Accumulation of Heavy Metal Ions from Urban Soil in Spontaneous Flora
Abstract
:1. Introduction
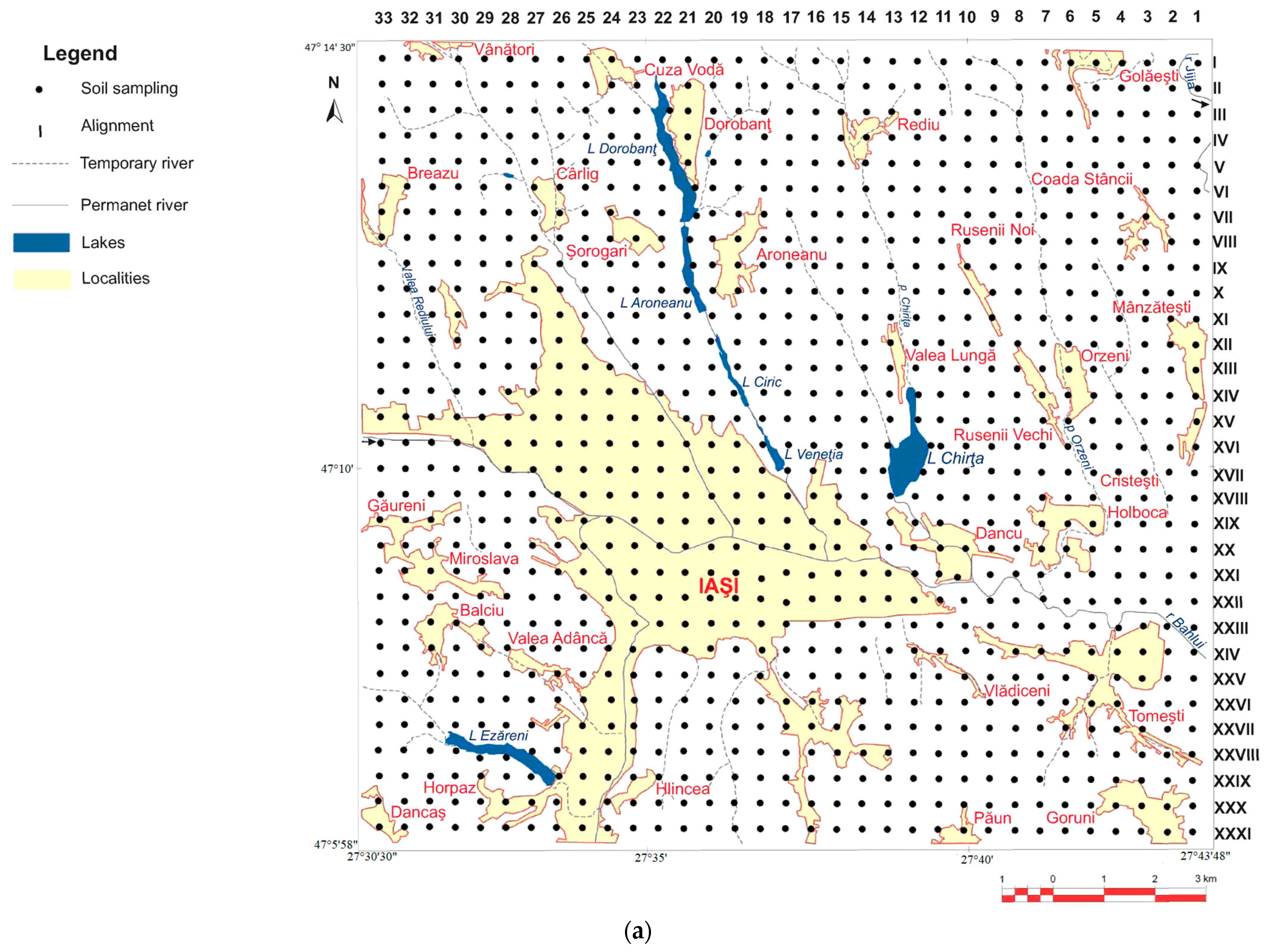
2. Materials and Methods
2.1. Soil Sample Preparation
2.2. Spontaneous Flora Sample Preparation
2.3. Methods for Heavy Metals’ Determination from Aqueous Extract
3. Results and Discussion
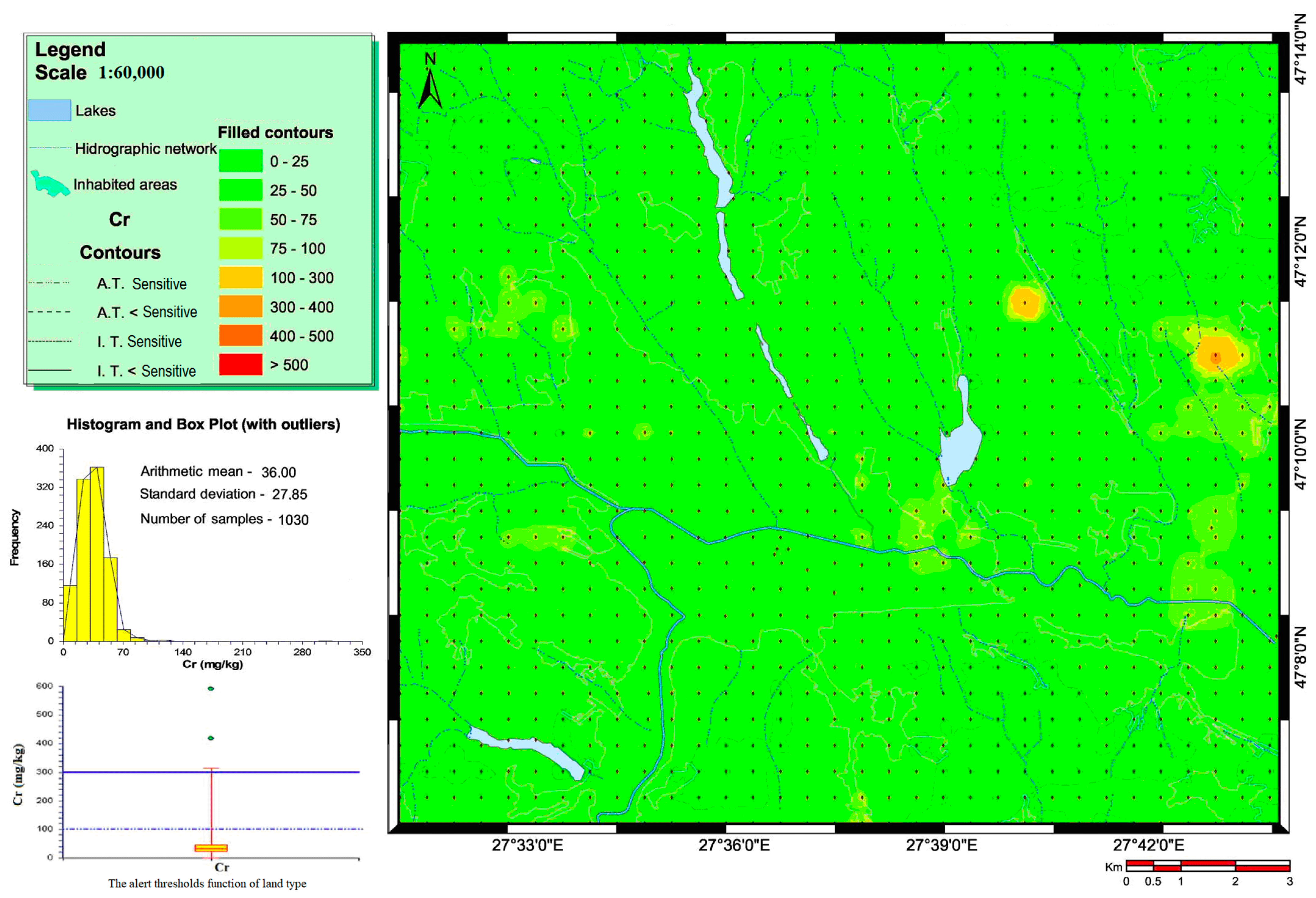
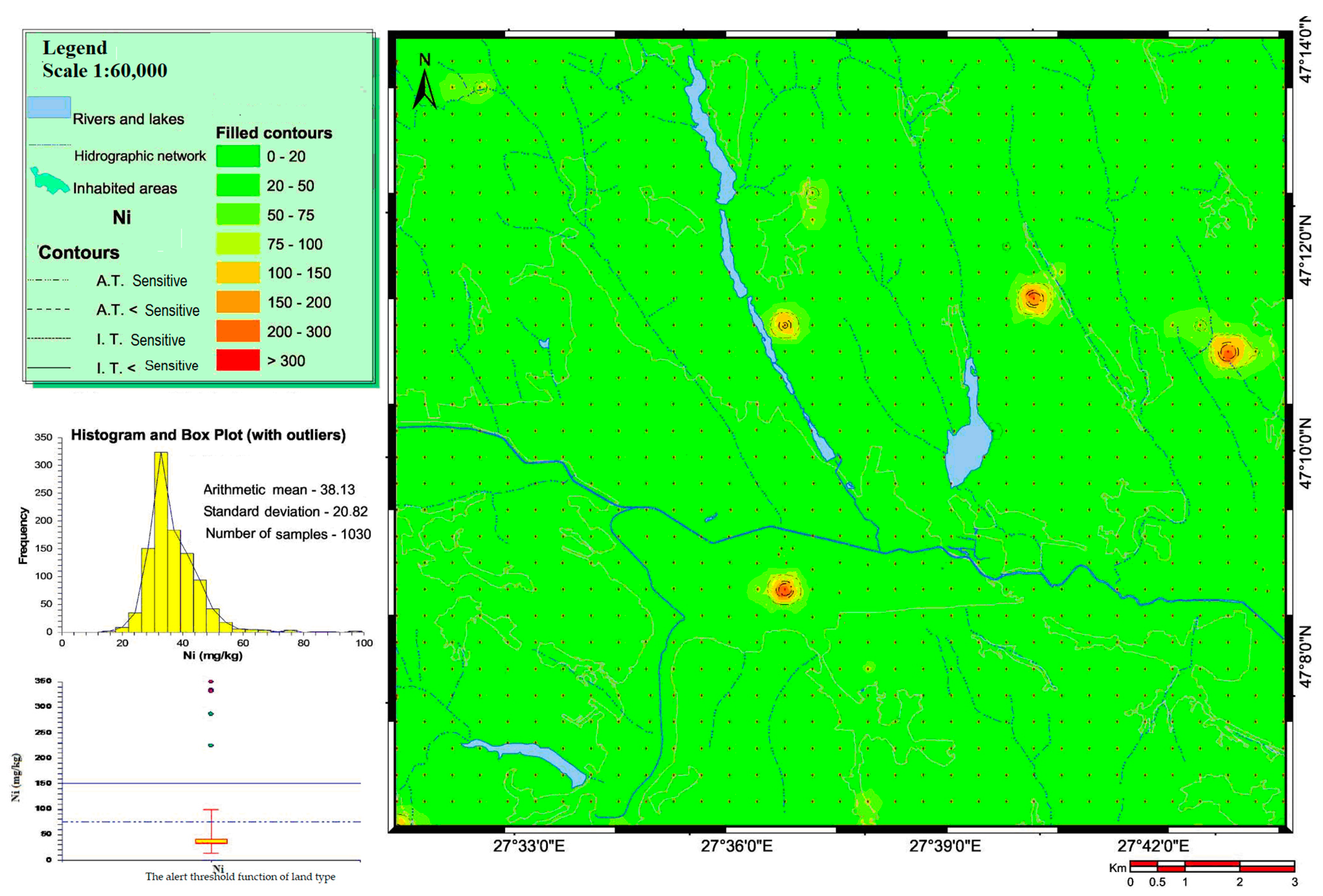
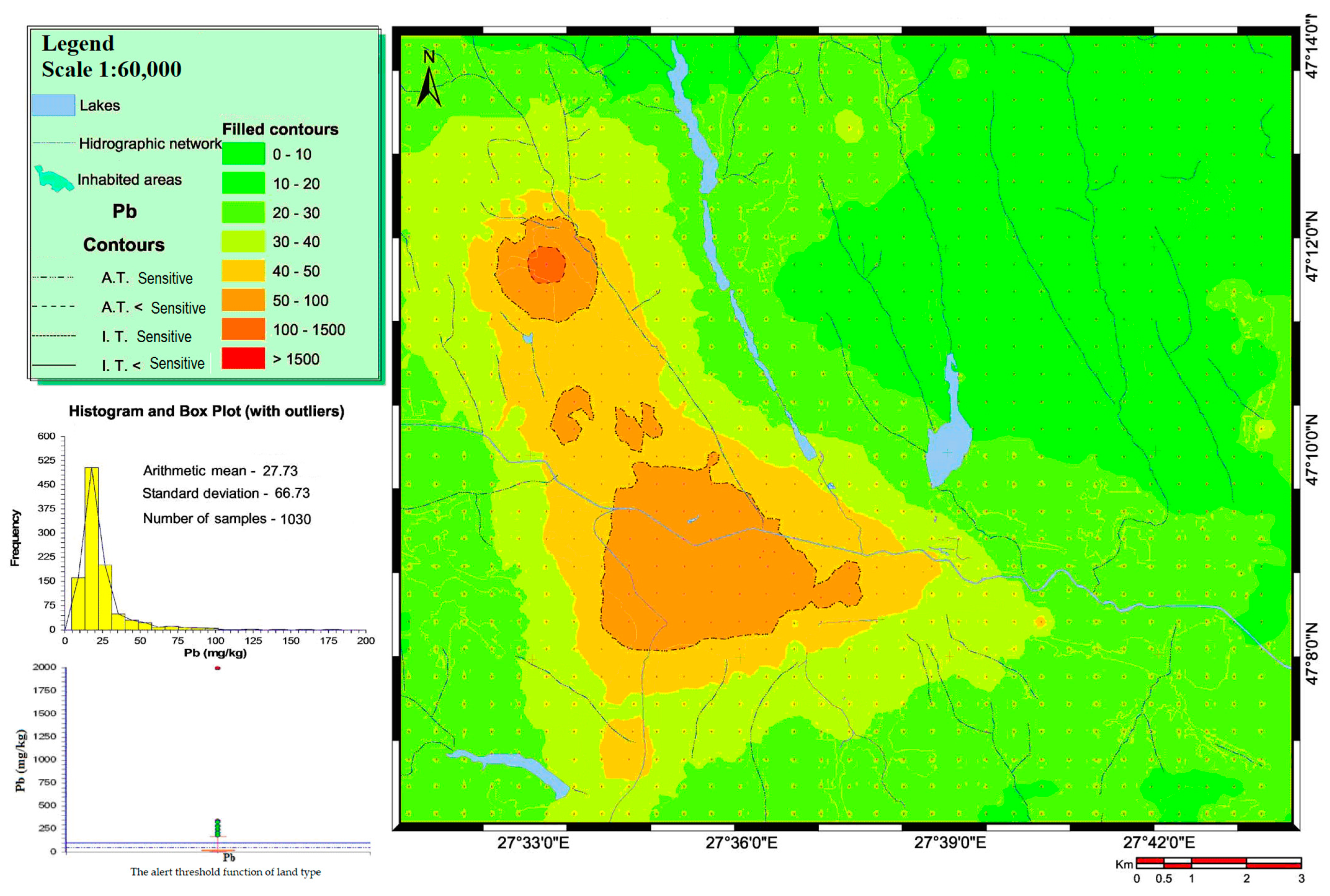
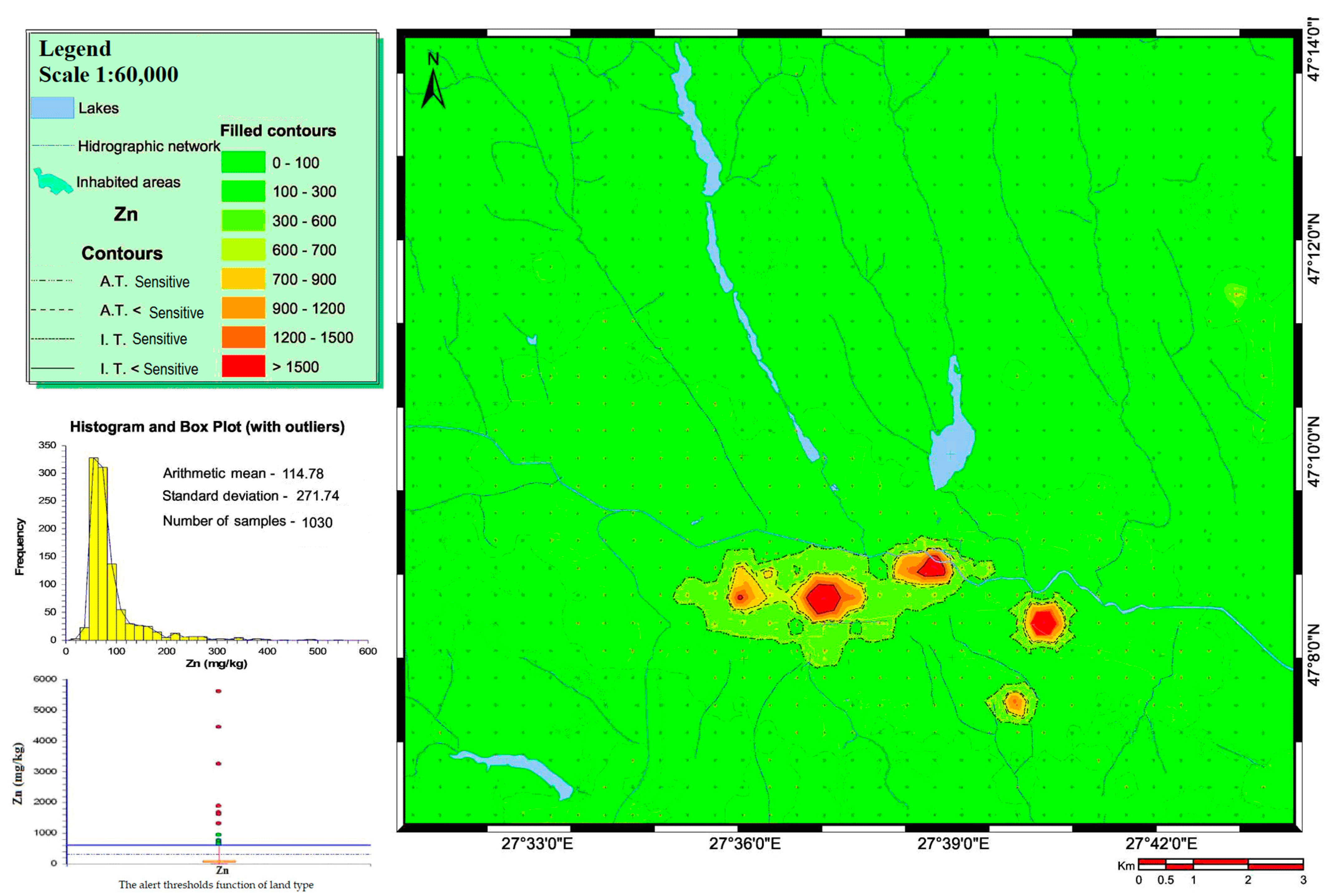
3.1. Heavy Metal Concentrations in Soils
3.2. Concentrations of Heavy Metals in Plants from the Spontaneous Flora
3.3. Concentrations of Heavy Metals in Aqueous Extract
4. Conclusions
- –
- The presence of heavy metals (Fe, Cu, Cr, Mn, Zn) was detected in variable concentrations, with mint and nettle being notable for their increased level of iron and thyme and rattle for an increased level of zinc;
- –
- Potentially toxic metals (Pb, Cd, Ni) are present at low concentrations in plants as well as in infusions, except in primula and plantain, which do not contain Pb and Cd.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wang, Z.; Qin, H.; Wang, J. Accumulation of uranium and heavy metals in the soil–plant system in Xiazhuang uranium ore field, Guangdong Province, China. Environ. Geochem. Health 2019, 41, 2413–2423. [Google Scholar] [CrossRef] [PubMed]
- Adimalla, N. Heavy metals contamination in urban surface soils of Medak province, India, and its risk assessment and spatial distribution. Environ. Geochem. Health 2019, 42, 59–75. [Google Scholar] [CrossRef] [PubMed]
- Vinceković, M.; Šegota, S.; Jurić, S.; Harja, M.; Ondrasek, G. Development and Characterization of a Novel Soil Amendment Based on Biomass Fly Ash Encapsulated in Calcium Alginate Microspheres. Int. J. Mol. Sci. 2022, 23, 9984. [Google Scholar] [CrossRef]
- Harja, M.; Cimpeanu, S.M.; Dirja, M.; Bucur, D. Synthesis of zeolites from fly ash and their use as soil amendment. In Zeolites—Useful Minerals; IntechOpen: London, UK, 2016. [Google Scholar]
- Meng, C.; Wang, P.; Hao, Z.; Gao, Z.; Li, Q.; Gao, H.; Feng, F. Ecological and health risk assessment of heavy metals in soil and Chinese herbal medicines. Environ. Geochem. Health 2022, 44, 817–828. [Google Scholar] [CrossRef]
