More about Persulfate-Assisted Ferrilanthanide-Mediated Photocatalysis of Textile Industry Dye Reactive Black 5: Surface Properties and Structural Assessment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Photocatalytic Experiments
2.3. Multiple-Reuse Experiments
2.4. Substrate Characterization, Photocatalyst Composition, and Morphology
2.4.1. UV-Vis and Fluorescence Spectroscopic Analyses
2.4.2. SEM/EDAX Analyses
2.4.3. FTIR Analyses
2.4.4. Raman Analyses
2.5. Other Analytical and Instrumental Procedures
3. Results and Discussion
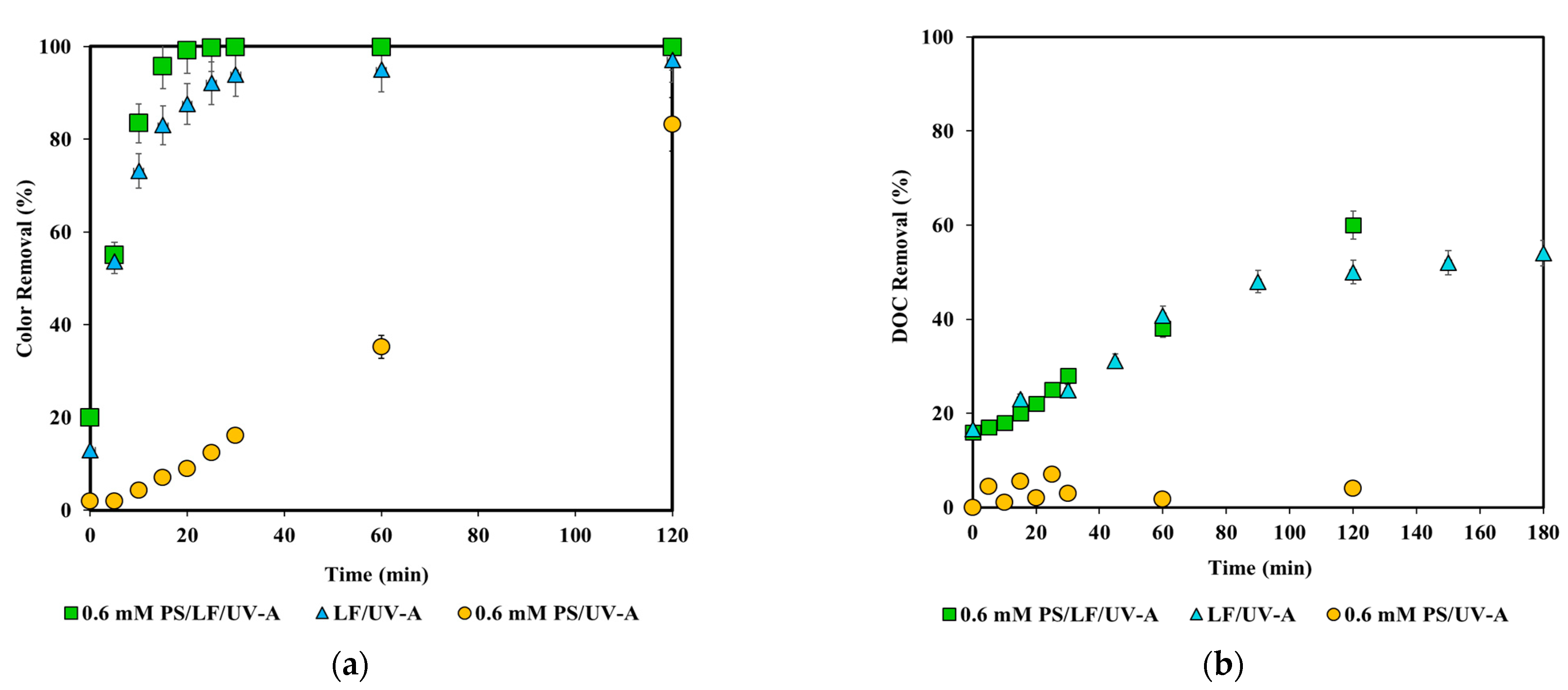
3.1. Control (Baseline) Experiments
3.2. Photocatalytic Treatment Experiments
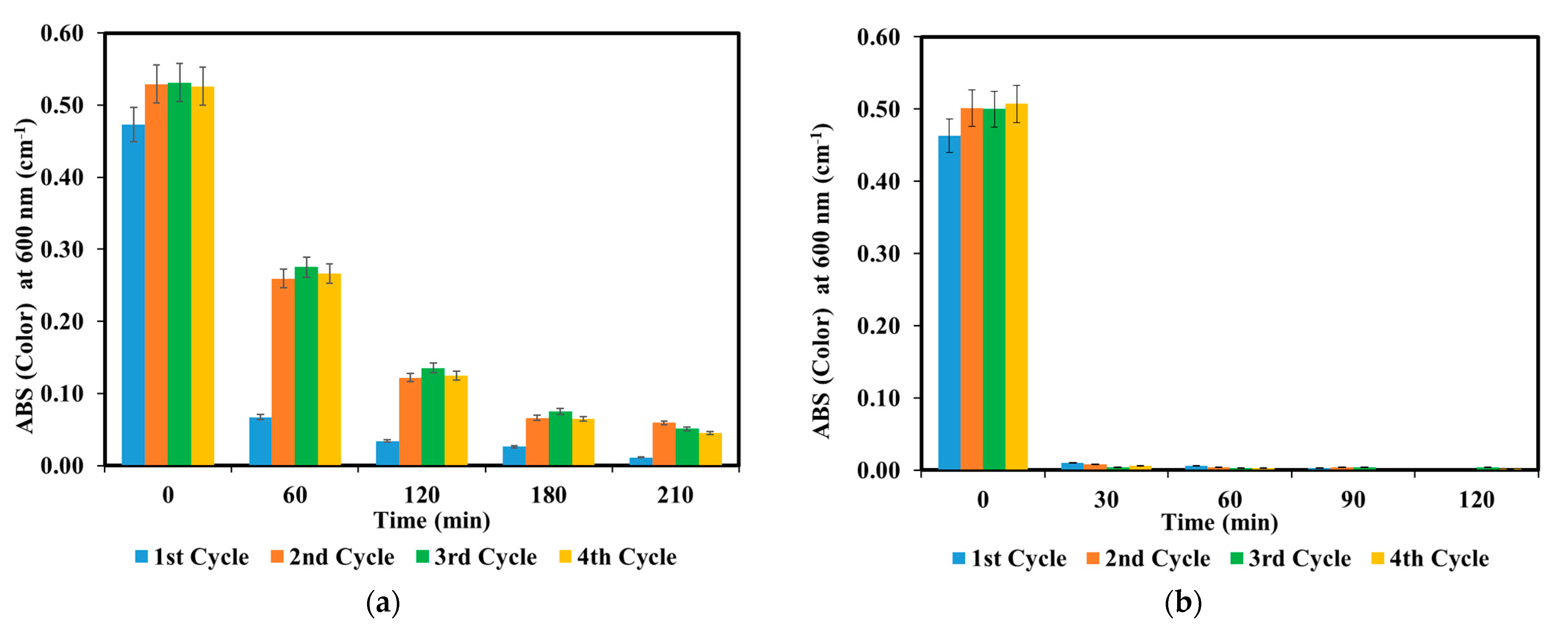
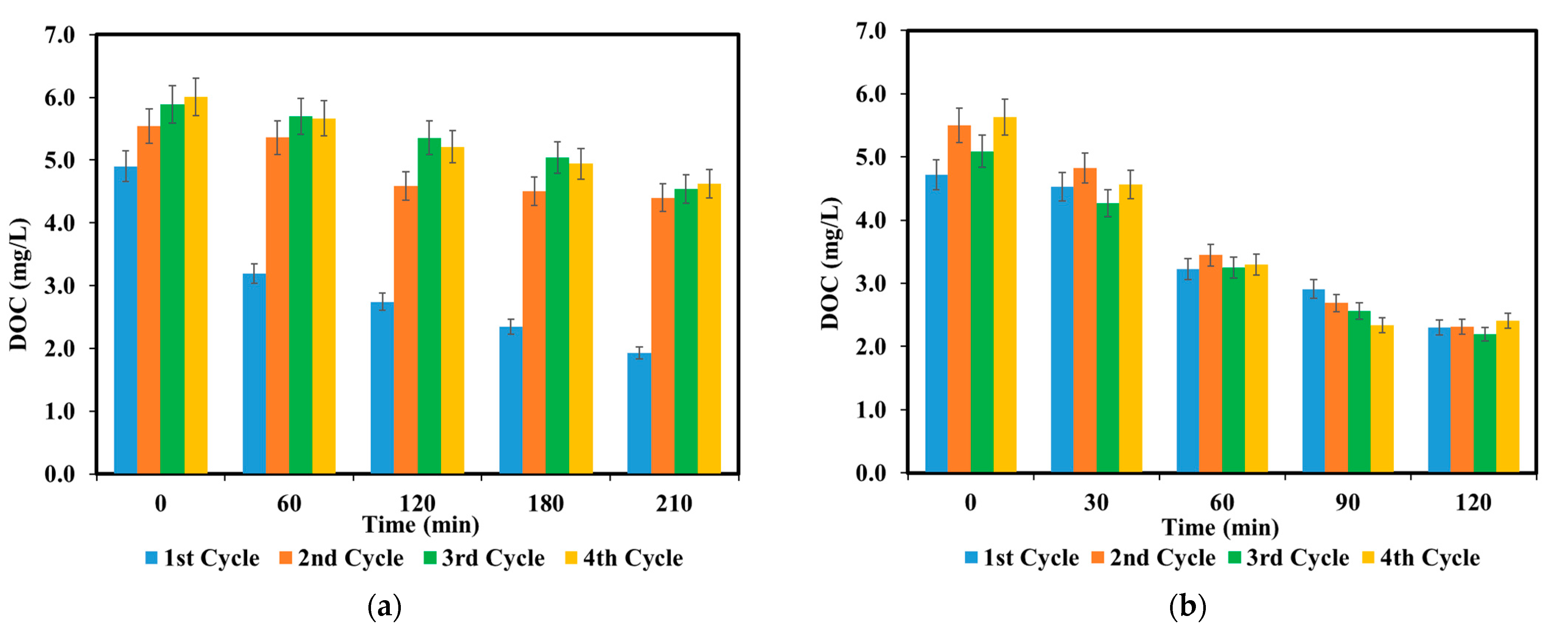
3.3. Photocatalyst Multiple-Reuse Experiments
3.4. Substrate Characterization, Photocatalyst Composition, and Morphology
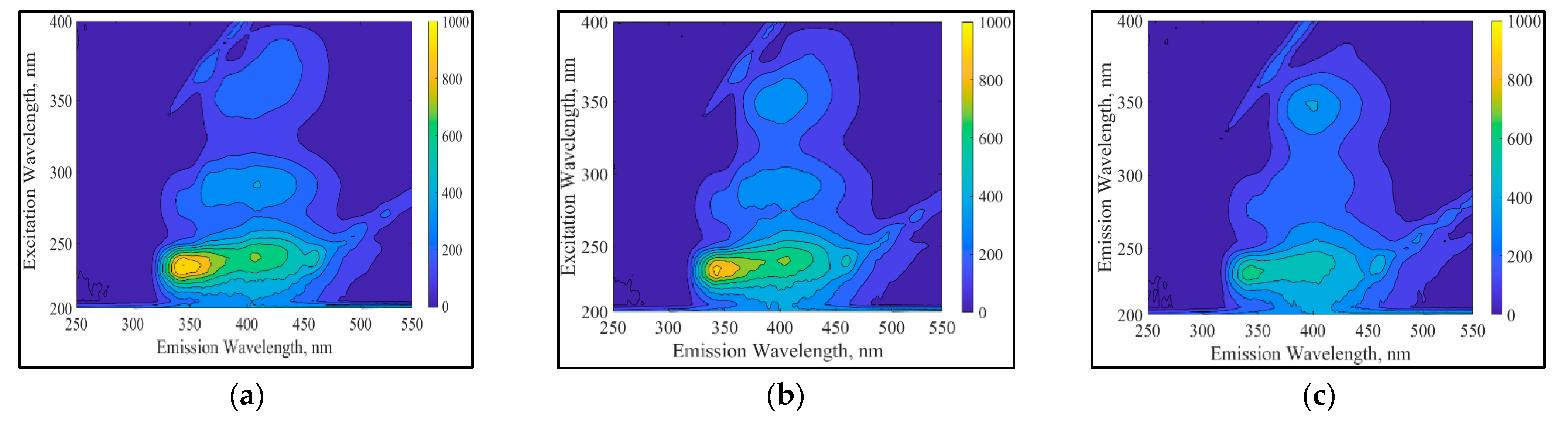
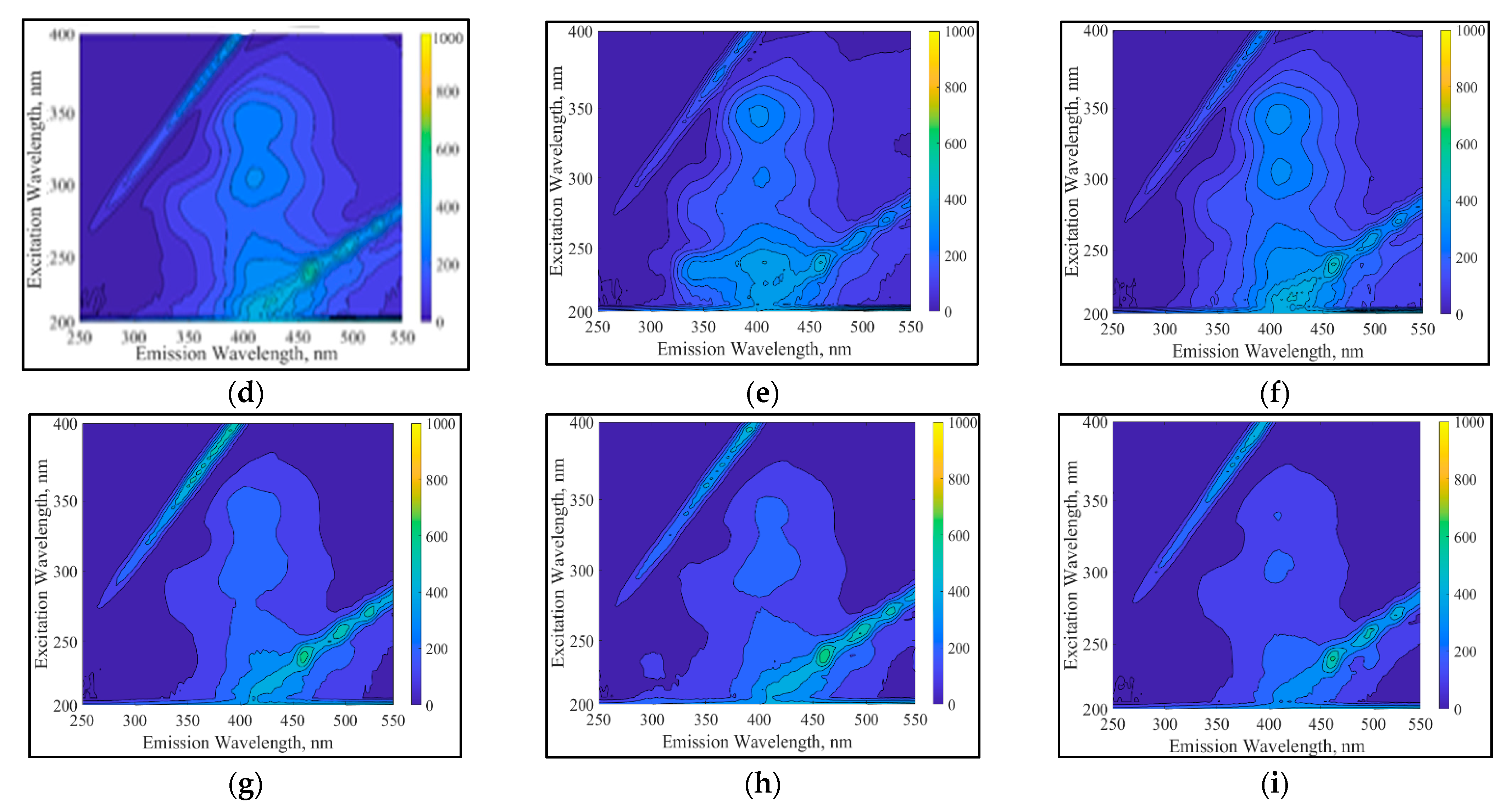
3.4.1. UV-Vis and Fluorescence Spectroscopic Analyses of RB5 Degradation
3.4.2. SEM/EDAX Analyses
3.4.3. FTIR Spectroscopy Analyses
3.4.4. Raman Spectroscopy Analyses
4. Conclusions and Recommendations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shore, J. Cellulosics Dyeing. In The Society of Dyers and Colorists; Alden Press: Oxford, UK, 1995. [Google Scholar]
- Freeman, H.S.; Reife, A. Dyes, Environmental Chemistry. In Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2003; pp. 431–463. [Google Scholar]
- Lucas, M.S.; Peres, J.A. Degradation of Reactive Black 5 by Fenton/UV-C and ferrioxalate/H2O2/solar light processes. Dyes Pigment. 2007, 7, 622–629. [Google Scholar]
- Bilinska, L.; Gmurek, M.; Ledakowicz, S. Comparison between industrial and simulated textile wastewater treatment by AOPs-biodegradability, toxicity and cost assessment. Chem. Eng. J. 2016, 306, 550–559. [Google Scholar]
