Cyanobacterial Blooms Increase Functional Diversity of Metazooplankton in a Shallow Eutrophic Lake
Abstract
1. Introduction
2. Materials and Methods
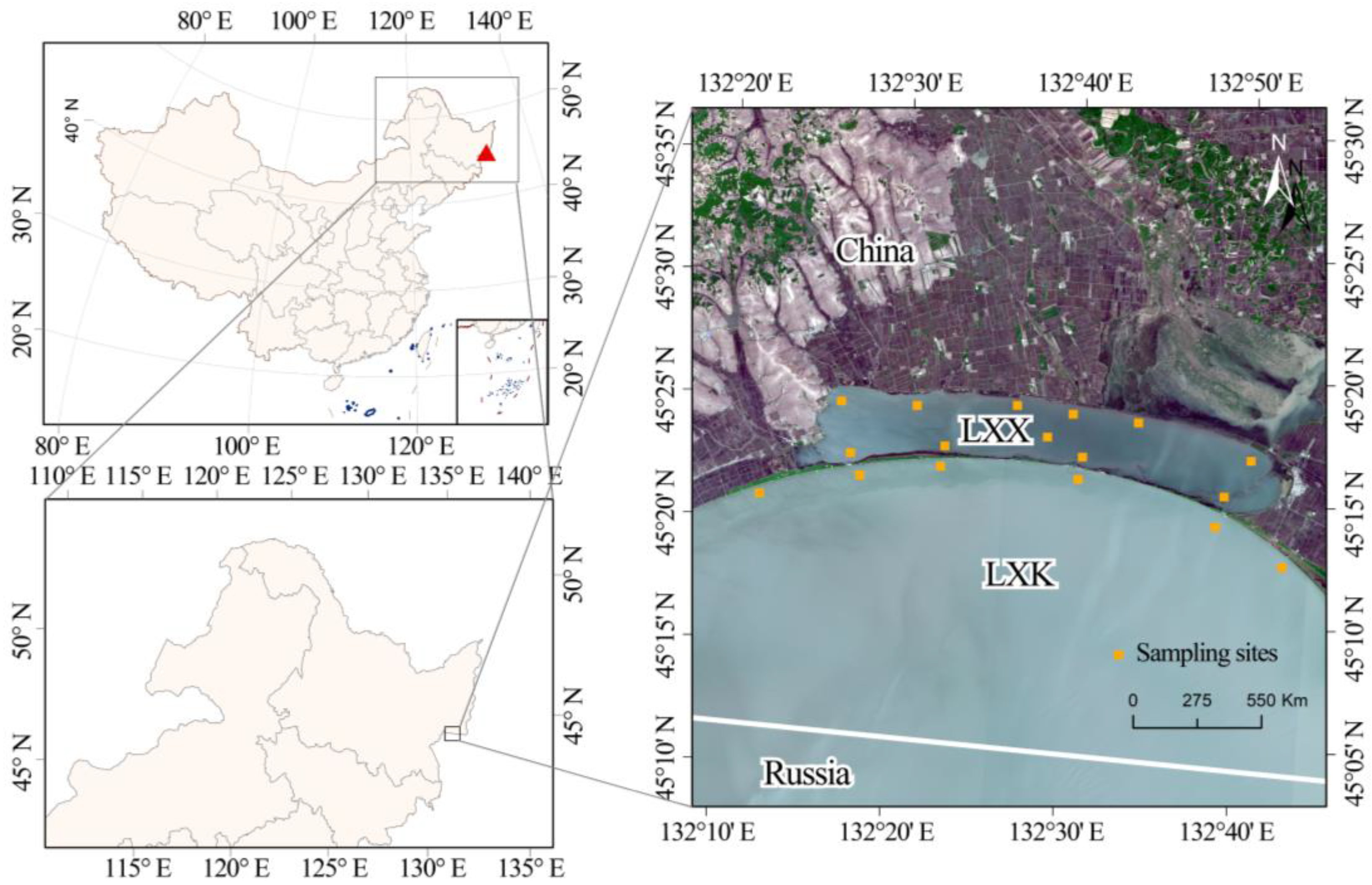
2.1. Study Area
2.2. Samplings
2.3. Data Analyses
2.3.1. Species Diversities
2.3.2. Functional Diversities
2.3.3. Statistical Analyses
3. Results
3.1. Variations in Environmental Variables and Cyanobacteria Communities
3.2. Biomass and Diversity of Metazooplankton during Cyanobacterial Bloom Period and Non-Bloom Period
3.3. Spatial Patterns of Metazooplankton Biomass and Diversity
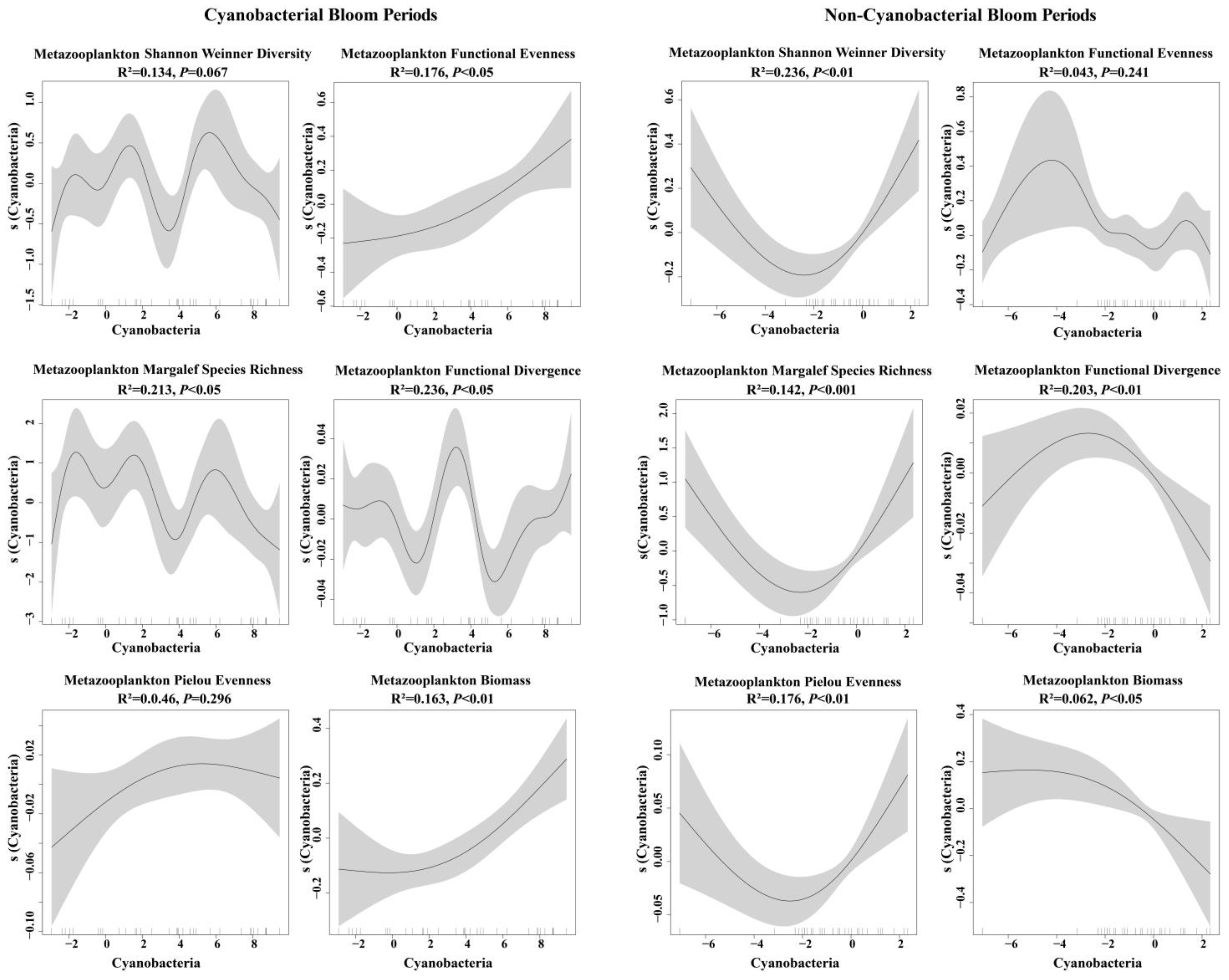
3.4. Impacts of Cyanobacteria on Metazooplankton Biomass and Diversity
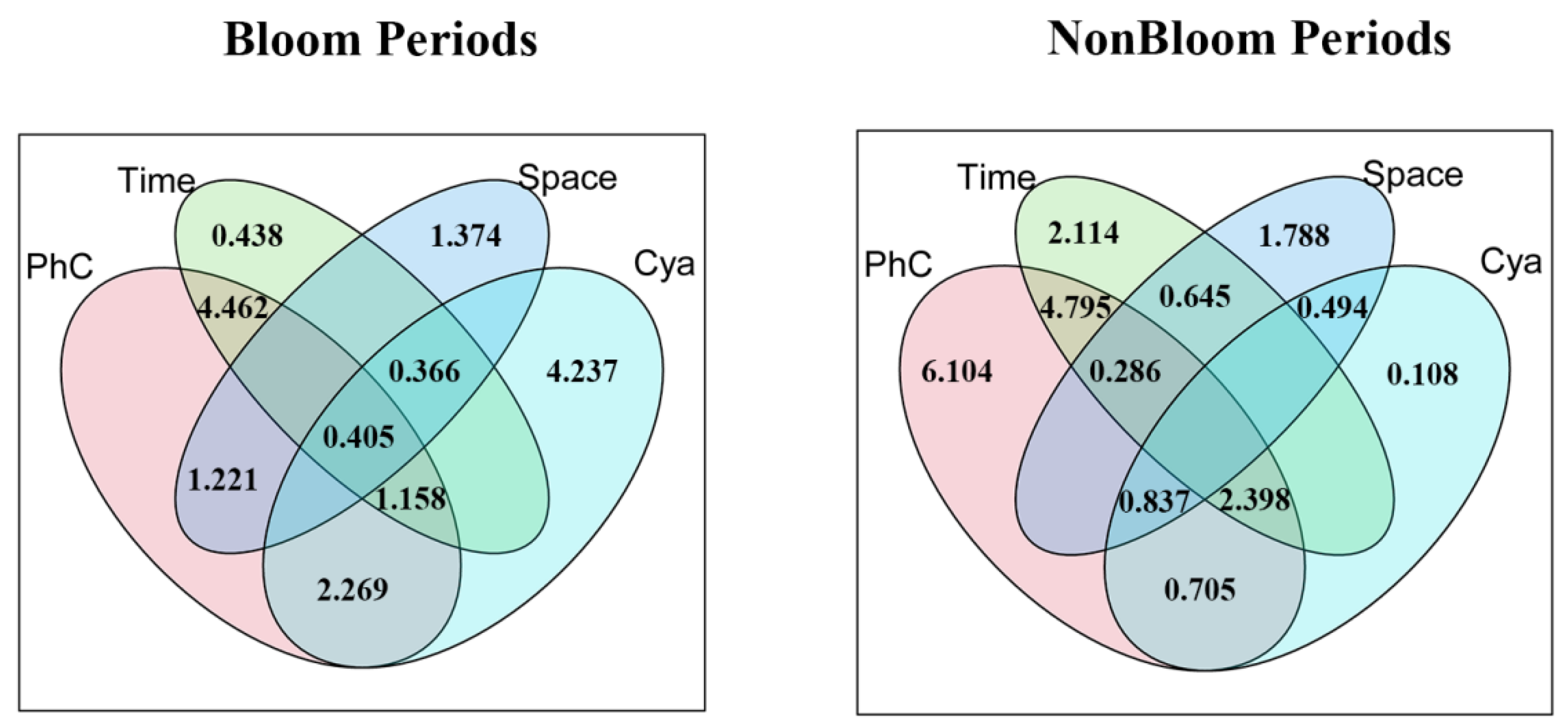
3.5. Partitioning Influences of Physicochemical, Cyanobacterial, and Spatio-Temporal Factors on Metazooplankton Community
4. Discussion
4.1. Variation in Abundance of Cyanobacteria and Metazooplankton
4.2. Variation in Metazooplankton Diversity and Its Interaction with Cyanobacteria
4.3. Integrated Analysis of the Influence of Physicochemical Factors, Period, Space, and Cyanobacteria on Metazooplankton
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Yin, L.P.; Xia, S.; Gu, J.; Li, T.; Chen, L. Nails float in the Qingcaosha Reservoir in Shanghai Characteristics of crustacean community structure. J. Shanghai Ocean. Univ. 2018, 27, 864–874. [Google Scholar]
- Gu, B.; Liu, Z.; Li, K. Limnology: Inland Water Ecosystem; Higher Education Press: Beijing, China, 2011; pp. 388–423. [Google Scholar]
