Benthic Macroinvertebrate Communities in Wadeable Rivers and Streams of Lao PDR as a Useful Tool for Biomonitoring Water Quality: A Multimetric Index Approach
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Measurement of Environmental Variables
2.3. Benthic Macroinvertebrate Sampling
2.4. Site Classification
2.5. Data Analysis
2.5.1. Habitat Score, Environmental Variables and Benthic Macroinvertebrate Composition
2.5.2. MMI Development
3. Results
3.1. Site Classification and Environmental Characteristics
3.2. Structure of Benthic Macroinvertebrate Community
3.3. Metric Sensitivity and Selection
3.4. Development of Lao MMI
3.5. Validation of the Multimetric Index
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- The World Factbook. Available online: https://www.cia.gov/the-world-factbook/ (accessed on 19 November 2022).
- Japan International Cooperation Agency. Profile on Environmental and Social Considerations in Lao P.D.R.; Japan International Cooperation Agency: Tokyo, Japan, 2013; pp. 1–333. [Google Scholar]
- MoNRE; IUCN. Fifth National Report to the United Nations Convention on Biological Diversity; Department of Forest and Natural Resources Management, Ministry of Natural Resource and Environment: Vientiane, Laos, 2016; pp. 1–91. [Google Scholar]
- Singkham, P. An Introduction to the Fisheries of Lao PDR; Mekong River Commission: Phnom Penh, Cambodia, 2013; pp. 1–56. [Google Scholar]
- UNDP. Available online: https://www.la.undp.org/ (accessed on 7 January 2023).
- Soutullo, J. The Mekong River: Geopolitics over Development, Hydropower and the Environment; European Union: Brussels, Belgium, 2019; pp. 1–55. [Google Scholar]
- Mekong River Commission Lao National Mekong Committee. Annual Report on Water Quality Data Assessment Lao PDR; Water Quality Monitoring Network in Mekong River Basin, Natural Resources and Environment Institute, Ministry of Natural Resources and Environment: Vientiane, Laos, 2016; pp. 1–46. [Google Scholar]
- Chea, R.; Grenouillet, G.; Lek, S. Evidence of Water Quality Degradation in Lower Mekong Basin Revealed by Self-Organizing Map. PLoS ONE 2016, 11, e0145527. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Kumar, R.; Satapathy, S.C.; Al-Ansari, N.; Singh, K.K.; Mahapatra, R.P.; Agarwal, A.K.; Le, H.V.; Pham, B.T. Analysis of Water Pollution Using Different Physicochemical Parameters: A Study of Yamuna River. Front. Environ. Sci. 2020, 8, 581591. [Google Scholar] [CrossRef]
- Braga, F.H.R.; Dutra, M.L.S.; Lima, N.S.; Silva, G.M.; Miranda, R.C.M.; Firmo, W.C.A.; Moura, A.R.L.; Monteiro, A.S.; Silva, L.C.N.; Silva, D.F.; et al. Study of the Influence of Physicochemical Parameters on the Water Quality Index (WQI) in the Maranhão Amazon, Brazil. Water 2022, 14, 1546. [Google Scholar] [CrossRef]
- Tampo, L.; Kaboré, I.; Alhassan, E.H.; Ouéda, A.; Bawa, L.M.; Djaneye-Boundjou, G. Benthic Macroinvertebrates as Ecological Indicators: Their Sensitivity to the Water Quality and Human Disturbances in a Tropical River. Front. Water 2021, 3, 662765. [Google Scholar] [CrossRef]
- Abdelkarim, M.S. Biomonitoring and bioassessment of running water quality in developing countries: A case study from Egypt. Egypt. J. Aquat. Res. 2020, 46, 313–324. [Google Scholar] [CrossRef]
- Mezgebu, A. A review on freshwater biomonitoring with benthic invertebrates in Ethiopia. Environ. Sustain. Indic. 2022, 14, 100174. [Google Scholar] [CrossRef]
- Morse, J.C.; Bae, Y.J.; Munkhjargal, G.; Sangpradub, N.; Tanida, K.; Vshivkova, T.S.; Wang, B.; Yang, L.; Yule, C.M. Freshwater Biomonitoring with Macroinvertebrates in East Asia. Front. Ecol. Environ. 2007, 5, 33–42. [Google Scholar] [CrossRef]
- Barbour, M.T.; Gerritsen, J.; Snyder, B.D.; Stribling, J.B. Rapid Bioassessment Protocols for Use in Streams and Wadeable Rivers: Periphyton, Benthic Macroinvertebrates and Fish, 2nd ed.; U.S. Environmental Protection Agency, Office of Water: Washington, DC, USA, 1999; pp. 1–339.
- Karr, J.R.; Allan, J.D.; Benke, A.C. River Conservation in the United States and Canada. Global Perspectives on River Conservation: Science, Policy, Practice; J. Wiley: Chichester, UK, 2000; pp. 3–39. [Google Scholar]
- Nguyen, H.H.; Everaert, G.; Gabriels, W.; Hoang, T.H.; Goethals, P.L.M. A multimetric macroinvertebrate index for assessing the waterquality of the Cau river basin in Vietnam. Limnologica 2014, 45, 16–23. [Google Scholar] [CrossRef]
- Boonsoong, B.; Sangpradub, N.; Barbour, M.T. Development of rapid bioassessment approaches using benthic macroinvertebrates for Thai streams. Environ. Monit. Assess. 2009, 155, 129–147. [Google Scholar] [CrossRef]
