Freshwater Diversity of Zooplankton from Mexico: Historical Review of Some of the Main Groups †
Abstract
:1. Introduction
2. Methods
3. Results
3.1. Phase 1: Early Studies Dominated by Foreign Researchers (Second Half of XIX Century to 1940s of XX Century)
3.1.1. Protists
3.1.2. Rotifers
3.1.3. Water Mites
3.1.4. Cladocerans
3.1.5. Large Branchiopods
3.1.6. Copepods
3.2. Phase 2: Towards the Formation of Mexican Limnologists (1950s to the End of XX Century)
3.2.1. Protists
3.2.2. Rotifers
3.2.3. Water Mites
3.2.4. Cladocerans
3.2.5. Large Branchiopods
3.2.6. Copepods
3.3. Phase 3: The Mature Phase of Zooplankton Studies (21st Century)
3.3.1. Protists
3.3.2. Rotifers
3.3.3. Water Mites
3.3.4. Cladocerans
3.3.5. Large Branchiopods
3.3.6. Copepods
3.4. Exotic/Invasive Zooplankton Species in Mexico
3.5. Conservation of the Zooplankton
4. Final Remarks and Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Haeckel, E. Generelle Morphologie der Organismen. Allgemeine Grundzüge der Organischen Formen-Wissenschaft, Mechanisch Begründet Durch Die von Charles Darwin Reformierte Descendenztheorie; De Gruyter: Berlin, Germany, 1866; Volume Bd. 1 Und 2, Available online: https://www.biodiversitylibrary.org/bibliography/3953 (accessed on 22 November 2022).
- Adl, S.M.; Bass, D.; Lane, C.E.; Lukes, J.; Schoch, C.L.; Smirnov, A.; Agatha, S.; Berney, C.; Brown, M.W.; Burki, F.; et al. Revisions to the Classification, Nomenclature, and Diversity of Eukaryotes. J. Eukaryot. Microbiol. 2019, 66, 4–119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azam, F.; Fenchel, T.; Field, J.G.; Gray, J.S.; Meyerreil, L.A.; Thingstad, F. The Ecological Role of Water-Column Microbes in the Sea. Mar. Ecol. Prog. Ser. 1983, 10, 257–263. [Google Scholar] [CrossRef]
- Chao, A.; Li, P.C.; Agatha, S.; Foissner, W. A Statistical Approach to Estimate Soil Ciliate Diversity and Distribution Based on Data from Five Continents. Oikos 2006, 114, 479–493. [Google Scholar] [CrossRef]
- Singer, D.; Seppey, C.V.W.; Lentendu, G.; Dunthorn, M.; Bass, D.; Belbahri, L.; Blandenier, Q.; Debroas, D.; de Groot, G.A.; de Vargas, C.; et al. Protist Taxonomic and Functional Diversity in Soil, Freshwater and Marine Ecosystems. Environ. Int. 2021, 146, 8. [Google Scholar] [CrossRef]
- Weisse, T. Functional Diversity of Aquatic Ciliates. Eur. J. Protistol. 2017, 61, 331–358. [Google Scholar] [CrossRef]
- Weisse, T.; Montagnes, D.J.S. Ecology of Planktonic Ciliates in a Changing World: Concepts, Methods, and Challenges. J. Eukaryot. Microbiol. 2022, 69, el2879. [Google Scholar] [CrossRef]
- Proctor, H.C.; Smith, I.M.; Cook, D.R.; Smith, B.P. Chapter 25—Subphylum Chelicerata, Class Arachnida. In Thorp and Covich’s Freshwater Invertebrates, 4th ed.; Thorp, J.H., Christopher Rogers, D., Eds.; Academic Press: Boston, MA, USA, 2015; pp. 599–660. [Google Scholar]
- Di Sabatino, A.; Smit, H.; Gerecke, R.; Goldschmidt, T.; Matsumoto, N.; Cicolani, B. Global Diversity of Water Mites (Acari, Hydrachnidia; Arachnida) in Freshwater. Hydrobiologia 2008, 59, 303–315. [Google Scholar] [CrossRef]
- Butler, M.I.; Burns, C.W. Prey Selectivity of Piona exigua, a Planktonic Water Mite. Oecologia 1991, 86, 210–222. [Google Scholar] [CrossRef]
- Modlin, R.F.; Gannon, J.E. Contribution to Ecology and Distribution of Aquatic Acari in St-Lawrence-Great-Lakes. Trans. Am. Microsc. Soc. 1973, 92, 217–224. [Google Scholar] [CrossRef]
- Montes-Ortiz, L.; Goldschmidt, T.; Elías-Gutiérrez, M. First Evidence of Parasitation of a Bosmina (Cladocera) by a Water Mite Larva in a Karst Sinkhole, in Quintana Roo (Yucatan Peninsula, Mexico). Acarologia 2019, 59, 111–114. [Google Scholar] [CrossRef]
- Riessen, H.P. Predatory Behavior and Prey Selectivity of the Pelagic Water Mite Piona constricta. Can. J. Fish. Aquat. Sci. 1982, 39, 1569–1579. [Google Scholar] [CrossRef]
- Cassano, C.R.; Castilho-Noll, M.S.M.; Arcifa, M. Water Mite Predation on Zooplankton of a Tropical Lake. Braz. J. Biol. 2002, 62, 565–571. [Google Scholar] [CrossRef] [Green Version]
- Matveev, V.F.; Martínez, C.C. Can Water Mites Control Populations of Planktonic Cladocera. Hydrobiologia 1990, 198, 227–231. [Google Scholar] [CrossRef]
- Proctor, H.C.; Smith, I.M.; Cook, D.R.; Smith, B.P. Subphylum Chelicerata, Class Arachnida. In Ecology and General Biology, Vol I: Thorp and Covich’s Freshwater Invertebrates, 4th ed.; Thorp, J.H., Christopher Rogers, D., Eds.; Academic Press Ltd.: London, UK; Elsevier Science Ltd.: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Smit, H. Water Mites of the World, with Keys to the Families, Subfamilies, Genera and Subgenera (Acari:Hydrachnidia). In Nederlandse Entomologische Vereniging; Nederlandse Entomologische Vereniging: Leiden, The Netherlands, 2020. [Google Scholar]
- Elías-Gutiérrez, M.; Suárez-Morales, E.; Gutiérrez-Aguirre, M.; Silva-Briano, M.; Granados-Ramirez, J.; Garfias-Espejo, T. Guía Ilustrada De Los Microcrustáceos (Cladocera Y Copepoda) De Las Aguas Continentales De México; Universidad Nacional Autónoma de México: Ciudad de México, México, 2008. [Google Scholar]
- Kotov, A.A.; Korovchinsky, N.; Petrusek, A. World Checklist of Freshwater Cladocera Species. Available online: https://web.archive.org/web/20180320182811/http%3A//fada.biodiversity.be/group/show/17 (accessed on 22 November 2022).
- Elías-Gutiérrez, M.; Martínez-Jerónimo, F.; Ivanova, N.; Valdez-Moreno, M. DNA Barcodes for Cladocera and Copepoda from Mexico and Guatemala, Highlights and New Discoveries. Zootaxa 2008, 1849, 1–42. [Google Scholar] [CrossRef] [Green Version]
- Maeda-Martínez, A.; Obregón-Barboza, H.; García-Velazco, H.; Prieto-Salazar, M. Branchiopoda Anostraca. In Biodiversidad, Taxonomía Y Biogeografía De Artrópodos De México; Llorente-Bousquet, J., Morrone, J.J., Eds.; Universidad Nacional Autónoma de México: Ciudad de México, México, 2002; pp. 305–322. [Google Scholar]
- Maeda-Martínez, A.; Obregón-Barboza, H.; García-Velazco, H. Branchiopoda: Notostraca. In Biodiversidad, Taxonomía Y Biogeografía De Artrópodos De México; Llorente-Bousquet, J., Morrone, J.J., Eds.; Universidad Nacional Autónoma de México: Ciudad de México, México, 2002; pp. 333–339. [Google Scholar]
- Llorente-Bousquet, J.; Morrone, J.J. Branchiopoda: Cyclestherida, Laevicaudata, and Spinicaudata. In Biodiversidad, Taxonomía Y Biogeografía De Artrópodos De México; Llorente-Bousquet, J., Morrone, J.J., Eds.; Universidad Nacional Autónoma de México: Ciudad de México, México, 2002; pp. 323–331. [Google Scholar]
- Gomez, S.; Morales-Serna, F.N. Updated Checklist of Published and Unpublished Records of Harpacticoid Copepods (Crustacea: Copepoda: Harpacticoida) from Mexico. Proc. Biol. Soc. Wash. 2014, 127, 99–121. [Google Scholar] [CrossRef]
- Gutiérrez-Aguirre, M.; Suárez-Morales, E. The Freshwater Centropagid Osphranticum Labronectum Forbes, 1882 (Crustacea: Copepoda: Calanoida) in Mexico with Description of a New Subspecies. Proc. Biol. Soc. Washigton 1999, 112, 687–694. [Google Scholar]
- Suarez-Morales, E.; Rodriguez-Almaraz, G.; Gutiérrez-Aguirre, M.A.; Walsh, E. The Coastal-Estuarine Copepod, Eurytemora affinis (Poppe) (Calanoida, Temoridae), from Arid Inland Areas of Mexico: An Expected Occurrence? Crustaceana 2008, 81, 679–694. [Google Scholar]
- Suárez-Morales, E.; Reid, J.W. An Updated List of the Free Living Freshwater Copepods (Crustacea) of Mexico. Southwest. Nat. 1998, 43, 256–265. [Google Scholar]
- Suárez-Morales, E.; Reid, J.W.; Elías-Gutiérrez, M. Diversity and Distributional Patterns of Neotropical Freshwater Copepods (Calanoida: Diaptomidae). Int. Rev. Hydrobiol. 2005, 90, 71–83. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Gutiérrez-Aguirre, M.A.; Gómez, S.; Perbiche-Neves, G.; Previattelli, D.; dos Santos-Silva, E.N.; da Rocha, C.E.; Mercado-Salas, N.F.; Marques, T.M.; Cruz-Quintana, Y.; et al. Class Copepoda. In Thorp and Covich’s Freshwater Invertebrates; Damborenea, C., Rogers, D.C., Thorp, H., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 665–790. [Google Scholar]
- Perbiche-Neves, G.; Previattelli, D.; Pie, M.R.; Duran, A.; Suárez-Morales, E.; Boxshall, G.A.; Nogueira, M.G.; da Rocha, C.E. Historical Biogeography of the Neotropical Diaptomidae (Crustacea: Copepoda). Front. Zool. 2014, 11, 36. [Google Scholar] [CrossRef] [Green Version]
- Gutiérrez-Aguirre, M.A.; Cervantes-Martínez, A.; Elías-Gutiérrez, M.; Lugo-Vazquez, A. Remarks on Mastigodiaptomus (Calanoida: Diaptomidae) from Mexico Using Integrative Taxonomy, with a Key of Identification and Three New Species. Peerj 2020, 8, e8416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suárez-Morales, E.; Reid, J.W. An Updated Checklist of the Continental Copepod Fauna of the Yucatan Peninsula, Mexico, with Notes on Its Regional Associations. Crustaceana 2003, 76, 977–991. [Google Scholar] [CrossRef]