- Armaković, S.J.; Savanović, M.M.; Armaković, S. Titanium dioxide as the most used photocatalyst for water purification: An overview. Catalysts 2023, 13, 26. [Google Scholar] [CrossRef]
- Chen, X.; Wang, W.; Xiao, H.; Hong, C.; Zhu, F.; Yao, Y.; Xue, Z. Accelerated TiO2 photocatalytic degradation of Acid Orange 7 under visible light mediated by peroxymonosulfate. Chem. Eng. J. 2012, 193–194, 290–295. [Google Scholar]
- Samy, M.; Kumi, A.G.; Salama, E.; ElKady, M.; Mensah, K.; Shokry, H. Heterogeneous activation of persulfate by a novel nano-magnetite/ZnO/activated carbon nanohybrid for carbofuran degradation: Toxicity assessment, water matrices, degradation mechanism and radical and non-radical pathways. Process Saf. Environ. Prot. 2023, 169, 337–351. [Google Scholar]
- Wang, X.; Chen, Z.; He, Y.; Yi, H.; Zhang, C.; Zhou, Q.; Xiang, X.; Gao, Y.; Huang, M. Activation of persulfate-based advanced oxidation processes by 1T-MoS2 for the degradation of imidacloprid: Performance and mechanism. Chem. Eng. J. 2023, 451, 138575. [Google Scholar] [CrossRef]
- Azouzi, W.; Sigle, W.; Labrim, H.; Benaissa, M. Sol-gel synthesis of nanoporous LaFeO3 powders for solar applications. Mater. Sci. Semicond. Process. 2019, 104, 104682. [Google Scholar] [CrossRef]
- Mokoen, M.; Koao, L.F.; Dejene, B.F.; Tshabalala, K.G.; Motloung, S.J. Structure, morphology, and optical properties of lanthanide orthovanadate powders prepared by chemical bath deposition. Mater. Today Commun. 2023, 34, 105001. [Google Scholar] [CrossRef]
- Zurlo, F.; Di Bartolomeo, E.; D’Epifanio, A.; Felice, V.; Sora, I.N.; Tortora, L.; Licoccia, S. La0.8Sr0.2Fe0.8Cu0.2O3−δ as “cobalt-free” cathode for La0.8Sr0.2Ga0.8Mg0.2O3−δ electrolyte. J. Power Source 2014, 271, 187–194. [Google Scholar] [CrossRef]