- Buema, G.; Harja, M.; Lupu, N.; Chiriac, H.; Forminte, L.; Ciobanu, G.; Bucur, R.D. Adsorption performance of modified fly ash for copper ion removal from aqueous solution. Water 2021, 13, 207. [Google Scholar] [CrossRef]
- Harja, M.; Buema, G.; Bucur, D. Recent advances in removal of Congo Red dye by adsorption using an industrial waste. Sci. Rep. 2022, 12, 6087. [Google Scholar] [CrossRef]
- Anaman, R.; Peng, C.; Jiang, Z.; Liu, X.; Zhou, Z.; Guo, Z.; Xiao, X. Identifying sources and transport routes of heavy metals in soil with different land uses around a smelting site by GIS based PCA and PMF. Sci. Total Environ. 2022, 823, 153759. [Google Scholar] [CrossRef]
- Haghnazar, H.; Belmont, P.; Johannesson, K.H.; Aghayani, E.; Mehraein, M. Human-induced pollution and toxicity of river sediment by potentially toxic elements (PTEs) and accumulation in a paddy soil-rice system: A comprehensive watershed-scale assessment. Chemosphere 2023, 311, 136842. [Google Scholar] [CrossRef]
- Wu, J.; Li, J.; Teng, Y.; Chen, H.; Wang, Y. A partition computing-based positive matrix factorization (PC-PMF) approach for the source apportionment of agricultural soil heavy metal contents and associated health risks. J. Hazard. Mater. 2020, 388, 121766. [Google Scholar] [CrossRef]
- Bisht, V.K.; Uniyal, R.C.; Sharma, S.M. Assessment of heavy metals content in herbal raw materials traded in India. S. Afr. J. Bot. 2022, 148, 154–161. [Google Scholar] [CrossRef]
- Harja, M.; Bucur, D.; Cimpeanu, S.M.; Ciocinta, R.C. Conversion of ash on zeolites for soil application. J. Food Agric. Environ. 2012, 10, 1056–1059. [Google Scholar]
- Jeong, H.I.; Han, J.E.; Shin, B.C.; Jang, S.B.; Won, J.H.; Kim, K.H.; Sung, S.H. Monitoring of Heavy Metals and Pesticide Residues of Herbal Decoctions in Traditional Korean Medicine Clinics in Korea. Int. J. Environ. Res. Public Health 2022, 19, 8523. [Google Scholar] [CrossRef] [PubMed]
- Savić, R.; Maksimović, L.; Cimpeanu, S.; Bucur, D.; Ondrašek, G.; Vasin, J.; Harja, M.; Milić, S. Hazardous and harmful substances in sediments of the Jegricka stream. J. Food Agric. Environ. 2013, 11, 1152–1156. [Google Scholar]