- Frantal, D.; Agatha, S.; Beisser, D.; Boenigk, J.; Darienko, T.; Dirren-Pitsch, G.; Filker, S.; Gruber, M.; Kammerlander, B.; Nachbaur, L.; et al. Molecular data reveal a cryptic diversity in the genus Urotricha (Alveolata, Ciliophora, Prostomatida), a key player in freshwate lakes, with remarks on morphology, food preferences, and distribution. Front. Microbiol. 2022, 12, 787290. [Google Scholar] [CrossRef] [PubMed]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.H.; Visser, P.M. Cyanobacterial blooms. Nat. Rev. Microbiol. 2018, 16, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Nwosu, E.C.; Brauer, A.; Monchamp, M.-E.; Pinkerneil, S.; Bartholomäus, A.; Theuerkauf, M.; Schmidt, J.-P.; Stoof-Leichsenring, K.R.; Wietelmann, T.; Kaiser, J.; et al. Early human impact on lake cyanobacteria revealed by a Holocene record of sedimentary ancient DNA. Commun. Biol. 2023, 6, 72. [Google Scholar] [CrossRef]
- Yang, J.R.; Lv, H.; Isabwe, A.; Liu, L.; Yu, X.; Chen, H.; Yang, J. Disturbance induced phytoplankton regime shifts and recovery of cyanobacteria dominance in two subtropical reservoirs. Water Res. 2017, 120, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.M.; Shi, W.Q.; Chen, Q.W.; Lauridsen, T.L. Spatial and temporal variations reveal the response of zooplankton to cyanobacteria. Harmful Algae 2017, 64, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.H.; Zhuang, Y.Y.; Chen, H.J.; Lu, S.H.; Li, Y.X.; Ge, R.P.; Chen, C.; Liu, G.X. Effects