- Parrino, F.; García-López, E.; Marcì, G.; Palmisano, L.; Felice, V.; Sora, I.N.; Armelao, L. Cu-substituted lanthanum ferrite perovskites: Preparation, characterization and photocatalytic activity in gas-solid regime under simulated solar light irradiation. J. Alloys Compd. 2016, 682, 686–694. [Google Scholar] [CrossRef]
- Natali Sora, I.; Fumagalli, D. Fast photocatalytic degradation of pharmaceutical micropollutants and ecotoxicological effects. Environ. Sci. Pollut. Res. 2017, 24, 12556–12561. [Google Scholar]
- Birben, N.C.; Lale, E.; Pelosato, R.; Turkten, N.; Sora, I.N.; Bekbolet, M. Photocatalytic bactericidal performance of LaFeO3 under solar light in the presence of natural organic matter: Spectroscopic and mechanistic evaluation. Water 2021, 13, 2785. [Google Scholar] [CrossRef]
- Birben, N.C.; Lale, E.; Pelosato, R.; Uyguner Demirel, C.S.; Natali Sora, I.; Bekbolet, M. Photocatalytic bactericidal performance of LaFeO3 under solar light: Kinetics, spectroscopic and mechanistic evaluation. Water 2021, 13, 1135. [Google Scholar] [CrossRef]
- Turkten, N.; Natali Sora, I.; Tomruk, A.; Bekbolet, M. Photocatalytic degradation of humic acids using LaFeO3. Catalysts 2018, 8, 630. [Google Scholar] [CrossRef] [Green Version]
- Koba-Ucun, O.; Arslan-Alaton, I.; Sora, I.N.; Bekbölet, M. Persulfate-enhanced lanthanum iron oxide-mediated photocatalysis can effectively degrade an aqueous industrial dye and mineralize water and wastewater. Desalin. Water Treat. 2022, 267, 215–230. [Google Scholar] [CrossRef]
- Arslan-Alaton, I.; Akmehmet-Balcioglu, I. Photochemical and heterogeneous photocatalytic degradation of waste vinylsulphone dyes: A case study with hydrolyzed Reactive Black 5. J. Photochem. Photobiol. A Chem. 2001, 141, 247–254. [Google Scholar] [CrossRef]