- Esfandbod, M.; Forghani, A.; Adhami, E.; Rezaei Rashti, M. The Role of CEC and pH in Cd Retention from Soils of North of Iran. Soil Sediment Contam. 2011, 20, 908–920. [Google Scholar] [CrossRef] [Green Version]
- Harja, M.; Rusu, L.; Bucur, D.; Ciocinta, R.C. Fly ash-derived zeolites as adsorbents for Ni removal from wastewater. Rev. Roum. Chim. 2012, 56, 84–90. [Google Scholar]
- Zhou, J.M.; Dang, Z.; Cai, M.F.; Liu, C.Q. Soil heavy metal pollution around the Dabaoshan Mine, Guangdong Province, China. Pedosphere 2007, 17, 588–594. [Google Scholar] [CrossRef]
- Sur, I.M.; Micle, V.; Polyak, E.T.; Gabor, T. Assessment of soil quality status and the ecological risk in the Baia Mare, Romania area. Sustainability 2022, 14, 3739. [Google Scholar] [CrossRef]
- Iosob, G.A.; Bute, A.; Calara, M.; Tremurici, A.A.; Benchea, C.M.; Bouruc, D.; Muscalu, S.P. Heavy metals contamination of soil and vegetables in three regions from Romania: A review. Sci. Study Res. Biol. 2020, 29, 79–87. [Google Scholar]
- Hoaghia, M.A.; Cadar, O.; Moisa, C.; Roman, C.; Kovacs, E. Heavy metals and health risk assessment in vegetables grown in the vicinity of a former non-metallic facility located in Romania. Environ. Sci. Pollut. Res. 2022, 29, 40079–40093. [Google Scholar] [CrossRef]
- Oprea, B.Ș.; Motelică, D.-M.; Vrînceanu, N.O.; Costea, M.; Plopeanu, G.I.; Carabulea, V. Research on the heavy metal content in onion bulbs correlated with soil from private households located in the Copşa Mică area, Central Romania. J. Appl. Life Sci. Environ. 2022, 55, 92–99. [Google Scholar] [CrossRef]
- Carabulea, V.; Motelică, D.-M.; Vrînceanu, N.O.; Costea, M.; Plopeanu, G.I.; Oprea, B.Ș. Bioaccumulation of heavy metals in garlic bulbs (Allium sativum L.) in correlation with soil from private household in the Copșa Mică area, Romania. J. Appl. Life Sci. Environ. 2022, 55, 245–255. [Google Scholar] [CrossRef]
- Secu, C.V.; Juravle, D.T.; Paiu, M.; Lesenciuc, D.C.; Costica, M.; Negru, D.; Breaban, I.G. Potentially toxic elements in urban soils of Iasi (Romania). Environ. Eng. Manag. J. 2016, 15, 687–698. [Google Scholar]