- Lakew, A.; Moog, O. A multimetric index based on benthic macroinvertebrates for assessing the ecological status of streams and rivers in central and southeast highlands of Ethiopia. Hydrobiologia 2015, 751, 229–242. [Google Scholar] [CrossRef]
- Rosenberg, D.M.; Resh, V.H. (Eds.) Introduction to freshwater biomonitoring and benthic macroinvertebrates. In Freshwater Biomonitoring and Benthic Macroinvertebrate; Chapmam and Hall: New York, NY, USA, 1993; pp. 1–10. [Google Scholar]
- Koudenoukpo, C.Z.; Chikou, A.; Toko, I.I.; Togouet, S.H.Z.; Tchakonté, S.; Hazoume, R.; Piscart, C. Diversity of aquatic macroinvertebrates in relationship with the environmental factors of a lotic ecosystem in tropical region: The Sô river in South-East of Benin (West Africa). J. Entomol. Zool. Stud. 2017, 5, 1–10. [Google Scholar]
- Souilmi, F.; Ghedda, K.; Fahde, A.; EL Fihri, F.Z.; Tahraoui, S.; Elasri, F.; Malki, M. Taxonomic diversity of benthic macroinvertebrates along the Oum Er Rbia River (Morocco): Implications for water quality bio-monitoring using indicator species. West Afr. J. Appl. Ecol. 2019, 27, 137–149. [Google Scholar]
- Brysiewicz, A.; Czerniejewski, P.; Dabrowski, J.; Formicki, K. Characterisation of Benthic Macroinvertebrate Communities in Small Watercourses of the European Central Plains Ecoregion and the Effect of Different Environmental Factors. Animals 2022, 12, 606. [Google Scholar] [CrossRef] [PubMed]
- Sripanya, J.; Rattanawilai, K.; Vongsombath, C.; Vannachak, V.; Hanjavanit, C.; Sangpradub, N. Benthic Macroinvertebrates and Trichoptera Adults for Bioassessment Approach in Streams and Wadeable Riversin Lao People’s Democratic Republic. Trop. Nat. Hist. 2022, 22, 12–24. [Google Scholar]
- Souto, R.M.G.; Facure, K.G.; Pavanin, L.A.; Jacobucci, G.B. Influence of environmental factors on benthic macroinvertebrate communities of urban streams in Vereda habitats, Central Brazil. Acta Limnol. Bras. 2011, 23, 293–306. [Google Scholar] [CrossRef]
- Leitner, P.; Hauer, C.; Ofenböck, T.; Pletterbauer, F.; Schmidt-Kloiber, A.; Graf, W. Fine sediment deposition affects biodiversity and density of benthic macroinvertebrates: A case study in the freshwater pearl mussel river Waldaist (Upper Austria). Limnologica 2015, 50, 54–57. [Google Scholar] [CrossRef]
- Pacioglu, O.; Duţu, F.; Pavel, A.B.; Duţu, L.T. The influence of hydrology and sediment grain-size on the spatial distribution of macroinvertebrate communities in two submerged dunes from the Danube Delta (Romania). Limnetica 2022, 41, 85–100. [Google Scholar] [CrossRef]
- Rezende, R.S.; Santos, A.M.; Henke-Oliveira, C.; Gonçalves, J.F., Jr. Effects of spatial and environmental factors on benthic a macroinvertebrate community. Zoologia 2014, 31, 426–434. [Google Scholar] [CrossRef]
- Zweig, L.D.; Rabeni, C.F. Biomonitoring for deposited sediment using benthic invertebrates: A test on 4 Missouri streams. J. N. Am. Benthol. Soc. 2001, 20, 643–657. [Google Scholar] [CrossRef]
- Shull, D.R.; Smith, Z.M.; Selckmann, G.M. Development of a benthic macroinvertebrate multimetric index for large semiwadeable rivers in the Mid-Atlantic region of the USA. Environ. Monit. Assess. 2019, 191, 22. [Google Scholar] [CrossRef]
- Indicators: Benthic Macroinvertebrates. Available online: https://www.epa.gov/national-aquatic-resource-surveys/indicators-benthic-macroinvertebrates (accessed on 19 November 2022).
- Gabriels, K.; Lock, N.; De Pauw, N.; Goethals, P.L.M. Multimetric macroinvertebrate index Flanders (MMIF) for biological assessment of rivers and lakes in Flanders (Belgium). Limnologica 2010, 40, 199–207. [Google Scholar] [CrossRef]
- Mereta, S.T.; Boets, P.; Meester, L.D.; Goethals, P.L.M. Development of a multimetric index based on benthic macroinvertebrates for the assessment of natural wetlands in Southwest Ethiopia. Ecol. Indic. 2013, 29, 510–521. [Google Scholar] [CrossRef]
- Barbour, M.T.; Gerritsen, J.; Griffith, G.E.; Frydenborg, R.; McCarron, E.; White, J.S.; Bastian, M.L. A framework for biological criteria for Florida streams using benthic macroinvertebrates. J. N. Am. Benthol. Soc. 1996, 15, 185–211. [Google Scholar] [CrossRef]
- Vlek, H.E.; Verdonschot, P.F.M.; Nijboer, R.C. Towards a multimetric index for the assessment of Dutch streams using benthic macroinvertebrates. Hydrobiologia 2004, 516, 173–189. [Google Scholar] [CrossRef]
- Hering, D.; Moog, O.; Sandin, L.; Verdonschot, P.F.M. Overview and application of the AQEM assessment system. Hydrobiologia 2004, 516, 1–20. [Google Scholar] [CrossRef]