- Arslan-Alaton, I.; Karatas, A.; Pehlivan, O.; Koba-Ucun, O.; Olmez-Hanci, T. Effect of UV-A-assisted iron-based and UV-C-driven oxidation processes on organic matter and antibiotic resistance removal in tertiary treated urban wastewater. Catal. Today 2020, 361, 152–158. [Google Scholar] [CrossRef]
- Wan, K.; Li, Y.; Wang, Y.; Wei, G. Recent advance in the fabrication of 2D and 3D metal carbides-based nanomaterials for energy and environmental applications. Nanomaterials 2021, 11, 246. [Google Scholar] [CrossRef]
- Wan, K.; Liu, C.; Wang, Y.; Zhang, W.; Qi, P.; Guo, L.; Wei, G.; Liu, X. Recent advances in the synthesis and regulation of 3D metal carbide-based hybrid architectures for water environmental remediation and monitoring. J. Environ. Chem. Eng. 2022, 10, 108994. [Google Scholar] [CrossRef]
- Wan, K.; Wang, Y.; Liu, C.; Wei, C.; Lv, S.; Tang, X.; Fang, T.; Zhao, J.; Wei, G.; Qi, P.; et al. Facile synthesis of hierarchical Ti3C2@FeOOH nanocomposites for antimony contaminated wastewater treatment: Performance, mechanisms, reutilization, and sustainability. Chem. Eng. J. 2022, 450, 138038. [Google Scholar] [CrossRef]
- Badvi, K.; Javanbakht, V. Enhanced photocatalytic degradation of dye contaminants with TiO2 immobilized on ZSM-5 zeolite modified with nickel nanoparticles. J. Clean. Prod. 2021, 280, 124518. [Google Scholar] [CrossRef]
- Chen, J.; Xiong, Y.; Duan, M.; Xiang, L.; Li, J.; Fang, S.; Qin, S.; Zhang, R. Insight into the Synergistic Effect of adsorption–photocatalysis for the removal of organic dye pollutants by Cr-Doped ZnO. Langmuir 2020, 36, 520–533. [Google Scholar] [CrossRef] [PubMed]
- Lau, Y.-Y.; Wong, Y.-S.; Ong, S.-A.; Ho, L.-N.; Hussin, K.; Lutpi, N.A. Intermolecular mechanistic treatment of recalcitrant environmental pollutants: Azo, benzene, naphthalene and vinyl sulfone. J. Taiwan Inst. Chem. Eng. 2017, 76, 27–34. [Google Scholar] [CrossRef]