of prorocentrum donghaiense bloom on zooplankton functional groups in the coastal waters of the east china sea. Mar. Pollut. Bull. 2021, 172, 112878. [Google Scholar] [CrossRef]
- Amorim, C.A.; Moura, A.N. Ecological impacts of freshwater algal blooms on water quality, plankton biodiversity, structure, and ecosystem functioning. Sci. Total Environ. 2020, 758, 143605. [Google Scholar] [CrossRef]
- Ger, K.A.; Urrutia-Cordero, P.; Frost, P.C.; Hansson, L.-A.; Sarnelle, O.; Wilson, A.E.; Lürling, M. The interaction between cyanobacteria and zooplankton in a more eutrophic world. Harmful Algae 2016, 54, 128–144. [Google Scholar] [CrossRef]
- Yao, C.; He, T.R.; Xu, Y.Y.; Ran, S.; Long, S.X. Mercury bioaccumulation in zooplankton and its relationship with eutrophication in the waters in the karst region of guizhou province, southwest china. Environ. Sci. Pollut. Res. 2020, 27, 8596–8610. [Google Scholar] [CrossRef]
- Kaur, R.P.; Sharma, A.; Sharma, A.K. Impact of fear effect on plankton-fish system dynamics incorporating zooplankton refuge. Chaos Solitons Fractals 2021, 143, 110563. [Google Scholar] [CrossRef]
- Zhao, K.; Wang, L.Z.; Wang, Q.X. Influence of cyanobacterial blooms and environmental variation on zooplankton and eukaryotic phytoplankton in a large, shallow, eutrophic lake in china. Sci. Total Environ. 2021, 773, 145421. [Google Scholar] [CrossRef] [PubMed]