- Jun, Y.C.; Won, D.H.; Lee, S.H.; Kong, D.S.; Hwang, S.J. A Multimetric Benthic Macroinvertebrate Index for the Assessment of Stream Biotic Integrity in Korea. Int. J. Environ. Res. Public Health 2012, 9, 3599–3628. [Google Scholar] [CrossRef]
- Karr, J.R. Assessment of Biotic Integrity Using Fish Communities. Fisheries 1981, 6, 21–27. [Google Scholar] [CrossRef]
- Vadas, R.L., Jr.; Hughes, R.M.; Bae, Y.J.; Baek, M.J.; Gonzáles, O.C.B.; Callisto, M.; de Carvalho, D.R.; Chen, K.; Ferreira, M.T.; Fierro, P.; et al. Assemblage-based biomonitoring of freshwater ecosystem health via multimetric indices: A critical review and suggestions for improving their applicability. Water Biol. Secur. 2022, 1, 100054. [Google Scholar] [CrossRef]
- Haase, P.; Lohse, S.; Pauls, S.; Schindehütte, K.; Sundermann, A.; Rolauffs, P.; Hering, D. Assessing streams in Germany with benthic invertebrates: Development of a practical standardised protocol for macroinvertebrate sampling and sorting. Limnologica 2004, 34, 349–365. [Google Scholar] [CrossRef]
- Arman, N.Z.; Salmiati, S.; Said, M.I.M.; Aris, A. Development of macroinvertebrate-based multimetric index and establishment of biocriteria for river health assessment in Malaysia. Ecol. Indic. 2019, 104, 449–458. [Google Scholar] [CrossRef]
- Huang, Q.; Gao, J.; Cai, Y.; Yin, H.; Gao, Y.; Zhao, J.; Liu, L.; Huang, J. Development and application of benthic macroinvertebrate-based multimetric indices for the assessment of streams and rivers in the Taihu Basin, China. Ecol. Indic. 2014, 48, 649–659. [Google Scholar] [CrossRef]
- Liu, L.; Xu, Z.; Yin, X.; Li, F.; Dou, T. Development of a multimetric index based on benthic macroinvertebrates for the assessment of urban stream health in Jinan City, China. Environ. Monit. Assess. 2017, 189, 205. [Google Scholar] [CrossRef] [PubMed]
- Rattanachan, K.; Boonsoong, B.; Getwongsa, P.; Uttaruk, Y.; Sangpradub, N. A benthic macroinvertebrates multimetric index for assessment of the ecological integrity of northeast streams, Thailand. Environ. Asia 2016, 9, 186–194. [Google Scholar]
- Mekong River Commission. 2018 Lower Mekong Water Quality Monitoring Report; Mekong River Commission: Vientiane, Laos, 2021; pp. 1–60. [Google Scholar]
- Brindha, K.; Pavelic, P.; Sotoukee, T. Environmental assessment of water and soil quality in the Vientiane Plain, Lao PDR. Groundw. Sustain. Dev. 2019, 8, 24–30. [Google Scholar] [CrossRef]
- Mekong River Commission. Report on the 2017 Biomonitoring Survey of the Lower Mekong River and Selected Tributaries; Mekong River Commission: Vientiane, Laos, 2019; pp. 1–85. [Google Scholar]
- Tetra Tech. A Stream Condition Index for West Virginia Wadeable Streams; Tetra Tech, Inc.: Baltimore, MD, USA, 2000; pp. 1–80. [Google Scholar]
- APHA. Standard Methods for the Examination of Water and Wastewater, 20th ed.; APHA: Washington, DC, USA, 2001. [Google Scholar]
- Krawczyk, D.; Gonglewski, N. Determining Suspended Solids Using a Spectrophotometer. Sewage. Ind. Waste 1959, 31, 1159–1164. [Google Scholar]
- Sangpradub, N.; Boonsoong, B. Identification of Freshwater Invertebrates of the Mekong River and Tributaries; Mekong River Commission: Vientiane, Laos, 2006; pp. 1–274. [Google Scholar]
- Morse, J.C.; Yang, L.; Tian, L. Aquatic Insects of China Useful for Monitoring Water Quality; Hohai University Press: Nanjing, China, 1994; pp. 1–570. [Google Scholar]
- Wiggins, G.B. Larvae of the North American Caddisfly Genera (Trichoptera); University of Toronto Press: Toronto, ON, Canada, 1996; pp. 1–457. [Google Scholar]
- Boonsoong, B. Rapid Bioassessment of Thai Streams with Benthic Macroinvertebrates. Ph.D. Thesis, Khon Kean University, Khon Kean, Thailand, 2007. [Google Scholar]
- Merritt, R.T.; Cummins, K.W.; Berg, M.B. An Introduction to the Aquatic Insects of North America, 5th ed.; Kendall Hunt Publishing Company: Dubuque, IA, USA, 2008; pp. 1–1158. [Google Scholar]
- Getwongsa, P. Development of Biotic Index for Rapid Bioassessment in Mekong 2 Basin. Ph.D. Thesis, Khon Kean University, Khon Kean, Thailand, 2010. [Google Scholar]
- Uttaruk, P. The Development of Ecological Indicators for Streams: Using Benthic Macroinvertebrates to Rapid Bioassessment for Wadeable Streams in Chi and Mun Headwater, the Northeast Thailand. Ph.D. Thesis, Khon Kean University, Khon Kaen, Thailand, 2011. [Google Scholar]
- Rattanachan, K. Benthic Macroinvertebrate Multimetric Index for Rapid Bioassessment of Streams in Thailand. Ph.D. Thesis, Khon Kean University, Khon Kaen, Thailand, 2017. [Google Scholar]