- Saroyan, H.; Ntagiou, D.; Rekos, K.; Deliyanni, E. Reactive Black 5 degradation on manganese oxides supported on sodium hydroxide modified graphene oxide. Appl. Sci. 2019, 9, 2167. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Xie, X.; Liu, Y.; Zheng, X.; Wang, Y.; Cong, J.; Yu, C.; Liu, N.; He, Z.; Liu, J.; et al. Sugar sources as co-substrates promoting the degradation of refractory dye: A comparative study. Ecotoxicol. Environ. Saf. 2019, 184, 109613. [Google Scholar] [CrossRef]
- Chen, Y.; D’Errico, G.; Fabbricino, M.; Gallucci, N.; Pontoni, L.; Race, M.; Yao, S. Role of organic nanoparticles on transport and fate of various dyes in aqueous solution. Environ. Res. 2022, 215, 114179. [Google Scholar] [CrossRef]
- Uyguner-Demirel, C.S.; Turkten, N.; Kaya, D.; Bekbolet, M. Effect of oxidative and non-oxidative conditions on molecular size fractionation of humic acids: TiO2 and Cu-doped TiO2 photocatalysis. Environ. Sci. Pollut. Res. 2022, 29, 85413–85432. [Google Scholar] [CrossRef]
- Kale, R.D.; Kane, P.B. Synthesis of PVP stabilized bimetallic nanoparticles for removal of azo based reactive dye from aqueous solution. Sustain. Chem. Pharm. 2018, 10, 153–162. [Google Scholar] [CrossRef]
- Bilal, M.; Rasheed, T.; Iqbal, H.M.N.; Hu, H.; Wang, W.; Zhang, X. Toxicological assessment and UV/TiO2-based induced degradation profile of Reactive Black 5 dye. Environ. Manag. 2018, 61, 171–180. [Google Scholar] [CrossRef]
- Neoh, C.H.; Lam, C.Y.; Lim, C.K.; Yahya, A.; Bay, H.H.; Ibrahim, Z.; Noor, Z.Z. Biodecolorization of recalcitrant dye as the sole sourceof nutrition using Curvularia clavata NZ2 and decolorization ability of its crude enzymes. Environ. Sci. Pollut. Res. 2015, 22, 11669–11678. [Google Scholar] [CrossRef]