- Li, C.C.; Feng, W.Y.; Chen, H.Y.; Li, X.F.; Song, F.H.; Guo, W.J.; Giesy, J.P.; Sun, F.H. Temporal variation in zooplankton and phytoplankton community species composition and the affecting factors in lake taihu-a large freshwater lake in china. Environ. Pollut. 2019, 245, 1050–1057. [Google Scholar] [CrossRef] [PubMed]
- Moustaka-Gouni, M.; Sommer, U. Effects of harmful blooms of large-sized and colonial cyanobacteria on aquatic food webs. Water 2020, 12, 1587. [Google Scholar] [CrossRef]
- Wang, K.; Mou, X.; Cao, H.; Struewing, I.; Allen, J.; Lu, J. Co-occurring microorganisms regulate the succession of cyanobacterial harmful algal blooms. Environ. Pollut. 2021, 288, 117682. [Google Scholar] [CrossRef] [PubMed]
- Carey, C.C.; Cottingham, K.L.; Weathers, K.C.; Brentrup, J.A.; Ruppertsberger, N.M.; Ewing, H.A.; Hairston, N.G., Jr. Experimental blooms of the cyanobacterium Gloeotrichia echinulata increase phytoplankton biomass, richness and diversity in an oligotrophic lake. J. Plankton Res. 2014, 36, 364–377. [Google Scholar] [CrossRef]
- Bockwoldt, K.A.; Nodine, E.R.; Mihuc, T.B.; Shambaugh, A.D.; Stockwell, J.D. Reduced phytoplankton and zooplankton diversity associated with increased cyanobacteria in Lake Champlain. USA. J. Contemp. Water Res. Edu. 2017, 160, 100–118. [Google Scholar] [CrossRef]
- Villéger, S.; Mason, N.W.H.; Mouillot, D. New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology 2008, 89, 2290–2301. [Google Scholar] [CrossRef] [PubMed]