- Pearse, A.S.; Creaser, E.P.; Hall, F.G. The Cenotes of Yucatan. A Zoological and Hydrographic Survey; Carnegie Institution of Washington: Washington, DC, USA, 1936. [Google Scholar]
- Bravo-Hollis, H. Contribuciones Al Conocimiento De Los Protozoarios Mexicanos. VI. Gastrostyla steinii Engelmann. Órgano De La Soc. Mex. De Biol. 1924, 4, 165–169. [Google Scholar]
- López-Ochoterena, E. La Protozoología Dentro De La Biología Actual. Rev. de la Soc. Mex. de Hist. Nat. 1991, 42, 267–274. [Google Scholar]
- Sámano, B.A.; Sokoloff, D. La Flora Y Fauna De Aguas Dulces Del Valle De México. Monogr. Inst. Biol. México 1931, 1, 5–49. [Google Scholar]
- Osorio Tafall, B.F. Biodinámica Del Lago De Pátzcuaro. I. Ensayo De Interpretación De Sus Relaciones Tróficas. Rev. Soc. Mex. Hist. Nat. 1944, 5, 197–227. [Google Scholar]
- Osorio-Tafall, B.F. Tintinnidos Nuevos O Poco Conocidos Del Plancton Nerítico De México. Rev. Soc. Mex. Hist. Nat. 1941, 2, 147–174. [Google Scholar]
- López-Ochoterena, E. Historia De Las Investigaciones Sobre Protozoarios De Vida Libre De México. Rev. Soc. Mex. Hist. Nat. 1970, 31, 1–15. [Google Scholar]
- Sámano, B.A. Contribución Al Conocimiento De Los Rotíferos De México. An. Del Inst. De Biol. 1931, 2, 157–163. [Google Scholar]
- Ahlstrom, E.H. Plankton Rotatoria from Mexico. Trans. Am. Microsc. Soc. 1932, 51, 242–251. [Google Scholar] [CrossRef]
- Carlin-Nilson, B. Rotatorien Aus México. Kungl. Fysiogr. Sällskapets I Lund Förhadlingar 1935, 5, 175–185. [Google Scholar]
- Sámano, B.A. Contribuciones Al Conocimiento De La Fauna De Actopan, Hgo. Iii. Nota Acerca De La Fauna De Rotíferos De Los Depósitos De Agua De Actopan Y Lugares Vecinos. An. Del Inst. De Biol. 1936, 7, 269–270. [Google Scholar]
- Hoffman, C.C.; Sámano, B.A. Nota Sobre Los Criaderos Invernales De Anopheles albimanus Wide En Los Pantanos De Veracruz. An. Del Inst. De Biol. 1938, 9, 193–208. [Google Scholar]
- Hoffmann, C.C.; Samano, A.B. Los Criaderos Invernales De Anopheles pseudopunctipennis En El Estado De Oaxaca. An. Del Inst. De Biol. 1938, 9, 181–192. [Google Scholar]
- Uéno, M. Zooplankton of Lago De Patzcuaro, Mexico. Annot. Zool. Jpn. 1939, 18, 105–114. [Google Scholar]
- Ahlstrom, E.H. A Revision of the Rotatorian Genera Brachionus and Platyas with Descriptions of One New Species and Two New Varieties. Bull. Am. Mus. Nat. Hist. 1940, 77, 143–184. [Google Scholar]
- Rioja, E. Observaciones Acerca Del Plancton Del Lago De Pátzcuaro. An. Del Inst. De Biol. 1940, 11, 417–425. [Google Scholar]
- Rioja, E. Estudios Hidrobiológicos Viii. Observaciones Acerca Del Plancton De La Laguna San Felipe Xochiltepec (Puebla). An. Del Inst. De Biol. 1942, 13, 519–526. [Google Scholar]
- Osorio-Tafall, B.F. Rotíferos Planctónicos De México I, Ii, Iii. Rev. Soc. Mex. Hist. Nat. 1942, 1–4, 23–79. [Google Scholar]
- Osorio Tafall, B.F. Observaciones Sobre La Fauna Acuática De Las Cuevas De La Región De Valles, San Luis Potosí (México). Rev. Soc. Mex. Hist. Nat. 1943, 4, 43–71. [Google Scholar]
- Dugès, A. Notes Zoologiques. Descriptions De Rhyncholophus albovittatus, N. Sp. Y Et De Antonides guanajuatensis, N. Sp. Notesur Pityophis Deppei, D. Et Bib., Et Sur Le Larynx De 1’ortalida Mac Calli. Rev. Des Sci. Nat. 1874, 2, 321–324. [Google Scholar]
- Dugès, A. Atax alzatei. Periódico Científico De La Soc. Méxicana De Hist. Nat. 1884, 6, 344–347. [Google Scholar]
- Marshall, R. The Arrhenuri of the United States. Trans. Am. Microsc. Soc. 1908, 28, 85–140. [Google Scholar] [CrossRef]
- Marshall, R. Hydracarina from Yucatan. In The Cenotes of Yucatan; a Zoological and Hydrographic Survey; Washington Carnegie Institution of Washington: Washington, DC, USA, 1936; pp. 133–137. Available online: https://catalog.libraries.wm.edu/discovery/fulldisplay?vid=01COWM_INST:01COWM_WM_NEWUI&mode=advanced&tab=LibraryCatalog&docid=alma991025734399703196&query=sub,exact,General%20Motors%20Corporation,AND&context=L&lang=en (accessed on 22 November 2022).
- Juday, C. Limnological Studies on Some Lakes in Central America. Trans. Wis. Acad. Sci. Arts Lett. 1915, 18, 214–250. [Google Scholar]
- Brehm, V. Notizen Zur Svwasserfauna Guatemalas Und Mexikos. Zool. Anz. 1932, 98, 63–66. [Google Scholar]
- Osorio Tafall, B.F. Prospecto Biológico Del Lago De Pátzcuaro. Rev. De La Soc. Mex. De Hist. Nat. 1942, 3, 435–513. [Google Scholar]
- Packard, A.S. Preliminary Notice of New North American Phyllopoda. Am. J. Sci. Arts 1871, 2, 108–113. [Google Scholar] [CrossRef] [Green Version]
- Richard, M. Sur Les Crustacés Phyllopodes Recueillis Par M. Diguet Dans La Basse-Californie. Bull. Du Muséum D’Histoire Nat. 1895, 1, 107–108. [Google Scholar]
- Daday, E. Monographie Systématique Des Phyllopodes Conchostracés. I. Ann. Des Sci. Nat. Zool. 9e Série 1914, 20, 39–330. [Google Scholar]
- Daday, E. Monographie Systématique Des Phyllopodes Conchostracés. I. Ann. Des Sci. Nat. Zooloogie 10e Série 1923, 6, 255–386. [Google Scholar]
- Linder, F. Contributions to the Morphology and the Taxonomy of the Branchiopoda Anostraca. Zool. Bidr. Fran Upps. 1941, 20, 101–302. [Google Scholar]
- Pearse, A.S. A New Species of Diaptomus from Mexico. Am. Nat. 1904, 38, 889–891. [Google Scholar] [CrossRef]
- Marsh, C.D. A Revision on the North American Species of Diaptomidae. Trans. Wis. Acad. Sci. Arts Lett. 1907, 15, 381–516. [Google Scholar]
- Marsh, C.D. Distribution and Key of the North American Copepods of the Genus Diaptomus, with the Description of a New Species. Proc. United States Natl. Mus. 1929, 75, 1–27. [Google Scholar] [CrossRef]
- Grimaldo-Ortega, D.; Elías-Gutiérrez, M.; Camacho-Lemus, M. Additions to Mexican Freshwater Copepods with the Description of the Female Leptodiaptomus mexicanus (Marsh). J. Mar. Syst. 1998, 15, 381–390. [Google Scholar] [CrossRef]
- Kiefer, F. Ruderfubkrebse (Crust. Cop.) Aus Mexico. Zool. Anz. 1938, 115, 274–279. [Google Scholar]
- Wilson, C.B. Copepods from the Cenotes and Caves of the Yucatan Peninsula, with Notes on Cladocerans. In The Cenotes of Yucatan. A Zoological and Hydrographic Survey; Pearse, A.S., Creaser, E.P., Hall, F.G., Eds.; Carnegie Institution of Washington: Washington, DC, USA, 1936; pp. 77–88. [Google Scholar]
- Pearse, A.S.; Wilson, C.B. Copepoda from Yucatan Caves. Carnegie Inst. Wash. Publ. 1938, 491, 153–154. [Google Scholar]
- Montiel-Martínez, A.; Ciros-Pérez, J.; Ortega-Mayagoitia, E.; Elías-Gutiérrez, M. Morphological, Ecological, Reproductive and Molecular Evidence for Leptodiaptomus garciai (Osorio-Tafall 1942) as a Valid Endemic Species. J. Plankton Res. 2008, 30, 1079–1093. [Google Scholar] [CrossRef] [Green Version]
- Elías-Gutiérrez, M.; Suárez-Morales, E. Redescription of Microdiaptomus cokeri (Crustacea: Copepoda: Diaptomidae) from Caves in Central Mexico, with the Description of a New Diaptomid Subfamily. Proc. Biol. Soc. Wash. 1998, 111, 199–208. [Google Scholar]
- Suárez-Morales, E.; Silva-Briano, M.; Elías-Gutiérrez, M. Redescription and Taxonomic Validity of Leptodiaptomus cuauhtemoci Osorio-Tafall, 1941 (Copepoda, Calanoida), with Notes on Its Known Distribution. J. Limnol. 2000, 59, 5–14. [Google Scholar] [CrossRef]
- Mayen-Estrada, R.; Olvera-Bautista, F.; Reyes-Santos, M.; Duran-Ramirez, C.A.; Medina-Duran, J.H. Tintinnids (Ciliophora: Tintinnida) from Mexico: A Checklist. Biologia 2020, 75, 969–987. [Google Scholar] [CrossRef]
- Aladro-Lubel, M.A.; Mayén-Estrada, R.; Reyes-Santos, M. Registro Actualizado De Ciliados (Agosto, 2004); Listados Faunísticos de México. XI. Inst. Biol. Universidad Nacional Autónoma de México: Ciudad de México, México, 2006. [Google Scholar]
- Aladro-Lubel, M.A.; Resyes-Santos, M.; Olvera-Bautista, F.; Briones, M.R. Ciliados Y Otros Protozoos. In Guía Ilustrada De La Cantera Oriente: Caracterización Ambiental E Inventario Biológico; Lot, A., Ed.; Universidad Nacional Autónoma de México: Ciudad de México, México, 2007; pp. 97–122. [Google Scholar]
- López-Ochoterena, E. Ciliados Mesosapróbicos De Chapultepec (Sistemática, Morfología, Ecología). Rev. Soc. Mex. Hist. Nat. 1966, 31, 1–15. [Google Scholar]
- Pérez-Reyes, R.; Salas-Gómez, E. Protozoarios Encontrados En Colecciones De Agua Del Valle De México. An. Esc. Nac. Cienc. Biológicas 1961, 10, 39–44. [Google Scholar]
- Lugo-Vázquez, A. Estudio de las Comunidades Litorales de Protozoarios En Seis Lagos Cráter Del Estado De Puebla Mediante El Método De Colonizacion De Sustratos Artificiales. Master’s Thesis, Universidad Nacional Autónoma de México, Tlalnepantla, México, 1993. [Google Scholar]
- Suárez-Morales, E.; Segura-Puertas, L.; Fernández-Alamo, M.A. Diversidad Y Abundancia Del Plancton En La Laguna De Catemaco, Veracruz Durante Un Ciclo Anual. An. Del Inst. De Cienc. Del Mar Y Limnol. 1986, 13, 313–316. [Google Scholar]