- Ziane, S.; Bessaha, F.; Marouf-Khelifa, K.; Khelifa, A. Single and binary adsorption of reactive black 5 and Congo red on modified dolomite: Performance and mechanism. J. Mol. Liq. 2018, 249, 1245–1253. [Google Scholar] [CrossRef]
- Gosavi, P.V.; Biniwale, R.B. Pure phase LaFeO3 perovskite with improved surface area synthesized using different routes and its characterization. Mater. Chem. Phys. 2010, 119, 324–329. [Google Scholar] [CrossRef]
- Li, S.; Jing, L.; Fu, W.; Yang, L.; Xin, B.; Fu, H. Photoinduced charge property of nanosized perovskite-type LaFeO3 and its relationships with photocatalytic activity under visible irradiation. Mater. Res. Bull. 2007, 42, 203–212. [Google Scholar] [CrossRef]
- Thirumalairajan, S.; Girija, K.; Ganesh, I.; Mangalaraj, D.; Viswanathan, C.; Balamurugan, A.; Ponpandian, N. Controlled synthesis of perovskite LaFeO3 microsphere composed of nanoparticles via self-assembly process and their associated photocatalytic activity. Chem. Eng. J. 2012, 209, 420–428. [Google Scholar] [CrossRef]
- Yahya, N.; Aziz, F.; Jaafar, J.; Lau, W.J.; Yusof, N.; Salleh, W.N.W.; Ismail, A.F.; Aziz, M. Impacts of annealing temperature on morphological, optical and photocatalytic properties of gel-combustion-derived LaFeO3 nanoparticles. Arab. J. Sci. Eng. 2021, 46, 6153–6165. [Google Scholar] [CrossRef]
- Abazari, R.; Sanati, S.; Saghatforoush, L.A. A unique and facile preparation of lanthanum ferrite nanoparticles in emulsion nanoreactors: Morphology, structure, and efficient photocatalysis. Mater. Sci. Semicond. Process. 2014, 25, 301–306. [Google Scholar] [CrossRef]
- Abdallah, F.B.; Benali, A.; Triki, M.; Dhahri, E.; Nomenyo, K.; Lerondel, G. Investigation of structural, morphological, optical and electrical properties of double-doping Lanthanum ferrite. J. Mater. Sci. Mater. Electron. 2019, 30, 349–3358. [Google Scholar] [CrossRef]