- Obertegger, U.; Smith, H.A.; Flaim, G.; Wallace, R.L. Using the guild ratio to characterize pelagicrotifer communities. Hydrobiologia 2011, 662, 157–162. [Google Scholar] [CrossRef]
- Litchman, E.; Ohman, M.D.; Kiørboe, T. Trait-based approaches to zooplankton communities. J. Plankton Res. 2013, 35, 473–484. [Google Scholar] [CrossRef]
- Boyer, J.; Rollwagen-Bollens, G.; Bollens, S.M. Microzooplankton grazing before, during and after a cyanobacterial bloom in Vancouver Lake, Washington. USA. Aquat. Microb. Ecol. 2011, 64, 163–174. [Google Scholar] [CrossRef]
- Yuan, Y.X.; Jiang, M.; Liu, X.T.; Yu, H.X.; Otte, M.L.; Ma, C.X.; Her, Y.G. Environmental variables influencing phytoplankton communities in hydrologically connected aquatic habitats in the lake xingkai basin. Ecol. Indic. 2018, 91, 1–12. [Google Scholar] [CrossRef]
- Ma, J.; Qin, B.; Paerl, H.W.; Brookes, J.D.; Hall, N.S.; Shi, K.; Zhou, Y.; Guo, J.; Li, Z.; Xu, H.; et al. The persistence of cyanobacterial (Microcystis spp.) blooms throughout winter in Lake Taihu, China. Limnol. Oceanogr. 2016, 61, 711–722. [Google Scholar] [CrossRef]
- Moustaka-Gouni, M.; Vardaka, E.; Michaloudi, E.; Kormas, K.A.; Tryfon, E.; Mihalatou, H.; Gkelis, S.; Lanaras, T. Plankton food web structure in a eutrophic polymictic lake with a history in toxic cyanobacterial blooms. Limnol. Oceanogr. 2006, 51, 715–727. [Google Scholar] [CrossRef]
- GB3838-2002; Surface Water Environmental Quality Standard. China Environmental Science Press: Beijing, China, 2002.