- Suárez-Morales, E.; Vázquez, A.; Solis, M.E. Variaciones Espacio-Temporales De Distribución Y Abundancia De Rotíferos Planctónicos En La Presa J. A. Alzate, México, Durante Un Ciclo Anual. An. Del Inst. De Cienc. Del Mar Y Limnol. 1991, 18, 217–227. [Google Scholar]
- Vilaclara, G.; Sladecek, B. Mexican Rotifers as Indicators of Water Quality with Description of Collotheca riverai, n. sp. Arch. Fur Hydrobiol. 1989, 115, 257–263. [Google Scholar] [CrossRef]
- Silva-Briano, M.; Segers, H. Una Nueva Especie Del Género Brachionus (Rotifera, Monogononta) Del Estado De Aguascalientes México. Rev. D’ Hydrobiol. Trop. 1992, 25, 283–285. [Google Scholar]
- Flores, F.J.; Martínez, J. Comparative Limnology of the Three Reservoirs on the Mexican Altiplano (a Transition Zone), Aguascalientes, Mexico. Trop. Freshw. Biol. 1993, 3, 319–329. [Google Scholar]
- Rico, M.R.; Silva-Briano, M. Contribution to the Knowledge of the Rotifera of Mexico. Hydrobiologia 1993, 255, 467–474. [Google Scholar] [CrossRef]
- Kutikova, L.A.; Silva-Briano, M. Keratella mexicana sp. nov., a New Planktonic Rotifer from Aguascalientes, México. Hydrobiologia 1994, 310, 119–122. [Google Scholar] [CrossRef]
- Örstan, A. A New Species of Bdelloid Rotifer from Sonora, Mexico. Southwest. Nat. 1995, 40, 255–258. [Google Scholar]
- Sarma, S.S.S.; Elías-Gutiérrez, M.; Serranía-Soto, C. Rotifers from High Altitude Crater-Lakes at Nevado De Toluca Volcano, México. Hidrobiologica 1996, 6, 33–38. [Google Scholar]
- Sarma, S.S.S.; Elías-Gutiérrez, M. Taxonomic Studies of Freshwater Rotifers (Rotifera) from Mexico. Pol. Arch. Hydrobiol. 1997, 44, 341–357. [Google Scholar]
- Sarma, S.S.S.; Elías-Gutiérrez, M. Rotifers (Rotifera) from Four Natural Water Bodies of Central Mexico. Limnologica 1999, 29, 475–483. [Google Scholar] [CrossRef] [Green Version]
- Sarma, S.S.S.; Elías-Gutiérrez, M. A Survey on the Rotifer (Rotifera) Fauna of the Yucatan Peninsula (Mexico). Rev. De Biol. Trop. 1999, 47, 187–196. [Google Scholar]
- Hernandez-Rodriguez, M.A.; Santos-Medrano, G.E.; Quintero-Diaz, G.; Rico-Martínez, R. Correlation between Rain and Zooplankton Density and Composition in Eight Dams of Aguascalientes, Mexico. Rev. De Biol. Trop. 1999, 47, 121–127. [Google Scholar]
- Lopez-Lopez, E.; Serna-Hernandez, J.A. Seasonal Zooplankton Variation in the Ignacio Allende Dam, Guanajuato, Mexico and Its Relation to Phytoplankton and Environmental Factors. Rev. De Biol. Trop. 1999, 47, 643–657. [Google Scholar]
- Sarma, S.S.S. Checklist of Rotifera (Rotifers) from Mexico. Environ. Ecol. 1999, 17, 978–983. [Google Scholar]
- Elías-Gutiérrez, M.; Suárez-Morales, E.; Sarma, S.S.S. Diversity of the Freshwater Zooplankton in the Neotropics: The Case of Mexico. Verh. Der Int. Ver. Der Limnol. 2001, 27, 4027–4031. [Google Scholar] [CrossRef]
- Cook, D.R. Water Mite Genera and Subgenera; Memoirs of the American Entomological Institute: Wisconsin, USA, 1974. [Google Scholar]
- Cook, D.R. Studies on Neotropical Water Mites; Memoirs of the American Entomological Institute: Wisconsin, USA, 1980. [Google Scholar]
- Vidrine, M.F. Nine New Species in the Subgenus Atacella (Acari: Unionicolidae: Unionicola) from Mexico and Brazil. Int. J. Acarol. 1985, 11, 255–271. [Google Scholar] [CrossRef]
- Otero-Colina, G. Datos Originales Sobre Los Ácaros Acuáticos (Prostigmata: Parasitengona) Del Sureste De México. Folia Entomológica Mex. 1988, 76, 195–223. [Google Scholar]
- Cramer-Hemkes, C. Koenikea (Notomideopsis) taninulensis, Una Nueva Especie De La Familia Unionicolidae (Acarida: Prostigmata). Folia Entomol. Mex. 1983, 55, 145–152. [Google Scholar]
- Cramer, C. Primer Registro De La Familia Feltriidae (Acarida: Hydrachnellae) Para México Y Descripción De Una Especie Nueva. Folia Entomol. Mex. 1986, 67, 37–43. [Google Scholar]
- Cramer-Hemkes, C.; Costero, A. Descripción De Una Especie Nueva De La Familia Hydrachnidae (Acari: Prostigmata) En Una Poza De Agua Temporal. An. Del Inst. De Biol. 1986, 56, 15–22. [Google Scholar]
- Cramer-Hemkes, C. Nuevas Especies Mexicanas De Hidracaridos Pertenecientes a Los Géneros Neomamersa, Kawamuracarus Y Neotorrenticola (Acarida: Limnesiidae). Folia Entomol. Mex. 1987, 73, 185–203. [Google Scholar]
- Cramer-Hemkes, C.; Cook, D.R. A New Species of Miraxonides (Aturidae: Axonopsinae) from Mexico. Int. J. Acarol. 1990, 17, 13–15. [Google Scholar] [CrossRef]
- Cramer-Hemkes, C.; Smith, I.M. New Species of Water Mites (Acari: Hydrachnida) of the Genera Bandakia (Anisitsiellidae), Chappuisides (Chappuisididae), and Neoacarus (Neocaridae) from Mexico. Can. Entomol. 1991, 123, 795–809. [Google Scholar] [CrossRef]
- Cramer-Hemkes, C. Estudios Sobre Hidracáridos Mexicanos. Especies Nuevas De La Familia Aturidae. Parte I. Folia Entomol. Mex. 1991, 82, 173–186. [Google Scholar]
- Cramer, C. Estudios Sobre Hidracáridos Mexicanos. Especies Nuevas De La Familia Aturidae. Parte II. Folia Entomol. Mex. 1992, 84, 113–129. [Google Scholar]
- Cramer-Hemkes, C.; Cook, D.R. New Species of Arrenurus (Acari, Arrenuridae) from Mexican Lakes. Acarologia 1992, 33, 349–366. [Google Scholar]
- Rivas, G.; Cramer-Hemkes, C. Especie Nueva De Hidracárido (Arrenurus truncaturus) (Acarida: Prostigmata) De Veracruz, México. An. Del Inst. De Biol. 1998, 69, 173–179. [Google Scholar]
- Brehm, V. Mexicanische Entomostraken. Osterreicher Zool. Z. 1955, 6, 412–420. [Google Scholar]
- Van de Velde, I.; Dumont, H.J.; Grootaert, P. Report on a Collection of Cladocera from Mexico and Guatemala. Arch. Fur Hydrobiol. 1978, 83, 391–404. [Google Scholar]
- Frey, D.G. Cladocera. Aquatic Biota of Mexico, Central America and the West Indies; Hurlbert, S.H., Villalobos-Figueroa, A., Eds.; San Diego State University: San Diego, CA, USA, 1982; pp. 177–186. [Google Scholar]
- Suárez-Morales, E.; Elías-Gutiérrez, M. Cladoceros (Crustacea: Branchiopoda) De La Reserva De La Biósfera De Sian Ka’an Quintana Roo Y Zonas Adyacentes. In Diversidad Biológica En La Reserva De Reserva De La Biósfera De Sian Ka’an Quintana Roo, México; Navarro, D., Suárez-Morales, E., Eds.; CIQRO/SEDESOL: Chetumal, México, 1992; pp. 145–161. [Google Scholar]
- Elías-Gutiérrez, M. Notas Sobre Los Cladóceros De Embalses a Gran Altitud En El Estado De México, México. An. De La Esc. Nac. De Cienc. Biológicas 1995, 40, 197–214. [Google Scholar]
- Ciros-Pérez, J.; Elías-Gutiérrez, M. Nuevos Registros De Cladóceros (Crustacea: Anomopoda) En México. Rev. De Biol. Trop. 1996, 44, 297–304. [Google Scholar]
- Ciros-Pérez, J.; Silva-Briano, M.; Elías-Gutiérrez, M. A New Species of Macrothrix (Anomopoda: Macrothricidae) from Central Mexico. Hydrobiologia 1996, 319, 159–166. [Google Scholar] [CrossRef]
- Hebert, P.D.N.; Finston, T.L. A Taxonomic Reevaluation of North American Daphnia (Crustacea: Cladocera) Ii. New Species in the Daphnia pulex Group from the South-Central United States and Mexico. Can. J. Zool. 1996, 74, 632–653. [Google Scholar] [CrossRef]
- Ciros-Pérez, J.; Elías-Gutiérrez, M. Spinalona anophtalma n. gen. N. sp. (Anomopoda, Chydoridae) a Blind Epigean Cladoceran from the Neovolcanic Province of Mexico. Hydrobiologia 1997, 353, 19–28. [Google Scholar] [CrossRef]
- CirosPerez, J.; EliasGutierrez, M. Macrothrix Smirnovi, a New Species (Crustacea: Anomopoda: Macrothricidae) from México, a Member of the M. triserialis-Group. Proc. Biol. Soc. Wash. 1997, 110, 115–127. [Google Scholar]
- Elías-Gutiérrez, M.; Suárez-Morales, E. Alona pectinata (Crustacea: Anomopoda; Chydoridae), a New Freshwater Cladoceran from Southeast Mexico. Rev. De Biol. Trop. 1999, 47, 105–111. [Google Scholar]
- Silva-Briano, M.; Dieu, N.Q.; Dumont, H.J. Redescription of Macrothrix laticornis (Jurine, 1820), and Description of Two New Species of the M. laticornis-Group. Hydrobiologia 1999, 403, 39–61. [Google Scholar] [CrossRef]
- Mattox, N.T. Conchostraca. In Freshwater Biology; Edmonson, W.T., Ed.; Wiley: New York, NY, USA, 1959; pp. 577–586. [Google Scholar]
- Moore, G. On the Occurrence of Streptocephalus similis Baird in Mexico and the United States. J. Wash. Acad. Sci. 1958, 48, 169–175. [Google Scholar]
- Moore, W.G. New World Fairy Shrimps of the Genus Streptocephalus (Branchiopoda, Anostraca). Southwest. Nat. 1966, 11, 24–48. [Google Scholar] [CrossRef]
- Belk, D. Streptocephalus moorei n.sp., a New Fairy Shrimp (Anostraca) from Mexico. Trans. Am. Microsc. Soc. 1973, 92, 507–512. [Google Scholar] [CrossRef]
- Bowen, S.T.; Durkin, J.P.; Sterling, G.; Clark, L.S. Artemia hemoglobins: Genetic Variation in Parthenogenetic and Zygogenetic Populations. Biol. Bull. 1978, 155, 273–287. [Google Scholar] [CrossRef]
- Abreu-Grobois, F.A.; Beardmore, J.A. Genetic Characterization of Artemia Populations: An Electrophoretic Approach. In International Study on Artemia. II. The Brine Shrimp Artemia; Persoone, G., Sorgeloos, P., Roels, P., Jaspers, O., Eds.; Universa Press: Wetteren, Belgium, 1980; pp. 133–146. [Google Scholar]