- Onache, P.A.; Ionete, R.E.; Iordache, A.M.; Florea, A.; Sumedrea, D.I.; Iliescu, M.; Tanase, A. Monitoring the Content of Heavy Metals in The Soil and Wines from Different Wine-Growing Areas of Romania. Int. Multidiscip. Res. Anal. 2022, 5, 343–354. [Google Scholar]
- Chhotu, J.; Fulekar, M. Phytoremediation of heavy metals: Recent techniques. Afr. J. Biotechnol. 2009, 8, 921–928. [Google Scholar]
- Grispen, V.M.J.; Nelissen, H.J.M.; Verkleij, J.A.C. Phytoextraction with Brassica napus L.: A tool for sustainable management of heavy metal contaminated soils. Environ. Pollut. 2006, 144, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.B.; Guo, P.T.; Wu, W.; Wang, Z.Y. Assessment of Soil Arsenic, Chromium, Mercury, and Lead at an Agricultural Landscape Scale. Soil Sediment Contam. 2011, 20, 995–1007. [Google Scholar] [CrossRef]
- Nadgorska-Socha, A.; Ptasinski, B.; Kita, A. Heavy metal bioaccumulation and antioxidative responses in Cardaminopsis arenosa and Plantago lanceolata leaves from metalliferous and non-metalliferous sites: A field study. Ecotoxicology 2013, 22, 1422–1434. [Google Scholar] [CrossRef]
- Schmidt, W. Biodiversity and plant productivity in a grassland succession: Effects of nutrient levels and disturbance regimes. Pol. Bot. Stud. 2006, 22, 437–448. [Google Scholar]
- Wang, G.; Yan, X.; Zhang, F.; Zeng, C.; Gao, D. Traffic-Related Trace Element Accumulation in Roadside Soils and Wild Grasses in the Qinghai-Tibet Plateau, China. Int. J. Environ. Res. Public Health 2013, 11, 456–472. [Google Scholar] [CrossRef] [Green Version]
- Yan, X.; Zhang, F.; Zeng, C.; Zhang, M.; Prasad Devkota, L.; Yao, T. Relationship between Heavy Metal Concentrations in Soils and Grasses of Roadside Farmland in Nepal. Int. J. Environ. Res. Public Health 2012, 9, 3029–3226. [Google Scholar] [CrossRef]
- Sarim, M.; Jan, T.; Khattak, S.A.; Mihoub, A.; Jamal, A.; Saeed, M.F.; Radicetti, E. Assessment of the Ecological and Health Risks of Potentially Toxic Metals in Agricultural Soils from the Drosh-Shishi Valley, Pakistan. Land 2022, 11, 1663. [Google Scholar] [CrossRef]