- Somnark, R. Community Structure of Freshwater Benthic Macroinvertebrates, Phenology of Some Species of Aquatic Insects and Leaf Litter Breakdown in Nam Nao National Park. Ph.D. Thesis, Khon Kean University, Khon Kaen, Thailand, 2018. [Google Scholar]
- Pollution Control Department of Thailand. Water Quality Criteria and Standards in Thailand; Pollution Control Department, Ministry of Natural Resources and Environment: Bangkok, Thailand, 1997. [Google Scholar]
- IBM Corp. IBM SPSS Statistics for Windows. Version 23.0; IBM Corp.: Armonk, NY, USA, 2015. [Google Scholar]
- McCune, B.; Mefford, M.J. PC-ORD. Mutivariate Analysis of Ecological Data. Version 5; MjM Software: Gleneden Beach, OR, USA, 2006. [Google Scholar]
- Hammer, O. PAST—PAlaeontological STatistics, Version 4.03. Reference Manual; Natural History Museum, University of Oslo: Oslo, Norway, 2020. [Google Scholar]
- Tetra Tech. Ecological Data Application System (EDAS): A User’s Manual; Tetra Tech, Inc.: Baltimore, MD, USA, 2000. [Google Scholar]
- Stribling, J.B.; Jessup, B.K.; Gerritsen, J. Development of Biological and Physical Habitat Criteria for Wyoming Streams and Their Use in the TMDL Process; Tetra Tech, Inc.: Baltimore, MD, USA, 2000. [Google Scholar]
- Stoddard, J.L.; Herlihy, A.T.; Peck, D.V.; Hughes, R.M.; Whittier, T.R.; Tarquinio, E. A process for creating multimetric indices for large-scale aquatic surveys. J. N. Am. Benthol. Soc. 2008, 27, 878–891. [Google Scholar] [CrossRef]
- Wu, H.; Lu, K.; Lu, X.; Xue, Z. A Macroinvertebrate Multimetric Index for the Bioassessment of Wetlands Adjacent to Agriculture Fields in the Sanjiang Plain, China. Chin. Geogr. Sci. 2019, 29, 974–984. [Google Scholar] [CrossRef]
- Ferreira, W.R.; Paiva, L.T.; Callisto, M. Development of a benthic multimetric index for biomonitoring of a neotropical watershed. Braz. J. Biol. 2011, 71, 15–25. [Google Scholar] [CrossRef]
- Department of Pollution Control. Standard of National Environment; Ministry of Natural Resources and Environment: Vientiane, Laos, 2017; pp. 15–19. [Google Scholar]
- Sánchez-Montoya, M.M.; Vidal-Abarca, M.R.; Puntı, T.; Poquet, J.M.; Prat, N.; Rieradevall, M.; Alba-Tercedor, J.; Zamora-Muñoz, C.; Toro, M.; Robles, S.; et al. Defining criteria to select reference sites in Mediterranean streams. Hydrobiologia 2009, 619, 39–54. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Xu, Z.; Li, L. Assessment of the Huntai River in China using a multimetric index based on fish and macroinvertebrate assemblages. J. Freshw. Ecol. 2016, 31, 169–190. [Google Scholar] [CrossRef]
- Ofenbӧck, T.; Otto Moog, O.; Gerritsen, J.; Barbour, M. A stressor specific multimetric approach for monitoring running waters in Austria using benthic macro-invertebrates. Hydrobiologia 2004, 516, 251–268. [Google Scholar] [CrossRef]
- Stoddard, J.L.; Larsen, D.P.; Hawkins, C.P.; Johnson, R.K.; Norris, R.H. Setting expectations for the ecological condition of streams: The concept of reference condition. Ecol. Appl. 2006, 16, 1267–1276. [Google Scholar] [CrossRef] [PubMed]
- Baptista, D.F.; Buss, D.F.; Egler, M.; Giovanelli, A.; Silveira, M.P.; Nessimian, J.L. A multimetric index based on benthic macroinvertebrates for evaluation of Atlantic Forest streams at Rio de Janeiro State, Brazil. Hydrobiologia 2007, 575, 83–94. [Google Scholar] [CrossRef]
- Hu, X.; Zuo, D.; Xu, Z.; Huang, Z.; Liu, B.; Han, Y.; Bi, Y. Response of macroinvertebrate community to water quality factors and aquatic ecosystem health assessment in a typical river in Beijing, China. Environ. Res. 2022, 212, 113474. [Google Scholar] [CrossRef] [PubMed]
- Karr, J.R.; Chu, E.W. Restoring Life in Running Waters: Better Biological Monitoring; Island Press: Washington, DC, USA, 1999. [Google Scholar]
- Clements, W.H. Metal Tolerance and Predator-Prey Interactions in Benthic Macroinvertebrate Stream Communities. Ecol. Appl. 1999, 9, 1073–1084. [Google Scholar]
- Jones, F.C. Taxonomic sufficiency: The influence of taxonomic resolution on freshwater bioassessments using benthic macroinvertebrates. Environ. Rev. 2008, 16, 45–69. [Google Scholar] [CrossRef]
- Helson, J.E.; Williams, D.D. Development of a macroinvertebrate multimetric index for the assessment of low-land streams in the neotropics. Ecol. Indic. 2013, 29, 167–178. [Google Scholar] [CrossRef]
- Suhaila, A.H.; Che Salmah, M.R. Application of aquatic insects (Ephemeroptera, Plecoptera and Trichoptera) in water quality assessment of Malaysian headwater. Trop. Life Sci. Res. 2017, 28, 143–162. [Google Scholar]