- Abreu-Grobois, F.A.; Beardmore, J.A. Chromosomes and Chromocenters in the Genus Artemia. An. Del Inst. De Cienc. Del Mar Y Limnol. Univ. Nac. Autónoma De México 1989, 16, 231–244. [Google Scholar]
- Spicer, G.S. A New Fairy Shrimp of the Genus Streptocephalus from Mexico with a Phylogenetic Analysis of the North-American Species (Anostraca). J. Crustacean Biol. 1985, 5, 168–174. [Google Scholar] [CrossRef]
- Martin, J.W. Eulimnadia belki, a New Clam Shrimp from Cozumel, Mexico (Conchostraca: Limnadiidae), with a Review of Central and South American Species of the Genus Eulimnadia. J. Crustacean Biol. 1989, 9, 104–114. [Google Scholar] [CrossRef]
- Eng, L.L.; Belk, D.; Eriksen, C.H. Californian Anostraca-Distribution, Habitat, and Status. J. Crustacean Biol. 1990, 10, 247–277. [Google Scholar] [CrossRef]
- Maeda-Martínez, A.M. Paralimnetis mapimi Nueva Especie (Crustacea: Conchostraca), Un Nuevo Camarón Almeja Del Desierto Chihuahuense En El Norte De México. Southwest. Nat. 1987, 32, 67–74. [Google Scholar] [CrossRef]
- Maeda-Martínez, A.M.; Obregón-Barboza, H.; Dumont, H.J. Branchinecta belki N. Sp. (Branchiopoda, Anostraca), a New Fairy Shrimp from Mexico, Hybridizing with B. packardi Pearse under Laboratory Conditions. Hydrobiologia 1992, 239, 151–162. [Google Scholar] [CrossRef]
- Maeda-Martínez, A.M.; Obregón-Barboza, H.; Dumont, H.J. Branchinecta mexicana, New Species (Branchiopoda: Anostraca) a Fairy Shrimp from Central México. J. Crustacean Biol. 1993, 13, 585–593. [Google Scholar] [CrossRef]
- Maeda-Martínez, A.M.; Obregón-Barboza, H.; Dumont, H.J. A Contribution to the Systematics of the Streptocephalidae (Branchiopoda: Anostraca). Hydrobiologia 1995, 298, 203–232. [Google Scholar] [CrossRef]
- Maeda-Martínez, A.M.; Obregón-Barboza, H.; Dumont, H.J. Large Branchiopod Assemblages Common to Mexico and the United States. Hydrobiologia 1997, 359, 45–62. [Google Scholar] [CrossRef]
- Fugate, M. Branchinecta sandiegonensis, a New Species of Fairy Shrimp (Crustacea, Anostraca) from Western North-America. Proc. Biol. Soc. Wash. 1993, 106, 296–304. [Google Scholar]
- Comita, G.W. Studies on Mexican Copepods. Trans. Am. Microsc. Soc. 1950, 69, 367–379. [Google Scholar] [CrossRef]
- Lindberg, K. Cyclopoïdes (Crustacés Copépodes) Du Mexike. Ark. För Zool. 1954, 7, 459–489. [Google Scholar]
- Fernando, C.H.; Smith, K.E. Copepoda. In Aquatic Biota of Mexico, Central America and West Indies; Hurlbert, S.H., Villalobos-Figueroa, A., Eds.; San Diego State University: San Diego, CA, USA, 1982; pp. 192–195. [Google Scholar]
- Reid, J.W. Cyclopoid and Harpacticoid Copepods (Crustacea) from México, Guatemala and Colombia. Transactions Am. Microsc. Soc. 1988, 107, 190–202. [Google Scholar] [CrossRef]
- Reid, J.W. Continental and Coastal Free-Living Copepoda (Crustacea) of Mexico, Central America and the Caribbean Region. In Diversidad Biológica En La Reserva De La Biósfera De Sian Ka’an Quintana Roo, México; Navarro, L.D., Robinson, J.G., Eds.; Centro de Investigaciones de Quintana Roo (CIQRO): Chetumal, México, 1990; pp. 175–213. [Google Scholar]
- Zamudio-Valdez, J.A.; Reid, J.W. A New Species of Leptocaris (Crustacea, Copepoda, Harpacticoida) from Inland Waters of México. An. Inst. Cienc. Mar Limnol. 1990, 17, 47–54. [Google Scholar]
- Reid, J.W. New Records and Redescriptions of American Species of Mesocyclops and of Diacyclops Bernardi (Petkovski, 1986) (Copepoda: Cyclopoida). Bijdr. Dierkd. 1993, 63, 173–191. [Google Scholar] [CrossRef] [Green Version]
- Suárez-Morales, E.; Reid, J.W.; Iliffe, T.M.; Fiers, F. Catálogo De Los Copépodos (Crustacea) Continentales De La Península De Yucatán; El Colegio de la Frontera Sur/Comisión Nacional Para el Conocimiento y Uso de la Biodiversidad: Ciudad de México, México, 1996. [Google Scholar]
- Fiers, F. Halicyclops caneki n. sp. (Copepoda, Cyclopoida) from Celestun Lagoon (Yucatan, Mexico). Belg. J. Zool. 1995, 125, 301–313. [Google Scholar]
- Suárez-Morales, E.; Reid, J.W. Adiciones a La Fauna Plánctica De Aguas Continentales De Quintana Roo. AvaCient 1994, 9, 29–33. [Google Scholar]
- Suárez-Morales, E.; Reid, J.W. Ampliación De Ámbito De Dos Copépodos De Aguas Continentales En La Península De Yucatán, México. Rev. De Biol. Trop. 1996, 44, 942–944. [Google Scholar]
- Fiers, F.; Reid, J.W.; Iliffe, T.M.; Suárez-Morales, E. New Hypogean Cyclopoid Copepods (Crustacea) from the Yucatan Peninsula, Mexico. Contrib. Zool. 1996, 66, 65–102. [Google Scholar] [CrossRef] [Green Version]
- Dayrat, B. Towards Integrative Taxonomy. Biol. J. Linn. Soc. 2005, 85, 407–415. [Google Scholar] [CrossRef] [Green Version]
- Mayén-Estrada, R.; Reyes-Santos, M.; Durán-Ramírez, C.A.; Medina-Durán, J.H.; Olvera-Bautista, F.; Vicencio, M.E.; Romero-Niembro, V. Protistas Ciliados Y Flagelados Heterótrofos; Informe Final SNIB-CONABIO. Proyecto No. KT003; Ciudad de México, México, 2020. Available online: http://www.conabio.gob.mx/institucion/proyectos/resultados/KT003_Anexo_Listado_Taxonomico.pdf (accessed on 22 November 2022).
- Fenchel, T.; Finlay, B.J. The Ubiquity of Small Species: Patterns of Local and Global Diversity. Bioscience 2004, 54, 777–784. [Google Scholar] [CrossRef] [Green Version]
- Foissner, W. Dispersal and Biogeography of Protists: Recent Advances. Jpn. J. Protozool. 2007, 40, 1–16. [Google Scholar]
- Debroas, D.; Domaizon, I.; Humbert, J.F.; Jardillier, L.; Lepere, C.; Oudart, A.; Taib, N. Overview of Freshwater Microbial Eukaryotes Diversity: A First Analysis of Publicly Available Metabarcoding Data. Fems Microbiol. Ecol. 2017, 93, 14. [Google Scholar] [CrossRef] [Green Version]
- De Luca, D.; Piredda, R.; Sarno, D.; Kooistra, W.H. Resolving Cryptic Species Complexes in Marine Protists: Phylogenetic Haplotype Networks Meet Global DNA Metabarcoding Datasets. Isme J. 2021, 15, 1931–1942. [Google Scholar] [CrossRef]
- Boenigk, J.; Ereshefsky, M.; Hoef-Emden, K.; Mallet, J.; Bass, D. Concepts in Protistology: Species Definitions and Boundaries. Eur. J. Protistol. 2012, 48, 96–102. [Google Scholar] [CrossRef]
- Olefeld, J.L.; Bock, C.; Jensen, M.; Vogt, J.C.; Sieber, G.; Albach, D.; Boenigk, J. Centers of Endemism of Freshwater Protists Deviate from Pattern of Taxon Richness on a Continental Scale. Sci. Rep. 2020, 10, 13. [Google Scholar] [CrossRef]
- Pestova, D.; Macek, M.; Perez, M.E.M. Ciliates and Their Picophytoplankton-Feeding Activity in a High-Altitude Warm-Monomictic Saline Lake. Eur. J. Protistol. 2008, 44, 13–25. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, M.R.; Lugo Vázquez, A.; Oliva _Martínez, M.G.; Verver y Vargas García, J.; Rodríguez-Rocha, A.; Peralta-Soriano, L. Planktonic Ciliates in a Hypertrophic Pond: Functional Role and Importance. J. Environ. Biol. 2011, 32, 497–503. [Google Scholar]
- Ponce-Márquez, M.E.; Ramírez-Rodríguez, R.; Ramírez-Vázquez, M. Algas De La Cantera Oriente, Reserva Ecológica Del Pedregal De San Ángel; Universidad Nacional Autónoma de México, Ed.; Las Prensas de Ciencias, Facultad de Ciencias: Ciudad de México, México, 2019. [Google Scholar]
- Macek, M.; Sánchez-Medina, X.; Vilaclara, G.; Lugo-Vázquez, A.; Bautista-Reyes, F.; Valdespino-Castillo, P.M. Protozooplankton. In Lake Alchichica Limnology: The Uniqueness of a Tropical Maar Lake; Alcocer, J., Ed.; Springer Nature PM AG: Berlin/Heildeberg, Germany, 2022; pp. 213–236. [Google Scholar]
- Ramírez-Ballesteros, M.; Durán-Ramírez, C.A.; Méndez-Sánchez, D.; Mayén-Estrada, R. Diversidad De Ciliados (Alveolata: Ciliophora) Del Parque Nacional Lagunas De Montebello, Chiapas. In Estudios Sobre La Biodiversidad Tropical Mexicana: Conservación Y Aprovechamiento Sustentable; Peralta, M.M.A., Ruan, S.J.F., De la Cruz, C.I., Diez, B.E.P., Castro, M.M., Than, M.B.A., Eds.; UNICACH: Tuxtla Gutiérrez, México, 2021; pp. 125–155. [Google Scholar]
- Méndez-Sánchez, D. Estudio Taxonómico De Ciliados De Vida Libre (Alveolata: Ciliophora) En Cuerpos De Agua Dulce De Dos Regiones Biogeográficas De México. Master’s Thesis, Universidad Nacional Autónoma de México, Ciudad de México, México, 2017. [Google Scholar]
- Potekhin, A.; Mayén-Estrada, R. Paramecium Diversity and a New Member of the Paramecium aurelia Species Complex Described from Mexico. Diversity 2020, 12, 20. [Google Scholar] [CrossRef]
- Silva-Briano, M.; Galvan-De la Rosa, R.D.; Perez-Legaspi, I.A.; Rico-Martínez, R. On the Description of Brachionus araceliae sp. nov. A New Species of Freshwater Rotifer from Mexico. Hidrobiologica 2007, 17, 179–183. [Google Scholar]
- García-Morales, A.; Elías-Gutiérrez, M. DNA Barcoding of Freshwater Rotifera in Mexico: Evidence of Cryptic Speciation in Common Rotifers. Mol. Ecol. Resour. 2013, 13, 1097–1107. [Google Scholar] [CrossRef]
- Guerrero-Jiménez, G.; Vannucchi, P.E.; Silva-Briano, M.; Adabache-Ortiz, A.; Rico-Martínez, R.; Roberts, D.; Neilson, R.; Elías-Gutiérrez, M. Brachionus paranguensis sp. nov. (Rotifera, Monogononta), a Member of the L Group of the Brachionus plicatilis Complex. Zookeys 2019, 880, 1–23. [Google Scholar] [CrossRef] [Green Version]
