy-metal pollution in three different Indian water bodies by combination of multivariate analysis and water pollution indices. Hum. Ecol. Risk Assess. 2020, 26, 1–16. [Google Scholar] [CrossRef]
- USEPA. Phthalates Action Plan. 2012. Available online: http://www.epa.gov/oppt/existingchemicals/pubs/actionplans/phthalates_actionplan_revised_2012-03-14.pdf (accessed on 20 December 2013).
- Ai, S.; Gao, X.; Wang, X.; Li, J.; Fan, B.; Zhao, S. Exposure and Tiered Ecological Risk Assessment of Phthalate Esters in the Surface Water of Poyang Lake, China. Chemosphere 2021, 262, 127864. [Google Scholar] [CrossRef] [PubMed]
- Baloyi, N.D.; Tekere, M.; Maphangwa, K.W.; Masindi, V. Insights into the prevalence and impacts of phthalate esters in aquatic ecosystems. Front. Environ. Sci. 2021, 9, 684190. [Google Scholar] [CrossRef]
- Harris, G.A.; Henttu, P.; Parker, M.G.; Sumpter, J.P. The estrogenic activity of phthalate esters in vitro. Environ. Health Perspect. 1997, 105, 802–811. [Google Scholar] [CrossRef]
- Ramzi, A.; Gireeshkumar, T.R.; Rahman, K.H.; Balachandran, K.K.; Shameem, K.; Chacko, J.; Chandramohanakumar, N. Phthalic acid esters—A grave ecological hazard in Cochin estuary, India. Mar. Pollut. Bull. 2020, 152, 110899. [Google Scholar] [CrossRef]
- Paluselli, A.; Aminot, Y.; Galgani, F.; Net, S.; Sempere, R. Occurrence of phthalate acid esters (PAEs) in the northwestern Mediterranean Sea and the Rhone River. Prog. Oceanogr. 2018, 163, 221–231. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y.; Alomirah, H.; Cho, H.-S.; Minh, T.B.; Mohd, M.A.; Nakata, H. Occurrence of phthalate metabolites in human urine from several Asian countries. Environ. Sci. Technol. 2011, 45, 3138–3144. [Google Scholar] [CrossRef]
- López-Carrillo, L.; Hernández-Ramírez, R.U.; Calafat, A.M.; Torres-Sánchez, L.; Galván-Portillo, M.; Needham, L.L.; Cebrián, M.E. Exposure to phthalates and breast cancer risk in northern Mexico. Environ. Health Perspect. 2010, 118, 539–544. [Google Scholar] [CrossRef]
- Hsieh, T.H.; Tsai, C.F.; Hsu, C.Y.; Kuo, P.L.; Lee, J.N.; Chai, C.Y. Phthalates induce proliferation and invasiveness of estrogen receptor-negative breast cancer through the AhR/HDAC6/c-Myc signaling pathway. FASEB J. 2012, 26, 778–787. [Google Scholar] [CrossRef] [Green Version]
- Sung, H.H.; Kao, W.Y.; Su, Y.J. Effects and toxicity of phthalate esters to hemocytes of giant freshwater prawn, Macrobrachium rosenbergii. Aquat. Toxicol. 2003, 64, 25–37. [Google Scholar] [CrossRef]
- Xu, H.; Shao, X.; Zhang, Z.; Zou, Y.; Wu, X.; Yang, L. Oxidative stress and immune related gene expression following exposure to di-n-butyl phthalate and diethyl phthalate in zebrafish embryos. Ecotoxicol. Environ. Saf. 2013, 93, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.L.; Sung, H.H. The toxic effect of phthalate esters on immune responses of giant freshwater prawn (Macrobrachium rosenbergii) via oral treatment. Aquat. Toxicol. 2005, 74, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhang, Z.; Zhang, L.; Tian, F.; Tang, Z.; Cai, W.; Jia, X. Effects of di-n-butyl phthalate on gills-and liver-specific EROD activities and CYP1A levels in juvenile red snapper (Lutjanus argentimaculatus). Comp. Biochem. Physiol. 2020, 232, 108757. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhu, H.; Kannan, K. A review of biomonitoring of phthalate exposures. Toxics 2019, 7, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| S. No. | Site Code | Latitude | Longitude | Location |
|---|---|---|---|---|
| 1 | 1 | 31°09′04″ N | 74°57′02″ E | Forest head station Harike wetland |
| 2 | 2 | 31°08′59″ N | 74°57′13″ E | Confluence point 1 |