- Zhang, K.; Chai, F.; Zheng, Z.; Yang, Q.; Zhong, X.; Fomba, K.W. Size distribution and source of heavy metals in particulate matter on the lead and zinc smelting affected area. J. Environ. Sci. 2018, 71, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Du, Q.; Guan, Q.; Luo, H.; Shan, Y.; Shao, W. A Monte Carlo simulation-based health risk assessment of heavy metals in soils of an oasis agricultural region in northwest China. Sci. Total Environ. 2023, 857, 159543. [Google Scholar] [CrossRef] [PubMed]
- Jurowski, K.; Krośniak, M. The Toxicological Assessment of Content and Exposure of Heavy Metals (Pb and Cd) in Traditional Herbal Medicinal Products with Marshmallow Root (Althaea officinalis L. radix) from Polish Pharmacies. Toxics 2022, 10, 188. [Google Scholar] [CrossRef]
- Wu, L.; Yue, W.; Wu, J.; Cao, C.; Liu, H.; Teng, Y. Metal-mining-induced sediment pollution presents a potential ecological risk and threat to human health across China: A meta-analysis. J. Environ. Manag. 2023, 329, 117058. [Google Scholar] [CrossRef]
- Xue, S.; Korna, R.; Fan, J.; Ke, W.; Lou, W.; Wang, J.; Zhu, F. Spatial distribution, environmental risks, and sources of potentially toxic elements in soils from a typical abandoned antimony smelting site. J. Environ. Sci. 2023, 127, 780–790. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhao, X.; Huang, X.; Guo, J.; Chen, G. Identifying a Period of Spatial Land Use Conflicts and Their Driving Forces in the Pearl River Delta. Sustainability 2023, 15, 392. [Google Scholar] [CrossRef]
- Safarova, V.I.; Shaidullina, G.F.; Mikheeva, T.N.; Kudasheva, F.K.; Nizamutdinova, N.R. Methods of sample preparation of soil, bottom sediments, and solid wastes for atomic absorption determination of heavy metals. Inorg. Mater. 2011, 47, 1512–1517. [Google Scholar] [CrossRef]
- Sadee, B.A.; Ali, R.J. Determination of heavy metals in edible vegetables and a human health risk assessment. Environ. Nanotechnol. Monit. Manag. 2023, 19, 100761. [Google Scholar]
- Sevik, H.; Ozel, H.B.; Cetin, M.; Özel, H.U.; Erdem, T. Determination of changes in heavy metal accumulation depending on plant species, plant organism, and traffic density in some landscape plants. Air Qual. Atmos. Health 2019, 12, 189–195. [Google Scholar] [CrossRef]