- García-Morales, A.E.; Domínguez-Domínguez, O.; Elías-Gutiérrez, M. Uncovering Hidden Diversity: Three New Species of the Keratella Genus (Rotifera, Monogononta, Brachionidae) of High Altitude Water Systems from Central Mexico. Diversity 2021, 13, 21. [Google Scholar] [CrossRef]
- Sarma, S.S.S.; Jimenez-Santos, M.A.; Nandini, S. Rotifer Species Diversity in Mexico: An Updated Checklist. Diversity 2021, 13, 18. [Google Scholar] [CrossRef]
- Cramer-Hemkes, C. New Species of the Water Mite Genus Kongsbergia (Acari: Hydrachnida: Aturidae) from Mexico. Int. J. Acarol. 2000, 26, 271–278. [Google Scholar] [CrossRef]
- Marin-Hernandez, C.H.; Cramer-Hemkes, C. New Species of Water Mites of the Genus Piona (Acari: Hydrachnidia: Pionidae), from Xochimilco’s Water Channel, Mexico. Rev. Mex. De Biodivers. 2009, 80, 51–54. [Google Scholar]
- Ramirez-Sánchez, M.M.; Rivas, G. New Species of Subgenus Megaluracarus (Acari: Hydrachnidiae: Arrenuridae: Arrenurus) from Mexico. Zootaxa 2013, 3718, 317–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montes-Ortiz, L.; Elías-Gutiérrez, M. Faunistic Survey of the Zooplankton Community in an Oligotrophic Sinkhole, Cenote Azul (Quintana Roo, Mexico), Using Different Sampling Methods, and Documented with DNA Barcodes. J. Limnol. 2018, 77, 428–440. [Google Scholar] [CrossRef]
- Montes-Ortiz, L.; Elías-Gutiérrez, M. Water Mite Diversity (Acariformes: Prostigmata: Parasitengonina: Hydrachnidiae) from Karst Ecosystems in Southern of Mexico: A Barcoding Approach. Diversity 2020, 12, 329. [Google Scholar] [CrossRef]
- Montes-Ortiz, L.; Goldschmidt, T.; Vasquez-Yeomans, L.; Elías-Gutiérrez, M. A New Species of Litarachna Walter, 1925 (Acari: Hydrachnidia: Pontarachnidae) from Corozal Bay (Belize), Described Based Upon and DNA Barcodes. Acarologia 2021, 61, 602–613. [Google Scholar] [CrossRef]
- Montes-Ortiz, L.; Elías-Gutiérrez, M.; Ramirez-Sanchez, M.M. Checklist of Arrenurids (Acari: Hydrachnidia: Arrenuridae) of Mexico, with New Records from the Yucatan Peninsula, and the Description of Five New Species of the Subgenera Megaluracarus and Dadayella. Diversity 2022, 14, 25. [Google Scholar] [CrossRef]
- Elías-Gutiérrez, M.; Montes-Ortiz, L. Present Day Kwnoledge on Diversity of Freshwater Zooplancton (Invertebrates) of the Yucatan Peninsula, Using Integrated Taxonomy. Teor. Y Prax. 2018, 14, 31–48. [Google Scholar]
- Elías-Gutiérrez, M.; Smirnov, N.N. Macrothrix marthae, a New Species (Crustacea: Anomopoda: Macrothricidae), a Highly Specialized Macrothricid from Mexico. Proc. Biol. Soc. Wash. 2000, 113, 652–660. [Google Scholar]
- Cervantes-Martínez, A.; Gutiérrez-Aguirre, M.; Elías-Gutiérrez, M. Description of Ilyocryptus nevadensis (Branchiopoda: Anomopoda), a New Species from a High Altitude Crater Lake in the Volcano Nevado De Toluca, Mexico. Crustaceana 2000, 73, 311–321. [Google Scholar]
- Kotov, A.A.; Elías-Gutiérrez, M.; Gutiérrez-Aguirre, M. Ilyocryptus paranaensis inarmatus subsp. nov. from Tabasco, Mexico (Cladocera. Anomopoda). Crustaceana 2001, 74, 1067–1082. [Google Scholar]
- Kotov, A.A.; Elías-Gutiérrez, M.; Nieto, M.G. Leydigia louisi louisi Jenkin, 1934 in the Neotropics, L. louisi mexicana n. subsp. in the Central Mexican Highlands. Hydrobiologia 2003, 510, 239–255. [Google Scholar] [CrossRef]
- Kotov, A.A.; Elías-Gutiérrez, M.; Granados-Ramirez, J.G. Moina dumonti sp. nov. (Cladocera, Anomopoda, Moinidae) from Southern Mexico and Cuba, with Comments on Moinid Limbs. Crustaceana 2005, 78, 41–57. [Google Scholar]
- Elías-Gutiérrez, M.; Kotov, A.A.; Garfias-Espejo, T. Cladocera (Crustacea: Ctenopoda, Anomopoda) from Southern Mexico, Belize and Northern Guatemala, with Some Biogeographical Notes. Zootaxa 2006, 1119, 1–27. [Google Scholar] [CrossRef]
- Elías-Gutiérrez, M.; Smirnov, N.N.; Suárez-Morales, E.; Dimas-Flores, N. New and Little Known Cladocerans (Crustacea: Anomopoda) from Southeastern Mexico. Hydrobiologia 2001, 442, 41–54. [Google Scholar] [CrossRef]
- Garfias-Espejo, T.; Elías-Gutiérrez, M. Taxonomy and Distribution of Macrothricidae (Crustacea: Anomopoda) in Southeastern Mexico, Northern Guatemala and Belize. An. Del Inst. De Biol. 2003, 74, 105–134. [Google Scholar]
- Elías-Gutiérrez, M.; Valdez-Moreno, M. A New Cryptic Species of Leberis Smirnov, 1989 (Crustacea, Cladocera, Chydoridae) from the Mexican Semi-Desert Region, Highlighted by DNA Barcoding. Hidrobiologica 2008, 18, 63–74. [Google Scholar]
- Quiroz-Vazquez, P.; Elías-Gutiérrez, M. A New Species of the Freshwater Cladoceran Genus Scapholeberis Schoedler, 1858 (Cladocera: Anomopoda) from the Semidesert Northern Mexico, Highlighted by DNA Barcoding. Zootaxa 2009, 2236, 50–64. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Caballero, A.L.; Morales-Gutiérrez, S.; Elías-Gutiérrez, M. First Record of the Genus Bunops Birge, 1893 (Cladocera: Macrotrichidae) in the Neotropical Highlands of Mexico with a Detailed Study of Morphology and DNA Barcodes. Zootaxa 2017, 4300, 589–600. [Google Scholar] [CrossRef]
- Montoliu-Elena, L.; Elias-Gutierrez, M.; Silva-Briano, M. Moina macrocopa (Straus, 1820): A Species Complex of a Common Cladocera, Highlighted by Morphology and DNA Barcodes. Limnetica 2019, 38, 253–277. [Google Scholar] [CrossRef]
- Maeda-Martínez, A.; Murugan, G.; Morales-Avila, R.; Obregón-Barboza, H.; Ramos, R.C.; Martínez Días, S.F. Biología Del Camarón De Salmuera Artemia De La Salina De Pichilingue, Bahía De La Paz, Baja California Sur, México. In La Bahía De La Paz: Biodiversidad, Procesos Ecológicos Y Sociales; Riosmena Rodríguez, R., López Vivas, J.M., Gómez Gallardo Unzueta, E.A., Cruz Piñón, G., Romo Piñera, A.K., Eds.; Universidad Autónoma de Baja California Sur: La Paz, México, 2020; pp. 409–439. [Google Scholar]
- Torrentera, L.; Abreu-Grobois, F.A. Cytogenetic Variability and Differentiation in Artemia (Branchiopoda: Anostraca) Populations from the Yucatan Peninsula, Mexico. Hydrobiologia 2002, 486, 303–314. [Google Scholar] [CrossRef]
- Tizol-Correa, R.; Maeda-Martínez, A.M.; Weekers, P.H.H.; Torrentera, L.; Murugan, G. Biodiversity of the Brine Shrimp Artemia from Tropical Salterns in Southern Mexico and Cuba. Curr. Sci. 2009, 96, 81–87. [Google Scholar]
- Sainz-Escudero, L.; Lopez-Estrada, E.K.; Rodriguez-Flores, P.C.; García-Paris, M. Settling Taxonomic and Nomenclatural Problems in Brine Shrimps, Artemia (Crustacea: Branchiopoda: Anostraca), by Integrating Mitogenomics, Marker Discordances and Nomenclature Rules. Peerj 2021, 9, 62. [Google Scholar] [CrossRef] [PubMed]
- Maeda-Martínez, A.M.; Obregón-Barboza, V.; Navarrete-Del Toro, M.A.; Obregón-Barboza, H.; García-Carreño, F.L. Trypsin-Like Enzymes from Two Morphotypes of the ‘Living Fossil’ Triops (Crustacea: Branchiopoda: Notostraca). Comp. Biochem. Physiol. B-Biochem. Mol. Biol. 2000, 126, 317–323. [Google Scholar] [CrossRef]
- Murugan, G.; Maeda-Martínez, A.M.; Obregón-Barboza, H.; Hernández-Saavedra, N.Y. Molecular Characterization of the Tadpole Shrimp Triops (Branchiopoda: Notostraca) from the Baja California Peninsula, Mexico: New Insights on Species Diversity and Phylogeny of the Genus. Hydrobiologia 2002, 486, 101–113. [Google Scholar] [CrossRef]
- García-Velazco, H.; Obregón-Barboza, H.; Rodriguez-Jaramillo, C.; Maeda-Martínez, A.M. Reproduction of the Tadpole Shrimp Triops (Notostraca) in Mexican Waters. Curr. Sci. 2009, 96, 91–97. [Google Scholar]
- Obregón-Barboza, H.; Maeda-Martínez, A.M.; Murugan, G. Reproduction, Molting, and Growth of Two Mexican Uniparental Forms of the Tadpole Shrimp Triops (Branchiopoda: Notostraca) under a Recirculating Culture System. Hydrobiologia 2001, 462, 173–184. [Google Scholar] [CrossRef]
- Obregón-Barboza, H.; Maeda-Martínez, A.M.; García-Velazco, H.; Dumont, H.J. Branchinecta oterosanvicentei N. Sp. (Branchiopoda: Anostraca), a New Fairy Shrimp from the Chihuahuan Desert, with a Proposal for the Conservation of the Branchinectidae of Mexico. Hydrobiologia 2002, 467, 45–56. [Google Scholar] [CrossRef]
- Maeda-Martínez, A.M.; Obregón-Barboza, H.; Prieto-Salazar, M.; García-Velazco, H. Two New Fairy Shrimp of the Genus Streptocephalus (Branchiopoda: Anostraca) from North America. J. Crustacean Biol. 2005, 25, 537–546. [Google Scholar] [CrossRef] [Green Version]
- Obregón-Barboza, H.; Murugan, G.; García-Velazco, H.; Maeda-Martínez, A.M. A Review of the Branchinecta (Branchiopoda: Anostraca) from the Baja California Peninsula: First Record of the Giant Fairy Shrimp B-Gigas Lynch, 1937 from Mexico. J. Crustacean Biol. 2015, 35, 433–440. [Google Scholar] [CrossRef] [Green Version]