| 3 | 3 | 31°09′05″ N | 74°57′49″ E | Confluence point 2 |
| 4 | 4 | 31°09′09″ N | 74°58′20″ E | Confluence point 3 |
| 5 | 5 | 31°09′04″ N | 74°58′45″ E | Sutlej River |
| 6 | 6 | 31°09′21″ N | 74°58′39″ E | Beas River |
| 7 | 7 | 31°08′38″ N | 74°56′58″ E | Near Gurudwara Nanaksar Sahib |
| 8 | 8 | 31°08′44″ N | 74°56′52″ E | Near Harike Barrage |
| 9 | 9 | 31°08′02″ N | 74°56′56″ E | Rajasthan Feeder |
| 10 | 10 | 31°07′57″ N | 74°57′01″ E | Ferozpur Feeder |
| Phthalate Esters | Winter | Summer | Rainy | |||
|---|---|---|---|---|---|---|
| Adult | Child | Adult | Child | Adult | Child | |
| DMP(mg/kg dw) | 1.125 | 2.701 | 1.465 | 3.516 | 1.323 | 3.175 |
| DEP(mg/kg dw) | 0.549 | 1.319 | 0.616 | 1.477 | 1.607 | 3.855 |
| BBP(mg/kg dw) | 1.730 | 4.153 | 0.205 | 0.489 | 6.143 | 14.741 |
| DBP(mg/kg dw) | 5.422 | 13.014 | 4.999 | 11.999 | 3.639 | 8.735 |
| Phthalate Esters | Winter | Summer | Rainy | |||
|---|---|---|---|---|---|---|
| Adult | Child | Adult | Child | Adult | Child | |
| DMP | 0.113 | 0.270 | 0.146 | 0.352 | 0.133 | 0.318 |
| DEP | 0.687 | 1.649 | 0.769 | 1.847 | 2.008 | 4.819 |
| BBP | 8.654 | 20.768 | 1.021 | 2.449 | 30.711 | 73.706 |
| DBP | 54.224 | 130.138 | 49.997 | 119.993 | 36.394 | 87.346 |
| Phthalate Esters | Winter Season | Summer Season | Rainy Season | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Fish | Daphnia | Green Algae | Fish | Daphnia | Green Algae | Fish | Daphnia | Green Algae | |
| DMP | 7.22 | 3.04 | 5.65 | 9.4 | 3.91 | 7.35 | 8.48 | 3.53 | 6.64 |
| DEP | 12.11 | 5.32 | 12.22 | 13.56 | 5.96 | 13.68 | 35.37 | 15.55 | 35.69 |
| DAP | 154.25 | 68.62 | 155.67 | 146.04 | 64.97 | 147.39 | 169.85 | 75.57 | 171.43 |
| DPP | 143.67 | 71.15 | 211.94 | 95.92 | 47.50 | 141.50 | 117.57 | 58.22 | 173.43 |
| BPP | 57.44 | 30.09 | 103.31 | 6.78 | 3.55 | 12.19 | 203.88 | 106.82 | 366.69 |
| DBP | 155.11 | 75.22 | 246.94 | 143.01 | 69.36 | 227.69 | 104.1 | 50.48 | 165.74 |
| DcHP | 422.7 | 245.17 | 1089.65 | 105.3 | 61.08 | 271.45 | 534.42 | 309.96 | 1377.59 |
| Phthalate | Winter Season | Summer Season | Rainy Season | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Fish | Daphnia | Green Algae | Fish | Daphnia | Green Algae | Fish | Daphnia | Green Algae | |
| DMP | 9.80 | 4.08 | 7.67 | 16.99 | 7.07 | 13.30 | 4.18 | 1.74 | 3.27 |
| DEP | 2.72 | 1.19 | 2.75 | 5.92 | 2.59 | 5.97 | 3.68 | 1.62 | 3.71 |
| DAP | 6.34 | 2.82 | 6.40 | 17.66 | 7.86 | 17.82 | 9.48 | 4.22 | 9.56 |
| DPP | 2.72 | 1.35 | 4.02 | 2.07 | 1.03 | 3.05 | 5.02 | 2.49 | 7.39 |
| BPP | 2.05 | 0.86 | 2.96 | 2.01 | 0.84 | 2.89 | 3.47 | 1.46 | 4.99 |
| DBP | 4.24 | 1.65 | 5.40 | 5.69 | 2.21 | 7.25 | 4.33 | 1.68 | 5.52 |
| DcHP | 0.25 | 0.14 | 0.63 | 0.36 | 0.21 | 0.91 | 0.74 | 0.43 | 1.89 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaudhary, G.; Jasrotia, A.; Raj, P.; Kaur, R.; Kumari, A.; Rajput, V.D.; Minkina, T.; Mandzhieva, S.; Kaur, R. Contamination of Water and Sediments of Harike Wetland with Phthalate Esters and Associated Risk Assessment. Water 2023, 15, 1009. https://doi.org/10.3390/w15061009
Chaudhary G, Jasrotia A, Raj P, Kaur R, Kumari A, Rajput VD, Minkina T, Mandzhieva S, Kaur R. Contamination of Water and Sediments of Harike Wetland with Phthalate Esters and Associated Risk Assessment. Water. 2023; 15(6):1009. https://doi.org/10.3390/w15061009
Chicago/Turabian StyleChaudhary, Gauri, Akanksha Jasrotia, Pushap Raj, Rajanbir Kaur, Arpna Kumari, Vishnu D. Rajput, Tatiana Minkina, Saglara Mandzhieva, and Rajinder Kaur. 2023. "Contamination of Water and Sediments of Harike Wetland with Phthalate Esters and Associated Risk Assessment" Water 15, no. 6: 1009. https://doi.org/10.3390/w15061009