- Casasanta, G.; Falcini, F.; Garra, R. Beer–Lambert law in photochemistry: A new approach. J. Photochem. Photobiol. A 2022, 432, 114086. [Google Scholar] [CrossRef]
- Santos-Francés, F.; Martínez-Graña, A.; Ávila Zarza, C.; García Sánchez, A.; Alonso Rojo, P. Spatial distribution of heavy metals and the environmental quality of soil in the Northern Plateau of Spain by geostatistical methods. Int. J. Environ. Res. Public Health 2017, 14, 568. [Google Scholar] [CrossRef] [PubMed]
- Agyeman, P.C.; Kebonye, N.M.; John, K.; Haghnazar, H.; Borůvka, L.; Vašát, R. Compositional mapping, uncertainty assessment, and source apportionment via pollution assessment-based receptor models in urban and peri-urban agricultural soils. J. Soils Sediments 2022, 1–22. [Google Scholar] [CrossRef]
- Petrean, I.A.; Micle, V.; Sur, I.M.; Șenilă, M. Characterization of Sterile Mining Dumps by the ICP-OES Analytical Method: A Case Study from Baia Mare Mining Area (Maramures, Romania). Sustainability 2023, 15, 1158. [Google Scholar] [CrossRef]
- Commission Regulation (EU) 2021/1323 of 10 August 2021 Amending Regulation (EC) No 1881/2006 as Regards Maximum Levels of Cadmium in Certain Foodstuff. Official Journal of the European. 11.08.2021 L228/13. Available online: http://data.europa.eu/eli/reg/2021/1323/oj (accessed on 10 December 2022).
- Shi, S.; Hou, M.; Gu, Z.; Jiang, C.; Zhang, W.; Hou, M.; Xi, Z. Estimation of Heavy Metal Content in Soil Based on Machine Learning Models. Land 2022, 11, 1037. [Google Scholar] [CrossRef]
- Cabrini, T.M.; Barboza, C.A.; Skinner, V.B.; Hauser-Davis, R.A.; Rocha, R.C.; Saint’ Pierre, T.D.; Cardoso, R.S. Investigating heavy metal bioaccumulation by macrofauna species from different feeding guilds from sandy beaches in Rio de Janeiro, Brazil. Ecotoxicol. Environ. Saf. 2018, 162, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Jiang, X.; Wang, Y.; Zhuang, D. Spatial characteristics of heavy metal pollution and the potential ecological risk of a typical mining area: A case study in China. Process Saf. Environ. Prot. 2018, 113, 204–219. [Google Scholar] [CrossRef]