- Aguilar, A.; Maeda-Martinez, A.M.; Murugan, G.; Obregón-Barboza, H.; Christopher Rogers, D.; McClintock, K.; Krumm, J.L. High Intraspecific Genetic Divergence in the Versatile Fairy Shrimp Branchinecta Lindahli with a Comment on Cryptic Species in the Genus Branchinecta (Crustacea: Anostraca). Hydrobiologia 2017, 801, 59–69. [Google Scholar] [CrossRef]
- Obregón-Barboza, H.; Murugan, G.; García-Velazco, H.; Maeda-Martínez, A.M. A Systematic Review of Mexican Populations of the Fairy Shrimp Genus Thamnocephalus (Branchiopoda: Anostraca). J. Crustacean Biol. 2015, 35, 407–432. [Google Scholar] [CrossRef] [Green Version]
- Mercado-Salas, N.; Mercado-Salas, N.F.; Khodami, S.; Kihara, T.; Elías-Gutiérrez, M.; Arbizu, P. Genetic Structure and Distributional Patterns of the Genus Mastigodiaptomus (Copepoda) in Mexico, with the Description of a New Species from the Yucatan Peninsula. Arthropod Syst. Phylogeny 2018, 76, 487–507. [Google Scholar]
- Miguez-Gutiérrez, A.; Castillo, J.; Marquez, J.; Goyenechea, I. Biogeography of the Mexican Transition Zone Based on a Reconciled Trees Analysis. Rev. Mex. De Biodivers. 2013, 84, 215–224. [Google Scholar] [CrossRef] [Green Version]
- Morrone, J.J. Biogeographic Regionalization and Biotic Evolution of Mexico: Biodiversity’s Crossroads of the New World. Rev. Mex. De Biodivers. 2019, 90, 68. [Google Scholar]
- Velázquez-Ornelas, K.; Juárez-Carrillo, E.; Ayón-Parente, M. Zooplancton (Cladocera Y Copepoda) De La Laguna De Cajititlán. e-CUCBA 2021, 16, 12–20. [Google Scholar] [CrossRef]
- Boxshall, G.A.; Defaye, D. Global Diversity of Copepods (Crustacea: Copepoda) in Freshwater. Hydrobiologia 2008, 595, 195–207. [Google Scholar] [CrossRef]
- Rocha, C.E.F.; Iliffe, T.M.; Reid, J.W.; Suárez-Morales, E. Prehendocyclops, a New Genus of the Subfamily Halicyclopinae (Copepoda, Cyclopoida, Cyclopidae) from Cenotes of the Yucatan Peninsula, Mexico. Sarsia 2000, 85, 119–140. [Google Scholar] [CrossRef]
- Mercado-Salas, N.F.; Suarez-Morales, E.; Maeda-Martínez, A.M.; Silva-Briano, M. A New Species of Metacyclops Kiefer, 1927 (Copepoda, Cyclopidae, Cyclopinae) from the Chihuahuan Desert, Northern Mexico. Zookeys 2013, 287, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Gutiérrez-Aguirre, M.; Suárez-Morales, E. The Eurasian Thermocyclops crassus (Fischer, 1853) (Copepoda, Cyclopoida) Found in Southeastern Mexico. Crustaceana 2000, 73, 705–713. [Google Scholar] [CrossRef] [Green Version]
- Suárez-Morales, E.; Gutiérrez-Aguirre, M.A.; Mendoza, F. The Afro-Asian Cyclopoid Mesocyclops aspericornis (Crustacea: Copepoda) in Eastern Mexico with Comments on the Distribution of Exotic Copepods. Rev. Mex. De Biodivers 2011, 82, 109–115. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Walsh, E.J. Two New Species of Eucyclops Claus (Copepoda: Cyclopoida) from the Chihuahuan Desert with a Redescription of E. pseudoensifer Dussart. Zootaxa 2009, 2206, 1–22. [Google Scholar] [CrossRef]
- Mercado-Salas, N.F.; Suárez-Morales, E.; Silva-Briano, M. Taxonomic Revision of the Mexican Eucyclops (Copepoda: Cyclopoida) with Comments on the Biogeography of the Genus. J. Nat. Hist. 2016, 50, 25–147. [Google Scholar] [CrossRef]
- Mercado-Salas, N.F.; Khodami, S.; Arbizu, P.M. Copepods and Ostracods Associated with Bromeliads in the Yucatan Peninsula, Mexico. PLoS ONE 2021, 16, e0248863. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Mendoza, F.; Mercado-Salas, N. A New Allocyclops (Crustacea, Copepoda, Cyclopoida) from Bromeliads and Records of Freshwater Copepods from Mexico. Zoosystema 2010, 32, 393–407. [Google Scholar] [CrossRef]
- Gomez, S.; Fleeger, J.W.; Rocha-Olivares, A.; Foltz, D. Four New Species of Cletocamptus Schmankewitsch, 1875, Closely Related to Cletocamptus deitersi (Richard, 1897) (Copepoda: Harpacticoida). J. Nat. Hist. 2004, 38, 2669–2732. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Iliffe, T.M. A New Stygonitocrella Petkovski (Copepoda: Harpacticoida) from a Cave in Northern Mexico with Comments on the Taxonomy of the Genus. Hydrobiologia 2005, 544, 215–228. [Google Scholar] [CrossRef] [Green Version]
- Suárez-Morales, E.; Barrera-Moreno, O.; Ciros-Perez, J. A New Species of Cletocamptus Schmankewitsch, 1875 (Crustacea, Copepoda, Harpacticoida) from a High Altitude Saline Lake in Central Mexico. J. Limnol. 2013, 72, 313–325. [Google Scholar] [CrossRef] [Green Version]
- Alcocer, J.; del Carmen Hernandez, M.; Oseguera, L.A.; Escobar, E. On the Ecology of Cletocamptus gomezi Suárez-Morales, Barrera-Moreno & Ciros-Perez 2013 (Crustacea, Copepoda, Harpacticoida) Micro-Endemic to Lake Alchichica, Central Mexico. J. Limnol. 2015, 74, 302–309. [Google Scholar]
- Nandini, S.; Silva-Briano, M.; García-García, G.; Sarma, S.S.S.; Adabache-Ortiz, A.R.; de la Rosa, R.G. First Record of the Temperate Species Daphnia curvirostris Eylmann, 1887 Emend. Johnson, 1952 (Cladocera: Daphniidae) in Mexico and Its Demographic Characteristics in Relation to Algal Food Density. Limnology 2009, 10, 87–94. [Google Scholar] [CrossRef]
- Elías-Gutiérrez, M. Zooplancton De Agua Dulce: Especies Exóticas, Posibles Vías De Introducción. In Especies Acuáticas Invasoras En México; Mendoza-Alfaro, R., Koleff-Osorio, P., Eds.; Comisión Nacional para el Conocimiento y Uso de la Biodiversidad: Ciudad de México, México, 2014; pp. 309–315. [Google Scholar]
- Silva-Briano, M.; Arroyo-Bustos, G.; Beltrán-Alvarez, R.; Adabache-Ortiz, A.; Galvan-De la Rosa, R.D. Daphnia Ctenodaphnia lumholtzi G. O. Sars, 1885 (Crustacea: Cladocera); Un Cladócero Exótico En México. Hidrobiologica 2010, 20, 275–280. [Google Scholar]
- Suárez-Morales, E.; Arroyo-Bustos, G. An Intra-Continental Invasion of the Temperate Freshwater Copepod Skistodiaptomus pallidus (Herrick, 1879) (Calanoida, Diaptomidae) in Tropical Mexico. BioInvasions Rec. 2012, 1, 255–262. [Google Scholar] [CrossRef]
- Suárez-Morales, E.; Gutiérrez-Aguirre, M.A.; Torres, J.L.; Hernandez, F. The Asian Mesocyclops pehpeiensis Hu, 1943 (Crustacea, Copepoda, Cyclopidae) in Southeast Mexico with Comments on the Distribution of the Species. Zoosystema 2005, 27, 245–256. [Google Scholar]
- Suárez-Morales, E.; Mercado-Salas, N.F.; Morales-Ramírez, Á. Morphological Variability and Distribution of the Exotic Asian Mesocyclops thermocyclopoides (Copepoda: Cyclopoida) in the Neotropical Region. Zoologia 2011, 28, 673–679. [Google Scholar] [CrossRef] [Green Version]
- Montoliu, L.; Miracle, M.R.; Elías-Gutiérrez, M. Using DNA Barcodes to Detect Non-Indigenous Species: The Case of the Asian Copepod Mesocyclops pehpeiensis Hu, 1943 (Cyclopidae) in Two Regions of the World. Crustaceana 2015, 88, 1323–1338. [Google Scholar] [CrossRef]
- Reid, J.W.; Bayly, I.A.; Pesce, G.L.; Rayner, N.A.; Reddy, Y.R.; Rocha, C.E.; Suárez-Morales, E.; Ueda, H. Conservation of Continental Copepod Crustaceans. In Modern Approaches to the Study of Crustacea; Escobar-Briones, E., Alvarez, F., Eds.; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2002; pp. 253–261. [Google Scholar]
- Martínez-Jeronimo, F.; Elías-Gutiérrez, M.; Suárez-Morales, E. A Redescription of Moina hutchinsoni, a Rare Cladoceran (Branchiopoda: Anomopoda) Found in Remnants of a Mexican Saline Lake, with Notes on Its Life History. J. Crustacean Biol. 2004, 24, 232–245. [Google Scholar] [CrossRef] [Green Version]
- Mercado-Salas, N.F.; Morales-Vela, B.; Suárez-Morales, E.; Iliffe, T.M. Conservation Status of the Inland Aquatic Crustaceans in the Yucatan Peninsula, Mexico: Shortcomings of a Protection Strategy. Aquat. Conserv. -Mar. Freshw. Ecosyst. 2013, 23, 939–951. [Google Scholar] [CrossRef]
- Makino, W.; Maruoka, N.; Nakagawa, M.; Takamura, N. DNA Barcoding of Freshwater Zooplankton in Lake Kasumigaura. Japan. Ecol. Res. 2017, 32, 481–493. [Google Scholar] [CrossRef]
- Jaime, S.; Cervantes-Martínez, A.; Gutiérrez-Aguirre, M.A.; Suárez-Morales, E.; Juárez-Pernillo, J.R.; Reyes-Solares, E.M.; Delgado-Blas, V.H. Historical Zooplankton Composition Indicates Eutrophication Stages in a Neotropical Aquatic System: The Case of Lake Amatitlan, Central America. Diversity 2021, 13, 10. [Google Scholar] [CrossRef]
- Macario-Gonzalez, L.; Cohuo, S.; Angyal, D.; Perez, L.; Mascaro, M. Subterranean Waters of Yucatan Peninsula, Mexico Reveal Epigean Species Dominance and Intraspecific Variability in Freshwater Ostracodes (Crustacea: Ostracoda). Diversity 2021, 13, 24. [Google Scholar] [CrossRef]
 Phase 1. Early studies dominated by foreign visitors;
Phase 1. Early studies dominated by foreign visitors;  Phase 2. Towards the formation of Mexican protistologists;
Phase 2. Towards the formation of Mexican protistologists;  Phase 3. Consolidation of Protist studies.
Phase 3. Consolidation of Protist studies.
 Phase 1. Early studies dominated by foreign visitors;
Phase 1. Early studies dominated by foreign visitors;  Phase 2. Towards the formation of Mexican protistologists;
Phase 2. Towards the formation of Mexican protistologists;  Phase 3. Consolidation of Protist studies.