- Kale, R.D.; Kane, P.B. Decolourization by PVP stabilized Fe-Ni nanoparticles of Reactive Black 5 dye. J. Environ. Chem. Eng. 2018, 6, 5961–5969. [Google Scholar] [CrossRef]
- Wawrzyniak, B.; Morawski, A.W. Solar-light-induced photocatalytic decomposition of two azo dyes on new TiO2 photocatalyst containing nitrogen. Appl. Catal. 2006, 62, 150–158. [Google Scholar] [CrossRef]
- Santos, P.B.; Dos Santos, H.F.; Andrade, G.F. Photodegradation mechanism of the RB5 dye: A theoretical and spectroscopic study. J. Photochem. Photobiol. A Chem. 2021, 416, 113315. [Google Scholar] [CrossRef]
- Turkten, N.; Cinar, Z. Photocatalytic decolorization of azo dyes on TiO2: Prediction of mechanism via conceptual DFT. Catal. Today 2017, 287, 169–175. [Google Scholar]
- Lin-Vien, D.; Colthup, N.B.; Fateley, W.G.; Grasselli, J.G. The Handbook of Infrared and Raman Characteristic Frequencies of Organic Molecules; Academic Press: San Diego, CA, USA, 1991. [Google Scholar]
- Aguayo, T.; Garrido, C.; Clavijo, R.E.; Gómez-Jeria, J.S.; Araya Monasterio, C.; Icaza, M.; Espinoza Moraga, F.; Campos Vallette, M.M. Raman and surface enhanced Raman scattering of a black dyed silk. J. Raman Spectrosc. 2013, 44, 1238–1245. [Google Scholar] [CrossRef]
- Santos, P.B.; Santos, J.J.; Corrêa, C.C.; Corio, P.; Andrade, G.F.S. Plasmonic photodegradation of textile dye Reactive Black 5 under visible light: A vibrational and electronic study. J. Photochem. Photobiol. A Chem. 2019, 371, 159–165. [Google Scholar] [CrossRef]
- Popa, M.; Frantti, J.; Kakihana, M. Lanthanum ferrite LaFeO3+d nanopowders obtained by the polymerizable complex method. Solid State Ion. 2002, 154–155, 437–445. [Google Scholar] [CrossRef]
- Sierra Gallego, G.; Marín Alzate, N.; Arnache, O. A novel LaFeO3−XNX oxynitride. Synthesis and characterization. J. Alloys Compd. 2013, 549, 163–169. [Google Scholar] [CrossRef]