- Bowles, D.E.; Cribbs, J.T.; Hinsey, J.A.; Williams, J.M. Aquatic Invertebrate Monitoring at Wilson’s Creek National Battlefield: 1996–2016 Status Report; National Park Service: Fort Collins, CO, USA, 2020; pp. 1–21. [Google Scholar]
- Lee, D.Y.; Lee, D.S.; Hwang, S.J.; Lee, K.L.; Park, Y.S. Distribution patterns and vulnerability of stoneflies (Plecoptera: Insecta) in South Korean streams with conservation perspectives. Glob. Ecol. Conserv. 2022, 34, e02030. [Google Scholar] [CrossRef]
- Alemneh, T.; Ambelu, A.; Zaitchik, B.F.; Bahrndorff, S.; Mereta, S.T.; Pertoldi, C. A macroinvertebrate multi-metric index for Ethiopian highland streams. Hydrobiologia 2019, 843, 125–141. [Google Scholar] [CrossRef]
- Arimoro, F.O.; Muller, W.J. Mayfly (Insecta: Ephemeroptera) community structure as an indicator of the ecological status of a stream in the Niger Delta area of Nigeria. Environ. Monit. Assess. 2010, 166, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Morse, J.C.; Frandsen, P.B.; Graf, W.; Thomas, J.A. Diversity and Ecosystem Services of Trichoptera. Insects 2019, 10, 125. [Google Scholar] [CrossRef] [PubMed]
- Edegbene, A.O.; Akamagwuna, F.C.; Odume, O.N.; Arimoro, F.O.; Edegbene Ovie, T.T.; Akumabor, E.C.; Ogidiaka, E.; Kaine, E.A.; Nwaka, K.H. A Macroinvertebrate-Based Multimetric Index for Assessing Ecological Condition of Forested Stream Sites Draining Nigerian Urbanizing Landscapes. Sustainability 2022, 14, 11289. [Google Scholar] [CrossRef]
- White, J.C.; Hill, M.J.; Bickerton, M.A.; Wood, P.J. Macroinvertebrate taxonomic and trait compositions within lotic habitats affected by river restoration practices. Environ. Manag. 2017, 60, 513–525. [Google Scholar] [CrossRef] [PubMed]
- Arimoro, F.O.; Odume, O.N.; Uhunoma, S.I.; Edegbene, A.O. Anthropogenic impact on water chemistry and benthic macroinvertebrate associated changes in a southern Nigeria stream. Environ. Monit. Assess. 2015, 187, 14. [Google Scholar] [CrossRef]
- Odume, O.N.; Palmer, C.G.; Arimoro, F.O.; Mensah, P.K. Chironomid assemblage structure and morphological response to pollution in an effluent-impacted river, Eastern Cape, South Africa. Ecol. Indic. 2016, 67, 391–402. [Google Scholar] [CrossRef]
- Harrison, E.T.; Norris, R.H.; Wilkinson, S.N. The impact of fine sediment accumulation on benthic macroinvertebrates: Implications for river management. In Proceedings of the 5th Australian Stream Management Conference. Australian Rivers: Making a Difference, Charles Sturt University, New South Wales, Australia, 21–25 May 2007; Wilson, A.L., Dehaan, R.L., Watts, R.J., Page, K.J., Bowmer, K.H., Curtis, A., Eds.; Charles Sturt University, Institute for Land, Water and Society: New South Wales, Australia, 2007; pp. 139–144. [Google Scholar]
- Park, Y.S.; Lek, S.; Chon, T.S.; Verdonschot, P.F.M. Evaluation of Environmental Factors to Determine the Distribution of Functional Feeding Groups of Benthic Macroinvertebrates Using an Artificial Neural Network. J. Ecol. Field Biol. 2008, 31, 233–241. [Google Scholar] [CrossRef] [Green Version]
- Addo-Bediako, A. Spatial distribution patterns of benthic macroinvertebrate functional feeding groups in two rivers of the olifants river system, South Africa. J. Freshw. Ecol. 2021, 36, 97–109. [Google Scholar] [CrossRef]
- Purcell, A.H.; Bressler, D.W.; Paul, M.J.; Barbour, M.T.; Rankin, E.T.; Carter, J.L.; Resh, V.H. Assessment Tools for Urban Catchments: Developing Biological Indicators Based on Benthic Macroinvertebrates. JAWRA 2009, 45, 306–319. [Google Scholar] [CrossRef]
- Uttaruk, P.; Voharndee, P.; Jusanit, P.; Bunyaadunyakit, P.; Suriya, S.; Jaengjaithum, T.; Nittaya, K.; Sangpradub, N. Development of biotic index based on rapid bioassessment approaches using benthic macroinvertebrates for Chi and Mun headwater streams, Northeast Thailand. AES Bioflux 2011, 3, 29–43. [Google Scholar]
- Ramírez, A.; Gutiérrez-Fonseca, P.E. Functional feeding groups of aquatic insect families in Latin America: A critical analysis and review of existing literature. Rev. Biol. Trop. 2014, 62, 155–167. [Google Scholar] [CrossRef] [PubMed]
- Gething, K.J.; Ripley, M.C.; Mathers, K.L.; Chadd, R.P.; Wood, P.J. The influence of substrate type on macroinvertebrate assemblages within agricultural drainage ditches. Hydrobiologia 2020, 847, 4273–4284. [Google Scholar] [CrossRef]
- Leung, A.S.L.; Li, A.O.Y.; Dudgeon, D. Scales of spatiotemporal variation in macroinvertebrate assemblage structure in monsoonal streams: The importance of season. Freshw. Biol. 2012, 57, 218–231. [Google Scholar] [CrossRef]