- Hu, H.J.; Wei, Y.X. Freshwater Algae in China: System, Classification and Ecology; Science Press: Beijing, China, 2006. [Google Scholar]
- Wang, J.J. Fauna of freshwater Rotifera of China; Institute of Hydrobiology, Chinese Academy of Sciences, Science Press: Beijing, China, 1961. [Google Scholar]
- Jiang, X.Z.; Du, N.S. Zoology of China: Arthropods Crustaceans Freshwater Clavicornis; Science Press: Beijing, China, 1979. [Google Scholar]
- Witek, Z.; Breuel, G.; Wolska-Py, M.; Gruszka, P.; Sujak, D. Comparison of Different Methods of Baltic Zooplankton Biomass Estimations. In Proceedings of the 13th Symposium of the Baltic Marine Biologists, Riga-Jurmala, Latvia, August 31–4 September 1993; pp. 87–92. [Google Scholar]
- Shannon, E.; Weaver, W. The Mathematical Theory of Communication; University Illinois Press: London, UK, 1949; pp. 296–297. [Google Scholar]
- Margalef, R. Information theory in ecology. Gen. Syst. 1958, 3, 36–71. [Google Scholar]
- Pielou, C. An Introduction to Mathematical Ecology; Wiley Interscience: New York, NY, USA, 1969. [Google Scholar]
- Shen, J.R. Fauma Sinica, Crustacea: Freshwater Copepoda; Science Press: Beijing, China, 1979. [Google Scholar]
- Lu, Z.; Ye, J.; Chen, Z.; Xiao, L.; Lei, L.; Han, B.P.; Paerl, H.W. Cyanophycin accumulated under nitrogen-fluctuating and high-nitrogen conditions facilitates the persistent dominance and blooms of raphidiopsis raciborskii in tropical waters. Water Res. 2022, 214, 118215. [Google Scholar] [CrossRef]
- Zhou, F.X.; Chen, J.H. Microbiological Map of Freshwater; Chemical Industry Press: Beijing, China, 2005. [Google Scholar]
- Kuo, Y.-M.; Yang, J.; Liu, W.-W.; Zhao, E.; Li, R.; Yao, L. Using generalized additive models to investigate factors influencing cyanobacterial abundance through phycocyanin fluorescence in East Lake, China. Environ. Monit. Assess. 2018, 190, 599. [Google Scholar] [CrossRef]
- Yu, X.; Zhuge, Y.S.; Liu, X.B.; Du, Q.; Tan, H. Evolution mechanism of dissolved oxygen stratification in a large deep reservoir. Lake Sci. 2020, 32, 1496–1507. [Google Scholar]
- Yang, W.H.; Feng, D.D.; Yang, F.; Li, W.; Zhou, X.; Yao, Z.; Wang, L. Variation characteristics of dissolved oxygen and metabolic rate during the ice-covered period. Lake Sci. 2022, 34, 2156–2168. [Google Scholar]
- Sommer, U.; Gliwicz, Z.M.; Lampert, W.; Duncan, A. The PEG-model of seasonal succession of planktonic events in fresh waters. Arch. Hydrobiol. 1986, 106, 433–471. [Google Scholar] [CrossRef]
- Miranda, M.; Noyma, N.; Pacheco, F.S.; de Magalhaes, L.; Pinto, E.; Santos, S.; Soares MF, A.; Huszar, V.L.; Lu, M.; Marinho, M.M. The efficiency of combined coagulant and ballast to remove harmful cyanobacterial blooms in a tropical shallow system. Harmful Algae 2017, 65, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Kosten, S.; Huszar, V.L.M.; Bécares, E.; Costa, L.S.; Donk, E.; Hansson, L.-A.; Jeppesen, E.; Kruk, C.; Lacerot, G.; Mazzeo, N.; et al. Warmer climates boost cyanobacterial dominance in shallow lakes. Glob. Chang. Biol. 2012, 18, 118–126. [Google Scholar] [CrossRef]
- Stein, L.Y. Microbiology: Cyanate fuels the nitrogen cycle. Nature 2015, 524, 43–44. [Google Scholar] [CrossRef] [PubMed]