Phase 3. Consolidation of Protist studies.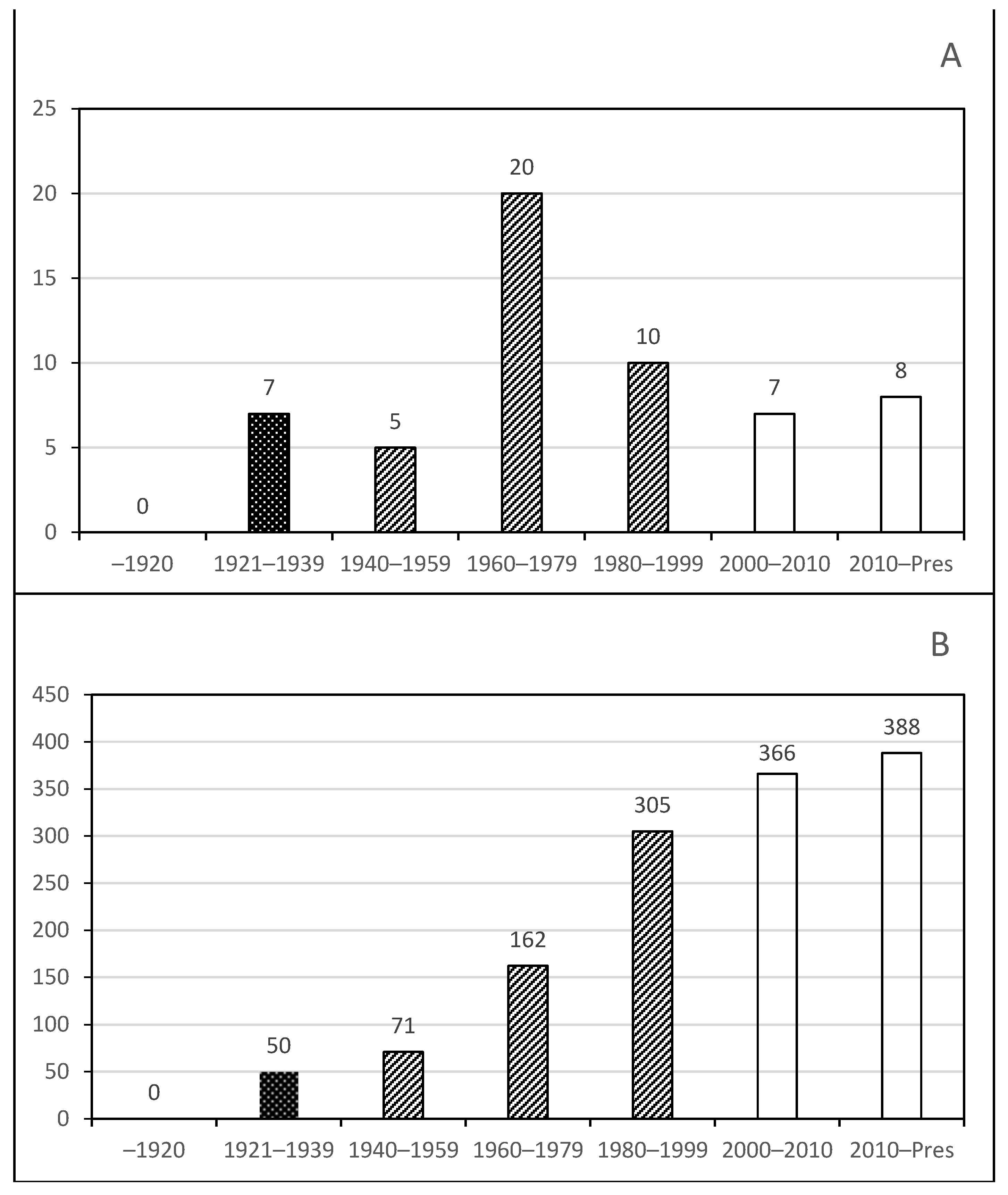
 Phase 1. Early studies included Mexican researchers but were dominated by foreign visitors;
Phase 1. Early studies included Mexican researchers but were dominated by foreign visitors;  Phase 2. Towards the formation of Mexican rotiferologists, working with foreign researchers, and first species descriptions;
Phase 2. Towards the formation of Mexican rotiferologists, working with foreign researchers, and first species descriptions;  Phase 3. Consolidation of rotifer studies and integrative taxonomy descriptions.
Phase 3. Consolidation of rotifer studies and integrative taxonomy descriptions.
 Phase 1. Early studies included Mexican researchers but were dominated by foreign visitors;
Phase 1. Early studies included Mexican researchers but were dominated by foreign visitors;  Phase 2. Towards the formation of Mexican rotiferologists, working with foreign researchers, and first species descriptions;
Phase 2. Towards the formation of Mexican rotiferologists, working with foreign researchers, and first species descriptions;  Phase 3. Consolidation of rotifer studies and integrative taxonomy descriptions.
Phase 3. Consolidation of rotifer studies and integrative taxonomy descriptions.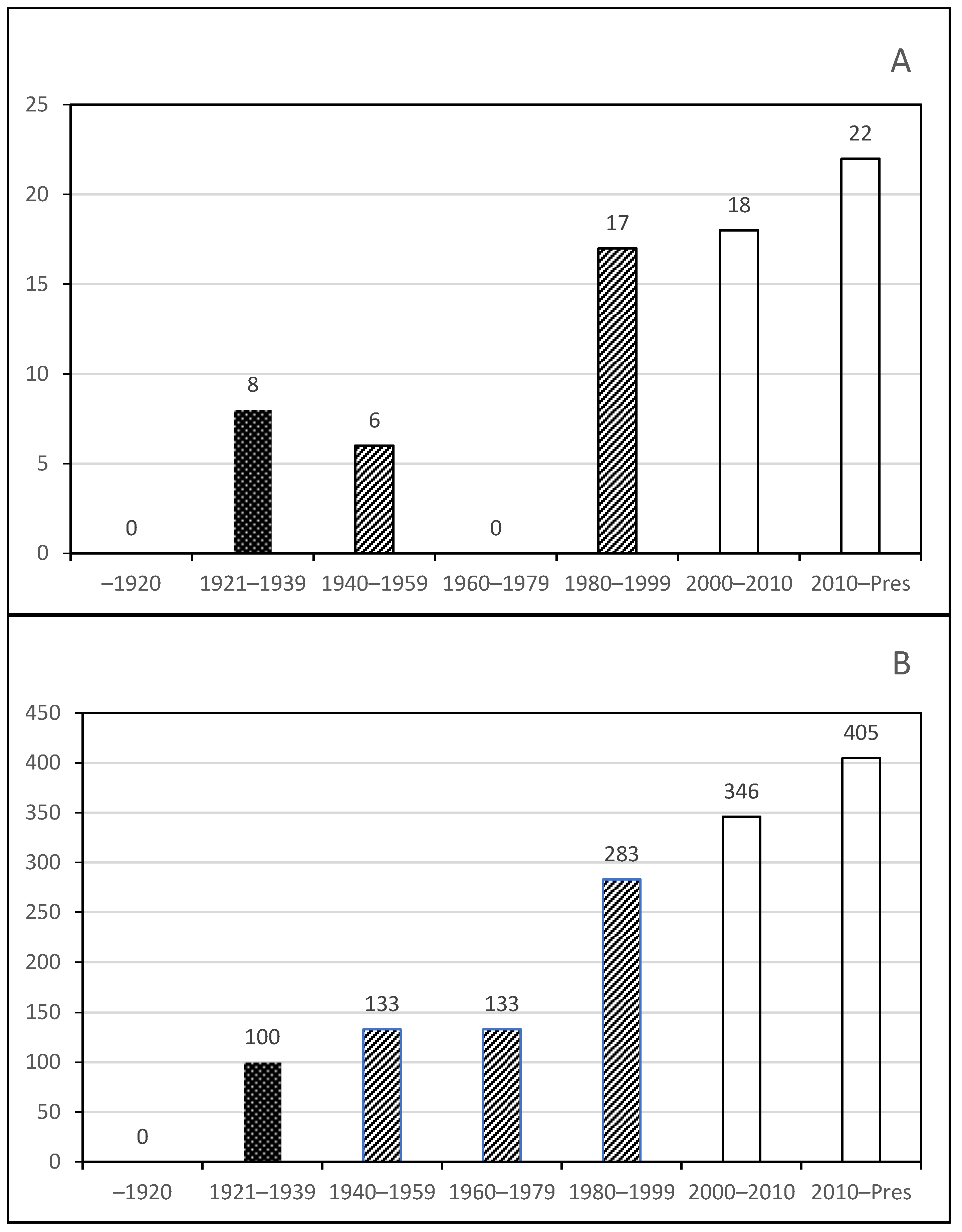
 Phase 1. Early studies by foreign visitors;
Phase 1. Early studies by foreign visitors;  Phase 2. The consolidation of Mexican cladocerologists in the last period (1980–1999);
Phase 2. The consolidation of Mexican cladocerologists in the last period (1980–1999);  Phase 3. Mature period and formation of 2nd and 3rd generation of researchers on this topic, and discovery of exotic species.
Phase 3. Mature period and formation of 2nd and 3rd generation of researchers on this topic, and discovery of exotic species.
 Phase 1. Early studies by foreign visitors;
Phase 1. Early studies by foreign visitors;  Phase 2. The consolidation of Mexican cladocerologists in the last period (1980–1999);
Phase 2. The consolidation of Mexican cladocerologists in the last period (1980–1999);  Phase 3. Mature period and formation of 2nd and 3rd generation of researchers on this topic, and discovery of exotic species.
Phase 3. Mature period and formation of 2nd and 3rd generation of researchers on this topic, and discovery of exotic species.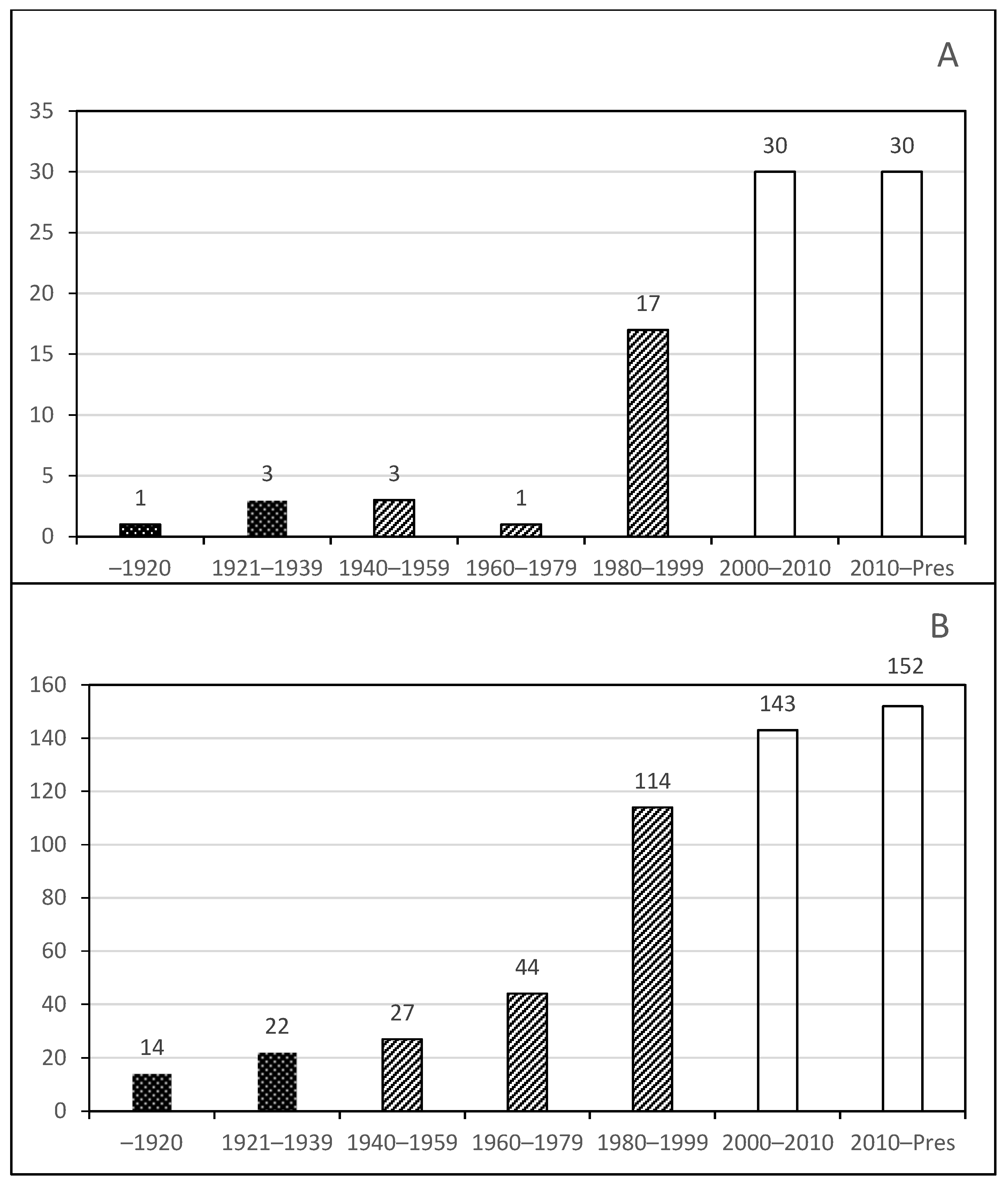
 Phase 1. Early studies by foreign visitors;
Phase 1. Early studies by foreign visitors;  Phase 2. First publications of Mexican researchers alone or with specialists from around the world;
Phase 2. First publications of Mexican researchers alone or with specialists from around the world;  Phase 3. Mature phase and discovery of unknown diversity.