- Marshall, J.C.; Steward, A.L.; Harch, B.D. Taxonomic resolution and quantification of freshwater macroinvertebrate samples from an Australian dryland river: The benefits and costs of using species abundance data. Hydrobiologia 2006, 572, 171–194. [Google Scholar] [CrossRef]



| Ecoregion | Study Localities | Type of Lotic Ecosystems | Province | Coordinates | Altitude (masl) | Activity | Substrate Types (%) |
|---|---|---|---|---|---|---|---|
| The Lower Lancang | 1. Nam Houng upstream (HU) | Wadeable river | Xaignabouly | 19°18′30.68″ N 101°43′24.94″ E | 294 | Forest areas, small agriculture patches | Cobble (60%), pebble (10%), gravel (20%), sand (10%) |
| 2. Nam Houng downstream (HL) | Wadeable river | Xaignabouly | 19°15′38.89″ N 101°42′46.01″ E | 283 | Residential areas, agriculture areas | Cobble (70%), gravel (10%), sand (15%), detritus (5%) | |
| 3. Nam Khod (KD) | Stream | Luangprabang | 19°44′14.12″ N 102°8′52.63″ E | 375 | Forest areas, small agriculture patches | Boulder (20%), cobble (30%), pebble (15%), gravel (20%), sand (5%), detritus (10%) | |
| 4. Nam Khan (NK) | Stream | Luangprabang | 19°43′55.58″ N 102°9′24.31″ E | 332 | Agriculture areas, intensive erosion | Cobble (50%), detritus (10%), muck-mud (40%) | |
| Khorat Plateau | 5. Nam Song (NS) | Wadeable river | Vientiane | 19°6′13.07″ N 102°30′3.16″ E | 315 | Forest areas, small agriculture patches | Boulder (10%), cobble (65%), pebble (15%), gravel (5%), sand (5%) |
| 6. Nam Thang (NT) | Stream | Vientiane | 19°6′15.12″ N 102°29′54.98″ E | 324 | Forest areas, small agriculture patches | Cobble (60%), pebble (15%), gravel (10%), sand (10%), detritus (5%) | |
| 7. Nam Hinboun upstream (BU) | Wadeable river | Khammouan | 17°57′19.47″ N 104°45′28.73″ E | 164 | Preserve areas | Cobble (15%), pebble (5%), gravel (60%), sand (20%) | |
| 8. Nam Hinboun downstream (BL) | Wadeable river | Khammouan | 17°57′33.58″ N 104°43′32.96″ E | 156 | Agriculture area | Gravel (20%), sand (40%), muck-mud (40%) | |
| Kratie-Stung Treng | 9. Xe Katam (KT) | Wadeable river | Champasak | 15°7′48.93″ N 106°40′09.92″ E | 257 | Forest areas, small agriculture patches | Boulder (60%), cobble (30%), gravel (5%), sand (5%) |
| 10. Xe Namnoy (NN) | Wadeable river | Xekong | 15°13′37.99″ N 106°44′45.30″ E | 132 | Forest areas, small agriculture patches | Boulder (50%), cobble (30%), pebble (10%), gravel (5%), sand (5%) |
| Parameters | Reference Group (n = 7) | Intermediate Group (n = 26) | Stressed Group (n = 7) | p-Value |
|---|---|---|---|---|
| Air temperature (°C) | 24.64 ± 4.53 | 27.12 ± 4.86 | 30.64 ± 4.19 | 0.152 |
| Water temperature (°C) | 22.59 ± 2.16 | 24.73 ± 2.69 | 25.91 ± 3.50 | 0.103 |
| Water channel width (m) | 19.85 ± 11.94 | 17.62 ± 10.20 | 14.18 ± 11.04 | 0.592 |
| Water depth (cm) | 21.35 ± 7.31 | 30.82 ± 13.99 | 33.43 ± 14.02 | 0.182 |
| Water velocity (m/s) | 0.49 ± 0.18 | 0.43 ± 0.18 | 0.32 ± 0.21 | 0.249 |
| Turbidity (NTU) | 2.83 ± 1.84 | 2.19 ± 1.93 | 2.56 ± 2.43 | 0.772 |
| Suspended solids (mg/L) | 5.38 ± 2.96 | 6.19 ± 3.98 | 6.05 ± 2.16 | 0.743 |
| Electrical conductivity (µS/cm) | 113.33 ± 65.56 a | 256.40 ± 124.29 b | 379.48 ± 101.02 b | 0.001 * |
| Total dissolved solids (mg/L) | 58.31 ± 34.23 a | 132.09 ± 64.03 b | 194.57 ± 53.45 b | 0.001 * |
| Dissolved oxygen (mg/L) | 6.30 ± 0.16 | 6.63 ± 0.75 | 6.62 ± 1.78 | 0.325 |
| pH | 7.74 ± 0.48 | 7.91 ± 0.36 | 7.85 ± 0.46 | 0.665 |
| Nitrate nitrogen (mg/L) | 0.20 ± 0.05 a | 0.30 ± 0.12 b | 0.32 ± 0.10 b | 0.021 * |
| Orthophosphate (mg/L) | 0.17 ± 0.06 | 0.19 ± 0.10 | 0.24 ± 0.13 | 0.647 |
| Biochemical oxygen demand (mg/L) | 1.45 ± 0.68 | 1.14 ± 0.81 | 1.11 ± 0.84 | 0.573 |
| Chlorophyll a (μg/L) | 0.83 ± 0.20 | 0.97 ± 0.43 | 1.15 ± 0.82 | 0.786 |
| THS | 152.57 ± 14.33 a | 137.27 ± 17.40 a | 80.71 ± 21.78 b | 0.000 * |
| Taxa | Reference Group | Intermediate Group | Stressed Group | p-Value |
|---|---|---|---|---|
| ANNELIDA | ||||
| Oligochaeta | 0.14 ± 0.38 | 0.08 ± 0.27 | 0.29 ± 0.49 | 0.338 |
| ARTHROPODA | ||||
| Decapoda | 0.43 ± 0.53 | 0.96 ± 0.96 | 1.14 ± 0.69 | 0.247 |
| Isopoda | 0.29 ± 0.49 | 0.04 ± 0.20 | 0 | 0.330 |
| Coleoptera | 8.14 ± 4.98 | 6.85 ± 2.94 | 4.14 ± 1.68 | 0.085 |
| Collembola | 0 | 0.04 ± 0.20 | 0 | - |
| Diptera | 3.71 ± 1.11 | 4.92 ± 2.33 | 4.57 ± 0.98 | 0.395 |
| Ephemeroptera | 14.29 ± 1.80 a | 13.15 ± 2.71 ab | 10.29 ± 3.20 b | 0.038 * |
| Hemiptera | 4.71 ± 2.43 | 4.96 ± 2.22 | 3.00 ± 2.24 | 0.109 |
| Lepidoptera | 0.71 ± 0.76 | 1.15 ± 1.05 | 0.57 ± 0.98 | 0.298 |
| Megaloptera | 0.71 ± 0.49 | 0.50 ± 0.51 | 0.43 ± 0.53 | 0.522 |