- Mortensen, L.H.; Rønn, R.; Vestergård, M. Bioaccumulation of cadmium in soil organisms–With focus on wood ash application. Ecotoxicol. Environ. Saf. 2018, 156, 452–462. [Google Scholar] [CrossRef]
- Wang, K.; Qiao, Y.; Zhang, H.; Yue, S.; Li, H.; Ji, X.; Liu, L. Bioaccumulation of heavy metals in earthworms from field contaminated soil in a subtropical area of China. Ecotoxicol. Environ. Saf. 2018, 148, 876–883. [Google Scholar] [CrossRef]
- Shi, H.; Wang, P.; Zheng, J.; Deng, Y.; Zhuang, C.; Huang, F.; Xiao, R. A comprehensive framework for identifying contributing factors of soil trace metal pollution using Geodetector and spatial bivariate analysis. Sci. Total Environ. 2023, 857, 159636. [Google Scholar] [CrossRef]
- Tang, Q.; Zhang, H.; Zhao, X.; Zheng, L.; Miao, C.; Liu, Y.; Fu, B. Chromium in Chinese coals: Geochemistry and environmental impacts associated with coal-fired power plants. Environ. Geochem. Health 2022, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Osei, L.B.; Fosu, S.; Ndur, S.A.; Nyarko, S.Y. Assessing heavy metal contamination and ecological risk of urban topsoils in Tarkwa, Ghana. Environ. Monit. Assess. 2022, 194, 710. [Google Scholar] [CrossRef] [PubMed]
- Adhami, E.; Salmanpour, A.; Omidi, A.; Khosravi, N.; Ghasemi-Fasaei, R.; Maftoun, M. Nickel adsorption characteristics of selected soils as related to some soil properties. Soil Sediment Contam. 2008, 17, 643–653. [Google Scholar] [CrossRef]
- Yang, X.; Wen, E.; Ge, C.; El-Naggar, A.; Yu, H.; Wang, S.; Rinklebe, J. Iron-modified phosphorus-and silicon-based biochars exhibited various influences on arsenic, cadmium, and lead accumulation in rice and enzyme activities in a paddy soil. J. Hazard. Mater. 2023, 443, 130203. [Google Scholar] [CrossRef]
- Xu, J.; Li, Y.; Wang, S.; Long, S.; Wu, Y.; Chen, Z. Sources, transfers and the fate of heavy metals in soil-wheat systems: The case of lead (Pb)/zinc (Zn) smelting region. J. Hazard. Mater. 2023, 441, 129863. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, S.K. Medicinal herbs: A potential source of toxic metal exposure for man and animals in India. Arch. Environ. Health 2002, 57, 229–231. [Google Scholar] [CrossRef]