- Sharma, N.; Sharma, S.K.; Sachdev, K. Effect of precursors on the morphology and surface area of LaFeO3. Ceram. Int. 2019, 45, 7217–7225. [Google Scholar] [CrossRef]
- Lee, W.-Y.; Yun, H.J.; Yoon, J.-W. Characterization and magnetic properties of LaFeO3 nanofibers synthesized by electrospinning. J. Alloys Compd. 2014, 583, 320–324. [Google Scholar] [CrossRef]
- Mohammadi, M.; Sabbaghi, S.; Sadeghi, H.; Zerafat, M.M.; Pooladi, R. Preparation and characterization of TiO2/ZnO/CuO nanocomposite and application for phenol removal from wastewaters. Desalin. Water Treat. 2016, 57, 799–809. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arslan-Alaton, I.; Koba-Ucun, O.; Turkten, N.; Sora, I.N.; Bekbolet, M. More about Persulfate-Assisted Ferrilanthanide-Mediated Photocatalysis of Textile Industry Dye Reactive Black 5: Surface Properties and Structural Assessment. Water 2023, 15, 906. https://doi.org/10.3390/w15050906
Arslan-Alaton I, Koba-Ucun O, Turkten N, Sora IN, Bekbolet M. More about Persulfate-Assisted Ferrilanthanide-Mediated Photocatalysis of Textile Industry Dye Reactive Black 5: Surface Properties and Structural Assessment. Water. 2023; 15(5):906. https://doi.org/10.3390/w15050906
Chicago/Turabian StyleArslan-Alaton, Idil, Olga Koba-Ucun, Nazli Turkten, Isabella Natali Sora, and Miray Bekbolet. 2023. "More about Persulfate-Assisted Ferrilanthanide-Mediated Photocatalysis of Textile Industry Dye Reactive Black 5: Surface Properties and Structural Assessment" Water 15, no. 5: 906. https://doi.org/10.3390/w15050906
APA StyleArslan-Alaton, I., Koba-Ucun, O., Turkten, N., Sora, I. N., & Bekbolet, M. (2023). More about Persulfate-Assisted Ferrilanthanide-Mediated Photocatalysis of Textile Industry Dye Reactive Black 5: Surface Properties and Structural Assessment. Water, 15(5), 906. https://doi.org/10.3390/w15050906