| Odonata | 5.14 ± 3.39 | 4.96 ± 2.24 | 4.00 ± 2.31 | 0.593 |
| Orthoptera | 0.29 ± 0.49 | 0.15 ± 0.37 | 0 | 0.620 |
| Plecoptera | 2.14 ± 0.90 a | 1.92 ± 1.74 ab | 0.71 ± 0.49 b | 0.029 * |
| Trichoptera | 10.57 ± 2.94 a | 7.85 ± 2.91 ab | 4.86 ± 2.48 b | 0.006 * |
| MOLLUSCA | ||||
| Mesogastropoda | 0.57 ± 0.53 | 1.19 ± 0.94 | 2.00 ± 1.41 | 0.072 |
| Unionoida | 0 | 0.04 ± 0.20 | 0.29 ± 0.49 | 0.330 |
| Veneroida | 0 | 0.31 ± 0.47 | 0.71 ± 0.49 | 0.109 |
| Total taxa | 51.86 ± 15.20 a | 49.08 ± 7.56 a | 37.00 ± 9.00 b | 0.009 * |
| Taxa | Reference Group | Intermediate Group | Stressed Group | p-Value |
|---|---|---|---|---|
| ANNELIDA | ||||
| Oligochaeta | 0.29 ± 0.76 | 0.12 ± 0.43 | 1.71 ± 4.11 | 0.320 |
| ARTHROPODA | ||||
| Decapoda | 0.43 ± 0.53 | 3.00 ± 4.85 | 5.86 ± 8.65 | 0.089 |
| Isopoda | 0.43 ± 0.79 | 0.12 ± 0.59 | 0 | 0.352 |
| Coleoptera | 35.29 ± 35.00 | 34.46 ± 27.87 | 25.00 ± 35.42 | 0.639 |
| Collembola | 0 | 0.04 ± 0.20 | 0 | - |
| Diptera | 32.00 ± 32.34 a | 38.50 ± 23.03 a | 78.57 ± 61.95 b | 0.045 * |
| Ephemeroptera | 126.57 ± 62.66 | 122.92 ± 51.90 | 96.57 ± 48.59 | 0.539 |
| Hemiptera | 19.14 ± 15.70 | 20.73 ± 13.53 | 18.57 ± 17.93 | 0.801 |
| Lepidoptera | 5.00 ± 10.65 | 2.23 ± 2.69 | 1.14 ± 2.04 | 0.489 |
| Megaloptera | 2.00 ± 2.16 | 1.42 ± 2.27 | 0.43 ± 0.53 | 0.311 |
| Odonata | 12.71 ± 8.60 | 17.69 ± 15.29 | 26.43 ± 23.96 | 0.352 |
| Orthoptera | 0.29 ± 0.49 | 0.23 ± 0.65 | 0 | 0.651 |
| Plecoptera | 11.71 ± 11.60 a | 13.81 ± 20.54 a | 1.00 ± 0.82 b | 0.020 * |
| Trichoptera | 84.29 ± 28.15 | 80.15 ± 51.44 | 62.43 ± 70.96 | 0.450 |
| MOLLUSCA | ||||
| Mesogastropoda | 10.71 ± 25.74 | 7.38 ± 9.74 | 9.43 ± 12.66 | 0.374 |
| Unionoida | 0 | 0.08 ± 0.39 | 1.86 ± 3.29 | 0.308 |
| Veneroida | 0 | 0.73 ± 1.28 | 4.00 ± 4.55 | 0.060 |
| Total individuals | 340.86 ± 19.99 | 343.62 ± 16.11 | 333.00 ± 17.30 | 0.168 |
| Chironomidae | 17.57 ± 21.83 a | 20.19 ± 10.68 b | 63.14 ± 68.04 c | 0.005 * |
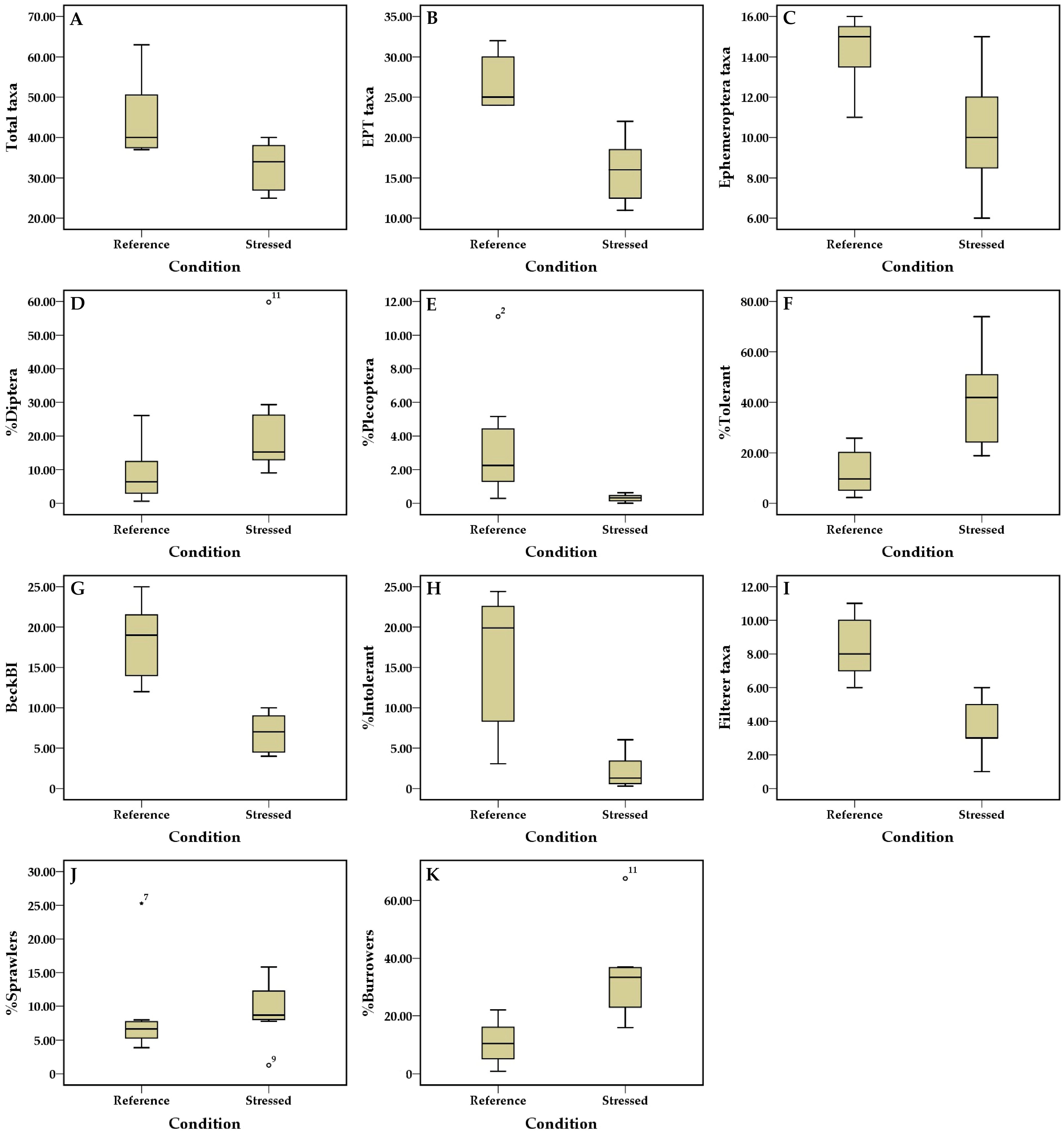
| Metric | Expected Response of Metrics to Pollution | Discrimination Power Score | DE Value | Metric Selection |
|---|---|---|---|---|
| Richness category | ||||
| 1. Total taxa | Decrease | 2 | 71.43 | / |
| 2. EPT taxa | Decrease | 3 | 100.00 | / |
| 3. Ephemeroptera taxa | Decrease | 3 | 71.43 | / |
| 4. Plecoptera taxa | Decrease | 3 | 100.00 | - |
| 5. Trichoptera taxa | Decrease | 3 | 85.71 | Redundant |
| 6. Coleoptera taxa | Decrease | 1 | 42.86 | - |
| 7. EPTC taxa | Decrease | 3 | 100.00 | Redundant |
| Composition category | ||||
| 8. %EPT | Decrease | 1 | 57.14 | - |
| 9. %Ephemeroptera | Decrease | 1 | 42.86 | - |
| 10. Margalef’s index | Decrease | 2 | 71.43 | - |
| 11. %Odonata | Increase | 2 | 57.14 | - |
| 12. %Chironomidae | Increase | 2 | 57.14 | - |
| 13. %Diptera | Increase | 3 | 71.43 | / |
| 14. %Plecoptera | Decrease | 3 | 100.00 | / |
| 15. %Trichoptera | Decrease | 2 | 57.14 | - |
| Tolerance value category | ||||
| 16. Intolerant taxa | Decrease | 3 | 100.00 | Redundant |
| 17. %Tolerant | Increase | 3 | 85.71 | / |
| 18. %Dominant taxon | Increase | 3 | 71.43 | - |
| 19. Beck’s Biotic Index | Decrease | 3 | 100.00 | / |
| 20. Simpson Index | Increase | 2 | 71.43 | - |
| 21. Hilsenhof’s Biotic Index | Increase | 3 | 100.00 | Redundant |
| 22. %Intolerant | Decrease | 3 | 100.00 | / |
| Functional feeding group category | ||||
| 23. %Filterers | Decrease | 1 | 57.14 | - |
| 24. %Scrapers | Decrease | 3 | 71.43 | - |
| 25. %Collectors | Increase | 2 | 57.14 | - |
| 26. Collector taxa | Decrease | 1 | 57.14 | - |
| 27. Filterer taxa | Decrease | 3 | 100.00 | / |
| 28. Predator taxa | Decrease | 2 | 42.86 | - |
| 29. Scraper taxa | Decrease | 3 | 57.14 | - |
| 30. %Shredders | Increase | 1 | 42.86 | - |
| 31. Shredder taxa | Decrease | 3 | 71.43 | - |
| Habit category | ||||
| 32. Clinger taxa | Decrease | 3 | 100.00 | Redundant |
| 33. %Clingers | Decrease | 2 | 71.43 | - |
| 34. %Sprawlers | Increase | 3 | 85.71 | / |
| 35. %Burrowers | Increase | 3 | 85.71 | / |
| Metric | Expected Response to Pollution | Statistic Value of Reference Sites | Scoring Criteria | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Min. | 25th | 50th | 75th | Max. | 5 | 3 | 1 | ||
| Richness category | |||||||||
| Total taxa | Decrease | 37 | 37 | 40 | 54 | 63 | ≥37 | 36–19 | <19 |
| EPT taxa | Decrease | 24 | 24 | 25 | 30 | 32 | ≥24 | 23–12 | <12 |
| Ephemeroptera taxa | Decrease | 11 | 13 | 15 | 16 | 16 | ≥13 | 12–7 | <7 |
| Composition category | |||||||||
| %Diptera | Increase | 0.6 | 1.9 | 6.3 | 14.4 | 26.1 | ≤14.4 | 14.5–21.6 | >21.6 |