Phase 3. Mature phase and discovery of unknown diversity.
 Phase 1. Early studies by foreign visitors;
Phase 1. Early studies by foreign visitors;  Phase 2. First publications of Mexican researchers alone or with specialists from around the world;
Phase 2. First publications of Mexican researchers alone or with specialists from around the world;  Phase 3. Mature phase and discovery of unknown diversity.
Phase 3. Mature phase and discovery of unknown diversity.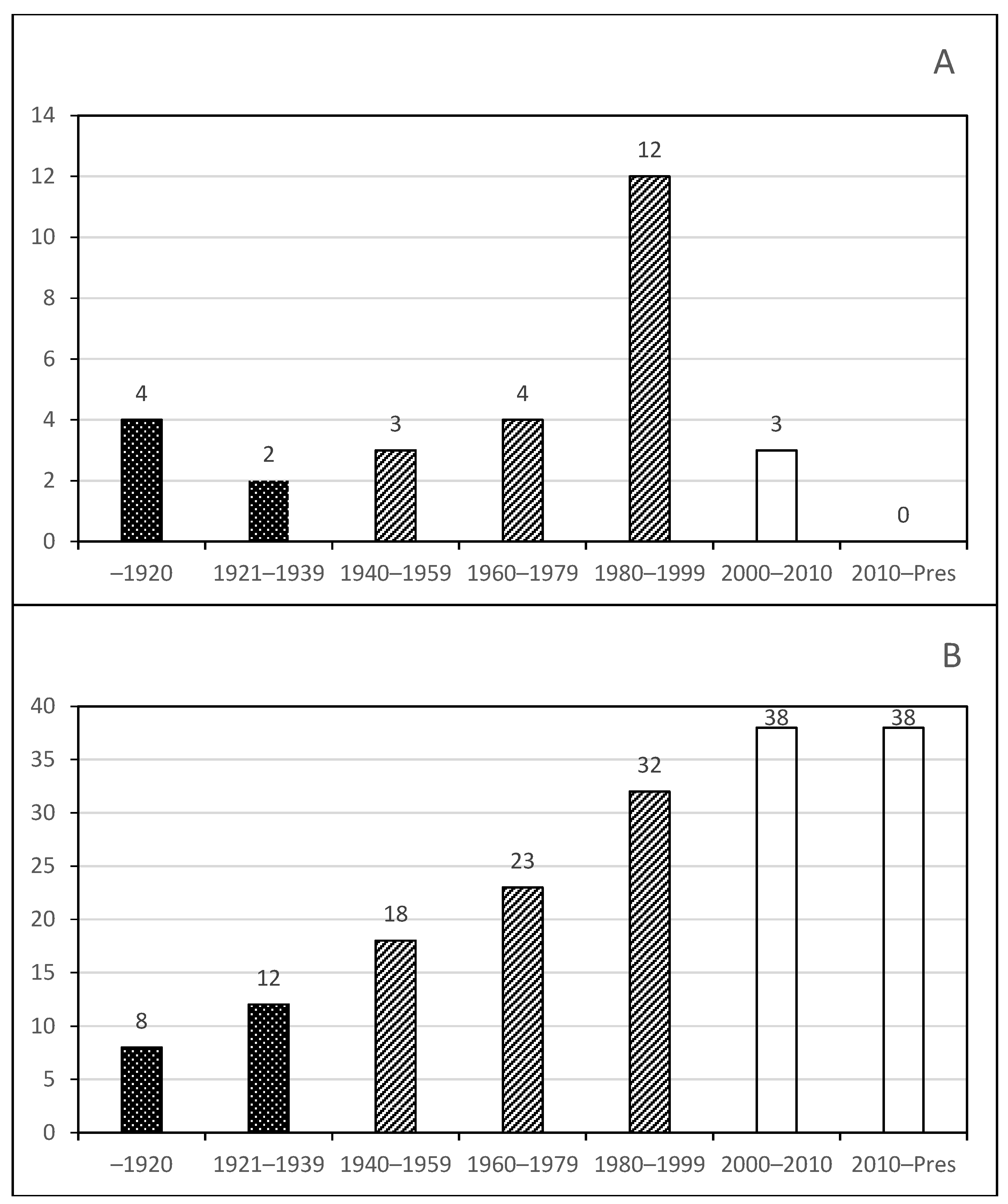
 Phase 1. Early studies by foreign visitors;
Phase 1. Early studies by foreign visitors;  Phase 2. The first generation of Mexican copepodologists, mainly in the last two decades (1980–1999);
Phase 2. The first generation of Mexican copepodologists, mainly in the last two decades (1980–1999);  Phase 3. Mature phase with integrative taxonomy and the discovery of exotic species.
Phase 3. Mature phase with integrative taxonomy and the discovery of exotic species.
 Phase 1. Early studies by foreign visitors;
Phase 1. Early studies by foreign visitors;  Phase 2. The first generation of Mexican copepodologists, mainly in the last two decades (1980–1999);
Phase 2. The first generation of Mexican copepodologists, mainly in the last two decades (1980–1999);  Phase 3. Mature phase with integrative taxonomy and the discovery of exotic species.
Phase 3. Mature phase with integrative taxonomy and the discovery of exotic species.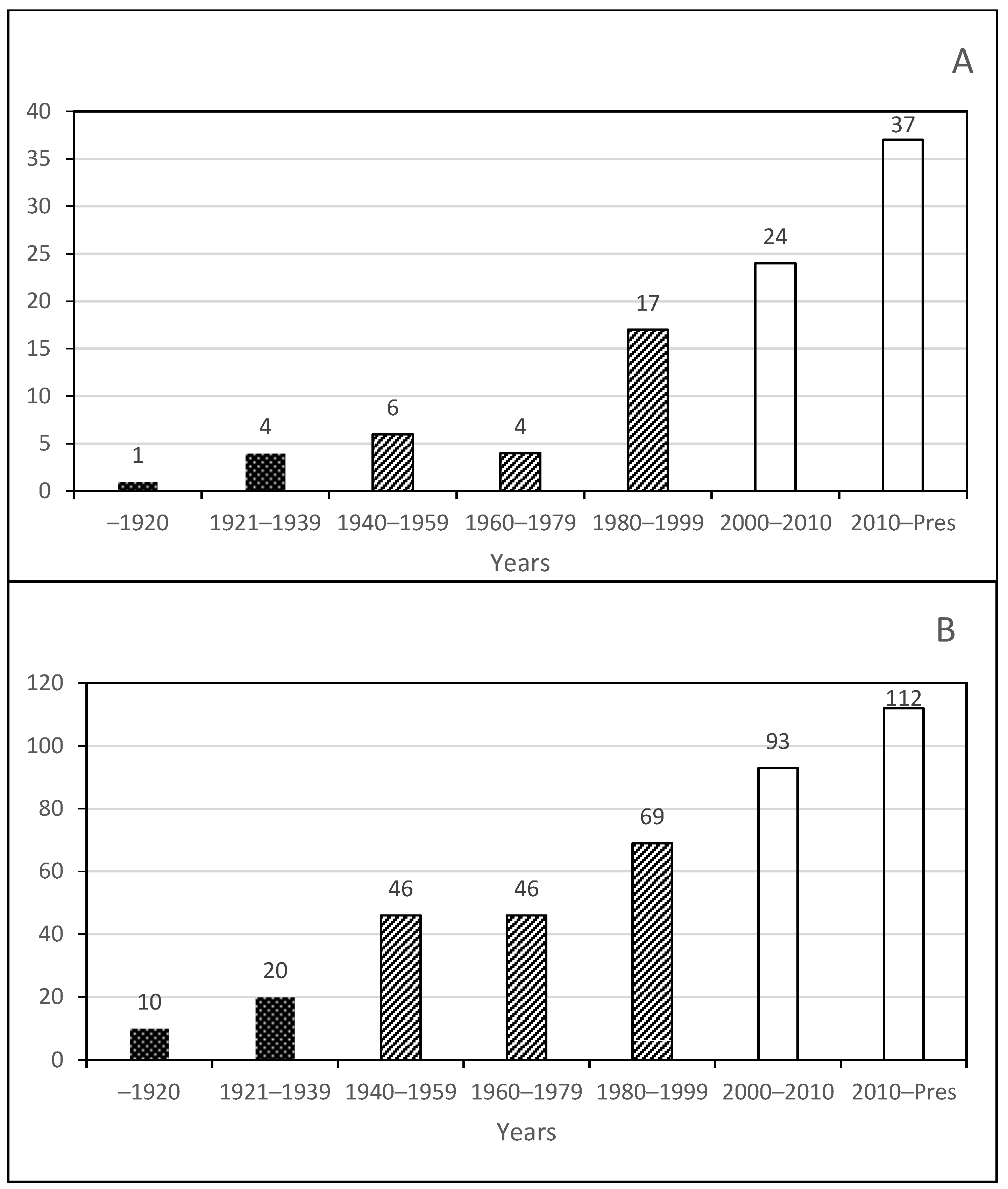
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cervantes-Martínez, A.; Durán Ramírez, C.A.; Elías-Gutiérrez, M.; García-Morales, A.E.; Gutiérrez-Aguirre, M.; Jaime, S.; Macek, M.; Maeda-Martínez, A.M.; Martínez-Jerónimo, F.; Mayén-Estrada, R.; et al. Freshwater Diversity of Zooplankton from Mexico: Historical Review of Some of the Main Groups. Water 2023, 15, 858. https://doi.org/10.3390/w15050858
Cervantes-Martínez A, Durán Ramírez CA, Elías-Gutiérrez M, García-Morales AE, Gutiérrez-Aguirre M, Jaime S, Macek M, Maeda-Martínez AM, Martínez-Jerónimo F, Mayén-Estrada R, et al. Freshwater Diversity of Zooplankton from Mexico: Historical Review of Some of the Main Groups. Water. 2023; 15(5):858. https://doi.org/10.3390/w15050858
Chicago/Turabian StyleCervantes-Martínez, Adrián, Carlos Alberto Durán Ramírez, Manuel Elías-Gutiérrez, Alma E. García-Morales, Martha Gutiérrez-Aguirre, Sarahi Jaime, Miroslav Macek, Alejandro M. Maeda-Martínez, Fernando Martínez-Jerónimo, Rosaura Mayén-Estrada, and et al. 2023. "Freshwater Diversity of Zooplankton from Mexico: Historical Review of Some of the Main Groups" Water 15, no. 5: 858. https://doi.org/10.3390/w15050858