- Frenken, T.; Wolinska, J.; Tao, Y.L.; Rohrlack, T.; Agha, R. Infection of filamentous phytoplankton by fungal parasites enhances herbivory in pelagic food webs. Limnol. Oceanogr. 2020, 65, 2618–2626. [Google Scholar] [CrossRef]
- DeMott, W.R.; Gulati, R.D.; Van Donk, E. Daphnia food limitation in three hypereutrophic Dutch lakes: Evidence for exclusion of large-bodied species by interfering filaments of cyanobacteria. Limnol. Oceanogr. 2001, 46, 2054–2060. [Google Scholar] [CrossRef]
- Brett MT Müller-Navarra, D.C. The role of highly unsaturated fatty acids in aquatic food web processes. Freshw. Biol. 1997, 38, 483–499. [Google Scholar] [CrossRef]
- Josue, I.I.P.; Cardoso, S.J.; Miranda, M.; Mucci, M.; Ger, K.A.; Roland, F.; Marinho, M.M. Cyanobacterial dominance drives zooplankton functional dispersion. Hydrobiologia 2019, 831, 149–161. [Google Scholar] [CrossRef]
- Josue, I.I.P.; Sodre, E.O.; Setubal, R.B.; Cardoso, S.J.; Roland, F.; Figueiredo-Barros, M.P.; Bozelli, R.L. Zooplankton functional diversity as an indicator of a long-term aquatic restoration in an amazonian lake. Restor. Ecol. 2021, 29, e13365. [Google Scholar] [CrossRef]
- Borics, G.; Tóthmérész, B.; Lukács, B.A.; Várbíró, G. Functional groups of phytoplankton shaping diversity of shallow lake ecosystems. Hydrobiologia 2012, 698, 251–262. [Google Scholar] [CrossRef]
- Krztoń, W.; Kosiba, J.; Pociecha, A.; Wilk-Woźniak, E. The effect of cyanobacterial blooms on bio-and functional diversity of zooplankton communities. Biodivers. Conserv. 2019, 28, 1815–1835. [Google Scholar] [CrossRef]
- Yang, Z.; Kong, F.; Shi, X.; Cao, H. Morphological response of Microcystis aeruginosa to grazing by different sorts of zooplankton. Hydrobiologia 2006, 563, 225–230. [Google Scholar] [CrossRef]
- Visser, P.M.; Verspagen JM, H.; Sandrini, G.; Stal, L.J.; Matthijs HC, P.; Davis, T.W.; Paerl, H.W.; Huisman, J. How rising CO2 and global warming may stimulate harmful cyanobacterial blooms. Harmful Algae 2016, 54, 145–159. [Google Scholar] [CrossRef] [PubMed]
- Druga, B.; Ukrainczyk, N.; Weise, W.; Koenders, K.; Lackner, S. Interaction between wastewater microorganisms and geopolymer or cementitious materials: Biofilm characterization and deterioration characteristics of mortars. Int. Biodeterior. Biodegrad. 2018, 134, 58–67. [Google Scholar] [CrossRef]
- Wang, K.; Razzano, M.; Mou, X. Cyanobacterial blooms alter the relative importance of neutral and selective processes in assembling freshwater bacterioplankton community. Sci. Total Environ. 2020, 706, 135724. [Google Scholar] [CrossRef]
- Konopka, A.; Lindemann, S.; Fredrickson, J. Dynamics in microbial communities: Unraveling mechanisms to identify principles. ISME J. 2015, 9, 1488. [Google Scholar] [CrossRef]
- Liu, L.; Chen, H.; Liu, M.; Yang, J.R.; Xiao, P.; Wilkinson, D.M.; Yang, J. Response of the eukaryotic plankton community to the cyanobacterial biomass cycle over 6 years in two subtropical reservoirs. ISME J. 2019, 13, 2196–2208. [Google Scholar] [CrossRef]
- Jiang, X.D.; Xie, J.H.; Xu, Y.; Zhong, W.F.; Zhu, X.; Zhu, C.D. Increasing dominance of small zooplankton with toxic cyanobacteria. Freshw. Biol. 2016, 62, 429–443. [Google Scholar] [CrossRef]
- Wang, S.B.; Shi, Z.J.; Geng, H.; Wu, L.Y.; Cao, Y.M. Effects of environmental factors on functional diversity of crustacean plankton community. J. Lake Sci. 2021, 33, 1220–1229. [Google Scholar] [CrossRef]