- Kumar, P.B.A.N.; Dushenkov, V.; Motto, H.; Raskin, I. Phytoextraction: The use of plants to remove heavy metals from soils. Environ. Sci. Technol. 1995, 29, 1232–1238. [Google Scholar] [CrossRef]
- Mrak, T.; Grašič, B.; Prislan, P.; Gričar, J.; Laznik, Ž.; Voglar, G.E. Soil contamination with potentially toxic elements and root herbivory: Effects on root surface area and stem secondary xylem of young common beech (Fagus sylvatica L.). Acta Physiol. Plant. 2023, 45, 23. [Google Scholar] [CrossRef]
- Gao, L.; Di, D.; Liu, X.; Teng, F. Comparative Study of Heavy Metals Analysis in Mongolian Medicines Based on High Sensitivity X-ray Fluorescence Spectroscopy and ICP-MS. Spectroscopy 2022, 37, 20–27. [Google Scholar] [CrossRef]



| Heavy Metals Ions | Iron | Chromium | Copper | Manganese | Zinc |
|---|---|---|---|---|---|
| Folium urticae | 49.62 ± 3.82 | 2.19 ± 0.13 | 6.74 ± 0.45 | 14.98 ± 0.49 | 30.40 ± 4.12 |
| Folium taraxaci | 31.11 ± 1.41 | 5.21 ± 0.42 | 2.78 ± 0.11 | 14.33 ± 0.34 | 49.35 ± 2.01 |
| Herba cichorii | 22.08 ± 1.63 | 10.44 ± 0.80 | 2.09 ± 0.24 | 20.54 ± 1.78 | 967.61 ± 6.28 |
| Herba hyperici | 10.23 ± 1.27 | 1.84 ± 0.82 | 3.31 ± 0.61 | 55.66 ± 5.15 | 811.09 ± 9.69 |
| Folium menthae | 80.52 ± 5.06 | 2.67 ± 0.84 | 4.15 ± 0.31 | 25.67 ± 0.96 | 309.63 ± 45.1 |
| Min | 8.96 | 1.02 | 1.85 | 13.99 | 26.28 |
| Max | 85.58 | 11.25 | 4.46 | 60.81 | 973.28 |
| Mean | 38.712 | 4.47 | 3.814 | 26.236 | 433.616 |
| Standard deviation | 27.44327 | 3.5897 | 1.80067 | 17.0806 | 26.35 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harja, M.; Ciocinta, R.C.; Ondrasek, G.; Bucur, D.; Dirja, M. Accumulation of Heavy Metal Ions from Urban Soil in Spontaneous Flora. Water 2023, 15, 768. https://doi.org/10.3390/w15040768
Harja M, Ciocinta RC, Ondrasek G, Bucur D, Dirja M. Accumulation of Heavy Metal Ions from Urban Soil in Spontaneous Flora. Water. 2023; 15(4):768. https://doi.org/10.3390/w15040768
Chicago/Turabian StyleHarja, Maria, Ramona Carla Ciocinta, Gabrijel Ondrasek, Daniel Bucur, and Marcel Dirja. 2023. "Accumulation of Heavy Metal Ions from Urban Soil in Spontaneous Flora" Water 15, no. 4: 768. https://doi.org/10.3390/w15040768