| %Plecoptera | Decrease | 0.3 | 0.6 | 2.3 | 5.2 | 11.1 | ≥0.6 | 0.5–0.3 | <0.3 |
| Tolerance value category | |||||||||
| %Tolerant | Increase | 2.3 | 4.4 | 9.6 | 25 | 25.8 | ≤25 | 25.1–37.5 | >37.5 |
| Beck’s Biotic Index | Decrease | 12 | 14 | 19 | 23 | 25 | ≥14 | 13–7 | <7 |
| %Intolerant | Decrease | 3.1 | 6 | 19.9 | 23.5 | 24.4 | ≥6 | 5.9–3.0 | <3.0 |
| Functional feeding category | |||||||||
| Filterers taxa | Decrease | 6 | 6 | 8 | 11 | 11 | ≥6 | 5.0–3.0 | <3 |
| Habit category | |||||||||
| %Sprawlers | Increase | 3.9 | 4.9 | 6.7 | 8 | 25.3 | ≤8.0 | 8.1–12.0 | >12.0 |
| %Burrowers | Increase | 0.9 | 1.3 | 10.5 | 19.7 | 22 | ≤19.7 | 19.8–28.5 | >28.5 |
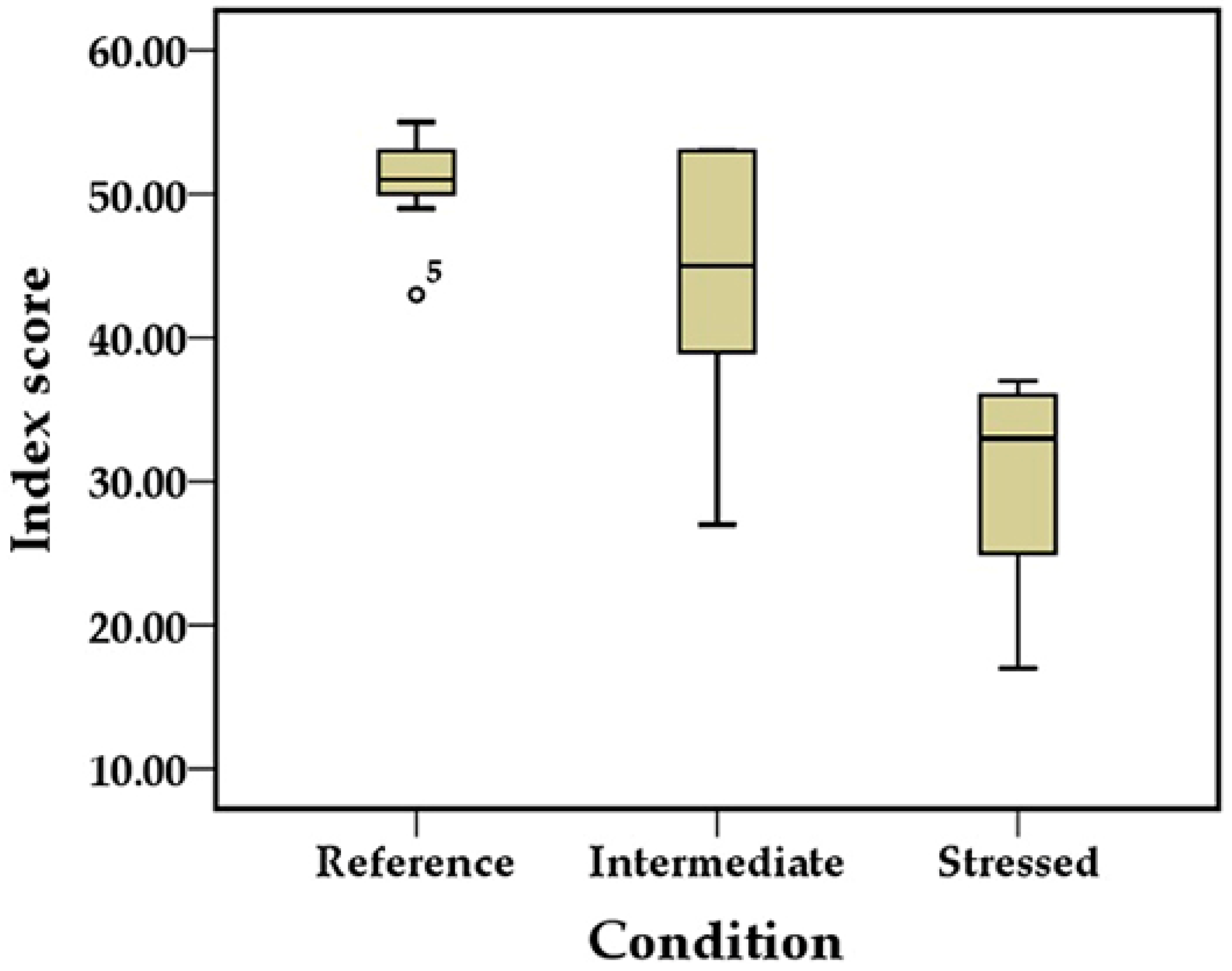
| Stream Quality Class | Percentile | Index Score |
|---|---|---|
| Excellent | ≥75th | 53 |
| Good | ≥25th | 49–52 |
| Fair | <25th | 25–48 |
| Poor | - | 13–24 |
| Impaired | - | <13 |
| Sampling Site | Condition | The Surface Water Quality Standard of Lao PDR | Lao MMI | ||
|---|---|---|---|---|---|
| Stream Class | Water Quality | Index Score | Water Quality | ||
| Cool season 2016 | |||||
| HUC16 | Reference | 2 | Good | 49 | Good |
| HLC16 | Stressed | 2 | Good | 33 | Fair |
| KDC16 | Intermediate | 2 | Good | 47 | Fair |
| KNC16 | Stressed | 2 | Good | 31 | Fair |
| NSC16 | Intermediate | 2 | Good | 53 | Excellent |
| NTC16 | Reference | 2 | Good | 55 | Excellent |
| BUC16 | Intermediate | 2 | Good | 45 | Fair |
| BLC16 | Intermediate | 2 | Good | 31 | Fair |
| KTC16 | Reference | 2 | Good | 53 | Excellent |
| NNC16 | Reference | 2 | Good | 53 | Excellent |
| Hot season 2017 | |||||
| HUH17 | Intermediate | 2 | Good | 35 | Fair |
| HLH17 | Intermediate | 2 | Good | 39 | Fair |
| KDH17 | Intermediate | 2 | Good | 53 | Excellent |
| KNH17 | Stressed | 2 | Good | 37 | Fair |
| NSH17 | Intermediate | 1 | Excellent | 43 | Fair |
| NTH17 | Intermediate | 2 | Good | 53 | Excellent |
| BUH17 | Intermediate | 2 | Good | 27 | Fair |
| BLH17 | Stressed | 2 | Good | 17 | Poor |
| KTH17 | Intermediate | 2 | Good | 53 | Excellent |
| NNH17 | Intermediate | 2 | Good | 53 | Excellent |
| Cool season 2017 | |||||
| HUC17 | Reference | 2 | Good | 43 | Fair |
| HLC17 | Intermediate | 2 | Good | 53 | Excellent |
| KDC17 | Intermediate | 2 | Good | 45 | Fair |
| KNC17 | Intermediate | 2 | Good | 39 | Fair |
| NSC17 | Intermediate | 2 | Good | 45 | Fair |
| NTC17 | Reference | 2 | Good | 51 | Good |
| BUC17 | Intermediate | 2 | Good | 33 | Fair |
| BLC17 | Stressed | 2 | Good | 37 | Fair |
| KTC17 | Intermediate | 2 | Good | 53 | Excellent |
| NNC17 | Intermediate | 2 | Good | 51 | Good |
| Hot season 2018 | |||||
| HUH18 | Intermediate | 2 | Good | 39 | Fair |
| HLH18 | Intermediate | 2 | Good | 41 | Fair |
| KDH18 | Intermediate | 2 | Good | 43 | Fair |
| KNH18 | Stressed | 3 | Fair | 35 | Fair |
| NSH18 | Intermediate | 2 | Good | 47 | Fair |
| NTH18 | Intermediate | 2 | Good | 45 | Fair |
| BUH18 | Intermediate | 2 | Good | 47 | Fair |
| BLH18 | Stressed | 2 | Good | 19 | Poor |
| KTH18 | Reference | 2 | Good | 51 | Good |
| NNH18 | Intermediate | 2 | Good | 47 | Fair |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sripanya, J.; Vongsombath, C.; Vannachak, V.; Rattanachan, K.; Hanjavanit, C.; Mahakham, W.; Sangpradub, N. Benthic Macroinvertebrate Communities in Wadeable Rivers and Streams of Lao PDR as a Useful Tool for Biomonitoring Water Quality: A Multimetric Index Approach. Water 2023, 15, 625. https://doi.org/10.3390/w15040625
Sripanya J, Vongsombath C, Vannachak V, Rattanachan K, Hanjavanit C, Mahakham W, Sangpradub N. Benthic Macroinvertebrate Communities in Wadeable Rivers and Streams of Lao PDR as a Useful Tool for Biomonitoring Water Quality: A Multimetric Index Approach. Water. 2023; 15(4):625. https://doi.org/10.3390/w15040625
Chicago/Turabian StyleSripanya, Jutamas, Chanda Vongsombath, Viengkhone Vannachak, Kaewpawika Rattanachan, Chutima Hanjavanit, Wuttipong Mahakham, and Narumon Sangpradub. 2023. "Benthic Macroinvertebrate Communities in Wadeable Rivers and Streams of Lao PDR as a Useful Tool for Biomonitoring Water Quality: A Multimetric Index Approach" Water 15, no. 4: 625. https://doi.org/10.3390/w15040625
APA StyleSripanya, J., Vongsombath, C., Vannachak, V., Rattanachan, K., Hanjavanit, C., Mahakham, W., & Sangpradub, N. (2023). Benthic Macroinvertebrate Communities in Wadeable Rivers and Streams of Lao PDR as a Useful Tool for Biomonitoring Water Quality: A Multimetric Index Approach. Water, 15(4), 625. https://doi.org/10.3390/w15040625