- Zhang, H.R.; Jiang, C.D. Resurrection of dormant zooplankton grazers reveals multiple evolutionary responses to toxic cyanobacteria. Limnol. Oceanogr. 2022, 67, 2000–2011. [Google Scholar] [CrossRef]
- Li, W.; Qin, B. Dynamics of spatiotemporal heterogeneity of cyanobacterial blooms in large eutrophic Lake Taihu, China. Hydrobiologia 2019, 833, 81–93. [Google Scholar] [CrossRef]
- Søndergaard, M.; Lauridsen, T.L.; Johansson, L.S.; Jeppesen, E. Repeated Fish Removal to Restore Lakes:Case Study of Lake Væng, Denmark—Two Biomanipulations during 30 Years of Monitoring. Water 2017, 9, 43. [Google Scholar] [CrossRef]


| Functional Traits | Ecological Functions | Variable Types | Classifications |
|---|---|---|---|
| Body length | Growth | Continuous variable | Body length (mm) |
| Feeding type | Feeding | Categorical variable | Bosmina, Chydorus, Daphnia, Sida, Raptorial, or Stationary suspension |
| Escape strategy | Survival | Categorical variable | Pausing and jumping, not moving, rapid swimming or reduced swimming |
| Habitat type | Growth | Categorical variable | Littoral or pelagic |
| Trophic group | Growth | Categorical variable | Herbivorous, omnivorous, or carnivorous |
| July | September | January | May | Mean | |
|---|---|---|---|---|---|
| DO (mg/L) | 7.07 ± 0.30 c | 9.47 ± 1.22 b | 13.65 ± 1.09 a | 9.62 ± 0.55 b | 9.95 ± 2.73 |
| Temp (°C) | 29.10 ± 3.20 a | 13.41 ± 0.93 b | 2.36 ± 1.61 c | 11.71 ± 3.68 b,c | 14.14 ± 11.09 |
| TN (mg/L) | 0.76 ± 0.21 c | 0.58 ± 0.16 c | 1.81 ± 0.83 b | 3.69 ± 2.04 a | 1.71 ± 1.439 |
| TP (mg/L) | 0.06 ± 0.02 c | 0.06 ± 0.02 c | 0.11 ± 0.04 a | 0.08 ± 0.04 b | 0.08 ± 0.03 |
| CODMn (mg/L) | 3.96 ± 1.10 b | 4.64 ± 1.52 a | 4.37 ± 1.43 a | 4.2 ± 1.29 a | 4.29 ± 0.29 |
| TN/TP | 14.6 ± 5.13 c | 10.48 ± 1.47 c | 19.58 ± 16.74 b | 87.89 ± 124.05 a | 33.14 ± 36.69 |
| Conductivity (S/cm) | 213 ± 44 b | 242 ± 86 a | 222 ± 57.16 b | 105 ± 33.42 c | 195 ± 79 |
| pH | 9.44 ± 0.26 a | 7.38 ± 0.68 c | 8.12 ± 0.36 b | 9.11 ± 0.35 a | 8.15 ± 0.93 |
| NH4-N (mg/L) | 0.06 ± 0.00 b | 0.07 ± 0.04 b | 0.16 ± 0.08 a | 0.03 ± 0.03 c | 0.08 ± 0.07 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, X.; Yuan, Y.; Zou, Y.; Qin, L.; Zhu, X.; Zhu, Y.; Zhao, Y.; Jiang, M.; Jiang, M. Cyanobacterial Blooms Increase Functional Diversity of Metazooplankton in a Shallow Eutrophic Lake. Water 2023, 15, 953. https://doi.org/10.3390/w15050953
Tian X, Yuan Y, Zou Y, Qin L, Zhu X, Zhu Y, Zhao Y, Jiang M, Jiang M. Cyanobacterial Blooms Increase Functional Diversity of Metazooplankton in a Shallow Eutrophic Lake. Water. 2023; 15(5):953. https://doi.org/10.3390/w15050953
Chicago/Turabian StyleTian, Xue, Yuxiang Yuan, Yuanchun Zou, Lei Qin, Xiaoyan Zhu, Yu Zhu, Yuxi Zhao, Mengyu Jiang, and Ming Jiang. 2023. "Cyanobacterial Blooms Increase Functional Diversity of Metazooplankton in a Shallow Eutrophic Lake" Water 15, no. 5: 953. https://doi.org/10.3390/w15050953
APA StyleTian, X., Yuan, Y., Zou, Y., Qin, L., Zhu, X., Zhu, Y., Zhao, Y., Jiang, M., & Jiang, M. (2023). Cyanobacterial Blooms Increase Functional Diversity of Metazooplankton in a Shallow Eutrophic Lake. Water, 15(5), 953. https://doi.org/10.3390/w15050953








